Abstract
The Arabian Gulf is considered as one of the most important sources for the crude oil all over the world. Due to the vast oil exploration and exploitation, huge amounts of organic pollutants infiltrate to the gulf. An important class of organic pollutants is polycyclic aromatic hydrocarbons (PAHs). One of the marine habitats in Arabian Gulf area is the mangrove stands, that are undoubtedly impacted by all anthropogenic factors like oil industries and sewage discharge. In the monitoring framework for mangrove ecosystem along Saudi coasts, nine mangrove stands were examined for the accumulation of PAHs in the Arabian Gulf coast. PAHs were measured using Gas Chromatography-Mass Spectrometry. The mean values detected for total PAHs in mangrove sediments, roots and leaf were 105.39, 680.0 and 282.4 ng/g, respectively. The trend of total PAHs concentrations in all sites showed the descending order: roots > leaf > sediments. Despite the sandy nature and low organic carbon contents of the mangrove sediments, moderate values of PAHs were detected in the major sites. PAH bio-accumulation factors for roots are higher than that in leaf. The diagnostic ratios revealed that the sources of PAHs are mainly pyrogenic, except for Damam and Damam Port that were found to be petrogenic.
1 Introduction
Polycyclic aromatic hydrocarbons (PAHs) are ubiquitous anthropogenic pollutants [1] characterized by high lipophilicity (accumulate in food chains) [2], high hydrophobicity (accumulate in sediments) [3]. Therefore, sediment could be reservoir and source for these pollutants [4]. PAHs also characterized by high toxicity, high stability and high resistance to microbial degradation [5]. Due to the probable cancer risk and mutagenic properties for PAHs; USEPA was listed 16 compounds of PAHs in the priority pollutants list [6]. Urban coastal areas often show elevated levels of PAHs [7, 8]. In addition to PAHs biogenic origin, there are two main anthropogenic sources for PAHs which are: pyrogenic that generated from incomplete combustion of foil, and petrogenic that derived directly from crude oil [9].
The Arabian Gulf is a major production area for oil and gas, not only in the Middle East but also all over the world; where almost two third of the oil resources of the world is found in this Gulf [10]. In addition to tremendous stress exerted to the marine environment due to the production and transportation of oil in this area; during 1991, the Gulf was impacted by one of the largest oil spills in history (10.8 million barrels of crude oil were deliberately spilled in the Gulf) [11], synchronously with large amount of ash input to the Gulf due to blowouts of about 8 million barrels and fires in Kuwait oil fields [12]. The environmental recovery of such huge accidents may extend for decades. Other moderate and small oil spill accidents surely occurred; as in Deylam (Iran) (west northern of the Gulf) during 2012, where oil leakage covered 160 km2 square area of surface water in the Gulf [13]. That is beside to urban sewage disposal, harbor activates, atmospheric deposition which are considered as source points for all kinds of organic pollutants include PAHs to the marine environment.
Mangroves are intertidal wetland plants usually found in tropical and subtropical coastal areas. The biological importance of mangroves is to provide foods for marine animals as the primary producers do, meanwhile several kinds of organic pollutants like PAHs can accumulate in mangroves due to its unique characters such as high productivity and organic carbon content [14]. The levels of PAHs in contaminated mangrove swamps could exceed 10,000 ng/g [15]. Moreover, the levels of PAHs detected in the sediments of contaminated mangrove swamps were found to be higher than the levels in marine harbors, where it acts as security guard protecting the coastal line from organic pollutants [16]. PAHs could uptake from the sediment to leaf via mangrove roots. The accumulated PAHs in mangrove transfer to higher trophic levels through food web and could accumulate in animals and humane [17].
Despite the dangerous environmental activities in the Arabian Gulf, most of the studies were done to assess the PAHs levels in the Gulf sediments [18,19,20,21] and rarely extend to seawater [13]; with negligence for the marine habitats like mangrove stands that are definitely affected seriously by the accumulation of PAHs. The present work aims to study the concentration levels of the priority 16 PAHs and its methylated derivatives in the surface sediments, roots and leaf of mangrove stands for 9 selected sites in the Arabian Gulf-Saudi coast. The study will extend to evaluate the bio-accumulation of PAHs in mangrove plants. Moreover, the PAHs origin will be investigated based on two diagnostic ratios.
2 Material and Methods
2.1 Study Area
The Arabian Gulf is located between Arabian Peninsula and Iran. The Saudi Arabian coastline of the Arabian Gulf extends for about 450 km. Avicenna marina is the only mangrove species existing in the Gulf. The present study cover mangrove stands in the Saudi coast of Arabia Gulf extended from 26° 42’ 54.616” N to 26° 26’ 14.69”N and from 50° 1’ 18.356” E to 50° 7’ 55.48”E. Some sites are located in remote areas far from direct anthropogenic effects (1, 5), while other sites directly affected by different anthropogenic sources like station 7 near sewage disposal area and sites 9 and 8 that are the Damam port and adjacent area which influenced by harbor activities. Mangrove samples mean heights were around 2.5-3.0 m and its age was estimated to be 25-30 years. The growth conditions of all swamps are comparable, therefore significant changes of PAHs contamination due to this factor is excluded. Figure 1 shows the map locations for the nine mangrove samples along the Arabian Gulf-Saudi coast.
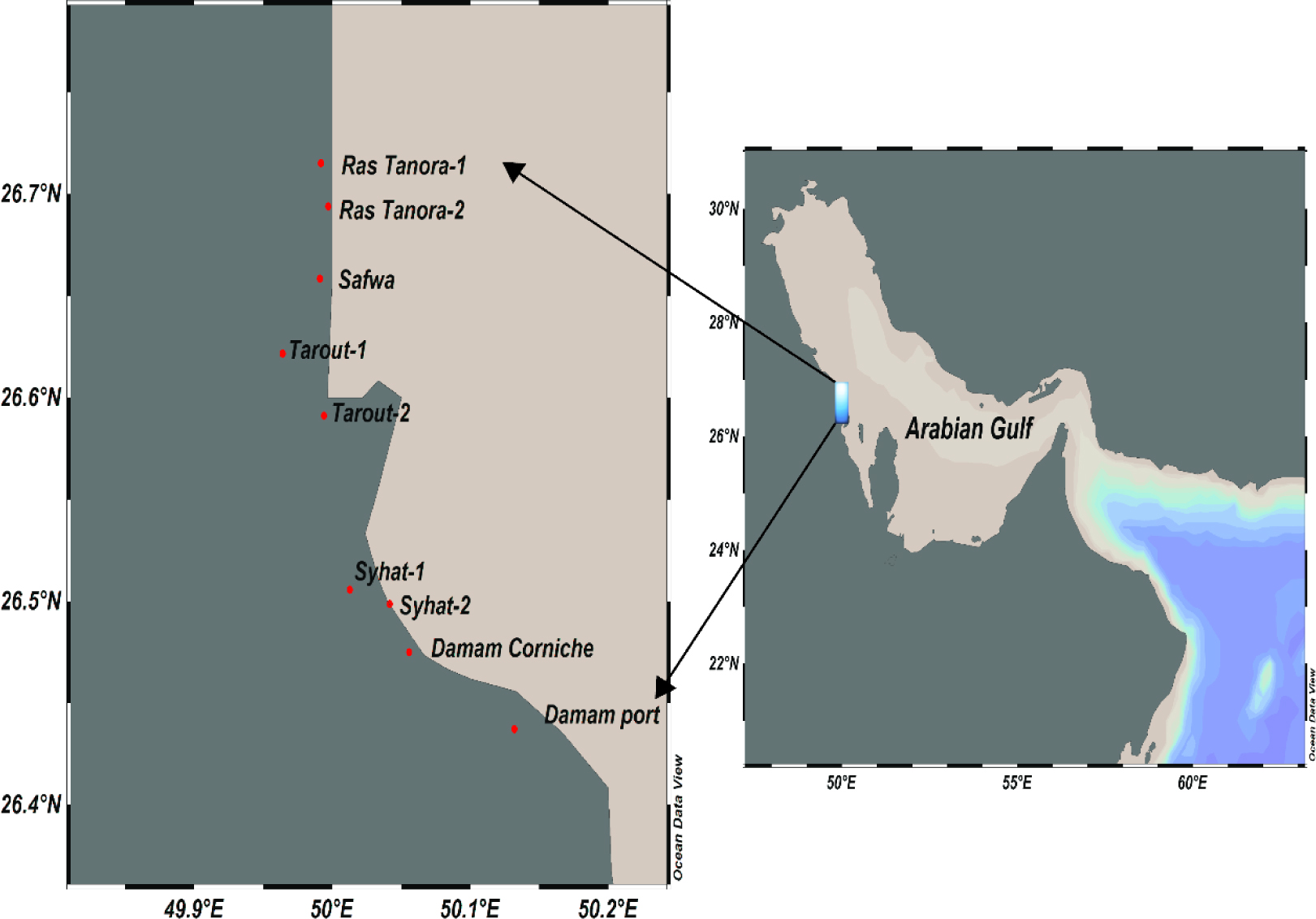
The map of the nine sampling locations of mangrove stands along Arabian Gulf.
2.2 Samples collection and pre-treatment
Approximately the top 0-5 cm of sediments were collected from each studied stand and placed in aluminum bags, refrigerated, and transported to the laboratory within 8 hours after collection and kept frozen in -20°C. Plant samples were washed by tap water followed by deionized water before being freeze-dried. The dried plant samples were grounded into powder using a blender. The sediment samples were freeze-dried for 24 hours, grounded into fine powder using a pestle and mortar, and filtered through 80-mesh screen-sieve. All samples (sediments and plants) were transferred to glass bottles and kept in 4°C refrigerator for further analysis.
2.3 Organic carbon and grain size analysis
Total organic carbon in the sediments was analyzed by wet dichromate-sulfuric acid oxidation method [22]. Standard dry sieving technique was used for sediment grain size fractions, where classified as fine mud (< 0.063 mm), sand (0.063 – 2 mm) and gravel (> 2 mm) [23].
