Abstract
C18H20N2O2, triclinic, P1̄ (no. 2), a = 9.9301(5) Å, b = 10.6446(5) Å, c = 15.2690(8) Å, α = 77.743(2)°, β = 87.460(2)°, γ = 89.139(2)°, V = 1575.60(14) Å3, Z = 4, R gt (F) = 0.0465, wR ref (F 2) = 0.1188, T = 150 K.
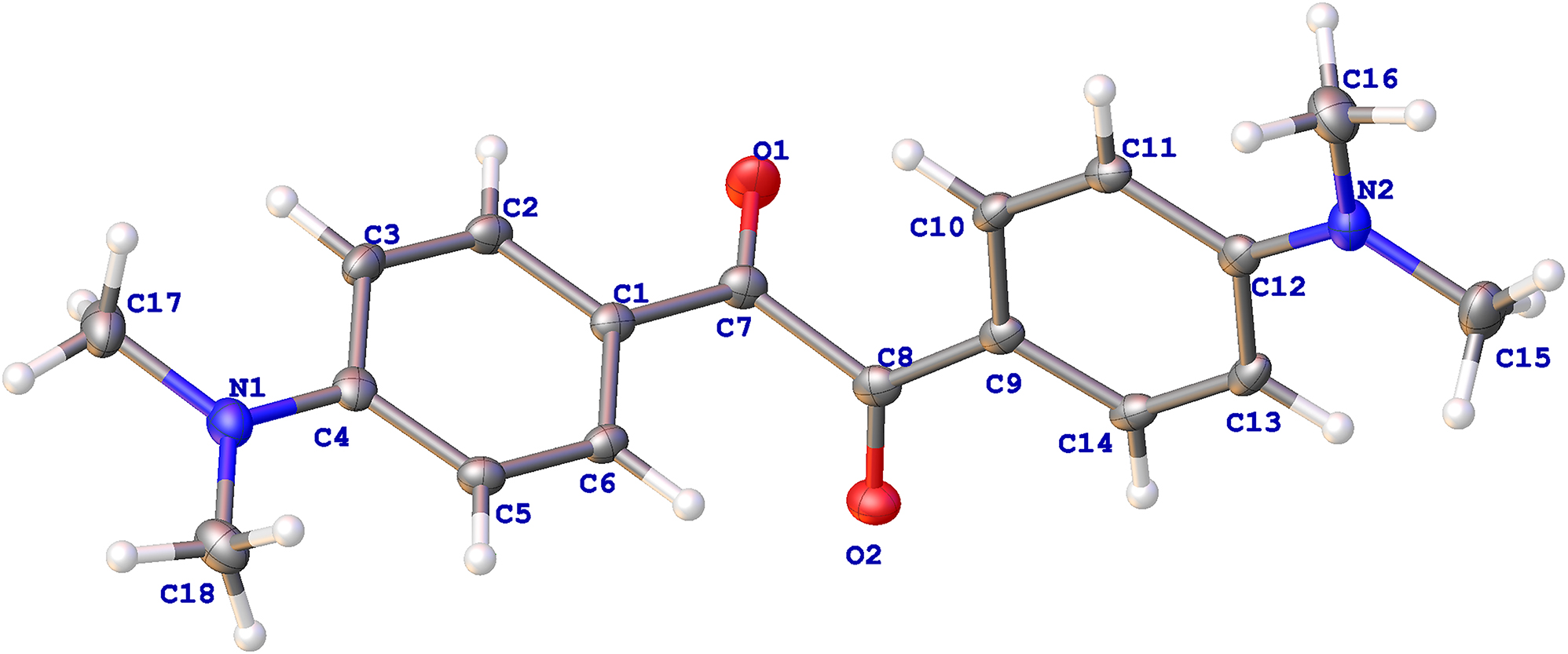
A part of the molecular structure is shown in the figure. Table 1 contains crystallographic data and Table 2 contains the list of the atoms including atomic coordinates and displacement parameters.
Data collection and handling.
| Crystal: | Yellow block |
| Size: | 0.15 × 0.12 × 0.11 mm |
| Wavelength: μ: |
Cu Kα radiation (1.54178 Å) 0.66 mm−1 |
| Diffractometer, scan mode: θ max, completeness: |
Bruker, 68.4°, 99 % |
| N(hkl)measured, N(hkl)unique, R int: | 30,030, 5722, 0.041 |
| Criterion for I obs, N(hkl)gt: | I obs > 2σ(I obs), 5146 |
| N(param)refined: | 405 |
| Programs: | Olex2, 1 SHELX 2 , 3 |
Fractional atomic coordinates and isotropic or equivalent isotropic displacement parameters (Å2).
