Abstract
Deep eutectic solvents (DESs), a new class of green solvents, have attracted considerable attention for CO2 capture. In this study, we synthesised DESs using a superbase, 1,5-diazabicyclo [4.3.0] non-5-ene (DBN), as the hydrogen bond acceptor and a weak acid, diethylene glycol (DEG), as the hydrogen bond donor in different molar ratios to achieve efficient and reversible CO2 absorption. Experimental results indicate that DES2 (DBN:DEG = 1:2) exhibits optimal CO2 absorption performance (a maximum absorption capacity of 0.275 gCO2/gDES. Deep eutectic solvents at 30°C and flow rate of 30 mL/min). Nuclear magnetic resonance (NMR) spectroscopy reveals that DEG activated by DBN in DESs can react with CO2 to form carbonates, thereby enabling CO2 capture. In addition, monoethanolamine was added to the DESs as a promoter, thereby enhancing the CO2 absorption performance, with a maximum absorption capacity of 0.316 gCO2/gDES. 13C NMR and Fourier transform infrared analyses revealed that the amino and hydroxyl groups in the DESs can simultaneously react with CO2 to form carbamate and carbonate, achieving synergistic absorption.
1 Introduction
The combustion of fossil fuels generates enormous industrial waste, increasing CO2 concentration in the atmosphere. CO2 is one of the major greenhouse gases; it can absorb and emit infrared radiation, warming the atmosphere. This greenhouse effect is the primary driver of global warming and climate change. In addition, atmospheric CO2 dissolves in oceans, forming carbonic acid, thereby increasing ocean acidity and harms marine ecosystems [1–3]. Therefore, CO2 emissions have become a global issue. Consequently, various measures, including developing renewable energy, improving energy efficiency, promoting carbon capture and storage technologies, carbon capture and use as well as afforestation, are being implemented globally to reduce CO2 emissions and mitigate the greenhouse effect [4,5]. Carbon capture technologies can be classified according to the sequence of carbon capture and combustion processes, separation methodologies, and technological advancements. This classification is fundamentally determined by the sequence of carbon capture and combustion processes. They can be categorised into pre-combustion capture, post-combustion capture, and oxy-fuel combustion. Pre-combustion capture and oxy-fuel combustion require the addition of new equipment to process carbon-containing components on an existing industrial base, posing a major challenge to already established industrial lines. In contrast, post-combustion capture only requires CO2 capture from flue gases after combustion; thus, it is more convenient and suitable for industrialisation [6,7]. Post-combustion capture techniques include chemical absorption, membrane separation, chemical and physical adsorption, ionic liquid (IL) capture, and deep eutectic solvent (DES) capture [8]. In chemical absorption, alkanolamine aqueous solutions are used for industrial-grade CO2 removal, primarily via the reaction of the amino groups in alkanolamine solutions with CO2 [9,10]. However, because of the high volatility, strong corrosiveness, and high energy for solvent regeneration during amine scrubbing, considerable efforts are required to develop promising and effective CO2 absorbents to replace alkanolamines [11,12].
ILs have attracted considerable attention as replacements for alkanolamines in CO2 absorption. ILs effectively capture CO2 by adjusting the ionic efficacy of anions and cations. Because of their wide liquid range, flexible DES design, low vapour pressure, broad temperature range, and high thermal stability, ILs can replace alkanolamines in CO2 capture [13,14,15]. Bates et al. first synthesised a functionalised ILs containing –NH2 group, i.e., [C3H7NH2-Bmin][BF4]. At ambient temperature and pressure, the CO2 absorption rate of [C3H7NH2-Bmim][BF4] was 7.4 wt%; however, its high viscosity hindered mass transfer [16]. Combining ILs with other organic solvents is considered a feasible approach. Recently, protic ionic liquids (PILs), created by mixing superbases and weak acids, effectively capture CO2 due to their high CO2 attraction and low viscosity. Typically, such mixtures are formed by neturalising strong proton acceptors, such as 1,8-diazabicyclo[5.4.0]undec-7-ene (DBU) and 1,5-diazabicyclo[4.3.0]non-5-ene (DBN), and weak proton donors, such as pyrrole, imidazole, phenol, and ethylene glycol (EG) [17,18]. Zhang et al. synthesised PIL-based DESs by adding DBN to DESs prepared by mixing imidazole and BmimCl at different molar ratios. They reported that the synergistic action of cations and anions facilitated CO2 capture, with the protonated superbase playing a pivotal role in the absorption process. Smith et al. designed DES systems by mixing Lewis or Brønsted acids as hydrogen bond donors (HBDs) and bases as hydrogen bond acceptors (HBAs) [19,20]. These systems can include various ions or molecules, and if hydrogen bonding is prominent, mixtures of superbases and weak acids seem more suitable for DESs. DESs exhibit structural parallels to ILs in physicochemical properties, while demonstrating distinctive merits including simple preparative methodologies, non-toxicity, biodegradability, 100% atomic efficiency, and high product purity. Based on these advantages, DESs are potential CO2-absorbent materials [21–23]. Fu et al. [24] signed and synthesised super basic DES systems – [DBUH][MLU]-EG and [DBUH]2[MLU]-EG. They investigated the single and dual active centres of these two super basic DES systems for CO2 capture. Results showed that DBUH+ could activate EG to absorb CO2, indicating CO₂ absorption by DES via amino and hydroxyl functional groups. Ruan et al. [25] designed and synthesised DES systems formed by DBN and Tz. They reported that [2DBN:Tz] had the highest weight capacity of 0.19 gCO2/gDES at 25°C. The DES systems absorb CO2 through the synergistic action of DBN molecules and Tz ions activated by DBN.
According to the literature, DESs comprising superbases and weak acids exhibit high CO2 capture performance. Therefore, we design a DES system for CO2 capture using a superbase – DBN and a weak acid – diethylene glycol (DEG) – as raw materials. The CO2 capture capacity of the DESs system was measured under different conditions. Based on absorption experiments and nuclear magnetic resonance (NMR) analysis, the CO2 absorption mechanism was proposed and its reversible decarbonisation was analysed according to the absorption-DES-desorption cycle performance. In addition, monoethanolamine (MEA) was added to DES as a promoter. Based on absorption experiments, 13C NMR data, Fourier transform infrared (FTIR) analysis, and the promotion mechanism was proposed.
2 Experimental sections
2.1 Materials
(Table 1)
Materials used in this work
| Chemical name | CAS | MW (g mol−1) | Purity (%) | Supplier |
|---|---|---|---|---|
| DBN | 3001-72-7 | 124.18 | 98 | Macklin |
| DEG | 111-46-6 | 106.11 | 98 | Macklin |
| MEA | 141-43-5 | 61.08 | 98 | Macklin |
| CO2 | 124-38-9 | 44.01 | 99.99 | Guizhou Sanhe Gas Co., Ltd, China |
| N2 | 7729-37-9 | 28.00 | 99.999 | Guizhou Sanhe Gas Co., Ltd, China |
2.2 Analytical testing equipment
Differential scanning calorimeter, Netzsch DSC2 14 from Germany; thermogravimetric (TG) analyser, Netzsch STA 449 F3 from Germany; NMR spectrometer, Bruker Ascend TM 600 MHz from Germany; FTIR spectrometer, Thermo Fisher Scientific Nicolet iS20 from the United States; electronic balance, AR2 24CN, accuracy of ±2 × 10−4 g, Ohaus Instrument Co., Ltd.
2.3 DES preparation
According to the literature [26], DESs was synthesised using the mixed heating method. DBN and DEG were poured into a flask at molar ratios of 1:2, 1:3, and 1:4. The flask was sealed, and the mixture was magnetically stirred in a water bath at 30°C until a uniform transparent liquid was formed. The prepared binary DESs were cooled at room temperature for 24 h. The resulting DESs with molar ratios of 1:1, 1:2, 1:3, and 1:4 are called DES1, DES2, DES3, and DES4, respectively. Using a similar method, MEA was added to the prepared binary DESs with different molar ratios.
