Abstract
To investigate the potential applications of bio-based epoxy resins across diverse fields, this study synthesized a bio-based epoxy resin using itaconic acid (EIA) as the precursor material and compared its thermal, mechanical, and electrical properties with those of epoxy acrylate (EA). The findings indicate that the glass transition temperature and the 5% thermal decomposition temperature of the EIA-cured system are higher than those of EA. The breakdown field strength of the EIA-cured system is slightly higher than that of EA (35.58 kV·mm−1), suggesting that EIA exhibits stronger electrical properties compared to EA. Mechanical property tests demonstrate that the tensile strength, elongation at the fracture point, and Shore hardness of the EIA-cured system are superior to those of EA. In conclusion, EIA, serving as a matrix resin, is influenced by cross-linking density and intramolecular ester bonding and exhibits close electrical strength but superior mechanical, thermal, and degradation properties than EA.
1 Introduction
UV curing technology involves the excitation of photo-initiator molecules by ultraviolet radiation, leading to the initiation of cationic or free radical activity, followed by the activation of these active groups. This, in turn, triggers a crosslinking reaction in a liquid system containing unsaturated double bonds or epoxy groups within the compound. This process transforms solvent-free liquid oligomers (including monomers) into crosslinked polymers at room temperature, making it the most rapid and effective method (1). Compared with previous curing technologies, UV curing technology is characterized by the “5E” principles (efficiency, economy, energy, ecology, and enabling) and is often referred to as the “twenty-first-century green industrial technology” (2,3). Furthermore, UV curing can be employed in environments with stringent temperature requirements, expanding the material’s scope of application. Commonly used light-curing resins include urethane acrylate, polyester-type acrylate, and epoxy acrylate (EA). EA resins, known for their exceptional adhesive properties, resistance to yellowing, hardness, and chemical resistance, find extensive use as UV-cured oligomers in corrosion-resistant materials and coatings (4). However, in some fields, the utilization of EA resins is restricted due to their weak mechanical properties, high brittleness, and inadequate resistance to aging caused by light exposure.
In recent years, the continuous increase in crude oil prices has led to consistently high prices for its derivative, propylene. This high cost of acrylic chemical products, primarily derived from propylene, results in substantial consumption of fossilized energy and environmental pollution during the production process. Such practices are no longer aligned with the principles of green chemistry and the need for sustainable development (5). Researchers worldwide have been increasingly emphasizing the substitution of chemical raw materials with environmentally friendly bio-based materials for resin preparation. These bio-based materials encompass a range of options, including tannins, vanillin, rosin, polyols, liquefied biomass, and lignin, among others (6,7,8). In the realm of bio-based epoxy resins, itaconic acid (IA) stands out as a bio-based raw material produced using bio-fermentation technology. It boasts a wide range of applications and cost-effectiveness and has been identified by the US Department of Energy as one of the 12 bio-based platform compounds with substantial development potential (9,10,11). As a dibasic carboxylic acid containing an unsaturated double bond, IA is chemically active. Beyond its inherent polymerization, it can undergo co-polymerization reactions with other olefinic monomers, in addition to various esterification, addition, and polymerization reactions, facilitating the creation of diverse new polymeric materials. In a study by Songqi and Xiaoqing (12), IA and 9,10-dihydro-9-oxa-10-phosphophenanthrene 10-oxide were used as raw materials to synthesize phosphorus-containing bio-based epoxy resin. This resin displayed comparable glass transition temperature and mechanical properties to the diglycidyl ether of the bisphenol A system. It also demonstrated excellent flame-retardant properties, achieving a UL94 V-0 rating in vertical combustion testing, showcasing its potential as a green flame retardant in epoxy resin systems. Guo (13) assessed the electrical properties of IA epoxy resins, focusing on breakdown field strength, dielectric loss angle, and leakage current. While the electrical properties of IA epoxy resin were slightly weaker than the widely used bisphenol A epoxy resin E-51, they did not significantly compromise its insulation properties. Additionally, when analyzing the experimental data using the Weibull distribution, the dispersion of breakdown field strength of IA-modified epoxy resin was found to be slightly lower than that of bisphenol A epoxy resin E-51. Consequently, IA-modified epoxy resin is considered to have significant potential in the field of electrical applications. EA, although widely employed, exhibits low strength and brittleness. Moreover, the escalating “oil crisis” has resulted in high costs associated with acrylic acid production, accompanied by severe environmental consequences.
The utilization of bio-based materials, whether in part or entirely replacing petroleum-based products, represents a promising avenue for enhancing environmental sustainability. In this study, IA was chosen as a substitute for acrylic acid in the formulation of light-curing polymers due to its outstanding performance. The two carboxylic acid groups in IA molecules can engage in a ring-opening reaction with epoxy groups in epoxy resins, expanding the epoxy resin’s chain and enhancing its flexibility. Simultaneously, the carbon–carbon double bond in IA permits UV polymerization under the influence of photo-initiators. This study compared the mechanical properties, thermal stability, and electrical insulation properties of epoxy itaconic esters with those of commercial EAs. It presents a promising method of combining IA-modified epoxy resin with 3D printing technology for the production of structurally complex dielectric components with functional gradients.
2 Experimental
2.1 Materials
IA with a purity of 99.7%; epoxy resin (model E-51), categorized as industrial grade; photo-initiator 1173, with a purity of 97%; thermal initiator methylhexahydrophthalic anhydride (MHHPA), with a purity of 95%; diluent dipropylene glycol diacrylate (DPGDA), with a purity of 99%; catalyst tetrabutylammonium bromide at a purity level of 99%; polymerization blocker tetra-methoxyphenol (MEHQ) with a purity of 99%; epoxy accelerator 1,8-diazabicyclo[5,5,8,8,8,8] (structure shown in Figure 1) with a purity of 99%; polymerization inhibitor MEHQ with a purity of 99%; and epoxy promoter 1,8-diazabicyclo[5,4,0]undec-7-ene (DBU) at 99% purity.

EIA reaction equation.
2.2 Preparation of specimens
In a 250 mL three-necked flask, 30 g of epoxy resin E-51, 0.06 g of polymerization inhibitor (4-methoxyphenyl), and 0.24 g of catalyst (tetrabutylammonium bromide) were combined. The temperature was increased to 90°C, and the mixture was stirred. About 5 g of IA was added to the three-necked flask and allowed to react for 30 min. Subsequently, the temperature was increased to 100°C, and the acid value was measured at 30 min intervals. The reaction continued until the acid value fell below 1 mg KOH·g−1. The reaction equation is depicted in Figure 1 (14).
