Abstract
Methylene blue dye has been widely used in many industries and usually released in natural water sources, which become a health thereat to human-beings and microbes. This paper demonstrates an oxidation method to remove methylene blue in water. The effect of natural organic matter humic acid, on the degradation of methylene blue using PMS was investigated. The results show that PMS could effectively degrade 50 mg/L methylene blue (>95%) when the PMS concentration was larger than 1.0 mM. Humic acid had either negative or positive impact on the degradation processes because of the co-existence of several competitive degradation mechanisms (I: humic acid competes with methylene blue for PMS; II: humic acid activates PMS to produce sulfate radicals; III: Cl–1 competes with methylene blue for sulfate radicals). This study is expected to provide valuable information to improve in situ remediation of dye-contaminated wastewater in the existence of natural organic matters.
1 Introduction
The control of water quality is of paramount importance to both environment and human health [1,2]. Thousands of pollutants can be found in wastewater, such as heavy metals, phenols, polychlorinated biphenyls, pesticides, and dyes [3,4,5]. It is reported that 10-15% of dyes are lost in the effluent process. Among these, organic dyes are highly colored, harmful to human beings, and result in significant increase of COD in water bodies [6]. Methylene blue is a cationic dye and widely used by industries including textile, printing, dye manufacturing, plastics, pharmaceutical, and photography industries. Its complicated chemical structure causes its recalcitrance to conventional biological treatment.
Chemical remediation has been widely applied for wastewater treatments due to its high efficiency for most pollutants and easy industrialized operation, among which, advanced oxidation process (AOP) based on peroxymonosulfate (PMS) or persulfate is a highly efficient method for a wide range of pollutants, such as phenol, dyes, and polychlorinated biphenyls [7,8,9]. Peroxymonosulfate is a strong oxidant with the standard potential of 1.82~2.01V. Transition metals, heat, radio-activation, alkali, and carbon based materials have been applied to activate PMS to produce sulfate radicals to promote the oxidation reaction [10,11,12,13]. However, the introduction of synthetic chemicals or metals into the system may lead to the operation difficulty or secondary pollution risk. The use of heat and radio-activation is complicated and difficult for large-scale operation. Previous studies also implied that the organic compounds such as ketones, primary alcohols, and low carbon chain aldehydes could activate PMS or persulfate for chemical oxidation applications in the field scale [14]. These organic compounds exist broadly in the natural water or soil. They are environmental friendly and derived from natural resources. In this research, the effect of humic acid which is a natural organic matter, on the methylene blue degradation using PMS was investigated for the first time.
This paper addresses an in-situ oxidative wastewater remediation method to degrade methylene blue using PMS in the absence of any activator and in the addition of a nature organic matter humic acid. The effect of humic acid on the degradation of the methylene blue effluent under different concentration of PMS was studied. We also conducted a list of experiments to elucidate the methylene blue degradation mechanism with the coexistence of humic acid and PMS. It is expected that this study could give some insights on how natural organic matters affect in situ oxidative remediation process of polluted natural water.
2 Materials and Methods
2.1 Chemicals and Materials
Methylene blue, humic acid, peroxymonosulfate, sodium hydrogen, sulfuric acid, ethanol, and sodium chloride were purchased from Sigma Aldrich and used as received. Deionized water from Milli-Q (Millipore) with resistivity >18.2MΩ·cm was used in all the experiments.
2.2 Experimental procedures and analysis
The batch experiments were conducted at room temperature (24°C). Different dosages of humic acid and PMS were added into wastewater that contained methylene blue, in a 50 mL tube. The pH value was adjusted to the desired level by adding either 0.1 M sulfuric acid or sodium hydroxide. Then the oxidative reaction was conducted on a reciprocating shaker with the shaking speed of 50 rpm under dark. At selected time intervals, about 10 mL of samples was taken out from the tube for analysis, and scavenger (0.1mL ethanol) was added into the sample immediately. The samples were placed inside a UV-VIS spectrophotometer cell (Thermo Scientific Evolution 60S) to determine the maximum absorption for the dye at an absorption wavelength of 660 nm. The dye removal efficiency (%), was expressed as follows:
Ethical approval: The conducted research is not related to either human or animals use.
3 Results and Discussion
3.1 Degradation of methylene blue by PMS at different pH value
The peroxymonosulfate (PMS) is a strong acidic oxidant due to the existing of HSO5–, which could oxidize methylene blue without the aid of any activator. Figure 1 shows the effects of solution pH values (2.3 to 9.0) and reaction time on the degradation of methylene blue. In the first half hour, concentration of methylene blue decreased from 50 mg/L to about 20 mg/L for all the pH values except pH of 9.0. After 24 h reaction, the methylene blue was significantly degraded (approximately 85% ~ 90% efficiency) for all conditions. And for the same pH sample, with the increase of reaction time, the methylene blue degradation efficiency enhanced accordingly. Obviously, reaction time but not the initial pH value greatly affects the degradation efficiency of methylene blue.
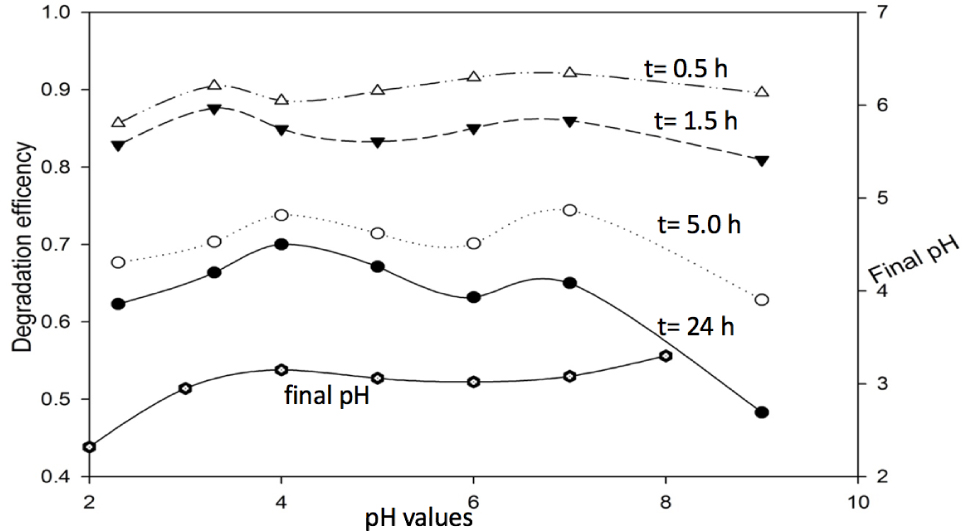
The effect of pH on the degradation of 50 mg/L methylene blue using 1.0 mM PMS and at different reaction time.
Another interesting phenomenon is that the dye degradation efficiency was much lower at pH 9 at a reaction time of 1.5 h. This result agreed well with previous study [15], and it can be explained by self-decomposition of PMS in alkaline conditions. At reaction time of 5 h and 24 h, the degradation efficiency was significantly improved for all pH values, and the final pH was found to be 3. Which indicated that in these runs the decomposition of methylene blue could decrease the pH values, resulting in the impedance of the self-decomposition of PMS.
It is believed that the relatively high concentration of PMS (1.0 mM) may hidden the effect of the pH value at a low loading of methylene blue. Therefore, the effect of the pH on the degradation of methylene blue at a relatively low concentration of PMS (0.5 mM) was further investigated. As shown in Figure 2, the degradation efficiency shows a similar trend as that using 1.0 mM PMS. But all the methylene blue degradation efficiencies were less than 60%. As shown in Figure 1 and 2, the pH value has slight impact on the degradation efficiency when the pH value was lower than 7, but the degradation efficiency decreased significantly, when the pH value was larger than 7. This indicates that the self-decomposition of PMS was not negligible at a low concentration of PMS. Since extremely low or high pH is very rare in the natural wastewater system, it can be concluded that the effect of pH was negligible in this methylene blue degradation process. In the following study, the pH values were adjusted to 7.0 ± 0.2 to mimic the natural water and remain high degradation efficiencies.
