Abstract
The abundant and renewable resources from fruit by-products are getting emphasis on their valorization. These by-products may contain toxic substances due to factors such as cultivation, harvesting, transportation, preservation, or processing. Hence, presenting scientific overviews of the toxicological qualities and detoxification trends of these by-products are critical for implicating their possible valorization. The present demand for valorization of fruit by-products requires emphasis and methodologies for the detoxification of any toxicants to develop healthier products. This review emphasized the toxicological qualities of by-products from fruits for which the maximum global production occurred in 2022. In this review, heavy metals (arsenic, cadmium, cobalt, chromium, nickel, lead, and mercury), mycotoxins, toxicant organic compounds, anti-nutritional factors, and pesticide/fungicide residues of the selected fruit byproducts were discussed. Current trends to reduce possible toxicants of these by-products during their valorization were emphasized. Novel functional foods valorized from these fruit by-products and future perspectives of detoxification were also focused on in this review.
Graphical abstract
1 Introduction
The global population is growing rapidly, which demands more resources. Alongside the expanding growth of fruit production, optimum utilization mechanisms of their by-products are very important. Valorizing fruit by-products means employing and ensuring methods to exploit fruit wastes such as skin, pomace, and seeds as excellent raw materials for small scale and industrial based usage. Toxicants are biological and chemical components present in fruit by-products that can be harmful for human consumption when their amount is beyond the permissible level. Improving the valorization of fruit by-products not only ensures proper utilization of agricultural products but also protects environmental pollution. However, some toxic components may be available in these fruit by-products. Sufficient data on their toxicity should be available for every fruit by-product. In case of available toxicant component, proper reduction mechanism are required before valorization.
Our current living style, food processing method adopted, nature of food sources, and food market globalization are susceptible to food poisoning. Therefore, many organizations such as the food and drug administration (FDA), Food Safety Commission of Japan, European Food Safety Authority, and World Health Organization (WHO) from different countries have been developing regulations on food toxicological qualities. Food poisoning agents in fruit by-products are chemical and natural toxins, which can originate from factors associated with the raw material of the food products, microorganisms, processing, and conservation conditions. Toxicants from excessive heavy metals, nitrates, oxalates, mycotoxins, and pesticide residues are the main sources of health hazards. Consumption of foods containing maximum concentrations of toxins leads to health hazards. Hence, food supplements, cosmetics, and pharmaceuticals produced from fruit by-products should ensure high food quality and safety levels.
Agro-food-by-products are susceptible to toxicants during maturation, harvesting, preservation, and processing. Although agro-food-by-products are cheaper sources, profitable for local economies, and contain potentially functional ingredients, consumers and the food industries need details of their nutritional and toxicological qualities to be able to make use of them. Health hazards mainly mycotoxins such as aflatoxins, ochratoxin A, patulin, zearalenone (ZEN), and deoxynivalenol (DON) present in fruits and its products cause health effects of mutagenic, neurotoxic, nephrotoxic, hepatic carcinoma, and immunodeficiency, respectively [1]. To minimize the risk of mycotoxin contamination, storing fruit by-products properly and discarding any visibly moldy or spoiled produce is important. Toxic heavy metals such as lead, cadmium, chromium, cobalt, and nickel can cause harmful damage to humans. These heavy metals excessively build up in edible plant and fruit bodies from the ecosystem where they are cultivated such as from downstream of leather factories (which use chromium and other chemicals). Besides, the range of hazard quotient for As, Cd, Cr, Co, Cu, Ni, Hg, and Pb should be determined [2,3]. Another potentially toxic component in fruit by-products is pesticide residue. To reduce pesticide exposure, it is advisable to wash the fruit thoroughly before consumption and consider purchasing organic produce whenever possible. Certain fruits also contain naturally occurring toxic compounds that can be harmful if consumed excessively [4,5]. For example, some nightshade fruits contain solanine, a glycoalkaloid that acts as a natural defense mechanism against pests and diseases [6,7]. Oxalates are another toxic component that can be present in fruit byproducts. Oxalates are naturally occurring substances found in many plants. Hence, ingesting high amounts of oxalates can lead to the formation of kidney stones in susceptible individuals [8]. Nitrites are an increased risk of cancer and other health issues. To minimize nitrite formation, storing these fruit by-products properly (refrigerated) and avoiding overheating them is recommended. Toxicological and anti-nutrient qualities of many fruit by-products have not been given much attention, and most of the time these parts are discarded with their hidden food, nutraceuticals, and pharmaceutical potential not realized. Hence, evaluating the toxicological and anti-nutrient contents of these by-products so that the knowledge derived can be used to encourage the use of the by-products, while also encouraging adequate consumption of fruits, is a value-added application that this review tries to address.
Liu et al. [9] applied shell thickness-dependent Raman enhancement of silver-coated gold nanoparticles to measure pesticide residues in apple, grape, pear, peach, and mango fruit peels. They reported that pesticide residues named as thiram (1.46–7.23 ng/cm2), chlorpyrifos (0.14–0.7 μg/cm2), and methyl parathion (0.025–0.5 μg/cm2) were found in each of the fruit peels, in which the maximum thiram (7.23 ng/cm2) was observed in mango peel.
Mycotoxins, toxic heavy metals such as arsenic (As), lead (Pb), cadmium (Cd), chromium (Cr), cobalt (Co), and nickel (Ni), pesticide residues, oxalates, and nitrate have recommended daily intake (RDI). Excessive presence of minerals (heavy minerals), saponin, alkaloid, hydrocyanic acid, oxalate, tannins, phytates, and nitrates in fruit by-products compared to recommended daily allowance (RDI) are considered as toxicant components. The range of hazard quotient (mg/kg) for As, Cd, Cr, Co, Cu, Ni, and Pb are 2.71–11.38, 0.60–3.32, 0.81–3.18, 0.03–0.09, 0.09–0.26, 0.08–0.34, and 0.83–2.23, respectively [2]. The acceptable daily intake (ADI) limit or toxicity level of dietary nitrates is 3.7 mg/kg body weight according to the regulations of the Joint Expert Committee of Food and Agriculture and the European Commission’s Scientific Committee on Food [10]. RDI values of potassium, magnesium, iron, zinc, manganese, copper, phosphorus, and calcium for adults are 4.6 g/day, 260 mg/day, 14 mg/day, 7 mg/day, 2.3 mg/day, 900 µg/day, 700 mg/day, and 1,000 mg/day, respectively [11]. Heavy metals like As, Pb, and Cd are considered toxicants if their concentrations are greater than the recommended level. Based on the WHO [12] recommendation, the monthly intake level should be <0.2 mg/100 g for aluminum; 0.21 mg/100 g for As and Pb, and 0.25 mg/100 g for Cd. Moreover, the maximum Pb limit for edible parts of crops for human health is 0.02 mg/100 g. Pineapple skin contained lead beyond this maximum limit (0.64 mg/100 g analyzed in dry base), whereas in fruit by-products such as orange peel, watermelon rind, banana peel, apple pomace, strawberry pomace, grape pomace, lead contents were insignificantly detected (below 0.1 µg/L) [13,14]. According to Oyeyinka and Afolayan [15], fresh banana (M. sinensis) peel contains a lower alkaloid (0.66 g/100 g), oxalate (37.0 g/100 g), phytate (2.78 g/100 g), and saponin (6.57 g/100 g) than boiled peel extract of the fruit that composed alkaloid (1.76 g/100 g), oxalate (40.2 g/100 g), and saponin (8.12 g/100 g). Except for oxalate, the alkaloid, phytate, and saponin in this study report are in a safe level for human consumption of the fruit peel. According to the study conducted on the local fruits in Incheon, Korea, the most frequently detected pesticide residues were chlorfenapyr, procymidone, etofenprox, pendimethalin, fluopyram, and azoxystrobin [7].
Anti-nutritional factors (ANFs) such as oxalates, hydrogen cyanides (HCN), alkaloids, phytates, tannins, and glycosides can be found in food products [16]. In particular, fruit by-products contain considerable amounts of ANFs although their level is different, which are summarized in Table 2. The anti-nutritional effect of oxalate (a salt formed from oxalic acid) is that it can bind to nutrients preventing their absorption. Hence, consuming foods containing high concentrations of oxalic acid can cause nutritional deficiencies and irritation of the lining of the gut. In addition to these sources, from the flavonoid groups, tannins are anti-nutrients that cause health effects. Mainly it involves in chelation of minerals like iron and zinc, which then reduce their absorption, as well as inhibit digestive enzymes, thus causing precipitate proteins [17]. Fruits and berries contain high amounts of oxalates, cyanide-inducing glycoside, amygdalin (AMG), sambunigrin, hydrocyanic acid, and toxins [18,19,20,21].
Our study tries to give a comprehensive review of the availability of heavy metals (As, Cd, Co, Cr, Ni, Pb, and Hg), mycotoxins, toxicant organic compounds, ANFs, and pesticide/fungicide residues in fruit by-products that have maximum (>1.2 million tons) global production from FAOSTAT during the year of 2022 [22]. Moreover, the current detoxification trends for the selected fruit by-products were discussed. We focused to review these selected fruit by-products since much of their by-products are being wasted even though these are important raw materials for small scale and industrial based exploitations. There is greater demand for valorization of these by-products due to their promising bioactive compound contents. Utilization of their by-products for better inspiration of future research and the discovery of biofuel, food ingredients, pharmaceutical, and cosmetic products are getting attention.
2 Fruits with maximum global production and their by-products
The global production, consumption, and generation of waste and fruits by-products are increasing from year to year. According to the FAOSTAT database, the fruits with large production during 2022 are bananas, watermelons, apples, oranges, grapes, and the remaining fruits are depicted in Figure 1a in decreasing order [22]. Many fruits are wasted during harvesting, transportation, and processing, and by-products are produced. Studies have been reporting that from the total fruit weight, about 30–50% accounts for fruit waste and by-products [23]. Percentage waste part of common fruits prioritized in the current review are depicted in Figure 1b. Nearly half of the fruit parts are discarded in terms of peels, seeds, rinds, husks, rags, roots, and pomace during the day-to-day activities in homes and agro-processing industries. It is important to consider that these fruit by-products are important plant sources containing many bioactive substances, dietary fiber, minerals, and others [24,25]. Moreover, fruit by-products contain bioactive substances that show potent ant-microbial activities [25]. Considering their maximum production and by-products that could be generated, we prioritized discussing the toxicity of the fruits with global production greater than 1.2 million tonnes.
![Figure 1
Global fruit production and their waste. (a) Global production of fruits by the year of 2022 [22]. (b) Percentage waste part of common fruits.](/document/doi/10.1515/biol-2025-1105/asset/graphic/j_biol-2025-1105_fig_001.jpg)
Global fruit production and their waste. (a) Global production of fruits by the year of 2022 [22]. (b) Percentage waste part of common fruits.
Potential toxicants that can persist in the bioactive compounds extracted from fruit by-products are mycotoxins, pesticides, biogenic amines, heavy metals, and microbial contaminations. These contaminants are the main causes of the extracted product’s safety, such as biological instability, potential for rapid auto-oxidation, potential pathogenic contaminations, high levels of active enzymes, and high water activity [26].
Many fruit by-products contain potential contaminants such as heavy metals mycotoxin, ant-nutritional contaminants, organic contaminants (biogenic amines), and pesticides [24]. The heavy metals (As, Cd, Co, Cr, Ni, and Pb) present in the prioritized fruit by-products are summarized in Table 1. Moreover, the selected fruit by-products containing mycotoxins, toxicant organic compounds, ANFs, and pesticide/fungicide residues are presented in Table 2.
Dominant heavy metals in fruit by-products
| Heavy metals (mg/kg) | References | |||||||
|---|---|---|---|---|---|---|---|---|
| Commodity | As | Cd | Cr | Co | Ni | Pb | ||
| Apple | Peel | <1 | 0.57–3.8 | <1 | <1 | [37,40,41] | ||
| Seed | ||||||||
| Pomace | ||||||||
| Apricot | Kernel | 0.1–6 | 2.7–35.7 | [42] | ||||
| Pomace | ||||||||
| Avocados | Seeds | 0.57–2.29 | 0.00 | 0.00 | [40,43] | |||
| Bananas | Peels | <0.0001 | 0.0013–0.18 | 1.42–4.04 | 0.4–47.2 | 0.0038–0.64 | [36,44,45] | |
| Blueberries | Pomace | 0.011 | 0.242 | 0.08 | 0.592 | 0.73 | [46] | |
| Grape | Peel/skin/pomace | <0.5 | 0.18–2.41 | <0.5 | 0.021–1.11 | [34,35,37] | ||
| Lemons | Peels by-products | 0.004 | 0.00047–0.25 | 1.04 | 0.038 | 0.973–1.24 | 0.0188–0.22 | [34,36,39] |
| Limes | By-products | ND | 0.003 | 0.073 | 1.678 | 0.128 | [39] | |
| Mangoes | 0.33 | 28.0 | [45] | |||||
| Orange | Peel | <0.5 | 1.04–4.14 | 0.015 | 0.05–2.36 | 0.01–1.75 | [34,36,37,39,47] | |
| Seed | ||||||||
| Papayas | Peel | 0.0287–0.03 | 0.0027–0.00685 | 0.278–7.36 | 0.4–219 | 0.246–13.2 | 0.03–0.044 | [45,48,49] |
| Seed | ||||||||
| Peaches | 0.17–1.38 | <0.10 | [50] | |||||
| Pineapples | Peels | <0.0001 | 0.0074 | 8.77 | 70.3 | 0.0027 | [44,45] | |
| Plums | Peels | 1.2 | ND | 2.8 | ND | [51,52] | ||
| Kernels | <0.1 | 0.13 | 0.2 | 1.7 | 0.13 | |||
| Pomelos | Peels | 1.36 × 10−3 | 0.0296 | [34] | ||||
| Raspberries | Pomace | 0.0084 | 0.116 | 0.073 | 0.762 | 0.047 | [46] | |
| Strawberry | Pomace | 0.0078 | <0.01 | 0.00336 | 0.0011 | [53,54] | ||
| Mandarins | 0.00062 | 0.020 | [38] | |||||
| Watermelons | Peel | 0.008–0.1 | 4.65 | 0.06–0.09 | [40,55] | |||
| Seed | ||||||||
ND: Not determined.
Toxicant compounds in fruit by-products
| Commodity | Mycotoxins (mg/kg) | Toxicant organic compounds (mg/kg) | Anti-nutritional contaminants (mg/kg) | Fungicide/pesticide residues (mg/kg) | References | |
|---|---|---|---|---|---|---|
| Apple | Peel/skin | AOH-3-S = 1.4–10.8 × 10−3 | Naphthaleneacetic acid = 0.433 | Oxalates = 890.7 | Acetamiprid = 72–81 | [57,58,59,60,61] |
| Seed | HCN = 960.4 | |||||
| Pomace | AME-3-S = 1.7–10 × 10−3 | AMG = 1,000–4,000 | ||||
| Alkaloids = 79.9 | ||||||
| Phytates = 14.2 | ||||||
| Apricots | Seeds | AFB1 and AFB2 = 0.0017–22.451 | AMG = 52,000 | Tannins = 1564.4 | [62,63,64] | |
| Phytic acid = 1171.5 | ||||||
| Oxalates = 156 | ||||||
| Avocados | Seeds | Tannins = 7.6 | [43] | |||
| Alkaloids = 54 | ||||||
| Phytates = 4.4 | ||||||
| Oxalates = 44 | ||||||
| Bananas | Peels | Phytate = 2.11–9,270 | Chlorpyrifos = 0.11–0.8 | [65,66] | ||
| Alkaloids = 0.45–5.45 | methiocarb = 0.014–0.183 | |||||
| Oxalate = 20–8,280 | ||||||
| Glycosides 149,020 | ||||||
| Tannin = 900 | ||||||
| Blueberries | Pomace | Chlorpyrifos-methyl = 4.27 × 10−3 | [67,68] | |||
| Thiametoxan 5.15 × 10−3 | ||||||
| Azoxytrobin 0.187 | ||||||
| Grapes | Peel/skin | OTA = 0.1–0.32 × 10−3 | Oxalate = 0.6–0.7 | Cyprodinil = 1.07–1.94 | [34,35,69] | |
| Pomace | Tannins = 0.274–0.41 | |||||
| Dimethomorph = 0.56–2.73 | ||||||
| Feamoxadone = 1.55 | ||||||
| Lemons | Peels/pomace | Oxalate = 0.4–0.5 | Pyriproxyfen = 0.039 | [34,39,69] | ||
| Tannins = 0.28 | Fludioxonil = 0.008 | |||||
| Propiconazole = 0.008 | ||||||
| Pyrimethanil = 3.8 | ||||||
| Limes | Pomace | Fludioxonil = 0.009 | [39] | |||
| Flutriafol = 0.11 | ||||||
| Propiconazole = 0.008 | ||||||
| Imazalil = 1.49 | ||||||
| Tebuconazole = 0.076 | ||||||
| Mangoes | Putrescine = 0.9 | Phytic acid = 254.8 | [70,71,72] | |||
| Oxalate = 724 | ||||||
| Tannin = 153 × 103 | ||||||
| Orange | Peel | Tryptoquialanine A = 248.1 | Putrescine = 11.34–151.1 | Tannins = 0.228 | Etoxazole = 0.010–0.637 | [34,60,69,72,73,74,75] |
| Pomace | Tryptoquialanine C = 375.80 | Oxalates = 1.2–997.8 | Imidacloprid = 162.16 | |||
| HCN = 397.9 | Carbendazim = 372.1 | |||||
| Alkaloids = 54.4 | Abamectin = 0.261 | |||||
| Phytates = 23.4 | Cypermethrin = 495.6 | |||||
| Prochloraz = 8.11.7 | ||||||
| Papayas | Peel | Putrescine = 5.3–19.3 | Tannin = 17.6–500 | [71,72,76] | ||
| Oxalate = 0.6 | ||||||
| Phytate = 0.6 | ||||||
| Peaches | Seeds | Putrescine = 1.82–2.02 | Tannins = 5137.6 | [63,72] | ||
| Phytic acid = 2126.3 | ||||||
| Oxalates = 385.9 | ||||||
| HCN = 372 | ||||||
| Pears | Putrescine = 23.6–24.2 | [72] | ||||
| Pineapples | Pineapple shell | Fusarium = 250 | Putrescine = 1.39–7.96 | Oxalates = 0.4–1290.6 | Carbaryl = 0.262 | [60,69,72,77,78,79] |
| HCN = 715 | Carbofuran = 14.3 | |||||
| Pomace | Aflatoxin B2 = 0.008 × 10−3 | Alkaloids = 161.9 | Fenobucarb = 0.01 | |||
| Aflatoxin G1 = 0.013–0.033 × 10−3 | Phytates = 19.9 | Isoprocarb = 0.313 | ||||
| OTA = 0.051 × 10−3 | Propachlor = 0.015 | |||||
| Pomelos | Peels | Tannins = 0.315 | Total pesticides = 0.216 | [34] | ||
| Strawberry | Pomace | Putrescine = 2.04–6.42 | Total pesticides = 2.143 | [53,72,80] | ||
| Procymidone = 0.7 | ||||||
| DEHP = 0.25 | ||||||
| acetamiprid = 0.212 | ||||||
| Boscalid = 0.745 | ||||||
| DIBP = 0.283 | Carbendazim = 0.13 | |||||
| and DBP = 0.222 | ||||||
| Tangerines | Peels | AOH = 0.003–0.017 | [39] | |||
| Watermelons | Peel | Phytate = 9,900 | Dimethoate = 1,730 | [60,81,82] | ||
Studying the amounts and the toxicity level of the waste and by-products of the prioritized fruits is demanded for better valorization. In particular, bananas have the maximum global production in 2022 (Figure 1a). The banana peel represents about 35% of the total fresh harvest mass of ripe fruit, a rich source of dietary fiber, protein, essential amino acids, polyunsaturated fatty acids, antioxidant compounds, and potassium (Figure 1b) [27]. About 25–30% of the apple’s total content is represented by by-products which include seeds and peels. Moreover, nearly 20% of the original grape weight accounts for its by-products mainly pomace (skins, stems, and residual pulp) and seeds. Besides, the mango processing produces by-products that range from 40 to 60%, from which about 10–20% represents peels, 10–25% is for seeds, and 15–20% is for kernels. Citrus fruit also creates about 50–70% by-products, which represent seed (1–10%), peel (flavedo and albedo) (range 60–65%), and internal tissues (30–35%) (juice sac residues and rag) [28,29]. The industrial processing of avocados produces by-products that account for 21–30% of the total, including residues like seeds and peels. Similarly, fresh processing of pineapples creates about 35–46% by-product mainly residual pulp, peels (30–42%), stems (5% core and stem share), and a core only (10%) [28,30]. As mentioned above, the fruits contained considerable by-products, which could be utilized for different types of product development. Much of the fruit by-products have promising nutritional qualities [31]. Hence, ensuring the toxicity of these by-products for better sustainability and feasibility of valorization is required.
3 Toxicological qualities of citrus fruit by-products
Citrus fruits are well-known sour fruits such as oranges, lemons, limes, tangerines, and grapefruit. Czech et al. [32] classified citrus fruits like orange, pomelo (Citrus maxima), mandarins, lemon, and grapefruits. The by-products of citrus fruits (Rag, peel, and seeds) account for about 50% [33]. These fruit by-products can contribute to many bioactive compound extractions, sources of pectin, molasses, fibers, oils, and animal feed [31]. Mainly, the citrus fruit peels are raw materials for the functional food products development such as jams, marmalade, yogurt, crackers (a thin flat crisp biscuit), and meatballs [32].
Heavy metals mainly lead, cadmium, chromium, copper, lead, and nickel were found in orange, lemon, and grapefruit peels as well as in grape skin extracts [34,35,36]. Bożym et al. [37] analyzed heavy metals (Cd, Cr, Ni, and Pb) present in grape and orange peels as shown in Table 1. Analysis of these heavy metals revealed that their concentrations are within the permissible limits for anaerobic digestion to produce biogas.
The heavy metals reported in citrus fruit peel such as orange (Cd = 0.00049, Pb = 0.01 mg/kg), lemon (Cd = 0.00047, Pb = 0.0188 mg/kg), red grapefruit (Cd = 0.0019, Pb = 0.03 mg/kg), mandarin (Cd = 0.00062, Pb = 0.02 mg/kg), lime (Cd = 0.0265, Pb = 0.00046 mg/kg), and pomelo (Cd = 0.00136, Pb = 0.0296 mg/kg) were considered. According to the norms for heavy metals (Regulation 1275/2013 and Ordinance 10/2009), the limit for animal feed consumption for cadmium and lead is 1 and 10 mg/kg, respectively. Hence, the reported heavy metal concentrations are below these limits [38].
Mateus et al. [39] investigated heavy metal concentrations in citrus by-products mainly from orange (Citrus sinensis), lemon (Citrus limon), and lime (Citrus aurantifulia) considering As, Cd, Co, Hg, Ni, and Pb. The concentrations of these heavy metals are summarized in Table 1. They reported that the Hg in lemon, orange, and lime and the As in orange and lime by-products were reported as below the detection limit.
The heavy metal (lead and cadmium) analysis conducted on citrus fruit peels (orange, pomelo, lemon, and grapefruits) as shown in Table 1 was reported by Czech et al. [34]. However, according to WHO (0.1–0.2 mg/kg fresh weight of fruit for lead and 0.05 mg/kg for cadmium), the reported results are not beyond the acceptable levels. Moreover, they reported that the peels of these fruits contained tannins (Table 2). The concentration of tannins in grape, orange, and pomelo fruit peels may limit the use of these fruit peels, which demand reduction mechanisms.
According to the European Commission [56], heavy metals and other metals of safety concern (mg/Kg) analyzed in lyophilized citrus by-products (lemon. orange, and lime) should have a maximum residual level (MRL) of As (0.02 mg/kg), Cd (0.02 mg/kg), Co (not applicable), Hg (not applicable), Ni (not applicable), and Pb (0.1 mg/kg). The citrus by-products reviewed in the present study are below this MRL.
The mineral binding anti-nutrients such as oxalates and phytic acid concentrations in citrus fruits (lemon, orange, and grapes) pomace were investigated as negligible amounts (Table 2). Hence, the pomace of these fruits can be valorized into value-added products [69]. However, the presence of tannins should be considered.
Mycotoxins such as aflatoxins (AFB1, AFB2, AFG1, and AFG2), ochratoxin A (OTA), ZEN, toxin T2 (T2), and fumonisins (FB1 and FB2) were found in orange, lemon, or lime pomace. Moreover, alternariol (AOH), and alternariol mono-methyl ether (AME) were determined in other citrus fruit by-products such as tangerine (Citrus reticulata) peels (range from 0.003 to 0.017 mg/kg) [39,83].
