Abstract
Gold extraction from the rock is generally carried out using mercury. However, the high toxicity of mercury has a very dangerous impact on the environment and health. Various efforts have been made to reduce the use of mercury in gold extraction, one of which is the leaching method using thiosulphate and thiourea solution because they have low toxicity and are environmentally friendly. This study aimed to determine the results of gold extraction with thiosulphate and thiourea solution and determined the optimum concentration and time of extraction. The yield of the gold extract with thiosulphate solution was greater than that of thiourea solution. The thiosulphate solution had an optimum concentration of 0.3 M and an optimum time of 2 h. While the thiourea solution had an optimum concentration of 0.2 M and an optimum time of 3 h. The results of the Friedman test on the leaching time and concentration parameters show that leaching time has a significant effect on the Au leaching process, and the concentration parameter does not affect the Au leaching process from solid samples.
1 Introduction
Gold processing carried out by various industrial sectors including small-scale gold mining generally uses mercury with amalgamation techniques. The use of mercury in the process of separating gold ore is because mercury has properties and characteristics that can be used to bind gold by forming amalgam compounds (Au2Hg3) [1,2]. The properties and characteristics of mercury include having a very high attractiveness to the element gold [3]. The advantages of this amalgamation technique are that it is easy to perform and fast [4]. However, mercury has the highest level of toxicity compared to other metals such as silver, arsenic, lead, nickel, and zinc [3,5,6].
Another method that is also used to separate gold from ore in industry and small-scale gold mining is the cyanide leaching method [7,8]. Leaching is the process of separating a solid that is dissolved with certain reagents [9,10,11]. The cyanide leaching process generally uses reagents such as NaCN and KCN to bind gold ions to form gold complex ions or aurocyanide (AuCN−) [12]. The cyanide method has low operating costs and high extraction yields [13,14]. However, this process also has a high level of toxicity and can pollute the environment [15].
The high impact and danger posed by the amalgamation method and the cyanide method commonly used by gold miners attract attention to be able to seek the development of alternative methods that are safe for the environment and health. Moreover, many gold mining activities are carried out because they can significantly increase people’s income [11]. The government has made various efforts to reduce the impact of the dangers, one of which is by issuing Presidential Regulation No. 21/2019 concerning the National Action Plan for Mercury Reduction and Elimination. Based on the regulation, by 2025, it is targeted to eliminate 100% of mercury, or in other words, mercury is no longer used in gold ore processing activities [16,17,18]. One more environmental friendly method in the recovery of gold from soil and rocks is phytomining. Phytomining is the use of plants to absorb metals in soil and rocks [19,20,21]. However, this method has drawbacks even though it is considered the most environmentally friendly. The disadvantage of phytomining is that the time or the process of taking metals, especially gold takes a relatively long process. This method is usually used to recover metals with relatively low grades to reduce the cost of mining these metals, which are deemed unprofitable due to low rates. The phytomining method uses a leaching process to extract the accumulated metals in the plant, so a reasonably efficient and effective method is to use environmentally friendly reagents. Another alternative method for processing gold ore is using more environmentally friendly reagents. Thiosulphate and thiourea are currently an alternative to cyanide reagents because they show the ability to extract gold and have a smaller negative impact on the environment and health [22,23,24,25,26]. The thiosulphate solution can form an anionic complex [Au(S2O3)2]3− with gold under alkaline conditions, while thiourea solution can form cationic complex Au[CS(NH2)]2 + in acidic conditions [27]. The advantages of these two solutions are the high dissolution rate of gold compared to cyanide solutions and fewer environmental problems [23,28,29]. In this study, the leaching process using thiosulphate and thiourea solutions is expected to increase the recovery of gold from ore by varying the contact time, varying the concentration, and determining the effect of using variations of reagent for leaching.
2 Method
2.1 Materials
The materials used include rock samples from the area mining West Sumbawa-Nusa Tenggara Barat, Indonesia. Chemicals consist of nitric acid (HNO3), hydrochloric acid (HCl), sodium thiosulphate (Na2S2O3), thiourea (CS(NH2)2), sulfuric acid (H2SO4), ferrous(iii)chloride (FeCl3·6H2O), copper(ii)pentahydrous sulfate (CuSO4·5H2O), and ammonium hydroxide (NH4OH), which were purchased from Merck-Millipore. The instruments used were scanning electron microscopy–energy-dispersive X-ray (SEM–EDX) and atomic absorption spectrophotometer (AAS) (Perkin Elmer PinAAcle 900T).
2.2 Preparation and pre-treatment
The gold rocks obtained from West Sumbawa Regency were crushed until it becomes powder. After that, it was sifted with a 100 mesh sieve (150 µm). The sampled in sieve were weighed as much as 100 g. Samples of sifted rocks were roasted for 45 min at a temperature of 500°C [30]. Rock samples were analysed using SEM–EDX to determine the morphology and the elemental content.
2.3 Analysis of gold (Au) content in rocks
A total of 2.5 g of rock samples (after pre-treatment) was placed into a beaker glass, followed by the addition of 25 mL of aqua regia solution. Aqua regia was made by mixing HCl and HNO3 (3:1) v/v [31]. The mixture was stirred and let stand for a while until all the metal dissolves. Then the solution was separated using the filter paper. The obtained filtrate was analysed using AAS to determine the Au content.
2.4 Preparation of leachate solution
2.4.1 Preparation of thiosulphate leachate solution
About 100 mL of sodium thiosulphate solution (Na2S2O3) for each concentration was placed into a 250 mL beaker glass, and then, 0.5 M NH4OH was added until the pH of the solution reached 10. Then, 20 mL of 0.03 M CuSO4·5H2O was added and stirred until a homogeneous solution was produced.
2.4.2 Preparation of thiourea leachate solution
About 100 mL of CS(NH2)2 solution was put into a 250 mL beaker. Then the pH was adjusted to 2 by adding 1 M H2SO4. After that, 30 mL of 0.025 M FeCl3 solution was added and stirred until homogeneous.
2.5 Leaching of Au with thiosulphate solution
2.5.1 Leaching process with contact time variations
A total of 2.5 g of samples were weighed and placed into a 100 mL beaker glass. Then, 25 mL of 0.2 M thiosulphate leachate solution was added. The mixture was sorted with variations in leaching time for 1, 2, 3, and 4 h. The leaching results were separated using filter paper, and the filtrate obtained was analysed using an AAS.
2.5.2 Leaching process with concentration variations
A total of 2.5 g of pre-treatment rock samples were weighed and placed into a 100 mL beaker glass. Then, a 25 mL of thiosulphate leachate solution with a concentration variation of 0.1 M, 0.2 M, and 0.3 M. The mixture was stirred for 3 h. The leaching results were filtered, and the filtrate obtained was analysed using an AAS.
2.6 Leaching of Au with thiourea solution
The same method presented earlier for leaching of Au with thiosulphate solution was followed for leaching of Au with thiourea solution.
3 Results and discussion
The rock samples used in this study were brownish-yellow obtained from the West Sumbawa mining area, West Nusa Tenggara. Rock samples are first carried out by crushing, smoothing, and sifting (Figure 1). The sample was crushed using a stone crusher, then smoothed using a set mortar and pestle, and then sifted. This process aims to obtain a smaller particle size. The small particle size will enlarge the surface area to improve the contact between solids and solution during the extraction process.
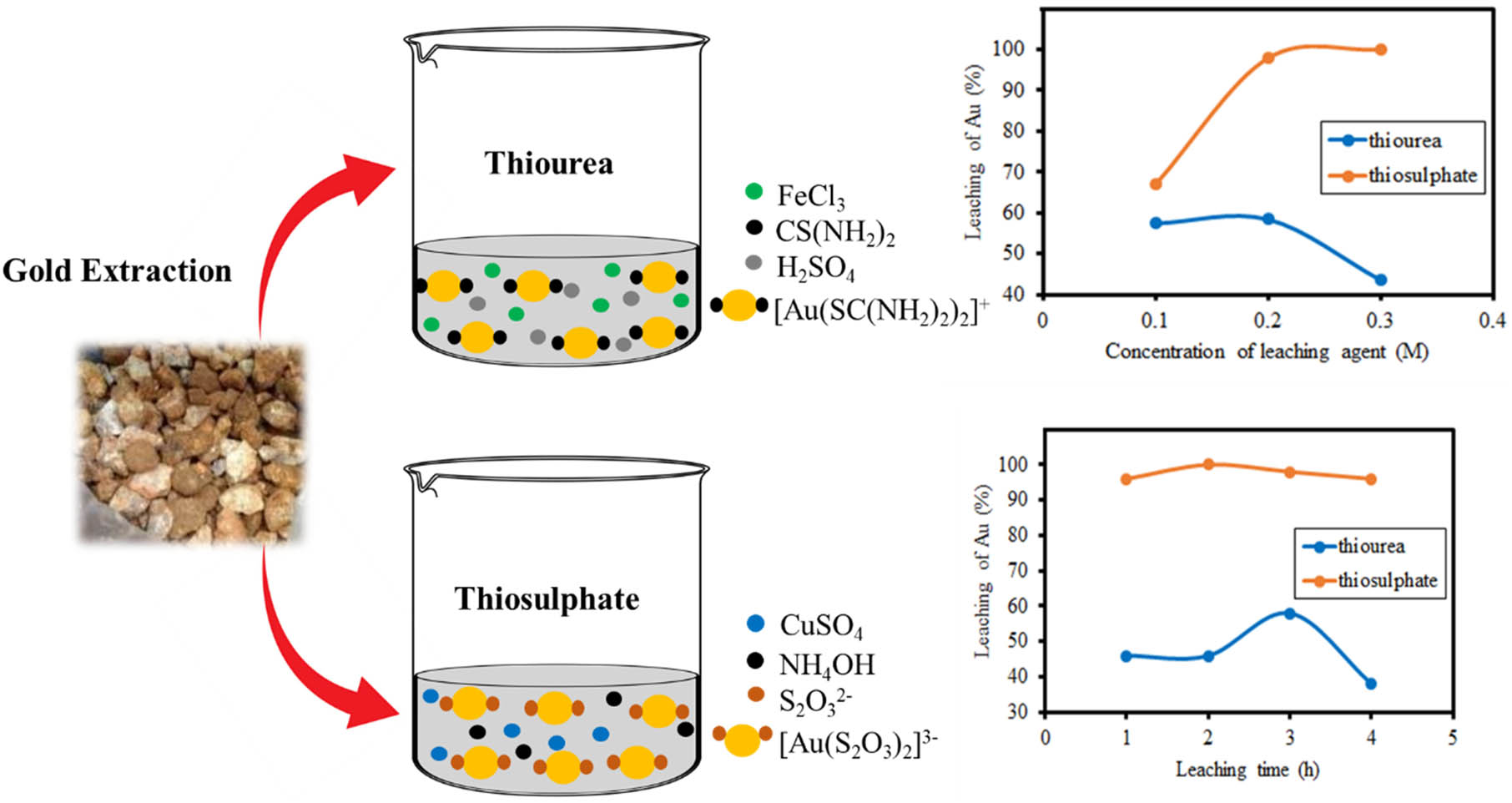
Gold extraction scheme.
