Acute and repeated dose 60-day oral toxicity assessment of chemically characterized Berberis hispanica Boiss. and Reut in Wistar rats
-
Loubna El Fakir
, Ahmad Mohammad Salamatullah
, Abdulhakeem Alzahrani
Abstract
Berberis hispanica Boiss. & Reut (B. hispanica) belongs to the family Berberidaceae, which is currently used in traditional medicines. This article aimed to study the phytochemical composition and acute and subacute toxicity of B. hispanica extract in rats. The phytochemical composition of B. hispanica extract was characterized using GC-MS. The acute toxicity was investigated in vivo via the oral administration of single doses including 400, 800, 1,000, 1,200, and 1,400 mg/kg for 14 days. The subchronic toxicity was studied through oral administration of 400 mg/kg for 60 days. The findings of the phytochemical analysis of B. hispanica extract showed the presence of various phytochemical compounds. Acute toxicity results revealed serious clinical symptoms and mortalities in rats treated with 800 mg/kg up to a maximum of 1,400 mg/kg. With acute toxicity, subchronic toxicity results showed also serious signs of toxicity including biochemical and histological alterations in animals treated with 1,400 mg/kg. B. hispanica extract revealed to be toxic in rats orally treated under both subacute (>400 mg/kg) and subchronic toxicity conditions (400 mg/kg). The outcome of this study serves the society as it provides toxicological evidence of B. hispanica used in alternative medicines
1 Introduction
Herbal medicines have been seen as an exhaustive source of therapeutic agents for many years ago. Nowadays, several developed drugs come from natural products or their derivatives [1]. The use of plants in traditional medicines for disease treatment goes back thousands of years ago. About 80% of people around the world use folk medicines for medication purposes. This huge use could be explained by the fact that most people based in developing countries have no access to modern medicine [2]. Moreover, 25% of prescription drugs used in the United States between 1959 and 1980 were derived from the plant kingdom [3], and about 60% of prescriptions in European countries come also directly or indirectly from plants [4]. The use of the traditional herbal practice can be explained by many reasons such as the high cost of advanced medicines, sociocultural practices of users, and the need for controlling resistant pathogens using natural alternative agents [4,5].
Nowadays, modern medicine has focused on natural products as unexhausted sources for drug production. Medicinal plants are extremely numerous, and more than 13,000 species of medicinal plants are used as traditional remedies by various cultures around the world [6,7]. The efficiency of medicine is due to secondary metabolites that vary due to environmental changes. Many secondary metabolites like polyphenols are antibiotics in the broad sense because they protect plants from fungi, bacteria, animals, and even other plants [8]. Toxic plants possess toxic substances to humans or animals, which induce serious clinical signs and even can be fatal after being ingested. The efficacy of plants versus diseases cannot exclude their potentially harmful effects on health, and therefore, any natural product should be used with precautions [9].
Berberis hispanica Boiss. & Reut (B. hispanica) is originally from Europe and the British Isles, which was introduced in North America and Mediterranean countries. It has been used as a medicinal plant for more than 2,500 years [10]. Numerous medicinal uses of B. hispanica were reported in the literature, such as analgesic, antimicrobial, antitonic, antiemetic, anti-pyretic, anti-pruritic, anti-arrhythmic, anticholinergic, anti-oxidant, antinociceptive, anti-arrhythmic, antijaundice, anticholelithiasis, antileishmaniasis, antihypertension, anticardiac arrhythmias, antidysentery, anti-inflammatory, antipruritic, anticholagogue, and antidigestive disorders [9,10,11,12,13,14].
The present study was conceptualized to study the phytochemical composition and acute and subchronic toxicity of B. hispanica used in traditional alternative medicine since no previous study has attempted this objective.
2 Materials and methods
2.1 Plant material and extract preparation
B. hispanica was collected in May 2016 from Middle Atlas (Morocco). The botanical authentication was done by Dr. Fennan and given the voucher specimen, which has been saved at the herbarium of the scientific institute # LHE.11. The bark of B. hispanica was removed, washed, dried in the shade, and then ground into fine powder. The plant extract was obtained by using maceration of 20 g of the plant powder in water for 24 h at ambient temperature. Afterward, the whole extract was undergone to filtration under reduced pressure [15,16].
2.2 Chemical characterization
Silylation (TMS) is a derivatization technique that converts nonvolatile compounds into volatile derivatives through chemical derivatization [17]. GC-MS characterization of the plant extract was performed using the Claus 580 chromatography apparatus possessing a capillary column (5% phenyl and 95% methyplisyloxane; 30.0 m × 250 μm) linked to a mass spectrometer (Polaris Q) (EI 70 eV). Helium was injected at 1 mL/min as a carrier gas. The injection volume was 1 µL, while the split was 1/75. Both injection and detection temperatures were 250 and 280°C, respectively. Furnace and regulating temperature was programmed as follows: first was set at 50 to 200°C with a rate of 11°C/min, and then from 200 to 240°C with a rate of 6°C/min. The identification of molecules was done by comparing the retention times obtained with those already used as database standards.
2.3 Animal material
Wistar albino rats weighing between 180 and 230 g were used as experimental animals for both acute and subchronic toxicity studies. Animals were acclimatized for 2 weeks under standard climatic conditions (22 ± 2°C and 12 hour light–dark cycle). Animals were fed with a chow pellet diet with free access to water. The Animal Ethics Review Committee of the Faculty of Sciences of Meknes, Morocco, reviewed and approved this study (#N° 201819K).
2.3.1 Acute toxicity
The acute toxicity study of B. hispanica was conducted according to the guidelines 423 [18]. Briefly, animals were divided into six groups each consisting of six rats (three females vs three males). Five different concentrations of the plant extract, 400, 800, 1,000, 1,200, and 1,400 mg/kg, were selected for administration into rats, and the control group received the same volume of vehicle (distilled water). Animals were studied for 14 days for recording clinical symptoms potentially induced by the plant extract. Clinical signs that occurred in mice including skin, mucous membranes, hair, eyes, respiratory, diarrhea, salivation, as well as lethargy were the most clinical symptoms noticed for treated rats under acute toxicity conditions [19]. The body weight of the treated animals was daily measured until the end of the experiment. Food and water intake alterations were also recorded for both female and male rats during the experimental period.
2.3.2 Subchronic toxicity
Subchronic toxicity of B. hispanica was assessed according to the guidelines 407 [20]. A group of 10 rats (five females vs five males) was treated with oral administration of 400 mg/kg/day of B. hispanica extract for 60 days, and the control group received the same volume of vehicle (distilled water) under subchronic toxicity conditions. Animals were controlled for clinical signs of toxicity during the whole experimental period, and the bodyweight was evaluated weekly.
2.3.2.1 Histopathological evaluation
At the end of the experiment, all animals were euthanized and subjected to cerebral dislocation. The kidney, liver, and lungs were excised to calculate the relative weight of organs.
The vital organs (kidney, lungs, and liver) were fixed in a 10% neutral formalin buffer for 24 h and then converted into paraffin cups using a rotary automaton. The tissue samples were subjected to the following process: dehydration into progressively increasing alcohol gradient, xylene cleaning, and immersion in wax. Afterward, the samples of organs were embedded in paraffin before thick sections being cut using a rotary microtome [21].
2.3.2.1.1 Hematoxylin and eosin staining
Histological sections were dewaxed in xylene and then rehydrated in decreasing concentrations of alcohol and then water. The slices were stained in hematoxylin for 5 min and washed with distilled water. Afterward, they were differentiated in 1% alcohol for 5 s and washed with tap water. The cups were colored in eosin for 10 min, washed with distilled water for 5 min, and then dehydrated in an increasing alcohol gradient. Finally, they were cleaned with xylene and covered with DPX support (a mixture of stilbene, tricresyl phosphate, and xylene) [22].
2.3.2.2 Analysis of serum biochemistry
The serum analysis was performed for rats treated under both acute and subacute toxicity conditions. At the end of the experiment, all survived rats were euthanized and then subjected to cerebral dislocation for blood collection using heparin tubes. Afterward, the recovered blood was centrifuged at 2,500 tr/min for 15 min, and the obtained serum was used to perform the analysis of AST, ALT, urea, creatinine, and blood sugar using an automated analyzer according to the earlier protocol [19].
2.4 Statistical analysis
Data were expressed as mean ± SD2 using BioStat 2009 Professional 5.8.4 software. Student’s t-test was used to perform analysis, and the values were statically considered at (p ≤ 0.05)
-
Ethical approval: This wok was approved by the Animal Ethical Committee of the Faculty of Sciences of Meknes, Morocco # N° 201819 K.
3 Results
3.1 Chemical analysis of B. hispanica extract
The findings of chemical compounds detected in B. hispanica extract by GC-MS showed the presence of 13 compounds that majorly constituted of glucaric acid whose area percentage is 11.823 (Figure 1 and Table 1).
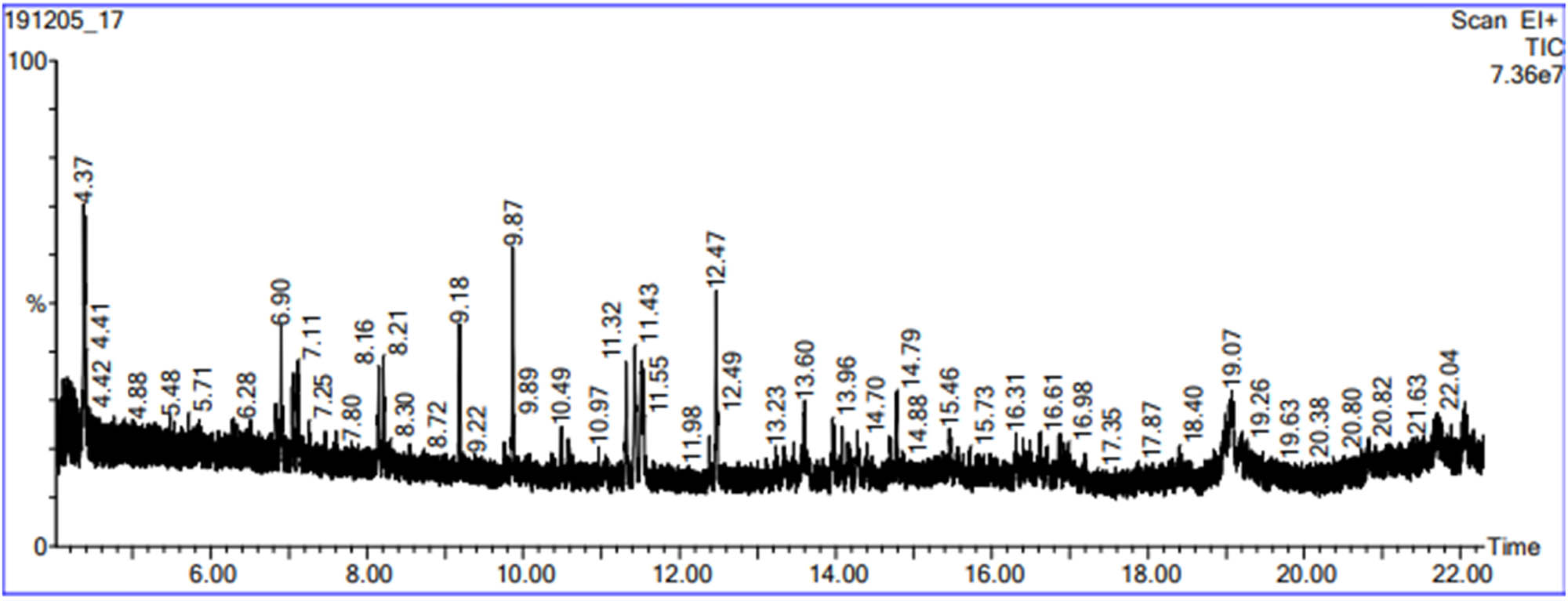
Chromatographic profile of B. hispanica bark extract.
