Abstract
Storage of wastes leads to severe problems of water pollution and neighboring matrices due to the infiltration of landfill leachate. Uncontrolled landfill and waste storage can lead to groundwater pollution, which can lead to serious health problems for the living. Engineered barriers can be a solution to these pollution problems. The purpose of this study was to develop novel composite materials – clay-based, activated carbon, cement, and PVA polymer. These composites were intended for the containment of waste in landfill. The clay (70–80%) and activated carbon (5–15%) contents were varied to obtain three different geomaterials – GM1, GM2, and GM3. In the preparation of GM3, the content of activated carbon used was higher than for GM1 and GM2, paracetamol removal capacity tested by adsorption, experiments were influenced by parameters such as the adsorbent mass, the initial solute concentration, contact time, temperature, and pH effect. The parameter of initial paracetamol concentrations was studied using a range of 50, 100, and 150 mg L−1. For a GM3 mass of 80 mg, the adsorbed amount is 14.67 mg g−1, and the contact time is 180 minutes. This study revealed that composites are efficient for the treatment of landfill leachates.
1 Introduction
The industrial development of certain industries such as pharmaceutical, chemical, petrochemical, and agrifood industries leads to the waste generation. This is an inevitable outcome, which represents a risk for the environment and the human health when they are not recoverable (recycling, energetic recovery). Therefore, the burying of these wastes can lead to groundwater pollution problems leading to leachate infiltration. Thus, these effluents require prior treatment to retain toxic pollutants. Drug consumption in developing and developed countries is becoming increasingly intense. The release of these pollutants in the environment affects water and soil. These drugs are present in trace amounts. Among the drugs detected in trace amounts are antidepressants, antibiotics, contraceptives, antiepileptics, anti-inflammatory, aspirin, and paracetamol. Numerous paracetamol-based medications are available over the counter and commonly used for pain and fevers [1]. Drug use is the main source of release to the environment. After administration, the drug is absorbed, metabolized (transformed by the body), excreted, and then released to wastewater [2]. Several studies have shown the presence of drugs in drinking water at a concentration of 211 ng L−1 [2,3]. Indeed, paracetamol is metabolized in the liver and excreted in the urine and then released in wastewater. In the case of paracetamol overdose or if the time of taking the drug is wrong, some of them (paracetamol molecules) are directly excreted. The metabolization process occurred by a biochemical process that transformed the original molecules [2,4,5]. The new products called metabolites are excreted and released into the natural environment and then transported to urban treatment plants [4,6,7], which partially degrades. Excreted molecules are found in wastewater systems via sewage treatment plant effluents and pollute water. Therefore, stream get loaded with residues, then aquatic organisms are infected. Thus, paracetamol is the priority drug in the aquatic environment based on criteria of toxicity, persistence, and fate in the environment [8]. The paracetamol overdose can attack the liver and induce liver failure (hepatotoxicity) and hepatic coma in animals and humans [4,9,10]. Paracetamol overdose induces the formation of a metabolite form (N-acetyl-p-benzoquinone imine, NAPQI) toxic to the liver. Since the neutralization capacity of NAPQI by glutathione is exceeded, oxidative stress is produced causing mitochondrial dysfunction, mediated by the activation of a cascade of cytosolic kinases and followed by DNA fragmentation [11]. The risks associated with paracetamol, such as cardiovascular, gastrointestinal, and kidney diseases, have increased. Risk of kidney problems increases when paracetamol is taken cumulatively over a lifetime, as reported in a recent study [12]. Therefore, efficient and low-cost methods to deal with the environmental and health problems of paracetamol must be implemented. To remedy the problem of pollutant infiltration, researchers carried out a study using the cellulosic waste product to improve the physicochemical properties of the soil. [13,14]. This study attempts to show the possibility of reducing risks in the environment. Therefore, the development of landfills requires a permanently sealed barrier to protect the surrounding natural environment by preventing leachates from polluting soil and groundwater. The engineered barrier of a waste storage site is intended to prevent the discharge of landfill juices into groundwater by collecting leachate. Functions are active sealing and drainage. For drainage, granular and synthetic materials are used and geomembranes to improve sealing. This barrier must be permanently sealed and has a high capacity to retain organic and inorganic pollution [15]. Clay minerals have very low permeability, due to the small particle size and complex porous structures; they also have a large surface area that gives rise to strong interactions with liquids and dissolved species [16]. These interactions allow the retention of various leachate components. The montmorillonite clay smectite has a layered structure – an octahedral layer between two tetrahedral layers. The atom substitution in clay structure produces a negative charge compensated by the ions in the interlayer space (Li+, Na+, Ca2+, K+). Metal ion retention such as heavy metals is due to the electrostatic between negatively charged montmorillonite and compensating ions facilitated by cation exchange called Cation Exchange Capacity (CEC) [17,18].
The clay can be negatively or positively charged depending on the environment where it is located either acidic or basic. This property can promote the adsorption of organic pollutants by the formation of strong hydrogen bonds with Si–OH groups on the clay surface or by dispersion forces, as is the case with polar aromatic organic compounds or aliphatic organic compounds [19,20,21,22]. Similarly, activated carbon with a large porous surface and thermostability is one of the treatments frequently used against the infiltration of landfill leachates [14,23,24]. The surface of activated carbon with several functional groups plays a significant role in the removal of organic pollutants [25,26,27]. The interaction of the material with organic compounds is classified into three types [27,28,29]:
Organic compound-activated carbon.
Organic compound-solution.
Activated carbon-solution.
The removal of organic compounds by activated carbon is due to the attraction that occurs between the π orbitals of the base planes of carbon and the electron density of aromatic rings (π–π interactions) [30,31,32,33]. The activated carbon properties provide a high capacity to remove organic pollutants; hence, the choice to be used as an engineered barrier.
The clay-based engineering barrier has been the subject of the work of several authors [34,35,36,37]. Studies have revealed that clay is very effective in removing heavy metals [38,39,40,41].
Clay and activated carbon characteristics confer retention properties to the engineered barrier. To improve the retention properties of pollutants and isolate the groundwater from the source of pollution in the landfill sites, authors mixed clay and activated carbon and added the cement and the polyvinyl alcohol PVA polymer to increase the strength and the lifespan of the barrier. However, by mixing these constituents with different mass percentages, a range of materials called geomaterials. The removal of paracetamol by adsorption in an aqueous medium is tested on these materials and their mineral constituents.
2 Materials and methods
2.1 Reagents
Paracetamol (C8H9NO2) – purity >99%; CAS Number: 103-90-2 – was purchased from Sigma-Aldrich. All other reagents used in this study are analytical grade and purchased from Riedel de Haën with a purity of 99%. A stock solution of 1 g/L paracetamol was prepared. A range of dilute solutions (25–700 mg/L) is prepared from the stock solution. Geomaterials used were prepared based on clay from western Algeria (Maghnia), activated carbon powder made from coconut purchased from Merck, Portland cement from Chlef (western Algeria), and PVA polymer from Fluka.
2.2 Geomaterial synthesis
The synthesis of geomaterials follows the following path. First, the clay was purified according to the Van Olphen process [42]. Then, a solution of NaCl 1 N was added to the purified clay. This step was performed to obtain a mono ionic clay. Then, the constituents of the geomaterials were added at different percentages by mass, mixed with 100 mL of distilled water with stirring for 48 h. The mixture was dried in an oven at 60°C, the resulting product was crushed. The final product is less than 1 µm in size. Figure 1 illustrates the geomaterial compositions.
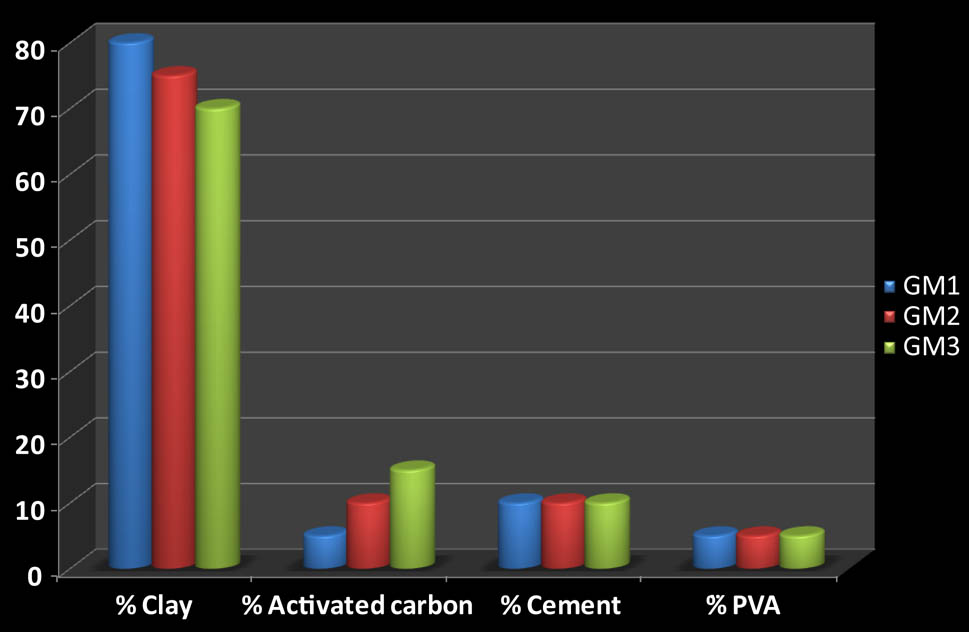
Composition of geomaterials.
2.3 Characterization
2.3.1 Nitrogen adsorption–desorption method
The studied adsorbents were previously degassed for 15 h at 150°C, then nitrogen adsorption–desorption was measured with the micrometric instrument ASAP 2010 at 77 K. The specific surface area of the samples was determined by the BET method [43].
2.3.2 X-ray diffraction (XRD)
Samples were analyzed by X-ray diffraction, curves recorded on an Xpert Pro Panalytical diffractometer using copper radiation of wavelength λ = 0.15418 nm. The crushed materials were placed on metal plates. Measurements were carried out in an angular range of 2θ from 0.03 to 70°, using a step of 0.02°.
2.3.3 Scanning electron microscopy (SEM)
The morphology of the samples was observed on a PHILIPS XL-30 FEG scanning electron microscope. Samples were powdered on an aluminum support previously covered with a self-adhesive pellet containing graphite.
2.4 Adsorption procedure
The stock solution of 1 g/L paracetamol was prepared with a purity of 99%. The studied solutions were prepared by dilution from the stock solution until the desired concentrations were obtained. The mass of the adsorbent is an important factor because of its effect on the adsorbent’s ability to fix the adsorbate. The experiment was performed by mixing a mass of adsorbent with a volume of 10 mL of 150 mg/L of paracetamol solution. The mass of adsorbent was 10, 20, 40, 60, 80, and 100 mg in batch mode.