2.4 Characterisation
The NMR spectra of the DESs were recorded before and after CO2 absorption. FTIR spectroscopy was performed in the attenuated total reflection mode to determine the CO2 absorption mechanism. The thermal stability of the absorbents was determined using TG analysis. The temperature was set from 30 to 500°C at a 10°C/min heating rate under a nitrogen atmosphere.
2.5 CO2 absorption and DES desorption experiment
According to the literature [27], the CO2 absorption experiment was performed using a gravimetric method. A DES was poured into a glass container, which was then placed in a water bath, ensuring the DES was below the water surface. Subsequently, CO2 was introduced into the DES at atmospheric pressure. Using an electronic analytical balance with a precision of 0.005 g, the weight of the glass container was measured every 10 min to obtain the amount of CO2 captured. Absorption equilibrium was reached when the weight difference between two consecutive measurements was less than 0.005 g. For the CO2 DES desorption experiment, a CO2 saturated DES was placed in a water bath and N2 was introduced at 70°C for DES desorption until the weight returned to the pre-absorption level.
3 Results and discussion
3.1 Effect of DBN:DEG ratio on CO2 absorption
The absorption curves of DES1(DBN:DEG = 1:1), DES2(DBN:DEG = 1:2), DES3(DBN:DEG = 1:3), and DES4(DBN:DEG = 1:4) were analysed (Figure 1a). At atmospheric pressure, a temperature of 30°C and a duration of 60 min, DES2 exhibited the maximum CO2 absorption capacity (0.275 gCO2/gDES; Figure 1a). Next, MEA was added to DES1 as a promoter (Figure 1b). The absorption capacity of DES1 containing MEA increased significantly because of the active absorption sites of MEA. However, increasing the MEA content did not continuously improve the CO2 absorption capacity because MEA tightened the hydrogen bond network of the DES, thereby increasing steric hindrance and hindering CO2 binding with the DES. Therefore, there is a balance, and DES containing MEA exhibited the maximum absorption capacity (0.316 gCO2/gDES) when [DBN][DEG]2-MEA = 1:2 (hereinafter, DES2-MEA).
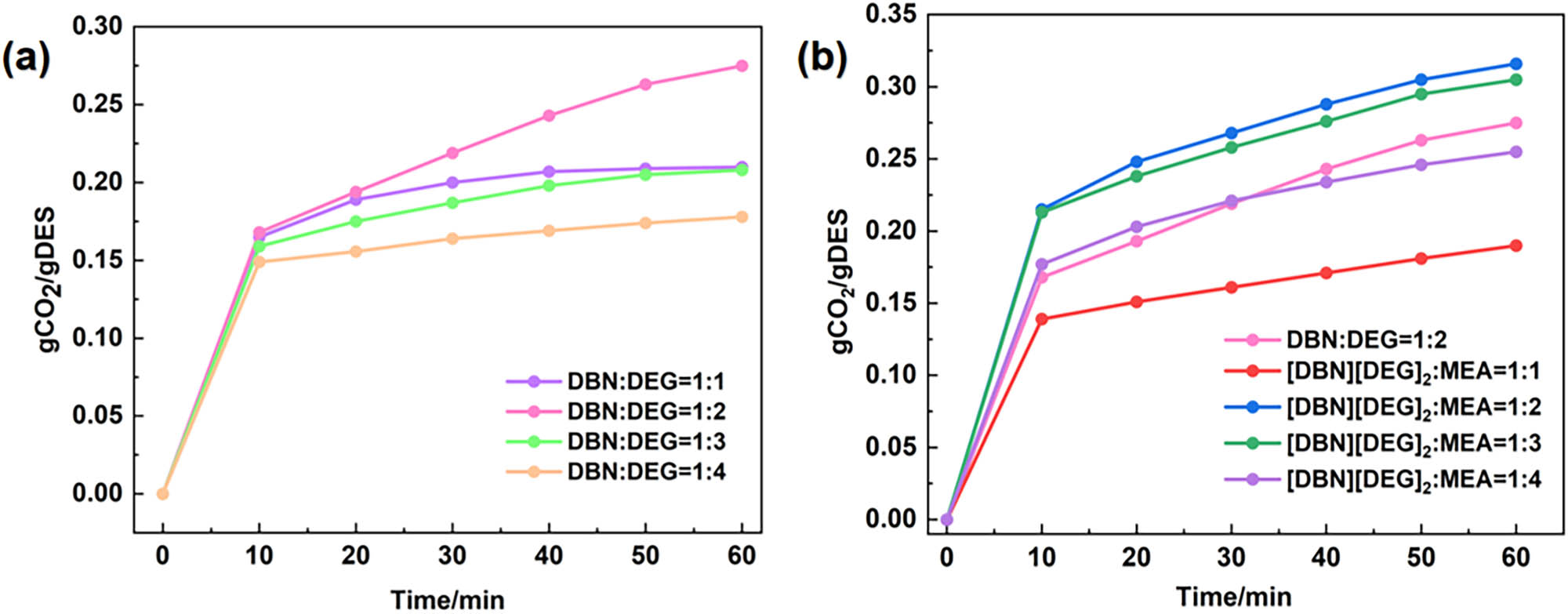
Absorption rate curves of different absorbents at 30°C and a flow rate of 30 mL/min.
3.2 Effect of temperature on CO2 absorption
We investigated the performance of DES2 for CO2 absorption at atmospheric pressure within 30–60°C. As shown in Figure 2a, as the temperature increased from 30 to 60°C, the CO2 absorption capacity decreased from 0.275 to 0.164 gCO2/gDES due to the exothermic nature of CO2 absorption. An increase in temperature significantly reduced the CO2 absorption performance, indicating a substantial effect of temperature on CO2 absorption by DESs. Notably, the absorption rate showed a marked increase with rising temperature, leading to a shorter time to reach absorption equilibrium, as higher temperature favours mass transfer. Given that CO2 absorption is an exothermic reaction, higher temperatures also facilitate DESs desorption, offering great potential for the regeneration and reusability of absorbents.
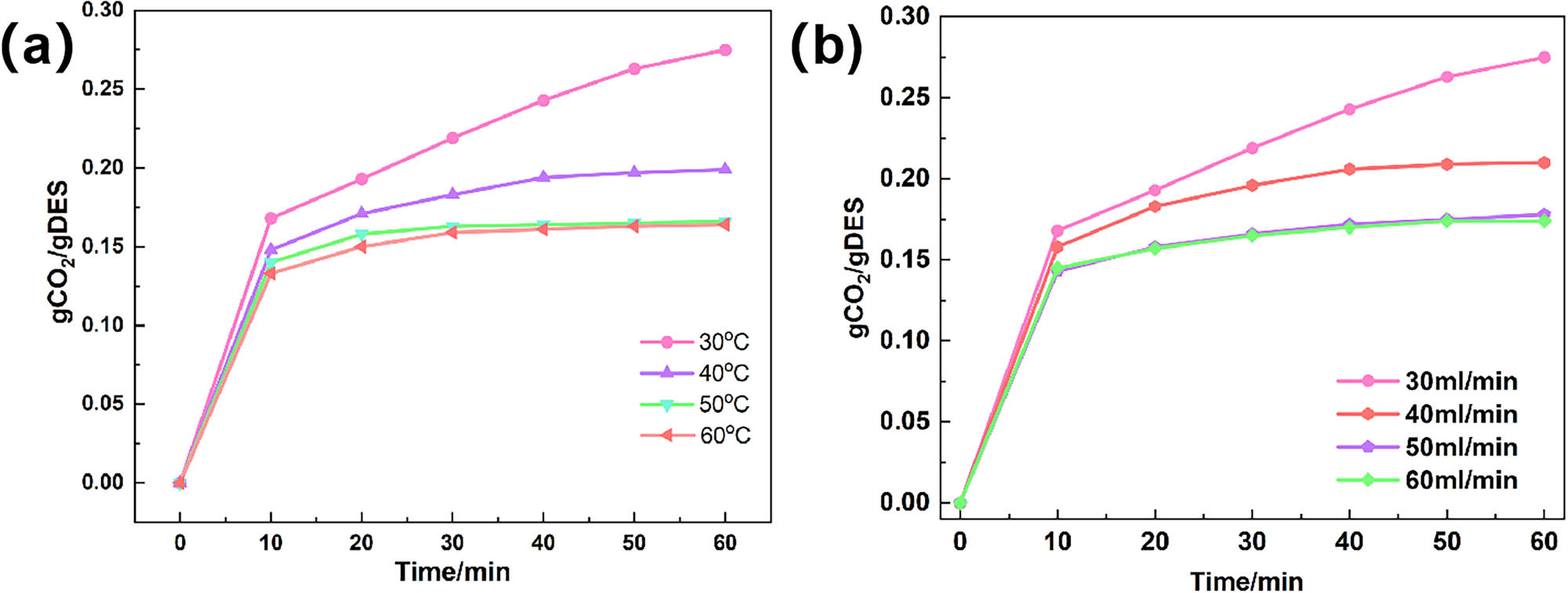
Absorption rate curves of DES2 under (a) different temperatures and (b) different flow rates.
