Abstract
This work investigated the mechanical properties of polyethylene terephthalate (PET) reinforced with calcium carbonate (CaCO3) and silica (SiO2) nanoparticles, respectively, and the improvement in toughness of the ternary system with the incorporation of graft-modified ethylene-1-octene copolymer (POE-g-GMA). PET nanocomposites were prepared by melt blending extrusion and injection molding. Molecular dynamics (MD) simulation was employed to construct models for binary system filled with nanoparticles and ternary system with the additional inclusion of POE-g-GMA elastomers. The results of mechanical property tests and MD simulation revealed that the binary system exhibited increased elastic modulus and tensile strength, mainly attributed to the effective reinforcement of rigid nanoparticles and the surface adsorption between nanoparticles and the PET matrix enhanced the interfacial interactions. CaCO3 indicated a more pronounced reinforcing effect, possibly due to the higher crystallinity of its composites. The incorporation of POE-g-GMA resulted in a significant improvement in impact strength and the elongation at break of PET nanocomposites. This enhancement in toughness is attributed to the elastomer’s ability to absorb a substantial amount of impact energy, while the elastic modulus is higher than that of pure PET.
1 Introduction
The rapid development of nanotechnology and nanomaterials has led to extensive research on polymer modification using nanoparticles (1). Calcium carbonate (CaCO3) is widely used as a filler in plastics due to its cost-effectiveness, superior mechanical properties, and thermal stability (2,3). Seyedzavvar et al. (4) incorporated CaCO3 nanoparticles into polybutylene terephthalate (PBT) and found that the thermal conductivity of PBT increased with the CaCO3 content, reaching up to 5 wt%. It is shown that the enhancement of the polymer’s mechanical properties is closely related to the interfacial interaction between the polymer and the filler (5,6,7,8). Similarly, silica (SiO2) is also commonly employed as a polymer modification due to its high stiffness, viscosity, and creep resistance (9). Guo et al. (10) showed that SiO2/epoxy resin systems modified with amino-containing silane coupling agents exhibited reduced viscosity and excellent thermal stability.
Although prior research has indicated that a higher concentration of nanoparticles increases the number of reinforcing sites (11), it may lead to a reduction in the fracture toughness of the polymer (12). Consequently, ternary systems that combine rigid nanofillers with elastomers possess higher application values and have been investigated by previous researchers in this area. For instance, the simultaneous addition of CaCO3 and ethylene-octene copolymer (POE) to polypropylene (PP) improved the impact toughness without decreasing the tensile strength (13). Pham and Nguyen (14) found that incorporating 2 wt% CaCO3 into styrene-ethylene/butylene-styrene (SEBS)/PET/PP resulted in increased tensile and flexural strength.
In addition, molecular dynamics (MD) simulation has proved to be a valuable tool in predicting the properties and interaction relationships of nanopolymer composites from the molecular scale (4,15). Fereidoon et al. (16) used MD simulation to show that the interaction between graphene and TiO2 surfaces increased Young’s modulus and shear modulus of TiO2. Guo et al. (17) investigated the effect of POE on the pyrolysis mechanism of PP by using MD methods.
However, there is a lack of research that integrates experimental studies and MD simulation to predict the mechanical properties of polyethylene terephthalate (PET) nanocomposites, spanning from binary system to ternary system with the inclusion of graft-modified ethylene-1-octene copolymer (POE-g-GMA). PET nanocomposites containing only added nanofillers exhibit significant brittleness, leading to a substantial decrease in fracture toughness. This situation diminishes the cost-effectiveness and utility of PET. Consequently, the incorporation of graft-modified elastomers serves to enhance material toughness while preserving its strength. In this study, the MD approach was employed to establish models of PET/CaCO3, PET/SiO2, PET/POE-g-GMA/CaCO3, and PET/POE-g-GMA/SiO2. Subsequently, PET binary and ternary blends were prepared through melt blending and injection molding. These methods were performed to analyze the influence of nanoparticles and elastomers on the mechanical properties of PET, as well as to elucidate the enhancement and toughening mechanisms involved.
2 Experimental method
2.1 Materials
PET with a characteristic viscosity of 0.83 dl·g−1 was purchased from Sanfame Co., Ltd (China). CaCO3 nanoparticles (CCU-200) were supplied by HUANA New Materials Co., Ltd (China). POE-g-GMA was provided by Fine-Blend Co., Ltd (China). SiO2 nanoparticles with a specific surface area of 195 m2·g−1 were supplied by Beesley Co., Ltd (China).
2.2 Nanocomposites preparation
PET, CaCO3, and SiO2 were placed in a vacuum drying oven and dried at 130°C. Subsequently, both binary composites containing 2 wt% of CaCO3 and SiO2, as well as ternary PET nanocomposites containing 10 wt% of POE-g-GMA, respectively, were thoroughly and homogeneously mixed. Afterward, the mixture was melt extruded in a twin-screw extruder at temperatures of 250°C, 255°C, 260°C, and 265°C along the extruder from barrel to head. After the extrusion process, tensile and impact samples were prepared by injection molding, with the barrel temperature of the injection molding machine set at 270°C.
2.3 Characterization
The notched impact strength of PET and its composites was tested using an XBL cantilever beam impact tester (Shanghai, China). The tensile properties of the specimens were measured by a Zwick/Roell Z005 (Germany) material testing machine at a rate of 10 mm·min−1. The bulk modulus, shear modulus, and Young’s modulus of PET and its composites were measured using the universal mechanical testing machine Instron 5982 (USA). Fourier transform infrared spectroscopy (FTIR) was used to characterize the functional groups and interaction properties of materials. The FTIR spectra were recorded using the Thermo Scientific Nicolet iS20 FTIR spectrometer (USA) with 32 scans and a resolution of 4 cm−1. The morphology of the nanoparticles was observed using a transmission electron microscope (TEM) model JEOL JEM-2100Plus (Japan). The morphology of the impact fracture was observed using a Sigma 300 (Germany) scanning electron microscope (SEM), and before testing, a thin layer of gold was sprayed on the samples. X-ray diffraction (XRD) was employed to characterize the crystal structure using Cu Kα radiation (λ CuKα = 1.5418 Å) on a Rigaku Ultima IV (Tokyo, Japan) X-ray diffractometer, the voltage was set at 40 KV, and the scanning rate was 2°·min−1. The material’s melting and crystallization processes were investigated using differential thermal scanning (DSC) with a TA instruments-Model 25 (USA) differential thermal scanner at a heating rate of 10°C·min−1. The samples were first heated from 25°C to 280°C and then cooled back to 25°C, subsequently, heated up to 280°C and cooled down again.
