Abstract
Neuroblastoma (NB) is a commonly occurring malignancy in children. Epithelial-mesenchymal transition (EMT) is an adaptive change in promoting tumor metastasis. As an important factor in regulating tumor metastasis, whether legumain could promote metastasis of NB by EMT is still unexplored. Legumain is the active form of prolegumain, abundant in tumor plasma. So in the current study, different forms of legumain were identified in NB. Second, correlation analysis of N-cadherin and active legumain was identified by western blot analysis. Third, legumain gene amplification or gene knockdown were proceeded to examine the effect of legumain on EMT by scratch and transwell assay; meanwhile, active mature legumain or its asparagine endopeptidase (AEP) inhibitor was also added in. Finally, legumain can be detected differently in NB cells. Changes in legumain could influence NB metastasis by regulating EMT markers (e.g., N-cadherin, vimentin, and slug). Besides, the effect of legumain on EMT by its AEP activity was proved by intervention experiment of AEP gene transfection and gene knockdown experiments or adding recombinant human legumain suspension or specific inhibitor of AEP in NB cells (p < 0.05). These results suggest that legumain can promote invasion and migration of NB by regulating EMT, and EMT of NB is regulated by AEP activity of legumain, which can be inhibited by a specific AEP inhibitor.
1 Introduction
Neuroblastoma (NB), a solid tumor caused by rapid division of undifferentiated neuroblasts, is the most common malignancy affecting children aged <5 years, with an incidence rate of 0.3/100,000–5.5/100,000 [1,2], accounting for 10% of pediatric malignancies and 15% of children’s deaths from tumors [3]. Approximately 650 new cases of NB are diagnosed in the USA every year, 90% of patients are diagnosed before 5 years of age, and 70% of patients have distal tumor metastasis while being diagnosed.
Adaptive changes in the tumor microenvironment and cells promote tumor migration and metastasis. A large number of processes influence the tumor metastasis and migration process, such as chemotaxis [4], microRNA [5], long non-coding RNAs [6], and also proteases.
Proteases present in the tumor microenvironment and within tumor cells can regulate tumor invasion and migration by regulating factors in the tumor cytoplasm and tumor stroma [7]. Legumain is a conservative cysteine protease belonging to the C13 family that is overexpressed in solid tumors but is scarce in normal tissues [8]. It can be presented with three different forms: prolegumain, asparagine carboxypeptidase (ACP), and asparagine endopeptidase (AEP, also called legumain) under different atmospheres. Correlation analysis in solid tumors, such as gastric tumors [9], colon cancer [10], cervical cancer [11], ovarian tumors [12], and breast cancer [13], showed that the overexpression of legumain is associated with a poor prognosis. Legumain is highly abundant in tumor stroma and is also found in the cytoplasm and on the surface of tumor cells. It is abundant in M2 macrophages (tumor-associated macrophages) [14] and tumor blood vessels that facilitate tumor metastasis and migration. It promotes tumor metastasis mainly by hydrolyzing metastasis-related substrates in the tumor stroma, for example, fibronectin [15], progelatinase A [16], cathepsin L [17], and matrix metalloproteinases (MMPs). In addition, it can act as a different type of protease in other situations [18]and also as a transcription factor in the nucleus of colon cancer cells [19].
Epithelial-mesenchymal transition (EMT) is an important process that allows tumor cells to change, escape, and migrate to distant sites [20,21]. As cells undergo EMT, they also develop a mesenchymal phenotype and start expressing mesenchymal markers such as N-cadherin and vimentin [22]. In a study based on a rapid autopsy program for patients with pancreatic cancer, 75% of primary tumors with mesenchymal features developed metastatic lesions in the liver and lung [23].
Legumain plays an important role in regulating metastasis-related factors in the tumor stroma, but it is unclear whether it plays a role in tumor metastasis-related EMT in NB. The purpose of this investigation was to determine whether legumain is involved in EMT of NB, and if so, through what mechanism. Plasmid transfection to produce legumain gene overexpression in SK-N-BE2 cells and gene knockdown by siRNA in SH-SY5Y and IMR32 cells were used to evaluate the legumain effect on the EMT of NB. In addition, whether its effect on EMT was based on its AEP activity was evaluated using activated legumain or specific legumain inhibitor experiments in NB.
2 Materials and methods
2.1 Legumain expression in NB cell lines and NB tumor tissues
The human NB cell lines SH-SY5Y, IMR32, and SK-N-BE2 were purchased from the Type Culture Collection of the Chinese Academy of Sciences, Shanghai, China. These three cell lines were, respectively, grown in DMEM/F12, MEM/EBSS, and RPMI 1640 (Gibco; Thermo Fisher Scientific, Inc., Waltham, MA, USA) culture medium containing 10% fetal bovine serum (FBS; Gibco; Thermo Fisher Scientific, Inc.) at 37°C in a humidified 5% CO2 atmosphere. Legumain expression of the three cell lines was assayed by western blot analysis. Adherent cells were resuspended in RIPA buffer mixed with a protease inhibitor cocktail (1:1,000; Thermo Fisher Scientific, Inc.) and then centrifuged at 10,000g for 15 min at 4°C. The protein supernatant was transferred to new tubes and qualified by a BCA kit. Equal amounts of protein samples (10 µg/well) were electrophoretized by 10% SDS-PAGE electrophoresis and transferred to polyvinylidene fluoride(PVDF) membranes (Roche; Mannheim, Germany). After blocking with 5% skimmed milk, incubated with a primary antibody against legumain (1:500; cat. no. ab125286, Abcam, Cambridge, MA, USA) overnight and secondary antibody for 1 h, the membrane was incubated with SuperSignal West Pico (Thermo Fisher Scientific, Inc.) and then exposed.
The location of legumain in tumor cells and solid tumor tissues was examined by immunofluorescence assays. Glass slides with attached SH-SY5Ycells were fixed in formaldehyde for at least 30 min, washed twice with phosphate-buffered saline (PBS), blocked with 5% FBS for 2 h, and incubated with a primary antibody against legumain (1:500; cat. no. ab125286) overnight at 4°C and then with a secondary antibody against anti-rabbit IgG fluorescein (1:500; cat. no. ab150077) for 1 h in the dark at 37°C. The cell nuclei were stained with DAPI (10 mg/mL, Beyotime, SHH, CHN) for 30 min, then mounted with coverslips and observed by fluorescence microscope. Paraffin-embedded tissue sections of a tumor-bearing mouse model of SH-SY5Y, which was deparaffinized, hydrated, and incubated first with a primary antibody against legumain at 4°C overnight, were performed and then proceeded with the procedures as above.
2.2 N-Cadherin and legumain expression in NB tumor samples
Tumor samples from nine cases of NB were collected in Shanghai Xinhua Hospital. Samples were pulverized and resuspended in RIPA buffer with a protease inhibitor cocktail and then centrifuged at 10,000g for 15 min at 4°C. The protein concentration was tested using a BCA kit. Equal amounts of protein samples (40 µg/well) were separated electrophoretically on 10% SDS-PAGE and transferred to PVDF membranes. After blocking in PBS-Tween 20 containing 5% skimmed milk (diluted with 0.1% PBST) for 1 h, the membranes were each incubated with a primary antibody against legumain (1:1,000; cat. no. ab125286), N-cadherin (1:1,000; cat. no. 8457, Cell Signaling Technology, Inc., Danvers, MA, USA), and GAPDH (1:1,000; cat. no. 5174, Cell Signaling Technology, Inc.) overnight at 4°C, washed three times with PBS-Tween 20, and incubated with a goat-anti-rabbit peroxidase-labeled secondary antibody for 1 h. After three washes with PBST, the membrane was incubated with SuperSignal West Pico and then exposed.
-
Informed consent: Informed consent has been obtained from all individuals included in this study.
-
Ethical approval: The research related to human use has been complied with all the relevant national regulations, institutional policies, and in accordance with the tenets of the Helsinki Declaration, and has been approved by the Ethics Committee of Xinhua Hospital Affiliated to Shanghai Jiao Tong University School of Medicine Approval No. XHEC-D-2015-103, 28 February, 2015.
2.3 Legumain gene knockdown by siRNA or overexpression by plasmid transfection
SiRNA was transfected to reduce legumain expression in NB cell lines SH-SY5Y and IMR32. Cells were seeded in a 6-well plate, grown to the confluence of 60–70%, and transfected with RNAimax 9 µL (Thermo Fisher Scientific, Inc.) mixed with siRNA 90 pmol (Genomeditech, SHH, CHN) for each well. Proteins in the cell lysate were extracted 72 h after transfection and used in the following experiments. Carboxyfluorescein and western blot analysis were used for assessing knockdown efficiency.
Plasmids carrying human legumain genes were transfected to increase legumain expression in the NB cell line SK-N-BE2. Cells were seeded in 6-well plates, grown to a confluence of 60–70%, and transfected with Lipo2000 5 µL (Thermo Fisher Scientific, Inc.) mixed with legumain containing 2.5 µg plasmid (GeneChem, SHH, CHN) in each well. Proteins in the cell lysate were extracted 72 h after transfection and used in the following experiments. The efficiency of transfection was examined by western blot analysis.
2.4 Invasiveness and migration assay
The invasiveness and migration ability of the different experimental groups below were tested by the scratch test or transwell test. NB cells (SH-SY5Y and IMR32) were seeded in a 6-well plate and grown to a confluence of 60–70%. After 48 h of legumain gene knockdown by siRNA, a line created by a 20 µL pipette tip was scratched into a confluent monolayer of cells to investigate cell motility. Pictures were taken immediately after the scratch and 24 h later, and distance measurements were carried out three times.
After legumain gene knockdown by siRNA or legumain gene overexpression by plasmid transfection for 48 h, cells that had invaded a polycarbonate membrane or Matrigel-coated polycarbonate membrane were fixed in 10% paraformaldehyde for 30 min, stained with crystal violet for 10 min, and then were counted under a microscope. Active mature recombinant human legumain (AEP, 2 µg) or legumain inhibitor (AEPI, 5 µL, 20 mmol/L) were added to the cells seeded in the transwell chambers, and pictures were taken 24 h after treatment.
2.5 Effects of recombinant human legumain (RhLegumain) and legumain inhibitors on EMT
RhLegumain purified protein (Novoprotein, SHH, CHN) and legumain inhibitor (AEPI, 5 µL, 20 mmol/L) were added into NB cell line SH-SY5Y to test their invasiveness and migration. RhLegumain (5 µg, 1 mg/mL) was activated under activation solution (50 mM citric acid, 121 mM Na2HPO4, 1 mM EDTA, and pH 4.1) for 3 h before adding to the cells [24], and legumain inhibitor (diluted in DMSO) was incubated with the culture medium for 30 min before adding to the cells, the hydrolyzing effect of AEP or enzyme-inhibition by AEPI was checked by hydrolyzing the substrate Z-ala-ala-asn-AMC (GL Biochem, SHH, CHN). The vehicle was used as a control. Transwell test and EMT markers were also examined.
