Abstract
Silver sulfide (Ag2S) nanomaterials are important chalcogenides and have potential applications in photovoltaic cells, thermoelectric sensors, solar cell batteries, and infrared detectors. However, the biological properties of green synthesized silver sulfide nanoparticles (Ag2S-NPs) have been rarely explored as compared to the extensively studied silver nanoparticles (AgNPs). The current study thus investigates and compares Fagonia arabica synthesized Ag2S-NPs and AgNPs for multiple biological properties. Biosynthesis of the NPs is achieved using an aqueous extract of F. arabica, whereas physicochemical, structural, morphological, and optical characterization of the materials was undertaken using UV–Vis spectroscopy, Fourier transform infrared spectroscopy, X-ray diffractometer, scanning electron microscopy, energy dispersive spectroscopy, photoluminescence, and pH-responsive dispersion studies. Our result reveals that both the NPs exhibit excellent biological properties, yet Ag2S-NPs exhibit relatively enhanced antioxidant and in vitro anticancer potential against MCF-7 cells. However, AgNPs possess comparatively better antibacterial, antileishmanial, and antifungal properties. Most importantly, both the synthesized NPs are exceptionally well compatible with human blood cells, ensuring their safe and non-toxic properties. Being highly potent and compatible antimicrobial and anticancer materials, the F. arabica synthesized G-Ag2S-NPs and G-AgNPs can be further explored in in vivo studies.
Graphical abstract

1 Introduction
Among the semiconductors, silver sulfide (Ag2S) holds a prominent position, along with lead, zinc, copper, cadmium, and mercury sulfides [1,2]. The in-depth exploration of nanostructured Ag2S stems from its promising roles in optoelectronics, bio-sensors, and catalysis [1,2]. In the past decade, extensive efforts were made in the synthesis of metallic nanoparticles (MNPs) for bio-imaging. Silver sulfide nanoparticles (Ag2S-NPs) with near-infrared optical properties have been investigated for fluorescence imaging and photo-thermal therapy. Notably, silver demonstrates superior contrast to iodine in dual-energy mammography and provides contrast for computed tomography [3].
Various methods, including hydro-chemical bath deposition, template synthesis, sol–gel technique, synthesis in micro-emulsions, sonochemical, hydrothermal, solvothermal, electrochemical, and microwave-assisted methods, are utilized for the synthesis of nanostructured Ag2S. Each method presents its advantages and drawbacks [4]. A physical method such as spray pyrolysis, lithography, and many other techniques offers multiple advantages as the methods are quick, less toxic, and use radiations as reducing agents; however, there are some drawbacks associated with such processes, including higher energy consumption, low yield, and contamination of solvents [5,6,7,8]. Similarly, chemical methods include chemical reduction, sol–gel, and micro-emulsion synthesis, to name a few [7]. Like physical methods, chemical techniques also offer some advantages as the processes are cheap, easy to carry out, and result in high yield and uniformity of the NPs, while drawbacks of using chemical methods are the high costs of chemical reagents, hazard associated with synthesis and toxic contamination to the environment [9].
Unlike physical and chemical synthesis, green synthesis involves the production of NPs using various types of biomass sources, including plants, bacteria, fungi, and algae or their extracts [7,9,10]. The plant-based synthesis or phytomediated synthesis of the NPs is particularly advantageous because it minimizes the risk of contamination from chemical reagents. Furthermore, plant extracts can be obtained at a low cost, are easily available, and the synthesis procedure is relatively simple, eco-friendly, and non-hazardous [11].
Fagonia arabica an important medicinal herb also known as Shawka al-Baidaa or Shukaaa in Arabic, Suchi booti in Urdu [12], belongs to the genus Fagonia which is the largest group of angiosperms [13]. Fagonia species are localized to the warm, dry regions of South Asia (India and Pakistan), Middle East, Central Europe, North Africa, and some regions of North America [14]. The species have been used as anticancer, analgesic, antioxidant, and preventing agents against smallpox. The plants are also used to cure fever, asthma, urine discharge, and renal problems [15]. Various alkaloids, glycosides, saponins, and triterpenes have been claimed to be found in the plant. Such rich therapeutic properties make F. arabica as an excellent candidate for the pharmacologically potent NPs [16,17,18].
In the current study, F. arabica has been explored for the synthesis of Ag2S and silver nanoparticles (AgNPs). To the best of our knowledge, Ag2S has been rarely explored for multifunctional biological properties, especially green synthesis, as compared to conventionally highly explored AgNPs. Thus, in the current study, an effort has been made to synthesize, characterize, and compare Ag2S NPs with AgNPs.
The facile, non-toxic, green synthesis was carried out using a simple aqueous extract of the herb. A comprehensive characterization analysis was carried out using various techniques such as UV, X-ray diffractometer (XRD), Fourier transform infrared spectroscopy (FTIR), scanning electron microscopy (SEM), energy dispersive X-ray (EDX), photoluminescence (PL), pH-responsive dispersion, and comparative yield studies. The NPs were investigated for detailed biological properties including antioxidant, antibacterial, antifungal, anticancer, and biocompatibility studies.
2 Experimental
2.1 Chemicals and reagents
Various chemicals and reagents were utilized according to standard laboratory protocols and procedures. Silver nitrate (AgNO3), sodium sulfide nonahydrate (Na2S·9H2O), dimethyl sulfoxide (DMSO), phosphate buffer saline (PBS), and 2,2-diphenylpicrylhydrazyl (DPPH) were procured from Sigma Aldrich. Nutrient agar, a standard antibacterial drug (Levofloxacin) and antifungal drug (Clotrimazole), was also obtained from Sigma Aldrich. Freshly prepared distilled water was used in the experiments when required.
2.2 Green synthesis of silver and Ag2S NPs
2.2.1 Plant collection, processing, and extract preparation
F. arabica was collected from Sakhi Sarwar (29.9802°N, 70.3063°E), which is located in the district of Dera Ghazi Khan-Punjab, Pakistan, as illustrated in Figure 1. The collected herb went through detailed identification via comprehensive physical examination, existing literature, and digital identification using Plant-Net software. After rinsing and washing the plant biomass with distilled water, it was cut into small pieces and then shade-dried for weeks. The well-dried herb was then ground into a fine powder and was utilized for aqueous extract preparation. To prepare the 10% aqueous extract, 10 g of the plant powder was taken in an Erlenmeyer flask and suspended in 100 mL of distilled water. After that, the mixture was continuously stirred with a magnetic stirrer with a hot plate at 55°C for 2 h. After stirring, the mixture was transferred into a shaking incubator for a 12–16 h period, followed by filtration. The freshly prepared aqueous extract was later used for the synthesis of NPs.

Graphical illustration of the geographical location (Dera Ghazi Khan-Pakistan) and collection point of F. arabica.
2.2.2 Synthesis of AgNPs (G-AgNPs)
Aqueous extract of F. arabica was utilized to synthesize Ag-NPs and Ag2S-NPs. In the experiment, a solution of AgNO3 at a molar concentration of 0.125 M was prepared by mixing 1.06 g of the precursor salt in 100 mL of distilled water. The solution was stirred on a magnetic stirrer with a hot plate until the temperature reached 55°C. The prepared plant extract (100 mL) was then added dropwise using a burette, and the reaction mixture was further preceded for 2 h. The resulting mixture was then collected and washed with distilled water thrice using centrifugation at 4,000 rpm for 8 min. The obtained material was then poured into a sterile clean petri plate and dried in an oven overnight at 80°C. The dried material was then collected from a petri plate, ground using a pestle and mortar, stored in a clean Eppendorf tube, and labeled as G-Ag-NPs.
2.2.3 Synthesis of Ag2S NPs (G-Ag2S-NPs)
The synthesis of Ag2S-NPs also followed a similar procedure with Na2S·9H2O as a sulfur source. In the experiment, 0.25 M Na2S·9H2O was prepared by adding 1.50 g into 100 mL of distilled water and kept on the magnetic stirrer until it dissolved completely. In a separate flask, 0.0625 M of AgNO3 was prepared by taking 1.06 g of the precursor salt in 100 mL of distilled water and was kept on a magnetic stirrer for 20 min to ensure complete dissolution. Following that, the prepared Na2S⸱9H2O solution was then added to the AgNO3 solution dropwise. After thorough mixing, the mixture temperature was taken to 55°C. When the obtained temperature was achieved, 200 mL of the aqueous extract of F. arabica was added to the reaction mixture dropwise. The mixture was further stirred at 55°C for 2 h. After reaction completion, the mixture was cooled down at room temperature, and the synthesized material was collected and washed thrice via centrifugation at 4,000 rpm for 8 min. The washed material was then oven-dried and collected, ground into fine powder in a mortar and pestle, stored in an Eppendorf tube, and labeled as G-Ag2S-NPs. The biosynthesis procedure was carried out multiple times and resulted in uniform yield with high reproducibility at the mentioned conditions. However, the production can be limited as the plant source is only native to South Asia, Middle East, Central Europe, North Africa, and some regions of North America [12,13,14,19].
2.3 Physicochemical, optical, and morphological characterization
A comprehensive characterization analysis was carried out using various techniques such as UV–Vis spectroscopy, XRD, FTIR, SEM, energy dispersive spectroscopy (EDS), and PL. Furthermore, comparative yield and pH-responsive dispersion analysis of the NPs were also carried out. UV–Vis analysis of the synthesized NPs was conducted to affirm the synthesis of NPs within the spectral range of 200–780 nm. To evaluate and compare the yield of NPs, the following formula was utilized for yield recovery:
where W stands for the weight of the added salt and w stands for the weight of the produced NPs. The prepared samples were then subjected to X-ray diffraction XRD analysis utilizing an XRD. Scanning of the samples was performed at room temperature over a 2θ diffraction angle range of 10°–80°, with a scanning speed of 0.4°·min−1. The widely used Scherer equation was then taken into account to determine the crystal size [20]. Functional group analysis of the NPs was carried out using FTIR spectroscopy on a Nexus Thermo FTIR spectrophotometer. Dried samples were mixed with KBr in a ratio of 1:200 (w/w) and pressed into pellets under vacuum, and the FTIR spectra were obtained in the transmittance mode within the wavenumber range of 4,000 to 500 cm−1. PL experiments were also conducted at room temperature using a Hitachi F-4500 FL spectrophotometer equipped with a Xenon lamp as the excitation source [5]. The surface features, morphology, and dimensions of the NPs were examined using a SEM. Before observation, a thin gold layer was applied to the samples using an ion sputter coater with a controlled deposition rate to enhance image quality. Images captured at various magnifications provided detailed information about surface morphologies and particle sizes. EDX analysis was also utilized to comprehensively analyze the elemental composition of the samples.
2.4 pH-responsive dispersion studies
The dispersion stability of the NPs in distilled water at different pH levels (2, 7, and 12) was assessed, highlighting the dispersion behavior of the prepared materials as a function of the storage time. In a glass vial (each 15 mL), 0.5 mg·mL−1 of the prepared NPs were suspended in an equivalent solvent, followed by sonication for 10–15 min. The dispersion behavior was observed visually, and images were captured immediately after sonication and at various time intervals thereafter.
2.5 Biological properties of the synthesized green materials
2.5.1 Antioxidant assays
2.5.1.1 DPPH-free radical scavenging assay (DPPH-FRSA)
The DPPH FRSA of the prepared NPs was assessed following a well-optimized literature-reported protocol [21,22]. The bioassay was utilized with various concentrations of NPs ranging from 12.5 to 400 µL. The evaluation involved observing the discoloration of the purple DPPH solution. Briefly, in a 96-well plate, 10 µL of the sample was added, mixed with 190 µL of DPPH reagent, and incubated in darkness for 1 h at 37°C. Absorbance at 525 nm was then measured using a microplate reader. FRSA, expressed as percent inhibition, was calculated using a provided equation [23]:
where “Abs sample” indicates the absorbance of the test sample and “Abs control” indicates the absorbance of the DPPH solution without the test sample.
2.5.1.2 Phosphomolybdenum-based total antioxidant capacity (TAC) assay
The prepared NPs were evaluated for TAC by using a phospho-molybdenum-based analysis technique according to the previously reported protocol in the literature with some modifications [24,25]. Briefly, 100 µL of the sample was transferred into an Eppendorf tube using a micropipette. Subsequently, it was mixed with 900 µL of TAC reagent, containing 0.6 M H2SO4, 28 mM Na3PO4, and 4 mM (NH4)2MoO4. The reaction mixture underwent incubation in a water bath for 45 min at a temperature of 80°C. Following incubation, the mixture was allowed to cool. Subsequently, the absorbance was carefully measured at a wavelength of 630 nm utilizing a microplate reader. The TAC was then quantified and expressed in micrograms of ascorbic acid equivalent per milligram of the dried NPs weight (µg AAE/mg).
2.5.1.3 Ferric-reducing potential assay (FRAP)
The prepared NPs were analyzed for total reducing power, which was carried out by using a ferric reducing analysis technique with a well-optimized reported protocol in the literature with some modifications [26,27,28]. About 100 µL of the sample aliquot, 400 µL phosphate buffer (pH 6.6), and 100 µL of 1% w/v K3[Fe(CN)6] were combined in an Eppendorf tube. The reaction mixture then underwent 30 min incubation in a water bath at 65°C. Following this, 200 µL of 10% w/v C2HCl3O2 was gently added, and the reaction mixture was centrifuged at 4,000 rpm for 12 min. Subsequently, 140 µL of the supernatant was carefully transferred to a well in a 96-well plate containing 60 µL of 0.1% w/v C3FeN3 solution. The absorbance was quantified at a wavelength of 630 nm using a microplate reader. The total reducing power was determined by calculating the micrograms of ascorbic acid equivalent per milligram of dried extract weight, expressed as µg AAE/mg.
2.5.2 Antimicrobial studies
2.5.2.1 Antibacterial assay against Gram-positive and Gram-negative strains and Methicillin-resistant Staphylococcus aureus (MRSA)
The in vitro antibacterial efficacy of the synthesized NPs was assessed using the agar well diffusion method, following a well-optimized protocol against three negative strains, i.e., Escherichia coli, Klebsiella pneumoniae, and Salmonella enterica, while four positive strains, i.e., Staphylococcus epidermidis, Staphylococcus aureus, Bacillus subtilis, and MRSA [29]. The seeding density of the tested isolates was adjusted to 1 × 106 CFU·mL−1 as per protocol standards. To create a bacterial turf, 100 μL of refreshed stock cultures were applied to solidified nutrient agar plates, and sterile wells were carefully made and labeled. Subsequently, 80 mL of the sample was poured into the corresponding well, with 15 mL of the standard antibiotic Levofloxacin (5 mg·mL−1) serving as the positive control, and 80 µL of DMSO as the negative control. Petri plates were then incubated for 20 h at 37°C. Following incubation, the antibacterial potential of various test samples was evaluated by measuring the zone of inhibition (ZOI) in millimeters. Samples that exhibited >10 mm ZOI were further examined for MIC using the micro broth dilution technique. Serial dilutions of the samples were prepared in a sterile nutrient broth, and selected bacterial culture was grown in nutrient broth with a seeding density of 5 × 104 CFU·mL−1. Following on, 20 µL of the tested dilution (with a final concentration of 2,500 to 39 µg·mL−1) and 180 µL of the microbial culture were smoothly in the respective well of a 96-well plate followed by incubation at 37°C for 18 h. After careful observation of the visible growth, the lowest concentration tested at which the NPs demonstrated visible and eminent growth inhibition of the strain was labeled as MIC of the test sample.
2.5.2.2 Antifungal assay against the selected spore-forming fungal strains
The agar well diffusion method was used to evaluate the antifungal activity of samples with some modifications to the protocol reported previously against four strains, i.e., Aspergillus flavus, Aspergillus fumigatus, Trichophyton rubrum, and Aspergillus terreus [30]. Fungal strains were grown in tested tubes (15 mL) containing Tween 20 solution (0.02% v/v). From each culture, 100 µL of the inoculum was taken and poured onto the sterilized SDA media-filled petri plates. Wells were carefully made and labeled accordingly using a sterile blue tip. Next, 90 mL of the sample was carefully poured into the corresponding well from the serial dilution of the test sample. Clotrimazole (20 µL of 5 mg·mL−1) served as a positive control, whereas 90 µL of DMSO was employed as the negative control. The plates were then incubated at 28°C for 34–48 h to facilitate fungal growth. The loaded wells carrying the tested materials and the control groups were examined after incubation to see if a ZOI had formed. A measurement scale was utilized to measure the ZOI’s circumference to the closest millimeter.
2.5.2.3 Antileishmanial assay against the promastigote and amastigote forms of the parasite (Leishmnia tropica)
The efficacy of the substances against L. tropica (KWH23) was assessed for both the promastigote and amastigote stages of the parasite. Utilizing established procedures, both forms of the parasite were cultured and sustained in specialized MI99 media containing 10% fetal bovine serum (FBS) for 12 h. Subsequently, minute quantities of the refreshed cultures and samples within the concentration range of up to 200 µg·mL−1 were cautiously combined in labeled wells of a 96-well plate. This incubation went for a 72-h incubation at 28 ± 2°C. DMSO served as the negative control. Conversely, Amphotericin-B acted as the positive control, exhibiting known leishmanicidal properties. The blank, consisting of media alone, provided a baseline for comparison. After incubation for 72 h at 28 ± 2°C, a small volume of 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) solution (5 mg·mL−1 DW) was introduced to the wells, followed by a 4-h re-incubation at the same temperature. This step allowed the viable cells to metabolize the MTT solution, resulting in the formation of a colored product. The activity was quantitatively gauged as a percentage of inhibition by measuring the absorbance at 560 nm, applying the formula
where Abs signifies the sample’s absorbance, and Abc corresponds to the absorbance of the negative control.
