Abstract
Being able to accurately monitor blood pressure in a reliable, truly non-invasive manner is a highly sought after goal within the biomedical community. In this paper we propose and assess a system, methodology and algorithm for unobtrusively obtaining true pulse transit time data from readily accessible peripheral locations, such as the hand, using a highly synchronous body-sensor-network encompassing an electrocardiogram- and dual mode photoplethysmogram sensor node. The results suggest the feasibility of acquiring such data, which strongly correlates with the recorded reference blood pressure, and can therefore be further employed to track changes thereof.
1 Introduction
Blood pressure issues are a serious health risk, not only for the elderly, but also for the population at large. Its monitoring and regulation is therefore crucial for keeping the human machinery running for as long as possible. Other than routine punctual check-ups in ambulant settings, close surveillance over extended periods of time may become necessary in case of intermittent ailments, and has also long been a part of every major surgical procedure. The need for a continuous and non-invasive method of assessing blood pressure (BP) is as high as ever, with various success in its resolution. The two most common procedures of measuring BP are the cuff sphygmomanometer and the intra-arterial catheter. The first one, while somewhat non-invasive, has the big deficit of being confined to burst sampling rates of a few per minute, and much lower for long-term monitoring (due to its cutting off of blood flow), and can only be reasonably used for punctual data acquisition. On the other side of the spectrum lies the arterial catheter – used almost exclusively during major surgical procedures, it delivers a continuous and accurate reading of BP, at the cost of being prohibitively invasive and risky for routine deployment. A few devices use finger cuffs to continuously, quasi-non-invasively measure BP, akin to the sphygmomanometer, with various accuracies and need for periodic calibration [1].
A truly non-invasive, continuous method of estimating BP is based on the following simple principle: pressure waves travel at different speeds through a medium at different pressures; i.e. the velocity, and therefore the time required between two points therein, is a function of system pressure. Therefore, one should be able, at least in theory, to draw conclusions about the arterial BP from attaining information about the propagation of the pressure (pulse) wave within the arterial system [2], [3]. The pulse wave originates in the heart’s left ventricle during systole, and sets on its way to propagate through the aorta to subsequent arteries, arterioles and finally capillaries, before it is almost completely dissipated in the venous system. The bulk of the vascular resistance the pressure wave is pushing against is implemented by the smallest of the blood vessels, the arterioles and capillaries. Nonetheless, all blood vessels can expand or contract to a certain degree, as a result of sympatho-vagal activity partly informed by baro- and shear receptors in the artery walls [13].

Measurement system.
The pulse arrival time (PAT) is composed of the pre-ejection period (PEP; isovolumetric ventricular contraction) and the pulse transit time (PTT) to the pick-up location [4]. PAT is usually measured from the preceding R-peak of the electrocardiogram (ECG) signal to a fiducial point (onset, peak, etc.) of the corresponding photoplethysmogram (PPG) pulse wave (see Figure 2). Since the PEP is difficult to predict or measure, and not a linear function of pressure, the PAT is a less accurate indicator overall. To rectify this, the aortic valve opening (AO) could be used, but this is an issue still under investigation [5]. Though more complicated to assess, the PTT exhibits much better tracking of the arterial BP [6], [7]. As can be seen in Figure 3, the PTT only describes the temporal offset between the arrival times of the pulse wave at two different locations along a blood vessel.

Arm positions.
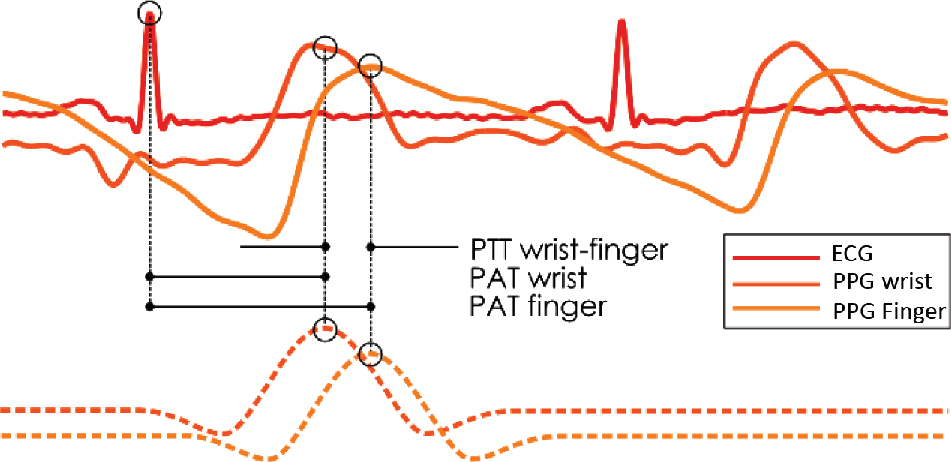
PAT and PTT wavelet method illustration.