Preliminary analysis of rock samples using the SEM–EDX instrument was aimed to determine the morphology of the surface as well as the composition contained in the rock. The results are shown in Figure 2, and the composition of the rocks is presented in Table 1. Based on these results, the surface of the rock appears relatively rough as well as large particles. The rock is dominantly made up of oxygen, silica, and carbon by 62.55, 15.12, and 10.91%, respectively. From these results, the composition of the rocks does not indicate the presence of the mineral gold (Au). In this case, gold particles are likely to be excluded from the dominant minerals [32]. The mineral can be pyrite (FeS2). This is in line with Munganyika’s research (2022), which stated that a significant iron content indicates a source of gold in pyrite [29].

The gold rocks after roasting at 500°C.
Chemical composition of rocks
| Element | % |
|---|---|
| O | 62.55 |
| Si | 15.12 |
| C | 10.91 |
| Al | 4.78 |
| Fe | 4.19 |
| Mg | 1.80 |
| K | 0.26 |
| Zn | 0.24 |
| Ca | 0.15 |
To determine the gold content contained in the rock sample, a preliminary analysis was carried out using an aqua regia solvent. The use of this solvent is based on the national gold testing standard contained in SNI 8880:2020. This is also in line with several previous studies such as those conducted by Piotr et al. [33] and Mooki et al. [34], which use aqua regia solvents to determine the initial content of gold and silver metal in rock samples [33,34]. Aqua regia solvents are made by mixing 37% HCl and 65% HNO3 in a ratio of 3:1. The mixture has obtained a solution of yellow colour. Furthermore, the gold leaching process is carried out for 10 min. The reaction that occurs in the process of leaching gold with the solvent of aqua regia is shown in equation (1) [23].
The dissolution result is in the form of a filtrate that has been separated from its residue and analysed using the AAS instrument. Thus, the gold content contained in the rock sample is known to be 0.0015%.
Thiosulphate is known to have the ability to dissolve gold by forming a complex [Au(S2O3)2]3−. Thiosulphate leachate solution is prepared by mixing the Na2S2O3 solution as the main solvent, CuSO4 solution as a catalyst, and ammonia solution, which is used to regulate the pH of the solution to pH 9–11 as well as acts a stabilizer of copper ions. The golden leaching system with thiosulphate occurs under alkaline conditions aimed at preventing the decomposition of thiosulphate and copper ions becomes ineffective when it is below pH 9. This mixture has obtained a solution of blue colour. Furthermore, the leaching process is carried out by adding a leachate solution to the pre-treatment rock sample. The leaching process is carried out based on a pre-determined time. Then the mixture is filtered using filter paper. The filtrate obtained from the filtering results is in the form of a colourless, and clear solution, which is then analysed using AAS to determine the concentration of gold, that has been obtained. The reaction of the formation of a gold-stable complex with thiosulphate occurs through two stages, which is indicated in equations (2) and (3).
The addition of ammonia and copper ions serves as a catalyst that accelerates the rate of leaching. Ammonia and copper ions in the solution form a complex [Cu(NH3)4]2+. This complex plays a role in oxidizing Au to Au+. Then there was a substitution of NH3 with S2O3 2−, so that a more stable [Au(S2O3)2]3− complex was formed. However, in the cathode region, the resulting [Cu(NH3)4]2+ is reduced to [Cu(S2O3)3]5− and [Cu(S2O3)3]5−, which is oxidized again rapidly by dissolved oxygen forming [Cu(NH3)4]2+ [35].
So that the total reaction of dissolution of gold in thiosulphate solution is as follows:
Thiourea is also known to have the ability to dissolve gold by forming the cationic complex [Au(CS(NH2)2)]2+. The thiourea leach solution is made by mixing thiourea solution, FeCl3 solution as an oxidizing agent, and H2SO4 solution as a pH regulator, so that the pH becomes 1–2 because in that pH range, the gold leaching process with thiourea solution is more efficient. If the solution does not have an acidic pH, thiourea becomes unstable and decomposes easily. The results of the mixture obtained a slightly yellowish and clear solution. After that, the leaching process was carried out by adding a leach solution to the pre-treatment rock samples. The leaching process is carried out based on predetermined time. The products obtained after leaching were filtered using filter paper to separate the filtrate and residue. The filtrate obtained was a colourless and clear solution which was then analysed using AAS to determine the concentration of gold that was successfully extracted with thiourea solution.
The reaction for the formation of the gold-thiourea cationic complex consists of two steps. The first step is that thiourea is converted to formamidine disulphide (FDS) by an oxidizing agent, namely Fe3+. Then FDS will react with gold in excess thiourea to form a gold-thiourea complex. The reactions that occur are presented as equations (5) and (6):
The total reaction is shown in equation (7).
Fe(iii) present in the solution from the addition of FeCl3 solution plays an important role in oxidizing thiourea to FDS (NH(NH2)CSSC(NH2)NH). Without the addition of an oxidizing agent, the leaching rate will be slower. Fe(iii) was chosen to be an effective oxidizing agent [36,37,38]. This is also supported by the value of the high standard reduction potential of Fe(iii), which is 0.77 V. FDS from thiourea oxidation can oxidize gold in solution, so that it can help increase the leaching speed.
Research on the effect of thiosulphate and thiourea concentrations on gold extraction was also carried out with a concentration variation of 0.1, 0.2, and 0.3 M using a solid sample of 2.5 g, 25 mL of leachate solution, and a leaching time of 3 h. The results of the leaching in the form of filtrate and residue were separated by filtration, and the filtrate was analysed using an AAS to determine the percentage of gold solubility obtained. The obtained gold extraction is due to leaching with varying concentrations of thiosulphate and thiourea, as shown in Figure 3 and Table 2.
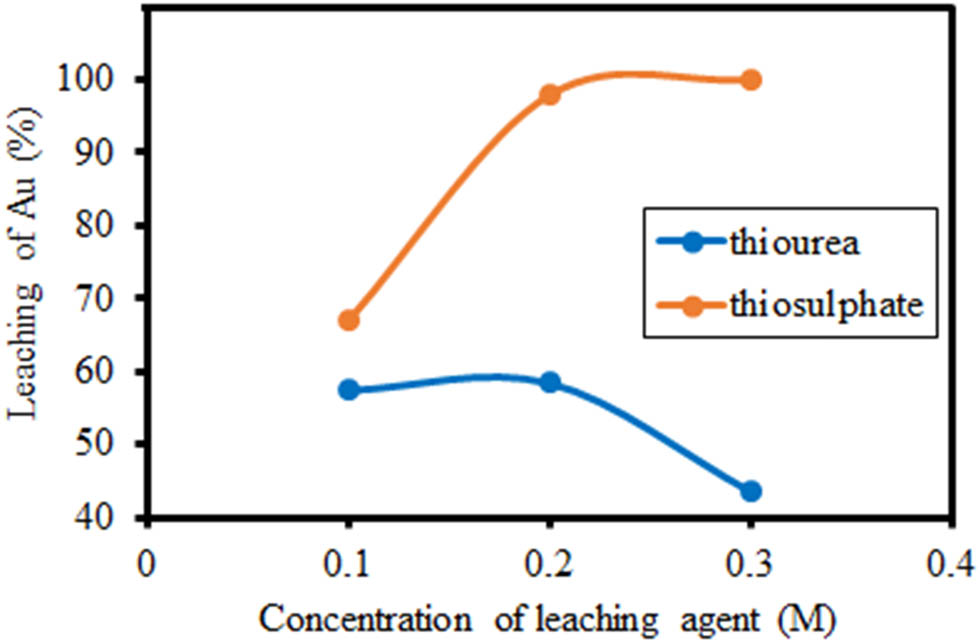
Effect of thiosulphate and thiourea concentration on Au extraction from 2.5 g sample in 25 mL of leachate solution.
Au fraction dissolved in thiosulphate and thiourea leachate solutions
| The concentration of leaching agent (M) | Fraction dissolved | |
|---|---|---|
| Thiosulphate | Thiourea | |
| 0.1 | 0.574 | 0.670 |
| 0.2 | 0.584 | 0.979 |
| 0.3 | 0.436 | 1.000 |
Figure 3 and Table 2 show the results of gold extraction against variations in thiosulphate concentrations. An increase in gold extraction was linearly observed with thiosulphate concentrations of 0.1–0.3 M. Gold extraction at a thiosulphate concentration of 0.3 M reached 100%. From these results, it is known that at the high concentration of thiosulphate, the gold extraction will also increase. The optimum concentration of thiosulphate can accelerate the rate of leaching. However, if the thiosulfate concentration is too low, it can lead to gold deposition [39].
Maximum gold extraction was achieved at 0.2 M thiourea concentration with a yield of 58.4% and decreased at 0.3 M concentration with an extraction percentage of 43.6%. Higher concentrations result in lower gold extraction. This occurs due to the increased decomposition of thiourea into FDS ((CS(NH2)NH)2), which then decomposes irreversibly into cyanamide (NH2CN) and elemental sulphur [38]. The sulphur then forms a passivation layer on the surface of the gold, so that it inhibits the extraction process [40,41,42]. The decomposition of thiourea into formamidine disulphide is shown in equation (8), and the reaction for the formation of cyanamide and elemental sulphur is shown in equation (9).
Thiosulphate solutions are better at extracting Au from solids than thiourea solutions (Table 3). This is due to Fe(iii) in the thiourea solution as an oxidizing agent. Fe(iii) will reduce to Fe(ii). As the leaching time increases in the extraction at pH 2, there is a continuous oxidation of Fe(ii) to Fe(iii), while thiosulfate remains consistently flow throughout the process (Figure 4). This triggers the reduction of Au(i) to Au(0), which is not identified during analysis with AAS. This is relevant to the previous research, which stated that Fe(ii) will undergo an oxidation reaction with O2 to become Fe(iii) through the formation of ferric superoxide (Fe(iii)–(O2˙−)) [43,44].
