Abstract
C76H65N13Cl2Ni3O9, monoclinic, P21/n, a = 15.937(4) Å, b = 16.805(4) Å, c = 27.463(7) Å, β = 94.613(3)°, V = 7332(3) Å3, Z = 4, Rgt (F) = 0.0490, wRref (F 2) = 0.1596, T = 298 K.
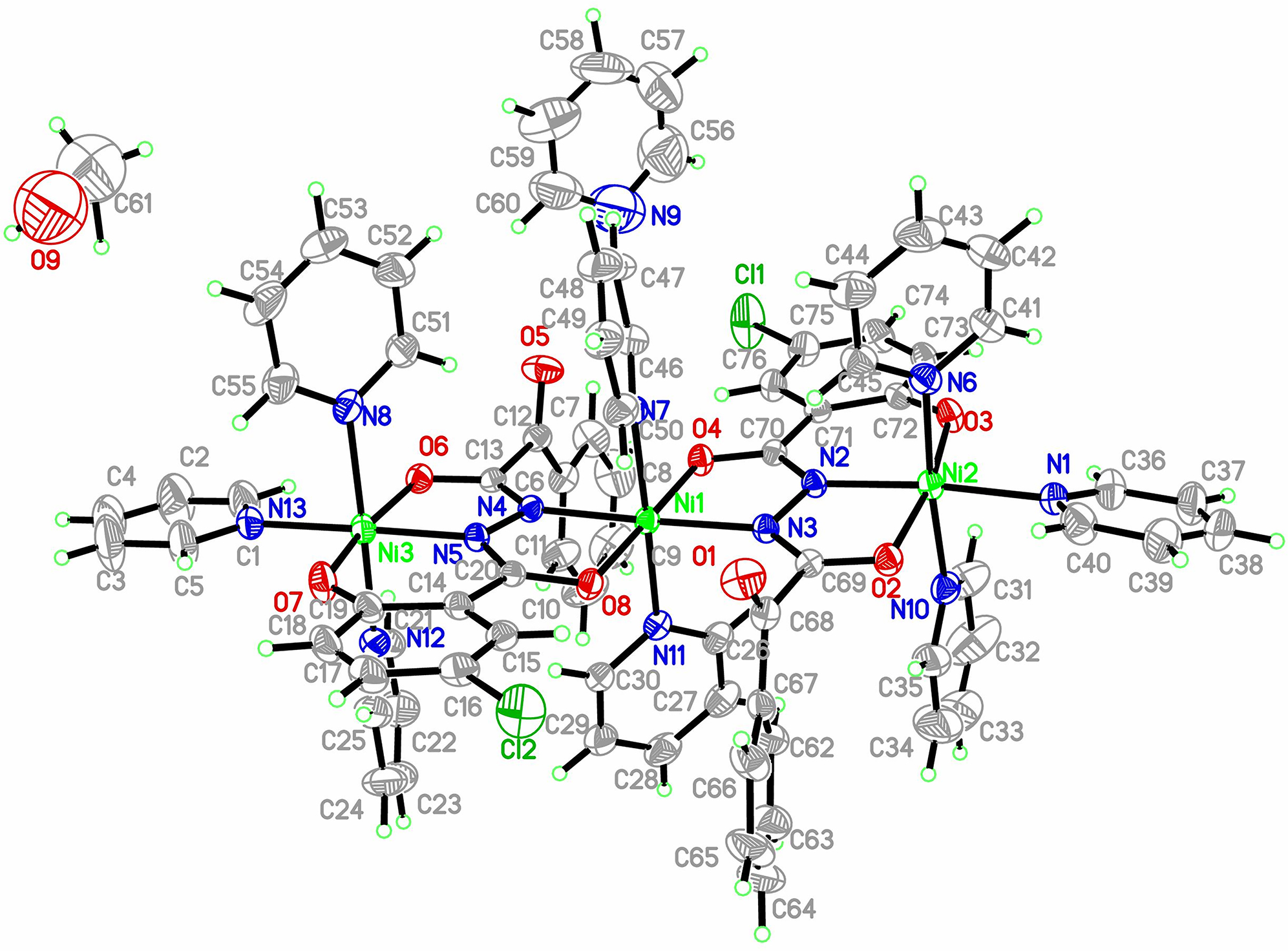
The crystal structure is shown in the figure. Table 1 contains crystallographic data and Table 2 contains the list of the atoms including atomic coordinates and displacement parameters.
