Crystal structure of 3-(adamantan-1-yl)-1-[(4-benzylpiperazin-1-yl)methyl]-4-[(E)-(2,6-difluorobenzylidene)amino]-1H-1,2,4-triazole-5(4H)-thione, C31H36F2N6S
Abstract
C31H36F2N6S, monoclinic, P21/c (no. 14), a = 14.7561(8) Å, b = 24.6766(13) Å, c = 7.7811(4) Å, β = 95.888(2)°, V = 2818.4(3) Å3, Z = 4, Rgt(F) = 0.0624, wRref(F2) = 0.1445, T = 100 K.
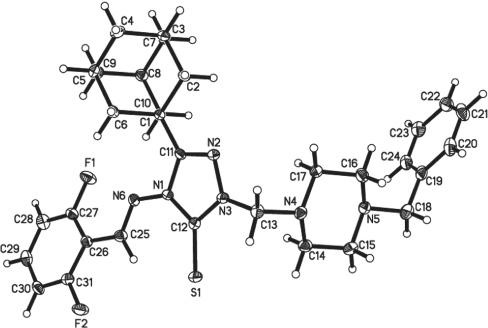
Tables 1 and 2 contain details of the measurement method and a list of the atoms including atomic coordinates and displacement parameters.
Data collection and handling.
| Crystal: | Yellow needles Size 0.49 × 0.21 × 0.16 |
| Wavelength: | Mo Kα radiation (0.71073 Å) |
| μ: | 1.61 cm−1 |
| Diffractometer, scan mode: | Bruker Apex II, φ and ω |
| 2θmax, completeness: | 55.0°, >99% |
| N(hkl)measured, N(hkl)unique: | 9934, 6468 |
| Criterion for Iobs, N(hkl)gt: | Iobs > 2 σ(Iobs), 4817 |
| N(param)refined: | 365 |
| Programs: | Bruker programs [25], SHELX [26] |
Fractional atomic coordinates and isotropic or equivalent isotropic displacement parameters (Å2).
| Atom | x | y | z | Uiso*/Ueq |
|---|---|---|---|---|
| S1 | 0.97668(5) | 0.46948(2) | 0.23439(8) | 0.02047(16) |
| F1 | 0.74649(10) | 0.30124(6) | 0.5218(2) | 0.0266(4) |
| F2 | 1.04001(10) | 0.30416(6) | 0.3347(2) | 0.0273(4) |
| N1 | 0.84177(14) | 0.45159(8) | 0.4548(2) | 0.0147(4) |
| N2 | 0.79924(14) | 0.53516(8) | 0.5099(2) | 0.0155(4) |
| N3 | 0.86593(14) | 0.53501(8) | 0.3977(3) | 0.0156(4) |
| N4 | 0.86437(14) | 0.61665(8) | 0.2129(3) | 0.0160(4) |
| N5 | 0.73701(15) | 0.64156(8) | −0.0791(3) | 0.0185(5) |
| N6 | 0.83721(14) | 0.39557(8) | 0.4738(3) | 0.0164(4) |
| C1 | 0.71672(16) | 0.46542(9) | 0.6608(3) | 0.0147(5) |
| C2 | 0.66778(17) | 0.51537(9) | 0.7275(3) | 0.0168(5) |
| H2A | 0.7131 | 0.5400 | 0.7892 | 0.020* |
| H2B | 0.6362 | 0.5354 | 0.6286 | 0.020* |
| C3 | 0.59822(18) | 0.49750(10) | 0.8503(3) | 0.0191(5) |
| H3A | 0.5672 | 0.5302 | 0.8927 | 0.023* |
| C4 | 0.64749(18) | 0.46709(10) | 1.0051(3) | 0.0205(5) |
| H4A | 0.6931 | 0.4911 | 1.0684 | 0.025* |
| H4B | 0.6031 | 0.4559 | 1.0853 | 0.025* |
| C5 | 0.69487(18) | 0.41699(10) | 0.9387(3) | 0.0189(5) |
| H5A | 0.7264 | 0.3968 | 1.0391 | 0.023* |
| C6 | 0.76539(17) | 0.43475(9) | 0.8181(3) | 0.0165(5) |
| H6A | 0.7973 | 0.4026 | 0.7779 | 0.020* |
| H6B | 0.8112 | 0.4587 | 0.8813 | 0.020* |
| C7 | 0.52722(17) | 0.46040(10) | 0.7538(3) | 0.0211(5) |
| H7A | 0.4945 | 0.4802 | 0.6555 | 0.025* |
| H7B | 0.4821 | 0.4489 | 0.8323 | 0.025* |
| C8 | 0.57500(17) | 0.41062(10) | 0.6874(3) | 0.0180(5) |
| H8A | 0.5286 | 0.3863 | 0.6245 | 0.022* |
| C9 | 0.62426(18) | 0.37990(10) | 0.8408(3) | 0.0200(5) |
| H9A | 0.5796 | 0.3677 | 0.9192 | 0.024* |
| H9B | 0.6548 | 0.3475 | 0.7988 | 0.024* |
| C10 | 0.64429(17) | 0.42812(9) | 0.5644(3) | 0.0166(5) |
| H10A | 0.6127 | 0.4477 | 0.4644 | 0.020* |
| H10B | 0.6742 | 0.3957 | 0.5205 | 0.020* |
| C11 | 0.78471(16) | 0.48445(9) | 0.5424(3) | 0.0141(5) |
| C12 | 0.89530(17) | 0.48463(9) | 0.3621(3) | 0.0160(5) |
| C13 | 0.90839(17) | 0.58716(9) | 0.3568(3) | 0.0172(5) |
| H13A | 0.9106 | 0.6106 | 0.4603 | 0.021* |
| H13B | 0.9721 | 0.5800 | 0.3340 | 0.021* |
| C14 | 0.86854(18) | 0.58909(10) | 0.0466(3) | 0.0187(5) |
| H14A | 0.8324 | 0.5552 | 0.0441 | 0.022* |
| H14B | 0.9325 | 0.5794 | 0.0323 | 0.022* |
| C15 | 0.83131(17) | 0.62573(10) | −0.0992(3) | 0.0201(5) |
| H15A | 0.8698 | 0.6586 | −0.1006 | 0.024* |
| H15B | 0.8333 | 0.6067 | −0.2108 | 0.024* |
| C16 | 0.73477(19) | 0.66966(10) | 0.0861(3) | 0.0207(5) |
| H16A | 0.6715 | 0.6806 | 0.1007 | 0.025* |
| H16B | 0.7725 | 0.7028 | 0.0872 | 0.025* |
| C17 | 0.77057(18) | 0.63294(10) | 0.2335(3) | 0.0189(5) |
| H17A | 0.7688 | 0.6522 | 0.3447 | 0.023* |
| H17B | 0.7315 | 0.6004 | 0.2350 | 0.023* |
| C18 | 0.70184(19) | 0.67613(11) | −0.2241(3) | 0.0270(6) |
| H18A | 0.7074 | 0.6566 | −0.3337 | 0.032* |
| H18B | 0.7396 | 0.7093 | −0.2241 | 0.032* |
| C19 | 0.60367(19) | 0.69217(10) | −0.2166(3) | 0.0212(6) |
| C20 | 0.5806(2) | 0.74361(10) | −0.1626(3) | 0.0264(6) |
| H20A | 0.6273 | 0.7693 | −0.1311 | 0.032* |
| C21 | 0.4907(2) | 0.75797(11) | −0.1542(3) | 0.0298(7) |
| H21A | 0.4761 | 0.7933 | −0.1174 | 0.036* |
| C22 | 0.4222(2) | 0.72115(11) | −0.1990(3) | 0.0272(6) |
| H22A | 0.3603 | 0.7313 | −0.1953 | 0.033* |
| C23 | 0.4439(2) | 0.66922(11) | −0.2498(3) | 0.0253(6) |
| H23A | 0.3970 | 0.6435 | −0.2786 | 0.030* |
| C24 | 0.53408(19) | 0.65506(10) | −0.2583(3) | 0.0235(6) |
| H24A | 0.5486 | 0.6195 | −0.2933 | 0.028* |
| C25 | 0.89500(17) | 0.36578(9) | 0.4068(3) | 0.0171(5) |
| H25A | 0.9396 | 0.3820 | 0.3436 | 0.021* |
| C26 | 0.89317(16) | 0.30654(9) | 0.4265(3) | 0.0155(5) |
| C27 | 0.82227(17) | 0.27572(10) | 0.4794(3) | 0.0178(5) |
| C28 | 0.82366(18) | 0.21974(10) | 0.4874(3) | 0.0227(6) |
| H28A | 0.7731 | 0.2002 | 0.5223 | 0.027* |
| C29 | 0.90022(18) | 0.19275(10) | 0.4437(3) | 0.0214(6) |
| H29A | 0.9025 | 0.1543 | 0.4505 | 0.026* |
| C30 | 0.97328(18) | 0.22065(10) | 0.3903(3) | 0.0205(6) |
| H30A | 1.0255 | 0.2021 | 0.3587 | 0.025* |
| C31 | 0.96806(17) | 0.27621(10) | 0.3845(3) | 0.0180(5) |
Source of material