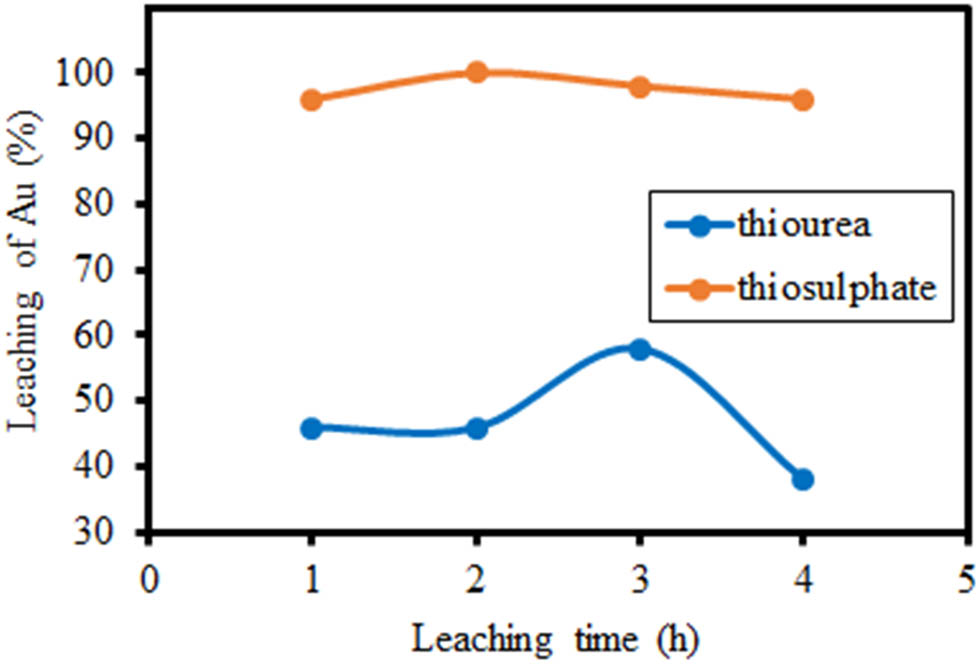
Effect of leaching time on Au extraction from 2.5 g sample in 25 mL of leachate solution (thiosulphate 0.3 M and thiourea 0.2 M).
Au fraction dissolved in thiosulphate and thiourea leachate solution at leaching time
| Time (h) | Fraction dissolved | |
|---|---|---|
| Thiosulphate | Thiourea | |
| 1 | 0.958 | 0.462 |
| 2 | 1.000 | 0.462 |
| 3 | 0.979 | 0.584 |
| 4 | 0.962 | 0.381 |
Based on Table 4, the results of the data normality test use the Kolmogorov–Smirnov and Shapiro–Wilk approach models. The Kolmogorov–Smirnov approach model is usually used for small sample data (<50 samples), and the Shapiro–Wilk approach model is usually used for more sample data (>50 samples) [45]. In Table 4, the Kolmogorov–Smirnov approach method does not show a significant value. It can be said that the Kolmogorov–Smirnov approach model can provide an overview for testing the normal distribution of data properly compared to the Shapiro–Wilk approach model. This is relevant to the previous research, which stated that the Kolmogorov–Smirnov approach model is less good at determining the normal distribution of data than the Shapiro–Wilk approach model [46]. Table 4 reveals that data obtained from different parameters, including leaching time and concentration, exhibit a normal distribution. However, there is one data point for the leachate solution with thiosulfate on the time parameter that is not normally distributed, as indicated by a p-value of 0.044.
Normality test results
| Variable | Kolmogorov–Smirnov | Shapiro–Wilk | |||||
|---|---|---|---|---|---|---|---|
| Statistic | df | Sig. | Statistic | df | Sig. | ||
| Thiosulphate | Time | 0.358 | 4 | 0.000 | 0.756 | 4 | 0.044 |
| Concentration | 0.268 | 3 | 0.000 | 0.950 | 3 | 0.571 | |
| Thiourea | Time | 0.299 | 4 | 0.000 | 0.926 | 4 | 0.569 |
| Concentration | 0.364 | 3 | 0.000 | 0.799 | 3 | 0.113 | |
The mean difference is significant at the 0.05 level.
Because there are data that are not normally distributed, the Friedman test was carried out to determine the parameters that affect the Au leaching process. Friedman’s test is a nonparametric test for data that are not normally distributed. The results of the Friedman test on the leaching time and concentration parameters presented in Table 5 show that leaching time has a significant effect on the Au leaching process. This is evidenced by the results of calculating the p ≤ 0.050, which means that H0 is accepted (the parameter of leaching time) and has a significant effect. It could be that the concentration parameter does not affect the Au leaching process from solid samples because the p > 0.050 value means that H0 is rejected. This research is relevant to the study by Rui et al. [47], which state that the time parameter is significant in the gold recovery process in a leaching agent [47]. The longer the leaching time, the greater the gold collected. The leaching time parameter plays a more critical role than concentration because there are conditions where less gold will be recovered at a specific concentration. As in the thiosulphate solution, the greater the thiosulphate concentration, the higher the gold recovery results. However, in the thiourea solution, the greater the thiourea concentration, the lower the gold recovery.
Friedman test results
| Friedman test | p Value | Conc. |
|---|---|---|
| Leaching time | 0.046 | Significant |
| Concentration | 0.083 | Not significant |
The mean difference is significant at the 0.050 level.
Using a thiosulphate solution has advantages over using a thiourea solution in extracting gold from the solid. In addition to the effectiveness of thiosulphate in extracting gold, it is also influenced by operational costs and the resulting impact (Table 6). The effectiveness of thiosulphate in extracting gold from solids is due to the strong interaction between thiosulphate and Au by producing a stable [Au(S2O3)2]3− complex. The strong S–Au–S bond results in a stable [Au(S2O3)2]3− complex [39].
Comparison of leaching with thiosulphate and thiourea
| No. | Solution | Ref. | |
|---|---|---|---|
| Thiosulphate | Thiourea | ||
| 1. | [Au(S2O3)2]3− complex is more stable | Au[SC(NH2)2]2 + complex is less stable | [39,48] |
| 2. | More selective for Au | Less selective for Au | [48] |
| 3. | Base system | Acid system | [48] |
| 4. | Non-corrosive to equipment | Corrosive to equipment | [49,50] |
| 5. | Inexpensive | Expensive (more reagents) | [51,52,53] |
4 Conclusions
The gold content contained in the rock sample is 0.0015%. The yield of the gold extract with thiosulphate solution was greater than that with the thiourea solution. Gold extraction at 0.3 M thiosulphate reached 100% and at 0.2 M thiourea reached 58%. The thiosulphate solution had an optimum concentration of 0.3 M and an optimum time of 2 h, while the thiourea solution had an optimum concentration of 0.2 M and an optimum time of 3 h. The results of the Friedman test on the leaching time and concentration parameters show that leaching time has a significant effect on the Au leaching process, and the concentration parameter does not affect the Au leaching process from solid samples. Thiosulphate solution is more effective and environmentally friendly than thiourea solution for the recovery of Au in solids.
-
Funding information: This work was not supported by any funding.
-
Author contributions: All authors contributed equally.
-
Conflict of interest: The authors declare no conflicts of interest.
-
Ethical approval: The conducted research is not related to either human or animal use.
-
Data availability statement: The datasets generated during and/or analysed during the current study are available from the corresponding author upon reasonable request.
References
[1] Marcello MV, Aaron JG. Gravity concentration in artisanal gold mining. Minerals. 2020;10:1026. 10.3390/min10111026.Search in Google Scholar
[2] Louise JE, Justin MC. The mercury problem in artisanal and small‐scale gold mining. Chemistry. 2018;24:6905–16. 10.1002/chem.201704840.Search in Google Scholar PubMed PubMed Central
[3] Erry S. Kompleksitas permasalahan merkuri dalam pengolahan bijih emas berdasarkan perspektif teknis lingkungan manusia dan masa depan. KURVATEK. 2020;5:113–22. 10.33579/krvtk.v5i1.1833.Search in Google Scholar
[4] Birgitte S, Peter WA, Leoncio DN-O, Asta SS, Ivan ZR, Rasmus K-R. Introduction of mercury-free gold extraction to smallscale miners in the Cabo Delgado province in Mozambique. J Health Pollut. 2018;8:1–5. 10.5696/2156-9614-8.19.180909.Search in Google Scholar PubMed PubMed Central
[5] Mahdi B-M, Kobra N, Zoya T, Mohammad RK, Mahmood S. Toxic mechanisms of five heavy metals: Mercury, lead, chromium, cadmium, and arsenic. Front Pharmacol. 2021;12:643972. 10.3389/fphar.2021.643972.Search in Google Scholar PubMed PubMed Central
[6] Jessica B, Emmanuel S, Renald B. Heavy metal pollution in the environment and their toxicological effects on humans. Heliyon. 2020;6:e04691. 10.1016/j.heliyon.2020.e04691.Search in Google Scholar PubMed PubMed Central
[7] Yahya FN, Ibrahim WH, Aziz BA, Suli LN. Simulation of leaching process of gold by cyanidation. IOP Conf Ser Mater Sci Eng (Energy Secur Chem Eng Congr). 2020;738:02211. 10.1088/1757-899X/736/2/022111.Search in Google Scholar
[8] Esthi K, Agus B, Flaminggo G. Recovery of gold with AgNO3 pretreatment by cyanidation at heap leaching cijiwa gold ore processing. Makara J Sci. 2018;22:77–81. 10.7454/mss.v22i2.8248.Search in Google Scholar
[9] Manuel S, Edelmira G, Pedro R, Jonathan C, Norman T. Copper mineral leaching mathematical models-A review. Minerals. 2022;15:1757. 10.3390/ma15051757.Search in Google Scholar PubMed PubMed Central
[10] Yuliusman Y, Amiliana RA, Wulandari PT, Ramadhan IT, Kusumadewi FA. Selection of organic acid leaching reagent for recovery of zinc and manganese from zinc-carbon and alkaline spent batteries. IOP Conf Ser Mater Sci Eng. 2018;333:012041. 10.1088/1757-899X/333/1/012041.Search in Google Scholar
[11] Putri ARY. Analisis kadar logam mekuri (Hg) dan (pH) air sungai kuantan terdampak penambangan emas tanpa izin (PETI). Orbital J Pendidik Kimia. 2018;2:28–36. 10.19109/ojpk.v2i1.2167.Search in Google Scholar
[12] Mufakhir FR, Sinaga JM, Oediyani S, Astuti W. Pelarutan emas pada pelindian konsentrat emas hasil roasting menggunakan reagen tiosianat. J Rekayasa Proses. 2019;13:24–30. 10.22146/jrekpros.41519.Search in Google Scholar
[13] La OA, Fika RM, Hendra S. Studi ekstraksi bijih emas asal pesawaran dengan metode pelindian agitasi dalam larutan sianida. J Sci Appl Technol. 2020;4:103. 10.35472/jsat.v4i2.365.Search in Google Scholar
[14] Franco B, Davide C, Beatrice B, Francesca DF. Reduction of potassium cyanide usage in a consolidated industrial process for gold recovery from wastes and scraps. J Clean Prod. 2017;142:1810–8. 10.1016/j.jclepro.2016.11.103.Search in Google Scholar
[15] Dongzhuang H, Lang L, Qixing Y, Bo Z, Huafu Q, Shishan R, et al. Decomposition of cyanide from gold leaching tailingsby using sodium metabisulphite and hydrogen peroxide. Adv Mater Sci Eng. 2020;2020:5640963. 10.1155/2020/5640963.Search in Google Scholar