Bisphenol A-type EA (structural formula shown in Figure 2), light curing agent 1173, and diluent DPGDA were weighed into a beaker and stirred at 60°C under light for 30 min, followed by a 30 min session of ultrasonic shock to eliminate air bubbles in the resin. The mixture was poured into a silicone mold, ensuring a flat and smooth liquid surface, and placed under the illumination of the lamp in a UV curing machine for 50 s. The EA light-cured samples were retrieved for performance comparison. EIA, diluent DPGDA, photo-initiator 1173, thermal initiator MHHPA, and epoxy accelerator DBU were weighed in a beaker and magnetically stirred for 30 min at 60°C, keeping the mixture protected from light. This was followed by a 30 min session of ultrasonic vibration to ensure thorough mixing of EIA, diluent, initiator, and other components while removing air bubbles. The well-mixed resin was poured into silicone molds, maintaining a level and smooth liquid surface. Subsequently, the molds were placed under the illumination of a UV curing machine for 50 s and then transferred to an oven set at 130°C for 5 h. Subsequently, the sample strips were removed. The UV curing machine process, as well as the subsequent oven treatment, are illustrated in Figure 3. Once the molds and sample strips had cooled to room temperature, as the final step in the process, the sample strips were removed.

Structural formula of EA.

Experimental flow chart for the preparation of IA epoxy resin and sample strips.
2.3 Characterization
2.3.1 Structural characterization
Fourier transform infrared (FT-IR): the structure of the prepared samples was analyzed using an FT-IR spectrometer (Nicolet iS20, Thermo Scientific, USA), with a spectral range of 4,000–500 cm⁻¹. Nuclear magnetic resonance (NMR) spectrometry: for NMR analysis, 5.0–10.0 mg of the sample to be tested was accurately weighed and dissolved in 0.5 mL of deuterated chloroform. The test frequency was set at 400 MHz. Gel permeation chromatography (GPC): tetrahydrofuran at a concentration of approximately 5 mg·mL−1 was employed as the test solvent. The system was operated at a pressure of 1.88 MPa, with the column oven and detector temperature set at 40°C and the mobile phase flow rate at 1.0 mL·min−1.
2.3.2 Gel content test
Approximately 0.5–1.0 g of cured resin was taken, and its mass was recorded as “m 1.” Acetone served as the solvent. The cured resin was placed in a Soxhlet extractor and extracted until a constant weight was achieved. After extraction, it was removed and dried in an oven, and its mass, denoted as “m 2,” was measured. The gel content (GC) of the cured resin was calculated using the following formula 1:
where m 1 is the mass of the resin before extraction (g) and m 2 is the mass of the resin after extraction (g).
2.3.3 Dynamic mechanical analysis
Dynamic thermomechanical analysis (DMA) was performed using a DMA-Q800 analyzer. Test conditions: single cantilever beam test mode was selected, the test frequency was 2 Hz, the heating rate was 5°C·min−1, and the test temperature range was 30–200°C.
2.3.4 Thermogravimetric (TGA) analysis
Thermal stability is a critical factor in assessing the suitability of epoxy resins. TGA is a widely accepted and convenient method for evaluating the thermal stability and degradation characteristics of polymer materials. A thermogravimetry/differential thermal analysis (TG/DTA) analyzer was employed to measure thermal weight loss. Test conditions: the specimens were placed within an Al2O3 crucible and subjected to testing in an air atmosphere, with a temperature ramp of 10°C·min−1. The testing temperature range spans from 10°C to 800°C. The TG/DTA analyzer was utilized to determine the specimen temperatures.
2.3.5 Mechanical properties testing
A universal tensile testing machine was employed to assess the mechanical properties of the cured resins. Dumbbell-type sample strips were prepared in accordance with GB/T 1040.2-2006, with sample dimensions of 115 mm × 25 mm × 4 mm. In each group of five specimens tested on the universal tensile material testing machine, the speed was set at 50 mm·min−1.
2.3.6 AC breakdown voltage test
For the dielectric breakdown tests, spherical electrodes were used, following IEC 60243-1-2013 standards. The specimens were subjected to a constant voltage rate of 2 kV·s−1 until material breakdown occurred. To effectively prevent surface flashover caused by voltage rise during the test, square sheet specimens measuring 100 mm × 100 mm × 1 mm were placed in transparent glass containers filled with dimethylsilicon oil. Thirteen valid data values were obtained for each model, and the experimental results were statistically analyzed using a Weibull distribution.
2.3.7 Broadband dielectric spectrum testing
Dielectric properties are a fundamental consideration for dielectric materials. In this study, a broadband dielectric spectrometer was employed to measure the relative permittivity and dielectric loss angle tangent of the specimens at room temperature. The specimens were 10 mm in diameter and 1 mm in thickness, and the test frequency range spanned from 10³ to 10⁶ Hz.
2.3.8 Double bond conversion rate
The double-bond conversion rates of the light-curing coating formulations were determined using a WQF-310 FT-IR spectrometer with an OmicCure S1000 UV point light source. The resin formulations were uniformly applied to potassium bromide salt tablets, with the light intensity of the UV point light source set at 45 mW·cm−² during an irradiation time of 600 s. The resolution of the scan was 4 cm⁻¹. This procedure was repeated four times. The double-bond conversion rate was calculated by measuring the change in the absorption peak area of the coating samples before and after irradiation, as per the following equation:
where A 0 represents the peak area of the carbon–carbon double bond at 810 cm⁻¹ before curing and A t is the peak area after curing. A ra corresponds to the peak area of the carbonyl group in the range 1,770–1,716 cm⁻¹ before curing, whereas A rb denotes the peak area after curing.
2.3.9 Scanning electron microscopy
For the analysis of sample surface micromorphology, a Zeiss Sigma 300 scanning electron microscope was employed. The microscope was operated at an accelerating voltage of 10 kV. Its primary function was to examine the surface morphology of the material post-tensile fracture and assess the fracture characteristics.
