Abstract
C27H23Fe2O5PS2, monoclinic, P21/c (no. 14), a = 11.3170(5) Å, b = 16.1774(8) Å, c = 15.2170(7) Å, β = 92.933(1)°, V = 2782.3(2) Å3, Z = 4, R gt (F) = 0.0537, wR ref(F 2) = 0.1636, T = 296(2) K.
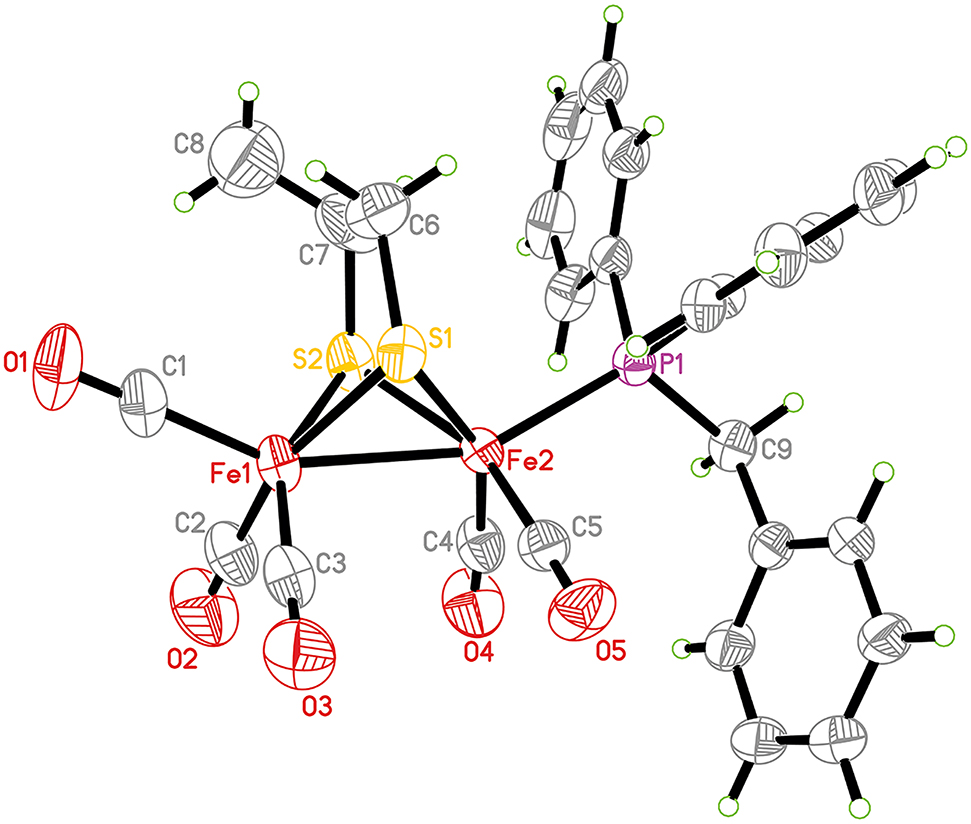
The molecular structure is shown in the figure. Table 1 contains crystallographic data and Table 2 contains the list of the atoms including atomic coordinates and displacement parameters.
Data collection and handling.
| Crystal: | Red block |
| Size: | 0.28 × 0.26 × 0.24 mm |
| Wavelength: | Mo Kα radiation (0.71073 Å) |
| μ: | 1.29 mm−1 |
| Diffractometer, scan mode: | Bruker D8 QUEST, ω |
| θ max, completeness: | 25.1°, 99% |
| N(hkl)measured, N(hkl)unique, R int: | 55006, 4918, 0.043 |
| Criterion for I obs, N(hkl)gt: | I obs > 2 σ(I obs), 3952 |
| N(param)refined: | 335 |
| Programs: | Bruker [1], Olex2 [2], SHELX [3, 4] |
Fractional atomic coordinates and isotropic or equivalent isotropic displacement parameters (Å2).
