Abstract
The objective of this research is to develop an unsaturated polyester (UPE) varnish with low-volatile organic compounds (VOCs). Instead of using a solvent, the solvent-free varnish incorporates a reactive diluent to reduce viscosity and a catalyst to accelerate curing. To achieve this, vinyl toluene and 1,4-butanediol dimethacrylate were employed as curing agents. Sebacic acid (SA) and fumaric acid were utilized to create UPE coatings for electrical insulation. Various tests and measurements were made to evaluate the physical, thermal, and chemical structure determination, and electrical properties of the synthesized resins. Given the increasing demand for eco-friendly and low-VOC products, gas chromatography was employed to determine VOC levels. The study demonstrated that the electrical volume resistance of cross-linked coatings containing FA was 1.58 × 1015 Ω·cm, whereas coatings containing SA exhibited a measurement of 6.96 × 1011 Ω·cm. VOC levels in the UPE coatings were found to be in the range of 2.10–3.60%.
1 Introduction
The coating insulation system, though regarded as a non-torque-generating and passive component, plays a significant role in places where electricity is utilized. The primary function of this coating is to serve as a fundamental thermal and electrical resistance element, greatly influencing the cooling and heating dynamics of the machinery (1). The approach employed to achieve this objective involves the creation of solvent-free resin systems, specifically focusing on unsaturated polyester (UPE) resins. In the context of polyester resins, the level of volatile organic compounds (VOC) can be primarily attributed to the presence of the toxic cross-linking monomer within the resin. This cross-linking monomer serves a dual role in the polymerization process: it functions as a solvent, reducing the viscosity of the base resin, while also actively participating in the curing process (2). Various polymeric structures, including polyesterimide (2), alkyd (3), epoxy (4), polyurethane (5), and UPE (6), have been synthesized as electrical insulation varnishes.
UPEs, known for their advantageous traits such as low VOC content, excellent dielectric insulation, thermal properties, and cost-effectiveness (7,8,9), are the preferred choice. UPE is a linear polymer formed through a polycondensation process, combining ester bonds and unsaturated double bonds, typically involving unsaturated dibasic acids with diols or saturated dibasic acids with unsaturated diols (10,11). Common starting materials include maleic anhydride (MA), phthalic anhydride, and propylene glycol for UPE production. Anhydrides are favored over diacids due to their faster esterification rates and lower water content requirements during polycondensation. Glycols are also added to enhance UPE chain flexibility. There exists a wide range of formulations for UPE, utilizing various combinations to optimize the properties of the cured resin (12,13). The curing process for UPE relies on radical polymerization, employing different techniques to initiate free radical formation, such as heat, light, electron beams, and ultrasonic waves. However, practical applications typically involve the use of free radical initiators (10).
UPEs are cured by initiating cross-linking among polymer chains with vinyl monomers like styrene, diallyl phthalate, methyl methacrylate, vinyl toluene (VT), divinyl benzene, or their combinations (14,15). While UPE resins combined with styrene are extensively used in coating technology, concerns over volatility and safety limit their use. In coating formulations, polyester resin is typically dissolved in the vinyl monomer, followed by cross-linking and curing facilitated by catalyst-induced stirring (16,17). To address these concerns, 1,4-butanediol dimethacrylate (BDDMA) emerges as a promising alternative. This is due to its compatibility with polyesters, low viscosity, and minimal volatility during polymerization (1–3%) (18). Jiang et al. conducted research on hydroxyethyl acrylate-blocking UPE solvent-less impregnating varnish, which demonstrated an electrical volume resistivity of 1.4 × 1014 Ω·cm, with a volatile content of 4.6%. In the study, it was emphasized that the impregnation varnish has excellent electrical insulation properties with low VOC (19). In a separate study by Sharma et al., the focus was on investigating the influence of nano/micro silica on the electrical properties of UPE composites. When 3% nano silica was added, the electrical volume resistivity was notably enhanced to 25 × 1015 Ω·cm. They found that the electrical characteristics were enhanced by the inclusion of nano-silica as compared with micro-silica (20).
In this research, UPE coatings are produced, which are tailored for electrical insulation using two distinct monomers: Sebacic acid (SA) and fumaric acid (FA). These UPE coatings underwent curing processes employing two diverse reactive monomers: VT and BDDMA. The primary goal of this study was to determine the combination of reactive monomer and acid monomer that would result in superior electrical and thermal insulation properties. As a result, the findings from this research will provide valuable insights for future endeavors in insulating varnish development by the authors. This investigation explored the influence of various monomer characteristics on UPE attributes, and to the best of the author’s knowledge, no similar study has been documented in the existing literature. To assess resin qualities, multiple physical tests and electrical measurements were conducted. Chemical structural analysis of the resin was carried out using Fourier transform infrared (FTIR) spectrophotometry. Thermal stability of the cured samples was evaluated using a Thermogravimetric Analyzer (TGA). Furthermore, in line with the demand for environmentally friendly products with very low VOC emissions, VOC tests were conducted using gas chromatography (GC).
2 Materials and methods
2.1 Materials
FA (commercial-grade), SA (commercial-grade), tetrahydrophthalic anhydride (THPA) (analytical-reagent-grade), MA (commercial-grade), neopentyl glycol (NPG) (commercial-grade), butylstannoic acid (catalyst) (analytical-reagent-grade), toluene (commercial-grade), hydroquinone (analytical-reagent-grade), VT (analytical-reagent-grade), and BDDMA (commercial-grade) were used.
2.2 Preparation of UPE varnish
Figure 1 shows that the experimental reaction system used for synthesizing UPE consists of a temperature-controlled glass polymerization reactor with a reflux condenser, mechanical stirrer, combined heating and cooling unit, nitrogen gas inlet, and outlet. Dean–Stark apparatus separates the water from the reaction by distillation.

The experimental system of the reaction.
In the formulation of the UPE resin, a ratio of 1.3:1 was deliberately chosen for the unsaturated component to the saturated component, representing the ratio of total −OH groups to total −COOH groups. The precise distribution of these compounds is outlined comprehensively in Table 1. The process commences with the initial combination of the required quantities of glycol and acid. Esterification is meticulously conducted under controlled conditions, utilizing a suitable catalyst, while maintaining the reaction temperature within the range of 160–170°C. To eliminate the water produced as a by-product during the reaction, azeotropic distillation is employed. Toluene serves as the distillation solvent for the purpose of removing water from the reaction mixture. The progression of the reaction is closely monitored by tracking the acid number in accordance with the ASTM D1639 standard test method. When the acid value of the reaction attains the range of 40–50 mg KOH g−1 sample, the reactor is promptly cooled to a temperature ranging from 100°C to 120°C. Subsequently, anhydrides are introduced, and the reaction temperature is diligently maintained between 170°C and 190°C, with continuous removal of the side product, water. Upon reaching an acid value within the range of 10–20 mg KOH g−1 sample, the reaction is halted, and hydroquinone is employed to quench it, followed by cooling to room temperature. Figure 2 provides an illustration of a conceivable esterification reaction mechanism.
