Abstract
Herein, the performance of silicone-modified 3D printing photosensitive resin was examined. Bisphenol-A epoxy acrylate (EA) was used as the substrate and isophorone diisocyanate, hydroxy-silicone oil, and hydroxyethyl acrylate were used as the raw materials. A silicone intermediate was synthesized to modify the substrate to prepare the 3D printing photosensitive material. The as-synthesized materials were characterized using Fourier transform infrared spectroscopy and scanning electron microscopy. The tensile fracture morphology was also analyzed. The effects of the addition of silicone intermediates on the mechanical properties, thermal stability, and shrinkage of the prepared 3D printing photosensitive resins were investigated. The results showed that an organosilicone group was successfully introduced into the side chain of EA. When the ratio of n(silicone):n(EA) is 0.3:1, the material has a high impact strength of 19.4 kJ·m−2, which is 32.8% higher than that of the pure resin; in addition, the elongation at break is 8.65% (compared to 6.56% of the pure resin). The maximum thermal weight loss temperature is 430.33°C, which is 6°C higher than that of the pure resin.
1 Introduction
Stereolithography (SLA) is a 3D printing technology that primarily uses photochemical reactions for curing. In traditional ultraviolet (UV) curing, a light-sensitive material is irradiated by UV light of a certain wavelength, transforming the material from liquid to solid state; generally, optical–thermal dual curing is used (1,2,3,4,5,6,7). SLA-3D printing involves a polymerization reaction initiated by an electron beam on a layer of resin or monomer solution; upon activation, the monomer is rapidly transformed into polymer chains and only the desired portion is cured within the resin layer at the end of the polymerization reaction (8,9,10,11,12,13). Epoxy resin has wide applications in various fields, including SLA-3D printing, because of its excellent performance. However, this resin has shortcomings, such as high curing shrinkage, poor toughness, unsatisfactory heat resistance, and low precision of molded parts, owing to which it often cannot meet various application requirements (14,15,16,17,18,19).
As an alternative, silicone has good thermal stability, oxidation resistance, weather resistance, and low-temperature characteristics, and it can compensate for the shortcomings of epoxy resin to some extent (20,21,22). Structurally, silicone is a compound with inorganic and organic characteristics, such as low surface tension, good compressibility, high- and low-temperature resistance, and good electrical insulation (23,24). Silicone resin is a type of cross-linked semi-inorganic polymer with an organic group containing a Si–O–Si structure as the main chain and connected with silicon atoms. The resin exhibits excellent heat-resistance performance. The bond energy of Si–O is 460 kJ·mol−1, which is considerably higher than that of C–C (358 kJ·mol−1); in addition, silicone resin exhibits excellent moisture resistance, good flexibility, and hydrophobicity (25,26,27).
By blending or copolymerizing an appropriate proportion of silicone resin into an epoxy resin, flexible Si–O bonds can be introduced into the cured product to reduce the internal stress of the epoxy resin; this treatment further improves the toughness, thermal stability, and moisture resistance while retaining the original properties of the epoxy resin (28,29,30,31,32,33). Yu et al. (34) prepared modified epoxy resins suitable for UV curing via silicone toughening; they added 10–15% silicone resin to the epoxy resin. The cured products exhibited good resistance to heat and moisture and elongation at break increased by 19.2%. Ma et al. (35) first synthesized polydimethylsiloxane intermediates (AGPMS) by reacting γ-aminopropyltriethoxysilane with 2,3-epoxypropoxypropyl-capped polydimethylsiloxane. Further, AGPMS and bisphenol-A epoxy resin were mixed evenly and cured to obtain the silicone-modified epoxy resin coating. Results showed that the impact strength, flexibility, and thermal stability of the silicone-modified epoxy resin coating improved considerably. Shen et al. (36) investigated the characterization and properties of the organosilicone intermediate RSN-6018-modified epoxy resin. A series of epoxy resins modified with RSN-6018 were prepared via the condensation reaction of C–OH of bisphenol-A epoxy resin with Si–O of the organosilicone intermediate. These resins have a curing content of up to 80% and can be cured at room temperature (25°C) with polyamide curing agents to obtain clear coatings. The paint film is more stable at temperatures below 348.96°C. The glass transition temperature (T g) of the cured coating decreases as the proportion of silicone intermediates increases. Currently, most of the research on silicone-modified epoxy resins has focused on the reaction of amine groups and halogen atoms in silicones with the epoxy groups to open rings. Although the epoxy resin was successfully modified and improved properties were realized after curing, the resulting resin was depleted of epoxy groups, which reduced the crosslink density of the system and, in turn, the glass transition temperature of the resin.
In this study, bisphenol-A epoxy acrylic acid was used as the substrate, and isophorone diisocyanate (IPDI), hydroxy-silicone oil, and hydroxyethyl acrylate (HEA) were used as the raw materials. A silicone intermediate was prepared to modify the substrate. The isocyanate of the intermediate product reacted with the hydroxyl group of the epoxy acrylate (EA), thereby introducing the –S–O– group in the side chain of EA. The effects of EAs modified with different n(silicone):n(EA) molar ratios on the viscosity, curing shrinkage, tensile strength, elongation at break, impact strength, hardness, thermal stability, and glass transition temperature of the 3D-printing light-curing systems were systematically investigated. The preparation and printing of the resin composite are shown in Figure 1.
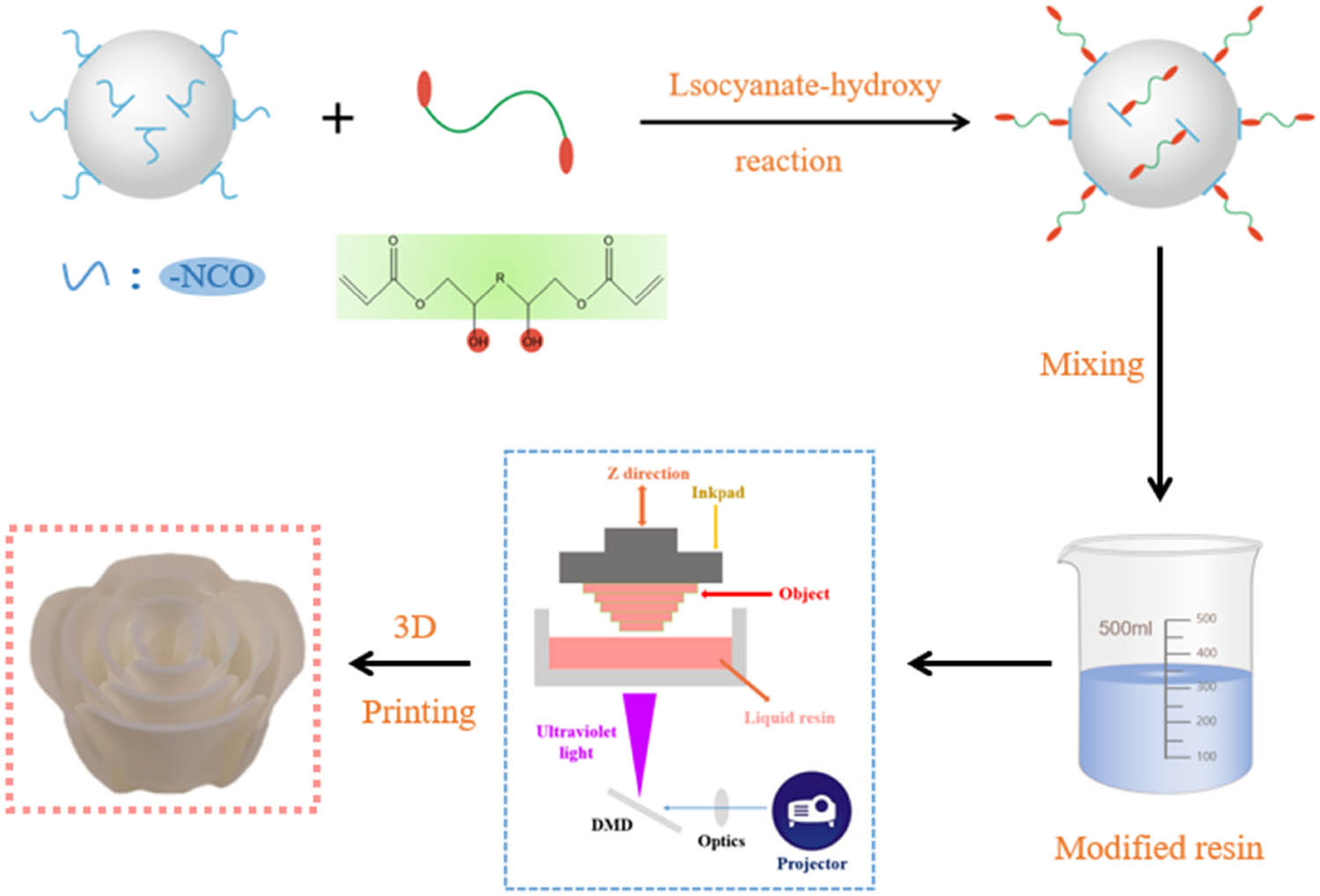
Illustration of the preparation and 3D printing of resin composite.