3 Results and discussion
3.1 Characterization of itaconic acid epoxy resin (EIA)
3.1.1 Infrared characterization of resins
As illustrated in Figure 4, the absorption peaks at 810 and 1,634 cm−1 in the EA are attributed to C═C stretching vibrations, whereas the absorption peak at 1,508 cm−1 confirms the presence of the benzene ring within bisphenol A EA. For the epoxy resin E51, subsequent to the reaction with IA, a reduction in the intensity of the epoxy group absorption peaks was observed, indicating a decrease in epoxy group content. Additionally, a new carbonyl peak was observed at 1,720 cm1. The broad peak at 3,480 cm−1 corresponds to the hydroxyl group resulting from the reaction between the epoxy group and carboxylic acid to form hydroxyl groups (–OH). Furthermore, a distinctive absorption peak of the carbonyl–carbon double bond was observed at 1,606 cm−1. These findings collectively signify the successful occurrence of a ring-opening reaction between the epoxy resin and IA (15).
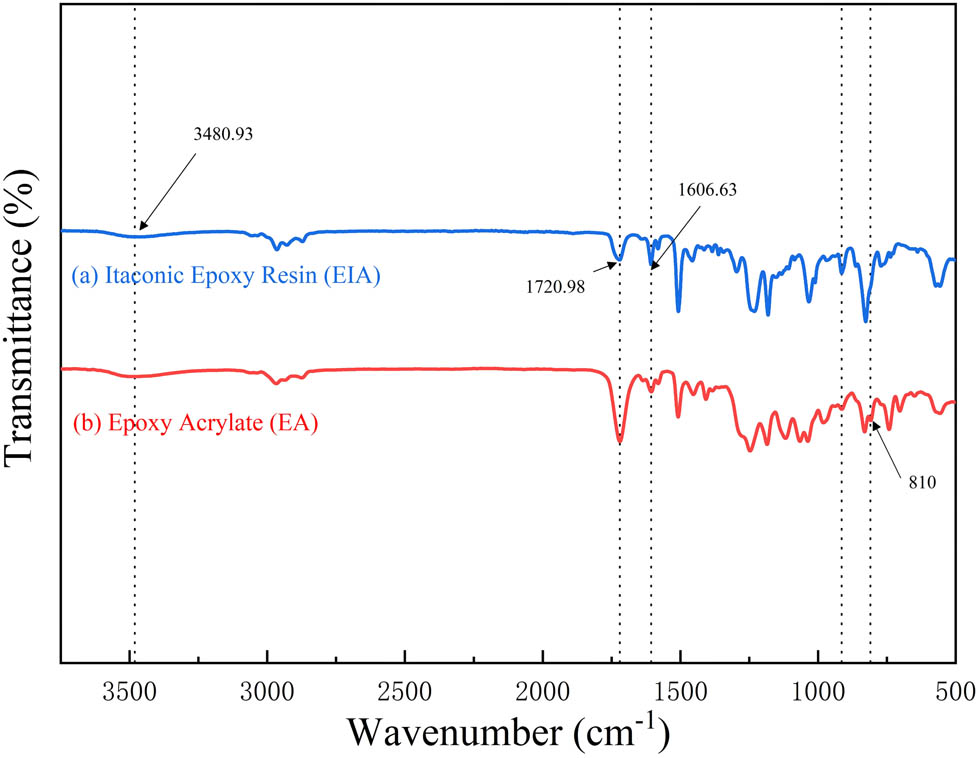
Infrared spectra of EIA and EA.
3.1.2 NMR characterization of resins
As shown in Figure 5(a), the benzene ring in the bisphenol A structure appeared at 6.8 and 7.1 ppm, the protons on the epoxy group appeared at 2.8, 2.9, and 3.3 ppm, and the methyl proton peak on the bisphenol A appeared at 1.6 ppm. When the epoxy resin was reacted with IA, the 1H NMR spectrum of EIA showed that the C═C bond appeared at 6.35 and 5.70 ppm, which indicated that the C═C double bond was successfully attached in the epoxy resin, suggesting the successful synthesis of the EIA epoxy resin. Figure 5(b) shows the 1H NMR spectrum of EA, which can be seen as the active hydrogen on the hydroxyl group appeared at 1.63 ppm, H on the bisphenol A benzene ring appeared at 6.80 and 7.12 ppm, and CH 2 ═CH– appeared at 6.47 and 5.89 ppm, while CH2═CH – appeared at 6.17 ppm.

NMR hydrogen spectra of (a) EIA and (b) EA.
3.1.3 Gel chromatogram analysis
The relative molecular mass (Mn) and molecular weight distribution index (PDI) were measured using GPC. As depicted in Figure 6, EA exhibited an average relative molecular mass of 2,301 with a molecular weight distribution index of 1.362. Upon subjecting epoxy resin E51 to a chain expansion reaction with IA, a notable increase in both the relative molecular mass (Mn) and molecular weight distribution index (PDI) was observed, reaching 3,447 and 1.171, respectively. These results confirm that IA effectively expanded the chain of epoxy resin E51 (16).

Gel permeation chromatograms of EIA and EA.
The EA showed an average relative molecular mass of 2,301 and molecular weight distribution index of 1.362, and the relative molecular mass (Mn) and molecular weight distribution index (PDI) of the EIA were 3,447 and 1.171, respectively. The molecular weight of EIA was significant, with a narrow distribution.
3.2 Characterization of the degree of curability
3.2.1 Double bond conversion rate
The double-bond conversion rate serves as a measure of the curing rate and extent of curing for the cured samples. Figure 7 illustrates the relationship between the double-bond conversion rate of EIA and the irradiation time. During the UV curing of both EIA and EA, a specific quantity of DPGDA reactive diluent was incorporated. To assess the double-bond conversion rate and extent of conversion, real-time infrared spectroscopy was employed to monitor changes in peak areas at the carbon–carbon double bond positions of EIA (1,606 cm−1) and EA (1,634 cm−1). The analysis of the variation curve of the double-bond conversion rate for EIA with UV irradiation time shows that the double-bond conversion rate of the EIA system reached 34.70% within 10 s. However, the final double-bond conversion rate remained relatively low due to the presence of fewer reactive double bonds in EIA, particularly within the structure of IA. In contrast, the overall double-bond conversion of EA peaked at 99.98% within the same 10 s timeframe. This high overall double-bond conversion can be attributed to the positioning of the double bonds at the two ends of the EA structure, rendering them more reactive in the reaction compared to EIA.