Formulation of UPE resin
| UPE-FA-VT | UPE-FA-BDDMA | UPE-SA-VT | UPE-SA-BDDMA | |
|---|---|---|---|---|
| FA | 14.84 (0.5) | 15.10 (0.5) | — | — |
| SA | – | – | 27.14 (0.5) | 27.24 (0.5) |
| NPG | 34.70 (1.3) | 35.14 (1.3) | 36.20 (1.3) | 36.50 (1.3) |
| THPA | 9.75 (0.25) | 9.90 (0.25) | 10.20 (0.25) | 10.30 (0.25) |
| MA | 6.30 (0.25) | 6.40 (0.25) | 6.55 (0.25) | 6.60 (0.25) |
| VT | 30.30 (1.00) | — | 15.80 (0.50) | — |
| BDDMA | — | 29.35 (0.50) | — | 15.25 (0.25) |
| Catalyst | 0.10 | 0.10 | 0.10 | 0.10 |
| Hydroquinone | 0.01 | 0.01 | 0.01 | 0.01 |
| Toluene | 4.00 | 4.00 | 4.00 | 4.00 |
| Total (%w) | 100.00 | 100.00 | 100.00 | 100.00 |
Note: The molar ratio of the compounds is shown in parentheses.
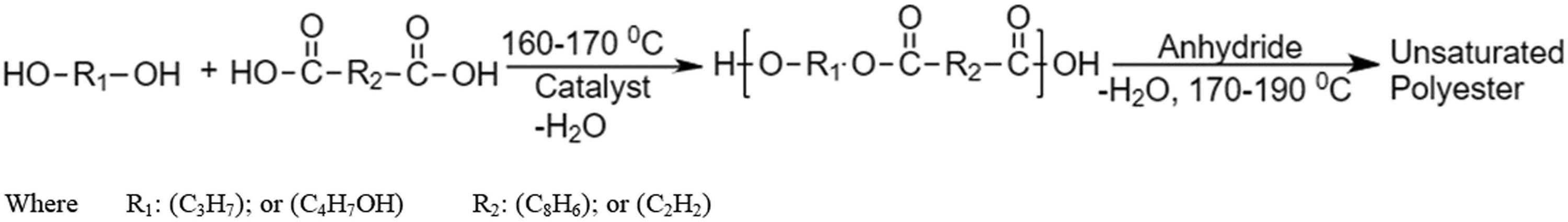
Esterification reaction mechanism.
2.3 Sample preparation
During the curing process, a curing catalyst, comprising 1–3% of the total weight, is introduced into the synthesized resin. Subsequently, the mixture undergoes agitation at 1,000 rpm for a duration of 1 h. In their uncured state, all liquid samples are then subjected to a comprehensive set of tests, including assessments of viscosity, density, VOC content, FTIR analysis, and flash point measurements. To thermal and electrical characterization, the samples are cast into glass Petri dishes and subsequently subjected to curing at a temperature of 200°C for a period ranging from 3 to 5 h. Notably, the curing duration for the VT-included samples is 5 h, whereas the BDDMA samples require 3 h to achieve complete curing. UPE samples synthesized in the study are coded as follows: UPE-SA-VT/UPE-SA-BDDMA and UPE-FA-VT/UPE-FA-BDDMA. The material at the end of the coding indicates the reactive monomer type.
2.4 Characterization
The characterization of the synthesized UPE samples involved several testing procedures.
Viscosity measurements were conducted using a Brookfield viscometer (ASTM D2196) in the uncured liquid state of UPE samples. Various speeds and spindles were employed for this purpose.
The density of the synthesized samples was determined by utilizing a pycnometer at a temperature of 25°C, following the guidelines outlined in the ASTM D1475 standard.
VOC content was assessed through GC in accordance with the ISO 11890-2 standard. Measurements were conducted using a SHIMADZU GC-2030 model GC device with an Rtx-624 column (30 m, 0.25 mm ID, Crossbond 6% cyanopropylphenyl/94% dimethyl polysiloxane) and a temperature range of 50–220°C.
To determine the percentage solid content, a 1.5–1.6 g sample was placed in an aluminum pan and subjected to an oven at 135°C for a duration of 3 h, following the ASTM D115 procedure.
Flash point measurements were carried out using the Abel closed cup method, adhering to the ISO 13736 standard.
To assess the chemical structure of the cured samples, FTIR spectrometry with an Attenuated total reflectance apparatus was utilized. This analysis was conducted using a Shimadzu Corp. Iraffinity-1S FTIR Spectrometer. The analysis spanned the range of 4,000–600 cm⁻¹.
Thermal degradation characteristics of the cured samples were investigated via thermogravimetric analysis (TGA), utilizing a Perkin Elmer (TGA 4000). TGA measurements were conducted in a nitrogen environment, spanning temperatures from 30°C to 900°C, with increments of 10°C per minute.
Electrical volumetric and surface resistivity measurements were performed using an electrometer (ASTM D257). The average thickness of each sample was taken into account for volumetric resistivity measurements. During the volumetric resistivity measurement, the conditions were as follows: temperature of 25°C, applied voltage of 500 V, and measurement duration of 10 s.
3 Results and discussion
An overview of the key properties of the utilized monomers, encompassing molecular weight, density, flash point, and molecular structures is given in Table 2. Notably, VT exhibits a cyclic structure with a molecular weight of 118 g·mol−1, while BDDMA presents a long-chain structure with a higher molecular weight of 226 g·mol−1. Furthermore, it is worth highlighting that BDDMA possesses a significantly elevated flash point. The influence of these distinctive characteristics was thoroughly investigated through a series of characterization analyses.