1-Benzylpiperazine (353 mg, 1 mmol) and a 37% formaldehyde solution (1.0 mL) were added to a stirred hot solution of 5-(adamantan-1-yl)-4-(2,6-difluorobenzylideneamino)-4H-1,2,4-triazole-\ 3-thiol (747 mg, 2 mmol), in ethanol (10 mL), and the mixture was heated under reflux for 15 min. Stirring was continued for 12 h at room temperature and the mixture was allowed to stand overnight. Cold water (5 mL) was gradually added and the mixture was stirred for 20 min. The precipitated crude product was filtered, washed with water, dried, and crystallized from ethanol to yield 900 mg (80%) of the title compound (C31H36F2N6S) as colourless needle crystals. M.P.: 421–423 K [17]. Single crystals suitable for X-ray analysis were obtained by slow evaporation of a CHCl3:EtOH (1:1; 5 mL) solution at room temperature. 1H NMR (CDCl3, 500.13 MHz): δ 1.80 (s, 6H, adamantane-H), 2.09 (s, 3H, adamantane-H), 2.18 (s, 6H, adamantane-H), 2.41–2.52 (m, 4H, piperazine-H), 2.95 (s, 4H, piperazine-H), 3.52 (s, 2H, PhCH2), 5.17 (s, 2H, CH2), 7.03 (t, 2H, Ar—H, J = 8.5 Hz), 7.25–7.32 (m, 5H, Ar—H), 7.45–7.49 (m, 1H, Ar—H), 10.65 (s, 1H, CH = N). 13C NMR (CDCl3, 125.76 MHz): δ 28.0, 35.52, 36.47, 38.36 (adamantane-C), 50.51, 53.12 (piperazine-C), 63.18 (PhCH2), 68.96 (CH2), 110.89, 112.17, 127.04, 128.18, 129.29, 133.11, 137.96, 152.11 (Ar—C), 155.47, 161.05 (triazole C-5 & CH = N), 163.19 (C = S). ESI-MS, m/z: 563 [M+H]+.
Discussion
Derivatives of adamantane have long been known for their antiviral activity against the influenza A [1–4], herpes simplex [5] and HIV [6–8] viruses. Several adamantane-based drugs are currently used as efficient therapies for the control central nervous disorders [9–12]. In addition, potent antimicrobial [13–18], anti-inflammatory [16–18] and anticancer [19, 20] activities were reported for adamantane-based derivatives. 1,2,4-Triazole derivatives were also recognized as a structural motif of particular value in medicinal chemistry possessing diverse biological activities [21–23]. In the present study, we report the crystal structure together with the synthesis of the title 1,2,4-triazole-N-Mannich base which was reported to exhibit marked antifungal activity [17]. This contribution is part of study on admantyl-based drugs [24]
There is one complete molecule in the asymmetric unit of the title structure. All bond lengths and angels are in the expected ranges.
The molecules packing in the crystal structure is stabilized via two intermolecular hydrogen bonds, of which S1 and F2 work as hydrogen bond acceptors and C14 and C15 work as hydrogen bond donors. The distance of the interactions between C14—H14B⋯S1i and C15—H15A⋯F2i are 2.85 and 2.54 Å, respectively and the angles are 135 and 130°, respectively. Symmetry codes: (i) −x+2, −y+1, −z.
Funding source: King Saud University
Award Identifier / Grant number: PRG-1436–23
Funding statement: The authors would like to extend their sincere appreciation to the Deanship of Scientific Research at King Saud University for funding this work through the Research Group Project No. PRG-1436–23.
Acknowledgements:
The authors would like to extend their sincere appreciation to the Deanship of Scientific Research at King Saud University for funding this work through the Research Group Project No. PRG-1436–23.
References
1. Togo, Y.; Hornick, R. B.; Dawkins, A. T.: Studies on induced influenza in man. I. Double blind studies designed to assess prophylactic efficacy of amantadine hydrochloride against A2/Rockville/1/65 strain. J. Am. Med. Assoc. 203 (1968) 1089–1094.10.1001/jama.203.13.1089Suche in Google Scholar
2. Davies, W. L.; Grunnert, R. R.; Haff, R. F.; McGahen, J. W.; Neumeyer, E. M.; Paulshock, M.; Watts, J. C.; Wood, T. R.; Hermann, E. C.; Hoffmann, C. E.: Antiviral activity of 1-adamantamine (amantadine). Science 144 (1964) 862–863.10.1126/science.144.3620.862Suche in Google Scholar
3. Rabinovich, S.; Baldini, J. T.; Bannister, R.: Treatment of influenza. The therapeutic efficacy of rimantadine HCl in a naturally occurring influenza A2 outbreak. Am. J. Med. Sci. 257 (1969) 328–335.10.1097/00000441-196905000-00005Suche in Google Scholar
4. Wendel, H. A.; Snyder, M. T.; Pell, S.: Trial of amantadine in epidemic influenza. Clin. Pharmacol. Therap. 7 (1966) 38–43.10.1002/cpt19667138Suche in Google Scholar
5. Rosenthal, K. S.; Sokol, M. S.; Ingram, R. L.; Subramanian, R.; Fort, R. C.: Tromantadine: Inhibitor of early and late events in herpes simplex virus replication. Antimicrob. Agents Chemother. 22 (1982) 1031–1036.10.1128/AAC.22.6.1031Suche in Google Scholar
6. Burstein, M. E.; Serbin, A. V.; Khakhulina, T. V.; Alymova, I. V.; Stotskaya, L. L.; Bogdan, O. P.; Manukchina, E. E.; Jdanov, V. V.; Sharova, N. K.: Inhibition of HIV-1 replication by newly developed adamantane-containing polyanionic agents. Antiviral Res. 41 (1999) 135–144.10.1016/S0166-3542(99)00006-6Suche in Google Scholar
7. Balzarini, J.; Orzeszko, B.; Mauri, J. K.; Orzeszko, A.: Synthesis and anti-HIV studies of 2-adamantyl-substituted thiazolidin-4-ones. Eur. J. Med. Chem. 42 (2007) 93–1003.10.1016/j.ejmech.2007.01.003Suche in Google Scholar
8. El-Emam, A. A.; Al-Deeb, O. A.; Al-Omar, M. A.; Lehmann, J.: Synthesis, antimicrobial, and anti-HIV-1 activity of certain 5-(1-adamantyl)-2-substituted thio-1,3,4-oxadiazoles and 5-(1-adamantyl)-3-substituted aminomethyl-1,3,4-oxadiazoline-2-thiones. Bioorg. Med. Chem. 12 (2004) 5107–5113.10.1016/j.bmc.2004.07.033Suche in Google Scholar