2 Methods
2.1 Hardware
To gain access to the PTT, two pick-up points for the pulse wave are needed at different distances from the heart along the same artery. We implement this using a dual photo plethysmography (PPG) node, encompassing a conceptually traditional, transmissive finger clip (though with adaptive gain and filtering) and a more intricate reflective sensor. These two are guaranteed by design to be absolutely synchronous. The 12 bit samples are acquired with a 500 Hz rate. The reflective sensor is placed either a top the radial or ulnar artery in close proximity to the wrist, dependent on the emerging signal quality. The finger clip can be positioned on whichever finger, as long as the arterial length in-between is roughly known (typ. 15–25 cm). This setup would, in theory, suffice for calculating the PTT. Nonetheless, for the sake of robustness, a wirelessly (Bluetooth) synchronized minimal ECG is recorded to be able to assess the different PAT at the two sites. These subsystems are part of a bigger robust body-sensor-network (rBSN) [8], [9].
The standard used for validation is the data retrieved from a Finapres Medical Systems Portapres system, which has been synchronously recorded via the Portapres analog output module and an rBSN universal data recorder. The finger cuff is attached to a different finger than the transmissive PPG clip, so as to avoid interference, and the hydrostatic pressure compensation of the Portapres has been deactivated. The Portapres is by no way a gold standard – unfortunately arterial catheter or cuff sphygmomanometer measurements were not viable because of their invasivity and low sample rate respectively.
2.2 Methodology
The experiment involves raising and holding the measured limb at various angles to the ground while standing: –90, –45, 0, 45 and 90 degrees respectively [10]. This effects different relative height to the heart and emulates a change in blood pressure. The limb can either be passively held in place by appropriate means, or actively held up, yielding two measurement scenarios.
2.3 Processing
The resulting signals (one ECG – preferably Einthoven II, and two PPGs) are processed in Matlab, at first using quasi-standardized algorithms: the R-peak detection is done via a Pan-Thompkins-like algorithm [11], albeit slightly adapted for noisy environments. The DC component of the PPG signals is discarded, as it bears no significance for the further proceedings, and different fiducial points thereof are computed; for example pulse wave onset and peak are extracted with a modified Zong algorithm [12].
From here on, two methods will be pursued: based on the aforementioned fiducial points, or employing the continuous wavelet transform. Since the expected fluctuation in PAT is very small, the task of reliably accessing this information is jeopardized by signal disturbances. Therefore the chosen reference point for the pulse wave arrival is halfway between onset and first maximum – much more immune to noise and artifacts due to its location on the sharply ascending upslope.
The second method relies on the continuous wavelet transform to identify the pulse wave location within the signal.
The advantage of this approach, though more computationally complex, is its relative imperviousness to noise and uncorrelated artifacts. To increase the temporal resolution of the results, a ten-fold up sampling is performed, followed by the CWT with an appropriate analysing wavelet. In the performed analysis, Mexican Hat employing rather large scaling factors has empirically been proven most appropriate so far. As pulse wave morphology can vary very strongly between individuals and even at different pick-up locations (Figure 3) of the same one, the effect of wavelet shape is difficult to evaluate. After each R-peak, the maximum of the resulting coefficients is an indicator for the most probable wave location. The resulting indexes from the two methods can now be subtracted to gain the PTT between wrist and finger. As obvious from Figure 3, the morphology of the pulse wave, or rather the change thereof, plays a role in the detected peak locations, and therefore the calculated PAT and PTT. This is, however, not problematic, but should much rather could have the added benefit of assessing two features at the same time – PTT and morphology variation related to BP changes. Nonetheless, further research needs to be undertaken to investigate the link.
3 Results
Over 800 heartbeats have been analysed so far, pertaining to five postures in each of the two experimental setups (passive vs. active arm sustain). The results fall in line with the expected behavior, as can be seen in Table 1: when the arm is lowered, and the net pressure high, the PTT remains low around 5 ms. With progressive heightening of the arm, the BP drops, and the PTT rises to in excess of 120 ms. Also, while the transitions between arm states in the passive scenario tended to be more gradual, the active raising and sustaining caused sharp and well-defined jumps in PTT. Figures 4 and 5 exemplarily show the PTT and PAT of a single measurement (male, healthy, 26, assisted arm raise) – the 90 degree segment data should be taken with a grain of salt, as the PPG quality degrades to a point where both methods fail to reliably detect most pulse waves.
Mean PAT and PTT and BP change vs. arm angle (90° data unreliable).
| Angle | PTT | PAT wrist | PAT finger | BP change |
|---|---|---|---|---|
| −90° | −31.5 ± 7 ms | −5 ± 30 ms | −14 ± 30 ms | +34.8 ± 29% |
| −45° | −23 ± 19 ms | −5 ± 34 ms | −10 ± 29 ms | +24.28 ± 19% |
| 0° | 0 ± 40 ms | 285 ± 41 ms | 295 ± 35 ms | 0 ± 41% |
| 45° | 25 ± 40 ms | +2 ± 57 ms | +35 ± 62 ms | −31.18 ± 39% |
| 90° | 85 ± 45 ms | −3 ± 36 ms | +130 ± 99 ms | −29.98 ± 29% |

PTT results with wavelet method.

