Abstract
C15H12Br0.2Cl0.8N2O3Re1, monoclinic, P21/c (no. 14), a = 14.6713(4) Å, b = 11.4724(3) Å, c = 9.6206(3) Å, β = 106.592 (3), V = 1551.87(8) Å3, Z = 4, Rgt(F) = 0.0452, wRref(F2) = 0.1110, T = 149.99(10) K.
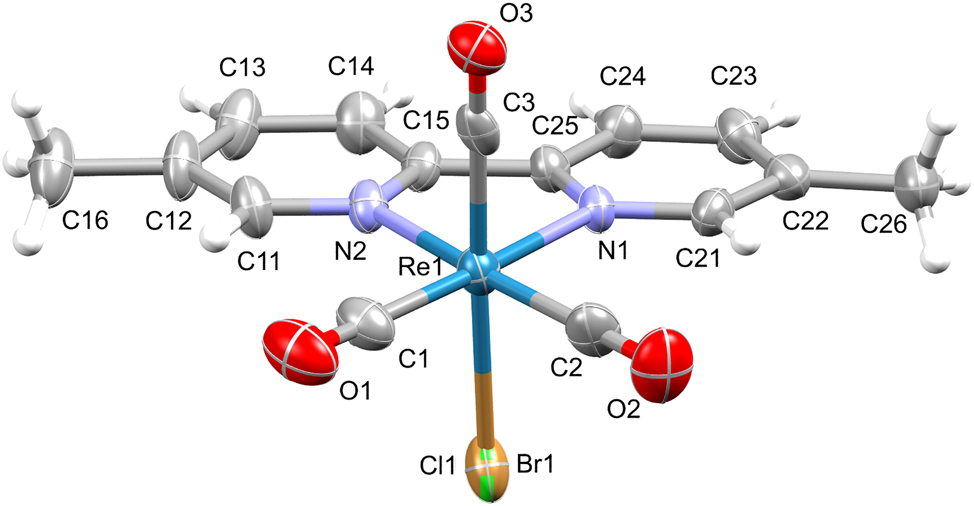
The molecular structure is shown in the figure. Table 1 contains crystallographic data and Table 2 contains the list of the atoms including atomic coordinates and displacement parameters.
Data collection and handling.
| Crystal: | Yellow block |
| Size: | 0.22 × 0.17 × 0.07 mm |
| Wavelength: | MoKα radiation (0.71073 Å) |
| μ: | 8.46 mm−1 |
| Diffractometer, scan mode: | XtaLAB Synergy R, ω |
| θmax, completeness: | 26.4°, >99 % |
| N(hkl)measured, N(hkl)unique, Rint: | 23,005, 3168, 0.078 |
| Criterion for Iobs, N(hkl)gt: | Iobs > 2 σ(Iobs), 2897 |
| N(param)refined: | 202 |
| Programs: | CrysAlisPro [1], Olex2 [2], WinGX [3], Shelx [4, 5] |
Fractional atomic coordinates and isotropic or equivalent isotropic displacement parameters (Å2).
| Atom | x | y | z | Uiso*/Ueq |
|---|---|---|---|---|
| Br1a | 0.65644 (13) | 0.53894 (17) | 0.9558 (2) | 0.0426 (8) |
| C1 | 0.7829 (7) | 0.7461 (9) | 0.8499 (11) | 0.042 (2) |
| C2 | 0.8801 (7) | 0.5910 (9) | 1.0348 (10) | 0.041 (2) |
| C3 | 0.9002 (6) | 0.6100 (7) | 0.7672 (9) | 0.0305 (19) |
| C11 | 0.6449 (6) | 0.6219 (8) | 0.5397 (11) | 0.037 (2) |
| H11 | 0.652842 | 0.701428 | 0.568228 | 0.044* |
| C12 | 0.5804 (7) | 0.5943 (9) | 0.4041 (11) | 0.043 (2) |
| C13 | 0.5696 (7) | 0.4785 (10) | 0.3664 (11) | 0.044 (3) |
| H13 | 0.526251 | 0.456159 | 0.276604 | 0.053* |
| C14 | 0.6222 (7) | 0.3930 (9) | 0.4599 (11) | 0.039 (2) |
| H14 | 0.616263 | 0.313091 | 0.433163 | 0.046* |
| C15 | 0.6833 (6) | 0.4276 (7) | 0.5925 (9) | 0.0281 (17) |
| C16 | 0.5270 (7) | 0.6895 (10) | 0.3069 (13) | 0.054 (3) |
| H16A | 0.505978 | 0.747948 | 0.365444 | 0.082* |
| H16B | 0.471405 | 0.656210 | 0.235714 | 0.082* |
| H16C | 0.568669 | 0.726326 | 0.256323 | 0.082* |
| C21 | 0.8566 (6) | 0.3210 (8) | 0.9190 (9) | 0.0329 (19) |
| H21 | 0.898313 | 0.355021 | 1.003397 | 0.039* |
| C22 | 0.8566 (7) | 0.2007 (8) | 0.9061 (10) | 0.034 (2) |
| C23 | 0.7947 (7) | 0.1515 (8) | 0.7810 (11) | 0.037 (2) |
| H23 | 0.791943 | 0.069321 | 0.767979 | 0.045* |
| C24 | 0.7379 (7) | 0.2236 (8) | 0.6772 (10) | 0.035 (2) |
| H24 | 0.696326 | 0.191307 | 0.591452 | 0.041* |
| C25 | 0.7417 (6) | 0.3438 (8) | 0.6981 (9) | 0.0282 (17) |
| C26 | 0.9212 (8) | 0.1282 (10) | 1.0244 (11) | 0.047 (3) |
| H26A | 0.986241 | 0.131901 | 1.016350 | 0.070* |
| H26B | 0.899333 | 0.047064 | 1.014601 | 0.070* |
| H26C | 0.920093 | 0.158389 | 1.119175 | 0.070* |
| Cl1b | 0.65644 (13) | 0.53894 (17) | 0.9558 (2) | 0.0426 (8) |
| N1 | 0.8012 (5) | 0.3927 (6) | 0.8196 (7) | 0.0263 (15) |
| N2 | 0.6953 (5) | 0.5412 (6) | 0.6300 (8) | 0.0298 (16) |
| O1 | 0.7824 (6) | 0.8488 (8) | 0.8583 (11) | 0.067 (2) |
| O2 | 0.9306 (6) | 0.5973 (7) | 1.1505 (8) | 0.060 (2) |
| O3 | 0.9638 (5) | 0.6324 (6) | 0.7276 (8) | 0.0437 (16) |
| Re1 | 0.79487 (2) | 0.57988 (3) | 0.84168 (4) | 0.02986 (14) |
-
aOccupancy: 0.184 (8), bOccupancy: 0.816 (8).