2.5.3 In vitro anticancer assay against MCF-7 cell line
To carry out the anticancer activity of the NPs, MCF-7 cells were cultured in a suitable medium containing 10% FBS and 1% penicillin–streptomycin. The cells were incubated at 37°C in a 5% CO2 humidified environment until they achieved a confluence of 70–80%. Varying concentrations of the NPs were prepared in the culture medium, spanning from low to high concentrations. Then, the prepared NP dilutions were applied to the MCF-7 cells and allowed for an incubation period lasting 72 h. In the assay, untreated cells, i.e., cells without being treated with test samples, were considered control. After the incubation period, the existing medium was removed and replaced with a fresh one. MTT reagents were introduced at a concentration of 0.5 mg·mL−1 to each well, incubating for 4 h at 37°C to facilitate formazan crystal formation. MTT solution was discarded, and DMSO was added to dissolve the formazan crystals. Finally, a microplate reader was utilized to measure absorbance at 570 nm, with a reference wavelength set at 690 nm for background absorbance correction, and the percentage of viability was calculated using the provided formula.
While % cytotoxicity was calculated as
2.5.4 In vitro biocompatibility assay against isolated blood cells
To determine the blood compatibility, ethylenediaminetetraacetic acid (EDTA) tubes were used to collect 2 mL of human blood after obtaining informed permission and following all applicable ethical guidelines as reported earlier [31,32]. Red blood cells (RBCs) from the centrifuged blood samples were subsequently washed thrice and re-suspended in PBS. After that, an EDTA solution of 2% was arranged and was added to the suspension to prevent the RBCs from coagulation. The preparation of the NPs involved dilutions ranging from 0.625 mg to 5 mg·mL−1 in distilled water and co-incubated with RBCs suspension in 1:1 for 1 h in an Eppendorf tube. Following this, microscopic slides were prepared by adding a considerable drop (20 µL) to the slide. To help RBCs and NPs interact, the slides were incubated at 37°C for 1 h. The samples were then examined under a high-magnification microscope to gauge the level of hemolysis. RBCs combined with dH2O served as the negative control, and RBCs treated with Triton X-100 served as the positive control. The slides were then observed at 1,000× magnification, and light micrographs were made.
2.5.5 Statistical analysis
Data from the antibacterial, anti-leishmanial, and cytotoxicity assays were evaluated using two-way analysis of variance (ANOVA) to examine the interactions between different treatment groups. The analyses were conducted using GraphPad Prism 9 (2022). Following the two-way ANOVA, a post hoc multiple comparison test was carried out to identify specific differences between group means. A p-value of less than 0.05 was considered statistically significant.
3 Results and discussion
3.1 Plausible mechanism of green synthesis of Ag2S-NPs and AgNPs
The aqueous extract of F. arabica worked as a reducing and stabilizing agent for the synthesis of green AgNPs and Ag2S-NPs. Numerous phytochemicals and secondary metabolites have been reported in the genera Fagonia, for instance, alkaloids, glycosides, saponins, carbohydrates, flavonoids, steroids, terpenoids, anthraquinones, and triterpenes, which have been associated with a range of pharmacological activities such as anticancer, antimicrobial, antiviral, analgesic, anti-inflammatory, antipyretic, and antioxidant properties. During synthesis, when the freshly prepared aqueous extract was added to the transparent solution of AgNO3, an obvious brown color change could be observed, which was more intense in G-Ag2S-NPs that testified to the formation of NPs. The change in color has been attributed to the reduction of salt solution after the addition of plant extract that acts as a reducing agent. As F. arabica contains abundant polyphenols, including but not limited to linoleic acid, aspartic acid, kaempferol-7-O-rhamnoside, d-pinitol, hederagenin, and ursolic acid, to name a few [7,13,15,33,34,35]. These metabolites not only served as reducing agents for the precursor salt but also functioned in stabilizing and capping the NPs at the nanometric scale. The plausible biosynthesis phenomenon is visually depicted in Figure 2.
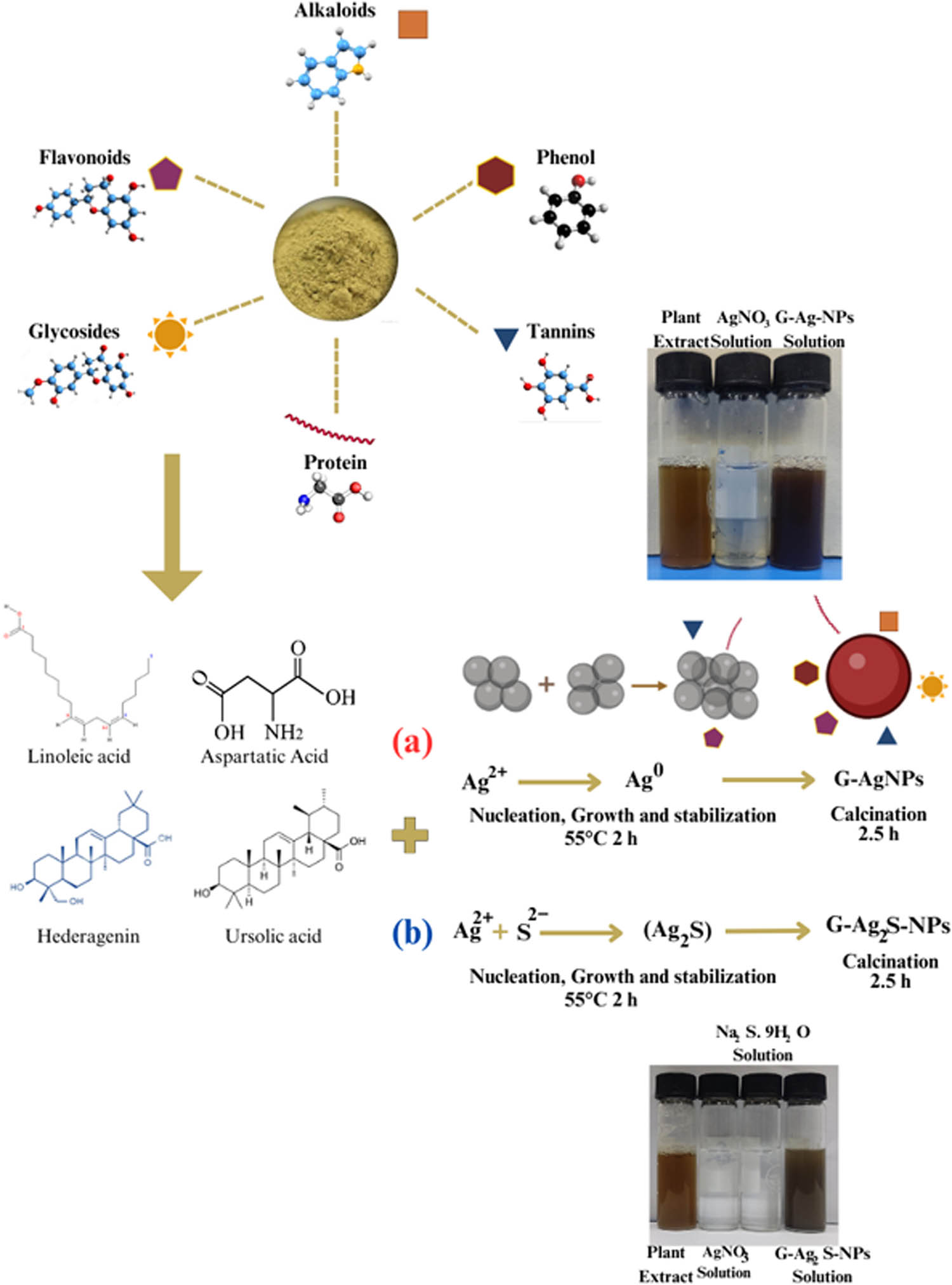
Graphical representation of the potential mechanism involved in the F. arabica mediated synthesis of (a) G-AgNPs and (b) G-Ag2S-NPs.
3.2 Physicochemical, optical, and morphological characterizations
3.2.1 UV–Vis spectroscopy
The F. arabica synthesized Ag2S-NPs and AgNPs were initially characterized using UV–Vis absorption spectra, as displayed in Figure 3. The UV–Vis spectra revealed broad surface plasma resonance (SPR) peaks between 350 and 490 nm [36]. The presence of a distinct SPR peak at 429 nm validates the formation of AgNPs, and a peak at 450 nm validates Ag2SNPs. Thus, the phytochemical constituents in the F. arabica extract, serving as bio-capping and bio-reducing agents, contributed to the successful synthesis and stabilization of both the NPs [36,37,38,39,40].
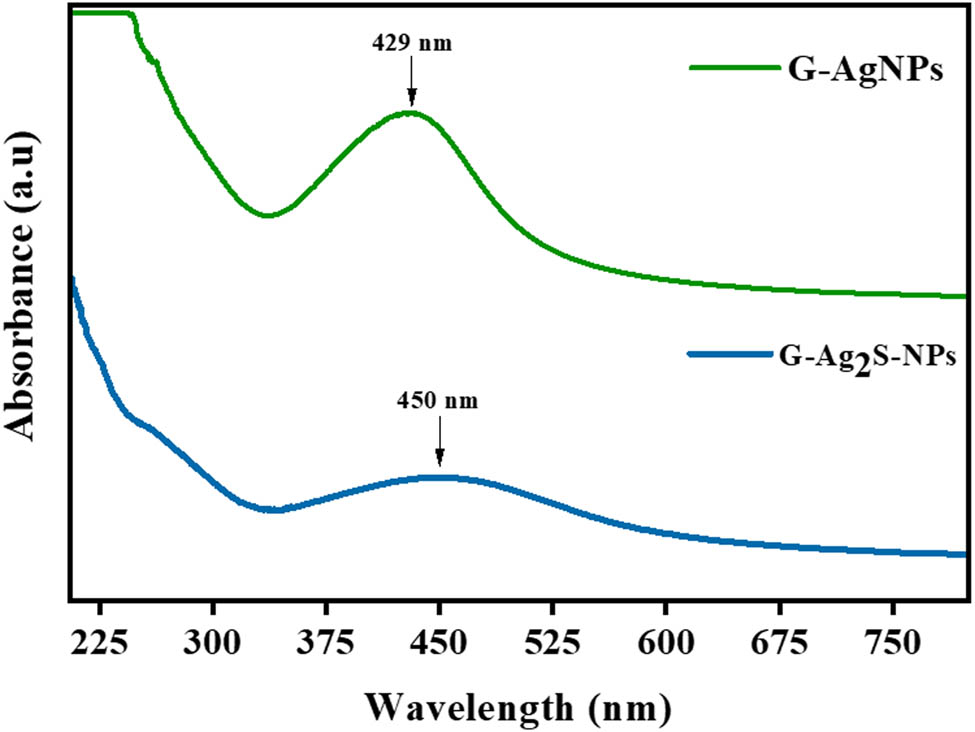
UV–Vis spectral patterns of F. arabica synthesized G-AgNPs and G-Ag2S-NPs.
3.2.2 % Yield recovery of the synthesized NPs
The yield of the NPs is affected by various factors including the synthesis route, concentration and the nature of the reducing and stabilizing agent, the nature of synthesized NPs, the precursor used, i.e. initial salt whose reduction takes place, and the physical and chemical conditions of the reaction, e.g. pH, temperature, pressure, catalyst presence, and the reaction time [41]. In our study, at the given reaction conditions (Table 1), F. arabica resulted in a higher yield of Ag2S-NPs, i.e. 43.86% as compared to the AgNPs, which was measured as 34.90%.
Percent yield (%) recovery of G-Ag-NPs and Ag2S-NPs after drying
| Samples | Precursor salt | Temp (°C) | Time (h) | Average yield (g) | (%) Yield |
|---|---|---|---|---|---|
| Ag-NPs | AgNO3 | 55 | 2 | 0.36 | 34.90 |
| Ag2S-NPs | AgNO3 + Na2S·9H2O | 55 | 2 | 0.93 | 43.86 |
3.2.3 X-ray diffraction analysis
XRD analysis of F. arabica synthesized Ag2S and AgNPs was performed to determine the phase structure, crystallite shape, and crystallite size, as presented in Figure 4. The XRD pattern of the green synthesized AgNPs exhibited four distinct diffraction peaks with 2θ degrees of 38.18°, 46.19°, 64.85°, and 78.07° were observed and compared with the JCPDS#04-0783. The peaks corresponded to the crystal planes of (111), (200), (220), and (311), confirming the face-centered cubic crystallite shape of G-AgNPs [42]. Furthermore, the average calculated crystallite size of green synthesized AgNPs was determined as 57.21 nm using Debye–Scherer’s equation. Some additional peaks are marked (*) as were observed, which might be possible because of the presence of the organic components in the plant extract, which capped the AgNPs. Similar observations have also been reported by other researchers [43]. As compared to AgNPs, the F. arabica synthesized Ag2S NPs displayed diffraction peaks at 2θ 25.49°, 28.39°, 34.45°, 38.27°, 44.47°, 57.72°, 64.63°, and 77.51° corresponding to the (111), (112), (121), (031), (210), (−223), (−134), and (311) crystalline planes of Ag2S, which were indexed to the JCPD#00-011-0688. The diffraction peaks confirmed the monoclinic shape of the crystallites. Furthermore, the average calculated size of the crystallites for G-Ag2S-NPs was noted as 41.82 nm [44,45,46,47,48,49,50]. Some extra peaks were also observed in the diffraction pattern of Ag2S NPs, marked as (*), indicating the presence of capped plant material (extract), which may have provided a layer or covering over synthesized NPs.
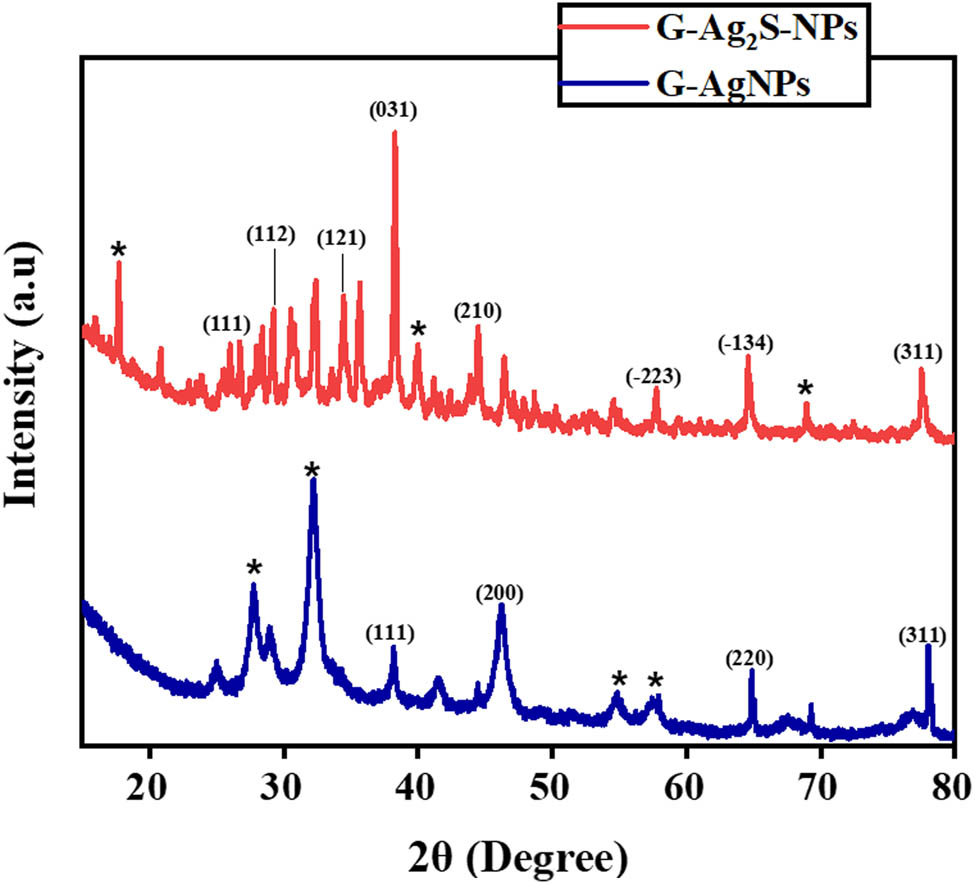
X-ray diffraction patterns of F. arabica synthesized G-AgNPs and G-Ag2S-NP.
3.2.4 FTIR analysis
FTIR analysis enables the identification of functional groups in a sample through their distinctive stretching and bending vibrations. The FTIR spectra for the F. arabica synthesized AgNPs and Ag2S-NPs were recorded in the spectral range of 4,000 to 500 cm−1, as displayed in Figure 5. In the FTIR spectra of G-AgNPs, significant peaks were observed at 566, 667.64, 1,054.47, 1,221.8, 1,384.69, 1,513.79, 1,643.03, and 2,325.53 cm−1. The distinctive peaks at 566.1 and 667 cm−1 confirm the presence of C–H bending due to the presence of carbohydrates in the F. arabica extract [51]. The peak at 1,054 cm−1 corresponds to C–O stretching, which is due to the alkaloid presence in the pant. The peak at 1,221 cm−1 corresponds to the C–C (═O)–O stretching of ester due to alkaloids, glycosides, saponins, and triterpenes present within the aqueous extract [52]. The presence of a peak at 2,325 cm−1 confirmed the stretching vibration associated with the C═N functional group found in nitrile compounds, which is due to the glycoside presence [53]. Similarly, FTIR spectra of G-Ag2S-NPs have shown prominent peaks at 671, 1,038.73, and 1,514.2 cm−1. For the fingerprint, the strong peak at 671 cm−1 represents the Ag–S elongation and deformation [54]. The bands at 1,038 cm−1 signify S═O stretch indicating sulfonamide and sulfoxide which is due to the sulfate esters present in plant extract [55]. The C═C–C aromatic ring, which is the functional group of aromatic compounds, exhibits a peak at 1,514 cm−1 due to the presence of polyphenols [53]. These particular groups serve a crucial role as stabilizing and capping agents during the synthesis of G-Ag2S-NPs that were present abundantly within the plant extract.