9. Bormann, J.: Memantine is a potent blocker of N-methyl-D-aspartate (NMDA) receptor channels. Eur. J. Pharmacol. 166 (1989) 59l–592.10.1016/0014-2999(89)90385-3Suche in Google Scholar
10. Abou-Gharbia, M. A.; Childers, W. E., Jr.; Fletcher, H.; McGaughey, G.; Patel, U.; Webb, M. B.; Yardley, J.; Andree, T.; Boast, C.; Kucharik, R. J., Jr., Marquis, K.; Morris, H.; Scerni, R.; Moyer, J. A.: Synthesis and SAR of adatanserin: novel adamantly aryl- and heteroarylpiperazines with dual serotonin 5-HT1A and 5-HT2 activity as potential anxiolytic and antidepressant agents. J. Med. Chem. 42 (1999) 5077–5094.10.1021/jm9806704Suche in Google Scholar PubMed
11. Narayanan, V. L.: Adamantyl analogs of the antidepressive, 5-(2-dimethylaminoethyl)-2,3-dihydro-2-phenylbenzothiazepin-4(5H)-one. J. Med. Chem. 15 (1972) 682–684.10.1021/jm00276a032Suche in Google Scholar PubMed
12. Sozio, P.; Cerasa, L. S.; Laserra, S.; Cacciatore, I.; Cornacchira, C.; Di Filippo, E. S.; Fulle, S.; Fontana, A.; Di Crescenzo, A.; Grillo, M.; Marchi, M.: Memantine-sulfur containing antioxidant conjugates as potential prodrugs to improve the treatment of Alzheimers disease. Eur. J. Med. Chem. 49 (2013) 187–198.10.1016/j.ejps.2013.02.013Suche in Google Scholar
13. Jia, L.; Tomaszewski, J. E.; Hanrahan, C.; Coward, L.; Noker, P.; Gorman, G.; Nikonenko, B.; Protopopova, M.: Pharmacodynamics and pharmacokinetics of SQ109, a new diamine-based antitubercular drug. Brit. J. Pharmacol. 144 (2005) 80–87.10.1038/sj.bjp.0705984Suche in Google Scholar PubMed PubMed Central
14. Omar, K.; Geronikaki, A.; Zoumpoulakis, P.; Camoutsis, C.; Soković, M.; Ćirić, A.; Glamoćlija, J.: Novel 4-thiazolidinone derivatives as potential antifungal and antibacterial drugs. Bioorg. Med. Chem. 18 (2010) 426–432.10.1016/j.bmc.2009.10.041Suche in Google Scholar PubMed
15. El-Emam, A. A.; Al-Tamimi, A. -M. S.; Al-Omar, M. A.; Alrashood, K. A.; Habib, E. E.: Synthesis and antimicrobial activity of novel 5-(1-adamantyl)-2-aminomethyl-4-substituted-1,2,4-triazoline-3-thiones. Eur. J. Med. Chem. 68 (2013) 96–102.10.1016/j.ejmech.2013.07.024Suche in Google Scholar PubMed
16. Al-Abdullah, E. S.; Asiri, H. H.; Lahsasni, S.; Habib, E. E.; Ibrahim, T. M.; El-Emam, A. A.: Synthesis, antimicrobial, and anti-inflammatory activity, of novel S-substituted and N-substituted 5-(1-adamantyl)-1,2,4-triazole-3-thiols. Drug Des. Dev. Ther. 8 (2014) 505–518.10.2147/DDDT.S62465Suche in Google Scholar PubMed PubMed Central
17. Al-Omar, M. A.; Al-Abdullah, E. S.; Shehata, I. S.; Habib. E. E.; Ibrahim, T. M.; El-Emam, A. A.: Synthesis, antimicrobial, and anti-inflammatory activities of novel 5-(1-adamantyl)-4-arylideneamino-3-mercapto-1,2,4-triazoles and related derivatives. Molecules 15 (2010) 2526–2550.10.3390/molecules15042526Suche in Google Scholar PubMed PubMed Central
18. Kadi, A. A.; Al-Abdullah, E. S.; Shehata, I. A.; Habib, E. E.; Ibrahim, T. M.; El-Emam, A. A.: Synthesis, antimicrobial and anti-inflammatory activities of novel 5-(1-adamantyl)-1,3,4-thiadiazole Derivatives. Eur. J. Med. Chem. 45 (2010) 5006–5011.10.1016/j.ejmech.2010.08.007Suche in Google Scholar PubMed
19. Sun, S. Y.; Yue, P.; Chen, X.; Hong, W. K.; Lotan, R.: The synthetic retinoid CD437 selectively induces apoptosis in human lung cancer cells while sparing normal human lung epithelial cells. Cancer Res. 62 (2002) 2430–2436.Suche in Google Scholar
20. Lorenzo, P.; Alvarez, R.; Ortiz, M. A.; Alvarez, S.; Piedrafita, F. J.; de Lira, Á. R.: Inhibition of IκB kinase-β and anticancer activities of novel chalcone adamantyl arotinoids. J. Med. Chem. 51 (2008) 5431–5440.10.1021/jm800285fSuche in Google Scholar PubMed
21. Navidpour, L.; Shafaroodi, H.; Abdi, K.; Amini, M.; Ghahremani, M. H.; Dehpour, A.R.; Shafiee, A.: Design, synthesis, and biological evaluation of substituted 3-alkylthio-4,5-diaryl-4H-1,2,4-triazoles as selective COX-2 inhibitors. Bioorg. Med. Chem. 14 (2006) 2507–2517.10.1016/j.bmc.2005.11.029Suche in Google Scholar PubMed
22. Bayrak, H.; Demirbas, A.; Karaoglu, S. A.; Demirbas, N.: Synthesis of some new 1,2,4-triazoles, their Mannich and Schiff bases and evaluation of their antimicrobial activities. Eur. J. Med. Chem. 44 (2009) 1057–1066.10.1016/j.ejmech.2008.06.019Suche in Google Scholar PubMed
23. Ashok, M.; Holla, B. S.; Boojary, B.: Convenient one pot synthesis and antimicrobial evaluation of some new Mannich bases carrying 4-methylthiobenzyl moiety. Eur. J. Med. Chem. 42 (2007) 1095–1101.10.1016/j.ejmech.2007.01.015Suche in Google Scholar PubMed
24. Al-Abdullah, E. S.; Ghabbour, H. A.; Al-Jabal, M. M. Hoong-Kun Fun H.-K.; El-Emam, A. A.: Crystal structure of N′-(adamantan-2-ylidene)isonicotinohydrazide, C16H19N3O. Z. Kristallogr. NCS 231 (2016) 273-275.10.1515/ncrs-2015-0118Suche in Google Scholar
25. Bruker. APEX2, SAINT and SADABS. Bruker AXS Inc., Madison, Wisconsin, USA, 2009.Suche in Google Scholar
26. Sheldrick, G. M.: A short history of SHELX. Acta Crystallogr. A64 (2008) 112–122.10.1107/S0108767307043930Suche in Google Scholar PubMed
©2016 Monirah A. Al-Alshaikh et al., published by De Gruyter.
This work is licensed under the Creative Commons Attribution-NonCommercial-NoDerivatives 3.0 License.