PAT results at wrist and finger.
3.1 Remarks
As stated, during the 90 and sometimes 45 degree raising of the arm, the PPG quality spontaneously degenerates, yielding an unsatisfactorily accurate signal for determining the pulse wave locations. This is due to multiple factors, amongst which is the experimental setup itself. By producing such a big height difference and, therefore, static pressure differential between heart and hand, the pulse strength is strongly diminished. Furthermore, due to the uncomfortable and somewhat unnatural pose, tremors and contractions are provoked in the subject, juddering the measurement apparatus and introducing disturbances.
The PTT does not, however, tell the whole truth. One would likely expect to find a simultaneous fluctuation of the PAT at both locations (i.e. wrist and finger) – interestingly, this is not the case. While the PAT at the fingertip does visibly scale with the height of the hand, the PAT at the wrist seems less influenced, remaining flat almost throughout all phases, as depicted in Figure 5. This would suggest that the big arteries (aorta, brachial and radial/ulnar) do not experience much pressure change at all, even when the BP as a whole does vary. This is consistent with the actuality that most of the vascular resistance occurs in the small blood vessels [13], making the changes in BP more visible therein.
Finally, it has been observed that the PAT in the fingertips (and accordingly the PTT) does not return to base level as soon as the limb is lowered in its incipient position, but does linger in a heightened state for some time. This is probably due to the latency of the capillary system’s reaction to abrupt state changes. Also, PAT at the wrist seems to rise only for the 90 degree test, but returns to normal instantaneously after release, hinting at a possible mechanical clamping of the arteries at the shoulder.
4 Conclusion
It has been proven possible to extract meaningful PTT information from non-invasive, continuous forms of pulse wave measurements via PPG. A compact system with sufficient synchronicity and sampling rate can be employed to record these variations even over relatively short pick-up distances. The evoked changes in blood pressure by posture of the measured limb are made well visible in the PTT with the use of the described processing methods and can be therefore used to predict BP.
Acknowledgement
The authors would like to thank all the people involved in creating and refining the rBSN over the years, and the volunteers partaking in the testing thereof.
Author’s Statement
Research funding: The author state no funding involved. Conflict of interest: Authors state no conflict of interest. Material and Methods: Informed consent: Informed consent has been obtained from all individuals included in this study. Ethical approval: The research related to human use complied with all the relevant national regulations, institutional policies and was performed in accordance with the tenets of the Helsinki Declaration, and has been approved by the authors’ institutional review board or equivalent committee.
References
[1] Gesche H, Grosskurth D, Küchler G, Patzak A. Continuous blood pressure measurement by using the PTT: comparison to a cuff-based method. Eur J Appl Physiol. 2012;112:309–15.10.1007/s00421-011-1983-3Search in Google Scholar PubMed
[2] Geddes LA, Voelz M, James S, Reiner D. Pulse arrival time as a method of obtaining systolic and diastolic blood pressure indirectly. Med Biol Eng Comput. 1981;19:671–2.10.1007/BF02442787Search in Google Scholar PubMed
[3] Geddes LA, Voelz MH, Babbs CF, Bourland JD, Tacker WA. Pulse transit time as an indicator of arterial blood pressure. Psychophysiology. 1981;18:71–4.10.1111/j.1469-8986.1981.tb01545.xSearch in Google Scholar PubMed
[4] Newlin DB. Relationships of pulse transmission times to pre-ejection period and blood pressure. Psychophysiology. 1981;18:316–21.10.1111/j.1469-8986.1981.tb03042.xSearch in Google Scholar PubMed
[5] Vaini E, Lombardi P, Di Rienzo M. “Aortic-finger pulse transit time vs. R-derived pulse arrival time: a beat-to-beat assessment.” Computing in Cardiology Conference (CinC) 2015, Nice, France, 2015, pp. 253–256. doi: 10.1109/CIC.2015.7408634.10.1109/CIC.2015.7408634Search in Google Scholar
[6] Payne RA, Symeonides CN, Webb DJ, Maxwell SRJ. Pulse transit time measured from the ECG: an unreliable marker of beat-to-beat blood pressure. J Appl Physiol. 2006;100:136–41.10.1152/japplphysiol.00657.2005Search in Google Scholar PubMed
[7] Zhang G, Gao M, Xu D, Olivier NB, Mukkamala R. Pulse arrival time is not an adequate surrogate for pulse transit time as a marker of blood pressure. J Appl Physiol (1985). 2011;111:1681–6.10.1152/japplphysiol.00980.2011Search in Google Scholar PubMed
[8] Pflugradt M, Fritzsch I, Mann S, Tigges T, Orglmeister R. “A novel pulseoximeter for bluetooth synchronized measurements in a body sensor network,” in Education and Research Conference (EDERC), 2014 6th European Embedded Design in, vol. no., pp. 21–25, 11–12 Sept. 2014. doi: 10.1109/EDERC.2014.6924351.10.1109/EDERC.2014.6924351Search in Google Scholar
[9] Volmer A, Orglmeister R. Wireless body sensor network for low-power motion-tolerant syncronized vital sign measurment. Conf Proc IEEE Eng Med Biol Soc. 2008;2008:3422–5.10.1109/IEMBS.2008.4649941Search in Google Scholar PubMed