1 Source of materials
The synthesis of the starting synthon, fac-[NEt4]2[Re(CO)3(Br)3], was strictly performed under Schlenk conditions. The reported complex was synthesized according to published procedures [6, 7]. To fac-[NEt4]2[Re(CO)3(Br)3] (251 mg, 0.325 mmol) in methanol 5,5′-dimethyl- 2,2′-bipyridyl (bid; 119 mg, 0.648 mmol) was added and refluxed for 6 h at room temperature. The light-yellow precipitate was filtered off, dried, and weighed to yield the title complex. Crystals were obtained by recrystallization using warm methanol and dilute HCl solution and allowing the resultant solution to crystallize at room temperature. The molecular structure of fac-[Re(CO)3(5,5′-DiMBpy)(Cl0.82Br0.18)] was confirmed with SCXRD. Yield: 289 mg, IR (ATR, cm−1): vCO = 2024.8, 1900.8.
2 Experimental details
The aromatic and methyl H atoms were place in geometrically idealized positions and constrained to ride on their parent atoms, with C–H = 0.93 (aromatic) and 0.96 Ang (methyl) and their Uiso values were set to 1.2Ueq (aromatic) and 1.5Ueq (methyl). The highest peak is 2.82 eAng−3 and deepest hole is −1.04 eAng−3. The graphics were generated using the Mercury program with 50 % probability ellipsoids.
3 Comment
In the previous five years, the chemistry of the relatively inert and low oxidation state rhenium(I) (Re) and technetium(I) (Tc) was derived from the fac-[M(CO)3(H2O)3]+ precursor. This precursor has been studied and employed by numerous research groups, mainly for their significance in the nuclear medicine arena, such as radiopharmacy, photocatalysis, and currently, chemotherapy [7, 8]. Furthermore, the fac-[Re(H2O)3(CO)3]+ precursor is very useful for designing molecules with different properties and applying various coordination models such as the “2 + 1” and most recently introduced “2 + 1 + 1” mixed ligand approach [9]. The various coordination models, enables a wide range of rhenium(I) tricarbonyl complexes to be obtained by displacing the three labile water ligands with alternative ligands [8]. Specifically, rhenium(I) tricarbonyl diimine complexes, [(N,N′)Re(CO)3(X)] (X = halide), exhibit remarkable photophysical and photochemical properties, which can be altered by either the nature of diimine ligand (N,N′) or the L ligand itself (Cl, Br) as indicated by Wrighton and co-workers in the 1970s [10, 11]. These rhenium(I) complexes with their associated properties, are often applied in anti-microbial, anti-inflammatory, anti-viral and anti-tumor studies [12], [13], [14], [15], [16].
The title structure is a rhenium(I) tricarbonyl complex with a bid ligand and a mixed halogen (Cl0.82Br0.12) ligand. The rhenium metal center is fulfilled by a bid bidentate ligand coordinated equatorially, three facially coordinated carbonyl ligands, and a halogen (Cl/Br) ligand on the axial position producing a distorted octahedral geometry. The complex has associated bond angles of 101.3(4)° (C1–Re1–N2), 97.3(3)° (C2–Re1–N1), 94.9(3)° (C3–Re1–N2), and 75.1(3)° (N1–Re1–N2), and are comparable to the same angles of similar structures previously reported [17]. The Re–C bond distances (Re1–C1, Re1–C2, and Re1–C3) are 1.919(10), 1.925(10), and 1.911(10) Å, respectively, and they are comparable to similar reported structures in literature [18], [19], [20], [21]. Furthermore, the rhenium to nitrogen bond distances is 2.163(7) Å (Re1–N1) and 2.186(7) Å (Re1–N2), respectively, again comparable to reported structures [22], [23], [24], [25], [26]. The rhenium to halogen bond distance of 2.612 Å(Re1–Cl1/Br1) is longer compared to Re–Cl bond distances in literature [27], while comparable to Re–Br bond distances which appear in the range of 2.59–2.64 Å of similar structures in literature [28]. Thus, the two atoms (Cl and Br) at the axial position appear as a substitutional disorder and has been treated as such to refine using a free variable (final occupancy: Cl1 = 0.816(8); Br1 = 0.184(8)) where Cl1/Br1 have been constrained to occupy the same position.
Funding source: National Research Foundation of South Africa
Award Identifier / Grant number: 129468
Funding source: Tshwane University of Technology
Funding source: University of Pretoria
Funding source: University of the Free State
-
Author contributions: All the authors have accepted responsibility for the entire content of this submitted manuscript and approved submission.
-
Research funding: This work was funded by National Research Foundation of South Africa (Grant No. 129468), Tshwane University of Technology, the University of Pretoria and the University of the Free State.
-
Conflict of interest statement: The authors declare no conflicts of interest.