3 MD simulation
3.1 MD method
All simulations were performed by the molecular simulation engine Materials Studio 6.0 software (35). During the geometry optimization and MD relaxation phases, the COMPASS force field was employed, which is based on ab initio calculations for condensed phase optimization (18,19). The potential function used in this force field consists of both valence and non-bonding interaction terms. The potential energy of the system is represented by Eq. 1, where the valence terms are composed of E b , E θ , E φ , and E χ , denoting the bonding (b), angle (θ), torsion (φ), and out-of-plane angle (χ), respectively. Additionally, the cross-coupling terms consist of E bb′ , E bθ , E bφ , E θθ′ , and E θθ′φ , and the non-bonding terms include the van der Waals interaction term (E vdw) (20) and the Coulomb term (E elec), which accounts for electrostatic interactions (21). These terms are represented by Eqs 2–4.
Verlet’s algorithm (22) is employed to integrate the molecular equations of motion during the simulation. The position of the particle at time t is represented by r i (t), and the position of the particle at time t + δt is expressed using Eq. 5.
During the equilibrium process of the model, NPT (isothermal and isobaric ensemble) and NVT (canonical ensemble) ensembles were employed. The temperature was controlled using the Nose thermostat (23), and the pressure was controlled using the Berendsen pressure control algorithm (24).
3.2 Simulation details for PET nanocomposites
The initial step involved constructing CaCO3 and SiO2 crystal models based on the structures of calcite and quartz crystal cells. The lattice parameters were set as follows: a = b = 4.99 Å, c = 17.061 Å, α = β = 90°, and γ = 120° for CaCO3, and a = b = 4.91 Å, c = 5.402 Å, α = β = 90°, and γ = 120° for SiO2 (25,26), as shown in Figure 1(a) and (c). Next geometrically optimized model of spherical CaCO3 and hydroxylated-silica nanoclusters with a radius of 5 Å were established, as shown in Figure 1(b) and (d). The geometry optimization was conducted using the Smart method, with a maximum iteration of 50,000 and a cutoff radius of 12.5 Å for non-bonded interactions. Subsequent processes involving the construction of packing model and equilibrium utilized an atom-based summation method for electrostatic and van der Waals interaction.

Optimized spherical nanocluster of (a) crystal structure of calcite, (b) CaCO3, (c) crystal structure of SiO2, and (d) hydroxylated SiO2.
The geometrically optimized structural model of PET with a repeat unit of 15 was constructed based on the molecular formula, as depicted in Figure 2(a). To illustrate the molecular structure of POE-g-GMA, the model of POE was built first, comprising two monomers, ethylene, and 1-octene, with the mass fraction of 1-octene accounting for 30%, as displayed in Figure 2(b). The potential chemical reaction pathways for grafting GMA onto the POE molecule are illustrated in Figure 3(a), and the chemical bonds and C atoms at the grafting site were labeled in yellow, as shown in Figure 3(b).

Optimized molecular model of (a) PET chains and (b) POE.
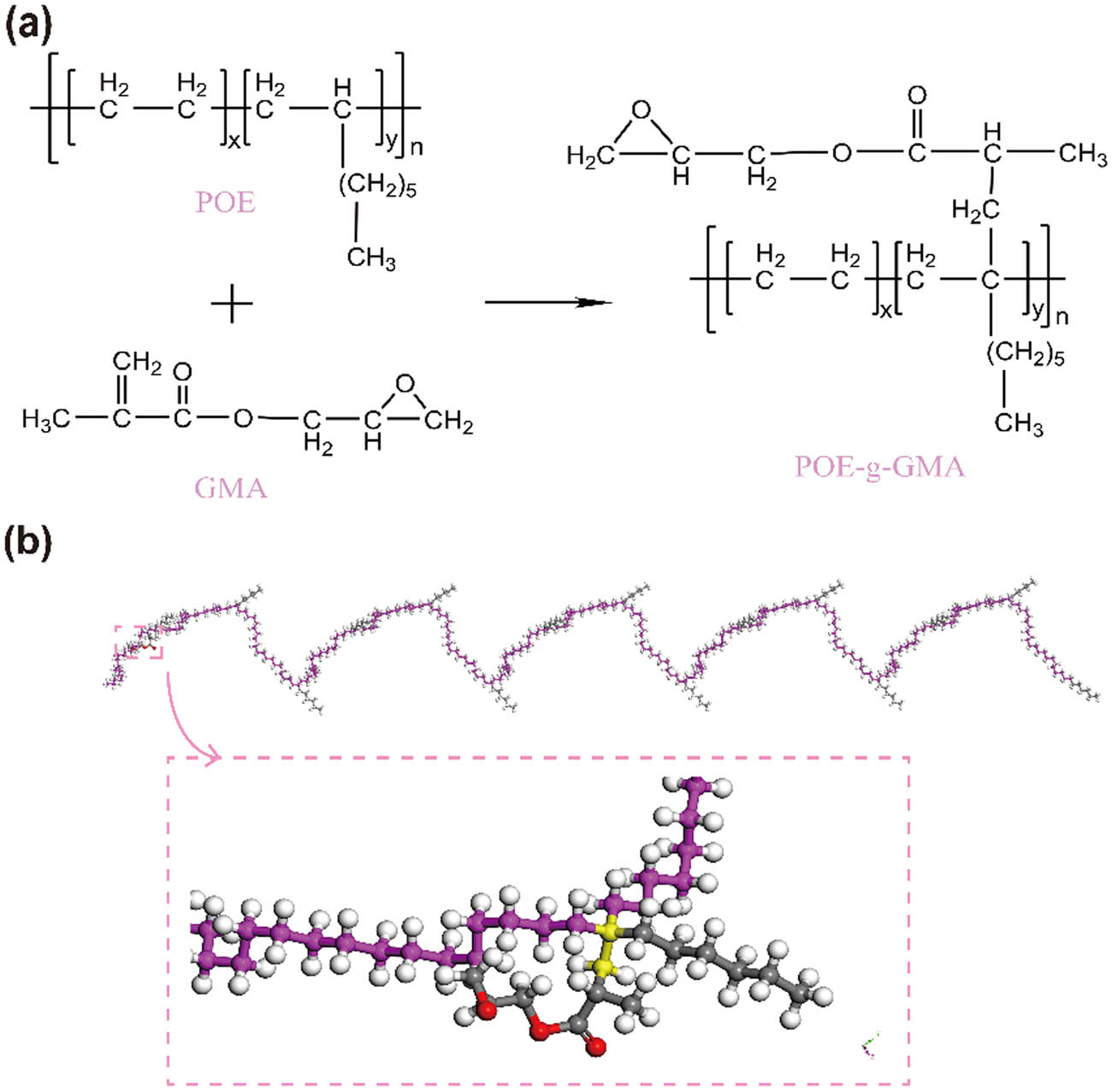
(a) Possible chemical grafting reaction of POE and GMA and (b) optimized molecular model of POE-g-GMA chains.