2 Materials and methods
2.1 Materials and instruments
Materials: Bisphenol-A epoxy acrylic acid (EA): 3100A, Shenzhen KLF Industrial Co; 2,4,6-Trimethylbenzoyl-diphenylphosphine oxide, 1,6-Hexanediol diacrylate (HDDA): Chengdu Si Cheng New Material Co; IPDI, dibutyltin dilaurate (DBTDL), HEA, hydroxysilicone oil, Shanghai Maclean Biochemical Technology Co; Acetone, Sinopharm Chemical Reagent Co; Di-n-butylamine, Shanghai Maclean Biochemical Technology Co; Hydroquinone, Texas Keanalysis Chemical Products Co.
Instruments: SLA-3D printer (Moai), Peopoly Moai, Hong Kong; Fourier infrared (IR) spectrometer (Tensor-II), Boruc, Germany; Field emission scanning electron microscope (JSM-6700), Japan Electronics Corporation; Thermogravimetric analyzer (TG209F1), NETZSCH Instrument Manufacturing GmbH, Germany; Electronic Universal Testing Machine (CMT6103), Sanshi Eternal Technology Co; Pendulum Impact Tester (ZBC-1400), New Sansi Enterprise Development Co.
2.2 Synthesis of organosilicone intermediates
IDPI (0.2 mol; 44.56 g) was introduced into a three-necked flask equipped with a spherical condenser tube, an electric stirrer, and a constant-pressure separatory funnel. The flask was heated to 65°C, and 2–3 drops of DBTDL were added; subsequently, 0.1 mol (9.217 g) of the organosilicone polymer, hydroxy-silicone oil, was added dropwise. After the dropwise addition, the –NCO (isocyanate) content of the solution was measured every 10 min with 0.1 mol·L−1 acetone-di-n-butylamine solution. When the –NCO content was reduced to half of its initial value, 0.1 mol (11.612 g) of HEA and an appropriate amount (1.308 g) of the blocking agent hydroquinone were added dropwise. After complete titration, the –NCO content of the solution was measured every 10 min. The reaction was completed when the –NCO content reached the theoretical value. The mechanism of synthesis of the silicone intermediate is depicted in Figure 2.

Synthesis mechanism of organosilicon intermediates.
2.3 Synthesis of silicone-modified EA
The as-prepared silicone intermediate product was cooled to room temperature. Further, the EA was added to the three-necked flask, and the flask was heated up to 50°C. Acetone-di-n-butylamine titration was performed and changes in the –NCO content were observed via IR monitoring. The reaction was completed when the characteristic absorption peak of –NCO at 2,270 cm−1 in the IR spectrum disappeared completely; subsequently, the acetone in the solution was distilled at atmospheric pressure. The reaction mechanism of silicone-modified EA is shown in Figure 3.

Synthesis mechanism of silicone-modified EA.
2.4 Preparation of silicone-modified 3D printing photosensitive materials and printing molding
The synthesized silicone samples were treated with EA in different molar ratios (0.1:1, 0.2:1, 0.3:1, 0.4:1, and 0.5:1). After the reaction was completed, a mixture of EA and HDDA with a mass ratio of 1.7 and a photoinitiator with a mass fraction of 4.5% were added to the silicone-modified EA. Subsequently, the samples were stirred magnetically for 4 h at 1,000 rpm, allowed to stand for 12 h to remove the bubbles, and stored away from light. Figure 4 shows the steps in 3D printing.
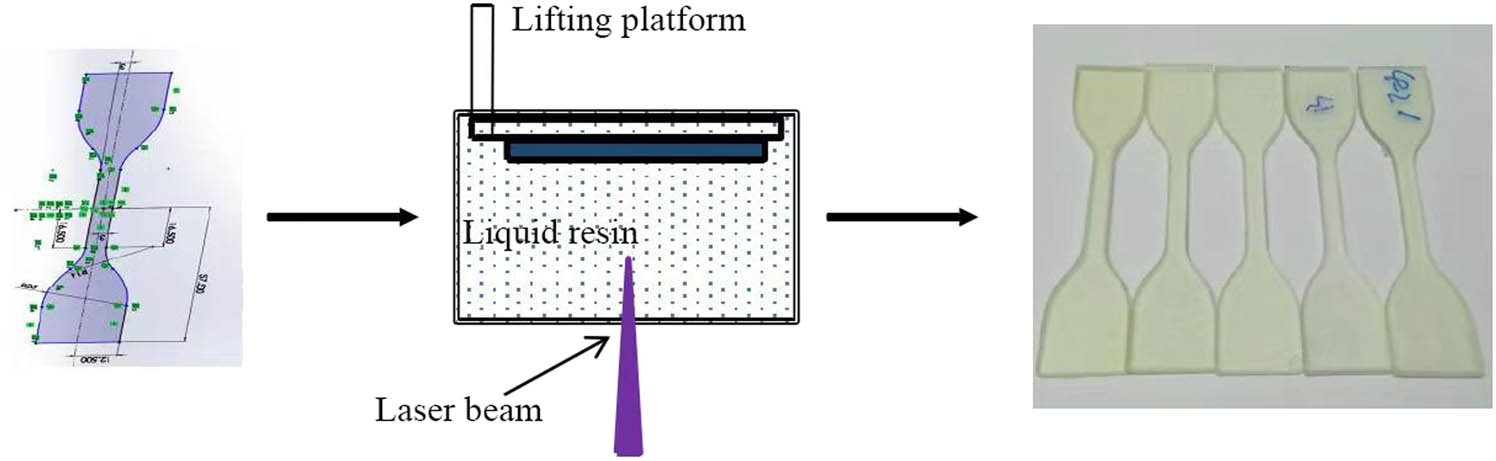
Curing process of epoxy based photocurable 3D printing materials.
3 Results and discussion
3.1 Determination of –NCO content
0.1 g of the synthesized silicone sample was placed in a 50 mL stoppered conical flask and 20 mL of a 0.1 mol·L−1 acetone-di-n-butylamine solution was added to the flask. Subsequently, the flask was closed with a stopper and stirred with a magnet for 20 min. After the reactants were completely dissolved, 3–4 drops of bromophenol blue reagent were added and the solution was titrated with 0.1 mol·L−1 of a HCl standard solution. The end point of titration was recorded when the solution changed from blue to yellow. Simultaneously, a blank experiment was performed. The isocyanate content can be calculated using Eq. 1.
where V 1 is the volume of HCl standard solution consumed by the blank experiment (mL), V 2 is the volume of HCl standard solution consumed by the specimen (mL), c is the concentration of the HCl standard solution (mol·L−1), and m is the mass of the sample (g).
3.2 Monitoring the preparation of the silicone intermediate
In the preparation of silicone intermediates and modified light-cured EAs, the changes in the –NCO content during the reaction were monitored via acetone-di-n-butylamine titration and IR testing to determine whether the reaction reached the end point. Table 1 summarizes the changes in –NCO content during the experiment.