Phytochemical compounds identified in B. hispanica bark extract
| Retention time | Molecular weight (g/mol) | Compound name | Structural formula |
|---|---|---|---|
| 4.371 | 126.24 | 2,4-Dimethyl-1-heptene | C9H18 |
| 6.90 | 184.36 | Decane, 2,5,6-trimethyl- | C13H28 |
| 7.71 | 286.41 | Oxalic acid, 2-ethylhexyl isohexyl ester | C16H30O4 |
| 8.16 | 196.37 | 6-Tridecene, 7-methyl- | C14H28 |
| 9.18 | 198.39 | Tetradecane | C14H30 |
| 9.87 | 128 | Nonane | C9H20 |
| 11.32 | 200.36 | 11-Methyldodecanol | C13H28O |
| 11.55 | 168.32 | 1-Nonene, 4,6,8-trimethyl- | C12H24 |
| 12.47 | 198.39 | Tetradecane, 7,8-dimethyl 7,8-dimethyltetradecane | C16H34 |
| 13.60 | 577.2 | 3-Isopropoxy-1,1,1,7,7,7-hexamethyl-3,5,5-tris(trimethylsiloxy)tetrasiloxane | C18H52O7Si7 |
| 14.79 | 184.36 | Undecane, 3,7-dimethyl- | C13H28 |
| 19.07 | 441.4 | Folic acid | C19H19N7O6 |
| 22.04 | 252.35 | Tertbutyloxyformamide, N-methyl-N-[4-(1-pyrrolidinyl)-2-butynyl]- | C14H24N2O2 |
3.2 Acute toxicity
No symptoms occurred in rats treated with 400 mg/kg of B. hispanica extract under acute toxicity conditions. However, animals treated with 1,000, 1,200, and 1,400 mg/kg of the plant extract showed serious clinical symptoms in a dose-dependent manner. Animals that were given oral administration of 1,400 mg/kg developed serious toxic symptoms like sedation and colored urine that could be due to inefficiency of renal elimination of the plant extract. Animals treated with the highest tested dose (1,400 mg/kg) also showed a dyspnea compared to the control group (Table 2). LD50 (lethal dose 50%) of B. hispanica extract was determined in 1016.16 mg/kg.
Clinical symptoms occurred in animals treated with B. hispanica extracts under acute toxicity conditions
| B. hispanica extract (mg/kg) | Number of rats treated | Mortality rate by sex | Total mortality rate | Clinical symptoms |
|---|---|---|---|---|
| 0 | 6 | 0/3 ♀ | 0 | No symptoms |
| 0/3 ♂ | ||||
| 400 | 6 | 0/3 ♀ | 0 | No symptoms |
| 0/3 ♂ | ||||
| 800 | 6 | 0/3 ♀ | 17 | Tachycardia, labored breathing, disorder, and loss of balance |
| 1/3 ♂ | ||||
| 1,000 | 6 | 1/3 ♀ | 50 | Sedation, movement disorder, unconscious, fast heartbeat, labored breathing |
| 2/3 ♂ | ||||
| 1,200 | 6 | 3/3 ♀ | 67 | Paralysis sedation |
| 1/3 ♂ | ||||
| 1,400 | 6 | 3/3 ♀ | 100 | Convulsion, total paralysis, coma, death |
| 3/3♂ |
3.2.1 Food and water intake
Rats treated (males and females) with doses of 400 and 800 mg/kg of the plant extract under subacute toxicity conditions revealed no significant variations in water and food intake when compared to the untreated group (p > 0.05). However, a significant decrease in food and water intake was observed for animals treated with 1,000, 1,200, and 1,400 mg/kg when compared to the untreated group (p < 0.05; Figures 2 and 3).
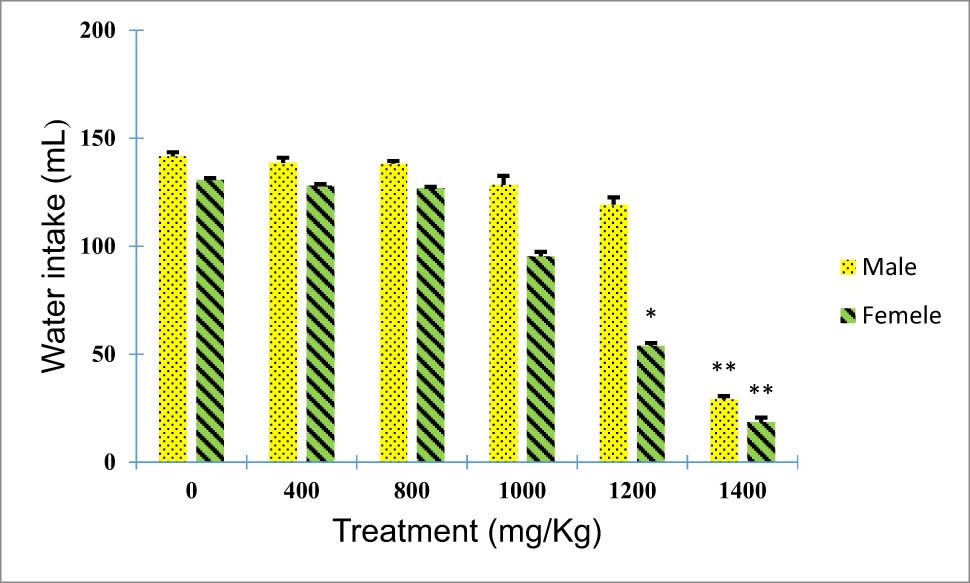
Effect of B. hispanica extracts on water intake (mL/100 g bodyweight) of animals treated with a dose up to 14,000 mg/kg.
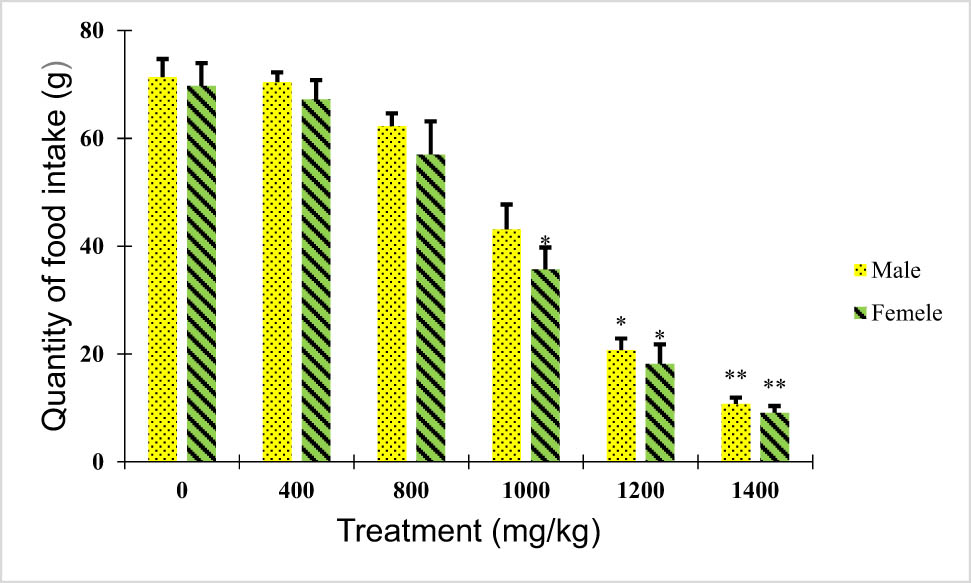
Effect of B. hispanica extracts on food intake (g/100 g bodyweight) of animals treated with a dose up to 14,000 mg/kg.
3.2.2 Bodyweight
During the whole period of dosing, acute toxicity findings showed a decrease in the bodyweight of animals (males and females) treated with a dose up to 1,400 mg/kg (Figures 4 and 5). Alteration of bodyweight of treated rats started progressively being significant from the eighth day of treatment to reach a maximum at the end of the experiment when compared to the untreated rats (p < 0.05). Measuring bodyweight for female rats treated with the highest doses 1,200 and 1,400 mg/kg was interrupted due to the death of rats before the end of the experiment (Figures 4 and 5).
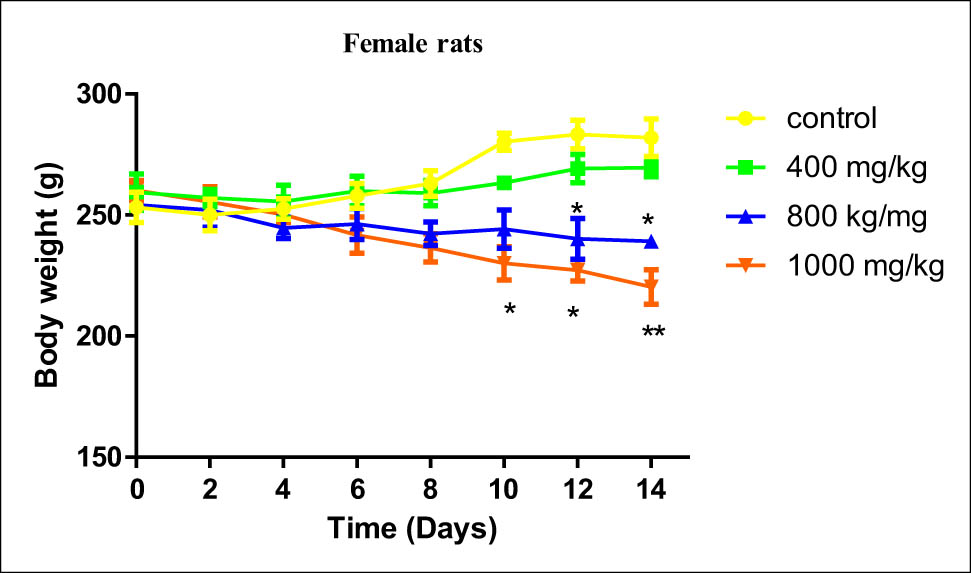
Effect of B. hispanica extracts on the bodyweight of female rats treated with a dose up to 14,000 mg/kg.