FTIR spectra of F. arabica synthesized G-AgNPs and G-Ag2S-NPs.
3.2.5 Morphological and elemental analysis
Surface morphology and size of the G-AgNPs and G-Ag2S-NPs were determined by analyzing the SEM micrographs, as illustrated in Figure 6a and b. The micrographs of G-AgNPs clearly show the mix of spherical and elliptical shaped features of G-AgNPs with a mean size of 81 ± 9 nm. At the same time, G-Ag2S-NP exhibited a mix of cubic and irregular morphology, while the average size of 92 ± 4 nm was measured using ImageJ. The elemental analysis of G-AgNPs for the presence of Ag and C in the NPs was analyzed by EDX. The EDX analysis (Figures 6c and 6d) indicated the atomic proportion of Ag, C, and O with 22.64%, 32.34%, and 45.02%, respectively. The mass proportion of Ag, C, and O was observed as 44.01%, 23.79%, and 32.20%, respectively. The elemental analysis showed the presence of C and O, which indicated the intense coating of the plant extract on the surface of NPs [56]. Similarly, elemental analysis of G-Ag2S-NPs for the presence of Ag and S confirms the formation of respective NPs. EDX spectra indicate the atomic proportion of Ag, S, C, O, Cl, and Ca with 23.92%, 10.38%, 30.48%, 31.54%, 1.39%, and 2.29%, respectively. In contrast, the mass proportion of Ag, S, C, O, Cl, and Ca was recorded as 58.83%, 11.75%, 10.25%, 14.93%, 2.44%, and 1.80%, respectively, in the samples. The elemental analysis of Ag2S NPs also showed the presence of C and O and very minute amounts of Cl and Ca, which may be due to the coating of plant extract and trace impurities in the sodium sulfide Na2S⸱9H2O, which was utilized as the sulfur source in the synthesis method [57].
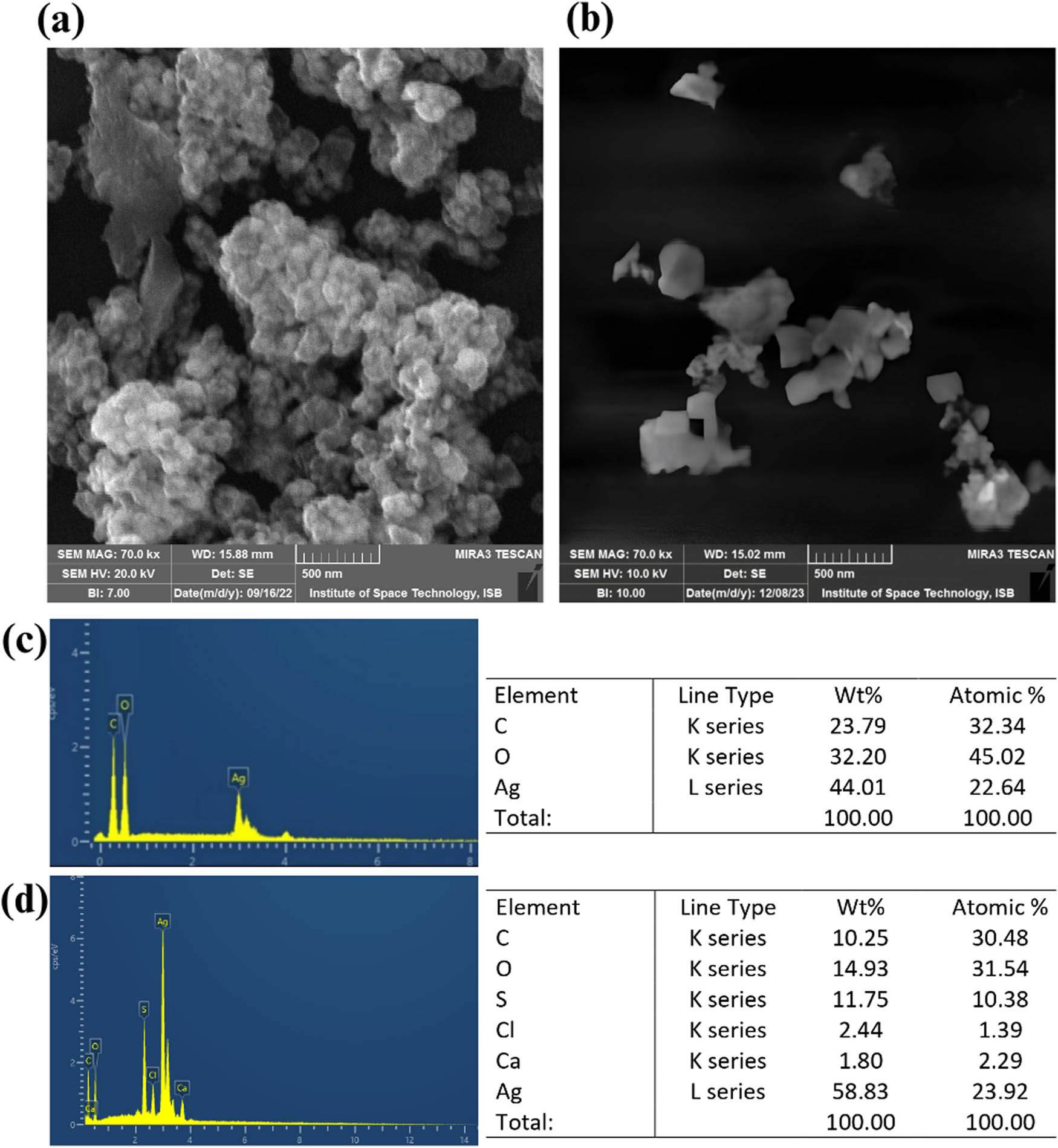
SEM micrograph of F. arabica (a) G-AgNPs and (b) G-Ag2S-NPs and EDX analysis of (c) G-AgNPs and (d) G-Ag2S-NPs.
3.2.6 pH-responsive dispersion behavior
Dispersion behavior of the nanoscale particles is affected by various factors such as pH, type of solvents, sonication parameter; surfactant used, and surface functionalized properties, to name a few. In the current study, the dispersion behavior of the NPs was observed in distilled water with different pH levels as a function of storage time, as presented in Figure 7. In general, both the NPs displayed considerable dispersion capacity, but relatively better property was observed for G-AgNPs as compared to G-Ag2S-NPs.

Pictorial presentation of pH-responsive dispersion behavior of F. arabica synthesized G-AgNPs and G-Ag2S-NPs at different pH values as a function of storage time.
For instance, the G-AgNPs did not show any type of sedimentation even after 24 h in particular at pH 12. However, the G-AgNPs at pH 2 started to sediment after 30 min, and after 24 h, complete sedimentation could be observed. In comparison, green synthesized Ag2S NPs displayed relatively better performance at pH 7, and eminent dispersion could be observed till the end of the experiment. Furthermore, different colors were also observed for G-AgNPs, but the change was most noticeable color at pH 12, giving intense blackish contrast. For G-Ag2S-NPs, a light brown color at pH 7 was observed as compared to dark brown at pH 7.
3.2.7 PL analysis
PL studies revealed that G-AgNPs exhibited a PL spectrum spanning 385–480 nm, as illustrated in Figure 8. The emission spectrum displayed a broad emission at 427 nm. These findings suggest that the reduction in particle size led to the generation of the PL peak [58]. In the case of G-Ag2S-NPs, the emission spectrum displayed a broad emission at 435 nm. G-Ag2S has gained attention in recent studies for its applications in bio-labeling and bio-imaging. Their optical properties, including remarkable photo-stability, high PL efficiency, and emission characteristics dependent on particle size, make them advantageous [59]. The luminescence peaks show a shift toward the green region of visible light, ranging from 390 to 525 nm.

PL spectra of F. arabica synthesized G-AgNPs and G-Ag2S-NPs.
3.3 Biological applications
3.3.1 F. arabica synthesized G-Ag2S-NPs exhibit higher antioxidant potential than G-AgNPs
The F. arabica synthesized G-AgNPs and G-Ag2S-NPs were explored for antioxidant and free radical scavenging potential using three distinct assays, i.e. TAC, TRP, and FRSA, as compiled in Figure 9. The evaluation of free radical scavenging potential utilized the DPPH FRSA, where the stable and purple DPPH turns into a pale-yellow color from purple solution upon reduction in the presence of antioxidant substances. This alteration in color thus signifies the scavenging activity of the test material against free radicals. In the analysis, it was observed that G-Ag2S exhibited more inhibition, i.e. 39.52%, as compared to the F. arabica synthesized G-AgNPs with 24.76% inhibition at the tested concentration. To support the findings, the antioxidant capacity was further assessed based on the phosphomolybdenum technique based on Mo (VI) reduction to (V) by the test sample. A similar trend was determined in the DPPH free radical scavenging activity as the TAC activity of 86.06 μg AAE/mg was recorded for G-Ag2S-NPs as compared to the 64.44 μg AAE/mg for the G-AgNPs. To further assess and support the antioxidant potential, a TRP assay, also known as FRAP assay, was utilized to determine the reducing potential of G-AgNPs and G-Ag2S. Here, in this technique, the sample reacts with the potassium ferricyanide (Fe3+) and reduces it to the potassium ferrocyanide (Fe2+) and forms a colored complex of ferric–ferrous by reacting with the ferric chloride. This can be computed at the wavelength of 630 nm by using a spectrophotometer. Our analysis found G-Ag2S-NPs with a high reducing power of 276.34 μg AAE/mg, while G-AgNPs exhibited a ferric reducing power of 170.53 μg AAE/mg of the sample. Thus, it was concluded that F. arabica synthesized G-Ag2S-NPs exhibit higher free radical and antioxidant potential as compared to the G-AgNPs. The increased antioxidant activity of the Ag2S NPs compared to AgNPs may be due to the synergetic effect of silver and sulfur ions along with the phytochemicals. It has been well reported that, like Ag, sulfur ions have considerable antioxidant activity [60].
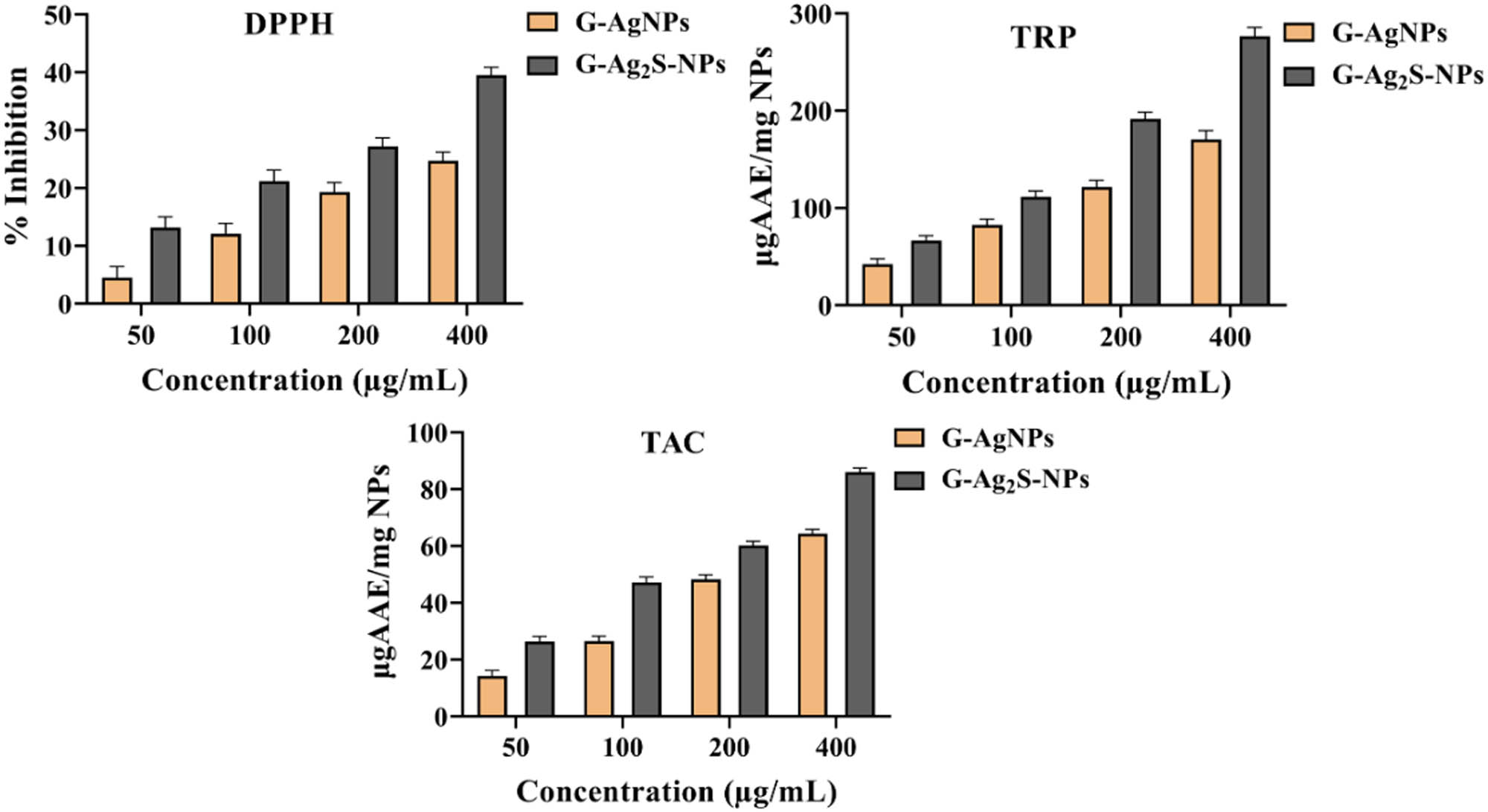
Antioxidant activities of F. arabica synthesized G-AgNPs and G-Ag2S-NPs. DPPH-FRSA, TAC, and TRP estimation.
3.3.2 F. arabica synthesized AgNPs possess enhanced bactericidal properties than Ag2S-NPs
Using MNPs as antimicrobial materials alternative to antibiotics is currently an intensively explored research area. The nanoscale materials may offer several benefits and advantages as antibacterial agents, including the ability to overcome current antibiotic resistance mechanisms, attack bacterial cells utilizing numerous methods and mechanisms simultaneously, and serve as effective antibiotic transporters or antibiotic delivery vehicles [31]. In the current study, different concentrations of the F. arabica synthesized G-AgNPs and G-Ag2S-NPs were tested for antibacterial properties via the well diffusion method. Three Gram-negative strains, i.e., E. coli, K. pneumoniae, and S. enterica, while four positive strains, i.e., MRSA, S. epidermidis, S. aureus, and B. subtilis were used in the susceptibility studies. Generally, both the green synthesized G-AgNPs and G-Ag2S-NPs showed considerable antibacterial activity in concentration-dependent pattern, as summarized in Figure 10. According to the results, the G-AgNPs displayed higher antibacterial activity as compared to the G-Ag2S-NPs. For instance, G-AgNPs showed significant activity against B. subtilis, with the determined ZOI of 25 ± 1.4 mm at the experimented concentration of 5 mg·mL−1. Furthermore, the NPs also resulted in excellent activity with ZOIs of 25 ± 1.4 mm and 24 ± 1.2 against MRSA and E. coli, respectively. Likewise, K. pneumonia, with an inhibition zone of 19 ± 0.88 mm, displayed moderate susceptibility. Among the strains, S. enterica was observed to be the least sensitive, with an inhibition zone of 18 ± 2.2 mm at the highest concentration used. However, at lower concentrations, E. coli was found to be more susceptible (Table 2) with the MIC of 39 µg·mL−1, followed by S. aureus and MRSA with the MIC of 78 and 156 µg·mL−1, respectively. K. pneumonia and S. epidermidis were found to be the least sensitive at lower concentrations with the MIC of 312 µg·mL−1 against each of the strains. As compared to AgNPs, the F. arabica synthesized G-Ag2S-NPs showed lower antibacterial properties. Among the strains used, S. aureus was the most sensitive that induced 23 ± 2.0 mm, ZOI against 5 mg·mL−1 while MIC for the strain was determined as 156 µg·mL−1. G-Ag2S-NPs also showed average activities against B. subtilis and E. coli with 15 ± 1.4 and 14.5 ± 1.2 mm, ZOIs, respectively, with the MIC of 312 µg·mL−1, each. While a weak activity was observed against the K. pneumoniae and S. enterica that induced 13 ± 1.6 and 13 ± 1.4 mm, ZOIs against 5 mg·mL−1 with MICs of 625 µg·mL−1 for each of the strains. Our extensive study thus concluded that AgNPs demonstrated higher antibacterial properties even at lower concentrations as compared to Ag2S-NPs.
