Abstract
This study evaluated the hepatoprotective potential of p-cymene (p-CYM) against two models of liver damage: ethanol (EtOH)-induced hepatocellular injury and diethylnitrosamine-carbon tetrachloride (DEN-CCl4)-induced liver fibrosis (LF). HepG2 cells were treated with p-CYM or silymarin (SIL) before exposure to 10% EtOH in order to induce cellular injury. LF was induced in Sprague–Dawley rats using a single dose of DEN followed by increasing doses of CCl4 over 60 days. Rats were treated twice weekly with p-CYM or SIL from day 21 to day 60. Results showed that p-CYM effectively mitigated EtOH-induced cell death in HepG2 cells by enhancing the activity of superoxide dismutase and glutathione reductase. In vivo findings revealed that p-CYM attenuated DEN– CCl4-induced liver damage by preventing weight loss, improving serum biomarkers (e.g., aspartate transaminase, alanine aminotransferase, alkaline phosphatase, and bilirubin), and reducing liver fibrotic changes. It also decreased the expression of pro-inflammatory and pro-fibrotic markers such as TNF-α, IL-1β, IL-6, TGF-β1, COL1A1, and TIMP-1. Molecular docking further supported the experimental findings, showing strong interactions between p-CYM and the target proteins. These results indicate that the hepatoprotective effects of p-CYM are likely due to its combined antioxidant, anti-inflammatory, and anti-fibrotic properties.
Graphical abstract

1 Introduction
Liver fibrosis (LF) is a condition characterized by the excessive accumulation of extracellular matrix (ECM) proteins, primarily type I and III cross-linked collagens, which form fibrous scars in response to chronic liver injury. This scarring replaces damaged tissue and impairs liver functionality [1]. LF typically results from two primary types of chronic liver damage: hepatotoxic and cholestatic. Cholestatic damage occurs due to bile flow obstruction caused by conditions such as primary and secondary biliary cholangitis, sclerosing cholangitis, and biliary atresia [2]. Hepatotoxic damage, on the other hand, is caused by factors such as alcohol, carbon tetrachloride (CCl4), paracetamol, and metabolic syndrome, which lead to steatohepatitis and chronic hepatocyte injury [3,4,5].
Alcohol consumption accounts for approximately 5% of deaths worldwide, with the liver being the primary site for ethanol (EtOH) metabolism [3]. EtOH is metabolized into acetaldehyde, a toxic byproduct, which is further broken down into acetate by acetaldehyde dehydrogenase in liver mitochondria. However, acetaldehyde accumulation and the production of ethyl esters of long-chain fatty acids through non-oxidative metabolic pathways disrupt mitochondrial function, making the liver particularly vulnerable to alcohol-induced damage [5,6,7]. Similarly, CCl4 exposure can cause centrilobular hepatic necrosis. Both EtOH and CCl4 are metabolized by cytochrome P450 2E1 (CYP2E1), which generates reactive free radicals that contribute to severe hepatotoxicity [4,5,8].
Despite advancements, treatment options for LF remain limited. A variety of drugs, including thalidomide, colchicine, corticosteroids, curcumin, glycyrrhizin, interferons, nitric oxide, resveratrol, silymarin (SIL), and sulfoadenosyl methionine, have gained attention for their anti-fibrotic properties [9,10]. Glycyrrhizin has also shown hepatoprotective effects in patients with sub-acute liver failure, but additional controlled clinical trials are needed [11]. Similarly, thalidomide, resveratrol, and curcumin have demonstrated potential as preventive and therapeutic agents for liver diseases, though their effectiveness across larger populations remains unproven. In cases of end-stage liver disease, liver transplantation remains the only definitive treatment, emphasizing the need for novel therapeutic options to enhance patient outcomes [12].
Medicinal herbs have been used for centuries to treat various diseases, and natural compounds derived from plants continue to attract significant interest in modern medicine [13,14]. One such compound, p-cymene (p-CYM), is an alkyl-substituted aromatic compound with a wide range of pharmacological properties, including antioxidant, anti-inflammatory, antibacterial, antifungal, antiviral, anti-parasitic, anti-diabetic, and anticancer effects [15,16]. A recent study highlighted that p-CYM enhanced the levels of anti-oxidants and reduced inflammatory cytokines in hyperlipidemic rats [17]. Given its reported anti-inflammatory activities and diverse therapeutic potential, p-CYM has been hypothesized to offer hepatoprotective benefits. This hypothesis was tested using a human hepatoma (HepG2) cell line and an LF model to assess its efficacy in mitigating hepatic damage and protecting liver function.
2 Materials and methods
2.1 Reagents
CCl4 (Cat. No. 289116) and SIL (Cat. No. 65666-07-01) were purchased from Sigma-Aldrich Company (St. Louis, MO, USA). Diethylnitrosamine (DEN; Cat. No. 55-18-5) was purchased from Rhawn Chemicals (Shanghai, China) and p-CYM (Cat. No. 99-87-6) from Tokyo Chemical Industry (Tokyo, Japan). All other chemicals used in this research were of standard analytical quality.
2.2 Culturing of HepG2 cells
HepG2 cell line was obtained from Cell and Tissue Culture Laboratory (The University of Lahore). Cells were cultured in Dulbecco’s modified Eagle’s medium (DMEM; Cat. No. D5030) supplemented with streptomycin (100 g/ml; Cat. No. S9137), penicillin (100 units/ml; Cat. No. P3032), and 10% fetal bovine serum (Cat. No. F4135) in a humidified incubator at 37°C. Subculturing was done when the cells attained a confluency of 70–80%. Cells were washed with 1× PBS, and adherent cells were detached with 1× trypsin (Cat. No. T4799). The cellular detachment was verified using a phase-contrast inverted microscope. The cell suspension was centrifuged at 2,000 rpm for 5 min, and the obtained cell pellets were resuspended in complete DMEM [18].
2.3 Cytotoxicity assessment of p-CYM and EtOH
Cell viability assay was conducted to determine the optimal concentrations of p-CYM and EtOH. Different concentrations of EtOH (1, 3, 5, 8, and 10%) were prepared in complete DMEM, and 1 M stock solution of p-CYM in DMSO (Cat. No. D8418) was prepared. Several dilutions of p-CYM (10, 50, 100, 500, and 1,000 µM) were later formulated from 1 M stock solution. HepG2 cells were seeded on a 96-well plate and incubated at 37℃ overnight. The next day, the medium was removed, and the cells were washed with 1× PBS. Various concentrations of EtOH and p-CYM (100 µl) were introduced into the wells. Cell viability of the treated cells was assessed using the 3-[4,5-dimethylthiazol-2-yl]-2,5 diphenyl tetrazolium bromide (MTT) assay according to the manufacturer’s protocol [19].
2.4 Determination of the hepatoprotective effect of p-CYM
HepG2 cells were cultivated on 96- and 6-well plates, and after overnight incubation, they were washed with 1× PBS and pretreated with 100 µl of either SIL (200 µg/ml) or various doses of p-CYM for 24 h. Following p-CYM/SIL treatments, cells were again rinsed with 1× PBS and subsequently treated with 10% EtOH for 24 h. After EtOH injury, cells grown on a 96-well plate were subjected to the MTT, Trypan blue, crystal violet, and PI staining assays. Cells grown on 6-well plates were harvested in TRIZOL reagent for gene expression study, while supernatants were collected for ELISA and anti-oxidant assays [19].
HepG2 cells were divided into the following groups (n = 3 in each group):
Control: complete DMEM
DMSO control: 0.1% DMSO dissolved in complete DMEM
Disease control: 10% EtOH dissolved in complete DMEM
SIL (200 µg/ml): 200 µg/ml SIL in complete DMEM
P-CYM 10 µM: 10 µM p-CYM in complete DMEM
P-CYM 50 µM: 50 µM p-CYM in complete DMEM
P-CYM 100 µM: 100 µM p-CYM in complete DMEM
P-CYM 500 µM: 500 µM p-CYM in complete DMEM
2.5 Cell viability assays
In order to calculate the cell viability of pretreated HepG2 cells, MTT and crystal violet assays were performed in which different concentrations of the abovementioned dilutions were tested on cells cultured in 96-well plates.
2.5.1 MTT assay
Pretreated cells were washed with 1× PBS followed by 3–4 h of incubation with 100 µl of DMEM and 25 µl of MTT (Cat. No. M5655) solution. Formazan crystals were solubilized with 10% sodium dodecyl sulfate (SDS), and absorbance at 570 nm was measured using a microplate reader. Percentage cell viability was calculated from the mean absorbance values [18].
2.5.2 Crystal violet assay
Pretreated cells were rinsed with 1× PBS and treated with a mixture of 0.1% crystal violet dye and 2% EtOH, followed by incubation for 15 min at room temperature (RT). Wells were thoroughly washed with 1× PBS, and the dye was carefully disposed of to prevent cells from lifting out of the wells. The stain was then solubilized by adding 100 µl of 1% SDS to each well. Finally, the absorbance was measured at 595 nm using a microplate reader [18].
2.6 Dead cell detection
For dead cell detection, a trypan blue assay was performed.
2.6.1 Trypan blue staining
Trypan blue reagent was used to distinguish between live and dead cells. Briefly, pretreated cells were washed three times with 1× PBS and subsequently stained with trypan blue (Cat. No. T6146). The blue-stained cells were designated as dead, which were counted using a compound microscope.
2.7 Antioxidant assays
2.7.1 Glutathione reductase (GSH) assay
GSH levels in the samples were quantified using the Bioassay Technology Laboratory ELISA Kit (Cat. No. EA0142Hu). Reagents, standard solutions, and samples were prepared according to the kit’s instructions and equilibrated to RT prior to use. For the assay, 50 µl of the standard solution was added to the standard wells, and 40 µl of the sample was added to the sample wells. Subsequently, 10 µl of anti-GSH antibody was added to each well, followed by 50 µl of streptavidin-HRP to the sample wells. The contents were mixed thoroughly, sealed with a plate sealer, and incubated at 37°C for 60 min. After incubation, the sealer was removed, and the plate was washed five times using 300 µl of wash buffer per well, with each wash lasting 30 s to 1 min. Following the washes, 50 µl of substrate solution A and 50 µl of substrate solution B were sequentially added to each well. The plate was then incubated in the dark for 10 min at 37°C. After incubation, 50 µl of stop solution was added to each well, resulting in an immediate color change from blue to yellow. The optical density (OD) of each well was measured immediately using a Bio-Rad microplate ELISA reader (Model PR4100) set to a wavelength of 450 nm [20].
2.7.2 Superoxide dismutase (SOD) assay
SOD was measured using the ELISA Kit from Bioassay Technology Laboratory (Cat. No. E4502Hu), and the same procedure was adopted, as described for the GSH assay.
2.8 Animals used
Male Sprague–Dawley rats weighing 150–200 g were purchased from the University of Veterinary and Animal Sciences (Lahore, Pakistan). Animals were kept under standard conditions (temperature: 22 ± 2°C and humidity: 60 ± 10%) with a 12 h light and dark cycle at the Animal House of the Faculty of Pharmacy (The University of Lahore). All the animals were given free access to food and water throughout the adaptation and experimental period. This study was performed in accordance with ARRIVE guidelines.
-
Ethical approval: The research related to animal use has complied with all the relevant national regulations and institutional policies for the care and use of animals and has been approved by the Institutional Research Ethics Committee (IREC) of the Department of Pharmacology, Faculty of Pharmacy, The University of Lahore, Lahore, Pakistan (Approval Number: IREC-2022-46).
2.9 LF experimental design
Animals were randomly divided into five groups (n = 4 in each group): control, disease (CCl4), standard (SIL), and treatment (p-CYM) groups. The control group received twice a week intraperitoneal (i.p.) injection of olive oil (0.5 ml/kg). LF was induced using a single dose of DEN, followed by increasing doses of CCl4 for 60 days (D). Briefly, a day after DEN administration, CCl4 (0.5 ml/kg) was injected intraperitoneally (i.p.) twice a week for 40D. On D41, animals were treated twice a week with 1 ml/kg of CCl4 for 18 D, i.e., D41–D58, which was later increased to 2 ml/kg for the last 2 days, i.e., D59–D60. To investigate the protective effects of p-CYM and SIL, animals were treated with SIL (100 mg/kg) and p-CYM (50 and 100 mg/kg) twice a week from D21 to D60. On D60, the body weights of animals were measured, and later, they were sacrificed by intraperitoneal injection of pentobarbital sodium (200 mg/kg) to collect blood and liver samples for subsequent biochemical, histopathological, and RT-qPCR analyses. Care was taken to minimize the suffering of animals.
2.10 Biochemical and histopathological analyses
Serum aspartate transferase (AST), alanine aminotransferase (ALT), alkaline phosphatase (ALP), and bilirubin levels were measured using standard ELISA kits. Liver samples fixed in 10% buffered formalin were sectioned using the paraffin embedding technique and stained with hematoxylin and eosin for histopathological analysis.
2.11 Real-time PCR analysis
Total RNA was isolated from liver samples using the Trizol method, which was later reverse-transcribed using the WizScript cDNA Synthesis Kit (Wizbio solutions, New Mexico, USA; Cat. No. W2202). The relative transcript levels of genes were measured by the ΔΔC T method using Zokeyo 2xSYBR Green qPCR mixture (Cat. No. HPR012-01). The following PCR conditions were used to measure the CT values: initial denaturation was carried out at 94°C for 2 min, followed by 40 cycles of denaturation at 94°C for 1 min, annealing at 60°C for 30 s, and elongation at 72°C for 15 s. Hypoxanthine guanine phosphoribosyltransferase (HPRT) was used as an internal standard. A list of primers (Macrogen, South Korea) used in the study is provided in Table S1.