2.4 Extraction and purification of PAHs
Around 20 g of each sediment sample (5 g for leaf; 5 g for root) was subjected to a soxhlet extraction using 300 ml dichloromethane (DCM) for 24 h. Sulfur was removed by the addition of metallic copper. All samples were spiked with 250µL deuterated surrogate standard mixture (naphthalene-d8, phenanthrene-d10, and chrysene-d12) before extraction [24]. The crude extract was concentrated on a rotavapor at 35°C. Particulate impurities for plant samples were filtered. The extracts were concentrated to approximately 1 ml and transferred to silica column for cleanup and fractionation. The column was packed from top and bottom with pre-combusted anhydrous Na2SO4 at 450°C for 6 hours. Silica gel (230-400 mesh) was activated at 230°C for 12 hours then partially deactivated with 5% deionized water. The elution was done using n-hexane for aliphatic fraction followed by n-hexane/ DCM (70:30 v/v) for PAH fraction [25]. PAH fractions were concentrated using gentle stream of pure N2 gas to nearly 1 ml DCM. Deuterated internal standard mixture (acenaphthene-d10, flourene-d10 and perylene-d12) (100 μl, 5 ppm) was added just before injection to GC-MS.
2.5 Identification and Quantification of PAHs
GC-MS (Schimatzu 2010) with DB-5MS column (30 m* 0.25 μm, RTX) was used for PAHs analysis. The initial temperature programmed at 100°C with 1-minute hold, and then ramped at 6°C/minute to 300°C then held for 3 minutes. The electron energy of the mass spectrometer was 70 eV. Individual PAHs were identified based on both retention time and mass spectrum of selected ions with the external calibrated standards for the priority 16 PAH parent targets. Methylated PAH homologs quantified using the response factor of the corresponding non-methylated parent PAH [26].
2.6 Calculation of Bio-accumulation Factors
The bio-accumulation factor (BAF) for roots and leaf was calculated by the formula: BAFroot = Croot/CS and BAFleaf = Cleaf/CS, where Croot and Cleaf are the concentration of total PAHs in mangrove roots and leaf, respectively, and CS = concentration of PAHs in sediment [27].
2.7 Quality control and quality assurance
Duplicate samples (10% of the analyzed samples) and procedural blanks (1 blank for each 5 samples) were performed at the same time with analysis. At the beginning of each working day, calibration standards were run before each sample batch to establish the calibration curves for PAHs. Before extraction, all samples were spiked with surrogate deuterated mixture for recovery calculations. Before GC-MS injection, deuterated internal standard mixture was added for all samples. The recoveries of samples were ranged between 69-102% with RSD% < 19%. The concentrations in the procedural blanks were no more than 3 times the method detection limit. Detection limits (DL) were calculated through five-point calibration curve for standers and extrapolated for determining the y axis intercepts [28]. All results were expressed as dry weight basis, and those samples with concentrations less than DL were reported as not detected (ND).
Ethical approval
The conducted research is not related to either human or animals use.
3 Results and discussion
3.1 Levels of PAHs in mangrove stands
Mangrove swamps protect coastal areas from erosion and storm waves. Meanwhile, mangrove trees act as an ocean safeguard from different pollutants [29]. It grows throughout the world, in the tropics and subtropics areas with low-oxygen soil, where slow-moving waters allow fine sediments to accumulate, and are adapted to life in harsh coastal systems. However, mangrove plants are able to grow in variable compositions of silt, clay and sand sediments contents [30]. Although several biogeochemical processes are influential in mangrove swamps to host organic pollutants [31], sediment particle size is principle factor affecting the levels of all organic pollutants [32]. Table 1 represents proportional ratios for gravel, sand, mud, CaCO3, in addition to total organic carbon (TOC) ratios in the mangrove sediments for the nine studied sites. The textural classification of sediments in the present study were found mainly sandy (sand range: 87.2-93.0%) with small surface area that diminish adsorption of organic pollutants [33]. Although low values of total organic carbon were detected in the mangrove sediments (range: 1.71-4.55%) that were consistent with the sandy nature; gradual increase for TOC % was recognized though northern sites to reach maximum at site 7 (Syhate 2) that affected by sewage drainage.
Sediment characteristics for the studied mangrove stands along Arabian Gulf.
| Site no. | Site name | Gravel% | Sand% | Mud% | CaCO3% | TOC% | Textural Classification |
|---|---|---|---|---|---|---|---|
| 1 | Ras Tanora 1 | 3.8 | 91.1 | 5.2 | 24.7 | 1.71 | Sand |
| 2 | Ras Tanora 2 | 6.2 | 92.7 | 1.1 | 35.2 | 2.86 | Sand |
| 3 | Safwa | 0.4 | 91.9 | 7.7 | 38.4 | 1.76 | Sand |
| 4 | Tarout 1 | 0.6 | 93 | 6.5 | 31.2 | 2.69 | Sand |
| 5 | Tarout 2 | 10.9 | 88.4 | 0.7 | 35.9 | 3.9 | Gravel sand |
| 6 | Syhat 1 | 8.6 | 87.2 | 4.3 | 31.2 | 2.86 | Slightly gravel sand |
| 7 | Syhate 2 | 0.8 | 90.5 | 8.8 | 30.9 | 4.55 | Sand |
| 8 | Damam Corniche | 0.4 | 91.9 | 7.7 | 34.5 | 2.31 | Sand |
| 9 | Damam Port | 8.9 | 88.6 | 2.4 | 22.9 | 2.01 | Slightly gravel sand |
Among the priority 16 inspected PAH congeners; 7 parent PAHs were detected either in sediments or in the mangrove plants which are: naphthalene, acenaphthene, phenanthrene, chrysene, fluoranthene, pyrene and benzo[k]fluorathene; while the rest of congeners remained below detection limit. Figure 2 represents the total PAHs concentrations in the nine studied mangrove stands (sediments, roots and leaf) along the Saudi coast of Arabian Gulf. The total PAHs concentration in sediment samples ranged from 4.39 to 533.40 ng/g with an average value of 105.39 ng/g.
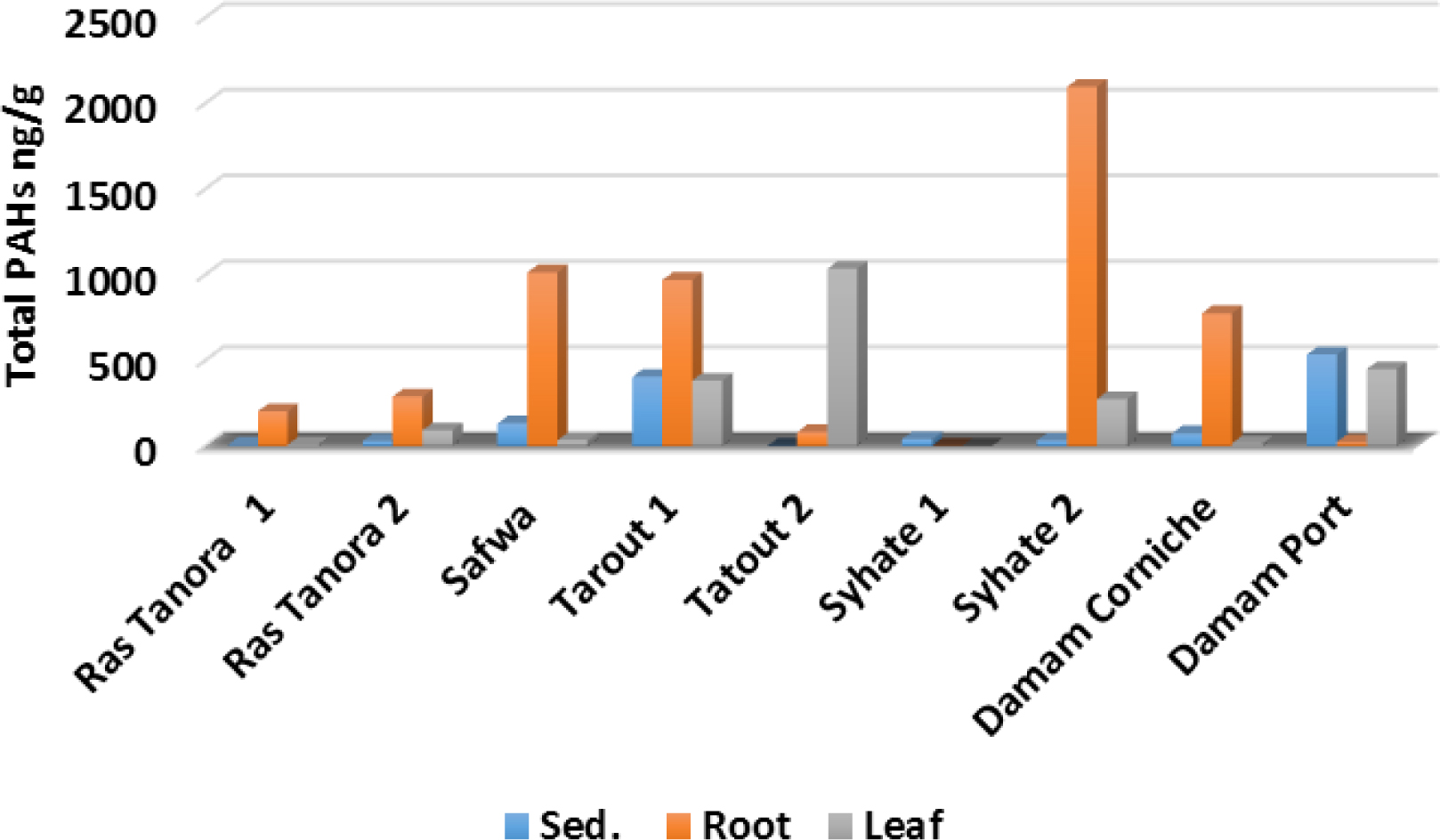
Total PAH concentrations (ng/g) for sediment, roots and leaf in the nine sites of the studied mangrove stands along Arabian Gulf.