The adsorption kinetic was studied at the pH of the solution in a thermostated bath at 25°C, using 10 mL of different concentrations of polluted solution put in contact with an adsorbent matrix (GM1, GM2, and GM3) with a mass of 80 mg. The samples were shaken for different times (2 min to 24 h), and then the samples were filtered and analyzed by the UV spectroscopy analysis using a Perkin Elmer Lambda 35 spectrophotometer at a detection wavelength of 241.7 nm. The adsorption isotherms were carried out at pH of the solution; a volume of 10 mL of 25, 50, 100, 150, 200, 300, 400, 500, 600, and 700 mg L−1 of paracetamol solution. The pH effect was studied only with GM3. Using flasks that contain a volume of 10 mL of the paracetamol solution with a concentration of 150 mg L−1 at pH values from 2 to 12, by separation put in contact with GM3 with a mass of 80 mg. The samples were shaken for 24 h at 25°C. The initial pH values were adjusted by adding 0.1 N hydrochloric acid HCl or 0.1 N potassium hydroxide KOH solutions. The temperature effect was carried out at 25, 35, and 45°C at the same operating conditions to the adsorption isotherm at 25°C. The adsorption capacity and the removal efficiency were expressed by the relations (1) and (2), respectively:
where, Q a is the adsorbed amount of pollutant per gram of adsorbent (mg/g), C 0 is the initial solute concentration (mg/L), C e is the residual concentration of the solute (mg/L), V is the solution volume (L), m is the mass of the adsorbent (g).
2.5 Error measurement
Reproducibility of paracetamol adsorption results on GM1, GM2, and GM3 was studied. Experiments were repeated three times under the same operating conditions used in this study. The uncertainty was calculated using the following expression:
Hence n = 3.
-
Ethical approval: The conducted research is not related to either human or animal use.
3 Result and discussion
3.1 Characterization
3.1.1 XRD and SEM methods
Numerous studies used new materials in the landfill to isolate the source of pollution from groundwater and reduce the toxicity of pollutants until their removal [44,45,46,47,48,49]. In this section, the characterization of the synthesized geomaterials in Section 2.2 was carried out. The studied geomaterials were analyzed by X-ray diffraction. The XRD patterns showed contain montmorillonite, kaolinite, quartz, and calcite, as shown in Figure 2. Scanning electron microscopy analysis allows visualization of the morphology of the studied materials as shown in Figure 3.
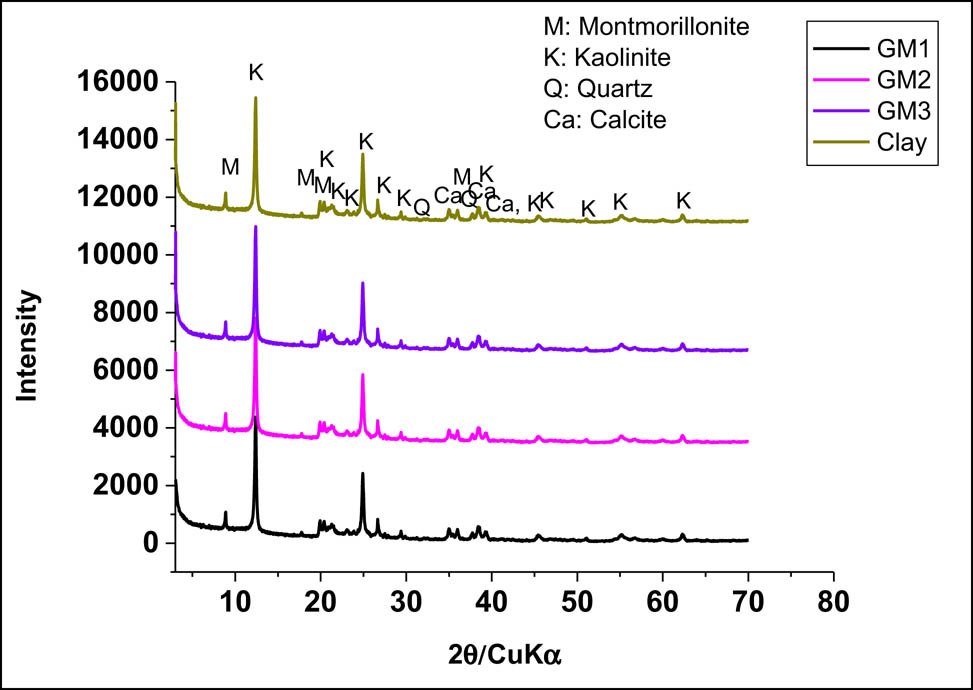
X ray diffraction patterns of geomaterials and clay.

SEM images of ATMa clay (a), GM1 (b), GM2 (c), GM3 (d).
The pure clay is in the form of leaves, shows particles of very variable sizes, and is most often aggregated (Figure 3a). With the addition of the geomaterials constituents, a smooth surface is obtained, which is pronounced in Figure 3d due to the activated carbon deposit [44,50].
3.1.2 Nitrogen adsorption–desorption isotherms
The nitrogen adsorption–desorption isotherm was studied. The result presented in Figure 4 shows that the adsorption isotherms were type 4 according to the IUPAC classification, which corresponds to mesoporous materials. It is also noticed that at low pressures, the volume of adsorbed nitrogen increased sharply and this is due to the microporosity which is related to the addition of activated carbon.
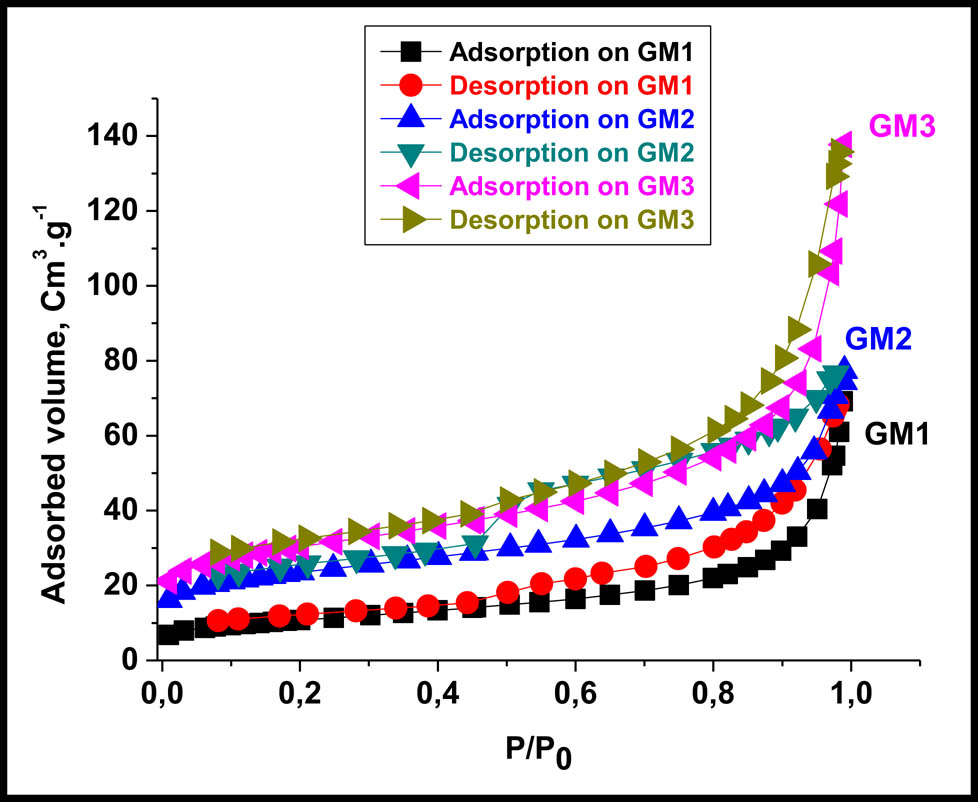
N2 adsorption–desorption isotherm on GM1, GM2, and GM3.
The specific surface areas of the studied materials were calculated by the BET method, from linear forms of nitrogen adsorption–desorption isotherms; the results are shown in Tables 1 and 2.
Source of reagents and characterization of the geomaterial constituents
| Constituents | Origin | S BET (N2/77 K) | V p (cm3 g−1) | CEC meq/100 g | Size of particles (μm) | Moisture (%) |
|---|---|---|---|---|---|---|
| Bentonite | Maghnia | 56.23 | 0.10 | 86.5 | <2 | 9 |
| Cement | Chlef | 6.68 | 0.01 | 92.8 | 200–250 | 2.5 |
| Activated carbon | Shell of coconut | 658.52 | 0.55 | — | <20 | 7.8 |
Physicochemical characteristics of geomaterials
| Geomaterial | S BET (N2/77 K) | V p (cm3 g−1) |
|---|---|---|
| GM1 | 63.82 | 0.10 |
| GM2 | 79.89 | 0.12 |
| GM3 | 102.9 | 0.21 |
From the results presented in Tables 1 and 2, it was noticed that the specific surface areas and pore volumes of the geomaterials increased by increasing the mass percentages of activated carbon.
3.2 Paracetamol adsorption
3.2.1 Adsorbent mass effect
Adsorption may be influenced by several parameters. In this section, the adsorbent mass effect on paracetamol adsorption was investigated and the result was reported in Figure 5.
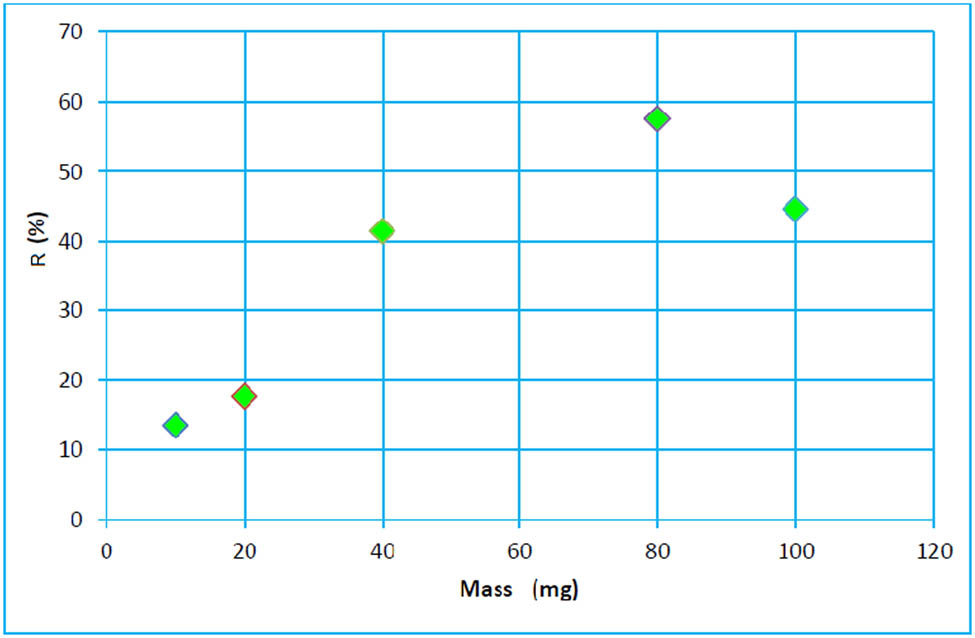
Influence of geomaterial mass on paracetamol removal yield.