Properties of monomers
| Monomer | Molecular weight (g·mol−1) (22) | Density (g·cm−3) (22) | Flash point (°C) (22) | Molecular structure (23) |
|---|---|---|---|---|
| FA | 116.07 | 1.64 | 230 |

|
| SA | 202.25 | 1.21 | 220 |

|
| BDDMA | 226.30 | 1.02 | 139 |

|
| VT | 118.18 | 0.89 | 52 |

|
Upon examining the results of the physical analyses presented in Table 3, it is apparent that the acid number and density values for UPE varnishes utilizing VT and BDDMA are closely aligned. However, in the realm of viscosity measurements, samples including BDDMA exhibited values approximately six times higher than those samples with VT (21). When comparing the viscosities of UPE-SA-VT and UPE-FA-VT, it is evident that the linear structure of the SA monomer (Table 2) has a substantial impact, resulting in viscosities almost twice as high as those of the FA-included samples. This disparity can be attributed to differences in molecular structure. The notably high flash point of 139°C associated with the BDDMA reactive monomer had a significant effect on the UPE-BDDMA varnish, resulting in a higher flash point when compared to the UPE-VT varnish (22). In consideration of the total solid content detailed in Table 1, it becomes evident that this aligns closely with the solid content observed when VT is employed in UPE synthesis. Conversely, there was an unexpected increase in solid content in UPE samples incorporating BDDMA. This distinction can be attributed to the fact that BDDMA is di-functional, whereas VT is mono-functional. This discrepancy is further substantiated by the differences in curing times, as explained in Section 2.3. Moreover, the %VOC content in the UPE-BDDMA samples consistently measured at 2.30%, falling within the specified range of 1–3% as per the literature (18). Importantly, this value was lower than that observed in the UPE-VT samples.
Results of physical properties
| Analysis | UPE-FA-VT | UPE-FA-BDDMA | UPE-SA-VT | UPE-SA-BDDMA |
|---|---|---|---|---|
| Acid number (mg KOH g−1) | 14.40 | 17.00 | 16.00 | 17.50 |
| Density (g·cm−3) | 1.0435 | 1.1030 | 1.0328 | 1.0623 |
| Viscosity (cP) | 290.0 | 1770.0 | 520.0 | 1860.0 |
| Solids content (%) | 67.05 | 92.35 | 76.11 | 92.30 |
| VOC (%) | 4.30 | 2.30 | 3.60 | 2.30 |
| Flash point (°C) | 47.20 | 64.30 | 47.10 | 64.50 |
FTIR analysis was utilized to analyze the structural characteristics of the samples. In the FTIR spectrums of UPE-FA-BDDMA, UPE-FA-VT, UPE-SA-BDDMA, and UPE-SA-VT samples, all had C═O stretching vibration of ester group at 1,714, 1,714, 1,716, and 1,730 cm−1, respectively. UPE-FA-BDDMA and UPE-FA-VT spectrums both had C-H stretching vibration at 771 cm−1. UPE-FA-VT and UPE-SA-VT spectrums both had C═O stretching vibration of aromatic ester groups at 1,298 and 1,255 cm−1. UPE-FA-BDDMA and UPE-SA-BDDMA spectrums both had C═C stretching vibration at 1,635 cm−1 (23). FTIR spectra of all samples are shown in Figure 3.

FTIR spectrums of samples.
In the existing literature, research on the thermal degradation of UPEs, resulting from the reaction of straight chain and branched chain glycols with MA and phthalic anhydride, isophthalic acid, or terephthalic acid, has been conducted using TGA (24,25). These studies have explored the impact of additional fillers, composition, and curing conditions on the thermal degradation of UPE, revealing that resins typically initiate degradation around 200°C and experience significant weight loss around 400°C. Notably, fillers and curing methods were found to have a limited effect on the stability of the resins. Furthermore, previous research (26) has established that the structure of the initiator can influence the thermal properties of UPEs (25,26). By using TGA, the decomposition initial temperature (T d5), the maximum decomposition temperature (T dmax, 50%), and residue amounts at 600°C were measured and the results of TGA are summarized in Table 4 in this study. UPEs with decomposition temperatures in the range of 234–276°C were synthesized in accordance with the literature. When the reactive monomer was changed in UPE resin samples containing SA, there was not much change in the initial and maximum decomposition temperature, only a 6°C change was observed. In samples containing FA, the change in reactive monomer significantly affected the decomposition initial temperature. The UPE-FA-VT sample began to decompose at 234°C, while the UPE-SA-VT sample began to decompose at 276°C. The UPE-SA-BDDMA sample showed a high value with a maximum decomposition temperature of about 436°C. UPE resin containing SA show main degradation at higher temperatures than UPE containing FA. It was found that the most effective parameters affecting the decomposition temperature are monomer type and chemical structure. UPE-FA-BDDMA varnish showed the highest char residue of 11% at 600°C. In addition, as seen in the TGA graph in Figure 4, all the varnishes decomposed in a single step.
