Abstract
Exosomes were enriched from plasma by ultracentrifugation, precipitation, and membrane-based approaches for yield and purity. Using the four isolation approaches, particles with mode sizes within the expected range (50–200 nm) can be isolated. By protein estimation, polymer precipitation resulted in a maximum yield (5610.59 ± 51.189 µg/mL), followed by membrane affinity (471.57 ± 12.16 µg/mL), ultracentrifugation (440.22 ± 11.71 µg/mL) and filter + ultracentrifugation (235.47 ± 13.27 µg/mL). By total RNA estimation, the yield of polymer precipitation (3.26 ± 0.42 ng/mL) was higher than that of ultracentrifugation (1.52 ± 0.06 ng/mL), filter + ultracentrifugation (1.21 ± 0.25 ng/mL) and membrane affinity (1.44 ± 0.14 ng/mL). The purity of exosomal preparations was determined as the ratio of the particle number to protein and of protein to RNA. According to the ratio of the particle number to protein concentration, the “purity” of the polymer precipitation method was similar to that of the membrane affinity method and higher than that of ultracentrifugation and filter + ultracentrifugation. When the ratio of RNA to protein was used, the “purity” of the polymer precipitation method was lower than that of the membrane affinity method. Differential methods can be employed to enrich specific exosome subpopulations. The steps of the methods affect the particle number, protein content, and even exosomal purity. The best extraction and evaluation methods for exosomes need to be selected in the laboratory according to their experimental needs.
1 Introduction
Exosomes, which are used to describe a population of small extracellular vesicles (50–200 nm), have emerged as an area of intense interest owing to their important role in orchestrating intercellular communication and molecular exchange [1]. Exosomes are actively secreted by most, if not all, cells into a variety of bodily fluids (e.g., blood, saliva, urine, bronchial lavage, synovial fluid, amniotic fluid, breast milk), where the diverse array of nucleic acids, proteins, and lipids packaged within them can relay signals between the cell of origin and recipient cells [2]. The growing interest in molecules carried by exosomes as potential circulating biomarkers has prompted extensive research and the development of methods for the isolation of plasma exosomes [3]. Exosome isolation from plasma is especially difficult because of the small volume available and the technical challenge, especially due to the high density and viscosity of the sample and the complex composition of different types of vesicles, proteins, ribonucleoproteins, and lipoproteins [3].
Currently, several methodologies exist for the isolation and analysis of exosomes, such as ultracentrifugation, polymer precipitation, and membrane affinity. Several issues remain unresolved, especially in terms of clinical applications, including the aggregation of vesicles, low recovery, necessity of a large sample volume, and contamination with soluble proteins and lipoproteins [4]. Ultracentrifugation is the most common technique and is considered the “gold standard” for general exosome isolation [5]. However, this method requires special centrifugal equipment and is time-consuming. The polymer precipitation method is used to obtain exosomes by mixing the polymer with the sample and centrifuging at a low speed. This method can be used to precipitate exosomes, and solvent precipitation may influence the experimental results. The membrane affinity method is also used to extract exosomes from plasma by binding the vesicle membrane to the reagent column [3]. In addition, with recent advances in novel materials [6,7,8], exosomes are expected to be better enriched. The characterization of efficient alternative methods for exosome isolation from plasma can affect research related to biomarker discovery and clinical translation. This work aims to compare exosomes enriched from plasma by ultracentrifugation, precipitation, and membrane-based approaches in terms of yield and purity, contributing to the characterization of exosome isolation methods.
2 Materials and methods
2.1 Plasma preparation
The plasma collection and preparation protocols were approved by the Ethics Committee of Zhongshan Hospital, Xiamen University, and the participants provided written informed consent. Twenty milliliters of blood was obtained from a healthy 27-year-old volunteer in EDTA-2k anticoagulant tubes. The blood was gently mixed upside down 5 times and then centrifuged at 2,500×g for 15 min to separate plasma. Plasma was transferred to a clean tube and centrifuged again at 2,500×g for 15 min before being aliquoted, snap-frozen on dry ice, and stored at −80°C until use.
2.2 Isolation of exosomes by ultracentrifugation
In this article, we adopted two ultracentrifugation methods, ultracentrifugation, and filter + ultracentrifugation. For ultracentrifugation, 2 mL of plasma was diluted in 18 mL of ice-cold phosphate-buffered saline (PBS) and centrifuged at 10,000×g for 30 min at 4°C to pellet and remove the microvesicles. The supernatants were then centrifuged in a Beckman Coulter Optima L-100XP Ultracentrifuge (Beckman Coulter, Fullerton, CA, USA) at 120,000×g for 2 h at 4°C with a Type 70 Ti rotor to pellet the exosomes. The supernatant was carefully removed, and crude exosome-containing pellets were resuspended in 1 mL of ice-cold PBS. A second round of ultracentrifugation (120,000×g for 2 h at 4°C with a Type 70 Ti rotor) was carried out, and the resulting exosome pellet was resuspended in 100 µL of ice-cold PBS (Figure 1).

Exosome isolation flow diagram.
For the filter + ultracentrifugation, after 2 mL of plasma was diluted in 18 mL of ice-cold PBS and centrifuged at 10,000×g for 30 min to pellet and remove the microvesicles, the supernatant was filtered using a 0.22 µm filter to remove particles larger than 0.22 µm and then subjected to ultracentrifugation as described above (Figure 1).
2.3 Isolation of exosomes by polymer precipitation
For polymer precipitation, 2 mL of plasma was centrifuged at 10,000×g for 30 min at 4°C to pellet and remove microvesicles. The supernatant was added to a 15 mL centrifuge tube, followed by a precipitation procedure. The supernatant was mixed with 2 mL of Blood PureExo Solution (Umibio, Shanghai, China) and 8 mL of cold PBS, incubated at 4°C for 2 h, and then centrifuged at 10,000×g for 1 h. The resulting supernatant was carefully removed, and crude exosome-containing pellets were resuspended in 0.8 mL of PBS. A second round of centrifugation (12,000×g for 2 min at 4°C) was carried out, and the resulting exosome supernatant was transferred to the upper part of the Exosome Purification Filter column (Umibio, Shanghai, China) and centrifuged at 3,000×g for 10 min. The eluate of the exosome column was collected for the following analysis (Figure 1).
2.4 Isolation of exosomes by the membrane affinity method
Two milliliters of plasma was centrifuged at 10,000×g for 30 min at 4°C to pellet and remove microvesicles. The supernatant was carefully collected and mixed 1:1 with buffer XBP (Qiagen, Hilden, Germany) and then added to an exoEasy spin column. After centrifugation at 500×g for 1 min, the flowthrough was discarded, and 10 mL of wash buffer was added to the column to wash away nonspecifically retained material. After another centrifugation (5,000×g for 5 min) and elimination of the flowthrough, the eluate with exosomes was collected by adding 400 µL of Buffer XE to the spin column and incubated for 1 min. After centrifugation at 500×g for 5 min, the eluate from the exosome column was collected for the following analysis (Figure 1).
2.5 Transmission electron microscopy
Exosomes were visualized using transmission electron microscopy as previously described, with slight modifications [9]. Briefly, exosome suspensions were diluted 1:50 with PBS, and 20 µL of exosome preparations was allowed to adsorb on a copper mesh for 3 min and was negatively stained with 2% (w/v) phosphotungstic acid for 5 min. Transmission electron microscopy analysis was performed at an acceleration voltage of 80 kV with a transmission electron microscope (H-7650; Hitachi, Ltd, Tokyo, Japan).
2.6 Protein quantification
Protein concentration was detected using a BCA protein assay kit (Thermo Fisher Scientific, Rockford, IL, USA) following the manufacturer's instructions.
2.7 Western blot analysis
The samples were mixed with 5× loading buffer and heated at 95°C for 5 min. Volumes corresponding to 25 mg of protein from isolates were separated on a 5–12% polyacrylamide gel. Samples were then transferred onto a nitrocellulose membrane (Bio-Rad Laboratories), which was blocked with 5% nonfat milk in Tris-buffered saline (TBS) for 1 h. The membrane was incubated with primary antibodies against calnexin (1:1,000; Abcam), albumin (1:500; Abcam), TSG101 (1:500; Abcam), galectin-3 BP (1:500; Abcam), and CD9 (1:1,000; Abcam) dissolved in 0.05% TBS-Tween overnight at 4°C, and then, the membrane was washed with 0.05% TBS-Tween for 10 min three times. Secondary antibodies were diluted in 0.05% nonfat milk in TBST and incubated for 1 h. The membrane was analyzed with ECL Prime Western Blotting Detection.