Subsequently, the polymer molecular chains were packed around the nanoparticles, periodic boxes of PET/CaCO3 and PET/SiO2 with a nanoparticle concentration of 2 wt% were constructed, respectively. Additionally, periodic boxes containing 10% POE-g-GMA were also constructed. This step was carried out in the Amorphous Cell Packing module, defining an initial density of 0.1 g·cm−3, as shown in Figure 4(a).

(a) Initial structure of PET/POE-g-GMA/SiO2 nanocomposite and (b) equilibrium structure of PET/POE-g-GMA/SiO2 nanocomposite.
After geometry optimization, the dynamic relaxation was conducted for 100 ps in the NPT ensemble at a pressure of 0.1 GPa. To enable sufficient movement and relaxation of the polymer chain segments, an annealing process was carried out in the NVT ensemble. The temperature was raised from 298 to 500 K and decreased back to 298 K in steps of 1 fs. Subsequently, a relaxation process was conducted in the NVT ensemble for 100 ps, resulting in a final configuration where both temperature and energy reached an equilibrium state, as illustrated in Figure 4(b).
3.3 Calculation details of mechanical properties
For a classical mechanical system containing a fixed number of atoms N, the equilibrium stress tensor can be obtained from the virial theorem (27), which is expressed as Eq. 6.
in which V represents the volume of the system, m denotes the mass, P i α is the momentum α component of particle i located at position r i , and F ij is the interaction force between particle i and particle j. The mechanical properties of the nanocomposites are calculated using the Forcite module. The elastic stiffness constant C ij is obtained by applying a constant strain. Eqs 7 and 8 express the Lame constant.
For isotropic materials, the mechanical property parameters such as bulk modulus K, shear modulus G, and Young’s modulus E are calculated as shown in Eq. 9.
4 Results and discussion
4.1 Crystallization and melting behavior
The crystalline behavior of PET and its nanocomposites was analyzed using DSC. As shown in Figure 5(a) and Table 1, the addition of SiO2 and CaCO3 resulted in an increase in the glass transition temperature (T g), crystallization temperature (T c), and crystallinity (X c) of the nanocomposites compared to pure PET. The increase in T c and X c can be attributed to the nanoparticles acting as effective nucleating agents in the PET matrix. The presence of more nucleation sites promoted the growth of PET crystals, and the significant enhancement of the crystallization peaks was also related to this phenomenon. Furthermore, the nanoparticles hindered the free movement of PET molecular chains at lower temperatures, resulting in a higher T g. However, with the addition of POE-g-GMA, the crystallinity of both nanocomposites decreased, which may be attributed to the generation of an amorphous mixed phase due to the blending of the elastomer and PET. This mixed phase inhibited the crystallization process and interfered with the regular arrangement of the PET chains, resulting in a decrease in the crystallinity of PET.

DSC curves of PET nanocomposites: (a) the first cooling cycle and (b) the second heating run.
Thermal properties of PET nanocomposites
| Sample | T g (°C) | T c (°C) | T m (°C) | ΔH c (J·g−1) | X c (%) |
|---|---|---|---|---|---|
| PET | 72.5 | 190.1 | 253.1 | 46.0 | 32.8 |
| PET/CaCO3 | 77.5 | 219.1 | 251.8 | 48.8 | 35.5 |
| PET/SiO2 | 72.9 | 217.5 | 252.1 | 48.3 | 35.2 |
| PET/POE-g-GMA/CaCO3 | 72.4 | 199.8 | 249.5 | 33.1 | 26.8 |
| PET/POE-g-GMA/SiO2 | 72.2 | 201.0 | 250.5 | 31.1 | 25.2 |
For the melting behavior, it can be observed from Figure 5(b) that two melting peaks appeared in all curves except for pure PET. This is a consequence of the material initially forming partially crystallized lamellae with incomplete structures during the cooling process, as the temperature rises over a sufficient duration, these crystals transform into more well-structured crystals (28). Within the blend system, two crystal forms are present: folded-chain crystals and partially elongated-chain crystals (29). Furthermore, the decrease in the melting temperature (T m) can be explained by the mutual constraints of PET and POE-g-GMA chains. There are reactions occurring between the polymer groups. Additionally, the presence of nanofillers disrupts crystal growth, leading to a reduction in the crystallinity of PET within the blend.
The XRD patterns presented in Figure 6 also explained the crystalline behavior. The diffraction peak of pure PET appeared broad around 2θ = 21.5°, corresponding to a crystallographic index of (−1 1 1). With the addition of CaCO3, the diffraction peaks in the binary and ternary composite systems were significantly enhanced, appearing at approximately 23.0°, 29.4°, 36°, 39.3°, 43.1°, 47.1°, and 48.5°, corresponding to the crystalline surface indices of (0 1 2), (1 0 4), (1 1 0), (1 1 3), (2 0 2), (0 2 4), and (1 1 6), respectively. However, the diffraction peaks observed in the PET composite system containing only CaCO3 were sharper than those in the ternary system containing elastomer, indicating that CaCO3 played a favorable role in promoting PET crystallization. For the composite system containing SiO2, diffraction peaks corresponding to the crystal surface indices of (1 1 0), (1 0 2), and (2 0 1) were observed at approximately 16.8°, 23.8°, and 26.0°, respectively. The diffraction peaks were not as prominent as those in the system containing CaCO3, probably because PET/CaCO3 composites exhibit higher crystallinity. Furthermore, CaCO3 contains heavier calcium elements with higher atomic scattering factors (30), resulting in relatively higher XRD intensities.

XRD patterns of PET nanocomposites.
4.2 Morphology of nanocomposites
The TEM images of CaCO3 and SiO2 nanoparticles are shown in Figure 7, reflecting the morphology and size distribution of the particles. The impact fracture morphology of PET, as well as its binary and ternary blends, was observed by SEM. Figure 8(a) presents the fracture surface of pure PET, which appeared relatively flat and smooth, exhibiting a brittle fracture morphology. With the incorporation of 2 wt% of CaCO3 and SiO2, respectively, the nanoparticles were uniformly dispersed within the polymer matrix, resulting in a flat fracture surface with a small number of cracks, which still exhibited brittle fracture characteristics, as shown in Figure 8(b) and (c). However, with the introduction of POE-g-GMA into the PET nano-binary system, the fracture surface became rougher and exhibited deformation. Stacked layer structure and tear-like ripples appeared. The material overcame the crack extension by absorbing a large amount of energy, and the matrix underwent shear yielding. As a result, the fracture morphology transformed from brittle fracture to tough fracture as illustrated in Figure 8(d) and (e) (31). Figure 8(f) shows the magnified POE-g-GMA elastomer morphology, observed to tear but not detach, owing to its strong interfacial bonding with the PET matrix. The elastomer particles effectively absorbed impact energy, reducing stress concentration during the tearing process and significantly enhancing the toughness of the polymer composites.

TEM images of (a) CaCO3 nanoparticles and (b) SiO2 nanoparticles.