These values were found comparable with the moderate values of PAHs detected in the mangrove sediments from the northern Arabian Gulf (Iran) (ranged from 75.24 to 581.94 ng/g) [34], Shantou mangrove wetlands (China) (ranged from 57 to 238 ng/ g) [35] and the Sunderban wetlands (Bay of Bengal, India) (ranged from 20 to 839 ng/g ) [36].While PAH values in the mangrove sediments in the present study seem to be relatively lower than the extremely high PAHs levels detected in highly polluted mangrove sediments for two swamps in Hong Kong (average: 11098 ng/g; 3785 ng/g) [15, 37] and in Shenzhen, China (average: 4480 ng/g) [38]. On the other hand, the detected levels in the present study seem extremely higher than the levels detected in mangrove sediments in the regional area along eastern side of Red Sea coast-Saudi Arabia ( ranged from 1.06 to 6.97 ng/g with an average value of 2.98 ng/g) [39].
Since the coastal areas of the Arabian Gulf are the largest source of crude oil and natural gases all over the world, and apart from mangrove stands; PAHs were studied in the sediments of different areas around Arabian Gulf extensively in the west northern part of the Gulf (Iran); recording very high values that ranged from 1,054 to 17,448 ng/g and 42.76 to 5596.49 ng/g in the industrial areas of Asaluyeh harbor [19] and Rural area [18] (Iran), respectively. While the moderate values recorded in Deylam, Busheher (Iran) (ranged between 15.3 to 759 ng/g) [13] and Qatar exclusive economic zone (ranged from 2.6 to 1025 ng/g, average: 117.3) [21] are comparable with the levels in the present study. It is worth to mention that the sediment core studied within Kuwait Bay for the depositional history of PAHs to the Arabian Gulf indicate low values fluctuated between 12 and 25 ng/g before 1970 [20].
Despite geochemical factors in the study area such as small particle size and low total organic carbon content, exclude hosting high concentrations of PAHs in the sediments; sites 3 and 4 showed moderate levels of total PAHs, in addition to site 9 (Damam port) that recorded the highest total PAH value (533.4 ng/g) which can be attributed to port activities like daily loading and washing of oil tankers, oil spills and leakages, and shipping wastes. Fluorancene appears as the highest detected individual parent congener (132.5 ng/g) in the sediment at site 9 (Damam port), while trimethyl phenanthrene recorded the highest concentration for methylated compound at site 3 (Safwa) (68.48 ng/g). PAH ring composition pattern covers 2, 3, 4 and 5 ring congeners. Average methylated congeners (mono-, di-, and tri-methyl) represented 22.5, 53.3, and 52.7% from total PAHs in the sediments, roots and leaf, respectively. Methylated PAHs ratios are distinctly increased in the mangrove roots and leaf; these compounds sometimes are more toxic than their parent PAHs, and tend to accumulate more than parent congeners [3, 26]. According to sediment quality guidelines (SQG), individual and total PAHs detected in the present study were found below the effect range low (ERL) [40]; hence, adverse biological effects hardly to occur.
Except sites 5, 6 and 9, mangrove roots showed significant increase in the total PAHs levels (Figure 2), where roots values ranged from 22.7 to 2096.36 ng/g with an average of 680 ng/g; while leaf recording range between 7.3 to 1033.2 ng/g with an average of 282.40 ng/g. The trend of total PAHs concentrations for the major sites showed the descending order: roots > leaf > sediments. The observed accumulation trend of PAHs in this study is compatible with the mangrove swamps in Mumbai (India) [16]. In contrary, the highest levels of PAHs were detected in the mangrove leaf followed by roots then sediments in Shenzhen (Chania) [38], and eastern coast of Red Sea [39].
3.2 Bio-accumulation of PAHs in mangrove plant
The substantial mechanism for accumulation of PAHs in mangrove plant take place through sediment/root uptake [41, 42]. Factors like sediment particle size around roots [43], the water solubility of PAHs and n-octanol-water partition coefficient (Kow) control the plant uptake of PAHs [44]. Another possible mechanism is leaf/atmospheric uptake, where PAHs horn to mangrove through the stomata of the large surface areas of mangrove leaf that usually covered with thick waxy layer, which can accumulate the lipophilic PAHs from atmosphere. The mean controlling factor in this mechanism is the volatility of PAH congeners and its partitioning between gaseous and practical forms [43]. It should be noted that both mechanisms are possible, that may lead to unsystematic correlations between PAHs in sediments and mangrove leaf and roots occasionally [45], as in the sites 5, 6 and 9 in the present study that showed different pattern from the major trend (Figures 2 and 3).
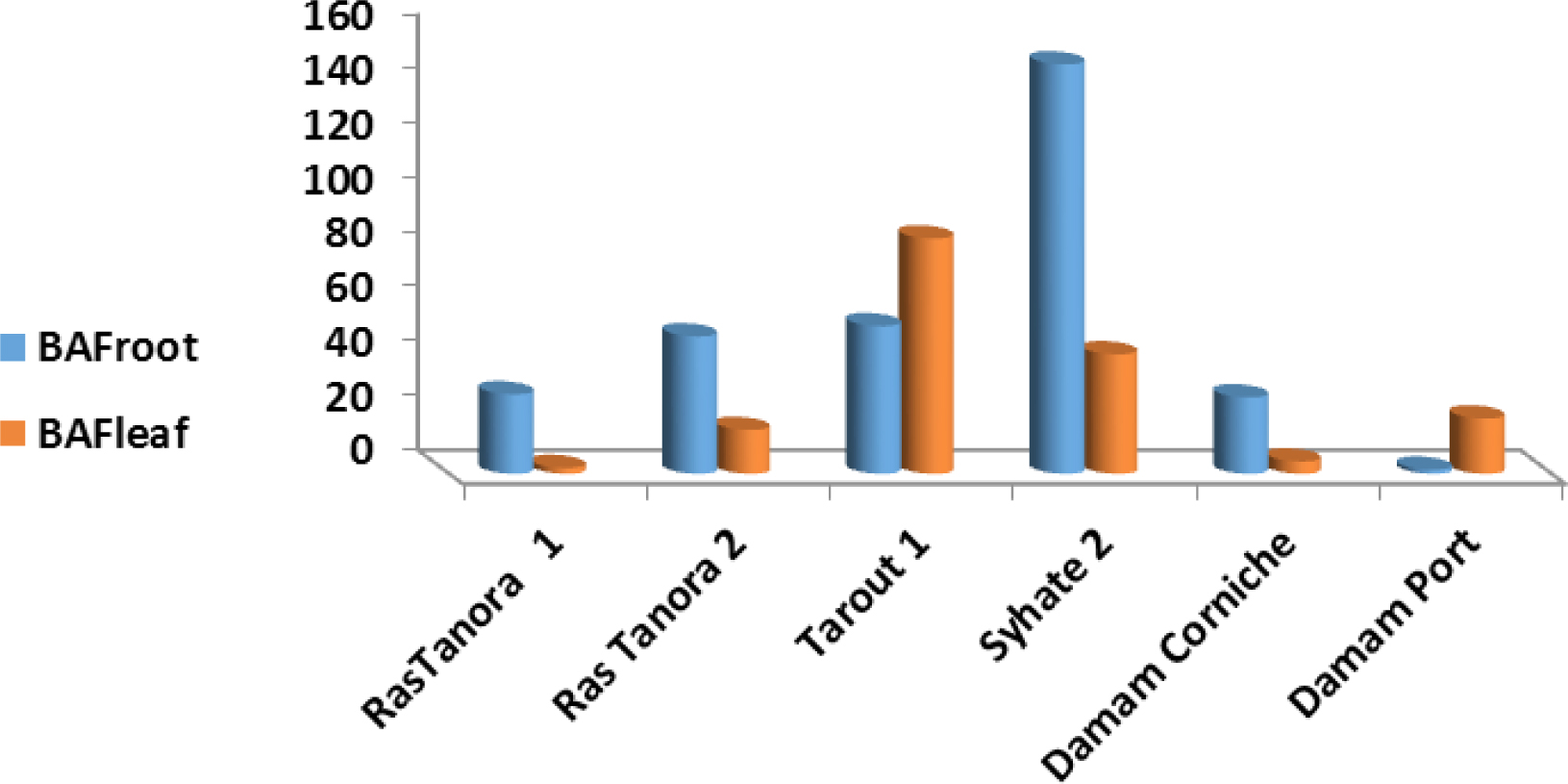
Bio-accumulation factors in roots and leaf for phenanthrene in mangrove plants in the study area.
Bio-accumulation factors for roots (BAFroot) and leaf (BAFleaf) were calculated for phenanthrene congener that was detected in most mangrove sediments, roots and leaf (Figure 3). Values of bio-accumulation factor (BAF)>1 mean that an organism absorbs PAH congener at a rate faster than that at which the substance is lost by catabolism and excretion. The results of the regression analysis for roots / sediments indicated p-values ≤ 0.05 that confirm a significant correlation for phenanthrene, harmonize with Sediment / root mechanism. BAFroot and BAFleaf values elucidate high accumulation in the major sites (Figure 3) and the highest value for BAFroot (150.48) was recorded at site 7 (Syhate 2) which affected by sewage derange; while the highest value for BAFleaf (86.50) was recorded at site 4 (Tarout 1) may support the presence of both mechanisms for phenanthrene accumulation in this site [42, 46]. BAFroot and BAFleaf values recorded in the present study for phenanthrene in mangrove roots and leaf are 60 and 12 times, respectively, higher than the accumulation factors recorded at the eastern coast of Red Sea [39].
It is worth to mention that, except for sites 3, 4 and 5; naphthalene and its methylated derivate were detected in the sediments of the rest sites, while they are absent in the corresponding roots and leaf. This phenomenon can be attributed to the extremely high temperatures that the Gulf area is exposed to all over the year. Despite the relative high hydrophilicity of these light congener, the high temperatures exceed solubility factor and allow these congener to evaporate and escape from sediments before absorption by roots. Hence, the harsh environmental conditions for the mangrove stands may prevent the bio-accumulation of some organic pollutants.