2.8 Nanoparticle tracking analysis
Exosome concentration was analyzed using a NanoSight LM10 system (NanoSight Ltd, Navato, CA) equipped with a blue laser (405 nm). Nanoparticles were illuminated by the laser, and their movement under Brownian motion was captured for 60 s. The process was repeated three times. Then, all three recorded videos were subjected to nanoparticle tracking analysis (NTA) using NanoSight particle tracking software to calculate exosome concentrations and size distribution.
2.9 Total RNA extraction
Total RNA was isolated using a miRNA Serum/Plasma Advanced Kit (Qiagen, Valencia, CA) according to the manufacturer’s protocol. The RNA concentration and integrity were determined by capillary electrophoresis using an Agilent 2100 Bioanalyzer (Agilent Technologies, Rockville, MD). For the detection of mRNA in exosomes, the total isolated RNA was converted to cDNA using a PrimeScriptTM RT reagent Kit (TaKaRa Bio, Inc., Kusatsu, Japan) and oligo (dT) primers. cDNA detection was performed using a Bioanalyzer. Quantification of miR-130 was performed using miR-130-specific forward primers and universal reverse primers.
2.10 Statistics
Data are expressed as the mean ± SD and were assessed using SPSS 18.0 statistical software (SPSS, Inc., Chicago, USA). One-way analysis of variance (ANOVA) was used to assess the differences between multiple groups, followed by Tukey’s post hoc test. A P value <0.05 was considered significant.
3 Results
3.1 Morphological characterization of exosomes extracted by different isolation procedures
Transmission electron microscopy analysis revealed that all isolation procedures successfully isolated exosomes with the expected size range and morphology. Cup-shaped vesicles were observed with heterogeneous sizes ranging from approximately 50–200 nm (Figure 2). The morphology of exosomes in samples was not different in the ultracentrifugation, filter + ultracentrifugation, and membrane affinity groups, and in the polymer precipitation group, the shape of exosomes was slightly irregular.

Electron microscopic image of exosomes following different isolation methods. Morphology of exosomes (white arrow indicate) isolated using (a) ultracentrifugation, (b) filter + ultracentrifugation, (c) membrane affinity, (d) and polymer precipitation. Cup-shaped vesicles with a heterogeneous size (50–200 nm diameter) were clearly visualized in all preparations. The bars indicate 100 nm.
3.2 The size and concentration of exosomes extracted with different isolation procedures
NTA profiles of exosomes obtained with the different isolation procedures revealed that the mode size was in the expected range of 50–200 nm. The major peaks of the ultracentrifugation, filter + ultracentrifugation, polymer precipitation, and membrane affinity groups were 96.6 ± 2.7, 97.9 ± 0.6, 96.6 ± 0.9, and 97.5 ± 0.5 nm, respectively (Figure 3a and b). In the exosomes enriched by membrane affinity, we observed several subpopulations of differently sized vesicles with multiple concentration peaks in NTA. NTA quantification confirmed that polymer precipitation resulted in the highest number of particles (6.20 ± 1.74 × 1011), followed by the membrane affinity (6.06 ± 1.90 × 1010), ultracentrifugation (5.86 ± 0.72 × 1010), and ultracentrifugation + filter methods (1.60 ± 0.40 × 1010). The number of enriched particles was not different among the membrane affinity, ultracentrifugation, and ultracentrifugation + filter groups (Figure 3c).

Size distribution and concentration of exosomes enriched with different isolation procedures. (a) Particle size for plasma exosomes measured by nanoparticle tracking analyses (NTA). Insets depict higher magnifications of the black dot box areas. (b) Size distribution as a percentage of vesicles larger than 195 nm (black), between 35 and 195 nm (gray) and smaller than 35 nm (white) based on the same data as in (a). (c) Particle concentration of exosomes extracted by different isolation procedures. The results are the mean ± standard error of the mean of three independent experiments. One-way analysis of variance (ANOVA) was used to assess the differences between multiple groups, followed by Tukey’s post hoc test. **P < 0.01. NS, P > 0.05.
3.3 The yield of exosomes from different isolation procedures
The total exosome yield was determined by protein estimation from intact exosomes using the BCA protein assay. We observed that the polymer precipitation method had the maximum yield (5610.59 ± 51.189 µg/mL), followed by the membrane affinity (471.57 ± 12.16 µg/mL), ultracentrifugation (440.22 ± 11.71 µg/mL), and filter + ultracentrifugation methods (235.47 ± 13.27 µg/mL). The filter + ultracentrifugation method yielded the lowest level of exosomes (Figure 4a).

The yield of exosomes from different isolation procedures. (a) The exosomal yield presented as the protein estimation. (b) The exosomal yield presented as the total RNA. (c) Representative bioanalyzer profiles of RNA analyzed by an Agilent 2100 Bioanalyzer; the y-axis shows fluorescence units (FU), and the x-axis shows the nucleotide length (nt) of the RNA. The results are the mean ± standard error of the mean of three independent experiments. One-way analysis of variance (ANOVA) was used to assess the differences among multiple groups, followed by Tukey’s post hoc test. **, P < 0.01; ***, P < 0.001. NS, P > 0.05.
As exosomes are considered an important source of RNA-based biomarkers, we also assessed exosome yield based on the total RNA content of exosomes determined by a Bioanalyzer. We found that RNA recovery by polymer precipitation (3.26 ± 0.42 ng/mL) was higher than that of ultracentrifugation (1.52 ± 0.06 ng/mL), filter + ultracentrifugation (1.21 ± 0.25 ng/mL), and membrane affinity (1.44 ± 0.14 ng/mL), and the RNA recovery by the latter three methods was not significantly different (P > 0.05) (Figure 4b). The Bioanalyzer profiles of isolated RNA showed the presence of small RNAs for different isolation procedures (Figure 4c).
In addition, to evaluate the yield of exosomes obtained from each isolation procedure, we performed an immunoblot with the loading of equal amounts of protein and demonstrated that the expression of Galectin-3 BP, CD9, and Tsg101 – all considered late endosomal markers enriched in exosomes – was positive in each isolation procedure. The expression levels of Galectin-3 BP, CD9, and Tsg101 were different in isolation procedures under the same protein loading. Galectin 3 BP was expressed at the lowest level in the polymer precipitation group, and TSG101 and CD9 were both expressed at the lowest level in the membrane affinity group (Figure 5).
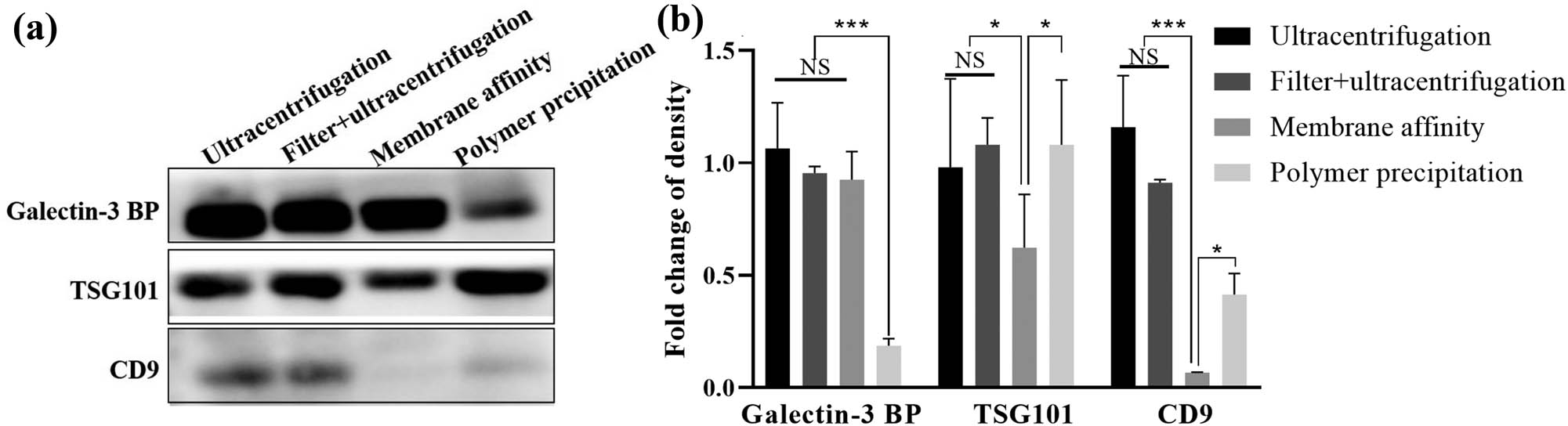
Surface marker expression profiles in exosome preparations. (a) Western blot for Galectin-3 BP, Tsg101, and CD9 expression in each exosome preparation (20 µL of total exosomal protein was loaded). (b) Quantification of the expression levels of Galectin-3 BP, Tsg101, and CD9 in each exosome preparation. The results are the mean ± SE of the mean of three independent experiments. One-way analysis of variance (ANOVA) was used to assess the differences among multiple groups, followed by Tukey’s post hoc test. * P < 0.05, ***P < 0.001.
