Abstract
In continuation of our efforts to fully utilize eggshell waste (ESW), here we report the possibility of ESW transformation to calcium chloride (CaCl2) anhydride of food-grade additive purity and eggshell membranes (ESMs) as potential enzyme immobilization carriers. ESW chemical transformation by 5% (w/v) hydrochloric acid to CaCl2 solution and ESM completely devoid of the remnants of ESW calcium carbonate was performed in the constructed 15 L batch reactor during 4 h at room temperature, followed by separation of ESM from CaCl2 solution by filtration. ESW-derived CaCl2 solution containing the excess hydrochloric acid was neutralized by adding calcium hydroxide, concentrated to approximately 1/8th of volume, and spray dried. Separated ESM was washed with water and acetone, dried, and ground to a size of less than 0.5 mm. The ESW transformation process produced 102.42 ± 3.31 g of CaCl2 anhydrous and 2.48 ± 0.28 g of ESM per 100 g of ESW dry matter. ESW-derived CaCl2 fulfilled all criteria for food-grade additive, while obtained ESM showed their suitability for Burkholderia cepacia lipase immobilization by adsorption.
Graphical abstract

1 Introduction
Calcium chloride (CaCl2) is an inorganic salt of calcium and chlorine, registered in the European Union (EU) under the Regulation on the Registration, Evaluation, Authorisation and Restriction of Chemicals (REACH), whose manufacturing and/or import to the European Economic Area comprise about 100,000 tons annually [1]. It has been used in road deicing, dust control, road stabilization, concrete curing, oil well drilling, tire ballasting, for the production of washing and cleaning products, anti-freeze products, fertilizers, plant protection products, adsorbents, water treatment chemicals, and heat transfer fluids, as well as food-grade additive [2,3,4,5].
Currently, the industrial production of CaCl2 is based on three different processes: (a) hydrochloric acid treatment of limestone as the dominant one, where CaCl2 is produced by chemical reaction between limestone calcium carbonate and hydrochloric acid; (b) Solvay process where the reaction between sodium chloride and calcium carbonate in the presence of ammonia as the catalyst results with sodium carbonate as the main product and CaCl2 as a by-product of the chemical reaction; and by (c) purification of naturally occurring brine CaCl2 (such as those one present in salt lakes) [2,3,4,5].
Besides limestone, which is a natural and geologically created deposit of calcium carbonate in the form of calcite crystals, another natural form of calcite crystal is the eggshell [6], accounting for the major part of eggshell waste (ESW) produced by households, restaurants, and various industries [7,8,9]. Based on the latest available report of the Food Agricultural Organization (FAO) on hen egg production for the year 2021 [10], and the most conservative estimate that ESW accounts for 10% of total egg mass [8,9], it is estimated that about 8,638,779 tons of ESW are generated worldwide, with 1,111,751 tons in the EU. Moreover, considering the fact that about 30% of totally generated ESW in the industrialized countries are the by-product of egg-breaking plants [11], it can be estimated that about 2,591,634 tons of worldwide generated ESW and about 333,525 tons of ESW in the EU has been generated by egg-breaking plants. Since the disposal of ESW generated by egg-breaking plants is quite costly [7,8,11], there is an obvious need to reduce such costs by implementing at least some of the strategies for ESW transformation. These include ESW transformation to the low-value-added products such as fertilizers or feed for animals and pets [7,12,13], or implementation of novel strategies of sustainable waste management oriented toward zero-waste model, where innovative waste transformation techniques for the production of chemicals, fine chemicals, bioactive compounds, enzymes, and functional materials have been applied [7,8,9,14].
Besides well-defined possibilities of ESW transformation to the value-added products reported in Cordeiro and Hincke [15], Waheed et al. [16], and Ahmed et al. [11], we would like to point out two recently published articles of our research group, where the possibility of the complete utilization of ESW collected from household and restaurants by chemical treatment with 5% (w/v) hydrochloric, 10% acetic (w/v), and 15% o-phosphoric acid (w/v) to the corresponding calcium salts and ESW-derived eggshell membranes (ESMs) were performed on a laboratory scale [8,9]. Therefore, it seems quite possible to extend current strategies of the ESW transformation process to the novel ones, more oriented toward zero-waste model of the complete ESW utilization.
In this regard, we have examined the possibility of ESW transformation by 5% (w/v) hydrochloric acid treatment upscaling in the constructed 15 L conical batch reactor, followed by the subsequent production of CaCl2 anhydride by spray drying, and ESM-derived ESMs as potential enzyme immobilization carrier. ESW-derived CaCl2 anhydride was tested on its suitability as food-grade additive, while eggshell-derived ESM was tested on the key characteristics as a potential enzyme immobilization carrier, as well as its suitability to be used as an enzyme carrier for Burkholderia cepacia lipase (BCL) immobilization by adsorption.
2 Materials and methods
2.1 Materials
Industrial ESW was generously supplied by Elcon-Nutritional Products Ltd. (Zlatar Bistrica, Croatia).
The transformation process of ESW was performed by 5% (w/v) HCl prepared from hydrochloric acid (37% w/v) purchased from Carlo Erba (Emmendingen, Germany), while the excess of hydrochloric acid in the obtained CaCl2 solution was neutralized by adding calcium hydroxide purchased from Across Organics (Geel, Belgium). Complexometric determination of calcium content was performed using calconcarboxylic acid obtained from Acros Organics (Geel, Belgium) and the disodium salt of ethylenediaminetetraacetic acid from Fischer Scientific (Leicestershire, UK).
Amano lipase PS from Burkholderia cepacia (Sigma–Aldrich product number: 534641) used for immobilization on the obtained ESMs was purchased from Sigma–Aldrich Chemicals (Saint Louis, MO, USA).
All other chemicals used in this research were of pro-analysis purity.
2.2 Chemical analysis of ESW
Chemical analysis of originally supplied ESW, as well as those prepared by tap water washing 3 × 10 min of originally supplied ESW, included determination of dry matter, total protein, lipid, and calcium carbonate content, all performed according to Strelec et al. [8].
2.3 ESW transformation process
The ESW transformation process using 5% (w/v) hydrochloric acid was modeled based on the previously reported ESW transformation with 5% HCl [8], but with some modifications. These included: (a) process scaling up (∼7.2-fold), (b) using 15 L conical batch reactor instead of glass beakers, (c) ESW washing with tap water instead of distilled one, (d) replacing calcium salt precipitation by acetone with spray-drying of concentrated solution, and (e) omitting the grinding step of obtained dried CaCl2.
The transformation process (Figure 1) started with 3 × 10 min washing of 650 g of ESW with 6.5 L of tap water in a constructed 15 L conical batch reactor equipped with an overhead variable speed stirrer set at 350 rpm·min−1, and the use of two double impulse type stirring elements set at 7 and 15 cm from the bottom of the reactor (Figure 2).
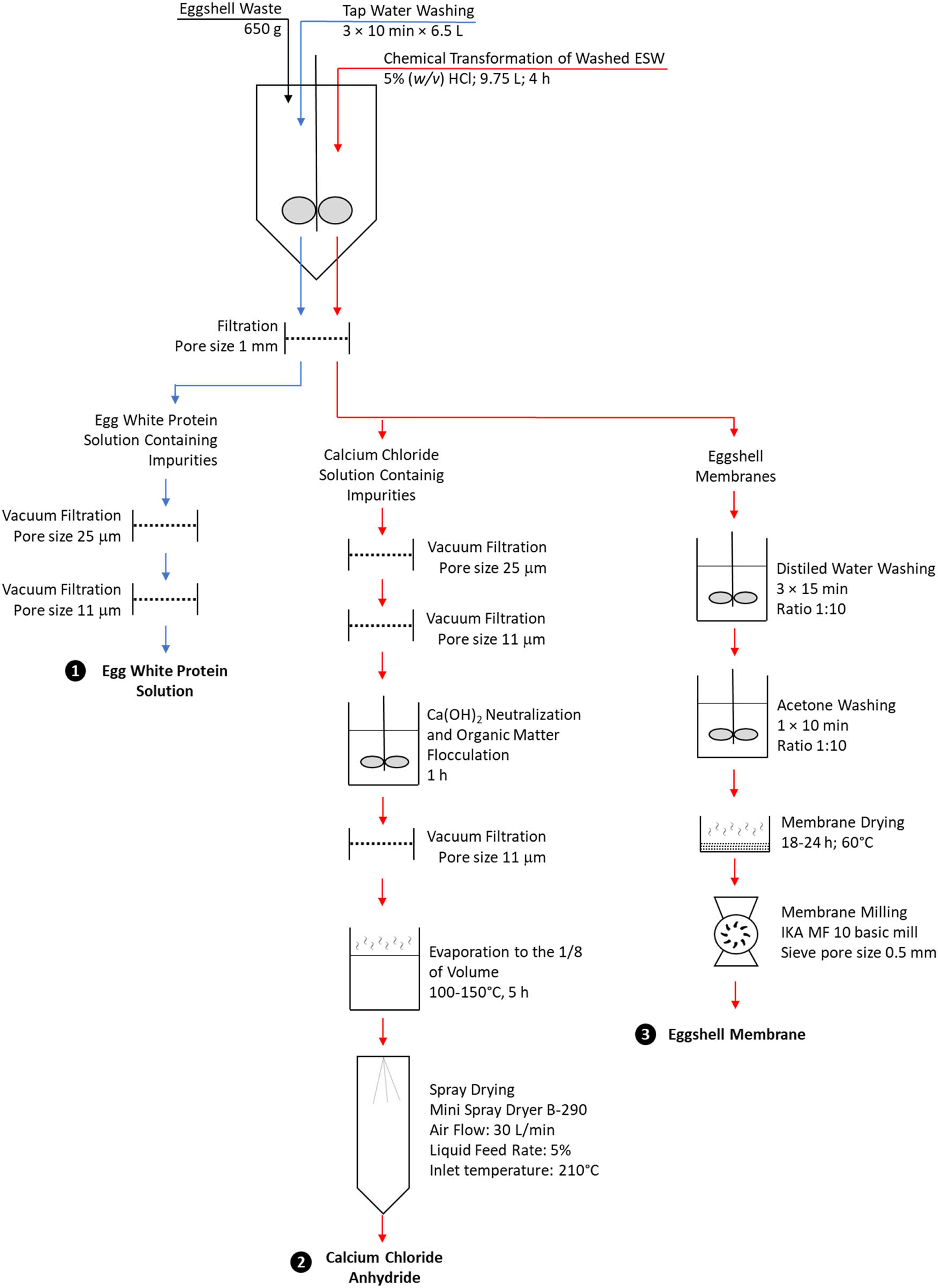
Schematic presentation of transformation process of industrial eggshell waste to egg white protein solution, calcium chloride anhydride, and eggshell membranes.

Schematic presentation of the constructed 15 L conical batch reactor for eggshell waste transformation. 1 – polyethylene terephthalate 15 L batch reactor (ϕ = 240 mm; h = 380 mm); 2 – conical batch reactor holder; 3 – poly(methyl methacrylate) overhead stirrer holder; 4 – variable speed overhead stirrer with the possibility of contrarotation; 5 – sampling opening; 6 – acid dosage opening; 7 – acid resistant stainless steel rotating shaft; 8 – acid resistant stainless steel double impulse stirring elements (ϕ = 110 mm); 9 – acid resistant stainless steel comb type stirring element (foam breakers; ϕ = 170 mm, h = 100 mm, 8 wires); and 10 – polyvinyl chloride un-plasticized (PVC-U) ball valve type 546 Pro.
Each of the protein solutions obtained after ESW washing was successively filtered through filter paper Whatman 114 followed by Whatman 1. The protein content of the clear protein solutions was determined using the Bradford technique [17].
The ESW obtained after three steps of washing was mixed with 9.75 L of 5% (w/v) hydrochloric acid in a 15 L batch reactor, but the addition of HCl was performed in aliquots as follows: 1 L of HCl was added to ESW at the beginning, followed by addition of 0.5 L each 10 min, and 0.75 L in the last step.
Mixing conditions of ESW suspension in HCl were performed at two different speed rates, as follows: 5% (w/v) HCl aliquot was added to washed ESW and mixed at 150 rpm·min−1 for 5 min in order to prevent excessive foaming; afterward, the mixing speed was then increased to 250 rpm·min−1. In both cases, excessive foaming was additionally reduced by the use of comb-type stirring elements. The process of the complete transformation of ESW calcium carbonate to CaCl2 by HCl and subsequent release of ESM completely devoid of the remnants of ESW-CaCO3 was finished at the fourth hour. Afterward, ESM was separated from the CaCl2 solution by filtration using a plastic mesh screen with a pore size of 1 mm.
The obtained ESM was washed with distilled water and acetone, dried, and milled to less than 0.5 mm, according to Strelec et al. [8].
The ESW-derived CaCl2 solution obtained after separation from ESM was subjected to two successive steps of vacuum filtration through filter paper Whatman 114 and then through Whatman 1. A clear solution of ESW-derived CaCl2 was then subjected to the neutralization of the excess of HCl by calcium hydroxide addition, followed by organic matter flocculation, clarification by vacuum filtration, and evaporation to the 1/8th of volume as previously reported by Strelec et al. [8].
The concentrated solution of ESW-derived CaCl2 was cooled to room temperature and then spray-dried with Mini Spray Dryer B-290 (Büchi, Postfach, Switzerland) at determined optimal conditions: an inlet temperature of 210°C, an airflow on nozzle of 30 L·min−1, an aspirator rate of 100% (equal to flow of 35 m3·h−1 of drying air), and a liquid feed rate of 1.5 mL·min−1. In addition, the determination of optimal conditions for spray drying of ESW-derived CaCl2 included variations in liquid feed rate between 1.5 and 6.0 mL·min−1, and airflow between 20 and 30 L·min−1.
CaCl2 production was measured by determining CaCl2, protein, and amino sugar content in the solution. Calcium content was determined by complexometric titration [8], protein content by the Bradford method [17], and amino sugar content by the Morgan–Elson method [18].
2.4 Characterization of ESW-derived CaCl2 anhydride
ESW-derived CaCl2 powder was spray-dried and examined for chemical composition, suitability as a food-grade additive, XRD pattern, and microbiological purity. Dry matter, CaCl2 and soluble protein content, CaCl2 powder solubility in water, pH of 10% CaCl2 powder solution, and free alkali content were determined according to Strelec et al. [8]. The total protein content in the obtained CaCl2 powder was determined by the standard Kjeldahl method, while the amino sugar content in CaCl2 solution of 100 mg·mL−1 by the Morgan–Elson method [18]. The Andrija Štampar Teaching Institute of Public Health (Zagreb, Croatia) assessed Mg and alkali salt content in CaCl2 powder using the gravimetric methods, fluoride content using an ion selective method, and Mg, F, As, Pb, and Hg using the ICP-MS technique.
FTIR-ATR analysis of CaCl2 powders produced on Mini Spray Dryer B-290 was performed on a Carry 630 FTIR ATR spectrometer (Agilent, Santa Clara, CA, USA) in the range of 650–4,000 cm−1.
Powder X-ray diffraction measurements of ESW-derived CaCl2 powders were performed using a Bruker Discover D8 diffractometer (Karlsruhe, Germany) supplied with a LYNXEYE XE-T detector in the angular 2θ range 10–70° with a step size of 0.02° and a measuring time of 1 s per step. The Eva software was used to identify the crystal phases, followed by a search and match method in the PDF4 database. Rietveld structural refinement was performed using the FULLPROF software [19].