Variation of double bond conversion rate of EIA formulation system with irradiation time.
3.2.2 Gel content test
The gel content was employed as a measure of the crosslinking density of the cured material. The gel contents of both EA and EIA after curing are presented in Figure 8. These gel contents are divided into four categories: EA-UV, EIA-UV, EA-UV-heat, and EIA-UV-heat. The former two represent test data following UV curing without additional heat treatment, whereas the latter two correspond to dual curing utilizing both UV and heat. Specifically, the gel content for EA-UV was 66.39%, whereas for EIA-UV, it reached 73.12%. This higher gel content in EIA-UV can be attributed to the greater presence of carbon–carbon double bonds in EIA, stemming from UV exposure. Following heat curing, the gel content for EIA-UV-heat significantly rose to 87.78%, surpassing that of EA. This substantial increase can be primarily attributed to the ring-opening curing of the epoxy groups within EIA during heat treatment, which effectively enhanced the crosslinking density of the cured material.

Gel content of EIA and EA after UV curing and UV/heat curing processes.
3.3 Mechanical property analysis of resins
The mechanical properties of the cured samples were evaluated by tensile tests and a Shore hardness tester. Table 1 provides a comparison between the tensile strength, elongation at break, and Shore hardness of EIA and EA. As observed in the table, for the UV/heat-cured samples, EIA exhibits superior tensile strength, Shore hardness, and elongation at break compared to E51 and EA. This enhancement in mechanical properties can be attributed to the presence of additional double bonds in EIA, resulting in a higher cross-linking density achieved through UV/heat curing. This observation aligns with the results obtained in the gel content test.
Mechanical properties of resins
| Name | Pencil hardness (H) | Tensile strength (MPa) | Elongation at break (%) | Shore hardness (HA) |
|---|---|---|---|---|
| EIA | 2H | 42 | 14 | 98 |
| EA | 3H | 21 | 14 | 74 |
3.4 Analysis of thermal properties of resins
3.4.1 Dynamic mechanical analysis
The crosslink density (ν e) and glass transition temperature (T g) of the cured systems were determined through DMA. Figures 9 and 10 depict the DMA curves for the EIA and EA curing systems, respectively. The crosslinking density (ν e) was calculated using the formula ν e = E′/3RT, yielding values of 19,710.72 and 8,287.79 mol·m−³ for EIA and EA, respectively. Notably, the network in EIA exhibits a significantly higher E at T g + 40°C, and ρ is notably greater compared to the EA system. The figure also reveals that the glass transition temperatures for EIA and EA are 83°C and 70°C, respectively. This discrepancy can be attributed to the fact that the glass transition temperature of the epoxy network is determined by its crosslinking density and the chemical structure of the chain segments. A higher crosslink density or more rigid chain segments result in a higher T g value. Moreover, EIA boasts a greater molecular weight than EA, leading to more molecular entanglement points, stronger intermolecular forces, and consequently higher T g values.

DMA curves of EIA cured system.

DMA curves of EA curing system.
3.4.2 TGA of resins
The thermal stability of a resin plays a pivotal role in evaluating its operational durability. As evident from Figure 11, the initial decomposition temperatures for a 5% weight loss of EIA and EA were measured at 323.86°C and 273.61°C, respectively. Subsequently, the temperatures at which the maximum decomposition rates occurred were recorded at 430.53°C for EIA and 378.12°C for EA. The residual mass fractions were found to be 7.6% and 3.9%, respectively, indicating the degradation of the polymer backbone structure throughout the process. Notably, while the curing network of EA disintegrated earlier and more thoroughly than that of EIA, the overall distinction remains relatively modest. This can be attributed to the presence of a substantial number of ester bonds in the molecules of EIA and EA polymers, which are susceptible to thermal cleavage. Additionally, the chain structure of EIA, to some extent, constrains its potential application scope. Therefore, its heat resistance must be enhanced through subsequent structural modifications.

Comparison of TG for EIA and EA.
3.5 Analysis of electrical properties of resins
3.5.1 Breakdown field strength analysis
AC breakdown voltage tests were performed on the EIA and EA curing systems. The Weibull distribution curves representing their breakdown field strengths are presented in Figure 12. For EIA and EA, the AC breakdown field strengths, with a breakdown probability of 63.2%, were measured at 40.15 and 35.58 kV·mm−1, respectively. Notably, these breakdown field strengths in EIA were higher than those in EA. This difference can be attributed to several factors. First, the lower cross-linking density of the curing resin in EA leads to the rapid expansion of voids within the molecules under voltage, ultimately resulting in material breakdown. Furthermore, the lower degree of cross-linking and the expansive nature of the ester group within EA contribute to its heightened susceptibility to water adsorption (17). Water adsorption on insulating materials can induce significant damage to their insulation properties as polar water molecules tend to conduct electricity.

Breakdown field strength Weibull distribution for EIA and EA.
3.5.2 Analysis of dielectric properties
The AC dielectric spectral curves of the EIA and EA curing systems are depicted in Figure 13. The figure shows that the fundamental component of the dielectric constant in both the EIA and EA curing systems gradually diminishes as the frequency rises. The difference between the dielectric constants of the EA and EIA curing systems is not substantial. In these systems, electronic polarization and atomic polarization contribute minimally compared to dipole polarization, which has a short relaxation time and primarily governs the high-frequency band dielectric constant of the polymer. The complex, non-uniform arrangement of high-frequency dipoles results in a reduction in permittivity. Both EIA and EA polymers contain molecular chains with O–H (hydroxyl), C–O (ether bond), and C–H bonds, alongside multiple polar ester bonds. The permanent dipole moments associated with polar bonds induce potential traps in their vicinity, and the relaxation time constant is determined by the trap depth of these polar bonds. The dielectric loss factor is a fundamental property of insulating materials, characterizing the extent of dielectric loss in a material following the application of an electric field. It stems from two sources: dielectric loss due to leakage current and dielectric loss due to polarization. At low frequencies, various polarization mechanisms have sufficient time to respond, resulting in minimal polarization loss. Consequently, dielectric loss mainly arises from leakage current. However, under high-frequency conditions, the dipoles within the dielectric fail to keep pace with the rapid changes in the electric field frequency, leading to relaxation and an increase in the dielectric loss of the composite. At this stage, the dielectric loss factor is contingent on the alignment of polar chain segments. When polarization loss predominates, the total dipole moment of the molecules drives the dielectric loss peaks observed in both the EIA and EA polymer molecules, owing to the prevalence of numerous ester bonds within their structures (18,19).