Changes in the content of –NCO during the experiment
| Reaction time (min) | –NCO (%) |
|---|---|
| 0 (HEA + hydroxyl silicone oil) | 41.8 |
| 30 (HEA + hydroxyl silicone oil) | 19.8 |
| 60 (30 min after HEA addition) | 10.7 |
Figure 5 shows the synthesis of organosilicone intermediates and the IR profiles of their modified epoxy resins. The figure clearly shows that after 30 min of reaction with hydroxyl silicone oil in IPDI, a clear characteristic absorption peak of −NCO appeared at 2,270 cm−1. After the addition of HEA for 30 min, the –NCO content (indicated by the peak observed at 2,270 cm−1) decreased considerably; the characteristic absorption peaks of C═C and N–H appeared at 1,670 and 3,510 cm−1, respectively, indicating the completion of the reaction between –NCO and –OH and the C═C group was successfully introduced into the organosilicon intermediate. After the completion of the abovementioned reaction, EA was added to the synthesized silicone for the 2 h reaction. Figure 6 shows that the characteristic absorption peak of –NCO (2,270 cm−1) basically disappeared after the reaction with EA was complete, and the characteristic absorption peak of N–H was present at 3,510 cm−1, indicating that –NCO reacted with the –OH on the side chain of EA. Thus, organosilicone groups were successfully introduced on the side chain of the EA.

Fourier transform infrared spectrum of organosilicon modified EA.

The effect of different n(Silicone):n(EA) molar ratios on resin curing shrinkage.
3.3 Effect of silicone content on curing shrinkage of photosensitive resins
Figure 6 shows the effect of different silicone-to-EA molar ratios on the curing shrinkage of 3D-printed light-cured resins. The figure shows that the volume shrinkage and linear shrinkage of the photosensitive resin decrease as the molar ratio n(Silicone):n(EA) increases. When the ratio exceeds 0.2:1, the curing shrinkage is less than 6%; this value meets the curing shrinkage requirement for these resins. At a ratio of 0.5:1, the curing shrinkage reaches a minimum value of 4.57% because the SLA-3D printing technology mainly utilizes photochemical reactions for curing. In addition to the shrinkage caused by the close alignment of atoms during curing, the free volume changes because of the transition from a monomer to a polymer. The monomer molecules are linearly and loosely arranged before curing, and the free volume is large. After curing, the density of intermolecular cross-linking increases and the movement of chain segments is restricted; the free volume decreases, resulting in a higher shrinkage rate. The addition of silicone intermediates can effectively improve this effect so that there is little difference between the free volume before and after curing. Thus, the silicone intermediates reduce the curing shrinkage of the system.
3.4 Effect of silicone content on the viscosity of photosensitive resins
Figure 7 shows the effect of different silicone-to-EA molar ratios on the viscosity of the 3D-printed light-cured resin. The shear rate and temperature at which the viscosity measurements were conducted were 50 s−1 and 25°C, respectively, and the viscosity at five different points was determined and averaged for each sample. The graph shows that the viscosity of the photosensitive material used for 3D printing before curing increases with the molar ratio. When n(silicone):n(EA) is 0:1, the viscosity of the pure epoxy resin is 249 mPa·s, and when n(silicone):n(EA) is 0.5:1, the viscosity reaches 1,320 mPa·s, which is 81.1% higher than that of the pure epoxy resin. There are two reasons for the increase in viscosity of the product: one is the condensation reaction between hydroxy-silicone oil and IPDI during the preparation of the silicone intermediate and chain expansion; this reaction leads to a considerable increase in the molecular weight of the modified EA, resulting in an increase in viscosity. The other reason is that because of the poor compatibility between EA and silicone intermediates, a certain degree of phase separation occurs, and the silicone intermediates contain aminomethyl acid bonds that undergo hydrogen bonding, making the photosensitive resin more viscous.

Effect of different n(Silicone)/n(EA) molar ratios on the viscosity silicone modified resins.
3.5 Effect of silicone content on the mechanical properties of photosensitive resins
Figure 8 shows the effect of modification of the epoxy resin with different silicone contents on the mechanical properties of the printed parts. Figure 8(a), (b), and (d) show that with a gradual increase in the n(Silicone)/n(EA) molar ratio, the tensile strength of the modified printed parts decreases and the impact strength and elongation at break improve. Figure 8(c) shows that the hardness decreases as the n(Silicone)/n(EA) molar ratio gradually increases, but the effect is not significant, and the hardness value is always above 80 HD. This indicates that silicone-modified epoxy resins can be used for floor coating and waterproofing in the construction sector, encapsulation, and protection of circuit boards in the electronics sector, and the manufacture of composite materials in the aerospace sector (37,38). At n(Silicone)/n(EA) of 0.3:1, the impact strength increases from 14.6 to 19.4 kJ·m−2, which is 32.8% higher compared to the impact strength of pure EA, and the elongation at break increased from 6.56% for pure EA to 8.65%. The decrease in tensile strength may be attributed to the fact that the flexible –Si–O– chains introduced in the resin act as new network nodes. With a gradual increase in the number of –Si–O– chains, the internal stress in the epoxy resin reduces; hence, the printed parts have enhanced toughness.

Effect of different contents of silicone-modified EA on the mechanical properties of molded parts: (a) tensile strength, (b) elongation at break, (c) hardness, and (d) impact strength.
3.6 Microstructure analysis of the tensile section of the silicone-modified photosensitive resin
The improvement in the mechanical properties can also be verified by testing the microstructure of the tensile section of the specimen. Figure 9(a–f) shows the microscopic morphology of the tensile section of the pure epoxy resin and the epoxy resin modified with different silicone contents. The comparison of microscopic morphology clearly shows that the tensile fracture surface of the unmodified resin is smoother; cracks are transmitted on similar surfaces, and the cracks on the fracture surface are finer and shallower as shown in Figure 9(a), which is a typical characteristic of brittle fracture (39). As can be seen from Figure 9(b–f), in contrast, the tensile section of the silicone-modified resin is rough with more creases; the cracks are deeper, and the ductile depression area is clearly observed. The appearance of all these features indicates that the –Si–O– bond energy in silicone is much greater than the –C–C– bond energy and –C–O– bond energy. Hence, compared to the unmodified resin, the modified resin absorbs more energy after curing, resulting in a shift from brittle fracture to ductile fracture. Because of the addition of the flexible –Si–O– chain segment, the proportion of the original rigid EA decreases, and to a certain extent, the chain segment acts as a toughening agent. In addition, when excessive silicone content is added, the toughness of the resin decreases after curing; this decrease occurs because the silicone intermediate synthesized by IPDI with hydroxy-silicone oil and HEA as raw materials in this experiment contains rigid cyclohexane groups, and as the silicone content increases, the number of rigid groups in the resin also gradually increases.

SEM images of tensile fracture surface of silicone modified epoxy resin. (a) Unmodified resin and (b–f) silicone-modified resin.
3.7 Thermal weight loss curves of the silicone-modified photosensitive resins
Figure 10 shows the thermal weight loss curves of pure and modified epoxy resins. The corresponding data, such as T 50% (decomposition temperature at 50% weight loss), T max (the maximum thermal weight loss temperature), and residual mass at 800°C are listed in Table 2. The figure shows that the thermal stability of the epoxy resin improves upon modification with silicone. For a n(Silicone):n(EA) ratio of 0.3:1, the T 50% and T max of the photosensitive resin increased by 1.9°C and 6°C, respectively, compared to the T 50% and T max of the unmodified epoxy resin. The increase in the maximum thermal weight loss temperature is due to the fact that the EA side link is grafted with silicone chain segments and double bonds, and the silicone chain segments decompose on heating to generate silica that coats the surface of the epoxy resin, forming a glassy protective layer (40,41). This layer prevents further decomposition of the epoxy resin to some extent. Thus, the modification with silicone has the effect of improving the thermal stability of the epoxy resin.