| Atom | x | y | z | U iso*/U eq |
|---|---|---|---|---|
| Fe1 | 0.48758 (6) | 0.19616 (5) | 0.81165 (5) | 0.0596 (2) |
| Fe2 | 0.30929 (5) | 0.18092 (4) | 0.70930 (4) | 0.0476 (2) |
| S1 | 0.31644 (11) | 0.14035 (9) | 0.84981 (8) | 0.0617 (3) |
| S2 | 0.46509 (11) | 0.09407 (9) | 0.71136 (10) | 0.0707 (4) |
| P1 | 0.16105 (10) | 0.11101 (7) | 0.64175 (7) | 0.0491 (3) |
| O1 | 0.6462 (5) | 0.1408 (4) | 0.9564 (4) | 0.1243 (19) |
| O2 | 0.6613 (5) | 0.2688 (4) | 0.7000 (4) | 0.135 (2) |
| O3 | 0.4179 (5) | 0.3563 (3) | 0.8819 (3) | 0.1078 (16) |
| O4 | 0.4108 (4) | 0.2613 (4) | 0.5605 (3) | 0.1104 (17) |
| O5 | 0.1726 (4) | 0.3239 (3) | 0.7601 (3) | 0.0965 (14) |
| C1 | 0.5868 (5) | 0.1603 (4) | 0.9000 (4) | 0.0828 (17) |
| C2 | 0.5941 (5) | 0.2409 (5) | 0.7446 (4) | 0.0854 (18) |
| C3 | 0.4463 (5) | 0.2935 (4) | 0.8558 (4) | 0.0745 (15) |
| C4 | 0.3666 (5) | 0.2292 (4) | 0.6171 (3) | 0.0714 (15) |
| C5 | 0.2245 (5) | 0.2677 (3) | 0.7377 (3) | 0.0622 (12) |
| C6 | 0.3421 (7) | 0.0276 (4) | 0.8472 (5) | 0.104 (2) |
| H6A | 0.3777 | 0.0097 | 0.9033 | 0.125* |
| H6B | 0.2670 | −0.0008 | 0.8378 | 0.125* |
| C7 | 0.4225 (7) | 0.0050 (4) | 0.7746 (6) | 0.108 (2) |
| H7 | 0.3782 | −0.0323 | 0.7343 | 0.130* |
| C8 | 0.5182 (10) | −0.0450 (7) | 0.8212 (7) | 0.157 (3) |
| H8A | 0.5748 | −0.0085 | 0.8501 | 0.236* |
| H8B | 0.5571 | −0.0784 | 0.7793 | 0.236* |
| H8C | 0.4841 | −0.0800 | 0.8641 | 0.236* |
| C9 | 0.0958 (5) | 0.1602 (3) | 0.5416 (3) | 0.0617 (12) |
| H9A | 0.1549 | 0.1618 | 0.4976 | 0.074* |
| H9B | 0.0307 | 0.1263 | 0.5185 | 0.074* |
| C10 | 0.0507 (4) | 0.2472 (3) | 0.5556 (3) | 0.0576 (11) |
| C11 | −0.0546 (4) | 0.2612 (3) | 0.5949 (3) | 0.0631 (13) |
| H11 | −0.1013 | 0.2168 | 0.6104 | 0.076* |
| C12 | −0.0911 (5) | 0.3408 (3) | 0.6113 (4) | 0.0743 (15) |
| H12 | −0.1627 | 0.3492 | 0.6372 | 0.089* |
| C13 | −0.0244 (5) | 0.4069 (4) | 0.5904 (4) | 0.0801 (16) |
| H13 | −0.0488 | 0.4602 | 0.6034 | 0.096* |
| C14 | 0.0800 (6) | 0.3940 (4) | 0.5497 (4) | 0.0845 (18) |
| H14 | 0.1260 | 0.4388 | 0.5343 | 0.101* |
| C15 | 0.1160 (5) | 0.3152 (4) | 0.5320 (4) | 0.0704 (15) |
| H15 | 0.1858 | 0.3072 | 0.5034 | 0.085* |
| C16 | 0.2047 (4) | 0.0104 (3) | 0.5995 (3) | 0.0589 (12) |
| C17 | 0.1654 (5) | −0.0640 (3) | 0.6293 (4) | 0.0746 (15) |
| H17 | 0.1105 | −0.0649 | 0.6727 | 0.090* |
| C18 | 0.2060 (7) | −0.1379 (4) | 0.5960 (5) | 0.096 (2) |
| H18 | 0.1794 | −0.1881 | 0.6174 | 0.115* |
| C19 | 0.2862 (8) | −0.1362 (5) | 0.5308 (6) | 0.112 (3) |
| H19 | 0.3113 | −0.1855 | 0.5063 | 0.134* |
| C20 | 0.3281 (6) | −0.0644 (5) | 0.5025 (4) | 0.096 (2) |
| H20 | 0.3842 | −0.0644 | 0.4600 | 0.116* |
| C21 | 0.2891 (5) | 0.0102 (4) | 0.5355 (4) | 0.0766 (16) |
| H21 | 0.3190 | 0.0598 | 0.5152 | 0.092* |
| C22 | 0.0277 (4) | 0.0902 (3) | 0.7011 (3) | 0.0527 (11) |
| C23 | −0.0656 (5) | 0.0447 (3) | 0.6618 (4) | 0.0695 (14) |
| H23 | −0.0588 | 0.0229 | 0.6058 | 0.083* |
| C24 | −0.1673 (5) | 0.0317 (4) | 0.7054 (4) | 0.0798 (17) |
| H24 | −0.2283 | 0.0005 | 0.6790 | 0.096* |
| C25 | −0.1794 (5) | 0.0640 (4) | 0.7861 (4) | 0.0796 (16) |
| H25 | −0.2489 | 0.0555 | 0.8148 | 0.095* |
| C26 | −0.0893 (4) | 0.1095 (4) | 0.8260 (4) | 0.0722 (14) |
| H26 | −0.0979 | 0.1317 | 0.8817 | 0.087* |