Microbiological quality criteria of ESW-derived CaCl2 powders included determination of total aerobic plate count, mesophilic and thermophilic spore count, yeasts, molds, as well as pathogenic microorganisms including fecal coliforms and Escherichia coli, Salmonella spp., and Staphylococcus aureus, all performed by standardized microbiological procedures [20,21,22,23,24].
2.5 Examination of ESW-derived ESMs as potential enzyme immobilization carrier
ESW-derived ESMs were evaluated for their suitability as potential enzyme immobilization carriers. This included the determination of particle size distribution, water and oil holding capacity (WHC and OHC), as well as the possibility of immobilization of BCL onto the produced ESM by adsorption.
Particle size distribution of the produced ESM grounded to a size less than 0.5 mm was investigated by the laser light scattering method using a Mastersizer Scirocco 2000 analyzer (Malvern Instruments, Malvern, UK). The obtained results are presented as two dependent parameters: volume weighted mean diameter and span value, which were averaged values from three measurements.
WHC and OHC of the obtained ESM was performed by the method from Ballesteros et al. [25] with minor modifications. In brief, 0.5 g of the ESM sample was mixed with 5 mL of distilled water or olive oil, vortexed for 1 min, and centrifuged at 2,795 × g during 30 min in a Centric 150 centrifuge (Tehtnica, Podplat, Slovenia). Afterward, the volume of supernatant was measured, with WHC represented as mL of water per gram of the ESM sample and OHC as mL of olive oil held per gram of the ESW sample.
BCL immobilization by adsorption on the produced ESM was performed by the combination of methods of Chattopadhyay and Sen [26] and Salleh et al. [27], during 1, 2, 3, 4, 5, and 6 h with constant 360° stirring/rotation of the BCL and ESM suspension (0.5 g of ESM mixed with 10 mL of BCL of desired total activity) in 15 mL Falcon tubes on Multi-Rotator PTR-60 (Grant-Instruments Ltd, Cambridgeshire, UK) set at 17 rpm·min−1, at room temperature. BCL solutions in 50 mM phosphate buffer at pH 7.5 (10 mL) exhibited desirable total lipase activity of 230, 450, 820, 1.190, and 1.430 U. The effect of time and BCL concentration on immobilization was monitored by titrimetric determination of lipase activity [28], where the activity of immobilized BCL was expressed in units per gram of the ESM carrier (U·g−1).
3 Results
Encouraged by our previous research on the possibility of ESW transformation, on a laboratory scale, to the egg white protein solution, CaCl2 dihydrate, and ESM powder of ESW collected from households and restaurants [8], here we report the possibility of transformation of “industrial” ESW collected from egg-breaking plant to CaCl2 anhydride as a food-grade additive, and ESMs as a promising enzyme immobilization carrier. Although the currently proposed ESW transformation procedure (Figure 1) included the majority of previously reported transformation steps [8], it should be pointed out that the novelty of the proposed ESW transformation lies in scaling-up (7.2-fold) where the glass beaker was replaced by a 15 L conical batch reactor (Figure 2) followed by subsequent changes in stirring conditions, and in the production of CaCl2 anhydride by spray drying. Therefore, it could be safely concluded that the currently proposed ESW transformation process does not represent a simple scale-up procedure, but a novel and upgraded version of the possibility of ESW transformation to the high-value added products oriented toward a zero waste model. Moreover, it seems that the currently proposed ESW transformation procedure in a 15 L conical batch reactor might present a true intermediate step for upscaling from the laboratory to industrial-scale production.
3.1 Chemical composition of industrial ESW
It is well established that hen egg and eggshell composition is dependent on several factors including genetics and hen breed, the mode of feeding, physiological status (hen-age, stress, and immune status), as well as eggshell color: brown or white [16,29,30,31,32]. Therefore, it was necessary to determine the basic chemical composition of the “industrial” ESW regardless of the existing data on ESW composition reported in the literature [8,16,30,33]. This was done in order to calculate the chemical yield of CaCl2 produced by 5% (w/v) hydrochloric acid ESW treatment and to evaluate the successes of the proposed ESW transformation process.
Table 1 shows the basic chemical composition of originally supplied ESW (unwashed eggshells) and ESW devoid of adherent egg white proteins (washed eggshells) collected from the egg-breaking plant. The major component of both examined ESWs was calcium carbonate (90–92%), while proteins were present in amounts lower than 5.5% and lipids lower than 0.6%. Originally supplied ESW washing with tap water resulted in a slight increase of CaCO3 in washed and dried ESW and a decrease in the protein content (Table 1), which was congruent with reports of Walton et al. [33] and Strelec et al. [8], and could be attributed to the removal of a thin layer of adherent egg white present in ESW.
Chemical composition of industrial eggshell waste1
| Content | Unwashed eggshells2 | Washed eggshells3 |
|---|---|---|
| Dry matter (g·100 g−1) | 85.97 ± 0.03 | 99.97 ± 0.02 |
| Proteins (g·100 gd.w.b. −1)4 | 5.35 ± 0.14 | 2.42 ± 0.15 |
| Lipids (g·100 gd.w.b. −1)4 | 0.59 ± 0.04 | 0.51 ± 0.05 |
| CaCO3 (g·100 gd.w.b. −1)4 | 90.39 ± 0.86 | 91.43 ± 0.88 |
1Results are shown as average value ± standard deviation of five independent determinations.
2Unwashed eggshells – originally supplied ESW.
3Washed eggshells – originally supplied ESW washed 3 × 10 min with distilled water and dried at 60°C for 24 h.
4d.w.b. – dry weight basis.
The obtained data on the basic chemical composition of examined ESWs were in agreement with findings by Strelec et al. [8], Waheed et al. [16], Ray et al. [30], and Walton et al. [33].
3.2 Optimization of ESW transformation process
The first step in the optimization of the ESW transformation process (Figure 1) was to determine the time necessary for adherent egg-white protein removal, followed by the time needed for the complete ESW calcium carbonate dissolution using 5% (w/v) hydrochloric acid.
Trial runs on the “industrial” ESW washing by tap water in a 15 L conical batch reactor revealed that 3 × 10 min washing was sufficient for the complete removal of the adherent egg white proteins (Figure 3a). It should be pointed out that the selected speed of stirring elements was sufficient to ensure suspension homogeneity. The majority of adherent egg white proteins were extracted during the first washing (∼92.89%), while remnants of ESW proteins were exhausted during the second (∼5.52%) and third (∼1.60%) phase of 10 min washing (Figure 3b). Therefore, 3 × 10 min washing of ESW with tap water was defined as the first step in ESW transformation in a 15 L conical batch reactor.

Optimization of industrial eggshell waste washing with tap water in 15 L batch reactor. (a) Mass concentration of proteins present in the solution after first, second, and third washing during 5, 10, and 15 min; (b) overall distribution of washed adherent egg white layer proteins during 10 min washing.
Obtained data on the percentage of adherent egg white proteins extracted during ESW washing were consistent with our previous report where ESW halves and pieces were washed 3 × 30 min in glass beaker with distilled water [8]. However, it should be noted that the currently proposed ESW washing time (3 × 10 min) clearly present improvement from the previous one [8], since ESW washing was threefold reduced (from 90 to 30 min).
Besides the reduction in washing time, “industrial” ESW water washing in a 15 L batch reactor by the use of two double impulse-type stirring elements set at 350 rpm·min −1 caused breaking of ESW halves and pieces to ESW particles smaller than 4 cm × 4 cm. The reduction in the size of washed ESW had a negative impact on the possibility of addition of the complete volume of 5% (w/v) hydrochloric acid due to uncontrolled foaming and spilling over the edge of the reactor. Therefore, it was necessary to conduct a few trial runs of washed ESW transformation treatment by 5% (w/v) hydrochloric acid, including time-dependent gradual acid dosage in combination with variations in stirring speed.
The combination of temporal acid dosage and changes in variations of stirring speed did not result in the desired foam reduction during the ESW transformation process after two trial runs. Therefore, it was necessary to introduce an additional element, a comb-type stirring element as a foam breaker/reducer (Figure 2). The use of foam breaker/reducer enabled adequate ESW-acid transformation, and after two additional trial runs, a complete protocol of washed ESW transformation by 5% (w/v) hydrochloric acid, defined in sub-Section 2.3, was established. Afterward, three independent “industrial” ESW-transformation batch procedures were performed.
Time-dosage dependent 5% (w/v) hydrochloric acid solvation of washed “industrial” ESW resulted in the prolongation of time from previously reported 3 h [8] to 4 h. Nevertheless, the total time of the ESW transformation process including ESW washing and hydrochloric acid treatment, performed at room temperature, was the same (4.5 h). While the previous report on the transformation of ESW by 5% (w/v) hydrochloric acid treatment included 3 × 30 min washing followed by 3 h acid treatment [8], the current research included 3 × 10 min washing and 4 h of 5% (w/v) hydrochloric acid treatment.
Once suspension of ESM completely devoid of the remnants of endogenous calcium carbonate in CaCl2 solution was obtained, it was subjected to filtration through a 1 mm pore size plastic screen (Figure 1) in order to separate ESW-derived CaCl2 solution from ESMs. Separated ESM was subjected to 3 × 15 min distilled water washing (mass-to-volume ratio: 1:10) in a 3 L glass beaker positioned on an electromagnetic stirrer set at 750 rpm. This was followed by single-volume acetone washing under the same conditions afterward obtained ESM were dried between 18 and 24 h at 60°C in a thermostatic incubator (Heraeus, Germany) and milled using a laboratory mill IKA MF 10.1 (IKA, Staufen, Germany), equipped with a 0.5 mm pore size sieve, at a speed rotation of 4,500 rpm.
ESW-derived CaCl2 solution obtained after separation from ESM through a 1 mm pore size plastic screen was subjected to two successive steps of vacuum filtration through filter paper Whatman 114 followed by Whatman 1 in order to achieve a clear solution. Afterward, the excess hydrochloric acid was neutralized by the addition of calcium hydroxide, and the solution was left to stand for 30 min for organic matter flocculation. Removal of precipitated organic matter was achieved by vacuum filtration through filter paper Whatman 1. The obtained clear solution of ESW-derived CaCl2 was then subjected to evaporation to approximately 1/8th of the volume in order to concentrate the solution, and afterward used for the production of CaCl2 powder by spray drying. It should be pointed out that evaporation of CaCl2 solution to the desired concentration was not selected arbitrarily but was based on the US 3433863 patent reported by Bowden and Terry [34], where 58% CaCl2 content in a slurry was found as the most prominent for the production of CaCl2 powder by spray drying.
Table 2 shows the changes in the CaCl2, protein, and amino sugar concentration in the solutions of ESW-derived CaCl2 during the transformation process, as well as the chemical yield of CaCl2 and percentage of protein and amino sugar removal. A complete conversion of ESW-calcium carbonate to CaCl2 by 5% (w/v) hydrochloric acid resulted with CaCl2 solution of 60.69 ± 1.05 g·L−1 containing small amount of proteins (γ = 0.35 ± 0.04 g·L−1) and amino sugars (0.12 ± 0.01 g·L−1). Neutralization of the excess of hydrochloric acid with calcium hydroxide followed by organic matter flocculation yielded partially purified CaCl2 solution of 72.02 ± 2.93 g·L−1 from which ∼68.6% of proteins and only ∼3.7% of amino sugars were removed. Further removal of proteins and amino sugars from CaCl2 solution occurred during concentration by evaporation to the ∼1/8th of volume where in total 99.14% of proteins and 83.25% of amino sugars present in the starting CaCl2 solution were removed. This can be attributed to the well-known process of the salting out. The data on CaCl2 and protein concentration in the obtained solution are well in agreement with our previous work [8]. However, there are no data on the concentration of amino sugars in the solution of ESW-derived CaCl2 so far. Nevertheless, the presence of amino sugars in the solutions of ESW-derived CaCl2 proved our previous assumption [8] that polysaccharides are probably the major non-proteinaceous organic matter extracted from ESM during the hydrochloric acid treatment of ESW. In fact, this was the main reason why have we monitored amino sugar concentration in the ESW-derived CaCl2 solutions.
Calcium chloride, soluble protein and amino sugar concentration changes during industrial eggshell waste transformation process1
| Production step | Calcium chloride | Proteins | Amino sugars | |||
|---|---|---|---|---|---|---|
| γ (g·L−1) | Yield (%)2 | γ (g·L−1) | Removed (%)3 | γ (g·L−1) | Removed (%)3 | |
| 5% (w/v) HCl solvation | 60.69 ± 1.05 | 100.53 ± 1.56 | 0.35 ± 0.02 | n.a.4 | 0.12 ± 0.01 | n.a.4 |
| Ca(OH)2 neutralization and organic matter flocculation | 72.02 ± 2.93 | 99.36 ± 1.24 | 0.11 ± 0.02 | 68.57 ± 4.10 | 0.12 ± 0.00 | 3.71 ± 1.34 |
| Evaporation to the ∼ 1/8th of the original concentration of calcium chloride | 590.89 ± 18.92 | n.a.4 | 0.03 ± 0.00 | 99.14 ± 0.46 | 0.19 ± 0.03 | 83.25 ± 1.36 |
1Results present the mean value ± standard deviations of three independent production batches each performed in triplicate.
2Chemical yield calculated as percentage of ratio of theoretical amount of calcium chloride and experimentally produced amount of calcium chloride.
3Cumulative percentage of removed proteins and sugars from the solution of completely dissolved eggshell waste by 5% (w/v) HCl.
4n.a. – not applicable.
Once a concentrated solution of ESW-derived CaCl2 was obtained, it was necessary to optimize the process of spray drying in order to produce CaCl2 anhydride of the lowest moisture content and the greatest yield in the collection beaker. This was done by changing the liquid feed rate from 1.5 to 6.0 mL·min−1, and airflow from 20 to 30 L·min−1. Among five different combinations of liquid feed rate and airflow used for spray drying of a concentrated solution of ESW-derived CaCl2, the best was the use of liquid feed rate of 1.5 mL·min−1 and airflow of 30 L·min−1 where CaCl2 anhydride of the lowest moisture content (2.15%) and the greatest yield (91.67%) in the collection beaker was obtained (Table 3). Therefore, the inlet temperature of 210°C, an airflow on the nozzle of 30 L·min−1, an aspirator rate of 100% (equal to the flow of 35 m3·h−1 of drying air), and a liquid feed rate of 1.5 mL·min−1 were chosen as optimal for spray drying of ESW-derived CaCl2 of three independent production batches.
Optimization of calcium chloride powder production by spray drying of ESW derived concentrated calcium chloride solutions (γ = 590.89 ± 18.92 g·L−1) using Mini Spray Dryer B-290
| Drying conditions | Liquid feed rate (mL·min−1) | Air flow (L·min−1) | Drying temperature (°C) | CaCl2 in the collection glass beaker | CaCl2 in the drying tower | |||
|---|---|---|---|---|---|---|---|---|
| Inlet | Outlet | Moisture (%) | Yield (%)1 | Moisture (%) | Yield (%)1 | |||
| I | 1.5 | 30 | 210 | 146 | 2.15 | 91.67 | 4.59 | 8.33 |
| II | 3.0 | 20 | 210 | 146 | 7.86 | 90.93 | 4.90 | 9.07 |
| III | 3.0 | 30 | 210 | 146 | 6.99 | 90.88 | 4.87 | 9.12 |
| IV | 4.5 | 25 | 210 | 132 | 8.44 | 88.30 | 6.86 | 11.70 |
| V | 6.0 | 25 | 210 | 132 | 8.22 | 80.33 | 11.56 | 19.67 |
1Yield of calcium chloride in the collection glass beaker and tower is calculated as percentage of ratio of the mass of obtained calcium chloride in the collection glass beaker or drying tower and the sum of masses of calcium chloride obtained in both.