The curve shown in Figure 5 indicates that the removal yield increases by increasing the mass of the geomaterials until a maximum was reached. It is noted that the yield rate decreases for higher adsorbent dose [51]. This decrease in rate remains constant according to a previous study, due to the increase in the adsorbent mass, which creates an obstruction to prevent pollutant molecules to access adsorbent sites.
3.2.2 Adsorption kinetic
The contact time effect on paracetamol adsorption is shown in Figure 6.
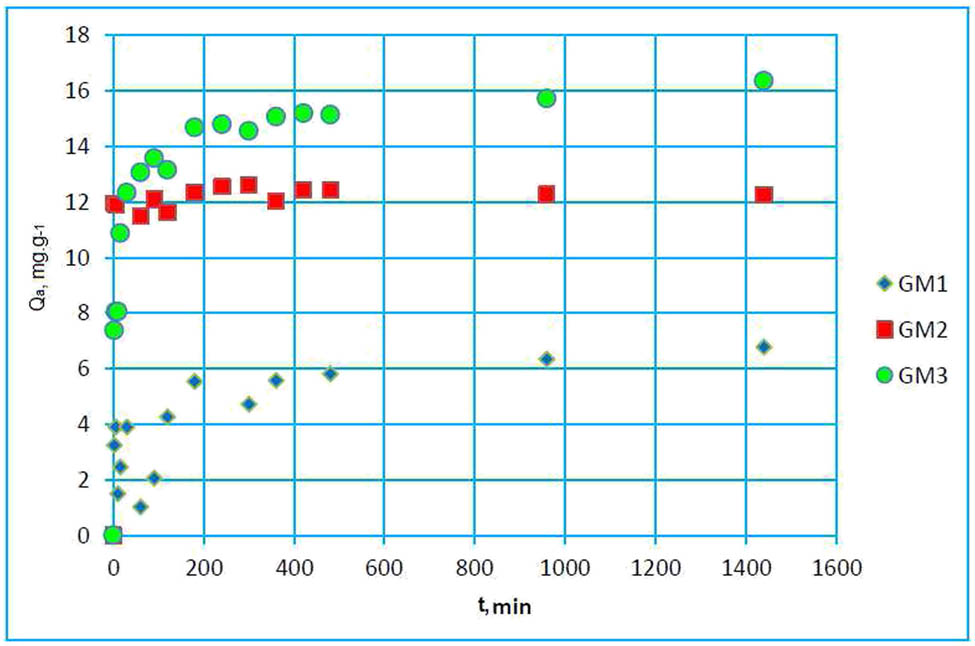
Variation of adsorbed amount as a function of contact time.
The stages of the paracetamol adsorption kinetic are shown in Figure 6. First, a fast increase of the adsorbed amount was observed due to the availability of sites on the adsorbent surface [52], then a plateau was reached, which can be explained by saturation of the adsorbent sites. These curves were used to determine the equilibrium time (t eq) and the adsorbed amount (Q a), which follow the succeeding order: t eq (GM3) = t eq (GM2) < t eq (GM1), Q a (GM1) < Q a (GM2) < Q a (GM3); with values: 120 and 180 min, 4, 5.42, and 5.96 mg g−1, respectively. This is related to the characteristics of geomaterials. Indeed, GM3 has a specific surface area of 102.90 m2 g−1 and a pore volume V p of 0.21 cm3 g−1, which are 63.82 m2 g−1 and 0.1 cm3 g−1 and S of 79.89 m2 g−1 and V p of 0.12 cm3 g−1, respectively, for GM1 and GM2. It has been demonstrated in a previous study [48] that the adsorption of pollutants on geomaterials is the contribution of mineral constituents, namely, clay, activated carbon, and cement.
3.2.3 Adsorption isotherm
The adsorption isotherm is a curve relating the equilibrium concentration of a solute on the surface of an adsorbent. The adsorption isotherms of GM1, GM2, and GM3 at 298 K are shown in Figure 7.
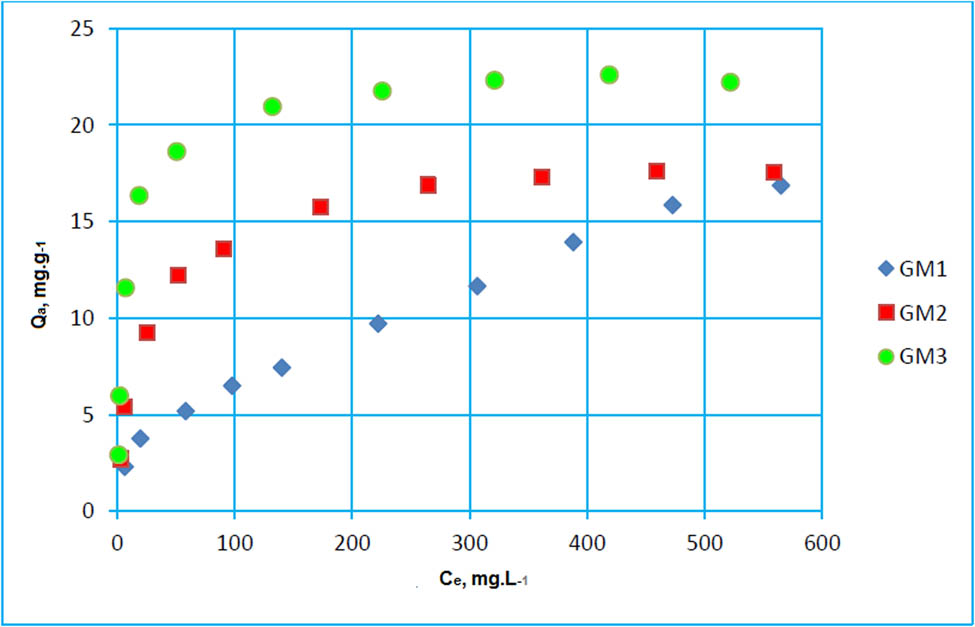
Paracetamol adsorption isotherm on GM1, GM2, and GM3 at 298 K.
The adsorption isotherm is of type L according to Giles classification [53], characteristic of high-affinity systems between adsorbate and adsorbent, and a weak competition between the solvent and the solute molecules.
3.2.4 Temperature effect
Temperature is one of the parameters that influence adsorption. Figure 8 illustrates the adsorption isotherms at 308 and 318 K.
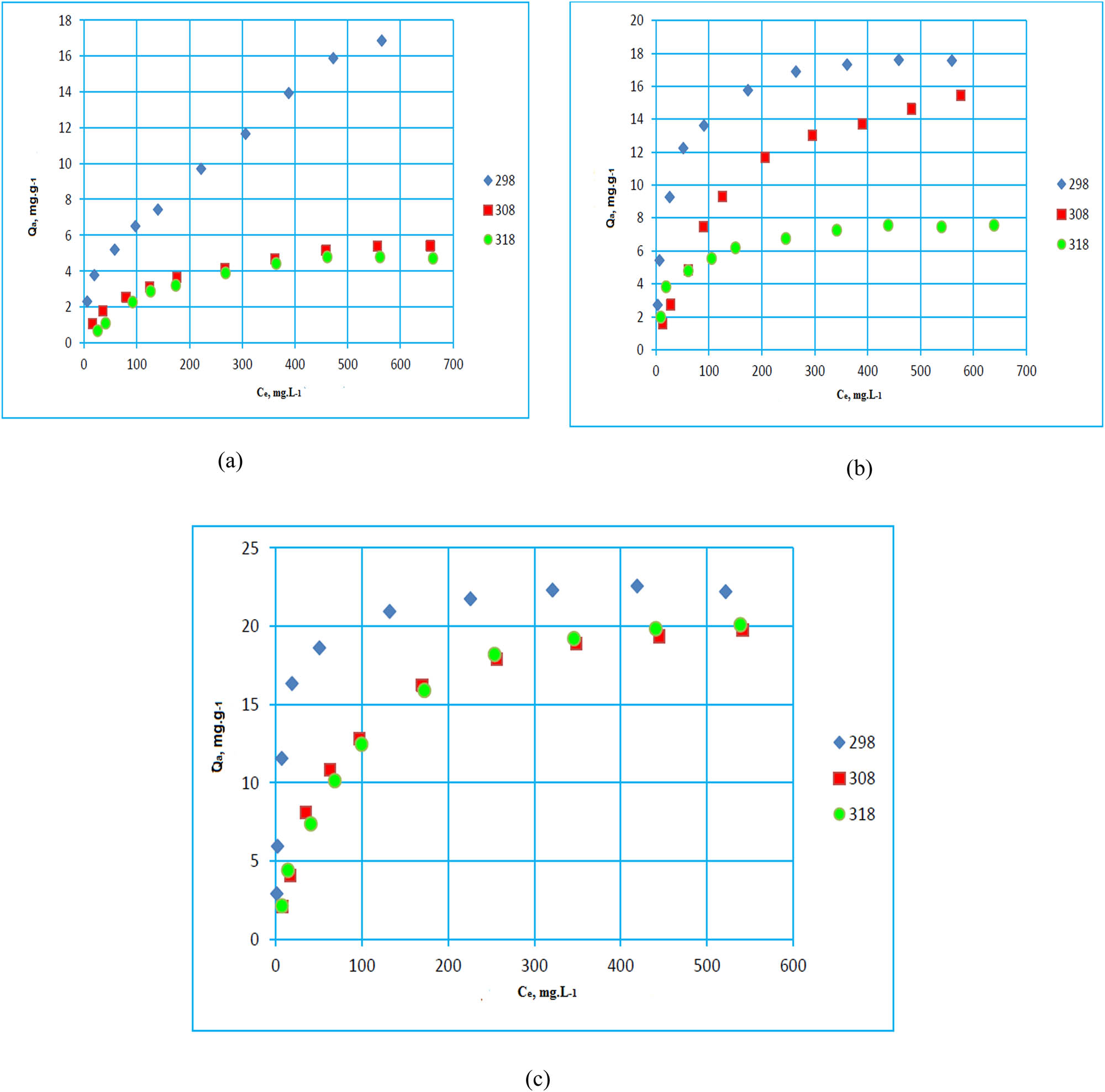
Paracetamol adsorption isotherm on GM1 (a), GM2 (b), and GM3 (c) at different temperatures.
