Abstract
It is quite necessary to develop a safe and efficient technique for disinfection of drinking water to avoid waterborne pathogens of infectious diseases. Herein, ZnO nanoarray electrodes with different sizes were investigated for low-voltage and high-efficiency electroporation disinfection. The results indicated that the ZnO nano-pyramid with small tip width and proper length exhibited over 99.9% disinfection efficiency against Escherichia coli under 1 V and a flow rate of 10 mL/min (contact time of 1.2 s). The suitable size of the nanoarray for electroporation disinfection was optimized by establishing the correlation between four kinds of ZnO nanoarrays and their efficiency of electroporation disinfection, which can guide the preparation of next-generation electroporation-disinfecting electrodes.
1 Introduction
Drinking water safety is closely related to human health [1]. Due to rapid economic development, water pollution has occurred in many parts of the world [2,3]. Waterborne pathogens can lead to various infectious diseases, such as diarrhea, typhoid, and cholera. Most deaths from waterborne pathogen infections occur in poor countries that still do not have access to sanitation and electricity [4,5]. To obtain safe drinking water, water disinfection treatment is required. Therefore, water disinfection treatment technology has always been the focus of attention [3,4,6]. Commonly used chlorination has low cost and is easy to apply, but there is a safety risk of producing carcinogenic by-products [7,8]. Ultraviolet disinfection and ozone disinfection have high energy consumption and limited disinfection capacity [9,10]. Moreover, traditional disinfection methods are not easily applicable in poor areas without sanitation and electricity supply [11]. To better protect human health, it is critical to develop efficient disinfection technology with low safety hazards, low energy consumption, and easy operation as a complement to modern disinfection technology.
A simple and effective application for inactivating different types of pathogens is electrodisinfection [12]. Electrodisinfection is of great interest because of its high efficiency in water treatment without generating products potentially toxics or bacterial resistance [13]. The effect of electrochemical disinfection is based mainly on the efficient generation of reactive oxidants (ROS), which not only inactivate bacteria but also degrade organic matter. In contrast, the effect of electroporation disinfection is based mainly on bacterial lysis and perforation triggered by a strong electric field, resulting in bacterial deactivation. Electroporation disinfection is more rapid compared to electrochemical disinfection [14]. The electroporation phenomenon of cell generally refers to the formation of nanoscale defects or pores in cell membrane under strong electric field, resulting in increased permeability of cell membrane to ions and other non-permeable molecules. Irreversible electroporation will be triggered when the strength of electric field is high enough (107 V/m), leading to cell death [15]. Traditional electroporation disinfection usually requires high applied voltage (10 kV) to generate strong electric field, which will not only lead to high energy consumption and high cost but also cause safety problems [16].
Since 2010, researchers have developed a bactericidal technique, which is using one-dimensional nanostructures to trigger electroporation for disinfection. Based on the locally enhanced electric field effect of the nanotips, irreversible electroporation of bacteria can achieve at a low applied voltage (<20 V), allowing for safe and low-cost electroporation disinfection [17]. Electroporation disinfection with nanoarray-modified electrodes is considered a reliable disinfection technology due to its ability to maintain good sterilization performance at low energy and low cost [18,19]. Electroporation disinfection is simultaneously performed with multiple disinfection mechanisms, including the production of ROS under electrical stimulation [14]. The contribution of ROS in disinfection depends on the experimental conditions and the nature of the electrode. It was reported that in electroporation disinfection performed at voltages above 10 V, more ROS were produced due to higher voltages, so that the disinfection mechanisms included electroporation and oxidative stress [20,21]. Under electrical stimulation, electrodes with longer contact time with bacteria, ROS became the main disinfection mechanism based on the electron transfer between the material and bacteria [22]. To avoid the problems of unnecessary water decomposition and corrosion occurring at the nanoelectrodes, researchers used a voltage below the typical voltage of electrolytic water (<2 V) for electroporation disinfection, in which case electroporation was the main disinfection mechanism [23,24,25].
How to improve the disinfection efficiency is the key issue of the electroporation-disinfecting technology [26,27]. Several kinds of nanoarray-modified porous electrodes of metal compounds were developed to achieve efficient disinfection [24,28]. Ag nanoparticle-loaded CuO nanoarray-modified (Ag NPs-CuO NWs) copper foam electrode was reported to exhibit high flow rate disinfection at 10 V applied voltage [29]. Carbon layer-coated Ag NPs-Cu2O NW-modified (C/Cu2O NWs-Ag NPs) copper foam electrode was prepared by coating a carbon layer on Ag NPs-Cu2O NWs, which enhanced the conductivity of the electrode while protecting the nanostructure using the carbon layer [21]. Branch-structured CuO-Co3O4 NWs were constructed on copper foam, and carbon film was coated on NWs to obtain a CuO-Co3O4@C NW-modified copper foam electrode, which further improved the efficiency of electroporation disinfection [30]. However, the use of precious metals, such as Ag, will bring more safety risks in the disinfection process, complex branch-structure construction and carbon layer loading processes will increase the difficulty and economic cost of the electrode preparation, while there are still problems such as hydrolysis during disinfection above 10 V [23,25,31]. These problems limit the practical application of electroporation disinfection technology. Therefore, it is important and necessary to find electroporation electrodes with simple preparation process, low material cost, and excellent disinfection performance to achieve efficient electroporation disinfection at low voltage.
As we all know, the efficiency of electroporation disinfection is closely related to the enhanced electric field generated from the nanotips under applied voltage, which may be determined by the size and morphology of the nanostructure. Therefore, morphology modulation of nanoarrays as a simple and effective means should be explored and applied to the optimization of electroporation electrodes. Up to now, there is few report on the effect of nanoarray size on the efficiency of electroporation disinfection. In this article, four types of ZnO nanoarrays (ZnO NRs) with different tip widths and lengths were prepared on copper foam electrodes. Efficiency for electroporation disinfection of ZnO NRs was realized by morphological modulation, size regulation, and the effect of electroporating disinfection under low voltage. The correlation between morphology of the nanoarray and efficiency of electroporation disinfection has been recognized, which can pave a way to the technology of electroporation disinfection.
2 Materials and methods
2.1 Chemicals and materials
Copper foams (pore size ∼200 μm) were cut into 2 cm × 2 cm as electrodes (thickness 1 mm), which were supplied by Shanghai Macklin Biotechnology Co. (China). Chemicals including hexamethylenetetramine (HMTA, (CH2)6N4), zinc nitrate hexahydrate (Zn(NO3)2·6H2O), iron nitrate nonahydrate (Fe(NO3)3·9H2O), and zinc acetate dihydrate (Zn(CH3COO)2·2H2O) were obtained from Aladdin Co. Ltd. (Shanghai). Polyethyleneimine (PEI, Mw 600) was purchased from Sigma-Aldrich. Escherichia coli (ATCC 25922) and Staphylococcus aureus (ATCC 6538) were purchased from the Guang Dong Detection Center of Microbiology (China).
2.2 Fabrication of ZnO nanoarray-modified electrodes
ZnO NRs were prepared by a two-step hydrothermal method, and the fabrication process of the ZnO seed layer referred to the sol–gel method [32] (Figure 1a). The ZnO seed layer was pre-deposited on copper foam by dip-coating from a zinc acetate ethanol colloidal solution (5 mM) and then annealing at 400°C for 40 min. To obtain a certain density of the seed layer, the coating step was selected four times. Then, chemical bath deposition was used to grow ZnO NRs [33]. A piece of copper foam substrate with the ZnO seed layer was immersed into a hydrothermal growth solution containing 50 mM of Zn(NO3)2·6H2O, 50 mM of HMTA, and PEI (1, 4, or 7 mM) at 95°C for 4 h. ZnO nano-prism, ZnO nano-prismoid, and ZnO nano-pyramid nanoarrays were prepared. ZnO nano-needle nanoarrays were grown by pre-oxidation (400°C, 20 min) of copper foam substrate before loading ZnO seed layer, and 7 mM PEI was added in the growth solution. After growth, all samples were washed with deionized water.