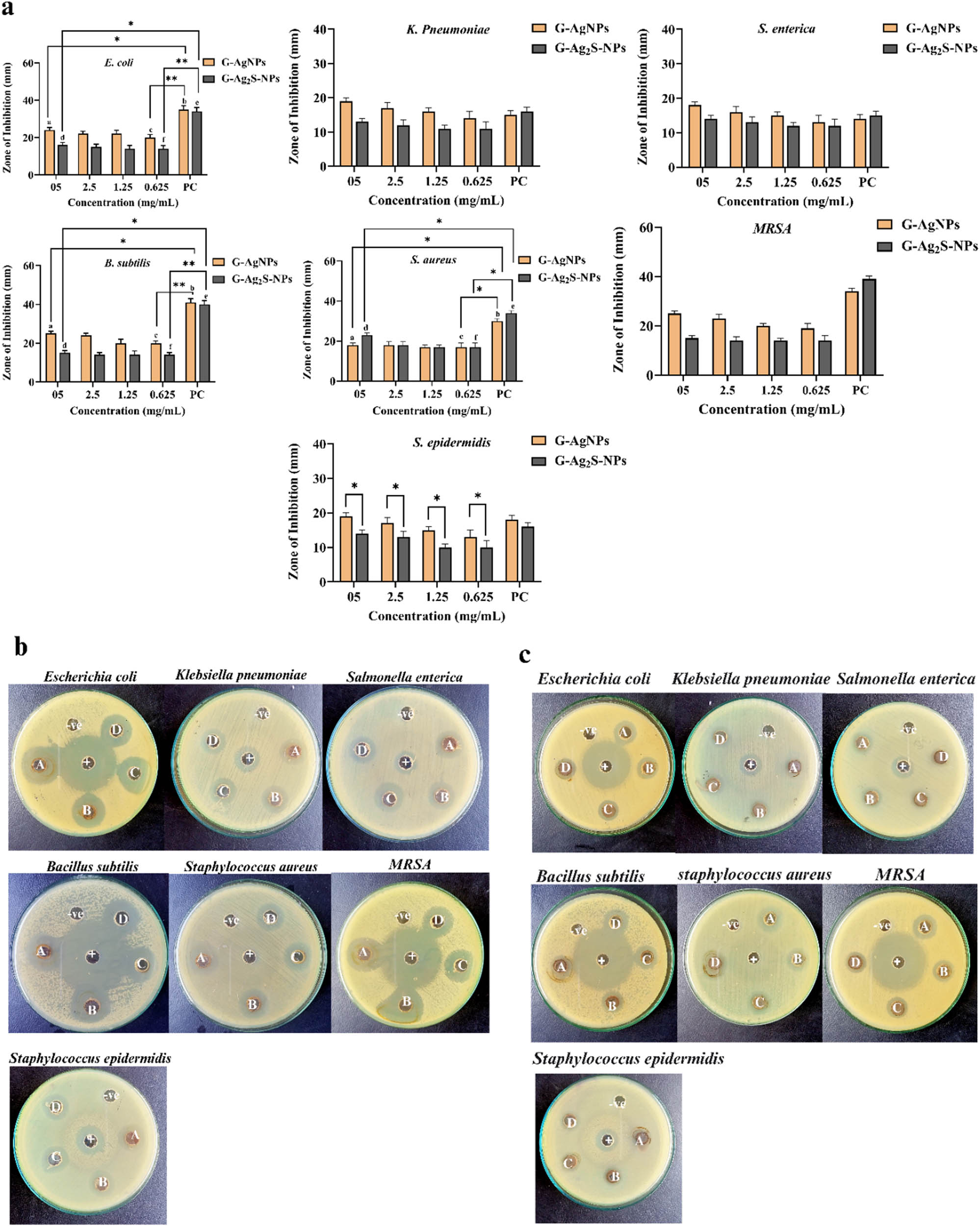
Graphical illustration of antibacterial activity of (a) G-AgNPs and (b) G-Ag2S-NPs against bacterial strains. Pictorial presentation of the antibacterial activity of (c) G-AgNPs and (d) G-Ag2S-NPs. The alphabets A, B, C, and D symbolize various concentrations, i.e. 5, 2.5, 1.25, and 0.625 mg·mL−1. NC (−) is the negative control, i.e. DMSO, and PC (+) is the positive control i.e. levofloxacin, the antibiotic. Bars represent the mean ± standard deviation (SD), and asterisks (*) denote significant differences between treatment groups (p < 0.05).
MIC of G-AgNPs and G-Ag2S-NPs against tested bacterial strains
| Sample | E. coli (µg·mL−1) | K. pneumoniae (µg·mL−1) | S. enterica (µg·mL−1) | MRSA (µg·mL−1) | S. epidermidis (µg·mL−1) | S. aureus (µg·mL−1) | B. subtilis (µg·mL−1) |
|---|---|---|---|---|---|---|---|
| G-AgNPs | 39 | 312 | 156 | 156 | 312 | 78 | 156 |
| G-Ag2S-NPs | 312 | 625 | 625 | 312 | 625 | 156 | 312 |
There are multiple factors that may have contributed to the distinct antibacterial properties of the materials. For instance, the reactivity, durability, and biological properties of NPs are greatly influenced by their specific size, shape, and structure [61]. Smaller sizes of the NPs have a higher surface area-to-volume ratio, enhancing the interaction with the biological systems. This increases the surface reactivity and improves the cellular uptake and thus antibacterial activity. AgNPs demonstrated stronger antibacterial effects as their smaller size allows them to penetrate bacterial cell walls more effectively [62]. The shape of NPs also impacts their cellular interactions, including uptake and distribution. Spherical AgNPs are generally more readily internalized by cells than rod-shaped or triangular or irregular-shaped shaped NPs, which may interact differently with cellular membranes [63]. Ag₂S-NPs, being slightly larger in size and with irregularly shaped morphologies, lead to different cellular responses and toxicity profiles compared to AgNPs, influencing their antimicrobial activities [64]. A thorough review of the literature suggests that multiple steps or mechanisms are involved for the antibacterial properties of MNPs including AgNPs, as illustrated in Figure 11. (1) The diffusion and absorption of nanoparticles take place on the cell membrane of micro-organisms, which cause accumulation and dissolution of nanoparticles on the cell membrane, leading to the cell leakage of micro-organisms resulting in cell death. (2) Nanoparticles interact with the DNA of micro-organisms, ceasing the replication and growth cycle, which causes apoptosis of the cell. (3) Nanoparticles may also interact and dysfunctional the endoplasmic reticulum and ribosomes, thus dismantle the production of proteins. (4) MNPs may also adhere to the thiol groups (–SH) and block the enzyme activations sites, making them inactive. (5) Finally, the NPs also influence the normal functioning of the mitochondria known as the powerhouse of the cells thus dismantle the energy production [65].

Graphical presentation of the plausible antibacterial mechanism of F. arabica synthesized G-AgNPs and G-Ag2S-NPs.
3.3.3 F. arabica synthesized AgNPs exhibited lower antifungal activity while Ag2S-NPs were inactive
Along with the antibacterial properties, F. arabica synthesized G-AgNPs and G-Ag2S-NPs were also assessed for antifungal potential in multiple concentrations via a well diffusion method. In the study, four fungal species, i.e., A. flavus, A. fumigatus, T. rubrum, and A. terreus, were used for the analysis, and the activity was measured as ZOIs in mm as summarized in Figure 12. In general, G-AgNPs showed considerable antifungal activity in a concentration-dependent pattern. Surprisingly, G-Ag2S-NPs did not possess any antifungal activity and were found to be inactive against the tested fungal strains. According to the observed results, the G-AgNPs exhibited considerable activity against T. rubrum with 22 ± 1.8 mm ZOI and MIC of 12 mg·mL−1. A. flavus was the second most sensitive strain, resulting in 20 ± 1.4 mm ZOI at the highest concentration but MIC for the strain was measured as 50 mg·mL−1. Similarly, both the A. fumigatus and A. terreus showed 17 ± 1.2 mm ZOIs with MICs of 12.5 and 25 mg·mL−1, respectively. Our study thus concluded that F. arabica synthesized G-AgNPs and G-Ag2S-NPs were highly active against bacterial strains, but the NPs did not induce significant fungicidal effects, especially at low concentrations.

(a) Graphical and (b) pictorial presentation of antifungal activity of G-AgNPs against tested fungal strains. The alphabets a, b, c, and d symbolize various concentrations, i.e. 100, 50, 25, 12.5 mg·mL−1. NC (−) is the negative control, i.e., DMSO, and PC (+) is the positive control, i.e. Clotrimazole.
3.3.4 F. arabica synthesized AgNPs exhibit slightly better antileishmanial potential than Ag2S-NPs
Leishmaniasis is caused by the intracellular protozoan parasite Leishmania and poses a significant threat to global health [66]. The parasitic disease affects approximately 12 million people worldwide, with an additional 350 million at risk [67]. The disease is prevalent in the poorest regions, exacerbated by underfunding, pharmaceutical disinterest, and inadequate healthcare. Till now, about 20 Leishmania species have been discovered that cause the disease in over 70 countries, with high incidences in Afghanistan, Algeria, Brazil, Pakistan, Peru, Saudi Arabia, and Syria [68,69]. Following transmission by sandfly bites, Leishmania swiftly locates host cells, residing in long-lived macrophages [70,71]. Adapting from promastigotes to amastigotes, the parasites survive in the harsh phago-lysosome-like environment [72]. The leishmanicidal efficiency of the F. arabica synthesized G-AgNPs and G-Ag2S-NPs was examined against the parasite L. tropica using multiple concentrations of the NPs via the MTT cell viability assay. We found a clear concentration-dependent pattern of growth, as summarized in Figure 13. While considerable cytotoxic potential was documented at all the concentrations, but % inhibition improved with increasing concentration. For G-AgNPs, 79.40 ± 1.2% inhibition of promastigote and 73.34 ± 1.4% inhibitions were noted for the amastigote forms of the parasite at 200 µg·mL−1. However, the inhibition reduced to 15.78 ± 1.0% and 12.12 ± 1.20%, respectively, when a lower concentration of 12.5 µg·mL−1 was taken into account. The IC50 was analyzed as 93.57 µg·mL−1 for amastigote, while IC50 of 88.39 µg·mL−1 was recorded for promastigote. Subsequently, for G-Ag2S-NPs, 72.3 ± 1.6% and 68.2% ± 1.4% inhibition for promastigote and amastigote were recorded, respectively. When the lower concentrations were tested, % inhibition dropped to 11.88 ± 1.0% and 07.22 ± 1.2%, respectively. Furthermore, the IC50 of 107.25 µg·mL−1 and 121.8 µg·mL−1, were calculated for the promastigote and amastigote, respectively. Amphotericin B was utilized as a benchmark of comparison for its leishmanicidal effectiveness as illustrated in the figure. Comparatively, F. arabica synthesized G-AgNPs exhibited slightly higher % inhibition than G-Ag2S-NPs at both the maximum and minimum tested concentrations. The anti-leishmanial mechanism can be assumed as widely discoursed for other metallic nanostructures. Literature review suggests that when metals interact with the parasite’s cell membrane at the nanoscale, the NPs enter the cell and prompt oxidative stress (OS) that eventually causes cell mortality. The sub-micron size and high surface area of MNPs allow them to interact with sensitive cellular components, including DNA and enzymes. Such interactions dysfunctional the cellular machinery and synthesize faulty proteins and or enzyme inactivation, ultimately resulting in parasite mortality [73].
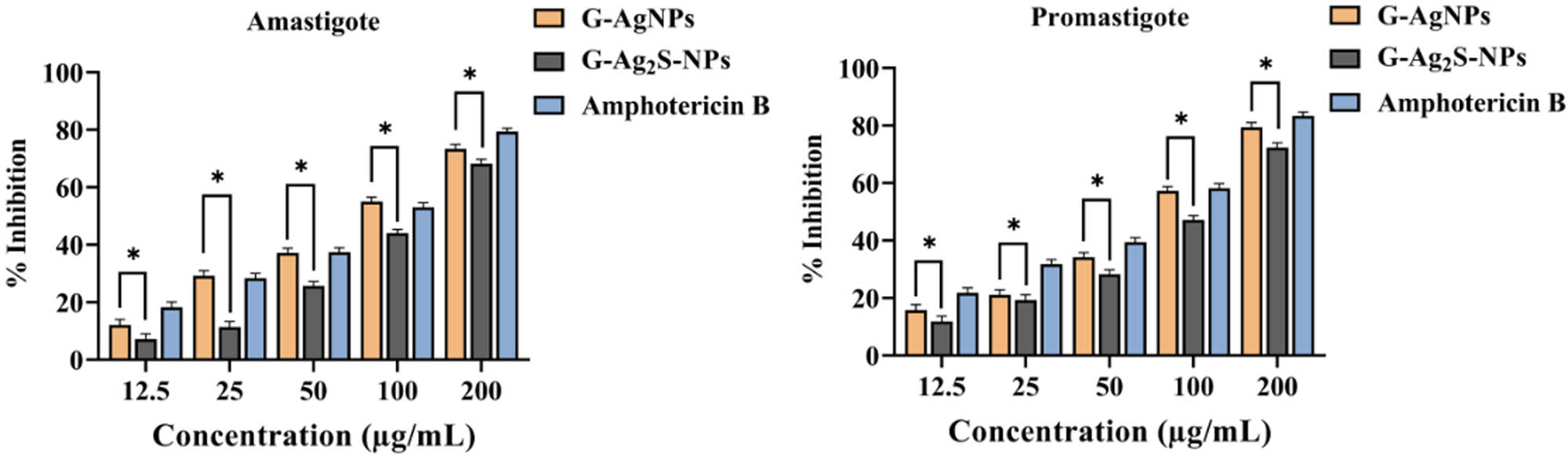
Antileishmanial activity of F. arabica synthesized G-AgNPs and G-Ag2S-NPs. Amastigote and promastigote forms of the parasite. Statistical analysis was performed using two-way ANOVA with a subsequent post hoc test for multiple comparisons. Bars represent the mean ± SD, and asterisks (*) indicate statistically significant differences between treatment groups (p < 0.05).
3.3.5 F. arabica synthesized Ag2S-NPs resulted in slightly better anticancer potential than AgNPs
An in vitro anticancer activity was conducted to evaluate the cytotoxicity of F. arabica synthesized G-AgNPs and G-Ag2S-NPs against the MCF-7 breast cancer cell line via the MTT assay. The prevalence of breast carcinoma in Asia, particularly in Asian women, has been ever-increasing. In Pakistan, the breast carcinoma rates are the highest in Asia, surpassing those in neighboring states by 2.5 times. Most importantly, this form of cancer ranks among the top 20 causes of mortality in Pakistan [74]. Thus, in the current study, a cytotoxicity assay was carried out against the MCF-7 cell line in multiple concentrations ranging from 50 to 400 µg mL−1. In general, a significant but concentration-dependent cytotoxicity potential was observed for both the NPs, with the G-Ag2S-NPs displaying a slightly improved effectiveness as summarized in Figure 14. For instance, at the highest tested concentration, the G-Ag2S-NPs resulted in a remarkable inhibition of 96.31 ± 1.8% of the MCF-Cells. However, the inhibition declined slightly at the decreasing concentration, and a deep decline was observed between 100 and 50 µg·mL−1 with IC50 of 73 µg·mL−1. It was observed that % cytotoxicity in the case of G-Ag2S-NPs was more effective than G-AgNPs, even at the very low concentration of 50 µg·mL−1. One reason for such significant cytotoxic potential is due to the intrinsic property of the plant extract having the potential to eliminate cancerous cells and its combination with Ag2S-NPs may have slightly enhanced its potential to eliminate cancer cells. According to a recent study, the crude extracts of F. arabica showed significant cytotoxic potential against cancerous cells of MCF-7 [35]. It has also been reported that sulfur ions have the potential to inhibit the proliferation of breast cancer (MCF-7) cells by inhibiting their ErB2, ErB3 protein, and mRNA expression [35,75]. Likewise, G-AgNPs also resulted in 93.97 ± 1.6% inhibition of the MCF-7 cells at the highest experimented concentration. However, their potential declined with decreasing concentration, especially between 100 and 50 µg·mL−1 with IC50 of 85.5 µg·mL−1. The study thus concluded that both the F. arabica synthesized G-AgNPs and G-Ag2S-NPs exhibited excellent anticancer potential, with Ag2S NPs exhibiting slightly improved performance.