| C27 | 0.0135 (4) | 0.1220 (3) | 0.7837 (3) | 0.0569 (11) |
| H27 | 0.0745 | 0.1524 | 0.8114 | 0.068* |
Source of material
To a solution of complex [Fe2(CO)6{μ–SCH2CH(CH3)S}] (1 mmol) and benzyldiphenylphosphine (1 mmol) in CH2Cl2 (10 mL) was added a solution of Me3NO⋅2H2O (1 mmol) in CH3CN (10 mL). The solution was stirred for 1 h and the solvent was reduced by rotary evaporator. The title complex was obtained by TLC separation and the single crystals were collected from slow evaporation of CH2Cl2/isopropanol solution at 4°.
Experimental details
The structure was solved by Direct Methods with the SHELXS program. Hydrogen atoms were positioned geometrically (C–H = 0.93–0.98 Å). Their U iso values were set to 1.2U eq or 1.5U eq of the parent atoms.
Comment
Over the past two decades, diiron propane-1,3-dithiolate complexes have been received much attention [5], nevertheless, diiron propane-1,2-dithiolate complexes have been received little attention [6], [7], [8].
The asymmetric unit of the title complex contains a diiron core ligated by a bridging propane-1,2-dithiolate, five carbonyls and a benzyldiphenylphosphine ligands. The orientation of the phosphine ligand is in an apical position of the distorted octahedral geometry of the Fe2 atom, is analogous to phosphine-containing compounds [9], [10], [11], [12], [13]. The Fe1–Fe2 bond length [2.4972(9) Å] is shorter than that of the parent complex [Fe2(CO)6{μ–SCH2CH(CH3)S}] [2.5196(7) Å] [14], inconsistent with the fact that the phosphine coordination will lengthen the Fe–Fe bond due to the phosphine ligand having stronger electron-donor than CO [15, 16]. Furthermore, the Fe1–Fe2 bond length is similar to other all-carbonyl analogues [17], [18], [19], and a directly related structure [22], but much shorter than those in natural enzymed [20, 21].
Funding source: Zhejiang Provincial Natural Science Foundation of China
Award Identifier / Grant number: LY19B020002
-
Author contributions: All the authors have accepted responsibility for the entire content of this submitted manuscript and approved submission.
-
Research funding: This research was supported by Zhejiang Provincial Natural Science Foundation of China under Grant LY19B020002.
-
Conflict of interest statement: The authors declare no conflicts of interest regarding this article.
References
1. BRUKER. SAINT, APEX2 and SADABS; Bruker AXS Inc.: Madison, Wisconsin, USA, 2009.Suche in Google Scholar
2. Dolomanov, O. V., Bourhis, L. J., Gildea, R. J., Howard, J. A. K., Puschmann, H. OLEX2: a complete structure solution, refinement and analysis program. J. Appl. Crystallogr. 2009, 42, 339–341; https://doi.org/10.1107/s0021889808042726.Suche in Google Scholar
3. Sheldrick, G. M. Crystal structure refinement with SHELXL. Acta Crystallogr. 2015, C71, 3–8; https://doi.org/10.1107/s2053229614024218.Suche in Google Scholar
4. Sheldrick, G. M. A short history of SHELX. Acta Crystallogr. 2008, A64, 112–122; https://doi.org/10.1107/s0108767307043930.Suche in Google Scholar PubMed
5. Tard, C., Pickett, C. J. Structural and functional analogues of the active sites of the [Fe]-, [NiFe]-, and [FeFe]-hydrogenases. Chem. Rev. 2009, 109, 2245–2274; https://doi.org/10.1021/cr800542q.Suche in Google Scholar PubMed