Data collection and handling.
| Crystal: | Red block |
| Size: | 0.17 × 0.12 × 0.08 mm |
| Wavelength: | Mo Kα radiation (0.71073 Å) |
| μ: | 0.90 mm−1 |
| Diffractometer, scan mode: | Bruker APEX-II, φ and ω-scans |
| θ max, completeness: | 27.6°, >99% |
| N(hkl)measured, N(hkl)unique, R int: | 78367, 16866, 0.041 |
| Criterion for I obs, N(hkl)gt: | I obs > 2 σ(I obs), 11675 |
| N(param)refined: | 928 |
| Programs: | Bruker programs [1], Shelx [2, 3] |
Fractional atomic coordinates and isotropic or equivalent isotropic displacement parameters (Å2).
| x | y | z | U iso*/U eq | |
|---|---|---|---|---|
| Ni1 | 0.57840 (2) | 0.26476 (2) | 0.38038 (2) | 0.03263 (10) |
| Cl1 | 0.48043 (7) | 0.31827 (11) | 0.12749 (4) | 0.1068 (5) |
| O1 | 0.76547 (18) | 0.32019 (16) | 0.48243 (9) | 0.0661 (7) |
| N1 | 0.96745 (16) | 0.34201 (16) | 0.30767 (10) | 0.0451 (6) |
| C1 | 0.1390 (2) | 0.1977 (3) | 0.41881 (16) | 0.0776 (13) |
| H1 | 0.1611 | 0.2011 | 0.3885 | 0.093* |
| Ni2 | 0.83732 (2) | 0.32671 (2) | 0.31229 (2) | 0.03728 (11) |
| Cl2 | 0.67252 (8) | 0.26969 (9) | 0.63572 (4) | 0.0850 (4) |
| O2 | 0.84896 (12) | 0.29712 (13) | 0.38655 (7) | 0.0428 (5) |
| N2 | 0.71812 (14) | 0.30550 (14) | 0.32390 (8) | 0.0331 (5) |
| C2 | 0.0556 (3) | 0.1781 (4) | 0.4200 (2) | 0.114 (2) |
| H2 | 0.0228 | 0.1673 | 0.3912 | 0.137* |
| Ni3 | 0.31677 (2) | 0.23389 (2) | 0.45099 (2) | 0.04128 (11) |
| O3 | 0.80597 (13) | 0.35326 (14) | 0.24237 (7) | 0.0461 (5) |
| N3 | 0.70463 (14) | 0.28382 (14) | 0.37274 (8) | 0.0339 (5) |
| C3 | 0.0221 (3) | 0.1747 (4) | 0.4632 (2) | 0.111 (2) |
| H3 | −0.0344 | 0.1623 | 0.4648 | 0.133* |
| O4 | 0.57639 (12) | 0.28586 (13) | 0.30787 (7) | 0.0392 (5) |
| N4 | 0.45166 (14) | 0.25054 (13) | 0.38845 (8) | 0.0324 (5) |
| C4 | 0.0716 (3) | 0.1894 (5) | 0.5039 (2) | 0.119 (2) |
| H4 | 0.0497 | 0.1877 | 0.5343 | 0.142* |
| O5 | 0.36931 (19) | 0.32895 (15) | 0.29200 (9) | 0.0650 (7) |
| N5 | 0.43664 (15) | 0.24795 (14) | 0.43871 (8) | 0.0343 (5) |
| C5 | 0.1562 (3) | 0.2071 (4) | 0.50020 (16) | 0.0871 (15) |
| H5 | 0.1907 | 0.2159 | 0.5287 | 0.105* |
| O6 | 0.30644 (12) | 0.23896 (13) | 0.37511 (7) | 0.0411 (5) |
| N6 | 0.82958 (17) | 0.45276 (16) | 0.33275 (9) | 0.0457 (6) |