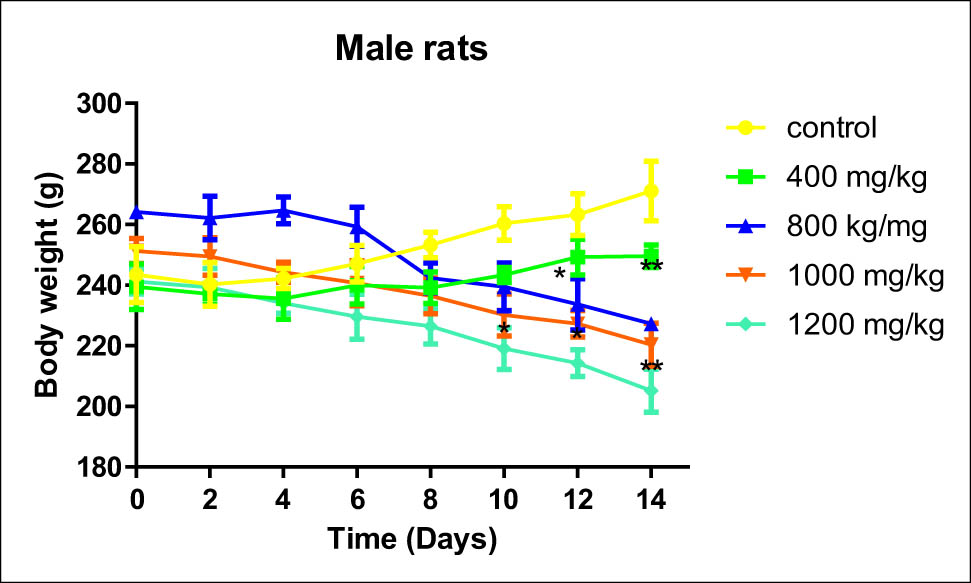
Effect of B. hispanica extracts on the bodyweight of male rats treated with a dose up to 14,000 mg/kg.
3.2.3 Serum biochemistry analysis
Regarding serum biochemistry analysis for glucose, creatinine, AST, and ALT at the end of the experiment, the results obtained showed only significant increase in creatinine in blood collected from female rats treated orally with the highest dose (1,400 mg/kg) compared to the control group (*p < 0.05) (Table 3). Otherwise, there were no significant changes in blood collected from treated animals (males and females) with a dose up to 1,400 mg/kg compared to the untreated group (p > 0.05; Tables 3 and 4).
Effect of B. hispanica extract on biochemical parameters in female rats under acute toxicity conditions
| Parameters | Control | B. hispanica extract (mg/kg) | ||||
|---|---|---|---|---|---|---|
| 400 | 800 | 1,000 | 1,200 | 1,400 | ||
| Glucose (mg/dL) | 71.04 ± 1.72 | 74.25 ± 2.24 | 64.13 ± 2.12 | 73.54 ± 2.25 | 70.19 ± 5.64 | 68.34 ± 8.23 |
| Creatinine (mg/dL) | 0.82 ± 0.05 | 0.78 ± 0.05 | 0.85 ± 0.10 | 0.79 ± 0.12 | 0.44 ± 0.05 | 1.19 * ± 0.25 |
| AST (U/L) | 182.4 ± 4.55 | 177.33 ± 5.47 | 179.6 ± 17.46 | 188.25 ± 3.90 | 185.31 ± 4.85 | 189.01 ± 5.24 |
| ALT (U/L) | 49.4 ± 1.78 | 50.25 ± 6.31 | 54.27 ± 7.53 | 51.02 ± 2.43 | 55.75 ± 4.76 | 46.52 ± 5.17 |
*Difference was significant.
Effect of B. hispanica extract on biochemical parameters in male rats under acute toxicity conditions
| Parameters | Control | B. hispanica extract (mg/kg) | ||||
|---|---|---|---|---|---|---|
| 200 | 400 | 600 | 800 | 1,000 | ||
| Glucose (mg/dL) | 69.25 ± 3.34 | 62.98 ± 1.47 | 71.45 ± 4.32 | 81.15 ± 3.25 | 76.61 ± 1.46 | 73.11 ± 6.83 |
| Creatinine (mg/dL) | 1.14 ± 1.14 | 0.91 ± 0.25 | 1.01 ± 0.13 | 1.10 ± 0.04 | 1.09 ± 0.54 | 0.88 ± 0.02 |
| AST (U/L) | 110.75 ± 4.58 | 104.47 ± 3.25 | 112.73 ± 3.84 | 101.95 ± 3.45 | 114.07 ± 3.96 | 99.21 ± 4.09 |
| ALT (U/L) | 52.50 ± 3.08 | 49.10 ± 4.63 | 51.32 ± 8.05 | 55.02 ± 2.43 | 46.52 ± 6.17 | 55.75 ± 6.47 |
3.3 Subchronic toxicity
Animals that received 400 mg/kg of B. hispanica extract orally under subchronic toxicity conditions showed serious clinical symptoms that adversely affected the animal bodyweight, which was progressively decreased throughout dosing when compared to the untreated group. There was a significant decrease in the bodyweight of the treated male rats from the first treatment days to reach a maximum at the end of the experiment compared to the untreated male rats (p < 0.05). There was a significant decrease in the bodyweight of the treated female rats from the 25th day of treatment more than ever before (p < 0.05; Figures 6 and 7). The findings obtained showed also a remarkable increase in the relative weight of organs (kidney, liver, stomach, and lungs) recovered from the treated animals when compared to the nontreated group (p ≤ 0.05; Figures 8 and 9).
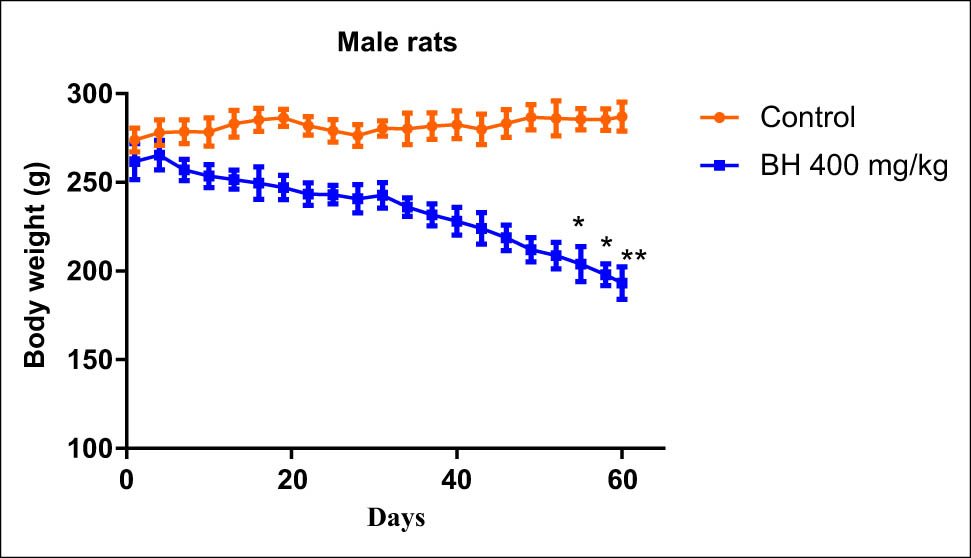
Effect of B. hispanica extracts on the bodyweight of the treated male rats under subchronic toxicity conditions.
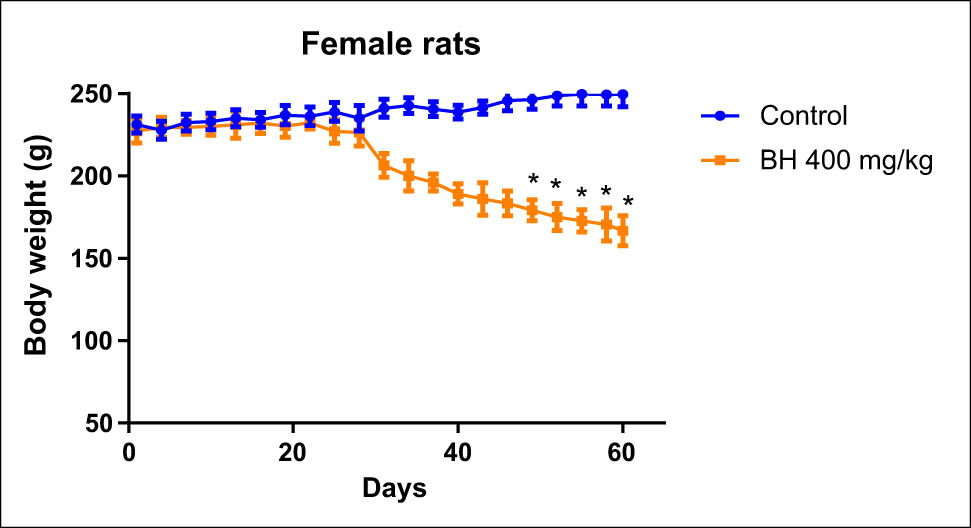
Effect of B. hispanica extracts on the bodyweight of the treated female rats under subchronic toxicity conditions.
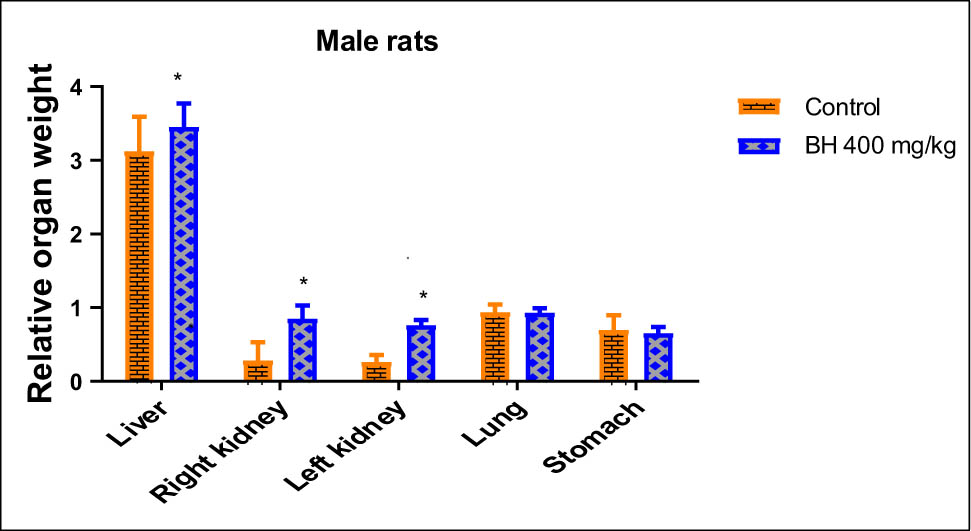
Effect of B. hispanica extract on the relative weight of organs (kidney, liver, stomach, and lungs) of the treated male rats under subchronic toxicity conditions.