2.6 Statistical analysis
All experiments were repeated at least three times. The results are expressed as the mean value ± SEM. Student’s paired t-test was used to analyze the differences between the two groups. A p-value <0.05 was defined to indicate statistical significance.
3 Results
3.1 Legumain expression in NB
Legumain expression in SK-N-BE2, SH-SY5Y, and IMR-32 cells was confirmed by western blotting (Figure 1a). SK-N-BE2 had more precursor forms than the active mature forms of legumain, but the other two cell lines, SH-SY5Y and IMR-32, had more active forms than precursor forms. In the immunofluorescence assay, legumain expression was mainly localized to the cytoplasm of the tumor cells. Besides, examining tumor xenograft models from SH-SY5Y also confirmed high levels of legumain, mainly in the cytoplasm and extracellular matrix of NB (Figure 1b).

Legumain expression in NB. (a) Legumain expression in NB cell lines SK-N-BE2, SH-SY5Y, and IMR32 by western blotting analysis. (b) Immunofluorescence staining in SH-SY5Y (20×) and tumor xenograft (10×). Legumain (green fluorescence) was localized in the cytoplasm and extracellular matrix. Cell nuclei are stained blue.
3.2 N-Cadherin was positively correlated with active legumain expression in NB tumor samples
Nine NB tumor samples were collected from nine patients at the Shanghai Xinhua Hospital. Clinical pathology confirmed the diagnosis of NB. Legumain was detected in all nine samples. Three forms of legumain were observed in the tumor samples and were identified as the precursor form (prolegumain, 56 kDa), intermediate form (ACP, 46 kDa), and mature active form (AEP, 36 kDa). As observed in our experiment, when the active form of legumain (36 kDa) levels were relatively high (sample numbers 6, 8, and 9 shown by a triangle), the corresponding N-cadherin expression was also at a relatively high level. On the contrary, while mature active legumain expression was low but prolegumain was high, N-cadherin expression was relatively low (sample numbers 2 and 3, shown by the arrow). Therefore, active mature legumain is positively correlated with N-cadherin expression, which suggests that active legumain may regulate N-cadherin expression (Figure 2).
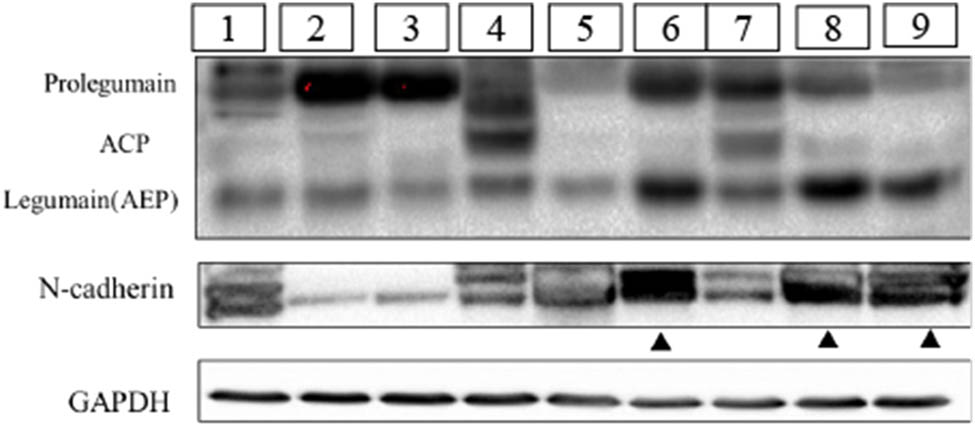
Correlation analysis of Legumain and N-cadherin in the clinical NB samples in the western blot assay. Three forms of legumain can be tested in NB, including precursor form (prolegumain, 56 kDa), intermediate form (ACP, 46 kDa), and mature active form (legumain, 36 kDa). Tumor samples were labeled from number 1 to number 9. Samples as the triangle pointed (numbers 6, 8, and 9) presented with a high level of active mature legumain (36 kDa) and N-cadherin. Samples pointed by the arrow (numbers 2 and 3) showed less expression of legumain and N-cadherin.
3.3 Legumain increases invasiveness and migration by modulating EMT
Western blot analysis showed a diversity of legumain expression in the three NB cell lines, a low level in SK-N-BE2 and a high level in SH-SY5Y and IMR32. Therefore, legumain was knocked down by siRNA in the cell lines SH-SY5Y and IMR32 (Figure 3a). Knockdown of legumain reduced the migration and invasion of SH-SY5Y and IMR32, which was examined by a transwell assay (Figure 4) and a scratch assay (p = 0.006 for SH-SY5Y; p = 0.0016 for IMR32) (Figure 5).
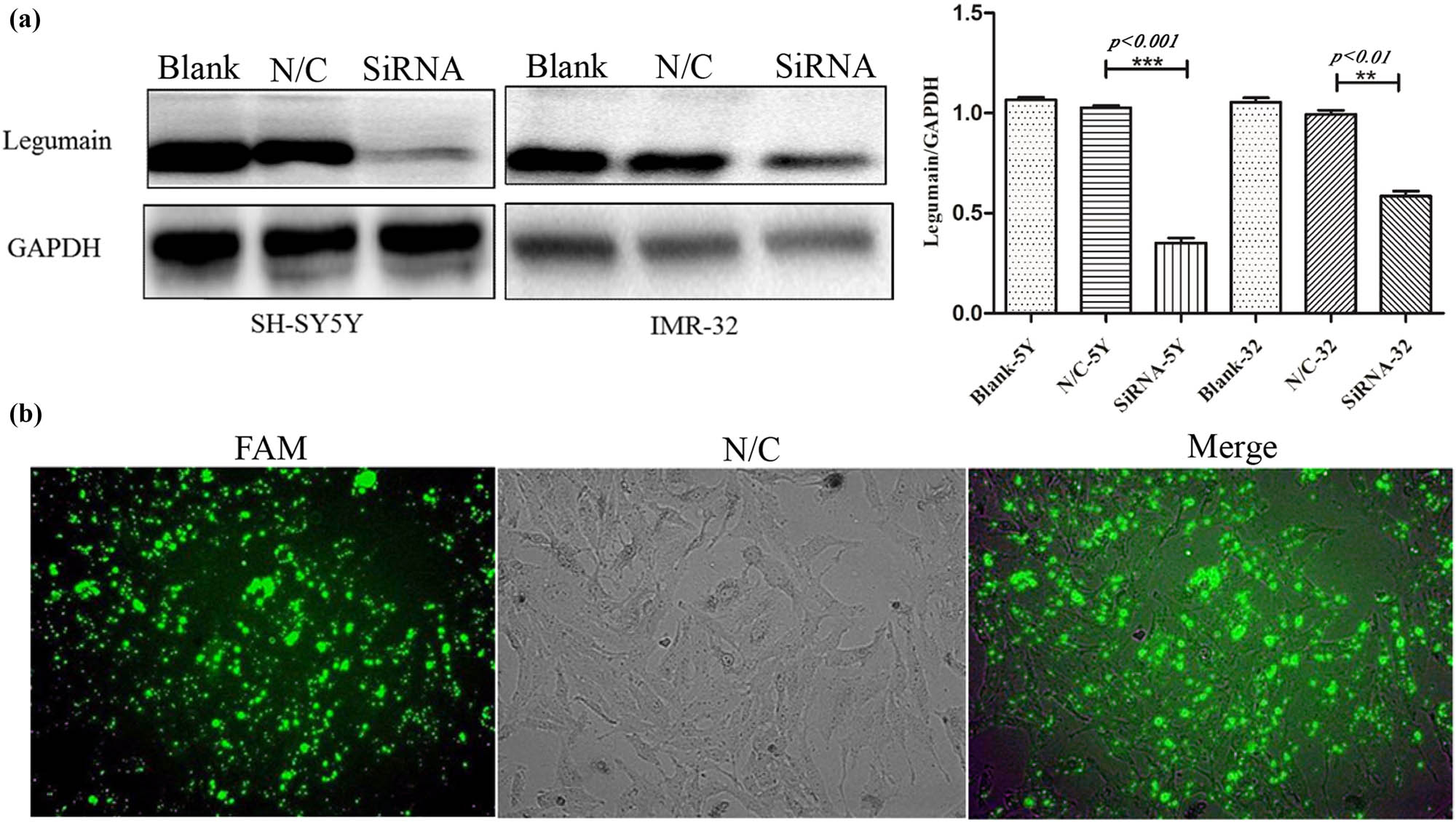
Knockdown of legumain by siRNA. (a) Knockdown of legumain by siRNA in SH-SY5Y and IMR32 cell lines was confirmed by western blot analysis. (b) Knockdown efficiency was also confirmed by FAM fluorescence coloration.

Migration and invasion tests by transwell assay. Cell counts (10×) passing through the transwell chamber after a legumain knockdown by siRNA decreased in cell lines SH-SY5Y and IMR32 compared with the control, p < 0.05 (***p < 0.001 and **p < 0.01). The procedure and statistic analysis is similar. (a) and (b) is different only in treatment of transwell membrane. In order to give brief explanation, a and b can be briefly summarized.

Gap distance (10×) increased after legumain knockdown by siRNA in SH-SY5Y and IMR32 in scratch assay compared with the control, p < 0.05 (***p < 0.001 and **p < 0.01).
After legumain knockdown or overexpression for 72 h, EMT markers were examined (Figure 6), such as E-cadherin (an epithelial marker), N-cadherin, slug, and vimentin (mesenchymal markers). After siRNA knockdown, MMP2 was also examined to evaluate the knockdown efficiency. All mesenchymal markers examined were downregulated after legumain knockdown, but epithelial markers were upregulated.