References
1. System C. S. CrysAlisPro Software System; Rigaku Oxford Diffraction: Yarnton, UK, 2021.Search in Google Scholar
2. Dolomanov, O. V., Bourhis, L. J., Gildea, R. J., Howard, J. A. K., Puschmann, H. OLEX2: a complete structure solution, refinement and analysis program. J. Appl. Crystallogr. 2009, 42, 339–341; https://doi.org/10.1107/s0021889808042726.Search in Google Scholar
3. Farrugia, L. J. WinGX suite for small-molecule single-crystal crystallography. J. Appl. Crystallogr. 1999, 32, 837–883; https://doi.org/10.1107/s0021889899006020.Search in Google Scholar
4. Sheldrick, G. M. Crystal structure refinement with SHELXL. Acta Crystallogr. 2015, C71, 3–8; https://doi.org/10.1107/s2053229614024218.Search in Google Scholar
5. Sheldrick, G. M. SHELXTL – integrated space-group and crystal-structure determination. Acta Crystallogr. 2015, A71, 3–8; https://doi.org/10.1107/s2053273314026370.Search in Google Scholar
6. Manicum, A., Schutte-Smith, M., Visser, H. G. The synthesis and structural comparison of fac-[Re(CO)3]+ containing complexes with altered β-diketone and phosphine ligands. Polyhedron 2018, 145, 80–87; https://doi.org/10.1016/j.poly.2018.01.022.Search in Google Scholar
7. Manicum, A., Alexander, O., Schutte-Smith, M., Visser, H. G. Synthesis, characterization and substitution reactions of fac-[Re(O,O- bid)(CO)3(P)] complexes, using the “2 + 1” mixed ligand model. J. Mol. Struct. 2020, 1209, 127953–127964; https://doi.org/10.1016/j.molstruc.2020.127953.Search in Google Scholar
8. Schutte-Smith, M., Marker, S. C., Wilson, J. J., Visser, H. G. Aquation and anation kinetics of rhenium(I) dicarbonyl complexes: relation to cell toxicity and bioavailability. Inorg. Chem. 2020, 59, 15888–15897; https://doi.org/10.1021/acs.inorgchem.0c02389.Search in Google Scholar PubMed
9. Manicum, A., Schutte-Smith, M., Malan, F. P., Visser, H. G. Steric and electronic influence of Re(I) tricarbonyl complexes with various coordinated β-diketones. J. Mol. Struct. 2022, 1264, 133278–133291; https://doi.org/10.1016/j.molstruc.2022.133278.Search in Google Scholar
10. Gallardo, H., Cepeda-Plaza, M., Nonell, S., Günther, G., Chamorro, E., Pizarro, N., Vega, A. Structural and photophysical properties of [(CO)3(phen)Re(l–Br)Re(phen)(CO)3] + [(CO)3Re(l–Br)3Re(CO)3]−: where does its luminescence come from? Polyhedron 2015, 97, 227–233; https://doi.org/10.1016/j.poly.2015.05.024.Search in Google Scholar
11. Kumar, A., Sun, S.-S., Lees, A. J. Photophysics and photochemistry of organometallic rhenium diimine complexes. Top. Organomet. Chem. 2010, 29, 1–35; https://doi.org/10.4103/0976-9668.71662.Search in Google Scholar PubMed PubMed Central
12. Kapp, L. E., Schutte-Smith, M., Twigge, L., Visser, H. G. Synthesis, characterization, and DNA binding of four imidazo[4,5-f]1,10-phenanthroline derivatives. J. Mol. Struct. 2022, 1247, 131235–131245; https://doi.org/10.1016/j.molstruc.2021.131235.Search in Google Scholar
13. Mkhatshwa, M., Moremi, J. M., Makgopa, K., Manicum, A. Nanoparticle functionalized with Re(I) tricarbonyl complexes for cancer theranostics. Int. J. Mol. Sci. 2021, 22, 6546–6563; https://doi.org/10.3390/ijms22126546.Search in Google Scholar PubMed PubMed Central
14. Matlou, M. L., Louis, H., Charlie, D. E., Agwamba, E. C., Amodu, I. O., Tembu, V. J., Manicum, A. Anticancer activities of Re(I) tricarbonyl and its imidazole-based ligands: insight from a theoretical approach. ACS Omega 2023, 8, 10242–10252; https://doi.org/10.1021/acsomega.2c07779.Search in Google Scholar PubMed PubMed Central
15. Moherane, L., Louis, H., Ekereke, E. E., Agwamba, E. C., Visser, H. G., Benjamin, I., Uwem, O., Edet, U. O., Manicum, A. Polypyridyl coordinated Re(I) complexes for human tenascin-C (TNC) as an antibreast cancer agent: an intuition from molecular modeling and simulations. Polycyclic Aromat. Compd. 2023, 1–18; https://doi.org/10.1080/10406638.2023.2189737.Search in Google Scholar
16. Manicum, A., Louis, H., Agwamba, E. C., Chima, C. M., Nzondomyo, W. J., Sithole, S. A. Acetylacetone and imidazole coordinated Re(I) tricarbonyl complexes: experimental, DFT studies, and molecular docking approach. Chem. Phys. Impact 2023, 6, 100165–100178; https://doi.org/10.1016/j.chphi.2023.100165.Search in Google Scholar