2.12 Molecular docking analysis
2.12.1 Retrieval of tumor necrosis factor-alpha (TNF-α) and matrix metalloproteinase-1 (MMP-1) structures from Protein Data Bank
The three-dimensional (3D) structures of human TNF-α and MMP-1 were obtained from the Protein Data Bank (PDB) using PDB IDs 2AZ5 and 4AUO, respectively. The target proteins were prepared for docking analysis using the Autodock Tool program. Proteins were reduced in energy, given Gasteiger charges, and saved in a pdbqt format. Discovery Studio 4.1 Client (2012) was used to generate Ramachandran plots. VADAR 1.8 was used to access the protein architecture and statistical percentages of helices, β-sheets, coils, and turns [21].
2.12.2 Ligand molecular docking
P-CYM was drawn in Discovery Studio Client and saved in a pdb format as a ligand. The most stable conformation of the ligand was prepared using Autodock Tools. The Kolman and Gasteiger charges were added before the ligand was saved in a pdbqt format. The synthetic ligand (p-CYM) was subjected to a molecular docking experiment using PyRx’s virtual screening tool and the Auto Dock VINA Wizard method [22].
2.13 Statistical analysis
Data of 3–4 biological replicates were presented as the mean ± SD and were analyzed by one-way ANOVA followed by Tukey’s multiple comparison test. Statistical analyses were performed using Graph Pad Prism 8.0 software (Graphpad Software, Inc., San Diego, USA). A probability of less than 0.05 was considered significant. The level of significance was expressed as *** ≤ 0.001, ** ≤ 0.01, * ≤ 0.05.
3 Results
3.1 P-CYM treatment did not alter the viability of HepG2 cells
The cytotoxicity of p-CYM in HepG2 cells was assessed using the MTT assay to determine its safe and tolerable concentrations. HepG2 cells were treated with increasing concentrations of p-CYM (10, 50, 100, 500, and 1,000 µM) for 24 h to evaluate its impact on cell viability. The assay results demonstrated that p-CYM did not significantly affect cell viability at lower concentrations, specifically 10, 50, 100, and 500 µM, indicating that these doses are non-toxic to the cells and can be considered safe for further experimental use. However, at a concentration of 1,000 µM, p-CYM exhibited cytotoxic effects, as evidenced by a significant reduction in cell viability. These findings suggest that while p-CYM is generally well tolerated at moderate doses, high concentrations may compromise cellular health, emphasizing the importance of dose optimization for its potential therapeutic applications (Figure S1).
3.2 Cytotoxic effects of EtOH in HepG2 cells
To determine the toxic effects of EtOH on HepG2 cells, the cells were exposed to increasing concentrations of EtOH (ranging from 1 to 10%) for a duration of 24 h. Cell viability was then assessed to evaluate the extent of toxicity. At lower concentrations, specifically between 1 and 4%, EtOH did not induce any significant reduction in cell viability. This suggests that these concentrations are relatively safe and do not cause notable cellular damage. However, as the concentration of EtOH increased, a gradual decrease in cell viability was observed. At 6% EtOH, the reduction in cell viability was minimal, but it became more pronounced at 8%. The most substantial decline in cell viability occurred at the highest concentration tested, 10% EtOH, where cell viability dropped by over 50%. This indicates that EtOH has a dose-dependent toxic effect on HepG2 cells, with significant cellular damage occurring at concentrations of 8% and 10%. These findings suggest that high concentrations of EtOH can induce severe cytotoxicity, potentially through mechanisms like oxidative stress or disruption of cellular functions (Figure S2).
3.3 P-CYM protected against EtOH-induced toxicity in HepG2 cells
The cytoprotective effects of p-CYM against EtOH-induced cell injury were investigated by treating HepG2 cells with various concentrations of p-CYM (10, 50, 100, and 500 µM) prior to EtOH exposure. EtOH exposure significantly reduced cell viability by approximately 50% compared to the p-CYM- and SIL-treated groups, demonstrating the toxic impact of EtOH on liver cells. However, pretreatment with p-CYM at concentrations ranging from 10 to 500 µM resulted in a dose-dependent attenuation of EtOH-induced cytotoxicity. The protective effect of p-CYM was most prominent at 500 µM, where it demonstrated cytoprotective effects comparable to those of SIL, a well-known hepatoprotective agent (Figure 1).

Cytoprotective effects of p-CYM against EtOH-induced damage in HepG2 cells. (a) The absorbance of MTT dye was measured. (b) Percentage cell viability was measured from the absorbance values. Pre-treatment of p-CYM (50, 100, and 500 µM) significantly reduced EtOH toxicity compared to the disease control group. *** ≤ 0.001, ** ≤ 0.01, * ≤ 0.05 (treated groups vs disease group); one-way ANOVA followed by Tukey’s multiple comparison test; n = 3.
To further assess the impact of p-CYM on cell viability, a crystal violet assay was performed, showing a reduction in cell viability of around 40% upon exposure to EtOH, which aligns with the initial findings. However, p-CYM treatment was able to effectively restore cell viability in a significant manner, suggesting its potential to counteract EtOH-induced damage. Interestingly, the cellular growth observed in the p-CYM-treated group was even more pronounced than in the SIL-treated group, indicating that p-CYM might have a stronger or more favorable effect on promoting cell recovery and proliferation after ethanol-induced injury. These results highlight the strong protective and restorative capabilities of p-CYM, positioning it as a promising agent for preventing or mitigating liver damage caused by EtOH (Figure S3).
3.4 P-CYM prevented EtOH-induced cell death
The Trypan blue assay was used to assess cell death in HepG2 cells following exposure to EtOH. The results showed that EtOH induced cell death in more than 50% of the HepG2 cells, confirming its toxic effects on the cells. However, pretreatment with p-CYM demonstrated a dose-dependent reduction in cell death compared to the disease control group. At concentrations of 500 µM, its cytoprotective effects were even more pronounced than those observed in the SIL-treated group. This indicates that p-CYM not only reduced cell death but also showed superior efficacy in protecting HepG2 cells from EtOH-induced toxicity, highlighting its potential as a more effective therapeutic agent compared to SIL (Figure 2).

P-CYM reduced EtOH-induced cell death. Trypan blue staining showed an increased cell death with EtOH treatment. P-CYM and SIL treatments significantly reduced EtOH-induced cell death in HepG2 cells. *** ≤ 0.001 (treated groups vs disease group); one-way ANOVA followed by Tukey’s multiple comparison test; n = 3.
3.5 P-CYM attenuated EtOH-induced oxidative stress and transcript levels of inflammatory and fibrotic modulators
SOD and GSH activity increased in p-CYM- and SIL-treated groups compared to the disease group showing that p-CYM reduced oxidative stress induced by EtOH. Moreover, higher doses of p-CYM displayed more potent effects compared to SIL (Figure 3). The relative mRNA expression of biomarkers, including TNF-α, transforming growth factor-beta1 (TGF-β1), interleukin-6 (IL-6), glutathione peroxidase-7 (GPX-7), collagen type 1 (COL1A1), MMP-1, and tissue inhibitor of MMP-1 (TIMP-1) were assessed to examine the molecular mechanism behind the hepatoprotective activity of p-CYM. The findings of this study showed that p-CYM significantly reduced the expression rate of the abovementioned biomarkers. These findings were equivalent to the standard drug “SIL.” In contrast to the treated groups, the expression rate of these biomarkers was higher in the disease group, which could be ascribed to toxicity induced by EtOH. The hepatoprotective role of p-CYM can be ascribed to the down-regulation of inflammatory and fibrotic markers (Figure 4).

P-CYM prevented EtOH-induced oxidative stress by inducing anti-oxidants. P-CYM and SIL induced the levels of SOD (a) and GSH (b). *** ≤ 0.001 (treated groups vs disease group); one-way ANOVA followed by Tukey’s multiple comparison test; n = 3.

P-CYM displayed hepatoprotective effects by reducing transcript levels of TNF-α, TGF-β1, IL-6, GPX-7, COL1A1, MMP-1, and TIMP-1. *** ≤ 0.001 (treated groups vs disease group); one-way ANOVA followed by Tukey’s multiple comparison test; n = 3.
3.6 P-CYM prevented CCl4-induced weight reduction and protected against CCl4-induced liver damage
One of the characteristics of chronic liver illness is weight loss, which may be caused by the liver’s metabolic dysfunction and a decrease in bile production, which in turn leads to a reduction in lipid emulsification and absorption [2,23]. The present findings also revealed that CCl4 reduced the body weight of rats, which was restored by treatment with SIL and p-CYM. Interestingly, the highest dose of p-CYM restored the body weight to normal levels, and this effect was much more prominent than in the SIL-treated group (Figure S4). Moreover, increased LFT levels reflect a variety of aberrant liver activities, including (a) hepatocellular instability, (b) decreased bile synthesis, and (c) altered protein synthesis, and are therefore indirect indicators of LF [2,23,24]. In current study, the exposure to CCl4 also led to increased levels of ALP, AST, ALT, and bilirubin, signaling liver damage. However, SIL and p-CYM treatment reduced these elevated markers, indicating their hepatoprotective effects (Figure S5).
3.7 Histopathological and real-time PCR analyses revealed anti-fibrotic effects of p-CYM
The liver tissue samples of the disease group showed fibrotic scarring enriched with collagen and swollen hepatocytes. Treatment with SIL showed mild infiltration of inflammatory cells, while p-CYM (50 mg/kg)-treated tissue appeared normal with mild swelling of hepatocytes. At a higher dose (100 mg/kg), p-CYM did not display any inflammation or scarring, and the tissue also had a normal appearance. The above findings clearly indicated the protective effects of p-CYM against DEN–CCl4-induced LF (Figure 5, Table S2). RT-qPCR findings also demonstrated that CCl4 induced transcript levels of pro-fibrotic markers (TIMP-1, IL-1β, COL1A1, and TGF-β1) and reduced anti-fibrotic markers (MMP-1). Treatment with SIL and p-CYM restored these markers, indicating their anti-fibrotic effects (Figure 6).

Histopathology of liver samples displayed anti-fibrotic effects of p-CYM.
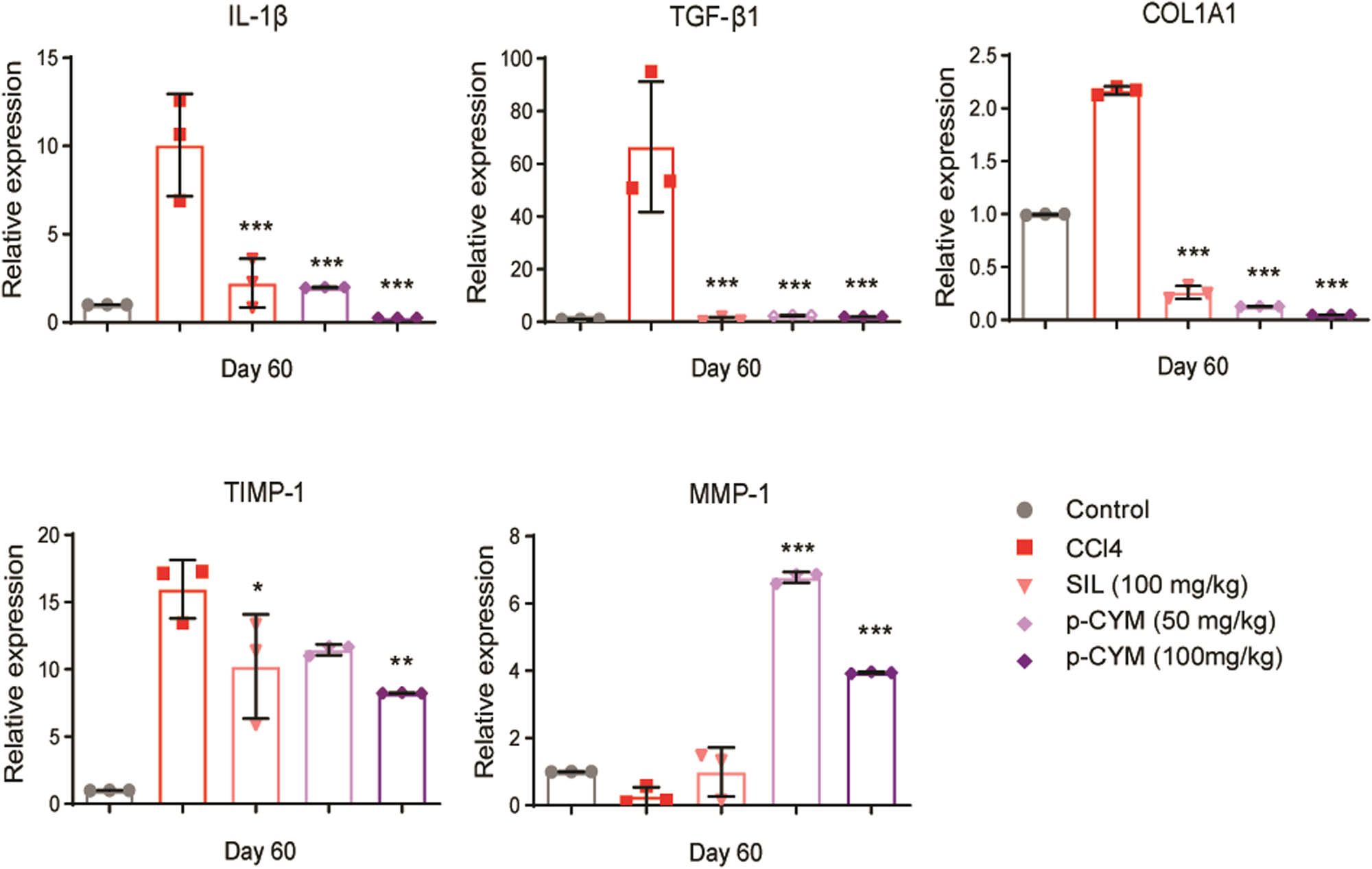
Modulation of pro- and anti-fibrotic markers by p-CYM. P-CYM and SIL reduced the transcript levels of TIMP-1, IL-1β, COL1A1, and TGF-β1 and induced MMP1, indicating its anti-fibrotic effects. *** ≤ 0.001, ** ≤ 0.01, and * ≤ 0.05 (treated groups vs disease group); one-way ANOVA followed by Tukey’s multiple comparison test; n = 3.