[16] Melania HA, Muhamad YH, Dewi R, Eva LN. Analisis scientometrics penelitian merkuri pada penambangan emas skala kecil di Indonesia tahun 2009–2019. J Ilmu Lingkung. 2020;18:185–92. 10.14710/jil.18.1.185-192.Search in Google Scholar
[17] Muhammad YH, Melania HA. Preliminary study: Local government readiness to remove mercury in artisanal and small-scale gold mining. ECOLAB. 2022;16:51–66. 10.20886/jklh.2022.16.2.51-66.Search in Google Scholar
[18] Hilman A, Suripto S, Ahmad J, Immy SR. Impact evaluation of the use of mercury (Hg) and cyanide (Cn) in gold processing activities on lombok island based on knowledge and experience of gold miners and processers on Lombok Island. J Biol Trop. 2022;22:1060–8. 10.29303/jbt.v22i3.4468.Search in Google Scholar
[19] Ajeng Arum Kusuma Di, Haeruddin H. Tinjauan terhadap perencanaan phytomining sebagai alternatif tambang emas ramah lingkungan. J Teknologi Sumber Mineral. 2022;3:81–91. 10.19184/jeneral.v3i2.34358.Search in Google Scholar
[20] Antonkiewicz J, Gworek B. Remediation of contaminated soils and lands. Warsaw: Wydawnictwo Naukowe PWN; 2023. p. 2002.Search in Google Scholar
[21] Sadia I, Jae-chun L. Gold metallurgy and the environment. 1st edn. Florida: CRC Press. 10.1201/9781315150475.Search in Google Scholar
[22] Alexander B, Srecko S, Bernd F. Kinetic investigation and dissolution behavior of cyanide alternative gold leaching reagents. Sci Rep. 2019;9:7191. 10.1038/s41598-019-43383-4.Search in Google Scholar PubMed PubMed Central
[23] Mertol G, Alexander B, Srecko S, Bernd F. A review on alternative gold recovery reagents to cyanide. J Mater Sci Chem Eng. 2016;4:8–17. 10.4236/msce.2016.48002.Search in Google Scholar
[24] Emilia N, Eniko K, Zamfira D, Anamaria IT, Cerasel V, Erika AL. Hydrometallurgical recovery of gold from mining wastes. Strateg Sustain Solid Waste Manag. 2020;2020:94597. 10.5772/intechopen.94597.Search in Google Scholar
[25] Jonovan VY, Naomi JB, Ka YC, Aleksandar NN, Navid RM, Anna HK. E-waste recycling and resource recovery: A review on technologies, barriers, and enablers with a focus on Oceania. Metals. 2021;11:1313. 10.3390/met11081313.Search in Google Scholar
[26] Daniel AR, Mahsa B, John EG, Alan G, Sebastian F. Thiourea leaching: An update on a sustainable approach for gold recovery from E‑waste. J Sustain Metall. 2022;8:597–612. 10.1007/s40831-022-00499-8.Search in Google Scholar
[27] Hesham M, Mohammed R, Mohamed GE-F, Yasser KA-M. Gold leaching using thiourea from uranium tailing material, gabal el-missikat, central eastern desert, Egypt. J Sustain Metall. 2020;6:599–611. 10.1007/s40831-020-00295-2.Search in Google Scholar
[28] Mark GA, David MM. Thiosulfate leaching of gold—A review. Miner Eng. 2001;14:135–74. 10.1016/S0892-6875(00)00172-2.Search in Google Scholar
[29] Jeanne PM, Jean BH, Gbetoglo CK, Placide U, Himanshu T, Grace O-S, et al. Optimization of gold dissolution parameters in acidified thiourea leaching solution with hydrogen peroxide as an oxidant: Implications of roasting pretreatment technology. Metals. 2022;12:1567. 10.3390/met12101567.Search in Google Scholar
[30] Hong Q, Xueyi G, Qinghua T, Dawei Y, Lei Z. Recovery of gold from sulfide refractory gold ore: Oxidation roasting pretreatment and gold extraction. Miner Eng. 2021;164:106822. 10.1016/j.mineng.2021.106822.Search in Google Scholar
[31] Oluwasanmi ST, Abraham AA, Simeon AI, Moshood DS. Leaching of a Nigerian refractory gold ore using aqua regia. Am J Eng Appl Sci. 2022;15:118–25. 10.3844/ajeassp.2022.118.125.Search in Google Scholar
[32] Erlina Y, Ardi G, Tatang W. Ekstraksi bijih emas sulfida tatelu minahasa utara menggunakan reagen ramah lingkungan tiosulfat. J TEKNIKA. 2018;12:97–106. 10.36055/tjst.v14i2.5865.Search in Google Scholar
[33] Piotr C, Kamil G, Anna L, Joanna MW, Pawel P, Dorota J-B. The recovery of gold from the aqua regia leachate of electronic parts using a core–shell type anion exchange resin. J Saudi Chem Soc. 2017;21:741–50. 10.1016/j.jscs.2017.03.007.Search in Google Scholar
[34] Mooki B, Jae-Chun L, Hyunju L, Sookyung K. Recovery of nitric acid and gold from gold-bearing aqua regia by tributyl-phosphate. Sep Purif Technol. 2020;235:116154. 10.1016/j.seppur.2019.116154.Search in Google Scholar
[35] Bin X, Wenhao K, Qian L, Yongbin Y, Tao J, Xiaoliang L. A review of thiosulfate leaching of gold: Focus on thiosulfate consumption and gold recovery from pregnant solution. Metals. 2017;7:222. 10.3390/met7060222.Search in Google Scholar
[36] Szabolcs F, Florica I-L, Petru I. Metals leaching from waste printed circuit boards. Part Ii: Influence Of thiourea, thiosulfate and thiocyanate concentration on the leaching process. Studia UBB Chem. 2012;LVII:41–9.Search in Google Scholar
[37] Yunji D, Huandong Z, Jiayi L, Shengen Z, Bo L, Christian E. An efficient leaching of palladium from spent catalysts through oxidation with Fe(III). Materials. 2019;12:1205. 10.3390/ma12081205.Search in Google Scholar PubMed PubMed Central
[38] Tran TT, Lee MS. Use Of ferric salt solutions as leaching agents of Co, Ni, Cu, Fe, and Mn from metallic alloys of spent lithium-ion batteries and separation of iron from the leaching solution. J Min Metall Sect B-Metall. 2022;58:405–15. 10.2298/JMMB220311023T.Search in Google Scholar
[39] Feng X, Jun-nan C, Jian W, Wei W. Review of gold leaching in thiosulfate-based solutions. Trans Nonferrous Met Soc China. 2021;31:3506–29. 10.1016/S1003-6326(21)65745-X.Search in Google Scholar
[40] Yasin Z, Sima M, Mahmood A. Gold passivation by sulfur species: A molecular picture. Miner Eng. 2019;134:215–21. 10.1016/j.mineng.2019.02.009.Search in Google Scholar
[41] Kanghee C, Hyunsoo K, Eunji M, Oyunbileg P, Nagchoul C, Cheonyoung P. Recovery of gold from the refractory gold concentrate using microwave assisted leaching. Metals. 2020;10:571. 10.3390/met10050571.Search in Google Scholar
[42] Chun-bao S, Xiao-liang Z, Jue K, Yi X. A review of gold extraction using noncyanide lixiviants: Fundamentals, advancements, and challenges toward alkaline sulfur-containing leaching agents. Int J Mineral Metall Mater. 2020;27:417–31. 10.1007/s12613-019-1955-x.Search in Google Scholar
[43] Michael LP, Daniel JM, Chaterine FW, Anna CB, Samantha IJ, Lewis EJ, et al. Mechanism of Catalytic O2 Reduction by Iron Tetraphenylporphyrin. J Am Chem Soc. 2019;141:8315–26. https://pubs.acs.org/doi/10.1021/jacs.9b02640.10.1021/jacs.9b02640Search in Google Scholar PubMed PubMed Central
[44] Markus RA, Stefan H, Alexander P, Mirza C, Valerio D, Manuel PH, et al. Binding of molecular oxygen by an artificial heme analogue: investigation on the formation of an Fe–tetracarbene superoxo complex. Dalton Trans. 2016;45:6449–55. 10.1039/c6dt00538a.Search in Google Scholar PubMed
[45] Prabhaker M, Chandra MP, Uttam S, Anshul G, Chinmoy S, Amit K. Descriptive statistics and normality tests for statistical data. Ann Card Anaesth. 2019;22:67–72. 10.4103/aca.ACA_157_18.Search in Google Scholar PubMed PubMed Central
[46] Shengping Y, Gilbert B. Normality tests. Southwest Respir Crit Care Chron. 2021;9:87–90. 10.12746/swrccc.v9i37.805.Search in Google Scholar
[47] Rui X, Xiaolong N, Feiyu M, Qian L, Xuling C, Yongbin Y, et al. Analysis and prediction of the thiourea gold leaching process using grey relational analysis and artificial neural networks. Minerals. 2020;10:811. 10.3390/min10090811.Search in Google Scholar
[48] Na Z, Jue K, Chunbao S. Investigation on Gold–Ligand interaction for complexes from gold leaching: A DFT study. Molecules. 2023;28:1508. 10.3390/molecules28031508.Search in Google Scholar PubMed PubMed Central
[49] Sadia I, Rajiv RS, Hyungjung K. Gold recovery from secondary waste of PCBs by electro-Cl2 leaching in brine solution and solvo-chemical separation with tri-butyl phosphate. J Clean Prod. 2021;295:126389. 10.1016/j.jclepro.2021.126389.Search in Google Scholar
[50] Song W, Lei L, ShiDing W, Heng W, GuoDong W. Extraction of platinum and gold from copper anode slimes by a process of chlorinating roasting followed by chlorinating leaching. J Min Met Sect B Met. 2020;56:193–202. 10.2298/JMMB190915015W.Search in Google Scholar
[51] Lianqun L, Changwei P, Jinju S, Weibing S, Xinyu Y, Chen J, et al. Pit-induced electrochemical layer dissolution and wave propagation on an Au(111) surface in an acidic thiourea solution. J Phys Chem C. 2020;124:19112–18. 10.1021/acs.jpcc.0c05329.Search in Google Scholar
[52] Wen-Juan L, He Z, An-Ping B, Yong-Sheng S, Liu-Lu C, Shui-Lin Z, et al. Electrochemical adsorption and passivation on gold surface in alkaline thiourea solutions. Rare Met. 2020;39:951–8. 10.1007/s12598-020-01422-4.Search in Google Scholar
[53] Bingqiong Y, Yanhong L, Xian P, Siyu H, Gangcheng Z, Kun Y, et al. Synthesis, characterization, and antitumor properties of Au(i)-thiourea complexes. Metallomics. 2020;12:104–13. 10.1039/C9MT00232D.Search in Google Scholar
© 2023 the author(s), published by De Gruyter
This work is licensed under the Creative Commons Attribution 4.0 International License.