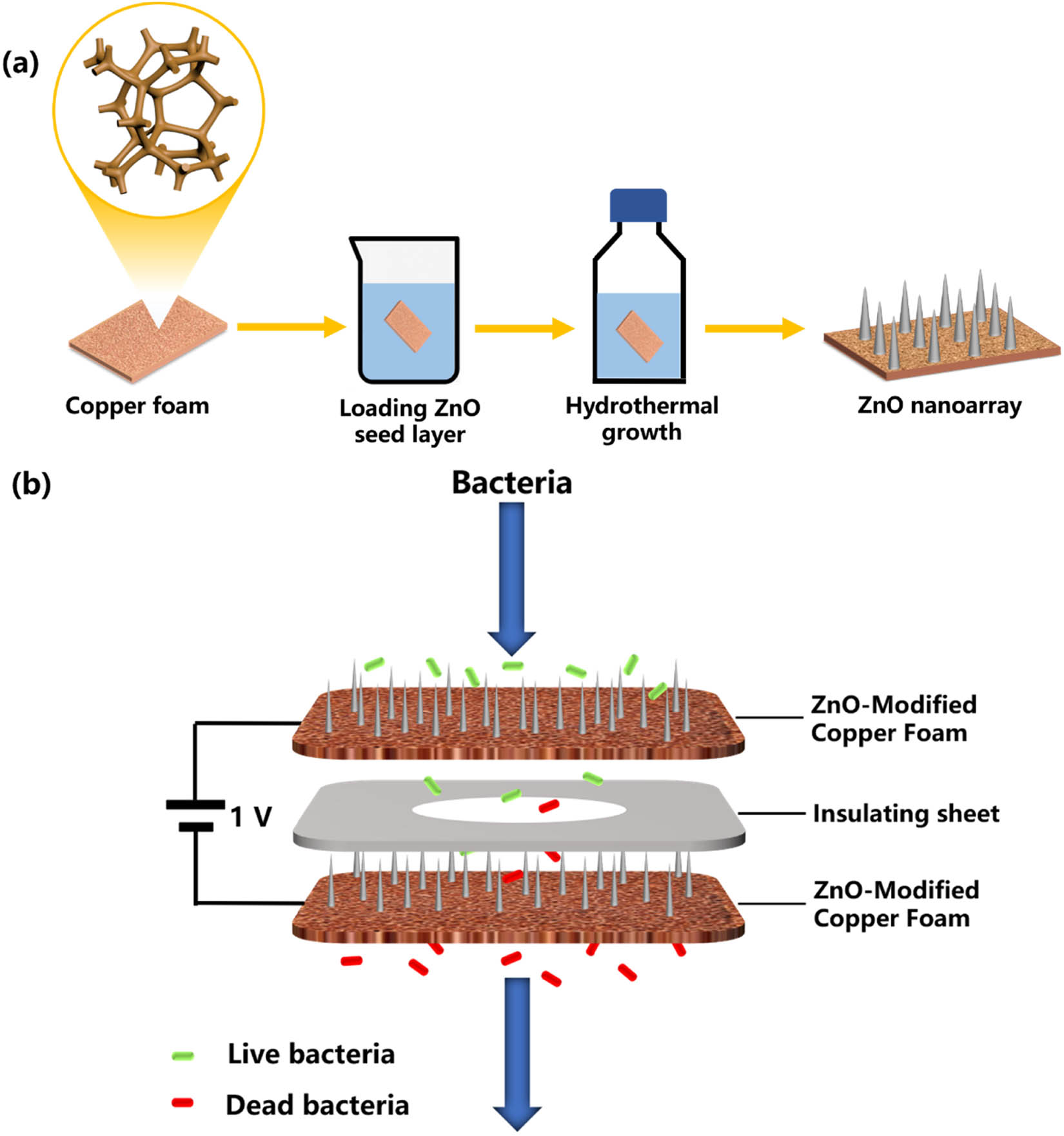
Electrode fabrication and construction of electroporation disinfection system. (a) Schematic illustration for the synthesis of ZnO NRs on copper foam electrodes. (b) Schematics showing the configuration of electroporation disinfection process.
2.3 Materials characterization
The morphology of the ZnO NR-modified electrodes was characterized by a scanning microscope (SEM, COXEM EM-30). The crystal structure of the samples was tested by X-ray diffraction (XRD, Empyrean), with CuKα radiation in the 2θ range of 5–85°.
2.4 Operation for water disinfection
Two model bacterial suspensions (E. coli [ATCC 25922] and S. aureus [ATCC 6538]) were selected and diluted with 0.9% NaCl solution to 106 colony-forming units (CFU)/mL. Before testing, two prepared ZnO NR-modified electrodes were assembled in an electroporation-disinfecting device in parallel, and the thickness of each electrode is 1 mm. The electroporation-disinfecting device is shown in Figure S1. Considering that the working surface of electroporation is about 1 cm2, flow rates were kept in the range of 5−20 mL/min, corresponding to contact times of 2.4–0.6 s.
The disinfection performance was investigated under different flow rates (corresponding to different contact times) and voltages. Among them, the flow rate was controlled by a peristaltic pump. and the voltage was controlled by a DC power supply. First, the electroporation-disinfecting device was connected to the water pipe controlled by the peristaltic pump, and the DC power supply was connected to the positive and negative copper wire. During testing, the electrodes were in the absence of an applied electric field and the presence of 1 and 2 V external voltages (the bacterial concentration is about 106 CFU/mL and the flow rate is 10 mL/min, corresponding to contact time of 1.2 s). Contact time between bacteria and electrode ranged from 0.6 to 2.4 s, which was controlled by the flow rate in the electroporation-disinfecting device (the bacterial concentration is about 106 CFU/mL, and the external voltage is 1 V). Colony counts were used to calculate bacterial removal efficiency. To ensure reproducibility, tests were conducted triple times for each group. The inactivated rate was calculated according to the following equation:
where E represents the inactivated rate, C i is the microorganism concentration in the influent, and C e is the microorganism concentration in the effluent (CFU/mL).
3 Results and discussion
3.1 Characterization of ZnO NR-modified electrodes
ZnO NRs on copper foam were fabricated by a two-step hydrothermal growth method. Among the four kinds of ZnO NRs on copper foam, ZnO nano-prisms, ZnO nano-prismoid, and ZnO nano-pyramids with similar density were obtained by introducing PEI to regulate the tip size of ZnO NRs. In addition, ZnO nano-needle with higher density was obtained by pre-oxidizing the copper foam substrate before growth. SEM images of four different tip widths of ZnO NRs are shown in Figure 2a–d. The PEI molecules tend to absorb on the crystalline surface of ZnO (100), increasing the relative c-axis growth rate, resulting in the formation of NR with uniform tip width [34]. Therefore, the tip width of ZnO NRs gradually decreased with the increase in PEI concentration. In addition, the surface roughness of copper foam increased after pre-oxidized, leading to an increase in the surface roughness of the subsequently prepared ZnO seed layer (Figure S2). The increased surface roughness of the ZnO seed layer led to an increase in the nucleation sites at the initial stage of ZnO NR growth, resulting in smaller tip width and length of nanoarrays [35].

SEM images of ZnO NRs hydrothermally grown on copper foam with PEI solution as (a) 1, (b) 4, and (c) 7 mM and (d) after pre-oxidation of copper foam, respectively.
The average value of tip width and length of the nanoarrays in the given area was measured using particle size statistics software, and the results are shown in Figure 3. The average tip width and length of ZnO nano-prism were 465 nm and 4.9 µm, respectively. The average tip width and length of ZnO nano-prismoid were 265 nm and 4.8 µm, respectively. The average tip width and length of ZnO nano-pyramid were 75 nm and 4.9 µm, respectively. And the average tip size and length of ZnO nano-needle were 35 nm and 1.3 µm, respectively. The aspect ratio of nanoarrays was calculated in the order of ZnO nano-pyramid > ZnO nano-needle > ZnO nano-prismoid > ZnO nano-prism.

Topographical parameters of the nanoarray: tip width and length, determined by SEM (according to Nano Measurer).
The XRD patterns of the ZnO NR are shown in Figure 4. It could be seen that the peaks of all four different tip size ZnO NRs are identical. When ZnO grew on the copper foam, the surface of the copper foam was oxidized to form a small amount of Cu2O, so the diffraction peaks of Cu2O and ZnO were weak compared with those of Cu. The strong peaks of Cu represented the original Cu substrate and could be designated as Cu (111), (200), and (220) planes (JCPDS card no. 04-0836). The Cu2O (JCPDS Card no. 05-0667) peaks corresponding to (110), (111), (200), (220), and (311) located at 29.5, 36.4, 42.2, 61.3, and 73.5°, respectively. The diffraction peaks corresponding to (100), (002), (101), (102), (110), (103), and (112) at 31.6, 34.3, 36.2, 47.4, 56.5, 62.8, and 67.9°, respectively, were associated with the hexagonal ZnO (JCPDS Card no. 36-1451), confirming the growth of ZnO on the copper foam.