SEM micrographs of the impact fracture surface of (a) PET and (b) PET/CaCO3, (c) PET/SiO2, (d) PET/POE-g-GMA/CaCO3, (e) PET/POE-g-GMA/SiO2, and (f) magnified section of (e).
4.3 Analysis of interactions in PET nanocomposites
The curves illustrating the evolution of energy and temperature over time for the initial model in the NPT simulations are depicted in Figure 9. Towards the end of the NPT run, both values exhibited fluctuations around a constant value, indicating that the system had reached an initial equilibrium state. After all simulations were completed, the distribution density of PET molecules near the nanoparticles was analyzed. The radial distribution function (RDF) of the PET/CaCO3 system is illustrated in Figure 10(a). A higher concentration of PET around the nano-CaCO3 can be observed, suggesting that PET exhibited a high ligand number density and formed a good interaction with CaCO3. The RDF showed significant peaks at r = 1.1 Å and r = 1.4 Å. Generally, peaks above 3.5 Å in RDF indicate van der Waals force interactions, while peaks below 3.5 Å primarily consist of hydrogen bonding interactions (26). When PET and nanoparticles were in contact, van der Waals forces and other interactions brought them closer and created interfacial adhesion. Figure 10(b) represents the RDF of CaCO3 in the composite model around PET molecules, indicating the dispersion state of CaCO3 particles located at the center of the centroid in the composite system. As the distance between CaCO3 and the polymer surface increased, the density distribution of PET molecules increased, especially at the peak positions. These peaks indicated a higher presence of PET molecules in these regions, the average distance between nanoparticles and molecules was closer, indicating good dispersion of CaCO3 particles. Beyond r > 4 Å, no distinct peaks were observed, indicating weak van der Waals interactions between CaCO3 particles and polymer functional groups, indicating poor dispersion of the particles.

PET/CaCO3 nanocomposite during NPT dynamics of (a) energy evolution and (b) temperature evolution.

RDF of (a) PET/CaCO3 nanocomposite and (b) CaCO3 around PET molecular chains in the composites.
Figure 11 displays the FTIR of PET and its composites, explaining the chemical bonding between nanoparticles and PET. In PET, the stretching vibrations of C═O, C–O, and C–O–C bonds appeared at 1,732, 1,242, and 1,060 cm−1, respectively. The characteristic peak at 3,631 cm−1 corresponded to the O–H stretching between hydroxyl groups in PET chains (5,32). Upon the addition of nano CaCO3, a stretching vibration of CO3 2− appeared at 1,452 cm−1 (33). The enhancement of the peak at 1,712 cm−1 might be related to physical adsorption interactions between CaCO3 and PET, causing a change in the vibration frequency of C═O. After adding SiO2 particles, no significant change in peak intensity was observed compared to pure PET. However, an absorption peak at 802 cm−1, corresponded to the stretching vibration of Si–O bonds. The diffraction peak intensity of PET/POE-g-GMA/CaCO3 and PET/POE-g-GMA/SiO2 significantly decreased compared to pure PET. This reduction might be due to the disruption of PET molecular chains’ integrity upon the addition of the elastomer, leading to a decrease in crystallinity. Moreover, in PET/POE-g-GMA/SiO2, strong hydrogen bonding interactions occurred between the free hydroxyl groups on the SiO2 surface and the carboxyl groups in PET, reducing the hydroxyl content and contributing to the weakened diffraction peak intensity. These interactions contributed to improving the mechanical properties of the composites.

FTIR spectra of PET nanocomposites.
4.4 Mechanical properties
The mechanical properties of PET and its binary and ternary composites obtained from experiments and MD simulations are shown in Table 2 and Figure 12, respectively. The experimental and MD results indicate that compared to pure PET, the tensile strength, bulk modulus (K), shear modulus (G), and Young’s modulus (E) of PET/CaCO3 and PET/SiO2 nanocomposites were significantly improved. This improvement can be attributed to the favorable interaction and uniform dispersion of nanoparticles within the PET matrix. The reinforcement of CaCO3 was more pronounced than that of SiO2, with a maximal tensile strength reaching 55.1 MPa and a maximal Young’s modulus of 2.51 GPa. This enhancement is likely due to the higher crystallinity with the addition of CaCO3, which contributed to the improvement of the polymer’s strength and stiffness. Additionally, the elongation at break and notched impact strength of both binary systems was reduced compared to pure PET, mainly due to the constraint imposed on the molecular chain activity of the polymer matrix by the rigid nanofillers (32).
Mechanical properties of PET nanocomposites by experiments
| Sample | Tensile strength (MPa) | Elongation at break (%) | Impact strength (kJ·m−2) | Bulk modulus (GPa) | Shear modulus (GPa) | Young’s modulus (GPa) |
|---|---|---|---|---|---|---|
| PET | 50.6 | 5.3 | 3.1 | 2.36 | 0.58 | 1.31 |
| PET/CaCO3 | 55.1 | 3.0 | 2.5 | 4.57 | 1.14 | 2.51 |
| PET/SiO2 | 51.6 | 1.4 | 1.9 | 3.16 | 0.96 | 2.37 |
| PET/POE-g-GMA/CaCO3 | 49.4 | 29.3 | 6.3 | 3.29 | 0.84 | 1.93 |
| PET/POE-g-GMA/SiO2 | 47.2 | 20.4 | 5.6 | 2.79 | 0.64 | 1.72 |

Mechanical properties of PET nanocomposites by MD simulation.
The incorporation of POE-g-GMA into the ternary system exhibited a remarkable increase in both elongation at break and notched impact strength, surpassing those of pure PET and PET nano-binary composites, up to 29.3% and 6.3 kJ·m−2, respectively. However, the tensile strength decreased compared to that of pure PET, and the elastic modulus was lower than that of the nano-binary system. This behavior can be attributed to the synergistic toughening effect of the soft elastomers within the matrix and the nanoparticles encapsulated around the elastomer (34). The blends underwent a brittle-tough transition from binary to ternary system, allowing the polymer molecular chains to move relatively easily and exhibit higher toughness. Although the rigidity and strength decreased compared to the binary system, the elastic modulus was higher than that of pure PET, and the decrease in tensile strength was not significant, indicating that the rigid nanoparticles served as an advantageous reinforcing phase in the matrix.