Thermal analysis results of samples
| Analysis | T d5 (°C) | T dmax,%50 (°C) | Residue amount (600°C, wt%) |
|---|---|---|---|
| UPE-FA-VT | 234 | 388 | 10.0 |
| UPE-FA-BDDMA | 264 | 414 | 11.0 |
| UPE-SA-VT | 276 | 430 | 5.0 |
| UPE-SA-BDDMA | 270 | 436 | 4.0 |
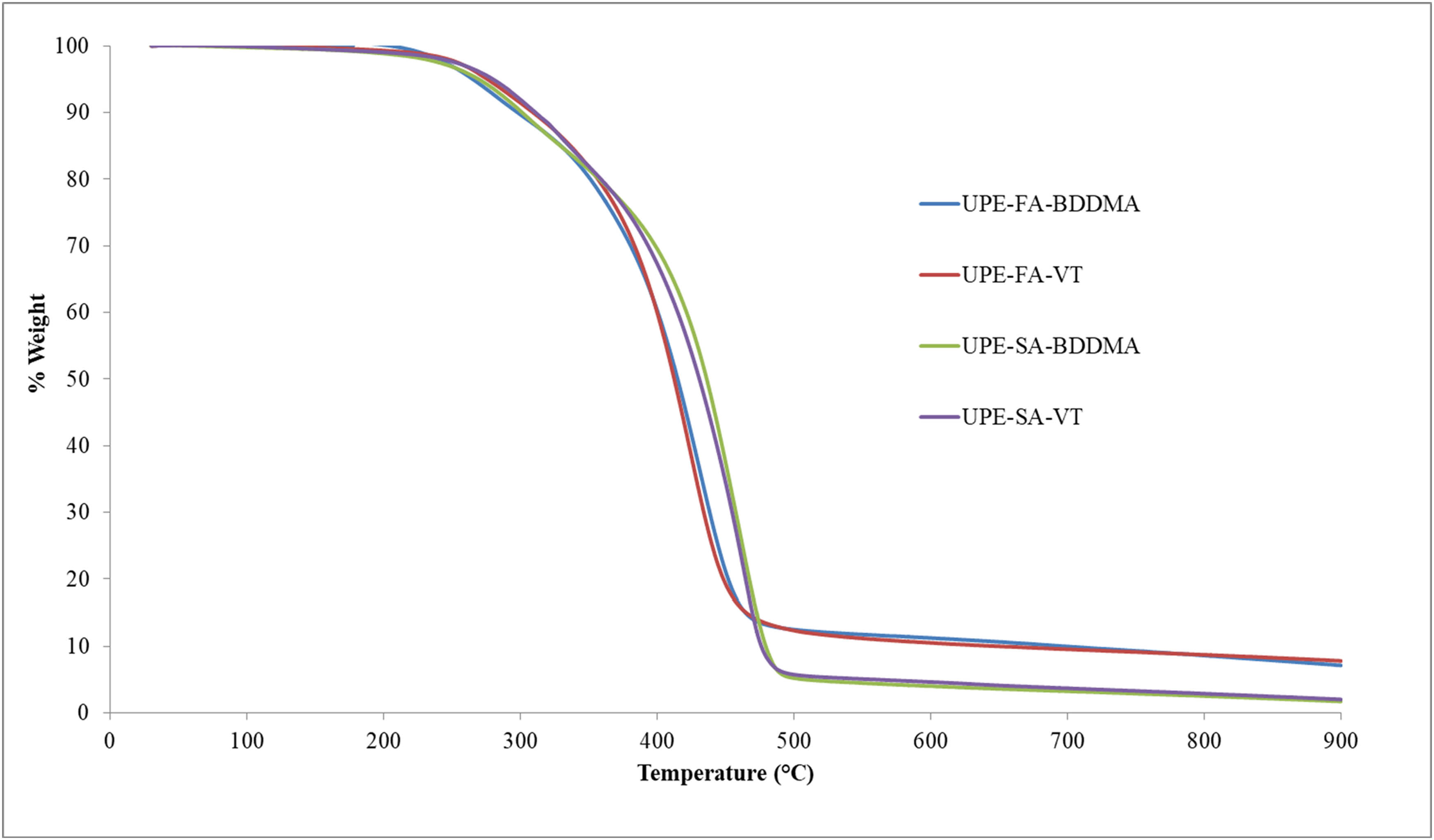
TGA graph of samples.
Analyzing the results presented in Table 5, it is evident that there are significant variations in electrical properties, particularly in electrical volumetric and surface resistivity measurements: For UPE-FA-VT, the electrical volumetric resistivity was measured to be 1.58 × 1015 Ω·cm, while for UPE-SA-VT it was 6.96 × 1011 Ω·cm. In terms of surface resistivity, UPE-FA-VT displayed a value of 1.96 × 1013 Ω, whereas UPE-SA-VT exhibited 7.47 × 1010 Ω. Unfortunately, it was not possible to obtain measurements for UPE-SA-BDDMA, as the varnish did not provide a suitable measurement surface after curing. Based on these findings, it can be concluded that varnishes incorporating the FA monomer tend to exhibit higher electrical resistance compared to those containing the SA monomer. However, it is notable that the choice of the reactive monomer does not seem to have a significant impact on these electrical properties. This disparity can be attributed to the presence of an unsaturated bond in the FA monomer’s structure, which facilitates cross-linking. In contrast, the linear structure of the SA monomer does not allow for such cross-linking. In the case of the UPE-SA-VT sample, cross-linking is facilitated by other compounds, such as MA and THPA, present in the structure. As a result, the use of the FA monomer provides an advantage over the SA monomer in terms of dielectric properties within UPE coatings.
Electrical properties of samples
| Analysis | UPE-FA-VT | UPE-FA-BDDMA | UPE-SA-VT | UPE-SA-BDDMA |
|---|---|---|---|---|
| Electrical volumetric resistivity (Ω·cm) | 1.58 × 1015 | 1.45 × 1015 | 6.96 × 1011 | — |
| Electrical surface resistance 500 V(Ω) | 1.96 × 1013 | 2.79 × 1013 | 7.47 × 1010 | — |
4 Conclusion
The study involved the synthesis of UPE resins and a comprehensive evaluation of their physical, thermal, electrical, and structural characteristics through various analytical methods. Additionally, the investigation delved into the impact of different reactive monomers, specifically acid monomers, on the properties of the synthesized varnishes. The incorporation of the BDDMA reactive monomer resulted in notable improvements in viscosity and flash point of the varnishes when compared to VT. Furthermore, the UPE varnishes containing the SA acid monomer exhibited higher initial degradation and maximum degradation values in comparison to the FA sample. In the realm of electrical analysis, varnishes containing the FA acid monomer showcased superior dielectric (insulating) properties when contrasted with varnishes containing the SA acid monomer. This research underscores the significant influence that specific reactive monomers can have on the properties of UPE resins and the resultant varnishes, thereby enhancing various aspects such as thermal stability, viscosity, and electrical insulating properties. As a result, our approach holds the potential to pave the way for the creation of innovative UPE resins with customizable properties, suitable for a wide spectrum of applications across diverse fields.
Anknowledgments
We would like to express our sincere gratitude to Betek Boya Ve Kimya Sanayi A.Ş for their support.
-
Funding information: The project is university-industry collaboration and was supported by TUBITAK within the scope of the research project numbered 1505-5210107.
-
Author contributions: (https://casrai.org/credit/). Selinay Gümüş: conceptualization, methodology, resources, formal analysis, data curation, writing – original draft, and visualization. Kaan Aksoy: conceptualization, writing – review and editing, and supervision. Ayşe Aytaç: conceptualization, methodology, formal analysis, data curation, writing – original draft, writing– review and editing, and visualization.
-
Conflict of interest: No potential conflict of interest was reported by the author(s).