3.3 Effect of velocity of flow on CO2 absorption
We investigated the performance of DES2 for CO2 absorption over 1 h at flow rates ranging from 30 to 70 mL/min (Figure 2b). As the flow rate increases, the performance of DES2 decreases significantly. Specifically, the CO2 absorption capacity decreases from 0.275 gCO2/gDES at a flow rate of 30 mL/min to 0.168 gCO2/gDES at a flow rate of 70 mL/min. The decrease in CO2 absorption by DES is attributed to the increased flow rate, which reduces the time available for gas molecules to diffuse into the absorbent. Consequently, the CO2 adsorption capacity decreases as the residence time decreases. This indicates that flow rate significantly influences CO₂ absorption by DESs [28].
3.4 Effect of moisture content on CO2 absorption
According to the absorption curve in Figure 3, a DES2 with a higher water content exhibits a more pronounced reduction in CO2 absorption efficiency, reaching saturation earlier. The addition of water to DESs reduces their viscosity, however, disrupting their hydrogen bond network and diminishing their absorption capacity. The reason in this study is that water competes with DEG for the activation sites of DBN, significantly reducing effective binding sites for CO2 absorption in DESs. Moreover, the reaction product of CO2 in DESs is aminoformate. Excess water in a DES system reacts with aminoformate to produce carbonate, which decomposes into water and CO2, thereby reducing the CO2 absorption capacity. As depicted in Figure 4, the variation in moisture content before and after DES absorption is minimal.
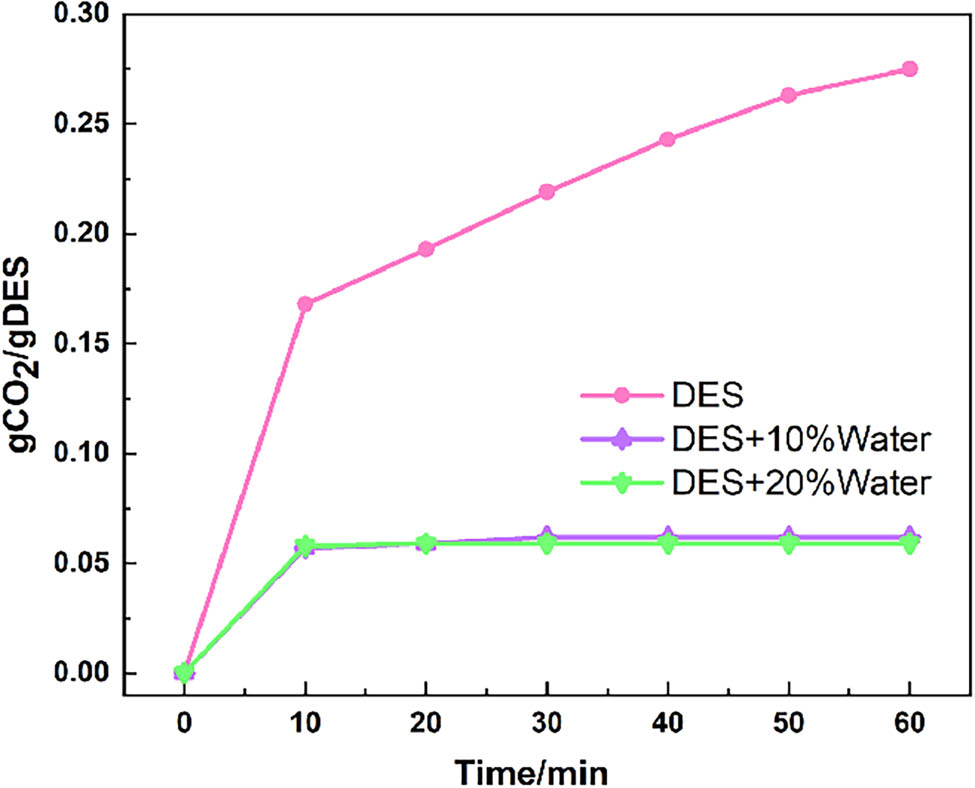
Absorption rate curves of DES2 under different water contents.

Moisture content of DESs before and after CO2 absorption.
3.5 Cyclic performance of DES
3.5.1 Thermal stability of DES
Figure 5a shows the TG curves of the synthesised DESs system at different molar ratios. The results indicate that DES2 starts to lose weight at 327.13 K and completely decomposes at around 442.73 K, with no significant change in mass observed with increasing temperature. DES2 exhibits a 5% mass loss at a temperature of 365.23 K, demonstrating good thermal stability. Meanwhile, DES3 and DES4 exhibit 5% mass loss at temperatures of 408.78 and 435.66 K, respectively, indicating enhanced stability with higher DEG contents. The main reason for this stability is the formation of a complex hydrogen bond network between HBA and HBD. Thermal stability is a crucial parameter for assessing an absorbent’s performance in practical applications, as higher thermal stability facilitates effective DESs desorption and recycling of the absorbent [22,29].

(a) Thermogravimetric analysis curves of DES2 at different molar ratios; (b) cyclic diagram of DES2 under N2 purge at 343.15 K.
3.5.2 DES cyclic performance
In industrial applications, the recyclability of decarbonising agents is crucial for their promotion and use. Excellent recyclability not only reduces production costs and emissions but also enhances economic feasibility. We investigated the cyclic absorption properties of DES2. As shown in Figure 5b, after four absorption-DES-desorption cycles, the absorption capacity remained at 0.251 gCO2/gDES. However, the absorption performance slightly decreased, indicating that some CO2 was not completely DES desorbed from the system. This is because the carbonates formed by DES2 and CO2 are relatively stable and do not completely decompose during regeneration, leaving a small amount of CO2 retained in DES2. Overall, after four cycles, DES2 maintained a stable and considerable absorption capacity, indicating that DESs have good absorption capacity and reusability [30,31].
3.6 Mechanism of CO2 absorption by DES
To explore the CO2 absorption mechanism of DES and the promoting effect of MEA on CO2 absorption, 13C NMR spectroscopy was used to characterise DESs before and after CO2 absorption. Considering DES2 and DES2-MEA as examples, the absorption mechanism of the synthesised DES system was studied (Figure 6b). After CO2 absorption, new peaks appeared at 63.35 and 156.7 ppm in DES2, attributable to the carbonate formed by the interaction between DEG and CO2 and the carbomethoxy (RO–COO–) group formed by the reaction of DEG with CO2, respectively. This indicates that the hydroxyl group of DEG is activated under the influence of DBN, enabling it to absorb CO2 [32]. As shown in Figure 6a, DES2-MEA exhibits new peaks at 59.85 and 162.14 ppm after CO2 absorption, attributable to the carbonate formed by the interaction between DEG and CO2 and the carbamate (–NH–COO–) formed by the interaction between CO2 and MEA, respectively [33].