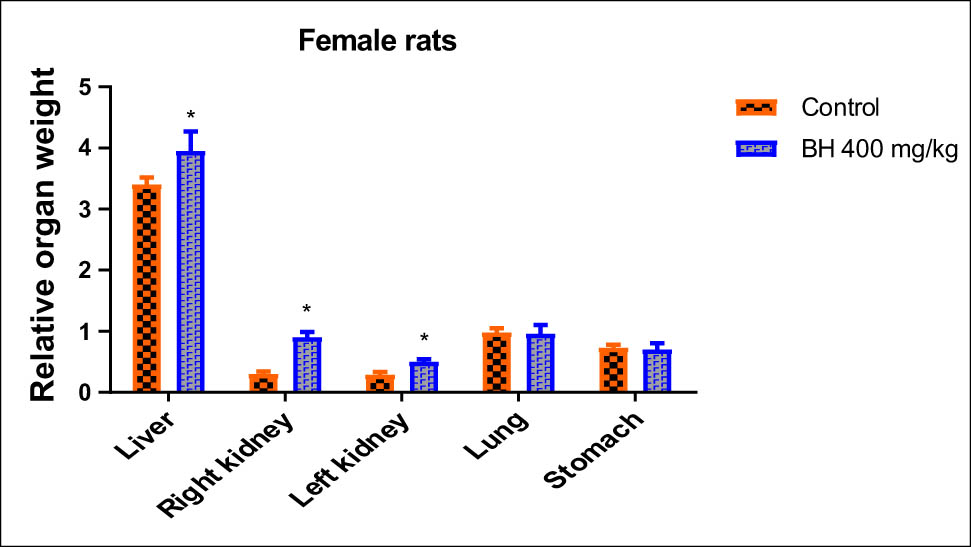
Effect of B. hispanica extract on the relative weight of organs (kidney, liver, stomach, and lungs) of the treated female rats under subchronic toxicity conditions.
3.3.1 Effect of B. hispanica extract on biochemical parameters
The findings of serum analysis showed a significant increase in the activity of plasma transaminase (ALT and AST) derived from the liver of both treated male and female rats when compared to the nontreated rats (p** < 0.01). However, the concentration of creatinine, urea, and blood sugar was not adversely affected when compared to the control rats (p* > 0.05; Table 5).
Effect of B. hispanica extract on biochemical parameters
| Biochemical parameters | Control | Treated rats (400 mg/kg) |
|---|---|---|
| Females | ||
| AST (U/L) | 103.5 ± 2.2 | 152.20 ± 5.4* |
| ALT (U/L) | 43.60 ± 1.1 | 93.75 ± 3.3* |
| Urea (mg/dL) | 63.96 ± 2.1 | 67.96 ± 3.21 |
| Blood sugar (g/L) | 1.18 ± 0.1 | 0.91 ± 0.1 |
| Creatinine (µmol/L) | 0.60 ± 0.12 | 0.83 ± 1.2 |
| Males | ||
| AST (U/L) | 131.25 ± 8.7 | 212.75 ± 5.4* |
| ALT (U/L) | 76.75 ± 4.4 | 123.75 ± 6.12** |
| Urea (mg/dL) | 58.06 ± 4.16 | 62.14 ± 6.13 |
| Blood sugar (g/L) | 1.02 ± 0.03 | 0.97 ± 0.07 |
| Creatinine (µmol/L) | 1.02 ± 0.8 | 1.36 ± 0.7 |
*Difference was highly significant.
**Difference was extremely significant.
3.3.2 Effect of B. hispanica extract on the internal organs
The results of the histological examination showed serious modifications in the tissues of liver, kidneys, and lungs in treated rats with B. hispanica extract (400 mg/kg/day) as follows: Regarding kidney parenchyma of animals treated with B. hispanica extract, serious histopathological injuries such as inflammation and renal and vascular congestion were detected (Figure 10d). The most histopathological modifications were found in the tissues of lungs, emphysema, and inflammation (Figure 10e). Moreover, the liver of the treated rats (Figure 10f) was also found to have degenerative alterations, sinusoidal and veins dilatation, vascular congestion, hemorrhagic foci, steatosis, and early periportal fibrosis.
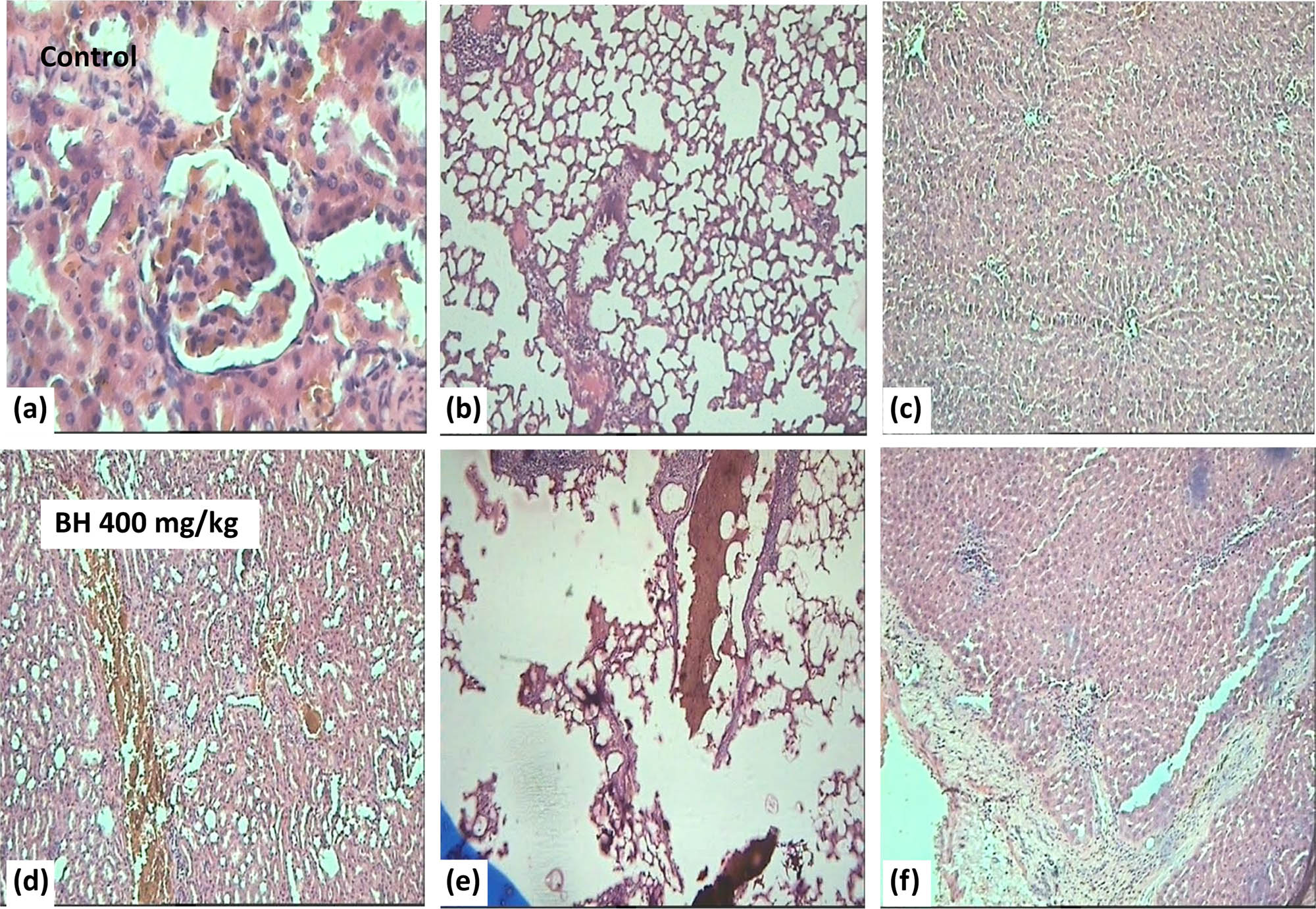
Effect of B. hispanica extracts on the internal organs (kidney, lungs, and liver) of animals treated with 400 mg/kg/day. (a) Sections of kidney parenchyma of untreated rats; (d) sections of kidney parenchyma of treated rats; (b) sections of lung parenchyma of untreated rats; (e) sections of lung parenchyma of treated rats; (c) sections of liver parenchyma of untreated rats; (f) sections of liver parenchyma of treated rats.
4 Discussion
Herbal medicine has been used in the treatment of diseases many years ago. The Moroccan flora has largely contributed to fighting diseases that occurred in the local population and overall the neighboring countries [23]. Plants used for medication without scientific validity could negatively affect the health of the users [24]. Hence, it is important to pay attention to plants used in the prevention and treatment of diseases or used as food.
This study was conducted to study the acute and subchronic toxicity of B. hispanica bark extract used in Morocco pharmacopeia. Under subchronic toxicity conditions, animals treated with B. hispanica extract with a dose up to 1,400 mg/kg showed serious clinical symptoms that could be attributed to toxic properties induced by the extract administration [19]. The dose of B. hispanica bark causing mortality in 50 percent of animals (LD50) was estimated at 1016.16 ± 224.57 mg/kg, and therefore, the plant extract is considered slightly toxic since 1016.16 ± 224.57 mg/kg falls within the scope 500–5,000 mg/kg [25]. Under subchronic toxicity conditions, it came to our attention that male rats were more sensible to plant extract toxicity compared to female rats. This observational remark was consistent with the previously reported findings, which showed that males have a longer gastric retention time that delays food absorption and, therefore, increases adverse drug reactions [26].