Artikel in diesem Heft
- Frontmatter
- Crystal structure of poly[aqua(μ2-2,5-bis(4-pyridyl)-1,3,4-oxadiazole-κ2N,N)(μ2-1,3-phenylenediacetato-κ3O,O′:O′′)cobalt(II)], C22H18CoN4O6
- Crystal structure of 2-amino-7,7-dimethyl-5-oxo-4-(3-phenoxy-phenyl)-5,6,7,8-tetrahydro-4H-chromene-3-carbonitrile
- Crystal structure of (E)-4,4′-(diazene-1,2-diyl)bis(1-nitro-1H-1,2,4-triazol-5(4H)-one)—acetonitrile (1:1), C6H5N11O6
- Crystal structure of potassium (E)-5-oxo-4-((5-oxo-1H-1,2,4-triazol-4(5H)-yl)diazenyl)-4,5-dihydro-1,2,4-triazol-1-ide – (E)-4,4-diazene-1,2-diylbis(2,4-dihydro-3H-1,2,4-triazol-3-one) – methanol (1/1/1), C9H11N16KO5
- Crystal structure of (N,N′-bis(2-(((2,6-diisopropylphenyl)imino)methyl)phenyl)benzene-1,2-diamido-κ4O,O′,O′′,O′′′)oxidovanadium(IV), C44H48N4OV
- Crystal structure of 5,5-bis(4-iodophenyl)-5H-cyclopenta[2,1-b:3,4-b′]dipyridine, C23H14I2N2
- Crystal structure of aquadichloridobis(1-((2-methyl-1H-imidazol-1-yl)methyl)-1H-benotriazole-κN)mercury(II), C22H24Cl2HgN10O
- Crystal structure of 2-[4-(1H-imidazol-1-yl)phenyl]-1H-benzimidazol-3-ium [2-(carboxymethyl)phenyl]acetate monohydrate, C26H24N4O5
- Crystal structure of 4,4′-bipiperidinium dichloride 0.12 hydrate, C10H22N2Cl2 · 0.12 H2O
- Crystal structure of catena-poly[tetraaqua(μ2-4,4′(E)-ethene-1,2-diyldipyridine-κ2N:N′)nickel(II)] bis(6-methyl-2-oxo-1,2-dihydro-pyridine-4-carboxylate) pentahydrate, C26H42N4O15Ni
- Crystal structure of ethyl-5-amino-1-(2,4-dinitrophenyl)-1H-pyrazole-4-carboxylate, C12H11N5O6
- Crystal structure of dicarbonyl(pyridin-2-olate-1-oxido-κ2O,O′)rhodium(I), C7H4NO4Rh
- Crystal structure of 1-(adamantan-1-yl)-3-(4-chlorophenyl)thiourea, C17H21ClN2S
- Crystal structure of 2-((2-chloropyridin-3-ylamino)methylene)malononitrile, C9H5ClN4
- Crystal structure of tetraaquabis(μ2-4-chlorobenzoato-κ2O:O,O′)bis(4-chlorobenzoato-κO)bis(1,10-phenanthroline-κ2N,N′)distrontium(II), C52H40Cl4N4O12Sr2
- Crystal structure of 3-hydroxy-3-phenyl-1,3-dihydro-2H-indol-2-one, C14H11NO2
- Crystal structures of bis(1,10-phenanthrolin-1-ium) aquapentakis(nitrato-κ2O,O′)neodym(III) monohydrate, C24H22N9NdO17
- Crystal structure of 2-ethyl-1-tert-butyl 3-oxo-2-[phenyl(tert-butoxycarbonylamino)methyl]-1,2-pyrrolidinedicarboxylate, C24H34N2O7
- Crystal structure of poly[diaquabis(μ4-benzene-1,3,5-tricarboxylato-κO1,κO2:κO3,κO4:κO5:κO6)-bis(μ2-4,4′-benzene-1,3-diylbis(4H-1,2,4-triazole-κ2N:N′)tricadmium(II)] tetrahydrate, C38H34Cd3N12O18
- Crystal structure of trans-dichlorido[1,3-bis(9-methyl-9H-fluoren-9-yl) benzimidazol-2-ylidene](pyridine)palladium(II) – a compound with anagostic CH–Pd interactions, C40H31Cl2N3Pd
- Crystal structure of catena-poly[(μ3-5-(4-(tetrazol-1-id-5-yl)phenoxy)benzene-1,3-dicarboxyato-κ3O:O′:N)(4-(3-(pyridin-4-yl)propyl)pyridinium-κN)zinc(II)], C28H22ZnN6O5
- Crystal structure of 3,6-di-2-pyridinyl-4-pyridazine carbonitrile, C15H9N5
- Crystal structure of 5,5,9,13-tetramethyltetracyclo[10·2·1·01,10·04,9]pentadecane-3,7,14-triol, C20H34O4
- Crystal structure of 2-amino-7-methyl-5-oxo-4-phenyl-4H,5H-pyrano[4,3-b]pyran-3-carbonitrile, C16H12N2O3
- Crystal structure of the poly[(1,10-phenanthroline-κ2N,N′)(μ3-carboxylatophenoxyacetato-κ4O,O′:O′′;O′′′)lead(II)] monohydrate, C21H16N2O6Pb
- Crystal structure of the poly[(μ4-biphenyl-4,4′-dicarboxylato-κ4O:O′:O′′:O′′′) bis(μ3-8-(11-(oxysulfonyl)-4-silbenyl)-2-(oxysulfonyl)stilbene-κ4O:O′:O′′,O′′′) bis(1,10-phenanthroline-k2N,N′) dipraseodymium(III)], C94H64N4O16Pr2S4
- Crystal structure of 3-iodo-5-methoxy-7-(methoxymethoxy)-4-(3-methoxyphenoxy)-2H-chromen-2-one, C19H17IO7
- Crystal structure of poly[(5-carboxy-2,6-dimethylpyridinium-3-carboxylato-κO)tris(μ2-2,6-dimethylpyridinium-3,5-dicarboxylato-κ3O,O′:O′′)erbium(III)], C36H33ErN4O16
- This molecule targets at type 2 diabetes - a single crystal study on (2R,3S,5R)-2-(2,5-difluorophenyl)-5-[2-(methylsulfonyl)-2,6-dihydropyrrolo[3,4-c]pyrazol-5(4H)-yl] tetrahydro-2H-pyran-3-amine (Omarigliptin), C17H20F2N4O3S
- Crystal structure of poly[(μ2-2,2′-benzene-1,2-diyldiacetato-κ2O:O′), (μ2-1-(4-(1H-imidazol-1-yl)benzyl)-1H-1,2,4-triazole-κ2N:N′)zinc(II)], C22H19N5O4Zn
- Crystal structure of tetraaqua(μ2-3-(3,5-dicarboxyphenoxy)benzene-1,2-dicarboxylato-κ2O:O′)manganese(II) dihydrate, C16H20O15Mn
- Crystal structure of 4,4′-sulfonyldipyridine, C10H8N2O2S
- Crystal structure of 4-[(E)-(2-chloro-6-fluorobenzylidene)amino]-1,2-dihydro-2,3-dimethyl-1-phenylpyrazol-5-one, C18H15ClFN3O
- Crystal structure of carbonyl(2-oxopyridin-1(2H)-olato-κ2O, O′)(triphenylphosphine-κP)rhodium(I), C24H19NO3PRh
- Crystal structure of diaquabis(2-(3-bromophenyl)-5-carboxy-1H-imidazol-4-carboxylato-κ2O,N) cobalt(II) trihydrate, C22H22Br2CoN4O13
- Crystal structure of 4-(pyridin-4-ylmethylsulfonyl)pyridine, C11H10N2O2S
- Crystal structure of poly[(μ4-1-methyl-1H-tetrazole-5-thiolato-κ3S:S:N:N′)copper(I)], C2H3CuN4S
- Crystal structure of poly[diaquabis(μ2-4,4′-sulfinyldipyridine-κ2N,N′)zinc(II)] diperchlorate dihydrate, C20H24N4O14S2Cl2Zn
- Crystal structure of (1-((1-benzylpyrrolidin-2-yl-κN)methyl)-3-isopropyl-1H-imidazol-2(3H)-ylidene–κC)dibromidopalladium(II), C18H25Br2N3Pd
- Crystal structure of pentacalcium tetranitridovanadate(V) mononitride based on a powder diffraction study, Ca5[VN4]N