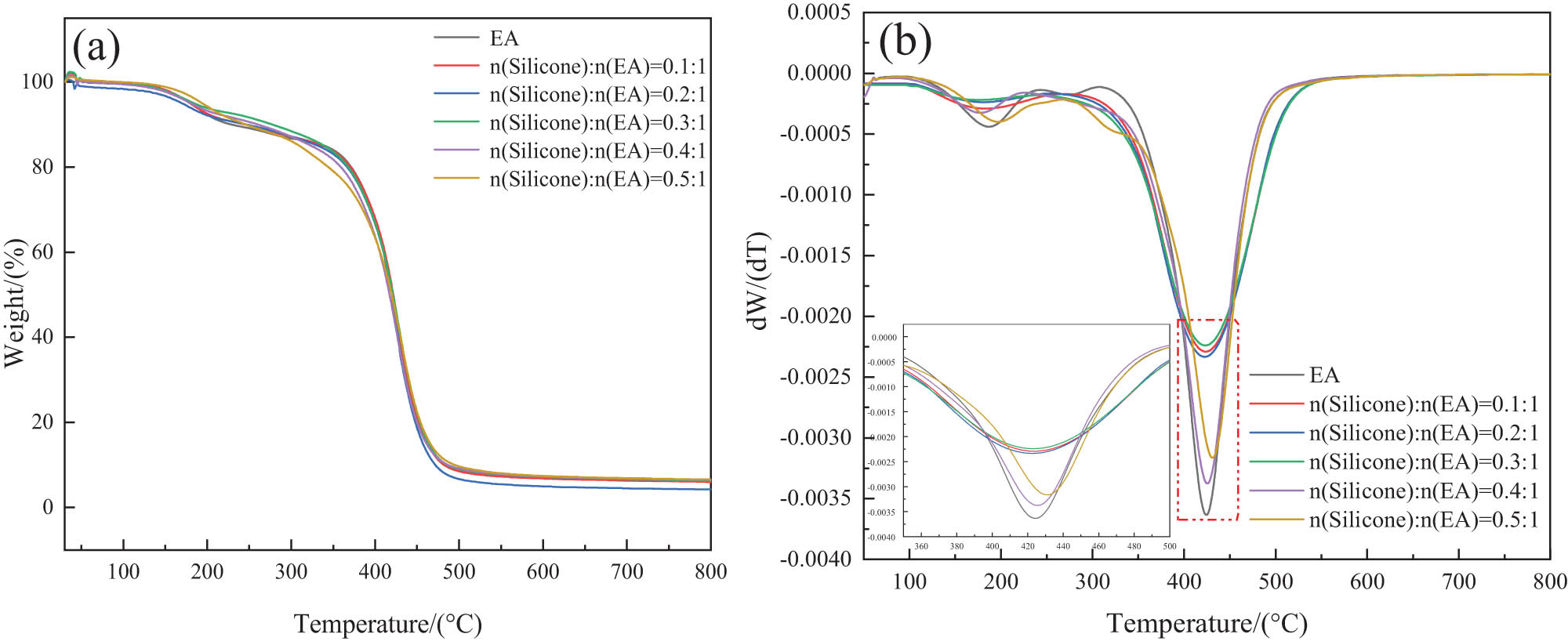
TG-DTG curves of the organic silicon-modified 3D printing photosensitive material: (a) TG and (b) DTG.
TGA data of pure epoxy resin and different proportions of silicone modification
| (n)Silicone/(n)EA | T 50% (°C) | T max (°C) | Residue (%) |
|---|---|---|---|
| 0 | 422 | 424 | 8 |
| 0.1:1 | 424 | 428 | 7 |
| 0.2:1 | 425 | 427 | 5 |
| 0.3:1 | 426 | 430 | 8 |
| 0.4:1 | 419 | 424 | 8 |
| 0.5:1 | 425 | 430 | 8 |
3.8 Effect of silicone modification on the glass transition temperature of resin
The glass transition temperature is an important index that characterizes the heat resistance of the resin. To investigate the effect of silicone modification at different molar ratios on the glass transition temperature after printing and curing, differential scanning calorimetry (DSC) analysis was performed on the epoxy resin with different silicone-to-EA molar ratios. Figure 11 shows the curves of the variations in the glass transition temperature of the silicone-modified epoxy resin. The measured glass transition temperatures are listed in Table 3. The test results show that with the increase in the silicone content, the glass transition temperature of the modified epoxy resin first increases and then decreases; the highest temperature was observed for a n(Silicone):n(EA) ratio of 0.4:1, and this temperature was 8.46°C higher than that of the unmodified resin. The increase in glass transition temperature can be explained as follows: the prepared silicone intermediates contain –NH–, which can easily form hydrogen bonds, and side chains contain rigid cyclic groups, which hinder the free movement of siloxane chain segments; their combined effect results in an increase in the glass transition temperature of the modified epoxy resin after curing.

T g change curves of the silicone-modified epoxy resin.
Glass transition temperature (T g) of epoxy resin before and after modification with silicone
| (n)Silicone/(n)EA | 0 | 0.1:1 | 0.2:1 | 0.3:1 | 0.4:1 | 0.5:1 |
|---|---|---|---|---|---|---|
| T g (°C) | 78 | 80 | 80 | 82 | 87 | 84 |
3.9 Influence of light intensity on the mechanical properties of resins
Figure 12 shows the trend of the effect of different light intensities on the mechanical properties of resin cured parts. The results of a series of aforementioned tests showed that 0.3:1 is the optimal n(silicone):n(EA) ratio and the comprehensive performance of the silicone-modified resin is optimal at this ratio. To investigate the effect of different light intensities on the mechanical properties of the resins, the resins configured with n(silicone):n(EA) of 0.3:1 were cured under different light intensities and their mechanical properties were evaluated. Figure 12(a) shows that the tensile strength and elongation at break tend to increase and subsequently decrease. The rate of increase and decrease in the elongation at break is slower than that of the tensile strength. The graph shows that the tensile strength and elongation at break are maximum when the light intensity is 60 mW·cm−2. This is because insufficient light intensity slows down the resin curing speed, resulting in a nonideal curing effect; therefore, the mechanical properties are not satisfactory. Excessive light intensity will make the resin surface uneven or undergo cracking, leading to uneven curing and thereby reducing the mechanical properties. Light intensity is another important factor affecting the hardness of light-curing resins. As shown in Figure 12(b), the hardness value increases rapidly between light intensities of 20 and 60 mW·cm−2. When the light intensity continues to increase to 80 mW·cm−2, the hardness value increases by only 1 HD. Substantially low light intensity will lead to an insufficient reaction and thereby to insufficiently strong hardness of the resin. The greater the light intensity, the faster the reaction speed, the shorter the curing time, and the higher the hardness. In summary, the mechanical properties of the resin are optimal when the light intensity is 60 mW·cm−2.

Effect of different light intensities on the mechanical properties of resins: (a) tensile strength and elongation at break and (b) hardness.
3.10 Rapid prototyping of photosensitive resin by SLA
The silicone-modified photosensitive resin materials prepared in this study were printed and molded using SLA equipment to obtain resin-based parts with complex structures. Figure 13 shows that the 3D printing resin that was modified by grafting the epoxy side chains with the silicone intermediates synthesized in the laboratory can be fully molded and can meet the performance requirements of the photosensitive resin for the SLA molding. Simple parts were molded with smooth surfaces, and complete structures were obtained without any obvious defects or residual resin.

Resin-based molded parts.
4 Conclusion
In this study, an organosilicone intermediate was prepared using bisphenol-A-based EA as a substrate and IPDI, hydroxy-silicone oil, and HEA as the raw materials. The substrate was modified without depleting the epoxy group.
(1) IR characterization showed that an organosilicone intermediate was successfully prepared and the –Si–O– chain ends and C═C bonds were successfully grafted onto the side chains of EA.
(2) Mechanical property tests showed that the introduction of silicone improved the overall mechanical properties of the epoxy resins. The impact strength and elongation at break of the silicone-modified 3D printed materials were improved. When the n(Silicone):n(EA) ratio was 0.3:1, the impact strength of the molded parts increased from 14.6 to 19.4 kJ·m−2, which is 32.8% higher than the impact strength of the unmodified resin; furthermore, the elongation at break increased from 6.56% (unmodified resin) to 8.65%. When the light intensity was 60 mW·cm−2, the resin underwent the optimal curing and exhibited optimal mechanical properties.