Changes in food and water intake were frequently used as indicators for the assessment of the general health status of animal experimentation [27]. Under subacute toxicity conditions, a decrease in water and food intake of animals (males and females) treated with an increasing dose up to 1,400 mg/kg was observed during the whole period of dosing, and therefore, we suggest that animals were negatively affected by the plant extract. A decrease in both food and water intake was observed for treated animals due to the deregulation of appetite under the plant extract effect.
Under acute and subchronic toxicity conditions, a remarkable decrease in the bodyweight of animals (males and females) treated with an increasing plant extract dose up to 1,400 mg/kg was observed. This result can be naturally explained by decreasing the food and water intake in animals treated with the plant extract. This sign of toxicity can also result from the deregulation of appetite. These findings were closely in accordance with the previously reported literature, which revealed a loss of animal weight due to drug-induced toxicity [28].
The analysis of biochemical parameters for treated animals provided data on plant extract-induced toxicity under acute and subchronic toxicity conditions. ALT and AST are enzymes found normally in the liver; however, when hepatic cells got damaged under diseases or toxic agents, these enzymes are discharged into surrounding extracellular space [29]. Under acute toxicity conditions, significant increase in only creatinine was observed in the blood collected from female rats treated orally with the highest tested dose (1,400 mg/kg), and therefore, we confirm that the tested extract has little toxicity vs kidneys when ingested in a single dose up to 1,400 mg/Kg. Under subchronic toxicity conditions, the plasmatic activities of ALT and AST dosed in recovered plasma of treated rats with extract were higher than those of the nontreated group (p ≤ 0.05), and consequently, the extract studied revealed to be drug-induced liver injury. The potential extract-induced kidney damage was also assessed by measuring the concentration of plasma urea and plasma creatinine, and the results revealed no significant variation when compared to the untreated group (p > 05). The results obtained were in agreement with earlier results, which showed the elevation of plasma creatinine and plasma urea concentration under chemical-induced kidney injury [19].
Risk assessment of B. hispanica bark extract in rats under chronic toxicity conditions was also investigated by analyzing the internal organ tissues such as liver, kidney, and lungs. Moderate clinical injuries were recorded in the kidney and lungs; meanwhile, the histopathological changes were more pronounced in the liver. These findings were in confirmation with those obtained from serum analyses since no changes were reported in urea and creatinine; however, serious elevation was detected in transaminase activities controlled by the liver. The major histopathological changes revealed in the liver of rats treated with 400 mg/kg/day were degenerative alteration, sinusoidal and veins dilatation, vascular congestion, hemorrhagic foci, and steatosis. These findings were consistent with those reported in the earlier data, which showed similar histological modifications that occurred in the internal organs of treated rats with Berberis vulgaris [30]. Kidneys of animals treated with B. hispanica extract showed inflammation and renal and vascular congestion, and therefore, these symptoms could indicate extract-induced nephrotoxicity as reported in the literature [19]. Toxicities occoured under both subacute and subchronic toxicity conditions in rats is mainly attributed to compounds detected in B. hispanica bark extract. These compounds can react individually or in synergy without excluding potential potentiation effects.
The genus Berberis belongs to the family Berberidaceae including more than 500 species [11]. The principal isolated compounds from Berberis were reported to be phenols, tannins, triterpenes, sterols, and alkaloids [29,30,31,32]. Alkaloids contained in Berberis are revealed to be toxic in mice under subchronic toxicity by inducing the increase of plasma activity of ALT and AST, which are liberated by damaged cells of the liver [32,33]. Hence, closer attention should be paid to plants used in traditional medicines for therapeutic purposes.
5 Conclusion
This study can provide more data on the phytochemical composition and in vivo toxicity of B. hispanica. The outcome of this study showed that B. hispanica extract revealed to be toxic in rats that received an oral dose higher than 400 mg/kg under subchronic toxicity conditions. The studied extract showed also serious toxicities in rats that received 400 mg/kg under subchronic toxicity conditions. Consequently, particular attention should be paid to the scientific validity of plants used in traditional medicines.
Acknowledgments
The authors extend their appreciation to the Deanship of Scientific Research at King Saud University for funding this work through research group no (RG-1441-360).
-
Funding information: This work is funded by the Deanship of Scientific Research at King Saud University via through research group no (RG-1441-360)
-
Author contributions: L. E. F. and M. B.: writing the original draft. S. G., and L. B.: formal analysis. A. M. S., A. A., R. U., E. E., G. A. M., A. B.: funding acquisition, reviewing, and editing; T. A.: supervision and data validation. A. Z.: visualization and data validation.
-
Conflict of interest: The authors declare that they have no conflicts of interest.
-
Data availability statement: The data used to support the findings of this study are available from the corresponding author upon request
-
Informed consent statement: Not applicable.
References
[1] Ullah R, Alqahtani AS, Noman OM, Alqahtani AM, Ibenmoussa S, Bourhia M. A review on ethno-medicinal plants used in traditional medicine in the Kingdom of Saudi Arabia. Saudi J Biol Sci. 2020;27:2706–18. 10.1016/j.sjbs.2020.06.020.Search in Google Scholar
[2] Ma WG, Tan RX, Fuzzati N, Li QS, Wolfender J-L, Hostettmann K. Wahlenbergioside, a phenylpropanoid glucoside from Wahlenbergia marginata. Phytochemistry. 1997;45(2):411–5. 10.1016/S0031-9422(96)00849-7.Search in Google Scholar
[3] Farnsworth NR. Screening plants for new medicines. Biodiversity. 1988;15(3):81–99.Search in Google Scholar
[4] Rao MR, Palada MC, Becker BN. Medicinal and aromatic plants in agroforestry systems. New vistas in agroforestry. Florida, USA: Springer; 2004. p. 107–22.10.1007/978-94-017-2424-1_8Search in Google Scholar
[5] Duke JA, Atchley AA. Proximate analysis. Boca Raton, FL: Handb Plant Sci Agric CRC Press Inc; 1984. p. 145–9.Search in Google Scholar
[6] Cox PA, Balick MJ. The ethnobotanical approach to drug discovery. Sci Am. 1994;270(6):82–7.10.1038/scientificamerican0694-82Search in Google Scholar
[7] Foster S. Tyler’s honest herbal: a sensible guide to the use of herbs and related remedies. New York, USA: Routledge; 1999.10.4324/9780203819692Search in Google Scholar
[8] Buchanan B, Gruissem W, Jones R. Biochemistry and molecular biology of plants, american society of plant physiologists. Waldorf MD: Courier Companies. Inc; 2000.Search in Google Scholar
[9] Fournier P. Les quatre flores de la France, Corse comprise:(generale, alpine, mediterraneenne, littorale)-v. 1: Texte.-v. 2: Atlas-2. 1977Search in Google Scholar
[10] Valdés B, Rejdali M, Achhal El Kadmiri A, Jury JL, Montserrat JM. (editors). Catalogue des plantes vasculaires du Nord du Maroc, incluant des cles d'identification. Checklist of vascular plants of N Morocco with identification keys. 2 volumes. Madrid: CSIC; 2002.Search in Google Scholar
[11] Rounsaville TJ, Ranney TG. Ploidy levels and genome sizes of Berberis L. and Mahonia Nutt. species, hybrids, and cultivars. HortScience. 2010;45(7):1029–33. 10.21273/HORTSCI.45.7.1029.Search in Google Scholar
[12] Mozaffarian V. Dict Iran plant names. Tehran: Farhang-e-Moaser; 2008.Search in Google Scholar
[13] Kafi M, Balandri A. Effects of gibberellic acid and ethephon on fruit characteristics and ease of harvest seed less barberry. Iran Res Organ Sci Technol Cent Khorasan. 1995.Search in Google Scholar
[14] El Youbi AEH, Bousta D, Ouahidi I, Aarab L. Effets antidépresseurs, antinociceptifs et immunomodulateurs des extraits aqueux et protéique de Berberishispanica Boiss. & Reut. du Maroc. Phytothérapie. 2011;9(1):25–32.10.1007/s10298-010-0606-1Search in Google Scholar
[15] Consortium LF-N. Understanding local Mediterranean diets: a multidisciplinary pharmacological and ethnobotanical approach. Pharmacol Res. 2005;52(4):353–66. 10.1016/j.phrs.2005.06.005.Search in Google Scholar PubMed
[16] Kpemissi M, Eklu-Gadegbeku K, Veerapur VP, Potârniche A-V, Adi K, Vijayakumar S, et al. Antioxidant and nephroprotection activities of Combretum micranthum: A phytochemical, in-vitro and ex-vivo studies. Heliyon. 2019;5(3):e01365. 10.1016/j.heliyon.2019.e01365.Search in Google Scholar PubMed PubMed Central
[17] Kabran GR, Mamyrbekova-Bekro JA, Pirat J-L, Bekro Y-A, Sommerer N, Verbaere A, Meudec E. Identification de composés phénoliques extraits de deux plantes de la pharmacopée ivoirienne. Journal de la Société Ouest-Africaine de Chimie. 2014;57-63.Search in Google Scholar
[18] OECD. Acute oral toxicity – fixed dose procedure (chptr). Villanova, USA: Oecd Guidel Test Chem; 2001 December. p. 1–14.Search in Google Scholar
[19] Bourhia M, Haj Said AA, Chaanoun A, El Gueddari F, Naamane A, Benbacer L, et al. Phytochemical screening and toxicological study of Aristolochia baetica linn roots: histopathological and biochemical evidence. J Toxicol. 2019;2019:2019. 10.1155/2019/8203832.Search in Google Scholar PubMed PubMed Central
[20] Oecd. OECD guidelines for the testing of chemicals. Fisheries Supply, USA: Organization for Economic; 1994.Search in Google Scholar
[21] Bourhia M, Bari A, Ali SS, Benbacer L. Phytochemistry and toxicological assessment of Bryonia dioica roots used in north-African alternative medicine. Open Chem. 2019;17(1):1403–11. 10.1515/chem-2019-0150.Search in Google Scholar
[22] Bourhia M, Lahmadi A, Achtak H, Touis A, Elbrahmi J, Ullah R, et al. Phytochemical analysis and toxicity study of Aristolochia paucinervis rhizomes decoction used in Moroccan alternative medicine: histopathological and biochemical profiles. Evid Based Compl Altern Med. 2019;2019:1398404. 10.1155/2019/1398404.Search in Google Scholar
[23] Makrane H, El Messaoudi M, Melhaoui A, El Mzibri M, Benbacer L, Aziz M. Cytotoxicity of the aqueous extract and organic fractions from Origanum majorana on human breast cell line MDA-MB-231 and human colon cell line HT-29. Adv Pharmacol Sci. 2018;2018:3297193.10.1155/2018/3297193Search in Google Scholar
[24] Makrane H, El Messaoudi M, Melhaoui A, El Mzibri M, Benbacer L, Aziz M. Étude de prévalence des effets indésirables liés à l’utilisation des plantes médicinales par les patients de l’Institut National d’Oncologie. Rabat Phytothérapie. 2014;12(1):25–32. 10.1155/2018/3297193.Search in Google Scholar
[25] Diezi J. Toxicologie: principes de base et répercussions cliniques dans Pharmacologie: des concepts fondamentaux aux applications thérapeutiques. Constantine, Algeria: Frisson-Roche; 1989.Search in Google Scholar
[26] Anderson GD. Chapter 1 gender differences in pharmacological response. Vol. 83. Int Rev Neurobiol. Washington, USA: Academic Press; 2008. p. 1–10. 10.1016/S0074-7742(08)00001-9.Search in Google Scholar
[27] El Hilaly J, Israili ZH, Lyoussi B. Acute and chronic toxicological studies of Ajuga iva in experimental animals. J Ethnopharmacol. 2004;91(1):43–50. 10.1016/j.jep.2003.11.009.Search in Google Scholar PubMed
[28] Raza M, Al-Shabanah OA, El-Hadiyah TM, Al-Majed AA. Effect of prolonged vigabatrin treatment on hematological and biochemical parameters in plasma, liver and kidney of Swiss albino mice. Sci Pharm. 2002;70(2):135–45. 10.3797/scipharm.aut-02-16.Search in Google Scholar
[29] Rahman MF, Siddiqui MK, Jamil K. Effects of Vepacide (Azadirachta indica) on asp artate and al anine aminotransferase profiles in a subchronic study with rats. Hum Exp Toxicol. 2001;20(5):243–9. 10.1191/096032701678227730.Search in Google Scholar PubMed
[30] Ahmed M, Azmat A. Acute toxicity (lethal dose 50 calculation) and histopathological effects of methanolic extract of Berberis vulgaris in mice. World J Pharmacol Res. 2014;3:1439–48.Search in Google Scholar
[31] Mokhber-Dezfuli N, Saeidnia S, Gohari AR, Kurepaz-Mahmoodabadi M. Phytochemistry and pharmacology of berberis species. Pharmacogn Rev. 2014;8(15):8–15. 10.4103/0973-7847.125517.Search in Google Scholar PubMed PubMed Central
[32] Rad SZK, Rameshrad M, Hosseinzadeh H. Toxicology effects of Berberis vulgaris (barberry) and its active constituent, berberine: a review. Iran J Basic Med Sci. 2017;20(5):516–29. 10.22038/IJBMS.2017.8676.Search in Google Scholar PubMed PubMed Central
[33] Ning N, Wang YZ, Zou ZY, Li XG. Pharmacological and safety evaluation of fibrous root of Rhizoma Coptidis. Environ Toxicol Pharmacol. 2015;39(1):53–69. 10.1016/j.etap.2014.11.006.Search in Google Scholar PubMed
© 2021 Loubna El Fakir et al., published by De Gruyter
This work is licensed under the Creative Commons Attribution 4.0 International License.