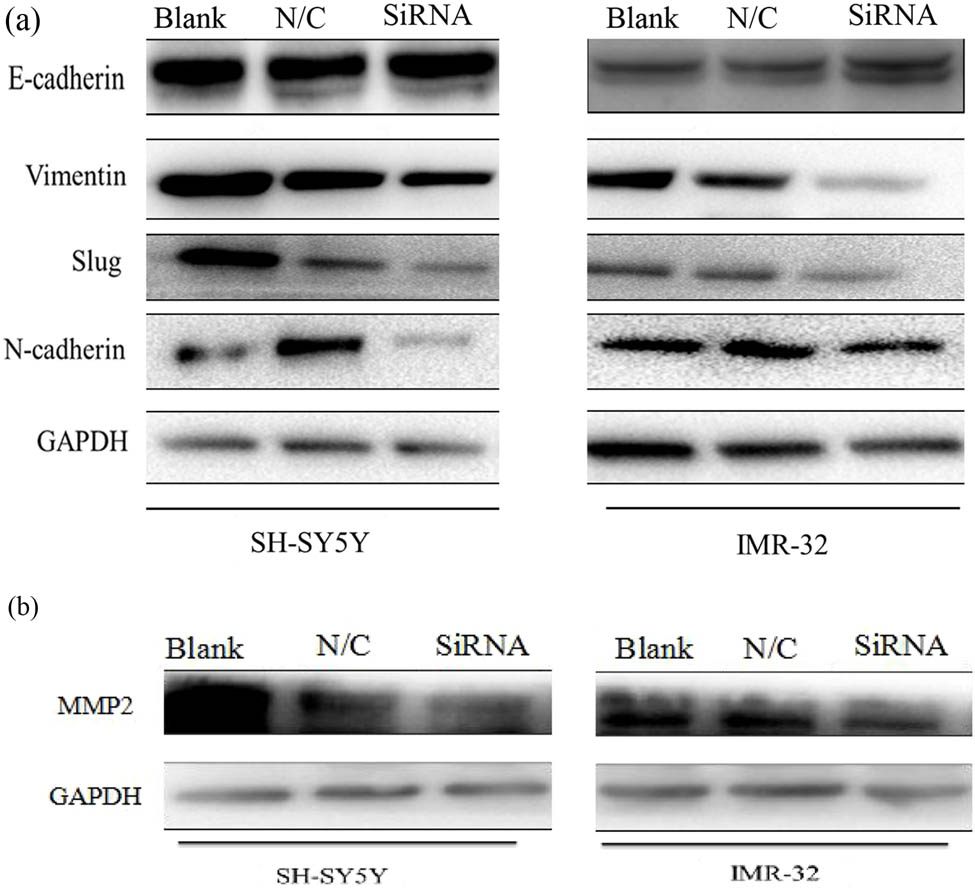
Changes in EMT markers after knockdown of the legumain gene. (a) Mesenchymal markers including vimentin, slug, and N-cadherin decreased in cell lines SH-SY5Y and IMR32 after knockdown of legumain in western blot analysis, but epithelial markers like E-cadherin increased accordingly. (b) MMP2 also decreased after the knockdown of legumain.
Legumain expression was amplified by plasmid transfection into the cell line SK-N-BE2 (Figure 7a), so the effect of legumain on migration and invasion was explored. Transfection of a plasmid carrying the legumain gene increased the migration and invasion of SK-N-BE2 cells (Figure 7b) in a transwell assay. When legumain was overexpressed, mesenchymal markers like N-cadherin, vimentin, and slug were upregulated significantly, while epithelial markers E-cadherin was downregulated slightly (Figure 7c).
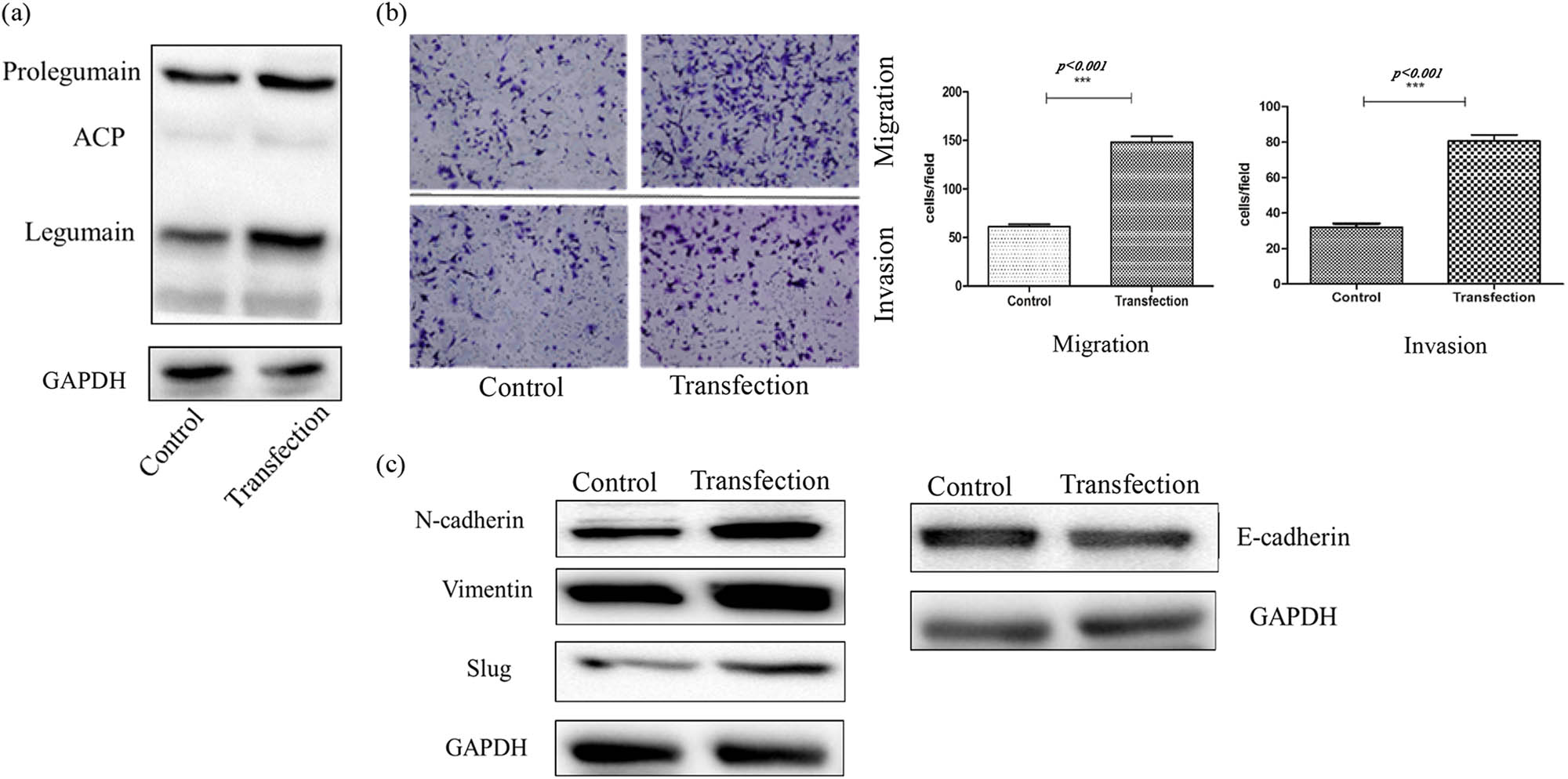
(a) Overexpression of legumain by a plasmid carrying legumain coding gene transfection in cell SK-N-BE2 was proved by western blot. (b) Migration and invasion ability increased in transwell assay after legumain overexpression by plasmid transfection, p < 0.05. (c) Mesenchymal markers above increased after legumain overexpression, but epithelial marker decreased.
3.4 Effect of legumain on EMT relies on its AEP activity and can be inhibited by AEPI
RhLegumain and the AEPI on EMT were introduced to examine AEP activity during EMT of NB cells. RhLegumain was activated into AEP in pH 4.1 activation buffer, and the effect of AEP and AEPI was confirmed by hydrolyzing the substrate Z-Ala-Ala-Asn-AMC. 24 h after activation of AEP or applying AEPI, migration and invasion abilities changed in SH-SY5Y cells (Figure 8a), with an enhancement of the migration and invasion ability by AEP, but inhibition of these by AEPI. Meanwhile, EMT markers examined (Figure 8b) with the above mesenchymal markers were upregulated by AEP and downregulated by AEPI. The epithelial marker E-cadherin was downregulated after treatment with AEP but upregulated after treatment with AEPI.
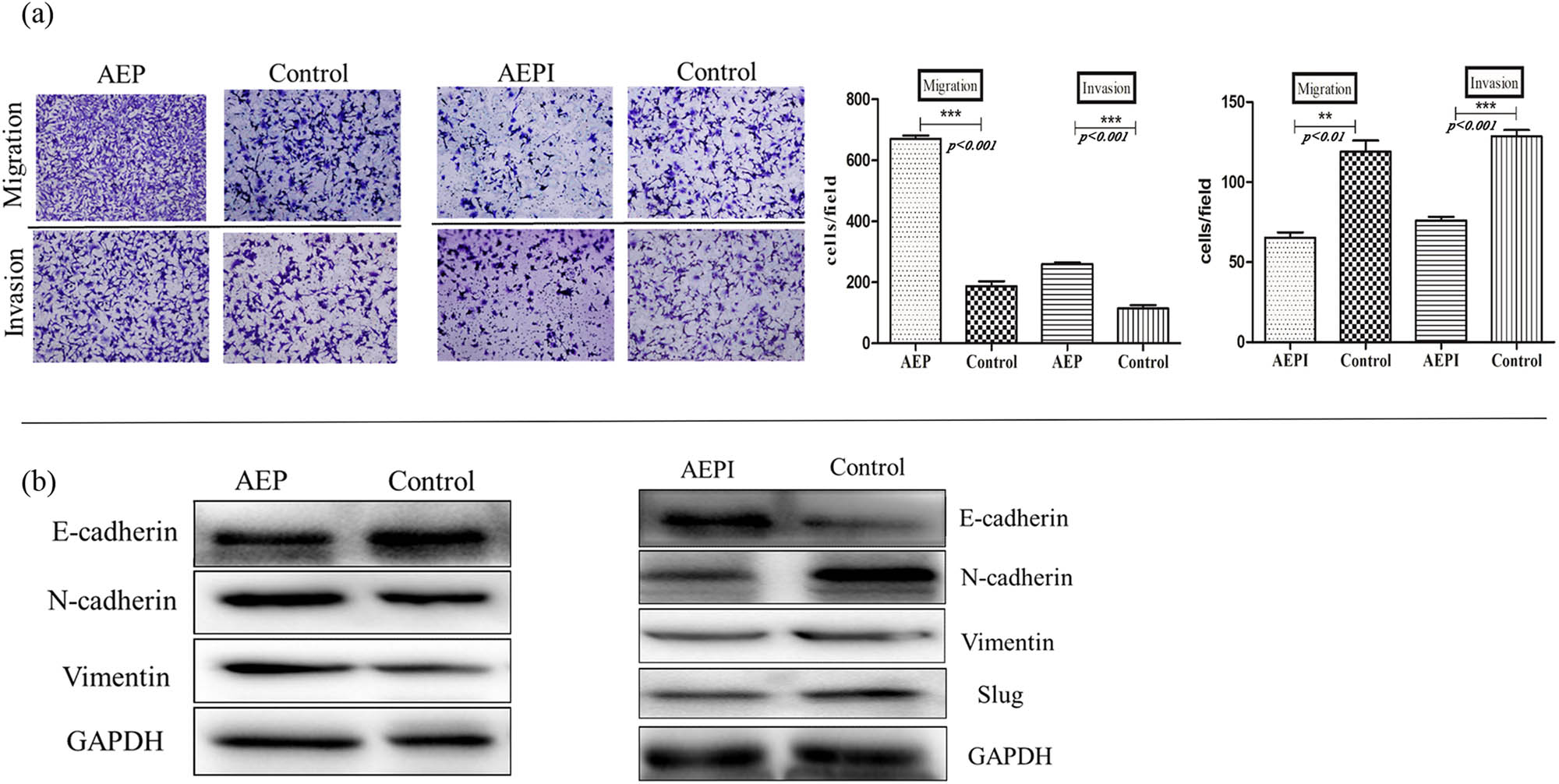
Changes in EMT markers after AEP or AEPI treatment. (a) Cell counts passing through the transwell chambers in transwell assay with or without Matrigel increased after AEP treatment in SH-SY5Y, but AEPI worked contrarily (10×), p < 0.05. (b) Mesenchymal markers increased after AEP treatment in SH-SY5Y in western blot assay, but epithelial markers decreased; mature active legumain (AEP) worked contrarily in the changes in EMT markers.