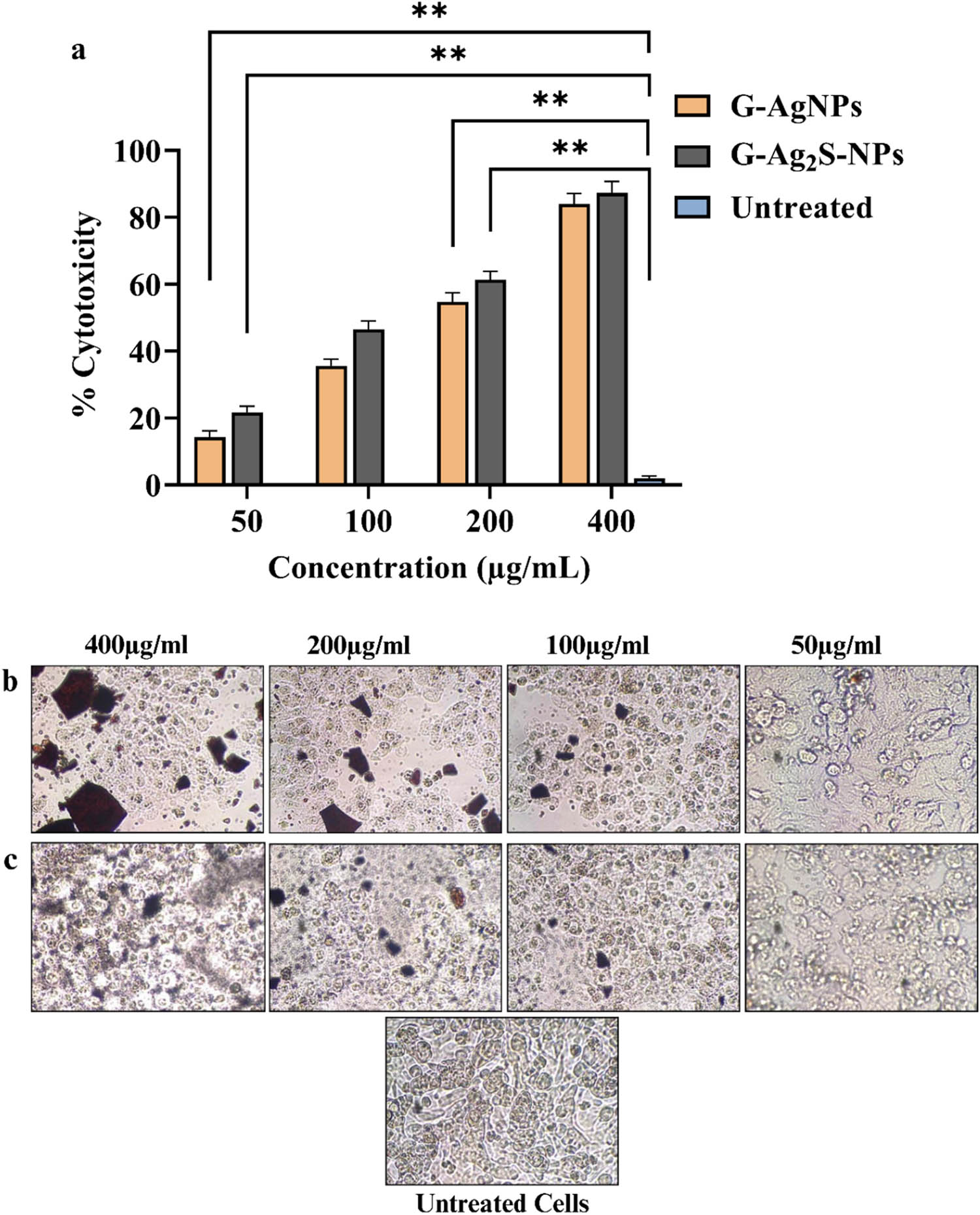
(a) Graphical illustration of the % cytotoxicity of MCF-7 cells against the G-AgNPs, and G-Ag2S-NPs. Pictorial presentation of the cytotoxicity of MCF-7 cells against (b) G-Ag-NPs and (c) G-Ag2S-NPs. The cytotoxicity assay was carried out thrice, and the data here is mean with standard deviation. Statistical analysis was evaluated using two-way ANOVA to examine the interactions between different treatment groups. Following the two-way ANOVA, a post hoc multiple comparison test was carried out to identify specific differences between group means. A p-value of less than 0.05 was considered statistically significant.
Various mechanisms have been associated with the anticancer properties of nano-scale materials, for instance, MNPs like AgNPs are known to induce the production of ROS upon entering the cancer cells. This ROS generation disrupts cellular signal transduction pathways, leading to mitochondrial dysfunction and uncoupling of respiration. The elevated ROS levels, coupled with decreased glutathione, result in activation of nuclear factor kB (NF-kB) and tumor necrosis factor (TNF-α), which damage cellular components such as DNA, lipids, and proteins. This OS triggers the activation of apoptotic pathways, leading to cell death as illustrated in Figure 15 [33,76,77,78,79,80]. The upregulation of the tumor suppressor protein p53 is another key mechanism of inducing apoptosis in cancer cells. The released silver ions from AgNPs trigger this upregulation, which is further associated with caspase activation, leading to programmed cell death [78,79,80].
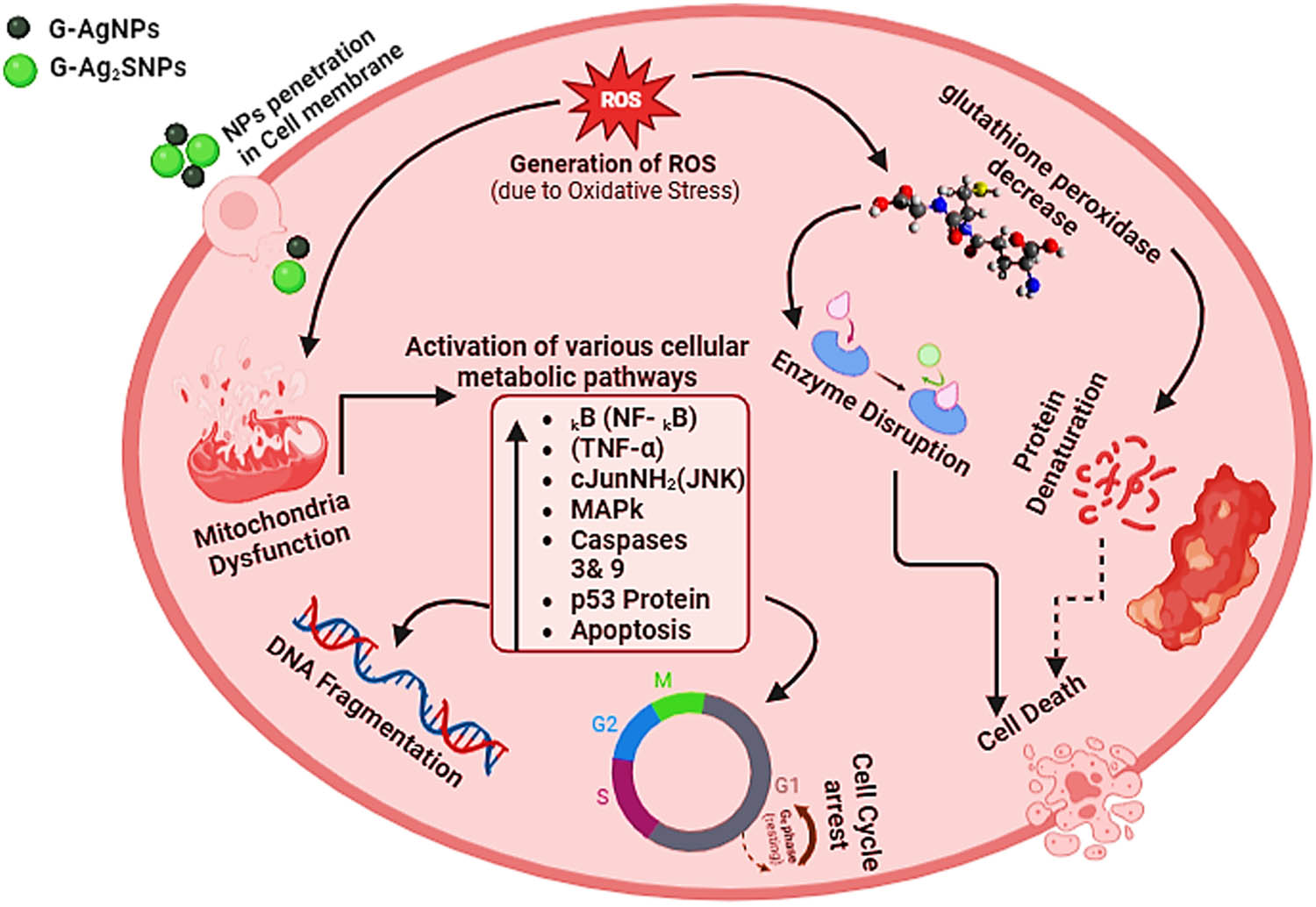
Plausible anticancer mechanism of the F. arabica synthesized G-AgNPs and G-Ag2S-NPs.
3.3.6 Both the F. arabica synthesized Ag2S-NPs and AgNPs are compatible with blood cells
The biocompatibility of emerging nanomaterials (ENMs) is essential to investigate and recognize the reaction of cells or tissues to newly prepared materials or nanomaterial-based implants. Such assessment not only confirms the safety of biological uses of emerging materials but also opens the door for the creation of novel treatments that pose little danger to essential blood components, leading the field of nanomedicine closer to safer and more efficient medical interventions [26,81]. In the current study, the toxicity of the F. arabica synthesized G-AgNPs and G-Ag2S-NPs against isolated human RBCs was investigated using an in vitro hemolysis assay. The NPs were tested against the blood cell lysis at various concentrations, as summarized in Figure 16. Our study found no eminent and obvious hemolytic activity, even at the highest measured concentrations. During the co-incubation with NPs, all of the blood cells maintained their integrity and failed to rupture. In contrast to our tested materials, Triton X-100, employed as a positive standard, caused ample hemolysis of the RBCs. The highly ruptured and damaged morphology can be observed in the light micrographs. The results hence suggest that the F. arabica synthesized G-AgNPs and G-Ag2S-NPs are safe and do not harm blood cells at the experimented concentrations.
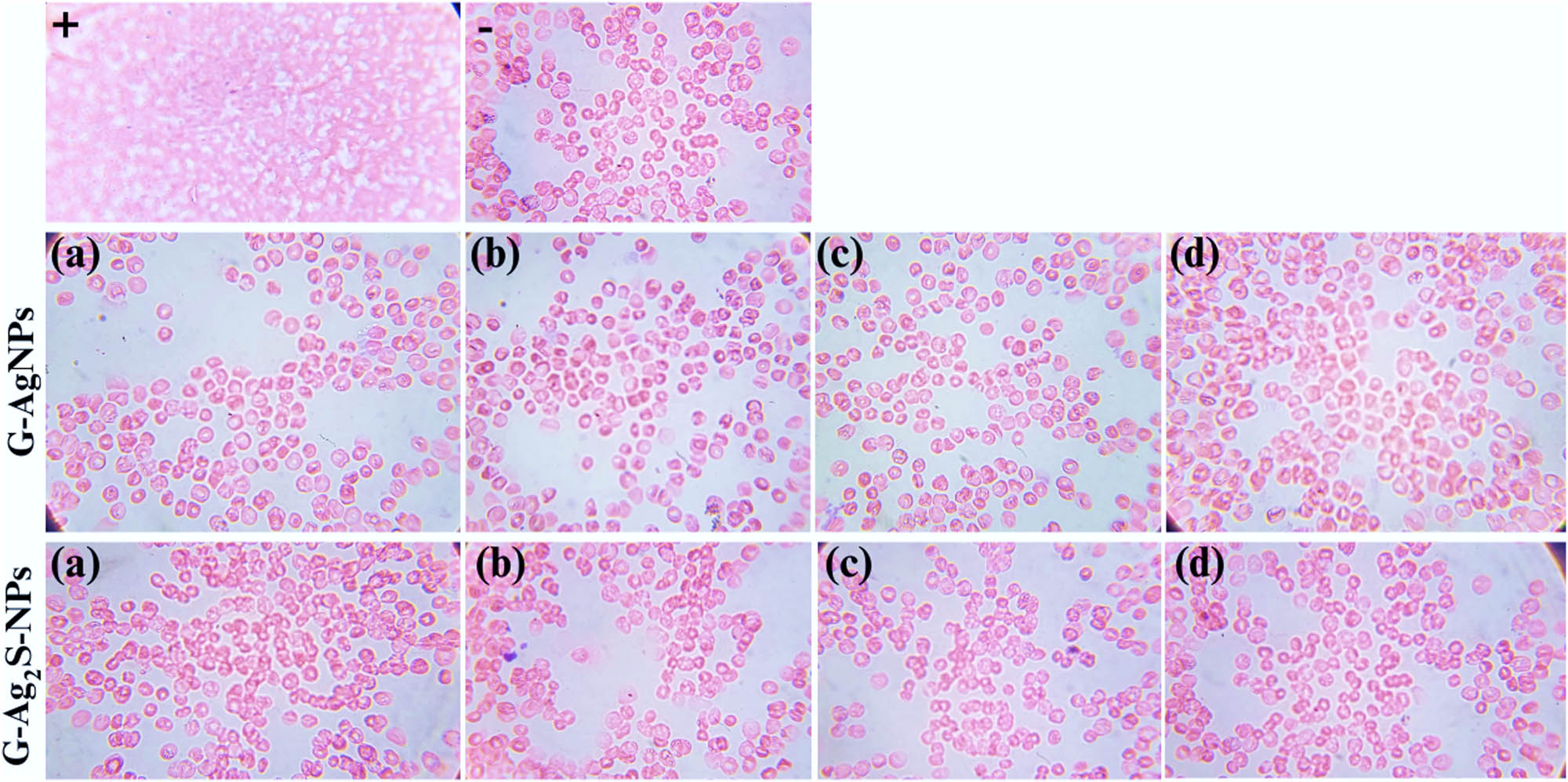
Microscopic visualization of the hemolytic behavior of F. arabica synthesized G-AgNPs and G-Ag2S-NPs at 1,000×. The alphabets a, b, c, and d symbolize various concentrations, i.e. 5, 2.5, 1.25, 0.625 mg·mL−1. (−) is the negative control, i.e. DMSO, and (+) is the positive control, i.e. Triton X-100.
4 Conclusions
The current study investigated rarely explored green synthesized Ag2S-NPs and extensively explored AgNPs for biological properties which were synthesized using an aqueous extract of F. arabica. After extensive characterization, our study found that both the NPs exhibited excellent biological properties; however, Ag2S-NPs possessed relatively enhanced antioxidant and anticancer potential against the MCF-7 cell line, demonstrating excellent ROS scavenging and cytotoxic potential against breast cancer. Contrarily, AgNPs resulted in relatively better antibacterial, anti-parasitic, and antifungal properties. We thus found that both the NPs unveiled distinct biological properties as biomedical applications. However, both the synthesized NPs demonstrated excellent compatibility against human blood cells in in vitro studies, thus ensuring their compatible and safe nature, in initial safety screening. Being highly potent antimicrobial and anticancer materials, the F. Arabica synthesized G-AgNPs and G-Ag2S-NPs can be further explored in in vivo disease models to uncover the mechanisms responsible for their excellent biological properties. Furthermore, the antibacterial potential of the NPs can also be exploited in a variety of applications, including dental materials, surgical applications, wounds and burns management, and biomedical devices.
Acknowledgements
The authors extend their appreciation to the Researchers supporting project number (RSP2024R185), King Saud University, Riyadh, Saudi Arabia.
-
Funding information: Authors state no funding involved.
-
Author contributions: N.R.J. carried out the experimental work, analyzed the data, and wrote the manuscript. M.A.K. perceived the idea, supervised the project, and wrote and reviewed the manuscript. M.F.H., K.M.A., M.S.E., D.A.F., H.M.M., and J.I. assisted in the experimental work and characterization analysis. S.A.I.B. supervised the project. The authors N.R.J. and M.A.K. contributed equally to this work.
-
Conflict of interest: Authors state no conflict of interest.