Mateus et al. [39] studied the presence of pesticide residues in fresh citrus pomace. Mainly they determined these pesticides in orange (pyriproxyfen = 0.027, fludioxonil = 0.355, imazalil = 0.007, and Pyrimethanil = 2.77 mg/kg), lemon (pyriproxyfen = 0.039, fludioxonil = 0.008, propiconazole = 0.008, and pyrimethanil = 3.8 mg/kg), and lime (fludioxonil = 0.009, flutriafol = 0.11, propiconazole = 0.008, imazalil = 1.49. and tebuconazole = 0.076 mg/kg) pomace.
Socas-Rodríguez et al. [84] studied the pesticides in citrus by-products. They reported that malathion (0.045 mg/kg) and pyriproxyfen (0.0495 mg/kg) were found dominantly in the studied citrus by-product samples. The malathion was below the MRL whereas the pyriproxyfen was found approximately near the MRL (0.05 mg/kg). However, other types of pesticides studied were below the detection limit (from 0.0085 to 0.1288 mg/kg).
Mycotoxins such as AOH and AME were found in citrus fruit by-products. In particular, AOH, a type of mycotoxin, was identified in tangerine (Citrus reticulata) peels [39].
According to the MRL developed by European Food Safety et al. [85] for some fungicides such as carbendazim (0.2 mg/kg), thiabendazole (7 mg/kg), imazalil (4 mg/kg), and insecticides such as λ-cyhalothrin (0.2 mg/kg), carbofuran (0.01 mg/kg), and chlorpyrifos (1.5 mg/kg); the reviewed results of the fungicides and insecticides in citrus fruits were below the MRLs.
3.1 Oranges
Orange fruit is the fourth globally produced fruit (Figure 1). Large amounts of orange fruits are utilized for juice production, which accounts for about 85% of total processed consumption [86]. Its by-products are peels, seeds, and pomace.
The heavy metals (Cr, Ni, and Pb) analyzed in orange fruit (Citrus sinensis) seeds and peels were reported as beyond the RDI set by WHO/FAO [87] (Cr = 0.05–0.2, Ni = 1.4, and Pb = 0.214 mg/day), which are summarized in Table 1 [47]. Hence, careful handling of these by-products is required when valorized in functional foods.
Citrus aurantium (L.) is commonly known as bitter orange, sour orange, Seville orange, bigarade orange, or marmalade orange, which is a rich source of many bioactive compounds. Oral administration of these fruit peel extracts did not cause mortality or signs of acute toxicity in mice at a 2,000 mg/kg dose. Hence, this fruit peel can extract many non-toxic phytoconstituents [88].
Essential oils (EOs) extracted from bitter orange peel were studied for their oral administration toxicity in mice from 48 h to 14 days with a concentration of 2,000 mg/kg. It is reported that any of clinical symptoms, acute toxicity, or mortality, as well as no change in food intake, behavior, or body weight were observed on the tested mice at the specified time. Other studies have also proven that oral administration of mice with a dose of 5,000 mg/kg EOs did not show any such toxicity except reducing serum total cholesterol of the mice at 10 mg/kg of EOs from these extracts [88,89].
The pesticide residues (imidacloprid, abamectin, cypermethrin, and prochloraz) in orange fruits were particularly distributed in its peels and the second most distributions were in the fruit pomace (Table 2). Washing during the fruit process can reduce the pesticide residues from 43.6 to 85.4%. The fruit pomace that passed the juice extraction process contained residues from 46.0 to 94.7% of imidacloprid, abamectin, cypermethrin, and prochloraz. However, the residue carbendazim was found to lower concentration. Hence, unless proper removal of these pesticide residues is applied valorizing the orange fruit by-products could have health effects [74].
Epicarp layer of orange fruit was found to contain tryptoquialanines A and C (Table 2). However, these concentrations were found within the range of limits of tremorgenic mycotoxins, which can cause deleterious effects on vertebrates [73].
Pesticides like imidacloprid, carbendazim, abamectin, cypermethrin, prochloraz, thiabendazole, and carbaryl, and mycotoxins such as fumonisin B1, tryptoquialanines A and C are present in peels and orange by-products [73,74,90].
The ANFs reported in orange fruit peels were oxalates (997.8 mg/kg), HCN (397.9 mg/kg), alkaloids (54.4 mg/kg), and phytates (23 mg/kg). The reported value for HCN in orange fruit peel is below the threshold value (below 3,500 mg/kg) reported as the safety limit [60].
In general, orange fruit peels can be valorized into valuable products by applying toxicity reduction mechanisms. Figure 2 shows some toxicant components present in orange fruit peels.

Toxicant elements and chemicals in orange fruit by-product (peel).
3.2 Pomelos
Pomelos is a type of citrus fruit, that includes sweet oranges (Citrus sinensis), lemons (Citrus limon), limes (Citrus aurantifulia), tangerines (Citrus reticulata), and grapefruit (Citrus paradisi). About 166.4 million tonnes of these fruits and other citrus fruits (mandarins, clementines, pomelos, grapefruits) were globally produced during 2022 [39]. About 9.6 million tonnes of pomelos were produced during 2022 (Figure 1). The pomelo peel extract accounts for 19.56% of the total fresh fruit weight [91].
Li et al. [92] analyzed the total concentration of pesticides in the pomelo waste parts (epicarp and seeds of pomelo fruit). They reported that the epicarp (0.216 mg/kg) contained the highest than the mesocarp (0.0095 mg/kg), endocarp (0.0044 mg/kg), seed (0.0038 mg/kg), and pulp (0.0011 mg/kg). The reported concentrations were below the ADI limit.
Pomelo peels contain heavy metals like Cd and Ni (Table 1), which are below the maximum level set by the European Commission [56].
Pu et al. [93] assessed pomelo seed oil cytotoxicity in human liver LO2 and human liver cancer Hep G2 [HEPG2] cells. They reported that IC50 values of the Pomelo seed oils were in the range of 16.31 and 28.8 mg/mL, which is acceptable since the non-cytotoxic compounds have IC50 values greater than 1 mg/mL. The cytotoxicity dose of the Majia pomelo seed oil concentration tested in human liver cancer HepG2 cells was in the range of 500–4,000 μg/mL, which were also found to be non-toxic.
3.3 Mandarin
The etoxazole concentration (0.010∼0.637 mg/kg), which is a type of pesticide in citrus (Citrus reticulata Blanco) peel, was found larger than its pulp part (0.010–0.011 mg/kg). However, the chronic dietary risk of etoxazole in this fruit peel was in an acceptable range (0.035–0.951%) of the daily consumption recommended dosage [75].
Rossi et al. [94] studied the cytotoxic effects of EO extracted from Citrus deliciosa fruit peels harvested at different stages of maturation (immature, intermediate, and mature) against HT-29 cells. They reported that the peel extract from mature fruit showed significant cytotoxic properties (IC50 of 110 μg/mL). Hence, bioactive compounds present in these fruit peels like monoterpenes and citronellol can kill or damage cells, and slow or stop the development of rapidly proliferating cancer cells [95].
3.4 Lemon
Lemon fruit structure is divided into albedo, flavedo, and pulp. The albedo and flavedo are by-products of lemon during its juice production, which are the main sources of pectin, cellulose, EOs, and pigments. Moreover, the albedo is rich in flavonoids like hesperidin and eriocitrin [96]. The heavy metal concentrations analyzed in kaffir lime peel considered were Cr, Co, Ni, Pb, Cd, Pb, and Hg as shown in Table 1. In this study, all except Hg (Hg = 0.0145 mg/kg) were reported below the detection limit [97]. However, other toxicity studies on fruit peels are scarcely studied. Hence, the valorization of the lemon fruit peel should be done with further investigations of toxicities.
3.5 Grape
Grapefruit is the fifth globally produced fruit with 74.9 million tons (Figure 1). The fruit skins, stems, and seeds of grapefruit account for about 20%, mostly found as pomace during grape juice processing [33].
Grape skin extract contained potential safety hazards such as pesticides (cyprodinil, dimethomorph, and feamoxadone), mycotoxins (OTA), biogenic amines (ethanolamine and ethylamine), and heavy metals (Cd and Pb) [35]. In particular, Moncalvo et al. [35] investigated the pesticide residues (cyprodinil, dimethomorph, and feamoxadone) as well as heavy metals in grape skin powders and extracts. The concentration of these pesticide residues shown in Table 2 is below the minimum recommended limit set by the European Union (cyprodinil = 15, dimethomorph = 15, and feamoxadone = 10 mg/kg). Similarly, the heavy metal concentrations are also below the minimum recommended limit (Pb = 0.23 and Cd = 0.23 mg/kg). The biogenic amines analyzed in waste grape skin powders were also found below the toxicity level. The mycotoxin (OTA = 0.1–0.32 × 10−3 mg/kg) content in grape skin powders was also below the maximum residue levels for ochratoxin (0.01 mg/kg). Furthermore, from the review reported by Georganas et al. [98], the potential hazards found in grape pomace were heavy metals such as As, Pb, Cd, and Ni), toxins like OTA, and biogenic amines.
Proanthocyanidins are part of polyphenols which are used as plant pigments and are found in grape seeds and skins. Proanthocyanidins found in bioactive extracts from grape seeds and skins were studied for their acute oral toxicity, genotoxicity, and lethal dose 50 (LD50). It is reported that their LD50 is greater than 5,000 mg/kg and up to 2,000 mg/kg did not show genotoxicity (micronucleated erythrocytes for 72 h treatments) [99,100]. Moreover, during daily administration of 1,420 mg polyphenols enriched with ellagitannin extracts for 4 weeks, human safety was also found safe [101]. Figure 3 depicts some toxicant components present in grapefruit pomace.

Toxicant elements and chemicals in grapefruit by-product (pomace).
4 Toxicological qualities of tropical fruit by-products
Tropical fruit by-products considered here are the peel, pomace and/or seed from avocado, pineapple, banana, papaya, watermelon, and melon fruits.
Tropical fruit by-products contained hazard heavy metals. de Matuoka e Chiocchetti et al. [45] studied the heavy metals (Co and Cr) present in banana, papaya, and pineapple peels, summarized in Table 1. The amounts are above the range of hazard quotient (Cr = 0.81–3.18 mg/kg and Co = 0.03–0.09 mg/kg) [2]. Moreover, the heavy metals analyzed in pineapple and banana peels were As, Cd, Sn, Hg, and Pb. In both peels, low concentrations of these heavy metals were reported (Table 1). In particular, the As, Sn, Hg, and Pb were <1 × 10−3 mg/kg [44].
Hassan et al. [40] studied heavy metal concentrations in avocado seeds and watermelon silage and considered Cr and Ni concentrations. They reported that both fruit by-products contained insignificant (negligible) Ni. However, the Cr concentrations in avocado seeds and watermelon silage were 0.57–2.29 and 4.65 mg/kg.
The anti-nutritional contaminants present in avocado seeds were tannins, alkaloids, phytates, and oxalates, the reported concentrations are summarized in Table 2 [43]. Moreover, the ANFs reported in pineapple, banana, and watermelon fruit peels were oxalates, HCN, alkaloids, and phytates, and their concentrations are presented in Table 2. The reported values for HCN in these fruit peels are below the threshold value (below 3,500 mg/kg) reported as the safety limit [60].
The amount of toxic substances summarized in this review can vary from place to place and in different measurement techniques applied. Hence, the reported toxicant components imply that the by-products should be valorized with the application of further toxicant reduction techniques.
4.1 Avocado
Avocados have nine million tons of global production (Figure 1). Avocados contain peel (7–15%) and stone (including the seed) (14–24%) waste parts of the total fresh fruit weight. These waste parts are sources of many functional components such as antioxidants, bio-oil, bioethanol, etc. [102].
The heavy metals analysis considering Co and Pb in avocado seeds was reported as insignificant [43]. Hence, avocado fruit seeds can be valorized for many applications.
Cytotoxicity study conducted on avocado agro-industrial by-products (peel and seed) of two varieties (Hass and Fuerte) did not exhibit a cytotoxicity effect on RAW 264.7 cells. Specifically, the cytotoxicity test was performed at different concentrations ranging from 0.1 to 100 μg/mL of the fruit varieties peels and seeds extract and concentrations up to 10 μg/mL have no cytotoxicity effect [103].
Other toxicant compositions in avocado fruit by-products are scarcely studied. Hence, further toxicity studies on these by-products are required to fully valorize them.
4.2 Pineapple
Pineapples have about 29.4 million tons of global production. Therefore, much of its by-products can be valorized into valuable products. The pineapple fruit waste accounts for 60% of the weight of pineapple fruit interns of peeled skin, core, crown end, etc. [104].
As, Cd, Cr, Co, and Pb were found in pineapple peels (Table 1) [44,45]. The As concentration is almost insignificant, whereas the Cr and Co amounts are considerable. Hence, careful treatment is required before valorization.
The main reasons for mycotoxins development in fruit by-products are the refrigeration temperature conditions and water activity during storage. In a study on mycotoxins toxicity (aflatoxin B2, aflatoxin G1, and OTA) levels (shown in Table 2) in pineapple shells were determined below the detection limits (2.9 × 10−3 mg/kg) [77].
Oxalates and phytic acid concentrations in pineapple pomace are negligible (Table 2), which assures its applicability for value-added products [69].
Stępień et al. [78] reported the pineapple skin infected with mycotoxin mainly containing fusarium (250 mg/kg). The US FDA mycotoxin exposure guidelines state that the total fusarium in cereal foods should be 2–4 mg/kg. Hence, the reported result can be hazardous to human health without proper pretreatment.
Wanwimolruk et al. [79] analyzed pesticide residues in unpeeled pineapples and reported that considering carbaryl (0.262 mg/kg), carbofuran (1.43 mg/kg), fenobucarb (0.01 mg/kg), isoprocarb (0.313 mg/kg), and propachlor (0.015 mg/kg). Moreover, they analyzed the peeled pineapples which contained pesticide residues much lower than the unpeeled.
In general, pineapple skins are not safe for valorizing into food products as well as for animal forages without detoxification of their toxicants. Other toxicological qualities of fruit by-products are scarcely studied.
4.3 Banana
According to the FAOSTAT database [22], bananas were the first globally produced fruit in 2022 (Figure 1). During the banana processing, the peel part accounts for about 35%. Hence, this percentage of the banana fruit is wasted throughout the processing unless it can be valorized.
The heavy metals analyzed in banana peels were As, Cd, Cr, Co, and Pb, and their concentrations are summarized in Table 1 [36,44,45]. In all the analyzed reports, the concentrations of the heavy metals are below the range of the hazardous limits [2].
Šeremet et al. [105] studied the cytotoxicity of banana peel extract on cells of tongue epithelium (CAL 27), colon epithelial cells (Caco-2), and liver cells (HepG2) in a range of 0.014–10 mg/mL extract concentration and treatment times of 0.5, 1, and 2 h. They reported that polysaccharides containing banana peel aqueous extract has no cytotoxic/proliferative effect on epithelium (CAL 27) and liver cells (HepG2) at any of the tested concentrations or treatment times, whereas 10 mg/mL of the polysaccharides-free aqueous extract has shown slight proliferative effect in all three treatment times on the colon epithelial cells (Caco-2).
Fusarium proliferatum is a fungal species that causes crown rot in banana fruits mainly occurring during infection on postharvest [106]. Studies have shown that fumonisin B1 can contaminate banana peels during postharvest [106,107]. Gomes et al. [65] reviewed pesticide concentrations in bananas. Accordingly, they reported that pesticides such as chlorpyrifos and methiocarb are within the minimum residual limit set by Codex. Moreover, Mohd Zaini et al. [66] summarized the anti-nutrient composition of banana peel processing using different processing conditions (Table 2). From their report, the phytate, alkaloids, oxalate, and glycoside content in the banana processed by fermentation has shown the maximum concentrations compared to the banana processed by microwave drying, boiling, and air and oven drying methods. A large concentration of glycosides was found in banana peel [108]. Cyanogenic glycosides are the main precursor of HCN, which can be converted through hydrolysis. Glycosides are carcinogens and HCN are also toxic substance, which are formed during the chemical reaction of acids with metal cyanides [66]. Furthermore, the banana and papaya peels contain about 900 and 500 mg/kg of tannins [71]. This value could be varied due to different conditions. Hence, careful reduction mechanisms of banana fruit peel contaminants are required during valorization.
4.4 Papaya
Papayas have 13.8 million tons of global production in 2022 as shown in Figure 1 [22]. The waste part (by-product) of papaya rind and seeds accounts for 10–20% [33]. Hence, much of the fruit waste can be valorized into valuable products.
Availability of heavy metals such as As, Cd, Cr, Co, Ni, and Pb were reported in papaya fruit by-products, and their concentrations are summarized in Table 1 [45,48,49]. Except for the Cr, which is slightly higher, all the reported heavy metals were below the range of the hazardous limits [2].
Kumar et al. [48] investigated the presence of higher nickel in a mature papaya peel (13.2 mg/kg) than in young peel (7.96 mg/kg), mature seed (7.41 mg/kg), and yang seed (5.99 mg/kg). Moreover, about 7.36 and 6.41 mg/kg of chromium in mature and young peel were determined, respectively. Similarly, the cobalt analyzed in mature seeds and young peels were 0.15 and 0.4 mg/kg, respectively. In another study reported by Vinha et al. [49], the heavy metal such as chromium, cobalt, arsenic, cadmium, nickel, and lead were determined as summarized in Table 1.
The ANFs such as tannins, oxalates, and phytate analyzed in pawpaw (Carica papaya) seed flour are presented in Table 2 [76]. Toxicants in papaya seed such as phytates (3.04%), glucosinolates (10%), tannins (6.35%), and isothiocyanate (0.03%) were analyzed as dry weight of defatted seed meal. Compared to other toxicants glucosinolates account for the highest proportion of the fruit seed. Besides, the presence of isothiocyanate in the papaya seed oil implies the thioglucosinolate present in the seed hydrolysis to some degree by the thioglycosidase enzyme [109,110]. Thus, the presence of these toxicants limits the use of papaya seed and its oil for animal or human consumption unless further processing to remove these toxicants could be adopted. Pineapple skin contains larger (597 mg/kg) total toxic metal contents than orange peel, watermelon rind, banana peel, apple pomace, strawberry pomace, and grape pomace, in all of which it is below 50 mg/kg [14].
4.5 Watermelon
Watermelon fruits have global production of 100 million tons in 2022 [22], which is the second most highly produced fruit. The watermelon fruit contains a high amount of water (91%) and sugar (6%). From its total weight of analysis, it consists of rind and seeds, which account for 40–45% [111].
Heavy metal concentrations of Citrullus lanatus (watermelon) and Citrullus colocynthis (egusi melon) seeds varieties were reported considering selenium (13–28 mg/kg), cadmium (0.008–0.1 mg/kg), and lead (0.06–0.09 mg/kg). These heavy metal concentrations are below the ADI level (Pb = 0.21–0.25 and Cd = 0.06–0.07 mg/day) developed by FAO/WHO regulations [55].
In a study conducted on pesticide residues (diazinon, dimethoate, and metalaxyl) in flesh and the flesh plus peel (rind) of watermelon samples, the dimethoate was found in higher concentration (diazinon = 2.1 × 10−5 mg/kg, dimethoate = 1.95 × 10−3 mg/kg, and metalaxyl = 2.9 × 10−5 mg/kg) in the rind part. However, this was much lower than the recommended MRL (dimethoate = 2 × 10−2 mg/kg) [82]. Hence, properly treated watermelon rind could be the main source of bioactive compound recovery, functional, nutraceutical, and industrial applications [112,113].
Jyothi Lakshmi and Kaul [81] investigated the anti-nutritional quality of watermelon whole meal seeds. They reported that the phytate (9,900 mg/kg), tannin (32 × 105 mg/kg), and oxalate (2,130 mg/kg). These amounts could cause health effects and require pretreatment of the watermelon by-products before consumption or valorization.
4.6 Melon
About 28.6 million tons of melon and other related fruit products were produced globally in 2022 (Figure 1). Melon by-products mainly peels and seeds, which account for 58–62% of the raw material, are discarded as residue. These waste parts are rich sources of nutritional qualities, bioactive compounds, and other valuable products [114]. Therefore, many of these fruit by-products could be valorized into valuable products.
Anti-nutritional contaminants such as saponins, oxalates, phytates, and tannins were found in melon by-products, and their concentrations are presented in Table 2 [17]. Other hazardous heavy metals, toxicant organic compounds, mycotoxins, and pesticides/fungicides were scarcely studied on fruit melon by-products. Hence, further studies on these issues are advisable for better valorization.
5 Toxicological qualities of fruit berries
Fruit berries contain by-products that can be valorized into many products. The inedible fractions of blueberries, cranberries, raspberries, and strawberries from different literature data were reviewed by De Laurentiis et al. [115]. They reported that fruit berries such as blueberries (9–15%), cranberries (14–17%), raspberries (0%), and strawberries (2–6%) fractions were found. Except for the blueberries and strawberries, other fruit berry by-product toxicity analysis was scarcely studied. Therefore, further investigations on toxicity analysis are required before valorizing them.
Some studies indicated that the berry fruit by-products are susceptible to mycotoxins. In particular, mycotoxins like AOH, monomethyl ether, tentoxin, aflatoxins, and OTA were found in fruit berry by-products [116]. Moreover, some fungicides such as carbendazim and thiophanatemethyl (Table 2) were found beyond the MRL developed by European Food Safety et al. [85], which are carbendazim (0.1 mg/kg) and thiophanatemethyl (0.1 mg/kg).
5.1 Blueberries
Shotyk [46] studied the heavy metals in wild blueberries and raspberries considering the metals such as Cd, Cr, and Co, whose amounts are summarized in Table 1. They reported that the raspberries contained slightly lower toxic elements considering the established limit of 0.01 mg/100 g (according to Regulation (EC) No. 396/2005 of the European Parliament and Council).
The chlorpyrifos-methyl concentration (4.27 × 10−3 mg/kg) in the blueberry bluecrop variety was reported below the established limit of 0.01 mg/100 g (according to the Regulation (EC) No. 396/2005 of the European Parliament and Council) [67]. Moreover, other blueberries from Serbia contained thiametoxan and azoxytrobin (shown in Table 2) below the abovementioned established limit [68].
5.2 Strawberries
The Cr (1.47 × 10−6 – 3.41 × 10−6 mg/kg) and Ni (2.33 × 10−6 – 3.56 × 10−6 mg/kg) analyzed in strawberry fruits grown in different mediums containing different minerals were found to be below the maximum daily intake limit (Ni = 0.002 mg/kg and Cr = 1.5 mg/kg) [54].
Shao et al. [53] studied pesticides, phthalates, and heavy metals in strawberries grown in Shanghai, China. Phthalates such as bis-2-ethylhexyl phthalate (DEHP), diisobutyl phthalate (DIBP), and dibutyl phthalate (DBP), as well as heavy metal residues like Pb, Cd, and Ni were detected in strawberry pomace. Moreover, the dominantly detected pesticides were procymidone, acetamiprid, boscalid, and carbendazim and their concentrations are summarized in Table 2. They reported that the pesticides, DEHP, DIBP, and DBP, as well as the lead, cadmium, and nickel, were below the estimated daily intake.
Strawberry press-cake is the main source of ellagitannins and dietary fiber, which are important for human health. Total pesticides (2.143 mg/kg) mainly containing fungicides and insecticides are present in this by-product. The ADI (% ADI) of pesticides in the ellagitannins should be between 0.2 and 4.1% [80]. Hence, this strawberry press-cake is in the acceptable range for a human dose, which is equivalent to 100 g of strawberries.
The MRL of some fungicides such as carbendazim (0.1 mg/kg), thiabendazole (0.05 mg/kg), imazalil (0.05 mg/kg), thiophanatemethyl (0.1 mg/kg), and insecticides such as λ-cyhalothrin (0.01 mg/kg), carbofuran (0.05 mg/kg), formethanate (0.05 mg/kg), and fenoxicarb (0.05 mg/kg) in strawberries was developed by European Food Safety et al. [85]. Except for the carbendazim (0.1 mg/kg) and thiophanatemethyl (0.1 mg/kg) in strawberries, which were reported equivalent to the MRL, all the analyzed fungicides and pesticides were below the MRLS [80]. Hence, since the concentrations of the above fungicides and pesticides can vary from area to area as well as during measurements, their reduction mechanisms is very much required during valorization.
6 Toxicological qualities of other fruits by-products
Many other fruit by-products listed below are also produced globally. According to the study reported by De Laurentiis et al. [115], conducted on the amount of fruit purchased and related unavoidable wastes by EU households in 2010, apples (1.2 metric tons), apricots (0.03 metric ton), cherries (0.05 metric ton), and sour cherries (0.01 metric ton) were reported as waste.
