Abstract
Breast and cervical cancers are the leading cause of death in women, and chemotherapy with cytotoxins is the usual treatment. This study evaluated the cytotoxicity of guava leaf (Psidium guajava L.) extracts as an alternative chemotherapeutic drug. Although many studies related to the cytotoxic effects of guava leaf (Psidium guajava L.) on cancer cells have been reported, the effects of guava leaf fractions on human breast and cervical cancer cells (T47D, MCF-7, and HeLa) have never been evaluated. Herein, we researched candidate activities of ethanol, ethyl acetate, and n-extracts from guava leaf fractions and their effect on various human cancer cell lines (T47D, MCF-7, and HeLa cells). The cytotoxicity test was carried out using the microtetrazolium assay for all fractions. We confirmed and showed the in vitro antitumor activity of guava leaf (Psidium guajava L.) fractions in human breast and cervical cancer cells. We found that the effectiveness of anticancer activity increased from ethanol to ethyl acetate to n-hexane fraction. This work underlines the potential of n-hexane fraction as a chemotherapeutic drug. These novel results have important implications for further isolation, identification, and characterization of Psidium guajava L.-based anti-cancer extracts.
1 Introduction
Cancer is ranked as the third leading cause of death in the world. Estimates from the IARC (International Agency for Research on Cancer) indicate that, by 2,040, the number of new cancer cases could reach 30.2 million cases with a death rate of up to 16.3 million cases. The most common cancers are breast cancer (11.7%), lung cancer (11.4%), colon or rectum cancer (10%), prostate cancer (7.3%), stomach cancer (5.6%), liver cancer (4.7%), cervical cancer (3.1%), and other cancers (46.2%) [1,2]. However, this situation has been an object of interest for many researchers and they have carried out impressive studies on the isolation and synthesis field to end the harms of cancer [3,4,5,6].
Breast and cervical cancer is malignant cancer, which is most commonly found in women and is one of the causes of death, especially in developing countries [7]. Because of the high prevalence of cancer, complementary and alternative medicines are needed in cancer treatment [8]. Several strategies in the discovery of new anticancer compounds have been performed such as isolating active compounds from natural products, exploring secondary metabolites to specifically inhibit cancer cells, and synthesizing potential organic compounds with anticancer activity [9,10,11,12,13].
It is known that 30,000–40,000 species of medicinal plants in the world are available in Indonesia. However, only a portion of these plants has been developed for therapeutic drugs [14], particularly cancer therapy [15]. Several plants have been investigated as therapeutic agents for leukemia, lung cancer, breast cancer, liver cancer, and prostate cancer [16,17,18,19,20,21,22,23]. Several anticancer compounds have also been isolated from other sources such as microbes (endophytic bacteria/yeast and fungi). For example, Pseudomonas putida can produce secondary metabolites that function as anticancer [24].
Guava (Psidium guajava L.) is a member of the Myrtaceae family and many investigations have reported that the leaf has antimicrobial [25,26,27], antidiabetic [28], antimalarial, [29], and antitumor activities [30,31]. In addition, it has been used to treat acne [32], coughs [33], and dental diseases [34]. Guava leaf contains mostly phenolic compounds, flavonoids (morin-3-O-glycoside, morin-3-O-arabinoside, quercetin, and quercetin-3-O-arabinoside), essential oils, lipids, gallic acid, catechin, epicatechin, rutin, tannin, saponin, alkaloids, and sesquiterpenes [31]. In particular, the flavonoids in guava leaf are reported to have strong antibacterial [25] and anticancer activities [5]. However, research on the activity of guava leaf as an anticancer drug is still very limited.
To the best of our knowledge, there are several studies related to examining the anticancer activity of guava Psidium guajava L. using cell cultures. The cancer cells that have been used to test the anticancer activity of guava are prostate cancer cells [35], cervical cancer [5], colon cancer [35], gastric cancer [36], and blood cell cancer [30]. Based on the results of the anticancer activity, we hypothesize that guava leaf has potential as an anticancer drug because it can prevent or inhibit the growth of cancer cells [6]. The anticancer potential of the extracts is evaluated using the microtetrazolium (MTT), MTS, SRB, thymidine incorporation, and trypan blue exclusion assay to observe their cytotoxicity to cancer cells [37,38,39,40]. Although some studies related to guava leaf (Psidium guajava L.) as an anticancer were reported to exhibit cytotoxic effects in human cancer cells [41,42,43,44,45,46,47], the effects of guava leaf fraction on human breast and cervical cancer cells (T47D, MCF-7, and HeLa) have never been evaluated.
In this study, we are interested in finding alternative medicine to chemotherapy based on guava leaves (Psidium guajava L.). Our research group explored and carried out the fractionation process to separate the active compounds in guava leaves based on their polarity levels using ethanol (polar), ethyl acetate (semi-polar), and n-hexane (non-polar) as solvents to find which fractions of the compounds are active as anti-cancer agents. This will be very useful for further exploration processes in the future, which include isolation, identification, and characterization of active anti-cancer compounds. The fractionation process in this study refers to several previous studies, where fractionation was carried out with a fractionation column using non-polar, semi-polar, and ending with polar solvents continuously [35,36]. We also reported that different fractions from guava leaves act as anticancer agents characteristically and selectively against three cancer cells. Then, we focused on anticancer tests that were carried out in vitro using the MTT assay. Furthermore, we confirmed the in vitro antitumor activity of the guava leaf (Psidium guajava L.) fraction in human breast and cervical cancer cells (T47D, MCF-7, and HeLa). By understanding the selectivity of each fraction, it can be developed into an anticancer agent. The fractions used were ethanol, ethyl acetate, and n-hexane fractions of guava leaves.
2 Materials and methods
2.1 Materials
All chemicals used were of analytical grade from JT Baker (n-hexane, ethyl acetate, ethanol, and chloroform), Merck and Co. Inc (methanol, acetic acid, acetonitrile, DMSO, silica gel 60 [0.063–0.200 mm], silica gel 60 PF254, Na2SO4, H2SO4, NaOH), Sigma-Aldrich (acetone, Mg metal, HCl 37%, Dragendroff reagent, Mayer’s reagent, FeCl3, phosphate buffer saline, RPMI culture media, trypsin-EDTA, dimethyl sulfoxide [DMSO], sodium dodecyl sulfate (SDS), Dulbecco's modified Eagle’s medium [DMEM], formaldehyde, MTT). The Psidium guajava L. leaves were obtained from a tree cultivated in the Sleman District and have passed the determination and identification test by our student at the Laboratory of the Faculty of Biology, Gadjah Mada University Indonesia, Yogyakarta Province. T47D, MCF-7, and HeLa cancer cells were obtained from the collection of Gadjah Mada University.
All analysis instruments used in this study are LC-MS/MS UPLC-QToF-MS/MS System (Waters), hemositometer, laminar air flow (Innotech V-800), ELISA reader (Bio Rad Benchmark), incubator (Memmert), centrifuge (Hettich Rotina 22R), analytical measure (Fujitsu), Rotary evaporator (Heidolph Laborata 4000 efficient), shaker (SCILOGEX SK-O330-Pro), micropipette (Eppendorf Research), microtube, and 96-well microplate.
2.2 Preparation of Psidium guajava L. leaves as raw materials
The Psidium guajava L. leaves were dried in an open space without direct sunlight until dry. A total of 700 g of dried samples were blended to powder. The powder was stored in a bottle to be used in the next step.
2.3 Preparation of ethanol, ethyl acetate, and n-hexane fraction of guava leaves
The extraction was carried out using the maceration technique. About 100 g of dry Simplicia powder was immersed in ethanol in an airtight container. After 1 day, the ethanol extract was separated from the macerate using filter paper. Then, the extract was concentrated using a rotary evaporator to obtain the ethanol crude extract. This crude extract was impregnated using silica gel and put in a glass chromatography column. The process was continued by fractionating the crude extract using a fractionation column chromatography using non-polar (n-hexane), semi-polar (ethyl acetate), and finally with polar (ethanol) solvents continuously. Then, the extract was concentrated to get n-hexane, ethyl acetate, and ethanol fractions of the guava leaf as non-polar, semi-polar, and polar fractions, respectively.
2.4 Phytochemical screening
Phytochemical screening in research is an initial stage that aims to obtain an overview of the class of compounds contained in a sample. The phytochemical screening method was performed by visualizing the color testing reaction. The screening included the content of tannin/polyphenol compounds, alkaloids, flavonoids, and terpenoids according to Singh et al. [48].
2.5 In vitro anticancer activity of guava leaf fractions on T47D, MCF-7 cells (breast cancer), and HeLa cells (cervical cancer) with MTT assay
The in vitro anticancer activity of guava leaf fractions (n-hexane, ethyl acetate, and ethanol fraction) on T47D, MCF-7, and HeLa cells was performed with the preparation of the RewI Park 1640 culture media for T47D, HeLa cells, and DMEM for MCF-7 cells, followed by cell activation, harvesting, and calculation as initial processes [49]. Afterward, each fraction (ethanol, ethyl acetate, and n-hexane fractions) was diluted into a solution using DMSO at concentrations of 200, 100, 50, 25, 12.5, 6.25, and 3.125 μg mL. Then, 100 μL of the fraction was added to a cell suspension in each well and incubated at 37°C. The test was replicated three times. After 24 h, culture media and the MTT reagent (3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide) were added in a ratio of 9:1 and then incubated. After 4 h, 100 μL of SDS as stopper solution was added to the well and then placed in a dark room for 24 h. The final stage was carried out by absorbance reading using an ELISA reader (wavelength 595 nm). Cytotoxicity tests were carried out by the same method, namely in vitro on Vero cells. After IC50 values were obtained for T47D, MCF-7, HeLa, and Vero cells, then the selectivity index (SI) was calculated (see the Supporting information) by the following formula:
where A is the absorbance of the treatment (cell + culture media + fraction), B is the absorbance of the media control (culture media), and C is the absorbance of the negative control (cell + culture media).
3 Results and discussion
3.1 Phytochemical screening
Phytochemical screening results on ethanol, ethyl acetate, and n-hexane fractions of guava leaves are shown in Table 1.
Phytochemical screening results of guava leaf fractions
| Material | Phytochemical screening | Reagent | Observation | Result |
|---|---|---|---|---|
| Ethanol fraction | Alkaloids | Dragendorff | Greenish-yellow solution (fixed) | − |
| Wagner | Greenish-yellow solution (fixed) | − | ||
| Flavonoids | HCl + Mg | Orange solution | + | |
| Tannins/polyphenols | FeCl3 | deposits (bluish-black solution) | + | |
| Terpenoids | Anhydrous acetic acid + H2SO4 | Purplish-red solution | + | |
| Ethyl acetate fraction | Alkaloids | Dragendorff | Brown solution (fixed) | − |
| Wagner | Greenish-yellow solution (fixed) | − | ||
| Flavonoids | HCl + Mg | Precipitate (green solution) | + | |
| Tannins/polyphenols | FeCl3 | Blackish-blue solution | + | |
| Terpenoids | Anhydrous acetic acid + H2SO4 | Dark-green solution | + | |
| n-Hexane fraction | Alkaloids | Dragendorff | Brown solution | − |
| Wagner | Blackish red solution | − | ||
| Flavonoids | HCl + Mg | Precipitate (red solution) | + | |
| Tannins/polyphenols | FeCl3 | Orangish-yellow solution | − | |
| Terpenoids | Anhydrous acetic acid + H2SO4 | Purplish-red solution | + |
Note: (+), positive; (–), negative.