| Atom | x | y | z | U iso*/U eq |
|---|---|---|---|---|
| O1 | 1.23894 (12) | 0.78017 (13) | 0.68555 (8) | 0.0455 (3) |
| O2 | 1.13676 (12) | 0.53098 (11) | 0.81983 (8) | 0.0378 (3) |
| N1 | 0.82578 (15) | 0.61685 (13) | 0.42010 (9) | 0.0366 (3) |
| N2 | 0.83089 (14) | 0.91823 (14) | 1.02825 (9) | 0.0364 (3) |
| C1 | 1.05585 (14) | 0.68721 (14) | 0.62821 (10) | 0.0257 (3) |
| C2 | 1.08659 (15) | 0.74582 (14) | 0.53854 (11) | 0.0292 (3) |
| H2 | 1.162068 | 0.801407 | 0.524804 | 0.035* |
| C3 | 1.01085 (16) | 0.72522 (15) | 0.47009 (10) | 0.0311 (3) |
| H3 | 1.034048 | 0.767286 | 0.410116 | 0.037* |
| C4 | 0.89825 (15) | 0.64186 (14) | 0.48770 (10) | 0.0283 (3) |
| C5 | 0.86580 (15) | 0.58495 (14) | 0.57858 (10) | 0.0281 (3) |
| H5 | 0.789520 | 0.530499 | 0.593051 | 0.034* |
| C6 | 0.94294 (14) | 0.60729 (14) | 0.64611 (10) | 0.0261 (3) |
| H6 | 0.919061 | 0.567511 | 0.706455 | 0.031* |
| C7 | 1.13906 (15) | 0.71170 (15) | 0.69876 (11) | 0.0299 (3) |
| C8 | 1.10577 (14) | 0.64524 (14) | 0.79635 (11) | 0.0283 (3) |
| C9 | 1.04526 (14) | 0.72190 (14) | 0.85620 (10) | 0.0259 (3) |
| C10 | 0.99421 (15) | 0.84651 (14) | 0.82431 (10) | 0.0263 (3) |
| H10 | 1.007460 | 0.885635 | 0.762602 | 0.032* |
| C11 | 0.92575 (15) | 0.91271 (14) | 0.88037 (10) | 0.0277 (3) |
| H11 | 0.891642 | 0.996366 | 0.856863 | 0.033* |
| C12 | 0.90528 (15) | 0.85770 (15) | 0.97304 (10) | 0.0279 (3) |
| C13 | 0.96391 (15) | 0.73503 (15) | 1.00579 (10) | 0.0293 (3) |
| H13 | 0.957029 | 0.697806 | 1.068120 | 0.035* |
| C14 | 1.03008 (14) | 0.67011 (14) | 0.94847 (10) | 0.0272 (3) |
| H14 | 1.066853 | 0.587502 | 0.971818 | 0.033* |
| C15 | 0.8113 (2) | 0.8599 (2) | 1.12282 (12) | 0.0466 (5) |
| H15A | 0.768948 | 0.775654 | 1.129384 | 0.070* |
| H15B | 0.752894 | 0.915351 | 1.151957 | 0.070* |
| H15C | 0.898666 | 0.849356 | 1.150966 | 0.070* |
| C16 | 0.7570 (2) | 1.03634 (18) | 0.99356 (13) | 0.0454 (4) |
| H16A | 0.820900 | 1.105342 | 0.968744 | 0.068* |
| H16B | 0.701725 | 1.060398 | 1.042274 | 0.068* |
| H16C | 0.698649 | 1.022699 | 0.946350 | 0.068* |
| C17 | 0.8564 (2) | 0.68052 (18) | 0.32760 (12) | 0.0444 (4) |
| H17A | 0.830981 | 0.771508 | 0.318866 | 0.067* |
| H17B | 0.805708 | 0.639827 | 0.287595 | 0.067* |
| H17C | 0.953195 | 0.673190 | 0.313923 | 0.067* |
| C18 | 0.70707 (18) | 0.53593 (17) | 0.43806 (13) | 0.0418 (4) |
| H18A | 0.731355 | 0.451534 | 0.473840 | 0.063* |
| H18B | 0.671490 | 0.525446 | 0.381203 | 0.063* |
| H18C | 0.638236 | 0.576177 | 0.471258 | 0.063* |
| O3 | 0.79844 (11) | 0.17197 (11) | 0.74342 (8) | 0.0392 (3) |
| O4 | 0.52753 (13) | 0.03587 (11) | 0.81602 (8) | 0.0415 (3) |
| N3 | 0.35184 (13) | 0.15516 (13) | 0.40725 (9) | 0.0333 (3) |
| N4 | 0.53808 (14) | 0.63604 (13) | 0.90466 (10) | 0.0350 (3) |
| C19 | 0.40304 (15) | 0.14698 (14) | 0.48922 (10) | 0.0276 (3) |
| C20 | 0.51740 (16) | 0.21841 (15) | 0.50091 (10) | 0.0299 (3) |
| H20 | 0.559755 | 0.273459 | 0.450372 | 0.036* |
| C21 | 0.56813 (15) | 0.20930 (14) | 0.58439 (10) | 0.0287 (3) |
| H21 | 0.643536 | 0.260126 | 0.590568 | 0.034* |
| C22 | 0.51133 (15) | 0.12686 (14) | 0.66060 (10) | 0.0266 (3) |
| C23 | 0.34462 (16) | 0.06484 (14) | 0.56680 (10) | 0.0289 (3) |
| H23 | 0.267089 | 0.016180 | 0.561565 | 0.035* |
| C24 | 0.39855 (16) | 0.05486 (14) | 0.64904 (10) | 0.0281 (3) |
| H24 | 0.358647 | −0.002221 | 0.699566 | 0.034* |
| C25 | 0.56669 (16) | 0.11399 (14) | 0.74874 (10) | 0.0300 (3) |
| C26 | 0.68209 (15) | 0.20315 (15) | 0.76078 (10) | 0.0294 (3) |
| C27 | 0.64576 (15) | 0.31497 (14) | 0.79775 (10) | 0.0263 (3) |
| C28 | 0.74333 (15) | 0.38696 (15) | 0.82865 (10) | 0.0289 (3) |