Some hazardous metals, mycotoxins, toxicant organic compounds, ANFs, and pesticides/fungicides that are present in fruit by-products were reported. For instance, pesticides such as thiram, chlorpyrifos, and methyl parathion were found in apple and mango fruit peels [9].
6.1 Cherry
Cherries have a global production of 2.8 million tons in 2022 that is shown in Figure 1 [22]. However, about 0.05 metric tons of fruits are unavoidable wastes [115]. Hence, many of these by-products can be valorized into many valuable products.
Pesticide residues in sweet Cherry pits analyzed beyond the MRL set by the European Commission, 2005 (0.01 mg/kg for imidacloprid and phosmet) were phosmet (0.023–0.439 mg/kg) and imidacloprid (0.029 mg/kg). However, many of the other types of pesticide residuals were reported below the maximum residual limit (Table 2). The total pesticide residues in this fruit seeds were reported ranging from 0.139 to 2.544 mg/kg [117].
Mateus et al. [117] analyzed the availability of mycotoxins in sweet cherry pits. They reported that none of the mycotoxins from the analyzed nine mycotoxins aflatoxins (AFs), ochratoxin A (OTA), fumonisins (FBs), zearalenone (ZEN), and trichothecenes (T-2) were found in the sweet cherry pits.
The valorization methods of cherry by-products should consider a further analysis of hazardous metals, mycotoxins, pesticides, ANFs, and toxicant organic components, since limited studies have been conducted.
6.2 Apple
Apple is the third globally most-produced with 95.8 million tons in 2022 (Figure 1). During the study conducted on the amount of fruit purchased and related unavoidable wastes by EU households in 2010, apples had 1.2 metric tons of unavoidable waste [115]. During the apple processing, its pomace, peel, and seeds are found as by-products.
Some studies have reported that apple peels contain heavy metals such as Cr and Cd. In particular, the heavy metal concentrations (Cu = 3.7, Zn = 4.13, Cr = 2.25, and Cd = 0.002 mg/kg) analyzed in apple peel were below the safety qualification for agricultural products except for chromium (Cr = 0.5 mg/kg) [41]. Moreover, heavy metals (Cd, Cr, Ni, and Pb) found in apple pomace were analyzed below the permissible limits to implicate their non-toxicity during biogas production (Table 1) [37].
Fruit by-products may contain naturally occurring plant toxins like cyanogenic glycosides, which include AMG. In particular, the AMG composition studied in apple seeds (from 1,000 to 4,000 mg/kg) is higher than its pulp fruit juice. Hence, excessive ingestion of this fruit seed can cause sub-acute cyanide poisoning. However, fruit processing methods such as crushing, fermentation, boiling, soaking, and drying help to reduce the cyanide contents in such fruits [57]. Similarly, the naphthaleneacetic acid residue on seven varieties of apple skin was found to be 43.3 mg/kg [61].
From the review reported by Georganas et al. [98], the potential hazards found in apple by-products were AMG, pesticides (e.g., neonicotinoids and arsenic-based pesticides), patulin. The AMG is found in the apple seeds, which can cause acute cyanide poisoning in humans with the consumption of about 800 g of apple pomace. Moreover, the pesticide residues like neonicotinoids and acetamiprid were detected in apple pomace. Fungicides such as thiophanate, carbendazim, and pyrimethanil in apple pomace were detected in apple pomace although the reported amounts were below the toxicity level set by the United States environmental protection agency [118].
Pavicich et al. [59] investigated Alternaria mycotoxins such as AOH, AME, AME 3-sulfate (AME-3-S), altenuene (ALT), tenuazonic acid (TeA), tentoxin (TEN), alternariol 3-glucoside (AOH-3-G), AME 3-glucoside (AME-3-G), and altertoxin-I (ATX-I), alternariol 3-sulfate (AOH-3-S) present in apple by-products (pomace). These by-products passed through different processing (grinding, clarification, centrifugation, and water evaporation) were found to contain AME-3-S, and AME-3-G (Table 2). Alternaria mycotoxins are considered relatively stable and their contamination levels can be reduced during apple concentrate processing listed above.
The ANFs reported in apple fruit peels were oxalates, HCN, alkaloids, and phytates, and their concentrations are summarized in Table 2. The reported value for HCN in this fruit peel is below the threshold value (below 3,500 mg/kg) reported as the safety limit [60].
Pesticides such as boscalid and deltamethrin mainly concentrate in the apple skin rather than in the pulp part. These pesticides can be removed employing thermal processing. However, pesticides like acetamiprid cannot be reduced by drying, wet pasteurization, and frying processes except by using the lyophilization method [58].
The overall analysis of the reported data on the toxicity of apple fruit by-products is healthy for further valorization. However, due to these data varying from production place to place and from measurement to measurement, the valorization method should be done concurrently with toxicity investigations.
6.3 Mango
Mangoes and their related fruits have a global production of 59.2 million tons in 2022 (Figure 1). Mango processing has by-products of mainly peel and stone, which account for 45% of the total fresh fruit weight.
de Matuoka e Chiocchetti et al. [45] investigated the heavy metals (Co = 28 and Cr = 0.33 mg/kg) present in mango fruit peels. The reported amounts are above the range of hazard quotient Cr = 0.81–3.18 mg/kg and Co = 0.03–0.09 mg/kg, respectively [2]. Hence, a careful reduction method of these heavy metals is required.
Mango peels contain higher phytic acid (254.8 mg/kg) and oxalate (724 mg/kg) than its pulp part [70]. Moreover, mango seed kernels contain a large amount (153 × 103 mg/kg) of tannins [71]. The ANFs reported in mango fruit peels were oxalates, HCN, alkaloids, and phytates, whose concentrations are summarized in Table 2. The reported HCN concentration in mango fruit peels is below the threshold value (below 3,500 mg/kg) reported as the safety limit [60]. Moreover, the anti-nutritional contaminants present in mango kernel flour were tannins, phytic acid, oxalates, and HCN. The reported concentrations (mg/kg) for tannins, phytic acid, oxalates, and HCN were 1027.4, 1149.8, 213.4, and 0.00, respectively [63]. These ANFs are below the safety limit stated above.
6.4 Plum
The plums and sloes together have a global production of 12.4 million tons in 2022 (Figure). According to the study conducted on the amount of fruit purchased and related unavoidable wastes by EU households in 2010, from the 0.58 metric tons of plums and sloes purchased, about 0.04 metric tons were reported as unavoidable waste [115].
Mohammadi-Moghaddam et al. [52] analyzed heavy metals such as As, Cd, Ni, Hg, and Pb in the black plum peel. They reported that the Hg, Cd, and Pb were absent, whereas As and Ni were 1.2 mg/kg and 2.8 mg/kg, respectively. Moreover, Akter et al. [51] reported the heavy metal such as As, Cd, Co, Ni, Pb, and Hg concentrations in plum kernels. They reported that the As and Hg concentrations were <0.1 mg/kg. These heavy metal concentrations were reported as below the tolerable upper intake level recommended by many Food Safety authorities.
Other toxicity reports were scarcely reported. Hence, further studies are required to fully valorize the plum fruit by-products.
6.5 Peaches
Peaches and nectarines together have a global production of 26.4 million tons in 2022 as shown in Figure 1. According to the study conducted on the amount of fruit purchased and related unavoidable wastes by EU households in 2010, from the 1.85 metric tons of peaches and nectarines purchased, about 0.16 metric tons were reported as unavoidable waste [115].
The mineral content of peach fruit homogenized with its peel and pulp was analyzed. The heavy metals considered in this study were reported as chromium (Cr = 0.17–1.38 mg/kg) and lead (Pb < 0.10 mg/kg). These results are below the maximum permissible limit value (2.3 mg/kg) for human consumption [50].
Other toxicity properties of the peach fruit by-products are scarcely investigated. Hence, further studies are required for better valorization of the by-products.
6.6 Apricot
Apricots have a global production of 3.9 million tons in 2022 (Figure 1). According to the study conducted on the amount of fruit purchased and related unavoidable wastes by EU households in 2010, from the 0.4 metric tons of apricot fruit purchased, about 0.03 metric tons were reported as unavoidable waste [115]. Hence, the unavoidable waste can be valorized into valuable products.
Heavy metals such as cobalt (Co), cadmium (Cd), and lead (Pb) were studied in apricot kernel and pomace. The heavy mineral concentration reported in the apricot kernel and pomace are summarized in Table 1. However, the concentration of lead (Pb) in these by-products is reported as insignificant [42]. The AMG content in apricot seeds (AMG = 52,000 mg/kg) did not induce any effect in rabbit spermatozoa conducted in vivo. In particular, the dose of apricot seeds (3.0 mg AMG/kg body weight) on the rabbit spermatozoa parameters did not induce any change [62].
The anti-nutritional contaminants present in apricot kernel flour were tannins, phytic acid, oxalates, and HCN. Moreover, these ANFs were investigated in raw peach fruit kernel flour. The concentrations of these ANFs are presented in Table 2 [63].
The aflatoxins B1 and B2 present in apricot kernels were measured in the range of 0.0017–22.451 mg/kg. In this study, removing the discolored kernels was able to remove 97.3–99.5% of the total aflatoxins [64].
Generally, the apricot by-products have less toxicity although further confirmation tests are important during valorization.
7 Current trends on fruit by-product toxicant reduction
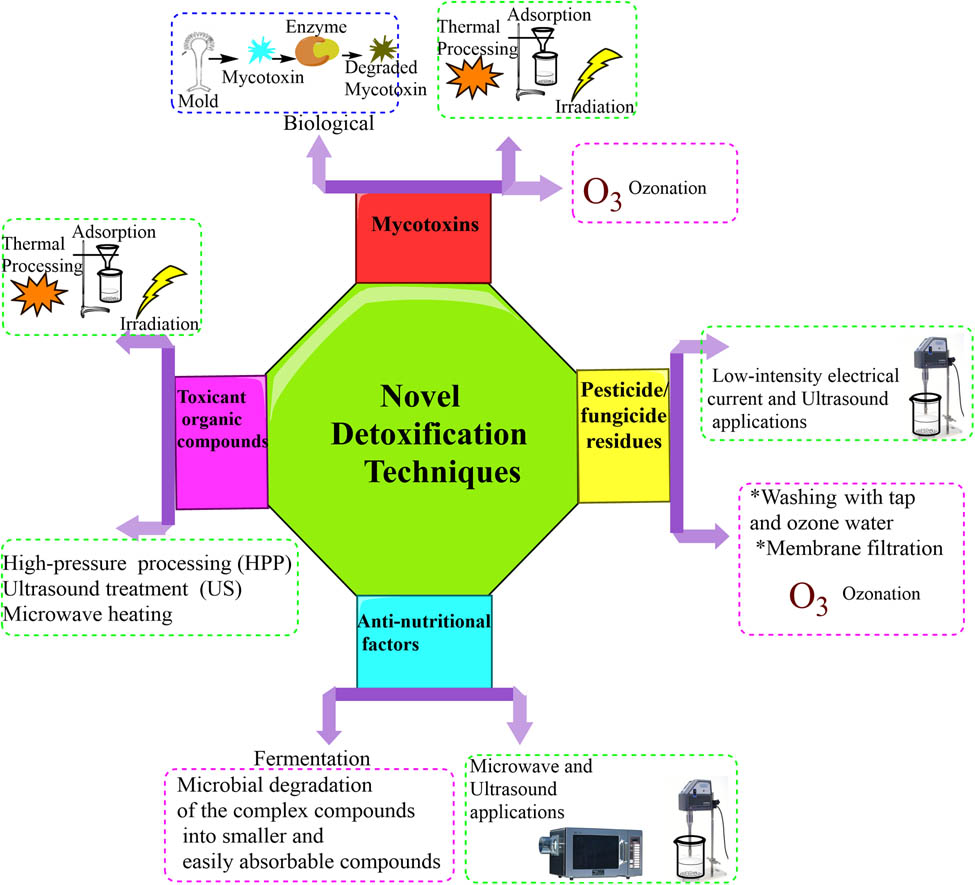
Several foods worldwide are contaminated by pesticides and mycotoxins due to the pollution of fruits, vegetables, and cereals [119,120,121,122,123,124]. Pesticide reduction constitutes one of the sustainable development goals. Gavahian and Cullen [125] reported on the use of innovative food processing technologies, including high-pressure processing (HPP), pulsed electric fields (PEF), cold plasma (CP), supercritical carbon dioxide, and ultrasound (USN) processing as those with good potential for mycotoxin and pesticide reduction. They depend on processing parameters, the type of pesticide/mycotoxin, and the food matrix. Some of the thermal, chemical, and non-thermal mechanisms of toxicant reduction are depicted in Figure 4.

Methods for fruit by-product toxicity reduction.
In the same context, Adebo et al. [126] studied novel non-thermal food processing techniques, particularly HPP, PEF, CP, and USN processing for the decontamination of mycotoxins in food with complete decontamination of mycotoxins in some cases. They also discussed the mechanisms by which reduction/elimination occurs. This takes place through the decomposition of toxins after collision with ions/electrons. Cleavage of bonds, structural degradation of the mycotoxin structure, and cleavage of functional groups are the immediate effects after this decomposition. Other mechanisms include Photolysis/photolytic damage leading to an attack on double bonds and/or heterocyclic moieties in the mycotoxin molecule, dihydroxylation, dehydrogenation, modification of terminal furan ring/alteration, and hydrolysis of lactone ring.
Natural and chemical decontamination of mycotoxins leading to significantly reduced levels in foods with no generation of degradable toxic by-products has been discussed by Agriopoulou et al. [127].
Prevention strategies should also be followed throughout the food production chain. Management should take place before any fungal infestation; the second step of control should be during the period of fungal invasion of plant material and mycotoxin production; and the third step should be initiated when the agricultural products have been identified as heavily contaminated as reported by Čolović et al. [128].
Wang et al. [129] suggested that protective agents, including plant extracts, yeast products, bacteria, peptides, enzymes, H2, oligosaccharides, amino acids, adsorbents, vitamins, and selenium could reduce effectively DON-induced organ toxicity.
Fu et al. [130] showed that ZEN exposure resulted in oxidative stress and ferroptosis by glutathione-dependency. Moreover, melatonin supplement through enhanced productions of glutathione peroxidase 4 and glutathione alleviate ZEN-induced abnormalities. Similarly, Zhang et al. [131] discussed the detoxification of AFB1 in ducks’ primary hepatocytes by the key glutathione S-transferase (GST) isozymes.
EOs with broad-range antimicrobial effectiveness, low toxicity, and diverse mechanisms of action have been discussed by Prakash et al. [132] with detailed mechanistic understanding, safety profile, and risk assessment. Singh et al. [133] referred to the use of Coleus aromaticus EO, with thymol as the major compound as a natural antimicrobial agent against food-spoilage bacteria and A. flavus and AFB1 contamination to extend the shelf-life of food products.
Toxic mechanisms of multiple mycotoxins have been reviewed in the editorial New insight into mycotoxins and bacterial toxins [134].
Lowering of pesticide residue levels in foods can be carried out by washing, blanching, peeling, thermal treatments, alkaline electrolyzed water washing, CP, ultrasonic cleaning, ozone treatment, and enzymatic treatment [135,136]. Significance of precision agricultural practices and integrated pest management (IPM) techniques has been mentioned in the review by Munir et al. [137].
Organic farming methods have been demonstrated to lower the amount of pesticides consumed through food [138]. In addition, IPM techniques – integrating chemical, biological, cultural, and physical approaches to control pests – reduce insecticide applications by 95% while leading to the preservation or increase in crop yields by conserving wild pollinators [139].
Different hormones and internal and external factors influence plant mechanisms ultimately effecting mycotoxin growth, which could be affected by antioxidants and fertilizers available during plantation [140]. For instance, the applications of sodium nitroprusside and melatonin for the distraction of heavy metal stress improve antioxidant response during plantation of crops [141,142].
Dietary fibers and prebiotics recovery from fruits and vegetable wastes and by-products display important biological activities, such as gut microbiota modulation, lowering the glycemic load. These have been mentioned in the review by Pop et al. [143], who also addressed aspects, such as recovery and extraction procedures, characterization, and utilization in different food matrixes.
Proper waste management practices, including waste reduction, safe handling, and appropriate treatment should be followed to alleviate from climate change, environmental degradation, and human health problems. Circularity and sustainable growth are solutions to these problems along with thermophilic microbes in the bioremediation of waste as reported by Najar et al. [144]. These thermophiles emphasize biotechnology and industrial bioprocess progressions toward the build-up of a zero-carbon maintainable bio-economy [145]. Since thermophiles (heat-loving bacteria) can endure extremely high temperatures, they are a major source of various industrial and biotechnological applications with production of enzymes such as amylase, cellulase, protease, xylanases, pullulanases, pectinases, chitinases, esterases, dehydrogenases, and isomerases [146,147].
Thermophilic microbes have been utilized as sources of therapeutic agents, in the food industries, bioremediation, and valorization [148,149].
Moreover, hazardous pollutant remediation from contaminated environments could be carried out by myco-remediation, a green and eco-friendly tool for pollution management as reported by Navina et al. [150], Bhattacharya et al. [151], and Kalia et al. [152]. This is due to the abundance in hyphal network, heavy metal resistance, generation of hydrolytic and degradative enzymes, high surface area to volume ratio, site for metal-binding proteins, high stability and flexibility toward different temperatures and pH. A wide range of contaminants, including pesticides, hydrocarbons, heavy metals, and various synthetic substances can be removed or reduced. Bioremediation refers to the use of organisms for elimination or reduction in pollutants [122].
Three different mechanisms for expunging the environmental pollutants and initiating a balance in the environment are followed by fungi and include bioconversion, biodegradation, and biosorption. Myco-remediation includes the involvement of fungi in the myco-extraction process to remove heavy metals from polluted materials. Fungi accumulate heavy metals and then extraction of heavy metals takes place along with their secure disposal from their biomass [153].
8 Novel detoxification techniques
The consumption of foods comprising phytonutrients provides a concurrent anti-oxidation, metal chelation, anti-inflammation, and genes activation related to detoxification. These arrangements are significant in stabilizing the cellular attack by heavy metals and persistent organic pollutants (POPs). As an illustration, the consumption of green tea appears to be a possible dietary line to detoxify heavy metals and POPs. In green tea, the well-investigated flavonoid (epigallocatechin-3-gallate, EGCG) established these protective actions. EGCG triggers the Nrf2-intervened detoxifying and anti-oxidant enzyme like glutathione peroxidase and GST [154]. By arylhydrocarbon receptor suppression and of Nrf2-regulated genes stimulation, Newsome et al. [155] reported that green tea EGCG decreases PCB 126 toxicities. Therefore, green tea consumption seems to be a potential dietary approach to detoxify heavy metals and POPs. On the other hand, it has been stated that quercetin plays a crucial role as a modulator of phase I and phase II detoxifying enzymes. Its molecular structure possesses three conceivable chelating sites, which explicate why it may act as a natural chelator of heavy metals and creates complex with transition metal ions [156]. An animal study displayed that quercetin powerfully regularized the As-induced toxicity in the liver and brain, and Al-induced oxidative harm in the brain [157]. Through its chelation, free radical scavenging, anti-inflammation, and trigger of anti-oxidant detoxification enzymes by upregulation of Nrf2 pathway, García-Niño and Pedraza-Chaverrí [158] confirmed that curcumin defends against heavy metal-induced liver harm. Similar trends reported the same protective effect of curcumin against Cd, As, and cisplatin toxicity [159]. An in vivo study concluded that curcumin repressed 2,3,7,8-tetrachlorodibenzo-p-dioxin-induced irregular intracellular signaling cascade of cytochrome P450 [157].
Many studies demonstrated the application of novel methods for detoxifications of toxicants in fruit and vegetable by-products. Some of the techniques are low-intensity electrical current and USN applications [160], membrane filtration [161] and application of free chlorine coupled with USN process for pesticide/fungicide residues detoxification [162], microwave, and USN applications for the removal of ANFs [16], and biological detoxification of mycotoxins [163] (Figure 4). Most studies addressed the applications of novel detoxifications for mycotoxins and pesticide/fungicide residues. However, novel technologies for the detoxification of heavy metals (As, Cd, Co, Cr, Ni, Pb, and Hg), toxicant organic compounds, and ANFs are scarcely studied. For instance, Adebo et al. [126] summarized novel non-thermal food processing techniques for mycotoxin reduction, which lead to decomposition of toxins occurred due to collision with ions/electrons by causing cleavage of bonds, degrade structural properties of the mycotoxins, and cleavage of its functional groups. Biological methods such as surface binding by extracellular polymeric substances, degradation by enzyme, and cellular metabolism are potent to reduce toxic metabolites originate from fungi [163]. Moreover, nanotechnology (nanoparticles), PEF, applying plant extracts, and using omics are being alternative solutions to chemical and physical methods for the distraction of mycotoxins in plant products after harvesting [164].
Novel technologies like low-intensity electrical current, membrane filtration, microwave, and USN applications are being more preferable to physicochemical techniques. The physicochemical techniques, are less supportive for mycotoxin detoxification due to causing loss of nutrients, require expensive equipment, and the process can be lengthy [164,165]. Hence, these novel technologies are being applicable for detoxification and preparation of fruit by-products during industrial exploitation.
In plants, consecutive hydroxylation, glycosylation and demethylation of fungal phytotoxins can evade plant cell death and stun the fungal invader [166]. In this line, detoxification of fungal phytotoxins through biotransformation by plants should be a significant plant defensive mechanism against fungal pathogens [166]. By the hydroxylase produced by cruciferous plants like Brassica napus, the Destruxin B was detoxified into hydroxydestruxin B [166]. According to Makhuvele et al. [167], ellagic acid and curcumin could avoid the AFB1 metabolism and develop the activity of GST involved in the detoxification of xenobiotics. In addition, numerous glycosyltransferases of mycotoxins existed in plants. A UDP-glucosyltransferase involved in the detoxification of DON was revealed from rice (Oryza sativa) [168]. The main detoxification reactions of Alternaria toxins were glycosylation and glucuronidation by plants [168]. In this line, the transformation pathways of Alternaria toxins are (1) glycosylation, (2) hydroxylation, (3) reduction, and (4) methylation and demethylation.
In corn plants, both 15-monoacetoxyscirpenol (15-MAS) and 4,15-diacetoxyscirpenol (4,15-DAS) were, respectively, converted to 15-MAS 3-glucoside and 4,15-DAS 3-glucoside, which are called masked mycotoxins [169]. Regarding citrinin, the chief transformation pathways are (1) decarboxylation: the citrinin was transformed to decarboxycitrinin and (2) oxido-reduction: the citrinin was converted to dihydrocitrinone [169]. In relation to the detoxification of trichothecenes, numerous biotransformation pathways: (1) Glycosylation or glucuronidation, (2) epimerization, (3) de-epoxidation, and (4) hydrolysis [169].
Studies on the removal of pesticide residues from vegetable samples were underscored although limited studies were conducted on fruit by-products [170]. In particular, fungicides and insecticides can be removed by washing with tap and ozone water, ultrasonic cleaning, which could be alternative to other chemical methods using acid/alkali [162]. Moreover, Yang et al. [171] studied the mechanism for the removal of pesticide residues from fresh vegetables by applying free chlorine coupled with USN process. They reported that from 20 to 40 kHz ultrasonic frequency and at 25 mg/L free chlorine concentration, the damage for the vegetable qualities was negligible. In a study reported by Cengiz et al. [27], combined low-intensity electrical current and USN applications were found to be novel techniques to reduce pesticide residues from lettuce samples. This technique was found effective at a current of 1,400 mA and USN frequency of 24 kHz at 10 min. At these combined conditions, the captan, thiamethoxam and metalaxyl residues were reduced by 92.57, 81.99, and 93.09%, respectively.
The hydroxylation usually takes place on pesticide detoxification [172]. For instance, the carbon hydroxylation convoluted in detoxification of chlorsulfuron and triasulfuron by CYP71C6v1 in wheat [173], triasulfuron, fluometuron, linuron, and diuron by CYP71A10 in soybean [174] and pesticide pelargonic acid by CYP72A18 in rice [175]. In addition, the plants Tau and Phi strongly contribute in the pesticides detoxification [176]. In this regard, biophysical and crystallographic valuations exposed that there are at least two ligand–binding sites in GSTs counting the G–site and GSH–binding region [177]. In certain leguminous species, β-alanine substitutes glycine to produce GSH analog homoglutathione (hGSH) [178]. In alfalfa, hGSH replacement of chlorine in atrazine was measured as one of the most essential detoxification pathways [178]. HmGSH conjugates of atrazine and acetochlor are also associated with detoxification of the pesticides in rice [178]. Both hmGSH and hGSH have a nucleophilic thiol group on cysteine residues which can act together with pesticides on their electrophilic metabolites, rendering them detoxified [178]. Additionally, pesticides methylation was described to be crucial for their detoxification in plants [179]. In Medicago sativa, C–methylation of an ethyl group on atrazine has been elucidated [178].