Phytochemical screenings are carried out to identify the class of active compounds in the sample, i.e., tannin/polyphenols, alkaloid, flavonoid, and terpenoid compounds. Based on visual observation, it was concluded that the n-hexane fraction contained flavonoids and terpenoids; the ethyl acetate fraction contained flavonoids, tannins/polyphenols, and terpenoids; the ethanol fraction contained terpenoids, flavonoids, and tannins/polyphenols, and these results are in line with previous studies from other researchers [50].
3.2 In vitro anticancer activity of guava leaf fractions on T47D, MCF-7 cells (breast cancer), and HeLa cells (cervical cancer) with MTT assay
The in vitro anticancer activity test is required to pass through several preliminary stages, i.e., preparation and harvesting of cancer cells, cytotoxicity test, MTT assay, and absorbance reading. Cell preparation was carried out by activating the cells and regenerating them until they were confluent (where the cell growth was homogeneously or equally distributed as monolayer cells covering the cover glass). In this study, our research group uses T47D and MCF-7 cells for breast cancer cells and HeLa cells for cervical cancer cells. T47D breast cancer cells are obtained from the breast tissue of adolescent and adult women who are affected by ductal carcinoma and grown in RPMI media (Roswell Park Memorial Institute). Different from T47D cells, MCF-7 cells (Michigan Cancer Foundation-7) are obtained from Caucasian female patients with DMEM. These cells were obtained from breast epithelial tissue with a pleural effusion breast adenocarcinoma metastatic point from a 69-year-old woman of Caucasian ethnicity with blood group O with Rh+. On the other hand, HeLa cells were derived from human cervical cancer epithelial cells. These cells have been isolated since 1951 from the uterus of the 31-year-old first woman, Henrietta Lack, with cervical cancer, from Baltimore, USA. Last, but not least, research related to the discovery of cancer drugs needs to be done with cytotoxic tests on normal cells to find out whether compounds that inhibit cancer cells are selective on cancer cells or attack normal body cells as well. The Vero cell is a cell line derived from the kidney of the African green monkey and used as a representative of normal cells [51].
In the harvesting step, cells are grown, multiplied using RewI Park 1640 culture media for T47D and HeLa cells and DMEM for MCF-7 cells and ended with the addition of trypsin to release the interaction between the cell to the matrix (cell–matrix) and cell to cell (cell–cell) to prevent cell damage. Cancer cell counting was carried out using a hemocytometer and observed using an inverted microscope to determine the number of cells in the initial step. The number of T47D and MCF-7 cells was 46 × 104/mL and 144.25 × 104/mL, respectively, and HeLa cells were 453.75 × 104/mL. The initial morphology of T47D, MCF-7, and HeLa cells are shown in Figure 1.
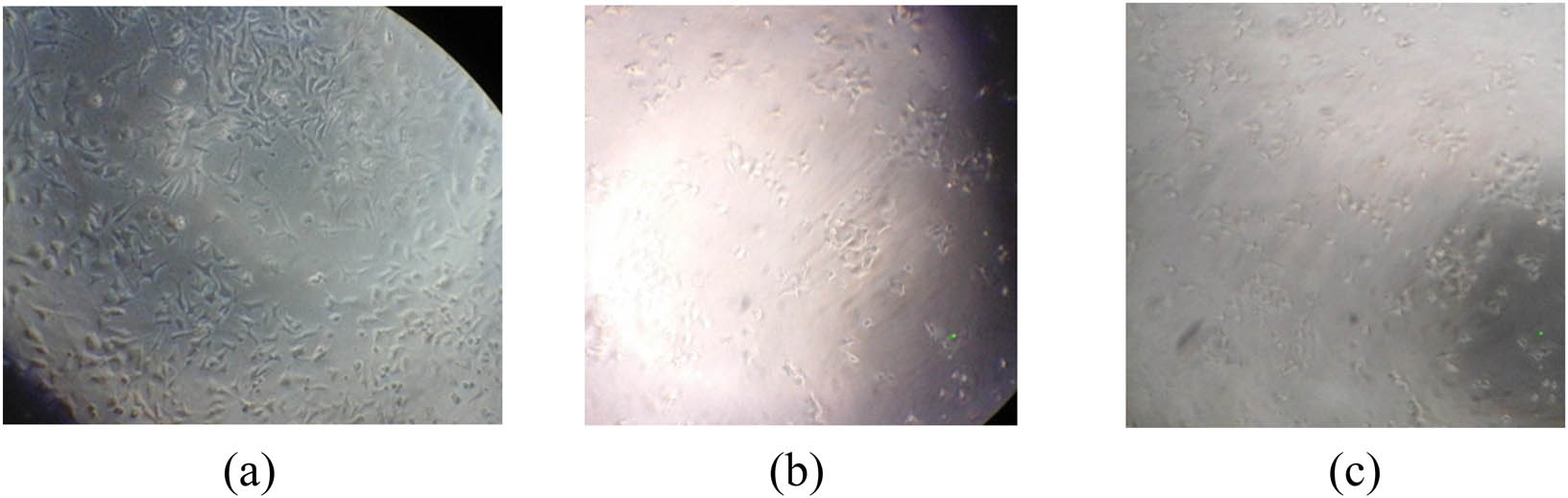
The initial morphology of T47D (a), MCF-7 (b), and HeLa (c) cells.
The cell subculture, sample preparation, and cell treatment were carried out as part of the cytotoxicity test. At the cell subculture, cells were transferred from confluent conditions to an empty place for growing optimally and generated 2.17 × 104 µL for T47D; 0.22 × 104 µL for MCF-7; and 0.69 × 104 µL for HeLa. These cells were used again in the harvesting step and divided into three groups, namely cell control, media control, and test group. The test group consisted of ethanol, ethyl acetate, and n-hexane fractions. About 100 µL of cell suspension was pipetted into plate 96, except for media control. Then, the plate was incubated in a 5% CO2 incubator at 37°C for 24 h. This enabled the cells to reach the logarithmic phase, where cells are in optimum growth conditions. The logarithmic phase is characterized by the condition of cells that are 80% confluent, covering the surface of the medium container.
The next step is the sample preparation including sample (fraction) dissolution and preparation of concentration series solutions with DMSO as a solvent. First, the sample solution was prepared by adding 10 mg of each fraction and dissolving it with 100 µL of DMSO. Then, the multilevel concentration of the solution was made (200, 100, 50, 25, 12.5, 6.25, and 3.125 µg/mL) with RPMI for T47D and HeLa cells and DMEM for MCF-7 cells. Later, 100 µL of the test solution was added into the wells and replicated three times for each concentration. For control cells, only 100 µL of the medium was added without the fraction solution. Then, the plates were incubated again in a 5% CO2 incubator at 37°C for 24 h.
After the treatment, an MTT assay was carried out by adding the MTT reagent into each well and then incubating for 4 h. The reaction was stopped using 10% SDS as a stopper reagent. The stopper reagent dissolves the formazan crystals which are reduced by the cell reductase system. Then, the plates were incubated at room temperature for 24 h. Furthermore, the absorbance was measured using an ELISA reader with a wavelength of 595 nm. From Figure 2, for T47D cells, it can be seen that the n-hexane fraction of guava leaves at 200, 100, and 50 µg/mL shows yellow color and purple color at 25, 12.5, 6.25, and 3.125 µg/mL. A change in the color from purple to yellow indicates that the cancer cells have died. There is no reductase enzyme produced in the mitochondria of the cell that reduces tetrazolium salt into purple formazan crystals. However, in the ethyl acetate fraction, the yellow color was only observed at a concentration of 200 µg/mL and purple at a lower concentration. Meanwhile, in the ethanol fraction, the observed color intensity was purple from high to low concentrations. This indicated that the number of living cells is still quite a lot.

The appearance of T47D (a), MCF-7 (b), Hela (c), and Vero (d) cells after treatment using the ethanol fraction, ethyl acetate fraction, and n-hexane fraction.
Based on the analysis using ELISA reader at 595 nm, the IC50 values were obtained and are presented in Table 2 (see the Supporting information for calculation).
Comparison of IC50 values of anticancer activity from guava leaf fractions and doxorubicin
| Fraction | IC50 (µg/mL) | |||
|---|---|---|---|---|
| T47D | MCF-7 | HeLa | Vero | |
| Ethanol | 236.15 | 22.61 | 143.33 | 285.17 |
| Ethyl acetate | 21.68 | 52.88 | 217.68 | 347.25 |
| n-Hexane | 8.92 | 4.28 | 85.98 | 266.88 |
| *Doxorubicin [52,53] | 8.53 | 0.69 | 1.69 | 88.23 |
*It means data from reference.
The IC50 values for potential anticancer is below 30 μg/mL, 30 < IC50 < 100 μg/mL for moderately active, and more than 100 μg/mL for less active [54,55]. Based on Table 2, it can be concluded that the ethanol fraction is inactive on T47D cells, active on MCF-7 cells, and moderately active on HeLa cells. The ethyl acetate fraction was active against T47D cells, moderately active against MCF-7 cells, and less active on HeLa cells. The n-hexane fraction has the best activity based on IC50 values, very active against T47D and MCF-7 cells but moderately active against HeLa cells.
A cytotoxicity test was also carried out on normal cells (Vero cells) to determine the selectivity of the fractions. The purpose of this selectivity value is to determine the level of safety of an anticancer compound against normal cells so that it can be further developed as a chemopreventive agent. The steps carried out were the same as the activity test on cancer cells for each fraction with seven concentration variations: 200, 100, 50, 25, 12.5, 6.25, and 3.125 µg/mL. Tables 2 and 3 show that the guava leaf fraction is not harmful to normal cells (Vero cells). This can be shown from the IC50 value, which is >100 μg/mL. Moreover, the selectivity value is very important. It was obtained from the IC50 values of normal cells divided by the IC50 values of the cancer cells for each fraction. For SI values of >10, it can be categorized to be good selectivity.
SI values of the guava leaf fraction
| Fraction | SI | ||
|---|---|---|---|
| T47D | MCF-7 | HeLa | |
| Ethanol | 1.20 | 12.61 | 1.98 |
| Ethyl acetate | 16.01 | 6.56 | 1.59 |
| n-Hexane | 29.91 | 62.35 | 3.10 |
Based on our analysis, the n-hexane fraction has high activity and selectivity to cancer cells. The cell morphology after n-hexane treatment as the best fraction for three cancer cells was observed under an inverted microscope for (a) T47D, (b) MCF-7, and (c) Hela cells, and shown in Figure 3. The morphology of Hela cells after interacting with the n-hexane fraction became long and spiky wrinkled. It looks different after the interaction with T47D where the cells look like little dots. Dramatically, the interaction with MCF-7 cells shrunk the structure into small pieces causing cell damage. This shows that there are differences in interactions between the fraction and breast cancer cells.

The morphology of (a) T47D, (b) MCF-7, and (c) Hela cells after treatment with the n-hexane fraction.
The inhibition analysis (IC50) values as a reference for the effectiveness of the fraction as a chemopreventive agent were strengthened by the cell viability tests, where cell viability indicates the number of living cells. The test results are presented in Tables 4–6.