Articles in the same Issue
- Regular Articles
- Qualitative and semi-quantitative assessment of anthocyanins in Tibetan hulless barley from different geographical locations by UPLC-QTOF-MS and their antioxidant capacities
- Effect of sodium chloride on the expression of genes involved in the salt tolerance of Bacillus sp. strain “SX4” isolated from salinized greenhouse soil
- GC-MS analysis of mango stem bark extracts (Mangifera indica L.), Haden variety. Possible contribution of volatile compounds to its health effects
- Influence of nanoscale-modified apatite-type calcium phosphates on the biofilm formation by pathogenic microorganisms
- Removal of paracetamol from aqueous solution by containment composites
- Investigating a human pesticide intoxication incident: The importance of robust analytical approaches
- Induction of apoptosis and cell cycle arrest by chloroform fraction of Juniperus phoenicea and chemical constituents analysis
- Recovery of γ-Fe2O3 from copper ore tailings by magnetization roasting and magnetic separation
- Effects of different extraction methods on antioxidant properties of blueberry anthocyanins
- Modeling the removal of methylene blue dye using a graphene oxide/TiO2/SiO2 nanocomposite under sunlight irradiation by intelligent system
- Antimicrobial and antioxidant activities of Cinnamomum cassia essential oil and its application in food preservation
- Full spectrum and genetic algorithm-selected spectrum-based chemometric methods for simultaneous determination of azilsartan medoxomil, chlorthalidone, and azilsartan: Development, validation, and application on commercial dosage form
- Evaluation of the performance of immunoblot and immunodot techniques used to identify autoantibodies in patients with autoimmune diseases
- Computational studies by molecular docking of some antiviral drugs with COVID-19 receptors are an approach to medication for COVID-19
- Synthesis of amides and esters containing furan rings under microwave-assisted conditions
- Simultaneous removal efficiency of H2S and CO2 by high-gravity rotating packed bed: Experiments and simulation
- Design, synthesis, and biological activities of novel thiophene, pyrimidine, pyrazole, pyridine, coumarin and isoxazole: Dydrogesterone derivatives as antitumor agents
- Content and composition analysis of polysaccharides from Blaps rynchopetera and its macrophage phagocytic activity
- A new series of 2,4-thiazolidinediones endowed with potent aldose reductase inhibitory activity
- Assessing encapsulation of curcumin in cocoliposome: In vitro study
- Rare norisodinosterol derivatives from Xenia umbellata: Isolation and anti-proliferative activity
- Comparative study of antioxidant and anticancer activities and HPTLC quantification of rutin in white radish (Raphanus sativus L.) leaves and root extracts grown in Saudi Arabia
- Comparison of adsorption properties of commercial silica and rice husk ash (RHA) silica: A study by NIR spectroscopy
- Sodium borohydride (NaBH4) as a high-capacity material for next-generation sodium-ion capacitors
- Aroma components of tobacco powder from different producing areas based on gas chromatography ion mobility spectrometry
- The effects of salinity on changes in characteristics of soils collected in a saline region of the Mekong Delta, Vietnam
- Synthesis, properties, and activity of MoVTeNbO catalysts modified by zirconia-pillared clays in oxidative dehydrogenation of ethane
- Synthesis and crystal structure of N,N′-bis(4-chlorophenyl)thiourea N,N-dimethylformamide
- Quantitative analysis of volatile compounds of four Chinese traditional liquors by SPME-GC-MS and determination of total phenolic contents and antioxidant activities
- A novel separation method of the valuable components for activated clay production wastewater
- On ve-degree- and ev-degree-based topological properties of crystallographic structure of cuprite Cu2O
- Antihyperglycemic effect and phytochemical investigation of Rubia cordifolia (Indian Madder) leaves extract
- Microsphere molecularly imprinted solid-phase extraction for diazepam analysis using itaconic acid as a monomer in propanol
- A nitric oxide-releasing prodrug promotes apoptosis in human renal carcinoma cells: Involvement of reactive oxygen species
- Machine vision-based driving and feedback scheme for digital microfluidics system
- Study on the application of a steam-foam drive profile modification technology for heavy oil reservoir development
- Ni–Ru-containing mixed oxide-based composites as precursors for ethanol steam reforming catalysts: Effect of the synthesis methods on the structural and catalytic properties
- Preparation of composite soybean straw-based materials by LDHs modifying as a solid sorbent for removal of Pb(ii) from water samples
- Synthesis and spectral characterizations of vanadyl(ii) and chromium(iii) mixed ligand complexes containing metformin drug and glycine amino acid
- In vitro evaluation of lactic acid bacteria with probiotic activity isolated from local pickled leaf mustard from Wuwei in Anhui as substitutes for chemical synthetic additives
- Utilization and simulation of innovative new binuclear Co(ii), Ni(ii), Cu(ii), and Zn(ii) diimine Schiff base complexes in sterilization and coronavirus resistance (Covid-19)
- Phosphorylation of Pit-1 by cyclin-dependent kinase 5 at serine 126 is associated with cell proliferation and poor prognosis in prolactinomas
- Molecularly imprinted membrane for transport of urea, creatinine, and vitamin B12 as a hemodialysis candidate membrane
- Optimization of Murrayafoline A ethanol extraction process from the roots of Glycosmis stenocarpa, and evaluation of its Tumorigenesis inhibition activity on Hep-G2 cells
- Highly sensitive determination of α-lipoic acid in pharmaceuticals on a boron-doped diamond electrode
- Synthesis, chemo-informatics, and anticancer evaluation of fluorophenyl-isoxazole derivatives
- In vitro and in vivo investigation of polypharmacology of propolis extract as anticancer, antibacterial, anti-inflammatory, and chemical properties
- Topological indices of bipolar fuzzy incidence graph
- Preparation of Fe3O4@SiO2–ZnO catalyst and its catalytic synthesis of rosin glycol ester
- Construction of a new luminescent Cd(ii) compound for the detection of Fe3+ and treatment of Hepatitis B
- Investigation of bovine serum albumin aggregation upon exposure to silver(i) and copper(ii) metal ions using Zetasizer
- Discoloration of methylene blue at neutral pH by heterogeneous photo-Fenton-like reactions using crystalline and amorphous iron oxides
- Optimized extraction of polyphenols from leaves of Rosemary (Rosmarinus officinalis L.) grown in Lam Dong province, Vietnam, and evaluation of their antioxidant capacity
- Synthesis of novel thiourea-/urea-benzimidazole derivatives as anticancer agents
- Potency and selectivity indices of Myristica fragrans Houtt. mace chloroform extract against non-clinical and clinical human pathogens
- Simple modifications of nicotinic, isonicotinic, and 2,6-dichloroisonicotinic acids toward new weapons against plant diseases
- Synthesis, optical and structural characterisation of ZnS nanoparticles derived from Zn(ii) dithiocarbamate complexes
- Presence of short and cyclic peptides in Acacia and Ziziphus honeys may potentiate their medicinal values
- The role of vitamin D deficiency and elevated inflammatory biomarkers as risk factors for the progression of diabetic nephropathy in patients with type 2 diabetes mellitus
- Quantitative structure–activity relationship study on prolonged anticonvulsant activity of terpene derivatives in pentylenetetrazole test
- GADD45B induced the enhancing of cell viability and proliferation in radiotherapy and increased the radioresistance of HONE1 cells
- Cannabis sativa L. chemical compositions as potential plasmodium falciparum dihydrofolate reductase-thymidinesynthase enzyme inhibitors: An in silico study for drug development
- Dynamics of λ-cyhalothrin disappearance and expression of selected P450 genes in bees depending on the ambient temperature
- Identification of synthetic cannabinoid methyl 2-{[1-(cyclohexylmethyl)-1H-indol-3-yl] formamido}-3-methylbutanoate using modern mass spectrometry and nuclear magnetic resonance techniques
- Study on the speciation of arsenic in the genuine medicinal material honeysuckle
- Two Cu(ii)-based coordination polymers: Crystal structures and treatment activity on periodontitis
- Conversion of furfuryl alcohol to ethyl levulinate in the presence of mesoporous aluminosilicate catalyst
- Review Articles
- Hsien Wu and his major contributions to the chemical era of immunology
- Overview of the major classes of new psychoactive substances, psychoactive effects, analytical determination and conformational analysis of selected illegal drugs
- An overview of persistent organic pollutants along the coastal environment of Kuwait
- Mechanism underlying sevoflurane-induced protection in cerebral ischemia–reperfusion injury
- COVID-19 and SARS-CoV-2: Everything we know so far – A comprehensive review
- Challenge of diabetes mellitus and researchers’ contributions to its control
- Advances in the design and application of transition metal oxide-based supercapacitors
- Color and composition of beauty products formulated with lemongrass essential oil: Cosmetics formulation with lemongrass essential oil
- The structural chemistry of zinc(ii) and nickel(ii) dithiocarbamate complexes
- Bioprospecting for antituberculosis natural products – A review
- Recent progress in direct urea fuel cell
- Rapid Communications
- A comparative morphological study of titanium dioxide surface layer dental implants
- Changes in the antioxidative properties of honeys during their fermentation
- Erratum
- Erratum to “Corrosion study of copper in aqueous sulfuric acid solution in the presence of (2E,5E)-2,5-dibenzylidenecyclopentanone and (2E,5E)-bis[(4-dimethylamino)benzylidene]cyclopentanone: Experimental and theoretical study”
- Erratum to “Modified TDAE petroleum plasticiser”
- Corrigendum
- Corrigendum to “A nitric oxide-releasing prodrug promotes apoptosis in human renal carcinoma cells: Involvement of reactive oxygen species”
- Special Issue on 3rd IC3PE 2020