ANFs can be minimized by applying novel detoxification technologies and using fermentation [16,180]. Novel detoxification techniques like microwave heating are being applicable to reduce heat labile ANFs such as phytic acid, trypsin inhibitors, tannins, saponins and oxalate in food products [16] as shown in Figure 4.
9 Novel functional foods from fruit by-products free from toxication
Many functional foods can be derived from different food by-products. Fruits contain vitamins, antioxidants, minerals, and dietary fiber.
Globally, 14% of food is lost during harvest. Food loss and waste affect food security and nutrition negatively and significantly lead to greenhouse gas emissions, environmental pollution, degradation of natural ecosystems, and biodiversity loss (https://www.fao.org/policy-support/policy-themes/food-loss-food-waste/en/).
Same for fruit waste where despite the reduction in fruit waste, there is still a need for an improvement in bio-waste utilization.
One such example is pineapple due to its excellent organoleptic quality and nutritional quality and good source of phenolic compounds. Pineapple by-products are used mainly in animal feed and the pharmaceutical industry and constitute 29–40% shell, 9–10% core, 2–6% stem, and 2–4% crown, representing approximately 50% (w/w) of the total weight of the pineapple [77]. The by-products contain dietary fiber, vitamins, minerals, phenolic compounds, and other bioactive compounds [181,182,183,184]. Natural sources of dietary fiber, antioxidants, pectin, enzymes, organic acids, food additives, EOs, etc., through different methods of extractions, purifications, and fermentations could be derived from pineapple waste utilization as reported by Roda and Lambri [185].
Peaches are primarily consumed fresh; however, some peaches are being processed and used as animal feed or fertilizers, or disposed of in landfills from peach by-products (PB) consisting of pulp and peel/skin, constituting 15–28% of the initial weight [186,187]. These by-products could be converted into higher-value byproducts, such as bioplastics at approximately $1,000 per ton of biomass [188]. Peach juice by-products are rich in bioactive compounds, particularly polyphenols, and can produce food additives, antioxidants for pharmaceuticals and cosmetics, and fermentable sugars for bioproducts [189].
Some challenges that need to be taken into account include the composition of the by-products from fruit processing, which depends on the fruit and the stage of processing [186]. Another significant problem might be the underestimation of the real antioxidant capacity and potential of the feedstocks arising from the ineffective characterization of the phenolic content in the extraction of the non-extractable phenolic compounds [177]. The polyphenol profile of both extractable polyphenols and hydrolyzable polyphenols (HPP) from peach by-products and peach peels has been determined [217,218].
Sample preparation and storage conditions are additional factors that need to be carefully designed to prevent perishability and preserve the bioactive components. Reduction in the water content to below 15% is suitable for maintenance of the microbiological quality of dehydrated vegetables while achieving volume reduction [219].
García-Aparicio et al. [220] aimed to assess the PB, generated at the pulp refinement stage for juice concentrate production, as feedstock for a biorefinery for production of fermentable sugars and novel functional products simultaneously. Different conventional and novel enzymatic extraction methods were applied to extract extractable and non-extractable bioactive compounds such as oven drying and freeze drying. They proposed that the solid residue enriched in recalcitrant phenolic compounds and proteins could be used to develop novel functional products for food/feed sectors. Table 3 presents the use of some fruit by-products in the food industry (Adapted from Teshome et al. [28]). Moreover, Table 4 presents medicinal and pharmaceutical use from the exploitation of fruit by-products.
Uses of some fruit by-products in the food industry
| Fruit by-products | Functional foods | References |
|---|---|---|
| Apple pomace | Dietary fiber source in baked foods, chicken-meat-based sausages, and yogurt products, stabilizer for oil-water emulsions | [190,191,192] |
| Avocado by-product and avocado peels | Antioxidants, antimicrobials, and food additives such as colorants, flavorings, thickening agents, and functional beverage formulation | [193,194] |
| Banana peel | Antioxidant, antibacterial, antifungal activity, reduction in blood sugar, lower cholesterol, and show anti-angiogenic activity and neuro-protective effect, synthesis of bio-inspired silver nanoparticles | [195,196] |
| Citrus peel | Source of molasses, pectin, oil, and limone, thickener, emulsifier, and stabilizer in many foods, pectin is a suitable polymeric matrix for edible films for active food pack by-product | [197,198] |
| Grape pomace | Meat and fish derivatives containing grape pomace powders, fiber in bakery products, oil from grape seed | [199,200,201] |
| Mango peel | Antioxidant and dietary fiber in macaroni, sources of phytochemicals in biscuits, edible films | [202,203,204,205] |
| Pineapple peel, core and stem | Pineapple peel is a rich source of sugar that can be used as a nutrient in fermentation processes, core can be used in pineapple juice concentrates, vinegar, and wine production, bromelain enzyme extracted from the pineapple stem used as a meat tenderizer, bread dough improver | [206,207] |
Medicinal and pharmaceutical used from the exploitation of fruit by-products
| Fruit by-products | Medicinal and pharmaceutical exploitation | References |
|---|---|---|
| Apple peel | Reduces metabolic syndrome and atherogenic progression | [208] |
| Avocado peel | Inhibitor for the inflammation mediator nitric oxide by a possible reduction of free radicals during inflammation, anticancer, antidiabetic, and antihypertensive effects | [103,209] |
| Banana peel | Antioxidant, antibacterial, antifungal activity, reduce blood sugar, lower cholesterol, and show anti-angiogenic activity and neuro-protective effect, silver nanoparticles, which are used as antimicrobials to pathogenic fungi | [195,210] |
| Citrus pulp and seed | d-limonene showing a therapeutic effect on lung cancer in mice and breast cancer in mice and rats | [210,211] |
| Mango | Anti-inflammatory and antioxidative properties during in vivo studies related to obesity, diabetes, cardiovascular disease, and skin cancer, reduction of carcinogenesis | [212,213,214] |
| Peach kernel | Phenols, carotenoids, and cyanogenic glycosides of peach kernel possess antidiabetic, antioxidative, and anti-aging properties | [215] |
Adapted from Teshome et al. [28].
10 Conclusion
Many of the prioritized fruit by-products have lower toxicity levels, which are analyzed in terms of heavy metals (As, Cd, Co, Cr, Ni, Pb, and Hg), mycotoxins, toxicant organic compounds, ANFs, and pesticide/fungicide residues. However, for full valorization of these by-products, pre-treatment mechanisms should be applied to reduce their toxicity. A holistic revalorization of these by-products with regard to major components, such as fermentable sugars, and value-added components such as the phenolic compounds is required. Details regarding composition, bioactive compounds profile (free and bound compounds), antioxidant capacity, and the impact of the drying process are essential for the development of processes and technologies for their reuse, and targeting of industrial sectors for their exploitation. Mycotoxin detoxification and pesticide reduction mechanisms and control strategies have been discussed as being highly beneficial for the development of food safety and security. The heavy metals, mycotoxins, toxicant organic compounds, anti-nutritional contaminants, and fungicide/pesticide residues of some by-products of fruits prioritized in our study were scarcely studied, Hence, toxicity analysis of each of these fruit by-products demands further investigations.
-
Funding information: Authors state no funding involved.
-
Author contribution: Conceptualization: Z.T. and T.V.; methodology: Z.T. and T.V; software: Z.T.; validation: Z.T. and T.V.; formal analysis: Z.T. and T.V.; investigation: Z.T. and T.V.; resources: Z.T. and T.V.; data curation: Z.T. and T.V.; writing – original draft preparation: Z.T. and T.V.; writing – review and editing: Z.T., S.S., and T.V.; visualization: Z.T., T.V., S.S., and T.V.; project administration: T.V.; funding acquisition: T.V. All authors accepted the responsibility for the entire content of the manuscript and consented to its submission, reviewed all the results, and approved the final version of the manuscript.
-
Conflict of interest: Authors state no conflict of interest.
-
Data availability statement: Data sharing is not applicable to this article as no datasets were generated or analyzed during the current study.
References
[1] Pushparaj K, Meyyazhagan A, Pappuswamy M, Mousavi Khaneghah A, Liu WC, Balasubramanian B. Occurrence, identification, and decontamination of potential mycotoxins in fruits and fruit by-products. Food Front. 2023;4(1):32–46. 10.1002/fft2.198.Search in Google Scholar
[2] Abuzed Sadee B, Jameel Ali R. Determination of heavy metals in edible vegetables and a human health risk assessment. Environ Nanotechnol Monit Manag. 2023;19:100761. 10.1016/j.enmm.2022.100761.Search in Google Scholar
[3] Gebrekidan A, Weldegebriel Y, Hadera A, Van der Bruggen B. Toxicological assessment of heavy metals accumulated in vegetables and fruits grown in Ginfel river near Sheba Tannery, Tigray, Northern Ethiopia. Ecotoxicol Environ Saf. 2013;95:171–8. 10.1016/j.ecoenv.2013.05.035.Search in Google Scholar PubMed
[4] Saqib S, Zaman W, Ayaz A, Habib S, Bahadur S, Hussain S, et al. Postharvest disease inhibition in fruit by synthesis and characterization of chitosan iron oxide nanoparticles. Biocatal Agric Biotechnol. 2020;28:101729. 10.1016/j.bcab.2020.101729.Search in Google Scholar
[5] Saqib S, Zaman W, Ullah F, Majeed I, Ayaz A, Hussain Munis MF. Organometallic assembling of chitosan-iron oxide nanoparticles with their antifungal evaluation against Rhizopus oryzae. Appl Organomet Chem. 2019;33(11):e5190. 10.1002/aoc.5190.Search in Google Scholar
[6] Nepal B, Stine KJ. Glycoalkaloids: Structure, properties, and interactions with model membrane systems. Process. 2019;7(8):513, https://www.mdpi.com/2227-9717/7/8/513.10.3390/pr7080513Search in Google Scholar
[7] Park BK, Kwon SH, Yeom MS, Joo KS, Heo MJ. Detection of pesticide residues and risk assessment from the local fruits and vegetables in Incheon, Korea. Sci Rep. 2022;12(1):9613. 10.1038/s41598-022-13576-5.Search in Google Scholar PubMed PubMed Central
[8] Salgado N, Silva MA, Figueira ME, Costa HS, Albuquerque TG. Oxalate in foods: Extraction conditions, analytical methods, occurrence, and health implications. Foods. 2023;12(17):3201, https://www.mdpi.com/2304-8158/12/17/3201.10.3390/foods12173201Search in Google Scholar
[9] Liu B, Han G, Zhang Z, Liu R, Jiang C, Wang S, et al. Shell thickness-dependent Raman enhancement for rapid identification and detection of pesticide residues at fruit peels. Anal Chem. 2012;84(1):255–61. 10.1021/ac202452t.Search in Google Scholar PubMed
[10] Uddin R, Thakur MU, Uddin MZ, Islam GMR. Study of nitrate levels in fruits and vegetables to assess the potential health risks in Bangladesh. Sci Rep. 2021;11(1):4704. 10.1038/s41598-021-84032-z.Search in Google Scholar PubMed PubMed Central
[11] Santos CMd, Abreu CMPd, Freire JM, Queiroz EdR, Mendonça MM. Chemical characterization of the flour of peel and seed from two papaya cultivars. Food Sci Technol. 2014;34(2):353–7.10.1590/fst.2014.0048Search in Google Scholar
[12] WHO. The public health impact of chemicals: Knowns and unknowns. WHO document production services, Geneva, Switzerland; 2016.Search in Google Scholar
[13] Hong C, Jia Y, Yang X, He Z, Stoffella P. Assessing lead thresholds for phytotoxicity and potential dietary toxicity in selected vegetable crops. Bull Environ Contam Toxicol. 2008;80(4):356–61.10.1007/s00128-008-9375-zSearch in Google Scholar
[14] Kuppusamy S, Venkateswarlu K, Megharaj M. Evaluation of nineteen food wastes for essential and toxic elements. Int J Recycl Org Waste Agric. 2017;6(4):367–73.10.1007/s40093-017-0178-2Search in Google Scholar
[15] Oyeyinka BO, Afolayan AJ. Comparative evaluation of the nutritive, mineral, and antinutritive composition of Musa sinensis L. (Banana) and Musa paradisiaca L. (Plantain) Fruit Compartments. Plants. 2019;8(12):598.10.3390/plants8120598Search in Google Scholar
[16] Suhag R, Dhiman A, Deswal G, Thakur D, Sharanagat VS, Kumar K, et al. Microwave processing: A way to reduce the anti-nutritional factors (ANFs) in food grains. LWT. 2021;150:111960. 10.1016/j.lwt.2021.111960.Search in Google Scholar
[17] Silva MA, Albuquerque TG, Alves RC, Oliveira MBPP, Costa HS. Melon (Cucumis melo L.) by-products: Potential food ingredients for novel functional foods? Trends Food Sci Technol. 2020;98:181–9. 10.1016/j.tifs.2018.07.005.Search in Google Scholar
[18] Dhanisha SS, Drishya S, Guruvayoorappan C. Pithecellobium dulce induces apoptosis and reduce tumor burden in experimental animals via regulating pro-inflammatory cytokines and anti-apoptotic gene expression. Food Chem Toxicol. 2022;161:112816. 10.1016/j.fct.2022.112816.Search in Google Scholar PubMed
[19] Erukainure OL, Msomi NZ, Beseni BK, Salau VF, Ijomone OM, Koorbanally NA, et al. Cola nitida infusion modulates cardiometabolic activities linked to cardiomyopathy in diabetic rats. Food Chem Toxicol. 2021;154:112335. 10.1016/j.fct.2021.112335.Search in Google Scholar PubMed
[20] Inanc ME, Gungor S, Yeni D, Avdatek F, Ipek V, Turkmen R, et al. Protective role of the dried white mulberry extract on the reproductive damage and fertility in rats treated with carmustine. Food Chem Toxicol. 2022;163:112979. 10.1016/j.fct.2022.112979.Search in Google Scholar PubMed
[21] Shendge AK, Panja S, Basu T, Ghate NB, Mandal N. Ameliorating effects of white mulberry on iron-overload-induced oxidative stress and liver fibrosis in Swiss albino mice. Food Chem Toxicol. 2021;156:112520. 10.1016/j.fct.2021.112520.Search in Google Scholar PubMed
[22] FAOSTAT. Food and agricultural data. FAOSTAT; 2022, https://www.fao.org/faostat/en/#data.Search in Google Scholar
[23] Jiménez-Moreno N, Esparza I, Bimbela F, Gandía LM, Ancín-Azpilicueta C. Valorization of selected fruit and vegetable wastes as bioactive compounds: Opportunities and challenges. Crit Rev Environ Sci Technol. 2020;50(20):2061–108. 10.1080/10643389.2019.1694819.Search in Google Scholar
[24] Hasan MM, Islam MR, Haque AR, Kabir MR, Khushe KJ, Hasan S. Trends and challenges of fruit by-products utilization: insights into safety, sensory, and benefits of the use for the development of innovative healthy food: a review. Bioresour Bioprocess. 2024;11(1):10. 10.1186/s40643-023-00722-8.Search in Google Scholar PubMed PubMed Central
[25] Tsegay ZT, Mulaw G. Antimicrobial activities and mode of action of bioactive substances from vegetable and fruit byproducts as a current option for valorization. Waste Biomass Valoriz. 2024;16:1–28. 10.1007/s12649-024-02587-0.Search in Google Scholar
[26] Vilas-Boas AA, Pintado M, Oliveira ALS. Natural bioactive compounds from food waste: toxicity and safety concerns. Foods. 2021;10(7):1564.10.3390/foods10071564Search in Google Scholar
[27] Gomes S, Vieira B, Barbosa C, Pinheiro R. Evaluation of mature banana peel flour on physical, chemical, and texture properties of a gluten-free Rissol. J Food Process Preserv. 2022;46(8):e14441. 10.1111/jfpp.14441.Search in Google Scholar
[28] Teshome E, Teka TA, Nandasiri R, Rout JR, Harouna DV, Astatkie T, et al. Fruit by-products and their industrial applications for nutritional benefits and health promotion: a comprehensive review. Sustainability. 2023;15(10):7840.10.3390/su15107840Search in Google Scholar
[29] Yu J, Ahmedna M. Functional components of grape pomace: their composition, biological properties and potential applications. Int J Food Sci Technol. 2013;48(2):221–37. 10.1111/j.1365-2621.2012.03197.x.Search in Google Scholar
[30] Choonut A, Saejong M, Sangkharak K. The production of ethanol and hydrogen from pineapple peel by Saccharomyces cerevisiae and Enterobacter aerogenes. Energy Procedia. 2014;52:242–9. 10.1016/j.egypro.2014.07.075.Search in Google Scholar
[31] Tsegay ZT, Gebreegziabher ST-B, Mulaw G. Nutritional qualities and valorization trends of vegetable and fruit byproducts: a comprehensive review. J Food Qual. 2024;2024(1):5518577. 10.1155/2024/5518577.Search in Google Scholar
[32] Czech A, Zarycka E, Yanovych D, Zasadna Z, Grzegorczyk I, Kłys S. Mineral content of the pulp and peel of various citrus fruit cultivars. Biol Trace Elem Res. 2020;193(2):555–63. 10.1007/s12011-019-01727-1.Search in Google Scholar PubMed PubMed Central
[33] Sagar NA, Pareek S, Sharma S, Yahia EM, Lobo MG. Fruit and vegetable waste: Bioactive compounds, their extraction, and possible utilization. Compr Rev Food Sci Food Saf. 2018;17(3):512–31. 10.1111/1541-4337.12330.Search in Google Scholar PubMed
[34] Czech A, Malik A, Sosnowska B, Domaradzki P. Bioactive substances, heavy metals, and antioxidant activity in whole fruit, peel, and pulp of citrus fruits. Int J Food Sci. 2021;2021(1):6662259. 10.1155/2021/6662259.Search in Google Scholar PubMed PubMed Central
[35] Moncalvo A, Marinoni L, Dordoni R, Duserm Garrido G, Lavelli V, Spigno G. Waste grape skins: evaluation of safety aspects for the production of functional powders and extracts for the food sector. Food Addit Contam: Part A. 2016;33(7):1116–26. 10.1080/19440049.2016.1191320.Search in Google Scholar PubMed
[36] Saleem M, Saeed MT. Potential application of waste fruit peels (orange, yellow lemon and banana) as wide range natural antimicrobial agent. J King Saud Univ - Sci. 2020;32(1):805–10. 10.1016/j.jksus.2019.02.013.Search in Google Scholar
[37] Bożym M, Florczak I, Zdanowska P, Wojdalski J, Klimkiewicz M. An analysis of metal concentrations in food wastes for biogas production. Renew Energy. 2015;77:467–72. 10.1016/j.renene.2014.11.010.Search in Google Scholar
[38] Neshovska H. Determination of heavy metal content (CD and PB) in citrus feed raw material. Tradit Mod Vet Med. 2023;1(14):59–64.Search in Google Scholar
[39] Mateus AR, Marino-Cortegoso S, Barros SC, Sendón R, Barbosa L, Pena A, et al. Citrus by-products: A dual assessment of antioxidant properties and food contaminants towards circular economy. Innov Food Sci Emerg Technol. 2024;95:103737. 10.1016/j.ifset.2024.103737.Search in Google Scholar
[40] Hassan M, Belanche A, Jiménez E, Rivelli I, Martín-García AI, Margolles A, et al. Evaluation of the nutritional value and presence of minerals and pesticides residues in agro-industrial by-products to replace conventional ingredients of small ruminant diets. Small Rumin Res. 2023;229:107117. 10.1016/j.smallrumres.2023.107117.Search in Google Scholar
[41] Wang Q, Liu J, Cheng S. Heavy metals in apple orchard soils and fruits and their health risks in Liaodong Peninsula, Northeast China. Environ Monit Assess. 2014;187(1):4178. 10.1007/s10661-014-4178-7.Search in Google Scholar PubMed
[42] Tareen AK, Panezai MA, Sajjad A, Achakzai JK, Kakar AM, Khan NY. Comparative analysis of antioxidant activity, toxicity, and mineral composition of kernel and pomace of apricot (Prunus armeniaca L.) grown in Balochistan, Pakistan. Saudi J Biol Sci. 2021;28(5):2830–9. 10.1016/j.sjbs.2021.02.015.Search in Google Scholar PubMed PubMed Central
[43] Okoye OOFNC. Comparative study of the constituents of the fruits pulps and seeds of Canarium ovatum, Persea americana and Dacryodes edulis. Jordan J Chem (JJC). 2017;12(2):113–25.Search in Google Scholar
[44] Aboul-Enein AM, Salama ZA, Gaafar AA, Aly HF, Abou-Elella F, Ahmed HA. Identification of phenolic compounds from banana peel (Musa paradaisica L.) as antioxidant and antimicrobial agents. J Chem Pharm Res. 2016;8(4):46–55.Search in Google Scholar
[45] de Matuoka e Chiocchetti G, De Nadai Fernandes EA, Bacchi MA, Pazim RA, Sarriés SR, Tome TM. Mineral composition of fruit by-products evaluated by neutron activation analysis. J Radioanal Nucl Chem. 2013;297(3):399–404. 10.1007/s10967-012-2392-8.Search in Google Scholar
[46] Shotyk W. Trace elements in wild berries from reclaimed lands: Biomonitors of contamination by atmospheric dust. Ecol Indic. 2020;110:105960. 10.1016/j.ecolind.2019.105960.Search in Google Scholar
[47] Simeon EO, Amamilom NS, Azuka IW. Metal assessment and phytochemical screening of orange fruit (Citrus sinensis) seeds and peels. J Pharmacogn Phytochem. 2018;7(3):709–14.Search in Google Scholar
[48] Kumar SS, V GT, K K, John M. Antioxidant potential and mineral elemental profiling of young and mature fruit and leaf of Carica papaya L. cultivar ‘Red Lady’. J Trace Elem Miner. 2024;9:100166. 10.1016/j.jtemin.2024.100166.Search in Google Scholar
[49] Vinha AF, Costa A, Espírito Santo L, Ferreira DM, Sousa C, Pinto E, et al. High-value compounds in papaya by-products (Carica papaya L. var. Formosa and Aliança): Potential sustainable use and exploitation. Plants. 2024;13(7):1009.10.3390/plants13071009Search in Google Scholar
[50] Mihaylova D, Popova A, Desseva I, Petkova N, Stoyanova M, Vrancheva R, et al. Comparative study of early- and mid-ripening peach (Prunus persica L.) varieties: Biological activity, macro-, and micro-nutrient profile. Foods. 2021;10(1):164.10.3390/foods10010164Search in Google Scholar