3.4 The purity of exosomes extracted by different isolation procedures
The purity of the exosomes was determined by the ratio between particle concentration and protein. Experiments showed that exosome extraction by membrane affinity and polymer precipitation showed excellent purity [(130.83 ± 43.50) × 107 vs (110.41 ± 30.47) × 107 particles/µg, P > 0.05], while ultracentrifugation [(13.36 ± 1.70) × 107 particles/µg] and filter + ultracentrifugation [(7.04 ± 2.22) × 107 particles/µg] resulted in the lowest purity (Figure 6a).

Purity of exosomes from different isolation procedures. (a) The exosomal purity presented as a ratio between particle concentration and protein. (b) Exosomal purity presented as the RNA (µg) to protein (µg) ratio. (c) Western blots for calnexin and albumin expression in each exosome preparation (20 µL of total exosomal protein was loaded). The results are the mean ± SE of the mean of three independent experiments. One-way analysis of variance (ANOVA) was used to assess the differences among multiple groups, followed by Tukey’s post hoc test. *, P < 0.05. **, P < 0.01. NS, P > 0.05.
The ratio of RNA to protein was previously found to be higher in exosomes than in microvesicles, suggesting that more RNA is associated with exosomes [10]. We also calculated the RNA to protein ratio to determine the exosomal purity. This ratio was significantly lower for the exosomes isolated by polymer precipitation than for the exosomes isolated by ultracentrifugation, filter + ultracentrifugation, or membrane affinity (Figure 6b). There was no difference in the ratio of RNA to protein among the latter three groups.
As plasma is a very complex and viscous fluid, the presence of contaminants is another method to determine the purity of exosomes. We assessed calnexin and albumin contamination by Western blotting. All samples were negative for calnexin, an endoplasmic reticulum component protein. Albumin was present in all exosome extracts analyzed, and the membrane affinity group had the highest albumin contamination (Figure 6c).
3.5 Practicability of different isolation procedures
For the practicability of different isolation procedures, ultracentrifugation requires special equipment (ultracentrifuge) and takes approximately 4 h. Special training is needed for researchers who are learning to perform exosome extraction by applying the ultracentrifugation method. After simple training, nonprofessionals can perform and master the membrane affinity and polymer precipitation approaches. The isolation time was approximately 3 h 12 min for polymer precipitation and only 25 min for membrane affinity (Table S1).
4 Discussion
Exosomes have emerged as a promising alternative source of biomarkers for several diseases since they can be obtained from almost all biological fluids [2]. As a result, efficient isolation of exosomes has been an active area of research to understand their biological properties and to explore their potential in biomarker development for early disease diagnosis [11]. In this article, we evaluated four exosome isolation methods and generated significant data on their relative efficacy with regard to exosome yield and purity. The NTA results showed that the diameters of the exosomes extracted by the four methods all met the standard exosome size of 50–200 nm. Among all the methods tested, the polymer precipitation method had the maximum yield (5610.59 ± 51.18 µg/mL), followed by membrane affinity (471.57 ± 12.16 µg/mL), ultracentrifugation (440.22 ± 11.71 µg/mL), and filter + ultracentrifugation (235.47 ± 13.27 µg/mL), as shown by protein estimation. Patel et al. also found that the precipitation-based method yielded the highest quantity [12], and the highest yield in the precipitation method could be likely due to precipitation of extracellular vesicles other than exosomes or additional aggregated proteins. The polymer precipitation method also provided the most particles and the maximum yield of total RNA. Differential protein, particle, or total RNA yields reflected different exosome quantities obtained from various methods.
Exosomes are nanometer-sized (50–200 nm) extracellular vesicles that are actively shed from cells into body fluids. In this article, the plasma was filtered using 0.22 µm filters to remove particles larger than 0.22 µm [13]. We accordingly investigated the purity and yield of exosomes extracted by ultracentrifugation and filter + ultracentrifugation. The protein concentration of the exosomes was low in the filter + ultracentrifugation group, but the number of particles and the total RNA yield did not decrease when using 0.22 µm filters. Therefore, we believe that although the diameter of the exosomes is less than 200 nm, a filter membrane with an aperture of 0.22 µm can still absorb a certain amount of exosomes, which may be related to the protein concentration of the exosome extract obtained by filter + ultracentrifugation.
Purity is an indispensable index for exosome evaluation. As previously reported, the purity of exosomal preparations was determined as a ratio of particle number to protein concentration [14,15] or a ratio of RNA to protein concentration [3]. Interestingly, for the membrane affinity method, the results of the two calculation methods were completely opposite. According to the ratio of the particle number to protein concentration, the “purity” of the polymer precipitation method was similar to that of the membrane affinity methods and higher than that of the ultracentrifugation and filter + ultracentrifugation methods. When the ratio of RNA to protein concentration was used, the “purity” of the polymer precipitation was lower than that of the membrane affinity method. Using different purity evaluation methods, we found that the exosomal preparations showed different levels of “purity”; thus, researchers need to perform appropriate exosome purity evaluations according to the experimental purpose.
As plasma is a complex and viscous fluid with a protein concentration of approximately 60–80 g/L, the isolation of exosomes to measure proteins that are in the range of 10−6 g/L is a major challenge [16,17]. For this reason, it is important not only to have a method that can purify exosomes efficiently but also have a method that can remove most impurities from plasma [4]. In this article, we did not detect contaminant debris from the endoplasmic reticulum marker calnexin, an endoplasmic reticulum component, thus eliminating the presence of cell contamination in the exosomal preparations. However, another serum contaminant, albumin, was present in all exosome extracts analyzed, and the membrane affinity method had the highest albumin level. The effect of albumin contamination on the function of extracted exosomes needs to be further evaluated. In addition, we did not assess the relevant marker of lipoprotein, which needs to be assessed in future studies.
Ultracentrifugation has been the most widely used method for exosome extraction but there are some limitations in the application of this method [18]. This strategy requires special equipment and takes a long time (approximately 4 h), and special training is needed for researchers who are learning to perform exosome extraction by applying the ultracentrifugation method [5]. The polymer precipitation method and membrane affinity method can effectively shorten the required time but albumin can be either nonspecifically bound to exosomes or obtained during the process of purification, which was confirmed by the polymer precipitation method and membrane affinity method showing higher albumin contamination than ultracentrifugation [9]. How to balance the ease of operation and albumin contamination is an urgent problem to be solved in the clinical application of plasma exosomes.
To uniformly compare the extraction efficiency of exosomes by different methods, we only used single samples for comparison but almost all cells could produce exosomes, and the interindividual variability could be high. A multiperson sample comparison of special samples (hemolytic, lipemic, or icteric samples) is also necessary.
5 Conclusion
Current research on exosomes is challenged by efficient isolation strategies with high specificity that are practicable. This study suggests that different methods can be employed to enrich specific exosome subpopulations. Our results also indicate that the exact steps of differential methods affect the particle number, protein content, and even purity of exosomes. The laboratory needs to select the best extraction and evaluation methods of exosomes according to their experimental needs.
Acknowledgements
This work was supported by the National Natural Science Foundation of China [grant numbers 82172331, 82102514, 81972028, 81871729, and 81672094] and the Key Projects for Province Science and Technology Program of Fujian Province, China [grant numbers 2020D017 and 2019D008]. The funders played no role in the study design, data collection, analyses, decision to publish, or manuscript preparation.