Viability test of the guava leaf fraction on T47D cells
| Fraction | Concentration (μg/mL) | Viability cells (%) |
|---|---|---|
| Ethanol | 200 | 52.86 |
| 100 | 74.64 | |
| 50 | 76.65 | |
| 25 | 77.18 | |
| Ethyl acetate | 50 | 13.19 |
| 25 | 26.19 | |
| 12.5 | 73.11 | |
| 6.25 | 76.66 | |
| n-Hexane | 25 | 4.38 |
| 12.5 | 15.22 | |
| 6.25 | 60.17 | |
| 3.125 | 82.05 |
Viability test of the guava leaf fraction on MCF-7 cells
| Fraction | Concentration (μg/mL) | Viability cells (%) |
|---|---|---|
| Ethanol | 25 | 46.82 |
| 12.5 | 61.64 | |
| 6.25 | 63.75 | |
| 3.125 | 67.46 | |
| Ethyl acetate | 100 | 26.01 |
| 50 | 54.72 | |
| 25 | 59.45 | |
| 12.5 | 71.62 | |
| n-Hexane | 25 | 15.87 |
| 12.5 | 32.77 | |
| 6.25 | 42.22 | |
| 3.125 | 57.43 |
Viability test of guava leaf fraction on HeLa cells
| Concentration (μg/mL) | Viability cells (%) | ||
|---|---|---|---|
| Ethanol | Ethyl acetate | n-Hexane | |
| 100 | 55.47 | 66.10 | 46.66 |
| 50 | 58.65 | 69.27 | 58.94 |
| 25 | 60.77 | 71.39 | 62.10 |
| 12.5 | 66.43 | 79.51 | 66.66 |
As seen from Table 4, at the same concentration (25 μg/mL) of ethanol fraction, the cell viability is around 77.18%, which means that more than 50% of the cells are alive, whereas 26.19% for ethyl acetate and n-hexane had lowest cell viability about 4.38%. The phenomena showed that at this concentration the n-hexane fraction had greater inhibition than the ethanol and ethyl acetate fraction. Therefore, it can be concluded that the n-hexane fraction is more effective in inhibiting T47D cancer cells than others.
As shown in Table 5, at the same concentration (25 μg/mL) of ethanol fraction, the viability or the number of living cells was 46.82% (less than 50%), and ethyl acetate fraction was not much different (59.45%), while the n-hexane fraction had much lower viability (15.87%). The phenomena also showed that the n-hexane fraction has a better inhibition ability to MCF-7 than the other two fractions at fairly low concentrations.
Based on Table 6, at the same concentration (25 μg/mL) of ethanol fraction, the viability was 60.77, 71.39% for ethyl acetate, and 62.10% n-hexane. This shows that the three fractions cannot inhibit the growth of HeLa cells at low enough concentrations so that cell viability is above 50%. In addition, when compared to T47D and MCF-7 cells, the three fractions showed better activity and effectiveness than in HeLa cells.
3.3 LCMS/MS analysis of dominant compounds in the n-hexane fraction
The n-hexane fraction, which is the most active fraction of three cancer cells, was analyzed using LC-MS/MS with positive and negative ionization modes. The chromatogram and mass analysis of the n-hexane fraction are given in the supporting information. This analysis showed that there are several high and sharp peaks with near retention times either in the negative mode or positive mode. These peaks were determined as the dominant compounds contained in the n-hexane fraction. They were analyzed and compared with some references from the previous research group to determine their masses and are presented in Tables 7 and 8 [56,57,58,59,60,61]. This analysis has provided valuable information regarding the content of secondary metabolites in the n-hexane fraction and is very useful for the development of further research to investigate which compounds have an important role as anticancer from the n-hexane fraction.
Results of LC-MS/MS analysis of the n-hexane fraction of guava leaves with a positive ionizing source and compared with other references
| Component name | [M + H]+ or [M + Na]+ (m/z) | Neutral mass (Da) | Observed RT (min) |
|---|---|---|---|
| Oleanolic acid [58]* | 203.19 | 201.19 | 5.70 |
| Quercetin isomer [57]* | 257.13 | 256.13 | 6.36 |
| Cyanidin-3-O-glucoside [57,60]* | 441.37 | 440.37 | 6.36 |
| Genistein [58]* | 271.12 | 270.12 | 6.36 |
| Quercetin hexoside [57]* | 179.07 | 178.07 | 6.36 |
| 3-tert-Butyl-4-methoxyphenol* | 181.12 | 180.12 | 8.86 |
| Hyptatic acid* | 511.34 | 488.34 | 9.19 |
| Petasitolone* | 259.16 | 236.16 | 9.37 |
| Stigmastan-3,6-dion* | 429.37 | 428.37 | 10.46 |
| Candidate C26H48O14* | 607.29 | 584.29 | 10.72 |
| Candidate C30H34O5* | 475.24 | 474.24 | 11.24 |
*LC-MS/MS data obtained from authors.
Results of LC-MS/MS analysis of the n-hexane fraction of guava leaves with a negative ionizing source and compared with other references
| Component name | [M–H]− (m/z) | Neutral mass (Da) | Observed RT (min) |
|---|---|---|---|
| Madecassic acid (triterpenoid) [57] | 503.31 | 504.31 | 4.54 |
| Gallic acid [57]* | 169.98 | 170.98 | 4.54 |
| Guavinoside [57]* | 543.21 | 544.21 | 4.91 |
| Cyanidin-3-O-glucoside* | 447.97 | 448.97 | 4.91 |
| Methylphloroglucinol-galloyl-hexoside [57] | 453.26 | 454.26 | 6.24 |
| Pinfaensin [61]* | 663.41 | 664.41 | 6.51 |
| Trihydroxycinnamoylquinic acid isomers [59]* | 369.01 | 370.01 | 6.51 |
| Guavenoic acid [57]* | 471.27 | 472.27 | 6.91 |
| Glycoside [59]* | 325.20 | 326.20 | 6.91 |
| Ellagitanin [57]* | 473.24 | 474.24 | 6.91 |
*LC-MS/MS data obtained from authors.
Based on the LC-MS/MS analysis using negative ionization (Table 8), the presence of ellagitannin compounds was shown in high concentration and it was suspected that these compounds had a role in preventing or inhibiting the growth of cancer cells. Ellagitannin is known as a chemoprevention agent for colon, skin, cervical, prostate, breast, and liver cancers [62]. Ellagitannin has specificity in inhibiting the growth of cancer cells by attacking the ER + point, where T47D cells have that point; therefore, the presence of ellagitannin makes the n-hexane fraction more active than other fractions and is effective for inhibiting T47D cells. Also in the positive ionization mode, genistein was identified to play a role in preventing the growth of breast cancer [63]. By identifying these compounds in the n-hexane fraction of guava leaves, it can be concluded that ellagitannin and genistein play a key role to inhibit the growth of cancer cells in this study.
4 Conclusion
The chemopreventive agent from guava leaves was successfully prepared via maceration and fractionation methods. Phytochemical screenings have demonstrated that the n-hexane fraction contained flavonoids and terpenoids; the ethyl acetate fraction contained flavonoids, tannins/polyphenols, and terpenoids; and the ethanol fraction contained terpenoids, flavonoids, and tannins/polyphenols. Based on the MTT assay, it was found that the n-hexane fraction has high activity and selectivity to cancer cells with the IC50 for T47D, MCF-7, and HeLa being 8.92, 4.28, and 85.98, respectively, and the selectivity value being>10, which was categorized to be good selectivity. Further development, isolating active compounds, and investigation of the n-hexane fraction to make it more suitable as a chemopreventive agent is still underway in our laboratory.
Acknowledgements
The authors thank to the Direktorat Penelitian dan Pengabdian Masyarakat (DPPM), Universitas Islam Indonesia, for the research funding support with research contract number 002/Dir/DPPM/70/Pen.Unggulan/PI/IV/2019.
-
Funding information: This research was financially supported by Universitas Islam Indonesia (project number: 002/Dir/DPPM/70/Pen.Unggulan/PI/IV/2019).
-
Author contributions: Nurcahyo Iman Prakoso: research design, identification, toxicity assay, and writing/reviewing/editing the draft. Mila Tria Nita: plant identification, separation, extraction, toxicity assay, and phytochemical screening.
-
Conflict of interest: The authors declare no conflict of interest.
-
Ethical approval: The conducted research is not related to either human or animal use.
-
Data availability statement: The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
[1] Arnold M, Morgan E, Rumgay H, Mafra A, Singh D, Laversanne M, et al. Current and future burden of breast cancer: Global statistics for 2020 and 2040. Breast. 2022;66:15–23. 10.1016/j.breast.2022.08.010.Suche in Google Scholar PubMed PubMed Central
[2] Ferlay J, Colombet M, Soerjomataram I, Parkin DM, Piñeros M, Znaor A, et al. Cancer statistics for the year 2020: An overview. Int J Cancer. 2021;149:778–89. 10.1002/ijc.33588.Suche in Google Scholar PubMed
[3] Ryu HN, Park RK, Kim MS, Yun MH, Nam D, Lee GS, et al. A hexane fraction of guava leaf (Psidium guajava L.) induces anticancer activity by suppressing AKT/mammalian target of rapamycin/ribosomal p70 S6 kinase in human prostate cancer cells. J Med Food. 2012;15:231–41. 10.1089/jmf.2011.1701.Suche in Google Scholar PubMed PubMed Central
[4] Prakoso NI, Matsuda F, Umezawa T. Efficient synthesis of α,β-dichlorinated ketones from α,β-dichlorinated weinreb amides through a simple work-up procedure. Org Biomol Chem. 2021;19:7822–6. 10.1039/d1ob01379c.Suche in Google Scholar PubMed
[5] Joseph B, Priya M. Review on nutritional, medicinal and pharmacological properties of guava (psidium guajava linn.). Int J Pharm Bio Sci. 2011;2:53–69.Suche in Google Scholar
[6] Rishika D, Sharma R. An update of pharmacological activity of Psidium guajava in the management of various disorder. Int J Pharm SRes. 2012;3:3577–84. 10.13040/IJPSR.0975-8232.3(10).3577-84.Suche in Google Scholar
[7] Richie RC, Swanson J. Breast cancer: A review of the literature. J Insur Med. 2003;35:85–101.Suche in Google Scholar
[8] Chrystal K, Allan S, Forgeson G, Isaacs R. The use of complementary/alternative medicine by cancer patients in a New Zealand regional cancer treatment centre. N Z Med J. 2003;116:1–7. 10.1186/1477-7800-4-10.Suche in Google Scholar PubMed PubMed Central
[9] Umezawa T, Prakoso NI, Tsuji K, Ogura Y, Sato T, Matsuda F. Model study toward total synthesis of mytilipin C. Bull Chem Soc Jpn. 2022;95:1491–500. 10.1246/bcsj.20220196.Suche in Google Scholar
[10] Keshava R, Muniyappa N, Rajalakshmi G, Ramaswamaiah AS. Anti-cancer effects of Imperata cylindrica leaf extract on human oral squamous carcinoma cell line SCC-9 in vitro. Asian Pac J Cancer. 2016;17:1891–8. 10.7314/apjcp.2016.17.4.1891.Suche in Google Scholar PubMed
[11] Kwok AHY, Wang Y, Ho WS. Cytotoxic and prooxidative effects of Imperata cylindrica aerial part ethyl acetate extract in colorectal cancer in vitro. Phytomedicine. 2016;23:558–65. 10.1016/j.phymed.2016.02.015.Suche in Google Scholar PubMed
[12] Allegra A, Innao V, Russo S, Gerace D, Alonci A, Musolino C. Anticancer activity of curcumin and its analogues: preclinical and clinical studies. Cancer Invest. 2017;35:1–22. 10.1080/07357907.2016.1247166.Suche in Google Scholar PubMed
[13] Gebashe F, Aremu AO, Gruz J, Finnie JF, Staden JV. Phytochemical profiles and antioxidant activity of grasses used in South African traditional medicine. Plants. 2020;9:1–23. 10.3390/plants9030371.Suche in Google Scholar PubMed PubMed Central
[14] Prakoso NI, Zakiyah ZN, Liyanita A, Rubiyanto D, Fitriastuti D, Ramadani AP, et al. Antimalarial activity of andrographis paniculata ness’s N-hexane extract and its major compounds. Open Chem. 2019;17:788–97. 10.1515/chem-2019-0086.Suche in Google Scholar
[15] Rahardjanto A, Ikhtira DA, Nuryady MM, Pantiwati Y, Widodo N, Husamah H. The medicinal plant potential parts and species diversity as antipyretic: Ethnobotany study at Senduro Lumajang. In: AIP Conference Proceedings. vol. 2353; 2021. p. 030018–1-8. 10.1063/5.0053124.Suche in Google Scholar