5 Conclusion
This study investigated the improvement of PET’s mechanical properties through the incorporation of two types of nanofillers, CaCO3 and SiO2, as well as the enhancement of toughness from binary to ternary system after the introduction of the elastomer POE-g-GMA. The investigation utilized a combination of experimental methods and MD simulation. Mechanical property tests and MD simulation showed that the tensile strength, bulk modulus, shear modulus, and Young’s modulus of PET/CaCO3 and PET/SiO2 nanocomposites were increased compared to pure PET. This reinforcing effect was mainly attributed to the CaCO3 and SiO2 nanoparticles acting as nucleating agents and the homogeneous dispersion in the polymer matrix, hindering the mobility of PET molecular chains. CaCO3 demonstrated a better enhancement, with the tensile strength reaching a maximum of 55.1 MPa, probably due to the higher crystallinity of its composites. With the incorporation of POE-g-GMA, PET nanocomposites exhibited an increase in impact strength and elongation at break, up to 29.3% and 6.3 kJ·m−2, respectively. The elastic modulus was higher than pure PET, while the tensile strength slightly decreased, achieving a balance between stiffness and toughness. The effective toughening effect was attributed to the elastomer absorbing a substantial amount of impact energy and reducing stress concentration when the material was subjected to stress.
Acknowledgements
The authors would like to acknowledge the College of Chemistry, Chemical Engineering and Biotechnology of Donghua University for the software support.
-
Funding information: Authors state no funding involved.
-
Author contributions: Xiaoying Cheng: conceptualization, data curation, software, validation, and writing – original draft; Qiuhui Liao: conceptualization, methodology, and supervision; Jiani Xu: resources and writing – review and editing; Zeen Gu: investigation and validation; Yibo He: investigation and visualization.
-
Conflict of interest: Authors state no conflict of interest.
-
Data availability statement: The datasets generated and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
(1) Biswal M, Mohanty S, Nayak SK, Kumar PS. Effect of functionalized nanosilica on the mechanical, dynamic-mechanical, and morphological performance of polycarbonate/nanosilica nanocomposites. Polym Eng Sci. 2013;53:1287–96. 10.1002/pen.23388.Suche in Google Scholar
(2) Sahoo A, Gayathri HN, Phanindra Sai T, Upasani PS, Raje V, Berkmans J, et al. Enhancement of thermal and mechanical properties of few layer boron nitride reinforced PET composite. Nanotechnology. 2020;31:315706. 10.1088/1361-6528/ab88ec.Suche in Google Scholar PubMed
(3) Gao W, Ma X, Liu Y, Wang Z, Zhu Y. Effect of calcium carbonate on PET physical properties and thermal stability. Powder Technol. 2013;244:45–51. 10.1016/j.powtec.2013.04.008.Suche in Google Scholar
(4) Seyedzavvar M, Boğa C, Akar S, Pashmforoush F. Molecular dynamic approach to predict thermo-mechanical properties of poly(butylene terephthalate)/CaCO3 nanocomposites. Mater Today Commun. 2021;28:102602. 10.1016/j.mtcomm.2021.102602.Suche in Google Scholar
(5) Parvinzadeh M, Moradian S, Rashidi A, Yazdanshenas M-E. Surface characterization of polyethylene terephthalate/silica nanocomposites. Appl Surf Sci. 2010;256:2792–802. 10.1016/j.apsusc.2009.11.030.Suche in Google Scholar
(6) Avella M, Cosco S, Lorenzo MLD, Pace ED, Errico ME, Gentile G. iPP based nanocomposites filled with calcium carbonate nanoparticles: structure/properties relationships. Macromol Symp. 2006;234:156–62. 10.1002/masy.200650220.Suche in Google Scholar
(7) Chen D, Tiwari SK, Ma Z, Wen J, Liu S, Li J, et al. Phase behavior and thermo-mechanical properties of IF-WS2 reinforced PP–PET blend-based nanocomposites. Polymers. 2020;12:2342. 10.3390/polym12102342.Suche in Google Scholar PubMed PubMed Central
(8) Rusu MA, Dan L, Consmin C, Miuta F, Marioara M, Laura SD, et al. Characterization polyethylene terephthalate nanocomposites mixing with nano-silica and titanium oxide. MATEC Web Conferences. 2017;137:08005. 10.1051/matecconf/201713708005.Suche in Google Scholar
(9) Salehi Vaziri H, Abadyan M, Nouri M, Omaraei IA, Sadredini Z, Ebrahimnia M. Investigation of the fracture mechanism and mechanical properties of polystyrene/silica nanocomposite in various silica contents. J Mater Sci. 2011;46:5628–38. 10.1007/s10853-011-5513-9.Suche in Google Scholar
(10) Guo Q, Zhu P, Li G, Wen J, Wang T, Lu D, et al. Study on the effects of interfacial interaction on the rheological and thermal performance of silica nanoparticles reinforced epoxy nanocomposites. Compos Part B: Eng. 2017;116:388–97. 10.1016/j.compositesb.2016.10.081.Suche in Google Scholar
(11) Chowreddy RR, Nord-Varhaug K, Rapp F. Recycled polyethylene terephthalate/carbon nanotube composites with improved processability and performance. J Mater Sci. 2018;53:7017–29. 10.1007/s10853-018-2014-0.Suche in Google Scholar
(12) Goudarzi L, Izadi-Vasafi H, Nikfar N. Investigation of the morphological and mechanical properties of polyethylene terephthalate (PET)/Ethylene propylene diene rubber (EPDM) blends in the presence of multi-walled carbon nanotubes. J Macromol Sci, Part B. 2018;57:585–94. 10.1080/00222348.2018.1493174.Suche in Google Scholar