-
Author contributions: WJ Li, H Chen, and L-R Lin conceived and designed the experiments. J-J Niu and X-Z Zhu performed the experiments. M-L Tong analyzed the data. L-R Lin wrote the paper.
-
Conflict of interest: The authors of this article have no conflicts of interest to disclose.
-
Ethical approval: The conducted research is not related to either human or animal use.
-
Data availability statement: The raw data supporting the conclusions of this article will be made available by the authors, without undue reservation.
References
[1] Hoshino A, Han SK, Bojmar L, Gyan KE, Lyden D. Extracellular vesicle and particle biomarkers define multiple human cancers. Cell. 2020;182(4):1–37.10.1016/j.cell.2020.07.009Search in Google Scholar PubMed PubMed Central
[2] Contreras-Naranjo JC, Wu HJ, Ugaz VM. Microfluidics for exosome isolation and analysis: enabling liquid biopsy for personalized medicine. Lab Chip. 2017;17(21):3558–77.10.1039/C7LC00592JSearch in Google Scholar PubMed PubMed Central
[3] Stranska R, Gysbrechts L, Wouters J, Vermeersch P, Bloch K, Dierickx D, et al. Comparison of membrane affinity-based method with size-exclusion chromatography for isolation of exosome-like vesicles from human plasma. J Transl Med. 2018;16(1):1–10.10.1186/s12967-017-1374-6Search in Google Scholar PubMed PubMed Central
[4] Gonzalez-Cao M, Alegre E, Luis P-G, Jose G, Alvaro Z. Circulating melanoma exosomes as diagnostic and prognosis biomarkers. Clin Chim Acta. 2016;454:28–32.10.1016/j.cca.2015.12.031Search in Google Scholar PubMed
[5] Nath Neerukonda S, Egan NA, Patria J, Assakhi I, Tavlarides-Hontz P, Modla S, et al. Comparison of exosomes purified via ultracentrifugation (UC) and total exosome isolation (TEI) reagent from the serum of Marek's disease virus (MDV)-vaccinated and tumor-bearing chickens. J Virol Methods. 2019;263:1–9.10.1016/j.jviromet.2018.10.004Search in Google Scholar PubMed
[6] Al Zoubi W, Ko YG. Freestanding anticorrosion hybrid materials based on coordination interaction between metal-quinoline compounds and TiO2-MgO film. J Colloid Interface Sci. 2020;565:86–95.10.1016/j.jcis.2020.01.017Search in Google Scholar PubMed
[7] Al Zoubi W, Kamil MP, Fatimah S, Nashrah N, Ko YG. Recent advances in hybrid organic–inorganic materials with spatial architecture for state-of-the-art applications. Prog Mater Sci. 2020;112:1–10.10.1016/j.pmatsci.2020.100663Search in Google Scholar
[8] Al Zoubi W, Kim MJ, Kim YG, Ko YG. Dual-functional crosslinked polymer-inorganic materials for robust electrochemical performance and antibacterial activity. Chem Eng J. 2020;392:1–10.10.1016/j.cej.2019.123654Search in Google Scholar
[9] Moon S, Shin DW, Kim S, Lee YS, Mankhong S, Yang SW, et al. Enrichment of exosome-like extracellular vesicles from plasma suitable for clinical vesicular miRNA biomarker research. J Clin Med. 2019;8(11):1–16.10.3390/jcm8111995Search in Google Scholar PubMed PubMed Central
[10] Lunavat TR, Cheng L, Kim DK, Bhadury J, Jang SC, Lässer C, et al. Small RNA deep sequencing discriminates subsets of extracellular vesicles released by melanoma cells – evidence of unique microRNA cargos. Rna Biol. 2015;S12:810–23.10.1080/15476286.2015.1056975Search in Google Scholar PubMed PubMed Central
[11] Pan S, Pei L, Zhang A, Zhang Y, Zhang C, Huang M, et al. Passion fruit-like exosome-PMA/Au-BSA@Ce6 nanovehicles for real-time fluorescence imaging and enhanced targeted photodynamic therapy with deep penetration and superior retention behavior in tumor. Biomaterials. 2020;230:119606.10.1016/j.biomaterials.2019.119606Search in Google Scholar PubMed
[12] Patel GK, Khan MA, Zubair H, Srivastava SK, Khushman M, Singh S, et al. Comparative analysis of exosome isolation methods using culture supernatant for optimum yield, purity and downstream applications. Sci Rep. 2019;9(1):5335.10.1038/s41598-019-41800-2Search in Google Scholar PubMed PubMed Central
[13] Rea AW, Zufall MJ, Williams RW, Sheldon L, Howard-Reed C. The influence of human activity patterns on personal PM exposure: a comparative analysis of filter-based and continuous particle measurements. Air Repair. 2001;51(9):1271–9.10.1080/10473289.2001.10464351Search in Google Scholar PubMed
[14] Lobb RJ, Becker M, Wen SW, Wong CS, Wiegmans AP, Leimgruber A, et al. Optimized exosome isolation protocol for cell culture supernatant and human plasma. J Extracell Vesicles. 2015;4:27031.10.3402/jev.v4.27031Search in Google Scholar PubMed PubMed Central
[15] Webber J, Clayton A. How pure are your vesicles? J Extracell Vesicles. 2013;2(1):1–10.10.3402/jev.v2i0.19861Search in Google Scholar PubMed PubMed Central
[16] Millioni R, Tolin S, Puricelli L, Sbrignadello S, Arrigoni G. High abundance proteins depletion vs low abundance proteins enrichment: comparison of methods to reduce the plasma proteome complexity. PLoS One. 2011;6(5):e19603.10.1371/journal.pone.0019603Search in Google Scholar PubMed PubMed Central
[17] Kalra H, Adda CG, Liem M, Ang CS, Mechler A, Simpson RJ, et al. Comparative proteomics evaluation of plasma exosome isolation techniques and assessment of the stability of exosomes in normal human blood plasma. Proteomics. 2013;13(22):3354–64.10.1002/pmic.201300282Search in Google Scholar PubMed
[18] Helwa I, Cai J, Drewry MD, Zimmerman A, Dinkins MB, Khaled ML, et al. A comparative study of serum exosome isolation using differential ultracentrifugation and three commercial reagents. PLoS One. 2017;12(1):e0170628.10.1371/journal.pone.0170628Search in Google Scholar PubMed PubMed Central
© 2022 Wei-Jian Li et al., published by De Gruyter
This work is licensed under the Creative Commons Attribution 4.0 International License.