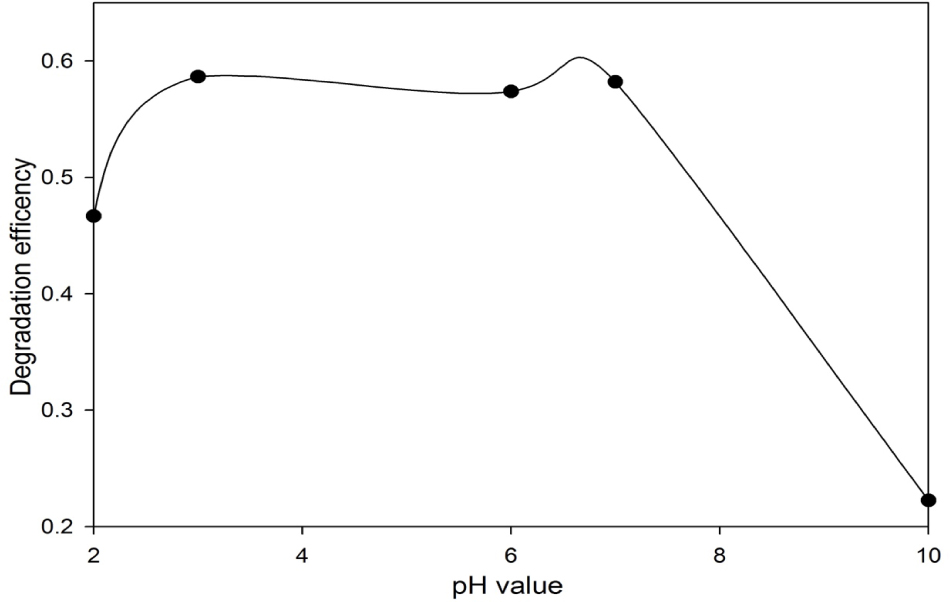
The effect of pH on the degradation of 50 mg/L methylene blue using 0.5 mM PMS at 24 h.
The degradation rate constant (kobs) based on the pseudo first order model was less than 0.1 min–1 calculated from the data (see Figure 1 which was much less than those previously reported in literatures. As demonstrated in Ghauch’s study, the degradation rate constant of methylene blue in a heated persulfate process was 4.81-9.05 min–1 at a pH range of 4 - 9 [16]. The distinctive difference in the reaction rate can be attributed to the absence of activators in this study. However, from the view of in situ remediation and environmental benefits, oxidation without additional chemicals should be a better choice.
3.2 Effect of the PMS concentration on the degradation of methylene blue
Figure 3 demonstrates the degradation effects of methylene blue with a concentration ranging from 50 mg/L to 200 mg/L under different PMS concentrations. The methylene blue degradation efficiency was increased with the enhancement of the PMS concentration in all the designed experiments. However, the degradation curve followed a slightly different trend depending on its initial concentration. A low dosage of PMS (0.5 mM) could not effectively degrade methylene blue at a relatively high concentration (larger than 50 mg/L). When the PMS concentration was increased from 1.0 mM to 2.5 mM, the degradation efficiency of 100, 150, and 200 mg/L methylene blue was increased to 28%, 47%, and 50%, respectively. In the addition of 6 mM PMS, the degradation efficiency for 150 and 200 mg/L methylene blue was 88% and 77%, respectively. At a high concentration of methylene blue (150 mg/L or 200 mg/L), the degradation efficiency was not significantly improved even in the addition of high concentration of PMS (6.0 mM in comparison with 3.5 mM PMS). This phenomenon could be attributed to the competitive consuming of PMS by intermediates [17,18].
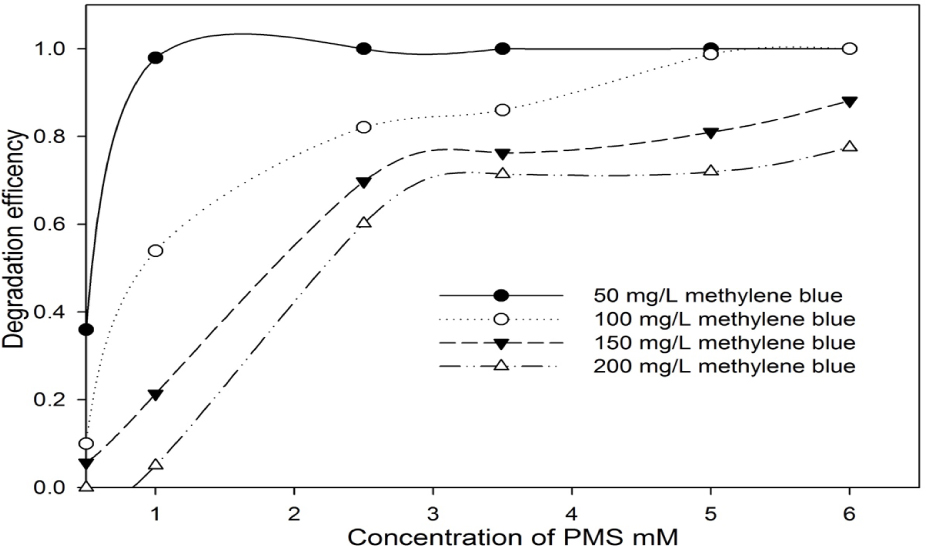
Degradation of different concentrations of methylene blue using PMS at different concentrations for 24 h.
3.3 Effect of humic acid
Humic acids occur in the natural water with the concentration ranging from 0.1~20 mg/L [19]. According to the previous studies, humic acid was one of the water pollutants that had impact on the water purification process due to its chelating properties [20,21]. Recently, Fang et al. reported that the redox active moiety of quinone in humic acid could act as an effective catalyst to activate persulfate to degrade polychlorinated biphenyls [22]. It is unclear whether humic acid activates PMS to produce sulfate radicals, which is possible to accelerate the degradation of methylene blue. Therefore, Herein, the effect of humic acid on degradation of methylene blue in the existence of PMS and its degradation mechanism was elucidated.
3.3.1 Effect of humic acid at different PMS concentrations
Based on our aforementioned results, the degradation efficiency of methylene blue was < 70% at a concentration of PMS < 2 mM and methylene blue ≥ 100 mg/L. Figure 4a shows the effects of different amount of humic acids (0, 2, 5, 10, 15 mg/L) on the degradation of methylene blue with the mole ratio of PMS to methylene blue of 3.2. When the concentration of humic acid was less than 2 mg/L, its effect on the degradation efficiency was negligible for all the designed methylene blue solutions. In a 50 mg/L methylene blue solution, the addition of 5 mg/L humic acid accelerated the degradation efficiency (from 31% to 60%). In a 100 mg/L methylene blue with 1mM PMS, the addition of 10 or 15 mg/L humic acid increased the degradation efficiency from initial 54% to more than 64%. The results shown in Figure 4b implies that 30 mg/L humic acid was beneficial for the degradation of 150 mg/L methylene blue (approximately 60% degradation efficiency, in comparison with that in the absence of humic acid 40%). In a 200 mg/L methylene blue effluent with 6 mM PMS, 35 mg/L humic acid could achieve the maximum degradation efficiency (95%), that was also greater than that in the absence of humic acid. It was noticeable that sometimes the addition of humic acid had the negative effect on the methylene blue degradation as shown in Figure 4b.
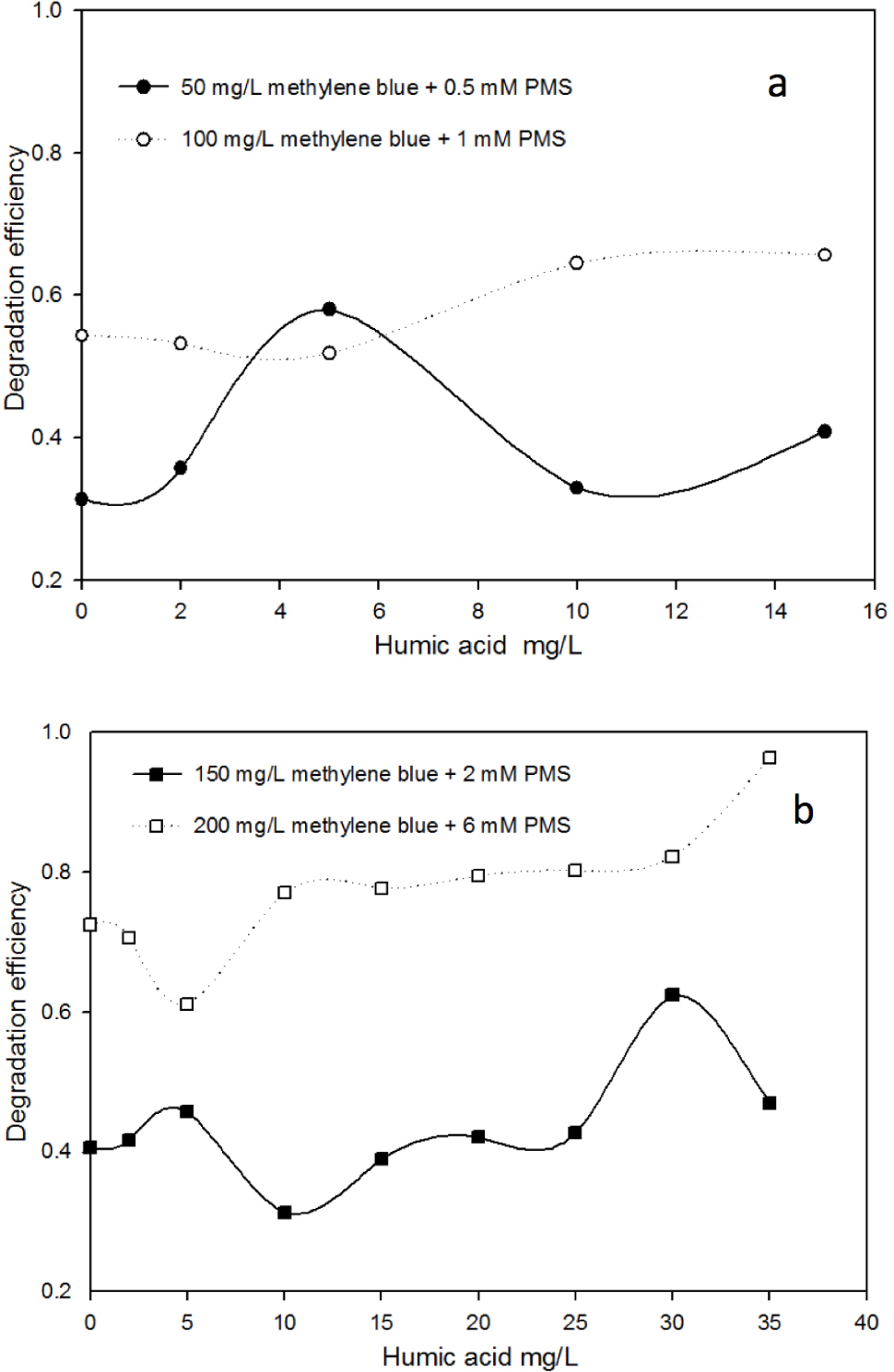
The effect of humic acid on the degradation of methylene blue using different PMS concentrations for 24 h.