| C6 | 0.38798 (19) | 0.1942 (2) | 0.27219 (11) | 0.0457 (7) |
| N7 | 0.56249 (16) | 0.39070 (15) | 0.39421 (9) | 0.0426 (6) |
| O7 | 0.34524 (15) | 0.23180 (16) | 0.52280 (8) | 0.0568 (6) |
| C7 | 0.3755 (3) | 0.2069 (3) | 0.22198 (13) | 0.0672 (10) |
| H7 | 0.3625 | 0.2577 | 0.2102 | 0.081* |
| N8 | 0.29200 (17) | 0.36276 (17) | 0.45093 (10) | 0.0494 (6) |
| O8 | 0.58011 (12) | 0.24454 (12) | 0.45332 (7) | 0.0383 (4) |
| C8 | 0.3822 (3) | 0.1455 (4) | 0.18979 (16) | 0.0930 (16) |
| H8 | 0.3746 | 0.1548 | 0.1563 | 0.112* |
| N9 | 0.3786 (6) | 0.4744 (5) | 0.2181 (3) | 0.179 (3) |
| C9 | 0.4000 (4) | 0.0706 (4) | 0.2069 (2) | 0.1001 (17) |
| H9 | 0.4039 | 0.0290 | 0.1849 | 0.120* |
| O9 | 0.0262 (5) | 0.5537 (5) | 0.5280 (3) | 0.236 (3) |
| H9C | −0.0109 | 0.5227 | 0.5348 | 0.354* |
| C10 | 0.4123 (3) | 0.0556 (3) | 0.2565 (2) | 0.0926 (16) |
| H10 | 0.4240 | 0.0043 | 0.2678 | 0.111* |
| N10 | 0.85686 (16) | 0.20157 (17) | 0.29187 (11) | 0.0488 (6) |
| C11 | 0.4071 (3) | 0.1184 (2) | 0.28947 (15) | 0.0647 (10) |
| H11 | 0.4164 | 0.1093 | 0.3229 | 0.078* |
| N11 | 0.59065 (15) | 0.13856 (15) | 0.36634 (9) | 0.0396 (5) |
| C13 | 0.38073 (18) | 0.24715 (16) | 0.36093 (10) | 0.0344 (6) |
| N13 | 0.18932 (17) | 0.21193 (18) | 0.45791 (11) | 0.0528 (7) |
| C12 | 0.38180 (19) | 0.26154 (19) | 0.30631 (11) | 0.0401 (6) |
| N12 | 0.33098 (17) | 0.10332 (17) | 0.44434 (11) | 0.0515 (7) |
| C14 | 0.49781 (19) | 0.24723 (17) | 0.52215 (10) | 0.0373 (6) |
| C15 | 0.5734 (2) | 0.25677 (18) | 0.55108 (11) | 0.0421 (7) |
| H15 | 0.6234 | 0.2619 | 0.5361 | 0.051* |
| C16 | 0.5754 (2) | 0.2587 (2) | 0.60129 (11) | 0.0501 (8) |
| C17 | 0.5022 (3) | 0.2505 (2) | 0.62461 (12) | 0.0575 (9) |
| H17 | 0.5038 | 0.2514 | 0.6585 | 0.069* |
| C18 | 0.4274 (2) | 0.2410 (2) | 0.59724 (12) | 0.0556 (9) |
| H18 | 0.3788 | 0.2347 | 0.6134 | 0.067* |
| C19 | 0.4200 (2) | 0.24027 (19) | 0.54488 (11) | 0.0444 (7) |
| C20 | 0.50583 (17) | 0.24621 (16) | 0.46825 (10) | 0.0324 (6) |
| C21 | 0.3062 (3) | 0.0655 (2) | 0.40349 (16) | 0.0654 (10) |
| H21 | 0.2793 | 0.0946 | 0.3780 | 0.078* |
| C22 | 0.3183 (3) | −0.0146 (3) | 0.3967 (2) | 0.0858 (14) |
| H22 | 0.3005 | −0.0386 | 0.3671 | 0.103* |