Articles in the same Issue
- Regular Articles
- Photocatalytic degradation of Rhodamine B in aqueous phase by bimetallic metal-organic framework M/Fe-MOF (M = Co, Cu, and Mg)
- Assessment of using electronic portal imaging device for analysing bolus material utilised in radiation therapy
- A detailed investigation on highly dense CuZr bulk metallic glasses for shielding purposes
- Simulation of gamma-ray shielding properties for materials of medical interest
- Environmental impact assesment regulation applications and their analysis in Turkey
- Sample age effect on parameters of dynamic nuclear polarization in certain difluorobenzen isomers/MC800 asphaltene suspensions
- Passenger demand forecasting for railway systems
- Design of a Robust sliding mode controller for bioreactor cultures in overflow metabolism via an interdisciplinary approach
- Gamma, neutron, and heavy charged ion shielding properties of Er3+-doped and Sm3+-doped zinc borate glasses
- Bridging chiral de-tert-butylcalix[4]arenes: Optical resolution based on column chromatography and structural characterization
- Petrology and geochemistry of multiphase post-granitic dikes: A case study from the Gabal Serbal area, Southwestern Sinai, Egypt
- Comparison of the yield and purity of plasma exosomes extracted by ultracentrifugation, precipitation, and membrane-based approaches
- Bioactive triterpenoids from Indonesian medicinal plant Syzygium aqueum
- Investigation of the effects of machining parameters on surface integrity in micromachining
- The mesoporous aluminosilicate application as support for bifunctional catalysts for n-hexadecane hydroconversion
- Gamma-ray shielding properties of Nd2O3-added iron–boron–phosphate-based composites
- Numerical investigation on perforated sheet metals under tension loading
- Statistical analysis on the radiological assessment and geochemical studies of granite rocks in the north of Um Taghir area, Eastern Desert, Egypt
- Two new polypodane-type bicyclic triterpenoids from mastic
- Structural, physical, and mechanical properties of the TiO2 added hydroxyapatite composites
- Tribological properties and characterization of borided Co–Mg alloys
- Studies on Anemone nemorosa L. extracts; polyphenols profile, antioxidant activity, and effects on Caco-2 cells by in vitro and in silico studies
- Mechanical properties, elastic moduli, transmission factors, and gamma-ray-shielding performances of Bi2O3–P2O5–B2O3–V2O5 quaternary glass system
- Cyclic connectivity index of bipolar fuzzy incidence graph
- The role of passage numbers of donor cells in the development of Arabian Oryx – Cow interspecific somatic cell nuclear transfer embryos
- Mechanical property evaluation of tellurite–germanate glasses and comparison of their radiation-shielding characteristics using EPICS2017 to other glass systems
- Molecular screening of ionic liquids for CO2 absorption and molecular dynamic simulation
- Microwave-assisted preparation of Ag/Fe magnetic biochar from clivia leaves for adsorbing daptomycin antibiotics
- Iminodisuccinic acid enhances antioxidant and mineral element accumulation in young leaves of Ziziphus jujuba
- Cytotoxic activity of guaiane-type sesquiterpene lactone (deoxycynaropicrin) isolated from the leaves of Centaurothamnus maximus
- Effects of welding parameters on the angular distortion of welded steel plates
- Simulation of a reactor considering the Stamicarbon, Snamprogetti, and Toyo patents for obtaining urea
- Effect of different ramie (Boehmeria nivea L. Gaud) cultivars on the adsorption of heavy metal ions cadmium and lead in the remediation of contaminated farmland soils
- Impact of a live bacterial-based direct-fed microbial (DFM) postpartum and weaning system on performance, mortality, and health of Najdi lambs
- Anti-tumor effect of liposomes containing extracted Murrayafoline A against liver cancer cells in 2D and 3D cultured models
- Physicochemical properties and some mineral concentration of milk samples from different animals and altitudes
- Copper(ii) complexes supported by modified azo-based ligands: Nucleic acid binding and molecular docking studies
- Diagnostic and therapeutic radioisotopes in nuclear medicine: Determination of gamma-ray transmission factors and safety competencies of high-dense and transparent glassy shields
- Calculation of NaI(Tl) detector efficiency using 226Ra, 232Th, and 40K radioisotopes: Three-phase Monte Carlo simulation study
- Isolation and identification of unstable components from Caesalpinia sappan by high-speed counter-current chromatography combined with preparative high-performance liquid chromatography
- Quantification of biomarkers and evaluation of antioxidant, anti-inflammatory, and cytotoxicity properties of Dodonaea viscosa grown in Saudi Arabia using HPTLC technique
- Characterization of the elastic modulus of ceramic–metal composites with physical and mechanical properties by ultrasonic technique
- GC-MS analysis of Vespa velutina auraria Smith and its anti-inflammatory and antioxidant activities in vitro
- Texturing of nanocoatings for surface acoustic wave-based sensors for volatile organic compounds
- Insights into the molecular basis of some chalcone analogues as potential inhibitors of Leishmania donovani: An integrated in silico and in vitro study
- (1R,2S,5R)-5-Methyl-2-(propan-2-yl)cyclohexyl 4-amino-3-phenylbutanoate hydrochloride: Synthesis and anticonvulsant activity
- On the relative extraction rates of colour compounds and caffeine during brewing, an investigation of tea over time and temperature
- Characterization of egg shell powder-doped ceramic–metal composites
- Rapeseed oil-based hippurate amide nanocomposite coating material for anticorrosive and antibacterial applications
- Chemically modified Teucrium polium (Lamiaceae) plant act as an effective adsorbent tool for potassium permanganate (KMnO4) in wastewater remediation
- Efficiency analysis of photovoltaic systems installed in different geographical locations
- Risk prioritization model driven by success factor in the light of multicriteria decision making
- Theoretical investigations on the excited-state intramolecular proton transfer in the solvated 2-hydroxy-1-naphthaldehyde carbohydrazone
- Mechanical and gamma-ray shielding examinations of Bi2O3–PbO–CdO–B2O3 glass system
- Machine learning-based forecasting of potability of drinking water through adaptive boosting model
- The potential effect of the Rumex vesicarius water seeds extract treatment on mice before and during pregnancy on the serum enzymes and the histology of kidney and liver
- Impact of benzimidazole functional groups on the n-doping properties of benzimidazole derivatives
- Extraction of red pigment from Chinese jujube peel and the antioxidant activity of the pigment extracts
- Flexural strength and thermal properties of carbon black nanoparticle reinforced epoxy composites obtained from waste tires
- A focusing study on radioprotective and antioxidant effects of Annona muricata leaf extract in the circulation and liver tissue: Clinical and experimental studies
- Clinical comprehensive and experimental assessment of the radioprotective effect of Annona muricata leaf extract to prevent cellular damage in the ileum tissue
- Effect of WC content on ultrasonic properties, thermal and electrical conductivity of WC–Co–Ni–Cr composites
- Influence of various class cleaning agents for prosthesis on Co–Cr alloy surface
- The synthesis of nanocellulose-based nanocomposites for the effective removal of hexavalent chromium ions from aqueous solution
- Study on the influence of physical interlayers on the remaining oil production under different development modes
- Optimized linear regression control of DC motor under various disturbances
- Influence of different sample preparation strategies on hypothesis-driven shotgun proteomic analysis of human saliva
- Determination of flow distance of the fluid metal due to fluidity in ductile iron casting by artificial neural networks approach
- Investigation of mechanical activation effect on high-volume natural pozzolanic cements
- In vitro: Anti-coccidia activity of Calotropis procera leaf extract on Eimeria papillata oocysts sporulation and sporozoite
- Determination of oil composition of cowpea (Vigna unguiculata L.) seeds under influence of organic fertilizer forms
- Activated partial thromboplastin time maybe associated with the prognosis of papillary thyroid carcinoma
- Treatment of rat brain ischemia model by NSCs-polymer scaffold transplantation
- Lead and cadmium removal with native yeast from coastal wetlands
- Characterization of electroless Ni-coated Fe–Co composite using powder metallurgy
- Ferrate synthesis using NaOCl and its application for dye removal
- Antioxidant, antidiabetic, and anticholinesterase potential of Chenopodium murale L. extracts using in vitro and in vivo approaches
- Study on essential oil, antioxidant activity, anti-human prostate cancer effects, and induction of apoptosis by Equisetum arvense
- Experimental study on turning machine with permanent magnetic cutting tool
- Numerical simulation and mathematical modeling of the casting process for pearlitic spheroidal graphite cast iron
- Design, synthesis, and cytotoxicity evaluation of novel thiophene, pyrimidine, pyridazine, and pyridine: Griseofulvin heterocyclic extension derivatives
- Isolation and identification of promising antibiotic-producing bacteria
- Ultrasonic-induced reversible blood–brain barrier opening: Safety evaluation into the cellular level
- Evaluation of phytochemical and antioxidant potential of various extracts from traditionally used medicinal plants of Pakistan
- Effect of calcium lactate in standard diet on selected markers of oxidative stress and inflammation in ovariectomized rats
- Identification of crucial salivary proteins/genes and pathways involved in pathogenesis of temporomandibular disorders
- Zirconium-modified attapulgite was used for removing of Cr(vi) in aqueous solution
- The stress distribution of different types of restorative materials in primary molar
- Reducing surface heat loss in steam boilers
- Deformation behavior and formability of friction stir processed DP600 steel
- Synthesis and characterization of bismuth oxide/commercial activated carbon composite for battery anode
- Phytochemical analysis of Ziziphus jujube leaf at different foliar ages based on widely targeted metabolomics
- Effects of in ovo injection of black cumin (Nigella sativa) extract on hatching performance of broiler eggs
- Separation and evaluation of potential antioxidant, analgesic, and anti-inflammatory activities of limonene-rich essential oils from Citrus sinensis (L.)