(a) Dielectric constant versus frequency for the EIA and EA cured systems. (b) Dielectric loss factor versus frequency for the EIA and EA cured systems.
3.6 Resin contact angle test
The contact angle (θ), which is the angle formed by a liquid droplet on the surface of a solid material, is a crucial parameter for assessing the material’s wetting performance. Two primary methods are employed for measuring the contact angle: the weighing method and the shape image analysis method. In this experiment, distilled water is utilized as the liquid for droplet placement, and the contact angle is determined using the shape image analysis method. A contact angle (θ) of less than 90° suggests that the material is hydrophilic, with smaller angles indicating superior wettability. Conversely, a contact angle (θ) greater than 90° indicates that the material is hydrophobic, meaning that water has difficulty wetting the material’s surface (20). As depicted in Figure 14, EIA exhibits a contact angle of 90.5°, signifying a hydrophobic state, whereas EA exhibits a contact angle of 87.5°, indicating a hydrophilic state.

Contact angles of (a) EIA and (b) EA.
3.7 Microscopic morphology analysis
Figure 15(a1) and (a2) shows the SEM images of the tensile section of the EIA resin after light curing. These images reveal a flush and perpendicular fracture in alignment with the positive stress direction. Cracks have propagated uniformly in the same direction, with minimal voids at the fracture sites, indicative of a characteristic brittle fracture. On the other hand, Figure 15(b1) and (b2) shows the SEM images of the tensile section of the EA resin after light curing. In these images, the crack direction single shallow, linear expansion, fracture is due to tensile stress, belonging to the brittle fracture.

SEM images of sections of (a1), (a2) EIA and (b1), (b2) EA.
4 Conclusions
To enhance the performance of EA and alleviate environmental concerns, a novel light-heat dual-curing epoxy resin, EIA, was synthesized. This was achieved by using the biomolecule IA, which features a double bond and a bisphenol A-type epoxy resin (E51) as raw materials. A comprehensive comparative investigation was conducted to assess the performance of EIA and EA. The cured EIA samples exhibited a higher gel content, attributable to the additional double bonds provided by IA. This increase in double bonds and molecular mass resulted in improved tensile strength, elongation at break, and Shore hardness, which, in turn, were attributed to the higher crosslink density. Thermal analysis results showed that the glass transition temperature of the EIA-cured system surpassed that of EA, measuring 83.17°C and 70.07°C, respectively. This superior heat resistance indicates that EIA exhibits better thermal stability. Furthermore, the breakdown strength of the EA curing system decreased compared to that of EIA, owing to insufficient crosslinking and increased water absorption. The presence of polar solid groups, such as ester bonds, in the molecule led to higher dielectric constants and increased dielectric loss in the EA material, but the overall difference is not that great.
-
Funding information: This work was financially supported by the Key Research and Development Program of Shaanxi, China (2023-YBGY-394), Science and Technology Program of Yulin, China (CXY-2022-160), and the Key Research and Development Plan of Shaanxi Province (2023-YBGY-448).
-
Conflict of interest: Authors state no conflict of interest.
-
Data availability statement: The raw data required to reproduce these findings are available to download from https://data.mendeley.com/datasets/htsp7r3g6f/1. The processed data required to reproduce these findings are available to download from https://data.mendeley.com/datasets/htsp7r3g6f/1.
References
(1) Sangermano M, Razza N, Crivello JV. Cationic UV-curing: technology and applications. Macromol Mater Eng. 2014;299(7):776–90. 10.1002/mame.201300349.Suche in Google Scholar
(2) Wang SY, Zou YQ. Synthesis, characterization, and UV-curing properties of silicon-containing(Meth)acrylate monomers. J Appl Polym Sci. 2013;129(6):3325–32. 10.1002/app.38943.Suche in Google Scholar
(3) Chang CW, Lu KT. Epoxy acrylate UV/PU dual-cured wood coatings. J Appl Polym Sci. 2010;115(4):2197–202. 10.1002/app.31326.Suche in Google Scholar
(4) Shen L, Li Y, Zheng J, Lu M, Wu K. Modified epoxy acrylate resin for photocurable temporary protective coatings. Prog Org Coat. 2015;89:17–8. 10.1016/j.porgcoat.2015.07.022.Suche in Google Scholar
(5) Cai Y, Chen G, Yu Z, Lu J, Fang X. Synthesis of a novel biodegradable curing agent modified by castor oil for polyacrylate adhesive applications. J Appl Polym Sci. 2023;140(12):1–11. 10.1002/app.53648.Suche in Google Scholar
(6) Ding C, Matharu AS. Recent developments on biobased curing agents: a review of their preparation and use. ACS Sustainable Chem Eng. 2014;2(10):2217–36. 10.1021/sc500478f.Suche in Google Scholar
(7) Kumar S, Samal SK, Mohanty S, Nayak SK. Recent development of biobased epoxy resins: a review. Polym-Plast Technol Eng. 2018;57(3):133–5. 10.1080/03602559.2016.1253742.Suche in Google Scholar
(8) Ramon E, Sguazzo C, Moreira PMGP. A review of recent research on bio-based epoxy systems for engineering applications and potentialities in the aviation sector. Aerospace. 2018;5(4):110–44. 10.3390/aerospace5040110.Suche in Google Scholar
(9) Zhang H, Quan L, Gao A, Tong Y, Shi F, Xu L. Thermal analysis and crystal structure of poly(acrylonitrile-coitaconic acid) copolymers synthesized in water. Polymers. 2020;12(1):221. 10.3390/polym12010221.Suche in Google Scholar PubMed PubMed Central
(10) Okabe M, Lies D, Kanamasa S, Park EY. Biotechnological production of itaconic acid and its biosynthesis in Aspergillus terreus. Appl Microbiol Biotechnol. 2009;84(4):597–606. 10.1007/s00253-009-2132-3.Suche in Google Scholar PubMed
(11) Ouyang Q, Cheng L, Wang H, Li K. Mechanism and kinetics of the stabilization reactions of itaconic acid-modified polyacrylonitrile. Polym Degrad Stab. 2008;93(8):1415–21. 10.1016/j.polymdegradstab.2008.05.021.Suche in Google Scholar
(12) Ma S, Liu X, Jiang Y, Fan L, Feng J, Zhu J. Synthesis and properties of phosphorus-containing bio-based epoxy resin from itaconic acid. Sci China Chem. 2014;57(3):379–87. 10.1007/s11426-013-5025-3.Suche in Google Scholar
(13) Guo Y. Study on the electrical properties of itaconic acid-based epoxy resin. J Phys Conf Ser. 2021;1986(1):1–5. 10.1088/1742-6596/1986/1/012007.Suche in Google Scholar