3.8 P-CYM displayed strong binding affinity with TNF-α and modest affinity with MMP-1
The affinity between the ligands and protein targets was examined by molecular docking. The docking analysis was conducted using the AutoDock Vina [22] tool and PyRx [25] user interface. The best-docked posture complex and protein’s affinity were evaluated using the E-value (kcal/mol). It offered a prediction of the binding constant and free energy for docked ligands. Usually, if the binding energy is more negative, stronger is the interaction between ligand and target protein. P-CYM’s docking tests displayed strong binding interactions with TNF-α and MMP-1, and the results were in line with the pharmacological effects. The binding energies of p-CYM with TNF-α and MMP-1 were −6.1 and −5.4 kcal/mol, respectively. These findings indicate that p-CYM-induced anti-fibrotic effects could be due to direct interaction with TNF-α and MMP-1 (Figure 7).

Binding interactions of p-CYM with TNF-α and MMP-1. 2D and 3D representation of binding interactions of p-CYM with the amino acid residues of the binding site of MMP-1 (a) and (b) and TNF-α (c) and (d).
4 Discussion
LF is a critical stage in the progression of chronic liver disease, occurring after the initial development of fatty liver disease. If left untreated, LF leads to significant liver damage, where the liver tissue undergoes structural changes, resulting in shrinkage and the formation of nodules, a condition known as cirrhosis. The progression from LF to cirrhosis and potentially to hepatocellular carcinoma (HCC) underscores the importance of targeting LF as a key therapeutic intervention to prevent further liver damage and mitigate the risk of life-threatening complications [26]. Despite its critical role in the progression of liver disease, LF remains a major clinical challenge due to the lack of effective treatments that can fully reverse or significantly alleviate fibrosis once it has developed. Currently, available therapies are limited in their ability to halt or reverse the fibrotic process, and no universally accepted treatment can cure or effectively manage LF on a long-term basis. This highlights the urgent need for the development of new, more effective therapies that can address LF at its core, prevent progression to cirrhosis, and reduce the risk of developing HCC.
In this study, we elucidated the cytoprotective ability of p-CYM against EtOH-induced injury in HepG2 cells and its anti-fibrotic potential against DEN–CCl4-induced LF in rats. Chronic consumption of EtOH accelerates liver damage, contributing to a range of liver diseases, such as cirrhosis, HCC, alcoholic hepatitis, and alcoholic steatosis [27,28]. High levels of EtOH intake induce oxidative stress and fat accumulation in hepatocytes, leading to liver injury. Reactive oxygen species (ROS) play a central role in oxidative stress-induced cell death [29,30]. Studies have shown that excessive ROS production in the liver causes abnormal protein expression, oxidative DNA damage, and disruption of cell membranes, worsening liver function [31,32]. To prevent the progression of alcoholic liver disease (ALD), reducing EtOH-induced oxidative stress and fat buildup in the liver could be beneficial [33]. In addition to EtOH, DEN and CCl4 are commonly used in animal models to induce LF. In both wild-type and transgenic animal models, administration of DEN and CCl4 for 3–4 weeks causes centrilobular and periportal LF. CCl4 specifically activates macrophages and hepatic stellate cells (HSCs), triggering the upregulation of pro-fibrotic mediators and increasing ROS generation. This leads to fibrogenesis and cirrhosis, primarily driven by the overproduction of TGF-β [26]. The key features of DEN–CCl4-induced LF include weight loss, increased LFTs, and fibrotic changes in the liver [23,24]. These findings highlight the importance of targeting oxidative stress and fibrotic pathways to prevent or treat ALD and associated liver damage.
Moreover, studies have shown that anti-oxidants alter the redox state of the cell and reduce the generation of free radicals. Some of these agents include vitamins E and C, N-acetylcysteine (NAC), mitoquinone, and polyenylphosphatidylcholine. Vitamin E stabilizes the free radical compounds by forming complexes with the unpaired electrons and prevents the activation of HSCs [34]. In a small open-label study, vitamin E treatment (1200 IU/day) for 8 weeks stopped the fibrogenesis cascade in six patients who were refractory to interferon. This was demonstrated by decreased levels of malonaldehyde and reduced activation of HSCs [34]. Similarly, a moderate reduction in serum ALT to 63 IU/l from baseline levels of 73 IU/l was observed in 17 patients who received vitamin E treatment (500 mg/day) for three months [35]. Previous studies report that humans experience severe functional and structural alterations after ingesting EtOH, which disrupts metabolism. GSH and SOD depletion occurs in case of EtOH exposure owing to their rapid utilization by ROS [36,37].
In our study, several assays also suggested that pre-treatment of HepG2 cells with p-CYM significantly reduced EtOH-induced oxidative stress and cell death. The present findings indicated that pre-treatment with p-CYM prevented the decrease in GSH and SOD levels in HepG2 cells. The restoration of the intracellular GSH and SOD levels in HepG2 cells pre-exposed with p-CYM indicated that increased amounts of EtOH-induced ROS could be scavenged, which subsequently resulted in reduced cellular damage. Furthermore, the pathological changes associated with DEN–CCl4-induced LF were significantly reduced by p-CYM, illustrating its beneficial effects in the resolution of LF.
In order to elucidate the possible molecular mechanism behind the hepatoprotective effects of p-CYM, we assessed the transcription levels of several pro- and anti-fibrotic biomarkers. Studies have revealed that pro-inflammatory mediators are crucial to the process of fibrogenesis, and those who suffer from chronic liver conditions have elevated IL-1β levels in their serum [38,39]. IL-1β is currently regarded as a key regulator of tissue injury and inflammation in chronic liver disorders because of its critical role in the transformation of steatosis into steatohepatitis and LF [40,41,42]. Our tested compound, p-CYM, showed potential anti-fibrotic properties by significantly reducing mRNA expression of IL-1β in rats.
Other cytokine family members that promote both acute and chronic inflammation in the liver include TNF-α and IL-6 [43,44,45]. The IKK and JNK pathways are activated by TNF receptor interactions by bringing in the adaptor molecules [46]. IKK phosphorylates IκB and p65, resulting in NF-κB activation [47]. Increased JNK activity tips the scales in favor of cell death by causing the E3 ligase to be phosphorylated, followed by the ubiquitination and degradation of the NF-κB-regulated caspase-8 inhibitor “c-Flip.” Prolonged activation of JNK necessitates TNF-α-induced ROS generation and moves the balance toward cell death [48]. Moreover, liver inflammation caused by EtOH is believed to be exacerbated by the pro-inflammatory cytokine IL-6, which plays a critical role in the progression of liver damage [49,50,51]. Elevated levels of IL-6 have been linked to the activation of several downstream signaling pathways, including the Janus kinase (JAK)/signal transducer and activator of the transcription 3 (STAT3) pathway. This pathway is a key mediator of LF and contributes to the chronic inflammatory environment associated with ALD. The activation of JAK/STAT3 by IL-6 leads to the transcription of target genes involved in fibrosis, inflammation, and cell survival, promoting the persistence of HSC activation and ECM deposition. This cascade not only worsens liver inflammation but also accelerates the progression of fibrosis, thereby playing a pivotal role in the pathogenesis of EtOH-induced liver injury [52]. Our study also revealed an increased expression rate of these biomarkers in EtOH-intoxicated HepG2 cells, and p-CYM effectively reduced the transcript levels of TNF-α and IL-6, reiterating its protective mechanism.
TGF-β1 is another significant regulator of liver cell growth and plays a role in the progression of chronic liver damage [53]. Numerous studies have shown that EtOH-induced inflammation results in the production of TGF-β1, which is thought to be crucial for the pathophysiology and development of ALD [54]. One of the primary proteins that promotes fibrogenesis is TGF-β1, which stimulates HSC and causes them to activate and produce ECM proteins [55]. Previous studies have indicated that LF is associated with high levels of TGF-β1 and COL1A1 expression, and the collagen deposition increases as the fibrosis progresses [56,57,58,59]. In the present study, we also witnessed increased levels of TGF-β1 and COL1A1 in EtOH- and DEN–CCl4-induced hepatic damage, while p-CYM significantly reduced these biomarkers, illustrating its anti-fibrotic effect.
TIMP and MMP proteins normally exist in equilibrium in healthy tissue; however, due to chronic liver damage, TIMP-1 levels increase than MMP-1 levels, which results in inhibition of ECM breakdown [60,61]. Advanced stages of LF include almost six times the normal amount of ECM, which includes proteoglycans, fibronectin, elastin, laminin, hyaluronan, and collagens I, III, and IV. Both increased synthesis and decreased degradation lead to the accumulation of ECM proteins [62]. The primary cause of the decreased activity of ECM-removing MMPs is the overexpression of their particular inhibitors (TIMPs). The breakdown of ECM proteins and programmed cell death of HSCs are the mechanisms underlying the MMP-derived inhibition of the fibrogenic response [63,64]. In this study, we also witnessed a reduction in TIMP-1 and induction in MMP-1 levels upon treatment with p-CYM, indicating that p-CYM has the ability to reduce fibrogenic response by promoting ECM degradation.
Overall, this study highlights the therapeutic potential of p-CYM against EtOH- and CCl₄-induced hepatotoxicity, suggesting its viability as a protective agent against liver damage. Despite its promise, one key limitation of p-CYM, like many natural compounds, may be its relatively low bioavailability, which can restrict its therapeutic efficacy. To address this challenge, future research could focus on developing a nanoformulation of p-CYM.
Nanoformulations have emerged as a cutting-edge approach in drug delivery, particularly for compounds that face challenges related to solubility, stability, or targeted delivery. By incorporating p-CYM into nanocarriers, its bioavailability can be significantly improved, ensuring more effective systemic circulation and cellular uptake. Moreover, nanomaterials offer several distinct advantages that make them highly suitable for this purpose. These include their large drug-loading capacity, which allows for the encapsulation of significant amounts of therapeutic agents, and their surface modification capabilities, enabling the development of targeted delivery systems. Such targeted systems could direct p-CYM specifically to the liver, minimizing off-target effects and enhancing therapeutic precision. Additionally, nanocarriers can offer controlled release profiles, ensuring sustained therapeutic levels of the drug over time [65,66,67].
Given the substantial progress in the field of nanotechnology, this approach could transform p-CYM from a promising natural compound into a highly effective therapeutic agent. Future investigations could explore various nanocarrier systems, such as liposomes, polymeric nanoparticles, or lipid-based nanocarriers, to identify the most suitable platform for p-CYM delivery. Collectively, these advancements could pave the way for the clinical translation of p-CYM as a novel hepatoprotective therapy.
5 Conclusion
Based on in vitro findings, it can be concluded from this study that EtOH intoxication in the HepG2 cell line induces oxidative stress, inflammation, and collagen synthesis, as evident by reduced anti-oxidants (GSH and SOD) and increased levels of inflammatory (IL-1β and TNF-α) and pro-fibrotic (TGF-β1 and COL1A1) biomarkers. P-CYM protected HepG2 cells from EtOH-induced cell death owing to its anti-oxidant and anti-inflammatory properties. Moreover, p-CYM also reduced the levels of pro-fibrotic mediators under in vitro settings. Similarly, under in vivo conditions, DEN- CCl4 induced oxidative stress and fibrosis, while treatment with p-CYM effectively reversed pro-fibrotic effects of DEN–CCl4. In a nutshell, it is conceivable from this study that the hepatoprotective effects of p-CYM could be attributed to its anti-oxidant, anti-inflammatory, and ECM modulatory activities.
Acknowledgments
The authors extend their appreciation to researchers supporting the project number (RSPD2025R885) at King Saud University, Riyadh, Saudi Arabia, for supporting this research. We would like to thank Dr. Ahsan Sattar (Institute of Molecular Biology, The University of Lahore) for providing us with the platform for cell culture studies.
-
Funding information: The authors extend their appreciation to researchers supporting the project number (RSPD2025R885) at King Saud University, Riyadh, Saudi Arabia, for supporting this research.
-
Author contributions: M.A. performed all the cell culture experiments, performed data analyses, and wrote the first version of the manuscript. M.N.H.M. designed and supervised the project, arranged all the resources, analyzed and validated the experimental data, and edited the final version of the manuscript. T.G.A. performed docking studies, validated all the data, and edited the final version of the manuscript. M.A. performed in vivo experiments, data analyses, and edited the final version of the manuscript. H.M.B. performed in vivo experiments and validated all data. W.Y., K.S.A., and S.I.A. performed ELISA and histopathological assays. B.A. and I.A. performed histopathological and statistical analyses. S.J. and T.M. performed qPCR and data analyses. G.F.B.S. analyzed and validated all data and edited the final version of the manuscript. All authors participated in editing the final version of the manuscript.
-
Conflict of interest: Authors state no conflict of interest.