(13) Ma CG, Mai YL, Rong MZ, Ruan WH, Zhang MQ. Phase structure and mechanical properties of ternary polypropylene/elastomer/nano-CaCO3 composites. Compos Sci Technol. 2007;67:2997–3005. 10.1016/j.compscitech.2007.05.022.Suche in Google Scholar
(14) Pham H-NT, Nguyen V-T. Effect of calcium carbonate on the mechanical properties of polyethylene terephthalate/polypropylene blends with styrene-ethylene/butylene-styrene. J Mech Sci Technol. 2020;34:3925–30. 10.1007/s12206-020-2201-1.Suche in Google Scholar
(15) Pashmforoush F, Ajori S, Azimi HR. Interfacial characteristics and thermo-mechanical properties of calcium carbonate/polystyrene nanocomposite. Mater Chem Phys. 2020;247:122871. 10.1016/j.matchemphys.2020.122871.Suche in Google Scholar
(16) Fereidoon A, Aleaghaee S, Taraghi I. Mechanical properties of hybrid graphene/TiO2 (rutile) nanocomposite: A molecular dynamics simulation. Comput Mater Sci. 2015;102:220–7. 10.1016/j.commatsci.2015.02.044.Suche in Google Scholar
(17) Guo G, Fan K, Guo Z, Guo W. Pyrolysis behavior of automotive polypropylene plastics: ReaxFF molecular dynamics study on the co-pyrolysis of polypropylene and EPDM/POE. Energy. 2023;280:128202. 10.1016/j.energy.2023.128202.Suche in Google Scholar
(18) Sun H, Ren P, Fried JR. The COMPASS force field: parameterization and validation for phosphazenes. Comput Theor Polym Sci. 1998;1–2:229–46. 10.1016/S1089-3156(98)00042-7.Suche in Google Scholar
(19) Sun H. Compass: An ab initio force-field optimized for condensed-phase applications overview with details on alkane and benzene compounds. JPhyChemB. 1998;102:7338–64. 10.1021/jp980939v.Suche in Google Scholar
(20) Andersson Y, Hult E, Rydberg H, Apell P, Lundqvist BI, Langreth DC. Van der Waals Interactions in density functional theory. In: Dobson JF, Vignale G, Das MP, editors. Electronic Density Functional Theory. Springer, Boston, MA; 1998. 10.1007/978-1-4899-0316-7_17.Suche in Google Scholar
(21) Darden T, York D, Pedersen L. Particle mesh Ewald: An N log( N) method for Ewald sums in large systems. J Chem Phys. 1993;98:10089–92. 10.1063/1.464397.Suche in Google Scholar
(22) Verlet L. Computer “experiments” on classical fluids. I. Thermodynamical properties of Lennard-Jones molecules. Phys Rev. 1967;98:159–1. 10.1103/PhysRev.159.98.Suche in Google Scholar
(23) Nosé S. A unified formulation of the constant temperature molecular dynamics methods. J Chem Phys. 1984;81:511–9. 10.1063/1.447334.Suche in Google Scholar
(24) Berendsen HJC, Postma JPM, Van Gunsteren WF, DiNola A, Haak JR. Molecular dynamics with coupling to an external bath. J Chem Phys. 1984;81:3684–90. 10.1063/1.448118.Suche in Google Scholar
(25) Zehir B, Boga C, Seyedzavvar M. Molecular dynamics simulation and experimental investigation of mechanical properties of calcium carbonate and graphene reinforced polylactic acid nanocomposites. J Mol Model. 2023;29:187. 10.1007/s00894-023-05598-1.Suche in Google Scholar PubMed
(26) Wei Q, Zhang Y, Wang Y, Yang M. A molecular dynamic simulation method to elucidate the interaction mechanism of nano-SiO2 in polymer blends. J Mater Sci. 2017;52:12889–901. 10.1007/s10853-017-1330-0 Suche in Google Scholar
(27) Swenson RJ. Comments on virial theorems for bounded systems. Am J Phys. 1983;51:940–2. 10.1119/1.13390.Suche in Google Scholar
(28) Zhou Y, Wang J, Cai S-Y, Wang Z-G, Zhang N-W, Ren J. Effect of POE-g-GMA on mechanical, rheological and thermal properties of poly(lactic acid)/poly(propylene carbonate) blends. Polym Bull. 2018;75:5437–54. 10.1007/s00289-018-2339-5.Suche in Google Scholar
(29) Zahir Rahem A, Douibi A, Lallam C, Delaite M. Guessoum, Synergistic combination of crystallization and addition of a toughening agent to promote recycled poly(ethylene terephthalate) performances. Polym Sci Ser A. 2019;61:635–49. 10.1134/S0965545X19050158.Suche in Google Scholar
(30) Maslen EN, Streltsov VA, Streltsova NR. X-ray study of the electron density in calcite, CaCo3. Acta Crystallogr B Struct Sci. 1993;49:636–41. 10.1107/S0108768193002575.Suche in Google Scholar
(31) Ke F, Jiang X, Xu H, Ji J, Su Y. Ternary nano-CaCO3/poly(ethylene terephthalate) fiber/polypropylene composites: Increased impact strength and reinforcing mechanism. Compos Sci Technol. 2012;72:574–9. 10.1016/j.compscitech.2012.01.001.Suche in Google Scholar
(32) Inuwa IM, Arjmandi R, Ibrahim AN, Haafiz MKM, Wong SL, Majeed K, et al. Enhanced mechanical and thermal properties of hybrid graphene nanoplatelets/multiwall carbon nanotubes reinforced polyethylene terephthalate nanocomposites. Fibers Polym. 2016;17:1657–66. 10.1007/s12221-016-6238-9.Suche in Google Scholar
(33) Lee CS, Yoon KH, Park JC, Kim H-U, Park Y-B. Thermal and mechanical properties of modified CaCO3 filled poly (ethylene terephthalate) nanocomposites. Fibers Polym. 2014;15:1493–9. 10.1007/s12221-014-1493-0.Suche in Google Scholar
(34) Hajibabazadeh S, Razavi Aghjeh M, Mehrabi Mazidi M. Stiffness-toughness balance in PP/EPDM/SiO2 ternary blend-nanocomposites: The role of microstructural evolution. J Compos Mater. 2021;55:265–75. 10.1177/0021998320948125.Suche in Google Scholar
(35) Accelrys Software Inc., Materials Studio Modeling Environment, Release6.0, Accelrys Software Inc., San Diego, 2011.Suche in Google Scholar
© 2024 the author(s), published by De Gruyter
This work is licensed under the Creative Commons Attribution 4.0 International License.