(14) Songqi M, Xiaoqing L. Bio-based epoxy resin from itaconic acid and its thermosets cured with anhydride and comonomers. Green Chem. 2013;15:245–54. 10.1039/c2gc36715g.Suche in Google Scholar
(15) Kumar S, Samal SK, Mohanty S, Nayak SK. Synthesis and characterization of itaconic-based epoxy resins. Polym Adv Technol. 2018;29(1):4–5. 10.1002/pat.4098.Suche in Google Scholar
(16) Pan Z, Chen M, Zeng K, Kang Y. Synthesis and application of cyclotetrasiloxane modified with epoxy resins. Polym Bull. 2021;79(9):1–16. 10.1007/s00289-021-03847-y.Suche in Google Scholar
(17) Aslam F, Li Z, Qu G, Feng Y, Li S, Li S, Mao H. Improvement of DC breakdown strength of the epoxy/POSS nanocomposite by tailoring interfacial electron trap characteristics. Materials. 2021 Mar 8;14(5):5–6. 10.3390/ma14051298.Suche in Google Scholar PubMed PubMed Central
(18) Zhong L, Liu W, Sun Y, Wang F, Chen S, Sun Q, et al. Electrically assisted stereolithography 3D printing of graded permittivity composites for in-situ encapsulation of insulated gate bipolar transistors (IGBTs). Mater Des. 2023;233:3–4. 10.1016/j.matdes.2023.112220.Suche in Google Scholar
(19) Shi L, Bao F, Liu Y, Cui J, Qiao Y, Yu J, et al. Biobased furan-functionalized high-performance poly (aryl ether ketone) with low dielectric constant and low dielectric loss. Chem Eng J Adv. 2023;16:2–7. 10.1016/J.CEJA.2023.100530.Suche in Google Scholar
(20) Pitzanti G, Mohylyuk V, Corduas F, Byrne NM, Coulter JA, Lamprou DA. Urethane dimethacrylate‑based photopolymerizable resins for stereolithography 3D printing: a physicochemical characterisation and biocompatibility evaluation. Drug Delivery Transl Res. 2023;14:177–90. 10.1007/s13346-023-01391-y.Suche in Google Scholar PubMed PubMed Central
© 2024 the author(s), published by De Gruyter
This work is licensed under the Creative Commons Attribution 4.0 International License.
Artikel in diesem Heft
- Research Articles
- Flame-retardant thermoelectric responsive coating based on poly(3,4-ethylenedioxythiphene) modified metal–organic frameworks
- Highly stretchable, durable, and reversibly thermochromic wrapped yarns induced by Joule heating: With an emphasis on parametric study of elastane drafts
- Molecular dynamics simulation and experimental study on the mechanical properties of PET nanocomposites filled with CaCO3, SiO2, and POE-g-GMA
- Multifunctional hydrogel based on silk fibroin/thermosensitive polymers supporting implant biomaterials in osteomyelitis
- Marine antifouling coating based on fluorescent-modified poly(ethylene-co-tetrafluoroethylene) resin
- Preparation and application of profiled luminescent polyester fiber with reversible photochromism materials
- Determination of pesticide residue in soil samples by molecularly imprinted solid-phase extraction method
- The die swell eliminating mechanism of hot air assisted 3D printing of GF/PP and its influence on the product performance
- Rheological behavior of particle-filled polymer suspensions and its influence on surface structure of the coated electrodes
- The effects of property variation on the dripping behaviour of polymers during UL94 test simulated by particle finite element method
- Experimental evaluation on compression-after-impact behavior of perforated sandwich panel comprised of foam core and glass fiber reinforced epoxy hybrid facesheets
- Synthesis, characterization and evaluation of a pH-responsive molecular imprinted polymer for Matrine as an intelligent drug delivery system
- Twist-related parametric optimization of Joule heating-triggered highly stretchable thermochromic wrapped yarns using technique for order preference by similarity to ideal solution
- Comparative analysis of flow factors and crystallinity in conventional extrusion and gas-assisted extrusion
- Simulation approach to study kinetic heterogeneity of gadolinium catalytic system in the 1,4-cis-polyisoprene production
- Properties of kenaf fiber-reinforced polyamide 6 composites
- Cellulose acetate filter rods tuned by surface engineering modification for typical smoke components adsorption
- A blue fluorescent waterborne polyurethane-based Zn(ii) complex with antibacterial activity
- Experimental investigation on damage mechanism of GFRP laminates embedded with/without steel wire mesh under low-velocity-impact and post-impact tensile loading
- Preparation and application research of composites with low vacuum outgassing and excellent electromagnetic sealing performance
- Assessing the recycling potential of thermosetting polymer waste in high-density polyethylene composites for safety helmet applications
- Mesoscale mechanics investigation of multi-component solid propellant systems
- Preparation of HTV silicone rubber with hydrophobic–uvioresistant composite coating and the aging research
- Experimental investigation on tensile behavior of CFRP bolted joints subjected to hydrothermal aging
- Structure and transition behavior of crosslinked poly(2-(2-methoxyethoxy) ethylmethacrylate-co-(ethyleneglycol) methacrylate) gel film on cellulosic-based flat substrate
- Mechanical properties and thermal stability of high-temperature (cooking temperature)-resistant PP/HDPE/POE composites
- Preparation of itaconic acid-modified epoxy resins and comparative study on the properties of it and epoxy acrylates
- Synthesis and properties of novel degradable polyglycolide-based polyurethanes
- Fatigue life prediction method of carbon fiber-reinforced composites
- Thermal, morphological, and structural characterization of starch-based bio-polymers for melt spinnability
- Robust biaxially stretchable polylactic acid films based on the highly oriented chain network and “nano-walls” containing zinc phenylphosphonate and calcium sulfate whisker: Superior mechanical, barrier, and optical properties
- ARGET ATRP of styrene with low catalyst usage in bio-based solvent γ-valerolactone
- New PMMA-InP/ZnS nanohybrid coatings for improving the performance of c-Si photovoltaic cells
- Impacts of the calcinated clay on structure and gamma-ray shielding capacity of epoxy-based composites
- Preparation of cardanol-based curing agent for underwater drainage pipeline repairs
- Preparation of lightweight PBS foams with high ductility and impact toughness by foam injection molding
- Gamma-ray shielding investigation of nano- and microstructures of SnO on polyester resin composites: Experimental and theoretical study
- Experimental study on impact and flexural behaviors of CFRP/aluminum-honeycomb sandwich panel