-
Data availability statement: The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
[1] Kisseleva T, Brenner D. Molecular and cellular mechanisms of liver fibrosis and its regression. Nat Rev Gastroenterol Hepatol. 2021;18(3):151–66.10.1038/s41575-020-00372-7Search in Google Scholar PubMed
[2] Benitez R, Caro M, Andres‐Leon E, O’Valle F, Delgado M. Cortistatin regulates fibrosis and myofibroblast activation in experimental hepatotoxic‐and cholestatic‐induced liver injury. Br J Pharmacol. 2022;179(10):2275–96.10.1111/bph.15752Search in Google Scholar PubMed
[3] Yoo ER, Cholankeril G, Ahmed A. Treating alcohol use disorder in chronic liver disease. Clin Liver Dis (Hoboken). 2020;15(2):77–80.10.1002/cld.881Search in Google Scholar PubMed PubMed Central
[4] Stashin AR, Fikse DJ, Orta AM, Briggs 3rd RP, Wheatley SM, Koons AL. You dropped the bomb on me: A case series of carbon tetrachloride toxicity. Cureus. 2023;15(4):e37879.10.7759/cureus.37879Search in Google Scholar PubMed PubMed Central
[5] Dey D, Chaskar S, Bhatt N, Chitre D. Hepatoprotective activity of BV-7310, a Proprietary herbal formulation of Phyllanthus niruri, Tephrosia purpurea, Boerhavia diffusa, and Andrographis paniculata, in alcohol-induced HepG2 cells and alcohol plus a haloalkane, CCl(4), induced liver damage in rats. Evid Based Complement Alternat Med. 2020;2020:6428906.10.1155/2020/6428906Search in Google Scholar PubMed PubMed Central
[6] Manzo-Avalos S, Saavedra-Molina A. Cellular and mitochondrial effects of alcohol - consumption. Int J Environ Res Public Health. 2010;7(12):4281–304.10.3390/ijerph7124281Search in Google Scholar PubMed PubMed Central
[7] Huang W, Booth DM, Cane MC, Chvanov M, Javed MA, Elliott VL, et al. Fatty acid ethyl ester synthase inhibition ameliorates ethanol-induced Ca2+ -dependent mitochondrial dysfunction and acute pancreatitis. Gut. 2014;63(8):1313–24.10.1136/gutjnl-2012-304058Search in Google Scholar PubMed PubMed Central
[8] Lee KJ, Choi JH, Jeong HG. Hepatoprotective and antioxidant effects of the coffee diterpenes kahweol and cafestol on carbon tetrachloride-induced liver damage in mice. Food Chem Toxicol. 2007;45(11):2118–25.10.1016/j.fct.2007.05.010Search in Google Scholar PubMed
[9] Tilg H, Day CP. Management strategies in alcoholic liver disease. Nat Clin Pract Gastroenterol Hepatol. 2007;4(1):24–34.10.1038/ncpgasthep0683Search in Google Scholar PubMed
[10] Muriel P, Rivera-Espinoza Y. Beneficial drugs for liver diseases. J Appl Toxicol. 2008;28(2):93–103.10.1002/jat.1310Search in Google Scholar PubMed
[11] Stickel F, Schuppan D. Herbal medicine in the treatment of liver diseases. Dig Liver Dis. 2007;39(4):293–304.10.1016/j.dld.2006.11.004Search in Google Scholar PubMed
[12] Osna NA, Donohue Jr TM, Kharbanda KK. Alcoholic liver disease: Pathogenesis and current management. Alcohol Res. 2017;38(2):147–61.10.35946/arcr.v38.2.01Search in Google Scholar
[13] Abe R, Ohtani K. An ethnobotanical study of medicinal plants and traditional therapies on Batan Island, the Philippines. J Ethnopharmacol. 2013;145(2):554–65.10.1016/j.jep.2012.11.029Search in Google Scholar PubMed
[14] Atanasov AG, Waltenberger B, Pferschy-Wenzig EM, Linder T, Wawrosch C, Uhrin P, et al. Discovery and resupply of pharmacologically active plant-derived natural products: A review. Biotechnol Adv. 2015;33(8):1582–614.10.1016/j.biotechadv.2015.08.001Search in Google Scholar PubMed PubMed Central
[15] Balahbib A, El Omari N, Hachlafi NE, Lakhdar F, El Menyiy N, Salhi N, et al. Health beneficial and pharmacological properties of p-cymene. Food Chem Toxicol. 2021;153:112259.10.1016/j.fct.2021.112259Search in Google Scholar PubMed
[16] Shareef SH, Al-Medhtiy MH, Ibrahim IAA, Alzahrani AR, Jabbar AA, Galali Y, et al. Gastroprophylactic effects of p-cymene in ethanol-induced gastric ulcer in rats. Processes. 2022;10(7):1314.10.3390/pr10071314Search in Google Scholar
[17] Wang S, Wang X, Wang YU, Leng Q, Sun YU, Hoffman RM, et al. The anti-oxidant monoterpene p-cymene reduced the occurrence of colorectal cancer in a hyperlipidemia rat model by reducing oxidative stress and expression of inflammatory cytokines. Anticancer Res. 2021;41(3):1213–8.10.21873/anticanres.14878Search in Google Scholar PubMed
[18] Maqbool T, Awan SJ, Malik S, Hadi F, Shehzadi S, Tariq K. In-vitro anti-proliferative, apoptotic and antioxidative activities of medicinal herb Kalonji (Nigella sativa). Curr Pharm Biotechnol. 2019;20(15):1288–308.10.2174/1389201020666190821144633Search in Google Scholar PubMed
[19] Lee JY, Kim H, Jeong Y, Kang CH. Lactic acid bacteria exert a hepatoprotective effect against ethanol-induced liver injury in HepG2 cells. Microorganisms. 2021;9(9):1844.10.3390/microorganisms9091844Search in Google Scholar PubMed PubMed Central
[20] Pouresmaeil V, Al Abudi AH, Mahimid AH, Sarafraz Yazdi M, Es-Haghi A. Evaluation of serum selenium and copper levels with inflammatory cytokines and indices of oxidative stress in type 2 diabetes. Biol Trace Elem Res. 2023;201(2):617–26.10.1007/s12011-022-03191-wSearch in Google Scholar PubMed
[21] Willard L, Ranjan A, Zhang H, Monzavi H, Boyko RF, Sykes BD, et al. VADAR: A web server for quantitative evaluation of protein structure quality. Nucleic Acids Res. 2003;31(13):3316–9.10.1093/nar/gkg565Search in Google Scholar PubMed PubMed Central
[22] Trott O, Olson AJ. AutoDock Vina: Improving the speed and accuracy of docking with a new scoring function, efficient optimization, and multithreading. J Comput Chem. 2010;31(2):455–61.10.1002/jcc.21334Search in Google Scholar PubMed PubMed Central
[23] Brattin WJ, Glende Jr EA, Recknagel RO. Pathological mechanisms in carbon tetrachloride hepatotoxicity. J Free Radic Biol Med. 1985;1(1):27–38.10.1016/0748-5514(85)90026-1Search in Google Scholar PubMed
[24] Nadkarni GD, D’Souza NB. Hepatic antioxidant enzymes and lipid peroxidation in carbon tetrachloride-induced liver cirrhosis in rats. Biochem Med Metab Biol. 1988;40(1):42–5.10.1016/0885-4505(88)90102-8Search in Google Scholar PubMed
[25] Dallakyan S, Olson AJ. Small-molecule library screening by docking with PyRx. In: Hempel JE. Chemical biology: Methods and protocols. New York, USA: Springer; 2015. pp. 243–50.10.1007/978-1-4939-2269-7_19Search in Google Scholar PubMed
[26] Gao HY, Li GY, Lou MM, Li XY, Wei XY, Wang JH. Hepatoprotective effect of matrine salvianolic acid B salt on carbon tetrachloride-induced hepatic fibrosis. J Inflamm (Lond). 2012;9(1):16.10.1186/1476-9255-9-16Search in Google Scholar PubMed PubMed Central
[27] Gao B, Bataller R. Alcoholic liver disease: Pathogenesis and new therapeutic targets. Gastroenterology. 2011;141(5):1572–85.10.1053/j.gastro.2011.09.002Search in Google Scholar PubMed PubMed Central
[28] Orman ES, Odena G, Bataller R. Alcoholic liver disease: Pathogenesis, management, and novel targets for therapy. J Gastroenterol Hepatol. 2013;28 Suppl 1(S1):77–84.10.1111/jgh.12030Search in Google Scholar PubMed PubMed Central
[29] Chen WM, Shaw LH, Chang PJ, Tung SY, Chang TS, Shen CH, et al. Hepatoprotective effect of resveratrol against ethanol-induced oxidative stress through induction of superoxide dismutase in vivo and in vitro. Exp Ther Med. 2016;11(4):1231–8.10.3892/etm.2016.3077Search in Google Scholar PubMed PubMed Central
[30] Kannan K, Jain SK. Oxidative stress and apoptosis. Pathophysiology. 2000;7(3):153–63.10.1016/S0928-4680(00)00053-5Search in Google Scholar PubMed
[31] Chen Z, Tian R, She Z, Cai J, Li H. Role of oxidative stress in the pathogenesis of nonalcoholic fatty liver disease. Free Radic Biol Med. 2020;152:116–41.10.1016/j.freeradbiomed.2020.02.025Search in Google Scholar PubMed
[32] Machida K, Cheng KT, Lai CK, Jeng KS, Sung VM, Lai MM. Hepatitis C virus triggers mitochondrial permeability transition with production of reactive oxygen species, leading to DNA damage and STAT3 activation. J Virol. 2006;80(14):7199–207.10.1128/JVI.00321-06Search in Google Scholar PubMed PubMed Central
[33] Nagappan A, Jung DY, Kim J-H, Lee H, Jung MH. Gomisin N alleviates ethanol-induced liver injury through ameliorating lipid metabolism and oxidative stress. Int J Mol Sci. 2018;19(9):2601.10.3390/ijms19092601Search in Google Scholar PubMed PubMed Central
[34] Houglum K, Venkataramani A, Lyche K, Chojkier M. A pilot study of the effects of d-alpha-tocopherol on hepatic stellate cell activation in chronic hepatitis C. Gastroenterology. 1997;113(4):1069–73.10.1053/gast.1997.v113.pm9322499Search in Google Scholar PubMed
[35] Mahmood S, Yamada G, Niiyama G, Kawanaka M, Togawa K, Sho M, et al. Effect of vitamin E on serum aminotransferase and thioredoxin levels in patients with viral hepatitis C. Free Radic Res. 2003;37(7):781–5.10.1080/1071576031000102141Search in Google Scholar PubMed
[36] Bae D, You Y, Yoon H-G, Kim K, Lee Y-H, Kim Y, et al. Protective effects of loquat (Eriobotrya japonica) leaves against ethanol-induced toxicity in HepG2 cells transfected with CYP2E1. Food Sci Biotechnol. 2010;19:1093–6.10.1007/s10068-010-0154-3Search in Google Scholar
[37] Hartley‐Whitaker J, Ainsworth G, Meharg AA. Copper‐and arsenate‐induced oxidative stress in Holcus lanatus L. clones with differential sensitivity. Plant Cell Eniron. 2001;24(7):713–22.10.1046/j.0016-8025.2001.00721.xSearch in Google Scholar
[38] Ludwiczek O, Vannier E, Moschen A, Salazar-Montes A, Borggraefe I, Gabay C, et al. Impaired counter-regulation of interleukin-1 by the soluble IL-1 receptor type II in patients with chronic liver disease. Scand J Gastroenterol. 2008;43(11):1360–5.10.1080/00365520802179925Search in Google Scholar PubMed
[39] Tilg H, Vogel W, Wiedermann CJ, Shapiro L, Herold M, Judmaier G, et al. Circulating interleukin-1 and tumor necrosis factor antagonists in liver disease. Hepatology. 1993;18(5):1132–8.10.1002/hep.1840180519Search in Google Scholar
[40] Kamari Y, Shaish A, Vax E, Shemesh S, Kandel-Kfir M, Arbel Y, et al. Lack of interleukin-1alpha or interleukin-1beta inhibits transformation of steatosis to steatohepatitis and liver fibrosis in hypercholesterolemic mice. J Hepatol. 2011;55(5):1086–94.10.1016/j.jhep.2011.01.048Search in Google Scholar PubMed PubMed Central
[41] Tilg H, Moschen AR, Szabo G. Interleukin-1 and inflammasomes in alcoholic liver disease/acute alcoholic hepatitis and nonalcoholic fatty liver disease/nonalcoholic steatohepatitis. Hepatology. 2016;64(3):955–65.10.1002/hep.28456Search in Google Scholar PubMed
[42] Gieling RG, Wallace K, Han YP. Interleukin-1 participates in the progression from liver injury to fibrosis. Am J Physiol Gastrointest Liver Physiol. 2009;296(6):G1324–31.10.1152/ajpgi.90564.2008Search in Google Scholar PubMed PubMed Central
[43] Shaikh PZ. Cytokines & their physiologic and pharmacologic functions in inflammation: A review. Int J Pharm Life Sci. 2011;2(11):1247–63.Search in Google Scholar
[44] Feghali CA, Wright TM. Cytokines in acute and chronic inflammation. Front Biosci. 1997;2(4):d12–26.10.2741/A171Search in Google Scholar PubMed
[45] Holtmann MH, Neurath MF. Differential TNF-signaling in chronic inflammatory disorders. Curr Mol Med. 2004;4(4):439–44.10.2174/1566524043360636Search in Google Scholar PubMed
[46] Baud V, Karin M. Signal transduction by tumor necrosis factor and its relatives. Trends Cell Biol. 2001;11(9):372–7.10.1016/S0962-8924(01)02064-5Search in Google Scholar
[47] Schwabe RF, Sakurai H. IKKβ phosphorylates p65 at S468 in transactivaton domain 2. FASEB J. 2005;19(12):1758–60.10.1096/fj.05-3736fjeSearch in Google Scholar PubMed
[48] Kamata H, Honda S, Maeda S, Chang L, Hirata H, Karin M. Reactive oxygen species promote TNFalpha-induced death and sustained JNK activation by inhibiting MAP kinase phosphatases. Cell. 2005;120(5):649–61.10.1016/j.cell.2004.12.041Search in Google Scholar PubMed
[49] GaoB. Cytokines, STATs and liver disease. Cell Mol Immunol. 2005;2(2):92–100.Search in Google Scholar
[50] Mitra SK, Varma SR, Godavarthi A, Nandakumar KS. Liv.52 regulates ethanol induced PPARgamma and TNF alpha expression in HepG2 cells. Mol Cell Biochem. 2008;315(1–2):9–15.10.1007/s11010-008-9782-9Search in Google Scholar PubMed
[51] Kawaratani H, Tsujimoto T, Douhara A, Takaya H, Moriya K, Namisaki T, et al. The effect of inflammatory cytokines in alcoholic liver disease. Mediators Inflamm. 2013;2013:495156.10.1155/2013/495156Search in Google Scholar PubMed PubMed Central