The spray drying optimization was also monitored by FTIR-ATR analysis. Figure 4 shows FTIR-ATR spectra of obtained ESW-derived CaCl2s during spray drying optimization in comparison with commercial CaCl2 anhydride and CaCl2 dihydrate. It could be seen that all examined CaCl2s (ESW-derived and commercial ones) show identical peak positions centered at 1,617, 3,451, and 3,488 cm−1, but differ in their absorbance intensities, where ESW-derived CaCl2s shows similar peak intensities to the commercial CaCl2 anhydride. Peaks centered at 3,451 and 3,488 cm−1 can be attributed to the symmetric or asymmetric O–H stretching in crystalline water, and peak centered at 1,617 cm−1 to the H–O–H bending vibration frequency in crystalline water [35].
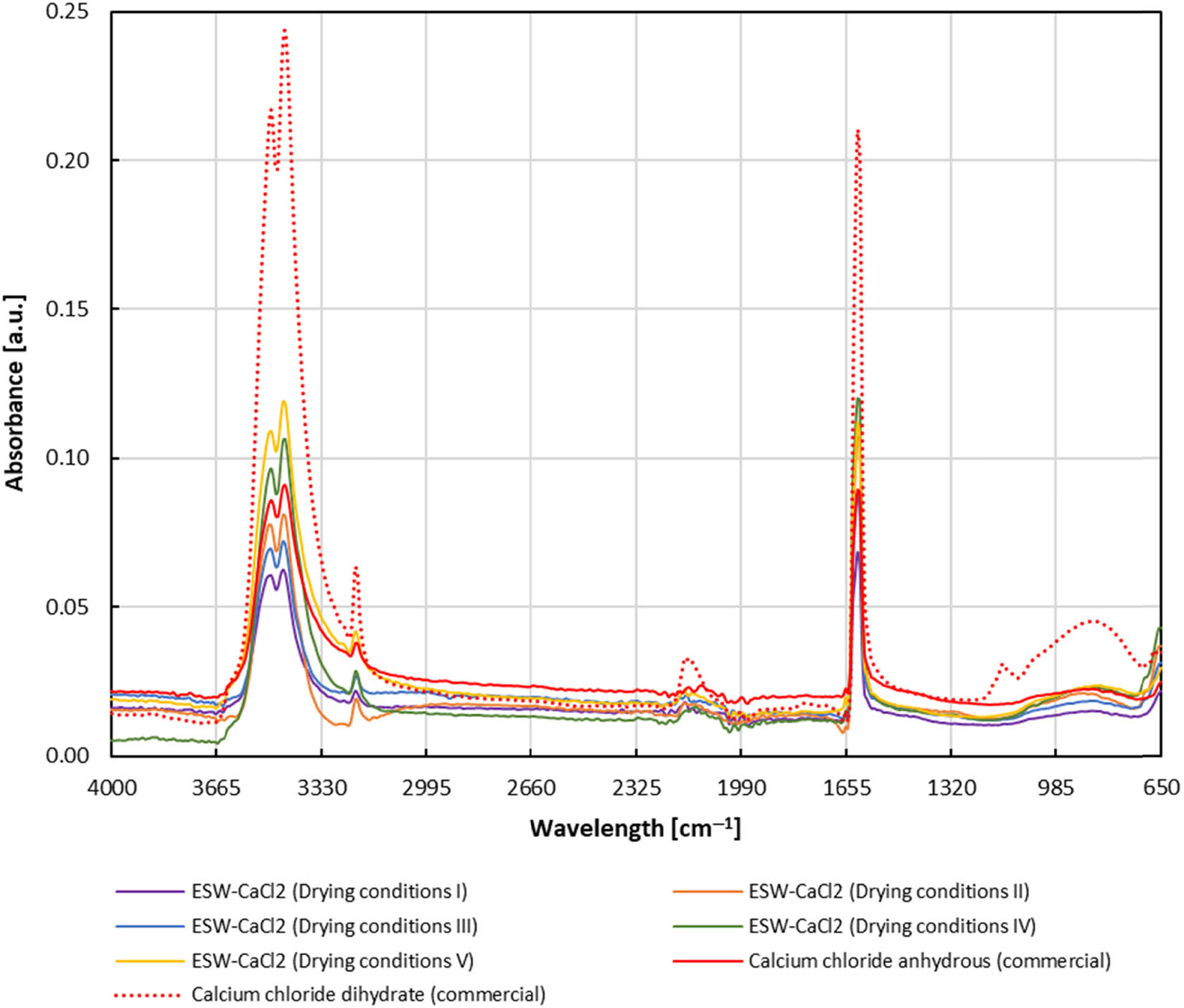
FTIR-ATR analysis of ESW derived calcium chloride in respect to spray-drying conditions. Drying conditions: I – liquid feed rate of 1.5 mL·min−1; air flow of 30 L·min−1; II – liquid feed rate of 3.0 mL·min−1; air flow of 20 L·min−1; III – liquid feed rate of 3.0 mL·min−1; air flow of 30 L·min−1; IV – liquid feed rate of 4.5 mL·min−1; air flow of 25 L·min−1; and V – liquid feed rate of 6.0 mL·min−1; air flow of 25 L·min−1.
Table 4 shows the mass yield of ESW-derived CaCl2 anhydride and ESMs obtained from three independent production batches. By the use of the proposed ESW transformation procedure, ∼ 102 g of CaCl2 anhydrous and ∼ 2.5 g of ESM can be produced from 100 g of ESW per dry weight basis. According to our knowledge, this is the first report on the possibility of the production of CaCl2 anhydride from ESW. On the other hand, the possibility of the production of ESM by 5% (w/v) hydrochloric acid treatment of ESW with concomitant production of CaCl2 dihydrate has been reported in our previous work [8], with somewhat higher ESM yield of 2.84 ± 0.16 g per 100 g of ESW, than those obtained in the current research where 2.48 ± 0.28 g of ESM was obtained from 100 g of ESW. The most probable reason for the somewhat lower ESM yield is a difference in the chemical composition of the “industrial” ESW (Table 1) and those collected from households and restaurants [8] attributable to the eggshell color [16,30], laying hen breed, as well as hen housing conditions [32]. Just for example, Kocetkovs et al. [32] reported a significant difference in the ESM thickness between the eggs of different hen breeds and hen housing conditions (cage and cage-free housing).
Mass yield of calcium chloride anhydride and corresponding eggshell membranes produced from industrial eggshell waste by proposed transformation process
| Transformation product | Mass yield (%)1,2 |
|---|---|
| Calcium chloride anhydride | 102.42 ± 3.31 |
| Eggshell membranes | 2.48 ± 0.28 |
1Results present the mean value ± standard deviations of three independent production batches.
2Mass yield calculated on dry weight basis.
3.3 ESW-derived CaCl2 anhydride as a food-grade additive
The ESW-derived CaCl2 powders were tested on their physicochemical properties, purity, food-grade additive criteria, as well as microbiological quality criteria. Complexometric titration of obtained powders revealed that ESW-derived CaCl2 contains 35.01 ± 0.63% of calcium (Table 5), which is slightly lower than the theoretically calculated amount of calcium (36.11%) in CaCl2 anhydride, indicating that the obtained CaCl2 is probably in anhydrous form. In this respect, based on the data of complexometric titration, it was found that obtained CaCl2 powders contain 96.96 ± 1.74% of CaCl2 anhydride. This was higher than the minimally prescribed content of CaCl2 (≥93.00) in CaCl2 anhydride as a food-grade additive (Table 5). Obtained CaCl2 had somewhat lower solubility in water at room temperature (≤0.8 g·mL−1) than expected (1 g·mL−1) [5], but pH of 10% water solution (9.46 ± 0.16) was quite similar to the pH of 10% water solution of commercial CaCl2 anhydride (9.65 ± 0.11). Identical solubility and somewhat lower pH of 10% water solution of ESW-derived CaCl2 dihydrate (pH = 9.34) was reported in our previous research [8]. Since the concentrated solution of CaCl2 (Table 2) contained a small amount of proteins and amino sugars, it was necessary to examine the content of both in the obtained CaCl2 powders. ESW-derived CaCl2 anhydride contained minor amounts of soluble proteins (0.05 ± 0.01 g·100 g−1 d.w.b.) and amino sugars (0.16 ± 0.01 g·100 g−1 d.w.b.), while total protein content determined by the Kjeldahl method was 0.21 ± 0.06% (Table 5). Somewhat lower amount of soluble proteins (0.02 ± 0.01 g·100 g−1 d.w.b.) has been reported for ESW-derived CaCl2 dihydrate powder [8], and a bit higher total protein content (0.29%) in ESW derived CaCl2·2H2O [36]. The ESW-derived CaCl2 fully met all criteria as a food-grade additive prescribed by the EU [37] and FAO [38], as shown in Table 5. Free alkali content was twofold lower than maximally allowed ≤0.15%, while Mg and alkali salt content almost tenfold (≤5.2 vs ≤50 mg·g−1). Moreover, at least 20-fold lower amount than defined maximal values for F, As, Pb, and Hg content in CaCl2 as a food-grade additive prescribed by the EU [37] and FAO [38] were detected in ESW-derived CaCl2 anhydride (Table 5). Based on the aforementioned, it can be safely concluded that ESW-derived CaCl2 anhydride, due to its purity [37,38], obviously has a great potential to be used as a food additive.
Physicochemical analysis of eggshell waste derived calcium chloride anhydride
| Parameter | Determined value | Food additive criteria value1 |
|---|---|---|
| Dry matter content (%) | 98.85 ± 0.01 | n.i.c.2 |
| Calcium content (%) | 35.01 ± 0.63 | n.i.c.2 |
| Calcium chloride content (%) | 96.96 ± 1.74 | ≥93.00 |
| Solubility in water (g·mL−1) | ≤0.8 | n.i.c. |
| pH of 10% solution | 9.46 ± 0.16 | n.i.c. |
| Free alkali (%) | <0.074 | ≤0.150 |
| Soluble protein content (g·100 g−1 d.w.b.)3 | 0.05 ± 0.01 | n.i.c. |
| Total protein content (%) | 0.21 ± 0.06 | n.i.c. |
| Amino sugar content (g·100 g−1 d.w.b.)3 | 0.16 ± 0.01 | n.i.c. |
| Mg and alkali salts content (mg·g−1) | ≤5.2 | ≤ 50 |
| Mg content (mg·kg−1) | 1975 ± 361 | n.i.c. |
| F content (mg·kg−1) | <2.0 | ≤40 |
| As content (mg·kg−1) | ≤0.031 | ≤3 |
| Pb content (mg·kg−1) | <0.05 | ≤2 |
| Hg content (mg·kg−1) | <0.01 | ≤1 |
In order to confirm the anhydrous form of ESW-derived CaCl2 powders, their purity, as well as uniformity of the ESW transformation process, XRD analysis of obtained CaCl2 powders from three production batches was performed. Table 6 shows crystallographic data of ESW-derived CaCl2 anhydrous analyzed and characterized by the Rietveld refinement of XRD patterns, while Figure 5 shows the Rietveld plots of XRD patterns. Obtained results clearly indicate that all three obtained CaCl2 powders are identified as anhydrous CaCl2 that crystallized in the orthorhombic Pnnm space group. Besides Bragg reflections describing anhydrous CaCl2 (vertical ticks in the Rietveld plot), XRD analysis reveals the presence of some unidentified impurities probably generated during the ESW transformation process. These peaks can be clearly resolved from the peaks of the main phase due to their sharper appearance. It is also worth mentioning that the sharp peak located at 35.1° is attributed to corundum originating from the XRD sample holder. Other crystallographic parameters, such as lattice parameters, cell volume, and calculated density, confirm that obtained CaCl2 powders are of the anhydrous form [39,40]. In addition, similar values of the crystallographic parameters for each ESW-derived CaCl2 (Table 6) confirm the uniformity of the ESW-transformation process. In order to visualize the obtained crystal structures, the VESTA software was utilized [41], and atomic arrangements in the obtained CaCl2 are shown in Figure 6.
Crystallographic data of ESW-derived calcium chloride production batches. Data are obtained from Rietveld refinement
| Compound | CaCl2 | CaCl2 | CaCl2 |
|---|---|---|---|
| Batch 1 | Batch 2 | Batch 3 | |
| Space group | Pnnm | ||
| Crystal system | Orthorhombic | ||
| Data collection range (2θ) | 10–70° | ||
| Phase composition (wt%) | 100 | ||
| Z | 2 | 2 | 2 |
| Lattice parameters (Å) | a = 6.2793 (2) | a = 6.2797 (2) | a = 6.2786 (2) |
| b = 6.4229 (2) | b = 6.4220 (2) | b = 6.4276 (2) | |
| c = 4.1612 (2) | c = 4.1598 (2) | c = 4.1600 (2) | |
| Cell volume (Å3) | 167.8264 (6) | 167.7592 (6) | 167. 8807 (6) |
| Calculated density (g·cm−3) | 2.196 | 2.197 | 2.196 |
| No. of parameters refined | 64 | 64 | 64 |
| Average crystallite size (nm) | 26 (2) | 24 (6) | 19 (2) |
| R B (%) | 7.0 | 6.72 | 6.44 |
| R p, R wp, R e (%) | 31.2, 29.8, 19.6 | 28.8, 29.1, 16.6 | 33.5, 30.9, 21.8 |
| GoF | 1.525 | 1.748 | 1.416 |
Legend: numbers is brackets - uncertainty of the last digit (+/−); R B – Bragg factor; R p – unweighted profile R-factor; R wp – weighted profile R-factor; R e – expected R factor; GoF = goodness of fit.