4 Discussion
The cysteine protease legumain (prolegumain) can develop into an AEP and an ACP in some aberrant situations [25,26]. Consistent with other reports on legumain in tumors [27], legumain was also found to be expressed in NB, where it is mainly distributed in the cytoplasm and extracellular matrix. There was a disparity in relative amounts of the 56 kDa prolegumain and 36 kDa active mature legumain in the three cell lines SK-N-BE2, SH-SY5Y, and IMR32, representing the inactive zymogen and mature active form, respectively. Expression of AEP is increased under the conditions of starvation, acidic pH, hypoxia, and other stress situations, so we used a relatively neutral culture medium and the same culture conditions (37°C, 5% CO2) to avoid factors like starvation, acid pH, and hypoxia so that different levels of mature active legumain could be correlated with different levels of legumain inhibitors-cystatins. Cystatin C and cystatin E/M are the most potent legumain inhibitors. Cystatins may regulate the activity of AEP through substrate competition for the active site [28]. The use of cystatin C to inhibit EMT and morphological transformation stimulated by transforming growth factor-beta was reported [29].
Wallin et al. [30] reported cystatin C in NBs. More cystatin C is present in the NB cell line SK-N-BE2 than in SH-SY5Y, consistent with our finding that less active mature legumain was present in SK-N-BE2 than in SH-SY5Y. Therefore, a disparity in cystatin C may explain the difference in levels of mature 36 kDa legumain between these cell lines. The IMR32 cell line is the most malignant type-I form of NB [31], and the abundant active legumain in IMR32 may also support legumain as a marker of tumor malignancy.
EMT has been shown to be important in regulating tumor metastasis and played a central role in tumor development [32,33]. A recent report also found that mesenchymal markers were correlated with a poor prognosis of NB, and mesenchymal markers should be regarded as potential markers for NB prognostication [34]. Slug is an important factor in NB because it is involved in gastrulation, development, and migration of neuronal precursors during embryonic development, and it also plays a role in tumor metastasis. Because NB is an embryonic neuroblastic tumor, slug may be involved in its development.
Vimentin is a typical marker of EMT [35]. Overexpression of vimentin correlates with accelerated tumor growth, invasion, and a poor prognosis in some cancers [36,37]. Vimentin’s overexpression during metastasis [38] suggests its central role as a metastasis promoter. In the regulation of EMT by legumain, either through gene overexpression of legumain or by extrinsic active mature legumain treatment, slug and vimentin were both upregulated and when legumain was inhibited by AEPI, slug and vimentin were downregulated, suggesting a major role of legumain in EMT of NB.
Lammens et al. [39] reported that all NB samples express N-cadherin and might be a valid target for treatment. Our experiments showed the expression of active mature legumain (AEP) and N-cadherin were positively correlated well in NB. Gene knockdown or plasmid transfection of legumain was carried out to verify the effect of legumain on EMT and N-cadherin expression, besides AEP and AEPI, were also introduced to elucidate the AEP activity of legumain changes in N-cadherin. Both gene knockdown and AEPI were able to downregulate N-cadherin expression in our in vitro experiments, and there was an upregulation of N-cadherin after legumain plasmid transfection and AEP treatment.
Legumain’s functions in digestion, antigen processing, and signaling via processing/activation are mainly attributed to its AEP activity [40]. Previous reports in adult solid tumors indicated a positive correlation between legumain and tumor malignancies, and our research proved legumain could regulate EMT through its AEP activity. The multi-branched and context-dependent activation process of legumain illustrates that proteases can act as signal transducers and decision-makers. Still, there is much we do not know. It is unclear how it triggers EMT as a transcriptional factor or just an interaction regulator between matrix proteins in the tumor stroma. Additional study of legumain in the future is essential.
Acknowledgments
Authors would like to thank Yongtao Xiao, Ph.D. from Shanghai Institute for Pediatric Research, and Mitchell Arico, Ph.D., from LiwenBianji, Edanz Group China, for editing the English text of a draft of this manuscript.
-
Funding information: The research was funded by Shanghai Pudong New Area Science and Technology Development Fund innovation Project [PKJ2020-Y105], Shanghai University of Medicine & Health Sciences Affiliated Zhoupu Hospital Research Project [ZPXM-2019G-02].
-
Author contributions: Min Zhang conducted a detailed experiment and drafted the manuscript. Jianhua Zhu revised the manuscript, analyzed data of the experiment and gave the preliminary framework of the paper. Wei Wang assisted to complete the experiment, especially the immunohistochemical detection. Zhiteng Jiang designed the study and analyzed the data.
-
Conflict of interest: Authors state no conflict of interest.
-
Data availability statement: The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
[1] Kumar MD, Dravid A, Kumar A. Gene therapy as a potential tool for treating neuroblastoma-a focused review. Cancer Gene Ther. 2016;23:115–24.10.1038/cgt.2016.16Suche in Google Scholar PubMed
[2] Newman EA, Nuchtern JG. Recent biologic and genetic advances in neuroblastoma: Implications for diagnostic, risk stratification, and treatment strategies. Semin Pediatr Surg. 2016;25:257–64.10.1053/j.sempedsurg.2016.09.007Suche in Google Scholar PubMed
[3] Rozen EJ, Shohet JM. Systematic review of the receptor tyrosine kinase superfamily in neuroblastoma pathophysiology. Cancer Metastasis Rev. 2022;41:33–52. 10.3390/cells10051153.Suche in Google Scholar PubMed PubMed Central
[4] Portella L, Bello AM, Scala S. CXCL12 signaling in the tumor microenvironment. Adv Exp Med Biol. 2021;1302:51–70.10.1007/978-3-030-62658-7_5Suche in Google Scholar PubMed
[5] Shi L, Wang M, Li H, You P. MicroRNAs in body fluids: a more promising biomarker for clear cell renal cell carcinoma. Cancer Manag Res. 2021;13:7663–75.10.2147/CMAR.S330881Suche in Google Scholar PubMed PubMed Central
[6] Yi Y, Wu M, Zeng H, Hu W, Zhao C, Xiong M, et al. Tumor-derived exosomal non-coding RNAs: the emerging mechanisms and potential clinical applications in breast cancer. Front Oncol. 2021;11:738945.10.3389/fonc.2021.738945Suche in Google Scholar PubMed PubMed Central
[7] Dhani S, Zhao Y, Zhivotovsky B. A long way to go: caspase inhibitors in clinical use. Cell Death Dis. 2021;12:949.10.1038/s41419-021-04240-3Suche in Google Scholar PubMed PubMed Central
[8] Reddy BD, Beeraka NM, Chitturi CMK, Madhunapantula SV. An overview of targeting legumain for inhibiting cancers. Curr Pharm Des. 2021;27:3337–48.10.2174/1381612826666201125111625Suche in Google Scholar PubMed
[9] Wang H, Chen B, Lin Y, Zhou Y, Li X. Legumain promotes gastric cancer progression through tumor-associated macrophages in vitro and in vivo. Int J Biol Sci. 2020;16:172–180.10.7150/ijbs.36467Suche in Google Scholar PubMed PubMed Central
[10] Haugen MH, Boye K, Nesland JM, Pettersen SJ, Egeland EV, Tamhane T, et al. High expression of the cysteine proteinase legumain in colorectal cancer – implications for therapeutic targeting. Eur J Cancer. 2015;51:9–17.10.1016/j.ejca.2014.10.020Suche in Google Scholar PubMed