| C23 | 0.3563 (3) | −0.0583 (3) | 0.4337 (2) | 0.0906 (15) |
| H23 | 0.3641 | −0.1128 | 0.4302 | 0.109* |
| C24 | 0.3825 (3) | −0.0208 (3) | 0.4759 (2) | 0.0881 (15) |
| H24 | 0.4096 | −0.0493 | 0.5016 | 0.106* |
| C25 | 0.3687 (2) | 0.0604 (2) | 0.48046 (17) | 0.0698 (11) |
| H25 | 0.3864 | 0.0856 | 0.5097 | 0.084* |
| C26 | 0.6347 (2) | 0.1100 (2) | 0.33113 (12) | 0.0502 (8) |
| H26 | 0.6642 | 0.1459 | 0.3131 | 0.060* |
| C28 | 0.5955 (2) | −0.0235 (2) | 0.34638 (17) | 0.0701 (11) |
| H28 | 0.5965 | −0.0776 | 0.3394 | 0.084* |
| C27 | 0.6393 (3) | 0.0302 (2) | 0.31987 (14) | 0.0643 (10) |
| H27 | 0.6711 | 0.0129 | 0.2950 | 0.077* |
| C29 | 0.5509 (2) | 0.0051 (2) | 0.38314 (17) | 0.0696 (11) |
| H29 | 0.5214 | −0.0295 | 0.4020 | 0.083* |
| C30 | 0.5501 (2) | 0.0852 (2) | 0.39186 (13) | 0.0511 (8) |
| H30 | 0.5195 | 0.1036 | 0.4170 | 0.061* |
| C31 | 0.8508 (3) | 0.1767 (3) | 0.24590 (17) | 0.0850 (14) |
| H31 | 0.8391 | 0.2142 | 0.2213 | 0.102* |
| C32 | 0.8609 (4) | 0.0983 (4) | 0.2324 (3) | 0.120 (2) |
| H32 | 0.8575 | 0.0832 | 0.1998 | 0.144* |
| C33 | 0.8760 (4) | 0.0434 (3) | 0.2693 (3) | 0.126 (3) |
| H33 | 0.8814 | −0.0103 | 0.2619 | 0.151* |
| C34 | 0.8830 (4) | 0.0677 (3) | 0.3155 (3) | 0.116 (2) |
| H34 | 0.8952 | 0.0316 | 0.3408 | 0.139* |
| C35 | 0.8721 (3) | 0.1462 (2) | 0.32525 (18) | 0.0758 (12) |
| H35 | 0.8757 | 0.1618 | 0.3578 | 0.091* |
| C36 | 1.0032 (2) | 0.3342 (2) | 0.26566 (14) | 0.0617 (10) |
| H36 | 0.9686 | 0.3255 | 0.2373 | 0.074* |
| C37 | 1.0884 (3) | 0.3383 (3) | 0.2622 (2) | 0.0800 (13) |
| H37 | 1.1108 | 0.3331 | 0.2321 | 0.096* |
| C38 | 1.1401 (3) | 0.3503 (3) | 0.3040 (2) | 0.0881 (15) |
| H38 | 1.1982 | 0.3528 | 0.3028 | 0.106* |
| C39 | 1.1046 (3) | 0.3585 (3) | 0.3471 (2) | 0.0884 (15) |
| H39 | 1.1382 | 0.3668 | 0.3759 | 0.106* |
| C40 | 1.0182 (2) | 0.3544 (3) | 0.34761 (15) | 0.0693 (11) |
| H40 | 0.9946 | 0.3605 | 0.3773 | 0.083* |
| C41 | 0.8741 (3) | 0.5083 (2) | 0.31126 (15) | 0.0631 (10) |
| H41 | 0.9101 | 0.4917 | 0.2882 | 0.076* |
| C42 | 0.8700 (3) | 0.5882 (3) | 0.3212 (2) | 0.0851 (14) |
| H42 | 0.9019 | 0.6245 | 0.3050 | 0.102* |
| C43 | 0.8184 (3) | 0.6134 (3) | 0.3553 (2) | 0.0866 (14) |