[51] Akter S, Netzel ME, Fletcher MT, Tinggi U, Sultanbawa Y. Chemical and nutritional composition of terminalia ferdinandiana (Kakadu Plum) Kernels: A novel nutrition source. Foods. 2018;7(4):60.10.3390/foods7040060Search in Google Scholar
[52] Mohammadi-Moghaddam T, Firoozzare A, Kariminejad M, Sorahi M, Tavakoli Z. Black plum peel as a useful by-product for the production of new foods: chemical, textural, and sensory characteristics of Halva Masghati. Int J Food Prop. 2020;23(1):2005–19. 10.1080/10942912.2020.1835953.Search in Google Scholar
[53] Shao W-C, Zang Y-Y, Ma H-Y, Ling Y, Kai Z-P. Concentrations and related health risk assessment of pesticides, phthalates, and heavy metals in strawberries from Shanghai, China. J Food Prot. 2021;84(12):2116–22. 10.4315/JFP-21-165.Search in Google Scholar PubMed
[54] Tozzi F, Renella G, Cristina M, Masciandaro G, Gonnelli C, Colzi I, et al. Agronomic performance and food safety of strawberry cultivated on a remediated sediment. Sci Total Environ. 2021;796:148803. 10.1016/j.scitotenv.2021.148803.Search in Google Scholar PubMed
[55] Falade OS, Otemuyiwa IO, Adekunle AS, Adewusi SA, Oluwasefunmi O. Nutrient composition of watermelon (Citrullis lanatus (Thunb.) Matsum. & Nakai) and egusi melon (Citrullus colocynthis (L.) Schrad.) seeds. Agric Conspec Sci. 2020;85(1):43–9.Search in Google Scholar
[56] Commission regulation (EU). 2023/915 of 25 April 2023 on maximum levels for certain contaminants in food and repealing regulation (EC), 103–157 § 119; 2023.Search in Google Scholar
[57] Bolarinwa IF, Orfila C, Morgan MRA. Determination of amygdalin in apple seeds, fresh apples and processed apple juices. Food Chem. 2015;170:437–42. 10.1016/j.foodchem.2014.08.083.Search in Google Scholar PubMed
[58] Hrynko I, Kaczyński P, Pietruszyńska M, Łozowicka B. The effect of food thermal processes on the residue concentration of systemic and non-systemic pesticides in apples. Food Control. 2023;143:109267. 10.1016/j.foodcont.2022.109267.Search in Google Scholar
[59] Pavicich MA, De Boevre M, Vidal A, Iturmendi F, Mikula H, Warth B, et al. Fate of free and modified Alternaria mycotoxins during the production of apple concentrates. Food Control. 2020;118:107388. 10.1016/j.foodcont.2020.107388.Search in Google Scholar
[60] Romelle FD, Rani A, Manohar RS. Chemical composition of some selected fruit peels. Eur J Food Sci Technol. 2016;4(4):12–21.Search in Google Scholar
[61] Skinner RC, Gigliotti JC, Ku K-M, Tou JC. A comprehensive analysis of the composition, health benefits, and safety of apple pomace. Nutr Rev. 2018;76(12):893–909. 10.1093/nutrit/nuy033.Search in Google Scholar PubMed
[62] Kolesar E, Tvrda E, Halenar M, Schneidgenova M, Chrastinova L, Ondruska L, et al. Assessment of rabbit spermatozoa characteristics after amygdalin and apricot seeds exposure in vivo. Toxicol Rep. 2018;5:679–86. 10.1016/j.toxrep.2018.05.015.Search in Google Scholar PubMed PubMed Central
[63] Sorour M, Mehanni A-H, Hussein SM, Mustafa MA. Utilization of treated seed kernel flours of some fruits in biscuit manufacture. Eur J Nutr Food Saf. 2022;14(3):1–13.10.9734/ejnfs/2022/v14i330481Search in Google Scholar
[64] Zivoli R, Gambacorta L, Piemontese L, Solfrizzo M. Reduction of aflatoxins in apricot kernels by electronic and manual color sorting. Toxins. 2016;8(1).10.3390/toxins8010026Search in Google Scholar
[65] Gomes HO, Menezes J, da Costa J, Coutinho H, Teixeira R, do Nascimento RF. Evaluating the presence of pesticides in bananas: An integrative review. Ecotoxicol Environ Saf. 2020;189:110016. 10.1016/j.ecoenv.2019.110016.Search in Google Scholar PubMed
[66] Mohd Zaini H, Roslan J, Saallah S, Munsu E, Sulaiman NS, Pindi W. Banana peels as a bioactive ingredient and its potential application in the food industry. J Funct Foods. 2022;92:105054. 10.1016/j.jff.2022.105054.Search in Google Scholar
[67] Dorosh O, Fernandes VC, Delerue-Matos C, Moreira MM. Blueberry pruning wastes: From an undervalued agricultural residue to a safe and valuable source of antioxidant compounds for the food industry. Foods. 2024;13(2):317.10.3390/foods13020317Search in Google Scholar
[68] Milinčić DD, Vojinović UD, Kostić AŽ, Pešić MB, Špirović Trifunović BD, Brkić DV, et al. In vitro assessment of pesticide residues bioaccessibility in conventionally grown blueberries as affected by complex food matrix. Chemosphere. 2020;252:126568. 10.1016/j.chemosphere.2020.126568.Search in Google Scholar PubMed
[69] Nagarajaiah SB, Prakash J. Chemical composition and bioactivity of pomace from selected fruits. Int J Fruit Sci. 2016;16(4):423–43. 10.1080/15538362.2016.1143433.Search in Google Scholar
[70] Madalageri DM, Bharati P, Kage U. Physicochemical properties, nutritional and antinutritional composition of pulp and peel of three mango varieties. Int J Educ Sci Res. 2017;7:81–94.10.24247/ijesrjun201710Search in Google Scholar
[71] Melesse A, Steingass H, Schollenberger M, Rodehutscord M. Component composition, in vitro gas and methane production profiles of fruit by-products and leaves of root crops. J Agric Sci. 2018;156(7):949–58. 10.1017/S0021859618000928.Search in Google Scholar
[72] Sánchez-Pérez S, Comas-Basté O, Rabell-González J, Veciana-Nogués MT, Latorre-Moratalla ML, Vidal-Carou MC. Biogenic amines in plant-origin foods: Are they frequently underestimated in low-histamine diets? Foods. 2018;7(12):205.10.3390/foods7120205Search in Google Scholar
[73] de Vilhena Araújo É, Vendramini PH, Costa JH, Eberlin MN, Montagner CC, Fill TP. Determination of tryptoquialanines A and C produced by Penicillium digitatum in oranges: Are we safe? Food Chem. 2019;301:125285. 10.1016/j.foodchem.2019.125285.Search in Google Scholar PubMed
[74] Li Y, Jiao B, Zhao Q, Wang C, Gong Y, Zhang Y, et al. Effect of commercial processing on pesticide residues in orange products. Eur Food Res Technol. 2012;234(3):449–56. 10.1007/s00217-011-1651-1.Search in Google Scholar
[75] Wang Z, Pang J, Liao C, Zhang Q, Sun D. Determination of etoxazole in different parts of citrus fruit and its potential dietary exposure risk assessment. Chemosphere. 2021;268:128832. 10.1016/j.chemosphere.2020.128832.Search in Google Scholar PubMed
[76] Ibrahim S, Inelo ED, Eke MO. Physico-chemical, alveograph and anti-nutritional properties of breads formulated from wheat and pawpaw (Carica papaya) seed flour blends. Asian Food Sci J. 2021;20(3):72–85.10.9734/afsj/2021/v20i330280Search in Google Scholar
[77] Santos DI, Martins CF, Amaral RA, Brito L, Saraiva JA, Vicente AA, et al. Pineapple (Ananas comosus L.) by-products valorization: Novel bio ingredients for functional foods. Molecules. 2021;26(11):3216.10.3390/molecules26113216Search in Google Scholar
[78] Stępień Ł, Koczyk G, Waśkiewicz A. Diversity of Fusarium species and mycotoxins contaminating pineapple. J Appl Genet. 2013;54(3):367–80. 10.1007/s13353-013-0146-0.Search in Google Scholar PubMed PubMed Central
[79] Wanwimolruk C, Wanwimolruk S, Kuaykaimuk K, Buddhaprom J, Saenserm P, Soikham S. Food safety of Thailand’s pineapples, bananas, and dragon fruits from pesticide contamination: a study using GC-MS analysis. Philipp J Sci. 2022;151(6B):2315–26.10.56899/151.6B.02Search in Google Scholar
[80] Sójka M, Miszczak A, Sikorski P, Zagibajło K, Karlińska E, Kosmala M. Pesticide residue levels in strawberry processing by-products that are rich in ellagitannins and an assessment of their dietary risk to consumers. NFS J. 2015;1:31–7. 10.1016/j.nfs.2015.09.001.Search in Google Scholar
[81] Jyothi Lakshmi A, Kaul P. Nutritional potential, bioaccessibility of minerals and functionality of watermelon (Citrullus vulgaris) seeds. LWT - Food Sci Technol. 2011;44(8):1821–6. 10.1016/j.lwt.2011.04.001.Search in Google Scholar
[82] Wanwimolruk S, Kanchanamayoon O, Boonpangrak S, Prachayasittikul V. Food safety in Thailand 1: it is safe to eat watermelon and durian in Thailand. Environ Health Prev Med. 2015;20(3):204–15. 10.1007/s12199-015-0452-8.Search in Google Scholar PubMed PubMed Central
[83] Soares Mateus AR, Barros S, Pena A, Sanches-Silva A. Chapter Two - The potential of citrus by-products in the development of functional food and active packaging. In: Capanoglu E, Navarro-Hortal MD, Forbes-Hernández TY, Battino M, editors. Advances in food and nutrition research. Vol. 107. Academic Press; 2023. p. 41–90. 10.1016/bs.afnr.2023.06.001.Search in Google Scholar PubMed
[84] Socas-Rodríguez B, Mendiola JA, Rodríguez-Delgado MÁ, Ibáñez E, Cifuentes A. Safety assessment of citrus and olive by-products using a sustainable methodology based on natural deep eutectic solvents. J Chromatogr A. 2022;1669:462922. 10.1016/j.chroma.2022.462922.Search in Google Scholar PubMed
[85] European Food Safety A, Carrasco Cabrera L, Medina Pastor P. The 2020 European Union report on pesticide residues in food. EFSA J. 2022;20(3):e07215. 10.2903/j.efsa.2022.7215.Search in Google Scholar PubMed PubMed Central
[86] Liu Y, Heying E, Tanumihardjo SA. History, global distribution, and nutritional importance of citrus fruits. Compr Rev Food Sci Food Saf. 2012;11(6):530–45. 10.1111/j.1541-4337.2012.00201.x.Search in Google Scholar
[87] WHO/FAO/UNU (2007) Protein and amino acid requirements in human nutrition: report of a joint WHO/FAO/UNU expert consultation. WHO Technical Report Series 935. Geneva. FAO/WHO/UNU, 276.Search in Google Scholar
[88] Benayad O, Bouhrim M, Tiji S, Kharchoufa L, Addi M, Drouet S, et al. Phytochemical profile, α-glucosidase, and α-amylase inhibition potential and toxicity evaluation of extracts from citrus aurantium (L) peel, a valuable by-product from Northeastern Morocco. Biomolecules. 2021;11(11):1555.10.3390/biom11111555Search in Google Scholar
[89] Costa CA, Cury TC, Cassettari BO, Takahira RK, Flório JC, Costa M. Citrus aurantium L. essential oil exhibits anxiolytic-like activity mediated by 5-HT1A-receptors and reduces cholesterol after repeated oral treatment. BMC Complement Altern Med. 2013;13(1):42. 10.1186/1472-6882-13-42.Search in Google Scholar PubMed PubMed Central
[90] Teixeira F, Santos B, Nunes G, Soares JM, Amaral L, Souza G, et al. Addition of orange peel in orange jam: evaluation of sensory, physicochemical, and nutritional characteristics. Molecules. 2020;25(7):1670.10.3390/molecules25071670Search in Google Scholar
[91] Phuong TNQ, Van Hung P, Phi NTL. Extraction of flavonoids in pomelos’ peels using Box-Behnken response surface design and their biological activities. Vietnam Journal of Science. Technol Eng. 2021;63(2):52–7.10.31276/VJSTE.63(2).52-57Search in Google Scholar
[92] Li X, Song S, Wei F, Huang X, Guo Y, Zhang T. Occurrence, distribution, and translocation of legacy and current-use pesticides in pomelo orchards in South China. Sci Total Environ. 2024;913:169674. 10.1016/j.scitotenv.2023.169674.Search in Google Scholar PubMed
[93] Pu S, Liang R, Chen J, Liu C, Xu C, Chen M, Chen J. Characterization and evaluation of Majia pomelo seed oil: A novel industrial by-product. Food Chem Adv. 2022;1:100051. 10.1016/j.focha.2022.100051.Search in Google Scholar
[94] Rossi RC, da Rosa SR, Weimer P, Lisbôa Moura JG, de Oliveira VR, de Castilhos J. Assessment of compounds and cytotoxicity of Citrus deliciosa Tenore essential oils: From an underexploited by-product to a rich source of high-value bioactive compounds. Food Biosci. 2020;38:100779. 10.1016/j.fbio.2020.100779.Search in Google Scholar
[95] Bhuia MS, Aktar MA, Chowdhury R, Ferdous J, Rahman MA, Hasan MSA, et al. Therapeutic potentials of ononin with mechanistic insights: A comprehensive review. Food Biosci. 2023;56:103302. 10.1016/j.fbio.2023.103302.Search in Google Scholar
[96] Martínez-Zamora L, Cano-Lamadrid M, Artés-Hernández F, Castillejo N. Flavonoid extracts from lemon by-products as a functional ingredient for new foods: a systematic review. Foods. 2023;12(19):3687.10.3390/foods12193687Search in Google Scholar
[97] Lubinska-Szczygeł M, Kuczyńska-Łażewska A, Rutkowska M, Polkowska Ż, Katrich E, Gorinstein S. Determination of the major by-products of citrus hystrix peel and their characteristics in the context of utilization in the industry. Molecules. 2023;28(6):2596.10.3390/molecules28062596Search in Google Scholar
[98] Georganas A, Giamouri E, Pappas AC, Zoidis E, Goliomytis M, Simitzis P. Utilization of agro-industrial by-products for sustainable poultry production. Sustainability. 2023;15(4):3679.10.3390/su15043679Search in Google Scholar
[99] Lluís L, Muñoz M, Nogués MR, Sánchez-Martos V, Romeu M, Giralt M, et al. Toxicology evaluation of a procyanidin-rich extract from grape skins and seeds. Food Chem Toxicol. 2011;49(6):1450–4. 10.1016/j.fct.2011.03.042.Search in Google Scholar PubMed
[100] Sano A. Safety assessment of 4-week oral intake of proanthocyanidin-rich grape seed extract in healthy subjects. Food Chem Toxicol. 2017;108:519–23. 10.1016/j.fct.2016.11.021.Search in Google Scholar PubMed
[101] Heber D, Seeram NP, Wyatt H, Henning SM, Zhang Y, Ogden LG, et al. Safety and antioxidant activity of a pomegranate ellagitannin-enriched polyphenol dietary supplement in overweight individuals with increased waist size. J Agric Food Chem. 2007;55(24):10050–4. 10.1021/jf071689v.Search in Google Scholar PubMed
[102] García-Vargas MC, Contreras MD, Castro E. Avocado-derived biomass as a source of bioenergy and bioproducts. Appl Sci. 2020;10(22):8195.10.3390/app10228195Search in Google Scholar
[103] Tremocoldi MA, Rosalen PL, Franchin M, Massarioli AP, Denny C, Daiuto ÉR, et al. Exploration of avocado by-products as natural sources of bioactive compounds. PLoS One. 2018;13(2):e0192577.10.1371/journal.pone.0192577Search in Google Scholar
[104] Yusof Y, Yahya SA, Adam A. Novel technology for sustainable pineapple leaf fibers productions. Procedia CIRP. 2015;26:756–60. 10.1016/j.procir.2014.07.160.Search in Google Scholar
[105] Šeremet D, Durgo K, Jokić S, Huđek A, Vojvodić Cebin A, Mandura A, et al. Valorization of banana and red beetroot peels: Determination of basic macrocomponent composition, application of novel extraction methodology and assessment of biological activity in vitro. Sustainability. 2020;12(11):4539.10.3390/su12114539Search in Google Scholar
[106] Xie L, Yang Q, Wu Y, Xiao J, Qu H, Jiang Y, et al. Fumonisin B1 biosynthesis is associated with oxidative stress and plays an important role in Fusarium proliferatum infection on Banana Fruit. J Agric Food Chem. 2023;71(13):5372–81. 10.1021/acs.jafc.3c00179.Search in Google Scholar PubMed
[107] Xie L, Wu Y, Wang Y, Jiang Y, Yang B, Duan X, et al. Fumonisin B1 induced aggressiveness and infection mechanism of Fusarium proliferatum on banana fruit. Environ Pollut. 2021;288:117793. 10.1016/j.envpol.2021.117793.Search in Google Scholar PubMed
[108] Ozabor P, Ojokoh A, Wahab A, Aramide O. Effect of fermentation on the proximate and antinutrient composition of banana peels. Int J Biotechnol. 2020;9(2):105–17.10.18488/journal.57.2020.92.105.117Search in Google Scholar
[109] Goriainov S, Orlova S, Nikitina E, Vandishev V, Ivlev V, Esparza C, et al. Study of the chemical composition of Carica papaya L. seed oils of various geographic origins. Horticulturae. 2023;9(11):1227.10.3390/horticulturae9111227Search in Google Scholar
[110] Marcinkowska M, Jeleń HH. Inactivation of thioglucosidase from sinapis alba (White Mustard) seed by metal salts. Molecules. 2020;25(19):4363. 10.3390/molecules25194363.Search in Google Scholar PubMed PubMed Central
[111] Capossio JP, Fabani MP, Román MC, Zhang X, Baeyens J, Rodriguez R, et al. Zero-waste watermelon production through nontraditional rind flour: multiobjective optimization of the fabrication process. Processes. 2022;10(10):1984.10.3390/pr10101984Search in Google Scholar
[112] Zia S, Khan MR, Aadil RM, Medina-Meza IG. Bioactive recovery from watermelon rind waste using ultrasound-assisted extraction. ACS Food Sci Technol. 2024;4(3):687–99. 10.1021/acsfoodscitech.3c00601.Search in Google Scholar
[113] Zia S, Khan MR, Shabbir MA, Aadil RM. An update on functional, nutraceutical and industrial applications of watermelon by-products: A comprehensive review. Trends Food Sci Technol. 2021;114:275–91. 10.1016/j.tifs.2021.05.039.Search in Google Scholar
[114] Rolim PM, Seabra LMAJ, de Macedo GR. Melon by-products: biopotential in human health and food processing. Food Rev Int. 2020;36(1):15–38. 10.1080/87559129.2019.1613662.Search in Google Scholar
[115] De Laurentiis V, Corrado S, Sala S. Quantifying household waste of fresh fruit and vegetables in the EU. Waste Manag. 2018;77:238–51. 10.1016/j.wasman.2018.04.001.Search in Google Scholar PubMed
[116] Juan C, Mañes J, Font G, Juan-García A. Determination of mycotoxins in fruit berry by-products using QuEChERS extraction method. LWT. 2017;86:344–51. 10.1016/j.lwt.2017.08.020.Search in Google Scholar
[117] Mateus A, Barros SC, Cortegoso SM, Sendón R, Barbosa-Pereira L, Khwaldia K, et al. Potential of fruit seeds: Exploring bioactives and ensuring food safety for sustainable management of food waste. Food Chem: X. 2024;23:101718. 10.1016/j.fochx.2024.101718.Search in Google Scholar PubMed PubMed Central
[118] Lyu F, Luiz SF, Azeredo DRP, Cruz AG, Ajlouni S, Ranadheera CS. Apple pomace as a functional and healthy ingredient in food products: a review. Processes. 2020;8(3):319.10.3390/pr8030319Search in Google Scholar
[119] Cámara MA, Cermeño S, Martínez G, Oliva J. Removal residues of pesticides in apricot, peach and orange processed and dietary exposure assessment. Food Chem. 2020;325:126936. 10.1016/j.foodchem.2020.126936.Search in Google Scholar PubMed
[120] Campagnollo FB, Ganev KC, Khaneghah AM, Portela JB, Cruz AG, Granato D, et al. The occurrence and effect of unit operations for dairy products processing on the fate of aflatoxin M1: A review. Food Control. 2016;68:310–29. 10.1016/j.foodcont.2016.04.007.Search in Google Scholar
[121] Gomiero T. Food quality assessment in organic vs. conventional agricultural produce: Findings and issues. Appl Soil Ecol. 2018;123:714–28. 10.1016/j.apsoil.2017.10.014.Search in Google Scholar
[122] González N, Marquès M, Nadal M, Domingo JL. Occurrence of environmental pollutants in foodstuffs: A review of organic vs. conventional food. Food Chem Toxicol. 2019;125:370–5. 10.1016/j.fct.2019.01.021.Search in Google Scholar PubMed
[123] Khaneghah AM, Fakhri Y, Abdi L, Coppa C, Franco LT, de Oliveira C. The concentration and prevalence of ochratoxin A in coffee and coffee-based products: A global systematic review, meta-analysis and meta-regression. Fungal Biol. 2019;123(8):611–7. 10.1016/j.funbio.2019.05.012.Search in Google Scholar PubMed
[124] Nabizadeh S, Shariatifar N, Shokoohi E, Shoeibi S, Gavahian M, Fakhri Y, et al. Prevalence and probabilistic health risk assessment of aflatoxins B1, B2, G1, and G2 in Iranian edible oils. Environ Sci Pollut Res. 2018;25(35):35562–70. 10.1007/s11356-018-3510-0.Search in Google Scholar PubMed
[125] Gavahian M, Cullen PJ. Cold plasma as an emerging technique for mycotoxin-free food: Efficacy, mechanisms, and trends. Environ Food Rev Int. 2019;36(2):193–214. 10.1080/87559129.2019.1630638.Search in Google Scholar
[126] Adebo OA, Molelekoa T, Makhuvele R, Adebiyi JA, Oyedeji AB, Gbashi S, et al. A review on novel non-thermal food processing techniques for mycotoxin reduction. Int J Food Sci Technol. 2021;56(1):13–27. 10.1111/ijfs.14734.Search in Google Scholar
[127] Agriopoulou S, Stamatelopoulou E, Varzakas T. Advances in occurrence, importance, and mycotoxin control strategies: Prevention and detoxification in foods. Foods. 2020;9(2):137.10.3390/foods9020137Search in Google Scholar
[128] Čolović R, Puvača N, Cheli F, Avantaggiato G, Greco D, Đuragić O, et al. Decontamination of mycotoxin-contaminated feedstuffs and compound feed. Toxins (Basel). 2019;11(11):617. 10.3390/toxins11110617.Search in Google Scholar PubMed PubMed Central
[129] Wang P, Yao Q, Meng X, Yang X, Wang X, Lu Q, et al. Effective protective agents against organ toxicity of deoxynivalenol and their detoxification mechanisms: A review. Food Chem Toxicol. 2023;182:114121. 10.1016/j.fct.2023.114121.Search in Google Scholar PubMed
[130] Fu W, Dai C, Ma Z, Li Q, Lan D, Sun C, et al. Enhanced glutathione production protects against zearalenone-induced oxidative stress and ferroptosis in female reproductive system. Food Chem Toxicol. 2024;185:114462. 10.1016/j.fct.2024.114462.Search in Google Scholar PubMed
[131] Zhang Y, Cao KX, Niu QJ, Deng J, Zhao L, Khalil MM, et al. Alpha-class glutathione S-transferases involved in the detoxification of aflatoxin B1 in ducklings. Food Chem Toxicol. 2023;174:113682. 10.1016/j.fct.2023.113682.Search in Google Scholar PubMed
[132] Prakash B, Singh PP, Gupta V, Raghuvanshi TS. Essential oils as green promising alternatives to chemical preservatives for agri-food products: New insight into molecular mechanism, toxicity assessment, and safety profile. Food Chem Toxicol. 2024;183:114241. 10.1016/j.fct.2023.114241.Search in Google Scholar PubMed
[133] Singh PP, Jaiswal AK, Raghuvanshi TS, Prakash B. Insights into the antimicrobial efficacy of Coleus aromaticus essential oil against food-borne microbes: Biochemical and molecular simulation approaches. Food Chem Toxicol. 2023;182:114111. 10.1016/j.fct.2023.114111.Search in Google Scholar PubMed
[134] Nie T, Wu Q, Long M, Wu W, Kuca K. New insight into mycotoxins and bacterial toxins: Toxicity assessment, molecular mechanism and food safety (preface to the special issue of food and chemical toxicology on the outcomes of Myco & bacterial toxin). Food Chem Toxicol. 2024;188:114655. 10.1016/j.fct.2024.114655.Search in Google Scholar PubMed
[135] Mir SA, Dar BN, Mir MM, Sofi SA, Shah MA, Sidiq T, et al. Current strategies for the reduction of pesticide residues in food products. J Food Compos Anal. 2022;106:104274. 10.1016/j.jfca.2021.104274.Search in Google Scholar