[10] Jeong G-Y, Yu K-H, Kim N-G. “Continuous blood pressure monitoring using pulse wave transit time”, ICCAS2005, June 2–5, Kintex, Gyeonggi-Do, Korea.Search in Google Scholar
[11] Pan J, Tompkins WJ. A real-time QRS detection algorithm. IEEE Trans Biomed Eng. 1985;32:230–6.10.1109/TBME.1985.325532Search in Google Scholar PubMed
[12] Zong W, Heldt T, Moody GB, Mark RG. An open-source algorithm to detect onset of arterial blood pressure pulses. Comput Cardiol. 2003;2003:259–62.10.1109/CIC.2003.1291140Search in Google Scholar
[13] Christensen KL, Mulvany MJ. Location of resistance arteries. J Vasc Res. 2001;38:1–12.10.1159/000051024Search in Google Scholar PubMed
©2016 Alexandru-Gabriel Pielmuş et al., licensee De Gruyter.
This work is licensed under the Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 License.
Articles in the same Issue
- Synthesis and characterization of PIL/pNIPAAm hybrid hydrogels
- Novel blood protein based scaffolds for cardiovascular tissue engineering
- Cell adhesion and viability of human endothelial cells on electrospun polymer scaffolds
- Effects of heat treatment and welding process on superelastic behaviour and microstructure of micro electron beam welded NiTi
- Long-term stable modifications of silicone elastomer for improved hemocompatibility
- The effect of thermal treatment on the mechanical properties of PLLA tubular specimens
- Biocompatible wear-resistant thick ceramic coating
- Protection of active implant electronics with organosilicon open air plasma coating for plastic overmolding
- Examination of dielectric strength of thin Parylene C films under various conditions
- Open air plasma deposited antimicrobial SiOx/TiOx composite films for biomedical applications
- Systemic analysis about residual chloroform in PLLA films
- A macrophage model of osseointegration
- Towards in silico prognosis using big data
- Technical concept and evaluation of a novel shoulder simulator with adaptive muscle force generation and free motion
- Usability evaluation of a locomotor therapy device considering different strategies
- Hypoxia-on-a-chip
- Integration of a semi-automatic in-vitro RFA procedure into an experimental setup
- Fabrication of MEMS-based 3D-μECoG-MEAs
- High speed digital interfacing for a neural data acquisition system
- Bionic forceps for the handling of sensitive tissue
- Experimental studies on 3D printing of barium titanate ceramics for medical applications
- Patient specific root-analogue dental implants – additive manufacturing and finite element analysis
- 3D printing – a key technology for tailored biomedical cell culture lab ware
- 3D printing of hydrogels in a temperature controlled environment with high spatial resolution
- Biocompatibility of photopolymers for additive manufacturing
- Biochemical piezoresistive sensors based on pH- and glucose-sensitive hydrogels for medical applications
- Novel wireless measurement system of pressure dedicated to in vivo studies
- Portable auricular device for real-time swallow and chew detection
- Detection of miRNA using a surface plasmon resonance biosensor and antibody amplification
- Simulation and evaluation of stimulation scenarios for targeted vestibular nerve excitation
- Deep brain stimulation: increasing efficiency by alternative waveforms
- Prediction of immediately occurring microsleep events from brain electric signals
- Determining cardiac vagal threshold from short term heart rate complexity
- Classification of cardiac excitation patterns during atrial fibrillation
- An algorithm to automatically determine the cycle length coverage to identify rotational activity during atrial fibrillation – a simulation study
- Deriving respiration from high resolution 12-channel-ECG during cycling exercise
- Reducing of gradient induced artifacts on the ECG signal during MRI examinations using Wilcoxon filter
- Automatic detection and mapping of double potentials in intracardiac electrograms
- Modeling the pelvic region for non-invasive pelvic intraoperative neuromonitoring
- Postprocessing algorithm for automated analysis of pelvic intraoperative neuromonitoring signals
- Best practice: surgeon driven application in pelvic operations
- Vasomotor assessment by camera-based photoplethysmography
- Classification of morphologic changes in photoplethysmographic waveforms
- Novel computation of pulse transit time from multi-channel PPG signals by wavelet transform
- Efficient design of FIR filter based low-pass differentiators for biomedical signal processing
- Nonlinear causal influences assessed by mutual compression entropy
- Comparative study of methods for solving the correspondence problem in EMD applications
- fNIRS for future use in auditory diagnostics
- Semi-automated detection of fractional shortening in zebrafish embryo heart videos
- Blood pressure measurement on the cheek
- Derivation of the respiratory rate from directly and indirectly measured respiratory signals using autocorrelation
- Left cardiac atrioventricular delay and inter-ventricular delay in cardiac resynchronization therapy responder and non-responder
- An automatic systolic peak detector of blood pressure waveforms using 4th order cumulants
- Real-time QRS detection using integrated variance for ECG gated cardiac MRI
- Preprocessing of unipolar signals acquired by a novel intracardiac mapping system
- In-vitro experiments to characterize ventricular electromechanics