Artikel in diesem Heft
- Research Articles
- Flame-retardant thermoelectric responsive coating based on poly(3,4-ethylenedioxythiphene) modified metal–organic frameworks
- Highly stretchable, durable, and reversibly thermochromic wrapped yarns induced by Joule heating: With an emphasis on parametric study of elastane drafts
- Molecular dynamics simulation and experimental study on the mechanical properties of PET nanocomposites filled with CaCO3, SiO2, and POE-g-GMA
- Multifunctional hydrogel based on silk fibroin/thermosensitive polymers supporting implant biomaterials in osteomyelitis
- Marine antifouling coating based on fluorescent-modified poly(ethylene-co-tetrafluoroethylene) resin
- Preparation and application of profiled luminescent polyester fiber with reversible photochromism materials
- Determination of pesticide residue in soil samples by molecularly imprinted solid-phase extraction method
- The die swell eliminating mechanism of hot air assisted 3D printing of GF/PP and its influence on the product performance
- Rheological behavior of particle-filled polymer suspensions and its influence on surface structure of the coated electrodes
- The effects of property variation on the dripping behaviour of polymers during UL94 test simulated by particle finite element method
- Experimental evaluation on compression-after-impact behavior of perforated sandwich panel comprised of foam core and glass fiber reinforced epoxy hybrid facesheets
- Synthesis, characterization and evaluation of a pH-responsive molecular imprinted polymer for Matrine as an intelligent drug delivery system
- Twist-related parametric optimization of Joule heating-triggered highly stretchable thermochromic wrapped yarns using technique for order preference by similarity to ideal solution
- Comparative analysis of flow factors and crystallinity in conventional extrusion and gas-assisted extrusion
- Simulation approach to study kinetic heterogeneity of gadolinium catalytic system in the 1,4-cis-polyisoprene production
- Properties of kenaf fiber-reinforced polyamide 6 composites
- Cellulose acetate filter rods tuned by surface engineering modification for typical smoke components adsorption
- A blue fluorescent waterborne polyurethane-based Zn(ii) complex with antibacterial activity
- Experimental investigation on damage mechanism of GFRP laminates embedded with/without steel wire mesh under low-velocity-impact and post-impact tensile loading
- Preparation and application research of composites with low vacuum outgassing and excellent electromagnetic sealing performance
- Assessing the recycling potential of thermosetting polymer waste in high-density polyethylene composites for safety helmet applications
- Mesoscale mechanics investigation of multi-component solid propellant systems
- Preparation of HTV silicone rubber with hydrophobic–uvioresistant composite coating and the aging research
- Experimental investigation on tensile behavior of CFRP bolted joints subjected to hydrothermal aging
- Structure and transition behavior of crosslinked poly(2-(2-methoxyethoxy) ethylmethacrylate-co-(ethyleneglycol) methacrylate) gel film on cellulosic-based flat substrate
- Mechanical properties and thermal stability of high-temperature (cooking temperature)-resistant PP/HDPE/POE composites
- Preparation of itaconic acid-modified epoxy resins and comparative study on the properties of it and epoxy acrylates
- Synthesis and properties of novel degradable polyglycolide-based polyurethanes
- Fatigue life prediction method of carbon fiber-reinforced composites
- Thermal, morphological, and structural characterization of starch-based bio-polymers for melt spinnability
- Robust biaxially stretchable polylactic acid films based on the highly oriented chain network and “nano-walls” containing zinc phenylphosphonate and calcium sulfate whisker: Superior mechanical, barrier, and optical properties
- ARGET ATRP of styrene with low catalyst usage in bio-based solvent γ-valerolactone
- New PMMA-InP/ZnS nanohybrid coatings for improving the performance of c-Si photovoltaic cells
- Impacts of the calcinated clay on structure and gamma-ray shielding capacity of epoxy-based composites
- Preparation of cardanol-based curing agent for underwater drainage pipeline repairs
- Preparation of lightweight PBS foams with high ductility and impact toughness by foam injection molding
- Gamma-ray shielding investigation of nano- and microstructures of SnO on polyester resin composites: Experimental and theoretical study
- Experimental study on impact and flexural behaviors of CFRP/aluminum-honeycomb sandwich panel
- Normal-hexane treatment on PET-based waste fiber depolymerization process
- Effect of tannic acid chelating treatment on thermo-oxidative aging property of natural rubber
- Design, synthesis, and characterization of novel copolymer gel particles for water-plugging applications
- Influence of 1,1′-Azobis(cyclohexanezonitrile) on the thermo-oxidative aging performance of diolefin elastomers
- Characteristics of cellulose nanofibril films prepared by liquid- and gas-phase esterification processes
- Investigation on the biaxial stretching deformation mechanism of PA6 film based on finite element method
- Simultaneous effects of temperature and backbone length on static and dynamic properties of high-density polyethylene-1-butene copolymer melt: Equilibrium molecular dynamics approach
- Research on microscopic structure–activity relationship of AP particle–matrix interface in HTPB propellant
- Three-layered films enable efficient passive radiation cooling of buildings
- Electrospun nanofibers membranes of La(OH)3/PAN as a versatile adsorbent for fluoride remediation: Performance and mechanisms
- Preparation and characterization of biodegradable polyester fibers enhanced with antibacterial and antiviral organic composites
- Preparation of hydrophobic silicone rubber composite insulators and the research of anti-aging performance
- Surface modification of sepiolite and its application in one-component silicone potting adhesive
- Study on hydrophobicity and aging characteristics of epoxy resin modified with nano-MgO
- Optimization of baffle’s height in an asymmetric twin-screw extruder using the response surface model
- Effect of surface treatment of nickel-coated graphite on conductive rubber
- Experimental investigation on low-velocity impact and compression after impact behaviors of GFRP laminates with steel mesh reinforced
- Development and characterization of acetylated and acetylated surface-modified tapioca starches as a carrier material for linalool
- Investigation of the compaction density of electromagnetic moulding of poly(ether-ketone-ketone) polymer powder
- Experimental investigation on low-velocity-impact and post-impact-tension behaviors of GFRP T-joints after hydrothermal aging
- The repeated low-velocity impact response and damage accumulation of shape memory alloy hybrid composite laminates
- Exploring a new method for high-performance TPSiV preparation through innovative Si–H/Pt curing system in VSR/TPU blends
- Large-scale production of highly responsive, stretchable, and conductive wrapped yarns for wearable strain sensors
- Preparation of natural raw rubber and silica/NR composites with low generation heat through aqueous silane flocculation
- Molecular dynamics simulation of the interaction between polybutylene terephthalate and A3 during thermal-oxidative aging
- Crashworthiness of GFRP/aluminum hybrid square tubes under quasi-static compression and single/repeated impact
- Review Articles
- Recent advancements in multinuclear early transition metal catalysts for olefin polymerization through cooperative effects
- Impact of ionic liquids on the thermal properties of polymer composites
- Recent progress in properties and application of antibacterial food packaging materials based on polyvinyl alcohol
- Additive manufacturing (3D printing) technologies for fiber-reinforced polymer composite materials: A review on fabrication methods and process parameters
- Rapid Communication
- Design, synthesis, characterization, and adsorption capacities of novel superabsorbent polymers derived from poly (potato starch xanthate-graft-acrylamide)
- Special Issue: Biodegradable and bio-based polymers: Green approaches (Guest Editors: Kumaran Subramanian, A. Wilson Santhosh Kumar, and Venkatajothi Ramarao)
- Development of smart core–shell nanoparticles-based sensors for diagnostics of salivary alpha-amylase in biomedical and forensics
- Thermoplastic-polymer matrix composite of banana/betel nut husk fiber reinforcement: Physico-mechanical properties evaluation
- Special Issue: Electrospun Functional Materials
- Electrospun polyacrylonitrile/regenerated cellulose/citral nanofibers as active food packagings