[11] Meng F, Liu W. Knockdown of legumain suppresses cervical cancer cell migration and invasion. Oncol Res. 2016;23:7–12.10.3727/096504015X14410238486649Suche in Google Scholar PubMed PubMed Central
[12] Li X, Tang M, Zhu Q, Wang X, Lin Y, Wang X. The exosomal integrin α5β1/AEP complex derived from epithelial ovarian cancer cells promotes peritoneal metastasis through regulating mesothelial cell proliferation and migration. Cell Oncol (Dordr). 2020;43:263–77.10.1007/s13402-019-00486-4Suche in Google Scholar PubMed
[13] Toss MS, Miligy IM, Gorringe KL, McCaffrey L, AlKawaz A, Abidi A, et al. Legumain is an independent predictor for invasive recurrence in breast ductal carcinoma in situ. Mod Pathol. 2019;32:639–49.10.1038/s41379-018-0180-xSuche in Google Scholar PubMed
[14] Shen L, Li H, Shi Y, Wang D, Gong J, Xun J, et al. M2 tumour-associated macrophages contribute to tumour progression via legumain remodelling the extracellular matrix in diffuse large B cell lymphoma. Sci Rep. 2016;28:30347.10.1038/srep30347Suche in Google Scholar PubMed PubMed Central
[15] Wang D, Xiong M, Chen C, Du L, Liu Z, Shi Y, et al. Legumain, an asparaginyl endopeptidase, mediates the effect of M2 macrophages on attenuating renal interstitial fibrosis in obstructive nephropathy. Kidney Int. 2018;94:91–101.10.1016/j.kint.2017.12.025Suche in Google Scholar PubMed
[16] Chen JM, Fortunato M, Stevens RA, Barrett AJ. Activation of progelatinase A by mammalian legumain, a recently discovered cysteine proteinase. Biol Chem. 2001;382:777–83.10.1515/bchm.2001.382.5.777Suche in Google Scholar
[17] Freeley S, Cardone J, Günther SC, West EE, Reinheckel T, Watts C, et al. Asparaginyl endopeptidase (Legumain) supports human Th1 induction via cathepsin L-mediated intracellular C3 activation. Front Immunol. 2018;9:2449.10.3389/fimmu.2018.02449Suche in Google Scholar PubMed PubMed Central
[18] Lunde NN, Bosnjak T, Solberg R, Johansen HT. Mammalian legumain – a lysosomal cysteine protease with extracellular functions? Biochimie. 2019;166:77-83-23.10.1016/j.biochi.2019.06.002Suche in Google Scholar PubMed
[19] Haugen MH, Johansen HT, Pettersen SJ, Solberg R, Brix K, Flatmark K, et al. Nuclear legumain activity in colorectal cancer. PLoS One. 2013;8(1):e52980.10.1371/journal.pone.0052980Suche in Google Scholar PubMed PubMed Central
[20] Pastushenko I, Blanpain C. EMT transition states during tumor progression and metastasis. Trends Cell Biol. 2019;29:212–26.10.1016/j.tcb.2018.12.001Suche in Google Scholar PubMed
[21] Cho ES, Kang HE, Kim NH, Yook JI. Therapeutic implications of cancer epithelial-mesenchymal transition (EMT). Arch Pharm Res. 2019;42:14–24.10.1007/s12272-018-01108-7Suche in Google Scholar PubMed
[22] Pastushenko I, Brisebarre A, Sifrim A, Fioramonti M, Revenco T, Boumahdi S, et al. Identification of the tumour transition states occurring during EMT. Nature. 2018;556:463–68.10.1038/s41586-018-0040-3Suche in Google Scholar PubMed
[23] Rasheed ZA, Yang J, Wang Q, Kowalski J, Freed I, Murter C, et al. Prognostic significance of tumorigenic cells with mesenchymal features in pancreatic adenocarcinoma. J Natl Cancer Inst. 2010;102:340–51.10.1093/jnci/djp535Suche in Google Scholar PubMed PubMed Central
[24] Liu Y, Bajjuri KM, Liu C, Sinha SC. Targeting cell surface alpha(v) beta(3) integrin increases therapeutic efficacies of a legumain protease-activated Auristatin Prodrug. Mol Pharm. 2012;9:168–75.10.1021/mp200434nSuche in Google Scholar PubMed PubMed Central
[25] Zhang W, Lin Y. The mechanism of asparagine endopeptidase in the progression of malignant tumors: a review. Cells. 2021;10:1153.10.3390/cells10051153Suche in Google Scholar PubMed PubMed Central
[26] Dall E, Brandstetter H. Mechanistic and structural studies on legumain explain its zymogenicity, distinct activation pathways, and regulation. Proc Natl Acad Sci. 2013;110:10940–5.10.1073/pnas.1300686110Suche in Google Scholar PubMed PubMed Central
[27] Zhao T, Liu Y, Hao Y, Zhang W, Tao L, Wang D, et al. Esomeprazole inhibits the lysosomal cysteine protease legumain to prevent cancer metastasis. Invest New Drugs. 2021;39:337–47.10.1007/s10637-020-01011-3Suche in Google Scholar PubMed
[28] Wallin H, Apelqvist J, Andersson F, Ekström U, Abrahamson M. Low-level internalization of cystatin E/M affects legumain activity and migration of melanoma cells. J Biol Chem. 2017;292:14413–24.10.1074/jbc.M117.776138Suche in Google Scholar PubMed PubMed Central
[29] Sokol JP, Neil JR, Schiemann BJ, Schiemann WP. The use of cystatin C to inhibit epithelial-mesenchymal transition and morphological transformation stimulated by transforming growth factor-beta. Breast Cancer Res. 2005;7:844–53.10.1186/bcr1312Suche in Google Scholar PubMed PubMed Central
[30] Wallin H, Bjarnadottir M, Vogel LK, Wassélius J, Ekström U, Abrahamson M. Cystatins extra-and intracellular cysteine protease inhibitors: high-level secretion and uptake of cystatin C in human neuroblastoma cells. Biochimie. 2010;92:1625–34.10.1016/j.biochi.2010.08.011Suche in Google Scholar PubMed
[31] Ross RA, Biedler JL, Spengler BA. A role for distinct cell types in determining malignancy in human neuroblastoma cell lines and tumors. Cancer Lett. 2003;197:35–9.10.1016/S0304-3835(03)00079-XSuche in Google Scholar
[32] Dongre A, Weinberg RA. New insights into the mechanisms of epithelial-mesenchymal transition and implications for cancer. Nat Rev Mol Cell Biol. 2019;20:69–84.10.1038/s41580-018-0080-4Suche in Google Scholar PubMed
[33] Bischoff J. Endothelial-to-mesenchymal transition. Circ Res. 2019;124:1163–5.10.1161/CIRCRESAHA.119.314813Suche in Google Scholar PubMed PubMed Central
[34] Nozato M, Kaneko S, Nakagawara A, Komuro H. Epithelial-mesenchymal transition-related gene expression as a new prognostic marker for neuroblastoma. Int J Oncol. 2013;42:134–40.10.3892/ijo.2012.1684Suche in Google Scholar PubMed PubMed Central
[35] Paolillo M, Schinelli S. Extracellular matrix alterations in metastatic processes. Int J Mol Sci. 2019;20:4947.10.3390/ijms20194947Suche in Google Scholar PubMed PubMed Central
[36] Navas T, Kinders RJ, Lawrence SM, Ferry-Galow KV, Borgel S, Hollingshead MG, et al. Clinical evolution of epithelial-mesenchymal transition in human carcinomas. Cancer Res. 2020;80:304–18.10.1158/0008-5472.CAN-18-3539Suche in Google Scholar PubMed PubMed Central
[37] Wu S, Du Y, Beckford J, Alachkar H. Upregulation of the EMT marker vimentin is associated with poor clinical outcome in acute myeloid leukemia. J Transl Med. 2018;16:170.10.1186/s12967-018-1539-ySuche in Google Scholar PubMed PubMed Central
[38] Wang W, Chen H, Gao W, Wang S, Wu K, Lu C, et al. Girdin interaction with vimentin induces EMT and promotes the growth and metastasis of pancreatic ductal adenocarcinoma. Oncol Rep. 2020;44:637–49.10.3892/or.2020.7615Suche in Google Scholar PubMed PubMed Central
[39] Lammens T, Swerts K, Derycke L, de Craemer A, de Brouwer S, de Preter K, et al. N-Cadherin in neuroblastoma disease: expression and clinical significance. PLoS One. 2012;7:1–8.10.1371/journal.pone.0031206Suche in Google Scholar PubMed PubMed Central
[40] Dall E, Brandstetter H. Structure and function of legumain in health and disease. Biochimie. 2016;122:126–50.10.1016/j.biochi.2015.09.022Suche in Google Scholar PubMed
© 2022 Min Zhang et al., published by De Gruyter
This work is licensed under the Creative Commons Attribution 4.0 International License.
Artikel in diesem Heft
- Biomedical Sciences
- Effects of direct oral anticoagulants dabigatran and rivaroxaban on the blood coagulation function in rabbits
- The mother of all battles: Viruses vs humans. Can humans avoid extinction in 50–100 years?