- Continuous non-invasive monitoring of blood pressure in the operating room: a cuffless optical technology at the fingertip
- Application of microwave sensor technology in cardiovascular disease for plaque detection
- Artificial blood circulatory and special Ultrasound Doppler probes for detecting and sizing gaseous embolism
- Detection of microsleep events in a car driving simulation study using electrocardiographic features
- A method to determine the kink resistance of stents and stent delivery systems according to international standards
- Comparison of stented bifurcation and straight vessel 3D-simulation with a prior simulated velocity profile inlet
- Transient Euler-Lagrange/DEM simulation of stent thrombosis
- Automated control of the laser welding process of heart valve scaffolds
- Automation of a test bench for accessing the bendability of electrospun vascular grafts
- Influence of storage conditions on the release of growth factors in platelet-rich blood derivatives
- Cryopreservation of cells using defined serum-free cryoprotective agents
- New bioreactor vessel for tissue engineering of human nasal septal chondrocytes
- Determination of the membrane hydraulic permeability of MSCs
- Climate retainment in carbon dioxide incubators
- Multiple factors influencing OR ventilation system effectiveness
- Evaluation of an app-based stress protocol
- Medication process in Styrian hospitals
- Control tower to surgical theater
- Development of a skull phantom for the assessment of implant X-ray visibility
- Surgical navigation with QR codes
- Investigation of the pressure gradient of embolic protection devices
- Computer assistance in femoral derotation osteotomy: a bottom-up approach
- Automatic depth scanning system for 3D infrared thermography
- A service for monitoring the quality of intraoperative cone beam CT images
- Resectoscope with an easy to use twist mechanism for improved handling
- In vitro simulation of distribution processes following intramuscular injection
- Adjusting inkjet printhead parameters to deposit drugs into micro-sized reservoirs
- A flexible standalone system with integrated sensor feedback for multi-pad electrode FES of the hand
- Smart control for functional electrical stimulation with optimal pulse intensity
- Tactile display on the remaining hand for unilateral hand amputees
- Effects of sustained electrical stimulation on spasticity assessed by the pendulum test
- An improved tracking framework for ultrasound probe localization in image-guided radiosurgery
- Improvement of a subviral particle tracker by the use of a LAP-Kalman-algorithm
- Learning discriminative classification models for grading anal intraepithelial neoplasia
- Regularization of EIT reconstruction based on multi-scales wavelet transforms
- Assessing MRI susceptibility artefact through an indicator of image distortion
- EyeGuidance – a computer controlled system to guide eye movements
- A framework for feedback-based segmentation of 3D image stacks
- Doppler optical coherence tomography as a promising tool for detecting fluid in the human middle ear
- 3D Local in vivo Environment (LivE) imaging for single cell protein analysis of bone tissue
- Inside-Out access strategy using new trans-vascular catheter approach
- US/MRI fusion with new optical tracking and marker approach for interventional procedures inside the MRI suite
- Impact of different registration methods in MEG source analysis
- 3D segmentation of thyroid ultrasound images using active contours
- Designing a compact MRI motion phantom
- Cerebral cortex classification by conditional random fields applied to intraoperative thermal imaging
- Classification of indirect immunofluorescence images using thresholded local binary count features
- Analysis of muscle fatigue conditions using time-frequency images and GLCM features
- Numerical evaluation of image parameters of ETR-1
- Fabrication of a compliant phantom of the human aortic arch for use in Particle Image Velocimetry (PIV) experimentation
- Effect of the number of electrodes on the reconstructed lung shape in electrical impedance tomography
- Hardware dependencies of GPU-accelerated beamformer performances for microwave breast cancer detection
- Computer assisted assessment of progressing osteoradionecrosis of the jaw for clinical diagnosis and treatment
- Evaluation of reconstruction parameters of electrical impedance tomography on aorta detection during saline bolus injection
- Evaluation of open-source software for the lung segmentation
- Automatic determination of lung features of CF patients in CT scans
- Image analysis of self-organized multicellular patterns
- Effect of key parameters on synthesis of superparamagnetic nanoparticles (SPIONs)
- Radiopacity assessment of neurovascular implants
- Development of a desiccant based dielectric for monitoring humidity conditions in miniaturized hermetic implantable packages
- Development of an artifact-free aneurysm clip
- Enhancing the regeneration of bone defects by alkalizing the peri-implant zone – an in vitro approach
- Rapid prototyping of replica knee implants for in vitro testing
- Protecting ultra- and hyperhydrophilic implant surfaces in dry state from loss of wettability
- Advanced wettability analysis of implant surfaces
- Patient-specific hip prostheses designed by surgeons
- Plasma treatment on novel carbon fiber reinforced PEEK cages to enhance bioactivity