- Normal-hexane treatment on PET-based waste fiber depolymerization process
- Effect of tannic acid chelating treatment on thermo-oxidative aging property of natural rubber
- Design, synthesis, and characterization of novel copolymer gel particles for water-plugging applications
- Influence of 1,1′-Azobis(cyclohexanezonitrile) on the thermo-oxidative aging performance of diolefin elastomers
- Characteristics of cellulose nanofibril films prepared by liquid- and gas-phase esterification processes
- Investigation on the biaxial stretching deformation mechanism of PA6 film based on finite element method
- Simultaneous effects of temperature and backbone length on static and dynamic properties of high-density polyethylene-1-butene copolymer melt: Equilibrium molecular dynamics approach
- Research on microscopic structure–activity relationship of AP particle–matrix interface in HTPB propellant
- Three-layered films enable efficient passive radiation cooling of buildings
- Electrospun nanofibers membranes of La(OH)3/PAN as a versatile adsorbent for fluoride remediation: Performance and mechanisms
- Preparation and characterization of biodegradable polyester fibers enhanced with antibacterial and antiviral organic composites
- Preparation of hydrophobic silicone rubber composite insulators and the research of anti-aging performance
- Surface modification of sepiolite and its application in one-component silicone potting adhesive
- Study on hydrophobicity and aging characteristics of epoxy resin modified with nano-MgO
- Optimization of baffle’s height in an asymmetric twin-screw extruder using the response surface model
- Effect of surface treatment of nickel-coated graphite on conductive rubber
- Experimental investigation on low-velocity impact and compression after impact behaviors of GFRP laminates with steel mesh reinforced
- Development and characterization of acetylated and acetylated surface-modified tapioca starches as a carrier material for linalool
- Investigation of the compaction density of electromagnetic moulding of poly(ether-ketone-ketone) polymer powder
- Experimental investigation on low-velocity-impact and post-impact-tension behaviors of GFRP T-joints after hydrothermal aging
- The repeated low-velocity impact response and damage accumulation of shape memory alloy hybrid composite laminates
- Exploring a new method for high-performance TPSiV preparation through innovative Si–H/Pt curing system in VSR/TPU blends
- Large-scale production of highly responsive, stretchable, and conductive wrapped yarns for wearable strain sensors
- Preparation of natural raw rubber and silica/NR composites with low generation heat through aqueous silane flocculation
- Molecular dynamics simulation of the interaction between polybutylene terephthalate and A3 during thermal-oxidative aging
- Crashworthiness of GFRP/aluminum hybrid square tubes under quasi-static compression and single/repeated impact
- Review Articles
- Recent advancements in multinuclear early transition metal catalysts for olefin polymerization through cooperative effects
- Impact of ionic liquids on the thermal properties of polymer composites
- Recent progress in properties and application of antibacterial food packaging materials based on polyvinyl alcohol
- Additive manufacturing (3D printing) technologies for fiber-reinforced polymer composite materials: A review on fabrication methods and process parameters
- Rapid Communication
- Design, synthesis, characterization, and adsorption capacities of novel superabsorbent polymers derived from poly (potato starch xanthate-graft-acrylamide)
- Special Issue: Biodegradable and bio-based polymers: Green approaches (Guest Editors: Kumaran Subramanian, A. Wilson Santhosh Kumar, and Venkatajothi Ramarao)
- Development of smart core–shell nanoparticles-based sensors for diagnostics of salivary alpha-amylase in biomedical and forensics
- Thermoplastic-polymer matrix composite of banana/betel nut husk fiber reinforcement: Physico-mechanical properties evaluation
- Special Issue: Electrospun Functional Materials
- Electrospun polyacrylonitrile/regenerated cellulose/citral nanofibers as active food packagings
Artikel in diesem Heft
- Research Articles
- Flame-retardant thermoelectric responsive coating based on poly(3,4-ethylenedioxythiphene) modified metal–organic frameworks
- Highly stretchable, durable, and reversibly thermochromic wrapped yarns induced by Joule heating: With an emphasis on parametric study of elastane drafts
- Molecular dynamics simulation and experimental study on the mechanical properties of PET nanocomposites filled with CaCO3, SiO2, and POE-g-GMA
- Multifunctional hydrogel based on silk fibroin/thermosensitive polymers supporting implant biomaterials in osteomyelitis
- Marine antifouling coating based on fluorescent-modified poly(ethylene-co-tetrafluoroethylene) resin
- Preparation and application of profiled luminescent polyester fiber with reversible photochromism materials
- Determination of pesticide residue in soil samples by molecularly imprinted solid-phase extraction method
- The die swell eliminating mechanism of hot air assisted 3D printing of GF/PP and its influence on the product performance
- Rheological behavior of particle-filled polymer suspensions and its influence on surface structure of the coated electrodes
- The effects of property variation on the dripping behaviour of polymers during UL94 test simulated by particle finite element method
- Experimental evaluation on compression-after-impact behavior of perforated sandwich panel comprised of foam core and glass fiber reinforced epoxy hybrid facesheets
- Synthesis, characterization and evaluation of a pH-responsive molecular imprinted polymer for Matrine as an intelligent drug delivery system
- Twist-related parametric optimization of Joule heating-triggered highly stretchable thermochromic wrapped yarns using technique for order preference by similarity to ideal solution
- Comparative analysis of flow factors and crystallinity in conventional extrusion and gas-assisted extrusion
- Simulation approach to study kinetic heterogeneity of gadolinium catalytic system in the 1,4-cis-polyisoprene production
- Properties of kenaf fiber-reinforced polyamide 6 composites
- Cellulose acetate filter rods tuned by surface engineering modification for typical smoke components adsorption
- A blue fluorescent waterborne polyurethane-based Zn(ii) complex with antibacterial activity