Articles in the same Issue
- Characteristics, source, and health risk assessment of aerosol polyaromatic hydrocarbons in the rural and urban regions of western Saudi Arabia
- Regular Articles
- A network-based correlation research between element electronegativity and node importance
- Pomegranate attenuates kidney injury in cyclosporine-induced nephrotoxicity in rats by suppressing oxidative stress
- Ab initio study of fundamental properties of XInO3 (X = K, Rb, Cs) perovskites
- Responses of feldspathic sandstone and sand-reconstituted soil C and N to freeze–thaw cycles
- Robust fractional control based on high gain observers design (RNFC) for a Spirulina maxima culture interfaced with an advanced oxidation process
- Study on arsenic speciation and redistribution mechanism in Lonicera japonica plants via synchrotron techniques
- Optimization of machining Nilo 36 superalloy parameters in turning operation
- Vacuum impregnation pre-treatment: A novel method for incorporating mono- and divalent cations into potato strips to reduce the acrylamide formation in French fries
- Characterization of effective constituents in Acanthopanax senticosus fruit for blood deficiency syndrome based on the chinmedomics strategy
- Comparative analysis of the metabolites in Pinellia ternata from two producing regions using ultra-high-performance liquid chromatography–electrospray ionization–tandem mass spectrometry
- The assessment of environmental parameter along the desalination plants in the Kingdom of Saudi Arabia
- Effects of harpin and carbendazim on antioxidant accumulation in young jujube leaves
- The effects of in ovo injected with sodium borate on hatching performance and small intestine morphology in broiler chicks
- Optimization of cutting forces and surface roughness via ANOVA and grey relational analysis in machining of In718
- Essential oils of Origanum compactum Benth: Chemical characterization, in vitro, in silico, antioxidant, and antibacterial activities
- Translocation of tungsten(vi) oxide/gadolinium(iii) fluoride in tellurite glasses towards improvement of gamma-ray attenuation features in high-density glass shields
- Mechanical properties, elastic moduli, and gamma ray attenuation competencies of some TeO2–WO3–GdF3 glasses: Tailoring WO3–GdF3 substitution toward optimum behavioral state range
- Comparison between the CIDR or sponge with hormone injection to induce estrus synchronization for twining and sex preselection in Naimi sheep
- Exergetic performance analyses of three different cogeneration plants
- Psoralea corylifolia (babchi) seeds enhance proliferation of normal human cultured melanocytes: GC–MS profiling and biological investigation
- A novel electrochemical micro-titration method for quantitative evaluation of the DPPH free radical scavenging capacity of caffeic acid
- Comparative study between supported bimetallic catalysts for nitrate remediation in water
- Persicaline, an alkaloid from Salvadora persica, inhibits proliferation and induces apoptosis and cell-cycle arrest in MCF-7 cells
- Determination of nicotine content in locally produced smokeless tobacco (Shammah) samples from Jazan region of Saudi Arabia using a convenient HPLC-MS/MS method
- Changes in oxidative stress markers in pediatric burn injury over a 1-week period
- Integrated geophysical techniques applied for petroleum basins structural characterization in the central part of the Western Desert, Egypt
- The impact of chemical modifications on gamma-ray attenuation properties of some WO3-reinforced tellurite glasses
- Microwave and Cs+-assisted chemo selective reaction protocol for synthesizing 2-styryl quinoline biorelevant molecules
- Structural, physical, and radiation absorption properties of a significant nuclear power plant component: A comparison between REX-734 and 316L SS austenitic stainless steels
- Effect of Moringa oleifera on serum YKL-40 level: In vivo rat periodontitis model
- Investigating the impact of CO2 emissions on the COVID-19 pandemic by generalized linear mixed model approach with inverse Gaussian and gamma distributions
- Influence of WO3 content on gamma rays attenuation characteristics of phosphate glasses at low energy range
- Study on CO2 absorption performance of ternary DES formed based on DEA as promoting factor
- Performance analyses of detonation engine cogeneration cycles
- Sterols from Centaurea pumilio L. with cell proliferative activity: In vitro and in silico studies
- Untargeted metabolomics revealing changes in aroma substances in flue-cured tobacco
- Effect of pumpkin enriched with calcium lactate on iron status in an animal model of postmenopausal osteoporosis
- Energy consumption, mechanical and metallographic properties of cryogenically treated tool steels
- Optimization of ultra-high pressure-assisted extraction of total phenols from Eucommia ulmoides leaves by response surface methodology
- Harpin enhances antioxidant nutrient accumulation and decreases enzymatic browning in stored soybean sprouts
- Physicochemical and biological properties of carvacrol
- Radix puerariae in the treatment of diabetic nephropathy: A network pharmacology analysis and experimental validation
- Anti-Alzheimer, antioxidants, glucose-6-phosphate dehydrogenase effects of Taverniera glabra mediated ZnO and Fe2O3 nanoparticles in alloxan-induced diabetic rats
- Experimental study on photocatalytic CO2 reduction performance of ZnS/CdS-TiO2 nanotube array thin films
- Epoxy-reinforced heavy metal oxides for gamma ray shielding purposes
- Black mulberry (Morus nigra L.) fruits: As a medicinal plant rich in human health-promoting compounds
- Promising antioxidant and antimicrobial effects of essential oils extracted from fruits of Juniperus thurifera: In vitro and in silico investigations
- Chloramine-T-induced oxidation of Rizatriptan Benzoate: An integral chemical and spectroscopic study of products, mechanisms and kinetics
- Study on antioxidant and antimicrobial potential of chemically profiled essential oils extracted from Juniperus phoenicea (L.) by use of in vitro and in silico approaches
- Screening and characterization of fungal taxol-producing endophytic fungi for evaluation of antimicrobial and anticancer activities
- Mineral composition, principal polyphenolic components, and evaluation of the anti-inflammatory, analgesic, and antioxidant properties of Cytisus villosus Pourr leaf extracts
- In vitro antiproliferative efficacy of Annona muricata seed and fruit extracts on several cancer cell lines
- An experimental study for chemical characterization of artificial anterior cruciate ligament with coated chitosan as biomaterial
- Prevalence of residual risks of the transfusion-transmitted infections in Riyadh hospitals: A two-year retrospective study
- Computational and experimental investigation of antibacterial and antifungal properties of Nicotiana tabacum extracts
- Reinforcement of cementitious mortars with hemp fibers and shives
- X-ray shielding properties of bismuth-borate glass doped with rare earth ions
- Green supported silver nanoparticles over modified reduced graphene oxide: Investigation of its antioxidant and anti-ovarian cancer effects
- Orthogonal synthesis of a versatile building block for dual functionalization of targeting vectors
- Thymbra spicata leaf extract driven biogenic synthesis of Au/Fe3O4 nanocomposite and its bio-application in the treatment of different types of leukemia
- The role of Ag2O incorporation in nuclear radiation shielding behaviors of the Li2O–Pb3O4–SiO2 glass system: A multi-step characterization study
- A stimuli-responsive in situ spray hydrogel co-loaded with naringenin and gentamicin for chronic wounds
- Assessment of the impact of γ-irradiation on the piperine content and microbial quality of black pepper
- Antioxidant, sensory, and functional properties of low-alcoholic IPA beer with Pinus sylvestris L. shoots addition fermented using unconventional yeast
- Screening and optimization of extracellular pectinase produced by Bacillus thuringiensis SH7
- Determination of polyphenols in Chinese jujube using ultra-performance liquid chromatography–mass spectrometry
- Synergistic effects of harpin and NaCl in determining soybean sprout quality under non-sterile conditions
- Field evaluation of different eco-friendly alternative control methods against Panonychus citri [Acari: Tetranychidae] spider mite and its predators in citrus orchards
- Exploring the antimicrobial potential of biologically synthesized zero valent iron nanoparticles
- NaCl regulates goldfish growth and survival at three food supply levels under hypoxia
- An exploration of the physical, optical, mechanical, and radiation shielding properties of PbO–MgO–ZnO–B2O3 glasses
- A novel statistical modeling of air pollution and the COVID-19 pandemic mortality data by Poisson, geometric, and negative binomial regression models with fixed and random effects
- Treatment activity of the injectable hydrogels loaded with dexamethasone In(iii) complex on glioma by inhibiting the VEGF signaling pathway
- An alternative approach for the excess lifetime cancer risk and prediction of radiological parameters
- Panax ginseng leaf aqueous extract mediated green synthesis of AgNPs under ultrasound condition and investigation of its anti-lung adenocarcinoma effects
- Study of hydrolysis and production of instant ginger (Zingiber officinale) tea
- Novel green synthesis of zinc oxide nanoparticles using Salvia rosmarinus extract for treatment of human lung cancer
- Evaluation of second trimester plasma lipoxin A4, VEGFR-1, IL-6, and TNF-α levels in pregnant women with gestational diabetes mellitus
- Antidiabetic, antioxidant and cytotoxicity activities of ortho- and para-substituted Schiff bases derived from metformin hydrochloride: Validation by molecular docking and in silico ADME studies
- Antioxidant, antidiabetic, antiglaucoma, and anticholinergic effects of Tayfi grape (Vitis vinifera): A phytochemical screening by LC-MS/MS analysis
- Identification of genetic polymorphisms in the stearoyl CoA desaturase gene and its association with milk quality traits in Najdi sheep
- Cold-acclimation effect on cadmium absorption and biosynthesis of polyphenolics, and free proline and photosynthetic pigments in Spirogyra aequinoctialis
- Analysis of secondary metabolites in Xinjiang Morus nigra leaves using different extraction methods with UPLC-Q/TOF-MS/MS technology
- Nanoarchitectonics and performance evaluation of a Fe3O4-stabilized Pickering emulsion-type differential pressure plugging agent
- Investigating pyrolysis characteristics of Shengdong coal through Py-GC/MS
- Extraction, phytochemical characterization, and antifungal activity of Salvia rosmarinus extract