| H28 | 0.835446 | 0.362410 | 0.825047 | 0.035* |
| C29 | 0.70920 (15) | 0.49209 (15) | 0.86399 (10) | 0.0292 (3) |
| H29 | 0.777804 | 0.538060 | 0.885003 | 0.035* |
| C30 | 0.57329 (15) | 0.53297 (14) | 0.86958 (10) | 0.0264 (3) |
| C31 | 0.47490 (15) | 0.46142 (14) | 0.83694 (10) | 0.0279 (3) |
| H31 | 0.382894 | 0.486925 | 0.838567 | 0.033* |
| C32 | 0.51113 (15) | 0.35578 (14) | 0.80303 (10) | 0.0275 (3) |
| H32 | 0.442994 | 0.308762 | 0.782479 | 0.033* |
| C33 | 0.39906 (18) | 0.68030 (18) | 0.90801 (14) | 0.0425 (4) |
| H33A | 0.373398 | 0.722801 | 0.847612 | 0.064* |
| H33B | 0.390178 | 0.741261 | 0.947868 | 0.064* |
| H33C | 0.339923 | 0.606659 | 0.930819 | 0.064* |
| C34 | 0.63866 (19) | 0.71408 (18) | 0.93383 (13) | 0.0443 (4) |
| H34A | 0.688265 | 0.661171 | 0.982591 | 0.067* |
| H34B | 0.594076 | 0.785391 | 0.955158 | 0.067* |
| H34C | 0.701534 | 0.748360 | 0.883417 | 0.067* |
| C35 | 0.41232 (19) | 0.23843 (19) | 0.32758 (11) | 0.0439 (4) |
| H35A | 0.401954 | 0.328365 | 0.332598 | 0.066* |
| H35B | 0.367340 | 0.225435 | 0.274370 | 0.066* |
| H35C | 0.508362 | 0.217546 | 0.322140 | 0.066* |
| C36 | 0.23161 (17) | 0.08491 (17) | 0.39588 (12) | 0.0368 (4) |
| H36A | 0.248203 | −0.007577 | 0.415473 | 0.055* |
| H36B | 0.209345 | 0.104323 | 0.332515 | 0.055* |
| H36C | 0.156266 | 0.110661 | 0.431994 | 0.055* |
1 Source of material
1,2-bis(4-(dimethylamino)phenyl)ethane-1,2-dione was synthesized according to the classical procedure. 1 The crystal for X-ray data collection was obtained by recrystallization from N, N-dimethylformamide.
2 Experimental details
Using Olex2, the structure was solved with the SHELXS structure solution program using Direct Methods and refined with the SHELXL refinement package. The C-bound H atoms were geometrically placed (C–H = 0.95–0.98 Å) and refined as riding with Uiso(H) = 1.2–1.5 Ueq(C). 2 , 3 , 4
3 Comment
Benzil is one of the important building blocks, with two adjacent carbonyl groups, which can be easily transformed into various heterocycles. These heterocycles and benzil derivatives are widely used in pharmaceuticals, polymer, and material chemistry. Such as biologically active compounds, 5 fluorescence probes, 6 dyes, 7 sensitizers in organic photovoltaics, 8 as well as organic nonlinear optical materials. 9 Benzils can be obtained through several synthetic methods, for instance, Friedel–Crafts acylation, 10 oxidation of benzoins, 11 oxidation of diarylalkenes, 12 oxidation of diarylalkynes, 13 etc. In benzils, some crystal structures of alkoxy group, halogenated or nitro group substitution on phenyl ring have been reported. 9 , 14 , 15
The crystal structure of 1,2-bis(4-(dimethylamino)phenyl) ethane-1,2-dione displays approximate bond lengths and angles compared to the similar structure. 8 The torsion angle of O(1)–C(7)–C(8)–O(2) is −101.53(18)∘ indicating that there is no conjugated π-bond between the two carbonyl groups. The torsion angle of C(2)–C(1)–C(7)–O(1) is −1.4(2)∘, exhibiting the carbonyl almost coplanar with the associated aromatic ring. The torsion angles of O(2)–C(8)–C(9)–C(14) and C(1)–C(7)–C(8)–C(9) are 5.9(2)∘ and −106.40(16)∘, respectively. The dimethylamino segments are approximately coplanar with the corresponding associated aromatic rings due to the p-π conjugation.
Acknowledgments
This work was supported by the National Natural Science Foundation of China (22172069), the Undergraduate Training Program for Innovative and Entrepreneurship in University (Grant No. 202310417006) and Research Start–Up Funding in University.