(3) Thermal weight loss analysis showed that silicone modification improved the heat resistance of the resin. When the n(Silicone):n(EA) ratio was 0.3:1, the values of T 50% and T max of the modified resin increased by 1.9°C and 6°C, respectively, compared to those of the pure epoxy resin; that is, the heat resistance was clearly enhanced. The DSC test showed that the addition of silicone could significantly increase the glass transition temperature of epoxy resin; with increase in the silicone content, the glass transition temperature first increased, attained a maximum value at a silicone-to-EA molar ratio of 0.4:1, and then decreased. The maximum value was higher than that of the unmodified resin by 8.46°C.
-
Funding information: Authors state no funding involved.
-
Author contributions: Lu Zheng: writing – original draft, writing – review and editing, methodology, and formal analysis; Jin Chen: writing – original draft and visualization; Wenwen Zhou: formal analysis and resources; Min Liu: supervision and validation; Yuyu Gao: software; and Jiaqiang Xie: project administration.
-
Conflict of interest: Authors state no conflict of interest.
References
(1) Mendes‐Felipe C, Oliveira J, Etxebarria I, Vilas-Vilela JL, Lanceros-Mendez S. State‐of‐the‐art and future challenges of UV curable polymer‐based smart materials for printing technologies. Adv Mater Technol. 2019;4(3):1800618.10.1002/admt.201800618Search in Google Scholar
(2) Chou WH, Gamboa A, Morales JO. Inkjet printing of small molecules, biologics, and nanoparticles. Int J Pharm. 2021;600:120462.10.1016/j.ijpharm.2021.120462Search in Google Scholar PubMed
(3) Hakeim OA, Arafa AA, Zahran MK, Abdou LAW. Characterisation and application of pigmented UV-curable inkjet inks. Pigment Resin Technol. 2018;47(2):164–72.10.1108/PRT-11-2016-0099Search in Google Scholar
(4) Scotton RS, Lília M, Guerrini, Oliveira MP. Evaluation of solvent-based and UV-curing inkjet inks on the adhesion and printing quality of different aircraft surfaces coating. Prog Org Coat. 2021;158:106389.10.1016/j.porgcoat.2021.106389Search in Google Scholar
(5) Quan H, Zhang T, Xu H, Luo S, Zhu X. Photo-curing 3D printing technique and its challenges. Bioact Mater. 2020;5(1):110–5.10.1016/j.bioactmat.2019.12.003Search in Google Scholar PubMed PubMed Central
(6) Xu X, Awad A, Martinez PR, Gaisford S, Basit AW. Vat photopolymerization 3D printing for advanced drug delivery and medical device applications. J Control Release. 2020;329(2):743–57.10.1016/j.jconrel.2020.10.008Search in Google Scholar PubMed
(7) Tammaro D, Gatta RD, Villone MM, Maffettone PL. Continuous 3D printing of hierarchically structured microfoamed objects. Adv Eng Mater. 2021;24(5):2101226.10.1002/adem.202101226Search in Google Scholar
(8) Waldbaur A, Rapp H, Lnge K, Rapp BE. Let there be chip–towards rapid prototyping of microfluidic devices: one-step manufacturing processes. Anal Methods-UK. 2011;3:2681–716.10.1039/c1ay05253eSearch in Google Scholar
(9) Au AK, Lee W, Folch A. Mail-order microfluidics: evaluation of stereolithography for the production of microfluidic devices. Lab Chip. 2014;14(7):1294–301.10.1039/C3LC51360BSearch in Google Scholar
(10) Gross BC, Erkal JL, Lockwood SY, Chen C, Spence DM. Evaluation of 3D printing and its potential impact on biotechnology and the chemical sciences. Anal Chem. 2014;86(7):3240–53.10.1021/ac403397rSearch in Google Scholar PubMed
(11) Ma XL. Research on application of SLA technology in the 3D printing technology. AMM. 2013;401–403:938–41.10.4028/www.scientific.net/AMM.401-403.938Search in Google Scholar
(12) Tumbleston JR, Shirvanyants D, Ermoshkin N, Janusziewicz R, Johnson AR, Kelly D, et al. Additive manufacturing. Continuous liquid interface production of 3D objects. Science. 2015;347(6228):1349–52.10.1126/science.aaa2397Search in Google Scholar
(13) Ngo TD, Kashani A, Imbalzano G, Nguyen KT, Hui D. Additive manufacturing (3D printing): A review of materials, methods, applications and challenges. Compos Part B. 2018;143:172–96.10.1016/j.compositesb.2018.02.012Search in Google Scholar
(14) Jawaid M, Awad S, Fouad H, Asim M, Saba N, Dhakal HN. Improvements in the thermal behaviour of date palm/bamboo fibres reinforced epoxy hybrid composites. Compos Struct. 2021;277:114644.10.1016/j.compstruct.2021.114644Search in Google Scholar
(15) Cavalcanti D, Banea MD, Neto J, Lima R. Comparative analysis of the mechanical and thermal properties of polyester and epoxy natural fibre-reinforced hybrid composites. J Compos Mater. 2021;55(12):1683–92.10.1177/0021998320976811Search in Google Scholar
(16) Wei W, Zhao Y, Zhang B, Cui Y, Liu X. Silica/poly(styrene-alt-maleic anhydride) hybrid particles as a reactive toughener for epoxy resin. J Appl Polym Sci. 2020;137(34):48986.10.1002/app.48986Search in Google Scholar
(17) Zotti A, Zuppolini S, Borriello A, Zarrelli M. Thermal properties and fracture toughness of epoxy nanocomposites loaded with hyperbranched-polymers-based core/shell nanoparticles. Nanomaterials-Basel. 2019;9(3):418.10.3390/nano9030418Search in Google Scholar
(18) Wen R, Huo J, Lv J, Liu Z, Yu Y. Effect of silicone resin modification on the performance of epoxy materials for LED encapsulation. J Mater Sci-Mater Electron. 2017;28(19):14522–35.10.1007/s10854-017-7316-5Search in Google Scholar
(19) Duan Y, Huo Y, Duan L. Preparation of acrylic resins modified with epoxy resins and their behaviors as binders of waterborne printing ink on plastic film. Colloid Surface A. 2017;535:225–31.10.1016/j.colsurfa.2017.09.041Search in Google Scholar
(20) Guenther J, Reibenspies J, Blümel J. Synthesis and characterization of tridentate phosphine ligands incorporating long methylene chains and ethoxysilane groups for immobilizing molecular rhodium catalysts. Mol Catal. 2019;479:110629.10.1016/j.mcat.2019.110629Search in Google Scholar
(21) Heng Z, Zeng Z, Chen Y, Zou H, Liang M. Silicone modified epoxy resins with good toughness, damping properties and high thermal residual weight. J Polym Res. 2015;22(11):1–7.10.1007/s10965-015-0852-xSearch in Google Scholar
(22) Huo S, Ma H, Liu G, Jin C, Chen C, Wu G, et al. Synthesis and properties of organosilicon-grafted cardanol novolac epoxy resin as a novel biobased reactive diluent and toughening agent. ACS Omega. 2018;3(12):16403–8.10.1021/acsomega.8b02401Search in Google Scholar PubMed PubMed Central
(23) Chen Y, Wu Y, Geng C, Li Z, Cui W. Curing kinetics and the properties of KH560-SiO2/polyethersulfone/bismaleimide-phenolic epoxy resin composite. J Inorg Organomet Polym Mater. 2019;30(18):1–9.10.1007/s10904-019-01290-1Search in Google Scholar
(24) Li J, Wang H, Li S. A novel phosphorus−silicon containing epoxy resin with enhanced thermal stability, flame retardancy and mechanical properties. Polym Degrad Stabil. 2019;164(Jun):36–45.10.1016/j.polymdegradstab.2019.03.020Search in Google Scholar
(25) Ge Z, Luo Y. Synthesis and characterization of siloxane-modified two-component waterborne polyurethane. Prog Org Coat. 2013;76(11):1522–6.10.1016/j.porgcoat.2013.06.007Search in Google Scholar
(26) Zhang M, Wu Y, Wu H, Zhang Q. Study on reactive polydimethylsiloxane-modified waterborne polyurethanes. J Polym Res. 2012;19(1):9807.10.1007/s10965-011-9807-zSearch in Google Scholar
(27) Yilgör E, Yilgör I. Silicone containing copolymers: Synthesis, properties and applications. Prog Polym Sci. 2014;39(6):1165–95.10.1016/j.progpolymsci.2013.11.003Search in Google Scholar
(28) Wu SY, Lee SL, Chang WL, Sung PH. Polysiloxane modified epoxy networks. IV. Catalytic effect on fracture behaviors of jointed interpenetrating polymer networks. J Appl Polym Sci. 2002;84(13):2352–7.10.1002/app.10429Search in Google Scholar
(29) Huang W, Yao Y, Huang Y, Yu Y. Surface modification of epoxy resin by polyether-polydimethylsiloxanes–polyether triblock copolymers. Polymer. 2001;42(4):1763–6.10.1016/S0032-3861(00)00393-1Search in Google Scholar