[52] Li Y, Zhao J, Yin Y, Li K, Zhang C, Zheng Y. The role of IL-6 in fibrotic Diseases: Molecular and cellular mechanisms. Int J Biol Sci. 2022;18(14):5405–14.10.7150/ijbs.75876Search in Google Scholar PubMed PubMed Central
[53] Dooley S, ten Dijke P. TGF-beta in progression of liver disease. Cell Tissue Res. 2012;347(1):245–56.10.1007/s00441-011-1246-ySearch in Google Scholar PubMed PubMed Central
[54] Song Z, Deaciuc I, Song M, Lee DY, Liu Y, Ji X, et al. Silymarin protects against acute ethanol-induced hepatotoxicity in mice. Alcohol Clin Exp Res. 2006;30(3):407–13.10.1111/j.1530-0277.2006.00063.xSearch in Google Scholar PubMed PubMed Central
[55] Dewidar B, Soukupova J, Fabregat I, Dooley S. TGF-β in hepatic stellate cell activation and liver fibrogenesis: Updated. Curr Pathobiol Rep. 2015;3(4):291–305.10.1007/s40139-015-0089-8Search in Google Scholar
[56] Badylak SF, Freytes DO, Gilbert TW. Extracellular matrix as a biological scaffold material: Structure and function. Acta Biomater. 2009;5(1):1–13.10.1016/j.actbio.2008.09.013Search in Google Scholar PubMed
[57] Karsdal MA, Nielsen SH, Leeming D, Langholm L, Nielsen M, Manon-Jensen T, et al. The good and the bad collagens of fibrosis–their role in signaling and organ function. Adv Drug Deliv Rev. 2017;121:43–56.10.1016/j.addr.2017.07.014Search in Google Scholar PubMed
[58] Liu X, Hu H, Yin JQ. Therapeutic strategies against TGF-beta signaling pathway in hepatic fibrosis. Liver Int. 2006;26(1):8–22.10.1111/j.1478-3231.2005.01192.xSearch in Google Scholar PubMed
[59] Liu M, Xu Y, Han X, Yin L, Xu L, Qi Y, et al. Dioscin alleviates alcoholic liver fibrosis by attenuating hepatic stellate cell activation via the TLR4/MyD88/NF-kappaB signaling pathway. Sci Rep. 2015;5(1):18038.10.1038/srep18038Search in Google Scholar PubMed PubMed Central
[60] Hemmann S, Graf J, Roderfeld M, Roeb E. Expression of MMPs and TIMPs in liver fibrosis - a systematic review with special emphasis on anti-fibrotic strategies. J Hepatol. 2007;46(5):955–75.10.1016/j.jhep.2007.02.003Search in Google Scholar PubMed
[61] Roeb E, Purucker E, Breuer B, Nguyen H, Heinrich PC, Rose-John S, et al. TIMP expression in toxic and cholestatic liver injury in rat. J Hepatol. 1997;27(3):535–44.10.1016/S0168-8278(97)80359-5Search in Google Scholar
[62] Robert S, Gicquel T, Bodin A, Lagente V, Boichot E. Characterization of the MMP/TIMP imbalance and collagen production induced by IL-1beta or TNF-alpha release from human hepatic stellate cells. PLoS One. 2016;11(4):e0153118.10.1371/journal.pone.0153118Search in Google Scholar PubMed PubMed Central
[63] Murphy FR, Issa R, Zhou X, Ratnarajah S, Arthur MJ, Benyon C, et al. Inhibition of apoptosis of activated hepatic stellate cells by tissue inhibitor of metalloproteinase-1 is mediated via effects on matrix metalloproteinase inhibition: Implications for reversibility of liver fibrosis. J Biol Chem. 2002;277(13):11069–76.10.1074/jbc.M111490200Search in Google Scholar PubMed
[64] Iimuro Y, Brenner DA. Matrix metalloproteinase gene delivery for liver fibrosis. Pharm Res. 2008;25(2):249–58.10.1007/s11095-007-9311-7Search in Google Scholar PubMed PubMed Central
[65] Zhu X, Li S. Nanomaterials in tumor immunotherapy: New strategies and challenges. Mol Cancer. 2023;22(1):94.10.1186/s12943-023-01797-9Search in Google Scholar PubMed PubMed Central
[66] Kang Y, Li S. Nanomaterials: Breaking through the bottleneck of tumor immunotherapy. Int J Biol Macromol. 2023;230:123159.10.1016/j.ijbiomac.2023.123159Search in Google Scholar PubMed
[67] Chen Z, Yue Z, Yang K, Li S. Nanomaterials: Small particles show huge possibilities for cancer immunotherapy. J Nanobiotechnology. 2022;20(1):484.10.1186/s12951-022-01692-3Search in Google Scholar PubMed PubMed Central
© 2025 the author(s), published by De Gruyter
This work is licensed under the Creative Commons Attribution 4.0 International License.
Articles in the same Issue
- Safety assessment and modulation of hepatic CYP3A4 and UGT enzymes by Glycyrrhiza glabra aqueous extract in female Sprague–Dawley rats
- Adult-onset Still’s disease with hemophagocytic lymphohistiocytosis and minimal change disease
- Role of DZ2002 in reducing corneal graft rejection in rats by influencing Th17 activation via inhibition of the PI3K/AKT pathway and downregulation of TRAF1
- Biomedical Sciences
- Mechanism of triptolide regulating proliferation and apoptosis of hepatoma cells by inhibiting JAK/STAT pathway
- Maslinic acid improves mitochondrial function and inhibits oxidative stress and autophagy in human gastric smooth muscle cells
- Comparative analysis of inflammatory biomarkers for the diagnosis of neonatal sepsis: IL-6, IL-8, SAA, CRP, and PCT
- Post-pandemic insights on COVID-19 and premature ovarian insufficiency
- Proteome differences of dental stem cells between permanent and deciduous teeth by data-independent acquisition proteomics
- Optimizing a modified cetyltrimethylammonium bromide protocol for fungal DNA extraction: Insights from multilocus gene amplification
- Preliminary analysis of the role of small hepatitis B surface proteins mutations in the pathogenesis of occult hepatitis B infection via the endoplasmic reticulum stress-induced UPR-ERAD pathway
- Efficacy of alginate-coated gold nanoparticles against antibiotics-resistant Staphylococcus and Streptococcus pathogens of acne origins
- Battling COVID-19 leveraging nanobiotechnology: Gold and silver nanoparticle–B-escin conjugates as SARS-CoV-2 inhibitors
- Neurodegenerative diseases and neuroinflammation-induced apoptosis
- Impact of fracture fixation surgery on cognitive function and the gut microbiota in mice with a history of stroke
- COLEC10: A potential tumor suppressor and prognostic biomarker in hepatocellular carcinoma through modulation of EMT and PI3K-AKT pathways
- High-temperature requirement serine protease A2 inhibitor UCF-101 ameliorates damaged neurons in traumatic brain-injured rats by the AMPK/NF-κB pathway
- SIK1 inhibits IL-1β-stimulated cartilage apoptosis and inflammation in vitro through the CRTC2/CREB1 signaling
- Rutin–chitooligosaccharide complex: Comprehensive evaluation of its anti-inflammatory and analgesic properties in vitro and in vivo
- Knockdown of Aurora kinase B alleviates high glucose-triggered trophoblast cells damage and inflammation during gestational diabetes
- Calcium-sensing receptors promoted Homer1 expression and osteogenic differentiation in bone marrow mesenchymal stem cells
- ABI3BP can inhibit the proliferation, invasion, and epithelial–mesenchymal transition of non-small-cell lung cancer cells
- Changes in blood glucose and metabolism in hyperuricemia mice
- Rapid detection of the GJB2 c.235delC mutation based on CRISPR-Cas13a combined with lateral flow dipstick
- IL-11 promotes Ang II-induced autophagy inhibition and mitochondrial dysfunction in atrial fibroblasts
- Short-chain fatty acid attenuates intestinal inflammation by regulation of gut microbial composition in antibiotic-associated diarrhea
- Application of metagenomic next-generation sequencing in the diagnosis of pathogens in patients with diabetes complicated by community-acquired pneumonia
- NAT10 promotes radiotherapy resistance in non-small cell lung cancer by regulating KPNB1-mediated PD-L1 nuclear translocation
- Phytol-mixed micelles alleviate dexamethasone-induced osteoporosis in zebrafish: Activation of the MMP3–OPN–MAPK pathway-mediating bone remodeling
- Association between TGF-β1 and β-catenin expression in the vaginal wall of patients with pelvic organ prolapse
- Primary pleomorphic liposarcoma involving bilateral ovaries: Case report and literature review
- Effects of de novo donor-specific Class I and II antibodies on graft outcomes after liver transplantation: A pilot cohort study
- Sleep architecture in Alzheimer’s disease continuum: The deep sleep question
- Ephedra fragilis plant extract: A groundbreaking corrosion inhibitor for mild steel in acidic environments – electrochemical, EDX, DFT, and Monte Carlo studies
- Langerhans cell histiocytosis in an adult patient with upper jaw and pulmonary involvement: A case report
- Inhibition of mast cell activation by Jaranol-targeted Pirin ameliorates allergic responses in mouse allergic rhinitis
- Aeromonas veronii-induced septic arthritis of the hip in a child with acute lymphoblastic leukemia
- Clusterin activates the heat shock response via the PI3K/Akt pathway to protect cardiomyocytes from high-temperature-induced apoptosis
- Research progress on fecal microbiota transplantation in tumor prevention and treatment
- Low-pressure exposure influences the development of HAPE
- Stigmasterol alleviates endplate chondrocyte degeneration through inducing mitophagy by enhancing PINK1 mRNA acetylation via the ESR1/NAT10 axis
- AKAP12, mediated by transcription factor 21, inhibits cell proliferation, metastasis, and glycolysis in lung squamous cell carcinoma
- Association between PAX9 or MSX1 gene polymorphism and tooth agenesis risk: A meta-analysis
- A case of bloodstream infection caused by Neisseria gonorrhoeae
- Case of nasopharyngeal tuberculosis complicated with cervical lymph node and pulmonary tuberculosis
- p-Cymene inhibits pro-fibrotic and inflammatory mediators to prevent hepatic dysfunction
- GFPT2 promotes paclitaxel resistance in epithelial ovarian cancer cells via activating NF-κB signaling pathway
- Transfer RNA-derived fragment tRF-36 modulates varicose vein progression via human vascular smooth muscle cell Notch signaling
- RTA-408 attenuates the hepatic ischemia reperfusion injury in mice possibly by activating the Nrf2/HO-1 signaling pathway
- Decreased serum TIMP4 levels in patients with rheumatoid arthritis
- Sirt1 protects lupus nephritis by inhibiting the NLRP3 signaling pathway in human glomerular mesangial cells
- Sodium butyrate aids brain injury repair in neonatal rats
- Interaction of MTHFR polymorphism with PAX1 methylation in cervical cancer
- Convallatoxin inhibits proliferation and angiogenesis of glioma cells via regulating JAK/STAT3 pathway
- The effect of the PKR inhibitor, 2-aminopurine, on the replication of influenza A virus, and segment 8 mRNA splicing
- Effects of Ire1 gene on virulence and pathogenicity of Candida albicans
- Small cell lung cancer with small intestinal metastasis: Case report and literature review
- GRB14: A prognostic biomarker driving tumor progression in gastric cancer through the PI3K/AKT signaling pathway by interacting with COBLL1
- 15-Lipoxygenase-2 deficiency induces foam cell formation that can be restored by salidroside through the inhibition of arachidonic acid effects
- FTO alleviated the diabetic nephropathy progression by regulating the N6-methyladenosine levels of DACT1
- Clinical relevance of inflammatory markers in the evaluation of severity of ulcerative colitis: A retrospective study
- Zinc valproic acid complex promotes osteoblast differentiation and exhibits anti-osteoporotic potential
- Primary pulmonary synovial sarcoma in the bronchial cavity: A case report
- Metagenomic next-generation sequencing of alveolar lavage fluid improves the detection of pulmonary infection
- Uterine tumor resembling ovarian sex cord tumor with extensive rhabdoid differentiation: A case report
- Genomic analysis of a novel ST11(PR34365) Clostridioides difficile strain isolated from the human fecal of a CDI patient in Guizhou, China
- Effects of tiered cardiac rehabilitation on CRP, TNF-α, and physical endurance in older adults with coronary heart disease
- Changes in T-lymphocyte subpopulations in patients with colorectal cancer before and after acupoint catgut embedding acupuncture observation
- Modulating the tumor microenvironment: The role of traditional Chinese medicine in improving lung cancer treatment
- Alterations of metabolites related to microbiota–gut–brain axis in plasma of colon cancer, esophageal cancer, stomach cancer, and lung cancer patients
- Research on individualized drug sensitivity detection technology based on bio-3D printing technology for precision treatment of gastrointestinal stromal tumors
- CEBPB promotes ulcerative colitis-associated colorectal cancer by stimulating tumor growth and activating the NF-κB/STAT3 signaling pathway
- Oncolytic bacteria: A revolutionary approach to cancer therapy
- A de novo meningioma with rapid growth: A possible malignancy imposter?