- Wear of a total intervertebral disc prosthesis
- Digital health and digital biomarkers – enabling value chains on health data
- Usability in the lifecycle of medical software development
- Influence of different test gases in a non-destructive 100% quality control system for medical devices
- Device development guided by user satisfaction survey on auricular vagus nerve stimulation
- Empirical assessment of the time course of innovation in biomedical engineering: first results of a comparative approach
- Effect of left atrial hypertrophy on P-wave morphology in a computational model
- Simulation of intracardiac electrograms around acute ablation lesions
- Parametrization of activation based cardiac electrophysiology models using bidomain model simulations
- Assessment of nasal resistance using computational fluid dynamics
- Resistance in a non-linear autoregressive model of pulmonary mechanics
- Inspiratory and expiratory elastance in a non-linear autoregressive model of pulmonary mechanics
- Determination of regional lung function in cystic fibrosis using electrical impedance tomography
- Development of parietal bone surrogates for parietal graft lift training
- Numerical simulation of mechanically stimulated bone remodelling
- Conversion of engineering stresses to Cauchy stresses in tensile and compression tests of thermoplastic polymers
- Numerical examinations of simplified spondylodesis models concerning energy absorption in magnetic resonance imaging
- Principle study on the signal connection at transabdominal fetal pulse oximetry
- Influence of Siluron® insertion on model drug distribution in the simulated vitreous body
- Evaluating different approaches to identify a three parameter gas exchange model
- Effects of fibrosis on the extracellular potential based on 3D reconstructions from histological sections of heart tissue
- From imaging to hemodynamics – how reconstruction kernels influence the blood flow predictions in intracranial aneurysms
- Flow optimised design of a novel point-of-care diagnostic device for the detection of disease specific biomarkers
- Improved FPGA controlled artificial vascular system for plethysmographic measurements
- Minimally spaced electrode positions for multi-functional chest sensors: ECG and respiratory signal estimation
- Automated detection of alveolar arches for nasoalveolar molding in cleft lip and palate treatment
- Control scheme selection in human-machine- interfaces by analysis of activity signals
- Event-based sampling for reducing communication load in realtime human motion analysis by wireless inertial sensor networks
- Automatic pairing of inertial sensors to lower limb segments – a plug-and-play approach
- Contactless respiratory monitoring system for magnetic resonance imaging applications using a laser range sensor
- Interactive monitoring system for visual respiratory biofeedback
- Development of a low-cost senor based aid for visually impaired people
- Patient assistive system for the shoulder joint
- A passive beating heart setup for interventional cardiology training
Articles in the same Issue
- Synthesis and characterization of PIL/pNIPAAm hybrid hydrogels
- Novel blood protein based scaffolds for cardiovascular tissue engineering
- Cell adhesion and viability of human endothelial cells on electrospun polymer scaffolds
- Effects of heat treatment and welding process on superelastic behaviour and microstructure of micro electron beam welded NiTi
- Long-term stable modifications of silicone elastomer for improved hemocompatibility
- The effect of thermal treatment on the mechanical properties of PLLA tubular specimens
- Biocompatible wear-resistant thick ceramic coating
- Protection of active implant electronics with organosilicon open air plasma coating for plastic overmolding
- Examination of dielectric strength of thin Parylene C films under various conditions
- Open air plasma deposited antimicrobial SiOx/TiOx composite films for biomedical applications
- Systemic analysis about residual chloroform in PLLA films
- A macrophage model of osseointegration
- Towards in silico prognosis using big data
- Technical concept and evaluation of a novel shoulder simulator with adaptive muscle force generation and free motion
- Usability evaluation of a locomotor therapy device considering different strategies
- Hypoxia-on-a-chip
- Integration of a semi-automatic in-vitro RFA procedure into an experimental setup
- Fabrication of MEMS-based 3D-μECoG-MEAs
- High speed digital interfacing for a neural data acquisition system
- Bionic forceps for the handling of sensitive tissue
- Experimental studies on 3D printing of barium titanate ceramics for medical applications
- Patient specific root-analogue dental implants – additive manufacturing and finite element analysis
- 3D printing – a key technology for tailored biomedical cell culture lab ware
- 3D printing of hydrogels in a temperature controlled environment with high spatial resolution
- Biocompatibility of photopolymers for additive manufacturing
- Biochemical piezoresistive sensors based on pH- and glucose-sensitive hydrogels for medical applications
- Novel wireless measurement system of pressure dedicated to in vivo studies
- Portable auricular device for real-time swallow and chew detection
- Detection of miRNA using a surface plasmon resonance biosensor and antibody amplification