6. Lin, H. M., Li, J. R., Mu, C., Li, A., Liu, X. F., Zhao, P. H., Li, Y. L., Jiang, Z. Q., Wu, H. K. Synthesis, characterization, and electrochemistry of monophosphine-containing diiron propane-1,2-dithiolate complexes related to the active site of [FeFe]-hydrogenases. Appl. Organomet. Chem. 2019, 33, e5196; https://doi.org/10.1002/aoc.5196.Suche in Google Scholar
7. Yan, L., Yang, J., Lü, S., Liu, X. F., Li, Y. L., Liu, X. H., Jiang, Z. Q. Phosphine-containing diiron propane-1,2-dithiolate derivatives: synthesis, spectroscopy, X-ray crystal structures, and electrochemistry. Catal. Lett. 2021, 151, 1857–1867; https://doi.org/10.1007/s10562-020-03450-2.Suche in Google Scholar
8. Yan, L., Wang, L. H., Yang, J., Liu, X. F., Li, Y. L., Liu, X. H., Jiang, Z. Q. Diiron propane-1,2-dithiolate complexes with monosubstituted tris(3-chlorophenyl)phosphine or tris(4-trifluoromethylphenyl)phosphine: synthesis, characterization, crystal structures, and electrochemistry. Inorg. Nano-Metal Chem. 2020, 50, 1137–1143; https://doi.org/10.1080/24701556.2020.1735431.Suche in Google Scholar
9. Chen, F. Y., He, J., Yu, X. Y., Wang, Z., Mu, C., Liu, X. F., Li, Y. L., Jiang, Z. Q., Wu, H. K. Electrocatalytic properties of diiron ethanedithiolate complexes containing benzoate ester. Appl. Organomet. Chem. 2018, 32, e4549; https://doi.org/10.1002/aoc.4549.Suche in Google Scholar
10. Yan, L., Hu, K., Liu, X. F., Li, Y. L., Liu, X. H., Jiang, Z. Q. Diiron ethane-1,2-dithiolate complexes with 1,2,3-thiadiazole moiety: synthesis, X-ray crystal structures, electrochemistry, and fungicidal activity. Appl. Organomet. Chem. 2021, 35, e6084; https://doi.org/10.1002/aoc.6084.Suche in Google Scholar
11. Liu, X. F., Li, Y. L., Liu, X. H. Heterocyclic pyrazole-containing diiron dithiolato analogues: synthesis, characterization, electrochemistry, and fungicidal activity. Appl. Organomet. Chem. 2022, 36, e6884; https://doi.org/10.1002/aoc.6884.Suche in Google Scholar
12. Yan, L., Yang, J., Liu, X. F., Li, Y. L., Liu, X. H., Jiang, Z. Q. Diiron toluene-3,4-dithiolate complexes with tris(3-fluorophenyl) phosphine or tris(4-trifluoromethylphenyl)phosphine: synthesis, spectroscopy, X-Ray crystal structures, and electrochemical properties. J. Chem. Crystallogr. 2021, 51, 183–190; https://doi.org/10.1007/s10870-020-00844-3.Suche in Google Scholar
13. Yan, L., Yang, J., Liu, X. F., Li, Y. L., Liu, X. H., Jiang, Z. Q. Di-1-adamantylphosphine-containing diiron propane-1,3-dithiolate pentacarbonyl complex: synthesis, structure, electrochemistry, and fungicidal activity. Phosphorus, Sulfur, Silicon Relat. Elem. 2022, 197, 62–66; https://doi.org/10.1080/10426507.2021.1989591.Suche in Google Scholar
14. Zhang, Q. Q., Dicson, R. S., Fallon, G. D., Mayadunne, R. A comparison of the reactions of pentacarbonyliron with cyclic thioethers and related dialkyl sulfides. J. Organomet. Chem. 2001, 627, 201–205; https://doi.org/10.1016/s0022-328x(01)00733-1.Suche in Google Scholar
15. Hu, M. Y., Zhao, P. H., Li, J. R., Gu, X. L., Jing, X. B., Liu, X. F. Synthesis, structures, and electrocatalytic properties of phosphine- monodentate, -chelate, and -bridge diiron 2,2-dimethylpropanedithiolate complexes related to [FeFe]-hydrogenases. Appl. Organomet. Chem. 2020, 34, e5523; https://doi.org/10.1002/aoc.5523.Suche in Google Scholar