- Visible light-responsive photocatalyst of SnO2/rGO prepared using Pometia pinnata leaf extract
- Antihyperglycemic activity of Centella asiatica (L.) Urb. leaf ethanol extract SNEDDS in zebrafish (Danio rerio)
- Selection of oil extraction process from Chlorella species of microalgae by using multi-criteria decision analysis technique for biodiesel production
- Special Issue on the 14th Joint Conference of Chemistry (14JCC)
- Synthesis and in vitro cytotoxicity evaluation of isatin-pyrrole derivatives against HepG2 cell line
- CO2 gas separation using mixed matrix membranes based on polyethersulfone/MIL-100(Al)
- Effect of synthesis and activation methods on the character of CoMo/ultrastable Y-zeolite catalysts
- Special Issue on Electrochemical Amplified Sensors
- Enhancement of graphene oxide through β-cyclodextrin composite to sensitive analysis of an antidepressant: Sulpiride
- Investigation of the spectroelectrochemical behavior of quercetin isolated from Zanthoxylum bungeanum
- An electrochemical sensor for high sensitive determination of lysozyme based on the aptamer competition approach
- An improved non-enzymatic electrochemical sensor amplified with CuO nanostructures for sensitive determination of uric acid
- Special Issue on Applied Biochemistry and Biotechnology 2020
- Fast discrimination of avocado oil for different extracted methods using headspace-gas chromatography-ion mobility spectroscopy with PCA based on volatile organic compounds
- Effect of alkali bases on the synthesis of ZnO quantum dots
- Quality evaluation of Cabernet Sauvignon wines in different vintages by 1H nuclear magnetic resonance-based metabolomics
- Special Issue on the Joint Science Congress of Materials and Polymers (ISCMP 2019)
- Diatomaceous Earth: Characterization, thermal modification, and application
- Electrochemical determination of atenolol and propranolol using a carbon paste sensor modified with natural ilmenite
- Special Issue on the Conference of Energy, Fuels, Environment 2020
- Assessment of the mercury contamination of landfilled and recovered foundry waste – a case study
- Primary energy consumption in selected EU Countries compared to global trends
- Modified TDAE petroleum plasticiser
- Use of glycerol waste in lactic acid bacteria metabolism for the production of lactic acid: State of the art in Poland
- Topical Issue on Applications of Mathematics in Chemistry
- Theoretical study of energy, inertia and nullity of phenylene and anthracene
- Banhatti, revan and hyper-indices of silicon carbide Si2C3-III[n,m]
- Topical Issue on Agriculture
- Occurrence of mycotoxins in selected agricultural and commercial products available in eastern Poland
- Special Issue on Ethnobotanical, Phytochemical and Biological Investigation of Medicinal Plants
- Acute and repeated dose 60-day oral toxicity assessment of chemically characterized Berberis hispanica Boiss. and Reut in Wistar rats
- Phytochemical profile, in vitro antioxidant, and anti-protein denaturation activities of Curcuma longa L. rhizome and leaves
- Antiplasmodial potential of Eucalyptus obliqua leaf methanolic extract against Plasmodium vivax: An in vitro study
- Prunus padus L. bark as a functional promoting component in functional herbal infusions – cyclooxygenase-2 inhibitory, antioxidant, and antimicrobial effects
- Molecular and docking studies of tetramethoxy hydroxyflavone compound from Artemisia absinthium against carcinogens found in cigarette smoke
- Special Issue on the Joint Science Congress of Materials and Polymers (ISCMP 2020)
- Preparation of cypress (Cupressus sempervirens L.) essential oil loaded poly(lactic acid) nanofibers
- Influence of mica mineral on flame retardancy and mechanical properties of intumescent flame retardant polypropylene composites
- Production and characterization of thermoplastic elastomer foams based on the styrene–ethylene–butylene–styrene (SEBS) rubber and thermoplastic material
- Special Issue on Applied Chemistry in Agriculture and Food Science
- Impact of essential oils on the development of pathogens of the Fusarium genus and germination parameters of selected crops
- Yield, volume, quality, and reduction of biotic stress influenced by titanium application in oilseed rape, winter wheat, and maize cultivations
- Influence of potato variety on polyphenol profile composition and glycoalcaloid contents of potato juice
- Carryover effect of direct-fed microbial supplementation and early weaning on the growth performance and carcass characteristics of growing Najdi lambs
- Special Issue on Applied Biochemistry and Biotechnology (ABB 2021)
- The electrochemical redox mechanism and antioxidant activity of polyphenolic compounds based on inlaid multi-walled carbon nanotubes-modified graphite electrode
- Study of an adsorption method for trace mercury based on Bacillus subtilis
- Special Issue on The 1st Malaysia International Conference on Nanotechnology & Catalysis (MICNC2021)
- Mitigating membrane biofouling in biofuel cell system – A review
- Mechanical properties of polymeric biomaterials: Modified ePTFE using gamma irradiation
Articles in the same Issue
- Regular Articles
- Qualitative and semi-quantitative assessment of anthocyanins in Tibetan hulless barley from different geographical locations by UPLC-QTOF-MS and their antioxidant capacities
- Effect of sodium chloride on the expression of genes involved in the salt tolerance of Bacillus sp. strain “SX4” isolated from salinized greenhouse soil
- GC-MS analysis of mango stem bark extracts (Mangifera indica L.), Haden variety. Possible contribution of volatile compounds to its health effects
- Influence of nanoscale-modified apatite-type calcium phosphates on the biofilm formation by pathogenic microorganisms
- Removal of paracetamol from aqueous solution by containment composites
- Investigating a human pesticide intoxication incident: The importance of robust analytical approaches
- Induction of apoptosis and cell cycle arrest by chloroform fraction of Juniperus phoenicea and chemical constituents analysis
- Recovery of γ-Fe2O3 from copper ore tailings by magnetization roasting and magnetic separation
- Effects of different extraction methods on antioxidant properties of blueberry anthocyanins
- Modeling the removal of methylene blue dye using a graphene oxide/TiO2/SiO2 nanocomposite under sunlight irradiation by intelligent system
- Antimicrobial and antioxidant activities of Cinnamomum cassia essential oil and its application in food preservation
- Full spectrum and genetic algorithm-selected spectrum-based chemometric methods for simultaneous determination of azilsartan medoxomil, chlorthalidone, and azilsartan: Development, validation, and application on commercial dosage form
- Evaluation of the performance of immunoblot and immunodot techniques used to identify autoantibodies in patients with autoimmune diseases
- Computational studies by molecular docking of some antiviral drugs with COVID-19 receptors are an approach to medication for COVID-19
- Synthesis of amides and esters containing furan rings under microwave-assisted conditions
- Simultaneous removal efficiency of H2S and CO2 by high-gravity rotating packed bed: Experiments and simulation
- Design, synthesis, and biological activities of novel thiophene, pyrimidine, pyrazole, pyridine, coumarin and isoxazole: Dydrogesterone derivatives as antitumor agents
- Content and composition analysis of polysaccharides from Blaps rynchopetera and its macrophage phagocytic activity
- A new series of 2,4-thiazolidinediones endowed with potent aldose reductase inhibitory activity
- Assessing encapsulation of curcumin in cocoliposome: In vitro study
- Rare norisodinosterol derivatives from Xenia umbellata: Isolation and anti-proliferative activity
- Comparative study of antioxidant and anticancer activities and HPTLC quantification of rutin in white radish (Raphanus sativus L.) leaves and root extracts grown in Saudi Arabia
- Comparison of adsorption properties of commercial silica and rice husk ash (RHA) silica: A study by NIR spectroscopy
- Sodium borohydride (NaBH4) as a high-capacity material for next-generation sodium-ion capacitors
- Aroma components of tobacco powder from different producing areas based on gas chromatography ion mobility spectrometry
- The effects of salinity on changes in characteristics of soils collected in a saline region of the Mekong Delta, Vietnam
- Synthesis, properties, and activity of MoVTeNbO catalysts modified by zirconia-pillared clays in oxidative dehydrogenation of ethane
- Synthesis and crystal structure of N,N′-bis(4-chlorophenyl)thiourea N,N-dimethylformamide
- Quantitative analysis of volatile compounds of four Chinese traditional liquors by SPME-GC-MS and determination of total phenolic contents and antioxidant activities
- A novel separation method of the valuable components for activated clay production wastewater
- On ve-degree- and ev-degree-based topological properties of crystallographic structure of cuprite Cu2O
- Antihyperglycemic effect and phytochemical investigation of Rubia cordifolia (Indian Madder) leaves extract
- Microsphere molecularly imprinted solid-phase extraction for diazepam analysis using itaconic acid as a monomer in propanol
- A nitric oxide-releasing prodrug promotes apoptosis in human renal carcinoma cells: Involvement of reactive oxygen species
- Machine vision-based driving and feedback scheme for digital microfluidics system
- Study on the application of a steam-foam drive profile modification technology for heavy oil reservoir development
- Ni–Ru-containing mixed oxide-based composites as precursors for ethanol steam reforming catalysts: Effect of the synthesis methods on the structural and catalytic properties
- Preparation of composite soybean straw-based materials by LDHs modifying as a solid sorbent for removal of Pb(ii) from water samples
- Synthesis and spectral characterizations of vanadyl(ii) and chromium(iii) mixed ligand complexes containing metformin drug and glycine amino acid
- In vitro evaluation of lactic acid bacteria with probiotic activity isolated from local pickled leaf mustard from Wuwei in Anhui as substitutes for chemical synthetic additives