13C NMR carbon spectra of different DESs before and after CO₂ absorption.
To elucidate the promoting mechanism of MEA, FTIR analysis was used to study the CO2 absorption mechanism of DES containing MEA (Figure 7). Comparing the FTIR spectra before and after CO2 absorption, new peaks appear at 1569.74 and 816.66 cm−1, attributable to the asymmetric stretching and bending vibrations of –COO– in –NHCOO–, respectively. The peak at 2864.7 cm−1 that corresponds to –NH– weakens, indicating carbamate generation. The disappearance of the peak at 3352.2 cm−1 that corresponds to the hydroxyl group after CO2 absorption suggests CO2 consumption, while the peak at 1299.82 cm−1 is attributed to the stretching vibration of carbonate, consistent with the previous 13C NMR analysis.

Infrared spectra of DES2-MEA before and after CO2 absorption.
Based on the above analysis, we propose a possible mechanism (Figure 8). The possible absorption mechanism of DESs is illustrated in Scheme 1: DBN deprotonates the hydroxyl group of DEG, leading to the formation of the HO–CH2–O–CH2–O− anion, which then combines with CO2 to form carbonate [32]. In the DES with added MEA, dual absorption of hydroxyl and amino groups occurs, as illustrated in Scheme 2. The activated DEG by DBN reacts with CO2, and the amino group in MEA reacts with CO2, achieving cooperative CO2 capture through amine-hydroxyl dual active sites in the system, thereby enhancing the CO2 absorption performance. The addition of MEA promotes the CO2 absorption capacity of DES.

Possible reaction mechanisms of CO2 with DES2 and DES2-MEA used in this work.
3.7 Comparison of CO2 absorption capacity with reported absorbents
To further evaluate the CO2 absorption potential of DBN/DEG, we compared the CO2 absorption capacity in this study with other absorbents reported in the literature. As shown in Table 2, the DESs employed in this work demonstrate competitive CO2 absorption capacities compared with most absorbents under relatively mild absorption conditions, indicating significant potential for efficient CO2 capture.
Comparison of CO2 capacity with other reported absorbents
| Absorbents | PCO2 (bar) | T (oC) | CO2 capacity (gCO2/g solvent) | Reference |
|---|---|---|---|---|
| DBN:DEG (1:1) | 1 | 30 | 0.19 | This work |
| DBN:DEG (1:2) | 1 | 30 | 0.275 | This work |
| [DBN][DEG]2:MEA (1:2) | 1 | 30 | 0.316 | This work |
| DBN/EG/MEA (3/1/1) | 1 | 40 | 0.248 | [32] |
| DBN/EG/MEA (1/1/3) | 1 | 40 | 0.267 | [32] |
| [DBUH][Car]:EG (1:2) | 1 | 25 | 1a | [31] |
| [EMIM][2-CNpyr]:EG (1:2) | 1 | 25 | 2.59a | [30] |
| K-GLY+MEA | 1 | 25 | 0.256 | [34] |
| [DBN]:[Tz] (2:1) | 1 | 25 | 0.19 | [25] |
| ChCl-DEA (1:6) | 1 | 30 | 0.15 | [35] |
| [DBUH][4-F-PhO] (1:5) | 1 | 25 | 0.9a | [23] |
| DBN-BmimCl-Im (1:1:1) | 1 | 25 | 1.02a | [36] |
| ChCl:MEA (1:2) | 1 | 30 | 23b | [37] |
| ChCl:MDEA (1:2) | 10 | 30 | 0.0896 | [38] |
| Be-EG-DEA (1:5:8) | 1 | 30 | 0.207 | [39] |
| Sodium carbonate-based solid sdsorbents | 1 | 0 | 10.56c | [40] |
| The CaO–MgO-based absorber | 1 | 650 | 0.768 | [41] |
aCO2 absorption capacity (mol CO2/mol solvent). bMass ratio. cCO2 absorption capacity (cm3 CO2/g absorbent).
4 Conclusions
In this study, a DES system comprising DBN and DEG as the HBA and HBD, respectively, was DES designed and synthesised. The CO2 absorption capacity of DESs was measured using a gravimetric method. The effects of adjusting the HBD and HBA molar ratio, water content, temperature, and flow rate on CO2 absorption performance were investigated. Optimal absorption conditions were determined; a molar ratio of 1:2, temperature of 30°C, and flow rate of 30 mL/min achieved the maximum CO2 absorption capacity (0.275 gCO2/gDES). The addition of water inhibited CO2 absorption. The reusability of DESs was also studied. After four absorption-DES-desorption cycles, DESs maintained a stable and considerable absorption capacity. Proton NMR spectroscopy indicated that DBN in DESs activated the hydroxyl groups of DEG, enabling them to react with CO2 to form carbonate and consequently absorb CO2. Furthermore, MEA was added to DESs as a promoter, achieving an optimal absorption capacity of 0.316 gCO2/gDES. According to NMR and FTIR analyses, activated DEG and MEA in DESs could simultaneously react with CO2 to form carbamate and carbonate. The addition of MEA increased the CO2 absorption capacity. Therefore, DESs can be considered promising absorbents. Moreover, the addition of the promoter MEA a new approach for DES designing efficient DESs for CO2 capture.
Acknowledgments
This work was financially supported by Science and Technology Department of Guizhou Province in China (project number: K22-0110-034).
-
Funding information: This work was financially supported by Science and Technology Department of Guizhou Province in China (project number: K22-0110-034).
-
Author contributions: Shuie Li: methodology; Xuzhong Zhou: investigation; Chengfang Liu: conceptualisation and methodology; Shihong Guo: data curation, software, writing – original and draft, writing – review. All the authors approved the final manuscript version submitted.
-
Conflict of interest: The authors state no conflict of interest.
-
Informed consent: Not applicable.
-
Ethical approval: The conducted research is not related to either human or animal use.
-
Supporting information: 1H NMR hydrogen spectra of DBN, DEG, and DBN-DEG(PDF).