3.3 The probable origin of PAHs in the studied mangrove stands
The anthropogenic activities related to pyrogenic and petrogenic processes are the common sources for PAHs in the marine environment [47]. Discrimination between different origins of PAH mixture is highly difficult due to the complexity governing PAHs distribution in marine environment and co-exist of the compounds from both origins. Numerous studies presented at least 13 diagnostic ratios that were applied to distinguish between PAH origins [9,47,48]. Some diagnostic ratios are based on parent PAHs, others on the proportions of alkyl-substituted to non-substituted molecules. However, in the present study, based on the abandance of different congeners, two diagnostic ratios were selected to discriminate PAH origins (Table 2). Values higher than 2 for methylated phenanthrene/phenanthrene (MP/P) ratio indicate petrogenic origin, while it pyrogenic origin is indicated if the ratio less than one [48]. On the other hand, values higher than 0.22 indicate petrogenic origin if pyrogenic index is applied, while it indicates pyrogenic origin when the ratio less than 0.8 [9]. MP/P values for sites 8 (Damam) and 9 (Damam port) recorded 11.90 and 5.91 respectively, which are the only values which exceeded 2 among all sites, and pointed out to petrogenic origin, while this index ratio for all other sites showed pyrogenic origin. These results are expected, where sites 8 (Damam) and 9 (Damam port) are the closest sites to the oil wells, in addition to the other port activities like discharges of the balanced water of the ships and oil tankers that introduce considerable amount of PAHs originated from oil source. On the other hand, pyrogenic index values in the study area (ranged between 0.23-2.22) are pointed out to pyrogenic sources for all sites, noting that, its value (0.23) at site 8 (Damam) is very close to the border line of petrogenic origin (0.22) which is consistence with MP/P index interpretation. Contribution percentages of pyrogenic and petrogenic origins were estimated at 78.8 and 22.2%, respectively, that are very close to the percentage recorded in the sediments northern of Arabian Gulf [34].
4 Conclusions
The ranges of total PAHs in the studied mangrove stands along Arabian Gulf-Saudi coast showed significant increase from sediment to leaf, and mangrove roots recorded the highest values. This trend support PAH uptake through sediment/root mechanism. Despite the sandy nature and low organic carbon content of sediments in the studied stands; moderate levels of PAHs were recorded either in sediments or mangrove plants that attributed to the presence of mangrove stands close to anthropogenic effects like sewage drainage, oil wells and port activates which increasing PAHs contamination probabilities. However, according to SQG adverse biological effects hardly to occur. Bio-accumulation in most sites showed high values reaching 150.50 for phenanthrene in the mangrove roots as the maximum recorded value for accumulation. PAH origin of the major mangrove stands in Arabia Gulf of Saudi coast was found mainly pyrogenic except two sites (Damam and Damam Port) that showed petrogenic origin.
Conflict of interest: Authors state no conflict of interest.
Acknowledgment
This project was funded by the Deanship of Scientific research (DSR) at King Abdulaziz University, Jeddah, under grant no. G-126-150-38. The authors, therefore, acknowledge with thanks DSR for technical and financial support. Authors would like to thank Prof. Mohsen El-Sherbiny, Department of Marine Biology, Faculty of Marine Sciences, King Abdulaziz University, for sampling and classification of Mangrove plants.
References
[1] CCME (Canadian Council of Ministers of the Environment). Interim Canadian environmental quality criteria for contaminated sites. CCME, Winnipeg. Canadian soil quality guidelines for potentially carcinogenic and other PAHs: Scientific criteria document. CCME, Winnipeg. 2010, P. 216.Suche in Google Scholar
[2] Neff J. M., Polycyclic Aromatic Hydrocarbons in the Aquatic Environment. Sources, Fates and Biological Effects, Applied Science Publishers Ltd., Essex, England,, 1979. p.262.Suche in Google Scholar
[3] Rabodonirina S., Net S., Ouddane B., Merhaby D., Dumoulin D., Popescu T., et al., Distribution of persistent organic pollutants (PAHs, Me-PAHs, PCBs) in dissolved, particulate and sedimentary phases in freshwater systems, Environ. Pollut., 2015, 206, 38-48.10.1016/j.envpol.2015.06.023Suche in Google Scholar
[4] Salomons W., De Rooij N. M., Kerdijk H., Bril J., Sediments as a source for contaminants. Hydrobiologia., 1987, 149, 13-30.10.1007/978-94-009-4053-6_3Suche in Google Scholar
[5] Nikolaou, A., Kostopoulou M., Petsas, A., Vagi M., Lofrano, G., Meric S., Levels and toxicity of polycyclic aromatic hydrocarbons in marine sediments. Anal. Chem., 2009, 28, 653–664.10.1016/j.trac.2009.04.004Suche in Google Scholar
[6] EPA (Environmental protection agency, US). Provisional guidance for quantitative risk assessment of polycyclic aromatic hydrocarbons. USEPA, Report, 1993.Suche in Google Scholar
[7] Boonyatumanond R., Wattayakorn G., Togo A., Takada H., Distribution and origins of polycyclic aromatic hydrocarbons (PAHs) in riverine, estuarine, and marine sediments in Thailand, Mar. Pollut. Bull., 2006, 52(8), 942–956. 10.1016/j.marpolbul.2005.12.015.Suche in Google Scholar
[8] Van Dolah R. F., Riekerk G. H. M., Levisen M. V., Scott G. I., Fulton M. H., Bearden D., et al., An evaluation of polycyclic aromatic hydrocarbon (PAH) runoff from highways into estuarine wetlands of South Carolina. Arch. Environ. Contam. Toxicol. 2005, 49, 362–370.10.1007/s00244-004-0210-1Suche in Google Scholar
[9] Wang Z. D., Fingas M., Shu Y. Y., Sigouin L., Landriault M., Lambert P., et al. Quantitative characterization of PAHs in burn residue and soot samples and differentiation of pyrogenic PAHs from petrogenic PAHs – the 1994 mobile burn study. Environ. Sci. Technol., 1999, 33, 3100–3109.10.1021/es990031ySuche in Google Scholar
[10] BP (British Petroleum), Sustainability review 2011. Available online at: http://www.bp.com/content/dam/bp/pdf/sustainability/groupreports/bp_sustainability_review_2011.Suche in Google Scholar
[11] Al-Ghadban A. N., Abdali F., Massoud M. S., . Sedimentation rate and bioturbation in the Arabian Gulf. Environ. Int., 1998, 24(1–2), 23–31.10.1016/S0160-4120(97)00118-9Suche in Google Scholar
[12] Literathy P., Considerations for the assessment of environmental consequences of the 1991 Gulf War. Mar. Pollut. Bull., 1993, 27, 349–356.10.1016/0025-326X(93)90042-ISuche in Google Scholar
[13] Aagh H., Rahmanpour S., Abedi E., Arebi I., Mahdinia A., Contamination of polycyclic aromatic hydrocarbons in seawater and sediments of West-northern coasts of the Persian Gulf, IJMS, 2016, 45(12), 1688-1695.Suche in Google Scholar
[14] Naidoo G., Naidoo Y., Achar P., Responses of the mangroves Avicennia marina and Bruguiera gymnorrhiza to oil contamination, Flora-Morphology, Distribution, Functional Ecology of Plants, 2010, 205, 357-362.10.1016/j.flora.2009.12.033Suche in Google Scholar
[15] Tam N., Ke L., Wang X., Wong Y., Contamination of polycyclic aromatic hydrocarbons in surface sediments of mangrove swamps, Enviro. Pollut., 2001, 114, 255-263.10.1016/S0269-7491(00)00212-8Suche in Google Scholar
[16] Shete A., Pandit G., Gunale V., Polycyclic Aromatic Hydrocarbons in the Mangrove Species: Avicennia Marina from Mumbai, India, J. Appl. Environ. Biol. Sci., 2016, 6, 6-11.Suche in Google Scholar
[17] Vane C., Harrison I., Kim A., Moss-Hayes V., Vickers B., Hong K., Organic and metal contamination in surface mangrove sediments of South China, Mar. Pollut. Bull., 2009, 58, 134-144.10.1016/j.marpolbul.2008.09.024Suche in Google Scholar PubMed