- Knockdown of G1P3 inhibits cell proliferation and enhances the cytotoxicity of dexamethasone in acute lymphoblastic leukemia
- LINC00665 regulates hepatocellular carcinoma by modulating mRNA via the m6A enzyme
- Association study of CLDN14 variations in patients with kidney stones
- Concanavalin A-induced autoimmune hepatitis model in mice: Mechanisms and future outlook
- Regulation of miR-30b in cancer development, apoptosis, and drug resistance
- Informatic analysis of the pulmonary microecology in non-cystic fibrosis bronchiectasis at three different stages
- Swimming attenuates tumor growth in CT-26 tumor-bearing mice and suppresses angiogenesis by mediating the HIF-1α/VEGFA pathway
- Characterization of intestinal microbiota and serum metabolites in patients with mild hepatic encephalopathy
- Functional conservation and divergence in plant-specific GRF gene family revealed by sequences and expression analysis
- Application of the FLP/LoxP-FRT recombination system to switch the eGFP expression in a model prokaryote
- Biomedical evaluation of antioxidant properties of lamb meat enriched with iodine and selenium
- Intravenous infusion of the exosomes derived from human umbilical cord mesenchymal stem cells enhance neurological recovery after traumatic brain injury via suppressing the NF-κB pathway
- Effect of dietary pattern on pregnant women with gestational diabetes mellitus and its clinical significance
- Potential regulatory mechanism of TNF-α/TNFR1/ANXA1 in glioma cells and its role in glioma cell proliferation
- Effect of the genetic mutant G71R in uridine diphosphate-glucuronosyltransferase 1A1 on the conjugation of bilirubin
- Quercetin inhibits cytotoxicity of PC12 cells induced by amyloid-beta 25–35 via stimulating estrogen receptor α, activating ERK1/2, and inhibiting apoptosis
- Nutrition intervention in the management of novel coronavirus pneumonia patients
- circ-CFH promotes the development of HCC by regulating cell proliferation, apoptosis, migration, invasion, and glycolysis through the miR-377-3p/RNF38 axis
- Bmi-1 directly upregulates glucose transporter 1 in human gastric adenocarcinoma
- Lacunar infarction aggravates the cognitive deficit in the elderly with white matter lesion
- Hydroxysafflor yellow A improved retinopathy via Nrf2/HO-1 pathway in rats
- Comparison of axon extension: PTFE versus PLA formed by a 3D printer
- Elevated IL-35 level and iTr35 subset increase the bacterial burden and lung lesions in Mycobacterium tuberculosis-infected mice
- A case report of CAT gene and HNF1β gene variations in a patient with early-onset diabetes
- Study on the mechanism of inhibiting patulin production by fengycin
- SOX4 promotes high-glucose-induced inflammation and angiogenesis of retinal endothelial cells by activating NF-κB signaling pathway
- Relationship between blood clots and COVID-19 vaccines: A literature review
- Analysis of genetic characteristics of 436 children with dysplasia and detailed analysis of rare karyotype
- Bioinformatics network analyses of growth differentiation factor 11
- NR4A1 inhibits the epithelial–mesenchymal transition of hepatic stellate cells: Involvement of TGF-β–Smad2/3/4–ZEB signaling
- Expression of Zeb1 in the differentiation of mouse embryonic stem cell
- Study on the genetic damage caused by cadmium sulfide quantum dots in human lymphocytes
- Association between single-nucleotide polymorphisms of NKX2.5 and congenital heart disease in Chinese population: A meta-analysis
- Assessment of the anesthetic effect of modified pentothal sodium solution on Sprague-Dawley rats
- Genetic susceptibility to high myopia in Han Chinese population
- Potential biomarkers and molecular mechanisms in preeclampsia progression
- Silencing circular RNA-friend leukemia virus integration 1 restrained malignancy of CC cells and oxaliplatin resistance by disturbing dyskeratosis congenita 1
- Endostar plus pembrolizumab combined with a platinum-based dual chemotherapy regime for advanced pulmonary large-cell neuroendocrine carcinoma as a first-line treatment: A case report
- The significance of PAK4 in signaling and clinicopathology: A review
- Sorafenib inhibits ovarian cancer cell proliferation and mobility and induces radiosensitivity by targeting the tumor cell epithelial–mesenchymal transition
- Characterization of rabbit polyclonal antibody against camel recombinant nanobodies
- Active legumain promotes invasion and migration of neuroblastoma by regulating epithelial-mesenchymal transition
- Effect of cell receptors in the pathogenesis of osteoarthritis: Current insights
- MT-12 inhibits the proliferation of bladder cells in vitro and in vivo by enhancing autophagy through mitochondrial dysfunction
- Study of hsa_circRNA_000121 and hsa_circRNA_004183 in papillary thyroid microcarcinoma
- BuyangHuanwu Decoction attenuates cerebral vasospasm caused by subarachnoid hemorrhage in rats via PI3K/AKT/eNOS axis
- Effects of the interaction of Notch and TLR4 pathways on inflammation and heart function in septic heart
- Monosodium iodoacetate-induced subchondral bone microstructure and inflammatory changes in an animal model of osteoarthritis
- A rare presentation of type II Abernethy malformation and nephrotic syndrome: Case report and review
- Rapid death due to pulmonary epithelioid haemangioendothelioma in several weeks: A case report
- Hepatoprotective role of peroxisome proliferator-activated receptor-α in non-cancerous hepatic tissues following transcatheter arterial embolization
- Correlation between peripheral blood lymphocyte subpopulations and primary systemic lupus erythematosus
- A novel SLC8A1-ALK fusion in lung adenocarcinoma confers sensitivity to alectinib: A case report
- β-Hydroxybutyrate upregulates FGF21 expression through inhibition of histone deacetylases in hepatocytes
- Identification of metabolic genes for the prediction of prognosis and tumor microenvironment infiltration in early-stage non-small cell lung cancer
- BTBD10 inhibits glioma tumorigenesis by downregulating cyclin D1 and p-Akt
- Mucormycosis co-infection in COVID-19 patients: An update
- Metagenomic next-generation sequencing in diagnosing Pneumocystis jirovecii pneumonia: A case report
- Long non-coding RNA HOXB-AS1 is a prognostic marker and promotes hepatocellular carcinoma cells’ proliferation and invasion
- Preparation and evaluation of LA-PEG-SPION, a targeted MRI contrast agent for liver cancer
- Proteomic analysis of the liver regulating lipid metabolism in Chaohu ducks using two-dimensional electrophoresis
- Nasopharyngeal tuberculosis: A case report
- Characterization and evaluation of anti-Salmonella enteritidis activity of indigenous probiotic lactobacilli in mice
- Aberrant pulmonary immune response of obese mice to periodontal infection
- Bacteriospermia – A formidable player in male subfertility
- In silico and in vivo analysis of TIPE1 expression in diffuse large B cell lymphoma
- Effects of KCa channels on biological behavior of trophoblasts
- Interleukin-17A influences the vulnerability rather than the size of established atherosclerotic plaques in apolipoprotein E-deficient mice
- Multiple organ failure and death caused by Staphylococcus aureus hip infection: A case report
- Prognostic signature related to the immune environment of oral squamous cell carcinoma
- Primary and metastatic squamous cell carcinoma of the thyroid gland: Two case reports
- Neuroprotective effects of crocin and crocin-loaded niosomes against the paraquat-induced oxidative brain damage in rats
- Role of MMP-2 and CD147 in kidney fibrosis
- Geometric basis of action potential of skeletal muscle cells and neurons
- Babesia microti-induced fulminant sepsis in an immunocompromised host: A case report and the case-specific literature review
- Role of cerebellar cortex in associative learning and memory in guinea pigs
- Application of metagenomic next-generation sequencing technique for diagnosing a specific case of necrotizing meningoencephalitis caused by human herpesvirus 2
- Case report: Quadruple primary malignant neoplasms including esophageal, ureteral, and lung in an elderly male
- Long non-coding RNA NEAT1 promotes angiogenesis in hepatoma carcinoma via the miR-125a-5p/VEGF pathway
- Osteogenic differentiation of periodontal membrane stem cells in inflammatory environments
- Knockdown of SHMT2 enhances the sensitivity of gastric cancer cells to radiotherapy through the Wnt/β-catenin pathway
- Continuous renal replacement therapy combined with double filtration plasmapheresis in the treatment of severe lupus complicated by serious bacterial infections in children: A case report
- Simultaneous triple primary malignancies, including bladder cancer, lymphoma, and lung cancer, in an elderly male: A case report
- Preclinical immunogenicity assessment of a cell-based inactivated whole-virion H5N1 influenza vaccine
- One case of iodine-125 therapy – A new minimally invasive treatment of intrahepatic cholangiocarcinoma
- S1P promotes corneal trigeminal neuron differentiation and corneal nerve repair via upregulating nerve growth factor expression in a mouse model
- Early cancer detection by a targeted methylation assay of circulating tumor DNA in plasma
- Calcifying nanoparticles initiate the calcification process of mesenchymal stem cells in vitro through the activation of the TGF-β1/Smad signaling pathway and promote the decay of echinococcosis
- Evaluation of prognostic markers in patients infected with SARS-CoV-2
- N6-Methyladenosine-related alternative splicing events play a role in bladder cancer
- Characterization of the structural, oxidative, and immunological features of testis tissue from Zucker diabetic fatty rats
- Effects of glucose and osmotic pressure on the proliferation and cell cycle of human chorionic trophoblast cells
- Investigation of genotype diversity of 7,804 norovirus sequences in humans and animals of China
- Characteristics and karyotype analysis of a patient with turner syndrome complicated with multiple-site tumors: A case report
- Aggravated renal fibrosis is positively associated with the activation of HMGB1-TLR2/4 signaling in STZ-induced diabetic mice
- Distribution characteristics of SARS-CoV-2 IgM/IgG in false-positive results detected by chemiluminescent immunoassay
- SRPX2 attenuated oxygen–glucose deprivation and reperfusion-induced injury in cardiomyocytes via alleviating endoplasmic reticulum stress-induced apoptosis through targeting PI3K/Akt/mTOR axis
- Aquaporin-8 overexpression is involved in vascular structure and function changes in placentas of gestational diabetes mellitus patients
- Relationship between CRP gene polymorphisms and ischemic stroke risk: A systematic review and meta-analysis
- Effects of growth hormone on lipid metabolism and sexual development in pubertal obese male rats
- Cloning and identification of the CTLA-4IgV gene and functional application of vaccine in Xinjiang sheep
- Antitumor activity of RUNX3: Upregulation of E-cadherin and downregulation of the epithelial–mesenchymal transition in clear-cell renal cell carcinoma
- PHF8 promotes osteogenic differentiation of BMSCs in old rat with osteoporosis by regulating Wnt/β-catenin pathway
- A review of the current state of the computer-aided diagnosis (CAD) systems for breast cancer diagnosis
- Bilateral dacryoadenitis in adult-onset Still’s disease: A case report
- A novel association between Bmi-1 protein expression and the SUVmax obtained by 18F-FDG PET/CT in patients with gastric adenocarcinoma
- The role of erythrocytes and erythroid progenitor cells in tumors
- Relationship between platelet activation markers and spontaneous abortion: A meta-analysis
- Abnormal methylation caused by folic acid deficiency in neural tube defects
- Silencing TLR4 using an ultrasound-targeted microbubble destruction-based shRNA system reduces ischemia-induced seizures in hyperglycemic rats
- Plant Sciences
- Seasonal succession of bacterial communities in cultured Caulerpa lentillifera detected by high-throughput sequencing
- Cloning and prokaryotic expression of WRKY48 from Caragana intermedia
- Novel Brassica hybrids with different resistance to Leptosphaeria maculans reveal unbalanced rDNA signal patterns
- Application of exogenous auxin and gibberellin regulates the bolting of lettuce (Lactuca sativa L.)
- Phytoremediation of pollutants from wastewater: A concise review
- Genome-wide identification and characterization of NBS-encoding genes in the sweet potato wild ancestor Ipomoea trifida (H.B.K.)