(30) Cabanelas JC, Serrano B, González-Benito J, Bravo J, Baselga J. Morphology of epoxy/polyorganosiloxane reactive blends. Macromol Rapid Commun. 2015;22(9):694–9.10.1002/1521-3927(20010601)22:9<694::AID-MARC694>3.0.CO;2-8Search in Google Scholar
(31) Shon MY, Kwon HS. Effects of surface modification with amino branched polydimethylsiloxane (ABP) on the corrosion protection of epoxy coating. Corros Sci. 2007;49(11):4259–75.10.1016/j.corsci.2007.05.006Search in Google Scholar
(32) Ji Y, Zhang Y, Wang P, Li Y, Sui J. Mechanical and thermal properties of epoxy resins modified by a novel thermoplastic-polyimide. Fiber Polym. 2021;22(1):205–12.10.1007/s12221-021-9142-xSearch in Google Scholar
(33) Chen L, Fan L, Gao L. Modification of epoxy resin with silicone for electronic encapsulation application. Adv Mater Res. 2014;936:627–34.10.4028/www.scientific.net/AMR.936.643Search in Google Scholar
(34) Yu Z, Cui A, Zhao P, Wei H, Hu F. Preparation and properties studies of UV-curable silicone modified epoxy resin composite system. J Appl Biomater Func. 2018;16(1):170–6.10.1177/2280800017753053Search in Google Scholar PubMed
(35) Ma S, Liu W, Li H, Tang C, Wei Z. Morphologies and mechanical and thermal properties of epoxy resins modified by a novel polysiloxane capped with silane coupling agent, epoxide, and imino groups. J Macromol Sci B. 2011;50(5):975–87.10.1080/00222348.2010.497124Search in Google Scholar
(36) Shen Z, Xia Z, Zhang Y. Characterization and properties of epoxy resin (E-20) modified with silicone intermediate RSN-6018. Prog Org Coat. 2018;114:115–22.10.1016/j.porgcoat.2017.10.014Search in Google Scholar
(37) Zhang X, Xu Y, Li L, Yan B, Bao J, Zhang A. Acrylate-based photosensitive resin for stereolithographic three-dimensional printing. J Appl Polym Sci. 2019;136(21a22):47487.10.1002/app.47487Search in Google Scholar
(38) Bhattacharjee N, Parra-Cabrera C, Kim YT, Kuo AP, Folch A. Desktop‐stereolithography 3D‐printing of a poly(dimethylsiloxane)‐based material with sylgard‐184 properties. Adv Mater. 2018;30(22):1800001.1–7.10.1002/adma.201800001Search in Google Scholar PubMed PubMed Central
(39) Liu Y, Li L, Liu H, Zhang M, Zhou S. Hollow polymeric microsphere-filled silicone-modified epoxy as an internally insulated material for composite cross-arm applications. Compos Sci Technol. 2020;200:108418.10.1016/j.compscitech.2020.108418Search in Google Scholar
(40) Ma S, Liu WQ, Yu D, Wang ZF. Modification of epoxy resin with polyether-grafted-polysiloxane and epoxy-miscible polysiloxane particles. Macromol Res. 2010;18(1):22–8.10.1007/s13233-009-0053-8Search in Google Scholar
(41) Zhang K, Shen MM, Wu K, Liu HF, Zhang Y Comparative study on flame retardancy and thermal degradation of phosphorus- and silicon-containing epoxy resin composites. J Polym Res. 2011;18(6):2061–70.10.1007/s10965-011-9615-5Search in Google Scholar
© 2023 the author(s), published by De Gruyter
This work is licensed under the Creative Commons Attribution 4.0 International License.
Articles in the same Issue
- Research Articles
- Chitosan nanocomposite film incorporating Nigella sativa oil, Azadirachta indica leaves’ extract, and silver nanoparticles
- Effect of Zr-doped CaCu3Ti3.95Zr0.05O12 ceramic on the microstructure, dielectric properties, and electric field distribution of the LDPE composites
- Effects of dry heating, acetylation, and acid pre-treatments on modification of potato starch with octenyl succinic anhydride (OSA)
- Loading conditions impact on the compression fatigue behavior of filled styrene butadiene rubber
- Characterization and compatibility of bio-based PA56/PET
- Study on the aging of three typical rubber materials under high- and low-temperature cyclic environment
- Numerical simulation and experimental research of electrospun polyacrylonitrile Taylor cone based on multiphysics coupling
- Experimental investigation of properties and aging behavior of pineapple and sisal leaf hybrid fiber-reinforced polymer composites
- Influence of temperature distribution on the foaming quality of foamed polypropylene composites
- Enzyme-catalyzed synthesis of 4-methylcatechol oligomer and preliminary evaluations as stabilizing agent in polypropylene
- Molecular dynamics simulation of the effect of the thermal and mechanical properties of addition liquid silicone rubber modified by carbon nanotubes with different radii
- Incorporation of poly(3-acrylamidopropyl trimethylammonium chloride-co-acrylic acid) branches for good sizing properties and easy desizing from sized cotton warps
- Effect of matrix composition on properties of polyamide 66/polyamide 6I-6T composites with high content of continuous glass fiber for optimizing surface performance
- Preparation and properties of epoxy-modified thermosetting phenolic fiber
- Thermal decomposition reaction kinetics and storage life prediction of polyacrylate pressure-sensitive adhesive
- Effect of different proportions of CNTs/Fe3O4 hybrid filler on the morphological, electrical and electromagnetic interference shielding properties of poly(lactic acid) nanocomposites
- Doping silver nanoparticles into reverse osmosis membranes for antibacterial properties
- Melt-blended PLA/curcumin-cross-linked polyurethane film for enhanced UV-shielding ability
- The affinity of bentonite and WO3 nanoparticles toward epoxy resin polymer for radiation shielding
- Prolonged action fertilizer encapsulated by CMC/humic acid
- Preparation and experimental estimation of radiation shielding properties of novel epoxy reinforced with Sb2O3 and PbO
- Fabrication of polylactic acid nanofibrous yarns for piezoelectric fabrics
- Copper phenyl phosphonate for epoxy resin and cyanate ester copolymer with improved flame retardancy and thermal properties
- Synergistic effect of thermal oxygen and UV aging on natural rubber
- Effect of zinc oxide suspension on the overall filler content of the PLA/ZnO composites and cPLA/ZnO composites
- The role of natural hybrid nanobentonite/nanocellulose in enhancing the water resistance properties of the biodegradable thermoplastic starch
- Performance optimization of geopolymer mortar blending in nano-SiO2 and PVA fiber based on set pair analysis
- Preparation of (La + Nb)-co-doped TiO2 and its polyvinylidene difluoride composites with high dielectric constants
- Effect of matrix composition on the performance of calcium carbonate filled poly(lactic acid)/poly(butylene adipate-co-terephthalate) composites
- Low-temperature self-healing polyurethane adhesives via dual synergetic crosslinking strategy
- Leucaena leucocephala oil-based poly malate-amide nanocomposite coating material for anticorrosive applications
- Preparation and properties of modified ammonium polyphosphate synergistic with tris(2-hydroxyethyl) isocynurate for flame-retardant LDPE
- Thermal response of double network hydrogels with varied composition
- The effect of coated calcium carbonate using stearic acid on the recovered carbon black masterbatch in low-density polyethylene composites
- Investigation of MXene-modified agar/polyurethane hydrogel elastomeric repair materials with tunable water absorption
- Damping performance analysis of carbon black/lead magnesium niobite/epoxy resin composites
- Molecular dynamics simulations of dihydroxylammonium 5,5′-bistetrazole-1,1′-diolate (TKX-50) and TKX-50-based PBXs with four energetic binders
- Preparation and characterization of sisal fibre reinforced sodium alginate gum composites for non-structural engineering applications
- Study on by-products synthesis of powder coating polyester resin catalyzed by organotin
- Ab initio molecular dynamics of insulating paper: Mechanism of insulating paper cellobiose cracking at transient high temperature
- Effect of different tin neodecanoate and calcium–zinc heat stabilizers on the thermal stability of PVC
- High-strength polyvinyl alcohol-based hydrogel by vermiculite and lignocellulosic nanofibrils for electronic sensing
- Impacts of micro-size PbO on the gamma-ray shielding performance of polyepoxide resin