- Introducing a novel and natural antibiotic for the treatment of oral pathogens: Abelmoschus esculentus green-formulated silver nanoparticles
- Optimization of gallic acid-enriched ultrasonic-assisted extraction from mango peels
- Effect of gamma rays irradiation in the structure, optical, and electrical properties of samarium doped bismuth titanate ceramics
- Combinatory in silico investigation for potential inhibitors from Curcuma sahuynhensis Škorničk. & N.S. Lý volatile phytoconstituents against influenza A hemagglutinin, SARS-CoV-2 main protease, and Omicron-variant spike protein
- Physical, mechanical, and gamma ray shielding properties of the Bi2O3–BaO–B2O3–ZnO–As2O3–MgO–Na2O glass system
- Twofold interpenetrated 3D Cd(ii) complex: Crystal structure and luminescent property
- Study on the microstructure and soil quality variation of composite soil with soft rock and sand
- Ancient spring waters still emerging and accessible in the Roman Forum area: Chemical–physical and microbiological characterization
- Extraction and characterization of type I collagen from scales of Mexican Biajaiba fish
- Finding small molecular compounds to decrease trimethylamine oxide levels in atherosclerosis by virtual screening
- Prefatory in silico studies and in vitro insecticidal effect of Nigella sativa (L.) essential oil and its active compound (carvacrol) against the Callosobruchus maculatus adults (Fab), a major pest of chickpea
- Polymerized methyl imidazole silver bromide (CH3C6H5AgBr)6: Synthesis, crystal structures, and catalytic activity
- Using calcined waste fish bones as a green solid catalyst for biodiesel production from date seed oil
- Influence of the addition of WO3 on TeO2–Na2O glass systems in view of the feature of mechanical, optical, and photon attenuation
- Naringin ameliorates 5-fluorouracil elicited neurotoxicity by curtailing oxidative stress and iNOS/NF-ĸB/caspase-3 pathway
- GC-MS profile of extracts of an endophytic fungus Alternaria and evaluation of its anticancer and antibacterial potentialities
- Green synthesis, chemical characterization, and antioxidant and anti-colorectal cancer effects of vanadium nanoparticles
- Determination of caffeine content in coffee drinks prepared in some coffee shops in the local market in Jeddah City, Saudi Arabia
- A new 3D supramolecular Cu(ii) framework: Crystal structure and photocatalytic characteristics
- Bordeaux mixture accelerates ripening, delays senescence, and promotes metabolite accumulation in jujube fruit
- Important application value of injectable hydrogels loaded with omeprazole Schiff base complex in the treatment of pancreatitis
- Color tunable benzothiadiazole-based small molecules for lightening applications
- Investigation of structural, dielectric, impedance, and mechanical properties of hydroxyapatite-modified barium titanate composites for biomedical applications
- Metal gel particles loaded with epidermal cell growth factor promote skin wound repair mechanism by regulating miRNA
- In vitro exploration of Hypsizygus ulmarius (Bull.) mushroom fruiting bodies: Potential antidiabetic and anti-inflammatory agent
- Alteration in the molecular structure of the adenine base exposed to gamma irradiation: An ESR study
- Comprehensive study of optical, thermal, and gamma-ray shielding properties of Bi2O3–ZnO–PbO–B2O3 glasses
- Lewis acids as co-catalysts in Pd-based catalyzed systems of the octene-1 hydroethoxycarbonylation reaction
- Synthesis, Hirshfeld surface analysis, thermal, and selective α-glucosidase inhibitory studies of Schiff base transition metal complexes
- Protective properties of AgNPs green-synthesized by Abelmoschus esculentus on retinal damage on the virtue of its anti-inflammatory and antioxidant effects in diabetic rat
- Effects of green decorated AgNPs on lignin-modified magnetic nanoparticles mediated by Cydonia on cecal ligation and puncture-induced sepsis
- Treatment of gastric cancer by green mediated silver nanoparticles using Pistacia atlantica bark aqueous extract
- Preparation of newly developed porcelain ceramics containing WO3 nanoparticles for radiation shielding applications
- Utilization of computational methods for the identification of new natural inhibitors of human neutrophil elastase in inflammation therapy
- Some anticancer agents as effective glutathione S-transferase (GST) inhibitors
- Clay-based bricks’ rich illite mineral for gamma-ray shielding applications: An experimental evaluation of the effect of pressure rates on gamma-ray attenuation parameters
- Stability kinetics of orevactaene pigments produced by Epicoccum nigrum in solid-state fermentation
- Treatment of denture stomatitis using iron nanoparticles green-synthesized by Silybum marianum extract
- Characterization and antioxidant potential of white mustard (Brassica hirta) leaf extract and stabilization of sunflower oil
- Characteristics of Langmuir monomolecular monolayers formed by the novel oil blends
- Strategies for optimizing the single GdSrFeO4 phase synthesis
- Oleic acid and linoleic acid nanosomes boost immunity and provoke cell death via the upregulation of beta-defensin-4 at genetic and epigenetic levels
- Unraveling the therapeutic potential of Bombax ceiba roots: A comprehensive study of chemical composition, heavy metal content, antibacterial activity, and in silico analysis
- Green synthesis of AgNPs using plant extract and investigation of its anti-human colorectal cancer application
- The adsorption of naproxen on adsorbents obtained from pepper stalk extract by green synthesis
- Treatment of gastric cancer by silver nanoparticles encapsulated by chitosan polymers mediated by Pistacia atlantica extract under ultrasound condition
- In vitro protective and anti-inflammatory effects of Capparis spinosa and its flavonoids profile
- Wear and corrosion behavior of TiC and WC coatings deposited on high-speed steels by electro-spark deposition
- Therapeutic effects of green-formulated gold nanoparticles by Origanum majorana on spinal cord injury in rats
- Melanin antibacterial activity of two new strains, SN1 and SN2, of Exophiala phaeomuriformis against five human pathogens
- Evaluation of the analgesic and anesthetic properties of silver nanoparticles supported over biodegradable acacia gum-modified magnetic nanoparticles
- Review Articles
- Role and mechanism of fruit waste polyphenols in diabetes management
- A comprehensive review of non-alkaloidal metabolites from the subfamily Amaryllidoideae (Amaryllidaceae)
- Discovery of the chemical constituents, structural characteristics, and pharmacological functions of Chinese caterpillar fungus
- Eco-friendly green approach of nickel oxide nanoparticles for biomedical applications
- Advances in the pharmaceutical research of curcumin for oral administration
- Rapid Communication
- Determination of the contents of bioactive compounds in St. John’s wort (Hypericum perforatum): Comparison of commercial and wild samples
- Retraction
- Retraction of “Two mixed-ligand coordination polymers based on 2,5-thiophenedicarboxylic acid and flexible N-donor ligands: The protective effect on periodontitis via reducing the release of IL-1β and TNF-α”
- Topical Issue on Phytochemicals, biological and toxicological analysis of aromatic medicinal plants
- Anti-plasmodial potential of selected medicinal plants and a compound Atropine isolated from Eucalyptus obliqua
- Anthocyanin extract from black rice attenuates chronic inflammation in DSS-induced colitis mouse model by modulating the gut microbiota
- Evaluation of antibiofilm and cytotoxicity effect of Rumex vesicarius methanol extract
- Chemical compositions of Litsea umbellata and inhibition activities
- Green synthesis, characterization of silver nanoparticles using Rhynchosia capitata leaf extract and their biological activities
- GC-MS analysis and antibacterial activities of some plants belonging to the genus Euphorbia on selected bacterial isolates
- The abrogative effect of propolis on acrylamide-induced toxicity in male albino rats: Histological study
- A phytoconstituent 6-aminoflavone ameliorates lipopolysaccharide-induced oxidative stress mediated synapse and memory dysfunction via p-Akt/NF-kB pathway in albino mice
- Anti-diabetic potentials of Sorbaria tomentosa Lindl. Rehder: Phytochemistry (GC-MS analysis), α-amylase, α-glucosidase inhibitory, in vivo hypoglycemic, and biochemical analysis
- Assessment of cytotoxic and apoptotic activities of the Cassia angustifolia aqueous extract against SW480 colon cancer
- Biochemical analysis, antioxidant, and antibacterial efficacy of the bee propolis extract (Hymenoptera: Apis mellifera) against Staphylococcus aureus-induced infection in BALB/c mice: In vitro and in vivo study
- Assessment of essential elements and heavy metals in Saudi Arabian rice samples underwent various processing methods
- Two new compounds from leaves of Capparis dongvanensis (Sy, B. H. Quang & D. V. Hai) and inhibition activities
- Hydroxyquinoline sulfanilamide ameliorates STZ-induced hyperglycemia-mediated amyleoid beta burden and memory impairment in adult mice
- An automated reading of semi-quantitative hemagglutination results in microplates: Micro-assay for plant lectins
- Inductively coupled plasma mass spectrometry assessment of essential and toxic trace elements in traditional spices consumed by the population of the Middle Eastern region in their recipes
- Phytochemical analysis and anticancer activity of the Pithecellobium dulce seed extract in colorectal cancer cells
- Impact of climatic disturbances on the chemical compositions and metabolites of Salvia officinalis
- Physicochemical characterization, antioxidant and antifungal activities of essential oils of Urginea maritima and Allium sativum
- Phytochemical analysis and antifungal efficiency of Origanum majorana extracts against some phytopathogenic fungi causing tomato damping-off diseases
- Special Issue on 4th IC3PE
- Graphene quantum dots: A comprehensive overview
- Studies on the intercalation of calcium–aluminium layered double hydroxide-MCPA and its controlled release mechanism as a potential green herbicide
- Synergetic effect of adsorption and photocatalysis by zinc ferrite-anchored graphitic carbon nitride nanosheet for the removal of ciprofloxacin under visible light irradiation
- Exploring anticancer activity of the Indonesian guava leaf (Psidium guajava L.) fraction on various human cancer cell lines in an in vitro cell-based approach
- The comparison of gold extraction methods from the rock using thiourea and thiosulfate
- Special Issue on Marine environmental sciences and significance of the multidisciplinary approaches
- Sorption of alkylphenols and estrogens on microplastics in marine conditions
- Cytotoxic ketosteroids from the Red Sea soft coral Dendronephthya sp.