[18] Valizadeh-Kakhki F., Zakaria M. P., Aris A. Z., Zulkifli S. Z., Mohammadi M., Tajik H., Polycyclic Aromatic Hydrocarbons Identification and Source Discrimination in Rural Soil of the Northern Persian Gulf Coast, Available online at www.tshe. org/EA, Environment Asia, 2014, 7(1), 104-111. 10.14456/ea.2014.14.Suche in Google Scholar
[19] Raeisi A., Arfaeinia H., Seifi M., Shirzad-Siboni M., Keshtkar M., Dobaradaran S., Polycyclic aromatic hydrocarbons (PAHs) in coastal sediments from urban and industrial areas of Asaluyeh Harbor, Iran: distribution, potential source and ecological risk assessment, Water Sci. Technol., 2016, 957-973, 10.2166/wst.2016.265.Suche in Google Scholar PubMed
[20] Gevao B., Boyle E. A., Carrasco G. G., Ghadban A., Zafar J., Bahloul M., Spatial and temporal distributions of polycyclic aromatic hydrocarbons in the Northern Arabian Gulf sediments, Mar. Pollut. Bull., 2016, 112, 218-224.10.1016/j.marpolbul.2016.08.016Suche in Google Scholar PubMed
[21] Soliman Y.S., Al Ansari E.M.S., Wade T.L., Concentration, composition and sources of PAHs in the coastal sediments of the exclusive economic zone (EEZ) of Qatar, Arabian Gulf, Mar. Pollut. Bull., 2014, 85, 542-548.10.1016/j.marpolbul.2014.04.027Suche in Google Scholar PubMed
[22] Aminot A., Chaussepied M., Manuel des analyses chimiques en milieu marin, 1983.Suche in Google Scholar
[23] Syvitski J. P., Principles, methods and application of particle size analysis: Cambridge University Press, 2007.Suche in Google Scholar
[24] Raza M., Zakaria M. P., Hashim N. R., Yim U. H., Kannan N., Ha S. Y., Composition and source identification of polycyclic aromatic hydrocarbons in mangrove sediments of Peninsular Malaysia: indication of anthropogenic input, Environ. Earth Sci., 2013, 70, 2425-2436.10.1007/s12665-013-2279-1Suche in Google Scholar
[25] Wu S., Tao S., Xu F., Dawson R., Lan T., Li B., et al., Polycyclic aromatic hydrocarbons in dust fall in Tianjin, China, Sci. Total Environ., 2005, 345, 115-126.10.1016/j.scitotenv.2004.11.003Suche in Google Scholar PubMed
[26] Douglas G., Emsbo-Mattingly S., Stout S., Uhler A., Mccarthy K., Murphy B., Morrison R., Chemical fingerprinting of hydrocarbons and polychlorinated biphenyls. Intr. Environ. Forensics., 2007, 317-459.10.1016/B978-012369522-2/50010-5Suche in Google Scholar
[27] Watts A. W., Ballestero T. P., Gardner K. H., Uptake of polycyclic aromatic hydrocarbons (PAHs) in salt marsh plants Spartina alterni flora grown in contaminated sediments, Chemosphere, 2006, 62, 1253-1260.10.1016/j.chemosphere.2005.07.006Suche in Google Scholar PubMed
[28] Victoria U., Determination of Polychlorinated Biphenyls (PCBs) in Waste Oils by Gas Chromatography with Electron Capture Detector, EPA Victoria method, 2003.Suche in Google Scholar
[29] Mazda Y., Kobashi D., Okada S., Tidal-Scale Hydrodynamics within Mangrove Swamps, Wetlands Ecol Manage., 2005, 13 (6), 647–655.10.1007/s11273-005-0613-4Suche in Google Scholar
[30] Ranjan R. K., Routh J., Ramanathan A., Bulk organic matter characteristics in the Pichavaram mangrove–estuarine complex, south-eastern India, Appl. Geochem., 2010, 25, 1176-1186.10.1016/j.apgeochem.2010.05.003Suche in Google Scholar
[31] Weissenfels W. D., Klewer H. J., Langhoff J., Adsorption of polycyclic aromatic hydrocarbons (PAHs) by soil particles: influence on biodegradability and bio-toxicity, Appl. Microbiol. Biotechnol., 1992, 36, 689-696.10.1007/BF00183251Suche in Google Scholar PubMed
[32] Zhao H., Li X., Wang X., Tian D., Grain size distribution of road-deposited sediment and its contribution to heavy metal pollution in urban runoff in Beijing, China, J. Hazard. Mater., 2010, 183, 203-210.10.1016/j.jhazmat.2010.07.012Suche in Google Scholar PubMed
[33] Bei L. B., Ping W., Li C., Yong Z., Effects of aging and flooding on sorption of PAHs in mangrove sediment, Fresenius Environ. Bull., 2011, 20, 623-630.Suche in Google Scholar
[34] Mohebbi-Nozar S. L., Zakaria M. P., Mortazavi M. S., Ismail W. R., Jokar K. K., Concentration and source identification of cyclic aromatic hydrocarbons (PAHs) in mangrove sediments from north of pertain gulf, Polycyclic Aromat. Compd., 2016, 5, 36.10.1080/10406638.2015.1037004Suche in Google Scholar
[35] Cao Q., Chen G., Wang H., Qin J., Huang X., Distribution and sources of PAHs in surface sediments of Shantou mangrove wetlands, China, Fresenius Environ. Bull., 2009, 18, 1788-1797.Suche in Google Scholar
[36] Binelli A., Sarkar S. K., Chatterjee M., Riva C., Parolini M., Bhattacharya B., et al., A comparison of sediment quality guidelines for toxicity assessment in the Sunderban wetlands (Bay of Bengal, India), Chemosphere, 2008, 73, 1129-1137.10.1016/j.chemosphere.2008.07.019Suche in Google Scholar PubMed
[37] Tam N. F., Wong T. W., Wong Y., A case study on fuel oil contamination in a mangrove swamp in Hong Kong, Mar. Pollut. Bull., 2005, 51, 1092-1100.10.1016/j.marpolbul.2005.06.005Suche in Google Scholar PubMed
[38] Li F., Zeng X., Yang J., Zhou K., Zan Q., Lei A., et al., Contamination of polycyclic aromatic hydrocarbons (PAHs) in surface sediments and plants of mangrove swamps in Shenzhen, China, Mar. Pollut. Bull., 2014, 85, 590-596.10.1016/j.marpolbul.2014.02.025Suche in Google Scholar PubMed
[39] Bashir M., El-Maradny A., El-Sherbiny M., Rasiq K. T., Orif M., Bio-concentration of Polycyclic Aromatic Hydrocarbons in the grey Mangrove (Avicennia marina) along eastern coast of the Red Sea, Open Chemistry, 2017, 15(1), 344-351.10.1515/chem-2017-0038Suche in Google Scholar
[40] Long E. R., Macdonald D. D., Smith S. L., Calder F. D., Incidence of adverse biological effects within ranges of chemical concentrations in marine and estuarine sediments, Environ Manage., 1995, 19, 81-97.10.1007/BF02472006Suche in Google Scholar
[41] Wang P., Zhang Y., Wu T. H., Novel method for in situ visualization of polycyclic aromatic hydrocarbons in mangrove plants, Toxicol. Environ. Chem., 2010, 92, 1825-1829.10.1080/02772248.2010.482833Suche in Google Scholar
[42] Lohmann R., Dapsis M., Morgan E. J., Dekany V., Luey P. J., Determining air– water exchange, spatial and temporal trends of freely dissolved PAHs in an urban estuary using passive polyethylene samplers, Environ. Sci. Technol., 2011, 45, 2655-2662.10.1021/es1025883Suche in Google Scholar PubMed
[43] Wang P., Du K. Z., Zhu Y. X., Zhang Y., A novel analytical approach for investigation of anthracene adsorption onto mangrove leaves, Talanta, 2008, 76, 1177-1182.10.1016/j.talanta.2008.05.021Suche in Google Scholar PubMed
[44] Chiou C. T., Sheng G., Manes M., A partition-limited model for the plant uptake of organic contaminants from soil and water, Environ. Sci. Technol., 2001, 35, 1437-1444.10.1021/es0017561Suche in Google Scholar PubMed
[45] Ahmed A., Ohlson M., Hoque S., Moula M. G., Chemical composition of leaves of a mangrove tree (Sonneratia apetala Buch.-Ham.) and their correlation with some soil variables, BANGL J BOT, 2010, 39, 61-69.10.3329/bjb.v39i1.5528Suche in Google Scholar
[46] Wang Y., Tao S., Jiao X., Coveney R., Wu S., Xing B., Polycyclic aromatic hydrocarbons in leaf cuticles and inner tissues of six species of trees in urban Beijing, Environ. Pollut., 2008, 151, 158-164.10.1016/j.envpol.2007.02.005Suche in Google Scholar PubMed
[47] Zakaria M. P., Takada H., Tsutsumi S., Ohno K., Yamada J., Kouno E., et al., Distribution of polycyclic aromatic hydrocarbons (PAHs) in rivers and estuaries in Malaysia: a widespread input of petrogenic PAHs, Environ. Sci. Technol., 2002, 36, 1907-1918.10.1021/es011278+Suche in Google Scholar PubMed
[48] Yan W., Chi J., Wang Z., Huang W., Zhang G., Spatial and temporal distribution of polycyclic aromatic hydrocarbons (PAHs) in sediments from Daya Bay, South China. Environ. Pollut., 2009, 157, 1823–1830.10.1016/j.envpol.2009.01.023Suche in Google Scholar PubMed
© 2018 Mohammed Orif, Amr El-Maradny, published by De Gruyter
This work is licensed under the Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 License.