- Influence of the molecular structure of phenylamine antioxidants on anti-migration and anti-aging behavior of high-performance nitrile rubber composites
- Fiber-reinforced polyvinyl alcohol hydrogel via in situ fiber formation
- Preparation and performance of homogenous braids-reinforced poly (p-phenylene terephthamide) hollow fiber membranes
- Synthesis of cadmium(ii) ion-imprinted composite membrane with a pyridine functional monomer and characterization of its adsorption performance
- Impact of WO3 and BaO nanoparticles on the radiation shielding characteristics of polydimethylsiloxane composites
- Comprehensive study of the radiation shielding feature of polyester polymers impregnated with iron filings
- Preparation and characterization of polymeric cross-linked hydrogel patch for topical delivery of gentamicin
- Mechanical properties of rCB-pigment masterbatch in rLDPE: The effect of processing aids and water absorption test
- Pineapple fruit residue-based nanofibre composites: Preparation and characterizations
- Effect of natural Indocalamus leaf addition on the mechanical properties of epoxy and epoxy-carbon fiber composites
- Utilization of biosilica for energy-saving tire compounds: Enhancing performance and efficiency
- Effect of capillary arrays on the profile of multi-layer micro-capillary films
- A numerical study on thermal bonding with preheating technique for polypropylene microfluidic device
- Development of modified h-BN/UPE resin for insulation varnish applications
- High strength, anti-static, thermal conductive glass fiber/epoxy composites for medical devices: A strategy of modifying fibers with functionalized carbon nanotubes
- Effects of mechanical recycling on the properties of glass fiber–reinforced polyamide 66 composites in automotive components
- Bentonite/hydroxyethylcellulose as eco-dielectrics with potential utilization in energy storage
- Study on wall-slipping mechanism of nano-injection polymer under the constant temperature fields
- Synthesis of low-VOC unsaturated polyester coatings for electrical insulation
- Enhanced apoptotic activity of Pluronic F127 polymer-encapsulated chlorogenic acid nanoparticles through the PI3K/Akt/mTOR signaling pathway in liver cancer cells and in vivo toxicity studies in zebrafish
- Preparation and performance of silicone-modified 3D printing photosensitive materials
- A novel fabrication method of slippery lubricant-infused porous surface by thiol-ene click chemistry reaction for anti-fouling and anti-corrosion applications
- Development of polymeric IPN hydrogels by free radical polymerization technique for extended release of letrozole: Characterization and toxicity evaluation
- Tribological characterization of sponge gourd outer skin fiber-reinforced epoxy composite with Tamarindus indica seed filler addition using the Box–Behnken method
- Stereocomplex PLLA–PBAT copolymer and its composites with multi-walled carbon nanotubes for electrostatic dissipative application
- Enhancing the therapeutic efficacy of Krestin–chitosan nanocomplex for cancer medication via activation of the mitochondrial intrinsic pathway
- Variation in tungsten(vi) oxide particle size for enhancing the radiation shielding ability of silicone rubber composites
- Damage accumulation and failure mechanism of glass/epoxy composite laminates subjected to repeated low velocity impacts
- Gamma-ray shielding analysis using the experimental measurements for copper(ii) sulfate-doped polyepoxide resins
- Numerical simulation into influence of airflow channel quantities on melt-blowing airflow field in processing of polymer fiber
- Cellulose acetate oleate-reinforced poly(butylene adipate-co-terephthalate) composite materials
- Radiation shielding capability and exposure buildup factor of cerium(iv) oxide-reinforced polyester resins
- Recyclable polytriazole resins with high performance based on Diels-Alder dynamic covalent crosslinking
- Adsorption and recovery of Cr(vi) from wastewater by Chitosan–Urushiol composite nanofiber membrane
- Comprehensive performance evaluation based on electromagnetic shielding properties of the weft-knitted fabrics made by stainless steel/cotton blended yarn
- Review Articles
- Preparation and application of natural protein polymer-based Pickering emulsions
- Wood-derived high-performance cellulose structural materials
- Flammability properties of polymers and polymer composites combined with ionic liquids
- Polymer-based nanocarriers for biomedical and environmental applications
- A review on semi-crystalline polymer bead foams from stirring autoclave: Processing and properties
- Rapid Communication
- Preparation and characterization of magnetic microgels with linear thermosensitivity over a wide temperature range
- Special Issue: Biodegradable and bio-based polymers: Green approaches (Guest Editors: Kumaran Subramanian, A. Wilson Santhosh Kumar, and Venkatajothi Ramarao)
- Synthesis and characterization of proton-conducting membranes based on bacterial cellulose and human nail keratin
- Fatigue behaviour of Kevlar/carbon/basalt fibre-reinforced SiC nanofiller particulate hybrid epoxy composite
- Effect of citric acid on thermal, phase morphological, and mechanical properties of poly(l-lactide)-b-poly(ethylene glycol)-b-poly(l-lactide)/thermoplastic starch blends
- Dose-dependent cytotoxicity against lung cancer cells via green synthesized ZnFe2O4/cellulose nanocomposites
Articles in the same Issue
- Research Articles
- Chitosan nanocomposite film incorporating Nigella sativa oil, Azadirachta indica leaves’ extract, and silver nanoparticles
- Effect of Zr-doped CaCu3Ti3.95Zr0.05O12 ceramic on the microstructure, dielectric properties, and electric field distribution of the LDPE composites
- Effects of dry heating, acetylation, and acid pre-treatments on modification of potato starch with octenyl succinic anhydride (OSA)
- Loading conditions impact on the compression fatigue behavior of filled styrene butadiene rubber
- Characterization and compatibility of bio-based PA56/PET
- Study on the aging of three typical rubber materials under high- and low-temperature cyclic environment
- Numerical simulation and experimental research of electrospun polyacrylonitrile Taylor cone based on multiphysics coupling
- Experimental investigation of properties and aging behavior of pineapple and sisal leaf hybrid fiber-reinforced polymer composites
- Influence of temperature distribution on the foaming quality of foamed polypropylene composites
- Enzyme-catalyzed synthesis of 4-methylcatechol oligomer and preliminary evaluations as stabilizing agent in polypropylene
- Molecular dynamics simulation of the effect of the thermal and mechanical properties of addition liquid silicone rubber modified by carbon nanotubes with different radii
- Incorporation of poly(3-acrylamidopropyl trimethylammonium chloride-co-acrylic acid) branches for good sizing properties and easy desizing from sized cotton warps
- Effect of matrix composition on properties of polyamide 66/polyamide 6I-6T composites with high content of continuous glass fiber for optimizing surface performance
- Preparation and properties of epoxy-modified thermosetting phenolic fiber
- Thermal decomposition reaction kinetics and storage life prediction of polyacrylate pressure-sensitive adhesive
- Effect of different proportions of CNTs/Fe3O4 hybrid filler on the morphological, electrical and electromagnetic interference shielding properties of poly(lactic acid) nanocomposites
- Doping silver nanoparticles into reverse osmosis membranes for antibacterial properties
- Melt-blended PLA/curcumin-cross-linked polyurethane film for enhanced UV-shielding ability
- The affinity of bentonite and WO3 nanoparticles toward epoxy resin polymer for radiation shielding
- Prolonged action fertilizer encapsulated by CMC/humic acid
- Preparation and experimental estimation of radiation shielding properties of novel epoxy reinforced with Sb2O3 and PbO