- Antibacterial and biofilm prevention metabolites from Acanthophora spicifera
- Characteristics, source, and health risk assessment of aerosol polyaromatic hydrocarbons in the rural and urban regions of western Saudi Arabia
- Special Issue on Advanced Nanomaterials for Energy, Environmental and Biological Applications - Part II
- Green synthesis, characterization, and evaluation of antibacterial activities of cobalt nanoparticles produced by marine fungal species Periconia prolifica
- Combustion-mediated sol–gel preparation of cobalt-doped ZnO nanohybrids for the degradation of acid red and antibacterial performance
- Perinatal supplementation with selenium nanoparticles modified with ascorbic acid improves hepatotoxicity in rat gestational diabetes
- Evaluation and chemical characterization of bioactive secondary metabolites from endophytic fungi associated with the ethnomedicinal plant Bergenia ciliata
- Enhancing photovoltaic efficiency with SQI-Br and SQI-I sensitizers: A comparative analysis
- Nanostructured p-PbS/p-CuO sulfide/oxide bilayer heterojunction as a promising photoelectrode for hydrogen gas generation
Articles in the same Issue
- Characteristics, source, and health risk assessment of aerosol polyaromatic hydrocarbons in the rural and urban regions of western Saudi Arabia
- Regular Articles
- A network-based correlation research between element electronegativity and node importance
- Pomegranate attenuates kidney injury in cyclosporine-induced nephrotoxicity in rats by suppressing oxidative stress
- Ab initio study of fundamental properties of XInO3 (X = K, Rb, Cs) perovskites
- Responses of feldspathic sandstone and sand-reconstituted soil C and N to freeze–thaw cycles
- Robust fractional control based on high gain observers design (RNFC) for a Spirulina maxima culture interfaced with an advanced oxidation process
- Study on arsenic speciation and redistribution mechanism in Lonicera japonica plants via synchrotron techniques
- Optimization of machining Nilo 36 superalloy parameters in turning operation
- Vacuum impregnation pre-treatment: A novel method for incorporating mono- and divalent cations into potato strips to reduce the acrylamide formation in French fries
- Characterization of effective constituents in Acanthopanax senticosus fruit for blood deficiency syndrome based on the chinmedomics strategy
- Comparative analysis of the metabolites in Pinellia ternata from two producing regions using ultra-high-performance liquid chromatography–electrospray ionization–tandem mass spectrometry
- The assessment of environmental parameter along the desalination plants in the Kingdom of Saudi Arabia
- Effects of harpin and carbendazim on antioxidant accumulation in young jujube leaves
- The effects of in ovo injected with sodium borate on hatching performance and small intestine morphology in broiler chicks
- Optimization of cutting forces and surface roughness via ANOVA and grey relational analysis in machining of In718
- Essential oils of Origanum compactum Benth: Chemical characterization, in vitro, in silico, antioxidant, and antibacterial activities
- Translocation of tungsten(vi) oxide/gadolinium(iii) fluoride in tellurite glasses towards improvement of gamma-ray attenuation features in high-density glass shields
- Mechanical properties, elastic moduli, and gamma ray attenuation competencies of some TeO2–WO3–GdF3 glasses: Tailoring WO3–GdF3 substitution toward optimum behavioral state range
- Comparison between the CIDR or sponge with hormone injection to induce estrus synchronization for twining and sex preselection in Naimi sheep
- Exergetic performance analyses of three different cogeneration plants
- Psoralea corylifolia (babchi) seeds enhance proliferation of normal human cultured melanocytes: GC–MS profiling and biological investigation
- A novel electrochemical micro-titration method for quantitative evaluation of the DPPH free radical scavenging capacity of caffeic acid
- Comparative study between supported bimetallic catalysts for nitrate remediation in water
- Persicaline, an alkaloid from Salvadora persica, inhibits proliferation and induces apoptosis and cell-cycle arrest in MCF-7 cells
- Determination of nicotine content in locally produced smokeless tobacco (Shammah) samples from Jazan region of Saudi Arabia using a convenient HPLC-MS/MS method
- Changes in oxidative stress markers in pediatric burn injury over a 1-week period
- Integrated geophysical techniques applied for petroleum basins structural characterization in the central part of the Western Desert, Egypt
- The impact of chemical modifications on gamma-ray attenuation properties of some WO3-reinforced tellurite glasses
- Microwave and Cs+-assisted chemo selective reaction protocol for synthesizing 2-styryl quinoline biorelevant molecules
- Structural, physical, and radiation absorption properties of a significant nuclear power plant component: A comparison between REX-734 and 316L SS austenitic stainless steels
- Effect of Moringa oleifera on serum YKL-40 level: In vivo rat periodontitis model
- Investigating the impact of CO2 emissions on the COVID-19 pandemic by generalized linear mixed model approach with inverse Gaussian and gamma distributions
- Influence of WO3 content on gamma rays attenuation characteristics of phosphate glasses at low energy range
- Study on CO2 absorption performance of ternary DES formed based on DEA as promoting factor
- Performance analyses of detonation engine cogeneration cycles
- Sterols from Centaurea pumilio L. with cell proliferative activity: In vitro and in silico studies
- Untargeted metabolomics revealing changes in aroma substances in flue-cured tobacco
- Effect of pumpkin enriched with calcium lactate on iron status in an animal model of postmenopausal osteoporosis
- Energy consumption, mechanical and metallographic properties of cryogenically treated tool steels
- Optimization of ultra-high pressure-assisted extraction of total phenols from Eucommia ulmoides leaves by response surface methodology
- Harpin enhances antioxidant nutrient accumulation and decreases enzymatic browning in stored soybean sprouts
- Physicochemical and biological properties of carvacrol
- Radix puerariae in the treatment of diabetic nephropathy: A network pharmacology analysis and experimental validation
- Anti-Alzheimer, antioxidants, glucose-6-phosphate dehydrogenase effects of Taverniera glabra mediated ZnO and Fe2O3 nanoparticles in alloxan-induced diabetic rats
- Experimental study on photocatalytic CO2 reduction performance of ZnS/CdS-TiO2 nanotube array thin films
- Epoxy-reinforced heavy metal oxides for gamma ray shielding purposes
- Black mulberry (Morus nigra L.) fruits: As a medicinal plant rich in human health-promoting compounds
- Promising antioxidant and antimicrobial effects of essential oils extracted from fruits of Juniperus thurifera: In vitro and in silico investigations
- Chloramine-T-induced oxidation of Rizatriptan Benzoate: An integral chemical and spectroscopic study of products, mechanisms and kinetics
- Study on antioxidant and antimicrobial potential of chemically profiled essential oils extracted from Juniperus phoenicea (L.) by use of in vitro and in silico approaches
- Screening and characterization of fungal taxol-producing endophytic fungi for evaluation of antimicrobial and anticancer activities
- Mineral composition, principal polyphenolic components, and evaluation of the anti-inflammatory, analgesic, and antioxidant properties of Cytisus villosus Pourr leaf extracts
- In vitro antiproliferative efficacy of Annona muricata seed and fruit extracts on several cancer cell lines
- An experimental study for chemical characterization of artificial anterior cruciate ligament with coated chitosan as biomaterial
- Prevalence of residual risks of the transfusion-transmitted infections in Riyadh hospitals: A two-year retrospective study
- Computational and experimental investigation of antibacterial and antifungal properties of Nicotiana tabacum extracts
- Reinforcement of cementitious mortars with hemp fibers and shives
- X-ray shielding properties of bismuth-borate glass doped with rare earth ions
- Green supported silver nanoparticles over modified reduced graphene oxide: Investigation of its antioxidant and anti-ovarian cancer effects
- Orthogonal synthesis of a versatile building block for dual functionalization of targeting vectors
- Thymbra spicata leaf extract driven biogenic synthesis of Au/Fe3O4 nanocomposite and its bio-application in the treatment of different types of leukemia
- The role of Ag2O incorporation in nuclear radiation shielding behaviors of the Li2O–Pb3O4–SiO2 glass system: A multi-step characterization study
- A stimuli-responsive in situ spray hydrogel co-loaded with naringenin and gentamicin for chronic wounds
- Assessment of the impact of γ-irradiation on the piperine content and microbial quality of black pepper
- Antioxidant, sensory, and functional properties of low-alcoholic IPA beer with Pinus sylvestris L. shoots addition fermented using unconventional yeast
- Screening and optimization of extracellular pectinase produced by Bacillus thuringiensis SH7
- Determination of polyphenols in Chinese jujube using ultra-performance liquid chromatography–mass spectrometry
- Synergistic effects of harpin and NaCl in determining soybean sprout quality under non-sterile conditions
- Field evaluation of different eco-friendly alternative control methods against Panonychus citri [Acari: Tetranychidae] spider mite and its predators in citrus orchards
- Exploring the antimicrobial potential of biologically synthesized zero valent iron nanoparticles
- NaCl regulates goldfish growth and survival at three food supply levels under hypoxia
- An exploration of the physical, optical, mechanical, and radiation shielding properties of PbO–MgO–ZnO–B2O3 glasses
- A novel statistical modeling of air pollution and the COVID-19 pandemic mortality data by Poisson, geometric, and negative binomial regression models with fixed and random effects
- Treatment activity of the injectable hydrogels loaded with dexamethasone In(iii) complex on glioma by inhibiting the VEGF signaling pathway
- An alternative approach for the excess lifetime cancer risk and prediction of radiological parameters
- Panax ginseng leaf aqueous extract mediated green synthesis of AgNPs under ultrasound condition and investigation of its anti-lung adenocarcinoma effects
- Study of hydrolysis and production of instant ginger (Zingiber officinale) tea
- Novel green synthesis of zinc oxide nanoparticles using Salvia rosmarinus extract for treatment of human lung cancer
- Evaluation of second trimester plasma lipoxin A4, VEGFR-1, IL-6, and TNF-α levels in pregnant women with gestational diabetes mellitus
- Antidiabetic, antioxidant and cytotoxicity activities of ortho- and para-substituted Schiff bases derived from metformin hydrochloride: Validation by molecular docking and in silico ADME studies
- Antioxidant, antidiabetic, antiglaucoma, and anticholinergic effects of Tayfi grape (Vitis vinifera): A phytochemical screening by LC-MS/MS analysis
- Identification of genetic polymorphisms in the stearoyl CoA desaturase gene and its association with milk quality traits in Najdi sheep
- Cold-acclimation effect on cadmium absorption and biosynthesis of polyphenolics, and free proline and photosynthetic pigments in Spirogyra aequinoctialis
- Analysis of secondary metabolites in Xinjiang Morus nigra leaves using different extraction methods with UPLC-Q/TOF-MS/MS technology
- Nanoarchitectonics and performance evaluation of a Fe3O4-stabilized Pickering emulsion-type differential pressure plugging agent
- Investigating pyrolysis characteristics of Shengdong coal through Py-GC/MS
- Extraction, phytochemical characterization, and antifungal activity of Salvia rosmarinus extract