-
Data availability statement: The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
References
[1] Chen Y, Mu T. Conversion of CO2 to value-added products mediated by ionic liquids. Green Chem. 2019;21:2544–74. 10.1039/c9gc00827f.Search in Google Scholar
[2] Feng Z, Wang N, Li X, Li Z, Li K, Jin H, et al. Process optimization, energy consumption analysis, and environmental assessment of total CO2 capture from syngas based on [NEMH][Ac] protic ionic liquid. Int J Greenh Gas Control. 2024;131:123–35. 10.1016/j.ijggc.2023.104037.Search in Google Scholar
[3] Tong J, Zhao Y, Huo F, Guo Y, Liang X, von Solms N, et al. The dynamic behavior and intrinsic mechanism of CO2 absorption by amino acid ionic liquids. Phys Chem Chem Phys. 2021;23:3246–55. 10.1039/d0cp05735e.Search in Google Scholar PubMed
[4] Wang J, Song Z, Cheng H, Chen L, Deng L, Qi Z. Multilevel screening of ionic liquid absorbents for simultaneous removal of CO2 and H2S from natural gas. Sep Purif Technol. 2020;248:117053. 10.1016/j.seppur.2020.117053.Search in Google Scholar
[5] Bui M, Adjiman CS, Bardow A, Anthony EJ, Boston A, Brown S, et al. Carbon capture and storage (CCS): the way forward. Energy Environ Sci. 2018;11:1062–176. 10.1039/c7ee02342a.Search in Google Scholar
[6] Subramanian N, Madejski P. Analysis of CO2 capture process from flue-gases in combined cycle gas turbine power plant using post-combustion capture technology. Energy. 2023;282:128311. 10.1016/j.energy.2023.128311.Search in Google Scholar
[7] Qian W, Hao J, Zhu M, Sun P, Zhang K, Wang X, et al. Development of green solvents for efficient post-combustion CO2 capture with good regeneration performance. J CO2 Util. 2022;59:101955. 10.1016/j.jcou.2022.101955.Search in Google Scholar
[8] Kassim MA, Sulaiman NA, Yusoff R, Aroua MK. Non-aqueous solvent mixtures for CO2 capture: choline hydroxide-based deep eutectic solvents absorbent performance at various temperatures and pressures. Sustainability. 2023;15(12):9191. 10.3390/su15129191.Search in Google Scholar
[9] Yan C, Wang C-H, Lin M, Bhalothia D, Yang S-S, Fan G-J, et al. Local synergetic collaboration between Pd and local tetrahedral symmetric Ni oxide enables ultra-high-performance CO2 thermal methanation. J Mater Chem A. 2020;8:12744–56. 10.1039/d0ta02957b.Search in Google Scholar
[10] Chen H, Tsai T-C, Tan C-S. CO2 capture using amino acid sodium salt mixed with alkanolamines. Int J Greenh Gas Control. 2018;79:127–33. 10.1016/j.ijggc.2018.10.002.Search in Google Scholar
[11] Khalifa O, Alkhatib III, Bahamon D, Alhajaj A, Abu-Zahra MRM, Vega LF. Modifying absorption process configurations to improve their performance for post-combustion CO2 capture – what have we learned and what is still missing? Chem Eng J. 2022;430:133096. 10.1016/j.cej.2021.133096.Search in Google Scholar
[12] Vega F, Baena-Moreno FM, Gallego Fernández LM, Portillo E, Navarrete B, Zhang Z. Current status of CO2 chemical absorption research applied to CCS: towards full deployment at industrial scale. Appl Energy. 2020;260:114313. 10.1016/j.apenergy.2019.114313.Search in Google Scholar
[13] Wang J, Song Z, Cheng H, Chen L, Deng L, Qi Z. Computer-aided design of ionic liquids as absorbent for gas separation exemplified by CO2 capture cases. ACS Sustain Chem Eng. 2018;6:12025–35. 10.1021/acssuschemeng.8b02321.Search in Google Scholar
[14] Zhang P, Ma X, Tu Z, Zhang X, Hu X, Wu Y. Constructing ether-rich and carboxylate hydrogen bonding sites in protic ionic liquids for efficient and simultaneous membrane separation of H2S and CO2 from CH4. Green Energy Environ. 2024;10:123–35. 10.1016/j.gee.2024.05.009.Search in Google Scholar
[15] Li F, Bai Y, Zeng S, Liang X, Wang H, Huo F, et al. Protic ionic liquids with low viscosity for efficient and reversible capture of carbon dioxide. Int J Greenh Gas Control. 2019;90:102801. 10.1016/j.ijggc.2019.102801.Search in Google Scholar
[16] Zhang R, Ke Q, Zhang Z, Zhou B, Cui G, Lu H. Tuning functionalized ionic liquids for CO2 capture. Int J Mol Sci. 2022;23:11401. 10.3390/ijms231911401.Search in Google Scholar PubMed PubMed Central
[17] Xiong W, Shi M, Peng L, Zhang X, Hu X, Wu Y. Low viscosity superbase protic ionic liquids for the highly efficient simultaneous removal of H2S and CO2 from CH4. Sep Purif Technol. 2021;263:118417. 10.1016/j.seppur.2021.118417.Search in Google Scholar
[18] Wang X, Wu C, Yang D. CO2 absorption mechanism by diamino protic ionic liquids (DPILs) containing azolide anions. Processes. 2021;9:1023. 10.3390/pr9061023.Search in Google Scholar
[19] Smith EL, Abbott AP, Ryder KS. Deep eutectic solvents (DESs) and their applications. Chem Rev. 2014;114:11060–82. 10.1021/cr300162p.Search in Google Scholar PubMed
[20] Hansen BB, Spittle S, Chen B, Poe D, Zhang Y, Klein JM, et al. Deep eutectic solvents: a review of fundamentals and applications. Chem Rev. 2021;121:1232–85. 10.1021/acs.chemrev.0c00385.Search in Google Scholar PubMed
[21] Jiang B, Ma J, Yang N, Huang Z, Zhang N, Tantai X, et al. Superbase/acylamido-based deep eutectic solvents for multiple-site efficient CO2 absorption. Energy Fuels. 2019;33:7569–77. 10.1021/acs.energyfuels.9b01361.Search in Google Scholar
[22] Shi S, Li S, Liu X. Mechanism study of imidazole-type deep eutectic solvents for efficient absorption of CO2. ACS Omega. 2022;7:48272–81. 10.1021/acsomega.2c06437.Search in Google Scholar PubMed PubMed Central
[23] Wang Z, Chen M, Lu B, Zhang S, Yang D. Effect of hydrogen bonds on CO2 capture by functionalized deep eutectic solvents derived from 4-fluorophenol. ACS Sustain Chem Eng. 2023;11:6272–9. 10.1021/acssuschemeng.2c07590.Search in Google Scholar
[24] Fu H, Wang X, Sang H, Liu J, Lin X, Zhang L. Highly efficient absorption of carbon dioxide by EG-assisted DBU-based deep eutectic solvents. J CO2 Util. 2021;43:101372. 10.1016/j.jcou.2020.101372.Search in Google Scholar
[25] Ruan J, Ye X, Wang R, Chen L, Deng L, Qi Z. Experimental and theoretical study on efficient CO2 absorption coordinated by molecules and ions of DBN and 1,2,4-triazole formed deep eutectic solvents. Fuel. 2023;334:126709. 10.1016/j.fuel.2022.126709.Search in Google Scholar
[26] Li P, Zhang Z, Zhang X, Li K, Jin Y, Wu W. DES: their effect on lignin and recycling performance. RSC Adv. 2023;13:3241–54. 10.1039/d2ra06033g.Search in Google Scholar PubMed PubMed Central
[27] Mei M, Hu X, Song Z, Chen L, Deng L, Qi Z. CO2 capture by 1-ethyl-3-methylimidazolium acetate: Solubility at low pressure and quantification of chemisorption and physisorption. J Mol Liq. 2022;348:118036. 10.1016/j.molliq.2021.118036.Search in Google Scholar
[28] Zhang Y, Zhu C, Fu T, Gao X, Ma Y, Li HZ. CO2 absorption and desorption performance by ChCl-MEA-PZ deep eutectic solvent aqueous solutions. Sep Purif Technol. 2024;330:125275. 10.1016/j.seppur.2023.125275.Search in Google Scholar
[29] Jangir AK, Bhawna B, Verma G, Pandey S, Kuperkar K. Design and thermophysical characterization of betaine hydrochloride-based deep eutectic solvents as a new platform for CO2 capture. N J Chem. 2022;46:5332–45. 10.1039/d1nj05373f.Search in Google Scholar
[30] Lee Y-Y, Penley D, Klemm A, Dean W, Gurkan B. Deep eutectic solvent formed by imidazolium cyanopyrrolide and ethylene glycol for reactive CO2 separations. ACS Sustain Chem Eng. 2021;9:1090–8. 10.1021/acssuschemeng.0c07217.Search in Google Scholar
[31] Wang Z, Wang Z, Huang X, Yang D, Wu C, Chen J. Deep eutectic solvents composed of bio-phenol-derived superbase ionic liquids and ethylene glycol for CO2 capture. Chem Commun. 2022;58:2160–3. 10.1039/d1cc06856c.Search in Google Scholar PubMed
[32] Yue W, Han W, Yuan M, Zhou X, Fu H. Three-component CO2 binding organic liquids for efficient and reversible CO2 capture: effect of molar ratio of component on mechanism. J Mol Liq. 2024;399:124400. 10.1016/j.molliq.2024.124400.Search in Google Scholar
[33] Cheng J, Wu C, Gao W, Li H, Ma Y, Liu S, et al. CO2 absorption mechanism by the deep eutectic solvents formed by monoethanolamine-based protic ionic liquid and ethylene glycol. Int J Mol Sci. 2022;23:1893. 10.3390/ijms23031893.Search in Google Scholar PubMed PubMed Central
[34] Jo J, Park J, Kwon S, Park M, Jung J, Yoo Y, et al. Revolutionizing CO2 capture: molecular complexes of deep eutectic solvents with enhanced hydrogen bond accepting interaction for superior absorption performance. J Environ Chem Eng. 2023;11:110108. 10.1016/j.jece.2023.110108.Search in Google Scholar