[136] Pathak VM, Verma VK, Rawat BS, Kaur B, Babu N, Sharma A, et al. Current status of pesticide effects on environment, human health and its eco-friendly management as bioremediation: A comprehensive review [Review]. Front Microbiol. 2022;13:962619, https://www.frontiersin.org/journals/microbiology/articles/10.3389/fmicb.2022.962619.10.3389/fmicb.2022.962619Search in Google Scholar
[137] Munir S, Azeem A, Sikandar Zaman M, Zia Ul Haq M. From field to table: Ensuring food safety by reducing pesticide residues in food. Sci Total Environ. 2024;922:171382. 10.1016/j.scitotenv.2024.171382.Search in Google Scholar PubMed
[138] Leskovac A, Petrović S. Pesticide use and degradation strategies: food safety, challenges and perspectives. Foods. 2023;12(14):2709.10.3390/foods12142709Search in Google Scholar
[139] Pecenka JR, Ingwell LL, Foster RE, Krupke CH, Kaplan I. IPM reduces insecticide applications by 95% while maintaining or enhancing crop yields through wild pollinator conservation. Proc Natl Acad Sci. 2021;118(44):e2108429118.10.1073/pnas.2108429118Search in Google Scholar
[140] Wahab A, Batool F, Muhammad M, Zaman W, Mikhlef RM, Qaddoori SM, et al. Unveiling the complex molecular dynamics of arbuscular mycorrhizae: A comprehensive exploration and future perspectives in harnessing phosphate-solubilizing microorganisms for sustainable progress. Environ Exp Bot. 2024;219:105633. 10.1016/j.envexpbot.2023.105633.Search in Google Scholar
[141] Ullah F, Saqib S, Khan W, Zhao L, Khan W, Li MY, et al. Sodium nitroprusside and melatonin improve physiological vitality and drought acclimation via synergistically enhancing antioxidant response in dryland maize. J Plant Growth Regul. 2025;44(2):891–908. 10.1007/s00344-024-11498-2.Search in Google Scholar
[142] Ullah F, Saqib S, Zaman W, Khan W, Zhao L, Khan A, et al. Mitigating drought and heavy metal stress in maize using melatonin and sodium nitroprusside. Plant Soil. 2024;44:891–908. 10.1007/s11104-024-07077-9.Search in Google Scholar
[143] Pop C, Suharoschi R, Pop OL. Dietary fiber and prebiotic compounds in fruits and vegetables food waste. Sustainability. 2021;13:7219.10.3390/su13137219Search in Google Scholar
[144] Najar IN, Sharma P, Das R, Tamang S, Mondal K, Thakur N, et al. From waste management to circular economy: Leveraging thermophiles for sustainable growth and global resource optimization. J Environ Manag. 2024;360:121136. 10.1016/j.jenvman.2024.121136.Search in Google Scholar PubMed
[145] Antranikian G, Streit WR. Microorganisms harbor keys to a circular bioeconomy making them useful tools in fighting plastic pollution and rising CO2 levels. Extremophiles. 2022;26(1):10. 10.1007/s00792-022-01261-4.Search in Google Scholar PubMed PubMed Central
[146] Najar IN, Sherpa MT, Das S, Das S, Thakur N. Microbial ecology of two hot springs of Sikkim: Predominate population and geochemistry. Sci Total Environ. 2018;637–638:730–45. 10.1016/j.scitotenv.2018.05.037.Search in Google Scholar PubMed
[147] Najar IN, Sherpa MT, Das S, Thakur N. Bacterial diversity and functional metagenomics expounding the diversity of xenobiotics, stress, defense and CRISPR gene ontology providing eco-efficiency to Himalayan Hot Springs. Funct Integr Genomics. 2020;20(4):479–96. 10.1007/s10142-019-00723-x.Search in Google Scholar PubMed
[148] Banerjee A, Sarkar S, Govil T, González-Faune P, Cabrera-Barjas G, Bandopadhyay R, et al. Extremophilic exopolysaccharides: biotechnologies and wastewater remediation [Mini Review]. Front Microbiol. 2021;12:721365, https://www.frontiersin.org/journals/microbiology/articles/10.3389/fmicb.2021.721365.10.3389/fmicb.2021.721365Search in Google Scholar
[149] Mehta R, Singhal P, Singh H, Damle D, Sharma AK. Insight into thermophiles and their wide-spectrum applications. 3 Biotech. 2016;6(1):81. 10.1007/s13205-016-0368-z.Search in Google Scholar PubMed PubMed Central
[150] Navina BK, Velmurugan NK, Senthil Kumar P, Rangasamy G, Palanivelu J, Thamarai P, et al. Fungal bioremediation approaches for the removal of toxic pollutants: Mechanistic understanding for biorefinery applications. Chemosphere. 2024;350:141123. 10.1016/j.chemosphere.2024.141123.Search in Google Scholar PubMed
[151] Bhattacharya A, Gola D, Dey P, Malik A. Synergistic and antagonistic effects on metal bioremediation with increasing metal complexity in a hexa-metal environment by Aspergillus fumigatus. Int J Environ Res. 2020;14(6):761–70. 10.1007/s41742-020-00295-w.Search in Google Scholar
[152] Kalia S, Bhattacharya A, Prajapati SK, Malik A. Utilization of starch effluent from a textile industry as a fungal growth supplement for enhanced α-amylase production for industrial application. Chemosphere. 2021;279:130554. 10.1016/j.chemosphere.2021.130554.Search in Google Scholar PubMed
[153] Khatua S, Simal-Gandara J, Acharya K. Myco-remediation of plastic pollution: current knowledge and future prospects. Biodegradation. 2024;35(3):249–79. 10.1007/s10532-023-10053-2.Search in Google Scholar PubMed PubMed Central
[154] Kanlaya R, Khamchun S, Kapincharanon C, Thongboonkerd V. Protective effect of epigallocatechin-3-gallate (EGCG) via Nrf2 pathway against oxalate-induced epithelial mesenchymal transition (EMT) of renal tubular cells. Sci Rep. 2016;6(1):30233. 10.1038/srep30233.Search in Google Scholar PubMed PubMed Central
[155] Newsome BJ, Petriello MC, Han SG, Murphy MO, Eske KE, Sunkara M, et al. Green tea diet decreases PCB 126-induced oxidative stress in mice by up-regulating antioxidant enzymes. J Nutr Biochem. 2014;25(2):126–35. 10.1016/j.jnutbio.2013.10.003.Search in Google Scholar PubMed PubMed Central
[156] Ravichandran R, Rajendran M, Devapiriam D. Antioxidant study of quercetin and their metal complex and determination of stability constant by spectrophotometry method. Food Chem. 2014;146:472–8. 10.1016/j.foodchem.2013.09.080.Search in Google Scholar PubMed
[157] Chung RTM. Detoxification effects of phytonutrients against environmental toxicants and sharing of clinical experience on practical applications. Environ Sci Pollut Res. 2017;24(10):8946–56. 10.1007/s11356-015-5263-3.Search in Google Scholar PubMed
[158] García-Niño WR, Pedraza-Chaverrí J. Protective effect of curcumin against heavy metals-induced liver damage. Food Chem Toxicol. 2014;69:182–201. 10.1016/j.fct.2014.04.016.Search in Google Scholar PubMed
[159] Flora G, Gupta D, Tiwari A. Preventive efficacy of bulk and nanocurcumin against lead-induced oxidative stress in mice. Biol Trace Elem Res. 2013;152(1):31–40. 10.1007/s12011-012-9586-3.Search in Google Scholar PubMed
[160] Cengiz MF, Basançelebi O, Başlar M, Certel M. A novel technique for the reduction of pesticide residues by a combination of low-intensity electrical current and ultrasound applications: A study on lettuce samples. Food Chem. 2021;354:129360. 10.1016/j.foodchem.2021.129360.Search in Google Scholar PubMed
[161] Jatoi AS, Hashmi Z, Adriyani R, Yuniarto A, Mazari SA, Akhter F, et al. Recent trends and future challenges of pesticide removal techniques – A comprehensive review. J Environ Chem Eng. 2021;9(4):105571. 10.1016/j.jece.2021.105571.Search in Google Scholar
[162] Lozowicka B, Jankowska M, Hrynko I, Kaczynski P. Removal of 16 pesticide residues from strawberries by washing with tap and ozone water, ultrasonic cleaning and boiling. Environ Monit Assess. 2015;188(1):51. 10.1007/s10661-015-4850-6.Search in Google Scholar PubMed PubMed Central
[163] Liu L, Xie M, Wei D. Biological detoxification of mycotoxins: current status and future advances. Int J Mol Sci. 2022;23(3):1064.10.3390/ijms23031064Search in Google Scholar
[164] Shekhar R, Raghvendra VB, Rachitha P. A comprehensive review of mycotoxins, their toxicity, and innovative detoxification methods. Toxicol Rep. 2025;14:101952. 10.1016/j.toxrep.2025.101952.Search in Google Scholar PubMed PubMed Central
[165] Karlovsky P, Suman M, Berthiller F, De Meester J, Eisenbrand G, Perrin I, et al. Impact of food processing and detoxification treatments on mycotoxin contamination. Mycotoxin Res. 2016;32(4):179–205. 10.1007/s12550-016-0257-7.Search in Google Scholar PubMed PubMed Central
[166] Pedras MSC, Khallaf I. Molecular interactions of the phytotoxins destruxin B and sirodesmin PL with crucifers and cereals: Metabolism and elicitation of plant defenses. Phytochemistry. 2012;77:129–39. 10.1016/j.phytochem.2012.02.010.Search in Google Scholar PubMed
[167] Makhuvele R, Naidu K, Gbashi S, Thipe VC, Adebo OA, Njobeh PB. The use of plant extracts and their phytochemicals for control of toxigenic fungi and mycotoxins. Heliyon. 2020;6(10):e05291. 10.1016/j.heliyon.2020.e05291.Search in Google Scholar PubMed PubMed Central
[168] Wetterhorn KM, Newmister SA, Caniza RK, Busman M, McCormick SP, Berthiller F, et al. Crystal Structure of Os79 (Os04g0206600) from Oryza sativa: A UDP-glucosyltransferase Involved in the Detoxification of Deoxynivalenol. Biochemistry. 2016;55(44):6175–86. 10.1021/acs.biochem.6b00709.Search in Google Scholar PubMed
[169] Li P, Su R, Yin R, Lai D, Wang M, Liu Y, et al. Detoxification of mycotoxins through biotransformation. Toxins. 2020;12(2):121.10.3390/toxins12020121Search in Google Scholar
[170] Zhang AA, Sutar PP, Bian Q, Fang XM, Ni JB, Xiao HW. Pesticide residue elimination for fruits and vegetables: the mechanisms, applications, and future trends of thermal and non-thermal technologies. J Future Foods. 2022;2(3):223–40. 10.1016/j.jfutfo.2022.06.004.Search in Google Scholar
[171] Yang L, Zhou J, Feng Y. Removal of pesticide residues from fresh vegetables by the coupled free chlorine/ultrasound process. Ultrason Sonochem. 2022;82:105891. 10.1016/j.ultsonch.2021.105891.Search in Google Scholar PubMed PubMed Central
[172] Rong Tan L, Chen Lu Y, Jing Zhang J, Luo F, Yang H. A collection of cytochrome P450 monooxygenase genes involved in modification and detoxification of herbicide atrazine in rice (Oryza sativa) plants. Ecotoxicol Environ Saf. 2015;119:25–34. 10.1016/j.ecoenv.2015.04.035.Search in Google Scholar PubMed
[173] Pandian BA, Sathishraj R, Djanaguiraman M, Prasad PVV, Jugulam M. Role of cytochrome P450 enzymes in plant stress response. Antioxidants. 2020;9(5):454.10.3390/antiox9050454Search in Google Scholar
[174] Ohkawa H, Inui H. Metabolism of agrochemicals and related environmental chemicals based on cytochrome P450s in mammals and plants. Pest Manag Sci. 2015;71(6):824–8. 10.1002/ps.3871.Search in Google Scholar PubMed
[175] Saika H, Horita J, Taguchi-Shiobara F, Nonaka S, Nishizawa-Yokoi A, Iwakami S, et al. A novel rice cytochrome P450 gene, CYP72A31, confers tolerance to acetolactate synthase-inhibiting herbicides in rice and Arabidopsis. Plant Physiol. 2014;166(3):1232–40. 10.1104/pp.113.231266.Search in Google Scholar PubMed PubMed Central
[176] Sylvestre-Gonon E, Law SR, Schwartz M, Robe K, Keech O, Didierjean C, et al. Functional, structural and biochemical features of plant serinyl-glutathione transferases [Review]. Front Plant Sci. 2019;10:608, https://www.frontiersin.org/journals/plant-science/articles/10.3389/fpls.2019.00608.10.3389/fpls.2019.00608Search in Google Scholar
[177] Perperopoulou F, Pouliou F, Labrou NE. Recent advances in protein engineering and biotechnological applications of glutathione transferases. Crit Rev Biotechnol. 2018;38(4):511–28. 10.1080/07388551.2017.1375890.Search in Google Scholar PubMed
[178] Zhang JJ, Yang H. Metabolism and detoxification of pesticides in plants. Sci Total Environ. 2021;790:148034. 10.1016/j.scitotenv.2021.148034.Search in Google Scholar PubMed
[179] Liscombe DK, Louie GV, Noel JP. Architectures, mechanisms and molecular evolution of natural product methyltransferases. Nat Prod Rep. 2012;29(10):1238–50.10.1039/c2np20029eSearch in Google Scholar
[180] Samtiya M, Aluko RE, Dhewa T. Plant food anti-nutritional factors and their reduction strategies: an overview. Food Prod Process Nutr. 2020;2(1):6. 10.1186/s43014-020-0020-5.Search in Google Scholar
[181] Banerjee S, Ranganathan V, Patti A, Arora A. Valorisation of pineapple wastes for food and therapeutic applications. Trends Food Sci Technol. 2018;82:60–70. 10.1016/j.tifs.2018.09.024.Search in Google Scholar
[182] de Toledo NMV, Nunes LP, da Silva PPM, Spoto MHF, Canniatti-Brazaca SG. Influence of pineapple, apple and melon by-products on cookies: physicochemical and sensory aspects. Int J Food Sci Technol. 2017;52(5):1185–92. 10.1111/ijfs.13383.Search in Google Scholar
[183] Debnath P, Dey P, Chanda A, Bhakta T. A survey on pineapple and its medicinal value. Scholars Acad J Pharm. 2012;1(1):24–9.Search in Google Scholar
[184] Ketnawa S, Chaiwut P, Rawdkuen S. Pineapple wastes: A potential source for bromelain extraction. Food Bioprod Process. 2012;90(3):385–91. 10.1016/j.fbp.2011.12.006.Search in Google Scholar
[185] Roda A, Lambri M. Food uses of pineapple waste and by-products: a review. Int J Food Sci Technol. 2019;54(4):1009–17. 10.1111/ijfs.14128.Search in Google Scholar
[186] Amariz A, Lima MA, Alves RE. Quality and antioxidant potential of byproducts from refining of fruit pulp. Food Sci Technol. 2018;38:203–9.10.1590/fst.25816Search in Google Scholar
[187] Plazzotta S, Ibarz R, Manzocco L, Martín-Belloso O. Optimizing the antioxidant biocompound recovery from peach waste extraction assisted by ultrasounds or microwaves. Ultrason Sonochem. 2020;63:104954. 10.1016/j.ultsonch.2019.104954.Search in Google Scholar PubMed
[188] Esparza I, Jiménez-Moreno N, Bimbela F, Ancín-Azpilicueta C, Gandía LM. Fruit and vegetable waste management: Conventional and emerging approaches. J Environ Manag. 2020;265:110510. 10.1016/j.jenvman.2020.110510.Search in Google Scholar PubMed
[189] Rudke CRM, Zielinski AAF, Ferreira SRS. From biorefinery to food product design: Peach (Prunus persica) by-products deserve attention. Food Bioprocess Technol. 2023;16(6):1197–215. 10.1007/s11947-022-02951-9.Search in Google Scholar PubMed PubMed Central
[190] Choi Y-S, Kim YB, Hwang KE, Song DH, Ham YK, Kim HW, et al. Effect of apple pomace fiber and pork fat levels on quality characteristics of uncured, reduced-fat chicken sausages. Poult Sci. 2016;95(6):1465–71. 10.3382/ps/pew096.Search in Google Scholar PubMed
[191] Huc-Mathis D, Journet C, Fayolle N, Bosc V. Emulsifying properties of food by-products: Valorizing apple pomace and oat bran. Colloids Surf A: Physicochem Eng Asp. 2019;568:84–91. 10.1016/j.colsurfa.2019.02.001.Search in Google Scholar
[192] Perussello CA, Zhang Z, Marzocchella A, Tiwari BK. Valorization of apple pomace by extraction of valuable compounds. Compr Rev Food Sci Food Saf. 2017;16(5):776–96. 10.1111/1541-4337.12290.Search in Google Scholar PubMed
[193] Ayala-Zavala JF, Vega-Vega V, Rosas-Domínguez C, Palafox-Carlos H, Villa-Rodriguez JA, Siddiqui MW, et al. Agro-industrial potential of exotic fruit byproducts as a source of food additives. Food Res Int. 2011;44(7):1866–74. 10.1016/j.foodres.2011.02.021.Search in Google Scholar
[194] Rotta EM, Morais DR, Biondo PBF, Santos VJ, Matsushita M, Visentainer JV. Use of avocado peel (Persea americana) in tea formulation: a functional product containing phenolic compounds with antioxidant activity. Acta Sci Technol. 2016;38(1):23–9.10.4025/actascitechnol.v38i1.27397Search in Google Scholar
[195] Bankar A, Joshi B, Kumar AR, Zinjarde S. Banana peel extract mediated novel route for the synthesis of silver nanoparticles. Colloids Surf A: Physicochem Eng Asp. 2010;368(1):58–63. 10.1016/j.colsurfa.2010.07.024.Search in Google Scholar
[196] Vu HT, Scarlett CJ, Vuong QV. Phenolic compounds within banana peel and their potential uses: A review. J Funct Foods. 2018;40:238–48. 10.1016/j.jff.2017.11.006.Search in Google Scholar
[197] Espitia PJ, Du WX, de Jesús Avena-Bustillos R, Soares ND, McHugh TH. Edible films from pectin: Physical-mechanical and antimicrobial properties – A review. Food Hydrocoll. 2014;35:287–96. 10.1016/j.foodhyd.2013.06.005.Search in Google Scholar
[198] Satari B, Karimi K. Citrus processing wastes: Environmental impacts, recent advances, and future perspectives in total valorization. Resour Conserv Recycl. 2018;129:153–67. 10.1016/j.resconrec.2017.10.032.Search in Google Scholar
[199] Acun S, Gül H. Effects of grape pomace and grape seed flours on cookie quality. Qual Assur Saf Crop Foods. 2014;6(1):81–8.10.3920/QAS2013.0264Search in Google Scholar
[200] Mainente F, Menin A, Alberton A, Zoccatelli G, Rizzi C. Evaluation of the sensory and physical properties of meat and fish derivatives containing grape pomace powders. Int J Food Sci Technol. 2019;54(4):952–8. 10.1111/ijfs.13850.Search in Google Scholar
[201] Mattos GN, Tonon RV, Furtado AAL, Cabral LMC. Grape by-product extracts against microbial proliferation and lipid oxidation: a review. J Sci Food Agric. 2017;97(4):1055–64. 10.1002/jsfa.8062.Search in Google Scholar PubMed
[202] Adilah AN, Jamilah B, Noranizan MA, Hanani ZAN. Utilization of mango peel extracts on the biodegradable films for active packaging. Food Packag Shelf Life. 2018;16:1–7. 10.1016/j.fpsl.2018.01.006.Search in Google Scholar
[203] Ajila CM, Aalami M, Leelavathi K, Rao UJSP. Mango peel powder: A potential source of antioxidant and dietary fiber in macaroni preparations. Innov Food Sci Emerg Technol. 2010;11(1):219–24. 10.1016/j.ifset.2009.10.004.Search in Google Scholar
[204] Ashoush IS, Gadallah MGE. Utilization of mango peels and seed kernels powders as sources of phytochemicals in biscuit. World J Dairy Food Sci. 2011;6(1):35–42.Search in Google Scholar
[205] Torres-León C, Vicente AA, Flores-López ML, Rojas R, Serna-Cock L, Alvarez-Pérez OB, et al. Edible films and coatings based on mango (var. Ataulfo) by-products to improve gas transfer rate of peach. LWT. 2018;97:624–31. 10.1016/j.lwt.2018.07.057.Search in Google Scholar
[206] Arshad ZI, Amid A, Yusof F, Jaswir I, Ahmad K, Loke SP. Bromelain: an overview of industrial application and purification strategies. Appl Microbiol Biotechnol. 2014;98(17):7283–97. 10.1007/s00253-014-5889-y.Search in Google Scholar PubMed
[207] Kodagoda K, Marapana R. Development of non-alcoholic wines from the wastes of Mauritius pineapple variety and its physicochemical properties. J Pharmacogn Phytochem. 2017;6(3):492–7.Search in Google Scholar
[208] Gonzalez J, Donoso W, Sandoval N, Reyes M, Gonzalez P, Gajardo M, et al. Apple peel supplemented diet reduces parameters of metabolic syndrome and atherogenic progression in ApoE−/− mice. Evid-Based Complement Altern Med. 2015;2015(1):918384. 10.1155/2015/918384.Search in Google Scholar PubMed PubMed Central
[209] Araújo RG, Rodriguez-Jasso RM, Ruiz HA, Pintado MME, Aguilar CN. Avocado by-products: Nutritional and functional properties. Trends Food Sci Technol. 2018;80:51–60. 10.1016/j.tifs.2018.07.027.Search in Google Scholar
[210] Yu X, Lin H, Wang Y, Lv W, Zhang S, Qian Y, et al. D-limonene exhibits antitumor activity by inducing autophagy and apoptosis in lung cancer. OncoTargets Ther. 2018;11:1833–47. 10.2147/OTT.S155716.Search in Google Scholar PubMed PubMed Central
[211] Miller JA, Thompson PA, Hakim IA, Chow HHS, Thomson CA. d-Limonene: a bioactive food component from citrus and evidence for a potential role in breast cancer prevention and treatment. Oncol Rev. 2011;5(1):31–42. 10.1007/s12156-010-0066-8.Search in Google Scholar
[212] Burton-Freeman BM, Sandhu AK, Edirisinghe I. Mangos and their bioactive components: adding variety to the fruit plate for health [10.1039/C7FO00190H]. Food Funct. 2017;8(9):3010–32. 10.1039/C7FO00190H.Search in Google Scholar
[213] Maxwell EG, Belshaw NJ, Waldron KW, Morris VJ. Pectin – An emerging new bioactive food polysaccharide. Trends Food Sci Technol. 2012;24(2):64–73. 10.1016/j.tifs.2011.11.002.Search in Google Scholar
[214] Wang X, Gao L, Lin H, Song J, Wang J, Yin Y, et al. Mangiferin prevents diabetic nephropathy progression and protects podocyte function via autophagy in diabetic rat glomeruli. Eur J Pharmacol. 2018;824:170–8. 10.1016/j.ejphar.2018.02.009.Search in Google Scholar PubMed
[215] Nowicka P, Wojdyło A. Content of bioactive compounds in the peach kernels and their antioxidant, anti-hyperglycemic, anti-aging properties. Eur Food Res Technol. 2019;245(5):1123–36. 10.1007/s00217-018-3214-1.Search in Google Scholar
[216] Pérez-Jiménez J, Saura-Calixto F. Fruit peels as sources of non-extractable polyphenols or macromolecular antioxidants: Analysis and nutritional implications. Food Res Int. 2018;111:148–52. 10.1016/j.foodres.2018.05.023.Search in Google Scholar PubMed
[217] Mihaylova D, Popova A, Desseva I, Dincheva I, Tumbarski Y. Valorization of peels of eight peach varieties: GC–MS profile, free and bound phenolics and corresponding biological activities. Antioxidants. 2023;12(1):205.10.3390/antiox12010205Search in Google Scholar
[218] Rodríguez-González S, Pérez-Ramírez IF, Amaya-Cruz DM, Gallegos-Corona MA, Ramos-Gomez M, Mora O, et al. Polyphenol-rich peach (Prunus persica L.) by-product exerts a greater beneficial effect than dietary fiber-rich by-product on insulin resistance and hepatic steatosis in obese rats. J Funct Foods. 2018;45:58–66. 10.1016/j.jff.2018.03.010.Search in Google Scholar
[219] Gamboa-Santos J, Soria AC, Fornari T, Villamiel M, Montilla A. Optimisation of convective drying of carrots using selected processing and quality indicators. Int J Food Sci Technol. 2013;48(10):1998–2006. 10.1111/ijfs.12076.Search in Google Scholar
[220] García-Aparicio MdP, Castro-Rubio F, Marina ML. Unlocking peach juice byproduct potential in food waste biorefineries: Phenolic compounds profile, antioxidant capacity and fermentable sugars. Bioresour Technol. 2024;396:130441. 10.1016/j.biortech.2024.130441.Search in Google Scholar PubMed
© 2025 the author(s), published by De Gruyter
This work is licensed under the Creative Commons Attribution 4.0 International License.