17. Moherane, L., Alexander, O. T., Schutte-Smith, M., Kroon, R. E., Mokolokolo, P. P., Biswas, S., Prince, S., Visser, H. G., Manicum, A. E. Polypyridyl coordinated rhenium(I) tricarbonyl complexes as model devices for cancer diagnosis and treatment. Polyhedron 2022, 228, 116178–116188; https://doi.org/10.1016/j.poly.2022.116178.Search in Google Scholar
18. Manicum, A. E., Schutte-Smith, M., Alexander, O. T., Twigge, L., Roodt, A., Visser, H. G. First kinetic data of the CO substitution in fac-[Re(L,L′-Bid)(CO)3(X)] complexes (L,L′-Bid = acacetylacetonate or tropolonate) by tertiary phosphines PTA and PPh3: synthesis and crystal structures of water-soluble rhenium(I) tri- and dicarbonyl complexes with 1,3,5-triaza-7- phosphaadamantane (PTA). Inorg. Chem. Commun. 2019, 101, 93–98; https://doi.org/10.1016/j.inoche.2019.01.014.Search in Google Scholar
19. Moherane, L., Alexander, O. T., Visser, H. G., Manicum, A. E. The crystal structure of [μ-hydroxido-bis[(5,5′-dimethyl-2,2′-bipyridine-κ2N,N′)- tricarbonylrhenium(I)] bromide hemihydrate, C30H26N4O9Re2Br. Z. Kristallogr. N. Cryst. Struct. 2021, 236, 1027–1029; https://doi.org/10.1515/ncrs-2021-0210.Search in Google Scholar
20. Makhakhayi, L., Malan, F. P., Tembu, V. J., Nkambule, C. M., Manicum, A. E. The crystal structure of fac-tricarbonyl(N-benzoyl-N,N- cyclohexylmethylcarbamimidothioato-κ2S,O)-(pyridine-κN)rhenium(I), C23H24N3O4ReS. Z. Kristallogr. N. Cryst. Struct. 2023, 238, 697–699; https://doi.org/10.1515/ncrs-2023-0157.Search in Google Scholar
21. Schutte-Smith, M., Visser, H. G. Crystal and molecular structures of fac-[Re(Bid)- (PPh3)(CO)3] [Bid is tropolone (TropH) and tribromotropolone (TropBr3H)]. Acta Crystallogr. 2022, C78, 351–359; https://doi.org/10.1107/s205322962200465x.Search in Google Scholar
22. Schutte-Smith, M., Roodt, A., Alberto, R., Twigge, L., Visser, H. G., Kirstena, L., Koen, R. Structures of rhenium(I) complexes with 3-hydroxyflavone and benzhydroxamic acid as O,O′- bidentate ligands and confirmation of p-stacking by solid-state NMR spectroscopy. Acta Crystallogr. 2019, C75, 378–387; https://doi.org/10.1107/s2053229619002717.Search in Google Scholar PubMed
23. Matlou, M. L., Malan, F. P., Nkadimeng, S., Mcgaw, L., Tembu, V. J., Manicum, A. E. Exploring the in vitro anticancer activities of Re(I) picolinic acid and its fluorinated complex derivatives on lung cancer cells: a structural study. J. Biol. Inorg. Chem. 2023, 28, 29–41; https://doi.org/10.1007/s00775-022-01971-2.Search in Google Scholar PubMed
24. Sithole, S. A., Malan, F. P., Katerere, D. R., Manicum, A. E. The crystal structure of fac-tricarbonyl(2-pyridin-2-yl-quinoline-κ2N,N′)- (pyrazole-κN)rhenium(I)nitrate, C20H14N4O3ReNO3. Z. Kristallogr. N. Cryst. Struct. 2023, 238, 685–687; https://doi.org/10.1515/ncrs-2023-0152.Search in Google Scholar
25. Mkhatshwa, M., Malan, F. P., Makgopa, K., Manicum, A. E. The crystal structure of fac-tricarbonyl(6-bromo-2,2-bipyridine-κ2N,N)- (nitrato-κO)rhenium(I), C13H7BrN3O6Re. Z. Kristallogr. N. Cryst. Struct. 2023, 238, 667–669; https://doi.org/10.1515/ncrs-2023-0141.Search in Google Scholar
26. Moremi, M. J., Alexander, O. T., Vatsha, B., Makgopa, K., Manicum, A. E. The crystal structure of fac-tricarbonyl(4,4-dimethyl-2,2-dipyridyl-κ2N,N′)- (pyrazole-κN)rhenium(I) nitrate, C18H16O3N4Re. Z. Kristallogr. N. Cryst. Struct. 2021, 236, 33–35; https://doi.org/10.1515/ncrs-2020-0458.Search in Google Scholar
27. Chabolla, S. A., Dellamary, E. A., Machan, C. W., Tezcan, F. A., Kubiak, C. P. Combined steric and electronic effects of positional substitution on dimethyl-bipyridine rhenium(I)tricarbonyl electrocatalysts for the reduction of CO2. Inorg. Chim. Acta 2014, 422, 109–113; https://doi.org/10.1016/j.ica.2014.07.007.Search in Google Scholar
28. Heard, P. J., Sroisuwan, P., Tocher, D. A. Synthesis and reactivity of N,N,N′,N′-tetramethyldiaminomethane complexes of tricarbonylrhenium(I). X-ray molecular structures of [ReBr(CO)3(TMDM)] and [{Re(bipy)(CO)3}2(μ-OH)] [SbF6]. Polyhedron 2003, 22, 1321–1327; https://doi.org/10.1016/s0277-5387(03)00102-5.Search in Google Scholar
© 2023 the author(s), published by De Gruyter, Berlin/Boston
This work is licensed under the Creative Commons Attribution 4.0 International License.