- Alleviative effects of magnetic Fe3O4 nanoparticles on the physiological toxicity of 3-nitrophenol to rice (Oryza sativa L.) seedlings
- Selection and functional identification of Dof genes expressed in response to nitrogen in Populus simonii × Populus nigra
- Study on pecan seed germination influenced by seed endocarp
- Identification of active compounds in Ophiopogonis Radix from different geographical origins by UPLC-Q/TOF-MS combined with GC-MS approaches
- The entire chloroplast genome sequence of Asparagus cochinchinensis and genetic comparison to Asparagus species
- Genome-wide identification of MAPK family genes and their response to abiotic stresses in tea plant (Camellia sinensis)
- Selection and validation of reference genes for RT-qPCR analysis of different organs at various development stages in Caragana intermedia
- Cloning and expression analysis of SERK1 gene in Diospyros lotus
- Integrated metabolomic and transcriptomic profiling revealed coping mechanisms of the edible and medicinal homologous plant Plantago asiatica L. cadmium resistance
- A missense variant in NCF1 is associated with susceptibility to unexplained recurrent spontaneous abortion
- Assessment of drought tolerance indices in faba bean genotypes under different irrigation regimes
- The entire chloroplast genome sequence of Asparagus setaceus (Kunth) Jessop: Genome structure, gene composition, and phylogenetic analysis in Asparagaceae
- Food Science
- Dietary food additive monosodium glutamate with or without high-lipid diet induces spleen anomaly: A mechanistic approach on rat model
- Binge eating disorder during COVID-19
- Potential of honey against the onset of autoimmune diabetes and its associated nephropathy, pancreatitis, and retinopathy in type 1 diabetic animal model
- FTO gene expression in diet-induced obesity is downregulated by Solanum fruit supplementation
- Physical activity enhances fecal lactobacilli in rats chronically drinking sweetened cola beverage
- Supercritical CO2 extraction, chemical composition, and antioxidant effects of Coreopsis tinctoria Nutt. oleoresin
- Functional constituents of plant-based foods boost immunity against acute and chronic disorders
- Effect of selenium and methods of protein extraction on the proteomic profile of Saccharomyces yeast
- Microbial diversity of milk ghee in southern Gansu and its effect on the formation of ghee flavor compounds
- Ecology and Environmental Sciences
- Effects of heavy metals on bacterial community surrounding Bijiashan mining area located in northwest China
- Microorganism community composition analysis coupling with 15N tracer experiments reveals the nitrification rate and N2O emissions in low pH soils in Southern China
- Genetic diversity and population structure of Cinnamomum balansae Lecomte inferred by microsatellites
- Preliminary screening of microplastic contamination in different marine fish species of Taif market, Saudi Arabia
- Plant volatile organic compounds attractive to Lygus pratensis
- Effects of organic materials on soil bacterial community structure in long-term continuous cropping of tomato in greenhouse
- Effects of soil treated fungicide fluopimomide on tomato (Solanum lycopersicum L.) disease control and plant growth
- Prevalence of Yersinia pestis among rodents captured in a semi-arid tropical ecosystem of south-western Zimbabwe
- Effects of irrigation and nitrogen fertilization on mitigating salt-induced Na+ toxicity and sustaining sea rice growth
- Bioengineering and Biotechnology
- Poly-l-lysine-caused cell adhesion induces pyroptosis in THP-1 monocytes
- Development of alkaline phosphatase-scFv and its use for one-step enzyme-linked immunosorbent assay for His-tagged protein detection
- Development and validation of a predictive model for immune-related genes in patients with tongue squamous cell carcinoma
- Agriculture
- Effects of chemical-based fertilizer replacement with biochar-based fertilizer on albic soil nutrient content and maize yield
- Genome-wide identification and expression analysis of CPP-like gene family in Triticum aestivum L. under different hormone and stress conditions
- Agronomic and economic performance of mung bean (Vigna radiata L.) varieties in response to rates of blended NPS fertilizer in Kindo Koysha district, Southern Ethiopia
- Influence of furrow irrigation regime on the yield and water consumption indicators of winter wheat based on a multi-level fuzzy comprehensive evaluation
- Discovery of exercise-related genes and pathway analysis based on comparative genomes of Mongolian originated Abaga and Wushen horse
- Lessons from integrated seasonal forecast-crop modelling in Africa: A systematic review
- Evolution trend of soil fertility in tobacco-planting area of Chenzhou, Hunan Province, China
- Animal Sciences
- Morphological and molecular characterization of Tatera indica Hardwicke 1807 (Rodentia: Muridae) from Pothwar, Pakistan
- Research on meat quality of Qianhua Mutton Merino sheep and Small-tail Han sheep
- SI: A Scientific Memoir
- Suggestions on leading an academic research laboratory group
- My scientific genealogy and the Toronto ACDC Laboratory, 1988–2022
- Erratum
- Erratum to “Changes of immune cells in patients with hepatocellular carcinoma treated by radiofrequency ablation and hepatectomy, a pilot study”
- Erratum to “A two-microRNA signature predicts the progression of male thyroid cancer”
- Retraction
- Retraction of “Lidocaine has antitumor effect on hepatocellular carcinoma via the circ_DYNC1H1/miR-520a-3p/USP14 axis”
Artikel in diesem Heft
- Biomedical Sciences
- Effects of direct oral anticoagulants dabigatran and rivaroxaban on the blood coagulation function in rabbits
- The mother of all battles: Viruses vs humans. Can humans avoid extinction in 50–100 years?
- Knockdown of G1P3 inhibits cell proliferation and enhances the cytotoxicity of dexamethasone in acute lymphoblastic leukemia
- LINC00665 regulates hepatocellular carcinoma by modulating mRNA via the m6A enzyme
- Association study of CLDN14 variations in patients with kidney stones
- Concanavalin A-induced autoimmune hepatitis model in mice: Mechanisms and future outlook
- Regulation of miR-30b in cancer development, apoptosis, and drug resistance
- Informatic analysis of the pulmonary microecology in non-cystic fibrosis bronchiectasis at three different stages
- Swimming attenuates tumor growth in CT-26 tumor-bearing mice and suppresses angiogenesis by mediating the HIF-1α/VEGFA pathway
- Characterization of intestinal microbiota and serum metabolites in patients with mild hepatic encephalopathy
- Functional conservation and divergence in plant-specific GRF gene family revealed by sequences and expression analysis
- Application of the FLP/LoxP-FRT recombination system to switch the eGFP expression in a model prokaryote
- Biomedical evaluation of antioxidant properties of lamb meat enriched with iodine and selenium
- Intravenous infusion of the exosomes derived from human umbilical cord mesenchymal stem cells enhance neurological recovery after traumatic brain injury via suppressing the NF-κB pathway
- Effect of dietary pattern on pregnant women with gestational diabetes mellitus and its clinical significance
- Potential regulatory mechanism of TNF-α/TNFR1/ANXA1 in glioma cells and its role in glioma cell proliferation
- Effect of the genetic mutant G71R in uridine diphosphate-glucuronosyltransferase 1A1 on the conjugation of bilirubin
- Quercetin inhibits cytotoxicity of PC12 cells induced by amyloid-beta 25–35 via stimulating estrogen receptor α, activating ERK1/2, and inhibiting apoptosis
- Nutrition intervention in the management of novel coronavirus pneumonia patients
- circ-CFH promotes the development of HCC by regulating cell proliferation, apoptosis, migration, invasion, and glycolysis through the miR-377-3p/RNF38 axis
- Bmi-1 directly upregulates glucose transporter 1 in human gastric adenocarcinoma
- Lacunar infarction aggravates the cognitive deficit in the elderly with white matter lesion
- Hydroxysafflor yellow A improved retinopathy via Nrf2/HO-1 pathway in rats
- Comparison of axon extension: PTFE versus PLA formed by a 3D printer
- Elevated IL-35 level and iTr35 subset increase the bacterial burden and lung lesions in Mycobacterium tuberculosis-infected mice
- A case report of CAT gene and HNF1β gene variations in a patient with early-onset diabetes
- Study on the mechanism of inhibiting patulin production by fengycin
- SOX4 promotes high-glucose-induced inflammation and angiogenesis of retinal endothelial cells by activating NF-κB signaling pathway
- Relationship between blood clots and COVID-19 vaccines: A literature review
- Analysis of genetic characteristics of 436 children with dysplasia and detailed analysis of rare karyotype
- Bioinformatics network analyses of growth differentiation factor 11
- NR4A1 inhibits the epithelial–mesenchymal transition of hepatic stellate cells: Involvement of TGF-β–Smad2/3/4–ZEB signaling
- Expression of Zeb1 in the differentiation of mouse embryonic stem cell
- Study on the genetic damage caused by cadmium sulfide quantum dots in human lymphocytes
- Association between single-nucleotide polymorphisms of NKX2.5 and congenital heart disease in Chinese population: A meta-analysis
- Assessment of the anesthetic effect of modified pentothal sodium solution on Sprague-Dawley rats
- Genetic susceptibility to high myopia in Han Chinese population
- Potential biomarkers and molecular mechanisms in preeclampsia progression
- Silencing circular RNA-friend leukemia virus integration 1 restrained malignancy of CC cells and oxaliplatin resistance by disturbing dyskeratosis congenita 1
- Endostar plus pembrolizumab combined with a platinum-based dual chemotherapy regime for advanced pulmonary large-cell neuroendocrine carcinoma as a first-line treatment: A case report
- The significance of PAK4 in signaling and clinicopathology: A review
- Sorafenib inhibits ovarian cancer cell proliferation and mobility and induces radiosensitivity by targeting the tumor cell epithelial–mesenchymal transition
- Characterization of rabbit polyclonal antibody against camel recombinant nanobodies
- Active legumain promotes invasion and migration of neuroblastoma by regulating epithelial-mesenchymal transition
- Effect of cell receptors in the pathogenesis of osteoarthritis: Current insights
- MT-12 inhibits the proliferation of bladder cells in vitro and in vivo by enhancing autophagy through mitochondrial dysfunction
- Study of hsa_circRNA_000121 and hsa_circRNA_004183 in papillary thyroid microcarcinoma
- BuyangHuanwu Decoction attenuates cerebral vasospasm caused by subarachnoid hemorrhage in rats via PI3K/AKT/eNOS axis
- Effects of the interaction of Notch and TLR4 pathways on inflammation and heart function in septic heart
- Monosodium iodoacetate-induced subchondral bone microstructure and inflammatory changes in an animal model of osteoarthritis
- A rare presentation of type II Abernethy malformation and nephrotic syndrome: Case report and review
- Rapid death due to pulmonary epithelioid haemangioendothelioma in several weeks: A case report
- Hepatoprotective role of peroxisome proliferator-activated receptor-α in non-cancerous hepatic tissues following transcatheter arterial embolization
- Correlation between peripheral blood lymphocyte subpopulations and primary systemic lupus erythematosus
- A novel SLC8A1-ALK fusion in lung adenocarcinoma confers sensitivity to alectinib: A case report
- β-Hydroxybutyrate upregulates FGF21 expression through inhibition of histone deacetylases in hepatocytes
- Identification of metabolic genes for the prediction of prognosis and tumor microenvironment infiltration in early-stage non-small cell lung cancer
- BTBD10 inhibits glioma tumorigenesis by downregulating cyclin D1 and p-Akt
- Mucormycosis co-infection in COVID-19 patients: An update