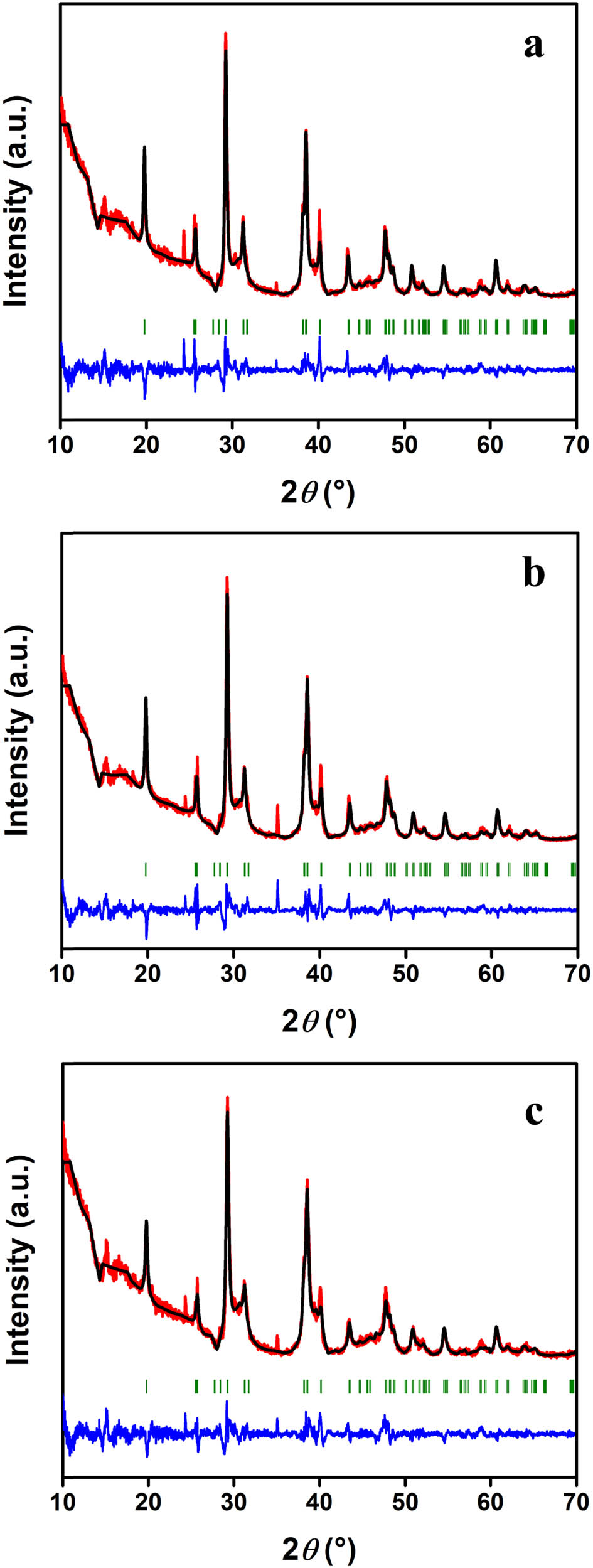
XRD pattern of ESW derived calcium chloride anhydride production batches. (a) The Rietveld plot of the investigated CaCl2 – 1, (b) the Rietveld plot of the investigated CaCl2 – 2, (c) the Rietveld plot of the investigated CaCl2 – 3. The Rietveld plot displays the Bragg reflections and includes the observed (black), calculated (red), and difference (blue) plot, while vertical bars (green) are the expected Bragg reflection positions.

Crystal structure visualization of ESW-derived calcium chloride anhydride. Calcium cation is depicted in dark blue and chloride anion in green.
Currently, the prescribed criteria for the purity of CaCl2 as a food-grade additive [37,38] do not involve microbiological quality criteria. This is not surprising since CaCl2 is mainly produced by hydrochloric acid treatment of limestone. However, the question remains: What if CaCl2 is produced from an organic source rich in calcium, such as ESW? Is there a need for prescribing microbiological quality criteria for food-grade additives derived from eggs? Considering the fact that egg and egg products are prone to microbiological contamination if improperly stored [20,22,42], and the report of EFSA Panel on Dietetic Products, Nutrition and Allergies (EFSA NDA Panel) on the safety of egg membrane hydrolysate as a novel food produced from eggshells [21], it seemed quite reasonable to check the microbial quality of ESW-derived CaCl2. In this regard, we have examined the microbiological quality of obtained ESW-derived CaCl2 powders using the same criteria as those reported by the EFSA panel [21]. The results of examined microbiological parameters in the ESW-derived CaCl2 powders from three production batches are shown in Table 7. Pathogenic microorganisms including fecal coliforms, Escherichia coli and Salmonella spp., and Staphylococcus aureus have not been detected in ESW-derived CaCl2 powders. The same situation was in the case of thermophilic spore count and yeasts. Conversely, the presence of bacterial population in all three samples of ESW-derived CaCl2 powder has been detected by aerobic plate count, while two of the three production batches contained mesophilic spore (batches 1 and 3) and molds (batches 1 and 2) lower than 100 CFU·g −1. When obtained data on tested microbiological parameters of ESW-derived CaCl2 were compared with prescribed microbiological quality criteria (Table 7), it could be seen that two of three production batches, batch 2 and 3, were in compliance with the EU Regulation [21], while production batch 1 fulfilled all criteria except mesophilic spore count (86 CFU·g −1) which was higher than prescribed (≤ 25 CFU·g −1). Considering the fact that ESW-derived CaCl2 solution was exposed to high temperature (100−150°C) during 5 h evaporation, and even higher temperature during spray drying (210°C), it is less likely that elevated mesophilic spore count in CaCl2 powder from the production batch 1 can be attributed to elevated contamination of starting material (ESW). Rather, it could be a consequence of unintended contamination of powder during handling.
Microbiological quality of eggshell waste derived calcium chloride anhydride of three production batches in respect to the prescribed criteria for egg derived products
| Parameter | Prescribed microbiological criteria | CaCl2 production Batch 1 | CaCl2 production Batch 2 | CaCl2 production Batch 3 |
|---|---|---|---|---|
| Aerobic plate count | ≤2,500 CFU·g −1 | 1,450 | 290 | 1,350 |
| Escherichia coli | ≤5 MPN·g−1 | 0 | 0 | 0 |
| Salmonella | Negative (in 25 g) | 0 | 0 | 0 |
| Coliforms | ≤10 MPN·g −1 | 0 | 0 | 0 |
| Staphylococcus aureus | ≤10 CFU·g −1 | 0 | 0 | 0 |
| Mesophilic spore count | ≤25 CFU·g −1 | 86 | 0 | 5 |
| Thermophilic spore count | ≤10 CFU·g −1 | 0 | 0 | 0 |
| Yeasts | ≤10 CFU·g −1 | 0 | 0 | 0 |
| Molds | ≤200 CFU·g −1 | 15 | 5 | 0 |
CFU – colony forming units; MPN – most probable number.
Based on aforementioned, it can be concluded that ESW-derived CaCl2 due to its acceptable microbiological quality has a great potential to be used as a food additive.
3.4 ESM as a promising carrier for lipase immobilization
Besides a multitude of potential applications in various fields of industry including medical, pharmaceutical, cosmetic, electric, and food industry, well described in reviews of Park et al. [43], Shi et al. [44], Kulshreshtha et al. [45], Mensah et al. [46], and Han et al. [47], ESM has been reported as a promising platform for the development of immobilized enzyme-based biosensors for glucose [48,49,50,51], hydrogen peroxide [52], aspartame [53], homocysteine [54], dopamine [55], urinary oxalate [56], and urea detection [57]. However, the majority of aforementioned reports used native ESM for the enzyme immobilization, where ESMs intended for biosensor development were prepared by ESW distilled water washing, stripping off by forceps, and enzyme immobilization performed on ESM pieces ranging in size from 1 × 1 up to 4 × 2 cm. This was quite different from the current research, where ESM was obtained by hydrochloric acid treatment of ESW and milled to a particle size less than 500 µm (Figure 1) prior to the lipase immobilization. Therefore, it was necessary to investigate whether hydrochloric acid-derived ESM can be used as a promising platform/carriers for enzyme immobilization, especially considering the fact that acid-induced release of ESM from ESW leads to the physical and chemical changes of ESMs [8,9,45,46,47,58].
The suitability of ESW-derived ESM for enzyme immobilization was examined by the simplest immobilization technique, immobilization by adsorption, using commercial BCL, as the enzyme of interest.
However, prior to the lipase immobilization, it was necessary to confirm that the particle size of the obtained ESM was well in accordance with the desired size range of carriers/beads for enzyme immobilization, as well as to define a minimal volume of enzyme solution, which should be added to the 1 g of dry ESW-derived ESM.
Figure 7 shows the volume-weighted particle size distribution curve of ESW-derived ESM, while Table 8 shows the WHC and OHC of obtained ESMs.

Particle size distribution of ESW derived eggshell membranes. Results are averaged values of three measurements.
WHC and OHC of ESW-derived ESMs
| Parameter | Value1 |
|---|---|
| WHC (mL·g−1) | 5.54 ± 0.12 |
| OHC (mL·g−1) | 3.15 ± 0.13 |
1Results present the mean value ± standard deviations of four independent determinations.
The volume-weighted particle size distribution curve of the ESW-derived ESMs (Figure 7) showed a clear monomodal particle size distribution with a relatively narrow particle size distribution confirmed by a span value of 1.586. The volume-weighted mean diameter of obtained ESMs was 291.89 μm, with the largest volume fraction of particles in the range of mean particle size diameters from 200 to 400 μm (Figure 7) confirming the suitability of obtained ESM for immobilization. According to the literature, the desirable particle size of enzyme immobilization carrier should range from 20 up to 500 µm [59,60,61,62].
Dependent on the extraction method used, it has been reported that ESMs show somewhat different capacities of fluid/water adsorption [58]. In this regard, it was necessary to determine the WHC of ESW-derived ESM obtained by 5% (w/v) hydrochloric acid treatment in order to determine the suitable volume of lipase solution that should be added to ESM during the immobilization process. Besides determination of WHC obtained ESM was also tested on OHC. Corici et al. [60] reported enhanced lipase immobilization on the rice husk when rape seed oil was used for enzyme immobilization in comparison with a buffered enzyme solution.
ESM obtained in the current research showed great WHC of 5.54 ± 0.12 mL of water per 1 g of dry ESMs, and almost twice-lower OHC of 3.15 ± 0.13 g of olive oil per 1 g of dried ESM (Table 8), indicating high water adsorption capacity of the produced membranes. While the exact data on the oil and water holding capacity of ESM in the currently available literature do not exist, the report of Mensah et al. [58] on ESM water adsorption capacity clearly supports the determined WHC of ESM.
Once WHC of ESM was determined, optimization of BCL immobilization by adsorption onto ESM combining the effect of immobilization duration (from 1 to 6 h) and lipase activity load (230, 450, 820, 1,190, and 1,430 U) was performed at 25°C. These two parameters of enzyme immobilization have been selected as the most important for defining the suitability of the obtained ESM for lipase immobilization by adsorption due to the significant effect of both on the immobilization efficiency. Immobilization by adsorption of various lipases (commercial or purified ones) onto various hydrophobic and hydrophilic carriers/supports of commercial and/or natural origin, ranging from 30 min up to 20 h, and enzyme activity load between 90 and 50,000 U, with immobilized lipase activities ranging from 0.1 up to 40,000 U per 1 g of the carrier have been reported in the available literature [26,27,60,63,64,65,66,67,68,69,70,71,72,73,74,75].
Figure 8 shows the effect of the immobilization duration and commercial BCL activity load on ESM-immobilized BCL activity. One hour of mixing of the suspension of ESM with BCL solutions of different activity loads was found sufficient to obtain maximal non-covalent binding of BCL onto ESM, i.e., immobilization by adsorption, where the maximal activity of lipase of ∼124 U per 1 g of wet ESM was obtained by the use of the total activity of lipase of 1,190 U (Figure 8).

Immobilization of BCL on ESW derived eggshell membranes by adsorption. Results present the mean value ± standard deviations of three independent determinations each performed in triplicate.
The prolonged time of immobilization, greater than 1 h, for all examined activity loads of BCL (230, 450, 820, 1,190, and 1,430 U) resulted in the decreased lipase activity per gram of ESM carriers (Figure 8). This was different from the report of Jiang et al. [73], where 8 h of immobilization by adsorption of BCL at the highest activity load of 1,500 U onto ESM has been found to be better than immobilization performed during 0.5 h. However, it should be pointed out that authors used ESM pieces of 1 cm × 1 cm size instead of milled ones and reported differences in the activity of immobilized lipase by adsorption regarding applied low and high enzyme activity load (300 vs 1,500 U) and low and high time of immobilization (0.5 vs 8 h) were relatively minimal (9.86 ± 0.33 vs 11.52 ± 0.63 mmol·min−1·g−1).
The most probable reason for the observed decrease in the immobilized BCL activity after 1 h of immobilization onto obtained ESM in the current research (Figure 8) is the multilayer adsorption of lipases, which leads to the steric interference of neighboring immobilized enzymes and subsequently hindered activity toward substrate [76,77].
The substantial lack of available literature reports regarding the activity of immobilized lipases onto ESM is one of the greatest obstacles to the actual comparison of currently observed ESM-immobilized BCL activity with previous ones. This is even more pronounced by the fact that various lipase activity assays differ in the determined lipase activity up to 20-fold [28,66,78,79,80,81], which additionally disables proper comparison. Nevertheless, ESM-immobilized BCL activity of 124 U per 1 g of wet ESM was found to be greater than those reported by Abdulla et al. [74], where 80 U of BCL per 1 g of ESM was immobilized by adsorption followed by glutaraldehyde crosslinking, but lower than reports of Chattopadhyay and Sen [26] and Salleh et al. [27] where lipase immobilized by adsorption on eggshells showed activity of ∼355 and ∼280 U·g−1 of carriers, but with tributyrin as lipase substrate. Regardless, the aforementioned pinpoints ESW-derived ESM by hydrochloric acid treatment as a promising/suitable carrier for lipase immobilization.
4 Discussion
The current research presents an upgraded innovative technique of ESW transformation by 5% (w/v) hydrochloric acid to the value-added products including CaCl2 anhydride of food-grade additive purity, and ESMs as promising enzyme immobilization carrier (Figure 1), oriented toward zero waste model of sustainable waste management strategy. Although it is partially based on the previously reported ESW transformation process by our research group, where ESW collected from households and restaurants was transformed to CaCl2 dihydrate and ESMs on a laboratory scale [8], here we report upgraded and upscaled ESW-transformation version of the complete process where the majority of previously mentioned future perspectives are fulfilled, and some assumptions proved. This includes the use of “industrial” ESW collected from egg-breaking plants, transformation process scaling-up (7.2-fold), confirmation that part of glycosaminoglycans have been extracted from ESM during 5% (w/v) hydrochloric acid treatment of ESW at room temperature (Table 2), the replacement of ESW-derived CaCl2 concentrated solution precipitation with acetone by spray-drying (Table 3), and subsequently omitted necessity of grinding of obtained dried CaCl2 preparations.
Taken together, it can be safely concluded that the currently proposed ESW transformation process (Figure 1) presents a novel and upgraded version of the possibility of ESW transformation by 5% (w/v) hydrochloric acid to the high-value-added products, as well as ready-to-be-used developed upgraded laboratory transformation process easily transferable to the industrial scale, with obvious possibility of upscaling from the laboratory to the pilot plant scale.
In general, current transformation (Figure 1) of 100 g of ESW on a dry weight basis (or ∼118 g ESW on a wet basis) yielded ∼102 g of CaCl2 anhydride of high purity as the main transformation product and ∼2.5 g of ESMs as by-product serving as a promising enzyme immobilization carrier (Table 4), which clearly justify the suitability of the proposed transformation process. Aforementioned is even more pronounced if one considers that price of 100 g of CaCl2 anhydride containing more than 96% of CaCl2 (ESW-derived CaCl2 anhydrous contains 96.96 ± 1.74% of CaCl2) ranges from 43.20 € up to 182 € [82].