3.3.2 Effect of humic acid on the methylene blue degradation under a relatively high concentration of PMS
Figure 5 presents the effect of humic acid on the degradation of methylene blue when the PMS concentration was relative high. More than 95% degradation efficiency was achieved in a 50 mg/L methylene blue effluent when the PMS was 2 mM for all the examined humic acid concentrations. The degradation efficiency was higher than 93% for 100 mg/L methylene blue with a 5 mM PMS. The increase of the humic acid concentration (0 to 30 mg/L) could slightly reduce the degradation efficiency. It could be explained by the competitive consuming of PMS by humic acid.
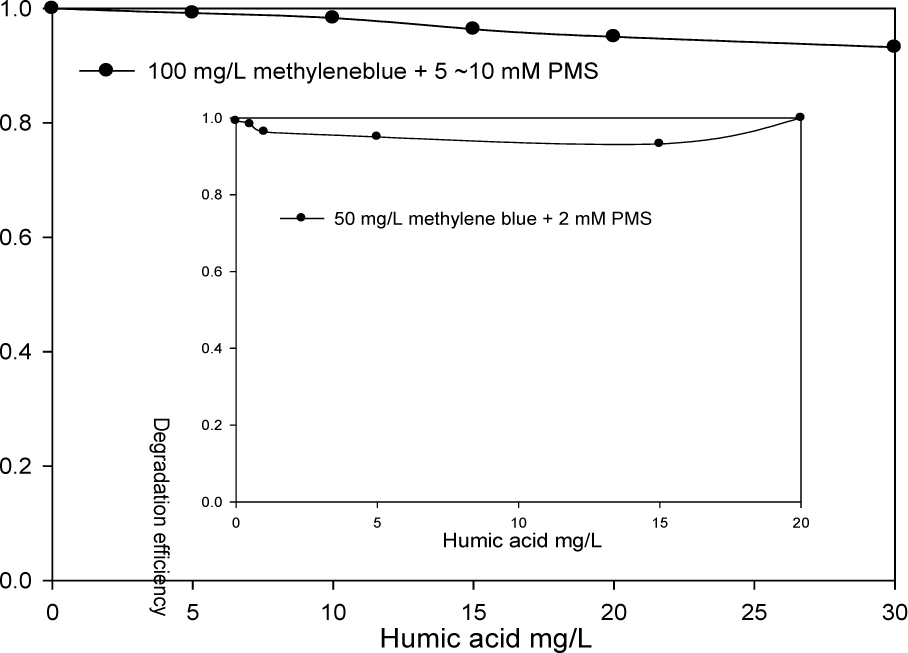
The effect of humic acid on the degradation of 50 mg/L and 100 mg/L methylene blue at high concentration of PMS (the mole ratio >12.8) for 24 h
3.3.3 The degradation mechanism of methylene blue with the coexistence of PMS and humic acid
The aforementioned results demonstrate that humic acids had either positive or slightly negative effects on the degradation of methylene blue. The negative effect may be attributed to the competitive reaction between humic acid and methylene blue with PMS, which was previously reported [23,24] and this was in accordance with the results obtained in this study (see Figure 1S).
Results shown in Figure 4 demonstrate that in a low loading of PMS and humic acid could accelerate the degradation of methylene blue. This result indicates that humic acid may act as the activator to promote the generation of free radicals from PMS. In order to elucidate the effect of humic acid on free radical generation, a radical quenching experiment was conducted. Ethanol, whose rate constant with sulfate radical is 1.6-7.7 ×107 M–1S–1 [25] was selected as the scavenger. Figure 6 compares the degradation efficiency of 50 mg/L methylene blue using 0.5 mM PMS in the absence and presence of humic acid and ethanol. The results illustrate that, only the addition of 5 mg/L humic acid increased the degradation of methylene blue from 39.3% to 62.8%, indicating that humic acid could greatly accelerate the methylene blue degradation. When ethanol was added, the degradation efficiency decreased significantly. Similar trend was observed in 100 mg/L methylene blue solution with the addition of 10 mg/L humic acid and 100 mM ethanol. Therefore, we can conclude that humic acid is an effective activator to initiate sulfate radicals to degrade methylene blue at a low loading of PMS, and this result agreed well with previous research [26]. Besides, methylene blue (C16H18N3ClS) is a cationic dye, with Cl– counter anion that is formed during its dissolving process. Previous research reported that sulfate radicals could be consumed by Cl–1. The reaction was described as follows:
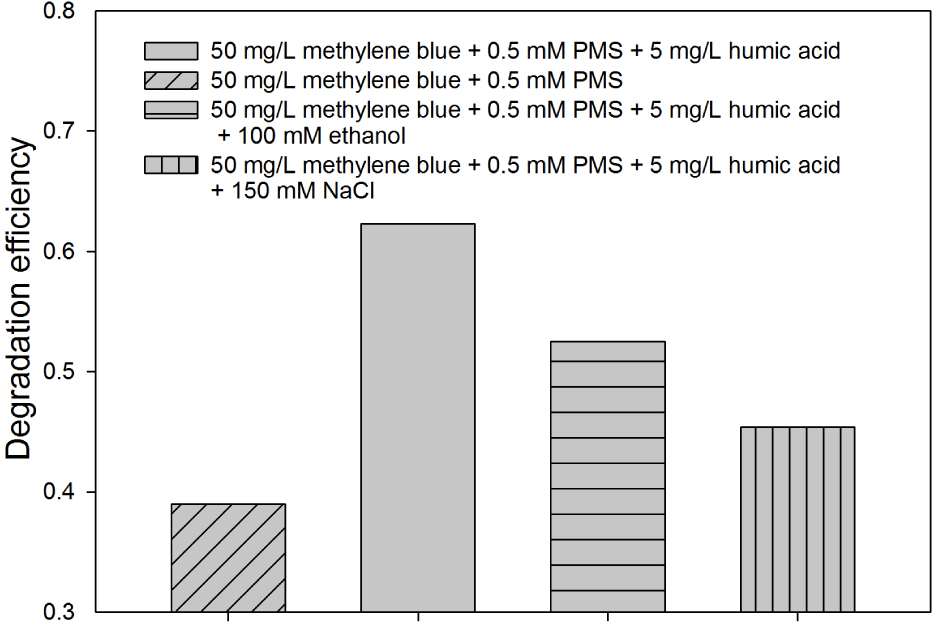
Radical scavenger study for 50 mg/L methylene blue with 0.5 mM PMS and 5 mg/L humic acid using ethanol and NaCl
with the reaction rate of k=3.0×108 M–1S–1 [27]. As shown in Figure 6, when 150 mM NaCl was added to the effluent, the degradation efficiency was decreased from 62.8% to 44.3%, proving that Cl–1 inhibited the methylene blue degradation process.