-
Author contributions: All the authors have accepted responsibility for the entire content of this submitted manuscript and approved submission.
-
Competing interests: The authors declare no conflicts of interest regarding this article.
-
Research funding: National Natural Science Foundation of China (22172069), the Undergraduate Training Program for Innovative and Entrepreneurship in University (Grant No. 202310417006) and Research Start–Up Funding in University.
References
1. Tuzun, C.; Ogliaruso, M.; Becker, E. I. 4, 4′-bis(dimethylamino)benzil. Org. Synth. 1973, 5, 111.Suche in Google Scholar
2. Dolomanov, O. V.; Bourhis, L. J.; Gildea, R. J.; Howard, J. A. K.; Puschmann, H. OLEX2: a Complete Structure Solution, Refinement and Analysis Program. J. Appl. Cryst. 2009, 42, 339–341; https://doi.org/10.1107/s0021889808042726.Suche in Google Scholar
3. Sheldrick, G. M. A Short History of SHELX. Acta Crystallogr. 2008, A64, 112–122; https://doi.org/10.1107/s0108767307043930.Suche in Google Scholar PubMed
4. Sheldrick, G. M. Crystal Structure Refinement with SHELXL. Acta Crystallogr. 2015, C71, 3–8; https://doi.org/10.1107/s2053229614024218.Suche in Google Scholar PubMed PubMed Central
5. Patel, A.; Sharp, S. Y.; Hall, K.; Lewis, W.; Stevens, M. F. G.; Workman, P.; Moody, C. J. Fused Imidazoles as Potential Chemical Scaffolds for Inhibition of Heat Shock Protein 70 and Induction of Apoptosis. Synthesis and Biological Evaluation of Phenanthro [9, 10-d] Imidazoles and Imidazo [4, 5-f] [1, 10] Phenanthrolines. Org. Biomol. Chem. 2016, 14, 3889–3905; https://doi.org/10.1039/c6ob00471g.Suche in Google Scholar PubMed
6. Jakku, R.; Reddy, G.; Trivedi, R.; Lingamallu, G.; Bhargava, S. K. Synthesis, Photophysical, Electrochemical, Theoretical and Self-Assembly Investigation of Tetraphenylethene Tethered Arylimidazoles. J. Photochem. Photobiol. A 2024, 455, 115788; https://doi.org/10.1016/j.jphotochem.2024.115788.Suche in Google Scholar
7. Liu, Z.; Li, C.; Chen, C.; Wang, T.; He, L.; Nan, X. Y.; Li, L.; Qian, Q.; Bai, P. L. The Danger of Dichloromethane in the Synthesis and Application of Imidazole-Based Dyes. Dyes Pigments 2022, 204, 110404; https://doi.org/10.1016/j.dyepig.2022.110404.Suche in Google Scholar
8. Lee, S. H.; Paredes, M. S. V.; Forster, P. M.; Lee, D. C. Side Group Dependent Room Temperature Crystallization-Induced Phosphorescence of Benzil Based All Organic Phosphors. RSC Adv. 2024, 14, 6285–6291; https://doi.org/10.1039/d4ra00816b.Suche in Google Scholar PubMed PubMed Central
9. Patel, A.; Bures, F.; Ludwig, M.; Kulhanek, J.; Pytela, O.; Ruzicka, A. Novel Charge-Transfer Chromophores Featuring Imidazole as π-Linkage. Heterocycles 2009, 78, 999–1013; https://doi.org/10.3987/com-08-11609.Suche in Google Scholar
10. Mohr, B.; Enkelmann, V.; Wegner, G. Synthesis of Alkyl-And Alkoxy-Substituted Benzils and Oxidative Coupling to Tetraalkoxyphenanthrene-9, 10-diones. J. Org. Chem. 1994, 59, 635–638; https://doi.org/10.1021/jo00082a022.Suche in Google Scholar
11. Huang, L. H.; Wang, Q.; Ma, Y. C.; Lou, J. D.; Zhang, C. H. Oxidation of Benzoins to Benzils with Chromium Trioxide under Viscous Conditions. Synth. Commun. 2011, 41, 1659–1663; https://doi.org/10.1080/00397911.2010.492071.Suche in Google Scholar
12. Zeng, X. H.; Miao, C. X.; Wang, S. F.; Xia, C. G.; Sun, W. Facile and Highly Chemoselective Synthesis of Benzil Derivatives via Oxidation of Stilbenes in an I2–H2o System. RSC Adv. 2013, 3, 9666–9669; https://doi.org/10.1039/c3ra41489b.Suche in Google Scholar
13. Jiang, S.; Li, Y.; Luo, X.; Huang, G. L.; Shao, Y.; Li, D. L.; Li, B. NH4 I/EtOCSSK Promoted Synthesis of Substituted Benzils from Diphenylacetylene Derivatives. Tetrahedron Lett. 2018, 59, 3249–3252; https://doi.org/10.1016/j.tetlet.2018.06.053.Suche in Google Scholar
14. Cannon, J. R.; Patrick, V. A.; White, A. H. Color and Conformation of Some Derivatives of Benzil in the Solid–State: Crystal–Structures of 2, 2′-Dimethoxy-4, 4′-Dimethoxy-4, 4′-Diethoxy- and 4, 4′-Dibenzyloxy-Benzil. Aust. J. Chem. 1989, 42, 1631–1645; https://doi.org/10.1071/ch9891631.Suche in Google Scholar
15. Staples, R. J. Crystal Structure of Bis (5-Bromo-2-Hydroxyphenyl) Ethanedione, C4H8Br2O4. Z. Kristallogr. N. Cryst. Struct. 2007, 222, 267–268; https://doi.org/10.1524/ncrs.2007.0113.Suche in Google Scholar
© 2024 the author(s), published by De Gruyter, Berlin/Boston
This work is licensed under the Creative Commons Attribution 4.0 International License.