- Simulation and evaluation of stimulation scenarios for targeted vestibular nerve excitation
- Deep brain stimulation: increasing efficiency by alternative waveforms
- Prediction of immediately occurring microsleep events from brain electric signals
- Determining cardiac vagal threshold from short term heart rate complexity
- Classification of cardiac excitation patterns during atrial fibrillation
- An algorithm to automatically determine the cycle length coverage to identify rotational activity during atrial fibrillation – a simulation study
- Deriving respiration from high resolution 12-channel-ECG during cycling exercise
- Reducing of gradient induced artifacts on the ECG signal during MRI examinations using Wilcoxon filter
- Automatic detection and mapping of double potentials in intracardiac electrograms
- Modeling the pelvic region for non-invasive pelvic intraoperative neuromonitoring
- Postprocessing algorithm for automated analysis of pelvic intraoperative neuromonitoring signals
- Best practice: surgeon driven application in pelvic operations
- Vasomotor assessment by camera-based photoplethysmography
- Classification of morphologic changes in photoplethysmographic waveforms
- Novel computation of pulse transit time from multi-channel PPG signals by wavelet transform
- Efficient design of FIR filter based low-pass differentiators for biomedical signal processing
- Nonlinear causal influences assessed by mutual compression entropy
- Comparative study of methods for solving the correspondence problem in EMD applications
- fNIRS for future use in auditory diagnostics
- Semi-automated detection of fractional shortening in zebrafish embryo heart videos
- Blood pressure measurement on the cheek
- Derivation of the respiratory rate from directly and indirectly measured respiratory signals using autocorrelation
- Left cardiac atrioventricular delay and inter-ventricular delay in cardiac resynchronization therapy responder and non-responder
- An automatic systolic peak detector of blood pressure waveforms using 4th order cumulants
- Real-time QRS detection using integrated variance for ECG gated cardiac MRI
- Preprocessing of unipolar signals acquired by a novel intracardiac mapping system
- In-vitro experiments to characterize ventricular electromechanics
- Continuous non-invasive monitoring of blood pressure in the operating room: a cuffless optical technology at the fingertip
- Application of microwave sensor technology in cardiovascular disease for plaque detection
- Artificial blood circulatory and special Ultrasound Doppler probes for detecting and sizing gaseous embolism
- Detection of microsleep events in a car driving simulation study using electrocardiographic features
- A method to determine the kink resistance of stents and stent delivery systems according to international standards
- Comparison of stented bifurcation and straight vessel 3D-simulation with a prior simulated velocity profile inlet
- Transient Euler-Lagrange/DEM simulation of stent thrombosis
- Automated control of the laser welding process of heart valve scaffolds
- Automation of a test bench for accessing the bendability of electrospun vascular grafts
- Influence of storage conditions on the release of growth factors in platelet-rich blood derivatives
- Cryopreservation of cells using defined serum-free cryoprotective agents
- New bioreactor vessel for tissue engineering of human nasal septal chondrocytes
- Determination of the membrane hydraulic permeability of MSCs
- Climate retainment in carbon dioxide incubators
- Multiple factors influencing OR ventilation system effectiveness
- Evaluation of an app-based stress protocol
- Medication process in Styrian hospitals
- Control tower to surgical theater
- Development of a skull phantom for the assessment of implant X-ray visibility
- Surgical navigation with QR codes
- Investigation of the pressure gradient of embolic protection devices
- Computer assistance in femoral derotation osteotomy: a bottom-up approach
- Automatic depth scanning system for 3D infrared thermography
- A service for monitoring the quality of intraoperative cone beam CT images
- Resectoscope with an easy to use twist mechanism for improved handling
- In vitro simulation of distribution processes following intramuscular injection
- Adjusting inkjet printhead parameters to deposit drugs into micro-sized reservoirs
- A flexible standalone system with integrated sensor feedback for multi-pad electrode FES of the hand
- Smart control for functional electrical stimulation with optimal pulse intensity
- Tactile display on the remaining hand for unilateral hand amputees
- Effects of sustained electrical stimulation on spasticity assessed by the pendulum test
- An improved tracking framework for ultrasound probe localization in image-guided radiosurgery
- Improvement of a subviral particle tracker by the use of a LAP-Kalman-algorithm
- Learning discriminative classification models for grading anal intraepithelial neoplasia
- Regularization of EIT reconstruction based on multi-scales wavelet transforms
- Assessing MRI susceptibility artefact through an indicator of image distortion
- EyeGuidance – a computer controlled system to guide eye movements
- A framework for feedback-based segmentation of 3D image stacks
- Doppler optical coherence tomography as a promising tool for detecting fluid in the human middle ear