Articles in the same Issue
- Frontmatter
- New Crystal Structures
- The crystal structure of (N-([1,1′:4′,1″-terphenyl]-4,4′-diethyl)-2-(bis(pyridin-2-ylmethyl)amino)acetamide-κ4N,N,N″, O)tri(nitrato-kO, O′) samarium(III) - methanol - acetonitrile (1/1/1), C40H39SmN8O14
- The crystal structure of 6,6′-(((2-(dimethylamino)ethyl)azanediyl)bis(methylene))bis(2-chloro-4-methyl phenolate-κ4N,N′,O,O′)-(pyridine-2,6-dicarboxylato-N,O,O′)-titanium(IV), C27H27Cl2N3O6Ti
- N′-[(1E)-(4–Fluorophenyl)methylidene]adamantane-1-carbohydrazide, C18H21FN2O
- Crystal structure of 4-bromo-3-nitro-1H-pyrazole-5-carboxylic acid monohydrate, C4H2N3BrO4·H2O
- Crystal structure of dipyridine-k1N-tris(2,2,6,6-tetramethyl-5-oxohept-3-en-3-olato-k2O,O′)dysprosium(III), DyC43H67O6N2
- Crystal structure of cyclo[tetraiodido-bis{μ2-1-[(benzotriazol-1-yl)methyl]-1-H-1,3-(2-isopropyl-imidazol)-k2N:N}dicadmiun(II)], C26H30N10Cd2I4
- The crystal structure of tert-butyl (E)-3-(2-(benzylideneamino)phenyl)-1H-indole-1-carboxylate, C26H24N2O2
- The crystal structure of 4-(3-carboxy-1-cyclopropyl-6-fluoro-8-methoxy-4-oxo-1,4- dihydroquinolin-7-yl)-2-methylpiperazin-1-ium 2,5-dihydroxybenzoate methanol solvate, C27H32FN3O9
- Crystal structure of (μ2-1-(4,4′-bipyridine-κ2N:N′)-bis[diaqua-(4-iodopyridine-2,6-dicarboxylato-κ3O,N,O′)–cobalt(II)], C24H20Co2I2N4O12
- The crystal structure of dimethyl 4,4′-(10,20-diphenylporphyrin-5,15-diyl)dibenzoate dichloromethane solvate, C49H36N4O4Cl2
- (E)-2-((E)-4-(2,6,6-trimethylcyclohex-1-en-1-yl)but-3-en-2-ylidene)hydrazine-1-carbothioamide C14H23N3S1
- The crystal structure of [1-(4-(trifluoromethyl)phenyl)-3,4-dihydroquinolin-2(1H)-one], C16H12F3NO
- Crystal structure of (E)-2-amino-N′-((3-hydroxy-5-(hydroxymethyl)-2-methylpyridin-4-yl)methylene)benzohydrazide – dimethylformamide – water (1/1/2), C15H16N4O3·C3H7NO·2H2O
- Crystal structure of 3-(4-bromophenyl)-5-methyl-1H-pyrazole, C10H9BrN2
- Crystal structure of 1,10-phenanthrolinium bromide dihydrate, C12H9N2Br
- Crystal structure of N-(4′-chloro-[1,1′-biphenyl]-2-yl)formamide, C13H10ClNO
- The crystal structure of nitroterephthalic acid, C8H5NO6
- Crystal structure of (2-((4-bromo-2,6-dichlorophenyl)amino)phenyl) (morpholino)methanone, C17H15BrCl2N2O2
- Crystal structure of tetraaqua-bis(ethanol-κO)-tetrakis(μ2-trifluoroacetate-κ2O:O′)-bis(trifluoroacetate-κ2O)digadolinium(III) Gd2C16H20O18F18
- The crystal structure of dimethyl 4,4′-[10,20-bis(2,6-difluorophenyl)porphyrin-5,15-diyl]dibenzoate chloroform solvate, C50H32Cl6F4N4O4
- The crystal structure of N,N′-((nitroazanediyl)bis(methylene))diacetamide, C6H12O4N4
- The crystal structure of [bis(2,2′-bipyridine-6-carboxylato-κ3N,N,O)magnesium(II)]dihydrate, C22H18N4O6Mg
- Crystal structure of poly[diaqua-(bis(μ2-1,4-bis(imidazol-1-ylmethyl)benzene)-κ2N,N′] cobalt(II)-tetraqua-bis(1,4-bis(imidazol-1-ylmethyl)benzene)-κ1N)-cobalt(II) di(2,5-thiophenedicarboxylate) dihydrate, C68H76Co2N16O16S2
- Crystal structure of poly[chlorido-μ2-chlorido-(μ2-1-[(2-ethyl-4-methyl-1H-imidazol-1-yl)methyl]-1H-benzotriazole-κN:N’)cadmium(II)], C13H15CdN5Cl2
- The crystal structure of (4-hydroxybenzenesulfonate)-k1O-6,6′-((1E,1′E)- (ethane-1,2-diylbis(azaneylylidene))bis(methaneylylidene)) bis(2-methoxyphenol)-κ2N,N,μ2O,O,κ2O, O)-(methanol)-cobalt(II) sodium(I), C25H27CoN2NaO9S
- Crystal structure of (1-methyl-3-(trifluoromethyl)-1H-pyrazol-4-yl)(4-((2-methyl-6-(trifluoromethyl)pyrimidin-4-yl)amino)piperidin-1-yl)methanone, C17H18F6N6O
- Crystal structure of bis{[(cyclohexylimino)(phenylimino)-l5-(methyl)diethylazane-κ2N:N′]-(ethyl)-zinc(II)]}, C38H62N6Zn2
- Crystal structure of 2-[(4-bromobenzyl)thio]-5-(5-bromothiophen-2-yl)-1,3,4-oxadiazole, C13H8Br2N2OS2
- Crystal structure of 10-methoxy-7,11b,12,13-tetrahydro-6H-pyrazino [2′,3′:5,6]pyrazino[2,1-a]isoquinoline, C15H16N4O