- Crystal structure of ((1-((1-benzylpyrrolidin-2-yl)methyl)-3-ethyl-1H-imidazol-2(3H)-ylidene)-κ2C,N)dichloridopalladium(II), C17H23Cl2N3Pd
- Crystal structure of 9-allyl-4,5-dichloro-12-cyano-9,10-dihydro-9,10-ethanoanthracen-12-yl acetate, C22H17Cl2NO2
- Crystal structure of 4-bromo-2-(8-(3-ethoxy-2-hydroxyphenyl)-3,6-dioxa-2,7-diazaocta-1,7-dien-1-yl)phenol, C18H19BrN2O5
- Crystal structure of 3-tert-butyl-3-hydroxy-1,3-dihydro-2H-pyrrolo[3,2-c]pyridin-2-one, C11H14N2O2
- Crystal structure of diaquabis(3-(3,5-dibromophenyl)-5-(pyridin-2-yl)-1,2,4-triazol-4-ido-κ2N,N′)nickel(II) mono hydrate, C26H20Br4N8NiO3
- Crystal structure of 5-(adamantan-1-yl)-3-[(2-trifluoromethylanilino)methyl]-2,3-dihydro-1,3,4-oxadiazole-2-thione, C20H22F3N3OS
- Crystal structure of 2-ethyl-1-tert-butyl-2-((4-fluorophenyl)(tert-butoxycarbonylamino)methyl)-3-oxo-pyrrolidine-1,2-dicarboxylate, C24H33FN2O7
- Crystal structure of bis(μ2-2-fluorobenzoato-κ2O:O:O′) bis(μ2-2-fluorobenzoato-κ2O:O′)dinitrato-κ2O,O′ bis(1,10-phenathroline-κ2N,N′)diterbium(III), C52H32F4N6O14Tb2
- Crystal structure of tetrabutylammonium 4-aminobenzenesulfonate 2/3 hydrate, C22H42N2O3S · 2/3 H2O
- Crystal structure of tetraethylammonium 4-aminobenzenesulfonate, C14H26N2O3S
- Crystal structure of bis(guanidinium) 3,3′-oxybis(6-carboxybenzoate), C18H20N6O9
- Crystal structure of N′-(4-methoxybenzylidene)-5-phenyl-1H-pyrazole-3-carbohydrazide, C18H16N4O2
- Crystal structure of N′-(4-nitrobenzylidene)-5-phenyl-1H-pyrazole-3-carbohydrazide, C17H13N5O3
- Crystal structure of 5-((4-bromophenyl)(2-hydroxy-6-oxocyclohex-1-en-1-yl)methyl)-6-hydroxy-1,3-dimethylpyrimidine-2,4(1H,3H)-dione, C19H19BrN2O5
- Crystal structure of 3-(((cyclohexyl(phenyl)methylidene)amino)oxy)-2-hydroxy-N-(propan-2-yl)propan-1-aminium chloride, C19H31ClN2O2
- Crystal structure of 6-hydroxy-5-((2-hydroxy-6-oxocyclohex-1-en-1-yl)(phenyl)methyl)-1,3-dimethylpyrimidine-2,4(1H,3H)-dione, C19H20N2O5
- Crystal structure of 2-(4-(4-bromophenyl)thiazol-2-yl)isoindoline-1,3-dione, C17H9BrN2O2S
- Crystal structure of 2-(4-methylbenzoyl)pyrene, C24H16O
- Crystal structure of N-(5-bromo-4-(p-tolyl)thiazol-2-yl)-4-chlorobutanamide, C14H14BrClN2OS
- Crystal structure of 4,5-diphenylthiazol-2-amine, C15H12N2S
- Crystal structure of poly[bis(μ2-4-(3-(pyridin-3-yl)-1H-1,2,4-triazol-5-yl)benzoato-κ3N:O,O′)-lead(II)], C28H18O4N8Pb
- Crystal structure of (Z)-4-(4-oxopent-2-en-2-ylamino)benzenesulfonamide, C11H14N2O3S
- Crystal structure of poly[(μ2-2-methyl-1-(4-(2-methyl-2H-benzo[d] imidazol-1(7aH)-yl)butyl)-1H-benzo[d]imidazole-κ2N:N′)bis(μ3-5-tert-butylbenzene-1,3-dicarboxylato-κ4O:O,O′:O′′,O′′′)dicadmium(II)] tetrahydrate, C44H54Cd2N4O12
- Crystal structure of catena-poly-[aqua(μ2-4,4′-bipyridine-κ2N:N′)bis(3′,5,5′-tricarboxybiphenyl-2-carboxylato-κ2O,O′)cadmium(II)], C42H28N2O17Cd
- Crystal structure of (2RS,3RS)-1-(4-chlorophenyl)-4,4-dimethyl-2-(1H-1,2,4-triazol-1-yl)pentan-3-ol, C15H20ClN3O
- Crystal structure of N′-(4-(dimethylamino)benzylidene)-5-phenyl-1H-pyrazole-3-carbohydrazide, C19H19N5O
- Crystal structure of 3-(benzofuran-2-yl)-5-(4-fluorophenyl)-4,5-dihydro-1Hpyrazole-1-carbothioamide, C18H14FN3OS
- Crystal structure of ent-1β-acetoxy-7α,14α-di-hydroxy-7β,20-epoxykaur-16-en-15-one, C22H30O6
- Crystal structure of 1α,7β-dihydroxy-11β-acetoxy-ent-7β,20-epoxykaur-16-en-15-one, C22H30O6
- Crystal structure of 7β,14β,15β-trihydroxy-1α-acetoxy-7α,20-epoxy-ent-kaurane, C22H32O6
- Crystal structure of ent-1β,7α,11α-trihydroxy-7β,20-epoxykaur-16-en-15-one, C20H28O5
- Crystal structure of poly-[tetraaquabis(μ8-benzene-1,2,4,5-tetracarboxylato-1κ3O4:O6:O8:2κ4O2:O2:O5:O5:3κ4O1:O3:O5:O7)(di-μ3-hydroxido)-pentazinc(II)] decahydrate, C20H34O32Zn5
- Crystal structure of 1-(4-methylthiazol-2-yl)-3-propylthiourea, C8H13N3S2
- Crystal structure of 2-((dimethylamino)methylene)-5,5-Dimethylcyclohexane-1,3-dione, C11H17NO2
- Crystal structure of poly[1,4-bis(2-methylbenzimidazol)butane-κ2N:N′)bis(4,4′-oxybis(benzoato-κ4O,O′:O′′,O′′′)dicadmium] monohydrate, C48H38Cd2N4O10
- Crystal structure of 2-(3-(benzofuran-2-yl)-5-(4-fluorophenyl)-4,5-dihydro-1H-pyrazol-1-yl)-4-(4-chlorophenyl)thiazole, C26H17ClFN3OS
- Crystal structure of bis(μ3-isophthalato-κ3O:O′:O′′)(μ2-1,4-\ bis((2-propyl-1H-benzo[d]imidazol-1-yl)methyl)benzene-κ2N:N′)dizinc(II), C22H19N2O4Zn
- Crystal structure of poly[bis(adipate-κ4O,O′:O′′, O′′′)(1,4-bis(2-methyl-1H-benzo[d]imidazol-1-yl)benzene-κ2N:N′)dizinc(II), C36H38N4O8Zn2
- Crystal structure of N-(2-(2-oxoindolin-4-yl)ethyl)-N-propylpropan-1-aminium tetraphenylborate, C40H45BN2O
- Crystal structure of 1-(2,3-dihydro-4-methyl-3-phenyl-2-thioxothiazol-5-yl)-1-ethanone, C12H11NOS2
- Crystal structure of 3-(adamantan-1-yl)-1-[(4-benzylpiperazin-1-yl)methyl]-4-[(E)-(2,6-difluorobenzylidene)amino]-1H-1,2,4-triazole-5(4H)-thione, C31H36F2N6S
- Crystal structure of 6-(4-chlorophenyl)-3-(thiophen-2-yl)-[1,2,4]triazolo[3,4-b][1,3,4]-thiadiazole, C13H7ClN4S2
- Crystal structure of bis(ethanaminium) poly[bis(hexaselenido-κ2Se1,Se6)palladate(II)], C4H16N2PdSe12
- Crystal structure of 2-(3-(benzofuran-2-yl)-5-phenyl-4,5-dihydro-1H-pyrazol-1-yl)-4-phenylthiazole, C26H19N3OS
- Crystal structure of poly-[bis(μ3-5-hydroxyisophtalato-κ3O:O′:O′′)(μ2-1,4-bis(2-ethylbenzimidazol-1-ylmethyl)benzene-κ2N:N′)dizinc(II)], C40H30N4O5Zn2