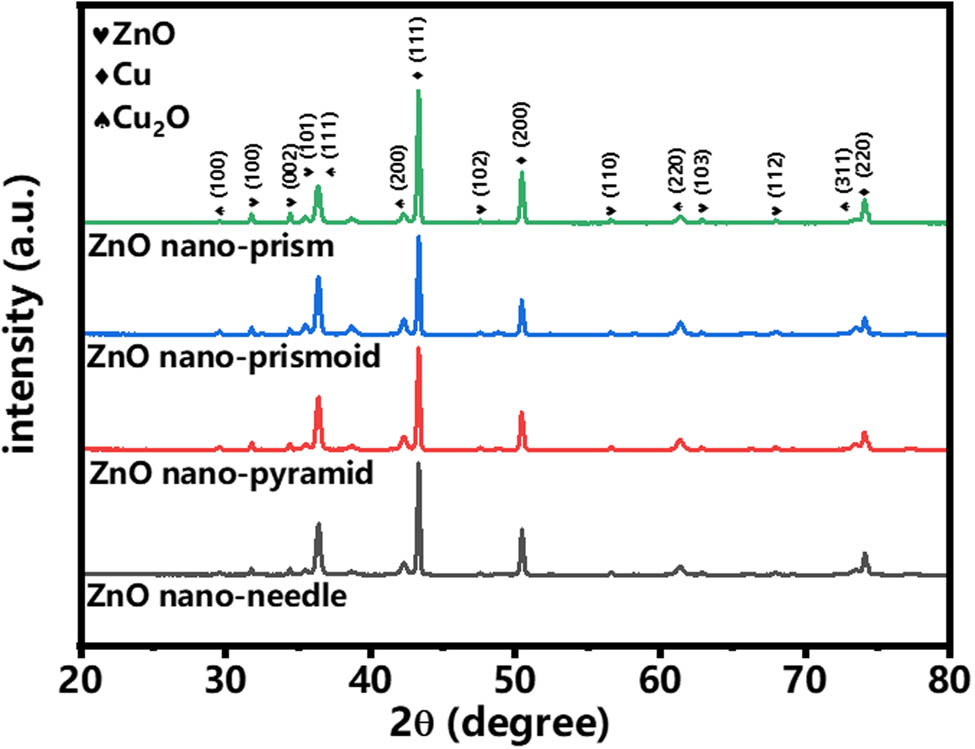
XRD patterns of ZnO NRs with different morphologies.
3.2 Performance for electroporation disinfection of ZnO nanoarrays
Applied voltage and flow rate were important parameters for evaluating the efficiency of electroporation disinfection. The disinfection efficiency of the four kinds of ZnO NR electrodes under different voltages and flow rates against two model bacterial suspensions was studied. Figure 5 shows the disinfection results of four ZnO NRs under different voltages. It could be seen that the disinfecting effect of all the four samples was not satisfied without added voltage, and the sterilization effect comes from the physical sterilization effect of ZnO NR [33]. Under the condition of applied voltage, the disinfection efficiency of the four kinds of ZnO NRs all increased. ZnO nano-pyramid had the best disinfection effect, which can achieve over 99.9% disinfection efficiency against E. coli under an applied voltage of 1 V. Moreover, the disinfection efficiency of all four kinds of ZnO NR under 2 V was significantly higher than that under 1 V, indicating that the voltage had a large effect on the efficiency of electroporation disinfection. Compared with the intact and smooth membrane of untreated E. coli, treated E. coli had significant damage on the surface (Figure S3).

Disinfection efficiency of ZnO nano-pyramid, ZnO nano-needle, ZnO nano-prismoid, and ZnO nano-prism electrodes at an applied voltage of 0–2 V and a flow rate of 10 mL/min (corresponding to contact time of 1.2 s), using 106 CFU/mL E. coli solution in a saline environment. (a) Optical images and (b) efficiency for electroporation disinfection of four ZnO NRs under 0–2 V applied voltages.
The voltage value of 1 V was selected for safety and low energy consumption, which is less than 10.5 × 10−3 J/S, as seen in Table S1. Figure 6 shows the disinfection results of four kinds of ZnO NR electrodes under different contact times. Due to the increase in the treatment flow rate, the contact time between bacteria and electrodes decreased, and the disinfection results show a gradual increase in the number of colonies in the counted Petri dishes (Figure 6a). This was due to the fact that at low flow rates, the bacteria were in contact with the ZnO electrode for a longer time and had a bigger chance for electroporation. We noticed that both ZnO nano-pyramid and ZnO nano-prismoid electrodes could achieve over 99% disinfection efficiency at a contact time of 2.4 s, illustrating the importance of sufficient contact time between bacteria and electrodes for electroporation disinfection. Meanwhile, the ZnO nano-pyramid had the best disinfection efficiency, which achieved 99.9% disinfection efficiency at a contact time of 1.2–2.4 s (flow rates 5–10 mL/min), maintained over 90% disinfection efficiency at a contact time of 0.6–0.8 s (flow rates 15–20 mL/min).

Disinfection efficiency of ZnO nano-pyramid, ZnO nano-needle, ZnO nano-prismoid, and ZnO nano-prism electrodes at an applied voltage of 1 V and a contact time of 0.6–2.4 s (corresponding to the flow rate of 5–20 mL/min), using 106 CFU/mL E. coli solution in a saline environment. (a) Optical images and (b) efficiency for electroporation disinfection of four ZnO NRs under 0.6–2.4 s contact time.
Figure 7 shows the disinfection results of all four kinds of ZnO NR electrodes against E. coli and S. aureus. It was obvious from the colony count results that the disinfection efficiency of ZnO NR electrodes against E. coli was better than that against S. aureus. Again, ZnO nano-pyramid exhibited the best electroporation disinfection efficiency for both bacteria, achieving 99.9% for E. coli and 84.4% for S. aureus under flow rate 10 mL/min (contact time of 1.2 s) and applied voltage 1 V, indicating the superiority of this morphological nanoarray in electroporation disinfection. And we found that despite a small amount of detachment and inclination of the nanorods, the surface of the ZnO nano-pyramid electrode still maintained a good morphology (Figure S4).

Disinfection efficiency of ZnO nano-pyramid, ZnO nano-needle, ZnO nano-prismoid, and ZnO nano-prism electrodes at an applied voltage of 1 V and a flow rate of 10 mL/min (corresponding to a contact time of 1.2 s), against 106 CFU/mL E. coli and S. aureus solutions in a saline environment. (a) Optical images and (b) efficiency for electroporation disinfection of four ZnO NRs against E. coli and S. aureus under 1 V and a contact time of 1.2 s.
We know that bacterial inactivation during electroporation of nanoelectrodes may be caused by a strong electric field and oxidative stress [20]. To explore the potential role of oxidative stress in bacterial inactivation, the bacteria were incubated with 10 mM glutathione (reduced form, GSH) as the influent microorganism to remove the contribution of oxidative stress to bacterial deactivation. The results show that, in this work, the disinfection efficiency is mainly attributed to the electroporation bactericidal effect caused by the strong electric field, and the oxidative stress caused by ROS is negligible (Figure S5). By comparing the disinfection performance of four kinds of ZnO NR electrodes, we found that the disinfection efficiency was in the order of ZnO nano-pyramid > ZnO nano-prismoid > ZnO nano-needle > ZnO nano-prism. Among them, the ZnO nano-pyramid with small tip width and the largest nanoarray length had the highest disinfection efficiency, which can achieve a disinfection efficiency of more than 99.9% for all 103–106 CFU/mL of E. coli (Figure S6). Disinfection efficiency of ZnO nano-needle with minimal tip width was not satisfied. This may be due to the fact that, although the tip width of ZnO nano-needle was minimal, its array length was short and dense, and such a morphology was not conducive to the electric field enhancement of the nanotips. These results indicated that for electroporation disinfection, the size of nanoarray should be pursued not only for the small size of the tip width but also for the suitable length. In addition, the comparison of the ZnO nano-pyramid electrode and other previously reported electrodes for water disinfection is shown in Table S1, further confirming the superior performance of the ZnO nano-pyramid electrode (Table S2).
4 Conclusions
In summary, the most suitable size of ZnO nanoarrays for electroporation disinfection was obtained by investigating the performance for electroporation disinfection of four kinds of ZnO NRs with different tip widths and lengths, and a simple and efficient ZnO electroporation electrode was prepared. The results indicated that the ZnO nano-pyramid with small tip width and proper length exhibited over 99.9% disinfection efficiency against E. coli under a low voltage of 1 V and a flow rate of 10 mL/min (a contact time of 1.2 s). Here, the electroporation disinfection performance was mainly derived from the locally enhanced electric field of the nanotip, rather than from the ROS. Lastly, we concluded that the smaller tip width of nanoarray was beneficial for electroporation disinfection within a certain range, but the length of nanoarray was also important. A tip width of below 100 nm and a length of above 4 μm should be a more suitable size of nanoarray for electroporation disinfection. A study on the effect of nanoarray size on the efficiency of electroporation disinfection is important for the development of electroporation-disinfecting technology.