The results reveal the role of temperature (T) on adsorption. Indeed, by increasing T, the adsorbed amount decreases. That breaks the bonds between the adsorbate and the adsorbent.
3.3 The thermodynamic aspect of adsorption
The temperature effect was studied to determine the thermodynamic parameters from the following equation:
where
The plot of ln K d as a function of 1/T (figure not shown) to determine ∆H°, ∆S°, and ∆G°; results are shown in Table 3.
Thermodynamic parameters of paracetamol adsorption on GM1, GM2, and GM3
| Materials | Temperatures (K) | ΔH° (kJ mol−1) | ΔS° (kJ K−1 mol−1) | ΔG° (kJ mol−1) |
|---|---|---|---|---|
| GM1 | 298 | −56.01 | −0.218 | −8.95 |
| 308 | −11.13 | |||
| 318 | −13.31 | |||
| GM2 | 298 | −38.13 | −0.155 | −8.06 |
| 308 | −9.61 | |||
| 318 | −11.16 | |||
| GM3 | 298 | −5.30 | −0.044 | −7.81 |
| 308 | −8.25 | |||
| 318 | −8.69 |
Values of the free enthalpy variations ΔG° are negative for all geomaterials and indicate the spontaneity of the adsorption.
3.4 pH effect
The pH of the solution is an important parameter that can influence the adsorption and the physicochemical parameters of adsorbates-adsorbents. Figure 9 represents the adsorbed amount as a function of the solution pH.
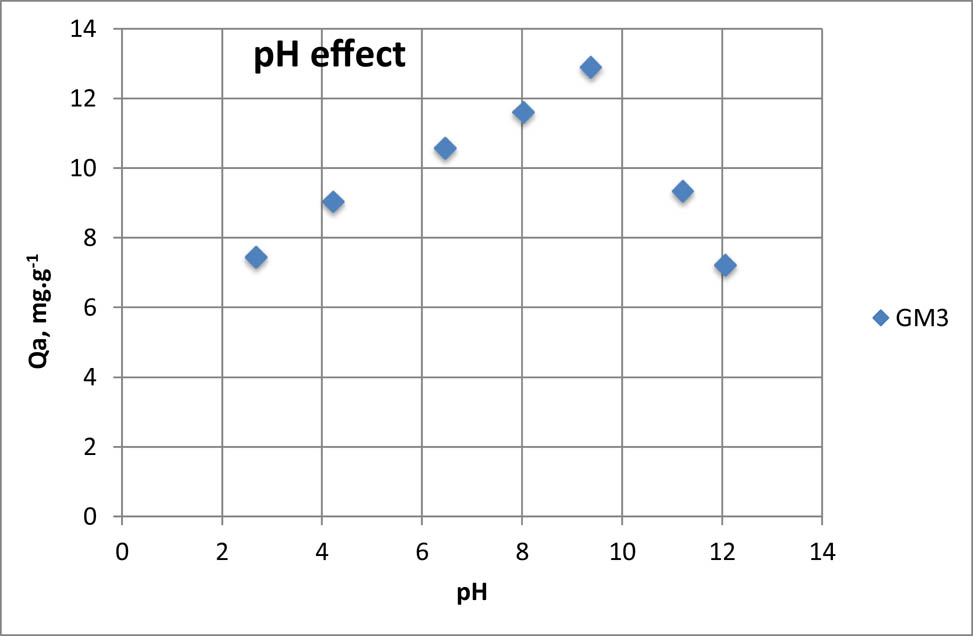
pH effect on paracetamol adsorption onto GM3.
The paracetamol adsorption on geomaterials is pH-dependent. The adsorbed amount increases until it reaches a maximum at pH 9 and decreases to a higher value. This mechanism is due to the surface load of the geomaterial. Indeed, at acidic pH < pK a, the ions resulting from the adsorbate were in competition with protons H of the medium. However, groups aluminols and silanols on the surface of GM3 (resulting from the clay) were more protonated [54], and thus less willing to receipt the studied pollutant. This protonation decreases more and more until pH = pK a. At pH > pK a very basic, the adsorption was inhibited due to the repulsion of the medium and the geomaterial. At pH = pK a, the paracetamol is in its molecular form [55]. Hence, the fixation of paracetamol is favored on the sites of GM3.
3.5 Results of error measurement calculation
The result of the uncertainty calculation was represented in Table 4.
Error calculation on paracetamol adsorption study on geomaterials GM1, GM2, and GM3
| Geomaterials | GM1 | GM2 | GM3 |
|---|---|---|---|
| Uncertainty on the kinetics of adsorption | 6.383 ± 0.195 | 12.272 ± 0.002 | 15.900 ± 0.195 |
| Uncertainty on the adsorption isotherm (298, 308, 318 K) | 15.215 ± 0.001 (298 K) | 17.2236 ± 0.0004 (298 K) | 22.0845 ± 0.0003 (298 K) |
| 5.203 ± 0.001 (308 K) | 14.7556 ± 0.0003 (308 K) | 19.40236 ± 7 × 10−5 (308 K) | |
| 4.792 ± 0.002 (318 K) | 7.302 ± 0.001 (318 K) | 19.7971 ± 0.0002 (318 K) | |
| Uncertainty on pH | 9.77 ± 2.09 | ||
| Uncertainty on ∆H° (kJ mol−1) | −48.25 ± 2.42 | −39.78 ± 1.30 | −10.67 ± 7.53 |
| Uncertainty on ∆S° (kJ mol−1 K−1) | −0.191 ± 0.008 | −0.159 ± 0.0028 | −0.06 ± 0.02 |
| Uncertainty on ∆G° (kJ mol−1) | −8.874 ± 0.13 (298 K) | −7.68 ± 0.41 (298 K) | −7.27 ± 0.64 (298 K) |
| −10.789 ± 0.15 (308 K) | −9.30 ± 0.42 (308 K) | −7.87 ± 0.43 (308 K) | |
| −12.912 ± 0.70 (318 K) | −10.88 ± 0.39 (318 K) | −8.47 ± 0.26 (318 K) |
4 Conclusion
The purpose of the present study is, first, to develop composite materials called geomaterials. These materials were based on Algerian clay, activated carbon, cement, and PVA polymer, then these materials were used in the retention of an organic pollutant such as paracetamol. The synthesized geomaterials were characterized by nitrogen adsorption and desorption, X-ray diffraction, and SEM. These methods were performed to investigate their textural properties, their porosities as well as the distribution of the pore size and the different mineral phases that exist in each material. Adsorption experiments were carried out and evaluate the parameters that could affect the paracetamol adsorption on GM1, GM2, and GM3 geomaterials, in particular adsorbent mass, contact time, and temperature. The obtained results showed that:
The equilibrium is reached after relatively short contact times 180 min for GM1 and 120 min for GM2 and GM3.
From results, the most suitable geomaterial for paracetamol removal is GM3, due to its physicochemical properties.
The adsorbed amount decreases by increasing the temperature. This behavior highlighted the exothermic nature of the process.
The optimum adsorption capacity value was obtained at a basic pH of 9.35, very close to that of the paracetamol acid constant (pK a).
The thermodynamic parameter values indicate the process is spontaneous and exothermic process.
According to the Algerian law of December 12, 2001, relating to the management, control, and elimination of waste, the geomaterial GM3 can be used for containment waste in a landfill and protect the environment.
Acknowledgments
This work was carried out at the LEPCMAE laboratory Faculty of Chemistry, University of Science and Technology Houari Boumediene (USTHB), and Scientific and Technical Research Center in Physicochemical Analysis (CRAPC).
-
Funding: It is funded by the Ministry of Higher Education and Research (Algeria).
-
Authors’ contribution: S. Ait Hamoudi – conceptualization, S. Ait Hamoudi – data curation, J. Arrar – formal analysis, B. Hamdi – funding acquisition, M. Boucha – investigation, S. Ait Hamoudi – methodology, J. Arrar – project administration, B. Hamdi – resources, M. Brahimi – software, S. Ait Hamoudi – supervision, M. Boucha and M. Brahimi – validation, S. Ait Hamoudi – visualization, M. Boucha and M. Brahimi – writing – original draft, S. Ait Hamoudi – writing – review and editing.
-
Competing interests: There is no competing interest.
-
Data availability statement: All data generated or analyzed during this study are included in this published article.