Artikel in diesem Heft
- Frontmatter
- New Crystal Structures
- Crystal structure of 3-nitrophenol-2,1,3-benzoselenadiazole (1/1), C12H9N3O3Se
- Crystal structure of diaqua-(hydroxido)-{μ-[2-(hydroxy)-5-[(4-nitrophenyl)diazenyl]benzoato]}-{2-hydroxy-5-[(4-nitrophenyl)diazenyl]benzoato}-(1,10-phenanthroline)-diterbium hydrate, C38H27.4N8O12.2Tb
- Crystal structure of poly[bis(μ3-3-fluoro-4-(1H-1,2,4-triazol-1-yl)benzoato-κ3 O:O′:N)cadmium(II)] – dimethylformamide (1/1), C21H17CdF2N7O5
- The crystal structure of 2-amino-N-(pyridin-2-yl)benzamide, C12H11N3O
- The crystal structure of 2,3-di(pyridin-2-yl)-2,3-dihydroquinazolin-4(1H)-one, C18H14N4O
- Crystal structure of 2-chloro-4-fluorobenzyl (R)-2-(6-methoxynaphthalen-2-yl)propanoate, C21H18ClFO3
- Crystal structure of [1-(4-carboxyphenyl)-4-oxo-1,4-dihydropyridazine-3-carboxylic acid]-(methylsulfinyl)methane, C15H16N2O6S
- The crystal structure of 2-ethyl-1,1-dimethyl-1H-benzo[e]indole, C16H17N
- The crystal structure of (Z)-5-amino-N ′-hydroxy-1H-pyrazole-4-carboximidamide, C4H7N5O
- The crystal structure of 2,2,5-trimethyl-3-(4-(4-(5-phenyl-4,5-dihydroisoxazol-3-yl)thiazol-2-yl)phenyl)imidazolidin-4-one, C24H24N4O2S
- The crystal structure of tetrakis(μ2-acetato-κ2 O:O′)-bis[(4′-phenyl-4,2′:6′,4″-terpyridine-κ1 N)dicopper(II)], C25H21CuN3O4
- Crystal structure of poly(3-thiophenecarboxylato-κ 3 O,O′:O′)-(methanol-κO)cadmium(II), C11H10O5S2Cd
- The crystal structure of dichloridobis[4′-(p-methoxylphenyl)-4,2′:6′,4″-terpyridine-κN] zinc(II), C44H34Cl2N6O2Zn
- The crystal structure of 1-(2-carboxyethyl)-1H-imidazole 3-oxide
- Crystal structure of 1,1′,1″-(nitrilotris(ethane-2,1-diyl))tris(3-(4-(((E)-pyridin-2-ylmethylene)amino)phenyl)urea), C45H47N13O4
- Crystal structure of a (E)-4-bromo-N-(4-(diethylamino)-2-hydroxybenzylidene) benzenaminium acetate ─ 4-bromoaniline (1/1)
- Crystal structure of 2,2′-(iminobis(methylene))bis(benzimidazolium) bis(p-toluenesulfonate), C30H31N5O6S2
- The crystal structure of alogliptinium meta-chlorobenzoate
- Crystal structure of 4-bromobenzyl 2-(6-methoxy-naphthalen-2-yl)propanoate, C21H19BrO3
- The hydrated double salt structure of (E)-4-(2-benzylidenehydrazine-1-carbonyl)pyridin-1-ium cation with 2-hydroxybenzoate and benzoate anions
- Crystal structure of (R)(R)-5-chloro-3-((S,1E,3E)-3,5-dimethyl-hepta-1,3-dien-1-yl)-7-methyl-6,8-dioxo-2,6,7,8-tetrahydroisoquinolin-7-yl acetate, C21H24ClNO4
- The crystal structure of bis(3-oxo-1,3-diphenylprop-1-en-1-olato-κ 2 O:O′)-bis(1,4-dioxane-κ 1 O)nickel(II), C38H38O8Ni
- Crystal structure of poly[aqua-(pyridine-3-carboxylato-κ1 N)(pyridine-3-carboxylato-κ2 O,O′) cadmium(II)] dihydrate, C12H14N2O7Cd
- The crystal structure of 4-(4-phenyl-5-(((1-(2,4,6-tribromophenyl)-1H-1,2,3-triazol-4-yl)methyl)thio)-4H-1,2,4-triazol-3-yl)pyridine, C22H14Br3N7S
- The crystal structure of N-benzylquinoline-2-carbothioamide, C17H14N2S
- Crystal structure of bis(3-isopropylphenyl)-4,4′-bipyridinium dichloride dihydrate, C28H30N2⋅2Cl⋅2H2O
- The crystal structure of ethyl 2-amino-4-(cyanophenyl)-5-oxo-5,6,7,8-tetrahydro-4H-chromene-3-carboxylate, C19H18N2O4
- Crystal structure of (4R,10S)-6-hydroxy-7-isopropyl-4,10-dimethyl-1,2,3,5-hexahydro-6,10-epoxyazulen-9-one, C15H22O3