- Introducing a novel and natural antibiotic for the treatment of oral pathogens: Abelmoschus esculentus green-formulated silver nanoparticles
- Optimization of gallic acid-enriched ultrasonic-assisted extraction from mango peels
- Effect of gamma rays irradiation in the structure, optical, and electrical properties of samarium doped bismuth titanate ceramics
- Combinatory in silico investigation for potential inhibitors from Curcuma sahuynhensis Škorničk. & N.S. Lý volatile phytoconstituents against influenza A hemagglutinin, SARS-CoV-2 main protease, and Omicron-variant spike protein
- Physical, mechanical, and gamma ray shielding properties of the Bi2O3–BaO–B2O3–ZnO–As2O3–MgO–Na2O glass system
- Twofold interpenetrated 3D Cd(ii) complex: Crystal structure and luminescent property
- Study on the microstructure and soil quality variation of composite soil with soft rock and sand
- Ancient spring waters still emerging and accessible in the Roman Forum area: Chemical–physical and microbiological characterization
- Extraction and characterization of type I collagen from scales of Mexican Biajaiba fish
- Finding small molecular compounds to decrease trimethylamine oxide levels in atherosclerosis by virtual screening
- Prefatory in silico studies and in vitro insecticidal effect of Nigella sativa (L.) essential oil and its active compound (carvacrol) against the Callosobruchus maculatus adults (Fab), a major pest of chickpea
- Polymerized methyl imidazole silver bromide (CH3C6H5AgBr)6: Synthesis, crystal structures, and catalytic activity
- Using calcined waste fish bones as a green solid catalyst for biodiesel production from date seed oil
- Influence of the addition of WO3 on TeO2–Na2O glass systems in view of the feature of mechanical, optical, and photon attenuation
- Naringin ameliorates 5-fluorouracil elicited neurotoxicity by curtailing oxidative stress and iNOS/NF-ĸB/caspase-3 pathway
- GC-MS profile of extracts of an endophytic fungus Alternaria and evaluation of its anticancer and antibacterial potentialities
- Green synthesis, chemical characterization, and antioxidant and anti-colorectal cancer effects of vanadium nanoparticles
- Determination of caffeine content in coffee drinks prepared in some coffee shops in the local market in Jeddah City, Saudi Arabia
- A new 3D supramolecular Cu(ii) framework: Crystal structure and photocatalytic characteristics
- Bordeaux mixture accelerates ripening, delays senescence, and promotes metabolite accumulation in jujube fruit
- Important application value of injectable hydrogels loaded with omeprazole Schiff base complex in the treatment of pancreatitis
- Color tunable benzothiadiazole-based small molecules for lightening applications
- Investigation of structural, dielectric, impedance, and mechanical properties of hydroxyapatite-modified barium titanate composites for biomedical applications
- Metal gel particles loaded with epidermal cell growth factor promote skin wound repair mechanism by regulating miRNA
- In vitro exploration of Hypsizygus ulmarius (Bull.) mushroom fruiting bodies: Potential antidiabetic and anti-inflammatory agent
- Alteration in the molecular structure of the adenine base exposed to gamma irradiation: An ESR study
- Comprehensive study of optical, thermal, and gamma-ray shielding properties of Bi2O3–ZnO–PbO–B2O3 glasses
- Lewis acids as co-catalysts in Pd-based catalyzed systems of the octene-1 hydroethoxycarbonylation reaction
- Synthesis, Hirshfeld surface analysis, thermal, and selective α-glucosidase inhibitory studies of Schiff base transition metal complexes
- Protective properties of AgNPs green-synthesized by Abelmoschus esculentus on retinal damage on the virtue of its anti-inflammatory and antioxidant effects in diabetic rat
- Effects of green decorated AgNPs on lignin-modified magnetic nanoparticles mediated by Cydonia on cecal ligation and puncture-induced sepsis
- Treatment of gastric cancer by green mediated silver nanoparticles using Pistacia atlantica bark aqueous extract
- Preparation of newly developed porcelain ceramics containing WO3 nanoparticles for radiation shielding applications
- Utilization of computational methods for the identification of new natural inhibitors of human neutrophil elastase in inflammation therapy
- Some anticancer agents as effective glutathione S-transferase (GST) inhibitors
- Clay-based bricks’ rich illite mineral for gamma-ray shielding applications: An experimental evaluation of the effect of pressure rates on gamma-ray attenuation parameters
- Stability kinetics of orevactaene pigments produced by Epicoccum nigrum in solid-state fermentation
- Treatment of denture stomatitis using iron nanoparticles green-synthesized by Silybum marianum extract
- Characterization and antioxidant potential of white mustard (Brassica hirta) leaf extract and stabilization of sunflower oil
- Characteristics of Langmuir monomolecular monolayers formed by the novel oil blends
- Strategies for optimizing the single GdSrFeO4 phase synthesis
- Oleic acid and linoleic acid nanosomes boost immunity and provoke cell death via the upregulation of beta-defensin-4 at genetic and epigenetic levels
- Unraveling the therapeutic potential of Bombax ceiba roots: A comprehensive study of chemical composition, heavy metal content, antibacterial activity, and in silico analysis
- Green synthesis of AgNPs using plant extract and investigation of its anti-human colorectal cancer application
- The adsorption of naproxen on adsorbents obtained from pepper stalk extract by green synthesis
- Treatment of gastric cancer by silver nanoparticles encapsulated by chitosan polymers mediated by Pistacia atlantica extract under ultrasound condition
- In vitro protective and anti-inflammatory effects of Capparis spinosa and its flavonoids profile
- Wear and corrosion behavior of TiC and WC coatings deposited on high-speed steels by electro-spark deposition
- Therapeutic effects of green-formulated gold nanoparticles by Origanum majorana on spinal cord injury in rats
- Melanin antibacterial activity of two new strains, SN1 and SN2, of Exophiala phaeomuriformis against five human pathogens
- Evaluation of the analgesic and anesthetic properties of silver nanoparticles supported over biodegradable acacia gum-modified magnetic nanoparticles
- Review Articles
- Role and mechanism of fruit waste polyphenols in diabetes management
- A comprehensive review of non-alkaloidal metabolites from the subfamily Amaryllidoideae (Amaryllidaceae)
- Discovery of the chemical constituents, structural characteristics, and pharmacological functions of Chinese caterpillar fungus
- Eco-friendly green approach of nickel oxide nanoparticles for biomedical applications
- Advances in the pharmaceutical research of curcumin for oral administration
- Rapid Communication
- Determination of the contents of bioactive compounds in St. John’s wort (Hypericum perforatum): Comparison of commercial and wild samples
- Retraction
- Retraction of “Two mixed-ligand coordination polymers based on 2,5-thiophenedicarboxylic acid and flexible N-donor ligands: The protective effect on periodontitis via reducing the release of IL-1β and TNF-α”
- Topical Issue on Phytochemicals, biological and toxicological analysis of aromatic medicinal plants
- Anti-plasmodial potential of selected medicinal plants and a compound Atropine isolated from Eucalyptus obliqua
- Anthocyanin extract from black rice attenuates chronic inflammation in DSS-induced colitis mouse model by modulating the gut microbiota
- Evaluation of antibiofilm and cytotoxicity effect of Rumex vesicarius methanol extract
- Chemical compositions of Litsea umbellata and inhibition activities
- Green synthesis, characterization of silver nanoparticles using Rhynchosia capitata leaf extract and their biological activities
- GC-MS analysis and antibacterial activities of some plants belonging to the genus Euphorbia on selected bacterial isolates
- The abrogative effect of propolis on acrylamide-induced toxicity in male albino rats: Histological study
- A phytoconstituent 6-aminoflavone ameliorates lipopolysaccharide-induced oxidative stress mediated synapse and memory dysfunction via p-Akt/NF-kB pathway in albino mice
- Anti-diabetic potentials of Sorbaria tomentosa Lindl. Rehder: Phytochemistry (GC-MS analysis), α-amylase, α-glucosidase inhibitory, in vivo hypoglycemic, and biochemical analysis
- Assessment of cytotoxic and apoptotic activities of the Cassia angustifolia aqueous extract against SW480 colon cancer
- Biochemical analysis, antioxidant, and antibacterial efficacy of the bee propolis extract (Hymenoptera: Apis mellifera) against Staphylococcus aureus-induced infection in BALB/c mice: In vitro and in vivo study
- Assessment of essential elements and heavy metals in Saudi Arabian rice samples underwent various processing methods
- Two new compounds from leaves of Capparis dongvanensis (Sy, B. H. Quang & D. V. Hai) and inhibition activities
- Hydroxyquinoline sulfanilamide ameliorates STZ-induced hyperglycemia-mediated amyleoid beta burden and memory impairment in adult mice
- An automated reading of semi-quantitative hemagglutination results in microplates: Micro-assay for plant lectins
- Inductively coupled plasma mass spectrometry assessment of essential and toxic trace elements in traditional spices consumed by the population of the Middle Eastern region in their recipes
- Phytochemical analysis and anticancer activity of the Pithecellobium dulce seed extract in colorectal cancer cells
- Impact of climatic disturbances on the chemical compositions and metabolites of Salvia officinalis
- Physicochemical characterization, antioxidant and antifungal activities of essential oils of Urginea maritima and Allium sativum
- Phytochemical analysis and antifungal efficiency of Origanum majorana extracts against some phytopathogenic fungi causing tomato damping-off diseases
- Special Issue on 4th IC3PE
- Graphene quantum dots: A comprehensive overview
- Studies on the intercalation of calcium–aluminium layered double hydroxide-MCPA and its controlled release mechanism as a potential green herbicide
- Synergetic effect of adsorption and photocatalysis by zinc ferrite-anchored graphitic carbon nitride nanosheet for the removal of ciprofloxacin under visible light irradiation
- Exploring anticancer activity of the Indonesian guava leaf (Psidium guajava L.) fraction on various human cancer cell lines in an in vitro cell-based approach
- The comparison of gold extraction methods from the rock using thiourea and thiosulfate
- Special Issue on Marine environmental sciences and significance of the multidisciplinary approaches
- Sorption of alkylphenols and estrogens on microplastics in marine conditions
- Cytotoxic ketosteroids from the Red Sea soft coral Dendronephthya sp.
- Antibacterial and biofilm prevention metabolites from Acanthophora spicifera
- Characteristics, source, and health risk assessment of aerosol polyaromatic hydrocarbons in the rural and urban regions of western Saudi Arabia
- Special Issue on Advanced Nanomaterials for Energy, Environmental and Biological Applications - Part II
- Green synthesis, characterization, and evaluation of antibacterial activities of cobalt nanoparticles produced by marine fungal species Periconia prolifica
- Combustion-mediated sol–gel preparation of cobalt-doped ZnO nanohybrids for the degradation of acid red and antibacterial performance
- Perinatal supplementation with selenium nanoparticles modified with ascorbic acid improves hepatotoxicity in rat gestational diabetes
- Evaluation and chemical characterization of bioactive secondary metabolites from endophytic fungi associated with the ethnomedicinal plant Bergenia ciliata
- Enhancing photovoltaic efficiency with SQI-Br and SQI-I sensitizers: A comparative analysis
- Nanostructured p-PbS/p-CuO sulfide/oxide bilayer heterojunction as a promising photoelectrode for hydrogen gas generation