[35] Liu X, Ao Q, Shi S, Li S. CO2 capture by alcohol ammonia based deep eutectic solvents with different water content. Mater Res Express. 2022;9:045601. 10.1088/2053-1591/ac47c6.Search in Google Scholar
[36] Zhang N, Huang Z, Zhang H, Ma J, Jiang B, Zhang L. Highly efficient and reversible CO2 capture by task-specific deep eutectic solvents. Ind Eng Chem Res. 2019;58:13321–9. 10.1021/acs.iecr.9b02041.Search in Google Scholar
[37] Li Z, Wang L, Li C, Cui Y, Li S, Yang G, et al. Absorption of carbon dioxide using ethanolamine-based deep eutectic solvents. ACS Sustain Chem Eng. 2019;7:10403–14. 10.1021/acssuschemeng.9b00555.Search in Google Scholar
[38] Haider MB, Jha D, Marriyappan Sivagnanam B, Kumar R. Thermodynamic and kinetic studies of CO2 capture by glycol and amine-based deep eutectic solvents. J Chem Eng Data. 2018;63(8):2671–80.10.1021/acs.jced.8b00015Search in Google Scholar
[39] Liu C, Zhou X, Guo S, Li S. Study on carbon dioxide capture using ternary betaine-based deep eutectic solvents. ACS Omega. 2024;9:51031–9. 10.1021/acsomega.4c05705.Search in Google Scholar PubMed PubMed Central
[40] Liu K, Adili G, Ding Z, Tuergong N, Shi J, Tuersun A, et al. Synthesis of sodium carbonate based solid adsorbents and their CO2 adsorption performance. ChemistrySelect. 2024;9:e202403048. 10.1002/slct.202403048.Search in Google Scholar
[41] Rajamathi R, Bhojaraj B, Nethravathi C. Porous CaO–MgO nanostructures for CO2 capture. ACS Appl Nano Mater. 2021;4:10969–75. 10.1021/acsanm.1c02428.Search in Google Scholar
© 2025 the author(s), published by De Gruyter
This work is licensed under the Creative Commons Attribution 4.0 International License.
Articles in the same Issue
- Research Articles
- Phytochemical investigation and evaluation of antioxidant and antidiabetic activities in aqueous extracts of Cedrus atlantica
- Influence of B4C addition on the tribological properties of bronze matrix brake pad materials
- Discovery of the bacterial HslV protease activators as lead molecules with novel mode of action
- Characterization of volatile flavor compounds of cigar with different aging conditions by headspace–gas chromatography–ion mobility spectrometry
- Effective remediation of organic pollutant using Musa acuminata peel extract-assisted iron oxide nanoparticles
- Analysis and health risk assessment of toxic elements in traditional herbal tea infusions
- Cadmium exposure in marine crabs from Jiaxing City, China: Insights into health risk assessment
- Green-synthesized silver nanoparticles of Cinnamomum zeylanicum and their biological activities
- Tetraclinis articulata (Vahl) Mast., Mentha pulegium L., and Thymus zygis L. essential oils: Chemical composition, antioxidant and antifungal properties against postharvest fungal diseases of apple, and in vitro, in vivo, and in silico investigation
- Exploration of plant alkaloids as potential inhibitors of HIV–CD4 binding: Insight into comprehensive in silico approaches
- Recovery of phenylethyl alcohol from aqueous solution by batch adsorption
- Electrochemical approach for monitoring the catalytic action of immobilized catalase
- Green synthesis of ZIF-8 for selective adsorption of dyes in water purification
- Optimization of the conditions for the preparation of povidone iodine using the response surface methodology
- A case study on the influence of soil amendment on ginger oil’s physicochemical properties, mineral contents, microbial load, and HPLC determination of its vitamin level
- Removal of antiviral favipiravir from wastewater using biochar produced from hazelnut shells
- Effect of biochar and soil amendment on bacterial community composition in the root soil and fruit of tomato under greenhouse conditions
- Bioremediation of malachite green dye using Sargassum wightii seaweed and its biological and physicochemical characterization
- Evaluation of natural compounds as folate biosynthesis inhibitors in Mycobacterium leprae using docking, ADMET analysis, and molecular dynamics simulation
- Novel insecticidal properties of bioactive zoochemicals extracted from sea urchin Salmacis virgulata
- Elevational gradients shape total phenolic content and bioactive potential of sweet marjoram (Origanum majorana L.): A comparative study across altitudinal zones
- Study on the CO2 absorption performance of deep eutectic solvents formed by superbase DBN and weak acid diethylene glycol
- Preparation and wastewater treatment performance of zeolite-modified ecological concrete
- Multifunctional chitosan nanoparticles: Zn2+ adsorption, antimicrobial activity, and promotion of aquatic health
- Comparative analysis of nutritional composition and bioactive properties of Chlorella vulgaris and Arthrospira platensis: Implications for functional foods and dietary supplements
- Growth kinetics and mechanical characterization of boride layers formed on Ti6Al4V
- Enhancement of water absorption properties of potassium polyacrylate-based hydrogels in CaCl2-rich soils using potassium di- and tri-carboxylate salts
- Electrochemical and microbiological effects of dumpsite leachates on soil and air quality
- Modeling benzene physicochemical properties using Zagreb upsilon indices
- Characterization and ecological risk assessment of toxic metals in mangrove sediments near Langen Village in Tieshan Bay of Beibu Gulf, China
- Protective effect of Helicteres isora, an efficient candidate on hepatorenal toxicity and management of diabetes in animal models
- Valorization of Juglans regia L. (Walnut) green husk from Jordan: Analysis of fatty acids, phenolics, antioxidant, and cytotoxic activities
- Molecular docking and dynamics simulations of bioactive terpenes from Catharanthus roseus essential oil targeting breast cancer
- Selection of a dam site by using AHP and VIKOR: The Sakarya Basin
- Characterization and modeling of kidney bean shell biochar as adsorbent for caffeine removal from aquatic environments
- The effects of short-term and long-term 2100 MHz radiofrequency radiation on adult rat auditory brainstem response
- Biochemical insights into the anthelmintic and anti-inflammatory potential of sea cucumber extract: In vitro and in silico approaches
- Resveratrol-derived MDM2 inhibitors: Synthesis, characterization, and biological evaluation against MDM2 and HCT-116 cells
- Phytochemical constituents, in vitro antibacterial activity, and computational studies of Sudanese Musa acuminate Colla fruit peel hydro-ethanol extract
- Chemical composition of essential oils reviewed from the height of Cajuput (Melaleuca leucadendron) plantations in Buru Island and Seram Island, Maluku, Indonesia
- Phytochemical analysis and antioxidant activity of Azadirachta indica A. Juss from the Republic of Chad: in vitro and in silico studies
- Stability studies of titanium–carboxylate complexes: A multi-method computational approach
- Efficient adsorption performance of an alginate-based dental material for uranium(vi) removal
- Synthesis and characterization of the Co(ii), Ni(ii), and Cu(ii) complexes with a 1,2,4-triazine derivative ligand
- Evaluation of the impact of music on antioxidant mechanisms and survival in salt-stressed goldfish
- Optimization and validation of UPLC method for dapagliflozin and candesartan cilexetil in an on-demand formulation: Analytical quality by design approach
- Biomass-based cellulose hydroxyapatite nanocomposites for the efficient sequestration of dyes: Kinetics, response surface methodology optimization, and reusability
- Multifunctional nitrogen and boron co-doped carbon dots: A fluorescent probe for Hg2+ and biothiol detection with bioimaging and antifungal applications
- Separation of sulphonamides on a C12-diol mixed-mode HPLC column and investigation of their retention mechanism
- Characterization and antioxidant activity of pectin from lemon peels
- Fast PFAS determination in honey by direct probe electrospray ionization tandem mass spectrometry: A health risk assessment insight
- Correlation study between GC–MS analysis of cigarette aroma compounds and sensory evaluation
- Synthesis, biological evaluation, and molecular docking studies of substituted chromone-2-carboxamide derivatives as anti-breast cancer agents