- Bioactivity of a polyhydroxy gorgostane steroid from Xenia umbellata
- BiCAM-based automated scoring system for digital logic circuit diagrams
- Analysis of standard systems with solar monitoring systems
- Structural and spectroscopic properties of voriconazole and fluconazole – Experimental and theoretical studies
- New plant resistance inducers based on polyamines
- Experimental investigation of single-lap bolted and bolted/bonded (hybrid) joints of polymeric plates
- Investigation of inlet air pressure and evaporative cooling of four different cogeneration cycles
- Review Articles
- Comprehensive review on synthesis, physicochemical properties, and application of activated carbon from the Arecaceae plants for enhanced wastewater treatment
- Research progress on speciation analysis of arsenic in traditional Chinese medicine
- Recent modified air-assisted liquid–liquid microextraction applications for medicines and organic compounds in various samples: A review
- An insight on Vietnamese bio-waste materials as activated carbon precursors for multiple applications in environmental protection
- Antimicrobial activities of the extracts and secondary metabolites from Clausena genus – A review
- Bioremediation of organic/heavy metal contaminants by mixed cultures of microorganisms: A review
- Sonodynamic therapy for breast cancer: A literature review
- Recent progress of amino acid transporters as a novel antitumor target
- Aconitum coreanum Rapaics: Botany, traditional uses, phytochemistry, pharmacology, and toxicology
- Corrigendum
- Corrigendum to “Petrology and geochemistry of multiphase post-granitic dikes: A case study from the Gabal Serbal area, Southwestern Sinai, Egypt”
- Corrigendum to “Design of a Robust sliding mode controller for bioreactor cultures in overflow metabolism via an interdisciplinary approach”
- Corrigendum to “Statistical analysis on the radiological assessment and geochemical studies of granite rocks in the north of Um Taghir area, Eastern Desert, Egypt”
- Corrigendum to “Aroma components of tobacco powder from different producing areas based on gas chromatography ion mobility spectrometry”
- Corrigendum to “Mechanical properties, elastic moduli, transmission factors, and gamma-ray-shielding performances of Bi2O3–P2O5–B2O3–V2O5 quaternary glass system”
- Erratum
- Erratum to “Copper(ii) complexes supported by modified azo-based ligands: Nucleic acid binding and molecular docking studies”
- Special Issue on Applied Biochemistry and Biotechnology (ABB 2021)
- Study of solidification and stabilization of heavy metals by passivators in heavy metal-contaminated soil
- Human health risk assessment and distribution of VOCs in a chemical site, Weinan, China
- Preparation and characterization of Sparassis latifolia β-glucan microcapsules
- Special Issue on the Conference of Energy, Fuels, Environment 2020
- Improving the thermal performance of existing buildings in light of the requirements of the EU directive 2010/31/EU in Poland
- Special Issue on Ethnobotanical, Phytochemical and Biological Investigation of Medicinal Plants
- Study of plant resources with ethnomedicinal relevance from district Bagh, Azad Jammu and Kashmir, Pakistan
- Studies on the chemical composition of plants used in traditional medicine in Congo
- Special Issue on Applied Chemistry in Agriculture and Food Science
- Strip spraying technology for precise herbicide application in carrot fields
- Special Issue on Pharmacology and Metabolomics of Ethnobotanical and Herbal Medicine
- Phytochemical profiling, antibacterial and antioxidant properties of Crocus sativus flower: A comparison between tepals and stigmas
- Antioxidant and antimicrobial properties of polyphenolics from Withania adpressa (Coss.) Batt. against selected drug-resistant bacterial strains
- Integrating network pharmacology and molecular docking to explore the potential mechanism of Xinguan No. 3 in the treatment of COVID-19
- Chemical composition and in vitro and in vivo biological assortment of fixed oil extracted from Ficus benghalensis L.
- A review of the pharmacological activities and protective effects of Inonotus obliquus triterpenoids in kidney diseases
- Ethnopharmacological study of medicinal plants in Kastamonu province (Türkiye)
- Protective effects of asperuloside against cyclophosphamide-induced urotoxicity and hematotoxicity in rats
- Special Issue on Essential Oil, Extraction, Phytochemistry, Advances, and Application
- Identification of volatile compounds and antioxidant, antibacterial, and antifungal properties against drug-resistant microbes of essential oils from the leaves of Mentha rotundifolia var. apodysa Briq. (Lamiaceae)
- Phenolic contents, anticancer, antioxidant, and antimicrobial capacities of MeOH extract from the aerial parts of Trema orientalis plant
- Chemical composition and antimicrobial activity of essential oils from Mentha pulegium and Rosmarinus officinalis against multidrug-resistant microbes and their acute toxicity study
- Special Issue on Marine Environmental Sciences and Significance of the Multidisciplinary Approaches
- An insightful overview of the distribution pattern of polycyclic aromatic hydrocarbon in the marine sediments of the Red Sea
- Antifungal–antiproliferative norcycloartane-type triterpenes from the Red Sea green alga Tydemania expeditionis
- Solvent effect, dipole moment, and DFT studies of multi donor–acceptor type pyridine derivative
- An extensive assessment on the distribution pattern of organic contaminants in the aerosols samples in the Middle East
- Special Issue on 4th IC3PE
- Energetics of carboxylic acid–pyridine heterosynthon revisited: A computational study of intermolecular hydrogen bond domination on phenylacetic acid–nicotinamide cocrystals
- A review: Silver–zinc oxide nanoparticles – organoclay-reinforced chitosan bionanocomposites for food packaging
- Green synthesis of magnetic activated carbon from peanut shells functionalized with TiO2 photocatalyst for Batik liquid waste treatment
- Coagulation activity of liquid extraction of Leucaena leucocephala and Sesbania grandiflora on the removal of turbidity
- Hydrocracking optimization of palm oil over NiMoO4/activated carbon catalyst to produce biogasoline and kerosine
- Special Issue on Pharmacology and metabolomics of ethnobotanical and herbal medicine
- Cynarin inhibits PDGF-BB-induced proliferation and activation in hepatic stellate cells through PPARγ
- Special Issue on The 1st Malaysia International Conference on Nanotechnology & Catalysis (MICNC2021)
- Surfactant evaluation for enhanced oil recovery: Phase behavior and interfacial tension
- Topical Issue on phytochemicals, biological and toxicological analysis of aromatic medicinal plants
- Phytochemical analysis of leaves and stems of Physalis alkekengi L. (Solanaceae)
- Phytochemical and pharmacological profiling of Trewia nudiflora Linn. leaf extract deciphers therapeutic potentials against thrombosis, arthritis, helminths, and insects
- Pergularia tomentosa coupled with selenium nanoparticles salvaged lead acetate-induced redox imbalance, inflammation, apoptosis, and disruption of neurotransmission in rats’ brain
- Protective effect of Allium atroviolaceum-synthesized SeNPs on aluminum-induced brain damage in mice
- Mechanism study of Cordyceps sinensis alleviates renal ischemia–reperfusion injury
- Plant-derived bisbenzylisoquinoline alkaloid tetrandrine prevents human podocyte injury by regulating the miR-150-5p/NPHS1 axis
- Network pharmacology combined with molecular docking to explore the anti-osteoporosis mechanisms of β-ecdysone derived from medicinal plants
- Chinese medicinal plant Polygonum cuspidatum ameliorates silicosis via suppressing the Wnt/β-catenin pathway
- Special Issue on Advanced Nanomaterials for Energy, Environmental and Biological Applications - Part I
- Investigation of improved optical and conductivity properties of poly(methyl methacrylate)–MXenes (PMMA–MXenes) nanocomposite thin films for optoelectronic applications
- Special Issue on Applied Biochemistry and Biotechnology (ABB 2022)
- Model predictive control for precision irrigation of a Quinoa crop
Articles in the same Issue
- Regular Articles
- Photocatalytic degradation of Rhodamine B in aqueous phase by bimetallic metal-organic framework M/Fe-MOF (M = Co, Cu, and Mg)
- Assessment of using electronic portal imaging device for analysing bolus material utilised in radiation therapy
- A detailed investigation on highly dense CuZr bulk metallic glasses for shielding purposes
- Simulation of gamma-ray shielding properties for materials of medical interest
- Environmental impact assesment regulation applications and their analysis in Turkey
- Sample age effect on parameters of dynamic nuclear polarization in certain difluorobenzen isomers/MC800 asphaltene suspensions
- Passenger demand forecasting for railway systems
- Design of a Robust sliding mode controller for bioreactor cultures in overflow metabolism via an interdisciplinary approach
- Gamma, neutron, and heavy charged ion shielding properties of Er3+-doped and Sm3+-doped zinc borate glasses
- Bridging chiral de-tert-butylcalix[4]arenes: Optical resolution based on column chromatography and structural characterization
- Petrology and geochemistry of multiphase post-granitic dikes: A case study from the Gabal Serbal area, Southwestern Sinai, Egypt