References
[1] Mane SN, Gadalkar SM, Rathod VK. Intensification of paracetamol (acetaminophen) synthesis from hydroquinone using ultrasound. Ultrason Sonochem. 2018;49:106–10.10.1016/j.ultsonch.2018.07.029Suche in Google Scholar
[2] Żur J, Wojcieszyńska D, Hupert-Kocurek K, Marchlewicz A, Guzik U. Paracetamol – toxicity and microbial utilization. Pseudomonas moorei KB4 as a case study for exploring degradation pathway. Chemosphere. 2018;206:192–202.10.1016/j.chemosphere.2018.04.179Suche in Google Scholar
[3] Rabiet M, Togola A, Brissaud F, Seidel J-L, Budzinski H, Elbaz-Poulichet F. Consequences of treated water recycling as regards pharmaceuticals and drugs in surface and ground waters of a medium-sized mediterranean catchment. Environ Sci Technol. 2006;40:5282–8.10.1021/es060528pSuche in Google Scholar
[4] Hinson JA, Reid AB, McCullough SS, James LP. Acetaminophen‐induced hepatotoxicity: role of metabolic activation, reactive oxygen/nitrogen species, and mitochondrial permeability transition. Drug Metab Rev. 2004;36:805–22.10.1081/DMR-200033494Suche in Google Scholar
[5] van der Marel CD, Anderson BJ, van Lingen RA, Holford NHG, Pluim MAL, Jansman FGA, et al. Paracetamol and metabolite pharmacokinetics in infants. Eur J Clin Pharmacol. 2003;59:243–51.10.1007/s00228-003-0608-0Suche in Google Scholar
[6] Dalgic G, Turkdogan I, Yetilmezsoy K, Kocak E. Treatment of real paracetamol wastewater by Fenton process. Chem Ind Chem Eng Q. 2016;23:29.10.2298/CICEQ150831032DSuche in Google Scholar
[7] Al-kaf A, Naji K, Abdullah Q, Edrees W. Occurrence of paracetamol in aquatic environments and transformation by microorganisms: a review; 2017. p. 1.Suche in Google Scholar
[8] Jaeschke H, Knight TR, Bajt ML. The role of oxidant stress and reactive nitrogen species in acetaminophen hepatotoxicity. Toxicol Lett. 2003;144:279–88.10.1016/S0378-4274(03)00239-XSuche in Google Scholar
[9] Brind AM. Drugs that damage the liver. Medicine. 2007;35:26–30.10.1053/j.mpmed.2006.10.005Suche in Google Scholar
[10] Xu JJ, Hendriks BS, Zhao J, de Graaf D. Multiple effects of acetaminophen and p38 inhibitors: towards pathway toxicology. FEBS Lett. 2008;582:1276–82.10.1016/j.febslet.2008.01.063Suche in Google Scholar PubMed
[11] Mégarbane B. Intoxication par le paracétamol: mécanismes de toxicité, facteurs prédictifs et modalités de prise en charge. Toxicol Anal Clin. 2016;28:240.10.1016/j.toxac.2016.05.014Suche in Google Scholar
[12] Wise J. True risks of paracetamol may be underestimated, say researchers. BMJ Br Med J. 2015;350:h1186.10.1136/bmj.h1186Suche in Google Scholar PubMed
[13] Antonkiewicz J, Pełka R, Bik-Małodzińska M, Żukowska G, Gleń-Karolczyk K. The effect of cellulose production waste and municipal sewage sludge on biomass and heavy metal uptake by a plant mixture. Environ Sci Pollut Res. 2018;25:31101–12.10.1007/s11356-018-3109-5Suche in Google Scholar PubMed
[14] Antonkiewicz J, Baran A, Pełka R, Wisła-Świder A, Nowak E, Konieczka P. A mixture of cellulose production waste with municipal sewage as new material for an ecological management of wastes. Ecotoxicol Environ Saf. 2019;169:607–14.10.1016/j.ecoenv.2018.11.070Suche in Google Scholar PubMed
[15] Erto A, Lancia A, Bortone I, Di Nardo A, Di Natale M, Musmarra D. A procedure to design a permeable adsorptive barrier (PAB) for contaminated groundwater remediation. J Environ Manag. 2011;92:23–30.10.1016/j.jenvman.2010.07.044Suche in Google Scholar PubMed
[16] Houari M, Hamdi B, Brendle J, Bouras O, Bollinger JC, Baudu M. Dynamic sorption of ionizable organic compounds (IOCs) and xylene from water using geomaterial-modified montmorillonite. J Hazard Mater. 2007;147:738–45.10.1016/j.jhazmat.2007.01.113Suche in Google Scholar PubMed
[17] Li Q, Zhai J, Zhang W, Wang M, Zhou J. Kinetic studies of adsorption of Pb(II), Cr(III) and Cu(II) from aqueous solution by sawdust and modified peanut husk. J Hazard Mater. 2007;141:163–7.10.1016/j.jhazmat.2006.06.109Suche in Google Scholar PubMed
[18] Akpomie KG, Dawodu FA. Physicochemical analysis of automobile effluent before and after treatment with an alkaline-activated montmorillonite. J Taibah Univ Sci. 2015;9:465–76.10.1016/j.jtusci.2014.10.005Suche in Google Scholar
[19] Domínguez JR, González T, Palo P, Cuerda-Correa EM. Removal of common pharmaceuticals present in surface waters by Amberlite XAD-7 acrylic-ester-resin: influence of pH and presence of other drugs. Desalination. 2011;269:231–8.10.1016/j.desal.2010.10.065Suche in Google Scholar
[20] Fernandez ME, Nunell GV, Bonelli PR, Cukierman AL. Batch and dynamic biosorption of basic dyes from binary solutions by alkaline-treated cypress cone chips. Bioresour Technol. 2012;106:55–62.10.1016/j.biortech.2011.12.003Suche in Google Scholar PubMed
[21] Suriyanon N, Punyapalakul P, Ngamcharussrivichai C. Mechanistic study of diclofenac and carbamazepine adsorption on functionalized silica-based porous materials. Chem Eng J. 2013;214:208–18.10.1016/j.cej.2012.10.052Suche in Google Scholar
[22] Cabrera-Lafaurie WA, Román FR, Hernández-Maldonado AJ. Single and multi-component adsorption of salicylic acid, clofibric acid, carbamazepine and caffeine from water onto transition metal modified and partially calcined inorganic–organic pillared clay fixed beds. J Hazard Mater. 2015;282:174–82.10.1016/j.jhazmat.2014.03.009Suche in Google Scholar
[23] Foo KY, Hameed BH. An overview of landfill leachate treatment via activated carbon adsorption process. J Hazard Mater. 2009;171:54–60.10.1016/j.jhazmat.2009.06.038Suche in Google Scholar
[24] Erabee IK, Ahsan A, Jose B, Aziz MMA, Ng AWM, Idrus S, et al. Adsorptive treatment of landfill leachate using activated carbon modified with three different methods. KSCE J Civ Eng. 2018;22:1083–95.10.1007/s12205-017-1430-zSuche in Google Scholar
[25] Pamidimukkala PS, Soni H. Efficient removal of organic pollutants with activated carbon derived from palm shell: spectroscopic characterisation and experimental optimisation. J Environ Chem Eng. 2018;6:3135–49.10.1016/j.jece.2018.04.013Suche in Google Scholar
[26] Jeirani Z, Niu C, Soltan J. Adsorption of emerging pollutants on activated carbon. Rev Chem Eng. 2017;33(5):491–522.10.1515/revce-2016-0027Suche in Google Scholar
[27] Zhou Y, Zhang L, Cheng Z. Removal of organic pollutants from aqueous solution using agricultural wastes: a review. J Mol Liq. 2015;212:739–62.10.1016/j.molliq.2015.10.023Suche in Google Scholar
[28] Karanfil T, Dastgheib SA. Trichloroethylene adsorption by fibrous and granular activated carbons: aqueous phase, gas phase, and water vapor adsorption studies. Environ Sci Technol. 2004;38:5834–41.10.1021/es0497936Suche in Google Scholar
[29] Lian F, Chang C, Du Y, Zhu L, Xing B, Liu C. Adsorptive removal of hydrophobic organic compounds by carbonaceous adsorbents: a comparative study of waste-polymer-based, coal-based activated carbon, and carbon nanotubes. J Environ Sci. 2012;24:1549–58.10.1016/S1001-0742(11)60984-4Suche in Google Scholar
[30] Moreno-Castilla C. Adsorption of organic molecules from aqueous solutions on carbon materials. Carbon. 2004;42:83–94.10.1016/j.carbon.2003.09.022Suche in Google Scholar
[31] Li B, Lei Z, Huang Z. Surface-treated activated carbon for removal of aromatic compounds from water. Chem Eng Technol. 2009;32:763–70.10.1002/ceat.200800535Suche in Google Scholar
[32] Cotoruelo LM, Marqués MD, Leiva A, Rodríguez-Mirasol J, Cordero T. Adsorption of oxygen-containing aromatics used in petrochemical, pharmaceutical and food industries by means of lignin based active carbons. Adsorption. 2011;17:539–50.10.1007/s10450-010-9319-xSuche in Google Scholar
[33] Gauden PA, Terzyk AP, Furmaniak S, Włoch J, Kowalczyk P, Zieliński WMD. simulation of organics adsorption from aqueous solution in carbon slit-like pores. Foundations of the pore blocking effect. J Phys Condens Matter. 2013;26:055008.10.1088/0953-8984/26/5/055008Suche in Google Scholar PubMed
[34] Sellin P, Leupin O. The use of clay as an engineered barrier in radioactive-waste management – a review. Clays Clay Miner. 2014;61:477–98.10.1346/CCMN.2013.0610601Suche in Google Scholar
[35] Bildstein O, Claret F. Chapter 5 – stability of clay barriers under chemical perturbations. In: Tournassat C, Steefel CI, Bourg IC, Bergaya F, eds., Developments in clay science. Amsterdam: Elsevier; 2015. p. 155–88.10.1016/B978-0-08-100027-4.00005-XSuche in Google Scholar
[36] Collin F, Charlier R. THM behaviour of engineered and natural clay barriers. Rev Européenne de Génie Civ. 2005;9:797–808.10.1080/17747120.2005.9692784Suche in Google Scholar
[37] Abootalebi P, Siemens G. Thermal properties of engineered barriers for a Canadian deep geological repository. Can Geotech J. 2018;55:759–76.10.1139/cgj-2017-0150Suche in Google Scholar
[38] Mnasri-Ghnimi S, Frini-Srasra N. Removal of heavy metals from aqueous solutions by adsorption using single and mixed pillared clays. Appl Clay Sci. 2019;179:105151.10.1016/j.clay.2019.105151Suche in Google Scholar
[39] Ghorbel-Abid I, Trabelsi-Ayadi M. Competitive adsorption of heavy metals on local landfill clay. Arab J Chem. 2015;8:25–31.10.1016/j.arabjc.2011.02.030Suche in Google Scholar
[40] Yadav VB, Gadi R, Kalra S. Clay based nanocomposites for removal of heavy metals from water: a review. J Environ Manag. 2019;232:803–17.10.1016/j.jenvman.2018.11.120Suche in Google Scholar PubMed
[41] Abukhadra MR, Bakry BM, Adlii A, Yakout SM, El-Zaidy ME. Facile conversion of kaolinite into clay nanotubes (KNTs) of enhanced adsorption properties for toxic heavy metals (Zn2+, Cd2+, Pb2+, andCr6+) from water. J Hazard Mater. 2019;374:296–308.10.1016/j.jhazmat.2019.04.047Suche in Google Scholar PubMed
[42] Van Olphen H. An introduction to clay colloid chemistry. 2nd edn. Toronto: John-Wiley & Sons; 1977.Suche in Google Scholar
[43] Brunauer S, Emmett PH, Teller E. Adsorption of gases in multimolecular layers. J Am Chem Soc. 1938;60:309–19.10.1021/ja01269a023Suche in Google Scholar
[44] Anadão P, Pajolli ILR, Hildebrando EA, Wiebeck H. Preparation and characterization of carbon/montmorillonite composites and nanocomposites from waste bleaching sodium montmorillonite clay. Adv Powder Technol. 2014;25:926–32.10.1016/j.apt.2014.01.010Suche in Google Scholar
[45] Watts RJ, Teel AL. 9.01 – groundwater and air contamination: risk, toxicity, exposure assessment, policy, and regulation. In: Holland HD, Turekian KK. Treatise on geochemistry. Oxford: Pergamon; 2003. p. 1–16.10.1016/B0-08-043751-6/09046-0Suche in Google Scholar
[46] Muralikrishna IV, Manickam V. Chapter seventeen – hazardous waste management. In: Muralikrishna IV, Manickam V, eds., Environmental management. Woburn: Butterworth-Heinemann; 2017. p. 463–94.10.1016/B978-0-12-811989-1.00017-8Suche in Google Scholar
[47] Ferronato N, Torretta V. Waste mismanagement in developing countries: a review of global issues. Int J Environ Res Public Health. 2019;16:1060.10.3390/ijerph16061060Suche in Google Scholar
[48] Peirce JJ, Weiner RF, Vesilind PA. Chapter 15 – hazardous waste. In: Peirce JJ, Weiner RF, Vesilind PA, eds., Environmental pollution and control. 4th edn. Woburn: Butterworth-Heinemann; 1998. p. 193–210.10.1016/B978-075069899-3/50016-XSuche in Google Scholar
[49] Martin EJ, Chawla RC, Swartzbaugh JT. VI.10 – Hazardous waste site remediation technology selection. In: Twardowska I, ed., Waste management series. Amsterdam: Elsevier; 2004. p. 1019–66.10.1016/S0713-2743(04)80040-0Suche in Google Scholar
[50] Ait Hamoudi S, Hamdi B, Brendlé J, Kessaissia Z. Adsorption of lead by geomaterial matrix: adsorption equilibrium and kinetics. Sep Sci Technol. 2014;49:1416–26.10.1080/01496395.2013.879313Suche in Google Scholar
[51] Bhattacharyya KG, Sen Gupta S. Pb(II) uptake by kaolinite and montmorillonite in aqueous medium: influence of acid activation of the clays. Colloid Surf A Physicochem Eng Asp. 2006;277:191–200.10.1016/j.colsurfa.2005.11.060Suche in Google Scholar
[52] Cotoruelo LM, Marqués MD, Rodríguez-Mirasol J, Cordero T, Rodríguez JJ. Adsorption of aromatic compounds on activated carbons from lignin: kinetic study. Ind Eng Chem Res. 2007;46:2853–60.10.1021/ie061445kSuche in Google Scholar
[53] Giles CH, Smith D, Huitson A. A general treatment and classification of the solute adsorption isotherm. I. Theoretical. J Colloid Interface Sci. 1974;47:755–65.10.1016/0021-9797(74)90252-5Suche in Google Scholar
[54] Hamilton AR, Roberts M, Hutcheon GA, Gaskell EE. Formulation and antibacterial properties of clay mineral-tetracycline and -doxycycline composites. Appl Clay Sci. 2019;179:105148.10.1016/j.clay.2019.105148Suche in Google Scholar
[55] Thiebault T, Boussafir M. Adsorption mechanisms of psychoactive drugs onto montmorillonite. Colloid Interface Sci Commun. 2019;30:100183.10.1016/j.colcom.2019.100183Suche in Google Scholar
© 2021 Souhila Ait Hamoudi et al., published by De Gruyter
This work is licensed under the Creative Commons Attribution 4.0 International License.