-
Data availability statement: The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
[1] Sadovnikov SI, Gusev AI, Rempel AA. Nanostructured lead sulfide: synthesis, structure and properties. Russian Chem Rev. 2016;85(7):731.10.1070/RCR4594Search in Google Scholar
[2] Shi X, Zheng S, Gao W, Wei W, Chen M, Deng F, et al. Excitation wavelength and intensity dependence of photo-spectral blue shift in single CdSe/ZnS quantum dots. J Nanopart Res. 2014;16:1–9.10.1007/s11051-014-2741-3Search in Google Scholar
[3] Hsu JC, Cruz ED, Lau KC, Bouché M, Kim J, Maidment AD, et al. Renally excretable and size-tunable silver sulfide nanoparticles for dual-energy mammography or computed tomography. Chem Mater. 2019;31(19):7845–54.10.1021/acs.chemmater.9b01750Search in Google Scholar PubMed PubMed Central
[4] Sadovnikov S. Thermal expansion of nanostructured solid solutions of lead and silver sulfides. Int J Nanosci. 2019;18(03n04):1940061.10.1142/S0219581X19400611Search in Google Scholar
[5] Hamedi S, Ghaseminezhad SM, Shojaosadati SA, Shokrollahzadeh S. Comparative study on silver nanoparticles properties produced by green methods. Iran J Biotechnol. 2012;10(3):191–7.Search in Google Scholar
[6] Elsupikhe RF, Shameli K, Ahmad MB, Ibrahim NA, Zainudin N. Green sonochemical synthesis of silver nanoparticles at varying concentrations of κ-carrageenan. Nanoscale Res Lett. 2015;10:1–8.10.1186/s11671-015-0916-1Search in Google Scholar PubMed PubMed Central
[7] Zhang XF, Liu ZG, Shen W, Gurunathan S. Silver nanoparticles: synthesis, characterization, properties, applications, and therapeutic approaches. Int J Mol Sci. 2016;17(9):1509–34.10.3390/ijms17091534Search in Google Scholar PubMed PubMed Central
[8] Hamedi S, Shojaosadati SA. Rapid and green synthesis of silver nanoparticles using Diospyros lotus extract: Evaluation of their biological and catalytic activities. Polyhedron. 2019;171:172–80.10.1016/j.poly.2019.07.010Search in Google Scholar
[9] Ahmed S, Ahmad M, Swami BL, Ikram S. A review on plants extract mediated synthesis of silver nanoparticles for antimicrobial applications: A green expertise. J Adv Res. 2016;7(1):17–28.10.1016/j.jare.2015.02.007Search in Google Scholar PubMed PubMed Central
[10] Malik A, O’Brien P, Revaprasadu N. A simple route to the synthesis of core/shell nanoparticles of chalcogenides. Chem Mater. 2002;14:2004–10.10.1021/cm011154wSearch in Google Scholar
[11] Patel P, Agarwal P, Kanawaria S, Kachhwaha S, Kothari S. Plant-based synthesis of silver nanoparticles and their characterization. Springer International Publishing; 2015. p. 271–88.10.1007/978-3-319-14502-0_13Search in Google Scholar
[12] Alamami A, Elshibani F, Elremali N, Daboub A, Zaed SB, Bumadian M. The species of genus fagonia in Libya:(A comprehensive review). J Pharmacogn Phytochem. 2022;11(5):28–37.10.22271/phyto.2022.v11.i5a.14501Search in Google Scholar
[13] El-Amier YA, Aisha IAA. Phytochemical constituents of common growing Fagonia species (Zygophyllaceae) in Egyptian deserts and its biological activities. Plant Arch (09725210). 2019;19(2):2213–9.Search in Google Scholar
[14] Prajapati R, Davra K, Kalariya M, Sailor G, Jain V. Pharmacognostic and phytochemical evaluation of the fagonia arabica stem-a potent indian medicinal plant. Int J Pharmacogn. 2020;8:193–7.10.4103/JISM.JISM_49_20Search in Google Scholar
[15] Iftikhar N, Chatha SA, Ahmad T, Ali Q, Hussain AI, Rathore HA. Fagonia arabica L.: a review of its phytochemistry, pharmacology and traditional uses. Comb Chem High Throughput Screening. 2022;25(7):1187–99.10.2174/1386207325666210923120957Search in Google Scholar PubMed
[16] Alqasoumi SI, Yusufoglu HS, Alam A. Anti-inflammatory and wound healing activity of Fagonia schweinfurthii alcoholic extract herbal gel onn albino rats. Afr J Pharm Pharmacol. 2011;5(17):1996–2001.10.5897/AJPP11.190Search in Google Scholar
[17] Qureshi H, Asif S, Ahmed H, Al-Kahtani H, Hayat K. Chemical composition and medicinal significance of Fagonia cretica: A review. Nat product Res. 2016;30:625–39.10.1080/14786419.2015.1036268Search in Google Scholar PubMed
[18] Shiekh F, Naz I, Rahman L, Younis T, Shinwari Z, Faisal R. Genus Fagonia mediated nanoparticles and their therapeutic potential: a review. Pak J Botany. 2024;56:1623–9.10.30848/PJB2024-4(13)Search in Google Scholar
[19] Puri D, Bhandari A. Fagonia: A potential medicinal desert plant. J Nepal Pharm Assoc. 2015;27(1):28–33.10.3126/jnpa.v27i1.12147Search in Google Scholar
[20] Awwad AM, Salem NM. Green synthesis of silver nanoparticles byMulberry LeavesExtract. Nanosci Nanotechnol. 2012;2(4):125–8.10.5923/j.nn.20120204.06Search in Google Scholar
[21] Aziz H, Saeed A, Jabeen F, Khan MA, Rehman AU, Khan MQ, et al. Design, synthesis, in vitro anti-oxidant evaluation, α-amylase inhibition assay, and molecular docking analysis of 2-(2-benzylidenehydrazinyl)-4, 4-diphenyl-1H-imidazol-5 (4H)-ones. J Mol Struct. 2023;1278:134924.10.1016/j.molstruc.2023.134924Search in Google Scholar
[22] Ullah H, Ihsan J, Mohamed RM, Khan MA, Ghani M, Rauf N, et al. Bionanocomposite scaffolds based on MnS-nanorods loaded acacia-Senegal-gum hydrogels: Fabrication, characterization and biological evaluation. Bioact Carbohydr Diet Fibre. 2023;30:100368.10.1016/j.bcdf.2023.100368Search in Google Scholar
[23] Faisal S, Shah SA, Shah S, Akbar MT, Jan F, Haq I, et al. In vitro biomedical and photo-catalytic application of bio-inspired zingiber officinale mediated silver nanoparticles. J Biomed Nanotechnol. 2020;16(4):492–504.10.1166/jbn.2020.2918Search in Google Scholar PubMed
[24] Hussain A, Sajid M, Rasheed H, Hassan M, Khan MA, Bokhari SAI. Phytochemistry and antibacterial efficacy of Northeastern Pakistani Artemisia rutifolia Stephan ex Spreng. extracts against some clinical and phyto-pathogenic bacterial strains. Acta Pharm Sci. 2022;60(3):247–71.10.23893/1307-2080.APS.6017Search in Google Scholar
[25] Ihsan J, Farooq M, Khan MA, Ghani M, Shah LA, Saeed S, et al. Synthesis, characterization, and biological screening of metal nanoparticles loaded gum acacia microgels. Microsc Res Tech. 2021;84(8):1673–84.10.1002/jemt.23726Search in Google Scholar PubMed
[26] Ahmad U, Khan MA, Zahid MU, Alarjani KM, Gawwad MRA, Hussain SJ, et al. Effect of surface capping on the biological properties of calcium oxide nanoparticles (CaO-NPs). Chem Pap. 2024;78:1–20.10.1007/s11696-024-03446-xSearch in Google Scholar
[27] Aziz A, Ali N, Khan A, Bilal M, Malik S, Ali N, et al. Chitosan‑zinc sulfide nanoparticles, characterization and their photocatalytic degradation efficiency for azo dyes. Int J Biol Macromol. 2020;153:502–12.10.1016/j.ijbiomac.2020.02.310Search in Google Scholar PubMed
[28] Siddique MAR, Khan MA, Bokhari SAI, Ismail M, Ahmad K, Haseeb HA, et al. Ascorbic acid-mediated selenium nanoparticles as potential antihyperuricemic, antioxidant, anticoagulant, and thrombolytic agents. Green Process Synth. 2024;13(1):20230158.10.1515/gps-2023-0158Search in Google Scholar
[29] Farooq M, Ihsan J, Mohamed RM, Khan MA, Rehman TU, Ullah H, et al. Highly biocompatible formulations based on Arabic gum Nano composite hydrogels: Fabrication, characterization, and biological investigation. Int J Biol Macromol. 2022;209:59–69.10.1016/j.ijbiomac.2022.03.162Search in Google Scholar PubMed
[30] Magaldi S, Mata-Essayag S, De Capriles CH, Pérez C, Colella M, Olaizola C, et al. Well diffusion for antifungal susceptibility testing. Int J Infect Dis. 2004;8(1):39–45.10.1016/j.ijid.2003.03.002Search in Google Scholar PubMed
[31] Khan MA, Siddique MAR, Sajid M, Karim S, Ali MU, Abid R, et al. A comparative study of green and chemical cerium oxide nanoparticles (CeO2-NPs): From synthesis, characterization, and electrochemical analysis to multifaceted biomedical applications. BioNanoScience. 2023;13(2):667–85.10.1007/s12668-023-01114-0Search in Google Scholar
[32] Zahid MU, Khan MA, Ahmad U, Alismail HA, Hussain SJ, Khan MI, et al. A comparative study of PEGylated cobalt oxide nanoparticles (Co3O4-NPs) and cobalt sulfide nanoparticles (Co9S8-NPs) for biological and photocatalytic applications. BioNanoScience. 2024;14:1–18.10.1007/s12668-024-01322-2Search in Google Scholar
[33] Fani S, Kamalidehghan B, Lo KM, Nigjeh SE, Keong YS, Dehghan F, et al. Anticancer activity of a monobenzyltin complex C1 against MDA-MB-231 cells through induction of apoptosis and inhibition of breast cancer stem cells. Sci Rep. 2016;6(1):38992.10.1038/srep38992Search in Google Scholar PubMed PubMed Central
[34] Kasture VS, Gosavi SA, Kolpe JB, Deshapande SG. Phytochemical and biological evaluation of Fagonia species: a review. World J Pharm Pharm Sci. 2014;9(5):1206–17.Search in Google Scholar
[35] Walbi IA, Alshabi AM, Alkahtani SA, Shaikh IA, Abdel-Wahab BA, Khateeb MM, et al. A preliminary cytotoxicity study of fagonia arabica against breast (MCF-7), oral (KB-3-1), AND lung cancer (A-549) cell lines: a study supported by molecular marker analysis using dual staining dyes. Separations. 2023;10(2):110.10.3390/separations10020110Search in Google Scholar
[36] Mani M, Pavithra S, Mohanraj K, Kumaresan S, Alotaibi SS, Eraqi MM, et al. Studies on the spectrometric analysis of metallic silver nanoparticles (Ag NPs) using Basella alba leaf for the antibacterial activities. Environ Res. 2021;199:111274.10.1016/j.envres.2021.111274Search in Google Scholar PubMed
[37] Jemal K, Sandeep B, Pola S. Synthesis, characterization, and evaluation of the antibacterial activity of Allophylus serratus leaf and leaf derived callus extracts mediated silver nanoparticles. J Nanomater. 2017;2017:1–11.10.1155/2017/4213275Search in Google Scholar
[38] Mason C, Vivekanandhan S, Misra M, Mohanty AK. Switchgrass (Panicum virgatum) extract mediated green synthesis of silver nanoparticles. World J Nano Sci Eng. 2012;2(2):47.10.4236/wjnse.2012.22008Search in Google Scholar
[39] Shaikh W, Chakraborty S, Islam R. Photocatalytic degradation of rhodamine B under UV irradiation using Shorea robusta leaf extract-mediated bio-synthesized silver nanoparticles. Int J Environ Sci Technol. 2020;17(4):2059–72.10.1007/s13762-019-02473-6Search in Google Scholar
[40] Sreelekha E, George B, Shyam A, Sajina N, Mathew B. A comparative study on the synthesis, characterization, and antioxidant activity of green and chemically synthesized silver nanoparticles. BioNanoScience. 2021;11:489–96.10.1007/s12668-021-00824-7Search in Google Scholar
[41] Natsuki J, Natsuki T, Hashimoto Y. A review of silver nanoparticles: synthesis methods, properties and applications. Int J Mater Sci Appl. 2015;4(5):325–32.10.11648/j.ijmsa.20150405.17Search in Google Scholar
[42] Lanje AS, Sharma SJ, Pode RB. Synthesis of silver nanoparticles: a safer alternative to conventional antimicrobial and antibacterial agents. J Chem Pharm Res. 2010;2(3):478–83.Search in Google Scholar
[43] Narayanan M, Divya S, Natarajan D, Senthil-Nathan S, Kandasamy S, Chinnathambi A, et al. Green synthesis of silver nanoparticles from aqueous extract of Ctenolepis garcini L. and assess their possible biological applications. Process Biochem. 2021;107:91–9.10.1016/j.procbio.2021.05.008Search in Google Scholar
[44] Awwad AM, Salem NM, Aqarbeh MM, Abdulaziz FM. Green synthesis, characterization of silver sulfide nanoparticles and antibacterial activity evaluation. Chem Int. 2020;6(1):42–8.Search in Google Scholar
[45] Christy RS, Kumaran J, Bansal C. Jump in band gap energy of Ag2S nanoparticles synthesized by solvothermal method. Adv Sci Focus. 2014;2(2):115–20.10.1166/asfo.2014.1087Search in Google Scholar
[46] Iqbal T, Munir K, Afsheen S, Zafar M, Abrar M, Qureshi MT, et al. Green synthesis of Ag2S/ZnS composites and their application for seeds germination to explore critical aspect. J Inorg Organomet Polym Mater. 2022;32(6):2221–34.10.1007/s10904-022-02293-1Search in Google Scholar
[47] Kothari R, Sharma D. Biofabrication of silver sulphide nanoparticles from cinnamomum tamala leaves: a next generation anti-inflammatory agent. World J Pharm Res. 2020;9(5):1918–39.Search in Google Scholar
[48] Wang G, Liu J, Zhu L, Guo Y, Yang L. Silver sulfide nanoparticles for photodynamic therapy of human lymphoma cells via disruption of energy metabolism. RSC Adv. 2019;9(51):29936–41.10.1039/C9RA05432DSearch in Google Scholar
[49] Xaba T, Moloto M, Nchoe O, Nate Z, Moloto N. Synthesis of silver sulfide nanoparticles through homogeneous precipitation route and the preparation of the Ag 2 S-chitosan nanocomposites for the removal of iron (ii) ion from wastewater. Chalcogenide Lett. 2017;14(8):337–46.Search in Google Scholar
[50] Zargar M, Hamid AA, Bakar FA, Shamsudin MN, Shameli K, Jahanshiri F, et al. Green synthesis and antibacterial effect of silver nanoparticles using Vitex negundo L. Molecules. 2011;16(8):6667–76.10.3390/molecules16086667Search in Google Scholar PubMed PubMed Central
[51] Bibi N, Ali Q, Tanveer ZI, Rahman H, Anees M. Antibacterial efficacy of silver nanoparticles prepared using Fagonia cretica L. leaf extract. Inorg Nano-Metal Chem. 2019;49(8):260–6.10.1080/24701556.2019.1661440Search in Google Scholar
[52] Wan Mat Khalir WKA, Shameli K, Jazayeri SD, Othman NA, Che Jusoh NW, Hassan NM. Biosynthesized silver nanoparticles by aqueous stem extract of Entada spiralis and screening of their biomedical activity. Front Chem. 2020;8:620.10.3389/fchem.2020.00620Search in Google Scholar PubMed PubMed Central
[53] Hemmalakshmi S, Priyanga S, Devaki K. Fourier Transform Infra-Red Spectroscopy Analysis of Erythrina variegata L. J Pharm Sci Res. 2017;9(11):2062–7.Search in Google Scholar
[54] Salem NM, Abedalaziz FM, Awwad AM. Nanoparticles and antifungal activity of iron sulfide synthesized by green route using Uncaria tomentosa leaves extract. Glob Adv Res J Agric Sci. 2018;7(6):176–82.Search in Google Scholar
[55] OgwekpeEgbeneje V, Hungwa AG, Dooshima S, Okhale SE, Ogbogo I, Ojo O. Green synthesis, characterisation and antibacterial activity evaluation of silver sulfide nanoparticles of Senna occidentalis (L.). Int J Pharmacogn. 2022;9(6):116–22.Search in Google Scholar
[56] Rao YS, Kotakadi VS, Prasad T, Reddy AV, Gopal DS. Green synthesis and spectral characterization of silver nanoparticles from Lakshmi tulasi (Ocimum sanctum) leaf extract. Spectrochim Acta Part A: Mol Biomol Spectrosc. 2013;103:156–9.10.1016/j.saa.2012.11.028Search in Google Scholar PubMed
[57] Salem NM, Albanna LS, Awwad AM. Green synthesis of sulfur nanoparticles using Punica granatum peels and the effects on the growth of tomato by foliar spray applications. Environ Nanotechnol, Monit & Manag. 2016;6:83–7.10.1016/j.enmm.2016.06.006Search in Google Scholar
[58] Vankudoth S, Dharavath S, Veera S, Maduru N, Chada R, Chirumamilla P, et al. Green synthesis, characterization, photoluminescence and biological studies of silver nanoparticles from the leaf extract of Muntingia calabura. Biochem Biophys Res Commun. 2022;630:143–50.10.1016/j.bbrc.2022.09.054Search in Google Scholar PubMed
[59] Fakhri A, Pourmand M, Khakpour R, Behrouz S. Structural, optical, photoluminescence and antibacterial properties of copper-doped silver sulfide nanoparticles. J Photochem Photobiol B: Biol. 2015;149:78–83.10.1016/j.jphotobiol.2015.05.013Search in Google Scholar PubMed
[60] Mukwevho E, Ferreira Z, Ayeleso A. Potential role of sulfur-containing antioxidant systems in highly oxidative environments. Molecules. 2014;19(12):19376–89. 10.3390/molecules191219376. From NLM.Search in Google Scholar
[61] Khan I, Saeed K, Khan I. Nanoparticles: properties, applications and toxicities. Arab J Chem. 2019;12(7):908–31.10.1016/j.arabjc.2017.05.011Search in Google Scholar
[62] Salayová A, Bedlovičová Z, Daneu N, Baláž M, Lukáčová Bujňáková Z, Balážová Ľ, et al. Green synthesis of silver nanoparticles with antibacterial activity using various medicinal plant extracts: morphology and antibacterial efficacy. Nanomaterials. 2021;11(4):1005.10.3390/nano11041005Search in Google Scholar PubMed PubMed Central
[63] Mohamed AA, Fouda A, Abdel-Rahman MA, Hassan SE-D, El-Gamal MS, Salem SS, et al. Fungal strain impacts the shape, bioactivity and multifunctional properties of green synthesized zinc oxide nanoparticles. Biocatal Agric Biotechnol. 2019;19:101103.10.1016/j.bcab.2019.101103Search in Google Scholar
[64] Bala Subramaniyan S, Megarajan S, Vijayakumar S, Mariappan M, Anbazhagan V. Evaluation of the toxicities of silver and silver sulfide nanoparticles against Gram-positive and Gram-negative bacteria. IET Nanobiotechnol. 2019;13(3):326–31.10.1049/iet-nbt.2018.5221Search in Google Scholar PubMed PubMed Central
[65] Bapat RA, Chaubal TV, Joshi CP, Bapat PR, Choudhury H, Pandey M, et al. An overview of application of silver nanoparticles for biomaterials in dentistry. Mater Sci Eng: C. 2018;91:881–98.10.1016/j.msec.2018.05.069Search in Google Scholar PubMed
[66] Kaufer A, Ellis J, Stark D, Barratt J. The evolution of trypanosomatid taxonomy. Parasites vectors. 2017;10:1–17.10.1186/s13071-017-2204-7Search in Google Scholar PubMed PubMed Central
[67] Lockard RD, Wilson ME, Rodríguez NE. Sex-related differences in immune response and symptomatic manifestations to infection with Leishmania species. J Immunol Res. 2019;2019:1–14.10.1155/2019/4103819Search in Google Scholar PubMed PubMed Central
[68] Choi HL, Jain S, Ruiz Postigo JA, Borisch B, Dagne DA. The global procurement landscape of leishmaniasis medicines. PLoS Neglected Trop Dis. 2021;15(2):e0009181.10.1371/journal.pntd.0009181Search in Google Scholar PubMed PubMed Central
[69] Torrado JJ, Cuquerella M, Olías-Molero AI, de la Fuente C, Alunda JM. Antileishmanial drug discovery and development: time to reset the model? Microorganisms. 2021.10.3390/microorganisms9122500Search in Google Scholar PubMed PubMed Central
[70] Knight CA, Harris DR, Alshammari SO, Gugssa A, Young T, Lee CM. Leishmaniasis: Recent epidemiological studies in the Middle East. Front Microbiol. 2023;13:5160.10.3389/fmicb.2022.1052478Search in Google Scholar PubMed PubMed Central
[71] Reithinger R, Dujardin J-C, Louzir H, Pirmez C, Alexander B, Brooker S. Cutaneous leishmaniasis-Reply. Lancet Infect Dis. 2008;8(8):458–9.10.1016/S1473-3099(08)70160-9Search in Google Scholar
[72] Rossi M, Fasel N. How to master the host immune system? Leishmania parasites have the solutions! Int Immunol. 2018;30(3):103–11.10.1093/intimm/dxx075Search in Google Scholar PubMed PubMed Central
[73] Guerra RO, do Carmo Neto JR, de Albuquerque Martins T, Farnesi de-Assunção TS, Junior VR, De Oliveira CJF, et al. Metallic nanoparticles: a new frontier in the fight against leishmaniasis. Curr Med Chem. 2022;29(26):4547–73.10.2174/0929867329666220225111052Search in Google Scholar PubMed
[74] Anjum F, Razvi N, Maqbool A, Jahan N. A review of breast cancer risk factors. Univ J Pharm Res. 2017;2(5):36–41.10.22270/ujpr.v2i5.RW2Search in Google Scholar
[75] Ha AW, Hong KH, Kim HS, Kim WK. Inorganic sulfur reduces cell proliferation by inhibiting of ErbB2 and ErbB3 protein and mRNA expression in MDA-MB-231 human breast cancer cells. Nutr Res Pract. 2013;7(2):89–95.10.4162/nrp.2013.7.2.89Search in Google Scholar PubMed PubMed Central
[76] Tortella G, Rubilar O, Durán N, Diez M, Martínez M, Parada J, et al. Silver nanoparticles: Toxicity in model organisms as an overview of its hazard for human health and the environment. J Hazard Mater. 2020;390:121974.10.1016/j.jhazmat.2019.121974Search in Google Scholar PubMed
[77] Wang Y, Chinnathambi A, Nasif O, Alharbi SA. Green synthesis and chemical characterization of a novel anti-human pancreatic cancer supplement by silver nanoparticles containing Zingiber officinale leaf aqueous extract. Arab J Chem. 2021;14(4):103081.10.1016/j.arabjc.2021.103081Search in Google Scholar
[78] Amaral JD, Xavier JM, Steer CJ, Rodrigues CM. The role of p53 in apoptosis. Discovery Med. 2010;9(45):145–52.Search in Google Scholar
[79] Eom H-J, Choi J. p38 MAPK activation, DNA damage, cell cycle arrest and apoptosis as mechanisms of toxicity of silver nanoparticles in Jurkat T cells. Environ Sci Technol. 2010;44(21):8337–42.10.1021/es1020668Search in Google Scholar PubMed
[80] Ovais M, Khalil AT, Raza A, Khan MA, Ahmad I, Islam NU, et al. Green synthesis of silver nanoparticles via plant extracts: beginning a new era in cancer theranostics. Nanomedicine. 2016;11(23):3157–77.10.2217/nnm-2016-0279Search in Google Scholar PubMed
[81] Haseeb HA, Khan MA, Rasheed H, Zahid MU, Doan TD, Siddique MAR, et al. Polygonum bistorta Linn. as a green source for synthesis of biocompatible selenium nanoparticles with potent antimicrobial and antioxidant properties. BioMetals. 2024;37:1–17.10.1007/s10534-024-00622-0Search in Google Scholar PubMed
© 2024 the author(s), published by De Gruyter
This work is licensed under the Creative Commons Attribution 4.0 International License.