[16] Mohan S, Abdul AB, Abdelwahab SI, Al-Zubairi AS, Aspollah SM, Abdullah R, et al. Typhonium flagelliforme inhibits the proliferation of murine leukemia WEHI-3 cells in vitro and induces apoptosis in vivo. Leuk Res. 2010;34:1483–92. 10.1016/j.leukres.2010.04.023.Suche in Google Scholar PubMed
[17] Mohan S, Bustamam A, Ibrahim S, Al-Zubairi AS, Aspollah SM, Abdullah R, et al. In vitro ultramorphological assessment of apoptosis on CEMss induced by linoleic acid-rich fraction from typhonium flagelliforme tuber. Evid Based Complement Altern Med. 2010;2011:1–13. 10.1093/ecam/neq010.Suche in Google Scholar PubMed PubMed Central
[18] Isa NM, Abdul AB, Abdelwahab SI, Abdullah R, Sukari MA, Boesenbergin A. a chalcone from Boesenbergia rotunda induces apoptosisvia mitochondrial dysregulation and cytochrome c release in A549 cells in vitro: Involvement of HSP70 and Bcl2/Baxsignalling pathways. J Funct Foods. 2012;5:87–97. 10.1016/j.jff.2012.08.008.Suche in Google Scholar
[19] Ho WY, Yeap SK, Ho CL, Raha AR, Suraini AA. Elephantopus scaber induces cytotoxicity in MCF-7 human breast cancer cells via p53-induced apoptosis. J Med Plants Res. 2011;5:5741–9.Suche in Google Scholar
[20] Syam S, Abdul AB, Sukari MA, Mohan S, Abdelwahab SI, Wah TS. The growth suppressing effects of girinimbine on HepG2 involve induction of apoptosis and cell cycle arrest. Molecules. 2011;16:7155–70. 10.3390/molecules16087155.Suche in Google Scholar PubMed PubMed Central
[21] Arbab IA, Abdul AB, Sukari MA, Abdullah R, Syam S, Kamalidehghan B, et al. Dentatin isolated from Clausena excavata induces apoptosis in MCF7 cells through the intrinsic pathway with involvement of NFkB signalling and G0/G1 cell cycle arrest: a bioassay-guided approach. J Ethno pharmacol. 2013;145:343–54. 10.1016/j.jep.2012.11.020.Suche in Google Scholar PubMed
[22] Anassamy T, Ahmad BA, Mohd AS, Siddig IA, Syam M, Behnam K. A Phenylbutenoid dimer, cis-3- (3′, 4′-dimethoxyphenyl)-4-[(E)3′′′, 4′′′-dimethoxystyryl] cyclohex-1-ene, exhibits apoptogenic properties in T-Acute lymphoblastic leukemia cells via induction of p53-independent mitochondrial signalling pathway. Evid Based Complement Altern Med. 2013;939810:1–14. 10.1155/2013/939810.Suche in Google Scholar PubMed PubMed Central
[23] Foo JB, Yazan LS, Chan KW, Tahir PM, Ismail M. Kenaf seed oil from supercritical carbon dioxide fluid extraction induced G1 phase cell cycle arrest and apoptosis in leukemia cells. Afr J Biotechnol. 2011;10:5389–97.Suche in Google Scholar
[24] Singh M, Kumar A, Singh R, Pandey KD. Endophytic bacteria: a new source of bioactive compounds. 3 Biotech. 2017;7:1–14. 10.1007/s13205-017-0942-z.Suche in Google Scholar PubMed PubMed Central
[25] Arima H, Danno G. Isolation of antimicrobial compounds from guava (Psidium guajava L.). Biosci Biotechnol Biochem. 2002;66:1727–30. 10.1271/bbb.66.1727.Suche in Google Scholar PubMed
[26] Chah KF, Eze CA, Emuelosi CE, Esimone CO. Antibacterial and wound healing properties of methanolic extracts of some Nigerian medicinal plants. J Ethnopharmacol. 2006;140:164–7. 10.1016/j.jep.2005.08.070.Suche in Google Scholar PubMed
[27] Prabu GR, Gnanamani A, Sadulla S. Guaijaverin a plants flavonoid as potential antiplaque agent against Streptococcusmutans. J Appl Microbiol. 2006;101:487–95. 10.1111/j.1365-2672.2006.02912.x.Suche in Google Scholar PubMed
[28] Ojewole JA. Hypoglycemic and hypotensive effects of Psidium guajava Linn. (Myrtaceae) leaf aqueous extract. Methods Find Exp Clin Pharmacol. 2005;27:689–95. 10.1358/mf.2005.27.10.948917.Suche in Google Scholar PubMed
[29] Tona L, Kambu K, Ngimbi N, Cimanga K, Vlietinck AJ. Antiamoebic and phytochemical screening of some Congolese medicinal plants. J Ethnopharmacol. 1998;61:57–65. 10.1016/s0378-8741(98)00015-4.Suche in Google Scholar PubMed
[30] Manosroi J, Dhumtanom P, Manosroi A. Anti-proliferative activity of essential oil extracted from Thai medicinal plants on KB and P388 cell lines. Cancer Lett. 2006;235:114–20. 10.1016/j.canlet.2005.04.021.Suche in Google Scholar PubMed
[31] Chen KC, Hsieh CL, Peng CC, Hsieh-Li HM, Chiang KD, Peng RY. Brain derived prostate cancer DU-145 cells are effectively inhibited in vitro by guava leaf extracts. Nutr Cancer. 2007;58:93–106. 10.1080/01635580701308240.Suche in Google Scholar PubMed
[32] Qadan F, Thewaini AJ, Ali DA, Afifi R, Elkhawad A, Matalka KZ. The antimicrobial activities of Psidium guajava and Junglans regia leaf extracts to acne-developing organisms. Am Chin Med. 2005;33:197–204. 10.1142/S0192415X05002783.Suche in Google Scholar PubMed
[33] Jairaj P, Khoohaswan P, Wongkrajang Y, Peungvicha P, Suriyawong P, Saraya ML, et al. Anticough and antimicrobial activities of Psidium guajava Linn leaf extract. J Ethnopharmacol. 1999;67:203–12. 10.1016/s0378-8741(99)00022-7.Suche in Google Scholar PubMed
[34] Razak FA, Othman RY, Rahim ZH. The effect of Piper betle and Psidium guajava extracts on the cell-surface hydrophobicity of selected early settlers of dental plaque. J Oral Sci. 2006;48:71–5. 10.2334/josnusd.48.71.Suche in Google Scholar PubMed
[35] Lee SB, Park HR. Anticancer activity of guava (Psidium guajava L.) branch extract against HT-29 human colon cancer cells. J Med Plant Res. 2010;4:891–6. 10.5897/JMPR10.043.Suche in Google Scholar
[36] Manthey JA, Grohmann K, Guthrie N. Biological properties of citrus flavonoids pertaining to cancer and inflammation. Curr Med Chem. 2001;8:135–53. 10.2174/0929867013373723.Suche in Google Scholar PubMed
[37] Morgan DM. Tetrazolium (MTT) assay for cellular viability and activityPolyamine Protocols. Methods in Molecular Biology. New York, USA: Springer; 1998. p. 179–84. 10.1385/0-89603-448-8:179.Suche in Google Scholar PubMed
[38] Cory AH, Owen TC, Barltrop JA, Cory JG. Use of an aqueous soluble tetrazolium/formazan assay for cell growth assays in culture. Cancer Commun. 1991;3:207–12. 10.3727/095535491820873191.Suche in Google Scholar PubMed
[39] Skehan P, Storeng R, Scudiero D, Monks A, McMahon J, Vistica D, et al. New colorimetric cytotoxicity assay for anticancer-drug screening. J Natl Cancer Inst. 1990;82:1107–12. 10.1093/jnci/82.13.1107.Suche in Google Scholar PubMed
[40] Borenfreund E, Puerner JA. A simple quantitative procedure using monolayer cultures for cytotoxicity assays (HTD/NR-90). J Tissue Cult Methods. 1985;9:7–9. 10.1007/BF01666038.Suche in Google Scholar
[41] Rizzo LY, Longato GB, Ruiz AL, Tinti SV, Possenti A, Vendramini-Costa DB, et al. In vitro, in vivo and in silico analysis of the anticancer and estrogen-like activity of guava leaf extracts. Curr Med Chem. 2014;21:2322–30. 10.2174/0929867321666140120120031.Suche in Google Scholar PubMed
[42] Qin XJ, Yu Q, Yan H, Khan A, Feng MY, Li PP, et al. Meroterpenoids with antitumor activities from guava (Psidium guajava). J Agric Food Chem. 2017;65:4993–9. 10.1021/acs.jafc.7b01762.Suche in Google Scholar PubMed
[43] Zhu X, Ouyang W, Pan C, Gao Z, Han Y, Song M, et al. Identification of a new benzophenone from Psidium guajava L. leaf and its antineoplastic effects on human colon cancer cells. Food Funct. 2019;10:4189–98. 10.1039/C9FO00569B.Suche in Google Scholar PubMed
[44] Bazioli JM, Costa JH, Shiozawa L. Ruiz ALTG, Foglio MA, Carvalho JED. Antiestrogenic activity of guajadial fraction, from guava leaf (Psidium guajava L.). Molecules. 2020;25(1525):1–21. 10.3390/molecules25071525.Suche in Google Scholar PubMed PubMed Central
[45] Feng XH, Wang ZH, Meng DL, Li X. Cytotoxic and antioxidant constituents from the leaf of Psidium guajava. Bioorganic Med Chem Lett. 2015;25:2193–8. 10.1016/j.bmcl.2015.03.058.Suche in Google Scholar PubMed
[46] Lok B, Sandai D, Baharetha HM, Nazari VM, Asif M, Tan CS, et al. Anticancer effect of Psidium guajava (Guava) leaf extracts against colorectal cancer through inhibition of angiogenesis. Asian Pac J Trop Biomed. 2020;10:293–307. 10.4103/2221-1691.284944.Suche in Google Scholar
[47] Manikyam HK, Joshi SK, Vakadi S, Patil SB. Anticancer activity of terpenoid saponin extract of Psidium guajava on MCF-7 cancer cell line using DAPI and MTT assays. Afr J Pharm Pharm. 2021;15:206–11. 10.5897/AJPP2020.5216 Suche in Google Scholar
[48] Singh PK, Singh J, Medhi T, Kumar A. Phytochemical screening, quantification, FT-IR analysis, and in silico characterization of potential bio-active compounds identified in HR-LC/MS analysis of the polyherbal formulation from Northeast India. ACS Omega. 2022;7:33067–78. 10.1021/acsomega.2c03117.Suche in Google Scholar PubMed PubMed Central
[49] Doyle A, Griffiths JB. Cell and tissue culture for medical research. New York: John Wiley & Sons; 2000.Suche in Google Scholar
[50] Adeyemi SO, Akanji MA, Oguntoye SA. Ethanolic leaf extract of Psidium Guajava: phytochemical and trypanocidal activity in rats infected with Trypanosoma Brucei Brucei. J Med Plant Res. 2009;3:420–3. 10.5897/JMPR.9000580.Suche in Google Scholar
[51] Adjo AJ, Lin SX. Comparison of functional proteomic analyses of human breast cancer cell lines T47D and MCF7. PLoS One. 2012;7(2):e31532. 10.1371/journal.pone.0031532.Suche in Google Scholar PubMed PubMed Central
[52] Wen SH, Su SC, Liou BH, Lin CH, Lee KR. Sulbactam-enhanced cytotoxicity of doxorubicin in breast cancer cells. Cancer Cell Int. 2018;18(128):1–18. 10.1186/s12935-018-0625-9.Suche in Google Scholar PubMed PubMed Central
[53] Seber S, Sirin DY, Yetisyigit T, Bilgen T. Piperlongumine increases the apoptotic effect of doxorubicin and paclitaxel in a cervical cancer cell line. Niger J Clin Pract. 2020;23(3):386–91. 10.4103/njcp.njcp_80_19.Suche in Google Scholar PubMed
[54] Hasoon MRA, Kadhim NJ. Improvement of the selectivity index (SI) and cytotoxicity activity of doxorubicin drug by panax ginseng plant extract. Arch Razi Inst. 2021;76(3):659–66. 10.22092/ari.2021.355413.1681.Suche in Google Scholar PubMed PubMed Central
[55] Boik J. Natural compounds in cancer therapy. Minnesota: Oregon Medical Press; 2001.Suche in Google Scholar
[56] Lee CC, Houghton P. Cytotoxicity of plants from Malaysia and Thailand used traditionally to treat cancer. J Ethnopharmacol. 2005;100(3):237–43. 10.1016/j.jep.2005.01.064.Suche in Google Scholar PubMed
[57] Rojas-Garbanzo C, Zimmermann BF, Schulze-Kaysers N, Schieber A. Characterization of phenolic and other polar compounds in peel and fleshof pink guava (Psidium guajava L. cv. ‘Criolla’) by ultra-high performanceliquid chromatography with diode array and massspectrometric detection. Food Res Int. 2017;100:445–53. 10.1016/j.foodres.2016.12.004.Suche in Google Scholar PubMed
[58] Moon JY, Mosaddik A, Kim H, Cho M, Choi HK, Kim YS, et al. The chloroform fraction of guava (Psidium cattleianum sabine) leaf extractinhibits human gastric cancer cell proliferation via induction of apoptosis. Food Chem. 2011;125(2):369–75. 10.1016/j.foodchem.2010.09.007.Suche in Google Scholar
[59] Fang N, Yu S, Prior RL. LC-MS/MS characterization of phenolic constituents in dried plums. J Agric Food Chem. 2002;50(12):3579–85. 10.1021/jf0201327.Suche in Google Scholar PubMed
[60] Díaz-de-Cerio E, Verardo V, Gómez-Caravaca AM, Fernández-Gutiérre A, Segura-Carretero A. Exploratory characterization of phenolic compounds with demonstrated anti-diabetic activity in guava leaves at different oxidation states. Int J Mol Sci. 2016;17(5):699–712.10.3390/ijms17050699Suche in Google Scholar PubMed PubMed Central