Based on these results, a degradation mechanism was proposed and described in Figure S2. Humic acid acts as the natural activator for PMS to produce sulfate radicals, which could promote the degradation efficiency of methylene blue. Meanwhile, both humic acid and methylene blue competitively react with PMS, resulting in a negative impact of humic acid on the methylene blue degradation. In addition, the Cl–1 from methylene blue acts as the radical scavenger to reduce the methylene blue degradation efficiency as well.
4 Conclusions
This paper addresses a method to degrade methylene blue in effluents using peroxymonosulfate (PMS) without the usage of any additional activator. The results demonstrate that the PMS was capable of degrading methylene blue effectively without any other activator and at a relatively high PMS concentration. In a low PMS concentration (0.5 mM PMS V.S. 50 mg/L methylene blue), the methylene blue degradation efficiency was significantly decreased and extremely low degradation efficiency was observed when the pH was greater than 7. Humic acid could activate the PMS to produce sulfate radicals to promote the methylene blue degradation. However, the degradation efficiency could be reduced by the competing reactions between humic acid and methylene blue with PMS when the PMS is at a high dosage. Other elements (e.g. Cl–1) in the natural water may also act as the radical scavenger for sulfate radicals and resulted in the reduction of the degradation efficiency. All these reactions synergically occurred in methylene blue effluent system and led to a complicated degradation mechanism. It is expected that this research can provide some insights to efficiently control the degradation of methylene blue in wastewater using low cost PMS as the oxidizer.
Acknowledgement
This work was financially supported by the National Natural Science Foundation of China (No. 51409024, 51579096), the Natural Science Foundation of Hu Nan province (No. 2017JJ3341), the China Scholarship Council, the Science and Technology Program of Hunan province (No. 2014FJ3063), and the National Program for Support of Top–Notch Young Professionals of China (2012).
Conflict of interest: Authors state no conflict of interest.
References
[1] Huang Y.Z., Tackling China’s Environmental Health Crisis. Council on foreign relations, 2015.Search in Google Scholar
[2] Yao W, Yu, S. J., Wang J, Zou Y D., Wang X. K., Enhanced removal of methyl orange on calcined glycerol-modified nanocrystallined Mg/Al layered double hydroxides, Chem. Eng. J., 2017, 307, 476-486.10.1016/j.cej.2016.08.117Search in Google Scholar
[3] Pang Y., Zeng G.M., Tang L., Preparation and application of stability enhanced magnetic nanoparticles for rapid removal of Cr(VI), Chem. Eng. J., 2011, 175, 222- 227.10.1016/j.cej.2011.09.098Search in Google Scholar
[4] Yu S. J., Wang X. X., Yao W., Wang J., Wang X. K., Macroscopic, Spectroscopic, and Theoretical Investigation for the Interaction of Phenol and Naphthol on Reduced Graphene Oxide, Environ. Sci. Technol., 2017, 51, 3278-3286.10.1021/acs.est.6b06259Search in Google Scholar
[5] Li P., Liu Z.P., Wang X.G., Guo Y.D., Wang L., Enhanced decolorization of methyl orange in aqueous solution using iron-carbon micro-electrolysis activation of sodium persulfate, Chemosphere, 2017, 180, 100-107.10.1016/j.chemosphere.2017.04.019Search in Google Scholar
[6] Shitu, A.M.M.A., Ibrahim, A., Removal of methylene blue using low cost adsorbent: a review, Res. J. Chem. Sci., 2014, 4, 91-102.Search in Google Scholar
[7] Andreozzi, R., Caprio, V., Insola, A., Marotta, R., Advanced oxidation processes (AOP) for water purification and recovery, Catal. Today, 1999, 53, 51-59.10.1016/S0920-5861(99)00102-9Search in Google Scholar
[8] Aaron J.J., Advanced Oxidation Processes in Water/Wastewater Treatment: Principles and Applications. A Review, Crit. Rev. Env. Sci. Tec., 2014, 44, 2577-2641.10.1080/10643389.2013.829765Search in Google Scholar
[9] Matzek L.W., Kimberly E. C., Activated persulfate for organic chemical degradation: A review, Chemosphere, 2016, 151, 178-18810.1016/j.chemosphere.2016.02.055Search in Google Scholar PubMed
[10] Corbin J.F., Mechanisms of Base, Mineral, and Soil Activation of Persulfate for Groundwater Treatment, PhD Theses, 2008.Search in Google Scholar
[11] Shukla P.R., Wang S., Sun H., Ang H.M., Tadé M., Activated carbon supported cobalt catalysts for advanced oxidation of organic contaminants in aqueous solution, Appl. Catal. B Environ. 2010, 100, 529-534.10.1016/j.apcatb.2010.09.006Search in Google Scholar
[12] Lee H., Lee H.J., Jeong J., Lee J., Park N.B., Lee C., Activation of persulfate by carbon nanotubes: Oxidation of organic compounds by nonradical mechanism, Chem. Eng. J., 2015, 266, 28-33.10.1016/j.cej.2014.12.065Search in Google Scholar
[13] Kang J., Duan X., Zhou L., Sun H., Tadé M.O., Wang S., Carbocatalytic activation of persulfate for removal of antibiotics in water solutions, Chem. Eng. J., 2016, 288, 399-405.10.1016/j.cej.2015.12.040Search in Google Scholar
[14] Ocampo A.M., Persulfate activation by organic compounds, PhD thesis, Washington state university, USA, 2009.Search in Google Scholar
[15] Farshid G., Mahsa M., Mohammad M., Textile wastewater decolorization by zero valent iron activated peroxymonosulfate: compared with zero valent copper, J. Environ. Chem. Eng., 2014, 23, 1846-1851.10.1016/j.jece.2014.08.003Search in Google Scholar
[16] Ghauch A., Tuqan A.M., Kibbi N., Geryes S., Methylene blue discoloration by heated persulfate in aqueous solution, Chem. Eng. J., 2012, 213, 259-271.10.1016/j.cej.2012.09.122Search in Google Scholar
[17] Wang D.G., Li Y.F., Yang M., Han M., Decomposition of polycyclic aromatic hydrocarbons in atmospheric aqueous droplets through sulfate anion radicals: An experimental and theoretical study, Sci. Total Environ., 2008, 393, 64-71.10.1016/j.scitotenv.2007.11.036Search in Google Scholar PubMed
[18] Chen Y. Q., Deng P. Y., Xie P. C., Shang R., Wang Z. P., Wang S. L., Heat-activated persulfate oxidation of methyl- and ethylparabens: Effect, kinetics, and mechanism, Chemosphere, 2017, 168, 1628-1636.10.1016/j.chemosphere.2016.11.143Search in Google Scholar PubMed
[19] Boggs S., Livermore D., Seitz M.G., Humic Substances in Natural Waters and their Complexation with Trace Metals and Radionuclides: A Review, Technical Report Archive & Image Library, 1985.10.2172/5569909Search in Google Scholar
[20] Wang J., Han X., Ma X., Ji Y., Bi L., Adsorptive removal of humic acid from aqueous solution on polyaniline/attapulgite composite, Chem. Eng. J., 2011, 173,171-177.10.1016/j.cej.2011.07.065Search in Google Scholar
[21] Fuhrman H.G., Mikkelsen P.S., Simultaneous removal of As, Cd, Cr, Cu, Ni and Zn from storm water using high-efficiency industrial sorbents: Effect of pH, contact time and humic acid, Sci. Total Environ., 2016, 566-567, 76-85.10.1016/j.scitotenv.2016.04.210Search in Google Scholar PubMed
[22] Fang G.D., Gao J., Dionysios D.D., Zhou D.M., Activation of Persulfate by Quinones: Free Radical Reactions and Implication for the Degradation of PCBs, Environ. Sci. Technol., 2013, 47, 4605-4611.10.1021/es400262nSearch in Google Scholar PubMed
[23] Chen Y., Zhang K., Zuo Y.G., Direct and indirect photodegradation of estriol in the presence of humic acid, nitrate and iron complexes in water solutions, Sci. Total Environ. 2013, 463-464,802-809.10.1016/j.scitotenv.2013.06.026Search in Google Scholar PubMed
[24] Oh W.D., Lu S.K., Dong Z.L., Lim T.T., Performance of magnetic activated carbon composite as peroxymonosulfate activator and regenerable adsorbent via sulfate radical-mediated oxidation processes, J. Hazard. Mater., 2015, 284,1-9.10.1016/j.jhazmat.2014.10.042Search in Google Scholar PubMed
[25] Zhu C.G., Fang G.D., Dionysios D.D., Liu C., Efficient transformation of DDTs with Persulfate Activation by Zerovalent Iron Nanoparticles: A Mechanistic Study, J. Hazard. Mater., 2016, 316, 232-241.10.1016/j.jhazmat.2016.05.040Search in Google Scholar PubMed
[26] George P.A., Dionysiou D.D., Degradation of Organic Contaminants in Water with Sulfate Radicals Generated by the Conjunction of Peroxymonosulfate with Cobalt, Environ. Sci. Technol., 2003, 37, 4790-4797.10.1021/es0263792Search in Google Scholar PubMed
[27] Yu X.Y., Bao Z.C., Baker J.R., Free Radical Reactions Involving Cl•●, Cl2● and SO4–● in the 248 nm photolysis of aqueous solutions containing S2O82– and Cl–, J. Phys. Chem. A, 2004, 108, 295-308.10.1021/jp036211iSearch in Google Scholar
Supplemental Material
The online version of this article offers supplementary material (https://doi.org/10.1515/chem-2018-0044).