-
Funding information: This work was financially supported by the National Natural Science Foundation of China (No. 51772251), Science and Technology Planning Project of Sichuan Province (Nos. 2023NSFSC1998, 2020ZDZX0008, and 2020ZDZX0005), and Space Station Engineering Aerospace Technology Test Field Project. And we would like to appreciate the Analysis and Testing Center of Southwest Jiaotong University for characterization (XRD, Empyrean).
-
Author contributions: All authors have accepted responsibility for the entire content of this article and approved its submission.
-
Conflict of interest: David Hui, who is the co-author of this article, is a current Editorial Board member of Nanotechnology Reviews. This fact did not affect the peer-review process. The authors declare no other conflict of interest.
References
[1] Elimelech M. The global challenge for adequate and safe water. J Water Supply Res Technology-Aqua. 2006;55(1):3–10.10.2166/aqua.2005.064Search in Google Scholar
[2] Ferreira Filho SS. A problem solving approach to aquatic chemistry. Eng Sanitaria e Ambiental. 2005;10(3):184–4.10.1590/S1413-41522005000300001Search in Google Scholar
[3] Basu H, Saha S, Kailasa SK, Singhal RK. Present status of hybrid materials for potable water decontamination: a review. Environ Sci-Water Res Technol. 2020;6(12):3214–48.10.1039/D0EW00619JSearch in Google Scholar
[4] Santosham M, Chandran A, Fitzwater S, Fischer-Walker C, Baqui AH, Black R. Progress and barriers for the control of diarrhoeal disease. Lancet. 2010;376(9734):63–7.10.1016/S0140-6736(10)60356-XSearch in Google Scholar PubMed
[5] WHO, UNICEF. Progress on drinking water and sanitation. Hydrol Und Wasserbewirtsch. 2014;58(4):244–5.Search in Google Scholar
[6] Shannon MA, Bohn PW, Elimelech M, Georgiadis JG, Mariñas BJ, Mayes AM. Science and technology for water purification in the coming decades. Nature. 2008;452(7185):301–10.10.1038/nature06599Search in Google Scholar PubMed
[7] Richardson SD, Plewa MJ, Wagner ED, Schoeny R, Demarini DM. Occurrence, genotoxicity, and carcinogenicity of regulated and emerging disinfection by-products in drinking water: A review and roadmap for research. Mutat Res-Reviews Mutat Research. 2007;636(1–3):178–242.10.1016/j.mrrev.2007.09.001Search in Google Scholar PubMed
[8] Richardson SD. Disinfection by-products and other emerging contaminants in drinking water. Trac-Trends Anal Chem. 2003;22(10):666–84.10.1016/S0165-9936(03)01003-3Search in Google Scholar
[9] Bodenes P, Bensalem S, Francais O, Pareau D, Le Pioufle B, Lopes F. Inducing reversible or irreversible pores in Chlamydomonas reinhardtii with electroporation: Impact of treatment parameters. Algal Res-Biomass Biofuels Bioprod. 2019;37:124–32.10.1016/j.algal.2018.11.016Search in Google Scholar
[10] Logan BE, Elimelech M. Membrane-based processes for sustainable power generation using water. Nature. 2012;488(7411):313–9.10.1038/nature11477Search in Google Scholar PubMed
[11] Jackson BR, Talkington DF, Pruckler JM, Fouché M, Lafosse E, Nygren B, et al. Seroepidemiologic survey of epidemic cholera in Haiti to assess spectrum of illness and risk factors for severe disease. Am J Tropical Med Hyg. 2013;89(4):654–64.10.4269/ajtmh.13-0208Search in Google Scholar PubMed PubMed Central
[12] Martinez-Huitle CA, Brillas E. A critical review over the electrochemical disinfection of bacteria in synthetic and real wastewaters using a boron-doped diamond anode. Curr Opin Solid State Mater Sci. 2021;25(4):100926.10.1016/j.cossms.2021.100926Search in Google Scholar
[13] Rems L, Miklavcic D. Tutorial: Electroporation of cells in complex materials and tissue. J Appl Phys. 2016;119(20):201101.10.1063/1.4949264Search in Google Scholar
[14] Martínez-Huitle CA, Rodrigo MA, Sirés I, Scialdone O. A critical review on latest innovations and future challenges of electrochemical technology for the abatement of organics in water. Appl Catal B: Environ. 2023;328:122430.10.1016/j.apcatb.2023.122430Search in Google Scholar
[15] Rolong A, Davalos RV, Rubinsky B. History of electroporation. In: Meijerink M, Scheffer H, Narayanan G, editors. Irreversible Electroporation in Clinical Practice. Cham: Springer; 2018.10.1007/978-3-319-55113-5_2Search in Google Scholar
[16] Kotnik T, Frey W, Sack M, Haberl Meglič S, Peterka M, Miklavčič D. Electroporation-based applications in biotechnology. Trends Biotechnol. 2015;33(8):480–8.10.1016/j.tibtech.2015.06.002Search in Google Scholar PubMed
[17] Schoen DT, Schoen AP, Hu L, Kim HS, Heilshorn SC, Cui Y. High speed water sterilization using one-dimensional nanostructures. Nano Lett. 2010;10(9):3628–32.10.1021/nl101944eSearch in Google Scholar PubMed
[18] Rojas-Chapana JA, Correa-Duarte MA, Ren Z, Kempa K, Giersig M. Enhanced introduction of gold nanoparticles into vital Acidothiobacillus ferrooxidans by carbon nanotube-based microwave electroporation. Nano Lett. 2004;4(5):985–8.10.1021/nl049699nSearch in Google Scholar
[19] Zhou J, Wang T, Yu C, Xie X. Locally enhanced electric field treatment (LEEFT) for water disinfection. Front Environ Sci Eng. 2020;14(5):78.10.1007/s11783-020-1253-xSearch in Google Scholar
[20] Wang C, Yue L, Wang S, Pu Y, Zhang X, Hao X, et al. Role of electric field and reactive oxygen species in enhancing antibacterial activity: A case study of 3D Cu foam electrode with branched CuO-ZnO NWs. J Phys Chem C. 2018;122(46):26454–63.10.1021/acs.jpcc.8b08232Search in Google Scholar
[21] Wang S, Wang W, Yue L, Cui S, Wang H, Wang C, Chen S. Hierarchical Cu2O nanowires covered by silver nanoparticles-doped carbon layer supported on Cu foam for rapid and efficient water disinfection with lower voltage. Chem Eng J. 2020;382:122855.10.1016/j.cej.2019.122855Search in Google Scholar
[22] Wang G, Feng H, Hu L, Jin W, Hao Q, Gao A, et al. An antibacterial platform based on capacitive carbon-doped TiO 2 nanotubes after direct or alternating currentcharging. Nat Commun. 2018;9(1):2055.10.1038/s41467-018-04317-2Search in Google Scholar PubMed PubMed Central
[23] Huo ZY, Liu H, Yu C, Wu YH, Hu HY, Xie X. Elevating the stability of nanowire electrodes by thin polydopamine coating for low-voltage electroporation-disinfection of pathogens in water. Chem Eng J. 2019;369:1005–13.10.1016/j.cej.2019.03.146Search in Google Scholar
[24] Huo ZY, Xie X, Yu T, Lu Y, Feng C, Hu HY. Nanowire-modified three-dimensional electrode enabling low-voltage electroporation for water disinfection. Environ Sci Technol. 2016;50(14):7641–9.10.1021/acs.est.6b01050Search in Google Scholar PubMed
[25] Huo ZY, Zhou JF, Wu Y, Wu YH, Liu H, Liu N, et al. A Cu3P nanowire enabling high-efficiency, reliable, and energy-efficient low-voltage electroporation-inactivation of pathogens in water. J Mater Chem A. 2018;6(39):18813–20.10.1039/C8TA06304DSearch in Google Scholar
[26] Zhou J, Yu C, Wang T, Xie X. Development of nanowire-modified electrodes applied in the locally enhanced electric field treatment (LEEFT) for water disinfection. J Mater Chem A. 2020;8(25):12262–77.10.1039/D0TA03750HSearch in Google Scholar
[27] Zhou JF, Wang T, Xie X. Locally enhanced electric field treatment (LEEFT) promotes the performance of ozonation for bacteria inactivation by disrupting the cell membrane. Environ Sci Technol. 2020;54(21):14017–25.10.1021/acs.est.0c03968Search in Google Scholar PubMed
[28] Tian J, Feng H, Yan L, Yu M, Ouyang H, Li H, et al. A self-powered sterilization system with both instant and sustainable antibacterial ability. Nano Energy. 2017;36:241–9.10.1016/j.nanoen.2017.04.030Search in Google Scholar
[29] Yue L, Chen S, Wang S, Wang C, Hao X, Cheng YF. Water disinfection using Ag nanoparticle-CuO nanowire co-modified 3D copper foam nanocomposites in high flow under low voltages. Environ Sci-Nano. 2019;6(9):2801–9.10.1039/C9EN00455FSearch in Google Scholar
[30] Wang H, Yu M, Cui S, Dong L, Wang S, Wei S, et al. Branched CuO-Co3O4 nanowires coated with carbon on Cu foam for water sterilization. J Environ Chem Eng. 2021;9(4):105629.10.1016/j.jece.2021.105629Search in Google Scholar
[31] Huo ZY, Liu H, Wang WL, Wang YH, Wu YH, Xie X, et al. Low-voltage alternating current powered polydopamine-protected copper phosphide nanowire for electroporation-disinfection in water. J Mater Chem A. 2019;7(13):7347–54.10.1039/C8TA10942GSearch in Google Scholar
[32] Xie Y, Wei W, Meng F, Qu X, Li J, Wang L, et al. Electric-field assisted growth and mechanical bactericidal performance of ZnO nanoarrays with gradient morphologies. Nanotechnol Rev. 2019;8(1):315–26.10.1515/ntrev-2019-0030Search in Google Scholar
[33] Xie Y, Qu X, Li J, Li D, Wei W, Hui D, et al. Ultrafast physical bacterial inactivation and photocatalytic self-cleaning of ZnO nanoarrays for rapid and sustainable bactericidal applications. Sci Total Environ. 2020;738:139714.10.1016/j.scitotenv.2020.139714Search in Google Scholar PubMed PubMed Central
[34] Zhou Y, Wu W, Hu G, Wu H, Cui S. Hydrothermal synthesis of ZnO nanorod arrays with the addition of polyethyleneimine. Mater Res Bull. 2008;43(8–9):2113–8.10.1016/j.materresbull.2007.09.024Search in Google Scholar
[35] Qiu J, Li X, Zhuge F, Gan X, Gao X, He W, et al. Solution-derived 40 mu m vertically aligned ZnO nanowire arrays as photoelectrodes in dye-sensitized solar cells. Nanotechnology. 2010;21(19):195602.10.1088/0957-4484/21/19/195602Search in Google Scholar PubMed
© 2023 the author(s), published by De Gruyter
This work is licensed under the Creative Commons Attribution 4.0 International License.