[61] Chiari BG, Trovatti E, Marto J, Almeida MGJ, Melero A, Corrêa MA, et al. Guava: phytochemical composition of a potential source of antioxidants for cosmetic and/or dermatological applications. Braz J Pharm Sci. 2017;53(2):1–10. 10.1590/s2175-97902017000216141.Suche in Google Scholar
[62] Flores G, Wu S, Negrin A, Kennelly EJ. Chemical composition and antioxidant activity of seven cultivars of guava (Psidium guajava) fruits. Food Chem. 2015;170:327–35. 10.1016/j.foodchem.2014.08.076.Suche in Google Scholar PubMed
[63] Ismail T, Calcabrini C, Diaz AR, Fimognari C, Turrini E, Catanzaro E, et al. Review: Ellagitannins in cancer chemopreventionand therapy. Toxins. 2016;8(5):151–73. 10.3390/toxins8050151.Suche in Google Scholar PubMed PubMed Central
© 2023 the author(s), published by De Gruyter
This work is licensed under the Creative Commons Attribution 4.0 International License.
Artikel in diesem Heft
- Characteristics, source, and health risk assessment of aerosol polyaromatic hydrocarbons in the rural and urban regions of western Saudi Arabia
- Regular Articles
- A network-based correlation research between element electronegativity and node importance
- Pomegranate attenuates kidney injury in cyclosporine-induced nephrotoxicity in rats by suppressing oxidative stress
- Ab initio study of fundamental properties of XInO3 (X = K, Rb, Cs) perovskites
- Responses of feldspathic sandstone and sand-reconstituted soil C and N to freeze–thaw cycles
- Robust fractional control based on high gain observers design (RNFC) for a Spirulina maxima culture interfaced with an advanced oxidation process
- Study on arsenic speciation and redistribution mechanism in Lonicera japonica plants via synchrotron techniques
- Optimization of machining Nilo 36 superalloy parameters in turning operation
- Vacuum impregnation pre-treatment: A novel method for incorporating mono- and divalent cations into potato strips to reduce the acrylamide formation in French fries
- Characterization of effective constituents in Acanthopanax senticosus fruit for blood deficiency syndrome based on the chinmedomics strategy
- Comparative analysis of the metabolites in Pinellia ternata from two producing regions using ultra-high-performance liquid chromatography–electrospray ionization–tandem mass spectrometry
- The assessment of environmental parameter along the desalination plants in the Kingdom of Saudi Arabia
- Effects of harpin and carbendazim on antioxidant accumulation in young jujube leaves
- The effects of in ovo injected with sodium borate on hatching performance and small intestine morphology in broiler chicks
- Optimization of cutting forces and surface roughness via ANOVA and grey relational analysis in machining of In718
- Essential oils of Origanum compactum Benth: Chemical characterization, in vitro, in silico, antioxidant, and antibacterial activities
- Translocation of tungsten(vi) oxide/gadolinium(iii) fluoride in tellurite glasses towards improvement of gamma-ray attenuation features in high-density glass shields
- Mechanical properties, elastic moduli, and gamma ray attenuation competencies of some TeO2–WO3–GdF3 glasses: Tailoring WO3–GdF3 substitution toward optimum behavioral state range
- Comparison between the CIDR or sponge with hormone injection to induce estrus synchronization for twining and sex preselection in Naimi sheep
- Exergetic performance analyses of three different cogeneration plants
- Psoralea corylifolia (babchi) seeds enhance proliferation of normal human cultured melanocytes: GC–MS profiling and biological investigation
- A novel electrochemical micro-titration method for quantitative evaluation of the DPPH free radical scavenging capacity of caffeic acid
- Comparative study between supported bimetallic catalysts for nitrate remediation in water
- Persicaline, an alkaloid from Salvadora persica, inhibits proliferation and induces apoptosis and cell-cycle arrest in MCF-7 cells
- Determination of nicotine content in locally produced smokeless tobacco (Shammah) samples from Jazan region of Saudi Arabia using a convenient HPLC-MS/MS method
- Changes in oxidative stress markers in pediatric burn injury over a 1-week period
- Integrated geophysical techniques applied for petroleum basins structural characterization in the central part of the Western Desert, Egypt
- The impact of chemical modifications on gamma-ray attenuation properties of some WO3-reinforced tellurite glasses
- Microwave and Cs+-assisted chemo selective reaction protocol for synthesizing 2-styryl quinoline biorelevant molecules
- Structural, physical, and radiation absorption properties of a significant nuclear power plant component: A comparison between REX-734 and 316L SS austenitic stainless steels
- Effect of Moringa oleifera on serum YKL-40 level: In vivo rat periodontitis model
- Investigating the impact of CO2 emissions on the COVID-19 pandemic by generalized linear mixed model approach with inverse Gaussian and gamma distributions
- Influence of WO3 content on gamma rays attenuation characteristics of phosphate glasses at low energy range
- Study on CO2 absorption performance of ternary DES formed based on DEA as promoting factor
- Performance analyses of detonation engine cogeneration cycles
- Sterols from Centaurea pumilio L. with cell proliferative activity: In vitro and in silico studies
- Untargeted metabolomics revealing changes in aroma substances in flue-cured tobacco
- Effect of pumpkin enriched with calcium lactate on iron status in an animal model of postmenopausal osteoporosis
- Energy consumption, mechanical and metallographic properties of cryogenically treated tool steels
- Optimization of ultra-high pressure-assisted extraction of total phenols from Eucommia ulmoides leaves by response surface methodology
- Harpin enhances antioxidant nutrient accumulation and decreases enzymatic browning in stored soybean sprouts
- Physicochemical and biological properties of carvacrol
- Radix puerariae in the treatment of diabetic nephropathy: A network pharmacology analysis and experimental validation
- Anti-Alzheimer, antioxidants, glucose-6-phosphate dehydrogenase effects of Taverniera glabra mediated ZnO and Fe2O3 nanoparticles in alloxan-induced diabetic rats
- Experimental study on photocatalytic CO2 reduction performance of ZnS/CdS-TiO2 nanotube array thin films
- Epoxy-reinforced heavy metal oxides for gamma ray shielding purposes
- Black mulberry (Morus nigra L.) fruits: As a medicinal plant rich in human health-promoting compounds
- Promising antioxidant and antimicrobial effects of essential oils extracted from fruits of Juniperus thurifera: In vitro and in silico investigations
- Chloramine-T-induced oxidation of Rizatriptan Benzoate: An integral chemical and spectroscopic study of products, mechanisms and kinetics
- Study on antioxidant and antimicrobial potential of chemically profiled essential oils extracted from Juniperus phoenicea (L.) by use of in vitro and in silico approaches
- Screening and characterization of fungal taxol-producing endophytic fungi for evaluation of antimicrobial and anticancer activities
- Mineral composition, principal polyphenolic components, and evaluation of the anti-inflammatory, analgesic, and antioxidant properties of Cytisus villosus Pourr leaf extracts
- In vitro antiproliferative efficacy of Annona muricata seed and fruit extracts on several cancer cell lines
- An experimental study for chemical characterization of artificial anterior cruciate ligament with coated chitosan as biomaterial
- Prevalence of residual risks of the transfusion-transmitted infections in Riyadh hospitals: A two-year retrospective study
- Computational and experimental investigation of antibacterial and antifungal properties of Nicotiana tabacum extracts
- Reinforcement of cementitious mortars with hemp fibers and shives
- X-ray shielding properties of bismuth-borate glass doped with rare earth ions
- Green supported silver nanoparticles over modified reduced graphene oxide: Investigation of its antioxidant and anti-ovarian cancer effects
- Orthogonal synthesis of a versatile building block for dual functionalization of targeting vectors
- Thymbra spicata leaf extract driven biogenic synthesis of Au/Fe3O4 nanocomposite and its bio-application in the treatment of different types of leukemia
- The role of Ag2O incorporation in nuclear radiation shielding behaviors of the Li2O–Pb3O4–SiO2 glass system: A multi-step characterization study
- A stimuli-responsive in situ spray hydrogel co-loaded with naringenin and gentamicin for chronic wounds
- Assessment of the impact of γ-irradiation on the piperine content and microbial quality of black pepper
- Antioxidant, sensory, and functional properties of low-alcoholic IPA beer with Pinus sylvestris L. shoots addition fermented using unconventional yeast
- Screening and optimization of extracellular pectinase produced by Bacillus thuringiensis SH7
- Determination of polyphenols in Chinese jujube using ultra-performance liquid chromatography–mass spectrometry
- Synergistic effects of harpin and NaCl in determining soybean sprout quality under non-sterile conditions
- Field evaluation of different eco-friendly alternative control methods against Panonychus citri [Acari: Tetranychidae] spider mite and its predators in citrus orchards
- Exploring the antimicrobial potential of biologically synthesized zero valent iron nanoparticles
- NaCl regulates goldfish growth and survival at three food supply levels under hypoxia
- An exploration of the physical, optical, mechanical, and radiation shielding properties of PbO–MgO–ZnO–B2O3 glasses
- A novel statistical modeling of air pollution and the COVID-19 pandemic mortality data by Poisson, geometric, and negative binomial regression models with fixed and random effects
- Treatment activity of the injectable hydrogels loaded with dexamethasone In(iii) complex on glioma by inhibiting the VEGF signaling pathway
- An alternative approach for the excess lifetime cancer risk and prediction of radiological parameters
- Panax ginseng leaf aqueous extract mediated green synthesis of AgNPs under ultrasound condition and investigation of its anti-lung adenocarcinoma effects
- Study of hydrolysis and production of instant ginger (Zingiber officinale) tea
- Novel green synthesis of zinc oxide nanoparticles using Salvia rosmarinus extract for treatment of human lung cancer
- Evaluation of second trimester plasma lipoxin A4, VEGFR-1, IL-6, and TNF-α levels in pregnant women with gestational diabetes mellitus
- Antidiabetic, antioxidant and cytotoxicity activities of ortho- and para-substituted Schiff bases derived from metformin hydrochloride: Validation by molecular docking and in silico ADME studies
- Antioxidant, antidiabetic, antiglaucoma, and anticholinergic effects of Tayfi grape (Vitis vinifera): A phytochemical screening by LC-MS/MS analysis
- Identification of genetic polymorphisms in the stearoyl CoA desaturase gene and its association with milk quality traits in Najdi sheep
- Cold-acclimation effect on cadmium absorption and biosynthesis of polyphenolics, and free proline and photosynthetic pigments in Spirogyra aequinoctialis