© 2018 Ya Pang et al., published by De Gruyter
This work is licensed under the Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 License.
Articles in the same Issue
- Regular Articles
- The effect of CuO modification for a TiO2 nanotube confined CeO2 catalyst on the catalytic combustion of butane
- The preparation and antibacterial activity of cellulose/ZnO composite: a review
- Linde Type A and nano magnetite/NaA zeolites: cytotoxicity and doxorubicin loading efficiency
- Performance and thermal decomposition analysis of foaming agent NPL-10 for use in heavy oil recovery by steam injection
- Spectroscopic (FT-IR, FT-Raman, UV, 1H and 13C NMR) insights, electronic profiling and DFT computations on ({(E)-[3-(1H-imidazol-1-yl)-1-phenylpropylidene] amino}oxy)(4-nitrophenyl)methanone, an imidazole-bearing anti-Candida agent
- A Simplistic Preliminary Assessment of Ginstling-Brounstein Model for Solid Spherical Particles in the Context of a Diffusion-Controlled Synthesis
- M-Polynomials And Topological Indices Of Zigzag And Rhombic Benzenoid Systems
- Photochemical Transformation of some 3-benzyloxy-2-(benzo[b]thiophen-2-yl)-4Hchromen-4-ones: A Remote Substituent Effect
- Dynamic Changes of Secondary Metabolites and Antioxidant Activity of Ligustrum lucidum During Fruit Growth
- Studies on the flammability of polypropylene/ammonium polyphosphate and montmorillonite by using the cone calorimeter test
- DSC, FT-IR, NIR, NIR-PCA and NIR-ANOVA for determination of chemical stability of diuretic drugs: impact of excipients
- Antioxidant and Hepatoprotective Effects of Methanolic Extracts of Zilla spinosa and Hammada elegans Against Carbon Tetrachlorideinduced Hepatotoxicity in Rats
- Prunus cerasifera Ehrh. fabricated ZnO nano falcates and its photocatalytic and dose dependent in vitro bio-activity
- Organic biocides hosted in layered double hydroxides: enhancing antimicrobial activity
- Experimental study on the regulation of the cholinergic pathway in renal macrophages by microRNA-132 to alleviate inflammatory response
- Synthesis, characterization, in-vitro antimicrobial properties, molecular docking and DFT studies of 3-{(E)-[(4,6-dimethylpyrimidin-2-yl)imino]methyl} naphthalen-2-ol and Heteroleptic Mn(II), Co(II), Ni(II) and Zn(II) complexes
- M-Polynomials and Topological Indices of Dominating David Derived Networks
- Human Health Risk Assessment of Trace Metals in Surface Water Due to Leachate from the Municipal Dumpsite by Pollution Index: A Case Study from Ndawuse River, Abuja, Nigeria
- Analysis of Bowel Diseases from Blood Serum by Autofluorescence and Atomic Force Microscopy Techniques
- Hydrographic parameters and distribution of dissolved Cu, Ni, Zn and nutrients near Jeddah desalination plant
- Relationships between diatoms and environmental variables in industrial water biotopes of Trzuskawica S.A. (Poland)
- Optimum Conversion of Major Ginsenoside Rb1 to Minor Ginsenoside Rg3(S) by Pulsed Electric Field-Assisted Acid Hydrolysis Treatment
- Antioxidant, Anti-microbial Properties and Chemical Composition of Cumin Essential Oils Extracted by Three Methods
- Regulatory mechanism of ulinastatin on autophagy of macrophages and renal tubular epithelial cells
- Investigation of the sustained-release mechanism of hydroxypropyl methyl cellulose skeleton type Acipimox tablets
- Bio-accumulation of Polycyclic Aromatic Hydrocarbons in the Grey Mangrove (Avicennia marina) along Arabian Gulf, Saudi Coast
- Dynamic Change of Secondary Metabolites and spectrum-effect relationship of Malus halliana Koehne flowers during blooming
- Lipids constituents from Gardenia aqualla Stapf & Hutch
- Effect of using microwaves for catalysts preparation on the catalytic acetalization of glycerol with furfural to obtain fuel additives
- Effect of Humic Acid on the Degradation of Methylene Blue by Peroxymonosulfate
- Serum containing drugs of Gua Lou Xie Bai decoction (GLXB-D) can inhibit TGF-β1-Induced Epithelial to Mesenchymal Transition (EMT) in A549 Cells
- Antiulcer Activity of Different Extracts of Anvillea garcinii and Isolation of Two New Secondary Metabolites
- Analysis of Metabolites in Cabernet Sauvignon and Shiraz Dry Red Wines from Shanxi by 1H NMR Spectroscopy Combined with Pattern Recognition Analysis
- Can water temperature impact litter decomposition under pollution of copper and zinc mixture
- Released from ZrO2/SiO2 coating resveratrol inhibits senescence and oxidative stress of human adipose-derived stem cells (ASC)
- Validated thin-layer chromatographic method for alternative and simultaneous determination of two anti-gout agents in their fixed dose combinations
- Fast removal of pollutants from vehicle emissions during cold-start stage
- Review Article
- Catalytic activities of heterogeneous catalysts obtained by copolymerization of metal-containing 2-(acetoacetoxy)ethyl methacrylate
- Antibiotic Residue in the Aquatic Environment: Status in Africa
- Regular Articles
- Mercury fractionation in gypsum using temperature desorption and mass spectrometric detection
- Phytosynthetic Ag doped ZnO nanoparticles: Semiconducting green remediators
- Epithelial–Mesenchymal Transition Induced by SMAD4 Activation in Invasive Growth Hormone-Secreting Adenomas
- Physicochemical properties of stabilized sewage sludge admixtures by modified steel slag
- In Vitro Cytotoxic and Antiproliferative Activity of Cydonia oblonga flower petals, leaf and fruit pellet ethanolic extracts. Docking simulation of the active flavonoids on anti-apoptotic protein Bcl-2
- Synthesis and Characterization of Pd exchanged MMT Clay for Mizoroki-Heck Reaction
- A new selective, and sensitive method for the determination of lixivaptan, a vasopressin 2 (V2)-receptor antagonist, in mouse plasma and its application in a pharmacokinetic study
- Anti-EGFL7 antibodies inhibit rat prolactinoma MMQ cells proliferation and PRL secretion
- Density functional theory calculations, vibration spectral analysis and molecular docking of the antimicrobial agent 6-(1,3-benzodioxol-5-ylmethyl)-5-ethyl-2-{[2-(morpholin-4-yl)ethyl] sulfanyl}pyrimidin-4(3H)-one
- Effect of Nano Zeolite on the Transformation of Cadmium Speciation and Its Uptake by Tobacco in Cadmium-contaminated Soil
- Effects and Mechanisms of Jinniu Capsule on Methamphetamine-Induced Conditioned Place Preference in Rats
- Calculating the Degree-based Topological Indices of Dendrimers
- Efficient optimization and mineralization of UV absorbers: A comparative investigation with Fenton and UV/H2O2
- Metabolites of Tryptophane and Phenylalanine as Markers of Small Bowel Ischemia-Reperfusion Injury
- Adsorption and determination of polycyclic aromatic hydrocarbons in water through the aggregation of graphene oxide
- The role of NR2C2 in the prolactinomas
- Chromium removal from industrial wastewater using Phyllostachys pubescens biomass loaded Cu-S nanospheres
- Hydrotalcite Anchored Ruthenium Catalyst for CO2 Hydrogenation Reaction
- Preparation of Calcium Fluoride using Phosphogypsum by Orthogonal Experiment
- The mechanism of antibacterial activity of corylifolinin against three clinical bacteria from Psoralen corylifolia L
- 2-formyl-3,6-bis(hydroxymethyl)phenyl benzoate in Electrochemical Dry Cell
- Electro-photocatalytic degradation of amoxicillin using calcium titanate
- Effect of Malus halliana Koehne Polysaccharides on Functional Constipation
- Structural Properties and Nonlinear Optical Responses of Halogenated Compounds: A DFT Investigation on Molecular Modelling
- DMFDMA catalyzed synthesis of 2-((Dimethylamino)methylene)-3,4-dihydro-9-arylacridin-1(2H)-ones and their derivatives: in-vitro antifungal, antibacterial and antioxidant evaluations
- Production of Methanol as a Fuel Energy from CO2 Present in Polluted Seawater - A Photocatalytic Outlook
- Study of different extraction methods on finger print and fatty acid of raw beef fat using fourier transform infrared and gas chromatography-mass spectrometry
- Determination of trace fluoroquinolones in water solutions and in medicinal preparations by conventional and synchronous fluorescence spectrometry
- Extraction and determination of flavonoids in Carthamus tinctorius