- The influence of feed space velocity and pressure on the cold flow properties of diesel fuel
- Acid etching behavior and mechanism in acid solution of iron components in basalt fibers
- Protective effect of green synthesized nanoceria on retinal oxidative stress and inflammation in streptozotocin-induced diabetic rat
- Evaluation of the antianxiety activity of green zinc nanoparticles mediated by Boswellia thurifera in albino mice by following the plus maze and light and dark exploration tests
- Yeast as an efficient and eco-friendly bifunctional porogen for biomass-derived nitrogen-doped carbon catalysts in the oxygen reduction reaction
- Novel descriptors for the prediction of molecular properties
- Synthesis and characterization of surfactants derived from phenolphthalein: In vivo and in silico studies of their antihyperlipidemic effect
- Turmeric oil-fortified nutraceutical-SNEDDS: An approach to boost therapeutic effectiveness of dapagliflozin during treatment of diabetic patients
- Analysis and study on volatile flavor compounds of three Yunnan cultivated cigars based on headspace-gas chromatography-ion mobility spectrometry
- Near-infrared IR780 dye-loaded poloxamer 407 micelles: Preparation and in vitro assessment of anticancer activity
- Study on the influence of the viscosity reducer solution on percolation capacity of thin oil in ultra-low permeability reservoir
- Detection method of Aristolochic acid I based on magnetic carrier Fe3O4 and gold nanoclusters
- Juglone’s apoptotic impact against eimeriosis-induced infection: a bioinformatics, in-silico, and in vivo approach
- Potential anticancer agents from genus Aerva based on tubulin targets: an in-silico integration of quantitative structure activity relationship (QSAR), molecular docking, simulation, drug-likeness, and density functional theory (DFT) analysis
- Hepatoprotective and PXR-modulating effects of Erodium guttatum extract in propiconazole-induced toxicity
- Studies on chemical composition of medicinal plants collected in natural locations in Ecuador
- A study of different pre-treatment methods for cigarettes and their aroma differences
- Cytotoxicity and molecular mechanisms of quercetin, gallic acid, and pinocembrin in Caco-2 cells: insights from cell viability assays, network pharmacology, and molecular docking
- Choline-based deep eutectic solvents for green extraction of oil from sour cherry seeds
- Green-synthesis of chromium (III) nanoparticles using garden fern and evaluation of its antibacterial and anticholinesterase activities
- Innovative functional mayonnaise formulations with watermelon seeds oil: evaluation of quality parameters and storage stability
- Molecular insights and biological evaluation of compounds isolated from Ferula oopoda against diabetes, advanced glycation end products and inflammation in diabetics
- Removal of cytotoxic tamoxifen from aqueous solutions using a geopolymer-based nepheline–cordierite adsorbent
- Unravelling the therapeutic effect of naturally occurring Bauhinia flavonoids against breast cancer: an integrated computational approach
- Characterization of organic arsenic residues in livestock and poultry meat and offal and consumption risks
- Synthesis and characterization of zinc sulfide nanoparticles and their genotoxic and cytotoxic effects on acute myeloid leukemia cells
- Activity of Coriandrum sativum methanolic leaf extracts against Eimeria papillata: a combined in vitro and in silico approach
- Special Issue on Advancing Sustainable Chemistry for a Greener Future
- One-pot fabrication of highly porous morphology of ferric oxide-ferric oxychloride/poly-O-chloroaniline nanocomposite seeded on poly-1H pyrrole: Photocathode for green hydrogen generation from natural and artificial seawater
- High-efficiency photocathode for green hydrogen generation from sanitation water using bismuthyl chloride/poly-o-chlorobenzeneamine nanocomposite
- Innovative synthesis of cobalt-based catalysts using ionic liquids and deep eutectic solvents: A minireview on electrocatalytic water splitting
- Special Issue on Phytochemicals, Biological and Toxicological Analysis of Plants
- Comparative analysis of fruit quality parameters and volatile compounds in commercially grown citrus cultivars
- Total phenolic, flavonoid, flavonol, and tannin contents as well as antioxidant and antiparasitic activities of aqueous methanol extract of Alhagi graecorum plant used in traditional medicine: Collected in Riyadh, Saudi Arabia
- Study on the pharmacological effects and active compounds of Apocynum venetum L.
- Chemical profile of Senna italica and Senna velutina seed and their pharmacological properties
- Essential oils from Brazilian plants: A literature analysis of anti-inflammatory and antimalarial properties and in silico validation
- Toxicological effects of green tea catechin extract on rat liver: Delineating safe and harmful doses
- Unlocking the potential of Trigonella foenum-graecum L. plant leaf extracts against diabetes-associated hypertension: A proof of concept by in silico studies
Articles in the same Issue
- Research Articles
- Phytochemical investigation and evaluation of antioxidant and antidiabetic activities in aqueous extracts of Cedrus atlantica
- Influence of B4C addition on the tribological properties of bronze matrix brake pad materials
- Discovery of the bacterial HslV protease activators as lead molecules with novel mode of action
- Characterization of volatile flavor compounds of cigar with different aging conditions by headspace–gas chromatography–ion mobility spectrometry
- Effective remediation of organic pollutant using Musa acuminata peel extract-assisted iron oxide nanoparticles
- Analysis and health risk assessment of toxic elements in traditional herbal tea infusions
- Cadmium exposure in marine crabs from Jiaxing City, China: Insights into health risk assessment
- Green-synthesized silver nanoparticles of Cinnamomum zeylanicum and their biological activities
- Tetraclinis articulata (Vahl) Mast., Mentha pulegium L., and Thymus zygis L. essential oils: Chemical composition, antioxidant and antifungal properties against postharvest fungal diseases of apple, and in vitro, in vivo, and in silico investigation
- Exploration of plant alkaloids as potential inhibitors of HIV–CD4 binding: Insight into comprehensive in silico approaches
- Recovery of phenylethyl alcohol from aqueous solution by batch adsorption
- Electrochemical approach for monitoring the catalytic action of immobilized catalase
- Green synthesis of ZIF-8 for selective adsorption of dyes in water purification
- Optimization of the conditions for the preparation of povidone iodine using the response surface methodology
- A case study on the influence of soil amendment on ginger oil’s physicochemical properties, mineral contents, microbial load, and HPLC determination of its vitamin level
- Removal of antiviral favipiravir from wastewater using biochar produced from hazelnut shells
- Effect of biochar and soil amendment on bacterial community composition in the root soil and fruit of tomato under greenhouse conditions
- Bioremediation of malachite green dye using Sargassum wightii seaweed and its biological and physicochemical characterization
- Evaluation of natural compounds as folate biosynthesis inhibitors in Mycobacterium leprae using docking, ADMET analysis, and molecular dynamics simulation
- Novel insecticidal properties of bioactive zoochemicals extracted from sea urchin Salmacis virgulata
- Elevational gradients shape total phenolic content and bioactive potential of sweet marjoram (Origanum majorana L.): A comparative study across altitudinal zones
- Study on the CO2 absorption performance of deep eutectic solvents formed by superbase DBN and weak acid diethylene glycol
- Preparation and wastewater treatment performance of zeolite-modified ecological concrete
- Multifunctional chitosan nanoparticles: Zn2+ adsorption, antimicrobial activity, and promotion of aquatic health
- Comparative analysis of nutritional composition and bioactive properties of Chlorella vulgaris and Arthrospira platensis: Implications for functional foods and dietary supplements
- Growth kinetics and mechanical characterization of boride layers formed on Ti6Al4V
- Enhancement of water absorption properties of potassium polyacrylate-based hydrogels in CaCl2-rich soils using potassium di- and tri-carboxylate salts