- Metagenomic next-generation sequencing in diagnosing Pneumocystis jirovecii pneumonia: A case report
- Long non-coding RNA HOXB-AS1 is a prognostic marker and promotes hepatocellular carcinoma cells’ proliferation and invasion
- Preparation and evaluation of LA-PEG-SPION, a targeted MRI contrast agent for liver cancer
- Proteomic analysis of the liver regulating lipid metabolism in Chaohu ducks using two-dimensional electrophoresis
- Nasopharyngeal tuberculosis: A case report
- Characterization and evaluation of anti-Salmonella enteritidis activity of indigenous probiotic lactobacilli in mice
- Aberrant pulmonary immune response of obese mice to periodontal infection
- Bacteriospermia – A formidable player in male subfertility
- In silico and in vivo analysis of TIPE1 expression in diffuse large B cell lymphoma
- Effects of KCa channels on biological behavior of trophoblasts
- Interleukin-17A influences the vulnerability rather than the size of established atherosclerotic plaques in apolipoprotein E-deficient mice
- Multiple organ failure and death caused by Staphylococcus aureus hip infection: A case report
- Prognostic signature related to the immune environment of oral squamous cell carcinoma
- Primary and metastatic squamous cell carcinoma of the thyroid gland: Two case reports
- Neuroprotective effects of crocin and crocin-loaded niosomes against the paraquat-induced oxidative brain damage in rats
- Role of MMP-2 and CD147 in kidney fibrosis
- Geometric basis of action potential of skeletal muscle cells and neurons
- Babesia microti-induced fulminant sepsis in an immunocompromised host: A case report and the case-specific literature review
- Role of cerebellar cortex in associative learning and memory in guinea pigs
- Application of metagenomic next-generation sequencing technique for diagnosing a specific case of necrotizing meningoencephalitis caused by human herpesvirus 2
- Case report: Quadruple primary malignant neoplasms including esophageal, ureteral, and lung in an elderly male
- Long non-coding RNA NEAT1 promotes angiogenesis in hepatoma carcinoma via the miR-125a-5p/VEGF pathway
- Osteogenic differentiation of periodontal membrane stem cells in inflammatory environments
- Knockdown of SHMT2 enhances the sensitivity of gastric cancer cells to radiotherapy through the Wnt/β-catenin pathway
- Continuous renal replacement therapy combined with double filtration plasmapheresis in the treatment of severe lupus complicated by serious bacterial infections in children: A case report
- Simultaneous triple primary malignancies, including bladder cancer, lymphoma, and lung cancer, in an elderly male: A case report
- Preclinical immunogenicity assessment of a cell-based inactivated whole-virion H5N1 influenza vaccine
- One case of iodine-125 therapy – A new minimally invasive treatment of intrahepatic cholangiocarcinoma
- S1P promotes corneal trigeminal neuron differentiation and corneal nerve repair via upregulating nerve growth factor expression in a mouse model
- Early cancer detection by a targeted methylation assay of circulating tumor DNA in plasma
- Calcifying nanoparticles initiate the calcification process of mesenchymal stem cells in vitro through the activation of the TGF-β1/Smad signaling pathway and promote the decay of echinococcosis
- Evaluation of prognostic markers in patients infected with SARS-CoV-2
- N6-Methyladenosine-related alternative splicing events play a role in bladder cancer
- Characterization of the structural, oxidative, and immunological features of testis tissue from Zucker diabetic fatty rats
- Effects of glucose and osmotic pressure on the proliferation and cell cycle of human chorionic trophoblast cells
- Investigation of genotype diversity of 7,804 norovirus sequences in humans and animals of China
- Characteristics and karyotype analysis of a patient with turner syndrome complicated with multiple-site tumors: A case report
- Aggravated renal fibrosis is positively associated with the activation of HMGB1-TLR2/4 signaling in STZ-induced diabetic mice
- Distribution characteristics of SARS-CoV-2 IgM/IgG in false-positive results detected by chemiluminescent immunoassay
- SRPX2 attenuated oxygen–glucose deprivation and reperfusion-induced injury in cardiomyocytes via alleviating endoplasmic reticulum stress-induced apoptosis through targeting PI3K/Akt/mTOR axis
- Aquaporin-8 overexpression is involved in vascular structure and function changes in placentas of gestational diabetes mellitus patients
- Relationship between CRP gene polymorphisms and ischemic stroke risk: A systematic review and meta-analysis
- Effects of growth hormone on lipid metabolism and sexual development in pubertal obese male rats
- Cloning and identification of the CTLA-4IgV gene and functional application of vaccine in Xinjiang sheep
- Antitumor activity of RUNX3: Upregulation of E-cadherin and downregulation of the epithelial–mesenchymal transition in clear-cell renal cell carcinoma
- PHF8 promotes osteogenic differentiation of BMSCs in old rat with osteoporosis by regulating Wnt/β-catenin pathway
- A review of the current state of the computer-aided diagnosis (CAD) systems for breast cancer diagnosis
- Bilateral dacryoadenitis in adult-onset Still’s disease: A case report
- A novel association between Bmi-1 protein expression and the SUVmax obtained by 18F-FDG PET/CT in patients with gastric adenocarcinoma
- The role of erythrocytes and erythroid progenitor cells in tumors
- Relationship between platelet activation markers and spontaneous abortion: A meta-analysis
- Abnormal methylation caused by folic acid deficiency in neural tube defects
- Silencing TLR4 using an ultrasound-targeted microbubble destruction-based shRNA system reduces ischemia-induced seizures in hyperglycemic rats
- Plant Sciences
- Seasonal succession of bacterial communities in cultured Caulerpa lentillifera detected by high-throughput sequencing
- Cloning and prokaryotic expression of WRKY48 from Caragana intermedia
- Novel Brassica hybrids with different resistance to Leptosphaeria maculans reveal unbalanced rDNA signal patterns
- Application of exogenous auxin and gibberellin regulates the bolting of lettuce (Lactuca sativa L.)
- Phytoremediation of pollutants from wastewater: A concise review
- Genome-wide identification and characterization of NBS-encoding genes in the sweet potato wild ancestor Ipomoea trifida (H.B.K.)
- Alleviative effects of magnetic Fe3O4 nanoparticles on the physiological toxicity of 3-nitrophenol to rice (Oryza sativa L.) seedlings
- Selection and functional identification of Dof genes expressed in response to nitrogen in Populus simonii × Populus nigra
- Study on pecan seed germination influenced by seed endocarp
- Identification of active compounds in Ophiopogonis Radix from different geographical origins by UPLC-Q/TOF-MS combined with GC-MS approaches
- The entire chloroplast genome sequence of Asparagus cochinchinensis and genetic comparison to Asparagus species
- Genome-wide identification of MAPK family genes and their response to abiotic stresses in tea plant (Camellia sinensis)
- Selection and validation of reference genes for RT-qPCR analysis of different organs at various development stages in Caragana intermedia
- Cloning and expression analysis of SERK1 gene in Diospyros lotus
- Integrated metabolomic and transcriptomic profiling revealed coping mechanisms of the edible and medicinal homologous plant Plantago asiatica L. cadmium resistance
- A missense variant in NCF1 is associated with susceptibility to unexplained recurrent spontaneous abortion
- Assessment of drought tolerance indices in faba bean genotypes under different irrigation regimes
- The entire chloroplast genome sequence of Asparagus setaceus (Kunth) Jessop: Genome structure, gene composition, and phylogenetic analysis in Asparagaceae
- Food Science
- Dietary food additive monosodium glutamate with or without high-lipid diet induces spleen anomaly: A mechanistic approach on rat model
- Binge eating disorder during COVID-19
- Potential of honey against the onset of autoimmune diabetes and its associated nephropathy, pancreatitis, and retinopathy in type 1 diabetic animal model
- FTO gene expression in diet-induced obesity is downregulated by Solanum fruit supplementation
- Physical activity enhances fecal lactobacilli in rats chronically drinking sweetened cola beverage
- Supercritical CO2 extraction, chemical composition, and antioxidant effects of Coreopsis tinctoria Nutt. oleoresin
- Functional constituents of plant-based foods boost immunity against acute and chronic disorders
- Effect of selenium and methods of protein extraction on the proteomic profile of Saccharomyces yeast
- Microbial diversity of milk ghee in southern Gansu and its effect on the formation of ghee flavor compounds
- Ecology and Environmental Sciences
- Effects of heavy metals on bacterial community surrounding Bijiashan mining area located in northwest China
- Microorganism community composition analysis coupling with 15N tracer experiments reveals the nitrification rate and N2O emissions in low pH soils in Southern China
- Genetic diversity and population structure of Cinnamomum balansae Lecomte inferred by microsatellites
- Preliminary screening of microplastic contamination in different marine fish species of Taif market, Saudi Arabia
- Plant volatile organic compounds attractive to Lygus pratensis
- Effects of organic materials on soil bacterial community structure in long-term continuous cropping of tomato in greenhouse
- Effects of soil treated fungicide fluopimomide on tomato (Solanum lycopersicum L.) disease control and plant growth
- Prevalence of Yersinia pestis among rodents captured in a semi-arid tropical ecosystem of south-western Zimbabwe
- Effects of irrigation and nitrogen fertilization on mitigating salt-induced Na+ toxicity and sustaining sea rice growth
- Bioengineering and Biotechnology
- Poly-l-lysine-caused cell adhesion induces pyroptosis in THP-1 monocytes
- Development of alkaline phosphatase-scFv and its use for one-step enzyme-linked immunosorbent assay for His-tagged protein detection
- Development and validation of a predictive model for immune-related genes in patients with tongue squamous cell carcinoma
- Agriculture
- Effects of chemical-based fertilizer replacement with biochar-based fertilizer on albic soil nutrient content and maize yield
- Genome-wide identification and expression analysis of CPP-like gene family in Triticum aestivum L. under different hormone and stress conditions
- Agronomic and economic performance of mung bean (Vigna radiata L.) varieties in response to rates of blended NPS fertilizer in Kindo Koysha district, Southern Ethiopia
- Influence of furrow irrigation regime on the yield and water consumption indicators of winter wheat based on a multi-level fuzzy comprehensive evaluation
- Discovery of exercise-related genes and pathway analysis based on comparative genomes of Mongolian originated Abaga and Wushen horse
- Lessons from integrated seasonal forecast-crop modelling in Africa: A systematic review
- Evolution trend of soil fertility in tobacco-planting area of Chenzhou, Hunan Province, China
- Animal Sciences
- Morphological and molecular characterization of Tatera indica Hardwicke 1807 (Rodentia: Muridae) from Pothwar, Pakistan
- Research on meat quality of Qianhua Mutton Merino sheep and Small-tail Han sheep
- SI: A Scientific Memoir
- Suggestions on leading an academic research laboratory group
- My scientific genealogy and the Toronto ACDC Laboratory, 1988–2022
- Erratum
- Erratum to “Changes of immune cells in patients with hepatocellular carcinoma treated by radiofrequency ablation and hepatectomy, a pilot study”
- Erratum to “A two-microRNA signature predicts the progression of male thyroid cancer”
- Retraction
- Retraction of “Lidocaine has antitumor effect on hepatocellular carcinoma via the circ_DYNC1H1/miR-520a-3p/USP14 axis”