16. Zhao, P. H., Hu, M. Y., Li, J. R., Wang, Y. Z., Lu, B. P., Han, H. F., Liu, X. F. Impacts of coordination modes (chelate versus bridge) of PNP-diphosphine ligands on the redox and electrocatalytic properties of diiron oxadithiolate complexes for proton reduction. Electrochim. Acta 2020, 353, 136615; https://doi.org/10.1016/j.electacta.2020.136615.Suche in Google Scholar
17. Lu, D. T., He, J., Yu, X. Y., Liu, X. F., Li, Y. L., Jiang, Z. Q. Diiron ethanedithiolate complexes with pendant ferrocene: synthesis, characterization and electrochemistry. Polyhedron 2018, 149, 1–6; https://doi.org/10.1016/j.poly.2018.04.015.Suche in Google Scholar
18. Lian, M., He, J., Yu, X. Y., Mu, C., Liu, X. F., Li, Y. L., Jiang, Z. Q. Diiron ethanedithiolate complexes with acetate ester: synthesis, characterization and electrochemical properties. J. Organomet. Chem. 2018, 870, 90–96; https://doi.org/10.1016/j.jorganchem.2018.06.023.Suche in Google Scholar
19. He, J., Gao, F., Li, Y. L., Liu, X. F., Wu, H., Jiang, Z. Q., Wu, H. K. Synthesis, characterization and electrochemistry of diiron 1,2-dithiolate complexes with a trans-cinnamate ester. J. Coord. Chem. 2019, 72, 452–467; https://doi.org/10.1080/00958972.2019.1569641.Suche in Google Scholar
20. Peters, J. W., Lanzilotta, W. N., Lemon, B. J., Seefeldt, L. C. X-ray crystal structure of the Fe-only hydrogenase (CpI) from clostridium pasteurianum to 1.8 angstrom resolution. Science 1998, 282, 1853–1857; https://doi.org/10.1126/science.282.5395.1853.Suche in Google Scholar PubMed
21. Nicolet, Y., Piras, C., Legrand, P., Hatchikian, C. E., Fontecilla-Camps, J. C. Desulfovibrio Desulfuricans iron hydrogenase: the structure shows unusual coordination to an active site Fe binuclear center. Structure 1999, 7, 13–23; https://doi.org/10.1016/s0969-2126(99)80005-7.Suche in Google Scholar PubMed
22. Sheng, Y.-D., Yu, X.-Y., Liu, X.-F., Li, Y.-L. 2-(diphenylphosphino)benzaldehyde or isopropyldiphenylphosphine substituted diiron dithiolate complexes. Polyhedron 2017, 137, 134–139; https://doi.org/10.1016/j.poly.2017.08.029.Suche in Google Scholar
© 2022 the author(s), published by De Gruyter, Berlin/Boston
This work is licensed under the Creative Commons Attribution 4.0 International License.
Artikel in diesem Heft
- Frontmatter
- New Crystal Structures
- Crystal structure of undecacalcium decaarsenide, Ca11As10
- Crystal structure of catena-poly[diiodido-(μ2-1,5-dimethyl-2-phenyl-4-((pyridin-4- ylmethylene)amino)-1,2-dihydro-3H -pyrazol-3-one-κ2 N: O)zinc(II)], C17H16I2N4OZn
- The crystal structure of 5,10,15,20-tetrakis(4-(tert-butyl)phenyl)porphyrin-21,23-diido-κ4 N 4-naphthalocyanido-κ4 N 4-neodymium(IV) - chloroform (1/6) C114H90N12Cl18Nd
- The crystal structure of 1-(4-bromophenyl)-3-(2-chlorobenzyl)urea, C14H12BrClN2O
- Crystal structure of bis[benzyl(methyl)carbamodithioato-κ 2 S,S′]-di-n-butyltin(IV), C26H38N2S4Sn
- Crystal structure of (E)-3-(2-(4-(diethylamino)-2-hydroxystyryl)-3,3-dimethyl-3H-indol-1-ium-1-yl)propane-1-sulfonate – methanol (1/2), C25H32N2O4S⋅2CH3OH