- Diagnosis of secondary tuberculosis infection in an asymptomatic elderly with cancer using next-generation sequencing: Case report
- Hesperidin and its zinc(ii) complex enhance osteoblast differentiation and bone formation: In vitro and in vivo evaluations
- Research progress on the regulation of autophagy in cardiovascular diseases by chemokines
- Anti-arthritic, immunomodulatory, and inflammatory regulation by the benzimidazole derivative BMZ-AD: Insights from an FCA-induced rat model
- Immunoassay for pyruvate kinase M1/2 as an Alzheimer’s biomarker in CSF
- The role of HDAC11 in age-related hearing loss: Mechanisms and therapeutic implications
- Evaluation and application analysis of animal models of PIPNP based on data mining
- Therapeutic approaches for liver fibrosis/cirrhosis by targeting pyroptosis
- Fabrication of zinc oxide nanoparticles using Ruellia tuberosa leaf extract induces apoptosis through P53 and STAT3 signalling pathways in prostate cancer cells
- Haplo-hematopoietic stem cell transplantation and immunoradiotherapy for severe aplastic anemia complicated with nasopharyngeal carcinoma: A case report
- Modulation of the KEAP1-NRF2 pathway by Erianin: A novel approach to reduce psoriasiform inflammation and inflammatory signaling
- The expression of epidermal growth factor receptor 2 and its relationship with tumor-infiltrating lymphocytes and clinical pathological features in breast cancer patients
- Innovations in MALDI-TOF Mass Spectrometry: Bridging modern diagnostics and historical insights
- BAP1 complexes with YY1 and RBBP7 and its downstream targets in ccRCC cells
- Hypereosinophilic syndrome with elevated IgG4 and T-cell clonality: A report of two cases
- Electroacupuncture alleviates sciatic nerve injury in sciatica rats by regulating BDNF and NGF levels, myelin sheath degradation, and autophagy
- Polydatin prevents cholesterol gallstone formation by regulating cholesterol metabolism via PPAR-γ signaling
- RNF144A and RNF144B: Important molecules for health
- Analysis of the detection rate and related factors of thyroid nodules in the healthy population
- Artesunate inhibits hepatocellular carcinoma cell migration and invasion through OGA-mediated O-GlcNAcylation of ZEB1
- Endovascular management of post-pancreatectomy hemorrhage caused by a hepatic artery pseudoaneurysm: Case report and review of the literature
- Efficacy and safety of anti-PD-1/PD-L1 antibodies in patients with relapsed refractory diffuse large B-cell lymphoma: A meta-analysis
- SATB2 promotes humeral fracture healing in rats by activating the PI3K/AKT pathway
- Overexpression of the ferroptosis-related gene, NFS1, corresponds to gastric cancer growth and tumor immune infiltration
- Understanding risk factors and prognosis in diabetic foot ulcers
- Atractylenolide I alleviates the experimental allergic response in mice by suppressing TLR4/NF-kB/NLRP3 signalling
- FBXO31 inhibits the stemness characteristics of CD147 (+) melanoma stem cells
- Immune molecule diagnostics in colorectal cancer: CCL2 and CXCL11
- Inhibiting CXCR6 promotes senescence of activated hepatic stellate cells with limited proinflammatory SASP to attenuate hepatic fibrosis
- Cadmium toxicity, health risk and its remediation using low-cost biochar adsorbents
- Pulmonary cryptococcosis with headache as the first presentation: A case report
- Solitary pulmonary metastasis with cystic airspaces in colon cancer: A rare case report
- RUNX1 promotes denervation-induced muscle atrophy by activating the JUNB/NF-κB pathway and driving M1 macrophage polarization
- Morphometric analysis and immunobiological investigation of Indigofera oblongifolia on the infected lung with Plasmodium chabaudi
- The NuA4/TIP60 histone-modifying complex and Hr78 modulate the Lobe2 mutant eye phenotype
- Experimental study on salmon demineralized bone matrix loaded with recombinant human bone morphogenetic protein-2: In vitro and in vivo study
- A case of IgA nephropathy treated with a combination of telitacicept and half-dose glucocorticoids
- Analgesic and toxicological evaluation of cannabidiol-rich Moroccan Cannabis sativa L. (Khardala variety) extract: Evidence from an in vivo and in silico study
- Wound healing and signaling pathways
- Combination of immunotherapy and whole-brain radiotherapy on prognosis of patients with multiple brain metastases: A retrospective cohort study
- To explore the relationship between endometrial hyperemia and polycystic ovary syndrome
- Research progress on the impact of curcumin on immune responses in breast cancer
- Biogenic Cu/Ni nanotherapeutics from Descurainia sophia (L.) Webb ex Prantl seeds for the treatment of lung cancer
- Dapagliflozin attenuates atrial fibrosis via the HMGB1/RAGE pathway in atrial fibrillation rats
- Glycitein alleviates inflammation and apoptosis in keratinocytes via ROS-associated PI3K–Akt signalling pathway
- ADH5 inhibits proliferation but promotes EMT in non-small cell lung cancer cell through activating Smad2/Smad3
- Apoptotic efficacies of AgNPs formulated by Syzygium aromaticum leaf extract on 32D-FLT3-ITD human leukemia cell line with PI3K/AKT/mTOR signaling pathway
- Novel cuproptosis-related genes C1QBP and PFKP identified as prognostic and therapeutic targets in lung adenocarcinoma
- Bee venom promotes exosome secretion and alters miRNA cargo in T cells
- Treatment of pure red cell aplasia in a chronic kidney disease patient with roxadustat: A case report
- Comparative bioinformatics analysis of the Wnt pathway in breast cancer: Selection of novel biomarker panels associated with ER status
- Kynurenine facilitates renal cell carcinoma progression by suppressing M2 macrophage pyroptosis through inhibition of CASP1 cleavage
- RFX5 promotes the growth, motility, and inhibits apoptosis of gastric adenocarcinoma cells through the SIRT1/AMPK axis
- ALKBH5 exacerbates early cardiac damage after radiotherapy for breast cancer via m6A demethylation of TLR4
- Phytochemicals of Roman chamomile: Antioxidant, anti-aging, and whitening activities of distillation residues
- Circadian gene Cry1 inhibits the tumorigenicity of hepatocellular carcinoma by the BAX/BCL2-mediated apoptosis pathway
- The TNFR-RIPK1/RIPK3 signalling pathway mediates the effect of lanthanum on necroptosis of nerve cells
- Longitudinal monitoring of autoantibody dynamics in patients with early-stage non-small-cell lung cancer undergoing surgery
- The potential role of rutin, a flavonoid, in the management of cancer through modulation of cell signaling pathways
- Construction of pectinase gene engineering microbe and its application in tobacco sheets
- Construction of a microbial abundance prognostic scoring model based on intratumoral microbial data for predicting the prognosis of lung squamous cell carcinoma
- Sepsis complicated by haemophagocytic lymphohistiocytosis triggered by methicillin-resistant Staphylococcus aureus and human herpesvirus 8 in an immunocompromised elderly patient: A case report
- Sarcopenia in liver transplantation: A comprehensive bibliometric study of current research trends and future directions
- Advances in cancer immunotherapy and future directions in personalized medicine
- Can coronavirus disease 2019 affect male fertility or cause spontaneous abortion? A two-sample Mendelian randomization analysis
- Heat stroke associated with novel leukaemia inhibitory factor receptor gene variant in a Chinese infant
- PSME2 exacerbates ulcerative colitis by disrupting intestinal barrier function and promoting autophagy-dependent inflammation
- Hyperosmolar hyperglycemic state with severe hypernatremia coexisting with central diabetes insipidus: A case report and literature review
- Efficacy and mechanism of escin in improving the tissue microenvironment of blood vessel walls via anti-inflammatory and anticoagulant effects: Implications for clinical practice
- Merkel cell carcinoma: Clinicopathological analysis of three patients and literature review
- Genetic variants in VWF exon 26 and their implications for type 1 Von Willebrand disease in a Saudi Arabian population
- Lipoxin A4 improves myocardial ischemia/reperfusion injury through the Notch1-Nrf2 signaling pathway
- High levels of EPHB2 expression predict a poor prognosis and promote tumor progression in endometrial cancer
- Knockdown of SHP-2 delays renal tubular epithelial cell injury in diabetic nephropathy by inhibiting NLRP3 inflammasome-mediated pyroptosis
- Exploring the toxicity mechanisms and detoxification methods of Rhizoma Paridis
- Concomitant gastric carcinoma and primary hepatic angiosarcoma in a patient: A case report
- YAP1 inhibition protects retinal vascular endothelial cells under high glucose by inhibiting autophagy
- Identification of secretory protein related biomarkers for primary biliary cholangitis based on machine learning and experimental validation
- Integrated genomic and clinical modeling for prognostic assessment of radiotherapy response in rectal neoplasms
- Stem cell-based approaches for glaucoma treatment: a mini review
- Bacteriophage titering by optical density means: KOTE assays
- Neutrophil-related signature characterizes immune landscape and predicts prognosis of esophageal squamous cell carcinoma
- Integrated bioinformatic analysis and machine learning strategies to identify new potential immune biomarkers for Alzheimer’s disease and their targeting prediction with geniposide
- TRIM21 accelerates ferroptosis in intervertebral disc degeneration by promoting SLC7A11 ubiquitination and degradation
- TRIM21 accelerates ferroptosis in intervertebral disc degeneration by promoting SLC7A11 ubiquitination and degradation
- Histone modification and non-coding RNAs in skin aging: emerging therapeutic avenues
- A multiplicative behavioral model of DNA replication initiation in cells
- Biogenic gold nanoparticles synthesized from Pergularia daemia leaves: a novel approach for nasopharyngeal carcinoma therapy
- Creutzfeldt-Jakob disease mimicking Hashimoto’s encephalopathy: steroid response followed by decline
- Impact of semaphorin, Sema3F, on the gene transcription and protein expression of CREB and its binding protein CREBBP in primary hippocampal neurons of rats
- Iron overloaded M0 macrophages regulate hematopoietic stem cell proliferation and senescence via the Nrf2/Keap1/HO-1 pathway
- Revisiting the link between NADPH oxidase p22phox C242T polymorphism and ischemic stroke risk: an updated meta-analysis
- Exercise training preferentially modulates α1D-adrenergic receptor expression in peripheral arteries of hypertensive rats
- Overexpression of HE4/WFDC2 gene in mice leads to keratitis and corneal opacity
- Tumoral calcinosis complicating CKD-MBD in hemodialysis: a case report
- Mechanism of KLF4 Inhibition of epithelial-mesenchymal transition in gastric cancer cells
- Dissecting the molecular mechanisms of T cell infiltration in psoriatic lesions via cell-cell communication and regulatory network analysis
- Circadian rhythm-based prognostic features predict immune infiltration and tumor microenvironment in molecular subtypes of hepatocellular carcinoma
- Ecology and Environmental Science
- Optimization and comparative study of Bacillus consortia for cellulolytic potential and cellulase enzyme activity
- The complete mitochondrial genome analysis of Haemaphysalis hystricis Supino, 1897 (Ixodida: Ixodidae) and its phylogenetic implications
- Epidemiological characteristics and risk factors analysis of multidrug-resistant tuberculosis among tuberculosis population in Huzhou City, Eastern China
- Indices of human impacts on landscapes: How do they reflect the proportions of natural habitats?
- Genetic analysis of the Siberian flying squirrel population in the northern Changbai Mountains, Northeast China: Insights into population status and conservation
- Diversity and environmental drivers of Suillus communities in Pinus sylvestris var. mongolica forests of Inner Mongolia
- Global assessment of the fate of nitrogen deposition in forest ecosystems: Insights from 15N tracer studies
- Fungal and bacterial pathogenic co-infections mainly lead to the assembly of microbial community in tobacco stems
- Influencing of coal industry related airborne particulate matter on ocular surface tear film injury and inflammatory factor expression in Sprague-Dawley rats
- Temperature-dependent development, predation, and life table of Sphaerophoria macrogaster (Thomson) (Diptera: Syrphidae) feeding on Myzus persicae (Sulzer) (Homoptera: Aphididae)
- Eleonora’s falcon trophic interactions with insects within its breeding range: A systematic review
- Agriculture
- Integrated analysis of transcriptome, sRNAome, and degradome involved in the drought-response of maize Zhengdan958
- Variation in flower frost tolerance among seven apple cultivars and transcriptome response patterns in two contrastingly frost-tolerant selected cultivars
- Heritability of durable resistance to stripe rust in bread wheat (Triticum aestivum L.)