- The crystal structure of (E)-(2-(2-hydroxy-3-methoxybenzylidene)aminophenyl)arsonic acid, C14H14AsNO5
- The crystal structure of poly[(μ 2-2-aminoisophthalato-κ4O,O′:O″:O″′)-(N-methylpyrrolidone κ1O)-dioxido-uranium(VI)], C13H14N2O7U
- The crystal structure of the co-crystal isonicotinamide · terephthalic acid, C8H6O4·2(C6H6N2O)
- The crystal structure of (E)-1-phenyl-3-(p-tolylthio)but-2-en-1-one, C17H16OS
- The crystal structure of 4,5-bis((Z)-chloro(hydroxyimino)methyl)-1H-imidazol-3-ium chloride monohydrate
- The crystal structure of 1,2-bis(4-(dimethylamino)phenyl)ethane-1,2-dione. C18H20N2O2
- Crystal structure of 2-chloro-4-fluorobenzyl 2-acetoxybenzoate, C16H12ClFO4
- Crystal structure of methyl 1-phenyl-9H-pyrido[3,4-b]indole-3-carboxylate, C19H14N2O2
- Crystal structure of (3-(dimethoxymethyl)-5-methoxy-1H-indol-1-yl)(5-fluoro-2-iodophenyl)methanone, C19H17FINO4
- Crystal structure of tetrachlorido-bis(1-[(1H-triazole-1-yl)methyl]-1H-benzotriazole-κ2 N:N′)dicopper, C36H32Cu2N24Cl4
- Crystal structure of 2-(2,3-bis(4-methoxyphenyl)-1H-pyrrolo[2,3-b]quinoxalin-1-yl)anilin, C30H24N4O2
- Crystal structure of 5,7-dihydroxy-2-phenyl-4H-chromen-4-one–N,N-dimethylformamide(1/1), C18H17NO5
- The crystal structure of bis(μ 2-biphenyl-2,2′-dicarboxylato)-diaqua-bis(nitrato)-bis(2,2′:6′,2′′-terpyridine)dineodymium(III), C46H32I2N8Nd2O16
- Crystal structure of (Z)-4-amino-N ′-((4-chlorophenyl)(phenyl)methylene)benzohydrazide, C20H16ClN3O
- Crystal structure of (E)-6,8-dimethoxy-4-(4-morpholinobenzylidene)-3,4-dihydro-1-benzoxepin-5(2H)-one, C23H25NO5
- Crystal structure of (R)-2-((3-(3-aminopiperidin-1-yl)-6-methyl-5-oxo-1,2,4-triazin-4(5H)-yl) methyl)-4-fluorobenzonitrile benzoate monohydrate, C24H27FN6O4
- The crystal structure of [triaqua-(8-carboxymethoxy-quinoline-2-carboxylato-κ 3 N,O,O)copper(II)]monohydrate, C12H15NO9Cu
- Crystal structure of (((4-chlorophenyl)sulfonyl)glycinato-κ 2 N,O)bis(1,10-phenanthroline-κ 2 N,N′)cobalt(II) tetrahydrate, C32H30ClCoN5O8S
- Crystal structure of (((3-nitrophenyl)sulfonyl)-β-alaninato-κO)bis(2,2′-bipyridine-κ 2 N, N′)copper(II) 3-nitrobenzenesulfonate, C35H29CuN7O11S2
- Crystal structure of 3-phenoxybenzyl 2-(6-methoxynaphthalen-2-yl)propanoate, C27H24O4
- 6-(2′,3′-Dihydroxy-3′-methylbutyl)-7-methoxy-8-(3″-methylbut-2″-en-1″-yl)-2H-chromen-2-one, C20H26O5
- Crystal structure of bromido-(2,2′:6′,2″-terpyridine-4′-onato-κ3N)palladium(II) methanol solvate
- The crystal structure of ethyl 2-amino-(4-nitrophenyl)-5-oxo-5,6,7,8-tetrahydro-4H-chromene-3-carboxylate, C20H22N2O6
- Crystal structure of (1E,3E,5E)-1,6-bis(4-(pentyloxy)phenyl)hexa-1,3,5-triene, C28H36O2
- The crystal structure of tris(2-bromo-4-methylphenyl)amine, C21H18Br3N
- The crystal structure of 3-(2,5-dimethylanilino)-1-(2,5-dimethylphenyl)-4-methyl-1H-pyrrole-2,5-dione, C21H22N2O2
- Crystal structure of dicarbonyl (μ2-indole-2-carboxylato κ2 O:O′)tris(triphenylarsine-κAs)dirhodium(I) acetone solvate, C68H56As3NO5Rh2
- The crystal structure of 4-chloro-2-formylphenyl 4-methylbenzenesulfonate, C14H11ClO4S
- Crystal structure of 4-iodobenzyl 2-(6-methoxynaphthalen-2-yl) propanoate, C21H19IO3