Articles in the same Issue
- Research Articles
- Preparation of CdS–Ag2S nanocomposites by ultrasound-assisted UV photolysis treatment and its visible light photocatalysis activity
- Significance of nanoparticle radius and inter-particle spacing toward the radiative water-based alumina nanofluid flow over a rotating disk
- Aptamer-based detection of serotonin based on the rapid in situ synthesis of colorimetric gold nanoparticles
- Investigation of the nucleation and growth behavior of Ti2AlC and Ti3AlC nano-precipitates in TiAl alloys
- Dynamic recrystallization behavior and nucleation mechanism of dual-scale SiCp/A356 composites processed by P/M method
- High mechanical performance of 3-aminopropyl triethoxy silane/epoxy cured in a sandwich construction of 3D carbon felts foam and woven basalt fibers
- Applying solution of spray polyurea elastomer in asphalt binder: Feasibility analysis and DSR study based on the MSCR and LAS tests
- Study on the chronic toxicity and carcinogenicity of iron-based bioabsorbable stents
- Influence of microalloying with B on the microstructure and properties of brazed joints with Ag–Cu–Zn–Sn filler metal
- Thermohydraulic performance of thermal system integrated with twisted turbulator inserts using ternary hybrid nanofluids
- Study of mechanical properties of epoxy/graphene and epoxy/halloysite nanocomposites
- Effects of CaO addition on the CuW composite containing micro- and nano-sized tungsten particles synthesized via aluminothermic coupling with silicothermic reduction
- Cu and Al2O3-based hybrid nanofluid flow through a porous cavity
- Design of functional vancomycin-embedded bio-derived extracellular matrix hydrogels for repairing infectious bone defects
- Study on nanocrystalline coating prepared by electro-spraying 316L metal wire and its corrosion performance
- Axial compression performance of CFST columns reinforced by ultra-high-performance nano-concrete under long-term loading
- Tungsten trioxide nanocomposite for conventional soliton and noise-like pulse generation in anomalous dispersion laser cavity
- Microstructure and electrical contact behavior of the nano-yttria-modified Cu-Al2O3/30Mo/3SiC composite
- Melting rheology in thermally stratified graphene-mineral oil reservoir (third-grade nanofluid) with slip condition
- Re-examination of nonlinear vibration and nonlinear bending of porous sandwich cylindrical panels reinforced by graphene platelets
- Parametric simulation of hybrid nanofluid flow consisting of cobalt ferrite nanoparticles with second-order slip and variable viscosity over an extending surface
- Chitosan-capped silver nanoparticles with potent and selective intrinsic activity against the breast cancer cells
- Multi-core/shell SiO2@Al2O3 nanostructures deposited on Ti3AlC2 to enhance high-temperature stability and microwave absorption properties
- Solution-processed Bi2S3/BiVO4/TiO2 ternary heterojunction photoanode with enhanced photoelectrochemical performance
- Electroporation effect of ZnO nanoarrays under low voltage for water disinfection
- NIR-II window absorbing graphene oxide-coated gold nanorods and graphene quantum dot-coupled gold nanorods for photothermal cancer therapy
- Nonlinear three-dimensional stability characteristics of geometrically imperfect nanoshells under axial compression and surface residual stress
- Investigation of different nanoparticles properties on the thermal conductivity and viscosity of nanofluids by molecular dynamics simulation
- Optimized Cu2O-{100} facet for generation of different reactive oxidative species via peroxymonosulfate activation at specific pH values to efficient acetaminophen removal
- Brownian and thermal diffusivity impact due to the Maxwell nanofluid (graphene/engine oil) flow with motile microorganisms and Joule heating
- Appraising the dielectric properties and the effectiveness of electromagnetic shielding of graphene reinforced silicone rubber nanocomposite
- Synthesis of Ag and Cu nanoparticles by plasma discharge in inorganic salt solutions
- Low-cost and large-scale preparation of ultrafine TiO2@C hybrids for high-performance degradation of methyl orange and formaldehyde under visible light
- Utilization of waste glass with natural pozzolan in the production of self-glazed glass-ceramic materials
- Mechanical performance of date palm fiber-reinforced concrete modified with nano-activated carbon
- Melting point of dried gold nanoparticles prepared with ultrasonic spray pyrolysis and lyophilisation
- Graphene nanofibers: A modern approach towards tailored gypsum composites
- Role of localized magnetic field in vortex generation in tri-hybrid nanofluid flow: A numerical approach
- Intelligent computing for the double-diffusive peristaltic rheology of magneto couple stress nanomaterials
- Bioconvection transport of upper convected Maxwell nanoliquid with gyrotactic microorganism, nonlinear thermal radiation, and chemical reaction
- 3D printing of porous Ti6Al4V bone tissue engineering scaffold and surface anodization preparation of nanotubes to enhance its biological property
- Bioinspired ferromagnetic CoFe2O4 nanoparticles: Potential pharmaceutical and medical applications
- Significance of gyrotactic microorganisms on the MHD tangent hyperbolic nanofluid flow across an elastic slender surface: Numerical analysis
- Performance of polycarboxylate superplasticisers in seawater-blended cement: Effect from chemical structure and nano modification
- Entropy minimization of GO–Ag/KO cross-hybrid nanofluid over a convectively heated surface
- Oxygen plasma assisted room temperature bonding for manufacturing SU-8 polymer micro/nanoscale nozzle
- Performance and mechanism of CO2 reduction by DBD-coupled mesoporous SiO2
- Polyarylene ether nitrile dielectric films modified by HNTs@PDA hybrids for high-temperature resistant organic electronics field
- Exploration of generalized two-phase free convection magnetohydrodynamic flow of dusty tetra-hybrid Casson nanofluid between parallel microplates
- Hygrothermal bending analysis of sandwich nanoplates with FG porous core and piezomagnetic faces via nonlocal strain gradient theory
- Design and optimization of a TiO2/RGO-supported epoxy multilayer microwave absorber by the modified local best particle swarm optimization algorithm
- Mechanical properties and frost resistance of recycled brick aggregate concrete modified by nano-SiO2
- Self-template synthesis of hollow flower-like NiCo2O4 nanoparticles as an efficient bifunctional catalyst for oxygen reduction and oxygen evolution in alkaline media
- High-performance wearable flexible strain sensors based on an AgNWs/rGO/TPU electrospun nanofiber film for monitoring human activities
- High-performance lithium–selenium batteries enabled by nitrogen-doped porous carbon from peanut meal
- Investigating effects of Lorentz forces and convective heating on ternary hybrid nanofluid flow over a curved surface using homotopy analysis method
- Exploring the potential of biogenic magnesium oxide nanoparticles for cytotoxicity: In vitro and in silico studies on HCT116 and HT29 cells and DPPH radical scavenging
- Enhanced visible-light-driven photocatalytic degradation of azo dyes by heteroatom-doped nickel tungstate nanoparticles
- A facile method to synthesize nZVI-doped polypyrrole-based carbon nanotube for Ag(i) removal
- Improved osseointegration of dental titanium implants by TiO2 nanotube arrays with self-assembled recombinant IGF-1 in type 2 diabetes mellitus rat model
- Functionalized SWCNTs@Ag–TiO2 nanocomposites induce ROS-mediated apoptosis and autophagy in liver cancer cells
- Triboelectric nanogenerator based on a water droplet spring with a concave spherical surface for harvesting wave energy and detecting pressure
- A mathematical approach for modeling the blood flow containing nanoparticles by employing the Buongiorno’s model
- Molecular dynamics study on dynamic interlayer friction of graphene and its strain effect
- Induction of apoptosis and autophagy via regulation of AKT and JNK mitogen-activated protein kinase pathways in breast cancer cell lines exposed to gold nanoparticles loaded with TNF-α and combined with doxorubicin
- Effect of PVA fibers on durability of nano-SiO2-reinforced cement-based composites subjected to wet-thermal and chloride salt-coupled environment
- Effect of polyvinyl alcohol fibers on mechanical properties of nano-SiO2-reinforced geopolymer composites under a complex environment
- In vitro studies of titanium dioxide nanoparticles modified with glutathione as a potential drug delivery system
- Comparative investigations of Ag/H2O nanofluid and Ag-CuO/H2O hybrid nanofluid with Darcy-Forchheimer flow over a curved surface
- Study on deformation characteristics of multi-pass continuous drawing of micro copper wire based on crystal plasticity finite element method