- 3D Local in vivo Environment (LivE) imaging for single cell protein analysis of bone tissue
- Inside-Out access strategy using new trans-vascular catheter approach
- US/MRI fusion with new optical tracking and marker approach for interventional procedures inside the MRI suite
- Impact of different registration methods in MEG source analysis
- 3D segmentation of thyroid ultrasound images using active contours
- Designing a compact MRI motion phantom
- Cerebral cortex classification by conditional random fields applied to intraoperative thermal imaging
- Classification of indirect immunofluorescence images using thresholded local binary count features
- Analysis of muscle fatigue conditions using time-frequency images and GLCM features
- Numerical evaluation of image parameters of ETR-1
- Fabrication of a compliant phantom of the human aortic arch for use in Particle Image Velocimetry (PIV) experimentation
- Effect of the number of electrodes on the reconstructed lung shape in electrical impedance tomography
- Hardware dependencies of GPU-accelerated beamformer performances for microwave breast cancer detection
- Computer assisted assessment of progressing osteoradionecrosis of the jaw for clinical diagnosis and treatment
- Evaluation of reconstruction parameters of electrical impedance tomography on aorta detection during saline bolus injection
- Evaluation of open-source software for the lung segmentation
- Automatic determination of lung features of CF patients in CT scans
- Image analysis of self-organized multicellular patterns
- Effect of key parameters on synthesis of superparamagnetic nanoparticles (SPIONs)
- Radiopacity assessment of neurovascular implants
- Development of a desiccant based dielectric for monitoring humidity conditions in miniaturized hermetic implantable packages
- Development of an artifact-free aneurysm clip
- Enhancing the regeneration of bone defects by alkalizing the peri-implant zone – an in vitro approach
- Rapid prototyping of replica knee implants for in vitro testing
- Protecting ultra- and hyperhydrophilic implant surfaces in dry state from loss of wettability
- Advanced wettability analysis of implant surfaces
- Patient-specific hip prostheses designed by surgeons
- Plasma treatment on novel carbon fiber reinforced PEEK cages to enhance bioactivity
- Wear of a total intervertebral disc prosthesis
- Digital health and digital biomarkers – enabling value chains on health data
- Usability in the lifecycle of medical software development
- Influence of different test gases in a non-destructive 100% quality control system for medical devices
- Device development guided by user satisfaction survey on auricular vagus nerve stimulation
- Empirical assessment of the time course of innovation in biomedical engineering: first results of a comparative approach
- Effect of left atrial hypertrophy on P-wave morphology in a computational model
- Simulation of intracardiac electrograms around acute ablation lesions
- Parametrization of activation based cardiac electrophysiology models using bidomain model simulations
- Assessment of nasal resistance using computational fluid dynamics
- Resistance in a non-linear autoregressive model of pulmonary mechanics
- Inspiratory and expiratory elastance in a non-linear autoregressive model of pulmonary mechanics
- Determination of regional lung function in cystic fibrosis using electrical impedance tomography
- Development of parietal bone surrogates for parietal graft lift training
- Numerical simulation of mechanically stimulated bone remodelling
- Conversion of engineering stresses to Cauchy stresses in tensile and compression tests of thermoplastic polymers
- Numerical examinations of simplified spondylodesis models concerning energy absorption in magnetic resonance imaging
- Principle study on the signal connection at transabdominal fetal pulse oximetry
- Influence of Siluron® insertion on model drug distribution in the simulated vitreous body
- Evaluating different approaches to identify a three parameter gas exchange model
- Effects of fibrosis on the extracellular potential based on 3D reconstructions from histological sections of heart tissue
- From imaging to hemodynamics – how reconstruction kernels influence the blood flow predictions in intracranial aneurysms
- Flow optimised design of a novel point-of-care diagnostic device for the detection of disease specific biomarkers
- Improved FPGA controlled artificial vascular system for plethysmographic measurements
- Minimally spaced electrode positions for multi-functional chest sensors: ECG and respiratory signal estimation
- Automated detection of alveolar arches for nasoalveolar molding in cleft lip and palate treatment
- Control scheme selection in human-machine- interfaces by analysis of activity signals
- Event-based sampling for reducing communication load in realtime human motion analysis by wireless inertial sensor networks
- Automatic pairing of inertial sensors to lower limb segments – a plug-and-play approach
- Contactless respiratory monitoring system for magnetic resonance imaging applications using a laser range sensor
- Interactive monitoring system for visual respiratory biofeedback
- Development of a low-cost senor based aid for visually impaired people
- Patient assistive system for the shoulder joint
- A passive beating heart setup for interventional cardiology training