- Crystal structure of poly-[(μ-1,4-bis(2-ethylbenzimidazol-1-ylmethyl)benzene-κ2N:N′)-bis(μ4-2,2′-(1,3-phenylene)diacetate-κ4O:O′:O′′:O′′′)dizinc(II)], C44H38N4O8Zn2
- Crystal structure of 5,17-bis-cyano-25,26,27,28-tetrapropyloxy-calix[4]arene, C42H46N2O4
- Crystal structure of poly-[μ-1,4-bis(2-ethylbenzimidazol-1-ylmethyl)benzene-κ2N:N′)-bis(μ3-5-hydroxyisophthalate(2–)-κ3O,O′:O′′)dicadmium(II)] monohydrate, C64H54N8O11Cd2
- Crystal structure of poly-[μ-1,4-bis(2-ethylbenzimidazol-1-ylmethyl)benzene-κ2N:N′)-bis(μ4-4,4′-sulfonyldibenzoato-κ4O:O′:O′′:O′′′)dicadmium(II)] monohydrate, C52H42N4O14Cd2
- Crystal structure of poly-[bis(μ4-adipato-κ4O:O′:O′′:O′′′)(μ2-1,4-bis((2-ethyl-1H-benzo[d]imidazol-1-yl)methyl)benzene-κ2N:N′)dizinc(II)], C38H42N4O8Zn2
- Crystal structure of (2-(2-chlorophenyl)-5-methyl-1,3-dioxan-5-yl)methanol, C12H15ClO3
- Crystal structure of 2-(4-Bromophenyl)-5-ethyl-1,3-dioxane-5-carboxylic acid, C13H15BrO4
- Crystal structure of (2-(4-bromophenyl)-5-ethyl-1,3-dioxan-5-yl)methanol, C13H17BrO3
- Crystal structure of 3-((1,3,5,7-tetraoxo-6-(pyridin-3-ylmethyl)-3,3a,4,4a,5,6,7,7a,8,8a-decahydro-4,8-ethenopyrrolo[3,4-f]isoindol-2(1H)-yl)methyl)pyridin-1-ium-κN-trichloridocobalt(II) hemihydrate, C24H22Cl3CoN4O4.5
- Crystal structure of 4-chloro-4′,6′-dichloro-2,2′-[propane-1,3-diyldioxybis(nitrilomethylidyne)]-diphenol, C17H14Cl3N2O4
- Crystal structure of 2-amino-4-(3-trifluoromethylphenyl)-5-oxo-5,6,7,8-tetrahydro-4H-chromene-3-carbonitrile, C17H13F3N2O2
- Crystal structure of bis(benzoato-κO)bis(4,4′-((1H-1,2,4-triazol-1-yl)methylene)dibenzonitrile-κN)zinc(II), C48H32N10O4Zn
- Crystal structure of 3-methyl-2H-chromen-2-one, C10H8O2
- Crystal structure of catena-poly-[aqua-(2-carboxy-5-(3-carboxy-5-carboxylatophenoxy)benzoato-κO)(μ2-4,4′-di(1H-imidazol-1-yl)-1,1′-biphenyl-κ2N:N′)cobalt(II)], C34H24N4O10Co
- Crystal structure of 4,4′-di(1H-imidazol-1-yl)-1,1′-biphenyl, C36H28N8
- Crystal structure of 1-(3-chloropropyl)piperidin-1-ium tetraphenylborate, C32H37BClN
- Crystal structure of dimethyl 5-(benzylamino)isophthalate, C17H17NO4
- Crystal structure of dimethyl 5-(dibenzylamino)isophthalate, C24H23NO4
- Crystal structure of N-(adamantan-1-yl)-4-phenylpiperazine-1-carbothioamide, C21H29N3S
- Crystal structure of (2,2′(cyclohexane-1,2-diylbis(nitrilo(E)methylylidene))diphenolato-κ4O,O′,N,N′)dimethanolmanganese(III) bromide, C22H28BrMnN2O4
- Crystal structure of 3,5,7-tris(morpholinomethyl)tropolone·0.67 hydrate, C22H33N3O5·0.67H2O
- Crystal structure of biphenyl-2,3′,5,5′-tetracarboxylic acid – 4,4′-biphenyl-4,4′-diyldipyridine (3/2), C49H34N3O8
Artikel in diesem Heft
- Frontmatter
- Crystal structure of poly[aqua(μ2-2,5-bis(4-pyridyl)-1,3,4-oxadiazole-κ2N,N)(μ2-1,3-phenylenediacetato-κ3O,O′:O′′)cobalt(II)], C22H18CoN4O6
- Crystal structure of 2-amino-7,7-dimethyl-5-oxo-4-(3-phenoxy-phenyl)-5,6,7,8-tetrahydro-4H-chromene-3-carbonitrile
- Crystal structure of (E)-4,4′-(diazene-1,2-diyl)bis(1-nitro-1H-1,2,4-triazol-5(4H)-one)—acetonitrile (1:1), C6H5N11O6
- Crystal structure of potassium (E)-5-oxo-4-((5-oxo-1H-1,2,4-triazol-4(5H)-yl)diazenyl)-4,5-dihydro-1,2,4-triazol-1-ide – (E)-4,4-diazene-1,2-diylbis(2,4-dihydro-3H-1,2,4-triazol-3-one) – methanol (1/1/1), C9H11N16KO5
- Crystal structure of (N,N′-bis(2-(((2,6-diisopropylphenyl)imino)methyl)phenyl)benzene-1,2-diamido-κ4O,O′,O′′,O′′′)oxidovanadium(IV), C44H48N4OV
- Crystal structure of 5,5-bis(4-iodophenyl)-5H-cyclopenta[2,1-b:3,4-b′]dipyridine, C23H14I2N2
- Crystal structure of aquadichloridobis(1-((2-methyl-1H-imidazol-1-yl)methyl)-1H-benotriazole-κN)mercury(II), C22H24Cl2HgN10O
- Crystal structure of 2-[4-(1H-imidazol-1-yl)phenyl]-1H-benzimidazol-3-ium [2-(carboxymethyl)phenyl]acetate monohydrate, C26H24N4O5
- Crystal structure of 4,4′-bipiperidinium dichloride 0.12 hydrate, C10H22N2Cl2 · 0.12 H2O
- Crystal structure of catena-poly[tetraaqua(μ2-4,4′(E)-ethene-1,2-diyldipyridine-κ2N:N′)nickel(II)] bis(6-methyl-2-oxo-1,2-dihydro-pyridine-4-carboxylate) pentahydrate, C26H42N4O15Ni
- Crystal structure of ethyl-5-amino-1-(2,4-dinitrophenyl)-1H-pyrazole-4-carboxylate, C12H11N5O6
- Crystal structure of dicarbonyl(pyridin-2-olate-1-oxido-κ2O,O′)rhodium(I), C7H4NO4Rh
- Crystal structure of 1-(adamantan-1-yl)-3-(4-chlorophenyl)thiourea, C17H21ClN2S
- Crystal structure of 2-((2-chloropyridin-3-ylamino)methylene)malononitrile, C9H5ClN4
- Crystal structure of tetraaquabis(μ2-4-chlorobenzoato-κ2O:O,O′)bis(4-chlorobenzoato-κO)bis(1,10-phenanthroline-κ2N,N′)distrontium(II), C52H40Cl4N4O12Sr2
- Crystal structure of 3-hydroxy-3-phenyl-1,3-dihydro-2H-indol-2-one, C14H11NO2
- Crystal structures of bis(1,10-phenanthrolin-1-ium) aquapentakis(nitrato-κ2O,O′)neodym(III) monohydrate, C24H22N9NdO17
- Crystal structure of 2-ethyl-1-tert-butyl 3-oxo-2-[phenyl(tert-butoxycarbonylamino)methyl]-1,2-pyrrolidinedicarboxylate, C24H34N2O7
- Crystal structure of poly[diaquabis(μ4-benzene-1,3,5-tricarboxylato-κO1,κO2:κO3,κO4:κO5:κO6)-bis(μ2-4,4′-benzene-1,3-diylbis(4H-1,2,4-triazole-κ2N:N′)tricadmium(II)] tetrahydrate, C38H34Cd3N12O18
- Crystal structure of trans-dichlorido[1,3-bis(9-methyl-9H-fluoren-9-yl) benzimidazol-2-ylidene](pyridine)palladium(II) – a compound with anagostic CH–Pd interactions, C40H31Cl2N3Pd