- Properties of ultra-high-performance self-compacting fiber-reinforced concrete modified with nanomaterials
- Prediction of lap shear strength of GNP and TiO2/epoxy nanocomposite adhesives
- A novel exploration of how localized magnetic field affects vortex generation of trihybrid nanofluids
- Fabrication and physicochemical characterization of copper oxide–pyrrhotite nanocomposites for the cytotoxic effects on HepG2 cells and the mechanism
- Thermal radiative flow of cross nanofluid due to a stretched cylinder containing microorganisms
- In vitro study of the biphasic calcium phosphate/chitosan hybrid biomaterial scaffold fabricated via solvent casting and evaporation technique for bone regeneration
- Insights into the thermal characteristics and dynamics of stagnant blood conveying titanium oxide, alumina, and silver nanoparticles subject to Lorentz force and internal heating over a curved surface
- Effects of nano-SiO2 additives on carbon fiber-reinforced fly ash–slag geopolymer composites performance: Workability, mechanical properties, and microstructure
- Energy bandgap and thermal characteristics of non-Darcian MHD rotating hybridity nanofluid thin film flow: Nanotechnology application
- Green synthesis and characterization of ginger-extract-based oxali-palladium nanoparticles for colorectal cancer: Downregulation of REG4 and apoptosis induction
- Abnormal evolution of resistivity and microstructure of annealed Ag nanoparticles/Ag–Mo films
- Preparation of water-based dextran-coated Fe3O4 magnetic fluid for magnetic hyperthermia
- Statistical investigations and morphological aspects of cross-rheological material suspended in transportation of alumina, silica, titanium, and ethylene glycol via the Galerkin algorithm
- Effect of CNT film interleaves on the flexural properties and strength after impact of CFRP composites
- Self-assembled nanoscale entities: Preparative process optimization, payload release, and enhanced bioavailability of thymoquinone natural product
- Structure–mechanical property relationships of 3D-printed porous polydimethylsiloxane films
- Nonlinear thermal radiation and the slip effect on a 3D bioconvection flow of the Casson nanofluid in a rotating frame via a homotopy analysis mechanism
- Residual mechanical properties of concrete incorporated with nano supplementary cementitious materials exposed to elevated temperature
- Time-independent three-dimensional flow of a water-based hybrid nanofluid past a Riga plate with slips and convective conditions: A homotopic solution
- Lightweight and high-strength polyarylene ether nitrile-based composites for efficient electromagnetic interference shielding
- Review Articles
- Recycling waste sources into nanocomposites of graphene materials: Overview from an energy-focused perspective
- Hybrid nanofiller reinforcement in thermoset and biothermoset applications: A review
- Current state-of-the-art review of nanotechnology-based therapeutics for viral pandemics: Special attention to COVID-19
- Solid lipid nanoparticles for targeted natural and synthetic drugs delivery in high-incidence cancers, and other diseases: Roles of preparation methods, lipid composition, transitional stability, and release profiles in nanocarriers’ development
- Critical review on experimental and theoretical studies of elastic properties of wurtzite-structured ZnO nanowires
- Polyurea micro-/nano-capsule applications in construction industry: A review
- A comprehensive review and clinical guide to molecular and serological diagnostic tests and future development: In vitro diagnostic testing for COVID-19
- Recent advances in electrocatalytic oxidation of 5-hydroxymethylfurfural to 2,5-furandicarboxylic acid: Mechanism, catalyst, coupling system
- Research progress and prospect of silica-based polymer nanofluids in enhanced oil recovery
- Review of the pharmacokinetics of nanodrugs
- Engineered nanoflowers, nanotrees, nanostars, nanodendrites, and nanoleaves for biomedical applications
- Research progress of biopolymers combined with stem cells in the repair of intrauterine adhesions
- Progress in FEM modeling on mechanical and electromechanical properties of carbon nanotube cement-based composites
- Antifouling induced by surface wettability of poly(dimethyl siloxane) and its nanocomposites
- TiO2 aerogel composite high-efficiency photocatalysts for environmental treatment and hydrogen energy production
- Structural properties of alumina surfaces and their roles in the synthesis of environmentally persistent free radicals (EPFRs)
- Nanoparticles for the potential treatment of Alzheimer’s disease: A physiopathological approach
- Current status of synthesis and consolidation strategies for thermo-resistant nanoalloys and their general applications
- Recent research progress on the stimuli-responsive smart membrane: A review
- Dispersion of carbon nanotubes in aqueous cementitious materials: A review
- Applications of DNA tetrahedron nanostructure in cancer diagnosis and anticancer drugs delivery
- Magnetic nanoparticles in 3D-printed scaffolds for biomedical applications
- An overview of the synthesis of silicon carbide–boron carbide composite powders
- Organolead halide perovskites: Synthetic routes, structural features, and their potential in the development of photovoltaic
- Recent advancements in nanotechnology application on wood and bamboo materials: A review
- Application of aptamer-functionalized nanomaterials in molecular imaging of tumors
- Recent progress on corrosion mechanisms of graphene-reinforced metal matrix composites
- Research progress on preparation, modification, and application of phenolic aerogel
- Application of nanomaterials in early diagnosis of cancer
- Plant mediated-green synthesis of zinc oxide nanoparticles: An insight into biomedical applications
- Recent developments in terahertz quantum cascade lasers for practical applications
- Recent progress in dielectric/metal/dielectric electrodes for foldable light-emitting devices
- Nanocoatings for ballistic applications: A review
- A mini-review on MoS2 membrane for water desalination: Recent development and challenges
- Recent updates in nanotechnological advances for wound healing: A narrative review
- Recent advances in DNA nanomaterials for cancer diagnosis and treatment
- Electrochemical micro- and nanobiosensors for in vivo reactive oxygen/nitrogen species measurement in the brain
- Advances in organic–inorganic nanocomposites for cancer imaging and therapy
- Advancements in aluminum matrix composites reinforced with carbides and graphene: A comprehensive review
- Modification effects of nanosilica on asphalt binders: A review
- Decellularized extracellular matrix as a promising biomaterial for musculoskeletal tissue regeneration
- Review of the sol–gel method in preparing nano TiO2 for advanced oxidation process
- Micro/nano manufacturing aircraft surface with anti-icing and deicing performances: An overview
- Cell type-targeting nanoparticles in treating central nervous system diseases: Challenges and hopes
- An overview of hydrogen production from Al-based materials
- A review of application, modification, and prospect of melamine foam
- A review of the performance of fibre-reinforced composite laminates with carbon nanotubes
- Research on AFM tip-related nanofabrication of two-dimensional materials
- Advances in phase change building materials: An overview
- Development of graphene and graphene quantum dots toward biomedical engineering applications: A review
- Nanoremediation approaches for the mitigation of heavy metal contamination in vegetables: An overview
- Photodynamic therapy empowered by nanotechnology for oral and dental science: Progress and perspectives
- Biosynthesis of metal nanoparticles: Bioreduction and biomineralization
- Current diagnostic and therapeutic approaches for severe acute respiratory syndrome coronavirus-2 (SARS-COV-2) and the role of nanomaterial-based theragnosis in combating the pandemic
- Application of two-dimensional black phosphorus material in wound healing
- Special Issue on Advanced Nanomaterials and Composites for Energy Conversion and Storage - Part I
- Helical fluorinated carbon nanotubes/iron(iii) fluoride hybrid with multilevel transportation channels and rich active sites for lithium/fluorinated carbon primary battery
- The progress of cathode materials in aqueous zinc-ion batteries
- Special Issue on Advanced Nanomaterials for Carbon Capture, Environment and Utilization for Energy Sustainability - Part I
- Effect of polypropylene fiber and nano-silica on the compressive strength and frost resistance of recycled brick aggregate concrete
- Mechanochemical design of nanomaterials for catalytic applications with a benign-by-design focus