- Comparison of the yield and purity of plasma exosomes extracted by ultracentrifugation, precipitation, and membrane-based approaches
- Bioactive triterpenoids from Indonesian medicinal plant Syzygium aqueum
- Investigation of the effects of machining parameters on surface integrity in micromachining
- The mesoporous aluminosilicate application as support for bifunctional catalysts for n-hexadecane hydroconversion
- Gamma-ray shielding properties of Nd2O3-added iron–boron–phosphate-based composites
- Numerical investigation on perforated sheet metals under tension loading
- Statistical analysis on the radiological assessment and geochemical studies of granite rocks in the north of Um Taghir area, Eastern Desert, Egypt
- Two new polypodane-type bicyclic triterpenoids from mastic
- Structural, physical, and mechanical properties of the TiO2 added hydroxyapatite composites
- Tribological properties and characterization of borided Co–Mg alloys
- Studies on Anemone nemorosa L. extracts; polyphenols profile, antioxidant activity, and effects on Caco-2 cells by in vitro and in silico studies
- Mechanical properties, elastic moduli, transmission factors, and gamma-ray-shielding performances of Bi2O3–P2O5–B2O3–V2O5 quaternary glass system
- Cyclic connectivity index of bipolar fuzzy incidence graph
- The role of passage numbers of donor cells in the development of Arabian Oryx – Cow interspecific somatic cell nuclear transfer embryos
- Mechanical property evaluation of tellurite–germanate glasses and comparison of their radiation-shielding characteristics using EPICS2017 to other glass systems
- Molecular screening of ionic liquids for CO2 absorption and molecular dynamic simulation
- Microwave-assisted preparation of Ag/Fe magnetic biochar from clivia leaves for adsorbing daptomycin antibiotics
- Iminodisuccinic acid enhances antioxidant and mineral element accumulation in young leaves of Ziziphus jujuba
- Cytotoxic activity of guaiane-type sesquiterpene lactone (deoxycynaropicrin) isolated from the leaves of Centaurothamnus maximus
- Effects of welding parameters on the angular distortion of welded steel plates
- Simulation of a reactor considering the Stamicarbon, Snamprogetti, and Toyo patents for obtaining urea
- Effect of different ramie (Boehmeria nivea L. Gaud) cultivars on the adsorption of heavy metal ions cadmium and lead in the remediation of contaminated farmland soils
- Impact of a live bacterial-based direct-fed microbial (DFM) postpartum and weaning system on performance, mortality, and health of Najdi lambs
- Anti-tumor effect of liposomes containing extracted Murrayafoline A against liver cancer cells in 2D and 3D cultured models
- Physicochemical properties and some mineral concentration of milk samples from different animals and altitudes
- Copper(ii) complexes supported by modified azo-based ligands: Nucleic acid binding and molecular docking studies
- Diagnostic and therapeutic radioisotopes in nuclear medicine: Determination of gamma-ray transmission factors and safety competencies of high-dense and transparent glassy shields
- Calculation of NaI(Tl) detector efficiency using 226Ra, 232Th, and 40K radioisotopes: Three-phase Monte Carlo simulation study
- Isolation and identification of unstable components from Caesalpinia sappan by high-speed counter-current chromatography combined with preparative high-performance liquid chromatography
- Quantification of biomarkers and evaluation of antioxidant, anti-inflammatory, and cytotoxicity properties of Dodonaea viscosa grown in Saudi Arabia using HPTLC technique
- Characterization of the elastic modulus of ceramic–metal composites with physical and mechanical properties by ultrasonic technique
- GC-MS analysis of Vespa velutina auraria Smith and its anti-inflammatory and antioxidant activities in vitro
- Texturing of nanocoatings for surface acoustic wave-based sensors for volatile organic compounds
- Insights into the molecular basis of some chalcone analogues as potential inhibitors of Leishmania donovani: An integrated in silico and in vitro study
- (1R,2S,5R)-5-Methyl-2-(propan-2-yl)cyclohexyl 4-amino-3-phenylbutanoate hydrochloride: Synthesis and anticonvulsant activity
- On the relative extraction rates of colour compounds and caffeine during brewing, an investigation of tea over time and temperature
- Characterization of egg shell powder-doped ceramic–metal composites
- Rapeseed oil-based hippurate amide nanocomposite coating material for anticorrosive and antibacterial applications
- Chemically modified Teucrium polium (Lamiaceae) plant act as an effective adsorbent tool for potassium permanganate (KMnO4) in wastewater remediation
- Efficiency analysis of photovoltaic systems installed in different geographical locations
- Risk prioritization model driven by success factor in the light of multicriteria decision making
- Theoretical investigations on the excited-state intramolecular proton transfer in the solvated 2-hydroxy-1-naphthaldehyde carbohydrazone
- Mechanical and gamma-ray shielding examinations of Bi2O3–PbO–CdO–B2O3 glass system
- Machine learning-based forecasting of potability of drinking water through adaptive boosting model
- The potential effect of the Rumex vesicarius water seeds extract treatment on mice before and during pregnancy on the serum enzymes and the histology of kidney and liver
- Impact of benzimidazole functional groups on the n-doping properties of benzimidazole derivatives
- Extraction of red pigment from Chinese jujube peel and the antioxidant activity of the pigment extracts
- Flexural strength and thermal properties of carbon black nanoparticle reinforced epoxy composites obtained from waste tires
- A focusing study on radioprotective and antioxidant effects of Annona muricata leaf extract in the circulation and liver tissue: Clinical and experimental studies
- Clinical comprehensive and experimental assessment of the radioprotective effect of Annona muricata leaf extract to prevent cellular damage in the ileum tissue
- Effect of WC content on ultrasonic properties, thermal and electrical conductivity of WC–Co–Ni–Cr composites
- Influence of various class cleaning agents for prosthesis on Co–Cr alloy surface
- The synthesis of nanocellulose-based nanocomposites for the effective removal of hexavalent chromium ions from aqueous solution
- Study on the influence of physical interlayers on the remaining oil production under different development modes
- Optimized linear regression control of DC motor under various disturbances
- Influence of different sample preparation strategies on hypothesis-driven shotgun proteomic analysis of human saliva
- Determination of flow distance of the fluid metal due to fluidity in ductile iron casting by artificial neural networks approach
- Investigation of mechanical activation effect on high-volume natural pozzolanic cements
- In vitro: Anti-coccidia activity of Calotropis procera leaf extract on Eimeria papillata oocysts sporulation and sporozoite
- Determination of oil composition of cowpea (Vigna unguiculata L.) seeds under influence of organic fertilizer forms
- Activated partial thromboplastin time maybe associated with the prognosis of papillary thyroid carcinoma
- Treatment of rat brain ischemia model by NSCs-polymer scaffold transplantation
- Lead and cadmium removal with native yeast from coastal wetlands
- Characterization of electroless Ni-coated Fe–Co composite using powder metallurgy
- Ferrate synthesis using NaOCl and its application for dye removal
- Antioxidant, antidiabetic, and anticholinesterase potential of Chenopodium murale L. extracts using in vitro and in vivo approaches
- Study on essential oil, antioxidant activity, anti-human prostate cancer effects, and induction of apoptosis by Equisetum arvense
- Experimental study on turning machine with permanent magnetic cutting tool
- Numerical simulation and mathematical modeling of the casting process for pearlitic spheroidal graphite cast iron
- Design, synthesis, and cytotoxicity evaluation of novel thiophene, pyrimidine, pyridazine, and pyridine: Griseofulvin heterocyclic extension derivatives
- Isolation and identification of promising antibiotic-producing bacteria
- Ultrasonic-induced reversible blood–brain barrier opening: Safety evaluation into the cellular level
- Evaluation of phytochemical and antioxidant potential of various extracts from traditionally used medicinal plants of Pakistan
- Effect of calcium lactate in standard diet on selected markers of oxidative stress and inflammation in ovariectomized rats
- Identification of crucial salivary proteins/genes and pathways involved in pathogenesis of temporomandibular disorders
- Zirconium-modified attapulgite was used for removing of Cr(vi) in aqueous solution
- The stress distribution of different types of restorative materials in primary molar
- Reducing surface heat loss in steam boilers
- Deformation behavior and formability of friction stir processed DP600 steel
- Synthesis and characterization of bismuth oxide/commercial activated carbon composite for battery anode
- Phytochemical analysis of Ziziphus jujube leaf at different foliar ages based on widely targeted metabolomics
- Effects of in ovo injection of black cumin (Nigella sativa) extract on hatching performance of broiler eggs
- Separation and evaluation of potential antioxidant, analgesic, and anti-inflammatory activities of limonene-rich essential oils from Citrus sinensis (L.)