References
(1) Hemmati R, Wu F, El-Refaie A. In survey of insulation systems in electrical machines, 2019. IEEE International Electric Machines & Drives Conference (IEMDC). San Diego, CA, USA: IEEE; 2019. pp. 2069–76. 10.1109/IEMDC.2019.8785099.Suche in Google Scholar
(2) Calabrese E, Raimondo M, Catauro M, Vertuccio L, Lamberti P, et al. Thermal and electrical characterization of polyester resins suitable for electric motor insulation. Polymers. 2023;15(6):1374. 10.3390/polym15061374.Suche in Google Scholar PubMed PubMed Central
(3) Patel V, Varughese J, Krishnamoorthy P, Jain R, Singh A, Ramamoorty M. Synthesis of alkyd resin from jatropha and rapeseed oils and their applications in electrical insulation. J Appl Polym Sci. 2008;107(3):1724–9. 10.1002/app.27195.Suche in Google Scholar
(4) Cao M, Wang H, Cai R, Ge Q, Jiang S, Zhai L, et al. Preparation and properties of epoxy‐modified tung oil waterborne insulation varnish. J Appl Polym Sci. 2015;132(45):1–8. 10.1002/app.42755.Suche in Google Scholar
(5) Yeganeh H, Razavi‐Nouri M, Ghaffari M. Synthesis and properties of polybenzoxazine modified polyurethanes as a new type of electrical insulators with improved thermal stability. Polym Eng & Sci. 2008;48(7):1329–38. 10.1002/pen.21098.Suche in Google Scholar
(6) Wang Y-Q, Guo Y, Cui R-X, Wang Z-M, Wu Y-L. Preparation and mechanical properties of nano-silica/UPR polymer composite. Sci Eng Composite Mater. 2014;21(4):471–7. 10.1515/secm-2013-0051.Suche in Google Scholar
(7) Shukla B, Dixit V, Singhal R, Nagpal A, Singh PK. Synthesis and characterization of solventless polyester varnish based on di cyclo penta diene (DCPD). Polym Technol Eng. 2007;46(3):191–8. 10.1080/03602550601152697.Suche in Google Scholar
(8) Yao H, Pan H, Li RR, Li X, Zhang Z. Unsaturated polyester resin/epoxy‐functionalised nanosilica composites constructed by in situ polymerisation. Micro Nano Lett. 2015;10(9):427–31. 10.1049/mnl.2015.0085.Suche in Google Scholar
(9) Gumus S, Aksoy K, Aytac A. Modification to unsaturated polyester resin with silica and silica/boron nitride mixture nanoparticles. Pigment & Resin Technol. 2023. 10.1108/PRT-11-2022-0140.Suche in Google Scholar
(10) Gao Y, Zhang H, Huang M, Lai F. Unsaturated polyester resin concrete: A review. Constr Build Mater. 2019;228:116709. 10.1016/j.conbuildmat.2019.116709.Suche in Google Scholar
(11) Long TE, Scheirs J. Modern polyesters: chemistry and technology of polyesters and copolyesters. The Atrium, Southern Gate, Chichester, West Sussex, England: John Wiley & Sons; 2005. 10.1002/0470090685.Suche in Google Scholar
(12) Jones FR. Unsaturated polyester resins. In: Brydson’s Plastics Materials. Oxford, United Kingdom: Elsevier; 2017. p. 743–72. 10.1016/B978-0-323-35824-8.00026-8.Suche in Google Scholar
(13) Licari JJ. Coating materials for electronic applications: polymers, processing, reliability, testing. Norwich, New York, U.S.A.: William Andrew Publishing; 2003.Suche in Google Scholar
(14) Fink JK. Reactive polymers: fundamentals and applications: a concise guide to industrial polymers. Leoben, Austria: William Andrew; 2017.Suche in Google Scholar
(15) Kricheldorf H. Ring-opening polycondensation. In: Polycondensation: History and new results. Berlin, Heidelberg: Springer; 2013. p. 135–45. 10.1007/978-3-642-39429-4.Suche in Google Scholar
(16) Thomas S, Hosur M, Chirayil CJ. Unsaturated polyester resins: Fundamentals, design, fabrication, and applications. London, UK: Elsevier; 2019.Suche in Google Scholar
(17) Mannari V, Patel CJ. Understanding coatings raw materials. Vincentz Network: 2019.10.1515/9783748600374Suche in Google Scholar
(18) Fetouhi L, Petitgas B, Dantras E, Martinez-Vega J. Mechanical, dielectric, and physicochemical properties of impregnating resin based on unsaturated polyesterimides. Eur Phys J Appl Phys. 2017;80(1):10901. 10.1051/epjap/2017160451.Suche in Google Scholar
(19) Jiang QB, Deng QS, Gao J, Wang YQ. Study on the HEA-blocking unsaturated polyester solventless impregnating varnish. Adv Mater Res. 2012;380:133–6. 10.4028/www.scientific.net/AMR.380.133.Suche in Google Scholar
(20) Sharma RA, Bhattacharya S, D'Melo D, Swain S, Chaudhari L. Effect of nano/micro silica on electrical property of unsaturated polyester resin composites. Trans Electr ElectrMater. 2012;13(1):31–4. 10.4313/TEEM.2012.13.1.31.Suche in Google Scholar
(21) Cousinet S, Ghadban A, Fleury E, Lortie F, Pascault JP, Portinha D. Toward replacement of styrene by bio-based methacrylates in unsaturated polyester resins. Eur Polym J. 2015;67:539–50. 10.1016/j.eurpolymj.2015.02.016.Suche in Google Scholar
(22) Kim S, Chen J, Cheng T, Gindulyte A, He J, He S, et al. PubChem 2023 update. Nucleic Acids Res. 2023;51(D1):D1373–80. 10.1093%2Fnar%2Fgkac956.Suche in Google Scholar
(23) Infrared Spectroscopy Absorption Table. 2020 [cited 2023 Sep 7]. https://chem.libretexts.org/@go/page/22645.Suche in Google Scholar
(24) Piotr P, Piotr C, Jan P. Unsaturated polyester resins: Chemistry and technology. Adv Polym Sci. 2005;184:1–95. 10.1007/b136243.Suche in Google Scholar
(25) Evans SJ, Haines PJ, Skinner GA. The thermal degradation of polyester resins II. The effects of cure and of fillers on degradation. Thermochim acta. 1997;291(1–2):43–9. 10.1016/S0040-6031(96)03102-4.Suche in Google Scholar
(26) Bansal RK, Mittal J, Singh P. Thermal stability and degradation studies of polyester resins. J Appl Polym Sci. 1989;37(7):1901–8. 10.1002/app.1989.070370713.Suche in Google Scholar
© 2023 the author(s), published by De Gruyter
This work is licensed under the Creative Commons Attribution 4.0 International License.