- Synthesis and crystal structure of {(N′,N″-(((ethane-1,2-diylbis(oxy))bis(2,1-phenylene))-bis(methaneylylidene))bis(2-hydroxybenzohydrazonato)-κ6 N 2 O 4}copper(II), C30H24CuN4O6
- The crystal structure of ((E)-2,4-dibromo-6-(((5-(nitro)-2-oxidophenyl)imino)methyl)phenolato-κ 3 N,O,O′)tris(pyridine-κN)manganese(II), C28H21Br2MnN5O4
- Synthesis and crystal structure of 1-((3R,10S,13R,17S)-10,13-dimethyl-3-(p-tolylamino)hexadecahydro-1H-cyclopenta-[a]phenanthren-17-yl)ethan-1-one, C28H41NO
- The crystal structure of 3-nitrobenzene-1,2-dicarboxylic acid—pyrazine-2-carboxamide(1/1), C13H10N4O7
- Crystal structure of poly[tetrakis(μ3-2-aminonicotinato-κ3N,O,O′)-(μ2-oxalato-κ4 O,O′:O″,O′″)-(μ4-oxalato-κ6 N:N′:O,O′:O″,O′″)dicopper(I)-disamarium(III)], [SmCu(C6N2H5O2)2(C2O4)] n
- The crystal structure of 2,3,4-trihydroxybenzoic- acid—pyrazine-2-carboxamide—water (1/1/1), C12H13N3O7
- Crystal structure of N-ethyl-4-[3-(trifluoromethyl)-phenyl]piperazine-1-carbothioamide, C14H18F3N3S
- The crystal structure of 3-anilino-1,4-diphenyl-4H-1,2,4-triazol-1-ium iodide, C20H17N4I
- The crystal structure of (tris(2-benzimidazolylmethyl)amine)-benzoato-copper(II) perchlorate monohydrate, CuC31H28N7O7Cl
- Crystal structure of [2-hydroxy-3-methyl-benzoato-k1 O-triphenyltin(IV)], C26H22O3Sn
- Crystal structure of diaqua-bis(4-(hydroxymethyl)-benzoato-k1 O)zinc(II), C16H18O8Zn
- The crystal structure of dicarbonyl-(N-nitroso-N-oxido-phenylamine-κ 2 O,O)-rhodium(I), C8H5N2O4Rh
- The crystal structure of oxalic acid – 2-ethoxybenzamide (2/1), C20H24N2O8
- The crystal structure of ethyl 7-ethyl-5-methyl-4,7-dihydrotetrazolo[1,5-a]pyrimidine-6-carboxylate, C10H15N5O2
- Crystal structure of poly[(N,N-dimethylacetamide-κO) (μ4-2-nitroisophthalato-κ 4 O:O′:O″:O′″)manganese(II)], C11H10N2O7Mn
- Crystal structure of 14-O-acetyldelcosine, C26H41NO8
- The crystal structure of poly[(1,10-phenanthroline-κ2 N,N′)-(μ 4-2-chlorobenzene-1,3-dicarboxylato-κ5 O:O′:O″:O‴) cadmium(II)] monohydrate, C20H13CdClN2O5
- Crystal structure of propane-1,3-diylbis(diphenylphosphine sulfide) ethanol solvate, C27H26P2S2
- Crystal structure of bis{[(4-diethylamino-2-hydroxy-benzylidene)-hydrazinocarbonylmethyl]-trimethylammonium} tetrabromozincate, C32H54N8O4ZnBr4
- Synthesis and crystal structure of dimethyl 2,2′-(2,5-bis(4-hydroxyphenyl)-2,5-dihydrofuran-3,4-diyl)dibenzoate, C34H30O7
- Synthesis and crystal structure of 2-(2-oxo-2-phenylethyl)-4H-chromen-4-one, C17H12O3
- The crystal structure of tetra(imidazole-κ1 N)zinc(II) μ2-oxido-hexaoxido-divanadium(VI) C12H16N8O6V2Zn
- Crystal structure of S-2-(1-(5-methylpyridin-2-ylamino)octyl)-3-hydroxynaphthalene-1,4-dione, C24H28N2O3
- Crystal structure of bis(μ2-5-chloro-2-oxido-N-(1-oxido-2-oxo-2-phenylethylidene)-benzohydrazonato-κ5 N,O,O′:N′,O′′)-oktakis(pyridine-κ1 N)trinickel(II) – methanol – pyridine (1/1/1) C76H65N13Cl2Ni3O9
- The crystal structure of methyl 3,5-diaminobenzoate, C8H10N2O2
- Crystal structure of 10-(9H-carbazol-9-yl)-5H-dibenzo[a,d][7]annelen-5-one, C27H17NO
- Crystal structure of ethyl 1-(2-hydroxyethyl)-4-((4-methoxyphenyl)amino)-5-oxo-2,5-dihydro-1H-pyrrole-3-carboxylate, C16H20N2O5
- The crystal structure of 1-(4-bromophenyl)-3-cycloheptylurea, C14H19BrN2O