Articles in the same Issue
- Research Articles
- Preparation of CdS–Ag2S nanocomposites by ultrasound-assisted UV photolysis treatment and its visible light photocatalysis activity
- Significance of nanoparticle radius and inter-particle spacing toward the radiative water-based alumina nanofluid flow over a rotating disk
- Aptamer-based detection of serotonin based on the rapid in situ synthesis of colorimetric gold nanoparticles
- Investigation of the nucleation and growth behavior of Ti2AlC and Ti3AlC nano-precipitates in TiAl alloys
- Dynamic recrystallization behavior and nucleation mechanism of dual-scale SiCp/A356 composites processed by P/M method
- High mechanical performance of 3-aminopropyl triethoxy silane/epoxy cured in a sandwich construction of 3D carbon felts foam and woven basalt fibers
- Applying solution of spray polyurea elastomer in asphalt binder: Feasibility analysis and DSR study based on the MSCR and LAS tests
- Study on the chronic toxicity and carcinogenicity of iron-based bioabsorbable stents
- Influence of microalloying with B on the microstructure and properties of brazed joints with Ag–Cu–Zn–Sn filler metal
- Thermohydraulic performance of thermal system integrated with twisted turbulator inserts using ternary hybrid nanofluids
- Study of mechanical properties of epoxy/graphene and epoxy/halloysite nanocomposites
- Effects of CaO addition on the CuW composite containing micro- and nano-sized tungsten particles synthesized via aluminothermic coupling with silicothermic reduction
- Cu and Al2O3-based hybrid nanofluid flow through a porous cavity
- Design of functional vancomycin-embedded bio-derived extracellular matrix hydrogels for repairing infectious bone defects
- Study on nanocrystalline coating prepared by electro-spraying 316L metal wire and its corrosion performance
- Axial compression performance of CFST columns reinforced by ultra-high-performance nano-concrete under long-term loading
- Tungsten trioxide nanocomposite for conventional soliton and noise-like pulse generation in anomalous dispersion laser cavity
- Microstructure and electrical contact behavior of the nano-yttria-modified Cu-Al2O3/30Mo/3SiC composite
- Melting rheology in thermally stratified graphene-mineral oil reservoir (third-grade nanofluid) with slip condition
- Re-examination of nonlinear vibration and nonlinear bending of porous sandwich cylindrical panels reinforced by graphene platelets
- Parametric simulation of hybrid nanofluid flow consisting of cobalt ferrite nanoparticles with second-order slip and variable viscosity over an extending surface
- Chitosan-capped silver nanoparticles with potent and selective intrinsic activity against the breast cancer cells
- Multi-core/shell SiO2@Al2O3 nanostructures deposited on Ti3AlC2 to enhance high-temperature stability and microwave absorption properties
- Solution-processed Bi2S3/BiVO4/TiO2 ternary heterojunction photoanode with enhanced photoelectrochemical performance
- Electroporation effect of ZnO nanoarrays under low voltage for water disinfection
- NIR-II window absorbing graphene oxide-coated gold nanorods and graphene quantum dot-coupled gold nanorods for photothermal cancer therapy
- Nonlinear three-dimensional stability characteristics of geometrically imperfect nanoshells under axial compression and surface residual stress
- Investigation of different nanoparticles properties on the thermal conductivity and viscosity of nanofluids by molecular dynamics simulation
- Optimized Cu2O-{100} facet for generation of different reactive oxidative species via peroxymonosulfate activation at specific pH values to efficient acetaminophen removal
- Brownian and thermal diffusivity impact due to the Maxwell nanofluid (graphene/engine oil) flow with motile microorganisms and Joule heating
- Appraising the dielectric properties and the effectiveness of electromagnetic shielding of graphene reinforced silicone rubber nanocomposite
- Synthesis of Ag and Cu nanoparticles by plasma discharge in inorganic salt solutions
- Low-cost and large-scale preparation of ultrafine TiO2@C hybrids for high-performance degradation of methyl orange and formaldehyde under visible light
- Utilization of waste glass with natural pozzolan in the production of self-glazed glass-ceramic materials
- Mechanical performance of date palm fiber-reinforced concrete modified with nano-activated carbon
- Melting point of dried gold nanoparticles prepared with ultrasonic spray pyrolysis and lyophilisation
- Graphene nanofibers: A modern approach towards tailored gypsum composites
- Role of localized magnetic field in vortex generation in tri-hybrid nanofluid flow: A numerical approach
- Intelligent computing for the double-diffusive peristaltic rheology of magneto couple stress nanomaterials
- Bioconvection transport of upper convected Maxwell nanoliquid with gyrotactic microorganism, nonlinear thermal radiation, and chemical reaction
- 3D printing of porous Ti6Al4V bone tissue engineering scaffold and surface anodization preparation of nanotubes to enhance its biological property
- Bioinspired ferromagnetic CoFe2O4 nanoparticles: Potential pharmaceutical and medical applications
- Significance of gyrotactic microorganisms on the MHD tangent hyperbolic nanofluid flow across an elastic slender surface: Numerical analysis
- Performance of polycarboxylate superplasticisers in seawater-blended cement: Effect from chemical structure and nano modification
- Entropy minimization of GO–Ag/KO cross-hybrid nanofluid over a convectively heated surface
- Oxygen plasma assisted room temperature bonding for manufacturing SU-8 polymer micro/nanoscale nozzle
- Performance and mechanism of CO2 reduction by DBD-coupled mesoporous SiO2
- Polyarylene ether nitrile dielectric films modified by HNTs@PDA hybrids for high-temperature resistant organic electronics field
- Exploration of generalized two-phase free convection magnetohydrodynamic flow of dusty tetra-hybrid Casson nanofluid between parallel microplates
- Hygrothermal bending analysis of sandwich nanoplates with FG porous core and piezomagnetic faces via nonlocal strain gradient theory
- Design and optimization of a TiO2/RGO-supported epoxy multilayer microwave absorber by the modified local best particle swarm optimization algorithm
- Mechanical properties and frost resistance of recycled brick aggregate concrete modified by nano-SiO2
- Self-template synthesis of hollow flower-like NiCo2O4 nanoparticles as an efficient bifunctional catalyst for oxygen reduction and oxygen evolution in alkaline media
- High-performance wearable flexible strain sensors based on an AgNWs/rGO/TPU electrospun nanofiber film for monitoring human activities
- High-performance lithium–selenium batteries enabled by nitrogen-doped porous carbon from peanut meal
- Investigating effects of Lorentz forces and convective heating on ternary hybrid nanofluid flow over a curved surface using homotopy analysis method
- Exploring the potential of biogenic magnesium oxide nanoparticles for cytotoxicity: In vitro and in silico studies on HCT116 and HT29 cells and DPPH radical scavenging
- Enhanced visible-light-driven photocatalytic degradation of azo dyes by heteroatom-doped nickel tungstate nanoparticles
- A facile method to synthesize nZVI-doped polypyrrole-based carbon nanotube for Ag(i) removal
- Improved osseointegration of dental titanium implants by TiO2 nanotube arrays with self-assembled recombinant IGF-1 in type 2 diabetes mellitus rat model
- Functionalized SWCNTs@Ag–TiO2 nanocomposites induce ROS-mediated apoptosis and autophagy in liver cancer cells
- Triboelectric nanogenerator based on a water droplet spring with a concave spherical surface for harvesting wave energy and detecting pressure
- A mathematical approach for modeling the blood flow containing nanoparticles by employing the Buongiorno’s model
- Molecular dynamics study on dynamic interlayer friction of graphene and its strain effect
- Induction of apoptosis and autophagy via regulation of AKT and JNK mitogen-activated protein kinase pathways in breast cancer cell lines exposed to gold nanoparticles loaded with TNF-α and combined with doxorubicin
- Effect of PVA fibers on durability of nano-SiO2-reinforced cement-based composites subjected to wet-thermal and chloride salt-coupled environment
- Effect of polyvinyl alcohol fibers on mechanical properties of nano-SiO2-reinforced geopolymer composites under a complex environment
- In vitro studies of titanium dioxide nanoparticles modified with glutathione as a potential drug delivery system
- Comparative investigations of Ag/H2O nanofluid and Ag-CuO/H2O hybrid nanofluid with Darcy-Forchheimer flow over a curved surface
- Study on deformation characteristics of multi-pass continuous drawing of micro copper wire based on crystal plasticity finite element method