- The crystal structure of 1-propyl-2-nitro-imidazole oxide, C6H9N3O3
- The crystal structure of 3-nitrobenzene-1,2-dicarboxylic acid–2-ethoxybenzamide (1/1), C17H16N2O8
- The structure of RUB-1, (C8H16N)6[B6Si48O108], a boron containing levyne-type zeolite, occluding N-methyl-quinuclidinium in the cage-like pores
- The crystal structure of diaqua-(naphthalene-4,5-dicarboxylate-1,8-dicarboxylic anhydride-κ1O)-(4′-(4-(1H-benzimidazolyl-1-yl)phenyl)-2,2′:6′,2″-terpyridine-κ3N,N′,N″)–manganese(II) dihydrate, C42H27MnN5O9·2H2O
- Crystal structure of 6,6′-((1E,1′E)-hydrazine-1,2-diylidenebis(methanylylidene))bis (3-(3-bromopropoxy)phenol), C20H22Br2N2O4
- The crystal structure of 3-(2-hydroxyphenyl)-4-phenyl-6-(p-tolyl)-2H-pyran-2-one, C24H18O3
- Crystal structure of bis(μ2-2-(1,5-dimethyl–3-oxo-2-phenyl-2,3-dihydro-1H-pyrazol-4-yl)imino)methyl)phenolato-κ4O:O,N,O′)-(nitrato-κ2O,O′)dicobalt(II), C36H32Co2N8O4
- Synthesis and crystal structure of (3E,5S,10S,13S,14S,17Z)-17-ethylidene-10,13-dimethylhexadecahydro-3H-cyclopenta[α] phenanthren-3-one O-(4-fluorobenzoyl) oxime, C28H36FNO2
- The crystal structure of 4-aminiumbiphenyl benzenesulfonate, C18H17NO3S
- Synthesis and crystal structure of 1-(7-hydroxy-3-(4-hydroxy-3-nitrophenyl)-4-oxo-4H-chromen-8-yl)-N,N-dimethylmethanaminiumnitrate, C18H17N3O9
- Crystal structure of N-(Ar)-N′-(Ar′)-formamidine, C14H12Br2N2O
- The crystal structure of 4-(2,4-dichlorophenyl)-2-(4-fluorophenyl)-5-methyl-1H-imidazole, C16H11Cl2FN2
- Crystal structure of 1-(4–chlorophenyl)-4-benzoyl-3-methyl-1H-pyrazol-5-ol, C17H13ClN2O2
- The crystal structure of 5-amino-1-methyl-4-nitroimidazole, C4H6O2N4
- Crystal structure of 1,3-diisopropyl-4,5-dimethylimidazol-2-ylidene-N,N′-bis(1,3-bis(2,6-diisopropylphenyl)-1,3-dihydro-2H-1,3,2-diazaborol-2-yl)-l2-germenediamine, C63H94B2GeN8
- The crystal structure of (bromido, chlorido)-tricarbonyl-(5,5′-dimethyl-2,2′-bipyridine)-rhenium(I), C15H12Br0.2Cl0.8N2O3Re1
- Crystal structure of [N(E),N′(E)]-N,N′-(1,4-phenylenedimethylidyne)bis-3,5-bis(propan-2-yl)-1H-pyrazol-4-amine, C26H36N6
- The crystal structure of poly[2-(4-carboxypyridin-3-yl)terephthalpoly[diaqua-(μ4-2-(6-carboxylatopyridin-3-yl)terephthalato-κ5O,N:O′:O″,O‴)]) cadmium(II)] dihydrate, C28H20Cd3N2O16
- Crystal structure of [tetraaqua-bis((3-carboxy-5-(pyridin-4-yl)benzoate-κ1N)cobalt(II)] tetrahydrate, C26H32CoN2O16
- Crystal structure of bis(μ2-azido-κ2N:N)-tetrakis(azido-κ1N)-tetrakis(1,10-phenanthroline-κ2N,N′)dibismuth(III), C48H32N26Bi2
- Crystal structure of (Z)-N-(4-(4-(4-((4,5,6-trimethoxy-3-oxobenzofuran-2(3H)-ylidene)methyl)phenoxy)butoxy)phenyl)acetamide, C30H31NO8
- Crystal structure of poly[diaqua-(μ2-1,3-di(1H-imidazol-1-yl)propane-κ2N:N′)-bis(μ2-5-carboxybenzene-1,3-dicarboxylato-O,O′:O″)-aqua-di-zinc dihydrate solvate], C27H28N4O16Zn2
- Crystal structure of 2-(3,5,5-trimethylcyclohex-2-en-1-ylidene)malononitrile, C12H14N2
- Crystal structure of chlorido-(5-nitro-2-phenylpyridine-κ2N,C)-[(methylsulfinyl)methane-κ1S]platinum(II), C13H13ClN2O3PtS
- The crystal structure of the co-crystal 1,4-dioxane–4,6-bis(nitroimino)-1,3,5-triazinan-2-one(2/1), C11H19N7O9
- Crystal structure of [N(E),N′(E)]-N,N′-(1,4-phenylenedimethylidyne)bis-3,5-dimethyl-1H-pyrazol-4-amine di-methanol solvate, C18H20N6·2(CH3OH)
- Crystal structure of catena-poly[bis(μ2-azido-k2N:N′)-(nitrato-K2N:N′)-bis(1,10-phenanthroline-K2N:N′)samarium(III)], C24H16N11O3Sm
- Crystal structure of (Z)-2-(4-((5-bromopentyl)oxy)benzylidene)-4,5,6-trimethoxybenzofuran-3(2H)-one, C23H25BrO6
- Crystal structure of bis(3,5-dimethyl-1H-pyrazol-4-ammonium) tetrafluoroterephthate, 2[C5H10N3][C8F4O4]
- Crystal structure of 2-amino-4-(2-fluoro-4-(trifluoromethyl)phenyl)-9-methoxy-1,4,5,6-tetrahydrobenzo[h]quinazolin-3-ium chloride, C20H18ClF4N3O
- Crystal structure of 6-(pyridin-3-yl)-1,3,5-triazine-2,4-diamine-sebacic acid (2/1), C13H17N6O2