- The crystal structure of 1,4-bis(1,2,3,4,5-pentamethylcyclopenta-2,4-dien-1-yl)-3,6-bis ((4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)methylene)-1,4-dialuminacyclohexane – benzene (1/2), C50H72Al2B2O4
- Crystal structure of bis(μ 3-diphenylphosphinato)-tetrakis(μ 2-diphenylphosphinato)-bis(diphenylphosphinato)-bis(μ 2-hydroxo)dicopper(II)-ditin(IV), C104H100O18P8Cu2Sn2
- Crystal structure of 3-((3,4-dichloroisothiazol-5-yl)methoxy)benzo[d] isothiazole 1,1-dioxide, C11H6Cl2N2O3S2
- Synthesis and crystal structure of 2-(2-(2-fluorophenyl)-2-oxoethyl)-4H-chromen-4-one, C17H11FO3
- The crystal structure of tris(carbonyl)-bis(carbonyl)-[μ-propane-1,2- dithiolato]-(benzyldiphenylphosphine)diiron (Fe—Fe), C27H23Fe2O5PS2
- Crystal structure of 1-(2-(4-chlorophenethyl)-2-hydroxy-3,3-dimethylbutyl)-1H-1,2,4-triazol-4-ium nitrate, C16H23N4O4Cl
- The crystal structure of 3,3′-disulfanediyldi(1H-1,2,4-triazol-5-amine) monohydrate, C4H8N8OS2
- The crystal structure of trans-[bis(4-methylpyridine-κN)bis(quinoline-2-carboxylato- κ 2 N,O)cadmium(II)], C32H26CdN4O4
- The crystal structure of ethyl 2′-hydroxy-4′,6′-dimethoxy-3-(4-methoxynaphthalen-1-yl)-5-oxo-2,3,4,5-tetrahydro-[1,1′-biphenyl]-4-carboxylate, C28H28O7
Artikel in diesem Heft
- Frontmatter
- New Crystal Structures
- Crystal structure of undecacalcium decaarsenide, Ca11As10
- Crystal structure of catena-poly[diiodido-(μ2-1,5-dimethyl-2-phenyl-4-((pyridin-4- ylmethylene)amino)-1,2-dihydro-3H -pyrazol-3-one-κ2 N: O)zinc(II)], C17H16I2N4OZn
- The crystal structure of 5,10,15,20-tetrakis(4-(tert-butyl)phenyl)porphyrin-21,23-diido-κ4 N 4-naphthalocyanido-κ4 N 4-neodymium(IV) - chloroform (1/6) C114H90N12Cl18Nd
- The crystal structure of 1-(4-bromophenyl)-3-(2-chlorobenzyl)urea, C14H12BrClN2O
- Crystal structure of bis[benzyl(methyl)carbamodithioato-κ 2 S,S′]-di-n-butyltin(IV), C26H38N2S4Sn
- Crystal structure of (E)-3-(2-(4-(diethylamino)-2-hydroxystyryl)-3,3-dimethyl-3H-indol-1-ium-1-yl)propane-1-sulfonate – methanol (1/2), C25H32N2O4S⋅2CH3OH
- Synthesis and crystal structure of {(N′,N″-(((ethane-1,2-diylbis(oxy))bis(2,1-phenylene))-bis(methaneylylidene))bis(2-hydroxybenzohydrazonato)-κ6 N 2 O 4}copper(II), C30H24CuN4O6
- The crystal structure of ((E)-2,4-dibromo-6-(((5-(nitro)-2-oxidophenyl)imino)methyl)phenolato-κ 3 N,O,O′)tris(pyridine-κN)manganese(II), C28H21Br2MnN5O4
- Synthesis and crystal structure of 1-((3R,10S,13R,17S)-10,13-dimethyl-3-(p-tolylamino)hexadecahydro-1H-cyclopenta-[a]phenanthren-17-yl)ethan-1-one, C28H41NO
- The crystal structure of 3-nitrobenzene-1,2-dicarboxylic acid—pyrazine-2-carboxamide(1/1), C13H10N4O7
- Crystal structure of poly[tetrakis(μ3-2-aminonicotinato-κ3N,O,O′)-(μ2-oxalato-κ4 O,O′:O″,O′″)-(μ4-oxalato-κ6 N:N′:O,O′:O″,O′″)dicopper(I)-disamarium(III)], [SmCu(C6N2H5O2)2(C2O4)] n
- The crystal structure of 2,3,4-trihydroxybenzoic- acid—pyrazine-2-carboxamide—water (1/1/1), C12H13N3O7
- Crystal structure of N-ethyl-4-[3-(trifluoromethyl)-phenyl]piperazine-1-carbothioamide, C14H18F3N3S
- The crystal structure of 3-anilino-1,4-diphenyl-4H-1,2,4-triazol-1-ium iodide, C20H17N4I
- The crystal structure of (tris(2-benzimidazolylmethyl)amine)-benzoato-copper(II) perchlorate monohydrate, CuC31H28N7O7Cl