- Utilization and simulation of innovative new binuclear Co(ii), Ni(ii), Cu(ii), and Zn(ii) diimine Schiff base complexes in sterilization and coronavirus resistance (Covid-19)
- Phosphorylation of Pit-1 by cyclin-dependent kinase 5 at serine 126 is associated with cell proliferation and poor prognosis in prolactinomas
- Molecularly imprinted membrane for transport of urea, creatinine, and vitamin B12 as a hemodialysis candidate membrane
- Optimization of Murrayafoline A ethanol extraction process from the roots of Glycosmis stenocarpa, and evaluation of its Tumorigenesis inhibition activity on Hep-G2 cells
- Highly sensitive determination of α-lipoic acid in pharmaceuticals on a boron-doped diamond electrode
- Synthesis, chemo-informatics, and anticancer evaluation of fluorophenyl-isoxazole derivatives
- In vitro and in vivo investigation of polypharmacology of propolis extract as anticancer, antibacterial, anti-inflammatory, and chemical properties
- Topological indices of bipolar fuzzy incidence graph
- Preparation of Fe3O4@SiO2–ZnO catalyst and its catalytic synthesis of rosin glycol ester
- Construction of a new luminescent Cd(ii) compound for the detection of Fe3+ and treatment of Hepatitis B
- Investigation of bovine serum albumin aggregation upon exposure to silver(i) and copper(ii) metal ions using Zetasizer
- Discoloration of methylene blue at neutral pH by heterogeneous photo-Fenton-like reactions using crystalline and amorphous iron oxides
- Optimized extraction of polyphenols from leaves of Rosemary (Rosmarinus officinalis L.) grown in Lam Dong province, Vietnam, and evaluation of their antioxidant capacity
- Synthesis of novel thiourea-/urea-benzimidazole derivatives as anticancer agents
- Potency and selectivity indices of Myristica fragrans Houtt. mace chloroform extract against non-clinical and clinical human pathogens
- Simple modifications of nicotinic, isonicotinic, and 2,6-dichloroisonicotinic acids toward new weapons against plant diseases
- Synthesis, optical and structural characterisation of ZnS nanoparticles derived from Zn(ii) dithiocarbamate complexes
- Presence of short and cyclic peptides in Acacia and Ziziphus honeys may potentiate their medicinal values
- The role of vitamin D deficiency and elevated inflammatory biomarkers as risk factors for the progression of diabetic nephropathy in patients with type 2 diabetes mellitus
- Quantitative structure–activity relationship study on prolonged anticonvulsant activity of terpene derivatives in pentylenetetrazole test
- GADD45B induced the enhancing of cell viability and proliferation in radiotherapy and increased the radioresistance of HONE1 cells
- Cannabis sativa L. chemical compositions as potential plasmodium falciparum dihydrofolate reductase-thymidinesynthase enzyme inhibitors: An in silico study for drug development
- Dynamics of λ-cyhalothrin disappearance and expression of selected P450 genes in bees depending on the ambient temperature
- Identification of synthetic cannabinoid methyl 2-{[1-(cyclohexylmethyl)-1H-indol-3-yl] formamido}-3-methylbutanoate using modern mass spectrometry and nuclear magnetic resonance techniques
- Study on the speciation of arsenic in the genuine medicinal material honeysuckle
- Two Cu(ii)-based coordination polymers: Crystal structures and treatment activity on periodontitis
- Conversion of furfuryl alcohol to ethyl levulinate in the presence of mesoporous aluminosilicate catalyst
- Review Articles
- Hsien Wu and his major contributions to the chemical era of immunology
- Overview of the major classes of new psychoactive substances, psychoactive effects, analytical determination and conformational analysis of selected illegal drugs
- An overview of persistent organic pollutants along the coastal environment of Kuwait
- Mechanism underlying sevoflurane-induced protection in cerebral ischemia–reperfusion injury
- COVID-19 and SARS-CoV-2: Everything we know so far – A comprehensive review
- Challenge of diabetes mellitus and researchers’ contributions to its control
- Advances in the design and application of transition metal oxide-based supercapacitors
- Color and composition of beauty products formulated with lemongrass essential oil: Cosmetics formulation with lemongrass essential oil
- The structural chemistry of zinc(ii) and nickel(ii) dithiocarbamate complexes
- Bioprospecting for antituberculosis natural products – A review
- Recent progress in direct urea fuel cell
- Rapid Communications
- A comparative morphological study of titanium dioxide surface layer dental implants
- Changes in the antioxidative properties of honeys during their fermentation
- Erratum
- Erratum to “Corrosion study of copper in aqueous sulfuric acid solution in the presence of (2E,5E)-2,5-dibenzylidenecyclopentanone and (2E,5E)-bis[(4-dimethylamino)benzylidene]cyclopentanone: Experimental and theoretical study”
- Erratum to “Modified TDAE petroleum plasticiser”
- Corrigendum
- Corrigendum to “A nitric oxide-releasing prodrug promotes apoptosis in human renal carcinoma cells: Involvement of reactive oxygen species”
- Special Issue on 3rd IC3PE 2020
- Visible light-responsive photocatalyst of SnO2/rGO prepared using Pometia pinnata leaf extract
- Antihyperglycemic activity of Centella asiatica (L.) Urb. leaf ethanol extract SNEDDS in zebrafish (Danio rerio)
- Selection of oil extraction process from Chlorella species of microalgae by using multi-criteria decision analysis technique for biodiesel production
- Special Issue on the 14th Joint Conference of Chemistry (14JCC)
- Synthesis and in vitro cytotoxicity evaluation of isatin-pyrrole derivatives against HepG2 cell line
- CO2 gas separation using mixed matrix membranes based on polyethersulfone/MIL-100(Al)
- Effect of synthesis and activation methods on the character of CoMo/ultrastable Y-zeolite catalysts
- Special Issue on Electrochemical Amplified Sensors
- Enhancement of graphene oxide through β-cyclodextrin composite to sensitive analysis of an antidepressant: Sulpiride
- Investigation of the spectroelectrochemical behavior of quercetin isolated from Zanthoxylum bungeanum
- An electrochemical sensor for high sensitive determination of lysozyme based on the aptamer competition approach
- An improved non-enzymatic electrochemical sensor amplified with CuO nanostructures for sensitive determination of uric acid
- Special Issue on Applied Biochemistry and Biotechnology 2020
- Fast discrimination of avocado oil for different extracted methods using headspace-gas chromatography-ion mobility spectroscopy with PCA based on volatile organic compounds
- Effect of alkali bases on the synthesis of ZnO quantum dots
- Quality evaluation of Cabernet Sauvignon wines in different vintages by 1H nuclear magnetic resonance-based metabolomics
- Special Issue on the Joint Science Congress of Materials and Polymers (ISCMP 2019)
- Diatomaceous Earth: Characterization, thermal modification, and application
- Electrochemical determination of atenolol and propranolol using a carbon paste sensor modified with natural ilmenite
- Special Issue on the Conference of Energy, Fuels, Environment 2020
- Assessment of the mercury contamination of landfilled and recovered foundry waste – a case study
- Primary energy consumption in selected EU Countries compared to global trends
- Modified TDAE petroleum plasticiser
- Use of glycerol waste in lactic acid bacteria metabolism for the production of lactic acid: State of the art in Poland
- Topical Issue on Applications of Mathematics in Chemistry
- Theoretical study of energy, inertia and nullity of phenylene and anthracene
- Banhatti, revan and hyper-indices of silicon carbide Si2C3-III[n,m]
- Topical Issue on Agriculture
- Occurrence of mycotoxins in selected agricultural and commercial products available in eastern Poland
- Special Issue on Ethnobotanical, Phytochemical and Biological Investigation of Medicinal Plants
- Acute and repeated dose 60-day oral toxicity assessment of chemically characterized Berberis hispanica Boiss. and Reut in Wistar rats
- Phytochemical profile, in vitro antioxidant, and anti-protein denaturation activities of Curcuma longa L. rhizome and leaves
- Antiplasmodial potential of Eucalyptus obliqua leaf methanolic extract against Plasmodium vivax: An in vitro study
- Prunus padus L. bark as a functional promoting component in functional herbal infusions – cyclooxygenase-2 inhibitory, antioxidant, and antimicrobial effects
- Molecular and docking studies of tetramethoxy hydroxyflavone compound from Artemisia absinthium against carcinogens found in cigarette smoke
- Special Issue on the Joint Science Congress of Materials and Polymers (ISCMP 2020)
- Preparation of cypress (Cupressus sempervirens L.) essential oil loaded poly(lactic acid) nanofibers
- Influence of mica mineral on flame retardancy and mechanical properties of intumescent flame retardant polypropylene composites
- Production and characterization of thermoplastic elastomer foams based on the styrene–ethylene–butylene–styrene (SEBS) rubber and thermoplastic material
- Special Issue on Applied Chemistry in Agriculture and Food Science
- Impact of essential oils on the development of pathogens of the Fusarium genus and germination parameters of selected crops
- Yield, volume, quality, and reduction of biotic stress influenced by titanium application in oilseed rape, winter wheat, and maize cultivations
- Influence of potato variety on polyphenol profile composition and glycoalcaloid contents of potato juice
- Carryover effect of direct-fed microbial supplementation and early weaning on the growth performance and carcass characteristics of growing Najdi lambs
- Special Issue on Applied Biochemistry and Biotechnology (ABB 2021)
- The electrochemical redox mechanism and antioxidant activity of polyphenolic compounds based on inlaid multi-walled carbon nanotubes-modified graphite electrode
- Study of an adsorption method for trace mercury based on Bacillus subtilis
- Special Issue on The 1st Malaysia International Conference on Nanotechnology & Catalysis (MICNC2021)
- Mitigating membrane biofouling in biofuel cell system – A review
- Mechanical properties of polymeric biomaterials: Modified ePTFE using gamma irradiation