Articles in the same Issue
- Safety assessment and modulation of hepatic CYP3A4 and UGT enzymes by Glycyrrhiza glabra aqueous extract in female Sprague–Dawley rats
- Adult-onset Still’s disease with hemophagocytic lymphohistiocytosis and minimal change disease
- Role of DZ2002 in reducing corneal graft rejection in rats by influencing Th17 activation via inhibition of the PI3K/AKT pathway and downregulation of TRAF1
- Biomedical Sciences
- Mechanism of triptolide regulating proliferation and apoptosis of hepatoma cells by inhibiting JAK/STAT pathway
- Maslinic acid improves mitochondrial function and inhibits oxidative stress and autophagy in human gastric smooth muscle cells
- Comparative analysis of inflammatory biomarkers for the diagnosis of neonatal sepsis: IL-6, IL-8, SAA, CRP, and PCT
- Post-pandemic insights on COVID-19 and premature ovarian insufficiency
- Proteome differences of dental stem cells between permanent and deciduous teeth by data-independent acquisition proteomics
- Optimizing a modified cetyltrimethylammonium bromide protocol for fungal DNA extraction: Insights from multilocus gene amplification
- Preliminary analysis of the role of small hepatitis B surface proteins mutations in the pathogenesis of occult hepatitis B infection via the endoplasmic reticulum stress-induced UPR-ERAD pathway
- Efficacy of alginate-coated gold nanoparticles against antibiotics-resistant Staphylococcus and Streptococcus pathogens of acne origins
- Battling COVID-19 leveraging nanobiotechnology: Gold and silver nanoparticle–B-escin conjugates as SARS-CoV-2 inhibitors
- Neurodegenerative diseases and neuroinflammation-induced apoptosis
- Impact of fracture fixation surgery on cognitive function and the gut microbiota in mice with a history of stroke
- COLEC10: A potential tumor suppressor and prognostic biomarker in hepatocellular carcinoma through modulation of EMT and PI3K-AKT pathways
- High-temperature requirement serine protease A2 inhibitor UCF-101 ameliorates damaged neurons in traumatic brain-injured rats by the AMPK/NF-κB pathway
- SIK1 inhibits IL-1β-stimulated cartilage apoptosis and inflammation in vitro through the CRTC2/CREB1 signaling
- Rutin–chitooligosaccharide complex: Comprehensive evaluation of its anti-inflammatory and analgesic properties in vitro and in vivo
- Knockdown of Aurora kinase B alleviates high glucose-triggered trophoblast cells damage and inflammation during gestational diabetes
- Calcium-sensing receptors promoted Homer1 expression and osteogenic differentiation in bone marrow mesenchymal stem cells
- ABI3BP can inhibit the proliferation, invasion, and epithelial–mesenchymal transition of non-small-cell lung cancer cells
- Changes in blood glucose and metabolism in hyperuricemia mice
- Rapid detection of the GJB2 c.235delC mutation based on CRISPR-Cas13a combined with lateral flow dipstick
- IL-11 promotes Ang II-induced autophagy inhibition and mitochondrial dysfunction in atrial fibroblasts
- Short-chain fatty acid attenuates intestinal inflammation by regulation of gut microbial composition in antibiotic-associated diarrhea
- Application of metagenomic next-generation sequencing in the diagnosis of pathogens in patients with diabetes complicated by community-acquired pneumonia
- NAT10 promotes radiotherapy resistance in non-small cell lung cancer by regulating KPNB1-mediated PD-L1 nuclear translocation
- Phytol-mixed micelles alleviate dexamethasone-induced osteoporosis in zebrafish: Activation of the MMP3–OPN–MAPK pathway-mediating bone remodeling
- Association between TGF-β1 and β-catenin expression in the vaginal wall of patients with pelvic organ prolapse
- Primary pleomorphic liposarcoma involving bilateral ovaries: Case report and literature review
- Effects of de novo donor-specific Class I and II antibodies on graft outcomes after liver transplantation: A pilot cohort study
- Sleep architecture in Alzheimer’s disease continuum: The deep sleep question
- Ephedra fragilis plant extract: A groundbreaking corrosion inhibitor for mild steel in acidic environments – electrochemical, EDX, DFT, and Monte Carlo studies
- Langerhans cell histiocytosis in an adult patient with upper jaw and pulmonary involvement: A case report
- Inhibition of mast cell activation by Jaranol-targeted Pirin ameliorates allergic responses in mouse allergic rhinitis
- Aeromonas veronii-induced septic arthritis of the hip in a child with acute lymphoblastic leukemia
- Clusterin activates the heat shock response via the PI3K/Akt pathway to protect cardiomyocytes from high-temperature-induced apoptosis
- Research progress on fecal microbiota transplantation in tumor prevention and treatment
- Low-pressure exposure influences the development of HAPE
- Stigmasterol alleviates endplate chondrocyte degeneration through inducing mitophagy by enhancing PINK1 mRNA acetylation via the ESR1/NAT10 axis
- AKAP12, mediated by transcription factor 21, inhibits cell proliferation, metastasis, and glycolysis in lung squamous cell carcinoma
- Association between PAX9 or MSX1 gene polymorphism and tooth agenesis risk: A meta-analysis
- A case of bloodstream infection caused by Neisseria gonorrhoeae
- Case of nasopharyngeal tuberculosis complicated with cervical lymph node and pulmonary tuberculosis
- p-Cymene inhibits pro-fibrotic and inflammatory mediators to prevent hepatic dysfunction
- GFPT2 promotes paclitaxel resistance in epithelial ovarian cancer cells via activating NF-κB signaling pathway
- Transfer RNA-derived fragment tRF-36 modulates varicose vein progression via human vascular smooth muscle cell Notch signaling
- RTA-408 attenuates the hepatic ischemia reperfusion injury in mice possibly by activating the Nrf2/HO-1 signaling pathway
- Decreased serum TIMP4 levels in patients with rheumatoid arthritis
- Sirt1 protects lupus nephritis by inhibiting the NLRP3 signaling pathway in human glomerular mesangial cells
- Sodium butyrate aids brain injury repair in neonatal rats
- Interaction of MTHFR polymorphism with PAX1 methylation in cervical cancer
- Convallatoxin inhibits proliferation and angiogenesis of glioma cells via regulating JAK/STAT3 pathway
- The effect of the PKR inhibitor, 2-aminopurine, on the replication of influenza A virus, and segment 8 mRNA splicing
- Effects of Ire1 gene on virulence and pathogenicity of Candida albicans
- Small cell lung cancer with small intestinal metastasis: Case report and literature review
- GRB14: A prognostic biomarker driving tumor progression in gastric cancer through the PI3K/AKT signaling pathway by interacting with COBLL1
- 15-Lipoxygenase-2 deficiency induces foam cell formation that can be restored by salidroside through the inhibition of arachidonic acid effects
- FTO alleviated the diabetic nephropathy progression by regulating the N6-methyladenosine levels of DACT1
- Clinical relevance of inflammatory markers in the evaluation of severity of ulcerative colitis: A retrospective study
- Zinc valproic acid complex promotes osteoblast differentiation and exhibits anti-osteoporotic potential
- Primary pulmonary synovial sarcoma in the bronchial cavity: A case report
- Metagenomic next-generation sequencing of alveolar lavage fluid improves the detection of pulmonary infection
- Uterine tumor resembling ovarian sex cord tumor with extensive rhabdoid differentiation: A case report
- Genomic analysis of a novel ST11(PR34365) Clostridioides difficile strain isolated from the human fecal of a CDI patient in Guizhou, China
- Effects of tiered cardiac rehabilitation on CRP, TNF-α, and physical endurance in older adults with coronary heart disease
- Changes in T-lymphocyte subpopulations in patients with colorectal cancer before and after acupoint catgut embedding acupuncture observation
- Modulating the tumor microenvironment: The role of traditional Chinese medicine in improving lung cancer treatment
- Alterations of metabolites related to microbiota–gut–brain axis in plasma of colon cancer, esophageal cancer, stomach cancer, and lung cancer patients
- Research on individualized drug sensitivity detection technology based on bio-3D printing technology for precision treatment of gastrointestinal stromal tumors
- CEBPB promotes ulcerative colitis-associated colorectal cancer by stimulating tumor growth and activating the NF-κB/STAT3 signaling pathway
- Oncolytic bacteria: A revolutionary approach to cancer therapy
- A de novo meningioma with rapid growth: A possible malignancy imposter?
- Diagnosis of secondary tuberculosis infection in an asymptomatic elderly with cancer using next-generation sequencing: Case report
- Hesperidin and its zinc(ii) complex enhance osteoblast differentiation and bone formation: In vitro and in vivo evaluations
- Research progress on the regulation of autophagy in cardiovascular diseases by chemokines
- Anti-arthritic, immunomodulatory, and inflammatory regulation by the benzimidazole derivative BMZ-AD: Insights from an FCA-induced rat model
- Immunoassay for pyruvate kinase M1/2 as an Alzheimer’s biomarker in CSF
- The role of HDAC11 in age-related hearing loss: Mechanisms and therapeutic implications
- Evaluation and application analysis of animal models of PIPNP based on data mining
- Therapeutic approaches for liver fibrosis/cirrhosis by targeting pyroptosis
- Fabrication of zinc oxide nanoparticles using Ruellia tuberosa leaf extract induces apoptosis through P53 and STAT3 signalling pathways in prostate cancer cells
- Haplo-hematopoietic stem cell transplantation and immunoradiotherapy for severe aplastic anemia complicated with nasopharyngeal carcinoma: A case report
- Modulation of the KEAP1-NRF2 pathway by Erianin: A novel approach to reduce psoriasiform inflammation and inflammatory signaling
- The expression of epidermal growth factor receptor 2 and its relationship with tumor-infiltrating lymphocytes and clinical pathological features in breast cancer patients
- Innovations in MALDI-TOF Mass Spectrometry: Bridging modern diagnostics and historical insights
- BAP1 complexes with YY1 and RBBP7 and its downstream targets in ccRCC cells
- Hypereosinophilic syndrome with elevated IgG4 and T-cell clonality: A report of two cases
- Electroacupuncture alleviates sciatic nerve injury in sciatica rats by regulating BDNF and NGF levels, myelin sheath degradation, and autophagy
- Polydatin prevents cholesterol gallstone formation by regulating cholesterol metabolism via PPAR-γ signaling
- RNF144A and RNF144B: Important molecules for health
- Analysis of the detection rate and related factors of thyroid nodules in the healthy population
- Artesunate inhibits hepatocellular carcinoma cell migration and invasion through OGA-mediated O-GlcNAcylation of ZEB1
- Endovascular management of post-pancreatectomy hemorrhage caused by a hepatic artery pseudoaneurysm: Case report and review of the literature
- Efficacy and safety of anti-PD-1/PD-L1 antibodies in patients with relapsed refractory diffuse large B-cell lymphoma: A meta-analysis
- SATB2 promotes humeral fracture healing in rats by activating the PI3K/AKT pathway
- Overexpression of the ferroptosis-related gene, NFS1, corresponds to gastric cancer growth and tumor immune infiltration
- Understanding risk factors and prognosis in diabetic foot ulcers
- Atractylenolide I alleviates the experimental allergic response in mice by suppressing TLR4/NF-kB/NLRP3 signalling
- FBXO31 inhibits the stemness characteristics of CD147 (+) melanoma stem cells
- Immune molecule diagnostics in colorectal cancer: CCL2 and CXCL11
- Inhibiting CXCR6 promotes senescence of activated hepatic stellate cells with limited proinflammatory SASP to attenuate hepatic fibrosis
- Cadmium toxicity, health risk and its remediation using low-cost biochar adsorbents
- Pulmonary cryptococcosis with headache as the first presentation: A case report
- Solitary pulmonary metastasis with cystic airspaces in colon cancer: A rare case report
- RUNX1 promotes denervation-induced muscle atrophy by activating the JUNB/NF-κB pathway and driving M1 macrophage polarization
- Morphometric analysis and immunobiological investigation of Indigofera oblongifolia on the infected lung with Plasmodium chabaudi
- The NuA4/TIP60 histone-modifying complex and Hr78 modulate the Lobe2 mutant eye phenotype
- Experimental study on salmon demineralized bone matrix loaded with recombinant human bone morphogenetic protein-2: In vitro and in vivo study
- A case of IgA nephropathy treated with a combination of telitacicept and half-dose glucocorticoids
- Analgesic and toxicological evaluation of cannabidiol-rich Moroccan Cannabis sativa L. (Khardala variety) extract: Evidence from an in vivo and in silico study
- Wound healing and signaling pathways
- Combination of immunotherapy and whole-brain radiotherapy on prognosis of patients with multiple brain metastases: A retrospective cohort study
- To explore the relationship between endometrial hyperemia and polycystic ovary syndrome
- Research progress on the impact of curcumin on immune responses in breast cancer
- Biogenic Cu/Ni nanotherapeutics from Descurainia sophia (L.) Webb ex Prantl seeds for the treatment of lung cancer
- Dapagliflozin attenuates atrial fibrosis via the HMGB1/RAGE pathway in atrial fibrillation rats
- Glycitein alleviates inflammation and apoptosis in keratinocytes via ROS-associated PI3K–Akt signalling pathway
- ADH5 inhibits proliferation but promotes EMT in non-small cell lung cancer cell through activating Smad2/Smad3
- Apoptotic efficacies of AgNPs formulated by Syzygium aromaticum leaf extract on 32D-FLT3-ITD human leukemia cell line with PI3K/AKT/mTOR signaling pathway
- Novel cuproptosis-related genes C1QBP and PFKP identified as prognostic and therapeutic targets in lung adenocarcinoma
- Bee venom promotes exosome secretion and alters miRNA cargo in T cells
- Treatment of pure red cell aplasia in a chronic kidney disease patient with roxadustat: A case report
- Comparative bioinformatics analysis of the Wnt pathway in breast cancer: Selection of novel biomarker panels associated with ER status
- Kynurenine facilitates renal cell carcinoma progression by suppressing M2 macrophage pyroptosis through inhibition of CASP1 cleavage
- RFX5 promotes the growth, motility, and inhibits apoptosis of gastric adenocarcinoma cells through the SIRT1/AMPK axis
- ALKBH5 exacerbates early cardiac damage after radiotherapy for breast cancer via m6A demethylation of TLR4
- Phytochemicals of Roman chamomile: Antioxidant, anti-aging, and whitening activities of distillation residues
- Circadian gene Cry1 inhibits the tumorigenicity of hepatocellular carcinoma by the BAX/BCL2-mediated apoptosis pathway
- The TNFR-RIPK1/RIPK3 signalling pathway mediates the effect of lanthanum on necroptosis of nerve cells
- Longitudinal monitoring of autoantibody dynamics in patients with early-stage non-small-cell lung cancer undergoing surgery
- The potential role of rutin, a flavonoid, in the management of cancer through modulation of cell signaling pathways
- Construction of pectinase gene engineering microbe and its application in tobacco sheets
- Construction of a microbial abundance prognostic scoring model based on intratumoral microbial data for predicting the prognosis of lung squamous cell carcinoma
- Sepsis complicated by haemophagocytic lymphohistiocytosis triggered by methicillin-resistant Staphylococcus aureus and human herpesvirus 8 in an immunocompromised elderly patient: A case report
- Sarcopenia in liver transplantation: A comprehensive bibliometric study of current research trends and future directions
- Advances in cancer immunotherapy and future directions in personalized medicine
- Can coronavirus disease 2019 affect male fertility or cause spontaneous abortion? A two-sample Mendelian randomization analysis
- Heat stroke associated with novel leukaemia inhibitory factor receptor gene variant in a Chinese infant
- PSME2 exacerbates ulcerative colitis by disrupting intestinal barrier function and promoting autophagy-dependent inflammation
- Hyperosmolar hyperglycemic state with severe hypernatremia coexisting with central diabetes insipidus: A case report and literature review
- Efficacy and mechanism of escin in improving the tissue microenvironment of blood vessel walls via anti-inflammatory and anticoagulant effects: Implications for clinical practice
- Merkel cell carcinoma: Clinicopathological analysis of three patients and literature review
- Genetic variants in VWF exon 26 and their implications for type 1 Von Willebrand disease in a Saudi Arabian population
- Lipoxin A4 improves myocardial ischemia/reperfusion injury through the Notch1-Nrf2 signaling pathway
- High levels of EPHB2 expression predict a poor prognosis and promote tumor progression in endometrial cancer
- Knockdown of SHP-2 delays renal tubular epithelial cell injury in diabetic nephropathy by inhibiting NLRP3 inflammasome-mediated pyroptosis
- Exploring the toxicity mechanisms and detoxification methods of Rhizoma Paridis
- Concomitant gastric carcinoma and primary hepatic angiosarcoma in a patient: A case report
- YAP1 inhibition protects retinal vascular endothelial cells under high glucose by inhibiting autophagy
- Identification of secretory protein related biomarkers for primary biliary cholangitis based on machine learning and experimental validation
- Integrated genomic and clinical modeling for prognostic assessment of radiotherapy response in rectal neoplasms
- Stem cell-based approaches for glaucoma treatment: a mini review
- Bacteriophage titering by optical density means: KOTE assays
- Neutrophil-related signature characterizes immune landscape and predicts prognosis of esophageal squamous cell carcinoma
- Integrated bioinformatic analysis and machine learning strategies to identify new potential immune biomarkers for Alzheimer’s disease and their targeting prediction with geniposide
- TRIM21 accelerates ferroptosis in intervertebral disc degeneration by promoting SLC7A11 ubiquitination and degradation
- TRIM21 accelerates ferroptosis in intervertebral disc degeneration by promoting SLC7A11 ubiquitination and degradation
- Histone modification and non-coding RNAs in skin aging: emerging therapeutic avenues
- A multiplicative behavioral model of DNA replication initiation in cells
- Biogenic gold nanoparticles synthesized from Pergularia daemia leaves: a novel approach for nasopharyngeal carcinoma therapy
- Creutzfeldt-Jakob disease mimicking Hashimoto’s encephalopathy: steroid response followed by decline
- Impact of semaphorin, Sema3F, on the gene transcription and protein expression of CREB and its binding protein CREBBP in primary hippocampal neurons of rats
- Iron overloaded M0 macrophages regulate hematopoietic stem cell proliferation and senescence via the Nrf2/Keap1/HO-1 pathway
- Revisiting the link between NADPH oxidase p22phox C242T polymorphism and ischemic stroke risk: an updated meta-analysis
- Exercise training preferentially modulates α1D-adrenergic receptor expression in peripheral arteries of hypertensive rats
- Overexpression of HE4/WFDC2 gene in mice leads to keratitis and corneal opacity
- Tumoral calcinosis complicating CKD-MBD in hemodialysis: a case report
- Mechanism of KLF4 Inhibition of epithelial-mesenchymal transition in gastric cancer cells
- Dissecting the molecular mechanisms of T cell infiltration in psoriatic lesions via cell-cell communication and regulatory network analysis
- Circadian rhythm-based prognostic features predict immune infiltration and tumor microenvironment in molecular subtypes of hepatocellular carcinoma
- Ecology and Environmental Science
- Optimization and comparative study of Bacillus consortia for cellulolytic potential and cellulase enzyme activity
- The complete mitochondrial genome analysis of Haemaphysalis hystricis Supino, 1897 (Ixodida: Ixodidae) and its phylogenetic implications
- Epidemiological characteristics and risk factors analysis of multidrug-resistant tuberculosis among tuberculosis population in Huzhou City, Eastern China
- Indices of human impacts on landscapes: How do they reflect the proportions of natural habitats?
- Genetic analysis of the Siberian flying squirrel population in the northern Changbai Mountains, Northeast China: Insights into population status and conservation
- Diversity and environmental drivers of Suillus communities in Pinus sylvestris var. mongolica forests of Inner Mongolia
- Global assessment of the fate of nitrogen deposition in forest ecosystems: Insights from 15N tracer studies
- Fungal and bacterial pathogenic co-infections mainly lead to the assembly of microbial community in tobacco stems
- Influencing of coal industry related airborne particulate matter on ocular surface tear film injury and inflammatory factor expression in Sprague-Dawley rats
- Temperature-dependent development, predation, and life table of Sphaerophoria macrogaster (Thomson) (Diptera: Syrphidae) feeding on Myzus persicae (Sulzer) (Homoptera: Aphididae)
- Eleonora’s falcon trophic interactions with insects within its breeding range: A systematic review
- Agriculture
- Integrated analysis of transcriptome, sRNAome, and degradome involved in the drought-response of maize Zhengdan958
- Variation in flower frost tolerance among seven apple cultivars and transcriptome response patterns in two contrastingly frost-tolerant selected cultivars
- Heritability of durable resistance to stripe rust in bread wheat (Triticum aestivum L.)