- Crystal structure of catena-poly[(μ3-5-(4-(tetrazol-1-id-5-yl)phenoxy)benzene-1,3-dicarboxyato-κ3O:O′:N)(4-(3-(pyridin-4-yl)propyl)pyridinium-κN)zinc(II)], C28H22ZnN6O5
- Crystal structure of 3,6-di-2-pyridinyl-4-pyridazine carbonitrile, C15H9N5
- Crystal structure of 5,5,9,13-tetramethyltetracyclo[10·2·1·01,10·04,9]pentadecane-3,7,14-triol, C20H34O4
- Crystal structure of 2-amino-7-methyl-5-oxo-4-phenyl-4H,5H-pyrano[4,3-b]pyran-3-carbonitrile, C16H12N2O3
- Crystal structure of the poly[(1,10-phenanthroline-κ2N,N′)(μ3-carboxylatophenoxyacetato-κ4O,O′:O′′;O′′′)lead(II)] monohydrate, C21H16N2O6Pb
- Crystal structure of the poly[(μ4-biphenyl-4,4′-dicarboxylato-κ4O:O′:O′′:O′′′) bis(μ3-8-(11-(oxysulfonyl)-4-silbenyl)-2-(oxysulfonyl)stilbene-κ4O:O′:O′′,O′′′) bis(1,10-phenanthroline-k2N,N′) dipraseodymium(III)], C94H64N4O16Pr2S4
- Crystal structure of 3-iodo-5-methoxy-7-(methoxymethoxy)-4-(3-methoxyphenoxy)-2H-chromen-2-one, C19H17IO7
- Crystal structure of poly[(5-carboxy-2,6-dimethylpyridinium-3-carboxylato-κO)tris(μ2-2,6-dimethylpyridinium-3,5-dicarboxylato-κ3O,O′:O′′)erbium(III)], C36H33ErN4O16
- This molecule targets at type 2 diabetes - a single crystal study on (2R,3S,5R)-2-(2,5-difluorophenyl)-5-[2-(methylsulfonyl)-2,6-dihydropyrrolo[3,4-c]pyrazol-5(4H)-yl] tetrahydro-2H-pyran-3-amine (Omarigliptin), C17H20F2N4O3S
- Crystal structure of poly[(μ2-2,2′-benzene-1,2-diyldiacetato-κ2O:O′), (μ2-1-(4-(1H-imidazol-1-yl)benzyl)-1H-1,2,4-triazole-κ2N:N′)zinc(II)], C22H19N5O4Zn
- Crystal structure of tetraaqua(μ2-3-(3,5-dicarboxyphenoxy)benzene-1,2-dicarboxylato-κ2O:O′)manganese(II) dihydrate, C16H20O15Mn
- Crystal structure of 4,4′-sulfonyldipyridine, C10H8N2O2S
- Crystal structure of 4-[(E)-(2-chloro-6-fluorobenzylidene)amino]-1,2-dihydro-2,3-dimethyl-1-phenylpyrazol-5-one, C18H15ClFN3O
- Crystal structure of carbonyl(2-oxopyridin-1(2H)-olato-κ2O, O′)(triphenylphosphine-κP)rhodium(I), C24H19NO3PRh
- Crystal structure of diaquabis(2-(3-bromophenyl)-5-carboxy-1H-imidazol-4-carboxylato-κ2O,N) cobalt(II) trihydrate, C22H22Br2CoN4O13
- Crystal structure of 4-(pyridin-4-ylmethylsulfonyl)pyridine, C11H10N2O2S
- Crystal structure of poly[(μ4-1-methyl-1H-tetrazole-5-thiolato-κ3S:S:N:N′)copper(I)], C2H3CuN4S
- Crystal structure of poly[diaquabis(μ2-4,4′-sulfinyldipyridine-κ2N,N′)zinc(II)] diperchlorate dihydrate, C20H24N4O14S2Cl2Zn
- Crystal structure of (1-((1-benzylpyrrolidin-2-yl-κN)methyl)-3-isopropyl-1H-imidazol-2(3H)-ylidene–κC)dibromidopalladium(II), C18H25Br2N3Pd
- Crystal structure of pentacalcium tetranitridovanadate(V) mononitride based on a powder diffraction study, Ca5[VN4]N
- Crystal structure of ((1-((1-benzylpyrrolidin-2-yl)methyl)-3-ethyl-1H-imidazol-2(3H)-ylidene)-κ2C,N)dichloridopalladium(II), C17H23Cl2N3Pd
- Crystal structure of 9-allyl-4,5-dichloro-12-cyano-9,10-dihydro-9,10-ethanoanthracen-12-yl acetate, C22H17Cl2NO2
- Crystal structure of 4-bromo-2-(8-(3-ethoxy-2-hydroxyphenyl)-3,6-dioxa-2,7-diazaocta-1,7-dien-1-yl)phenol, C18H19BrN2O5
- Crystal structure of 3-tert-butyl-3-hydroxy-1,3-dihydro-2H-pyrrolo[3,2-c]pyridin-2-one, C11H14N2O2
- Crystal structure of diaquabis(3-(3,5-dibromophenyl)-5-(pyridin-2-yl)-1,2,4-triazol-4-ido-κ2N,N′)nickel(II) mono hydrate, C26H20Br4N8NiO3
- Crystal structure of 5-(adamantan-1-yl)-3-[(2-trifluoromethylanilino)methyl]-2,3-dihydro-1,3,4-oxadiazole-2-thione, C20H22F3N3OS
- Crystal structure of 2-ethyl-1-tert-butyl-2-((4-fluorophenyl)(tert-butoxycarbonylamino)methyl)-3-oxo-pyrrolidine-1,2-dicarboxylate, C24H33FN2O7
- Crystal structure of bis(μ2-2-fluorobenzoato-κ2O:O:O′) bis(μ2-2-fluorobenzoato-κ2O:O′)dinitrato-κ2O,O′ bis(1,10-phenathroline-κ2N,N′)diterbium(III), C52H32F4N6O14Tb2
- Crystal structure of tetrabutylammonium 4-aminobenzenesulfonate 2/3 hydrate, C22H42N2O3S · 2/3 H2O
- Crystal structure of tetraethylammonium 4-aminobenzenesulfonate, C14H26N2O3S
- Crystal structure of bis(guanidinium) 3,3′-oxybis(6-carboxybenzoate), C18H20N6O9
- Crystal structure of N′-(4-methoxybenzylidene)-5-phenyl-1H-pyrazole-3-carbohydrazide, C18H16N4O2
- Crystal structure of N′-(4-nitrobenzylidene)-5-phenyl-1H-pyrazole-3-carbohydrazide, C17H13N5O3
- Crystal structure of 5-((4-bromophenyl)(2-hydroxy-6-oxocyclohex-1-en-1-yl)methyl)-6-hydroxy-1,3-dimethylpyrimidine-2,4(1H,3H)-dione, C19H19BrN2O5
- Crystal structure of 3-(((cyclohexyl(phenyl)methylidene)amino)oxy)-2-hydroxy-N-(propan-2-yl)propan-1-aminium chloride, C19H31ClN2O2
- Crystal structure of 6-hydroxy-5-((2-hydroxy-6-oxocyclohex-1-en-1-yl)(phenyl)methyl)-1,3-dimethylpyrimidine-2,4(1H,3H)-dione, C19H20N2O5
- Crystal structure of 2-(4-(4-bromophenyl)thiazol-2-yl)isoindoline-1,3-dione, C17H9BrN2O2S
- Crystal structure of 2-(4-methylbenzoyl)pyrene, C24H16O
- Crystal structure of N-(5-bromo-4-(p-tolyl)thiazol-2-yl)-4-chlorobutanamide, C14H14BrClN2OS
- Crystal structure of 4,5-diphenylthiazol-2-amine, C15H12N2S
- Crystal structure of poly[bis(μ2-4-(3-(pyridin-3-yl)-1H-1,2,4-triazol-5-yl)benzoato-κ3N:O,O′)-lead(II)], C28H18O4N8Pb
- Crystal structure of (Z)-4-(4-oxopent-2-en-2-ylamino)benzenesulfonamide, C11H14N2O3S
- Crystal structure of poly[(μ2-2-methyl-1-(4-(2-methyl-2H-benzo[d] imidazol-1(7aH)-yl)butyl)-1H-benzo[d]imidazole-κ2N:N′)bis(μ3-5-tert-butylbenzene-1,3-dicarboxylato-κ4O:O,O′:O′′,O′′′)dicadmium(II)] tetrahydrate, C44H54Cd2N4O12