Artikel in diesem Heft
- Research Articles
- Flame-retardant thermoelectric responsive coating based on poly(3,4-ethylenedioxythiphene) modified metal–organic frameworks
- Highly stretchable, durable, and reversibly thermochromic wrapped yarns induced by Joule heating: With an emphasis on parametric study of elastane drafts
- Molecular dynamics simulation and experimental study on the mechanical properties of PET nanocomposites filled with CaCO3, SiO2, and POE-g-GMA
- Multifunctional hydrogel based on silk fibroin/thermosensitive polymers supporting implant biomaterials in osteomyelitis
- Marine antifouling coating based on fluorescent-modified poly(ethylene-co-tetrafluoroethylene) resin
- Preparation and application of profiled luminescent polyester fiber with reversible photochromism materials
- Determination of pesticide residue in soil samples by molecularly imprinted solid-phase extraction method
- The die swell eliminating mechanism of hot air assisted 3D printing of GF/PP and its influence on the product performance
- Rheological behavior of particle-filled polymer suspensions and its influence on surface structure of the coated electrodes
- The effects of property variation on the dripping behaviour of polymers during UL94 test simulated by particle finite element method
- Experimental evaluation on compression-after-impact behavior of perforated sandwich panel comprised of foam core and glass fiber reinforced epoxy hybrid facesheets
- Synthesis, characterization and evaluation of a pH-responsive molecular imprinted polymer for Matrine as an intelligent drug delivery system
- Twist-related parametric optimization of Joule heating-triggered highly stretchable thermochromic wrapped yarns using technique for order preference by similarity to ideal solution
- Comparative analysis of flow factors and crystallinity in conventional extrusion and gas-assisted extrusion
- Simulation approach to study kinetic heterogeneity of gadolinium catalytic system in the 1,4-cis-polyisoprene production
- Properties of kenaf fiber-reinforced polyamide 6 composites
- Cellulose acetate filter rods tuned by surface engineering modification for typical smoke components adsorption
- A blue fluorescent waterborne polyurethane-based Zn(ii) complex with antibacterial activity
- Experimental investigation on damage mechanism of GFRP laminates embedded with/without steel wire mesh under low-velocity-impact and post-impact tensile loading
- Preparation and application research of composites with low vacuum outgassing and excellent electromagnetic sealing performance
- Assessing the recycling potential of thermosetting polymer waste in high-density polyethylene composites for safety helmet applications
- Mesoscale mechanics investigation of multi-component solid propellant systems
- Preparation of HTV silicone rubber with hydrophobic–uvioresistant composite coating and the aging research
- Experimental investigation on tensile behavior of CFRP bolted joints subjected to hydrothermal aging
- Structure and transition behavior of crosslinked poly(2-(2-methoxyethoxy) ethylmethacrylate-co-(ethyleneglycol) methacrylate) gel film on cellulosic-based flat substrate
- Mechanical properties and thermal stability of high-temperature (cooking temperature)-resistant PP/HDPE/POE composites
- Preparation of itaconic acid-modified epoxy resins and comparative study on the properties of it and epoxy acrylates
- Synthesis and properties of novel degradable polyglycolide-based polyurethanes
- Fatigue life prediction method of carbon fiber-reinforced composites
- Thermal, morphological, and structural characterization of starch-based bio-polymers for melt spinnability
- Robust biaxially stretchable polylactic acid films based on the highly oriented chain network and “nano-walls” containing zinc phenylphosphonate and calcium sulfate whisker: Superior mechanical, barrier, and optical properties
- ARGET ATRP of styrene with low catalyst usage in bio-based solvent γ-valerolactone
- New PMMA-InP/ZnS nanohybrid coatings for improving the performance of c-Si photovoltaic cells
- Impacts of the calcinated clay on structure and gamma-ray shielding capacity of epoxy-based composites
- Preparation of cardanol-based curing agent for underwater drainage pipeline repairs
- Preparation of lightweight PBS foams with high ductility and impact toughness by foam injection molding
- Gamma-ray shielding investigation of nano- and microstructures of SnO on polyester resin composites: Experimental and theoretical study
- Experimental study on impact and flexural behaviors of CFRP/aluminum-honeycomb sandwich panel
- Normal-hexane treatment on PET-based waste fiber depolymerization process
- Effect of tannic acid chelating treatment on thermo-oxidative aging property of natural rubber
- Design, synthesis, and characterization of novel copolymer gel particles for water-plugging applications
- Influence of 1,1′-Azobis(cyclohexanezonitrile) on the thermo-oxidative aging performance of diolefin elastomers
- Characteristics of cellulose nanofibril films prepared by liquid- and gas-phase esterification processes
- Investigation on the biaxial stretching deformation mechanism of PA6 film based on finite element method
- Simultaneous effects of temperature and backbone length on static and dynamic properties of high-density polyethylene-1-butene copolymer melt: Equilibrium molecular dynamics approach
- Research on microscopic structure–activity relationship of AP particle–matrix interface in HTPB propellant
- Three-layered films enable efficient passive radiation cooling of buildings
- Electrospun nanofibers membranes of La(OH)3/PAN as a versatile adsorbent for fluoride remediation: Performance and mechanisms
- Preparation and characterization of biodegradable polyester fibers enhanced with antibacterial and antiviral organic composites
- Preparation of hydrophobic silicone rubber composite insulators and the research of anti-aging performance
- Surface modification of sepiolite and its application in one-component silicone potting adhesive
- Study on hydrophobicity and aging characteristics of epoxy resin modified with nano-MgO
- Optimization of baffle’s height in an asymmetric twin-screw extruder using the response surface model
- Effect of surface treatment of nickel-coated graphite on conductive rubber
- Experimental investigation on low-velocity impact and compression after impact behaviors of GFRP laminates with steel mesh reinforced
- Development and characterization of acetylated and acetylated surface-modified tapioca starches as a carrier material for linalool
- Investigation of the compaction density of electromagnetic moulding of poly(ether-ketone-ketone) polymer powder
- Experimental investigation on low-velocity-impact and post-impact-tension behaviors of GFRP T-joints after hydrothermal aging
- The repeated low-velocity impact response and damage accumulation of shape memory alloy hybrid composite laminates
- Exploring a new method for high-performance TPSiV preparation through innovative Si–H/Pt curing system in VSR/TPU blends
- Large-scale production of highly responsive, stretchable, and conductive wrapped yarns for wearable strain sensors
- Preparation of natural raw rubber and silica/NR composites with low generation heat through aqueous silane flocculation
- Molecular dynamics simulation of the interaction between polybutylene terephthalate and A3 during thermal-oxidative aging
- Crashworthiness of GFRP/aluminum hybrid square tubes under quasi-static compression and single/repeated impact
- Review Articles
- Recent advancements in multinuclear early transition metal catalysts for olefin polymerization through cooperative effects
- Impact of ionic liquids on the thermal properties of polymer composites
- Recent progress in properties and application of antibacterial food packaging materials based on polyvinyl alcohol
- Additive manufacturing (3D printing) technologies for fiber-reinforced polymer composite materials: A review on fabrication methods and process parameters
- Rapid Communication
- Design, synthesis, characterization, and adsorption capacities of novel superabsorbent polymers derived from poly (potato starch xanthate-graft-acrylamide)
- Special Issue: Biodegradable and bio-based polymers: Green approaches (Guest Editors: Kumaran Subramanian, A. Wilson Santhosh Kumar, and Venkatajothi Ramarao)
- Development of smart core–shell nanoparticles-based sensors for diagnostics of salivary alpha-amylase in biomedical and forensics
- Thermoplastic-polymer matrix composite of banana/betel nut husk fiber reinforcement: Physico-mechanical properties evaluation
- Special Issue: Electrospun Functional Materials
- Electrospun polyacrylonitrile/regenerated cellulose/citral nanofibers as active food packagings