Articles in the same Issue
- Frontmatter
- New Crystal Structures
- The crystal structure of (N-([1,1′:4′,1″-terphenyl]-4,4′-diethyl)-2-(bis(pyridin-2-ylmethyl)amino)acetamide-κ4N,N,N″, O)tri(nitrato-kO, O′) samarium(III) - methanol - acetonitrile (1/1/1), C40H39SmN8O14
- The crystal structure of 6,6′-(((2-(dimethylamino)ethyl)azanediyl)bis(methylene))bis(2-chloro-4-methyl phenolate-κ4N,N′,O,O′)-(pyridine-2,6-dicarboxylato-N,O,O′)-titanium(IV), C27H27Cl2N3O6Ti
- N′-[(1E)-(4–Fluorophenyl)methylidene]adamantane-1-carbohydrazide, C18H21FN2O
- Crystal structure of 4-bromo-3-nitro-1H-pyrazole-5-carboxylic acid monohydrate, C4H2N3BrO4·H2O
- Crystal structure of dipyridine-k1N-tris(2,2,6,6-tetramethyl-5-oxohept-3-en-3-olato-k2O,O′)dysprosium(III), DyC43H67O6N2
- Crystal structure of cyclo[tetraiodido-bis{μ2-1-[(benzotriazol-1-yl)methyl]-1-H-1,3-(2-isopropyl-imidazol)-k2N:N}dicadmiun(II)], C26H30N10Cd2I4
- The crystal structure of tert-butyl (E)-3-(2-(benzylideneamino)phenyl)-1H-indole-1-carboxylate, C26H24N2O2
- The crystal structure of 4-(3-carboxy-1-cyclopropyl-6-fluoro-8-methoxy-4-oxo-1,4- dihydroquinolin-7-yl)-2-methylpiperazin-1-ium 2,5-dihydroxybenzoate methanol solvate, C27H32FN3O9
- Crystal structure of (μ2-1-(4,4′-bipyridine-κ2N:N′)-bis[diaqua-(4-iodopyridine-2,6-dicarboxylato-κ3O,N,O′)–cobalt(II)], C24H20Co2I2N4O12
- The crystal structure of dimethyl 4,4′-(10,20-diphenylporphyrin-5,15-diyl)dibenzoate dichloromethane solvate, C49H36N4O4Cl2
- (E)-2-((E)-4-(2,6,6-trimethylcyclohex-1-en-1-yl)but-3-en-2-ylidene)hydrazine-1-carbothioamide C14H23N3S1
- The crystal structure of [1-(4-(trifluoromethyl)phenyl)-3,4-dihydroquinolin-2(1H)-one], C16H12F3NO
- Crystal structure of (E)-2-amino-N′-((3-hydroxy-5-(hydroxymethyl)-2-methylpyridin-4-yl)methylene)benzohydrazide – dimethylformamide – water (1/1/2), C15H16N4O3·C3H7NO·2H2O
- Crystal structure of 3-(4-bromophenyl)-5-methyl-1H-pyrazole, C10H9BrN2
- Crystal structure of 1,10-phenanthrolinium bromide dihydrate, C12H9N2Br
- Crystal structure of N-(4′-chloro-[1,1′-biphenyl]-2-yl)formamide, C13H10ClNO
- The crystal structure of nitroterephthalic acid, C8H5NO6
- Crystal structure of (2-((4-bromo-2,6-dichlorophenyl)amino)phenyl) (morpholino)methanone, C17H15BrCl2N2O2
- Crystal structure of tetraaqua-bis(ethanol-κO)-tetrakis(μ2-trifluoroacetate-κ2O:O′)-bis(trifluoroacetate-κ2O)digadolinium(III) Gd2C16H20O18F18
- The crystal structure of dimethyl 4,4′-[10,20-bis(2,6-difluorophenyl)porphyrin-5,15-diyl]dibenzoate chloroform solvate, C50H32Cl6F4N4O4
- The crystal structure of N,N′-((nitroazanediyl)bis(methylene))diacetamide, C6H12O4N4
- The crystal structure of [bis(2,2′-bipyridine-6-carboxylato-κ3N,N,O)magnesium(II)]dihydrate, C22H18N4O6Mg
- Crystal structure of poly[diaqua-(bis(μ2-1,4-bis(imidazol-1-ylmethyl)benzene)-κ2N,N′] cobalt(II)-tetraqua-bis(1,4-bis(imidazol-1-ylmethyl)benzene)-κ1N)-cobalt(II) di(2,5-thiophenedicarboxylate) dihydrate, C68H76Co2N16O16S2
- Crystal structure of poly[chlorido-μ2-chlorido-(μ2-1-[(2-ethyl-4-methyl-1H-imidazol-1-yl)methyl]-1H-benzotriazole-κN:N’)cadmium(II)], C13H15CdN5Cl2
- The crystal structure of (4-hydroxybenzenesulfonate)-k1O-6,6′-((1E,1′E)- (ethane-1,2-diylbis(azaneylylidene))bis(methaneylylidene)) bis(2-methoxyphenol)-κ2N,N,μ2O,O,κ2O, O)-(methanol)-cobalt(II) sodium(I), C25H27CoN2NaO9S
- Crystal structure of (1-methyl-3-(trifluoromethyl)-1H-pyrazol-4-yl)(4-((2-methyl-6-(trifluoromethyl)pyrimidin-4-yl)amino)piperidin-1-yl)methanone, C17H18F6N6O
- Crystal structure of bis{[(cyclohexylimino)(phenylimino)-l5-(methyl)diethylazane-κ2N:N′]-(ethyl)-zinc(II)]}, C38H62N6Zn2
- Crystal structure of 2-[(4-bromobenzyl)thio]-5-(5-bromothiophen-2-yl)-1,3,4-oxadiazole, C13H8Br2N2OS2
- Crystal structure of 10-methoxy-7,11b,12,13-tetrahydro-6H-pyrazino [2′,3′:5,6]pyrazino[2,1-a]isoquinoline, C15H16N4O