Artikel in diesem Heft
- Regular Articles
- Qualitative and semi-quantitative assessment of anthocyanins in Tibetan hulless barley from different geographical locations by UPLC-QTOF-MS and their antioxidant capacities
- Effect of sodium chloride on the expression of genes involved in the salt tolerance of Bacillus sp. strain “SX4” isolated from salinized greenhouse soil
- GC-MS analysis of mango stem bark extracts (Mangifera indica L.), Haden variety. Possible contribution of volatile compounds to its health effects
- Influence of nanoscale-modified apatite-type calcium phosphates on the biofilm formation by pathogenic microorganisms
- Removal of paracetamol from aqueous solution by containment composites
- Investigating a human pesticide intoxication incident: The importance of robust analytical approaches
- Induction of apoptosis and cell cycle arrest by chloroform fraction of Juniperus phoenicea and chemical constituents analysis
- Recovery of γ-Fe2O3 from copper ore tailings by magnetization roasting and magnetic separation
- Effects of different extraction methods on antioxidant properties of blueberry anthocyanins
- Modeling the removal of methylene blue dye using a graphene oxide/TiO2/SiO2 nanocomposite under sunlight irradiation by intelligent system
- Antimicrobial and antioxidant activities of Cinnamomum cassia essential oil and its application in food preservation
- Full spectrum and genetic algorithm-selected spectrum-based chemometric methods for simultaneous determination of azilsartan medoxomil, chlorthalidone, and azilsartan: Development, validation, and application on commercial dosage form
- Evaluation of the performance of immunoblot and immunodot techniques used to identify autoantibodies in patients with autoimmune diseases
- Computational studies by molecular docking of some antiviral drugs with COVID-19 receptors are an approach to medication for COVID-19
- Synthesis of amides and esters containing furan rings under microwave-assisted conditions
- Simultaneous removal efficiency of H2S and CO2 by high-gravity rotating packed bed: Experiments and simulation
- Design, synthesis, and biological activities of novel thiophene, pyrimidine, pyrazole, pyridine, coumarin and isoxazole: Dydrogesterone derivatives as antitumor agents
- Content and composition analysis of polysaccharides from Blaps rynchopetera and its macrophage phagocytic activity
- A new series of 2,4-thiazolidinediones endowed with potent aldose reductase inhibitory activity
- Assessing encapsulation of curcumin in cocoliposome: In vitro study
- Rare norisodinosterol derivatives from Xenia umbellata: Isolation and anti-proliferative activity
- Comparative study of antioxidant and anticancer activities and HPTLC quantification of rutin in white radish (Raphanus sativus L.) leaves and root extracts grown in Saudi Arabia
- Comparison of adsorption properties of commercial silica and rice husk ash (RHA) silica: A study by NIR spectroscopy
- Sodium borohydride (NaBH4) as a high-capacity material for next-generation sodium-ion capacitors
- Aroma components of tobacco powder from different producing areas based on gas chromatography ion mobility spectrometry
- The effects of salinity on changes in characteristics of soils collected in a saline region of the Mekong Delta, Vietnam
- Synthesis, properties, and activity of MoVTeNbO catalysts modified by zirconia-pillared clays in oxidative dehydrogenation of ethane
- Synthesis and crystal structure of N,N′-bis(4-chlorophenyl)thiourea N,N-dimethylformamide
- Quantitative analysis of volatile compounds of four Chinese traditional liquors by SPME-GC-MS and determination of total phenolic contents and antioxidant activities
- A novel separation method of the valuable components for activated clay production wastewater
- On ve-degree- and ev-degree-based topological properties of crystallographic structure of cuprite Cu2O
- Antihyperglycemic effect and phytochemical investigation of Rubia cordifolia (Indian Madder) leaves extract
- Microsphere molecularly imprinted solid-phase extraction for diazepam analysis using itaconic acid as a monomer in propanol
- A nitric oxide-releasing prodrug promotes apoptosis in human renal carcinoma cells: Involvement of reactive oxygen species
- Machine vision-based driving and feedback scheme for digital microfluidics system
- Study on the application of a steam-foam drive profile modification technology for heavy oil reservoir development
- Ni–Ru-containing mixed oxide-based composites as precursors for ethanol steam reforming catalysts: Effect of the synthesis methods on the structural and catalytic properties
- Preparation of composite soybean straw-based materials by LDHs modifying as a solid sorbent for removal of Pb(ii) from water samples
- Synthesis and spectral characterizations of vanadyl(ii) and chromium(iii) mixed ligand complexes containing metformin drug and glycine amino acid
- In vitro evaluation of lactic acid bacteria with probiotic activity isolated from local pickled leaf mustard from Wuwei in Anhui as substitutes for chemical synthetic additives
- Utilization and simulation of innovative new binuclear Co(ii), Ni(ii), Cu(ii), and Zn(ii) diimine Schiff base complexes in sterilization and coronavirus resistance (Covid-19)
- Phosphorylation of Pit-1 by cyclin-dependent kinase 5 at serine 126 is associated with cell proliferation and poor prognosis in prolactinomas
- Molecularly imprinted membrane for transport of urea, creatinine, and vitamin B12 as a hemodialysis candidate membrane
- Optimization of Murrayafoline A ethanol extraction process from the roots of Glycosmis stenocarpa, and evaluation of its Tumorigenesis inhibition activity on Hep-G2 cells
- Highly sensitive determination of α-lipoic acid in pharmaceuticals on a boron-doped diamond electrode
- Synthesis, chemo-informatics, and anticancer evaluation of fluorophenyl-isoxazole derivatives
- In vitro and in vivo investigation of polypharmacology of propolis extract as anticancer, antibacterial, anti-inflammatory, and chemical properties
- Topological indices of bipolar fuzzy incidence graph
- Preparation of Fe3O4@SiO2–ZnO catalyst and its catalytic synthesis of rosin glycol ester
- Construction of a new luminescent Cd(ii) compound for the detection of Fe3+ and treatment of Hepatitis B
- Investigation of bovine serum albumin aggregation upon exposure to silver(i) and copper(ii) metal ions using Zetasizer
- Discoloration of methylene blue at neutral pH by heterogeneous photo-Fenton-like reactions using crystalline and amorphous iron oxides
- Optimized extraction of polyphenols from leaves of Rosemary (Rosmarinus officinalis L.) grown in Lam Dong province, Vietnam, and evaluation of their antioxidant capacity
- Synthesis of novel thiourea-/urea-benzimidazole derivatives as anticancer agents
- Potency and selectivity indices of Myristica fragrans Houtt. mace chloroform extract against non-clinical and clinical human pathogens
- Simple modifications of nicotinic, isonicotinic, and 2,6-dichloroisonicotinic acids toward new weapons against plant diseases
- Synthesis, optical and structural characterisation of ZnS nanoparticles derived from Zn(ii) dithiocarbamate complexes
- Presence of short and cyclic peptides in Acacia and Ziziphus honeys may potentiate their medicinal values
- The role of vitamin D deficiency and elevated inflammatory biomarkers as risk factors for the progression of diabetic nephropathy in patients with type 2 diabetes mellitus
- Quantitative structure–activity relationship study on prolonged anticonvulsant activity of terpene derivatives in pentylenetetrazole test
- GADD45B induced the enhancing of cell viability and proliferation in radiotherapy and increased the radioresistance of HONE1 cells
- Cannabis sativa L. chemical compositions as potential plasmodium falciparum dihydrofolate reductase-thymidinesynthase enzyme inhibitors: An in silico study for drug development
- Dynamics of λ-cyhalothrin disappearance and expression of selected P450 genes in bees depending on the ambient temperature
- Identification of synthetic cannabinoid methyl 2-{[1-(cyclohexylmethyl)-1H-indol-3-yl] formamido}-3-methylbutanoate using modern mass spectrometry and nuclear magnetic resonance techniques
- Study on the speciation of arsenic in the genuine medicinal material honeysuckle
- Two Cu(ii)-based coordination polymers: Crystal structures and treatment activity on periodontitis
- Conversion of furfuryl alcohol to ethyl levulinate in the presence of mesoporous aluminosilicate catalyst
- Review Articles
- Hsien Wu and his major contributions to the chemical era of immunology
- Overview of the major classes of new psychoactive substances, psychoactive effects, analytical determination and conformational analysis of selected illegal drugs
- An overview of persistent organic pollutants along the coastal environment of Kuwait
- Mechanism underlying sevoflurane-induced protection in cerebral ischemia–reperfusion injury
- COVID-19 and SARS-CoV-2: Everything we know so far – A comprehensive review
- Challenge of diabetes mellitus and researchers’ contributions to its control
- Advances in the design and application of transition metal oxide-based supercapacitors
- Color and composition of beauty products formulated with lemongrass essential oil: Cosmetics formulation with lemongrass essential oil
- The structural chemistry of zinc(ii) and nickel(ii) dithiocarbamate complexes
- Bioprospecting for antituberculosis natural products – A review
- Recent progress in direct urea fuel cell
- Rapid Communications
- A comparative morphological study of titanium dioxide surface layer dental implants
- Changes in the antioxidative properties of honeys during their fermentation
- Erratum
- Erratum to “Corrosion study of copper in aqueous sulfuric acid solution in the presence of (2E,5E)-2,5-dibenzylidenecyclopentanone and (2E,5E)-bis[(4-dimethylamino)benzylidene]cyclopentanone: Experimental and theoretical study”
- Erratum to “Modified TDAE petroleum plasticiser”
- Corrigendum
- Corrigendum to “A nitric oxide-releasing prodrug promotes apoptosis in human renal carcinoma cells: Involvement of reactive oxygen species”
- Special Issue on 3rd IC3PE 2020