Artikel in diesem Heft
- Regular Articles
- The effect of CuO modification for a TiO2 nanotube confined CeO2 catalyst on the catalytic combustion of butane
- The preparation and antibacterial activity of cellulose/ZnO composite: a review
- Linde Type A and nano magnetite/NaA zeolites: cytotoxicity and doxorubicin loading efficiency
- Performance and thermal decomposition analysis of foaming agent NPL-10 for use in heavy oil recovery by steam injection
- Spectroscopic (FT-IR, FT-Raman, UV, 1H and 13C NMR) insights, electronic profiling and DFT computations on ({(E)-[3-(1H-imidazol-1-yl)-1-phenylpropylidene] amino}oxy)(4-nitrophenyl)methanone, an imidazole-bearing anti-Candida agent
- A Simplistic Preliminary Assessment of Ginstling-Brounstein Model for Solid Spherical Particles in the Context of a Diffusion-Controlled Synthesis
- M-Polynomials And Topological Indices Of Zigzag And Rhombic Benzenoid Systems
- Photochemical Transformation of some 3-benzyloxy-2-(benzo[b]thiophen-2-yl)-4Hchromen-4-ones: A Remote Substituent Effect
- Dynamic Changes of Secondary Metabolites and Antioxidant Activity of Ligustrum lucidum During Fruit Growth
- Studies on the flammability of polypropylene/ammonium polyphosphate and montmorillonite by using the cone calorimeter test
- DSC, FT-IR, NIR, NIR-PCA and NIR-ANOVA for determination of chemical stability of diuretic drugs: impact of excipients
- Antioxidant and Hepatoprotective Effects of Methanolic Extracts of Zilla spinosa and Hammada elegans Against Carbon Tetrachlorideinduced Hepatotoxicity in Rats
- Prunus cerasifera Ehrh. fabricated ZnO nano falcates and its photocatalytic and dose dependent in vitro bio-activity
- Organic biocides hosted in layered double hydroxides: enhancing antimicrobial activity
- Experimental study on the regulation of the cholinergic pathway in renal macrophages by microRNA-132 to alleviate inflammatory response
- Synthesis, characterization, in-vitro antimicrobial properties, molecular docking and DFT studies of 3-{(E)-[(4,6-dimethylpyrimidin-2-yl)imino]methyl} naphthalen-2-ol and Heteroleptic Mn(II), Co(II), Ni(II) and Zn(II) complexes
- M-Polynomials and Topological Indices of Dominating David Derived Networks
- Human Health Risk Assessment of Trace Metals in Surface Water Due to Leachate from the Municipal Dumpsite by Pollution Index: A Case Study from Ndawuse River, Abuja, Nigeria
- Analysis of Bowel Diseases from Blood Serum by Autofluorescence and Atomic Force Microscopy Techniques
- Hydrographic parameters and distribution of dissolved Cu, Ni, Zn and nutrients near Jeddah desalination plant
- Relationships between diatoms and environmental variables in industrial water biotopes of Trzuskawica S.A. (Poland)
- Optimum Conversion of Major Ginsenoside Rb1 to Minor Ginsenoside Rg3(S) by Pulsed Electric Field-Assisted Acid Hydrolysis Treatment
- Antioxidant, Anti-microbial Properties and Chemical Composition of Cumin Essential Oils Extracted by Three Methods
- Regulatory mechanism of ulinastatin on autophagy of macrophages and renal tubular epithelial cells
- Investigation of the sustained-release mechanism of hydroxypropyl methyl cellulose skeleton type Acipimox tablets
- Bio-accumulation of Polycyclic Aromatic Hydrocarbons in the Grey Mangrove (Avicennia marina) along Arabian Gulf, Saudi Coast
- Dynamic Change of Secondary Metabolites and spectrum-effect relationship of Malus halliana Koehne flowers during blooming
- Lipids constituents from Gardenia aqualla Stapf & Hutch
- Effect of using microwaves for catalysts preparation on the catalytic acetalization of glycerol with furfural to obtain fuel additives
- Effect of Humic Acid on the Degradation of Methylene Blue by Peroxymonosulfate
- Serum containing drugs of Gua Lou Xie Bai decoction (GLXB-D) can inhibit TGF-β1-Induced Epithelial to Mesenchymal Transition (EMT) in A549 Cells
- Antiulcer Activity of Different Extracts of Anvillea garcinii and Isolation of Two New Secondary Metabolites
- Analysis of Metabolites in Cabernet Sauvignon and Shiraz Dry Red Wines from Shanxi by 1H NMR Spectroscopy Combined with Pattern Recognition Analysis
- Can water temperature impact litter decomposition under pollution of copper and zinc mixture
- Released from ZrO2/SiO2 coating resveratrol inhibits senescence and oxidative stress of human adipose-derived stem cells (ASC)
- Validated thin-layer chromatographic method for alternative and simultaneous determination of two anti-gout agents in their fixed dose combinations
- Fast removal of pollutants from vehicle emissions during cold-start stage
- Review Article
- Catalytic activities of heterogeneous catalysts obtained by copolymerization of metal-containing 2-(acetoacetoxy)ethyl methacrylate
- Antibiotic Residue in the Aquatic Environment: Status in Africa
- Regular Articles
- Mercury fractionation in gypsum using temperature desorption and mass spectrometric detection
- Phytosynthetic Ag doped ZnO nanoparticles: Semiconducting green remediators
- Epithelial–Mesenchymal Transition Induced by SMAD4 Activation in Invasive Growth Hormone-Secreting Adenomas
- Physicochemical properties of stabilized sewage sludge admixtures by modified steel slag
- In Vitro Cytotoxic and Antiproliferative Activity of Cydonia oblonga flower petals, leaf and fruit pellet ethanolic extracts. Docking simulation of the active flavonoids on anti-apoptotic protein Bcl-2
- Synthesis and Characterization of Pd exchanged MMT Clay for Mizoroki-Heck Reaction
- A new selective, and sensitive method for the determination of lixivaptan, a vasopressin 2 (V2)-receptor antagonist, in mouse plasma and its application in a pharmacokinetic study
- Anti-EGFL7 antibodies inhibit rat prolactinoma MMQ cells proliferation and PRL secretion
- Density functional theory calculations, vibration spectral analysis and molecular docking of the antimicrobial agent 6-(1,3-benzodioxol-5-ylmethyl)-5-ethyl-2-{[2-(morpholin-4-yl)ethyl] sulfanyl}pyrimidin-4(3H)-one
- Effect of Nano Zeolite on the Transformation of Cadmium Speciation and Its Uptake by Tobacco in Cadmium-contaminated Soil
- Effects and Mechanisms of Jinniu Capsule on Methamphetamine-Induced Conditioned Place Preference in Rats
- Calculating the Degree-based Topological Indices of Dendrimers
- Efficient optimization and mineralization of UV absorbers: A comparative investigation with Fenton and UV/H2O2
- Metabolites of Tryptophane and Phenylalanine as Markers of Small Bowel Ischemia-Reperfusion Injury
- Adsorption and determination of polycyclic aromatic hydrocarbons in water through the aggregation of graphene oxide
- The role of NR2C2 in the prolactinomas
- Chromium removal from industrial wastewater using Phyllostachys pubescens biomass loaded Cu-S nanospheres
- Hydrotalcite Anchored Ruthenium Catalyst for CO2 Hydrogenation Reaction
- Preparation of Calcium Fluoride using Phosphogypsum by Orthogonal Experiment
- The mechanism of antibacterial activity of corylifolinin against three clinical bacteria from Psoralen corylifolia L
- 2-formyl-3,6-bis(hydroxymethyl)phenyl benzoate in Electrochemical Dry Cell
- Electro-photocatalytic degradation of amoxicillin using calcium titanate
- Effect of Malus halliana Koehne Polysaccharides on Functional Constipation
- Structural Properties and Nonlinear Optical Responses of Halogenated Compounds: A DFT Investigation on Molecular Modelling
- DMFDMA catalyzed synthesis of 2-((Dimethylamino)methylene)-3,4-dihydro-9-arylacridin-1(2H)-ones and their derivatives: in-vitro antifungal, antibacterial and antioxidant evaluations
- Production of Methanol as a Fuel Energy from CO2 Present in Polluted Seawater - A Photocatalytic Outlook
- Study of different extraction methods on finger print and fatty acid of raw beef fat using fourier transform infrared and gas chromatography-mass spectrometry
- Determination of trace fluoroquinolones in water solutions and in medicinal preparations by conventional and synchronous fluorescence spectrometry
- Extraction and determination of flavonoids in Carthamus tinctorius
- Therapeutic Application of Zinc and Vanadium Complexes against Diabetes Mellitus a Coronary Disease: A review
- Study of calcined eggshell as potential catalyst for biodiesel formation using used cooking oil
- Manganese oxalates - structure-based Insights
- Topological Indices of H-Naphtalenic Nanosheet
- Long-Term Dissolution of Glass Fibers in Water Described by Dissolving Cylinder Zero-Order Kinetic Model: Mass Loss and Radius Reduction
- Topological study of the para-line graphs of certain pentacene via topological indices
- A brief insight into the prediction of water vapor transmissibility in highly impermeable hybrid nanocomposites based on bromobutyl/epichlorohydrin rubber blends
- Comparative sulfite assay by voltammetry using Pt electrodes, photometry and titrimetry: Application to cider, vinegar and sugar analysis
- MicroRNA delivery mediated by PEGylated polyethylenimine for prostate cancer therapy
- Reversible Fluorescent Turn-on Sensors for Fe3+ based on a Receptor Composed of Tri-oxygen Atoms of Amide Groups in Water
- Sonocatalytic degradation of methyl orange in aqueous solution using Fe-doped TiO2 nanoparticles under mechanical agitation
- Hydrotalcite Anchored Ruthenium Catalyst for CO2 Hydrogenation Reaction
- Production and Analysis of Recycled Ammonium Perrhenate from CMSX-4 superalloys
- Topical Issue on Agriculture
- New phosphorus biofertilizers from renewable raw materials in the aspect of cadmium and lead contents in soil and plants
- Survey of content of cadmium, calcium, chromium, copper, iron, lead, magnesium, manganese, mercury, sodium and zinc in chamomile and green tea leaves by electrothermal or flame atomizer atomic absorption spectrometry
- Biogas digestate – benefits and risks for soil fertility and crop quality – an evaluation of grain maize response
- A numerical analysis of heat transfer in a cross-current heat exchanger with controlled and newly designed air flows
- Freshwater green macroalgae as a biosorbent of Cr(III) ions
- The main influencing factors of soil mechanical characteristics of the gravity erosion environment in the dry-hot valley of Jinsha river
- Free amino acids in Viola tricolor in relation to different habitat conditions
- The influence of filler amount on selected properties of new experimental resin dental composite
- Effect of poultry wastewater irrigation on nitrogen, phosphorus and carbon contents in farmland soil
- Response of spring wheat to NPK and S fertilization. The content and uptake of macronutrients and the value of ionic ratios
- The Effect of Macroalgal Extracts and Near Infrared Radiation on Germination of Soybean Seedlings: Preliminary Research Results
- Content of Zn, Cd and Pb in purple moor-grass in soils heavily contaminated with heavy metals around a zinc and lead ore tailing landfill
- Topical Issue on Research for Natural Bioactive Products