- Fabrication of polylactic acid nanofibrous yarns for piezoelectric fabrics
- Copper phenyl phosphonate for epoxy resin and cyanate ester copolymer with improved flame retardancy and thermal properties
- Synergistic effect of thermal oxygen and UV aging on natural rubber
- Effect of zinc oxide suspension on the overall filler content of the PLA/ZnO composites and cPLA/ZnO composites
- The role of natural hybrid nanobentonite/nanocellulose in enhancing the water resistance properties of the biodegradable thermoplastic starch
- Performance optimization of geopolymer mortar blending in nano-SiO2 and PVA fiber based on set pair analysis
- Preparation of (La + Nb)-co-doped TiO2 and its polyvinylidene difluoride composites with high dielectric constants
- Effect of matrix composition on the performance of calcium carbonate filled poly(lactic acid)/poly(butylene adipate-co-terephthalate) composites
- Low-temperature self-healing polyurethane adhesives via dual synergetic crosslinking strategy
- Leucaena leucocephala oil-based poly malate-amide nanocomposite coating material for anticorrosive applications
- Preparation and properties of modified ammonium polyphosphate synergistic with tris(2-hydroxyethyl) isocynurate for flame-retardant LDPE
- Thermal response of double network hydrogels with varied composition
- The effect of coated calcium carbonate using stearic acid on the recovered carbon black masterbatch in low-density polyethylene composites
- Investigation of MXene-modified agar/polyurethane hydrogel elastomeric repair materials with tunable water absorption
- Damping performance analysis of carbon black/lead magnesium niobite/epoxy resin composites
- Molecular dynamics simulations of dihydroxylammonium 5,5′-bistetrazole-1,1′-diolate (TKX-50) and TKX-50-based PBXs with four energetic binders
- Preparation and characterization of sisal fibre reinforced sodium alginate gum composites for non-structural engineering applications
- Study on by-products synthesis of powder coating polyester resin catalyzed by organotin
- Ab initio molecular dynamics of insulating paper: Mechanism of insulating paper cellobiose cracking at transient high temperature
- Effect of different tin neodecanoate and calcium–zinc heat stabilizers on the thermal stability of PVC
- High-strength polyvinyl alcohol-based hydrogel by vermiculite and lignocellulosic nanofibrils for electronic sensing
- Impacts of micro-size PbO on the gamma-ray shielding performance of polyepoxide resin
- Influence of the molecular structure of phenylamine antioxidants on anti-migration and anti-aging behavior of high-performance nitrile rubber composites
- Fiber-reinforced polyvinyl alcohol hydrogel via in situ fiber formation
- Preparation and performance of homogenous braids-reinforced poly (p-phenylene terephthamide) hollow fiber membranes
- Synthesis of cadmium(ii) ion-imprinted composite membrane with a pyridine functional monomer and characterization of its adsorption performance
- Impact of WO3 and BaO nanoparticles on the radiation shielding characteristics of polydimethylsiloxane composites
- Comprehensive study of the radiation shielding feature of polyester polymers impregnated with iron filings
- Preparation and characterization of polymeric cross-linked hydrogel patch for topical delivery of gentamicin
- Mechanical properties of rCB-pigment masterbatch in rLDPE: The effect of processing aids and water absorption test
- Pineapple fruit residue-based nanofibre composites: Preparation and characterizations
- Effect of natural Indocalamus leaf addition on the mechanical properties of epoxy and epoxy-carbon fiber composites
- Utilization of biosilica for energy-saving tire compounds: Enhancing performance and efficiency
- Effect of capillary arrays on the profile of multi-layer micro-capillary films
- A numerical study on thermal bonding with preheating technique for polypropylene microfluidic device
- Development of modified h-BN/UPE resin for insulation varnish applications
- High strength, anti-static, thermal conductive glass fiber/epoxy composites for medical devices: A strategy of modifying fibers with functionalized carbon nanotubes
- Effects of mechanical recycling on the properties of glass fiber–reinforced polyamide 66 composites in automotive components
- Bentonite/hydroxyethylcellulose as eco-dielectrics with potential utilization in energy storage
- Study on wall-slipping mechanism of nano-injection polymer under the constant temperature fields
- Synthesis of low-VOC unsaturated polyester coatings for electrical insulation
- Enhanced apoptotic activity of Pluronic F127 polymer-encapsulated chlorogenic acid nanoparticles through the PI3K/Akt/mTOR signaling pathway in liver cancer cells and in vivo toxicity studies in zebrafish
- Preparation and performance of silicone-modified 3D printing photosensitive materials
- A novel fabrication method of slippery lubricant-infused porous surface by thiol-ene click chemistry reaction for anti-fouling and anti-corrosion applications
- Development of polymeric IPN hydrogels by free radical polymerization technique for extended release of letrozole: Characterization and toxicity evaluation
- Tribological characterization of sponge gourd outer skin fiber-reinforced epoxy composite with Tamarindus indica seed filler addition using the Box–Behnken method
- Stereocomplex PLLA–PBAT copolymer and its composites with multi-walled carbon nanotubes for electrostatic dissipative application
- Enhancing the therapeutic efficacy of Krestin–chitosan nanocomplex for cancer medication via activation of the mitochondrial intrinsic pathway
- Variation in tungsten(vi) oxide particle size for enhancing the radiation shielding ability of silicone rubber composites
- Damage accumulation and failure mechanism of glass/epoxy composite laminates subjected to repeated low velocity impacts
- Gamma-ray shielding analysis using the experimental measurements for copper(ii) sulfate-doped polyepoxide resins
- Numerical simulation into influence of airflow channel quantities on melt-blowing airflow field in processing of polymer fiber
- Cellulose acetate oleate-reinforced poly(butylene adipate-co-terephthalate) composite materials
- Radiation shielding capability and exposure buildup factor of cerium(iv) oxide-reinforced polyester resins
- Recyclable polytriazole resins with high performance based on Diels-Alder dynamic covalent crosslinking
- Adsorption and recovery of Cr(vi) from wastewater by Chitosan–Urushiol composite nanofiber membrane
- Comprehensive performance evaluation based on electromagnetic shielding properties of the weft-knitted fabrics made by stainless steel/cotton blended yarn
- Review Articles
- Preparation and application of natural protein polymer-based Pickering emulsions
- Wood-derived high-performance cellulose structural materials
- Flammability properties of polymers and polymer composites combined with ionic liquids
- Polymer-based nanocarriers for biomedical and environmental applications
- A review on semi-crystalline polymer bead foams from stirring autoclave: Processing and properties
- Rapid Communication
- Preparation and characterization of magnetic microgels with linear thermosensitivity over a wide temperature range
- Special Issue: Biodegradable and bio-based polymers: Green approaches (Guest Editors: Kumaran Subramanian, A. Wilson Santhosh Kumar, and Venkatajothi Ramarao)
- Synthesis and characterization of proton-conducting membranes based on bacterial cellulose and human nail keratin
- Fatigue behaviour of Kevlar/carbon/basalt fibre-reinforced SiC nanofiller particulate hybrid epoxy composite
- Effect of citric acid on thermal, phase morphological, and mechanical properties of poly(l-lactide)-b-poly(ethylene glycol)-b-poly(l-lactide)/thermoplastic starch blends
- Dose-dependent cytotoxicity against lung cancer cells via green synthesized ZnFe2O4/cellulose nanocomposites