Artikel in diesem Heft
- Research Articles
- Chitosan nanocomposite film incorporating Nigella sativa oil, Azadirachta indica leaves’ extract, and silver nanoparticles
- Effect of Zr-doped CaCu3Ti3.95Zr0.05O12 ceramic on the microstructure, dielectric properties, and electric field distribution of the LDPE composites
- Effects of dry heating, acetylation, and acid pre-treatments on modification of potato starch with octenyl succinic anhydride (OSA)
- Loading conditions impact on the compression fatigue behavior of filled styrene butadiene rubber
- Characterization and compatibility of bio-based PA56/PET
- Study on the aging of three typical rubber materials under high- and low-temperature cyclic environment
- Numerical simulation and experimental research of electrospun polyacrylonitrile Taylor cone based on multiphysics coupling
- Experimental investigation of properties and aging behavior of pineapple and sisal leaf hybrid fiber-reinforced polymer composites
- Influence of temperature distribution on the foaming quality of foamed polypropylene composites
- Enzyme-catalyzed synthesis of 4-methylcatechol oligomer and preliminary evaluations as stabilizing agent in polypropylene
- Molecular dynamics simulation of the effect of the thermal and mechanical properties of addition liquid silicone rubber modified by carbon nanotubes with different radii
- Incorporation of poly(3-acrylamidopropyl trimethylammonium chloride-co-acrylic acid) branches for good sizing properties and easy desizing from sized cotton warps
- Effect of matrix composition on properties of polyamide 66/polyamide 6I-6T composites with high content of continuous glass fiber for optimizing surface performance
- Preparation and properties of epoxy-modified thermosetting phenolic fiber
- Thermal decomposition reaction kinetics and storage life prediction of polyacrylate pressure-sensitive adhesive
- Effect of different proportions of CNTs/Fe3O4 hybrid filler on the morphological, electrical and electromagnetic interference shielding properties of poly(lactic acid) nanocomposites
- Doping silver nanoparticles into reverse osmosis membranes for antibacterial properties
- Melt-blended PLA/curcumin-cross-linked polyurethane film for enhanced UV-shielding ability
- The affinity of bentonite and WO3 nanoparticles toward epoxy resin polymer for radiation shielding
- Prolonged action fertilizer encapsulated by CMC/humic acid
- Preparation and experimental estimation of radiation shielding properties of novel epoxy reinforced with Sb2O3 and PbO
- Fabrication of polylactic acid nanofibrous yarns for piezoelectric fabrics
- Copper phenyl phosphonate for epoxy resin and cyanate ester copolymer with improved flame retardancy and thermal properties
- Synergistic effect of thermal oxygen and UV aging on natural rubber
- Effect of zinc oxide suspension on the overall filler content of the PLA/ZnO composites and cPLA/ZnO composites
- The role of natural hybrid nanobentonite/nanocellulose in enhancing the water resistance properties of the biodegradable thermoplastic starch
- Performance optimization of geopolymer mortar blending in nano-SiO2 and PVA fiber based on set pair analysis
- Preparation of (La + Nb)-co-doped TiO2 and its polyvinylidene difluoride composites with high dielectric constants
- Effect of matrix composition on the performance of calcium carbonate filled poly(lactic acid)/poly(butylene adipate-co-terephthalate) composites
- Low-temperature self-healing polyurethane adhesives via dual synergetic crosslinking strategy
- Leucaena leucocephala oil-based poly malate-amide nanocomposite coating material for anticorrosive applications
- Preparation and properties of modified ammonium polyphosphate synergistic with tris(2-hydroxyethyl) isocynurate for flame-retardant LDPE
- Thermal response of double network hydrogels with varied composition
- The effect of coated calcium carbonate using stearic acid on the recovered carbon black masterbatch in low-density polyethylene composites
- Investigation of MXene-modified agar/polyurethane hydrogel elastomeric repair materials with tunable water absorption
- Damping performance analysis of carbon black/lead magnesium niobite/epoxy resin composites
- Molecular dynamics simulations of dihydroxylammonium 5,5′-bistetrazole-1,1′-diolate (TKX-50) and TKX-50-based PBXs with four energetic binders
- Preparation and characterization of sisal fibre reinforced sodium alginate gum composites for non-structural engineering applications
- Study on by-products synthesis of powder coating polyester resin catalyzed by organotin
- Ab initio molecular dynamics of insulating paper: Mechanism of insulating paper cellobiose cracking at transient high temperature
- Effect of different tin neodecanoate and calcium–zinc heat stabilizers on the thermal stability of PVC
- High-strength polyvinyl alcohol-based hydrogel by vermiculite and lignocellulosic nanofibrils for electronic sensing
- Impacts of micro-size PbO on the gamma-ray shielding performance of polyepoxide resin
- Influence of the molecular structure of phenylamine antioxidants on anti-migration and anti-aging behavior of high-performance nitrile rubber composites
- Fiber-reinforced polyvinyl alcohol hydrogel via in situ fiber formation
- Preparation and performance of homogenous braids-reinforced poly (p-phenylene terephthamide) hollow fiber membranes
- Synthesis of cadmium(ii) ion-imprinted composite membrane with a pyridine functional monomer and characterization of its adsorption performance
- Impact of WO3 and BaO nanoparticles on the radiation shielding characteristics of polydimethylsiloxane composites
- Comprehensive study of the radiation shielding feature of polyester polymers impregnated with iron filings
- Preparation and characterization of polymeric cross-linked hydrogel patch for topical delivery of gentamicin
- Mechanical properties of rCB-pigment masterbatch in rLDPE: The effect of processing aids and water absorption test
- Pineapple fruit residue-based nanofibre composites: Preparation and characterizations
- Effect of natural Indocalamus leaf addition on the mechanical properties of epoxy and epoxy-carbon fiber composites
- Utilization of biosilica for energy-saving tire compounds: Enhancing performance and efficiency
- Effect of capillary arrays on the profile of multi-layer micro-capillary films
- A numerical study on thermal bonding with preheating technique for polypropylene microfluidic device
- Development of modified h-BN/UPE resin for insulation varnish applications
- High strength, anti-static, thermal conductive glass fiber/epoxy composites for medical devices: A strategy of modifying fibers with functionalized carbon nanotubes
- Effects of mechanical recycling on the properties of glass fiber–reinforced polyamide 66 composites in automotive components
- Bentonite/hydroxyethylcellulose as eco-dielectrics with potential utilization in energy storage