- Molecular mechanism of follicular development in laying hens based on the regulation of water metabolism
- Molecular identification and control studies on Coridius sp. (Hemiptera: Dinidoridae) in Al-Khamra, south of Jeddah, Saudi Arabia
- 10.1515/biol-2025-1218
- Animal Science
- Effect of sex ratio on the life history traits of an important invasive species, Spodoptera frugiperda
- Plant Sciences
- Hairpin in a haystack: In silico identification and characterization of plant-conserved microRNA in Rafflesiaceae
- Widely targeted metabolomics of different tissues in Rubus corchorifolius
- The complete chloroplast genome of Gerbera piloselloides (L.) Cass., 1820 (Carduoideae, Asteraceae) and its phylogenetic analysis
- Field trial to correlate mineral solubilization activity of Pseudomonas aeruginosa and biochemical content of groundnut plants
- Correlation analysis between semen routine parameters and sperm DNA fragmentation index in patients with semen non-liquefaction: A retrospective study
- Plasticity of the anatomical traits of Rhododendron L. (Ericaceae) leaves and its implications in adaptation to the plateau environment
- Effects of Piriformospora indica and arbuscular mycorrhizal fungus on growth and physiology of Moringa oleifera under low-temperature stress
- Effects of different sources of potassium fertiliser on yield, fruit quality and nutrient absorption in “Harward” kiwifruit (Actinidia deliciosa)
- Comparative efficiency and residue levels of spraying programs against powdery mildew in grape varieties
- The DREB7 transcription factor enhances salt tolerance in soybean plants under salt stress
- Using plant electrical signals of water hyacinth (Eichhornia crassipes) for water pollution monitoring
- Response of hybrid grapes (Vitis spp.) to two biotic stress factors and their seedlessness status
- Metabolomic profiling reveals systemic metabolic reprogramming in Alternaria alternata under salt stress
- Effects of mixed salinity and alkali stress on photosynthetic characteristics and PEPC gene expression of vegetable soybean seedlings
- Food Science
- Phytochemical analysis of Stachys iva: Discovering the optimal extract conditions and its bioactive compounds
- Review on role of honey in disease prevention and treatment through modulation of biological activities
- Computational analysis of polymorphic residues in maltose and maltotriose transporters of a wild Saccharomyces cerevisiae strain
- Optimization of phenolic compound extraction from Tunisian squash by-products: A sustainable approach for antioxidant and antibacterial applications
- Liupao tea aqueous extract alleviates dextran sulfate sodium-induced ulcerative colitis in rats by modulating the gut microbiota
- Toxicological qualities and detoxification trends of fruit by-products for valorization: A review
- Polyphenolic spectrum of cornelian cherry fruits and their health-promoting effect
- Optimizing the encapsulation of the refined extract of squash peels for functional food applications: A sustainable approach to reduce food waste
- Advancements in curcuminoid formulations: An update on bioavailability enhancement strategies curcuminoid bioavailability and formulations
- Impact of saline sprouting on antioxidant properties and bioactive compounds in chia seeds
- The dilemma of food genetics and improvement
- Causal effects of trace elements on congenital foot deformities and their subtypes: a Mendelian randomization study with gut microbiota mediation
- Honey meets acidity: a novel biopreservative approach against foodborne pathogens
- Bioengineering and Biotechnology
- Impact of hyaluronic acid-modified hafnium metalorganic frameworks containing rhynchophylline on Alzheimer’s disease
- Emerging patterns in nanoparticle-based therapeutic approaches for rheumatoid arthritis: A comprehensive bibliometric and visual analysis spanning two decades
- Application of CRISPR/Cas gene editing for infectious disease control in poultry
- Preparation of hafnium nitride-coated titanium implants by magnetron sputtering technology and evaluation of their antibacterial properties and biocompatibility
- Preparation and characterization of lemongrass oil nanoemulsion: Antimicrobial, antibiofilm, antioxidant, and anticancer activities
- Fluorescent detection of sialic acid–binding lectins using functionalized quantum dots in ELISA format
- Smart tectorigenin-loaded ZnO hydrogel nanocomposites for targeted wound healing: synthesis, characterization, and biological evaluation
- Corrigendum
- Corrigendum to “Utilization of convolutional neural networks to analyze microscopic images for high-throughput screening of mesenchymal stem cells”
- Corrigendum to “Effects of Ire1 gene on virulence and pathogenicity of Candida albicans”
- Retraction
- Retraction of “Down-regulation of miR-539 indicates poor prognosis in patients with pancreatic cancer”
Articles in the same Issue
- Safety assessment and modulation of hepatic CYP3A4 and UGT enzymes by Glycyrrhiza glabra aqueous extract in female Sprague–Dawley rats
- Adult-onset Still’s disease with hemophagocytic lymphohistiocytosis and minimal change disease
- Role of DZ2002 in reducing corneal graft rejection in rats by influencing Th17 activation via inhibition of the PI3K/AKT pathway and downregulation of TRAF1
- Biomedical Sciences
- Mechanism of triptolide regulating proliferation and apoptosis of hepatoma cells by inhibiting JAK/STAT pathway
- Maslinic acid improves mitochondrial function and inhibits oxidative stress and autophagy in human gastric smooth muscle cells
- Comparative analysis of inflammatory biomarkers for the diagnosis of neonatal sepsis: IL-6, IL-8, SAA, CRP, and PCT
- Post-pandemic insights on COVID-19 and premature ovarian insufficiency
- Proteome differences of dental stem cells between permanent and deciduous teeth by data-independent acquisition proteomics
- Optimizing a modified cetyltrimethylammonium bromide protocol for fungal DNA extraction: Insights from multilocus gene amplification
- Preliminary analysis of the role of small hepatitis B surface proteins mutations in the pathogenesis of occult hepatitis B infection via the endoplasmic reticulum stress-induced UPR-ERAD pathway
- Efficacy of alginate-coated gold nanoparticles against antibiotics-resistant Staphylococcus and Streptococcus pathogens of acne origins
- Battling COVID-19 leveraging nanobiotechnology: Gold and silver nanoparticle–B-escin conjugates as SARS-CoV-2 inhibitors
- Neurodegenerative diseases and neuroinflammation-induced apoptosis
- Impact of fracture fixation surgery on cognitive function and the gut microbiota in mice with a history of stroke
- COLEC10: A potential tumor suppressor and prognostic biomarker in hepatocellular carcinoma through modulation of EMT and PI3K-AKT pathways
- High-temperature requirement serine protease A2 inhibitor UCF-101 ameliorates damaged neurons in traumatic brain-injured rats by the AMPK/NF-κB pathway
- SIK1 inhibits IL-1β-stimulated cartilage apoptosis and inflammation in vitro through the CRTC2/CREB1 signaling
- Rutin–chitooligosaccharide complex: Comprehensive evaluation of its anti-inflammatory and analgesic properties in vitro and in vivo
- Knockdown of Aurora kinase B alleviates high glucose-triggered trophoblast cells damage and inflammation during gestational diabetes
- Calcium-sensing receptors promoted Homer1 expression and osteogenic differentiation in bone marrow mesenchymal stem cells
- ABI3BP can inhibit the proliferation, invasion, and epithelial–mesenchymal transition of non-small-cell lung cancer cells
- Changes in blood glucose and metabolism in hyperuricemia mice
- Rapid detection of the GJB2 c.235delC mutation based on CRISPR-Cas13a combined with lateral flow dipstick
- IL-11 promotes Ang II-induced autophagy inhibition and mitochondrial dysfunction in atrial fibroblasts
- Short-chain fatty acid attenuates intestinal inflammation by regulation of gut microbial composition in antibiotic-associated diarrhea
- Application of metagenomic next-generation sequencing in the diagnosis of pathogens in patients with diabetes complicated by community-acquired pneumonia
- NAT10 promotes radiotherapy resistance in non-small cell lung cancer by regulating KPNB1-mediated PD-L1 nuclear translocation
- Phytol-mixed micelles alleviate dexamethasone-induced osteoporosis in zebrafish: Activation of the MMP3–OPN–MAPK pathway-mediating bone remodeling
- Association between TGF-β1 and β-catenin expression in the vaginal wall of patients with pelvic organ prolapse
- Primary pleomorphic liposarcoma involving bilateral ovaries: Case report and literature review
- Effects of de novo donor-specific Class I and II antibodies on graft outcomes after liver transplantation: A pilot cohort study
- Sleep architecture in Alzheimer’s disease continuum: The deep sleep question
- Ephedra fragilis plant extract: A groundbreaking corrosion inhibitor for mild steel in acidic environments – electrochemical, EDX, DFT, and Monte Carlo studies
- Langerhans cell histiocytosis in an adult patient with upper jaw and pulmonary involvement: A case report
- Inhibition of mast cell activation by Jaranol-targeted Pirin ameliorates allergic responses in mouse allergic rhinitis
- Aeromonas veronii-induced septic arthritis of the hip in a child with acute lymphoblastic leukemia
- Clusterin activates the heat shock response via the PI3K/Akt pathway to protect cardiomyocytes from high-temperature-induced apoptosis
- Research progress on fecal microbiota transplantation in tumor prevention and treatment
- Low-pressure exposure influences the development of HAPE
- Stigmasterol alleviates endplate chondrocyte degeneration through inducing mitophagy by enhancing PINK1 mRNA acetylation via the ESR1/NAT10 axis
- AKAP12, mediated by transcription factor 21, inhibits cell proliferation, metastasis, and glycolysis in lung squamous cell carcinoma
- Association between PAX9 or MSX1 gene polymorphism and tooth agenesis risk: A meta-analysis
- A case of bloodstream infection caused by Neisseria gonorrhoeae
- Case of nasopharyngeal tuberculosis complicated with cervical lymph node and pulmonary tuberculosis
- p-Cymene inhibits pro-fibrotic and inflammatory mediators to prevent hepatic dysfunction
- GFPT2 promotes paclitaxel resistance in epithelial ovarian cancer cells via activating NF-κB signaling pathway
- Transfer RNA-derived fragment tRF-36 modulates varicose vein progression via human vascular smooth muscle cell Notch signaling
- RTA-408 attenuates the hepatic ischemia reperfusion injury in mice possibly by activating the Nrf2/HO-1 signaling pathway
- Decreased serum TIMP4 levels in patients with rheumatoid arthritis
- Sirt1 protects lupus nephritis by inhibiting the NLRP3 signaling pathway in human glomerular mesangial cells
- Sodium butyrate aids brain injury repair in neonatal rats
- Interaction of MTHFR polymorphism with PAX1 methylation in cervical cancer
- Convallatoxin inhibits proliferation and angiogenesis of glioma cells via regulating JAK/STAT3 pathway
- The effect of the PKR inhibitor, 2-aminopurine, on the replication of influenza A virus, and segment 8 mRNA splicing
- Effects of Ire1 gene on virulence and pathogenicity of Candida albicans
- Small cell lung cancer with small intestinal metastasis: Case report and literature review
- GRB14: A prognostic biomarker driving tumor progression in gastric cancer through the PI3K/AKT signaling pathway by interacting with COBLL1
- 15-Lipoxygenase-2 deficiency induces foam cell formation that can be restored by salidroside through the inhibition of arachidonic acid effects
- FTO alleviated the diabetic nephropathy progression by regulating the N6-methyladenosine levels of DACT1
- Clinical relevance of inflammatory markers in the evaluation of severity of ulcerative colitis: A retrospective study
- Zinc valproic acid complex promotes osteoblast differentiation and exhibits anti-osteoporotic potential
- Primary pulmonary synovial sarcoma in the bronchial cavity: A case report
- Metagenomic next-generation sequencing of alveolar lavage fluid improves the detection of pulmonary infection
- Uterine tumor resembling ovarian sex cord tumor with extensive rhabdoid differentiation: A case report
- Genomic analysis of a novel ST11(PR34365) Clostridioides difficile strain isolated from the human fecal of a CDI patient in Guizhou, China
- Effects of tiered cardiac rehabilitation on CRP, TNF-α, and physical endurance in older adults with coronary heart disease
- Changes in T-lymphocyte subpopulations in patients with colorectal cancer before and after acupoint catgut embedding acupuncture observation
- Modulating the tumor microenvironment: The role of traditional Chinese medicine in improving lung cancer treatment
- Alterations of metabolites related to microbiota–gut–brain axis in plasma of colon cancer, esophageal cancer, stomach cancer, and lung cancer patients
- Research on individualized drug sensitivity detection technology based on bio-3D printing technology for precision treatment of gastrointestinal stromal tumors
- CEBPB promotes ulcerative colitis-associated colorectal cancer by stimulating tumor growth and activating the NF-κB/STAT3 signaling pathway
- Oncolytic bacteria: A revolutionary approach to cancer therapy
- A de novo meningioma with rapid growth: A possible malignancy imposter?