The obtained ESW-derived CaCl2 anhydride was of high purity and met all prescribed criteria to be used as a food-grade additive (Table 5). Besides prescribed criteria for use as a food-grade additive [37,38], ESW-derived CaCl2 was tested on its microbial quality criteria (Table 7), simply due to the fact that it was not produced from limestone but from ESW where the necessity for microbial quality testing should be applied [21]. The obtained results (Table 7) have shown the suitability of the proposed ESW transformation process for obtaining microbiologically safe ESW-derived CaCl2 as a food-grade additive. These findings are even more pronounced if one considers a bit of strengthening EU legislation related to the possibility of reintroducing transformation products derived from by-products of animal origin of category III, such as ESW, back to human consumption [83]. However, it should be pointed out that ESW-derived ESM hydrolysate has been accepted by the EU as a novel food [21], although produced from eggshells. Thus, it seems quite possible that currently obtained ESW-derived CaCl2 anhydride of high purity has a chance to be considered as a food-grade additive. Nevertheless, it should be noted that CaCl2 besides its possible use as a food-grade additive has a multitude of alternative uses, including road deicing and stabilization, dust control, concrete curing, oil well drilling, tire ballasting, the production of washing, cleaning and anti-freeze products, fertilizers and water treatment chemicals [2,3,4,5], which clearly justifies the proposed ESW-transformation process. This is even more important if one considers that ∼333,525 tons of ESW in the EU are generated by egg-breaking plants annually and the fact that ∼118 g of ESW per wet basis ∼102 g of CaCl2 anhydride of high purity can be produced (Table 4). Considering aforementioned, the simplest calculation shows that about 1/3rd of the EU market demands for CaCl2 of 100,000 tons annually [1] might be ensured by the proposed ESW transformation process using 5% (w/v) hydrochloric acid treatment if it is going to be applied on the industrial scale.
Besides CaCl2 anhydride as the major product obtained, the proposed ESW transformation process (Figure 1) generates at least two valuable by-products present in much lower amounts (lesser than 3%): adherent egg white protein solution (Figures 1 and 3) and ESMs (Figure 1, Table 4). While the possibility of the use of obtained adherent egg white protein solution was not examined in the current research, ESW-derived ESMs were tested on their suitability to be used as enzyme immobilization carriers. Obtained data (Figure 8) showed a great potential for ESW-derived ESM as BCL immobilization carrier, where up to 124 U of lipase activity per 1 g of wet ESM was achieved with immobilization by adsorption, implying ESM suitability to be used as an enzyme immobilization carrier. The observed is even more pronounced, considering the fact that ESM was found as a promising platform for the production of enzyme-based biosensors [48,49,50,51,52,53,54,55,56,57,58]. Thus, it seems quite possible that the currently proposed production of ESM (Figure 1) might end with the production of ESM pieces suitable for the development of biosensors, omitting the energy-demanding step of ESM milling to a size less than 0.5 mm necessary for successful enzyme immobilization. Nevertheless, the use of ESW-derived ESM for lipase immobilization (Figure 8) justifies the final step in ESM production, a milling procedure, where suitable enzyme immobilization carrier was achieved.
While the suitability of ESW-derived ESM for BCL immobilization by adsorption combining the effect of enzyme activity load and time of immobilization was proved (Figure 8), it should be noted that enzyme immobilization depends not only on the duration of immobilization and enzyme loading but on the other important factors including (a) carrier properties such as particle size, surface area, porosity, pore size, pore volume, and the presence of functional groups necessary for interaction with functional group of enzyme of interest; (b) properties of “free” enzyme such as solubility, pH and temperature stability, enzyme purity, the possibility of aggregation, etc.; (c) the buffer used for immobilization; and (d) pH and ionic strength of the buffer solution [76,84,85,86,87,88,89]. Therefore, examination of the applicability of ESM as a promising lipase immobilization carrier using various immobilization techniques is one of the future perspectives.
5 Conclusions
Innovative transformation technique of “industrial” ESW by 5% (w/v) hydrochloric acid treatment to CaCl2 anhydrous as suitable food-grade additive and ESMs as promising enzyme immobilization carrier, easily transferable to the industrial scale has been proposed. Besides its obvious potential in reducing ESW and subsequent reduction of costs of egg-breaking plants regarding ESW disposal, it clearly offers the possibility of achieving additional profit, if the currently proposed ESW transformation process is upscaled to the industrial scale ending with the production of high-value-added products including food-grade additive CaCl2 and ESMs as an enzyme immobilization carrier. Although the currently proposed ESW-transformation process might ensure aforementioned, there is still a space for achieving zero-waste model approach. This includes the necessity of resolving the fate of adherent egg white protein solutions and its potential application, as well as the possibility of the use of solid residue obtained after organic matter flocculation and subsequent filtration during the preparation of clarified ESW-derived CaCl2 solutions intended for spray drying. Aforementioned is even more pronounced if one considers that adherent egg white contains valuable proteins such as lysozyme and conalbumin, and that true eggshells (calcified matrix) contain highly valuable antimicrobial proteins which have been obviously precipitated during organic matter flocculation from CaCl2 solution. Therefore, it seems quite possible that solving currently unresolved issues of the proposed ESW transformation process might lead to the closing the circle, i.e., achieving profitable zero waste model of ESW transformation.
Acknowledgements
Authors are grateful to the Elcon-nutritional products Ltd. (Zlatar Bistrica, Croatia) for generous supply of industrial ESW. Ž.S. gratefully acknowledges the support of project CeNIKS co-financed by the Croatian Government and the EU through the European Regional Development Fund – Competitiveness and Cohesion Operational Program (Grant No. KK.01.1.1.02.0013).
-
Funding information: This work has been fully supported by the Croatian Science Foundation under the project IP-2020-02-6878.
-
Author contributions: Ivica Strelec: Conceptualization, investigation, methodology, supervision, validation, visualization, writing – original draft, writing – review and editing; Katarina Peranović: formal analysis, writing – original draft; Marta Ostojčić: formal analysis, writing – original draft; Krunoslav Aladić: methodology, writing – original draft; Hrvoje Pavlović: formal analysis, methodology, validation, writing – original draft; Igor Djerdj: methodology, validation, visualization, writing – review and editing; Dalibor Tatar: formal analysis, writing – original draft; Nikola Maravić: formal analysis, validation, writing – original draft; Željko Skoko: formal analysis, methodology, validation, visualization, writing – original draft, writing – review and editing; Sandra Budžaki: funding acquisition, investigation, project administration, resources, supervision, writing – review and editing.
-
Conflict of interest: Authors state no conflict of interest.
-
Data availability statement: The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
[1] European Chemicals Agency Substance Infocard – Calcium Chlorde. 2022. https://echa.europa.eu/substance-information/-/substanceinfo/100.030.115 (accessed on 17 June 2023).Search in Google Scholar
[2] Garrett DE. Part 2 – calcium chloride. In: Garrett DE, Ed. Handbook of Lithium and Natural Calcium Chloride. Oxford: Academic Press; 2004. p. 237–457.10.1016/B978-012276152-2/50038-4Search in Google Scholar
[3] Han, J. What Is Calcium Chloride (E509) in Food and Its Common Uses?. 2020. https://foodadditives.net/firming-agent/calcium-chloride/ (accessed on 15 June 2023).Search in Google Scholar
[4] PubChem Calcium Chloride. 2004. https://pubchem.ncbi.nlm.nih.gov/compound/5284359 (accessed on 17 June 2023).Search in Google Scholar
[5] Ropp RC. Chapter 2 – group 17 (H, F, Cl, Br, I) alkaline earth compounds. In: Ropp RC, editor. Encyclopedia of the Alkaline Earth Compounds. Amsterdam: Elsevier; 2013. p. 25–104.10.1016/B978-0-444-59550-8.00002-8Search in Google Scholar
[6] Nys Y, Gautron J. Structure and formation of the eggshell. In: Huopalahti R, López-Fandiño R, Anton M, Schade R, editor. Bioactive Egg Compounds. Berlin, Heidelberg: Springer; 2007. p. 99–102.10.1007/978-3-540-37885-3_15Search in Google Scholar
[7] Strelec I, Ostojčić M, Budžaki S. Transformacija ljuske kokošjih jaja u proizvode dodane vrijednosti. In: Neke mogućnosti iskorištenja nusproizvoda prehrambene industrije – Kn 3. Osijek: Sveučilište Josipa Jurja Strossmayera u Osijeku, Prehrambeno-tehnološki fakultet Osijek, Veleučilište u Požegi; 2021. p. 303–27.Search in Google Scholar
[8] Strelec I, Ostojčić M, Brekalo M, Hajra S, Kim H-J, Stanojev J, et al. Transformation of Eggshell waste to egg white protein solution, calcium chloride dihydrate, and eggshell membrane powder. Green Process Synth. 2023;12:20228151. 10.1515/gps-2022-8151.Search in Google Scholar
[9] Strelec I, Tomičić K, Zajec M, Ostojčić M, Budžaki S. Eggshell-waste-derived calcium acetate, calcium hydrogen phosphate and corresponding eggshell membranes. Appl Sci. 2023;13:7372. 10.3390/app13137372.Search in Google Scholar
[10] Food and Agriculture Organization of the United Nations(FAO) FAOSTAT. 1961. https://www.fao.org/faostat/en/#data/QCL (accessed on 15 June 2023).Search in Google Scholar
[11] Ahmed TAE, Wu L, Younes M, Hincke M. Biotechnological applications of eggshell: Recent advances. Front Bioeng Biotechnol. 2021;9:675364.10.3389/fbioe.2021.675364Search in Google Scholar PubMed PubMed Central
[12] Garcia-Garcia G, Stone J, Rahimifard S. Opportunities for waste valorisation in the food industry – A case study with four UK food manufacturers. J Clean Prod. 2019;211:1339–56. 10.1016/j.jclepro.2018.11.269.Search in Google Scholar
[13] Borges R, Soares Giroto A, Klaic R, Wypych F, Ribeiro C. Mechanochemical Synthesis of Eco-Friendly Fertilizer from Eggshell (Calcite) and KH2PO4. Adv Powder Technol. 2021;32:4070–7. 10.1016/j.apt.2021.09.013.Search in Google Scholar
[14] Gontard N, Sonesson U, Birkved M, Majone M, Bolzonella D, Celli A, et al. A research challenge vision regarding management of agricultural waste in a circular bio-based economy. Crit Rev Environ Sci Technol. 2018;48:614–54. 10.1080/10643389.2018.1471957.Search in Google Scholar
[15] Cordeiro CMM, Hincke MT. Recent patents on eggshell: shell and membrane applications. Recent Pat Food Nutr Agric. 2011;3:1–8. 10.2174/2212798411103010001.Search in Google Scholar PubMed
[16] Waheed M, Butt MS, Shehzad A, Adzahan NM, Shabbir MA, Suleria HAR, et al. Eggshell calcium: A cheap alternative to expensive supplements. Trends Food Sci Technol. 2019;91:219–30. 10.1016/j.tifs.2019.07.021.Search in Google Scholar
[17] Bradford MM. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem. 1976;72:248–54. 10.1006/abio.1976.9999.Search in Google Scholar PubMed
[18] Wrolstad RE, Acree TE, Decker EA, Penner MH, Reid DS, Schwartz SJ, et al. Handbook of Food Analytical Chemistry: Water, Proteins, Enzymes, Lipids, and Carbohydrates. Hoboken, New Jersey, USA: John Wiley and Sons, Inc; 2005.Search in Google Scholar
[19] Rodríguez-Carvajal J. Recent advances in magnetic structure determination by neutron powder diffraction. Phys B Condens Matter. 1993;192:55–69. 10.1016/0921-4526(93)90108-I.Search in Google Scholar
[20] Damena A, Mikru A, Adane M, Dobo B. Microbial profile and safety of chicken eggs from a poultry farm and small-scale vendors in hawassa, Southern Ethiopia. J Food Qual. 2022;2022:e7483253. 10.1155/2022/7483253 Search in Google Scholar
[21] EFSA Panel on Dietetic Products, Nutrition and Allergies (EFSA NDA Panel), Turck D, Bresson J-L, Burlingame B, Dean T, Fairweather-Tait S, et al. Safety of egg membrane hydrolysate as a novel food pursuant to regulation (EU) 2015/2283. EFSA J. 2018;16:e05363. 10.2903/j.efsa.2018.5363.Search in Google Scholar PubMed PubMed Central
[22] Németh C, Lang D, Tóth A, Surányi J, Friedrich L. Microbiological condition of “eggshell flour” in the food industry. J Hyg Eng Des. 2015;13:3–6.Search in Google Scholar
[23] Vanderzant C. Compendium of Methods for the Microbiological Examination of Foods. 3rd edn. Washington, DC: American Public Health Association; 1992.Search in Google Scholar
[24] International Standard Organization (ISO) Microbiology of the Food Chain — Horizontal Method for the Detection, Enumeration and Serotyping of Salmonella — Part 1: Detection of Salmonella Spp. 2017. https://www.iso.org/standard/56712.html (accessed on 17 October 2023).Search in Google Scholar
[25] Ballesteros LF, Teixeira JA, Mussatto SI. Chemical, functional, and structural properties of spent coffee grounds and coffee silverskin. Food Bioprocess Technol. 2014;7:3493–3503. 10.1007/s11947-014-1349-z.Search in Google Scholar
[26] Chattopadhyay S, Sen R. A comparative performance evaluation of jute and eggshell matrices to immobilize pancreatic lipase. Process Biochem. 2012;47:749–57. 10.1016/j.procbio.2012.02.003.Search in Google Scholar
[27] Salleh S, Serri NA, Hena S, Tajarudin HA. Preliminary studies on immobilization of lipase using chicken eggshell. IOP Conf Ser Earth Environ Sci. 2016;36:012026. 10.1088/1755-1315/36/1/012026.Search in Google Scholar
[28] Ostojčić M, Budžaki S, Flanjak I, Bilić Rajs B, Barišić I, Tran NN, et al. Production of biodiesel by Burkholderia cepacia lipase as a function of process parameters. Biotechnol Prog. 2021;37:e3109. 10.1002/btpr.3109.Search in Google Scholar PubMed
[29] Banaszewska D, Biesiada-Drzazga B, Ostrowski D, Drabik K, Batkowska J. The Impact of Breeder Age on Egg Quality and Lysozyme Activity. Turk J Vet Anim Sci. 2019;43:583–9. 10.3906/vet-1904-20.Search in Google Scholar
[30] Ray S, Barman A, Roy PK, Singh B. Chicken eggshell powder as dietary calcium source in chocolate cakes. Pharma Innov J. 2017;6(9):1–4.Search in Google Scholar