- Electrochemical and microbiological effects of dumpsite leachates on soil and air quality
- Modeling benzene physicochemical properties using Zagreb upsilon indices
- Characterization and ecological risk assessment of toxic metals in mangrove sediments near Langen Village in Tieshan Bay of Beibu Gulf, China
- Protective effect of Helicteres isora, an efficient candidate on hepatorenal toxicity and management of diabetes in animal models
- Valorization of Juglans regia L. (Walnut) green husk from Jordan: Analysis of fatty acids, phenolics, antioxidant, and cytotoxic activities
- Molecular docking and dynamics simulations of bioactive terpenes from Catharanthus roseus essential oil targeting breast cancer
- Selection of a dam site by using AHP and VIKOR: The Sakarya Basin
- Characterization and modeling of kidney bean shell biochar as adsorbent for caffeine removal from aquatic environments
- The effects of short-term and long-term 2100 MHz radiofrequency radiation on adult rat auditory brainstem response
- Biochemical insights into the anthelmintic and anti-inflammatory potential of sea cucumber extract: In vitro and in silico approaches
- Resveratrol-derived MDM2 inhibitors: Synthesis, characterization, and biological evaluation against MDM2 and HCT-116 cells
- Phytochemical constituents, in vitro antibacterial activity, and computational studies of Sudanese Musa acuminate Colla fruit peel hydro-ethanol extract
- Chemical composition of essential oils reviewed from the height of Cajuput (Melaleuca leucadendron) plantations in Buru Island and Seram Island, Maluku, Indonesia
- Phytochemical analysis and antioxidant activity of Azadirachta indica A. Juss from the Republic of Chad: in vitro and in silico studies
- Stability studies of titanium–carboxylate complexes: A multi-method computational approach
- Efficient adsorption performance of an alginate-based dental material for uranium(vi) removal
- Synthesis and characterization of the Co(ii), Ni(ii), and Cu(ii) complexes with a 1,2,4-triazine derivative ligand
- Evaluation of the impact of music on antioxidant mechanisms and survival in salt-stressed goldfish
- Optimization and validation of UPLC method for dapagliflozin and candesartan cilexetil in an on-demand formulation: Analytical quality by design approach
- Biomass-based cellulose hydroxyapatite nanocomposites for the efficient sequestration of dyes: Kinetics, response surface methodology optimization, and reusability
- Multifunctional nitrogen and boron co-doped carbon dots: A fluorescent probe for Hg2+ and biothiol detection with bioimaging and antifungal applications
- Separation of sulphonamides on a C12-diol mixed-mode HPLC column and investigation of their retention mechanism
- Characterization and antioxidant activity of pectin from lemon peels
- Fast PFAS determination in honey by direct probe electrospray ionization tandem mass spectrometry: A health risk assessment insight
- Correlation study between GC–MS analysis of cigarette aroma compounds and sensory evaluation
- Synthesis, biological evaluation, and molecular docking studies of substituted chromone-2-carboxamide derivatives as anti-breast cancer agents
- The influence of feed space velocity and pressure on the cold flow properties of diesel fuel
- Acid etching behavior and mechanism in acid solution of iron components in basalt fibers
- Protective effect of green synthesized nanoceria on retinal oxidative stress and inflammation in streptozotocin-induced diabetic rat
- Evaluation of the antianxiety activity of green zinc nanoparticles mediated by Boswellia thurifera in albino mice by following the plus maze and light and dark exploration tests
- Yeast as an efficient and eco-friendly bifunctional porogen for biomass-derived nitrogen-doped carbon catalysts in the oxygen reduction reaction
- Novel descriptors for the prediction of molecular properties
- Synthesis and characterization of surfactants derived from phenolphthalein: In vivo and in silico studies of their antihyperlipidemic effect
- Turmeric oil-fortified nutraceutical-SNEDDS: An approach to boost therapeutic effectiveness of dapagliflozin during treatment of diabetic patients
- Analysis and study on volatile flavor compounds of three Yunnan cultivated cigars based on headspace-gas chromatography-ion mobility spectrometry
- Near-infrared IR780 dye-loaded poloxamer 407 micelles: Preparation and in vitro assessment of anticancer activity
- Study on the influence of the viscosity reducer solution on percolation capacity of thin oil in ultra-low permeability reservoir
- Detection method of Aristolochic acid I based on magnetic carrier Fe3O4 and gold nanoclusters
- Juglone’s apoptotic impact against eimeriosis-induced infection: a bioinformatics, in-silico, and in vivo approach
- Potential anticancer agents from genus Aerva based on tubulin targets: an in-silico integration of quantitative structure activity relationship (QSAR), molecular docking, simulation, drug-likeness, and density functional theory (DFT) analysis
- Hepatoprotective and PXR-modulating effects of Erodium guttatum extract in propiconazole-induced toxicity
- Studies on chemical composition of medicinal plants collected in natural locations in Ecuador
- A study of different pre-treatment methods for cigarettes and their aroma differences
- Cytotoxicity and molecular mechanisms of quercetin, gallic acid, and pinocembrin in Caco-2 cells: insights from cell viability assays, network pharmacology, and molecular docking
- Choline-based deep eutectic solvents for green extraction of oil from sour cherry seeds
- Green-synthesis of chromium (III) nanoparticles using garden fern and evaluation of its antibacterial and anticholinesterase activities
- Innovative functional mayonnaise formulations with watermelon seeds oil: evaluation of quality parameters and storage stability
- Molecular insights and biological evaluation of compounds isolated from Ferula oopoda against diabetes, advanced glycation end products and inflammation in diabetics
- Removal of cytotoxic tamoxifen from aqueous solutions using a geopolymer-based nepheline–cordierite adsorbent
- Unravelling the therapeutic effect of naturally occurring Bauhinia flavonoids against breast cancer: an integrated computational approach
- Characterization of organic arsenic residues in livestock and poultry meat and offal and consumption risks
- Synthesis and characterization of zinc sulfide nanoparticles and their genotoxic and cytotoxic effects on acute myeloid leukemia cells
- Activity of Coriandrum sativum methanolic leaf extracts against Eimeria papillata: a combined in vitro and in silico approach
- Special Issue on Advancing Sustainable Chemistry for a Greener Future
- One-pot fabrication of highly porous morphology of ferric oxide-ferric oxychloride/poly-O-chloroaniline nanocomposite seeded on poly-1H pyrrole: Photocathode for green hydrogen generation from natural and artificial seawater
- High-efficiency photocathode for green hydrogen generation from sanitation water using bismuthyl chloride/poly-o-chlorobenzeneamine nanocomposite
- Innovative synthesis of cobalt-based catalysts using ionic liquids and deep eutectic solvents: A minireview on electrocatalytic water splitting
- Special Issue on Phytochemicals, Biological and Toxicological Analysis of Plants
- Comparative analysis of fruit quality parameters and volatile compounds in commercially grown citrus cultivars
- Total phenolic, flavonoid, flavonol, and tannin contents as well as antioxidant and antiparasitic activities of aqueous methanol extract of Alhagi graecorum plant used in traditional medicine: Collected in Riyadh, Saudi Arabia
- Study on the pharmacological effects and active compounds of Apocynum venetum L.
- Chemical profile of Senna italica and Senna velutina seed and their pharmacological properties
- Essential oils from Brazilian plants: A literature analysis of anti-inflammatory and antimalarial properties and in silico validation
- Toxicological effects of green tea catechin extract on rat liver: Delineating safe and harmful doses
- Unlocking the potential of Trigonella foenum-graecum L. plant leaf extracts against diabetes-associated hypertension: A proof of concept by in silico studies