- Analysis of secondary metabolites in Xinjiang Morus nigra leaves using different extraction methods with UPLC-Q/TOF-MS/MS technology
- Nanoarchitectonics and performance evaluation of a Fe3O4-stabilized Pickering emulsion-type differential pressure plugging agent
- Investigating pyrolysis characteristics of Shengdong coal through Py-GC/MS
- Extraction, phytochemical characterization, and antifungal activity of Salvia rosmarinus extract
- Introducing a novel and natural antibiotic for the treatment of oral pathogens: Abelmoschus esculentus green-formulated silver nanoparticles
- Optimization of gallic acid-enriched ultrasonic-assisted extraction from mango peels
- Effect of gamma rays irradiation in the structure, optical, and electrical properties of samarium doped bismuth titanate ceramics
- Combinatory in silico investigation for potential inhibitors from Curcuma sahuynhensis Škorničk. & N.S. Lý volatile phytoconstituents against influenza A hemagglutinin, SARS-CoV-2 main protease, and Omicron-variant spike protein
- Physical, mechanical, and gamma ray shielding properties of the Bi2O3–BaO–B2O3–ZnO–As2O3–MgO–Na2O glass system
- Twofold interpenetrated 3D Cd(ii) complex: Crystal structure and luminescent property
- Study on the microstructure and soil quality variation of composite soil with soft rock and sand
- Ancient spring waters still emerging and accessible in the Roman Forum area: Chemical–physical and microbiological characterization
- Extraction and characterization of type I collagen from scales of Mexican Biajaiba fish
- Finding small molecular compounds to decrease trimethylamine oxide levels in atherosclerosis by virtual screening
- Prefatory in silico studies and in vitro insecticidal effect of Nigella sativa (L.) essential oil and its active compound (carvacrol) against the Callosobruchus maculatus adults (Fab), a major pest of chickpea
- Polymerized methyl imidazole silver bromide (CH3C6H5AgBr)6: Synthesis, crystal structures, and catalytic activity
- Using calcined waste fish bones as a green solid catalyst for biodiesel production from date seed oil
- Influence of the addition of WO3 on TeO2–Na2O glass systems in view of the feature of mechanical, optical, and photon attenuation
- Naringin ameliorates 5-fluorouracil elicited neurotoxicity by curtailing oxidative stress and iNOS/NF-ĸB/caspase-3 pathway
- GC-MS profile of extracts of an endophytic fungus Alternaria and evaluation of its anticancer and antibacterial potentialities
- Green synthesis, chemical characterization, and antioxidant and anti-colorectal cancer effects of vanadium nanoparticles
- Determination of caffeine content in coffee drinks prepared in some coffee shops in the local market in Jeddah City, Saudi Arabia
- A new 3D supramolecular Cu(ii) framework: Crystal structure and photocatalytic characteristics
- Bordeaux mixture accelerates ripening, delays senescence, and promotes metabolite accumulation in jujube fruit
- Important application value of injectable hydrogels loaded with omeprazole Schiff base complex in the treatment of pancreatitis
- Color tunable benzothiadiazole-based small molecules for lightening applications
- Investigation of structural, dielectric, impedance, and mechanical properties of hydroxyapatite-modified barium titanate composites for biomedical applications
- Metal gel particles loaded with epidermal cell growth factor promote skin wound repair mechanism by regulating miRNA
- In vitro exploration of Hypsizygus ulmarius (Bull.) mushroom fruiting bodies: Potential antidiabetic and anti-inflammatory agent
- Alteration in the molecular structure of the adenine base exposed to gamma irradiation: An ESR study
- Comprehensive study of optical, thermal, and gamma-ray shielding properties of Bi2O3–ZnO–PbO–B2O3 glasses
- Lewis acids as co-catalysts in Pd-based catalyzed systems of the octene-1 hydroethoxycarbonylation reaction
- Synthesis, Hirshfeld surface analysis, thermal, and selective α-glucosidase inhibitory studies of Schiff base transition metal complexes
- Protective properties of AgNPs green-synthesized by Abelmoschus esculentus on retinal damage on the virtue of its anti-inflammatory and antioxidant effects in diabetic rat
- Effects of green decorated AgNPs on lignin-modified magnetic nanoparticles mediated by Cydonia on cecal ligation and puncture-induced sepsis
- Treatment of gastric cancer by green mediated silver nanoparticles using Pistacia atlantica bark aqueous extract
- Preparation of newly developed porcelain ceramics containing WO3 nanoparticles for radiation shielding applications
- Utilization of computational methods for the identification of new natural inhibitors of human neutrophil elastase in inflammation therapy
- Some anticancer agents as effective glutathione S-transferase (GST) inhibitors
- Clay-based bricks’ rich illite mineral for gamma-ray shielding applications: An experimental evaluation of the effect of pressure rates on gamma-ray attenuation parameters
- Stability kinetics of orevactaene pigments produced by Epicoccum nigrum in solid-state fermentation
- Treatment of denture stomatitis using iron nanoparticles green-synthesized by Silybum marianum extract
- Characterization and antioxidant potential of white mustard (Brassica hirta) leaf extract and stabilization of sunflower oil
- Characteristics of Langmuir monomolecular monolayers formed by the novel oil blends
- Strategies for optimizing the single GdSrFeO4 phase synthesis
- Oleic acid and linoleic acid nanosomes boost immunity and provoke cell death via the upregulation of beta-defensin-4 at genetic and epigenetic levels
- Unraveling the therapeutic potential of Bombax ceiba roots: A comprehensive study of chemical composition, heavy metal content, antibacterial activity, and in silico analysis
- Green synthesis of AgNPs using plant extract and investigation of its anti-human colorectal cancer application
- The adsorption of naproxen on adsorbents obtained from pepper stalk extract by green synthesis
- Treatment of gastric cancer by silver nanoparticles encapsulated by chitosan polymers mediated by Pistacia atlantica extract under ultrasound condition
- In vitro protective and anti-inflammatory effects of Capparis spinosa and its flavonoids profile
- Wear and corrosion behavior of TiC and WC coatings deposited on high-speed steels by electro-spark deposition
- Therapeutic effects of green-formulated gold nanoparticles by Origanum majorana on spinal cord injury in rats
- Melanin antibacterial activity of two new strains, SN1 and SN2, of Exophiala phaeomuriformis against five human pathogens
- Evaluation of the analgesic and anesthetic properties of silver nanoparticles supported over biodegradable acacia gum-modified magnetic nanoparticles
- Review Articles
- Role and mechanism of fruit waste polyphenols in diabetes management
- A comprehensive review of non-alkaloidal metabolites from the subfamily Amaryllidoideae (Amaryllidaceae)
- Discovery of the chemical constituents, structural characteristics, and pharmacological functions of Chinese caterpillar fungus
- Eco-friendly green approach of nickel oxide nanoparticles for biomedical applications
- Advances in the pharmaceutical research of curcumin for oral administration
- Rapid Communication
- Determination of the contents of bioactive compounds in St. John’s wort (Hypericum perforatum): Comparison of commercial and wild samples
- Retraction
- Retraction of “Two mixed-ligand coordination polymers based on 2,5-thiophenedicarboxylic acid and flexible N-donor ligands: The protective effect on periodontitis via reducing the release of IL-1β and TNF-α”
- Topical Issue on Phytochemicals, biological and toxicological analysis of aromatic medicinal plants
- Anti-plasmodial potential of selected medicinal plants and a compound Atropine isolated from Eucalyptus obliqua
- Anthocyanin extract from black rice attenuates chronic inflammation in DSS-induced colitis mouse model by modulating the gut microbiota
- Evaluation of antibiofilm and cytotoxicity effect of Rumex vesicarius methanol extract
- Chemical compositions of Litsea umbellata and inhibition activities
- Green synthesis, characterization of silver nanoparticles using Rhynchosia capitata leaf extract and their biological activities
- GC-MS analysis and antibacterial activities of some plants belonging to the genus Euphorbia on selected bacterial isolates
- The abrogative effect of propolis on acrylamide-induced toxicity in male albino rats: Histological study
- A phytoconstituent 6-aminoflavone ameliorates lipopolysaccharide-induced oxidative stress mediated synapse and memory dysfunction via p-Akt/NF-kB pathway in albino mice
- Anti-diabetic potentials of Sorbaria tomentosa Lindl. Rehder: Phytochemistry (GC-MS analysis), α-amylase, α-glucosidase inhibitory, in vivo hypoglycemic, and biochemical analysis
- Assessment of cytotoxic and apoptotic activities of the Cassia angustifolia aqueous extract against SW480 colon cancer
- Biochemical analysis, antioxidant, and antibacterial efficacy of the bee propolis extract (Hymenoptera: Apis mellifera) against Staphylococcus aureus-induced infection in BALB/c mice: In vitro and in vivo study
- Assessment of essential elements and heavy metals in Saudi Arabian rice samples underwent various processing methods
- Two new compounds from leaves of Capparis dongvanensis (Sy, B. H. Quang & D. V. Hai) and inhibition activities
- Hydroxyquinoline sulfanilamide ameliorates STZ-induced hyperglycemia-mediated amyleoid beta burden and memory impairment in adult mice
- An automated reading of semi-quantitative hemagglutination results in microplates: Micro-assay for plant lectins
- Inductively coupled plasma mass spectrometry assessment of essential and toxic trace elements in traditional spices consumed by the population of the Middle Eastern region in their recipes
- Phytochemical analysis and anticancer activity of the Pithecellobium dulce seed extract in colorectal cancer cells
- Impact of climatic disturbances on the chemical compositions and metabolites of Salvia officinalis
- Physicochemical characterization, antioxidant and antifungal activities of essential oils of Urginea maritima and Allium sativum
- Phytochemical analysis and antifungal efficiency of Origanum majorana extracts against some phytopathogenic fungi causing tomato damping-off diseases
- Special Issue on 4th IC3PE
- Graphene quantum dots: A comprehensive overview
- Studies on the intercalation of calcium–aluminium layered double hydroxide-MCPA and its controlled release mechanism as a potential green herbicide
- Synergetic effect of adsorption and photocatalysis by zinc ferrite-anchored graphitic carbon nitride nanosheet for the removal of ciprofloxacin under visible light irradiation
- Exploring anticancer activity of the Indonesian guava leaf (Psidium guajava L.) fraction on various human cancer cell lines in an in vitro cell-based approach
- The comparison of gold extraction methods from the rock using thiourea and thiosulfate
- Special Issue on Marine environmental sciences and significance of the multidisciplinary approaches
- Sorption of alkylphenols and estrogens on microplastics in marine conditions
- Cytotoxic ketosteroids from the Red Sea soft coral Dendronephthya sp.