[31] Réhault-Godbert S, Guyot N, Nys Y. The golden egg: Nutritional value, bioactivities, and emerging benefits for human health. Nutrients. 2019;11:684. 10.3390/nu11030684.Search in Google Scholar PubMed PubMed Central
[32] Kocetkovs V, Radenkovs V, Juhnevica-Radenkova K, Jakovlevs D, Muizniece-Brasava S. The impact of eggshell thickness on the qualitative characteristics of stored eggs produced by three breeds of laying hens of the cage and cage-free housed systems. Appl Sci. 2022;12:11539. 10.3390/app122211539.Search in Google Scholar
[33] Walton HV, Cotterill OJ, Vandepopuliere JM. Composition of shell waste from egg breaking plants1. Poult Sci. 1973;52:1836–41. 10.3382/ps.0521836.Search in Google Scholar
[34] Bowden JH, Terry CT. Process for Preparing Powder and Granular Calcium Chloride Products. Alexandria, Virginia, USA: United State Patent and Trademark Office (USPTO); 1967.Search in Google Scholar
[35] Karunadasa KSP, Manoratne CH, Pitawala HMTGA, Rajapakse RMG. Relative stability of hydrated/anhydrous products of calcium chloride during complete dehydration as examined by high-temperature X-Ray powder diffraction. J Phys Chem Solids. 2018;120:167–72. 10.1016/j.jpcs.2018.04.034.Search in Google Scholar
[36] Garnjanagoonchorn W, Changpuak A. Preparation and partial characterization of eggshell calcium chloride. Int J Food Prop. 2007;10:497–503. 10.1080/10942910600919484.Search in Google Scholar
[37] European Union Commission Regulation (EU) No 231/2012 of 9 March 2012 Laying down Specifications for Food Additives Listed in Annexes II and III to Regulation (EC) No 1333/2008 of the European Parliament and of the Council (Text with EEA Relevance) - Publications Office of the EU. 2015. https://op.europa.eu/en/publication-detail/-/publication/a42dd9b2-b63f-438b-a790-1fa5995b7d41 (accessed on 23 April 2023).Search in Google Scholar
[38] Food and Agriculture Organization of the United Nations Combined Compendium of Food Additive Specification; Food and Agriculutre Organization of the United Nations, Italy. Vol. 4; 2006.Search in Google Scholar
[39] van Bever AK, Nieuwenkamp W. Die kristallstruktur von calciumchlorid, CaCl2. Z Für Krist - Cryst Mater. 1935;90:374–6. 10.1524/zkri.1935.90.1.374.Search in Google Scholar
[40] Wyckoff RWG. Distorted rutile structure. 2nd edn. New York, New York: Interscience Publishers. https://www.crystallography.net/cod/9009084.html (accessed on 7 November 2023).Search in Google Scholar
[41] Momma K, Izumi F. VESTA: A three-dimensional visualization system for electronic and structural analysis. J Appl Crystallogr. 2008;41:653–8. 10.1107/S0021889808012016.Search in Google Scholar
[42] Chousalkar KK, Khan S, McWhorter AR. Microbial quality, safety and storage of eggs. Curr Opin Food Sci. 2021;38:91–5. 10.1016/j.cofs.2020.10.022.Search in Google Scholar
[43] Park S, Choi KS, Lee D, Kim D, Lim KT, Lee K-H, et al. Eggshell membrane: Review and impact on engineering. Biosyst Eng. 2016;151:446–63. 10.1016/j.biosystemseng.2016.10.014.Search in Google Scholar
[44] Shi Y, Zhou K, Li D, Guyonnet V, Hincke MT, Mine Y. Avian eggshell membrane as a novel biomaterial: A review. Foods. 2021;10:2178. 10.3390/foods10092178.Search in Google Scholar PubMed PubMed Central
[45] Kulshreshtha G, Diep T, Hudson H-A, Hincke MT. High value applications and current commercial market for eggshell membranes and derived bioactives. Food Chem. 2022;382:132270. 10.1016/j.foodchem.2022.132270.Search in Google Scholar PubMed
[46] Mensah RA, Salim K, Peszko K, Diop S, Wong TH, Chau DY. The chicken eggshell membrane: A versatile, sustainable, biological material for translational biomedical applications. Biomed Mater Bristol Engl. 2023;18:042001. 10.1088/1748-605X/acd316.Search in Google Scholar PubMed
[47] Han C, Chen Y, Shi L, Chen H, Li L, Ning Z, et al. Advances in Eggshell Membrane Separation and Solubilization Technologies. Front Vet Sci. 2023;10:1116126.10.3389/fvets.2023.1116126Search in Google Scholar PubMed PubMed Central
[48] Choi MMF, Pang WSH, Xiao D, Wu X. An optical glucose biosensor with eggshell membrane as an enzyme immobilisation platform. Analyst. 2001;126:1558–63. 10.1039/B103205B.Search in Google Scholar
[49] Wu B, Zhang G, Shuang S, Choi MMF. Biosensors for determination of glucose with glucose oxidase immobilized on an eggshell membrane. Talanta. 2004;64:546–53. 10.1016/j.talanta.2004.03.050.Search in Google Scholar PubMed
[50] Aini BN, Siddiquee S, Ampon K, Rodrigues KF, Suryani S. Development of glucose biosensor based on ZnO nanoparticles film and glucose oxidase-immobilized eggshell membrane. Sens Bio-Sens Res. 2015;4:46–56. 10.1016/j.sbsr.2015.03.004.Search in Google Scholar
[51] Singh AK, Roychoudhury A, Jha SK. Reusable glucose sensor based on enzyme immobilized egg-shell membrane. Anal Sci Int J Jpn Soc Anal Chem. 2016;32:1077–82. 10.2116/analsci.32.1077.Search in Google Scholar PubMed
[52] Choi MMF, Yiu TP. Immobilization of beef liver catalase on eggshell membrane for fabrication of hydrogen peroxide biosensor. Enzyme Microb Technol. 2004;34:41–7. 10.1016/j.enzmictec.2003.08.005.Search in Google Scholar
[53] Xiao D, Choi MMF. Aspartame optical biosensor with bienzyme-immobilized eggshell membrane and oxygen-sensitive optode membrane. Anal Chem. 2002;74:863–70. 10.1021/ac001097a.Search in Google Scholar PubMed
[54] Zhang G, Liu D, Shuang S, Choi MMF. A homocysteine biosensor with eggshell membrane as an enzyme immobilization platform. Sens Actuators B Chem. 2006;114:936–42. 10.1016/j.snb.2005.08.011.Search in Google Scholar
[55] Joshi P, Joshi HC, Sanghi SK, Kundu S. Immobilization of monoamine oxidase on eggshell membrane and its application in designing an amperometric biosensor for dopamine. Microchim Acta. 2010;169:383–8. 10.1007/s00604-010-0346-9.Search in Google Scholar
[56] Pundir CS, Bhambi M, Chauhan NS. Chemical activation of egg shell membrane for covalent immobilization of enzymes and its evaluation as inert support in urinary oxalate determination. Talanta. 2009;77:1688–93. 10.1016/j.talanta.2008.10.004.Search in Google Scholar PubMed
[57] D’Souza SF, Kumar J, Jha SK, Kubal BS. Immobilization of the urease on eggshell membrane and its application in biosensor. Mater Sci Eng C. 2013;33:850–4. 10.1016/j.msec.2012.11.010.Search in Google Scholar PubMed
[58] Mensah RA, Jo SB, Kim H, Park S-M, Patel KD, Cho KJ, et al. The eggshell membrane: A potential biomaterial for corneal wound healing. J Biomater Appl. 2021;36:912–29. 10.1177/08853282211024040.Search in Google Scholar PubMed PubMed Central
[59] Brekalo M, Rajs BB, Aladić K, Jakobek L, Šereš Z, Krstović S, et al. Multistep extraction transformation of spent coffee grounds to the cellulose-based enzyme immobilization carrier. Sustainability. 2023;15:13142. 10.3390/su151713142.Search in Google Scholar
[60] Corici L, Ferrario V, Pellis A, Ebert C, Lotteria S, Cantone S, et al. Large scale applications of immobilized enzymes call for sustainable and inexpensive solutions: Rice husks as renewable alternatives to fossil-based organic resins. RSC Adv. 2016;6:63256–70. 10.1039/C6RA12065B.Search in Google Scholar
[61] Biró E, Németh AS, Sisak C, Feczkó T, Gyenis J. Preparation of chitosan particles suitable for enzyme immobilization. J Biochem Biophys Methods. 2008;70:1240–6. 10.1016/j.jprot.2007.11.005.Search in Google Scholar PubMed
[62] Ferrario V, Veny H, De Angelis E, Navarini L, Ebert C, Gardossi L. Lipases immobilization for effective synthesis of biodiesel starting from coffee waste oils. Biomolecules. 2013;3:514–34. 10.3390/biom3030514.Search in Google Scholar PubMed PubMed Central
[63] Brígida AIS, Pinheiro ÁDT, Ferreira ALO, Gonçalves LRB. Immobilization of candida antarctica lipase b by adsorption to green coconut fiber. Appl Biochem Biotechnol. 2008;146:173–87. 10.1007/s12010-007-8072-4.Search in Google Scholar PubMed
[64] Bonet-Ragel K, López-Pou L, Tutusaus G, Benaiges MD, Valero F. Rice husk ash as a potential carrier for the immobilization of lipases applied in the enzymatic production of biodiesel. Biocatal Biotransform. 2018;36:151–8. 10.1080/10242422.2017.1308498.Search in Google Scholar
[65] Cui C, Tao Y, Li L, Chen B, Tan T. Improving the Activity and stability of yarrowia lipolytica lipase lip2 by immobilization on polyethyleneimine-coated polyurethane foam. J Mol Catal B Enzym. 2013;91:59–66. 10.1016/j.molcatb.2013.03.001.Search in Google Scholar
[66] Ittrat P, Chacho T, Pholprayoon J, Suttiwarayanon N, Charoenpanich J. Application of Agriculture Waste as a Support for Lipase Immobilization. Biocatal Agric Biotechnol. 2014;3:77–82. 10.1016/j.bcab.2014.02.002.Search in Google Scholar
[67] Jasińska K, Zieniuk B, Jankiewicz U, Fabiszewska A. Bio-based materials versus synthetic polymers as a support in lipase immobilization: Impact on versatile enzyme activity. Catalysts. 2023;13:395. 10.3390/catal13020395.Search in Google Scholar
[68] Kandasamy R, John Kennedy L, Vidya C, Boopathy R, Sekaran G. Immobilization of acidic lipase derived from pseudomonas gessardii onto mesoporous activated carbon for the hydrolysis of olive oil. J Mol Catal B Enzym. 2010;62:58–65. 10.1016/j.molcatb.2009.09.004.Search in Google Scholar
[69] Lv J-S, Liu X-Y, Xu J-X, Deng Y-F, Wu Z, Wang Y-M, et al. Preparation and properties of adsorption material from corn stalks core when used for enzyme immobilization and the subsequent activities of the adsorbed enzymes. Ind Crop Prod. 2013;50:787–96. 10.1016/j.indcrop.2013.08.068.Search in Google Scholar
[70] Salis A, Sanjust E, Solinas V, Monduzzi M. Commercial lipase immobilization on accurel MP 1004 porous polypropylene. Biocatal Biotransform. 2005;23:381–6. 10.1080/10242420500296337.Search in Google Scholar
[71] Trbojević Ivić J, Veličković D, Dimitrijević A, Bezbradica D, Dragačević V, Gavrović Jankulović M, et al. Design of biocompatible immobilized candida rugosa lipase with potential application in food industry. J Sci Food Agric. 2016;96:4281–7. 10.1002/jsfa.7641.Search in Google Scholar PubMed
[72] Ulker C, Gokalp N, Guvenilir Y. Immobilization of candida antarzctica lipase B (CALB) on surface-modified rice husk ashes (RHA) via physical adsorption and cross-linking methods. Biocatal Biotransform. 2016;34:172–80. 10.1080/10242422.2016.1247818.Search in Google Scholar
[73] Jiang C, Cheng C, Hao M, Wang H, Wang Z, Shen C, et al. Enhanced catalytic stability of lipase immobilized on oxidized and disulfide-rich eggshell membrane for esters hydrolysis and transesterification. Int J Biol Macromol. 2017;105:1328–36. 10.1016/j.ijbiomac.2017.07.166.Search in Google Scholar PubMed
[74] Abdulla R, Sanny SA, Derman E. Stability studies of immobilized lipase on rice husk and eggshell membrane. IOP Conf Ser Mater Sci Eng. 2017;206:012032. 10.1088/1757-899X/206/1/012032.Search in Google Scholar
[75] Işık C, Saraç N, Teke M, Uğur A. A new bioremediation method for removal of wastewater containing oils with high oleic acid composition: Acinetobacter haemolyticus lipase immobilized on eggshell membrane with improved stabilities. N J Chem. 2021;45:1984–92. 10.1039/D0NJ05175F.Search in Google Scholar
[76] Boudrant J, Woodley JM, Fernandez-Lafuente R. Parameters necessary to define an immobilized enzyme preparation. Process Biochem. 2020;90:66–80. 10.1016/j.procbio.2019.11.026.Search in Google Scholar
[77] de Andrade Silva T, Keijok WJ, Guimarães MCC, Cassini STA, de Oliveira JP. Impact of immobilization strategies on the activity and recyclability of lipases in nanomagnetic supports. Sci Rep. 2022;12:6815. 10.1038/s41598-022-10721-y.Search in Google Scholar PubMed PubMed Central
[78] Stoytcheva M, Montero G, Zlatev R, Leon JA, Gochev V. Analytical methods for lipases activity determination: A review. Curr Anal Chem. 2012;8:400–7. 10.2174/157341112801264879.Search in Google Scholar
[79] Vorderwülbecke T, Kieslich K, Erdmann H. Comparison of lipases by different assays. Enzyme Microb Technol. 1992;14:631–9. 10.1016/0141-0229(92)90038-P.Search in Google Scholar
[80] Lopes DB, Fraga LP, Fleuri LF, Macedo GA. Lipase and esterase: To what extent can this classification be applied accurately. Food Sci Technol. 2011;31:603–13. 10.1590/S0101-20612011000300009.Search in Google Scholar
[81] Dalal S, Singh PK, Raghava S, Rawat S, Gupta MN. Purification and properties of the alkaline lipase from Burkholderia Cepacia A.T.C.C. 25609. Biotechnol Appl Biochem. 2008;51:23–31. 10.1042/BA20070186.Search in Google Scholar PubMed
[82] Sigma-Aldrich Calcium Chloride Anhydrous | Sigma-Aldrich. 2024. https://www.sigmaaldrich.com/HR/en/search/calcium-chloride-anhydrous?focus=products&page=1&perpage=30&sort=relevance&term=calcium%20chloride%20anhydrous&type=product (accessed on 12 March 2024).Search in Google Scholar
[83] European Parliament and Council of European Union Regulation (EC) No 1069/2009 of the European Parliament and of the Council of 21 October 2009 Laying down Health Rules as Regards Animal by-Products and Derived Products Not Intended for Human Consumption and Repealing Regulation (EC) No 1774/2002 (Animal by-Products Regulation). Off. J. Eur. Union vol. 48; 2009. p. 1–34. doi: 10.3000/17252555.L_2009.300.eng.Search in Google Scholar
[84] Bié J, Sepodes B, Fernandes PCB, Ribeiro MHL. Enzyme immobilization and co-immobilization: Main framework, advances and some applications. Processes. 2022;10:494. 10.3390/pr10030494.Search in Google Scholar
[85] Jesionowski T, Zdarta J, Krajewska B. Enzyme immobilization by adsorption: A review. Adsorption. 2014;20:801–21. 10.1007/s10450-014-9623-y.Search in Google Scholar
[86] Messing RA. Immobilized enzymes for industrial reactors. Cambridge, Massachusetts, USA: Academic Press; 1975.Search in Google Scholar
[87] Mokhtar NF, Rahman RNZR, Muhd Noor ND, Mohd Shariff F, Mohamad Ali MS. The immobilization of lipases on porous support by adsorption and hydrophobic interaction method. Catalysts. 2020;10:744. 10.3390/catal10070744.Search in Google Scholar
[88] Yuan Y, Shen J, Salmon S. Developing enzyme immobilization with fibrous membranes: Longevity and characterization considerations. Membranes. 2023;13:532. 10.3390/membranes13050532.Search in Google Scholar PubMed PubMed Central
[89] Zdarta J, Meyer AS, Jesionowski T, Pinelo M. A general overview of support materials for enzyme immobilization: Characteristics, properties, practical utility. Catalysts. 2018;8:92. 10.3390/catal8020092.Search in Google Scholar
© 2024 the author(s), published by De Gruyter
This work is licensed under the Creative Commons Attribution 4.0 International License.