- Therapeutic Application of Zinc and Vanadium Complexes against Diabetes Mellitus a Coronary Disease: A review
- Study of calcined eggshell as potential catalyst for biodiesel formation using used cooking oil
- Manganese oxalates - structure-based Insights
- Topological Indices of H-Naphtalenic Nanosheet
- Long-Term Dissolution of Glass Fibers in Water Described by Dissolving Cylinder Zero-Order Kinetic Model: Mass Loss and Radius Reduction
- Topological study of the para-line graphs of certain pentacene via topological indices
- A brief insight into the prediction of water vapor transmissibility in highly impermeable hybrid nanocomposites based on bromobutyl/epichlorohydrin rubber blends
- Comparative sulfite assay by voltammetry using Pt electrodes, photometry and titrimetry: Application to cider, vinegar and sugar analysis
- MicroRNA delivery mediated by PEGylated polyethylenimine for prostate cancer therapy
- Reversible Fluorescent Turn-on Sensors for Fe3+ based on a Receptor Composed of Tri-oxygen Atoms of Amide Groups in Water
- Sonocatalytic degradation of methyl orange in aqueous solution using Fe-doped TiO2 nanoparticles under mechanical agitation
- Hydrotalcite Anchored Ruthenium Catalyst for CO2 Hydrogenation Reaction
- Production and Analysis of Recycled Ammonium Perrhenate from CMSX-4 superalloys
- Topical Issue on Agriculture
- New phosphorus biofertilizers from renewable raw materials in the aspect of cadmium and lead contents in soil and plants
- Survey of content of cadmium, calcium, chromium, copper, iron, lead, magnesium, manganese, mercury, sodium and zinc in chamomile and green tea leaves by electrothermal or flame atomizer atomic absorption spectrometry
- Biogas digestate – benefits and risks for soil fertility and crop quality – an evaluation of grain maize response
- A numerical analysis of heat transfer in a cross-current heat exchanger with controlled and newly designed air flows
- Freshwater green macroalgae as a biosorbent of Cr(III) ions
- The main influencing factors of soil mechanical characteristics of the gravity erosion environment in the dry-hot valley of Jinsha river
- Free amino acids in Viola tricolor in relation to different habitat conditions
- The influence of filler amount on selected properties of new experimental resin dental composite
- Effect of poultry wastewater irrigation on nitrogen, phosphorus and carbon contents in farmland soil
- Response of spring wheat to NPK and S fertilization. The content and uptake of macronutrients and the value of ionic ratios
- The Effect of Macroalgal Extracts and Near Infrared Radiation on Germination of Soybean Seedlings: Preliminary Research Results
- Content of Zn, Cd and Pb in purple moor-grass in soils heavily contaminated with heavy metals around a zinc and lead ore tailing landfill
- Topical Issue on Research for Natural Bioactive Products
- Synthesis of (±)-3,4-dimethoxybenzyl-4-methyloctanoate as a novel internal standard for capsinoid determination by HPLC-ESI-MS/MS(QTOF)
- Repellent activity of monoterpenoid esters with neurotransmitter amino acids against yellow fever mosquito, Aedes aegypti
- Effect of Flammulina velutipes (golden needle mushroom, eno-kitake) polysaccharides on constipation
- Bioassay-directed fractionation of a blood coagulation factor Xa inhibitor, betulinic acid from Lycopus lucidus
- Antifungal and repellent activities of the essential oils from three aromatic herbs from western Himalaya
- Chemical composition and microbiological evaluation of essential oil from Hyssopus officinalis L. with white and pink flowers
- Bioassay-guided isolation and identification of Aedes aegypti larvicidal and biting deterrent compounds from Veratrum lobelianum
- α-Terpineol, a natural monoterpene: A review of its biological properties
- Utility of essential oils for development of host-based lures for Xyleborus glabratus (Coleoptera: Curculionidae: Scolytinae), vector of laurel wilt
- Phenolic composition and antioxidant potential of different organs of Kazakh Crataegus almaatensis Pojark: A comparison with the European Crataegus oxyacantha L. flowers
- Isolation of eudesmane type sesquiterpene ketone from Prangos heyniae H.Duman & M.F.Watson essential oil and mosquitocidal activity of the essential oils
- Comparative analysis of the polyphenols profiles and the antioxidant and cytotoxicity properties of various blue honeysuckle varieties
- Special Issue on ICCESEN 2017
- Modelling world energy security data from multinomial distribution by generalized linear model under different cumulative link functions
- Pine Cone and Boron Compounds Effect as Reinforcement on Mechanical and Flammability Properties of Polyester Composites
- Artificial Neural Network Modelling for Prediction of SNR Effected by Probe Properties on Ultrasonic Inspection of Austenitic Stainless Steel Weldments
- Calculation and 3D analyses of ERR in the band crack front contained in a rectangular plate made of multilayered material
- Improvement of fuel properties of biodiesel with bioadditive ethyl levulinate
- Properties of AlSi9Cu3 metal matrix micro and nano composites produced via stir casting
- Investigation of Antibacterial Properties of Ag Doped TiO2 Nanofibers Prepared by Electrospinning Process
- Modeling of Total Phenolic contents in Various Tea samples by Experimental Design Methods
- Nickel doping effect on the structural and optical properties of indium sulfide thin films by SILAR
- The effect mechanism of Ginnalin A as a homeopathic agent on various cancer cell lines
- Excitation functions of proton induced reactions of some radioisotopes used in medicine
- Oxide ionic conductivity and microstructures of Pr and Sm co-doped CeO2-based systems
- Rapid Synthesis of Metallic Reinforced in Situ Intermetallic Composites in Ti-Al-Nb System via Resistive Sintering
- Oxidation Behavior of NiCr/YSZ Thermal Barrier Coatings (TBCs)
- Clustering Analysis of Normal Strength Concretes Produced with Different Aggregate Types
- Magnetic Nano-Sized Solid Acid Catalyst Bearing Sulfonic Acid Groups for Biodiesel Synthesis
- The biological activities of Arabis alpina L. subsp. brevifolia (DC.) Cullen against food pathogens
- Humidity properties of Schiff base polymers
- Free Vibration Analysis of Fiber Metal Laminated Straight Beam
- Comparative study of in vitro antioxidant, acetylcholinesterase and butyrylcholinesterase activity of alfalfa (Medicago sativa L.) collected during different growth stages
- Isothermal Oxidation Behavior of Gadolinium Zirconate (Gd2Zr2O7) Thermal Barrier Coatings (TBCs) produced by Electron Beam Physical Vapor Deposition (EB-PVD) technique
- Optimization of Adsorption Parameters for Ultra-Fine Calcite Using a Box-Behnken Experimental Design
- The Microstructural Investigation of Vermiculite-Infiltrated Electron Beam Physical Vapor Deposition Thermal Barrier Coatings
- Modelling Porosity Permeability of Ceramic Tiles using Fuzzy Taguchi Method
- Experimental and theoretical study of a novel naphthoquinone Schiff base
- Physicochemical properties of heat treated sille stone for ceramic industry
- Sand Dune Characterization for Preparing Metallurgical Grade Silicon
- Catalytic Applications of Large Pore Sulfonic Acid-Functionalized SBA-15 Mesoporous Silica for Esterification
- One-photon Absorption Characterizations, Dipole Polarizabilities and Second Hyperpolarizabilities of Chlorophyll a and Crocin
- The Optical and Crystallite Characterization of Bilayer TiO2 Films Coated on Different ITO layers
- Topical Issue on Bond Activation
- Metal-mediated reactions towards the synthesis of a novel deaminolysed bisurea, dicarbamolyamine
- The structure of ortho-(trifluoromethyl)phenol in comparison to its homologues – A combined experimental and theoretical study
- Heterogeneous catalysis with encapsulated haem and other synthetic porphyrins: Harnessing the power of porphyrins for oxidation reactions
- Recent Advances on Mechanistic Studies on C–H Activation Catalyzed by Base Metals
- Reactions of the organoplatinum complex [Pt(cod) (neoSi)Cl] (neoSi = trimethylsilylmethyl) with the non-coordinating anions SbF6– and BPh4–
- Erratum