Artikel in diesem Heft
- Frontmatter
- New Crystal Structures
- Crystal structure of 3-nitrophenol-2,1,3-benzoselenadiazole (1/1), C12H9N3O3Se
- Crystal structure of diaqua-(hydroxido)-{μ-[2-(hydroxy)-5-[(4-nitrophenyl)diazenyl]benzoato]}-{2-hydroxy-5-[(4-nitrophenyl)diazenyl]benzoato}-(1,10-phenanthroline)-diterbium hydrate, C38H27.4N8O12.2Tb
- Crystal structure of poly[bis(μ3-3-fluoro-4-(1H-1,2,4-triazol-1-yl)benzoato-κ3 O:O′:N)cadmium(II)] – dimethylformamide (1/1), C21H17CdF2N7O5
- The crystal structure of 2-amino-N-(pyridin-2-yl)benzamide, C12H11N3O
- The crystal structure of 2,3-di(pyridin-2-yl)-2,3-dihydroquinazolin-4(1H)-one, C18H14N4O
- Crystal structure of 2-chloro-4-fluorobenzyl (R)-2-(6-methoxynaphthalen-2-yl)propanoate, C21H18ClFO3
- Crystal structure of [1-(4-carboxyphenyl)-4-oxo-1,4-dihydropyridazine-3-carboxylic acid]-(methylsulfinyl)methane, C15H16N2O6S
- The crystal structure of 2-ethyl-1,1-dimethyl-1H-benzo[e]indole, C16H17N
- The crystal structure of (Z)-5-amino-N ′-hydroxy-1H-pyrazole-4-carboximidamide, C4H7N5O
- The crystal structure of 2,2,5-trimethyl-3-(4-(4-(5-phenyl-4,5-dihydroisoxazol-3-yl)thiazol-2-yl)phenyl)imidazolidin-4-one, C24H24N4O2S
- The crystal structure of tetrakis(μ2-acetato-κ2 O:O′)-bis[(4′-phenyl-4,2′:6′,4″-terpyridine-κ1 N)dicopper(II)], C25H21CuN3O4
- Crystal structure of poly(3-thiophenecarboxylato-κ 3 O,O′:O′)-(methanol-κO)cadmium(II), C11H10O5S2Cd
- The crystal structure of dichloridobis[4′-(p-methoxylphenyl)-4,2′:6′,4″-terpyridine-κN] zinc(II), C44H34Cl2N6O2Zn
- The crystal structure of 1-(2-carboxyethyl)-1H-imidazole 3-oxide
- Crystal structure of 1,1′,1″-(nitrilotris(ethane-2,1-diyl))tris(3-(4-(((E)-pyridin-2-ylmethylene)amino)phenyl)urea), C45H47N13O4
- Crystal structure of a (E)-4-bromo-N-(4-(diethylamino)-2-hydroxybenzylidene) benzenaminium acetate ─ 4-bromoaniline (1/1)
- Crystal structure of 2,2′-(iminobis(methylene))bis(benzimidazolium) bis(p-toluenesulfonate), C30H31N5O6S2
- The crystal structure of alogliptinium meta-chlorobenzoate
- Crystal structure of 4-bromobenzyl 2-(6-methoxy-naphthalen-2-yl)propanoate, C21H19BrO3
- The hydrated double salt structure of (E)-4-(2-benzylidenehydrazine-1-carbonyl)pyridin-1-ium cation with 2-hydroxybenzoate and benzoate anions
- Crystal structure of (R)(R)-5-chloro-3-((S,1E,3E)-3,5-dimethyl-hepta-1,3-dien-1-yl)-7-methyl-6,8-dioxo-2,6,7,8-tetrahydroisoquinolin-7-yl acetate, C21H24ClNO4
- The crystal structure of bis(3-oxo-1,3-diphenylprop-1-en-1-olato-κ 2 O:O′)-bis(1,4-dioxane-κ 1 O)nickel(II), C38H38O8Ni
- Crystal structure of poly[aqua-(pyridine-3-carboxylato-κ1 N)(pyridine-3-carboxylato-κ2 O,O′) cadmium(II)] dihydrate, C12H14N2O7Cd
- The crystal structure of 4-(4-phenyl-5-(((1-(2,4,6-tribromophenyl)-1H-1,2,3-triazol-4-yl)methyl)thio)-4H-1,2,4-triazol-3-yl)pyridine, C22H14Br3N7S
- The crystal structure of N-benzylquinoline-2-carbothioamide, C17H14N2S
- Crystal structure of bis(3-isopropylphenyl)-4,4′-bipyridinium dichloride dihydrate, C28H30N2⋅2Cl⋅2H2O
- The crystal structure of ethyl 2-amino-4-(cyanophenyl)-5-oxo-5,6,7,8-tetrahydro-4H-chromene-3-carboxylate, C19H18N2O4
- Crystal structure of (4R,10S)-6-hydroxy-7-isopropyl-4,10-dimethyl-1,2,3,5-hexahydro-6,10-epoxyazulen-9-one, C15H22O3