- Synthesis of (±)-3,4-dimethoxybenzyl-4-methyloctanoate as a novel internal standard for capsinoid determination by HPLC-ESI-MS/MS(QTOF)
- Repellent activity of monoterpenoid esters with neurotransmitter amino acids against yellow fever mosquito, Aedes aegypti
- Effect of Flammulina velutipes (golden needle mushroom, eno-kitake) polysaccharides on constipation
- Bioassay-directed fractionation of a blood coagulation factor Xa inhibitor, betulinic acid from Lycopus lucidus
- Antifungal and repellent activities of the essential oils from three aromatic herbs from western Himalaya
- Chemical composition and microbiological evaluation of essential oil from Hyssopus officinalis L. with white and pink flowers
- Bioassay-guided isolation and identification of Aedes aegypti larvicidal and biting deterrent compounds from Veratrum lobelianum
- α-Terpineol, a natural monoterpene: A review of its biological properties
- Utility of essential oils for development of host-based lures for Xyleborus glabratus (Coleoptera: Curculionidae: Scolytinae), vector of laurel wilt
- Phenolic composition and antioxidant potential of different organs of Kazakh Crataegus almaatensis Pojark: A comparison with the European Crataegus oxyacantha L. flowers
- Isolation of eudesmane type sesquiterpene ketone from Prangos heyniae H.Duman & M.F.Watson essential oil and mosquitocidal activity of the essential oils
- Comparative analysis of the polyphenols profiles and the antioxidant and cytotoxicity properties of various blue honeysuckle varieties
- Special Issue on ICCESEN 2017
- Modelling world energy security data from multinomial distribution by generalized linear model under different cumulative link functions
- Pine Cone and Boron Compounds Effect as Reinforcement on Mechanical and Flammability Properties of Polyester Composites
- Artificial Neural Network Modelling for Prediction of SNR Effected by Probe Properties on Ultrasonic Inspection of Austenitic Stainless Steel Weldments
- Calculation and 3D analyses of ERR in the band crack front contained in a rectangular plate made of multilayered material
- Improvement of fuel properties of biodiesel with bioadditive ethyl levulinate
- Properties of AlSi9Cu3 metal matrix micro and nano composites produced via stir casting
- Investigation of Antibacterial Properties of Ag Doped TiO2 Nanofibers Prepared by Electrospinning Process
- Modeling of Total Phenolic contents in Various Tea samples by Experimental Design Methods
- Nickel doping effect on the structural and optical properties of indium sulfide thin films by SILAR
- The effect mechanism of Ginnalin A as a homeopathic agent on various cancer cell lines
- Excitation functions of proton induced reactions of some radioisotopes used in medicine
- Oxide ionic conductivity and microstructures of Pr and Sm co-doped CeO2-based systems
- Rapid Synthesis of Metallic Reinforced in Situ Intermetallic Composites in Ti-Al-Nb System via Resistive Sintering
- Oxidation Behavior of NiCr/YSZ Thermal Barrier Coatings (TBCs)
- Clustering Analysis of Normal Strength Concretes Produced with Different Aggregate Types
- Magnetic Nano-Sized Solid Acid Catalyst Bearing Sulfonic Acid Groups for Biodiesel Synthesis
- The biological activities of Arabis alpina L. subsp. brevifolia (DC.) Cullen against food pathogens
- Humidity properties of Schiff base polymers
- Free Vibration Analysis of Fiber Metal Laminated Straight Beam
- Comparative study of in vitro antioxidant, acetylcholinesterase and butyrylcholinesterase activity of alfalfa (Medicago sativa L.) collected during different growth stages
- Isothermal Oxidation Behavior of Gadolinium Zirconate (Gd2Zr2O7) Thermal Barrier Coatings (TBCs) produced by Electron Beam Physical Vapor Deposition (EB-PVD) technique
- Optimization of Adsorption Parameters for Ultra-Fine Calcite Using a Box-Behnken Experimental Design
- The Microstructural Investigation of Vermiculite-Infiltrated Electron Beam Physical Vapor Deposition Thermal Barrier Coatings
- Modelling Porosity Permeability of Ceramic Tiles using Fuzzy Taguchi Method
- Experimental and theoretical study of a novel naphthoquinone Schiff base
- Physicochemical properties of heat treated sille stone for ceramic industry
- Sand Dune Characterization for Preparing Metallurgical Grade Silicon
- Catalytic Applications of Large Pore Sulfonic Acid-Functionalized SBA-15 Mesoporous Silica for Esterification
- One-photon Absorption Characterizations, Dipole Polarizabilities and Second Hyperpolarizabilities of Chlorophyll a and Crocin
- The Optical and Crystallite Characterization of Bilayer TiO2 Films Coated on Different ITO layers
- Topical Issue on Bond Activation
- Metal-mediated reactions towards the synthesis of a novel deaminolysed bisurea, dicarbamolyamine
- The structure of ortho-(trifluoromethyl)phenol in comparison to its homologues – A combined experimental and theoretical study
- Heterogeneous catalysis with encapsulated haem and other synthetic porphyrins: Harnessing the power of porphyrins for oxidation reactions
- Recent Advances on Mechanistic Studies on C–H Activation Catalyzed by Base Metals
- Reactions of the organoplatinum complex [Pt(cod) (neoSi)Cl] (neoSi = trimethylsilylmethyl) with the non-coordinating anions SbF6– and BPh4–
- Erratum
- Investigation on Two Compounds of O, O’-dithiophosphate Derivatives as Corrosion Inhibitors for Q235 Steel in Hydrochloric Acid Solution
Artikel in diesem Heft
- Regular Articles
- The effect of CuO modification for a TiO2 nanotube confined CeO2 catalyst on the catalytic combustion of butane
- The preparation and antibacterial activity of cellulose/ZnO composite: a review
- Linde Type A and nano magnetite/NaA zeolites: cytotoxicity and doxorubicin loading efficiency
- Performance and thermal decomposition analysis of foaming agent NPL-10 for use in heavy oil recovery by steam injection
- Spectroscopic (FT-IR, FT-Raman, UV, 1H and 13C NMR) insights, electronic profiling and DFT computations on ({(E)-[3-(1H-imidazol-1-yl)-1-phenylpropylidene] amino}oxy)(4-nitrophenyl)methanone, an imidazole-bearing anti-Candida agent
- A Simplistic Preliminary Assessment of Ginstling-Brounstein Model for Solid Spherical Particles in the Context of a Diffusion-Controlled Synthesis
- M-Polynomials And Topological Indices Of Zigzag And Rhombic Benzenoid Systems
- Photochemical Transformation of some 3-benzyloxy-2-(benzo[b]thiophen-2-yl)-4Hchromen-4-ones: A Remote Substituent Effect
- Dynamic Changes of Secondary Metabolites and Antioxidant Activity of Ligustrum lucidum During Fruit Growth
- Studies on the flammability of polypropylene/ammonium polyphosphate and montmorillonite by using the cone calorimeter test
- DSC, FT-IR, NIR, NIR-PCA and NIR-ANOVA for determination of chemical stability of diuretic drugs: impact of excipients
- Antioxidant and Hepatoprotective Effects of Methanolic Extracts of Zilla spinosa and Hammada elegans Against Carbon Tetrachlorideinduced Hepatotoxicity in Rats
- Prunus cerasifera Ehrh. fabricated ZnO nano falcates and its photocatalytic and dose dependent in vitro bio-activity
- Organic biocides hosted in layered double hydroxides: enhancing antimicrobial activity
- Experimental study on the regulation of the cholinergic pathway in renal macrophages by microRNA-132 to alleviate inflammatory response
- Synthesis, characterization, in-vitro antimicrobial properties, molecular docking and DFT studies of 3-{(E)-[(4,6-dimethylpyrimidin-2-yl)imino]methyl} naphthalen-2-ol and Heteroleptic Mn(II), Co(II), Ni(II) and Zn(II) complexes
- M-Polynomials and Topological Indices of Dominating David Derived Networks
- Human Health Risk Assessment of Trace Metals in Surface Water Due to Leachate from the Municipal Dumpsite by Pollution Index: A Case Study from Ndawuse River, Abuja, Nigeria
- Analysis of Bowel Diseases from Blood Serum by Autofluorescence and Atomic Force Microscopy Techniques
- Hydrographic parameters and distribution of dissolved Cu, Ni, Zn and nutrients near Jeddah desalination plant
- Relationships between diatoms and environmental variables in industrial water biotopes of Trzuskawica S.A. (Poland)
- Optimum Conversion of Major Ginsenoside Rb1 to Minor Ginsenoside Rg3(S) by Pulsed Electric Field-Assisted Acid Hydrolysis Treatment
- Antioxidant, Anti-microbial Properties and Chemical Composition of Cumin Essential Oils Extracted by Three Methods
- Regulatory mechanism of ulinastatin on autophagy of macrophages and renal tubular epithelial cells
- Investigation of the sustained-release mechanism of hydroxypropyl methyl cellulose skeleton type Acipimox tablets
- Bio-accumulation of Polycyclic Aromatic Hydrocarbons in the Grey Mangrove (Avicennia marina) along Arabian Gulf, Saudi Coast
- Dynamic Change of Secondary Metabolites and spectrum-effect relationship of Malus halliana Koehne flowers during blooming
- Lipids constituents from Gardenia aqualla Stapf & Hutch
- Effect of using microwaves for catalysts preparation on the catalytic acetalization of glycerol with furfural to obtain fuel additives
- Effect of Humic Acid on the Degradation of Methylene Blue by Peroxymonosulfate
- Serum containing drugs of Gua Lou Xie Bai decoction (GLXB-D) can inhibit TGF-β1-Induced Epithelial to Mesenchymal Transition (EMT) in A549 Cells
- Antiulcer Activity of Different Extracts of Anvillea garcinii and Isolation of Two New Secondary Metabolites
- Analysis of Metabolites in Cabernet Sauvignon and Shiraz Dry Red Wines from Shanxi by 1H NMR Spectroscopy Combined with Pattern Recognition Analysis
- Can water temperature impact litter decomposition under pollution of copper and zinc mixture
- Released from ZrO2/SiO2 coating resveratrol inhibits senescence and oxidative stress of human adipose-derived stem cells (ASC)
- Validated thin-layer chromatographic method for alternative and simultaneous determination of two anti-gout agents in their fixed dose combinations
- Fast removal of pollutants from vehicle emissions during cold-start stage
- Review Article
- Catalytic activities of heterogeneous catalysts obtained by copolymerization of metal-containing 2-(acetoacetoxy)ethyl methacrylate
- Antibiotic Residue in the Aquatic Environment: Status in Africa
- Regular Articles
- Mercury fractionation in gypsum using temperature desorption and mass spectrometric detection
- Phytosynthetic Ag doped ZnO nanoparticles: Semiconducting green remediators
- Epithelial–Mesenchymal Transition Induced by SMAD4 Activation in Invasive Growth Hormone-Secreting Adenomas
- Physicochemical properties of stabilized sewage sludge admixtures by modified steel slag
- In Vitro Cytotoxic and Antiproliferative Activity of Cydonia oblonga flower petals, leaf and fruit pellet ethanolic extracts. Docking simulation of the active flavonoids on anti-apoptotic protein Bcl-2
- Synthesis and Characterization of Pd exchanged MMT Clay for Mizoroki-Heck Reaction