- Antibacterial and biofilm prevention metabolites from Acanthophora spicifera
- Characteristics, source, and health risk assessment of aerosol polyaromatic hydrocarbons in the rural and urban regions of western Saudi Arabia
- Special Issue on Advanced Nanomaterials for Energy, Environmental and Biological Applications - Part II
- Green synthesis, characterization, and evaluation of antibacterial activities of cobalt nanoparticles produced by marine fungal species Periconia prolifica
- Combustion-mediated sol–gel preparation of cobalt-doped ZnO nanohybrids for the degradation of acid red and antibacterial performance
- Perinatal supplementation with selenium nanoparticles modified with ascorbic acid improves hepatotoxicity in rat gestational diabetes
- Evaluation and chemical characterization of bioactive secondary metabolites from endophytic fungi associated with the ethnomedicinal plant Bergenia ciliata
- Enhancing photovoltaic efficiency with SQI-Br and SQI-I sensitizers: A comparative analysis
- Nanostructured p-PbS/p-CuO sulfide/oxide bilayer heterojunction as a promising photoelectrode for hydrogen gas generation
Artikel in diesem Heft
- Characteristics, source, and health risk assessment of aerosol polyaromatic hydrocarbons in the rural and urban regions of western Saudi Arabia
- Regular Articles
- A network-based correlation research between element electronegativity and node importance
- Pomegranate attenuates kidney injury in cyclosporine-induced nephrotoxicity in rats by suppressing oxidative stress
- Ab initio study of fundamental properties of XInO3 (X = K, Rb, Cs) perovskites
- Responses of feldspathic sandstone and sand-reconstituted soil C and N to freeze–thaw cycles
- Robust fractional control based on high gain observers design (RNFC) for a Spirulina maxima culture interfaced with an advanced oxidation process
- Study on arsenic speciation and redistribution mechanism in Lonicera japonica plants via synchrotron techniques
- Optimization of machining Nilo 36 superalloy parameters in turning operation
- Vacuum impregnation pre-treatment: A novel method for incorporating mono- and divalent cations into potato strips to reduce the acrylamide formation in French fries
- Characterization of effective constituents in Acanthopanax senticosus fruit for blood deficiency syndrome based on the chinmedomics strategy
- Comparative analysis of the metabolites in Pinellia ternata from two producing regions using ultra-high-performance liquid chromatography–electrospray ionization–tandem mass spectrometry
- The assessment of environmental parameter along the desalination plants in the Kingdom of Saudi Arabia
- Effects of harpin and carbendazim on antioxidant accumulation in young jujube leaves
- The effects of in ovo injected with sodium borate on hatching performance and small intestine morphology in broiler chicks
- Optimization of cutting forces and surface roughness via ANOVA and grey relational analysis in machining of In718
- Essential oils of Origanum compactum Benth: Chemical characterization, in vitro, in silico, antioxidant, and antibacterial activities
- Translocation of tungsten(vi) oxide/gadolinium(iii) fluoride in tellurite glasses towards improvement of gamma-ray attenuation features in high-density glass shields
- Mechanical properties, elastic moduli, and gamma ray attenuation competencies of some TeO2–WO3–GdF3 glasses: Tailoring WO3–GdF3 substitution toward optimum behavioral state range
- Comparison between the CIDR or sponge with hormone injection to induce estrus synchronization for twining and sex preselection in Naimi sheep
- Exergetic performance analyses of three different cogeneration plants
- Psoralea corylifolia (babchi) seeds enhance proliferation of normal human cultured melanocytes: GC–MS profiling and biological investigation
- A novel electrochemical micro-titration method for quantitative evaluation of the DPPH free radical scavenging capacity of caffeic acid
- Comparative study between supported bimetallic catalysts for nitrate remediation in water
- Persicaline, an alkaloid from Salvadora persica, inhibits proliferation and induces apoptosis and cell-cycle arrest in MCF-7 cells
- Determination of nicotine content in locally produced smokeless tobacco (Shammah) samples from Jazan region of Saudi Arabia using a convenient HPLC-MS/MS method
- Changes in oxidative stress markers in pediatric burn injury over a 1-week period
- Integrated geophysical techniques applied for petroleum basins structural characterization in the central part of the Western Desert, Egypt
- The impact of chemical modifications on gamma-ray attenuation properties of some WO3-reinforced tellurite glasses
- Microwave and Cs+-assisted chemo selective reaction protocol for synthesizing 2-styryl quinoline biorelevant molecules
- Structural, physical, and radiation absorption properties of a significant nuclear power plant component: A comparison between REX-734 and 316L SS austenitic stainless steels
- Effect of Moringa oleifera on serum YKL-40 level: In vivo rat periodontitis model
- Investigating the impact of CO2 emissions on the COVID-19 pandemic by generalized linear mixed model approach with inverse Gaussian and gamma distributions
- Influence of WO3 content on gamma rays attenuation characteristics of phosphate glasses at low energy range
- Study on CO2 absorption performance of ternary DES formed based on DEA as promoting factor
- Performance analyses of detonation engine cogeneration cycles
- Sterols from Centaurea pumilio L. with cell proliferative activity: In vitro and in silico studies
- Untargeted metabolomics revealing changes in aroma substances in flue-cured tobacco
- Effect of pumpkin enriched with calcium lactate on iron status in an animal model of postmenopausal osteoporosis
- Energy consumption, mechanical and metallographic properties of cryogenically treated tool steels
- Optimization of ultra-high pressure-assisted extraction of total phenols from Eucommia ulmoides leaves by response surface methodology
- Harpin enhances antioxidant nutrient accumulation and decreases enzymatic browning in stored soybean sprouts
- Physicochemical and biological properties of carvacrol
- Radix puerariae in the treatment of diabetic nephropathy: A network pharmacology analysis and experimental validation
- Anti-Alzheimer, antioxidants, glucose-6-phosphate dehydrogenase effects of Taverniera glabra mediated ZnO and Fe2O3 nanoparticles in alloxan-induced diabetic rats
- Experimental study on photocatalytic CO2 reduction performance of ZnS/CdS-TiO2 nanotube array thin films
- Epoxy-reinforced heavy metal oxides for gamma ray shielding purposes
- Black mulberry (Morus nigra L.) fruits: As a medicinal plant rich in human health-promoting compounds
- Promising antioxidant and antimicrobial effects of essential oils extracted from fruits of Juniperus thurifera: In vitro and in silico investigations
- Chloramine-T-induced oxidation of Rizatriptan Benzoate: An integral chemical and spectroscopic study of products, mechanisms and kinetics
- Study on antioxidant and antimicrobial potential of chemically profiled essential oils extracted from Juniperus phoenicea (L.) by use of in vitro and in silico approaches
- Screening and characterization of fungal taxol-producing endophytic fungi for evaluation of antimicrobial and anticancer activities
- Mineral composition, principal polyphenolic components, and evaluation of the anti-inflammatory, analgesic, and antioxidant properties of Cytisus villosus Pourr leaf extracts
- In vitro antiproliferative efficacy of Annona muricata seed and fruit extracts on several cancer cell lines
- An experimental study for chemical characterization of artificial anterior cruciate ligament with coated chitosan as biomaterial
- Prevalence of residual risks of the transfusion-transmitted infections in Riyadh hospitals: A two-year retrospective study
- Computational and experimental investigation of antibacterial and antifungal properties of Nicotiana tabacum extracts
- Reinforcement of cementitious mortars with hemp fibers and shives
- X-ray shielding properties of bismuth-borate glass doped with rare earth ions
- Green supported silver nanoparticles over modified reduced graphene oxide: Investigation of its antioxidant and anti-ovarian cancer effects
- Orthogonal synthesis of a versatile building block for dual functionalization of targeting vectors
- Thymbra spicata leaf extract driven biogenic synthesis of Au/Fe3O4 nanocomposite and its bio-application in the treatment of different types of leukemia
- The role of Ag2O incorporation in nuclear radiation shielding behaviors of the Li2O–Pb3O4–SiO2 glass system: A multi-step characterization study
- A stimuli-responsive in situ spray hydrogel co-loaded with naringenin and gentamicin for chronic wounds
- Assessment of the impact of γ-irradiation on the piperine content and microbial quality of black pepper
- Antioxidant, sensory, and functional properties of low-alcoholic IPA beer with Pinus sylvestris L. shoots addition fermented using unconventional yeast
- Screening and optimization of extracellular pectinase produced by Bacillus thuringiensis SH7
- Determination of polyphenols in Chinese jujube using ultra-performance liquid chromatography–mass spectrometry
- Synergistic effects of harpin and NaCl in determining soybean sprout quality under non-sterile conditions
- Field evaluation of different eco-friendly alternative control methods against Panonychus citri [Acari: Tetranychidae] spider mite and its predators in citrus orchards
- Exploring the antimicrobial potential of biologically synthesized zero valent iron nanoparticles
- NaCl regulates goldfish growth and survival at three food supply levels under hypoxia
- An exploration of the physical, optical, mechanical, and radiation shielding properties of PbO–MgO–ZnO–B2O3 glasses
- A novel statistical modeling of air pollution and the COVID-19 pandemic mortality data by Poisson, geometric, and negative binomial regression models with fixed and random effects
- Treatment activity of the injectable hydrogels loaded with dexamethasone In(iii) complex on glioma by inhibiting the VEGF signaling pathway
- An alternative approach for the excess lifetime cancer risk and prediction of radiological parameters
- Panax ginseng leaf aqueous extract mediated green synthesis of AgNPs under ultrasound condition and investigation of its anti-lung adenocarcinoma effects
- Study of hydrolysis and production of instant ginger (Zingiber officinale) tea
- Novel green synthesis of zinc oxide nanoparticles using Salvia rosmarinus extract for treatment of human lung cancer
- Evaluation of second trimester plasma lipoxin A4, VEGFR-1, IL-6, and TNF-α levels in pregnant women with gestational diabetes mellitus
- Antidiabetic, antioxidant and cytotoxicity activities of ortho- and para-substituted Schiff bases derived from metformin hydrochloride: Validation by molecular docking and in silico ADME studies
- Antioxidant, antidiabetic, antiglaucoma, and anticholinergic effects of Tayfi grape (Vitis vinifera): A phytochemical screening by LC-MS/MS analysis
- Identification of genetic polymorphisms in the stearoyl CoA desaturase gene and its association with milk quality traits in Najdi sheep
- Cold-acclimation effect on cadmium absorption and biosynthesis of polyphenolics, and free proline and photosynthetic pigments in Spirogyra aequinoctialis
- Analysis of secondary metabolites in Xinjiang Morus nigra leaves using different extraction methods with UPLC-Q/TOF-MS/MS technology
- Nanoarchitectonics and performance evaluation of a Fe3O4-stabilized Pickering emulsion-type differential pressure plugging agent
- Investigating pyrolysis characteristics of Shengdong coal through Py-GC/MS
- Extraction, phytochemical characterization, and antifungal activity of Salvia rosmarinus extract