- The crystal structure of (E)-(2-(2-hydroxy-3-methoxybenzylidene)aminophenyl)arsonic acid, C14H14AsNO5
- The crystal structure of poly[(μ 2-2-aminoisophthalato-κ4O,O′:O″:O″′)-(N-methylpyrrolidone κ1O)-dioxido-uranium(VI)], C13H14N2O7U
- The crystal structure of the co-crystal isonicotinamide · terephthalic acid, C8H6O4·2(C6H6N2O)
- The crystal structure of (E)-1-phenyl-3-(p-tolylthio)but-2-en-1-one, C17H16OS
- The crystal structure of 4,5-bis((Z)-chloro(hydroxyimino)methyl)-1H-imidazol-3-ium chloride monohydrate
- The crystal structure of 1,2-bis(4-(dimethylamino)phenyl)ethane-1,2-dione. C18H20N2O2
- Crystal structure of 2-chloro-4-fluorobenzyl 2-acetoxybenzoate, C16H12ClFO4
- Crystal structure of methyl 1-phenyl-9H-pyrido[3,4-b]indole-3-carboxylate, C19H14N2O2
- Crystal structure of (3-(dimethoxymethyl)-5-methoxy-1H-indol-1-yl)(5-fluoro-2-iodophenyl)methanone, C19H17FINO4
- Crystal structure of tetrachlorido-bis(1-[(1H-triazole-1-yl)methyl]-1H-benzotriazole-κ2 N:N′)dicopper, C36H32Cu2N24Cl4
- Crystal structure of 2-(2,3-bis(4-methoxyphenyl)-1H-pyrrolo[2,3-b]quinoxalin-1-yl)anilin, C30H24N4O2
- Crystal structure of 5,7-dihydroxy-2-phenyl-4H-chromen-4-one–N,N-dimethylformamide(1/1), C18H17NO5
- The crystal structure of bis(μ 2-biphenyl-2,2′-dicarboxylato)-diaqua-bis(nitrato)-bis(2,2′:6′,2′′-terpyridine)dineodymium(III), C46H32I2N8Nd2O16
- Crystal structure of (Z)-4-amino-N ′-((4-chlorophenyl)(phenyl)methylene)benzohydrazide, C20H16ClN3O
- Crystal structure of (E)-6,8-dimethoxy-4-(4-morpholinobenzylidene)-3,4-dihydro-1-benzoxepin-5(2H)-one, C23H25NO5
- Crystal structure of (R)-2-((3-(3-aminopiperidin-1-yl)-6-methyl-5-oxo-1,2,4-triazin-4(5H)-yl) methyl)-4-fluorobenzonitrile benzoate monohydrate, C24H27FN6O4
- The crystal structure of [triaqua-(8-carboxymethoxy-quinoline-2-carboxylato-κ 3 N,O,O)copper(II)]monohydrate, C12H15NO9Cu
- Crystal structure of (((4-chlorophenyl)sulfonyl)glycinato-κ 2 N,O)bis(1,10-phenanthroline-κ 2 N,N′)cobalt(II) tetrahydrate, C32H30ClCoN5O8S
- Crystal structure of (((3-nitrophenyl)sulfonyl)-β-alaninato-κO)bis(2,2′-bipyridine-κ 2 N, N′)copper(II) 3-nitrobenzenesulfonate, C35H29CuN7O11S2
- Crystal structure of 3-phenoxybenzyl 2-(6-methoxynaphthalen-2-yl)propanoate, C27H24O4
- 6-(2′,3′-Dihydroxy-3′-methylbutyl)-7-methoxy-8-(3″-methylbut-2″-en-1″-yl)-2H-chromen-2-one, C20H26O5
- Crystal structure of bromido-(2,2′:6′,2″-terpyridine-4′-onato-κ3N)palladium(II) methanol solvate
- The crystal structure of ethyl 2-amino-(4-nitrophenyl)-5-oxo-5,6,7,8-tetrahydro-4H-chromene-3-carboxylate, C20H22N2O6
- Crystal structure of (1E,3E,5E)-1,6-bis(4-(pentyloxy)phenyl)hexa-1,3,5-triene, C28H36O2
- The crystal structure of tris(2-bromo-4-methylphenyl)amine, C21H18Br3N
- The crystal structure of 3-(2,5-dimethylanilino)-1-(2,5-dimethylphenyl)-4-methyl-1H-pyrrole-2,5-dione, C21H22N2O2
- Crystal structure of dicarbonyl (μ2-indole-2-carboxylato κ2 O:O′)tris(triphenylarsine-κAs)dirhodium(I) acetone solvate, C68H56As3NO5Rh2
- The crystal structure of 4-chloro-2-formylphenyl 4-methylbenzenesulfonate, C14H11ClO4S
- Crystal structure of 4-iodobenzyl 2-(6-methoxynaphthalen-2-yl) propanoate, C21H19IO3