- The crystal structure of 1-propyl-2-nitro-imidazole oxide, C6H9N3O3
- The crystal structure of 3-nitrobenzene-1,2-dicarboxylic acid–2-ethoxybenzamide (1/1), C17H16N2O8
- The structure of RUB-1, (C8H16N)6[B6Si48O108], a boron containing levyne-type zeolite, occluding N-methyl-quinuclidinium in the cage-like pores
- The crystal structure of diaqua-(naphthalene-4,5-dicarboxylate-1,8-dicarboxylic anhydride-κ1O)-(4′-(4-(1H-benzimidazolyl-1-yl)phenyl)-2,2′:6′,2″-terpyridine-κ3N,N′,N″)–manganese(II) dihydrate, C42H27MnN5O9·2H2O
- Crystal structure of 6,6′-((1E,1′E)-hydrazine-1,2-diylidenebis(methanylylidene))bis (3-(3-bromopropoxy)phenol), C20H22Br2N2O4
- The crystal structure of 3-(2-hydroxyphenyl)-4-phenyl-6-(p-tolyl)-2H-pyran-2-one, C24H18O3
- Crystal structure of bis(μ2-2-(1,5-dimethyl–3-oxo-2-phenyl-2,3-dihydro-1H-pyrazol-4-yl)imino)methyl)phenolato-κ4O:O,N,O′)-(nitrato-κ2O,O′)dicobalt(II), C36H32Co2N8O4
- Synthesis and crystal structure of (3E,5S,10S,13S,14S,17Z)-17-ethylidene-10,13-dimethylhexadecahydro-3H-cyclopenta[α] phenanthren-3-one O-(4-fluorobenzoyl) oxime, C28H36FNO2
- The crystal structure of 4-aminiumbiphenyl benzenesulfonate, C18H17NO3S
- Synthesis and crystal structure of 1-(7-hydroxy-3-(4-hydroxy-3-nitrophenyl)-4-oxo-4H-chromen-8-yl)-N,N-dimethylmethanaminiumnitrate, C18H17N3O9
- Crystal structure of N-(Ar)-N′-(Ar′)-formamidine, C14H12Br2N2O
- The crystal structure of 4-(2,4-dichlorophenyl)-2-(4-fluorophenyl)-5-methyl-1H-imidazole, C16H11Cl2FN2
- Crystal structure of 1-(4–chlorophenyl)-4-benzoyl-3-methyl-1H-pyrazol-5-ol, C17H13ClN2O2
- The crystal structure of 5-amino-1-methyl-4-nitroimidazole, C4H6O2N4
- Crystal structure of 1,3-diisopropyl-4,5-dimethylimidazol-2-ylidene-N,N′-bis(1,3-bis(2,6-diisopropylphenyl)-1,3-dihydro-2H-1,3,2-diazaborol-2-yl)-l2-germenediamine, C63H94B2GeN8
- The crystal structure of (bromido, chlorido)-tricarbonyl-(5,5′-dimethyl-2,2′-bipyridine)-rhenium(I), C15H12Br0.2Cl0.8N2O3Re1
- Crystal structure of [N(E),N′(E)]-N,N′-(1,4-phenylenedimethylidyne)bis-3,5-bis(propan-2-yl)-1H-pyrazol-4-amine, C26H36N6
- The crystal structure of poly[2-(4-carboxypyridin-3-yl)terephthalpoly[diaqua-(μ4-2-(6-carboxylatopyridin-3-yl)terephthalato-κ5O,N:O′:O″,O‴)]) cadmium(II)] dihydrate, C28H20Cd3N2O16
- Crystal structure of [tetraaqua-bis((3-carboxy-5-(pyridin-4-yl)benzoate-κ1N)cobalt(II)] tetrahydrate, C26H32CoN2O16
- Crystal structure of bis(μ2-azido-κ2N:N)-tetrakis(azido-κ1N)-tetrakis(1,10-phenanthroline-κ2N,N′)dibismuth(III), C48H32N26Bi2
- Crystal structure of (Z)-N-(4-(4-(4-((4,5,6-trimethoxy-3-oxobenzofuran-2(3H)-ylidene)methyl)phenoxy)butoxy)phenyl)acetamide, C30H31NO8
- Crystal structure of poly[diaqua-(μ2-1,3-di(1H-imidazol-1-yl)propane-κ2N:N′)-bis(μ2-5-carboxybenzene-1,3-dicarboxylato-O,O′:O″)-aqua-di-zinc dihydrate solvate], C27H28N4O16Zn2
- Crystal structure of 2-(3,5,5-trimethylcyclohex-2-en-1-ylidene)malononitrile, C12H14N2
- Crystal structure of chlorido-(5-nitro-2-phenylpyridine-κ2N,C)-[(methylsulfinyl)methane-κ1S]platinum(II), C13H13ClN2O3PtS
- The crystal structure of the co-crystal 1,4-dioxane–4,6-bis(nitroimino)-1,3,5-triazinan-2-one(2/1), C11H19N7O9
- Crystal structure of [N(E),N′(E)]-N,N′-(1,4-phenylenedimethylidyne)bis-3,5-dimethyl-1H-pyrazol-4-amine di-methanol solvate, C18H20N6·2(CH3OH)
- Crystal structure of catena-poly[bis(μ2-azido-k2N:N′)-(nitrato-K2N:N′)-bis(1,10-phenanthroline-K2N:N′)samarium(III)], C24H16N11O3Sm
- Crystal structure of (Z)-2-(4-((5-bromopentyl)oxy)benzylidene)-4,5,6-trimethoxybenzofuran-3(2H)-one, C23H25BrO6
- Crystal structure of bis(3,5-dimethyl-1H-pyrazol-4-ammonium) tetrafluoroterephthate, 2[C5H10N3][C8F4O4]
- Crystal structure of 2-amino-4-(2-fluoro-4-(trifluoromethyl)phenyl)-9-methoxy-1,4,5,6-tetrahydrobenzo[h]quinazolin-3-ium chloride, C20H18ClF4N3O
- Crystal structure of 6-(pyridin-3-yl)-1,3,5-triazine-2,4-diamine-sebacic acid (2/1), C13H17N6O2