- Introducing a novel and natural antibiotic for the treatment of oral pathogens: Abelmoschus esculentus green-formulated silver nanoparticles
- Optimization of gallic acid-enriched ultrasonic-assisted extraction from mango peels
- Effect of gamma rays irradiation in the structure, optical, and electrical properties of samarium doped bismuth titanate ceramics
- Combinatory in silico investigation for potential inhibitors from Curcuma sahuynhensis Škorničk. & N.S. Lý volatile phytoconstituents against influenza A hemagglutinin, SARS-CoV-2 main protease, and Omicron-variant spike protein
- Physical, mechanical, and gamma ray shielding properties of the Bi2O3–BaO–B2O3–ZnO–As2O3–MgO–Na2O glass system
- Twofold interpenetrated 3D Cd(ii) complex: Crystal structure and luminescent property
- Study on the microstructure and soil quality variation of composite soil with soft rock and sand
- Ancient spring waters still emerging and accessible in the Roman Forum area: Chemical–physical and microbiological characterization
- Extraction and characterization of type I collagen from scales of Mexican Biajaiba fish
- Finding small molecular compounds to decrease trimethylamine oxide levels in atherosclerosis by virtual screening
- Prefatory in silico studies and in vitro insecticidal effect of Nigella sativa (L.) essential oil and its active compound (carvacrol) against the Callosobruchus maculatus adults (Fab), a major pest of chickpea
- Polymerized methyl imidazole silver bromide (CH3C6H5AgBr)6: Synthesis, crystal structures, and catalytic activity
- Using calcined waste fish bones as a green solid catalyst for biodiesel production from date seed oil
- Influence of the addition of WO3 on TeO2–Na2O glass systems in view of the feature of mechanical, optical, and photon attenuation
- Naringin ameliorates 5-fluorouracil elicited neurotoxicity by curtailing oxidative stress and iNOS/NF-ĸB/caspase-3 pathway
- GC-MS profile of extracts of an endophytic fungus Alternaria and evaluation of its anticancer and antibacterial potentialities
- Green synthesis, chemical characterization, and antioxidant and anti-colorectal cancer effects of vanadium nanoparticles
- Determination of caffeine content in coffee drinks prepared in some coffee shops in the local market in Jeddah City, Saudi Arabia
- A new 3D supramolecular Cu(ii) framework: Crystal structure and photocatalytic characteristics
- Bordeaux mixture accelerates ripening, delays senescence, and promotes metabolite accumulation in jujube fruit
- Important application value of injectable hydrogels loaded with omeprazole Schiff base complex in the treatment of pancreatitis
- Color tunable benzothiadiazole-based small molecules for lightening applications
- Investigation of structural, dielectric, impedance, and mechanical properties of hydroxyapatite-modified barium titanate composites for biomedical applications
- Metal gel particles loaded with epidermal cell growth factor promote skin wound repair mechanism by regulating miRNA
- In vitro exploration of Hypsizygus ulmarius (Bull.) mushroom fruiting bodies: Potential antidiabetic and anti-inflammatory agent
- Alteration in the molecular structure of the adenine base exposed to gamma irradiation: An ESR study
- Comprehensive study of optical, thermal, and gamma-ray shielding properties of Bi2O3–ZnO–PbO–B2O3 glasses
- Lewis acids as co-catalysts in Pd-based catalyzed systems of the octene-1 hydroethoxycarbonylation reaction
- Synthesis, Hirshfeld surface analysis, thermal, and selective α-glucosidase inhibitory studies of Schiff base transition metal complexes
- Protective properties of AgNPs green-synthesized by Abelmoschus esculentus on retinal damage on the virtue of its anti-inflammatory and antioxidant effects in diabetic rat
- Effects of green decorated AgNPs on lignin-modified magnetic nanoparticles mediated by Cydonia on cecal ligation and puncture-induced sepsis
- Treatment of gastric cancer by green mediated silver nanoparticles using Pistacia atlantica bark aqueous extract
- Preparation of newly developed porcelain ceramics containing WO3 nanoparticles for radiation shielding applications
- Utilization of computational methods for the identification of new natural inhibitors of human neutrophil elastase in inflammation therapy
- Some anticancer agents as effective glutathione S-transferase (GST) inhibitors
- Clay-based bricks’ rich illite mineral for gamma-ray shielding applications: An experimental evaluation of the effect of pressure rates on gamma-ray attenuation parameters
- Stability kinetics of orevactaene pigments produced by Epicoccum nigrum in solid-state fermentation
- Treatment of denture stomatitis using iron nanoparticles green-synthesized by Silybum marianum extract
- Characterization and antioxidant potential of white mustard (Brassica hirta) leaf extract and stabilization of sunflower oil
- Characteristics of Langmuir monomolecular monolayers formed by the novel oil blends
- Strategies for optimizing the single GdSrFeO4 phase synthesis
- Oleic acid and linoleic acid nanosomes boost immunity and provoke cell death via the upregulation of beta-defensin-4 at genetic and epigenetic levels
- Unraveling the therapeutic potential of Bombax ceiba roots: A comprehensive study of chemical composition, heavy metal content, antibacterial activity, and in silico analysis
- Green synthesis of AgNPs using plant extract and investigation of its anti-human colorectal cancer application
- The adsorption of naproxen on adsorbents obtained from pepper stalk extract by green synthesis
- Treatment of gastric cancer by silver nanoparticles encapsulated by chitosan polymers mediated by Pistacia atlantica extract under ultrasound condition
- In vitro protective and anti-inflammatory effects of Capparis spinosa and its flavonoids profile
- Wear and corrosion behavior of TiC and WC coatings deposited on high-speed steels by electro-spark deposition
- Therapeutic effects of green-formulated gold nanoparticles by Origanum majorana on spinal cord injury in rats
- Melanin antibacterial activity of two new strains, SN1 and SN2, of Exophiala phaeomuriformis against five human pathogens
- Evaluation of the analgesic and anesthetic properties of silver nanoparticles supported over biodegradable acacia gum-modified magnetic nanoparticles
- Review Articles
- Role and mechanism of fruit waste polyphenols in diabetes management
- A comprehensive review of non-alkaloidal metabolites from the subfamily Amaryllidoideae (Amaryllidaceae)
- Discovery of the chemical constituents, structural characteristics, and pharmacological functions of Chinese caterpillar fungus
- Eco-friendly green approach of nickel oxide nanoparticles for biomedical applications
- Advances in the pharmaceutical research of curcumin for oral administration
- Rapid Communication
- Determination of the contents of bioactive compounds in St. John’s wort (Hypericum perforatum): Comparison of commercial and wild samples
- Retraction
- Retraction of “Two mixed-ligand coordination polymers based on 2,5-thiophenedicarboxylic acid and flexible N-donor ligands: The protective effect on periodontitis via reducing the release of IL-1β and TNF-α”
- Topical Issue on Phytochemicals, biological and toxicological analysis of aromatic medicinal plants
- Anti-plasmodial potential of selected medicinal plants and a compound Atropine isolated from Eucalyptus obliqua
- Anthocyanin extract from black rice attenuates chronic inflammation in DSS-induced colitis mouse model by modulating the gut microbiota
- Evaluation of antibiofilm and cytotoxicity effect of Rumex vesicarius methanol extract
- Chemical compositions of Litsea umbellata and inhibition activities
- Green synthesis, characterization of silver nanoparticles using Rhynchosia capitata leaf extract and their biological activities
- GC-MS analysis and antibacterial activities of some plants belonging to the genus Euphorbia on selected bacterial isolates
- The abrogative effect of propolis on acrylamide-induced toxicity in male albino rats: Histological study
- A phytoconstituent 6-aminoflavone ameliorates lipopolysaccharide-induced oxidative stress mediated synapse and memory dysfunction via p-Akt/NF-kB pathway in albino mice
- Anti-diabetic potentials of Sorbaria tomentosa Lindl. Rehder: Phytochemistry (GC-MS analysis), α-amylase, α-glucosidase inhibitory, in vivo hypoglycemic, and biochemical analysis
- Assessment of cytotoxic and apoptotic activities of the Cassia angustifolia aqueous extract against SW480 colon cancer
- Biochemical analysis, antioxidant, and antibacterial efficacy of the bee propolis extract (Hymenoptera: Apis mellifera) against Staphylococcus aureus-induced infection in BALB/c mice: In vitro and in vivo study
- Assessment of essential elements and heavy metals in Saudi Arabian rice samples underwent various processing methods
- Two new compounds from leaves of Capparis dongvanensis (Sy, B. H. Quang & D. V. Hai) and inhibition activities
- Hydroxyquinoline sulfanilamide ameliorates STZ-induced hyperglycemia-mediated amyleoid beta burden and memory impairment in adult mice
- An automated reading of semi-quantitative hemagglutination results in microplates: Micro-assay for plant lectins
- Inductively coupled plasma mass spectrometry assessment of essential and toxic trace elements in traditional spices consumed by the population of the Middle Eastern region in their recipes
- Phytochemical analysis and anticancer activity of the Pithecellobium dulce seed extract in colorectal cancer cells
- Impact of climatic disturbances on the chemical compositions and metabolites of Salvia officinalis
- Physicochemical characterization, antioxidant and antifungal activities of essential oils of Urginea maritima and Allium sativum
- Phytochemical analysis and antifungal efficiency of Origanum majorana extracts against some phytopathogenic fungi causing tomato damping-off diseases
- Special Issue on 4th IC3PE
- Graphene quantum dots: A comprehensive overview
- Studies on the intercalation of calcium–aluminium layered double hydroxide-MCPA and its controlled release mechanism as a potential green herbicide
- Synergetic effect of adsorption and photocatalysis by zinc ferrite-anchored graphitic carbon nitride nanosheet for the removal of ciprofloxacin under visible light irradiation
- Exploring anticancer activity of the Indonesian guava leaf (Psidium guajava L.) fraction on various human cancer cell lines in an in vitro cell-based approach
- The comparison of gold extraction methods from the rock using thiourea and thiosulfate
- Special Issue on Marine environmental sciences and significance of the multidisciplinary approaches
- Sorption of alkylphenols and estrogens on microplastics in marine conditions
- Cytotoxic ketosteroids from the Red Sea soft coral Dendronephthya sp.
- Antibacterial and biofilm prevention metabolites from Acanthophora spicifera
- Characteristics, source, and health risk assessment of aerosol polyaromatic hydrocarbons in the rural and urban regions of western Saudi Arabia
- Special Issue on Advanced Nanomaterials for Energy, Environmental and Biological Applications - Part II
- Green synthesis, characterization, and evaluation of antibacterial activities of cobalt nanoparticles produced by marine fungal species Periconia prolifica
- Combustion-mediated sol–gel preparation of cobalt-doped ZnO nanohybrids for the degradation of acid red and antibacterial performance
- Perinatal supplementation with selenium nanoparticles modified with ascorbic acid improves hepatotoxicity in rat gestational diabetes
- Evaluation and chemical characterization of bioactive secondary metabolites from endophytic fungi associated with the ethnomedicinal plant Bergenia ciliata
- Enhancing photovoltaic efficiency with SQI-Br and SQI-I sensitizers: A comparative analysis
- Nanostructured p-PbS/p-CuO sulfide/oxide bilayer heterojunction as a promising photoelectrode for hydrogen gas generation