- Experimental investigation on damage mechanism of GFRP laminates embedded with/without steel wire mesh under low-velocity-impact and post-impact tensile loading
- Preparation and application research of composites with low vacuum outgassing and excellent electromagnetic sealing performance
- Assessing the recycling potential of thermosetting polymer waste in high-density polyethylene composites for safety helmet applications
- Mesoscale mechanics investigation of multi-component solid propellant systems
- Preparation of HTV silicone rubber with hydrophobic–uvioresistant composite coating and the aging research
- Experimental investigation on tensile behavior of CFRP bolted joints subjected to hydrothermal aging
- Structure and transition behavior of crosslinked poly(2-(2-methoxyethoxy) ethylmethacrylate-co-(ethyleneglycol) methacrylate) gel film on cellulosic-based flat substrate
- Mechanical properties and thermal stability of high-temperature (cooking temperature)-resistant PP/HDPE/POE composites
- Preparation of itaconic acid-modified epoxy resins and comparative study on the properties of it and epoxy acrylates
- Synthesis and properties of novel degradable polyglycolide-based polyurethanes
- Fatigue life prediction method of carbon fiber-reinforced composites
- Thermal, morphological, and structural characterization of starch-based bio-polymers for melt spinnability
- Robust biaxially stretchable polylactic acid films based on the highly oriented chain network and “nano-walls” containing zinc phenylphosphonate and calcium sulfate whisker: Superior mechanical, barrier, and optical properties
- ARGET ATRP of styrene with low catalyst usage in bio-based solvent γ-valerolactone
- New PMMA-InP/ZnS nanohybrid coatings for improving the performance of c-Si photovoltaic cells
- Impacts of the calcinated clay on structure and gamma-ray shielding capacity of epoxy-based composites
- Preparation of cardanol-based curing agent for underwater drainage pipeline repairs
- Preparation of lightweight PBS foams with high ductility and impact toughness by foam injection molding
- Gamma-ray shielding investigation of nano- and microstructures of SnO on polyester resin composites: Experimental and theoretical study
- Experimental study on impact and flexural behaviors of CFRP/aluminum-honeycomb sandwich panel
- Normal-hexane treatment on PET-based waste fiber depolymerization process
- Effect of tannic acid chelating treatment on thermo-oxidative aging property of natural rubber
- Design, synthesis, and characterization of novel copolymer gel particles for water-plugging applications
- Influence of 1,1′-Azobis(cyclohexanezonitrile) on the thermo-oxidative aging performance of diolefin elastomers
- Characteristics of cellulose nanofibril films prepared by liquid- and gas-phase esterification processes
- Investigation on the biaxial stretching deformation mechanism of PA6 film based on finite element method
- Simultaneous effects of temperature and backbone length on static and dynamic properties of high-density polyethylene-1-butene copolymer melt: Equilibrium molecular dynamics approach
- Research on microscopic structure–activity relationship of AP particle–matrix interface in HTPB propellant
- Three-layered films enable efficient passive radiation cooling of buildings
- Electrospun nanofibers membranes of La(OH)3/PAN as a versatile adsorbent for fluoride remediation: Performance and mechanisms
- Preparation and characterization of biodegradable polyester fibers enhanced with antibacterial and antiviral organic composites
- Preparation of hydrophobic silicone rubber composite insulators and the research of anti-aging performance
- Surface modification of sepiolite and its application in one-component silicone potting adhesive
- Study on hydrophobicity and aging characteristics of epoxy resin modified with nano-MgO
- Optimization of baffle’s height in an asymmetric twin-screw extruder using the response surface model
- Effect of surface treatment of nickel-coated graphite on conductive rubber
- Experimental investigation on low-velocity impact and compression after impact behaviors of GFRP laminates with steel mesh reinforced
- Development and characterization of acetylated and acetylated surface-modified tapioca starches as a carrier material for linalool
- Investigation of the compaction density of electromagnetic moulding of poly(ether-ketone-ketone) polymer powder
- Experimental investigation on low-velocity-impact and post-impact-tension behaviors of GFRP T-joints after hydrothermal aging
- The repeated low-velocity impact response and damage accumulation of shape memory alloy hybrid composite laminates
- Exploring a new method for high-performance TPSiV preparation through innovative Si–H/Pt curing system in VSR/TPU blends
- Large-scale production of highly responsive, stretchable, and conductive wrapped yarns for wearable strain sensors
- Preparation of natural raw rubber and silica/NR composites with low generation heat through aqueous silane flocculation
- Molecular dynamics simulation of the interaction between polybutylene terephthalate and A3 during thermal-oxidative aging
- Crashworthiness of GFRP/aluminum hybrid square tubes under quasi-static compression and single/repeated impact
- Review Articles
- Recent advancements in multinuclear early transition metal catalysts for olefin polymerization through cooperative effects
- Impact of ionic liquids on the thermal properties of polymer composites
- Recent progress in properties and application of antibacterial food packaging materials based on polyvinyl alcohol
- Additive manufacturing (3D printing) technologies for fiber-reinforced polymer composite materials: A review on fabrication methods and process parameters
- Rapid Communication
- Design, synthesis, characterization, and adsorption capacities of novel superabsorbent polymers derived from poly (potato starch xanthate-graft-acrylamide)
- Special Issue: Biodegradable and bio-based polymers: Green approaches (Guest Editors: Kumaran Subramanian, A. Wilson Santhosh Kumar, and Venkatajothi Ramarao)
- Development of smart core–shell nanoparticles-based sensors for diagnostics of salivary alpha-amylase in biomedical and forensics
- Thermoplastic-polymer matrix composite of banana/betel nut husk fiber reinforcement: Physico-mechanical properties evaluation
- Special Issue: Electrospun Functional Materials
- Electrospun polyacrylonitrile/regenerated cellulose/citral nanofibers as active food packagings