Articles in the same Issue
- Research Articles
- Green polymer electrolyte and activated charcoal-based supercapacitor for energy harvesting application: Electrochemical characteristics
- Research on the adsorption of Co2+ ions using halloysite clay and the ability to recover them by electrodeposition method
- Simultaneous estimation of ibuprofen, caffeine, and paracetamol in commercial products using a green reverse-phase HPTLC method
- Isolation, screening and optimization of alkaliphilic cellulolytic fungi for production of cellulase
- Functionalized gold nanoparticles coated with bacterial alginate and their antibacterial and anticancer activities
- Comparative analysis of bio-based amino acid surfactants obtained via Diels–Alder reaction of cyclic anhydrides
- Biosynthesis of silver nanoparticles on yellow phosphorus slag and its application in organic coatings
- Exploring antioxidant potential and phenolic compound extraction from Vitis vinifera L. using ultrasound-assisted extraction
- Manganese and copper-coated nickel oxide nanoparticles synthesized from Carica papaya leaf extract induce antimicrobial activity and breast cancer cell death by triggering mitochondrial caspases and p53
- Insight into heating method and Mozafari method as green processing techniques for the synthesis of micro- and nano-drug carriers
- Silicotungstic acid supported on Bi-based MOF-derived metal oxide for photodegradation of organic dyes
- Synthesis and characterization of capsaicin nanoparticles: An attempt to enhance its bioavailability and pharmacological actions
- Synthesis of Lawsonia inermis-encased silver–copper bimetallic nanoparticles with antioxidant, antibacterial, and cytotoxic activity
- Facile, polyherbal drug-mediated green synthesis of CuO nanoparticles and their potent biological applications
- Zinc oxide-manganese oxide/carboxymethyl cellulose-folic acid-sesamol hybrid nanomaterials: A molecularly targeted strategy for advanced triple-negative breast cancer therapy
- Exploring the antimicrobial potential of biogenically synthesized graphene oxide nanoparticles against targeted bacterial and fungal pathogens
- Biofabrication of silver nanoparticles using Uncaria tomentosa L.: Insight into characterization, antibacterial activities combined with antibiotics, and effect on Triticum aestivum germination
- Membrane distillation of synthetic urine for use in space structural habitat systems
- Investigation on mechanical properties of the green synthesis bamboo fiber/eggshell/coconut shell powder-based hybrid biocomposites under NaOH conditions
- Green synthesis of magnesium oxide nanoparticles using endophytic fungal strain to improve the growth, metabolic activities, yield traits, and phenolic compounds content of Nigella sativa L.
- Estimation of greenhouse gas emissions from rice and annual upland crops in Red River Delta of Vietnam using the denitrification–decomposition model
- Synthesis of humic acid with the obtaining of potassium humate based on coal waste from the Lenger deposit, Kazakhstan
- Ascorbic acid-mediated selenium nanoparticles as potential antihyperuricemic, antioxidant, anticoagulant, and thrombolytic agents
- Green synthesis of silver nanoparticles using Illicium verum extract: Optimization and characterization for biomedical applications
- Antibacterial and dynamical behaviour of silicon nanoparticles influenced sustainable waste flax fibre-reinforced epoxy composite for biomedical application
- Optimising coagulation/flocculation using response surface methodology and application of floc in biofertilisation
- Green synthesis and multifaceted characterization of iron oxide nanoparticles derived from Senna bicapsularis for enhanced in vitro and in vivo biological investigation
- Potent antibacterial nanocomposites from okra mucilage/chitosan/silver nanoparticles for multidrug-resistant Salmonella Typhimurium eradication
- Trachyspermum copticum aqueous seed extract-derived silver nanoparticles: Exploration of their structural characterization and comparative antibacterial performance against gram-positive and gram-negative bacteria
- Microwave-assisted ultrafine silver nanoparticle synthesis using Mitragyna speciosa for antimalarial applications
- Green synthesis and characterisation of spherical structure Ag/Fe2O3/TiO2 nanocomposite using acacia in the presence of neem and tulsi oils
- Green quantitative methods for linagliptin and empagliflozin in dosage forms
- Enhancement efficacy of omeprazole by conjugation with silver nanoparticles as a urease inhibitor
- Residual, sequential extraction, and ecological risk assessment of some metals in ash from municipal solid waste incineration, Vietnam
- Green synthesis of ZnO nanoparticles using the mangosteen (Garcinia mangostana L.) leaf extract: Comparative preliminary in vitro antibacterial study
- Simultaneous determination of lesinurad and febuxostat in commercial fixed-dose combinations using a greener normal-phase HPTLC method
- A greener RP-HPLC method for quaternary estimation of caffeine, paracetamol, levocetirizine, and phenylephrine acquiring AQbD with stability studies
- Optimization of biomass durian peel as a heterogeneous catalyst in biodiesel production using microwave irradiation
- Thermal treatment impact on the evolution of active phases in layered double hydroxide-based ZnCr photocatalysts: Photodegradation and antibacterial performance
- Preparation of silymarin-loaded zein polysaccharide core–shell nanostructures and evaluation of their biological potentials
- Preparation and characterization of composite-modified PA6 fiber for spectral heating and heat storage applications
- Preparation and electrocatalytic oxygen evolution of bimetallic phosphates (NiFe)2P/NF
- Rod-shaped Mo(vi) trichalcogenide–Mo(vi) oxide decorated on poly(1-H pyrrole) as a promising nanocomposite photoelectrode for green hydrogen generation from sewage water with high efficiency
- Green synthesis and studies on citrus medica leaf extract-mediated Au–ZnO nanocomposites: A sustainable approach for efficient photocatalytic degradation of rhodamine B dye in aqueous media
- Cellulosic materials for the removal of ciprofloxacin from aqueous environments
- The analytical assessment of metal contamination in industrial soils of Saudi Arabia using the inductively coupled plasma technology
- The effect of modified oily sludge on the slurry ability and combustion performance of coal water slurry
- Eggshell waste transformation to calcium chloride anhydride as food-grade additive and eggshell membranes as enzyme immobilization carrier
- Synthesis of EPAN and applications in the encapsulation of potassium humate
- Biosynthesis and characterization of silver nanoparticles from Cedrela toona leaf extracts: An exploration into their antibacterial, anticancer, and antioxidant potential
- Enhancing mechanical and rheological properties of HDPE films through annealing for eco-friendly agricultural applications
- Immobilisation of catalase purified from mushroom (Hydnum repandum) onto glutaraldehyde-activated chitosan and characterisation: Its application for the removal of hydrogen peroxide from artificial wastewater
- Sodium titanium oxide/zinc oxide (STO/ZnO) photocomposites for efficient dye degradation applications
- Effect of ex situ, eco-friendly ZnONPs incorporating green synthesised Moringa oleifera leaf extract in enhancing biochemical and molecular aspects of Vicia faba L. under salt stress
- Biosynthesis and characterization of selenium and silver nanoparticles using Trichoderma viride filtrate and their impact on Culex pipiens
- Photocatalytic degradation of organic dyes and biological potentials of biogenic zinc oxide nanoparticles synthesized using the polar extract of Cyperus scariosus R.Br. (Cyperaceae)
- Assessment of antiproliferative activity of green-synthesized nickel oxide nanoparticles against glioblastoma cells using Terminalia chebula
- Chlorine-free synthesis of phosphinic derivatives by change in the P-function
- Anticancer, antioxidant, and antimicrobial activities of nanoemulsions based on water-in-olive oil and loaded on biogenic silver nanoparticles
- Study and mechanism of formation of phosphorus production waste in Kazakhstan
- Synthesis and stabilization of anatase form of biomimetic TiO2 nanoparticles for enhancing anti-tumor potential
- Microwave-supported one-pot reaction for the synthesis of 5-alkyl/arylidene-2-(morpholin/thiomorpholin-4-yl)-1,3-thiazol-4(5H)-one derivatives over MgO solid base
- Screening the phytochemicals in Perilla leaves and phytosynthesis of bioactive silver nanoparticles for potential antioxidant and wound-healing application
- Graphene oxide/chitosan/manganese/folic acid-brucine functionalized nanocomposites show anticancer activity against liver cancer cells
- Nature of serpentinite interactions with low-concentration sulfuric acid solutions
- Multi-objective statistical optimisation utilising response surface methodology to predict engine performance using biofuels from waste plastic oil in CRDi engines
- Microwave-assisted extraction of acetosolv lignin from sugarcane bagasse and electrospinning of lignin/PEO nanofibres for carbon fibre production
- Biosynthesis, characterization, and investigation of cytotoxic activities of selenium nanoparticles utilizing Limosilactobacillus fermentum
- Highly photocatalytic materials based on the decoration of poly(O-chloroaniline) with molybdenum trichalcogenide oxide for green hydrogen generation from Red Sea water
- Highly efficient oil–water separation using superhydrophobic cellulose aerogels derived from corn straw
- Beta-cyclodextrin–Phyllanthus emblica emulsion for zinc oxide nanoparticles: Characteristics and photocatalysis
- Assessment of antimicrobial activity and methyl orange dye removal by Klebsiella pneumoniae-mediated silver nanoparticles
- Influential eradication of resistant Salmonella Typhimurium using bioactive nanocomposites from chitosan and radish seed-synthesized nanoselenium
- Antimicrobial activities and neuroprotective potential for Alzheimer’s disease of pure, Mn, Co, and Al-doped ZnO ultra-small nanoparticles
- Green synthesis of silver nanoparticles from Bauhinia variegata and their biological applications
- Synthesis and optimization of long-chain fatty acids via the oxidation of long-chain fatty alcohols
- Eminent Red Sea water hydrogen generation via a Pb(ii)-iodide/poly(1H-pyrrole) nanocomposite photocathode
- Green synthesis and effective genistein production by fungal β-glucosidase immobilized on Al2O3 nanocrystals synthesized in Cajanus cajan L. (Millsp.) leaf extracts
- Green stability-indicating RP-HPTLC technique for determining croconazole hydrochloride
- Green synthesis of La2O3–LaPO4 nanocomposites using Charybdis natator for DNA binding, cytotoxic, catalytic, and luminescence applications
- Eco-friendly drugs induce cellular changes in colistin-resistant bacteria
- Tangerine fruit peel extract mediated biogenic synthesized silver nanoparticles and their potential antimicrobial, antioxidant, and cytotoxic assessments
- Green synthesis on performance characteristics of a direct injection diesel engine using sandbox seed oil
- A highly sensitive β-AKBA-Ag-based fluorescent “turn off” chemosensor for rapid detection of abamectin in tomatoes
- Green synthesis and physical characterization of zinc oxide nanoparticles (ZnO NPs) derived from the methanol extract of Euphorbia dracunculoides Lam. (Euphorbiaceae) with enhanced biosafe applications
- Detection of morphine and data processing using surface plasmon resonance imaging sensor
- Effects of nanoparticles on the anaerobic digestion properties of sulfamethoxazole-containing chicken manure and analysis of bio-enzymes
- Bromic acid-thiourea synergistic leaching of sulfide gold ore
- Green chemistry approach to synthesize titanium dioxide nanoparticles using Fagonia Cretica extract, novel strategy for developing antimicrobial and antidiabetic therapies
- Green synthesis and effective utilization of biogenic Al2O3-nanocoupled fungal lipase in the resolution of active homochiral 2-octanol and its immobilization via aluminium oxide nanoparticles
- Eco-friendly RP-HPLC approach for simultaneously estimating the promising combination of pentoxifylline and simvastatin in therapeutic potential for breast cancer: Appraisal of greenness, whiteness, and Box–Behnken design
- Use of a humidity adsorbent derived from cockleshell waste in Thai fried fish crackers (Keropok)
- One-pot green synthesis, biological evaluation, and in silico study of pyrazole derivatives obtained from chalcones
- Bio-sorption of methylene blue and production of biofuel by brown alga Cystoseira sp. collected from Neom region, Kingdom of Saudi Arabia
- Synthesis of motexafin gadolinium: A promising radiosensitizer and imaging agent for cancer therapy
- The impact of varying sizes of silver nanoparticles on the induction of cellular damage in Klebsiella pneumoniae involving diverse mechanisms
- Microwave-assisted green synthesis, characterization, and in vitro antibacterial activity of NiO nanoparticles obtained from lemon peel extract
- Rhus microphylla-mediated biosynthesis of copper oxide nanoparticles for enhanced antibacterial and antibiofilm efficacy
- Harnessing trichalcogenide–molybdenum(vi) sulfide and molybdenum(vi) oxide within poly(1-amino-2-mercaptobenzene) frameworks as a photocathode for sustainable green hydrogen production from seawater without sacrificial agents
- Magnetically recyclable Fe3O4@SiO2 supported phosphonium ionic liquids for efficient and sustainable transformation of CO2 into oxazolidinones
- A comparative study of Fagonia arabica fabricated silver sulfide nanoparticles (Ag2S) and silver nanoparticles (AgNPs) with distinct antimicrobial, anticancer, and antioxidant properties
- Visible light photocatalytic degradation and biological activities of Aegle marmelos-mediated cerium oxide nanoparticles
- Physical intrinsic characteristics of spheroidal particles in coal gasification fine slag
- Exploring the effect of tea dust magnetic biochar on agricultural crops grown in polycyclic aromatic hydrocarbon contaminated soil
- Crosslinked chitosan-modified ultrafiltration membranes for efficient surface water treatment and enhanced anti-fouling performances
- Study on adsorption characteristics of biochars and their modified biochars for removal of organic dyes from aqueous solution
- Zein polymer nanocarrier for Ocimum basilicum var. purpurascens extract: Potential biomedical use
- Green synthesis, characterization, and in vitro and in vivo biological screening of iron oxide nanoparticles (Fe3O4) generated with hydroalcoholic extract of aerial parts of Euphorbia milii
- Novel microwave-based green approach for the synthesis of dual-loaded cyclodextrin nanosponges: Characterization, pharmacodynamics, and pharmacokinetics evaluation
- Bi2O3–BiOCl/poly-m-methyl aniline nanocomposite thin film for broad-spectrum light-sensing
- Green synthesis and characterization of CuO/ZnO nanocomposite using Musa acuminata leaf extract for cytotoxic studies on colorectal cancer cells (HCC2998)
- Review Articles
- Materials-based drug delivery approaches: Recent advances and future perspectives
- A review of thermal treatment for bamboo and its composites
- An overview of the role of nanoherbicides in tackling challenges of weed management in wheat: A novel approach
- An updated review on carbon nanomaterials: Types, synthesis, functionalization and applications, degradation and toxicity
- Special Issue: Emerging green nanomaterials for sustainable waste management and biomedical applications
- Green synthesis of silver nanoparticles using mature-pseudostem extracts of Alpinia nigra and their bioactivities
- Special Issue: New insights into nanopythotechnology: current trends and future prospects
- Green synthesis of FeO nanoparticles from coffee and its application for antibacterial, antifungal, and anti-oxidation activity
- Dye degradation activity of biogenically synthesized Cu/Fe/Ag trimetallic nanoparticles
- Special Issue: Composites and green composites
- Recent trends and advancements in the utilization of green composites and polymeric nanocarriers for enhancing food quality and sustainable processing
- Retraction
- Retraction of “Biosynthesis and characterization of silver nanoparticles from Cedrela toona leaf extracts: An exploration into their antibacterial, anticancer, and antioxidant potential”
- Retraction of “Photocatalytic degradation of organic dyes and biological potentials of biogenic zinc oxide nanoparticles synthesized using the polar extract of Cyperus scariosus R.Br. (Cyperaceae)”
- Retraction to “Green synthesis on performance characteristics of a direct injection diesel engine using sandbox seed oil”
Articles in the same Issue
- Research Articles
- Green polymer electrolyte and activated charcoal-based supercapacitor for energy harvesting application: Electrochemical characteristics
- Research on the adsorption of Co2+ ions using halloysite clay and the ability to recover them by electrodeposition method
- Simultaneous estimation of ibuprofen, caffeine, and paracetamol in commercial products using a green reverse-phase HPTLC method
- Isolation, screening and optimization of alkaliphilic cellulolytic fungi for production of cellulase
- Functionalized gold nanoparticles coated with bacterial alginate and their antibacterial and anticancer activities
- Comparative analysis of bio-based amino acid surfactants obtained via Diels–Alder reaction of cyclic anhydrides
- Biosynthesis of silver nanoparticles on yellow phosphorus slag and its application in organic coatings
- Exploring antioxidant potential and phenolic compound extraction from Vitis vinifera L. using ultrasound-assisted extraction
- Manganese and copper-coated nickel oxide nanoparticles synthesized from Carica papaya leaf extract induce antimicrobial activity and breast cancer cell death by triggering mitochondrial caspases and p53
- Insight into heating method and Mozafari method as green processing techniques for the synthesis of micro- and nano-drug carriers
- Silicotungstic acid supported on Bi-based MOF-derived metal oxide for photodegradation of organic dyes
- Synthesis and characterization of capsaicin nanoparticles: An attempt to enhance its bioavailability and pharmacological actions
- Synthesis of Lawsonia inermis-encased silver–copper bimetallic nanoparticles with antioxidant, antibacterial, and cytotoxic activity
- Facile, polyherbal drug-mediated green synthesis of CuO nanoparticles and their potent biological applications
- Zinc oxide-manganese oxide/carboxymethyl cellulose-folic acid-sesamol hybrid nanomaterials: A molecularly targeted strategy for advanced triple-negative breast cancer therapy
- Exploring the antimicrobial potential of biogenically synthesized graphene oxide nanoparticles against targeted bacterial and fungal pathogens
- Biofabrication of silver nanoparticles using Uncaria tomentosa L.: Insight into characterization, antibacterial activities combined with antibiotics, and effect on Triticum aestivum germination
- Membrane distillation of synthetic urine for use in space structural habitat systems
- Investigation on mechanical properties of the green synthesis bamboo fiber/eggshell/coconut shell powder-based hybrid biocomposites under NaOH conditions
- Green synthesis of magnesium oxide nanoparticles using endophytic fungal strain to improve the growth, metabolic activities, yield traits, and phenolic compounds content of Nigella sativa L.