- Bioactivity of a polyhydroxy gorgostane steroid from Xenia umbellata
- BiCAM-based automated scoring system for digital logic circuit diagrams
- Analysis of standard systems with solar monitoring systems
- Structural and spectroscopic properties of voriconazole and fluconazole – Experimental and theoretical studies
- New plant resistance inducers based on polyamines
- Experimental investigation of single-lap bolted and bolted/bonded (hybrid) joints of polymeric plates
- Investigation of inlet air pressure and evaporative cooling of four different cogeneration cycles
- Review Articles
- Comprehensive review on synthesis, physicochemical properties, and application of activated carbon from the Arecaceae plants for enhanced wastewater treatment
- Research progress on speciation analysis of arsenic in traditional Chinese medicine
- Recent modified air-assisted liquid–liquid microextraction applications for medicines and organic compounds in various samples: A review
- An insight on Vietnamese bio-waste materials as activated carbon precursors for multiple applications in environmental protection
- Antimicrobial activities of the extracts and secondary metabolites from Clausena genus – A review
- Bioremediation of organic/heavy metal contaminants by mixed cultures of microorganisms: A review
- Sonodynamic therapy for breast cancer: A literature review
- Recent progress of amino acid transporters as a novel antitumor target
- Aconitum coreanum Rapaics: Botany, traditional uses, phytochemistry, pharmacology, and toxicology
- Corrigendum
- Corrigendum to “Petrology and geochemistry of multiphase post-granitic dikes: A case study from the Gabal Serbal area, Southwestern Sinai, Egypt”
- Corrigendum to “Design of a Robust sliding mode controller for bioreactor cultures in overflow metabolism via an interdisciplinary approach”
- Corrigendum to “Statistical analysis on the radiological assessment and geochemical studies of granite rocks in the north of Um Taghir area, Eastern Desert, Egypt”
- Corrigendum to “Aroma components of tobacco powder from different producing areas based on gas chromatography ion mobility spectrometry”
- Corrigendum to “Mechanical properties, elastic moduli, transmission factors, and gamma-ray-shielding performances of Bi2O3–P2O5–B2O3–V2O5 quaternary glass system”
- Erratum
- Erratum to “Copper(ii) complexes supported by modified azo-based ligands: Nucleic acid binding and molecular docking studies”
- Special Issue on Applied Biochemistry and Biotechnology (ABB 2021)
- Study of solidification and stabilization of heavy metals by passivators in heavy metal-contaminated soil
- Human health risk assessment and distribution of VOCs in a chemical site, Weinan, China
- Preparation and characterization of Sparassis latifolia β-glucan microcapsules
- Special Issue on the Conference of Energy, Fuels, Environment 2020
- Improving the thermal performance of existing buildings in light of the requirements of the EU directive 2010/31/EU in Poland
- Special Issue on Ethnobotanical, Phytochemical and Biological Investigation of Medicinal Plants
- Study of plant resources with ethnomedicinal relevance from district Bagh, Azad Jammu and Kashmir, Pakistan
- Studies on the chemical composition of plants used in traditional medicine in Congo
- Special Issue on Applied Chemistry in Agriculture and Food Science
- Strip spraying technology for precise herbicide application in carrot fields
- Special Issue on Pharmacology and Metabolomics of Ethnobotanical and Herbal Medicine
- Phytochemical profiling, antibacterial and antioxidant properties of Crocus sativus flower: A comparison between tepals and stigmas
- Antioxidant and antimicrobial properties of polyphenolics from Withania adpressa (Coss.) Batt. against selected drug-resistant bacterial strains
- Integrating network pharmacology and molecular docking to explore the potential mechanism of Xinguan No. 3 in the treatment of COVID-19
- Chemical composition and in vitro and in vivo biological assortment of fixed oil extracted from Ficus benghalensis L.
- A review of the pharmacological activities and protective effects of Inonotus obliquus triterpenoids in kidney diseases
- Ethnopharmacological study of medicinal plants in Kastamonu province (Türkiye)
- Protective effects of asperuloside against cyclophosphamide-induced urotoxicity and hematotoxicity in rats
- Special Issue on Essential Oil, Extraction, Phytochemistry, Advances, and Application
- Identification of volatile compounds and antioxidant, antibacterial, and antifungal properties against drug-resistant microbes of essential oils from the leaves of Mentha rotundifolia var. apodysa Briq. (Lamiaceae)
- Phenolic contents, anticancer, antioxidant, and antimicrobial capacities of MeOH extract from the aerial parts of Trema orientalis plant
- Chemical composition and antimicrobial activity of essential oils from Mentha pulegium and Rosmarinus officinalis against multidrug-resistant microbes and their acute toxicity study
- Special Issue on Marine Environmental Sciences and Significance of the Multidisciplinary Approaches
- An insightful overview of the distribution pattern of polycyclic aromatic hydrocarbon in the marine sediments of the Red Sea
- Antifungal–antiproliferative norcycloartane-type triterpenes from the Red Sea green alga Tydemania expeditionis
- Solvent effect, dipole moment, and DFT studies of multi donor–acceptor type pyridine derivative
- An extensive assessment on the distribution pattern of organic contaminants in the aerosols samples in the Middle East
- Special Issue on 4th IC3PE
- Energetics of carboxylic acid–pyridine heterosynthon revisited: A computational study of intermolecular hydrogen bond domination on phenylacetic acid–nicotinamide cocrystals
- A review: Silver–zinc oxide nanoparticles – organoclay-reinforced chitosan bionanocomposites for food packaging
- Green synthesis of magnetic activated carbon from peanut shells functionalized with TiO2 photocatalyst for Batik liquid waste treatment
- Coagulation activity of liquid extraction of Leucaena leucocephala and Sesbania grandiflora on the removal of turbidity
- Hydrocracking optimization of palm oil over NiMoO4/activated carbon catalyst to produce biogasoline and kerosine
- Special Issue on Pharmacology and metabolomics of ethnobotanical and herbal medicine
- Cynarin inhibits PDGF-BB-induced proliferation and activation in hepatic stellate cells through PPARγ
- Special Issue on The 1st Malaysia International Conference on Nanotechnology & Catalysis (MICNC2021)
- Surfactant evaluation for enhanced oil recovery: Phase behavior and interfacial tension
- Topical Issue on phytochemicals, biological and toxicological analysis of aromatic medicinal plants
- Phytochemical analysis of leaves and stems of Physalis alkekengi L. (Solanaceae)
- Phytochemical and pharmacological profiling of Trewia nudiflora Linn. leaf extract deciphers therapeutic potentials against thrombosis, arthritis, helminths, and insects
- Pergularia tomentosa coupled with selenium nanoparticles salvaged lead acetate-induced redox imbalance, inflammation, apoptosis, and disruption of neurotransmission in rats’ brain
- Protective effect of Allium atroviolaceum-synthesized SeNPs on aluminum-induced brain damage in mice
- Mechanism study of Cordyceps sinensis alleviates renal ischemia–reperfusion injury
- Plant-derived bisbenzylisoquinoline alkaloid tetrandrine prevents human podocyte injury by regulating the miR-150-5p/NPHS1 axis
- Network pharmacology combined with molecular docking to explore the anti-osteoporosis mechanisms of β-ecdysone derived from medicinal plants
- Chinese medicinal plant Polygonum cuspidatum ameliorates silicosis via suppressing the Wnt/β-catenin pathway
- Special Issue on Advanced Nanomaterials for Energy, Environmental and Biological Applications - Part I
- Investigation of improved optical and conductivity properties of poly(methyl methacrylate)–MXenes (PMMA–MXenes) nanocomposite thin films for optoelectronic applications
- Special Issue on Applied Biochemistry and Biotechnology (ABB 2022)
- Model predictive control for precision irrigation of a Quinoa crop