- Investigation on Two Compounds of O, O’-dithiophosphate Derivatives as Corrosion Inhibitors for Q235 Steel in Hydrochloric Acid Solution
Articles in the same Issue
- Regular Articles
- The effect of CuO modification for a TiO2 nanotube confined CeO2 catalyst on the catalytic combustion of butane
- The preparation and antibacterial activity of cellulose/ZnO composite: a review
- Linde Type A and nano magnetite/NaA zeolites: cytotoxicity and doxorubicin loading efficiency
- Performance and thermal decomposition analysis of foaming agent NPL-10 for use in heavy oil recovery by steam injection
- Spectroscopic (FT-IR, FT-Raman, UV, 1H and 13C NMR) insights, electronic profiling and DFT computations on ({(E)-[3-(1H-imidazol-1-yl)-1-phenylpropylidene] amino}oxy)(4-nitrophenyl)methanone, an imidazole-bearing anti-Candida agent
- A Simplistic Preliminary Assessment of Ginstling-Brounstein Model for Solid Spherical Particles in the Context of a Diffusion-Controlled Synthesis
- M-Polynomials And Topological Indices Of Zigzag And Rhombic Benzenoid Systems
- Photochemical Transformation of some 3-benzyloxy-2-(benzo[b]thiophen-2-yl)-4Hchromen-4-ones: A Remote Substituent Effect
- Dynamic Changes of Secondary Metabolites and Antioxidant Activity of Ligustrum lucidum During Fruit Growth
- Studies on the flammability of polypropylene/ammonium polyphosphate and montmorillonite by using the cone calorimeter test
- DSC, FT-IR, NIR, NIR-PCA and NIR-ANOVA for determination of chemical stability of diuretic drugs: impact of excipients
- Antioxidant and Hepatoprotective Effects of Methanolic Extracts of Zilla spinosa and Hammada elegans Against Carbon Tetrachlorideinduced Hepatotoxicity in Rats
- Prunus cerasifera Ehrh. fabricated ZnO nano falcates and its photocatalytic and dose dependent in vitro bio-activity
- Organic biocides hosted in layered double hydroxides: enhancing antimicrobial activity
- Experimental study on the regulation of the cholinergic pathway in renal macrophages by microRNA-132 to alleviate inflammatory response
- Synthesis, characterization, in-vitro antimicrobial properties, molecular docking and DFT studies of 3-{(E)-[(4,6-dimethylpyrimidin-2-yl)imino]methyl} naphthalen-2-ol and Heteroleptic Mn(II), Co(II), Ni(II) and Zn(II) complexes
- M-Polynomials and Topological Indices of Dominating David Derived Networks
- Human Health Risk Assessment of Trace Metals in Surface Water Due to Leachate from the Municipal Dumpsite by Pollution Index: A Case Study from Ndawuse River, Abuja, Nigeria
- Analysis of Bowel Diseases from Blood Serum by Autofluorescence and Atomic Force Microscopy Techniques
- Hydrographic parameters and distribution of dissolved Cu, Ni, Zn and nutrients near Jeddah desalination plant
- Relationships between diatoms and environmental variables in industrial water biotopes of Trzuskawica S.A. (Poland)
- Optimum Conversion of Major Ginsenoside Rb1 to Minor Ginsenoside Rg3(S) by Pulsed Electric Field-Assisted Acid Hydrolysis Treatment
- Antioxidant, Anti-microbial Properties and Chemical Composition of Cumin Essential Oils Extracted by Three Methods
- Regulatory mechanism of ulinastatin on autophagy of macrophages and renal tubular epithelial cells
- Investigation of the sustained-release mechanism of hydroxypropyl methyl cellulose skeleton type Acipimox tablets
- Bio-accumulation of Polycyclic Aromatic Hydrocarbons in the Grey Mangrove (Avicennia marina) along Arabian Gulf, Saudi Coast
- Dynamic Change of Secondary Metabolites and spectrum-effect relationship of Malus halliana Koehne flowers during blooming
- Lipids constituents from Gardenia aqualla Stapf & Hutch
- Effect of using microwaves for catalysts preparation on the catalytic acetalization of glycerol with furfural to obtain fuel additives
- Effect of Humic Acid on the Degradation of Methylene Blue by Peroxymonosulfate
- Serum containing drugs of Gua Lou Xie Bai decoction (GLXB-D) can inhibit TGF-β1-Induced Epithelial to Mesenchymal Transition (EMT) in A549 Cells
- Antiulcer Activity of Different Extracts of Anvillea garcinii and Isolation of Two New Secondary Metabolites
- Analysis of Metabolites in Cabernet Sauvignon and Shiraz Dry Red Wines from Shanxi by 1H NMR Spectroscopy Combined with Pattern Recognition Analysis
- Can water temperature impact litter decomposition under pollution of copper and zinc mixture
- Released from ZrO2/SiO2 coating resveratrol inhibits senescence and oxidative stress of human adipose-derived stem cells (ASC)
- Validated thin-layer chromatographic method for alternative and simultaneous determination of two anti-gout agents in their fixed dose combinations
- Fast removal of pollutants from vehicle emissions during cold-start stage
- Review Article
- Catalytic activities of heterogeneous catalysts obtained by copolymerization of metal-containing 2-(acetoacetoxy)ethyl methacrylate
- Antibiotic Residue in the Aquatic Environment: Status in Africa
- Regular Articles
- Mercury fractionation in gypsum using temperature desorption and mass spectrometric detection
- Phytosynthetic Ag doped ZnO nanoparticles: Semiconducting green remediators
- Epithelial–Mesenchymal Transition Induced by SMAD4 Activation in Invasive Growth Hormone-Secreting Adenomas
- Physicochemical properties of stabilized sewage sludge admixtures by modified steel slag
- In Vitro Cytotoxic and Antiproliferative Activity of Cydonia oblonga flower petals, leaf and fruit pellet ethanolic extracts. Docking simulation of the active flavonoids on anti-apoptotic protein Bcl-2
- Synthesis and Characterization of Pd exchanged MMT Clay for Mizoroki-Heck Reaction
- A new selective, and sensitive method for the determination of lixivaptan, a vasopressin 2 (V2)-receptor antagonist, in mouse plasma and its application in a pharmacokinetic study
- Anti-EGFL7 antibodies inhibit rat prolactinoma MMQ cells proliferation and PRL secretion
- Density functional theory calculations, vibration spectral analysis and molecular docking of the antimicrobial agent 6-(1,3-benzodioxol-5-ylmethyl)-5-ethyl-2-{[2-(morpholin-4-yl)ethyl] sulfanyl}pyrimidin-4(3H)-one
- Effect of Nano Zeolite on the Transformation of Cadmium Speciation and Its Uptake by Tobacco in Cadmium-contaminated Soil
- Effects and Mechanisms of Jinniu Capsule on Methamphetamine-Induced Conditioned Place Preference in Rats
- Calculating the Degree-based Topological Indices of Dendrimers
- Efficient optimization and mineralization of UV absorbers: A comparative investigation with Fenton and UV/H2O2
- Metabolites of Tryptophane and Phenylalanine as Markers of Small Bowel Ischemia-Reperfusion Injury
- Adsorption and determination of polycyclic aromatic hydrocarbons in water through the aggregation of graphene oxide
- The role of NR2C2 in the prolactinomas
- Chromium removal from industrial wastewater using Phyllostachys pubescens biomass loaded Cu-S nanospheres
- Hydrotalcite Anchored Ruthenium Catalyst for CO2 Hydrogenation Reaction
- Preparation of Calcium Fluoride using Phosphogypsum by Orthogonal Experiment
- The mechanism of antibacterial activity of corylifolinin against three clinical bacteria from Psoralen corylifolia L
- 2-formyl-3,6-bis(hydroxymethyl)phenyl benzoate in Electrochemical Dry Cell
- Electro-photocatalytic degradation of amoxicillin using calcium titanate
- Effect of Malus halliana Koehne Polysaccharides on Functional Constipation
- Structural Properties and Nonlinear Optical Responses of Halogenated Compounds: A DFT Investigation on Molecular Modelling
- DMFDMA catalyzed synthesis of 2-((Dimethylamino)methylene)-3,4-dihydro-9-arylacridin-1(2H)-ones and their derivatives: in-vitro antifungal, antibacterial and antioxidant evaluations
- Production of Methanol as a Fuel Energy from CO2 Present in Polluted Seawater - A Photocatalytic Outlook
- Study of different extraction methods on finger print and fatty acid of raw beef fat using fourier transform infrared and gas chromatography-mass spectrometry
- Determination of trace fluoroquinolones in water solutions and in medicinal preparations by conventional and synchronous fluorescence spectrometry
- Extraction and determination of flavonoids in Carthamus tinctorius
- Therapeutic Application of Zinc and Vanadium Complexes against Diabetes Mellitus a Coronary Disease: A review