- Molecular mechanism of follicular development in laying hens based on the regulation of water metabolism
- Molecular identification and control studies on Coridius sp. (Hemiptera: Dinidoridae) in Al-Khamra, south of Jeddah, Saudi Arabia
- 10.1515/biol-2025-1218
- Animal Science
- Effect of sex ratio on the life history traits of an important invasive species, Spodoptera frugiperda
- Plant Sciences
- Hairpin in a haystack: In silico identification and characterization of plant-conserved microRNA in Rafflesiaceae
- Widely targeted metabolomics of different tissues in Rubus corchorifolius
- The complete chloroplast genome of Gerbera piloselloides (L.) Cass., 1820 (Carduoideae, Asteraceae) and its phylogenetic analysis
- Field trial to correlate mineral solubilization activity of Pseudomonas aeruginosa and biochemical content of groundnut plants
- Correlation analysis between semen routine parameters and sperm DNA fragmentation index in patients with semen non-liquefaction: A retrospective study
- Plasticity of the anatomical traits of Rhododendron L. (Ericaceae) leaves and its implications in adaptation to the plateau environment
- Effects of Piriformospora indica and arbuscular mycorrhizal fungus on growth and physiology of Moringa oleifera under low-temperature stress
- Effects of different sources of potassium fertiliser on yield, fruit quality and nutrient absorption in “Harward” kiwifruit (Actinidia deliciosa)
- Comparative efficiency and residue levels of spraying programs against powdery mildew in grape varieties
- The DREB7 transcription factor enhances salt tolerance in soybean plants under salt stress
- Using plant electrical signals of water hyacinth (Eichhornia crassipes) for water pollution monitoring
- Response of hybrid grapes (Vitis spp.) to two biotic stress factors and their seedlessness status
- Metabolomic profiling reveals systemic metabolic reprogramming in Alternaria alternata under salt stress
- Effects of mixed salinity and alkali stress on photosynthetic characteristics and PEPC gene expression of vegetable soybean seedlings
- Food Science
- Phytochemical analysis of Stachys iva: Discovering the optimal extract conditions and its bioactive compounds
- Review on role of honey in disease prevention and treatment through modulation of biological activities
- Computational analysis of polymorphic residues in maltose and maltotriose transporters of a wild Saccharomyces cerevisiae strain
- Optimization of phenolic compound extraction from Tunisian squash by-products: A sustainable approach for antioxidant and antibacterial applications
- Liupao tea aqueous extract alleviates dextran sulfate sodium-induced ulcerative colitis in rats by modulating the gut microbiota
- Toxicological qualities and detoxification trends of fruit by-products for valorization: A review
- Polyphenolic spectrum of cornelian cherry fruits and their health-promoting effect
- Optimizing the encapsulation of the refined extract of squash peels for functional food applications: A sustainable approach to reduce food waste
- Advancements in curcuminoid formulations: An update on bioavailability enhancement strategies curcuminoid bioavailability and formulations
- Impact of saline sprouting on antioxidant properties and bioactive compounds in chia seeds
- The dilemma of food genetics and improvement
- Causal effects of trace elements on congenital foot deformities and their subtypes: a Mendelian randomization study with gut microbiota mediation
- Honey meets acidity: a novel biopreservative approach against foodborne pathogens
- Bioengineering and Biotechnology
- Impact of hyaluronic acid-modified hafnium metalorganic frameworks containing rhynchophylline on Alzheimer’s disease
- Emerging patterns in nanoparticle-based therapeutic approaches for rheumatoid arthritis: A comprehensive bibliometric and visual analysis spanning two decades
- Application of CRISPR/Cas gene editing for infectious disease control in poultry
- Preparation of hafnium nitride-coated titanium implants by magnetron sputtering technology and evaluation of their antibacterial properties and biocompatibility
- Preparation and characterization of lemongrass oil nanoemulsion: Antimicrobial, antibiofilm, antioxidant, and anticancer activities
- Fluorescent detection of sialic acid–binding lectins using functionalized quantum dots in ELISA format
- Smart tectorigenin-loaded ZnO hydrogel nanocomposites for targeted wound healing: synthesis, characterization, and biological evaluation
- Corrigendum
- Corrigendum to “Utilization of convolutional neural networks to analyze microscopic images for high-throughput screening of mesenchymal stem cells”
- Corrigendum to “Effects of Ire1 gene on virulence and pathogenicity of Candida albicans”
- Retraction
- Retraction of “Down-regulation of miR-539 indicates poor prognosis in patients with pancreatic cancer”
Articles in the same Issue
- Safety assessment and modulation of hepatic CYP3A4 and UGT enzymes by Glycyrrhiza glabra aqueous extract in female Sprague–Dawley rats
- Adult-onset Still’s disease with hemophagocytic lymphohistiocytosis and minimal change disease
- Role of DZ2002 in reducing corneal graft rejection in rats by influencing Th17 activation via inhibition of the PI3K/AKT pathway and downregulation of TRAF1
- Biomedical Sciences
- Mechanism of triptolide regulating proliferation and apoptosis of hepatoma cells by inhibiting JAK/STAT pathway
- Maslinic acid improves mitochondrial function and inhibits oxidative stress and autophagy in human gastric smooth muscle cells
- Comparative analysis of inflammatory biomarkers for the diagnosis of neonatal sepsis: IL-6, IL-8, SAA, CRP, and PCT
- Post-pandemic insights on COVID-19 and premature ovarian insufficiency
- Proteome differences of dental stem cells between permanent and deciduous teeth by data-independent acquisition proteomics
- Optimizing a modified cetyltrimethylammonium bromide protocol for fungal DNA extraction: Insights from multilocus gene amplification
- Preliminary analysis of the role of small hepatitis B surface proteins mutations in the pathogenesis of occult hepatitis B infection via the endoplasmic reticulum stress-induced UPR-ERAD pathway
- Efficacy of alginate-coated gold nanoparticles against antibiotics-resistant Staphylococcus and Streptococcus pathogens of acne origins
- Battling COVID-19 leveraging nanobiotechnology: Gold and silver nanoparticle–B-escin conjugates as SARS-CoV-2 inhibitors
- Neurodegenerative diseases and neuroinflammation-induced apoptosis
- Impact of fracture fixation surgery on cognitive function and the gut microbiota in mice with a history of stroke
- COLEC10: A potential tumor suppressor and prognostic biomarker in hepatocellular carcinoma through modulation of EMT and PI3K-AKT pathways
- High-temperature requirement serine protease A2 inhibitor UCF-101 ameliorates damaged neurons in traumatic brain-injured rats by the AMPK/NF-κB pathway
- SIK1 inhibits IL-1β-stimulated cartilage apoptosis and inflammation in vitro through the CRTC2/CREB1 signaling
- Rutin–chitooligosaccharide complex: Comprehensive evaluation of its anti-inflammatory and analgesic properties in vitro and in vivo
- Knockdown of Aurora kinase B alleviates high glucose-triggered trophoblast cells damage and inflammation during gestational diabetes
- Calcium-sensing receptors promoted Homer1 expression and osteogenic differentiation in bone marrow mesenchymal stem cells
- ABI3BP can inhibit the proliferation, invasion, and epithelial–mesenchymal transition of non-small-cell lung cancer cells
- Changes in blood glucose and metabolism in hyperuricemia mice
- Rapid detection of the GJB2 c.235delC mutation based on CRISPR-Cas13a combined with lateral flow dipstick
- IL-11 promotes Ang II-induced autophagy inhibition and mitochondrial dysfunction in atrial fibroblasts
- Short-chain fatty acid attenuates intestinal inflammation by regulation of gut microbial composition in antibiotic-associated diarrhea
- Application of metagenomic next-generation sequencing in the diagnosis of pathogens in patients with diabetes complicated by community-acquired pneumonia
- NAT10 promotes radiotherapy resistance in non-small cell lung cancer by regulating KPNB1-mediated PD-L1 nuclear translocation
- Phytol-mixed micelles alleviate dexamethasone-induced osteoporosis in zebrafish: Activation of the MMP3–OPN–MAPK pathway-mediating bone remodeling
- Association between TGF-β1 and β-catenin expression in the vaginal wall of patients with pelvic organ prolapse
- Primary pleomorphic liposarcoma involving bilateral ovaries: Case report and literature review
- Effects of de novo donor-specific Class I and II antibodies on graft outcomes after liver transplantation: A pilot cohort study
- Sleep architecture in Alzheimer’s disease continuum: The deep sleep question
- Ephedra fragilis plant extract: A groundbreaking corrosion inhibitor for mild steel in acidic environments – electrochemical, EDX, DFT, and Monte Carlo studies
- Langerhans cell histiocytosis in an adult patient with upper jaw and pulmonary involvement: A case report
- Inhibition of mast cell activation by Jaranol-targeted Pirin ameliorates allergic responses in mouse allergic rhinitis
- Aeromonas veronii-induced septic arthritis of the hip in a child with acute lymphoblastic leukemia
- Clusterin activates the heat shock response via the PI3K/Akt pathway to protect cardiomyocytes from high-temperature-induced apoptosis
- Research progress on fecal microbiota transplantation in tumor prevention and treatment
- Low-pressure exposure influences the development of HAPE
- Stigmasterol alleviates endplate chondrocyte degeneration through inducing mitophagy by enhancing PINK1 mRNA acetylation via the ESR1/NAT10 axis
- AKAP12, mediated by transcription factor 21, inhibits cell proliferation, metastasis, and glycolysis in lung squamous cell carcinoma
- Association between PAX9 or MSX1 gene polymorphism and tooth agenesis risk: A meta-analysis
- A case of bloodstream infection caused by Neisseria gonorrhoeae
- Case of nasopharyngeal tuberculosis complicated with cervical lymph node and pulmonary tuberculosis
- p-Cymene inhibits pro-fibrotic and inflammatory mediators to prevent hepatic dysfunction
- GFPT2 promotes paclitaxel resistance in epithelial ovarian cancer cells via activating NF-κB signaling pathway
- Transfer RNA-derived fragment tRF-36 modulates varicose vein progression via human vascular smooth muscle cell Notch signaling
- RTA-408 attenuates the hepatic ischemia reperfusion injury in mice possibly by activating the Nrf2/HO-1 signaling pathway
- Decreased serum TIMP4 levels in patients with rheumatoid arthritis
- Sirt1 protects lupus nephritis by inhibiting the NLRP3 signaling pathway in human glomerular mesangial cells
- Sodium butyrate aids brain injury repair in neonatal rats
- Interaction of MTHFR polymorphism with PAX1 methylation in cervical cancer
- Convallatoxin inhibits proliferation and angiogenesis of glioma cells via regulating JAK/STAT3 pathway
- The effect of the PKR inhibitor, 2-aminopurine, on the replication of influenza A virus, and segment 8 mRNA splicing
- Effects of Ire1 gene on virulence and pathogenicity of Candida albicans
- Small cell lung cancer with small intestinal metastasis: Case report and literature review
- GRB14: A prognostic biomarker driving tumor progression in gastric cancer through the PI3K/AKT signaling pathway by interacting with COBLL1
- 15-Lipoxygenase-2 deficiency induces foam cell formation that can be restored by salidroside through the inhibition of arachidonic acid effects
- FTO alleviated the diabetic nephropathy progression by regulating the N6-methyladenosine levels of DACT1
- Clinical relevance of inflammatory markers in the evaluation of severity of ulcerative colitis: A retrospective study
- Zinc valproic acid complex promotes osteoblast differentiation and exhibits anti-osteoporotic potential
- Primary pulmonary synovial sarcoma in the bronchial cavity: A case report
- Metagenomic next-generation sequencing of alveolar lavage fluid improves the detection of pulmonary infection
- Uterine tumor resembling ovarian sex cord tumor with extensive rhabdoid differentiation: A case report
- Genomic analysis of a novel ST11(PR34365) Clostridioides difficile strain isolated from the human fecal of a CDI patient in Guizhou, China
- Effects of tiered cardiac rehabilitation on CRP, TNF-α, and physical endurance in older adults with coronary heart disease
- Changes in T-lymphocyte subpopulations in patients with colorectal cancer before and after acupoint catgut embedding acupuncture observation
- Modulating the tumor microenvironment: The role of traditional Chinese medicine in improving lung cancer treatment
- Alterations of metabolites related to microbiota–gut–brain axis in plasma of colon cancer, esophageal cancer, stomach cancer, and lung cancer patients
- Research on individualized drug sensitivity detection technology based on bio-3D printing technology for precision treatment of gastrointestinal stromal tumors
- CEBPB promotes ulcerative colitis-associated colorectal cancer by stimulating tumor growth and activating the NF-κB/STAT3 signaling pathway
- Oncolytic bacteria: A revolutionary approach to cancer therapy
- A de novo meningioma with rapid growth: A possible malignancy imposter?
- Diagnosis of secondary tuberculosis infection in an asymptomatic elderly with cancer using next-generation sequencing: Case report
- Hesperidin and its zinc(ii) complex enhance osteoblast differentiation and bone formation: In vitro and in vivo evaluations
- Research progress on the regulation of autophagy in cardiovascular diseases by chemokines
- Anti-arthritic, immunomodulatory, and inflammatory regulation by the benzimidazole derivative BMZ-AD: Insights from an FCA-induced rat model
- Immunoassay for pyruvate kinase M1/2 as an Alzheimer’s biomarker in CSF
- The role of HDAC11 in age-related hearing loss: Mechanisms and therapeutic implications
- Evaluation and application analysis of animal models of PIPNP based on data mining
- Therapeutic approaches for liver fibrosis/cirrhosis by targeting pyroptosis
- Fabrication of zinc oxide nanoparticles using Ruellia tuberosa leaf extract induces apoptosis through P53 and STAT3 signalling pathways in prostate cancer cells
- Haplo-hematopoietic stem cell transplantation and immunoradiotherapy for severe aplastic anemia complicated with nasopharyngeal carcinoma: A case report
- Modulation of the KEAP1-NRF2 pathway by Erianin: A novel approach to reduce psoriasiform inflammation and inflammatory signaling
- The expression of epidermal growth factor receptor 2 and its relationship with tumor-infiltrating lymphocytes and clinical pathological features in breast cancer patients
- Innovations in MALDI-TOF Mass Spectrometry: Bridging modern diagnostics and historical insights
- BAP1 complexes with YY1 and RBBP7 and its downstream targets in ccRCC cells
- Hypereosinophilic syndrome with elevated IgG4 and T-cell clonality: A report of two cases
- Electroacupuncture alleviates sciatic nerve injury in sciatica rats by regulating BDNF and NGF levels, myelin sheath degradation, and autophagy
- Polydatin prevents cholesterol gallstone formation by regulating cholesterol metabolism via PPAR-γ signaling
- RNF144A and RNF144B: Important molecules for health
- Analysis of the detection rate and related factors of thyroid nodules in the healthy population
- Artesunate inhibits hepatocellular carcinoma cell migration and invasion through OGA-mediated O-GlcNAcylation of ZEB1
- Endovascular management of post-pancreatectomy hemorrhage caused by a hepatic artery pseudoaneurysm: Case report and review of the literature
- Efficacy and safety of anti-PD-1/PD-L1 antibodies in patients with relapsed refractory diffuse large B-cell lymphoma: A meta-analysis
- SATB2 promotes humeral fracture healing in rats by activating the PI3K/AKT pathway
- Overexpression of the ferroptosis-related gene, NFS1, corresponds to gastric cancer growth and tumor immune infiltration
- Understanding risk factors and prognosis in diabetic foot ulcers
- Atractylenolide I alleviates the experimental allergic response in mice by suppressing TLR4/NF-kB/NLRP3 signalling
- FBXO31 inhibits the stemness characteristics of CD147 (+) melanoma stem cells
- Immune molecule diagnostics in colorectal cancer: CCL2 and CXCL11
- Inhibiting CXCR6 promotes senescence of activated hepatic stellate cells with limited proinflammatory SASP to attenuate hepatic fibrosis
- Cadmium toxicity, health risk and its remediation using low-cost biochar adsorbents
- Pulmonary cryptococcosis with headache as the first presentation: A case report
- Solitary pulmonary metastasis with cystic airspaces in colon cancer: A rare case report
- RUNX1 promotes denervation-induced muscle atrophy by activating the JUNB/NF-κB pathway and driving M1 macrophage polarization
- Morphometric analysis and immunobiological investigation of Indigofera oblongifolia on the infected lung with Plasmodium chabaudi
- The NuA4/TIP60 histone-modifying complex and Hr78 modulate the Lobe2 mutant eye phenotype
- Experimental study on salmon demineralized bone matrix loaded with recombinant human bone morphogenetic protein-2: In vitro and in vivo study
- A case of IgA nephropathy treated with a combination of telitacicept and half-dose glucocorticoids
- Analgesic and toxicological evaluation of cannabidiol-rich Moroccan Cannabis sativa L. (Khardala variety) extract: Evidence from an in vivo and in silico study
- Wound healing and signaling pathways
- Combination of immunotherapy and whole-brain radiotherapy on prognosis of patients with multiple brain metastases: A retrospective cohort study
- To explore the relationship between endometrial hyperemia and polycystic ovary syndrome
- Research progress on the impact of curcumin on immune responses in breast cancer
- Biogenic Cu/Ni nanotherapeutics from Descurainia sophia (L.) Webb ex Prantl seeds for the treatment of lung cancer
- Dapagliflozin attenuates atrial fibrosis via the HMGB1/RAGE pathway in atrial fibrillation rats
- Glycitein alleviates inflammation and apoptosis in keratinocytes via ROS-associated PI3K–Akt signalling pathway
- ADH5 inhibits proliferation but promotes EMT in non-small cell lung cancer cell through activating Smad2/Smad3
- Apoptotic efficacies of AgNPs formulated by Syzygium aromaticum leaf extract on 32D-FLT3-ITD human leukemia cell line with PI3K/AKT/mTOR signaling pathway
- Novel cuproptosis-related genes C1QBP and PFKP identified as prognostic and therapeutic targets in lung adenocarcinoma
- Bee venom promotes exosome secretion and alters miRNA cargo in T cells
- Treatment of pure red cell aplasia in a chronic kidney disease patient with roxadustat: A case report
- Comparative bioinformatics analysis of the Wnt pathway in breast cancer: Selection of novel biomarker panels associated with ER status
- Kynurenine facilitates renal cell carcinoma progression by suppressing M2 macrophage pyroptosis through inhibition of CASP1 cleavage
- RFX5 promotes the growth, motility, and inhibits apoptosis of gastric adenocarcinoma cells through the SIRT1/AMPK axis
- ALKBH5 exacerbates early cardiac damage after radiotherapy for breast cancer via m6A demethylation of TLR4
- Phytochemicals of Roman chamomile: Antioxidant, anti-aging, and whitening activities of distillation residues
- Circadian gene Cry1 inhibits the tumorigenicity of hepatocellular carcinoma by the BAX/BCL2-mediated apoptosis pathway
- The TNFR-RIPK1/RIPK3 signalling pathway mediates the effect of lanthanum on necroptosis of nerve cells
- Longitudinal monitoring of autoantibody dynamics in patients with early-stage non-small-cell lung cancer undergoing surgery
- The potential role of rutin, a flavonoid, in the management of cancer through modulation of cell signaling pathways
- Construction of pectinase gene engineering microbe and its application in tobacco sheets
- Construction of a microbial abundance prognostic scoring model based on intratumoral microbial data for predicting the prognosis of lung squamous cell carcinoma
- Sepsis complicated by haemophagocytic lymphohistiocytosis triggered by methicillin-resistant Staphylococcus aureus and human herpesvirus 8 in an immunocompromised elderly patient: A case report
- Sarcopenia in liver transplantation: A comprehensive bibliometric study of current research trends and future directions
- Advances in cancer immunotherapy and future directions in personalized medicine
- Can coronavirus disease 2019 affect male fertility or cause spontaneous abortion? A two-sample Mendelian randomization analysis
- Heat stroke associated with novel leukaemia inhibitory factor receptor gene variant in a Chinese infant
- PSME2 exacerbates ulcerative colitis by disrupting intestinal barrier function and promoting autophagy-dependent inflammation
- Hyperosmolar hyperglycemic state with severe hypernatremia coexisting with central diabetes insipidus: A case report and literature review
- Efficacy and mechanism of escin in improving the tissue microenvironment of blood vessel walls via anti-inflammatory and anticoagulant effects: Implications for clinical practice
- Merkel cell carcinoma: Clinicopathological analysis of three patients and literature review
- Genetic variants in VWF exon 26 and their implications for type 1 Von Willebrand disease in a Saudi Arabian population
- Lipoxin A4 improves myocardial ischemia/reperfusion injury through the Notch1-Nrf2 signaling pathway
- High levels of EPHB2 expression predict a poor prognosis and promote tumor progression in endometrial cancer
- Knockdown of SHP-2 delays renal tubular epithelial cell injury in diabetic nephropathy by inhibiting NLRP3 inflammasome-mediated pyroptosis
- Exploring the toxicity mechanisms and detoxification methods of Rhizoma Paridis
- Concomitant gastric carcinoma and primary hepatic angiosarcoma in a patient: A case report
- YAP1 inhibition protects retinal vascular endothelial cells under high glucose by inhibiting autophagy
- Identification of secretory protein related biomarkers for primary biliary cholangitis based on machine learning and experimental validation
- Integrated genomic and clinical modeling for prognostic assessment of radiotherapy response in rectal neoplasms
- Stem cell-based approaches for glaucoma treatment: a mini review
- Bacteriophage titering by optical density means: KOTE assays
- Neutrophil-related signature characterizes immune landscape and predicts prognosis of esophageal squamous cell carcinoma
- Integrated bioinformatic analysis and machine learning strategies to identify new potential immune biomarkers for Alzheimer’s disease and their targeting prediction with geniposide
- TRIM21 accelerates ferroptosis in intervertebral disc degeneration by promoting SLC7A11 ubiquitination and degradation
- TRIM21 accelerates ferroptosis in intervertebral disc degeneration by promoting SLC7A11 ubiquitination and degradation
- Histone modification and non-coding RNAs in skin aging: emerging therapeutic avenues
- A multiplicative behavioral model of DNA replication initiation in cells
- Biogenic gold nanoparticles synthesized from Pergularia daemia leaves: a novel approach for nasopharyngeal carcinoma therapy
- Creutzfeldt-Jakob disease mimicking Hashimoto’s encephalopathy: steroid response followed by decline
- Impact of semaphorin, Sema3F, on the gene transcription and protein expression of CREB and its binding protein CREBBP in primary hippocampal neurons of rats
- Iron overloaded M0 macrophages regulate hematopoietic stem cell proliferation and senescence via the Nrf2/Keap1/HO-1 pathway
- Revisiting the link between NADPH oxidase p22phox C242T polymorphism and ischemic stroke risk: an updated meta-analysis
- Exercise training preferentially modulates α1D-adrenergic receptor expression in peripheral arteries of hypertensive rats
- Overexpression of HE4/WFDC2 gene in mice leads to keratitis and corneal opacity
- Tumoral calcinosis complicating CKD-MBD in hemodialysis: a case report
- Mechanism of KLF4 Inhibition of epithelial-mesenchymal transition in gastric cancer cells
- Dissecting the molecular mechanisms of T cell infiltration in psoriatic lesions via cell-cell communication and regulatory network analysis
- Circadian rhythm-based prognostic features predict immune infiltration and tumor microenvironment in molecular subtypes of hepatocellular carcinoma
- Ecology and Environmental Science
- Optimization and comparative study of Bacillus consortia for cellulolytic potential and cellulase enzyme activity
- The complete mitochondrial genome analysis of Haemaphysalis hystricis Supino, 1897 (Ixodida: Ixodidae) and its phylogenetic implications
- Epidemiological characteristics and risk factors analysis of multidrug-resistant tuberculosis among tuberculosis population in Huzhou City, Eastern China
- Indices of human impacts on landscapes: How do they reflect the proportions of natural habitats?
- Genetic analysis of the Siberian flying squirrel population in the northern Changbai Mountains, Northeast China: Insights into population status and conservation
- Diversity and environmental drivers of Suillus communities in Pinus sylvestris var. mongolica forests of Inner Mongolia
- Global assessment of the fate of nitrogen deposition in forest ecosystems: Insights from 15N tracer studies
- Fungal and bacterial pathogenic co-infections mainly lead to the assembly of microbial community in tobacco stems
- Influencing of coal industry related airborne particulate matter on ocular surface tear film injury and inflammatory factor expression in Sprague-Dawley rats
- Temperature-dependent development, predation, and life table of Sphaerophoria macrogaster (Thomson) (Diptera: Syrphidae) feeding on Myzus persicae (Sulzer) (Homoptera: Aphididae)
- Eleonora’s falcon trophic interactions with insects within its breeding range: A systematic review
- Agriculture
- Integrated analysis of transcriptome, sRNAome, and degradome involved in the drought-response of maize Zhengdan958
- Variation in flower frost tolerance among seven apple cultivars and transcriptome response patterns in two contrastingly frost-tolerant selected cultivars
- Heritability of durable resistance to stripe rust in bread wheat (Triticum aestivum L.)
- Molecular mechanism of follicular development in laying hens based on the regulation of water metabolism
- Molecular identification and control studies on Coridius sp. (Hemiptera: Dinidoridae) in Al-Khamra, south of Jeddah, Saudi Arabia
- 10.1515/biol-2025-1218
- Animal Science
- Effect of sex ratio on the life history traits of an important invasive species, Spodoptera frugiperda
- Plant Sciences
- Hairpin in a haystack: In silico identification and characterization of plant-conserved microRNA in Rafflesiaceae
- Widely targeted metabolomics of different tissues in Rubus corchorifolius
- The complete chloroplast genome of Gerbera piloselloides (L.) Cass., 1820 (Carduoideae, Asteraceae) and its phylogenetic analysis
- Field trial to correlate mineral solubilization activity of Pseudomonas aeruginosa and biochemical content of groundnut plants
- Correlation analysis between semen routine parameters and sperm DNA fragmentation index in patients with semen non-liquefaction: A retrospective study
- Plasticity of the anatomical traits of Rhododendron L. (Ericaceae) leaves and its implications in adaptation to the plateau environment
- Effects of Piriformospora indica and arbuscular mycorrhizal fungus on growth and physiology of Moringa oleifera under low-temperature stress
- Effects of different sources of potassium fertiliser on yield, fruit quality and nutrient absorption in “Harward” kiwifruit (Actinidia deliciosa)
- Comparative efficiency and residue levels of spraying programs against powdery mildew in grape varieties
- The DREB7 transcription factor enhances salt tolerance in soybean plants under salt stress
- Using plant electrical signals of water hyacinth (Eichhornia crassipes) for water pollution monitoring
- Response of hybrid grapes (Vitis spp.) to two biotic stress factors and their seedlessness status
- Metabolomic profiling reveals systemic metabolic reprogramming in Alternaria alternata under salt stress
- Effects of mixed salinity and alkali stress on photosynthetic characteristics and PEPC gene expression of vegetable soybean seedlings
- Food Science
- Phytochemical analysis of Stachys iva: Discovering the optimal extract conditions and its bioactive compounds
- Review on role of honey in disease prevention and treatment through modulation of biological activities
- Computational analysis of polymorphic residues in maltose and maltotriose transporters of a wild Saccharomyces cerevisiae strain
- Optimization of phenolic compound extraction from Tunisian squash by-products: A sustainable approach for antioxidant and antibacterial applications
- Liupao tea aqueous extract alleviates dextran sulfate sodium-induced ulcerative colitis in rats by modulating the gut microbiota
- Toxicological qualities and detoxification trends of fruit by-products for valorization: A review
- Polyphenolic spectrum of cornelian cherry fruits and their health-promoting effect
- Optimizing the encapsulation of the refined extract of squash peels for functional food applications: A sustainable approach to reduce food waste
- Advancements in curcuminoid formulations: An update on bioavailability enhancement strategies curcuminoid bioavailability and formulations
- Impact of saline sprouting on antioxidant properties and bioactive compounds in chia seeds
- The dilemma of food genetics and improvement
- Causal effects of trace elements on congenital foot deformities and their subtypes: a Mendelian randomization study with gut microbiota mediation
- Honey meets acidity: a novel biopreservative approach against foodborne pathogens
- Bioengineering and Biotechnology
- Impact of hyaluronic acid-modified hafnium metalorganic frameworks containing rhynchophylline on Alzheimer’s disease
- Emerging patterns in nanoparticle-based therapeutic approaches for rheumatoid arthritis: A comprehensive bibliometric and visual analysis spanning two decades
- Application of CRISPR/Cas gene editing for infectious disease control in poultry
- Preparation of hafnium nitride-coated titanium implants by magnetron sputtering technology and evaluation of their antibacterial properties and biocompatibility
- Preparation and characterization of lemongrass oil nanoemulsion: Antimicrobial, antibiofilm, antioxidant, and anticancer activities
- Fluorescent detection of sialic acid–binding lectins using functionalized quantum dots in ELISA format
- Smart tectorigenin-loaded ZnO hydrogel nanocomposites for targeted wound healing: synthesis, characterization, and biological evaluation
- Corrigendum
- Corrigendum to “Utilization of convolutional neural networks to analyze microscopic images for high-throughput screening of mesenchymal stem cells”
- Corrigendum to “Effects of Ire1 gene on virulence and pathogenicity of Candida albicans”
- Retraction
- Retraction of “Down-regulation of miR-539 indicates poor prognosis in patients with pancreatic cancer”