- Estimation of greenhouse gas emissions from rice and annual upland crops in Red River Delta of Vietnam using the denitrification–decomposition model
- Synthesis of humic acid with the obtaining of potassium humate based on coal waste from the Lenger deposit, Kazakhstan
- Ascorbic acid-mediated selenium nanoparticles as potential antihyperuricemic, antioxidant, anticoagulant, and thrombolytic agents
- Green synthesis of silver nanoparticles using Illicium verum extract: Optimization and characterization for biomedical applications
- Antibacterial and dynamical behaviour of silicon nanoparticles influenced sustainable waste flax fibre-reinforced epoxy composite for biomedical application
- Optimising coagulation/flocculation using response surface methodology and application of floc in biofertilisation
- Green synthesis and multifaceted characterization of iron oxide nanoparticles derived from Senna bicapsularis for enhanced in vitro and in vivo biological investigation
- Potent antibacterial nanocomposites from okra mucilage/chitosan/silver nanoparticles for multidrug-resistant Salmonella Typhimurium eradication
- Trachyspermum copticum aqueous seed extract-derived silver nanoparticles: Exploration of their structural characterization and comparative antibacterial performance against gram-positive and gram-negative bacteria
- Microwave-assisted ultrafine silver nanoparticle synthesis using Mitragyna speciosa for antimalarial applications
- Green synthesis and characterisation of spherical structure Ag/Fe2O3/TiO2 nanocomposite using acacia in the presence of neem and tulsi oils
- Green quantitative methods for linagliptin and empagliflozin in dosage forms
- Enhancement efficacy of omeprazole by conjugation with silver nanoparticles as a urease inhibitor
- Residual, sequential extraction, and ecological risk assessment of some metals in ash from municipal solid waste incineration, Vietnam
- Green synthesis of ZnO nanoparticles using the mangosteen (Garcinia mangostana L.) leaf extract: Comparative preliminary in vitro antibacterial study
- Simultaneous determination of lesinurad and febuxostat in commercial fixed-dose combinations using a greener normal-phase HPTLC method
- A greener RP-HPLC method for quaternary estimation of caffeine, paracetamol, levocetirizine, and phenylephrine acquiring AQbD with stability studies
- Optimization of biomass durian peel as a heterogeneous catalyst in biodiesel production using microwave irradiation
- Thermal treatment impact on the evolution of active phases in layered double hydroxide-based ZnCr photocatalysts: Photodegradation and antibacterial performance
- Preparation of silymarin-loaded zein polysaccharide core–shell nanostructures and evaluation of their biological potentials
- Preparation and characterization of composite-modified PA6 fiber for spectral heating and heat storage applications
- Preparation and electrocatalytic oxygen evolution of bimetallic phosphates (NiFe)2P/NF
- Rod-shaped Mo(vi) trichalcogenide–Mo(vi) oxide decorated on poly(1-H pyrrole) as a promising nanocomposite photoelectrode for green hydrogen generation from sewage water with high efficiency
- Green synthesis and studies on citrus medica leaf extract-mediated Au–ZnO nanocomposites: A sustainable approach for efficient photocatalytic degradation of rhodamine B dye in aqueous media
- Cellulosic materials for the removal of ciprofloxacin from aqueous environments
- The analytical assessment of metal contamination in industrial soils of Saudi Arabia using the inductively coupled plasma technology
- The effect of modified oily sludge on the slurry ability and combustion performance of coal water slurry
- Eggshell waste transformation to calcium chloride anhydride as food-grade additive and eggshell membranes as enzyme immobilization carrier
- Synthesis of EPAN and applications in the encapsulation of potassium humate
- Biosynthesis and characterization of silver nanoparticles from Cedrela toona leaf extracts: An exploration into their antibacterial, anticancer, and antioxidant potential
- Enhancing mechanical and rheological properties of HDPE films through annealing for eco-friendly agricultural applications
- Immobilisation of catalase purified from mushroom (Hydnum repandum) onto glutaraldehyde-activated chitosan and characterisation: Its application for the removal of hydrogen peroxide from artificial wastewater
- Sodium titanium oxide/zinc oxide (STO/ZnO) photocomposites for efficient dye degradation applications
- Effect of ex situ, eco-friendly ZnONPs incorporating green synthesised Moringa oleifera leaf extract in enhancing biochemical and molecular aspects of Vicia faba L. under salt stress
- Biosynthesis and characterization of selenium and silver nanoparticles using Trichoderma viride filtrate and their impact on Culex pipiens
- Photocatalytic degradation of organic dyes and biological potentials of biogenic zinc oxide nanoparticles synthesized using the polar extract of Cyperus scariosus R.Br. (Cyperaceae)
- Assessment of antiproliferative activity of green-synthesized nickel oxide nanoparticles against glioblastoma cells using Terminalia chebula
- Chlorine-free synthesis of phosphinic derivatives by change in the P-function
- Anticancer, antioxidant, and antimicrobial activities of nanoemulsions based on water-in-olive oil and loaded on biogenic silver nanoparticles
- Study and mechanism of formation of phosphorus production waste in Kazakhstan
- Synthesis and stabilization of anatase form of biomimetic TiO2 nanoparticles for enhancing anti-tumor potential
- Microwave-supported one-pot reaction for the synthesis of 5-alkyl/arylidene-2-(morpholin/thiomorpholin-4-yl)-1,3-thiazol-4(5H)-one derivatives over MgO solid base
- Screening the phytochemicals in Perilla leaves and phytosynthesis of bioactive silver nanoparticles for potential antioxidant and wound-healing application
- Graphene oxide/chitosan/manganese/folic acid-brucine functionalized nanocomposites show anticancer activity against liver cancer cells
- Nature of serpentinite interactions with low-concentration sulfuric acid solutions
- Multi-objective statistical optimisation utilising response surface methodology to predict engine performance using biofuels from waste plastic oil in CRDi engines
- Microwave-assisted extraction of acetosolv lignin from sugarcane bagasse and electrospinning of lignin/PEO nanofibres for carbon fibre production
- Biosynthesis, characterization, and investigation of cytotoxic activities of selenium nanoparticles utilizing Limosilactobacillus fermentum
- Highly photocatalytic materials based on the decoration of poly(O-chloroaniline) with molybdenum trichalcogenide oxide for green hydrogen generation from Red Sea water
- Highly efficient oil–water separation using superhydrophobic cellulose aerogels derived from corn straw
- Beta-cyclodextrin–Phyllanthus emblica emulsion for zinc oxide nanoparticles: Characteristics and photocatalysis
- Assessment of antimicrobial activity and methyl orange dye removal by Klebsiella pneumoniae-mediated silver nanoparticles
- Influential eradication of resistant Salmonella Typhimurium using bioactive nanocomposites from chitosan and radish seed-synthesized nanoselenium
- Antimicrobial activities and neuroprotective potential for Alzheimer’s disease of pure, Mn, Co, and Al-doped ZnO ultra-small nanoparticles
- Green synthesis of silver nanoparticles from Bauhinia variegata and their biological applications
- Synthesis and optimization of long-chain fatty acids via the oxidation of long-chain fatty alcohols
- Eminent Red Sea water hydrogen generation via a Pb(ii)-iodide/poly(1H-pyrrole) nanocomposite photocathode
- Green synthesis and effective genistein production by fungal β-glucosidase immobilized on Al2O3 nanocrystals synthesized in Cajanus cajan L. (Millsp.) leaf extracts
- Green stability-indicating RP-HPTLC technique for determining croconazole hydrochloride
- Green synthesis of La2O3–LaPO4 nanocomposites using Charybdis natator for DNA binding, cytotoxic, catalytic, and luminescence applications
- Eco-friendly drugs induce cellular changes in colistin-resistant bacteria
- Tangerine fruit peel extract mediated biogenic synthesized silver nanoparticles and their potential antimicrobial, antioxidant, and cytotoxic assessments
- Green synthesis on performance characteristics of a direct injection diesel engine using sandbox seed oil
- A highly sensitive β-AKBA-Ag-based fluorescent “turn off” chemosensor for rapid detection of abamectin in tomatoes
- Green synthesis and physical characterization of zinc oxide nanoparticles (ZnO NPs) derived from the methanol extract of Euphorbia dracunculoides Lam. (Euphorbiaceae) with enhanced biosafe applications
- Detection of morphine and data processing using surface plasmon resonance imaging sensor
- Effects of nanoparticles on the anaerobic digestion properties of sulfamethoxazole-containing chicken manure and analysis of bio-enzymes
- Bromic acid-thiourea synergistic leaching of sulfide gold ore
- Green chemistry approach to synthesize titanium dioxide nanoparticles using Fagonia Cretica extract, novel strategy for developing antimicrobial and antidiabetic therapies
- Green synthesis and effective utilization of biogenic Al2O3-nanocoupled fungal lipase in the resolution of active homochiral 2-octanol and its immobilization via aluminium oxide nanoparticles
- Eco-friendly RP-HPLC approach for simultaneously estimating the promising combination of pentoxifylline and simvastatin in therapeutic potential for breast cancer: Appraisal of greenness, whiteness, and Box–Behnken design
- Use of a humidity adsorbent derived from cockleshell waste in Thai fried fish crackers (Keropok)
- One-pot green synthesis, biological evaluation, and in silico study of pyrazole derivatives obtained from chalcones
- Bio-sorption of methylene blue and production of biofuel by brown alga Cystoseira sp. collected from Neom region, Kingdom of Saudi Arabia
- Synthesis of motexafin gadolinium: A promising radiosensitizer and imaging agent for cancer therapy
- The impact of varying sizes of silver nanoparticles on the induction of cellular damage in Klebsiella pneumoniae involving diverse mechanisms
- Microwave-assisted green synthesis, characterization, and in vitro antibacterial activity of NiO nanoparticles obtained from lemon peel extract
- Rhus microphylla-mediated biosynthesis of copper oxide nanoparticles for enhanced antibacterial and antibiofilm efficacy
- Harnessing trichalcogenide–molybdenum(vi) sulfide and molybdenum(vi) oxide within poly(1-amino-2-mercaptobenzene) frameworks as a photocathode for sustainable green hydrogen production from seawater without sacrificial agents
- Magnetically recyclable Fe3O4@SiO2 supported phosphonium ionic liquids for efficient and sustainable transformation of CO2 into oxazolidinones
- A comparative study of Fagonia arabica fabricated silver sulfide nanoparticles (Ag2S) and silver nanoparticles (AgNPs) with distinct antimicrobial, anticancer, and antioxidant properties
- Visible light photocatalytic degradation and biological activities of Aegle marmelos-mediated cerium oxide nanoparticles
- Physical intrinsic characteristics of spheroidal particles in coal gasification fine slag
- Exploring the effect of tea dust magnetic biochar on agricultural crops grown in polycyclic aromatic hydrocarbon contaminated soil
- Crosslinked chitosan-modified ultrafiltration membranes for efficient surface water treatment and enhanced anti-fouling performances
- Study on adsorption characteristics of biochars and their modified biochars for removal of organic dyes from aqueous solution
- Zein polymer nanocarrier for Ocimum basilicum var. purpurascens extract: Potential biomedical use
- Green synthesis, characterization, and in vitro and in vivo biological screening of iron oxide nanoparticles (Fe3O4) generated with hydroalcoholic extract of aerial parts of Euphorbia milii
- Novel microwave-based green approach for the synthesis of dual-loaded cyclodextrin nanosponges: Characterization, pharmacodynamics, and pharmacokinetics evaluation
- Bi2O3–BiOCl/poly-m-methyl aniline nanocomposite thin film for broad-spectrum light-sensing
- Green synthesis and characterization of CuO/ZnO nanocomposite using Musa acuminata leaf extract for cytotoxic studies on colorectal cancer cells (HCC2998)
- Review Articles
- Materials-based drug delivery approaches: Recent advances and future perspectives
- A review of thermal treatment for bamboo and its composites
- An overview of the role of nanoherbicides in tackling challenges of weed management in wheat: A novel approach
- An updated review on carbon nanomaterials: Types, synthesis, functionalization and applications, degradation and toxicity
- Special Issue: Emerging green nanomaterials for sustainable waste management and biomedical applications
- Green synthesis of silver nanoparticles using mature-pseudostem extracts of Alpinia nigra and their bioactivities
- Special Issue: New insights into nanopythotechnology: current trends and future prospects
- Green synthesis of FeO nanoparticles from coffee and its application for antibacterial, antifungal, and anti-oxidation activity
- Dye degradation activity of biogenically synthesized Cu/Fe/Ag trimetallic nanoparticles
- Special Issue: Composites and green composites
- Recent trends and advancements in the utilization of green composites and polymeric nanocarriers for enhancing food quality and sustainable processing
- Retraction
- Retraction of “Biosynthesis and characterization of silver nanoparticles from Cedrela toona leaf extracts: An exploration into their antibacterial, anticancer, and antioxidant potential”
- Retraction of “Photocatalytic degradation of organic dyes and biological potentials of biogenic zinc oxide nanoparticles synthesized using the polar extract of Cyperus scariosus R.Br. (Cyperaceae)”
- Retraction to “Green synthesis on performance characteristics of a direct injection diesel engine using sandbox seed oil”