- Diagnosis of secondary tuberculosis infection in an asymptomatic elderly with cancer using next-generation sequencing: Case report
- Hesperidin and its zinc(ii) complex enhance osteoblast differentiation and bone formation: In vitro and in vivo evaluations
- Research progress on the regulation of autophagy in cardiovascular diseases by chemokines
- Anti-arthritic, immunomodulatory, and inflammatory regulation by the benzimidazole derivative BMZ-AD: Insights from an FCA-induced rat model
- Immunoassay for pyruvate kinase M1/2 as an Alzheimer’s biomarker in CSF
- The role of HDAC11 in age-related hearing loss: Mechanisms and therapeutic implications
- Evaluation and application analysis of animal models of PIPNP based on data mining
- Therapeutic approaches for liver fibrosis/cirrhosis by targeting pyroptosis
- Fabrication of zinc oxide nanoparticles using Ruellia tuberosa leaf extract induces apoptosis through P53 and STAT3 signalling pathways in prostate cancer cells
- Haplo-hematopoietic stem cell transplantation and immunoradiotherapy for severe aplastic anemia complicated with nasopharyngeal carcinoma: A case report
- Modulation of the KEAP1-NRF2 pathway by Erianin: A novel approach to reduce psoriasiform inflammation and inflammatory signaling
- The expression of epidermal growth factor receptor 2 and its relationship with tumor-infiltrating lymphocytes and clinical pathological features in breast cancer patients
- Innovations in MALDI-TOF Mass Spectrometry: Bridging modern diagnostics and historical insights
- BAP1 complexes with YY1 and RBBP7 and its downstream targets in ccRCC cells
- Hypereosinophilic syndrome with elevated IgG4 and T-cell clonality: A report of two cases
- Electroacupuncture alleviates sciatic nerve injury in sciatica rats by regulating BDNF and NGF levels, myelin sheath degradation, and autophagy
- Polydatin prevents cholesterol gallstone formation by regulating cholesterol metabolism via PPAR-γ signaling
- RNF144A and RNF144B: Important molecules for health
- Analysis of the detection rate and related factors of thyroid nodules in the healthy population
- Artesunate inhibits hepatocellular carcinoma cell migration and invasion through OGA-mediated O-GlcNAcylation of ZEB1
- Endovascular management of post-pancreatectomy hemorrhage caused by a hepatic artery pseudoaneurysm: Case report and review of the literature
- Efficacy and safety of anti-PD-1/PD-L1 antibodies in patients with relapsed refractory diffuse large B-cell lymphoma: A meta-analysis
- SATB2 promotes humeral fracture healing in rats by activating the PI3K/AKT pathway
- Overexpression of the ferroptosis-related gene, NFS1, corresponds to gastric cancer growth and tumor immune infiltration
- Understanding risk factors and prognosis in diabetic foot ulcers
- Atractylenolide I alleviates the experimental allergic response in mice by suppressing TLR4/NF-kB/NLRP3 signalling
- FBXO31 inhibits the stemness characteristics of CD147 (+) melanoma stem cells
- Immune molecule diagnostics in colorectal cancer: CCL2 and CXCL11
- Inhibiting CXCR6 promotes senescence of activated hepatic stellate cells with limited proinflammatory SASP to attenuate hepatic fibrosis
- Cadmium toxicity, health risk and its remediation using low-cost biochar adsorbents
- Pulmonary cryptococcosis with headache as the first presentation: A case report
- Solitary pulmonary metastasis with cystic airspaces in colon cancer: A rare case report
- RUNX1 promotes denervation-induced muscle atrophy by activating the JUNB/NF-κB pathway and driving M1 macrophage polarization
- Morphometric analysis and immunobiological investigation of Indigofera oblongifolia on the infected lung with Plasmodium chabaudi
- The NuA4/TIP60 histone-modifying complex and Hr78 modulate the Lobe2 mutant eye phenotype
- Experimental study on salmon demineralized bone matrix loaded with recombinant human bone morphogenetic protein-2: In vitro and in vivo study
- A case of IgA nephropathy treated with a combination of telitacicept and half-dose glucocorticoids
- Analgesic and toxicological evaluation of cannabidiol-rich Moroccan Cannabis sativa L. (Khardala variety) extract: Evidence from an in vivo and in silico study
- Wound healing and signaling pathways
- Combination of immunotherapy and whole-brain radiotherapy on prognosis of patients with multiple brain metastases: A retrospective cohort study
- To explore the relationship between endometrial hyperemia and polycystic ovary syndrome
- Research progress on the impact of curcumin on immune responses in breast cancer
- Biogenic Cu/Ni nanotherapeutics from Descurainia sophia (L.) Webb ex Prantl seeds for the treatment of lung cancer
- Dapagliflozin attenuates atrial fibrosis via the HMGB1/RAGE pathway in atrial fibrillation rats
- Glycitein alleviates inflammation and apoptosis in keratinocytes via ROS-associated PI3K–Akt signalling pathway
- ADH5 inhibits proliferation but promotes EMT in non-small cell lung cancer cell through activating Smad2/Smad3
- Apoptotic efficacies of AgNPs formulated by Syzygium aromaticum leaf extract on 32D-FLT3-ITD human leukemia cell line with PI3K/AKT/mTOR signaling pathway
- Novel cuproptosis-related genes C1QBP and PFKP identified as prognostic and therapeutic targets in lung adenocarcinoma
- Bee venom promotes exosome secretion and alters miRNA cargo in T cells
- Treatment of pure red cell aplasia in a chronic kidney disease patient with roxadustat: A case report
- Comparative bioinformatics analysis of the Wnt pathway in breast cancer: Selection of novel biomarker panels associated with ER status
- Kynurenine facilitates renal cell carcinoma progression by suppressing M2 macrophage pyroptosis through inhibition of CASP1 cleavage
- RFX5 promotes the growth, motility, and inhibits apoptosis of gastric adenocarcinoma cells through the SIRT1/AMPK axis
- ALKBH5 exacerbates early cardiac damage after radiotherapy for breast cancer via m6A demethylation of TLR4
- Phytochemicals of Roman chamomile: Antioxidant, anti-aging, and whitening activities of distillation residues
- Circadian gene Cry1 inhibits the tumorigenicity of hepatocellular carcinoma by the BAX/BCL2-mediated apoptosis pathway
- The TNFR-RIPK1/RIPK3 signalling pathway mediates the effect of lanthanum on necroptosis of nerve cells
- Longitudinal monitoring of autoantibody dynamics in patients with early-stage non-small-cell lung cancer undergoing surgery
- The potential role of rutin, a flavonoid, in the management of cancer through modulation of cell signaling pathways
- Construction of pectinase gene engineering microbe and its application in tobacco sheets
- Construction of a microbial abundance prognostic scoring model based on intratumoral microbial data for predicting the prognosis of lung squamous cell carcinoma
- Sepsis complicated by haemophagocytic lymphohistiocytosis triggered by methicillin-resistant Staphylococcus aureus and human herpesvirus 8 in an immunocompromised elderly patient: A case report
- Sarcopenia in liver transplantation: A comprehensive bibliometric study of current research trends and future directions
- Advances in cancer immunotherapy and future directions in personalized medicine
- Can coronavirus disease 2019 affect male fertility or cause spontaneous abortion? A two-sample Mendelian randomization analysis
- Heat stroke associated with novel leukaemia inhibitory factor receptor gene variant in a Chinese infant
- PSME2 exacerbates ulcerative colitis by disrupting intestinal barrier function and promoting autophagy-dependent inflammation
- Hyperosmolar hyperglycemic state with severe hypernatremia coexisting with central diabetes insipidus: A case report and literature review
- Efficacy and mechanism of escin in improving the tissue microenvironment of blood vessel walls via anti-inflammatory and anticoagulant effects: Implications for clinical practice
- Merkel cell carcinoma: Clinicopathological analysis of three patients and literature review
- Genetic variants in VWF exon 26 and their implications for type 1 Von Willebrand disease in a Saudi Arabian population
- Lipoxin A4 improves myocardial ischemia/reperfusion injury through the Notch1-Nrf2 signaling pathway
- High levels of EPHB2 expression predict a poor prognosis and promote tumor progression in endometrial cancer
- Knockdown of SHP-2 delays renal tubular epithelial cell injury in diabetic nephropathy by inhibiting NLRP3 inflammasome-mediated pyroptosis
- Exploring the toxicity mechanisms and detoxification methods of Rhizoma Paridis
- Concomitant gastric carcinoma and primary hepatic angiosarcoma in a patient: A case report
- YAP1 inhibition protects retinal vascular endothelial cells under high glucose by inhibiting autophagy
- Identification of secretory protein related biomarkers for primary biliary cholangitis based on machine learning and experimental validation
- Integrated genomic and clinical modeling for prognostic assessment of radiotherapy response in rectal neoplasms
- Stem cell-based approaches for glaucoma treatment: a mini review
- Bacteriophage titering by optical density means: KOTE assays
- Neutrophil-related signature characterizes immune landscape and predicts prognosis of esophageal squamous cell carcinoma
- Integrated bioinformatic analysis and machine learning strategies to identify new potential immune biomarkers for Alzheimer’s disease and their targeting prediction with geniposide
- TRIM21 accelerates ferroptosis in intervertebral disc degeneration by promoting SLC7A11 ubiquitination and degradation
- TRIM21 accelerates ferroptosis in intervertebral disc degeneration by promoting SLC7A11 ubiquitination and degradation
- Histone modification and non-coding RNAs in skin aging: emerging therapeutic avenues
- A multiplicative behavioral model of DNA replication initiation in cells
- Biogenic gold nanoparticles synthesized from Pergularia daemia leaves: a novel approach for nasopharyngeal carcinoma therapy
- Creutzfeldt-Jakob disease mimicking Hashimoto’s encephalopathy: steroid response followed by decline
- Impact of semaphorin, Sema3F, on the gene transcription and protein expression of CREB and its binding protein CREBBP in primary hippocampal neurons of rats
- Iron overloaded M0 macrophages regulate hematopoietic stem cell proliferation and senescence via the Nrf2/Keap1/HO-1 pathway
- Revisiting the link between NADPH oxidase p22phox C242T polymorphism and ischemic stroke risk: an updated meta-analysis
- Exercise training preferentially modulates α1D-adrenergic receptor expression in peripheral arteries of hypertensive rats
- Overexpression of HE4/WFDC2 gene in mice leads to keratitis and corneal opacity
- Tumoral calcinosis complicating CKD-MBD in hemodialysis: a case report
- Mechanism of KLF4 Inhibition of epithelial-mesenchymal transition in gastric cancer cells
- Dissecting the molecular mechanisms of T cell infiltration in psoriatic lesions via cell-cell communication and regulatory network analysis
- Circadian rhythm-based prognostic features predict immune infiltration and tumor microenvironment in molecular subtypes of hepatocellular carcinoma
- Ecology and Environmental Science
- Optimization and comparative study of Bacillus consortia for cellulolytic potential and cellulase enzyme activity
- The complete mitochondrial genome analysis of Haemaphysalis hystricis Supino, 1897 (Ixodida: Ixodidae) and its phylogenetic implications
- Epidemiological characteristics and risk factors analysis of multidrug-resistant tuberculosis among tuberculosis population in Huzhou City, Eastern China
- Indices of human impacts on landscapes: How do they reflect the proportions of natural habitats?
- Genetic analysis of the Siberian flying squirrel population in the northern Changbai Mountains, Northeast China: Insights into population status and conservation
- Diversity and environmental drivers of Suillus communities in Pinus sylvestris var. mongolica forests of Inner Mongolia
- Global assessment of the fate of nitrogen deposition in forest ecosystems: Insights from 15N tracer studies
- Fungal and bacterial pathogenic co-infections mainly lead to the assembly of microbial community in tobacco stems
- Influencing of coal industry related airborne particulate matter on ocular surface tear film injury and inflammatory factor expression in Sprague-Dawley rats
- Temperature-dependent development, predation, and life table of Sphaerophoria macrogaster (Thomson) (Diptera: Syrphidae) feeding on Myzus persicae (Sulzer) (Homoptera: Aphididae)
- Eleonora’s falcon trophic interactions with insects within its breeding range: A systematic review
- Agriculture
- Integrated analysis of transcriptome, sRNAome, and degradome involved in the drought-response of maize Zhengdan958
- Variation in flower frost tolerance among seven apple cultivars and transcriptome response patterns in two contrastingly frost-tolerant selected cultivars
- Heritability of durable resistance to stripe rust in bread wheat (Triticum aestivum L.)
- Molecular mechanism of follicular development in laying hens based on the regulation of water metabolism
- Molecular identification and control studies on Coridius sp. (Hemiptera: Dinidoridae) in Al-Khamra, south of Jeddah, Saudi Arabia
- 10.1515/biol-2025-1218
- Animal Science
- Effect of sex ratio on the life history traits of an important invasive species, Spodoptera frugiperda
- Plant Sciences
- Hairpin in a haystack: In silico identification and characterization of plant-conserved microRNA in Rafflesiaceae
- Widely targeted metabolomics of different tissues in Rubus corchorifolius
- The complete chloroplast genome of Gerbera piloselloides (L.) Cass., 1820 (Carduoideae, Asteraceae) and its phylogenetic analysis
- Field trial to correlate mineral solubilization activity of Pseudomonas aeruginosa and biochemical content of groundnut plants
- Correlation analysis between semen routine parameters and sperm DNA fragmentation index in patients with semen non-liquefaction: A retrospective study
- Plasticity of the anatomical traits of Rhododendron L. (Ericaceae) leaves and its implications in adaptation to the plateau environment
- Effects of Piriformospora indica and arbuscular mycorrhizal fungus on growth and physiology of Moringa oleifera under low-temperature stress
- Effects of different sources of potassium fertiliser on yield, fruit quality and nutrient absorption in “Harward” kiwifruit (Actinidia deliciosa)
- Comparative efficiency and residue levels of spraying programs against powdery mildew in grape varieties
- The DREB7 transcription factor enhances salt tolerance in soybean plants under salt stress
- Using plant electrical signals of water hyacinth (Eichhornia crassipes) for water pollution monitoring
- Response of hybrid grapes (Vitis spp.) to two biotic stress factors and their seedlessness status
- Metabolomic profiling reveals systemic metabolic reprogramming in Alternaria alternata under salt stress
- Effects of mixed salinity and alkali stress on photosynthetic characteristics and PEPC gene expression of vegetable soybean seedlings
- Food Science
- Phytochemical analysis of Stachys iva: Discovering the optimal extract conditions and its bioactive compounds
- Review on role of honey in disease prevention and treatment through modulation of biological activities
- Computational analysis of polymorphic residues in maltose and maltotriose transporters of a wild Saccharomyces cerevisiae strain
- Optimization of phenolic compound extraction from Tunisian squash by-products: A sustainable approach for antioxidant and antibacterial applications
- Liupao tea aqueous extract alleviates dextran sulfate sodium-induced ulcerative colitis in rats by modulating the gut microbiota
- Toxicological qualities and detoxification trends of fruit by-products for valorization: A review
- Polyphenolic spectrum of cornelian cherry fruits and their health-promoting effect
- Optimizing the encapsulation of the refined extract of squash peels for functional food applications: A sustainable approach to reduce food waste
- Advancements in curcuminoid formulations: An update on bioavailability enhancement strategies curcuminoid bioavailability and formulations
- Impact of saline sprouting on antioxidant properties and bioactive compounds in chia seeds
- The dilemma of food genetics and improvement
- Causal effects of trace elements on congenital foot deformities and their subtypes: a Mendelian randomization study with gut microbiota mediation
- Honey meets acidity: a novel biopreservative approach against foodborne pathogens
- Bioengineering and Biotechnology
- Impact of hyaluronic acid-modified hafnium metalorganic frameworks containing rhynchophylline on Alzheimer’s disease
- Emerging patterns in nanoparticle-based therapeutic approaches for rheumatoid arthritis: A comprehensive bibliometric and visual analysis spanning two decades
- Application of CRISPR/Cas gene editing for infectious disease control in poultry
- Preparation of hafnium nitride-coated titanium implants by magnetron sputtering technology and evaluation of their antibacterial properties and biocompatibility
- Preparation and characterization of lemongrass oil nanoemulsion: Antimicrobial, antibiofilm, antioxidant, and anticancer activities
- Fluorescent detection of sialic acid–binding lectins using functionalized quantum dots in ELISA format
- Smart tectorigenin-loaded ZnO hydrogel nanocomposites for targeted wound healing: synthesis, characterization, and biological evaluation
- Corrigendum
- Corrigendum to “Utilization of convolutional neural networks to analyze microscopic images for high-throughput screening of mesenchymal stem cells”
- Corrigendum to “Effects of Ire1 gene on virulence and pathogenicity of Candida albicans”
- Retraction
- Retraction of “Down-regulation of miR-539 indicates poor prognosis in patients with pancreatic cancer”