- Study on wall-slipping mechanism of nano-injection polymer under the constant temperature fields
- Synthesis of low-VOC unsaturated polyester coatings for electrical insulation
- Enhanced apoptotic activity of Pluronic F127 polymer-encapsulated chlorogenic acid nanoparticles through the PI3K/Akt/mTOR signaling pathway in liver cancer cells and in vivo toxicity studies in zebrafish
- Preparation and performance of silicone-modified 3D printing photosensitive materials
- A novel fabrication method of slippery lubricant-infused porous surface by thiol-ene click chemistry reaction for anti-fouling and anti-corrosion applications
- Development of polymeric IPN hydrogels by free radical polymerization technique for extended release of letrozole: Characterization and toxicity evaluation
- Tribological characterization of sponge gourd outer skin fiber-reinforced epoxy composite with Tamarindus indica seed filler addition using the Box–Behnken method
- Stereocomplex PLLA–PBAT copolymer and its composites with multi-walled carbon nanotubes for electrostatic dissipative application
- Enhancing the therapeutic efficacy of Krestin–chitosan nanocomplex for cancer medication via activation of the mitochondrial intrinsic pathway
- Variation in tungsten(vi) oxide particle size for enhancing the radiation shielding ability of silicone rubber composites
- Damage accumulation and failure mechanism of glass/epoxy composite laminates subjected to repeated low velocity impacts
- Gamma-ray shielding analysis using the experimental measurements for copper(ii) sulfate-doped polyepoxide resins
- Numerical simulation into influence of airflow channel quantities on melt-blowing airflow field in processing of polymer fiber
- Cellulose acetate oleate-reinforced poly(butylene adipate-co-terephthalate) composite materials
- Radiation shielding capability and exposure buildup factor of cerium(iv) oxide-reinforced polyester resins
- Recyclable polytriazole resins with high performance based on Diels-Alder dynamic covalent crosslinking
- Adsorption and recovery of Cr(vi) from wastewater by Chitosan–Urushiol composite nanofiber membrane
- Comprehensive performance evaluation based on electromagnetic shielding properties of the weft-knitted fabrics made by stainless steel/cotton blended yarn
- Review Articles
- Preparation and application of natural protein polymer-based Pickering emulsions
- Wood-derived high-performance cellulose structural materials
- Flammability properties of polymers and polymer composites combined with ionic liquids
- Polymer-based nanocarriers for biomedical and environmental applications
- A review on semi-crystalline polymer bead foams from stirring autoclave: Processing and properties
- Rapid Communication
- Preparation and characterization of magnetic microgels with linear thermosensitivity over a wide temperature range
- Special Issue: Biodegradable and bio-based polymers: Green approaches (Guest Editors: Kumaran Subramanian, A. Wilson Santhosh Kumar, and Venkatajothi Ramarao)
- Synthesis and characterization of proton-conducting membranes based on bacterial cellulose and human nail keratin
- Fatigue behaviour of Kevlar/carbon/basalt fibre-reinforced SiC nanofiller particulate hybrid epoxy composite
- Effect of citric acid on thermal, phase morphological, and mechanical properties of poly(l-lactide)-b-poly(ethylene glycol)-b-poly(l-lactide)/thermoplastic starch blends
- Dose-dependent cytotoxicity against lung cancer cells via green synthesized ZnFe2O4/cellulose nanocomposites
Artikel in diesem Heft
- Research Articles
- Chitosan nanocomposite film incorporating Nigella sativa oil, Azadirachta indica leaves’ extract, and silver nanoparticles
- Effect of Zr-doped CaCu3Ti3.95Zr0.05O12 ceramic on the microstructure, dielectric properties, and electric field distribution of the LDPE composites
- Effects of dry heating, acetylation, and acid pre-treatments on modification of potato starch with octenyl succinic anhydride (OSA)
- Loading conditions impact on the compression fatigue behavior of filled styrene butadiene rubber
- Characterization and compatibility of bio-based PA56/PET
- Study on the aging of three typical rubber materials under high- and low-temperature cyclic environment
- Numerical simulation and experimental research of electrospun polyacrylonitrile Taylor cone based on multiphysics coupling
- Experimental investigation of properties and aging behavior of pineapple and sisal leaf hybrid fiber-reinforced polymer composites
- Influence of temperature distribution on the foaming quality of foamed polypropylene composites
- Enzyme-catalyzed synthesis of 4-methylcatechol oligomer and preliminary evaluations as stabilizing agent in polypropylene
- Molecular dynamics simulation of the effect of the thermal and mechanical properties of addition liquid silicone rubber modified by carbon nanotubes with different radii
- Incorporation of poly(3-acrylamidopropyl trimethylammonium chloride-co-acrylic acid) branches for good sizing properties and easy desizing from sized cotton warps
- Effect of matrix composition on properties of polyamide 66/polyamide 6I-6T composites with high content of continuous glass fiber for optimizing surface performance
- Preparation and properties of epoxy-modified thermosetting phenolic fiber
- Thermal decomposition reaction kinetics and storage life prediction of polyacrylate pressure-sensitive adhesive
- Effect of different proportions of CNTs/Fe3O4 hybrid filler on the morphological, electrical and electromagnetic interference shielding properties of poly(lactic acid) nanocomposites
- Doping silver nanoparticles into reverse osmosis membranes for antibacterial properties
- Melt-blended PLA/curcumin-cross-linked polyurethane film for enhanced UV-shielding ability
- The affinity of bentonite and WO3 nanoparticles toward epoxy resin polymer for radiation shielding
- Prolonged action fertilizer encapsulated by CMC/humic acid
- Preparation and experimental estimation of radiation shielding properties of novel epoxy reinforced with Sb2O3 and PbO
- Fabrication of polylactic acid nanofibrous yarns for piezoelectric fabrics
- Copper phenyl phosphonate for epoxy resin and cyanate ester copolymer with improved flame retardancy and thermal properties
- Synergistic effect of thermal oxygen and UV aging on natural rubber
- Effect of zinc oxide suspension on the overall filler content of the PLA/ZnO composites and cPLA/ZnO composites
- The role of natural hybrid nanobentonite/nanocellulose in enhancing the water resistance properties of the biodegradable thermoplastic starch
- Performance optimization of geopolymer mortar blending in nano-SiO2 and PVA fiber based on set pair analysis
- Preparation of (La + Nb)-co-doped TiO2 and its polyvinylidene difluoride composites with high dielectric constants
- Effect of matrix composition on the performance of calcium carbonate filled poly(lactic acid)/poly(butylene adipate-co-terephthalate) composites