- Visible light-responsive photocatalyst of SnO2/rGO prepared using Pometia pinnata leaf extract
- Antihyperglycemic activity of Centella asiatica (L.) Urb. leaf ethanol extract SNEDDS in zebrafish (Danio rerio)
- Selection of oil extraction process from Chlorella species of microalgae by using multi-criteria decision analysis technique for biodiesel production
- Special Issue on the 14th Joint Conference of Chemistry (14JCC)
- Synthesis and in vitro cytotoxicity evaluation of isatin-pyrrole derivatives against HepG2 cell line
- CO2 gas separation using mixed matrix membranes based on polyethersulfone/MIL-100(Al)
- Effect of synthesis and activation methods on the character of CoMo/ultrastable Y-zeolite catalysts
- Special Issue on Electrochemical Amplified Sensors
- Enhancement of graphene oxide through β-cyclodextrin composite to sensitive analysis of an antidepressant: Sulpiride
- Investigation of the spectroelectrochemical behavior of quercetin isolated from Zanthoxylum bungeanum
- An electrochemical sensor for high sensitive determination of lysozyme based on the aptamer competition approach
- An improved non-enzymatic electrochemical sensor amplified with CuO nanostructures for sensitive determination of uric acid
- Special Issue on Applied Biochemistry and Biotechnology 2020
- Fast discrimination of avocado oil for different extracted methods using headspace-gas chromatography-ion mobility spectroscopy with PCA based on volatile organic compounds
- Effect of alkali bases on the synthesis of ZnO quantum dots
- Quality evaluation of Cabernet Sauvignon wines in different vintages by 1H nuclear magnetic resonance-based metabolomics
- Special Issue on the Joint Science Congress of Materials and Polymers (ISCMP 2019)
- Diatomaceous Earth: Characterization, thermal modification, and application
- Electrochemical determination of atenolol and propranolol using a carbon paste sensor modified with natural ilmenite
- Special Issue on the Conference of Energy, Fuels, Environment 2020
- Assessment of the mercury contamination of landfilled and recovered foundry waste – a case study
- Primary energy consumption in selected EU Countries compared to global trends
- Modified TDAE petroleum plasticiser
- Use of glycerol waste in lactic acid bacteria metabolism for the production of lactic acid: State of the art in Poland
- Topical Issue on Applications of Mathematics in Chemistry
- Theoretical study of energy, inertia and nullity of phenylene and anthracene
- Banhatti, revan and hyper-indices of silicon carbide Si2C3-III[n,m]
- Topical Issue on Agriculture
- Occurrence of mycotoxins in selected agricultural and commercial products available in eastern Poland
- Special Issue on Ethnobotanical, Phytochemical and Biological Investigation of Medicinal Plants
- Acute and repeated dose 60-day oral toxicity assessment of chemically characterized Berberis hispanica Boiss. and Reut in Wistar rats
- Phytochemical profile, in vitro antioxidant, and anti-protein denaturation activities of Curcuma longa L. rhizome and leaves
- Antiplasmodial potential of Eucalyptus obliqua leaf methanolic extract against Plasmodium vivax: An in vitro study
- Prunus padus L. bark as a functional promoting component in functional herbal infusions – cyclooxygenase-2 inhibitory, antioxidant, and antimicrobial effects
- Molecular and docking studies of tetramethoxy hydroxyflavone compound from Artemisia absinthium against carcinogens found in cigarette smoke
- Special Issue on the Joint Science Congress of Materials and Polymers (ISCMP 2020)
- Preparation of cypress (Cupressus sempervirens L.) essential oil loaded poly(lactic acid) nanofibers
- Influence of mica mineral on flame retardancy and mechanical properties of intumescent flame retardant polypropylene composites
- Production and characterization of thermoplastic elastomer foams based on the styrene–ethylene–butylene–styrene (SEBS) rubber and thermoplastic material
- Special Issue on Applied Chemistry in Agriculture and Food Science
- Impact of essential oils on the development of pathogens of the Fusarium genus and germination parameters of selected crops
- Yield, volume, quality, and reduction of biotic stress influenced by titanium application in oilseed rape, winter wheat, and maize cultivations
- Influence of potato variety on polyphenol profile composition and glycoalcaloid contents of potato juice
- Carryover effect of direct-fed microbial supplementation and early weaning on the growth performance and carcass characteristics of growing Najdi lambs
- Special Issue on Applied Biochemistry and Biotechnology (ABB 2021)
- The electrochemical redox mechanism and antioxidant activity of polyphenolic compounds based on inlaid multi-walled carbon nanotubes-modified graphite electrode
- Study of an adsorption method for trace mercury based on Bacillus subtilis
- Special Issue on The 1st Malaysia International Conference on Nanotechnology & Catalysis (MICNC2021)
- Mitigating membrane biofouling in biofuel cell system – A review
- Mechanical properties of polymeric biomaterials: Modified ePTFE using gamma irradiation
Artikel in diesem Heft
- Regular Articles
- Qualitative and semi-quantitative assessment of anthocyanins in Tibetan hulless barley from different geographical locations by UPLC-QTOF-MS and their antioxidant capacities
- Effect of sodium chloride on the expression of genes involved in the salt tolerance of Bacillus sp. strain “SX4” isolated from salinized greenhouse soil
- GC-MS analysis of mango stem bark extracts (Mangifera indica L.), Haden variety. Possible contribution of volatile compounds to its health effects
- Influence of nanoscale-modified apatite-type calcium phosphates on the biofilm formation by pathogenic microorganisms
- Removal of paracetamol from aqueous solution by containment composites
- Investigating a human pesticide intoxication incident: The importance of robust analytical approaches
- Induction of apoptosis and cell cycle arrest by chloroform fraction of Juniperus phoenicea and chemical constituents analysis
- Recovery of γ-Fe2O3 from copper ore tailings by magnetization roasting and magnetic separation
- Effects of different extraction methods on antioxidant properties of blueberry anthocyanins
- Modeling the removal of methylene blue dye using a graphene oxide/TiO2/SiO2 nanocomposite under sunlight irradiation by intelligent system
- Antimicrobial and antioxidant activities of Cinnamomum cassia essential oil and its application in food preservation
- Full spectrum and genetic algorithm-selected spectrum-based chemometric methods for simultaneous determination of azilsartan medoxomil, chlorthalidone, and azilsartan: Development, validation, and application on commercial dosage form
- Evaluation of the performance of immunoblot and immunodot techniques used to identify autoantibodies in patients with autoimmune diseases
- Computational studies by molecular docking of some antiviral drugs with COVID-19 receptors are an approach to medication for COVID-19
- Synthesis of amides and esters containing furan rings under microwave-assisted conditions
- Simultaneous removal efficiency of H2S and CO2 by high-gravity rotating packed bed: Experiments and simulation
- Design, synthesis, and biological activities of novel thiophene, pyrimidine, pyrazole, pyridine, coumarin and isoxazole: Dydrogesterone derivatives as antitumor agents
- Content and composition analysis of polysaccharides from Blaps rynchopetera and its macrophage phagocytic activity
- A new series of 2,4-thiazolidinediones endowed with potent aldose reductase inhibitory activity
- Assessing encapsulation of curcumin in cocoliposome: In vitro study
- Rare norisodinosterol derivatives from Xenia umbellata: Isolation and anti-proliferative activity
- Comparative study of antioxidant and anticancer activities and HPTLC quantification of rutin in white radish (Raphanus sativus L.) leaves and root extracts grown in Saudi Arabia
- Comparison of adsorption properties of commercial silica and rice husk ash (RHA) silica: A study by NIR spectroscopy
- Sodium borohydride (NaBH4) as a high-capacity material for next-generation sodium-ion capacitors
- Aroma components of tobacco powder from different producing areas based on gas chromatography ion mobility spectrometry
- The effects of salinity on changes in characteristics of soils collected in a saline region of the Mekong Delta, Vietnam
- Synthesis, properties, and activity of MoVTeNbO catalysts modified by zirconia-pillared clays in oxidative dehydrogenation of ethane
- Synthesis and crystal structure of N,N′-bis(4-chlorophenyl)thiourea N,N-dimethylformamide
- Quantitative analysis of volatile compounds of four Chinese traditional liquors by SPME-GC-MS and determination of total phenolic contents and antioxidant activities
- A novel separation method of the valuable components for activated clay production wastewater
- On ve-degree- and ev-degree-based topological properties of crystallographic structure of cuprite Cu2O
- Antihyperglycemic effect and phytochemical investigation of Rubia cordifolia (Indian Madder) leaves extract
- Microsphere molecularly imprinted solid-phase extraction for diazepam analysis using itaconic acid as a monomer in propanol
- A nitric oxide-releasing prodrug promotes apoptosis in human renal carcinoma cells: Involvement of reactive oxygen species
- Machine vision-based driving and feedback scheme for digital microfluidics system
- Study on the application of a steam-foam drive profile modification technology for heavy oil reservoir development
- Ni–Ru-containing mixed oxide-based composites as precursors for ethanol steam reforming catalysts: Effect of the synthesis methods on the structural and catalytic properties
- Preparation of composite soybean straw-based materials by LDHs modifying as a solid sorbent for removal of Pb(ii) from water samples
- Synthesis and spectral characterizations of vanadyl(ii) and chromium(iii) mixed ligand complexes containing metformin drug and glycine amino acid
- In vitro evaluation of lactic acid bacteria with probiotic activity isolated from local pickled leaf mustard from Wuwei in Anhui as substitutes for chemical synthetic additives