- A new selective, and sensitive method for the determination of lixivaptan, a vasopressin 2 (V2)-receptor antagonist, in mouse plasma and its application in a pharmacokinetic study
- Anti-EGFL7 antibodies inhibit rat prolactinoma MMQ cells proliferation and PRL secretion
- Density functional theory calculations, vibration spectral analysis and molecular docking of the antimicrobial agent 6-(1,3-benzodioxol-5-ylmethyl)-5-ethyl-2-{[2-(morpholin-4-yl)ethyl] sulfanyl}pyrimidin-4(3H)-one
- Effect of Nano Zeolite on the Transformation of Cadmium Speciation and Its Uptake by Tobacco in Cadmium-contaminated Soil
- Effects and Mechanisms of Jinniu Capsule on Methamphetamine-Induced Conditioned Place Preference in Rats
- Calculating the Degree-based Topological Indices of Dendrimers
- Efficient optimization and mineralization of UV absorbers: A comparative investigation with Fenton and UV/H2O2
- Metabolites of Tryptophane and Phenylalanine as Markers of Small Bowel Ischemia-Reperfusion Injury
- Adsorption and determination of polycyclic aromatic hydrocarbons in water through the aggregation of graphene oxide
- The role of NR2C2 in the prolactinomas
- Chromium removal from industrial wastewater using Phyllostachys pubescens biomass loaded Cu-S nanospheres
- Hydrotalcite Anchored Ruthenium Catalyst for CO2 Hydrogenation Reaction
- Preparation of Calcium Fluoride using Phosphogypsum by Orthogonal Experiment
- The mechanism of antibacterial activity of corylifolinin against three clinical bacteria from Psoralen corylifolia L
- 2-formyl-3,6-bis(hydroxymethyl)phenyl benzoate in Electrochemical Dry Cell
- Electro-photocatalytic degradation of amoxicillin using calcium titanate
- Effect of Malus halliana Koehne Polysaccharides on Functional Constipation
- Structural Properties and Nonlinear Optical Responses of Halogenated Compounds: A DFT Investigation on Molecular Modelling
- DMFDMA catalyzed synthesis of 2-((Dimethylamino)methylene)-3,4-dihydro-9-arylacridin-1(2H)-ones and their derivatives: in-vitro antifungal, antibacterial and antioxidant evaluations
- Production of Methanol as a Fuel Energy from CO2 Present in Polluted Seawater - A Photocatalytic Outlook
- Study of different extraction methods on finger print and fatty acid of raw beef fat using fourier transform infrared and gas chromatography-mass spectrometry
- Determination of trace fluoroquinolones in water solutions and in medicinal preparations by conventional and synchronous fluorescence spectrometry
- Extraction and determination of flavonoids in Carthamus tinctorius
- Therapeutic Application of Zinc and Vanadium Complexes against Diabetes Mellitus a Coronary Disease: A review
- Study of calcined eggshell as potential catalyst for biodiesel formation using used cooking oil
- Manganese oxalates - structure-based Insights
- Topological Indices of H-Naphtalenic Nanosheet
- Long-Term Dissolution of Glass Fibers in Water Described by Dissolving Cylinder Zero-Order Kinetic Model: Mass Loss and Radius Reduction
- Topological study of the para-line graphs of certain pentacene via topological indices
- A brief insight into the prediction of water vapor transmissibility in highly impermeable hybrid nanocomposites based on bromobutyl/epichlorohydrin rubber blends
- Comparative sulfite assay by voltammetry using Pt electrodes, photometry and titrimetry: Application to cider, vinegar and sugar analysis
- MicroRNA delivery mediated by PEGylated polyethylenimine for prostate cancer therapy
- Reversible Fluorescent Turn-on Sensors for Fe3+ based on a Receptor Composed of Tri-oxygen Atoms of Amide Groups in Water
- Sonocatalytic degradation of methyl orange in aqueous solution using Fe-doped TiO2 nanoparticles under mechanical agitation
- Hydrotalcite Anchored Ruthenium Catalyst for CO2 Hydrogenation Reaction
- Production and Analysis of Recycled Ammonium Perrhenate from CMSX-4 superalloys
- Topical Issue on Agriculture
- New phosphorus biofertilizers from renewable raw materials in the aspect of cadmium and lead contents in soil and plants
- Survey of content of cadmium, calcium, chromium, copper, iron, lead, magnesium, manganese, mercury, sodium and zinc in chamomile and green tea leaves by electrothermal or flame atomizer atomic absorption spectrometry
- Biogas digestate – benefits and risks for soil fertility and crop quality – an evaluation of grain maize response
- A numerical analysis of heat transfer in a cross-current heat exchanger with controlled and newly designed air flows
- Freshwater green macroalgae as a biosorbent of Cr(III) ions
- The main influencing factors of soil mechanical characteristics of the gravity erosion environment in the dry-hot valley of Jinsha river
- Free amino acids in Viola tricolor in relation to different habitat conditions
- The influence of filler amount on selected properties of new experimental resin dental composite
- Effect of poultry wastewater irrigation on nitrogen, phosphorus and carbon contents in farmland soil
- Response of spring wheat to NPK and S fertilization. The content and uptake of macronutrients and the value of ionic ratios
- The Effect of Macroalgal Extracts and Near Infrared Radiation on Germination of Soybean Seedlings: Preliminary Research Results
- Content of Zn, Cd and Pb in purple moor-grass in soils heavily contaminated with heavy metals around a zinc and lead ore tailing landfill
- Topical Issue on Research for Natural Bioactive Products
- Synthesis of (±)-3,4-dimethoxybenzyl-4-methyloctanoate as a novel internal standard for capsinoid determination by HPLC-ESI-MS/MS(QTOF)
- Repellent activity of monoterpenoid esters with neurotransmitter amino acids against yellow fever mosquito, Aedes aegypti
- Effect of Flammulina velutipes (golden needle mushroom, eno-kitake) polysaccharides on constipation
- Bioassay-directed fractionation of a blood coagulation factor Xa inhibitor, betulinic acid from Lycopus lucidus
- Antifungal and repellent activities of the essential oils from three aromatic herbs from western Himalaya
- Chemical composition and microbiological evaluation of essential oil from Hyssopus officinalis L. with white and pink flowers
- Bioassay-guided isolation and identification of Aedes aegypti larvicidal and biting deterrent compounds from Veratrum lobelianum
- α-Terpineol, a natural monoterpene: A review of its biological properties
- Utility of essential oils for development of host-based lures for Xyleborus glabratus (Coleoptera: Curculionidae: Scolytinae), vector of laurel wilt
- Phenolic composition and antioxidant potential of different organs of Kazakh Crataegus almaatensis Pojark: A comparison with the European Crataegus oxyacantha L. flowers
- Isolation of eudesmane type sesquiterpene ketone from Prangos heyniae H.Duman & M.F.Watson essential oil and mosquitocidal activity of the essential oils
- Comparative analysis of the polyphenols profiles and the antioxidant and cytotoxicity properties of various blue honeysuckle varieties
- Special Issue on ICCESEN 2017
- Modelling world energy security data from multinomial distribution by generalized linear model under different cumulative link functions
- Pine Cone and Boron Compounds Effect as Reinforcement on Mechanical and Flammability Properties of Polyester Composites
- Artificial Neural Network Modelling for Prediction of SNR Effected by Probe Properties on Ultrasonic Inspection of Austenitic Stainless Steel Weldments
- Calculation and 3D analyses of ERR in the band crack front contained in a rectangular plate made of multilayered material
- Improvement of fuel properties of biodiesel with bioadditive ethyl levulinate
- Properties of AlSi9Cu3 metal matrix micro and nano composites produced via stir casting
- Investigation of Antibacterial Properties of Ag Doped TiO2 Nanofibers Prepared by Electrospinning Process
- Modeling of Total Phenolic contents in Various Tea samples by Experimental Design Methods
- Nickel doping effect on the structural and optical properties of indium sulfide thin films by SILAR
- The effect mechanism of Ginnalin A as a homeopathic agent on various cancer cell lines
- Excitation functions of proton induced reactions of some radioisotopes used in medicine
- Oxide ionic conductivity and microstructures of Pr and Sm co-doped CeO2-based systems
- Rapid Synthesis of Metallic Reinforced in Situ Intermetallic Composites in Ti-Al-Nb System via Resistive Sintering
- Oxidation Behavior of NiCr/YSZ Thermal Barrier Coatings (TBCs)
- Clustering Analysis of Normal Strength Concretes Produced with Different Aggregate Types
- Magnetic Nano-Sized Solid Acid Catalyst Bearing Sulfonic Acid Groups for Biodiesel Synthesis
- The biological activities of Arabis alpina L. subsp. brevifolia (DC.) Cullen against food pathogens
- Humidity properties of Schiff base polymers
- Free Vibration Analysis of Fiber Metal Laminated Straight Beam
- Comparative study of in vitro antioxidant, acetylcholinesterase and butyrylcholinesterase activity of alfalfa (Medicago sativa L.) collected during different growth stages
- Isothermal Oxidation Behavior of Gadolinium Zirconate (Gd2Zr2O7) Thermal Barrier Coatings (TBCs) produced by Electron Beam Physical Vapor Deposition (EB-PVD) technique
- Optimization of Adsorption Parameters for Ultra-Fine Calcite Using a Box-Behnken Experimental Design
- The Microstructural Investigation of Vermiculite-Infiltrated Electron Beam Physical Vapor Deposition Thermal Barrier Coatings
- Modelling Porosity Permeability of Ceramic Tiles using Fuzzy Taguchi Method
- Experimental and theoretical study of a novel naphthoquinone Schiff base
- Physicochemical properties of heat treated sille stone for ceramic industry
- Sand Dune Characterization for Preparing Metallurgical Grade Silicon
- Catalytic Applications of Large Pore Sulfonic Acid-Functionalized SBA-15 Mesoporous Silica for Esterification
- One-photon Absorption Characterizations, Dipole Polarizabilities and Second Hyperpolarizabilities of Chlorophyll a and Crocin
- The Optical and Crystallite Characterization of Bilayer TiO2 Films Coated on Different ITO layers
- Topical Issue on Bond Activation
- Metal-mediated reactions towards the synthesis of a novel deaminolysed bisurea, dicarbamolyamine
- The structure of ortho-(trifluoromethyl)phenol in comparison to its homologues – A combined experimental and theoretical study
- Heterogeneous catalysis with encapsulated haem and other synthetic porphyrins: Harnessing the power of porphyrins for oxidation reactions
- Recent Advances on Mechanistic Studies on C–H Activation Catalyzed by Base Metals
- Reactions of the organoplatinum complex [Pt(cod) (neoSi)Cl] (neoSi = trimethylsilylmethyl) with the non-coordinating anions SbF6– and BPh4–
- Erratum
- Investigation on Two Compounds of O, O’-dithiophosphate Derivatives as Corrosion Inhibitors for Q235 Steel in Hydrochloric Acid Solution