- Study of calcined eggshell as potential catalyst for biodiesel formation using used cooking oil
- Manganese oxalates - structure-based Insights
- Topological Indices of H-Naphtalenic Nanosheet
- Long-Term Dissolution of Glass Fibers in Water Described by Dissolving Cylinder Zero-Order Kinetic Model: Mass Loss and Radius Reduction
- Topological study of the para-line graphs of certain pentacene via topological indices
- A brief insight into the prediction of water vapor transmissibility in highly impermeable hybrid nanocomposites based on bromobutyl/epichlorohydrin rubber blends
- Comparative sulfite assay by voltammetry using Pt electrodes, photometry and titrimetry: Application to cider, vinegar and sugar analysis
- MicroRNA delivery mediated by PEGylated polyethylenimine for prostate cancer therapy
- Reversible Fluorescent Turn-on Sensors for Fe3+ based on a Receptor Composed of Tri-oxygen Atoms of Amide Groups in Water
- Sonocatalytic degradation of methyl orange in aqueous solution using Fe-doped TiO2 nanoparticles under mechanical agitation
- Hydrotalcite Anchored Ruthenium Catalyst for CO2 Hydrogenation Reaction
- Production and Analysis of Recycled Ammonium Perrhenate from CMSX-4 superalloys
- Topical Issue on Agriculture
- New phosphorus biofertilizers from renewable raw materials in the aspect of cadmium and lead contents in soil and plants
- Survey of content of cadmium, calcium, chromium, copper, iron, lead, magnesium, manganese, mercury, sodium and zinc in chamomile and green tea leaves by electrothermal or flame atomizer atomic absorption spectrometry
- Biogas digestate – benefits and risks for soil fertility and crop quality – an evaluation of grain maize response
- A numerical analysis of heat transfer in a cross-current heat exchanger with controlled and newly designed air flows
- Freshwater green macroalgae as a biosorbent of Cr(III) ions
- The main influencing factors of soil mechanical characteristics of the gravity erosion environment in the dry-hot valley of Jinsha river
- Free amino acids in Viola tricolor in relation to different habitat conditions
- The influence of filler amount on selected properties of new experimental resin dental composite
- Effect of poultry wastewater irrigation on nitrogen, phosphorus and carbon contents in farmland soil
- Response of spring wheat to NPK and S fertilization. The content and uptake of macronutrients and the value of ionic ratios
- The Effect of Macroalgal Extracts and Near Infrared Radiation on Germination of Soybean Seedlings: Preliminary Research Results
- Content of Zn, Cd and Pb in purple moor-grass in soils heavily contaminated with heavy metals around a zinc and lead ore tailing landfill
- Topical Issue on Research for Natural Bioactive Products
- Synthesis of (±)-3,4-dimethoxybenzyl-4-methyloctanoate as a novel internal standard for capsinoid determination by HPLC-ESI-MS/MS(QTOF)
- Repellent activity of monoterpenoid esters with neurotransmitter amino acids against yellow fever mosquito, Aedes aegypti
- Effect of Flammulina velutipes (golden needle mushroom, eno-kitake) polysaccharides on constipation
- Bioassay-directed fractionation of a blood coagulation factor Xa inhibitor, betulinic acid from Lycopus lucidus
- Antifungal and repellent activities of the essential oils from three aromatic herbs from western Himalaya
- Chemical composition and microbiological evaluation of essential oil from Hyssopus officinalis L. with white and pink flowers
- Bioassay-guided isolation and identification of Aedes aegypti larvicidal and biting deterrent compounds from Veratrum lobelianum
- α-Terpineol, a natural monoterpene: A review of its biological properties
- Utility of essential oils for development of host-based lures for Xyleborus glabratus (Coleoptera: Curculionidae: Scolytinae), vector of laurel wilt
- Phenolic composition and antioxidant potential of different organs of Kazakh Crataegus almaatensis Pojark: A comparison with the European Crataegus oxyacantha L. flowers
- Isolation of eudesmane type sesquiterpene ketone from Prangos heyniae H.Duman & M.F.Watson essential oil and mosquitocidal activity of the essential oils
- Comparative analysis of the polyphenols profiles and the antioxidant and cytotoxicity properties of various blue honeysuckle varieties
- Special Issue on ICCESEN 2017
- Modelling world energy security data from multinomial distribution by generalized linear model under different cumulative link functions
- Pine Cone and Boron Compounds Effect as Reinforcement on Mechanical and Flammability Properties of Polyester Composites
- Artificial Neural Network Modelling for Prediction of SNR Effected by Probe Properties on Ultrasonic Inspection of Austenitic Stainless Steel Weldments
- Calculation and 3D analyses of ERR in the band crack front contained in a rectangular plate made of multilayered material
- Improvement of fuel properties of biodiesel with bioadditive ethyl levulinate
- Properties of AlSi9Cu3 metal matrix micro and nano composites produced via stir casting
- Investigation of Antibacterial Properties of Ag Doped TiO2 Nanofibers Prepared by Electrospinning Process
- Modeling of Total Phenolic contents in Various Tea samples by Experimental Design Methods
- Nickel doping effect on the structural and optical properties of indium sulfide thin films by SILAR
- The effect mechanism of Ginnalin A as a homeopathic agent on various cancer cell lines
- Excitation functions of proton induced reactions of some radioisotopes used in medicine
- Oxide ionic conductivity and microstructures of Pr and Sm co-doped CeO2-based systems
- Rapid Synthesis of Metallic Reinforced in Situ Intermetallic Composites in Ti-Al-Nb System via Resistive Sintering
- Oxidation Behavior of NiCr/YSZ Thermal Barrier Coatings (TBCs)
- Clustering Analysis of Normal Strength Concretes Produced with Different Aggregate Types
- Magnetic Nano-Sized Solid Acid Catalyst Bearing Sulfonic Acid Groups for Biodiesel Synthesis
- The biological activities of Arabis alpina L. subsp. brevifolia (DC.) Cullen against food pathogens
- Humidity properties of Schiff base polymers
- Free Vibration Analysis of Fiber Metal Laminated Straight Beam
- Comparative study of in vitro antioxidant, acetylcholinesterase and butyrylcholinesterase activity of alfalfa (Medicago sativa L.) collected during different growth stages
- Isothermal Oxidation Behavior of Gadolinium Zirconate (Gd2Zr2O7) Thermal Barrier Coatings (TBCs) produced by Electron Beam Physical Vapor Deposition (EB-PVD) technique
- Optimization of Adsorption Parameters for Ultra-Fine Calcite Using a Box-Behnken Experimental Design
- The Microstructural Investigation of Vermiculite-Infiltrated Electron Beam Physical Vapor Deposition Thermal Barrier Coatings
- Modelling Porosity Permeability of Ceramic Tiles using Fuzzy Taguchi Method
- Experimental and theoretical study of a novel naphthoquinone Schiff base
- Physicochemical properties of heat treated sille stone for ceramic industry
- Sand Dune Characterization for Preparing Metallurgical Grade Silicon
- Catalytic Applications of Large Pore Sulfonic Acid-Functionalized SBA-15 Mesoporous Silica for Esterification
- One-photon Absorption Characterizations, Dipole Polarizabilities and Second Hyperpolarizabilities of Chlorophyll a and Crocin
- The Optical and Crystallite Characterization of Bilayer TiO2 Films Coated on Different ITO layers
- Topical Issue on Bond Activation
- Metal-mediated reactions towards the synthesis of a novel deaminolysed bisurea, dicarbamolyamine
- The structure of ortho-(trifluoromethyl)phenol in comparison to its homologues – A combined experimental and theoretical study
- Heterogeneous catalysis with encapsulated haem and other synthetic porphyrins: Harnessing the power of porphyrins for oxidation reactions
- Recent Advances on Mechanistic Studies on C–H Activation Catalyzed by Base Metals
- Reactions of the organoplatinum complex [Pt(cod) (neoSi)Cl] (neoSi = trimethylsilylmethyl) with the non-coordinating anions SbF6– and BPh4–
- Erratum
- Investigation on Two Compounds of O, O’-dithiophosphate Derivatives as Corrosion Inhibitors for Q235 Steel in Hydrochloric Acid Solution