- Utilization and simulation of innovative new binuclear Co(ii), Ni(ii), Cu(ii), and Zn(ii) diimine Schiff base complexes in sterilization and coronavirus resistance (Covid-19)
- Phosphorylation of Pit-1 by cyclin-dependent kinase 5 at serine 126 is associated with cell proliferation and poor prognosis in prolactinomas
- Molecularly imprinted membrane for transport of urea, creatinine, and vitamin B12 as a hemodialysis candidate membrane
- Optimization of Murrayafoline A ethanol extraction process from the roots of Glycosmis stenocarpa, and evaluation of its Tumorigenesis inhibition activity on Hep-G2 cells
- Highly sensitive determination of α-lipoic acid in pharmaceuticals on a boron-doped diamond electrode
- Synthesis, chemo-informatics, and anticancer evaluation of fluorophenyl-isoxazole derivatives
- In vitro and in vivo investigation of polypharmacology of propolis extract as anticancer, antibacterial, anti-inflammatory, and chemical properties
- Topological indices of bipolar fuzzy incidence graph
- Preparation of Fe3O4@SiO2–ZnO catalyst and its catalytic synthesis of rosin glycol ester
- Construction of a new luminescent Cd(ii) compound for the detection of Fe3+ and treatment of Hepatitis B
- Investigation of bovine serum albumin aggregation upon exposure to silver(i) and copper(ii) metal ions using Zetasizer
- Discoloration of methylene blue at neutral pH by heterogeneous photo-Fenton-like reactions using crystalline and amorphous iron oxides
- Optimized extraction of polyphenols from leaves of Rosemary (Rosmarinus officinalis L.) grown in Lam Dong province, Vietnam, and evaluation of their antioxidant capacity
- Synthesis of novel thiourea-/urea-benzimidazole derivatives as anticancer agents
- Potency and selectivity indices of Myristica fragrans Houtt. mace chloroform extract against non-clinical and clinical human pathogens
- Simple modifications of nicotinic, isonicotinic, and 2,6-dichloroisonicotinic acids toward new weapons against plant diseases
- Synthesis, optical and structural characterisation of ZnS nanoparticles derived from Zn(ii) dithiocarbamate complexes
- Presence of short and cyclic peptides in Acacia and Ziziphus honeys may potentiate their medicinal values
- The role of vitamin D deficiency and elevated inflammatory biomarkers as risk factors for the progression of diabetic nephropathy in patients with type 2 diabetes mellitus
- Quantitative structure–activity relationship study on prolonged anticonvulsant activity of terpene derivatives in pentylenetetrazole test
- GADD45B induced the enhancing of cell viability and proliferation in radiotherapy and increased the radioresistance of HONE1 cells
- Cannabis sativa L. chemical compositions as potential plasmodium falciparum dihydrofolate reductase-thymidinesynthase enzyme inhibitors: An in silico study for drug development
- Dynamics of λ-cyhalothrin disappearance and expression of selected P450 genes in bees depending on the ambient temperature
- Identification of synthetic cannabinoid methyl 2-{[1-(cyclohexylmethyl)-1H-indol-3-yl] formamido}-3-methylbutanoate using modern mass spectrometry and nuclear magnetic resonance techniques
- Study on the speciation of arsenic in the genuine medicinal material honeysuckle
- Two Cu(ii)-based coordination polymers: Crystal structures and treatment activity on periodontitis
- Conversion of furfuryl alcohol to ethyl levulinate in the presence of mesoporous aluminosilicate catalyst
- Review Articles
- Hsien Wu and his major contributions to the chemical era of immunology
- Overview of the major classes of new psychoactive substances, psychoactive effects, analytical determination and conformational analysis of selected illegal drugs
- An overview of persistent organic pollutants along the coastal environment of Kuwait
- Mechanism underlying sevoflurane-induced protection in cerebral ischemia–reperfusion injury
- COVID-19 and SARS-CoV-2: Everything we know so far – A comprehensive review
- Challenge of diabetes mellitus and researchers’ contributions to its control
- Advances in the design and application of transition metal oxide-based supercapacitors
- Color and composition of beauty products formulated with lemongrass essential oil: Cosmetics formulation with lemongrass essential oil
- The structural chemistry of zinc(ii) and nickel(ii) dithiocarbamate complexes
- Bioprospecting for antituberculosis natural products – A review
- Recent progress in direct urea fuel cell
- Rapid Communications
- A comparative morphological study of titanium dioxide surface layer dental implants
- Changes in the antioxidative properties of honeys during their fermentation
- Erratum
- Erratum to “Corrosion study of copper in aqueous sulfuric acid solution in the presence of (2E,5E)-2,5-dibenzylidenecyclopentanone and (2E,5E)-bis[(4-dimethylamino)benzylidene]cyclopentanone: Experimental and theoretical study”
- Erratum to “Modified TDAE petroleum plasticiser”
- Corrigendum
- Corrigendum to “A nitric oxide-releasing prodrug promotes apoptosis in human renal carcinoma cells: Involvement of reactive oxygen species”
- Special Issue on 3rd IC3PE 2020
- Visible light-responsive photocatalyst of SnO2/rGO prepared using Pometia pinnata leaf extract
- Antihyperglycemic activity of Centella asiatica (L.) Urb. leaf ethanol extract SNEDDS in zebrafish (Danio rerio)
- Selection of oil extraction process from Chlorella species of microalgae by using multi-criteria decision analysis technique for biodiesel production
- Special Issue on the 14th Joint Conference of Chemistry (14JCC)
- Synthesis and in vitro cytotoxicity evaluation of isatin-pyrrole derivatives against HepG2 cell line
- CO2 gas separation using mixed matrix membranes based on polyethersulfone/MIL-100(Al)
- Effect of synthesis and activation methods on the character of CoMo/ultrastable Y-zeolite catalysts
- Special Issue on Electrochemical Amplified Sensors
- Enhancement of graphene oxide through β-cyclodextrin composite to sensitive analysis of an antidepressant: Sulpiride
- Investigation of the spectroelectrochemical behavior of quercetin isolated from Zanthoxylum bungeanum
- An electrochemical sensor for high sensitive determination of lysozyme based on the aptamer competition approach
- An improved non-enzymatic electrochemical sensor amplified with CuO nanostructures for sensitive determination of uric acid
- Special Issue on Applied Biochemistry and Biotechnology 2020
- Fast discrimination of avocado oil for different extracted methods using headspace-gas chromatography-ion mobility spectroscopy with PCA based on volatile organic compounds
- Effect of alkali bases on the synthesis of ZnO quantum dots
- Quality evaluation of Cabernet Sauvignon wines in different vintages by 1H nuclear magnetic resonance-based metabolomics
- Special Issue on the Joint Science Congress of Materials and Polymers (ISCMP 2019)
- Diatomaceous Earth: Characterization, thermal modification, and application
- Electrochemical determination of atenolol and propranolol using a carbon paste sensor modified with natural ilmenite
- Special Issue on the Conference of Energy, Fuels, Environment 2020
- Assessment of the mercury contamination of landfilled and recovered foundry waste – a case study
- Primary energy consumption in selected EU Countries compared to global trends
- Modified TDAE petroleum plasticiser
- Use of glycerol waste in lactic acid bacteria metabolism for the production of lactic acid: State of the art in Poland
- Topical Issue on Applications of Mathematics in Chemistry
- Theoretical study of energy, inertia and nullity of phenylene and anthracene
- Banhatti, revan and hyper-indices of silicon carbide Si2C3-III[n,m]
- Topical Issue on Agriculture
- Occurrence of mycotoxins in selected agricultural and commercial products available in eastern Poland
- Special Issue on Ethnobotanical, Phytochemical and Biological Investigation of Medicinal Plants
- Acute and repeated dose 60-day oral toxicity assessment of chemically characterized Berberis hispanica Boiss. and Reut in Wistar rats
- Phytochemical profile, in vitro antioxidant, and anti-protein denaturation activities of Curcuma longa L. rhizome and leaves
- Antiplasmodial potential of Eucalyptus obliqua leaf methanolic extract against Plasmodium vivax: An in vitro study
- Prunus padus L. bark as a functional promoting component in functional herbal infusions – cyclooxygenase-2 inhibitory, antioxidant, and antimicrobial effects
- Molecular and docking studies of tetramethoxy hydroxyflavone compound from Artemisia absinthium against carcinogens found in cigarette smoke
- Special Issue on the Joint Science Congress of Materials and Polymers (ISCMP 2020)
- Preparation of cypress (Cupressus sempervirens L.) essential oil loaded poly(lactic acid) nanofibers
- Influence of mica mineral on flame retardancy and mechanical properties of intumescent flame retardant polypropylene composites
- Production and characterization of thermoplastic elastomer foams based on the styrene–ethylene–butylene–styrene (SEBS) rubber and thermoplastic material
- Special Issue on Applied Chemistry in Agriculture and Food Science
- Impact of essential oils on the development of pathogens of the Fusarium genus and germination parameters of selected crops
- Yield, volume, quality, and reduction of biotic stress influenced by titanium application in oilseed rape, winter wheat, and maize cultivations
- Influence of potato variety on polyphenol profile composition and glycoalcaloid contents of potato juice
- Carryover effect of direct-fed microbial supplementation and early weaning on the growth performance and carcass characteristics of growing Najdi lambs
- Special Issue on Applied Biochemistry and Biotechnology (ABB 2021)
- The electrochemical redox mechanism and antioxidant activity of polyphenolic compounds based on inlaid multi-walled carbon nanotubes-modified graphite electrode
- Study of an adsorption method for trace mercury based on Bacillus subtilis
- Special Issue on The 1st Malaysia International Conference on Nanotechnology & Catalysis (MICNC2021)
- Mitigating membrane biofouling in biofuel cell system – A review
- Mechanical properties of polymeric biomaterials: Modified ePTFE using gamma irradiation