- Crystal structure of catena-poly-[aqua(μ2-4,4′-bipyridine-κ2N:N′)bis(3′,5,5′-tricarboxybiphenyl-2-carboxylato-κ2O,O′)cadmium(II)], C42H28N2O17Cd
- Crystal structure of (2RS,3RS)-1-(4-chlorophenyl)-4,4-dimethyl-2-(1H-1,2,4-triazol-1-yl)pentan-3-ol, C15H20ClN3O
- Crystal structure of N′-(4-(dimethylamino)benzylidene)-5-phenyl-1H-pyrazole-3-carbohydrazide, C19H19N5O
- Crystal structure of 3-(benzofuran-2-yl)-5-(4-fluorophenyl)-4,5-dihydro-1Hpyrazole-1-carbothioamide, C18H14FN3OS
- Crystal structure of ent-1β-acetoxy-7α,14α-di-hydroxy-7β,20-epoxykaur-16-en-15-one, C22H30O6
- Crystal structure of 1α,7β-dihydroxy-11β-acetoxy-ent-7β,20-epoxykaur-16-en-15-one, C22H30O6
- Crystal structure of 7β,14β,15β-trihydroxy-1α-acetoxy-7α,20-epoxy-ent-kaurane, C22H32O6
- Crystal structure of ent-1β,7α,11α-trihydroxy-7β,20-epoxykaur-16-en-15-one, C20H28O5
- Crystal structure of poly-[tetraaquabis(μ8-benzene-1,2,4,5-tetracarboxylato-1κ3O4:O6:O8:2κ4O2:O2:O5:O5:3κ4O1:O3:O5:O7)(di-μ3-hydroxido)-pentazinc(II)] decahydrate, C20H34O32Zn5
- Crystal structure of 1-(4-methylthiazol-2-yl)-3-propylthiourea, C8H13N3S2
- Crystal structure of 2-((dimethylamino)methylene)-5,5-Dimethylcyclohexane-1,3-dione, C11H17NO2
- Crystal structure of poly[1,4-bis(2-methylbenzimidazol)butane-κ2N:N′)bis(4,4′-oxybis(benzoato-κ4O,O′:O′′,O′′′)dicadmium] monohydrate, C48H38Cd2N4O10
- Crystal structure of 2-(3-(benzofuran-2-yl)-5-(4-fluorophenyl)-4,5-dihydro-1H-pyrazol-1-yl)-4-(4-chlorophenyl)thiazole, C26H17ClFN3OS
- Crystal structure of bis(μ3-isophthalato-κ3O:O′:O′′)(μ2-1,4-\ bis((2-propyl-1H-benzo[d]imidazol-1-yl)methyl)benzene-κ2N:N′)dizinc(II), C22H19N2O4Zn
- Crystal structure of poly[bis(adipate-κ4O,O′:O′′, O′′′)(1,4-bis(2-methyl-1H-benzo[d]imidazol-1-yl)benzene-κ2N:N′)dizinc(II), C36H38N4O8Zn2
- Crystal structure of N-(2-(2-oxoindolin-4-yl)ethyl)-N-propylpropan-1-aminium tetraphenylborate, C40H45BN2O
- Crystal structure of 1-(2,3-dihydro-4-methyl-3-phenyl-2-thioxothiazol-5-yl)-1-ethanone, C12H11NOS2
- Crystal structure of 3-(adamantan-1-yl)-1-[(4-benzylpiperazin-1-yl)methyl]-4-[(E)-(2,6-difluorobenzylidene)amino]-1H-1,2,4-triazole-5(4H)-thione, C31H36F2N6S
- Crystal structure of 6-(4-chlorophenyl)-3-(thiophen-2-yl)-[1,2,4]triazolo[3,4-b][1,3,4]-thiadiazole, C13H7ClN4S2
- Crystal structure of bis(ethanaminium) poly[bis(hexaselenido-κ2Se1,Se6)palladate(II)], C4H16N2PdSe12
- Crystal structure of 2-(3-(benzofuran-2-yl)-5-phenyl-4,5-dihydro-1H-pyrazol-1-yl)-4-phenylthiazole, C26H19N3OS
- Crystal structure of poly-[bis(μ3-5-hydroxyisophtalato-κ3O:O′:O′′)(μ2-1,4-bis(2-ethylbenzimidazol-1-ylmethyl)benzene-κ2N:N′)dizinc(II)], C40H30N4O5Zn2
- Crystal structure of poly-[(μ-1,4-bis(2-ethylbenzimidazol-1-ylmethyl)benzene-κ2N:N′)-bis(μ4-2,2′-(1,3-phenylene)diacetate-κ4O:O′:O′′:O′′′)dizinc(II)], C44H38N4O8Zn2
- Crystal structure of 5,17-bis-cyano-25,26,27,28-tetrapropyloxy-calix[4]arene, C42H46N2O4
- Crystal structure of poly-[μ-1,4-bis(2-ethylbenzimidazol-1-ylmethyl)benzene-κ2N:N′)-bis(μ3-5-hydroxyisophthalate(2–)-κ3O,O′:O′′)dicadmium(II)] monohydrate, C64H54N8O11Cd2
- Crystal structure of poly-[μ-1,4-bis(2-ethylbenzimidazol-1-ylmethyl)benzene-κ2N:N′)-bis(μ4-4,4′-sulfonyldibenzoato-κ4O:O′:O′′:O′′′)dicadmium(II)] monohydrate, C52H42N4O14Cd2
- Crystal structure of poly-[bis(μ4-adipato-κ4O:O′:O′′:O′′′)(μ2-1,4-bis((2-ethyl-1H-benzo[d]imidazol-1-yl)methyl)benzene-κ2N:N′)dizinc(II)], C38H42N4O8Zn2
- Crystal structure of (2-(2-chlorophenyl)-5-methyl-1,3-dioxan-5-yl)methanol, C12H15ClO3
- Crystal structure of 2-(4-Bromophenyl)-5-ethyl-1,3-dioxane-5-carboxylic acid, C13H15BrO4
- Crystal structure of (2-(4-bromophenyl)-5-ethyl-1,3-dioxan-5-yl)methanol, C13H17BrO3
- Crystal structure of 3-((1,3,5,7-tetraoxo-6-(pyridin-3-ylmethyl)-3,3a,4,4a,5,6,7,7a,8,8a-decahydro-4,8-ethenopyrrolo[3,4-f]isoindol-2(1H)-yl)methyl)pyridin-1-ium-κN-trichloridocobalt(II) hemihydrate, C24H22Cl3CoN4O4.5
- Crystal structure of 4-chloro-4′,6′-dichloro-2,2′-[propane-1,3-diyldioxybis(nitrilomethylidyne)]-diphenol, C17H14Cl3N2O4
- Crystal structure of 2-amino-4-(3-trifluoromethylphenyl)-5-oxo-5,6,7,8-tetrahydro-4H-chromene-3-carbonitrile, C17H13F3N2O2
- Crystal structure of bis(benzoato-κO)bis(4,4′-((1H-1,2,4-triazol-1-yl)methylene)dibenzonitrile-κN)zinc(II), C48H32N10O4Zn
- Crystal structure of 3-methyl-2H-chromen-2-one, C10H8O2
- Crystal structure of catena-poly-[aqua-(2-carboxy-5-(3-carboxy-5-carboxylatophenoxy)benzoato-κO)(μ2-4,4′-di(1H-imidazol-1-yl)-1,1′-biphenyl-κ2N:N′)cobalt(II)], C34H24N4O10Co
- Crystal structure of 4,4′-di(1H-imidazol-1-yl)-1,1′-biphenyl, C36H28N8
- Crystal structure of 1-(3-chloropropyl)piperidin-1-ium tetraphenylborate, C32H37BClN
- Crystal structure of dimethyl 5-(benzylamino)isophthalate, C17H17NO4
- Crystal structure of dimethyl 5-(dibenzylamino)isophthalate, C24H23NO4
- Crystal structure of N-(adamantan-1-yl)-4-phenylpiperazine-1-carbothioamide, C21H29N3S
- Crystal structure of (2,2′(cyclohexane-1,2-diylbis(nitrilo(E)methylylidene))diphenolato-κ4O,O′,N,N′)dimethanolmanganese(III) bromide, C22H28BrMnN2O4
- Crystal structure of 3,5,7-tris(morpholinomethyl)tropolone·0.67 hydrate, C22H33N3O5·0.67H2O
- Crystal structure of biphenyl-2,3′,5,5′-tetracarboxylic acid – 4,4′-biphenyl-4,4′-diyldipyridine (3/2), C49H34N3O8