- Properties of ultra-high-performance self-compacting fiber-reinforced concrete modified with nanomaterials
- Prediction of lap shear strength of GNP and TiO2/epoxy nanocomposite adhesives
- A novel exploration of how localized magnetic field affects vortex generation of trihybrid nanofluids
- Fabrication and physicochemical characterization of copper oxide–pyrrhotite nanocomposites for the cytotoxic effects on HepG2 cells and the mechanism
- Thermal radiative flow of cross nanofluid due to a stretched cylinder containing microorganisms
- In vitro study of the biphasic calcium phosphate/chitosan hybrid biomaterial scaffold fabricated via solvent casting and evaporation technique for bone regeneration
- Insights into the thermal characteristics and dynamics of stagnant blood conveying titanium oxide, alumina, and silver nanoparticles subject to Lorentz force and internal heating over a curved surface
- Effects of nano-SiO2 additives on carbon fiber-reinforced fly ash–slag geopolymer composites performance: Workability, mechanical properties, and microstructure
- Energy bandgap and thermal characteristics of non-Darcian MHD rotating hybridity nanofluid thin film flow: Nanotechnology application
- Green synthesis and characterization of ginger-extract-based oxali-palladium nanoparticles for colorectal cancer: Downregulation of REG4 and apoptosis induction
- Abnormal evolution of resistivity and microstructure of annealed Ag nanoparticles/Ag–Mo films
- Preparation of water-based dextran-coated Fe3O4 magnetic fluid for magnetic hyperthermia
- Statistical investigations and morphological aspects of cross-rheological material suspended in transportation of alumina, silica, titanium, and ethylene glycol via the Galerkin algorithm
- Effect of CNT film interleaves on the flexural properties and strength after impact of CFRP composites
- Self-assembled nanoscale entities: Preparative process optimization, payload release, and enhanced bioavailability of thymoquinone natural product
- Structure–mechanical property relationships of 3D-printed porous polydimethylsiloxane films
- Nonlinear thermal radiation and the slip effect on a 3D bioconvection flow of the Casson nanofluid in a rotating frame via a homotopy analysis mechanism
- Residual mechanical properties of concrete incorporated with nano supplementary cementitious materials exposed to elevated temperature
- Time-independent three-dimensional flow of a water-based hybrid nanofluid past a Riga plate with slips and convective conditions: A homotopic solution
- Lightweight and high-strength polyarylene ether nitrile-based composites for efficient electromagnetic interference shielding
- Review Articles
- Recycling waste sources into nanocomposites of graphene materials: Overview from an energy-focused perspective
- Hybrid nanofiller reinforcement in thermoset and biothermoset applications: A review
- Current state-of-the-art review of nanotechnology-based therapeutics for viral pandemics: Special attention to COVID-19
- Solid lipid nanoparticles for targeted natural and synthetic drugs delivery in high-incidence cancers, and other diseases: Roles of preparation methods, lipid composition, transitional stability, and release profiles in nanocarriers’ development
- Critical review on experimental and theoretical studies of elastic properties of wurtzite-structured ZnO nanowires
- Polyurea micro-/nano-capsule applications in construction industry: A review
- A comprehensive review and clinical guide to molecular and serological diagnostic tests and future development: In vitro diagnostic testing for COVID-19
- Recent advances in electrocatalytic oxidation of 5-hydroxymethylfurfural to 2,5-furandicarboxylic acid: Mechanism, catalyst, coupling system
- Research progress and prospect of silica-based polymer nanofluids in enhanced oil recovery
- Review of the pharmacokinetics of nanodrugs
- Engineered nanoflowers, nanotrees, nanostars, nanodendrites, and nanoleaves for biomedical applications
- Research progress of biopolymers combined with stem cells in the repair of intrauterine adhesions
- Progress in FEM modeling on mechanical and electromechanical properties of carbon nanotube cement-based composites
- Antifouling induced by surface wettability of poly(dimethyl siloxane) and its nanocomposites
- TiO2 aerogel composite high-efficiency photocatalysts for environmental treatment and hydrogen energy production
- Structural properties of alumina surfaces and their roles in the synthesis of environmentally persistent free radicals (EPFRs)
- Nanoparticles for the potential treatment of Alzheimer’s disease: A physiopathological approach
- Current status of synthesis and consolidation strategies for thermo-resistant nanoalloys and their general applications
- Recent research progress on the stimuli-responsive smart membrane: A review
- Dispersion of carbon nanotubes in aqueous cementitious materials: A review
- Applications of DNA tetrahedron nanostructure in cancer diagnosis and anticancer drugs delivery
- Magnetic nanoparticles in 3D-printed scaffolds for biomedical applications
- An overview of the synthesis of silicon carbide–boron carbide composite powders
- Organolead halide perovskites: Synthetic routes, structural features, and their potential in the development of photovoltaic
- Recent advancements in nanotechnology application on wood and bamboo materials: A review
- Application of aptamer-functionalized nanomaterials in molecular imaging of tumors
- Recent progress on corrosion mechanisms of graphene-reinforced metal matrix composites
- Research progress on preparation, modification, and application of phenolic aerogel
- Application of nanomaterials in early diagnosis of cancer
- Plant mediated-green synthesis of zinc oxide nanoparticles: An insight into biomedical applications
- Recent developments in terahertz quantum cascade lasers for practical applications
- Recent progress in dielectric/metal/dielectric electrodes for foldable light-emitting devices
- Nanocoatings for ballistic applications: A review
- A mini-review on MoS2 membrane for water desalination: Recent development and challenges
- Recent updates in nanotechnological advances for wound healing: A narrative review
- Recent advances in DNA nanomaterials for cancer diagnosis and treatment
- Electrochemical micro- and nanobiosensors for in vivo reactive oxygen/nitrogen species measurement in the brain
- Advances in organic–inorganic nanocomposites for cancer imaging and therapy
- Advancements in aluminum matrix composites reinforced with carbides and graphene: A comprehensive review
- Modification effects of nanosilica on asphalt binders: A review
- Decellularized extracellular matrix as a promising biomaterial for musculoskeletal tissue regeneration
- Review of the sol–gel method in preparing nano TiO2 for advanced oxidation process
- Micro/nano manufacturing aircraft surface with anti-icing and deicing performances: An overview
- Cell type-targeting nanoparticles in treating central nervous system diseases: Challenges and hopes
- An overview of hydrogen production from Al-based materials
- A review of application, modification, and prospect of melamine foam
- A review of the performance of fibre-reinforced composite laminates with carbon nanotubes
- Research on AFM tip-related nanofabrication of two-dimensional materials
- Advances in phase change building materials: An overview
- Development of graphene and graphene quantum dots toward biomedical engineering applications: A review
- Nanoremediation approaches for the mitigation of heavy metal contamination in vegetables: An overview
- Photodynamic therapy empowered by nanotechnology for oral and dental science: Progress and perspectives
- Biosynthesis of metal nanoparticles: Bioreduction and biomineralization
- Current diagnostic and therapeutic approaches for severe acute respiratory syndrome coronavirus-2 (SARS-COV-2) and the role of nanomaterial-based theragnosis in combating the pandemic
- Application of two-dimensional black phosphorus material in wound healing
- Special Issue on Advanced Nanomaterials and Composites for Energy Conversion and Storage - Part I
- Helical fluorinated carbon nanotubes/iron(iii) fluoride hybrid with multilevel transportation channels and rich active sites for lithium/fluorinated carbon primary battery
- The progress of cathode materials in aqueous zinc-ion batteries
- Special Issue on Advanced Nanomaterials for Carbon Capture, Environment and Utilization for Energy Sustainability - Part I
- Effect of polypropylene fiber and nano-silica on the compressive strength and frost resistance of recycled brick aggregate concrete
- Mechanochemical design of nanomaterials for catalytic applications with a benign-by-design focus