Articles in the same Issue
- Research Articles
- Green polymer electrolyte and activated charcoal-based supercapacitor for energy harvesting application: Electrochemical characteristics
- Research on the adsorption of Co2+ ions using halloysite clay and the ability to recover them by electrodeposition method
- Simultaneous estimation of ibuprofen, caffeine, and paracetamol in commercial products using a green reverse-phase HPTLC method
- Isolation, screening and optimization of alkaliphilic cellulolytic fungi for production of cellulase
- Functionalized gold nanoparticles coated with bacterial alginate and their antibacterial and anticancer activities
- Comparative analysis of bio-based amino acid surfactants obtained via Diels–Alder reaction of cyclic anhydrides
- Biosynthesis of silver nanoparticles on yellow phosphorus slag and its application in organic coatings
- Exploring antioxidant potential and phenolic compound extraction from Vitis vinifera L. using ultrasound-assisted extraction
- Manganese and copper-coated nickel oxide nanoparticles synthesized from Carica papaya leaf extract induce antimicrobial activity and breast cancer cell death by triggering mitochondrial caspases and p53
- Insight into heating method and Mozafari method as green processing techniques for the synthesis of micro- and nano-drug carriers
- Silicotungstic acid supported on Bi-based MOF-derived metal oxide for photodegradation of organic dyes
- Synthesis and characterization of capsaicin nanoparticles: An attempt to enhance its bioavailability and pharmacological actions
- Synthesis of Lawsonia inermis-encased silver–copper bimetallic nanoparticles with antioxidant, antibacterial, and cytotoxic activity
- Facile, polyherbal drug-mediated green synthesis of CuO nanoparticles and their potent biological applications
- Zinc oxide-manganese oxide/carboxymethyl cellulose-folic acid-sesamol hybrid nanomaterials: A molecularly targeted strategy for advanced triple-negative breast cancer therapy
- Exploring the antimicrobial potential of biogenically synthesized graphene oxide nanoparticles against targeted bacterial and fungal pathogens
- Biofabrication of silver nanoparticles using Uncaria tomentosa L.: Insight into characterization, antibacterial activities combined with antibiotics, and effect on Triticum aestivum germination
- Membrane distillation of synthetic urine for use in space structural habitat systems
- Investigation on mechanical properties of the green synthesis bamboo fiber/eggshell/coconut shell powder-based hybrid biocomposites under NaOH conditions
- Green synthesis of magnesium oxide nanoparticles using endophytic fungal strain to improve the growth, metabolic activities, yield traits, and phenolic compounds content of Nigella sativa L.
- Estimation of greenhouse gas emissions from rice and annual upland crops in Red River Delta of Vietnam using the denitrification–decomposition model
- Synthesis of humic acid with the obtaining of potassium humate based on coal waste from the Lenger deposit, Kazakhstan
- Ascorbic acid-mediated selenium nanoparticles as potential antihyperuricemic, antioxidant, anticoagulant, and thrombolytic agents
- Green synthesis of silver nanoparticles using Illicium verum extract: Optimization and characterization for biomedical applications
- Antibacterial and dynamical behaviour of silicon nanoparticles influenced sustainable waste flax fibre-reinforced epoxy composite for biomedical application
- Optimising coagulation/flocculation using response surface methodology and application of floc in biofertilisation
- Green synthesis and multifaceted characterization of iron oxide nanoparticles derived from Senna bicapsularis for enhanced in vitro and in vivo biological investigation
- Potent antibacterial nanocomposites from okra mucilage/chitosan/silver nanoparticles for multidrug-resistant Salmonella Typhimurium eradication
- Trachyspermum copticum aqueous seed extract-derived silver nanoparticles: Exploration of their structural characterization and comparative antibacterial performance against gram-positive and gram-negative bacteria
- Microwave-assisted ultrafine silver nanoparticle synthesis using Mitragyna speciosa for antimalarial applications
- Green synthesis and characterisation of spherical structure Ag/Fe2O3/TiO2 nanocomposite using acacia in the presence of neem and tulsi oils
- Green quantitative methods for linagliptin and empagliflozin in dosage forms
- Enhancement efficacy of omeprazole by conjugation with silver nanoparticles as a urease inhibitor
- Residual, sequential extraction, and ecological risk assessment of some metals in ash from municipal solid waste incineration, Vietnam
- Green synthesis of ZnO nanoparticles using the mangosteen (Garcinia mangostana L.) leaf extract: Comparative preliminary in vitro antibacterial study
- Simultaneous determination of lesinurad and febuxostat in commercial fixed-dose combinations using a greener normal-phase HPTLC method
- A greener RP-HPLC method for quaternary estimation of caffeine, paracetamol, levocetirizine, and phenylephrine acquiring AQbD with stability studies
- Optimization of biomass durian peel as a heterogeneous catalyst in biodiesel production using microwave irradiation
- Thermal treatment impact on the evolution of active phases in layered double hydroxide-based ZnCr photocatalysts: Photodegradation and antibacterial performance
- Preparation of silymarin-loaded zein polysaccharide core–shell nanostructures and evaluation of their biological potentials
- Preparation and characterization of composite-modified PA6 fiber for spectral heating and heat storage applications
- Preparation and electrocatalytic oxygen evolution of bimetallic phosphates (NiFe)2P/NF
- Rod-shaped Mo(vi) trichalcogenide–Mo(vi) oxide decorated on poly(1-H pyrrole) as a promising nanocomposite photoelectrode for green hydrogen generation from sewage water with high efficiency
- Green synthesis and studies on citrus medica leaf extract-mediated Au–ZnO nanocomposites: A sustainable approach for efficient photocatalytic degradation of rhodamine B dye in aqueous media
- Cellulosic materials for the removal of ciprofloxacin from aqueous environments
- The analytical assessment of metal contamination in industrial soils of Saudi Arabia using the inductively coupled plasma technology
- The effect of modified oily sludge on the slurry ability and combustion performance of coal water slurry
- Eggshell waste transformation to calcium chloride anhydride as food-grade additive and eggshell membranes as enzyme immobilization carrier
- Synthesis of EPAN and applications in the encapsulation of potassium humate
- Biosynthesis and characterization of silver nanoparticles from Cedrela toona leaf extracts: An exploration into their antibacterial, anticancer, and antioxidant potential
- Enhancing mechanical and rheological properties of HDPE films through annealing for eco-friendly agricultural applications
- Immobilisation of catalase purified from mushroom (Hydnum repandum) onto glutaraldehyde-activated chitosan and characterisation: Its application for the removal of hydrogen peroxide from artificial wastewater
- Sodium titanium oxide/zinc oxide (STO/ZnO) photocomposites for efficient dye degradation applications
- Effect of ex situ, eco-friendly ZnONPs incorporating green synthesised Moringa oleifera leaf extract in enhancing biochemical and molecular aspects of Vicia faba L. under salt stress
- Biosynthesis and characterization of selenium and silver nanoparticles using Trichoderma viride filtrate and their impact on Culex pipiens
- Photocatalytic degradation of organic dyes and biological potentials of biogenic zinc oxide nanoparticles synthesized using the polar extract of Cyperus scariosus R.Br. (Cyperaceae)
- Assessment of antiproliferative activity of green-synthesized nickel oxide nanoparticles against glioblastoma cells using Terminalia chebula
- Chlorine-free synthesis of phosphinic derivatives by change in the P-function
- Anticancer, antioxidant, and antimicrobial activities of nanoemulsions based on water-in-olive oil and loaded on biogenic silver nanoparticles
- Study and mechanism of formation of phosphorus production waste in Kazakhstan
- Synthesis and stabilization of anatase form of biomimetic TiO2 nanoparticles for enhancing anti-tumor potential
- Microwave-supported one-pot reaction for the synthesis of 5-alkyl/arylidene-2-(morpholin/thiomorpholin-4-yl)-1,3-thiazol-4(5H)-one derivatives over MgO solid base
- Screening the phytochemicals in Perilla leaves and phytosynthesis of bioactive silver nanoparticles for potential antioxidant and wound-healing application
- Graphene oxide/chitosan/manganese/folic acid-brucine functionalized nanocomposites show anticancer activity against liver cancer cells
- Nature of serpentinite interactions with low-concentration sulfuric acid solutions
- Multi-objective statistical optimisation utilising response surface methodology to predict engine performance using biofuels from waste plastic oil in CRDi engines
- Microwave-assisted extraction of acetosolv lignin from sugarcane bagasse and electrospinning of lignin/PEO nanofibres for carbon fibre production
- Biosynthesis, characterization, and investigation of cytotoxic activities of selenium nanoparticles utilizing Limosilactobacillus fermentum
- Highly photocatalytic materials based on the decoration of poly(O-chloroaniline) with molybdenum trichalcogenide oxide for green hydrogen generation from Red Sea water
- Highly efficient oil–water separation using superhydrophobic cellulose aerogels derived from corn straw
- Beta-cyclodextrin–Phyllanthus emblica emulsion for zinc oxide nanoparticles: Characteristics and photocatalysis
- Assessment of antimicrobial activity and methyl orange dye removal by Klebsiella pneumoniae-mediated silver nanoparticles
- Influential eradication of resistant Salmonella Typhimurium using bioactive nanocomposites from chitosan and radish seed-synthesized nanoselenium
- Antimicrobial activities and neuroprotective potential for Alzheimer’s disease of pure, Mn, Co, and Al-doped ZnO ultra-small nanoparticles
- Green synthesis of silver nanoparticles from Bauhinia variegata and their biological applications
- Synthesis and optimization of long-chain fatty acids via the oxidation of long-chain fatty alcohols
- Eminent Red Sea water hydrogen generation via a Pb(ii)-iodide/poly(1H-pyrrole) nanocomposite photocathode
- Green synthesis and effective genistein production by fungal β-glucosidase immobilized on Al2O3 nanocrystals synthesized in Cajanus cajan L. (Millsp.) leaf extracts
- Green stability-indicating RP-HPTLC technique for determining croconazole hydrochloride
- Green synthesis of La2O3–LaPO4 nanocomposites using Charybdis natator for DNA binding, cytotoxic, catalytic, and luminescence applications
- Eco-friendly drugs induce cellular changes in colistin-resistant bacteria
- Tangerine fruit peel extract mediated biogenic synthesized silver nanoparticles and their potential antimicrobial, antioxidant, and cytotoxic assessments
- Green synthesis on performance characteristics of a direct injection diesel engine using sandbox seed oil
- A highly sensitive β-AKBA-Ag-based fluorescent “turn off” chemosensor for rapid detection of abamectin in tomatoes
- Green synthesis and physical characterization of zinc oxide nanoparticles (ZnO NPs) derived from the methanol extract of Euphorbia dracunculoides Lam. (Euphorbiaceae) with enhanced biosafe applications
- Detection of morphine and data processing using surface plasmon resonance imaging sensor
- Effects of nanoparticles on the anaerobic digestion properties of sulfamethoxazole-containing chicken manure and analysis of bio-enzymes
- Bromic acid-thiourea synergistic leaching of sulfide gold ore
- Green chemistry approach to synthesize titanium dioxide nanoparticles using Fagonia Cretica extract, novel strategy for developing antimicrobial and antidiabetic therapies
- Green synthesis and effective utilization of biogenic Al2O3-nanocoupled fungal lipase in the resolution of active homochiral 2-octanol and its immobilization via aluminium oxide nanoparticles
- Eco-friendly RP-HPLC approach for simultaneously estimating the promising combination of pentoxifylline and simvastatin in therapeutic potential for breast cancer: Appraisal of greenness, whiteness, and Box–Behnken design
- Use of a humidity adsorbent derived from cockleshell waste in Thai fried fish crackers (Keropok)
- One-pot green synthesis, biological evaluation, and in silico study of pyrazole derivatives obtained from chalcones
- Bio-sorption of methylene blue and production of biofuel by brown alga Cystoseira sp. collected from Neom region, Kingdom of Saudi Arabia
- Synthesis of motexafin gadolinium: A promising radiosensitizer and imaging agent for cancer therapy
- The impact of varying sizes of silver nanoparticles on the induction of cellular damage in Klebsiella pneumoniae involving diverse mechanisms
- Microwave-assisted green synthesis, characterization, and in vitro antibacterial activity of NiO nanoparticles obtained from lemon peel extract
- Rhus microphylla-mediated biosynthesis of copper oxide nanoparticles for enhanced antibacterial and antibiofilm efficacy
- Harnessing trichalcogenide–molybdenum(vi) sulfide and molybdenum(vi) oxide within poly(1-amino-2-mercaptobenzene) frameworks as a photocathode for sustainable green hydrogen production from seawater without sacrificial agents
- Magnetically recyclable Fe3O4@SiO2 supported phosphonium ionic liquids for efficient and sustainable transformation of CO2 into oxazolidinones
- A comparative study of Fagonia arabica fabricated silver sulfide nanoparticles (Ag2S) and silver nanoparticles (AgNPs) with distinct antimicrobial, anticancer, and antioxidant properties
- Visible light photocatalytic degradation and biological activities of Aegle marmelos-mediated cerium oxide nanoparticles
- Physical intrinsic characteristics of spheroidal particles in coal gasification fine slag
- Exploring the effect of tea dust magnetic biochar on agricultural crops grown in polycyclic aromatic hydrocarbon contaminated soil
- Crosslinked chitosan-modified ultrafiltration membranes for efficient surface water treatment and enhanced anti-fouling performances
- Study on adsorption characteristics of biochars and their modified biochars for removal of organic dyes from aqueous solution
- Zein polymer nanocarrier for Ocimum basilicum var. purpurascens extract: Potential biomedical use
- Green synthesis, characterization, and in vitro and in vivo biological screening of iron oxide nanoparticles (Fe3O4) generated with hydroalcoholic extract of aerial parts of Euphorbia milii
- Novel microwave-based green approach for the synthesis of dual-loaded cyclodextrin nanosponges: Characterization, pharmacodynamics, and pharmacokinetics evaluation
- Bi2O3–BiOCl/poly-m-methyl aniline nanocomposite thin film for broad-spectrum light-sensing
- Green synthesis and characterization of CuO/ZnO nanocomposite using Musa acuminata leaf extract for cytotoxic studies on colorectal cancer cells (HCC2998)
- Review Articles
- Materials-based drug delivery approaches: Recent advances and future perspectives
- A review of thermal treatment for bamboo and its composites
- An overview of the role of nanoherbicides in tackling challenges of weed management in wheat: A novel approach
- An updated review on carbon nanomaterials: Types, synthesis, functionalization and applications, degradation and toxicity
- Special Issue: Emerging green nanomaterials for sustainable waste management and biomedical applications
- Green synthesis of silver nanoparticles using mature-pseudostem extracts of Alpinia nigra and their bioactivities
- Special Issue: New insights into nanopythotechnology: current trends and future prospects
- Green synthesis of FeO nanoparticles from coffee and its application for antibacterial, antifungal, and anti-oxidation activity
- Dye degradation activity of biogenically synthesized Cu/Fe/Ag trimetallic nanoparticles
- Special Issue: Composites and green composites
- Recent trends and advancements in the utilization of green composites and polymeric nanocarriers for enhancing food quality and sustainable processing
- Retraction
- Retraction of “Biosynthesis and characterization of silver nanoparticles from Cedrela toona leaf extracts: An exploration into their antibacterial, anticancer, and antioxidant potential”
- Retraction of “Photocatalytic degradation of organic dyes and biological potentials of biogenic zinc oxide nanoparticles synthesized using the polar extract of Cyperus scariosus R.Br. (Cyperaceae)”
- Retraction to “Green synthesis on performance characteristics of a direct injection diesel engine using sandbox seed oil”
Articles in the same Issue
- Research Articles
- Green polymer electrolyte and activated charcoal-based supercapacitor for energy harvesting application: Electrochemical characteristics
- Research on the adsorption of Co2+ ions using halloysite clay and the ability to recover them by electrodeposition method
- Simultaneous estimation of ibuprofen, caffeine, and paracetamol in commercial products using a green reverse-phase HPTLC method
- Isolation, screening and optimization of alkaliphilic cellulolytic fungi for production of cellulase
- Functionalized gold nanoparticles coated with bacterial alginate and their antibacterial and anticancer activities
- Comparative analysis of bio-based amino acid surfactants obtained via Diels–Alder reaction of cyclic anhydrides
- Biosynthesis of silver nanoparticles on yellow phosphorus slag and its application in organic coatings
- Exploring antioxidant potential and phenolic compound extraction from Vitis vinifera L. using ultrasound-assisted extraction
- Manganese and copper-coated nickel oxide nanoparticles synthesized from Carica papaya leaf extract induce antimicrobial activity and breast cancer cell death by triggering mitochondrial caspases and p53
- Insight into heating method and Mozafari method as green processing techniques for the synthesis of micro- and nano-drug carriers
- Silicotungstic acid supported on Bi-based MOF-derived metal oxide for photodegradation of organic dyes
- Synthesis and characterization of capsaicin nanoparticles: An attempt to enhance its bioavailability and pharmacological actions
- Synthesis of Lawsonia inermis-encased silver–copper bimetallic nanoparticles with antioxidant, antibacterial, and cytotoxic activity
- Facile, polyherbal drug-mediated green synthesis of CuO nanoparticles and their potent biological applications
- Zinc oxide-manganese oxide/carboxymethyl cellulose-folic acid-sesamol hybrid nanomaterials: A molecularly targeted strategy for advanced triple-negative breast cancer therapy
- Exploring the antimicrobial potential of biogenically synthesized graphene oxide nanoparticles against targeted bacterial and fungal pathogens
- Biofabrication of silver nanoparticles using Uncaria tomentosa L.: Insight into characterization, antibacterial activities combined with antibiotics, and effect on Triticum aestivum germination
- Membrane distillation of synthetic urine for use in space structural habitat systems
- Investigation on mechanical properties of the green synthesis bamboo fiber/eggshell/coconut shell powder-based hybrid biocomposites under NaOH conditions
- Green synthesis of magnesium oxide nanoparticles using endophytic fungal strain to improve the growth, metabolic activities, yield traits, and phenolic compounds content of Nigella sativa L.