- Crystal structure of [2-hydroxy-3-methyl-benzoato-k1 O-triphenyltin(IV)], C26H22O3Sn
- Crystal structure of diaqua-bis(4-(hydroxymethyl)-benzoato-k1 O)zinc(II), C16H18O8Zn
- The crystal structure of dicarbonyl-(N-nitroso-N-oxido-phenylamine-κ 2 O,O)-rhodium(I), C8H5N2O4Rh
- The crystal structure of oxalic acid – 2-ethoxybenzamide (2/1), C20H24N2O8
- The crystal structure of ethyl 7-ethyl-5-methyl-4,7-dihydrotetrazolo[1,5-a]pyrimidine-6-carboxylate, C10H15N5O2
- Crystal structure of poly[(N,N-dimethylacetamide-κO) (μ4-2-nitroisophthalato-κ 4 O:O′:O″:O′″)manganese(II)], C11H10N2O7Mn
- Crystal structure of 14-O-acetyldelcosine, C26H41NO8
- The crystal structure of poly[(1,10-phenanthroline-κ2 N,N′)-(μ 4-2-chlorobenzene-1,3-dicarboxylato-κ5 O:O′:O″:O‴) cadmium(II)] monohydrate, C20H13CdClN2O5
- Crystal structure of propane-1,3-diylbis(diphenylphosphine sulfide) ethanol solvate, C27H26P2S2
- Crystal structure of bis{[(4-diethylamino-2-hydroxy-benzylidene)-hydrazinocarbonylmethyl]-trimethylammonium} tetrabromozincate, C32H54N8O4ZnBr4
- Synthesis and crystal structure of dimethyl 2,2′-(2,5-bis(4-hydroxyphenyl)-2,5-dihydrofuran-3,4-diyl)dibenzoate, C34H30O7
- Synthesis and crystal structure of 2-(2-oxo-2-phenylethyl)-4H-chromen-4-one, C17H12O3
- The crystal structure of tetra(imidazole-κ1 N)zinc(II) μ2-oxido-hexaoxido-divanadium(VI) C12H16N8O6V2Zn
- Crystal structure of S-2-(1-(5-methylpyridin-2-ylamino)octyl)-3-hydroxynaphthalene-1,4-dione, C24H28N2O3
- Crystal structure of bis(μ2-5-chloro-2-oxido-N-(1-oxido-2-oxo-2-phenylethylidene)-benzohydrazonato-κ5 N,O,O′:N′,O′′)-oktakis(pyridine-κ1 N)trinickel(II) – methanol – pyridine (1/1/1) C76H65N13Cl2Ni3O9
- The crystal structure of methyl 3,5-diaminobenzoate, C8H10N2O2
- Crystal structure of 10-(9H-carbazol-9-yl)-5H-dibenzo[a,d][7]annelen-5-one, C27H17NO
- Crystal structure of ethyl 1-(2-hydroxyethyl)-4-((4-methoxyphenyl)amino)-5-oxo-2,5-dihydro-1H-pyrrole-3-carboxylate, C16H20N2O5
- The crystal structure of 1-(4-bromophenyl)-3-cycloheptylurea, C14H19BrN2O
- The crystal structure of 1,4-bis(1,2,3,4,5-pentamethylcyclopenta-2,4-dien-1-yl)-3,6-bis ((4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)methylene)-1,4-dialuminacyclohexane – benzene (1/2), C50H72Al2B2O4
- Crystal structure of bis(μ 3-diphenylphosphinato)-tetrakis(μ 2-diphenylphosphinato)-bis(diphenylphosphinato)-bis(μ 2-hydroxo)dicopper(II)-ditin(IV), C104H100O18P8Cu2Sn2
- Crystal structure of 3-((3,4-dichloroisothiazol-5-yl)methoxy)benzo[d] isothiazole 1,1-dioxide, C11H6Cl2N2O3S2
- Synthesis and crystal structure of 2-(2-(2-fluorophenyl)-2-oxoethyl)-4H-chromen-4-one, C17H11FO3
- The crystal structure of tris(carbonyl)-bis(carbonyl)-[μ-propane-1,2- dithiolato]-(benzyldiphenylphosphine)diiron (Fe—Fe), C27H23Fe2O5PS2
- Crystal structure of 1-(2-(4-chlorophenethyl)-2-hydroxy-3,3-dimethylbutyl)-1H-1,2,4-triazol-4-ium nitrate, C16H23N4O4Cl
- The crystal structure of 3,3′-disulfanediyldi(1H-1,2,4-triazol-5-amine) monohydrate, C4H8N8OS2
- The crystal structure of trans-[bis(4-methylpyridine-κN)bis(quinoline-2-carboxylato- κ 2 N,O)cadmium(II)], C32H26CdN4O4
- The crystal structure of ethyl 2′-hydroxy-4′,6′-dimethoxy-3-(4-methoxynaphthalen-1-yl)-5-oxo-2,3,4,5-tetrahydro-[1,1′-biphenyl]-4-carboxylate, C28H28O7