- Low-temperature self-healing polyurethane adhesives via dual synergetic crosslinking strategy
- Leucaena leucocephala oil-based poly malate-amide nanocomposite coating material for anticorrosive applications
- Preparation and properties of modified ammonium polyphosphate synergistic with tris(2-hydroxyethyl) isocynurate for flame-retardant LDPE
- Thermal response of double network hydrogels with varied composition
- The effect of coated calcium carbonate using stearic acid on the recovered carbon black masterbatch in low-density polyethylene composites
- Investigation of MXene-modified agar/polyurethane hydrogel elastomeric repair materials with tunable water absorption
- Damping performance analysis of carbon black/lead magnesium niobite/epoxy resin composites
- Molecular dynamics simulations of dihydroxylammonium 5,5′-bistetrazole-1,1′-diolate (TKX-50) and TKX-50-based PBXs with four energetic binders
- Preparation and characterization of sisal fibre reinforced sodium alginate gum composites for non-structural engineering applications
- Study on by-products synthesis of powder coating polyester resin catalyzed by organotin
- Ab initio molecular dynamics of insulating paper: Mechanism of insulating paper cellobiose cracking at transient high temperature
- Effect of different tin neodecanoate and calcium–zinc heat stabilizers on the thermal stability of PVC
- High-strength polyvinyl alcohol-based hydrogel by vermiculite and lignocellulosic nanofibrils for electronic sensing
- Impacts of micro-size PbO on the gamma-ray shielding performance of polyepoxide resin
- Influence of the molecular structure of phenylamine antioxidants on anti-migration and anti-aging behavior of high-performance nitrile rubber composites
- Fiber-reinforced polyvinyl alcohol hydrogel via in situ fiber formation
- Preparation and performance of homogenous braids-reinforced poly (p-phenylene terephthamide) hollow fiber membranes
- Synthesis of cadmium(ii) ion-imprinted composite membrane with a pyridine functional monomer and characterization of its adsorption performance
- Impact of WO3 and BaO nanoparticles on the radiation shielding characteristics of polydimethylsiloxane composites
- Comprehensive study of the radiation shielding feature of polyester polymers impregnated with iron filings
- Preparation and characterization of polymeric cross-linked hydrogel patch for topical delivery of gentamicin
- Mechanical properties of rCB-pigment masterbatch in rLDPE: The effect of processing aids and water absorption test
- Pineapple fruit residue-based nanofibre composites: Preparation and characterizations
- Effect of natural Indocalamus leaf addition on the mechanical properties of epoxy and epoxy-carbon fiber composites
- Utilization of biosilica for energy-saving tire compounds: Enhancing performance and efficiency
- Effect of capillary arrays on the profile of multi-layer micro-capillary films
- A numerical study on thermal bonding with preheating technique for polypropylene microfluidic device
- Development of modified h-BN/UPE resin for insulation varnish applications
- High strength, anti-static, thermal conductive glass fiber/epoxy composites for medical devices: A strategy of modifying fibers with functionalized carbon nanotubes
- Effects of mechanical recycling on the properties of glass fiber–reinforced polyamide 66 composites in automotive components
- Bentonite/hydroxyethylcellulose as eco-dielectrics with potential utilization in energy storage
- Study on wall-slipping mechanism of nano-injection polymer under the constant temperature fields
- Synthesis of low-VOC unsaturated polyester coatings for electrical insulation
- Enhanced apoptotic activity of Pluronic F127 polymer-encapsulated chlorogenic acid nanoparticles through the PI3K/Akt/mTOR signaling pathway in liver cancer cells and in vivo toxicity studies in zebrafish
- Preparation and performance of silicone-modified 3D printing photosensitive materials
- A novel fabrication method of slippery lubricant-infused porous surface by thiol-ene click chemistry reaction for anti-fouling and anti-corrosion applications
- Development of polymeric IPN hydrogels by free radical polymerization technique for extended release of letrozole: Characterization and toxicity evaluation
- Tribological characterization of sponge gourd outer skin fiber-reinforced epoxy composite with Tamarindus indica seed filler addition using the Box–Behnken method
- Stereocomplex PLLA–PBAT copolymer and its composites with multi-walled carbon nanotubes for electrostatic dissipative application
- Enhancing the therapeutic efficacy of Krestin–chitosan nanocomplex for cancer medication via activation of the mitochondrial intrinsic pathway
- Variation in tungsten(vi) oxide particle size for enhancing the radiation shielding ability of silicone rubber composites
- Damage accumulation and failure mechanism of glass/epoxy composite laminates subjected to repeated low velocity impacts
- Gamma-ray shielding analysis using the experimental measurements for copper(ii) sulfate-doped polyepoxide resins
- Numerical simulation into influence of airflow channel quantities on melt-blowing airflow field in processing of polymer fiber
- Cellulose acetate oleate-reinforced poly(butylene adipate-co-terephthalate) composite materials
- Radiation shielding capability and exposure buildup factor of cerium(iv) oxide-reinforced polyester resins
- Recyclable polytriazole resins with high performance based on Diels-Alder dynamic covalent crosslinking
- Adsorption and recovery of Cr(vi) from wastewater by Chitosan–Urushiol composite nanofiber membrane
- Comprehensive performance evaluation based on electromagnetic shielding properties of the weft-knitted fabrics made by stainless steel/cotton blended yarn
- Review Articles
- Preparation and application of natural protein polymer-based Pickering emulsions
- Wood-derived high-performance cellulose structural materials
- Flammability properties of polymers and polymer composites combined with ionic liquids
- Polymer-based nanocarriers for biomedical and environmental applications
- A review on semi-crystalline polymer bead foams from stirring autoclave: Processing and properties
- Rapid Communication
- Preparation and characterization of magnetic microgels with linear thermosensitivity over a wide temperature range
- Special Issue: Biodegradable and bio-based polymers: Green approaches (Guest Editors: Kumaran Subramanian, A. Wilson Santhosh Kumar, and Venkatajothi Ramarao)
- Synthesis and characterization of proton-conducting membranes based on bacterial cellulose and human nail keratin
- Fatigue behaviour of Kevlar/carbon/basalt fibre-reinforced SiC nanofiller particulate hybrid epoxy composite
- Effect of citric acid on thermal, phase morphological, and mechanical properties of poly(l-lactide)-b-poly(ethylene glycol)-b-poly(l-lactide)/thermoplastic starch blends
- Dose-dependent cytotoxicity against lung cancer cells via green synthesized ZnFe2O4/cellulose nanocomposites