- Estimation of greenhouse gas emissions from rice and annual upland crops in Red River Delta of Vietnam using the denitrification–decomposition model
- Synthesis of humic acid with the obtaining of potassium humate based on coal waste from the Lenger deposit, Kazakhstan
- Ascorbic acid-mediated selenium nanoparticles as potential antihyperuricemic, antioxidant, anticoagulant, and thrombolytic agents
- Green synthesis of silver nanoparticles using Illicium verum extract: Optimization and characterization for biomedical applications
- Antibacterial and dynamical behaviour of silicon nanoparticles influenced sustainable waste flax fibre-reinforced epoxy composite for biomedical application
- Optimising coagulation/flocculation using response surface methodology and application of floc in biofertilisation
- Green synthesis and multifaceted characterization of iron oxide nanoparticles derived from Senna bicapsularis for enhanced in vitro and in vivo biological investigation
- Potent antibacterial nanocomposites from okra mucilage/chitosan/silver nanoparticles for multidrug-resistant Salmonella Typhimurium eradication
- Trachyspermum copticum aqueous seed extract-derived silver nanoparticles: Exploration of their structural characterization and comparative antibacterial performance against gram-positive and gram-negative bacteria
- Microwave-assisted ultrafine silver nanoparticle synthesis using Mitragyna speciosa for antimalarial applications
- Green synthesis and characterisation of spherical structure Ag/Fe2O3/TiO2 nanocomposite using acacia in the presence of neem and tulsi oils
- Green quantitative methods for linagliptin and empagliflozin in dosage forms
- Enhancement efficacy of omeprazole by conjugation with silver nanoparticles as a urease inhibitor
- Residual, sequential extraction, and ecological risk assessment of some metals in ash from municipal solid waste incineration, Vietnam
- Green synthesis of ZnO nanoparticles using the mangosteen (Garcinia mangostana L.) leaf extract: Comparative preliminary in vitro antibacterial study
- Simultaneous determination of lesinurad and febuxostat in commercial fixed-dose combinations using a greener normal-phase HPTLC method
- A greener RP-HPLC method for quaternary estimation of caffeine, paracetamol, levocetirizine, and phenylephrine acquiring AQbD with stability studies
- Optimization of biomass durian peel as a heterogeneous catalyst in biodiesel production using microwave irradiation
- Thermal treatment impact on the evolution of active phases in layered double hydroxide-based ZnCr photocatalysts: Photodegradation and antibacterial performance
- Preparation of silymarin-loaded zein polysaccharide core–shell nanostructures and evaluation of their biological potentials
- Preparation and characterization of composite-modified PA6 fiber for spectral heating and heat storage applications
- Preparation and electrocatalytic oxygen evolution of bimetallic phosphates (NiFe)2P/NF
- Rod-shaped Mo(vi) trichalcogenide–Mo(vi) oxide decorated on poly(1-H pyrrole) as a promising nanocomposite photoelectrode for green hydrogen generation from sewage water with high efficiency
- Green synthesis and studies on citrus medica leaf extract-mediated Au–ZnO nanocomposites: A sustainable approach for efficient photocatalytic degradation of rhodamine B dye in aqueous media
- Cellulosic materials for the removal of ciprofloxacin from aqueous environments
- The analytical assessment of metal contamination in industrial soils of Saudi Arabia using the inductively coupled plasma technology
- The effect of modified oily sludge on the slurry ability and combustion performance of coal water slurry
- Eggshell waste transformation to calcium chloride anhydride as food-grade additive and eggshell membranes as enzyme immobilization carrier
- Synthesis of EPAN and applications in the encapsulation of potassium humate
- Biosynthesis and characterization of silver nanoparticles from Cedrela toona leaf extracts: An exploration into their antibacterial, anticancer, and antioxidant potential
- Enhancing mechanical and rheological properties of HDPE films through annealing for eco-friendly agricultural applications
- Immobilisation of catalase purified from mushroom (Hydnum repandum) onto glutaraldehyde-activated chitosan and characterisation: Its application for the removal of hydrogen peroxide from artificial wastewater
- Sodium titanium oxide/zinc oxide (STO/ZnO) photocomposites for efficient dye degradation applications
- Effect of ex situ, eco-friendly ZnONPs incorporating green synthesised Moringa oleifera leaf extract in enhancing biochemical and molecular aspects of Vicia faba L. under salt stress
- Biosynthesis and characterization of selenium and silver nanoparticles using Trichoderma viride filtrate and their impact on Culex pipiens
- Photocatalytic degradation of organic dyes and biological potentials of biogenic zinc oxide nanoparticles synthesized using the polar extract of Cyperus scariosus R.Br. (Cyperaceae)
- Assessment of antiproliferative activity of green-synthesized nickel oxide nanoparticles against glioblastoma cells using Terminalia chebula
- Chlorine-free synthesis of phosphinic derivatives by change in the P-function
- Anticancer, antioxidant, and antimicrobial activities of nanoemulsions based on water-in-olive oil and loaded on biogenic silver nanoparticles
- Study and mechanism of formation of phosphorus production waste in Kazakhstan
- Synthesis and stabilization of anatase form of biomimetic TiO2 nanoparticles for enhancing anti-tumor potential
- Microwave-supported one-pot reaction for the synthesis of 5-alkyl/arylidene-2-(morpholin/thiomorpholin-4-yl)-1,3-thiazol-4(5H)-one derivatives over MgO solid base
- Screening the phytochemicals in Perilla leaves and phytosynthesis of bioactive silver nanoparticles for potential antioxidant and wound-healing application
- Graphene oxide/chitosan/manganese/folic acid-brucine functionalized nanocomposites show anticancer activity against liver cancer cells
- Nature of serpentinite interactions with low-concentration sulfuric acid solutions
- Multi-objective statistical optimisation utilising response surface methodology to predict engine performance using biofuels from waste plastic oil in CRDi engines
- Microwave-assisted extraction of acetosolv lignin from sugarcane bagasse and electrospinning of lignin/PEO nanofibres for carbon fibre production
- Biosynthesis, characterization, and investigation of cytotoxic activities of selenium nanoparticles utilizing Limosilactobacillus fermentum
- Highly photocatalytic materials based on the decoration of poly(O-chloroaniline) with molybdenum trichalcogenide oxide for green hydrogen generation from Red Sea water
- Highly efficient oil–water separation using superhydrophobic cellulose aerogels derived from corn straw
- Beta-cyclodextrin–Phyllanthus emblica emulsion for zinc oxide nanoparticles: Characteristics and photocatalysis
- Assessment of antimicrobial activity and methyl orange dye removal by Klebsiella pneumoniae-mediated silver nanoparticles
- Influential eradication of resistant Salmonella Typhimurium using bioactive nanocomposites from chitosan and radish seed-synthesized nanoselenium
- Antimicrobial activities and neuroprotective potential for Alzheimer’s disease of pure, Mn, Co, and Al-doped ZnO ultra-small nanoparticles
- Green synthesis of silver nanoparticles from Bauhinia variegata and their biological applications
- Synthesis and optimization of long-chain fatty acids via the oxidation of long-chain fatty alcohols
- Eminent Red Sea water hydrogen generation via a Pb(ii)-iodide/poly(1H-pyrrole) nanocomposite photocathode
- Green synthesis and effective genistein production by fungal β-glucosidase immobilized on Al2O3 nanocrystals synthesized in Cajanus cajan L. (Millsp.) leaf extracts
- Green stability-indicating RP-HPTLC technique for determining croconazole hydrochloride
- Green synthesis of La2O3–LaPO4 nanocomposites using Charybdis natator for DNA binding, cytotoxic, catalytic, and luminescence applications
- Eco-friendly drugs induce cellular changes in colistin-resistant bacteria
- Tangerine fruit peel extract mediated biogenic synthesized silver nanoparticles and their potential antimicrobial, antioxidant, and cytotoxic assessments
- Green synthesis on performance characteristics of a direct injection diesel engine using sandbox seed oil
- A highly sensitive β-AKBA-Ag-based fluorescent “turn off” chemosensor for rapid detection of abamectin in tomatoes
- Green synthesis and physical characterization of zinc oxide nanoparticles (ZnO NPs) derived from the methanol extract of Euphorbia dracunculoides Lam. (Euphorbiaceae) with enhanced biosafe applications
- Detection of morphine and data processing using surface plasmon resonance imaging sensor
- Effects of nanoparticles on the anaerobic digestion properties of sulfamethoxazole-containing chicken manure and analysis of bio-enzymes
- Bromic acid-thiourea synergistic leaching of sulfide gold ore
- Green chemistry approach to synthesize titanium dioxide nanoparticles using Fagonia Cretica extract, novel strategy for developing antimicrobial and antidiabetic therapies
- Green synthesis and effective utilization of biogenic Al2O3-nanocoupled fungal lipase in the resolution of active homochiral 2-octanol and its immobilization via aluminium oxide nanoparticles
- Eco-friendly RP-HPLC approach for simultaneously estimating the promising combination of pentoxifylline and simvastatin in therapeutic potential for breast cancer: Appraisal of greenness, whiteness, and Box–Behnken design
- Use of a humidity adsorbent derived from cockleshell waste in Thai fried fish crackers (Keropok)
- One-pot green synthesis, biological evaluation, and in silico study of pyrazole derivatives obtained from chalcones
- Bio-sorption of methylene blue and production of biofuel by brown alga Cystoseira sp. collected from Neom region, Kingdom of Saudi Arabia
- Synthesis of motexafin gadolinium: A promising radiosensitizer and imaging agent for cancer therapy
- The impact of varying sizes of silver nanoparticles on the induction of cellular damage in Klebsiella pneumoniae involving diverse mechanisms
- Microwave-assisted green synthesis, characterization, and in vitro antibacterial activity of NiO nanoparticles obtained from lemon peel extract
- Rhus microphylla-mediated biosynthesis of copper oxide nanoparticles for enhanced antibacterial and antibiofilm efficacy
- Harnessing trichalcogenide–molybdenum(vi) sulfide and molybdenum(vi) oxide within poly(1-amino-2-mercaptobenzene) frameworks as a photocathode for sustainable green hydrogen production from seawater without sacrificial agents
- Magnetically recyclable Fe3O4@SiO2 supported phosphonium ionic liquids for efficient and sustainable transformation of CO2 into oxazolidinones
- A comparative study of Fagonia arabica fabricated silver sulfide nanoparticles (Ag2S) and silver nanoparticles (AgNPs) with distinct antimicrobial, anticancer, and antioxidant properties
- Visible light photocatalytic degradation and biological activities of Aegle marmelos-mediated cerium oxide nanoparticles
- Physical intrinsic characteristics of spheroidal particles in coal gasification fine slag
- Exploring the effect of tea dust magnetic biochar on agricultural crops grown in polycyclic aromatic hydrocarbon contaminated soil
- Crosslinked chitosan-modified ultrafiltration membranes for efficient surface water treatment and enhanced anti-fouling performances
- Study on adsorption characteristics of biochars and their modified biochars for removal of organic dyes from aqueous solution
- Zein polymer nanocarrier for Ocimum basilicum var. purpurascens extract: Potential biomedical use
- Green synthesis, characterization, and in vitro and in vivo biological screening of iron oxide nanoparticles (Fe3O4) generated with hydroalcoholic extract of aerial parts of Euphorbia milii
- Novel microwave-based green approach for the synthesis of dual-loaded cyclodextrin nanosponges: Characterization, pharmacodynamics, and pharmacokinetics evaluation
- Bi2O3–BiOCl/poly-m-methyl aniline nanocomposite thin film for broad-spectrum light-sensing
- Green synthesis and characterization of CuO/ZnO nanocomposite using Musa acuminata leaf extract for cytotoxic studies on colorectal cancer cells (HCC2998)
- Review Articles
- Materials-based drug delivery approaches: Recent advances and future perspectives
- A review of thermal treatment for bamboo and its composites
- An overview of the role of nanoherbicides in tackling challenges of weed management in wheat: A novel approach
- An updated review on carbon nanomaterials: Types, synthesis, functionalization and applications, degradation and toxicity
- Special Issue: Emerging green nanomaterials for sustainable waste management and biomedical applications
- Green synthesis of silver nanoparticles using mature-pseudostem extracts of Alpinia nigra and their bioactivities
- Special Issue: New insights into nanopythotechnology: current trends and future prospects
- Green synthesis of FeO nanoparticles from coffee and its application for antibacterial, antifungal, and anti-oxidation activity
- Dye degradation activity of biogenically synthesized Cu/Fe/Ag trimetallic nanoparticles
- Special Issue: Composites and green composites
- Recent trends and advancements in the utilization of green composites and polymeric nanocarriers for enhancing food quality and sustainable processing
- Retraction
- Retraction of “Biosynthesis and characterization of silver nanoparticles from Cedrela toona leaf extracts: An exploration into their antibacterial, anticancer, and antioxidant potential”
- Retraction of “Photocatalytic degradation of organic dyes and biological potentials of biogenic zinc oxide nanoparticles synthesized using the polar extract of Cyperus scariosus R.Br. (Cyperaceae)”
- Retraction to “Green synthesis on performance characteristics of a direct injection diesel engine using sandbox seed oil”