| H43 | 0.8147 | 0.6671 | 0.3631 | 0.104* |
| C44 | 0.7716 (3) | 0.5574 (3) | 0.37818 (16) | 0.0736 (11) |
| H44 | 0.7356 | 0.5728 | 0.4015 | 0.088* |
| C45 | 0.7791 (2) | 0.4789 (2) | 0.36584 (13) | 0.0563 (9) |
| H45 | 0.7473 | 0.4417 | 0.3814 | 0.068* |
| C46 | 0.5251 (2) | 0.4385 (2) | 0.36033 (13) | 0.0553 (8) |
| H46 | 0.5102 | 0.4180 | 0.3294 | 0.066* |
| C47 | 0.5078 (3) | 0.5176 (2) | 0.36945 (17) | 0.0719 (11) |
| H47 | 0.4816 | 0.5495 | 0.3451 | 0.086* |
| C48 | 0.5297 (3) | 0.5479 (2) | 0.41478 (19) | 0.0774 (12) |
| H48 | 0.5178 | 0.6006 | 0.4218 | 0.093* |
| C49 | 0.5694 (3) | 0.5000 (2) | 0.45000 (16) | 0.0655 (10) |
| H49 | 0.5852 | 0.5197 | 0.4811 | 0.079* |
| C50 | 0.5850 (2) | 0.4229 (2) | 0.43813 (13) | 0.0516 (8) |
| H50 | 0.6128 | 0.3907 | 0.4618 | 0.062* |
| C51 | 0.3181 (3) | 0.4109 (2) | 0.41701 (15) | 0.0650 (10) |
| H51 | 0.3510 | 0.3892 | 0.3939 | 0.078* |
| C52 | 0.2999 (3) | 0.4900 (2) | 0.41382 (17) | 0.0758 (12) |
| H52 | 0.3197 | 0.5207 | 0.3890 | 0.091* |
| C53 | 0.2526 (3) | 0.5238 (3) | 0.44738 (18) | 0.0796 (13) |
| H53 | 0.2387 | 0.5775 | 0.4457 | 0.096* |
| C54 | 0.2257 (3) | 0.4763 (3) | 0.48373 (18) | 0.0875 (15) |
| H54 | 0.1941 | 0.4973 | 0.5077 | 0.105* |
| C55 | 0.2468 (3) | 0.3967 (3) | 0.48392 (14) | 0.0717 (11) |
| H55 | 0.2284 | 0.3648 | 0.5086 | 0.086* |
| C56 | 0.4011 (6) | 0.5255 (8) | 0.1837 (3) | 0.164 (3) |
| H56 | 0.4207 | 0.5064 | 0.1549 | 0.197* |
| C57 | 0.3952 (6) | 0.6013 (9) | 0.1908 (4) | 0.194 (6) |
| H57 | 0.4259 | 0.6365 | 0.1731 | 0.233* |
| C58 | 0.3503 (7) | 0.6263 (5) | 0.2202 (6) | 0.201 (6) |
| H58 | 0.3326 | 0.6790 | 0.2180 | 0.242* |
| C59 | 0.3244 (5) | 0.5803 (5) | 0.2566 (3) | 0.143 (3) |
| H59 | 0.2993 | 0.6025 | 0.2828 | 0.171* |
| C60 | 0.3359 (6) | 0.5039 (5) | 0.2537 (3) | 0.151 (3) |
| H60 | 0.3142 | 0.4700 | 0.2763 | 0.181* |
| C61 | 0.0062 (7) | 0.5620 (7) | 0.4726 (4) | 0.222 (4) |
| H61A | −0.0425 | 0.5953 | 0.4665 | 0.334* |
| H61B | −0.0045 | 0.5109 | 0.4578 | 0.334* |
| H61C | 0.0536 | 0.5863 | 0.4590 | 0.334* |
| C62 | 0.7834 (2) | 0.1190 (2) | 0.44030 (13) | 0.0563 (9) |
| H62 | 0.7638 | 0.1269 | 0.4078 | 0.068* |
| C63 | 0.8043 (3) | 0.0431 (3) | 0.45651 (18) | 0.0789 (12) |
| H63 | 0.7980 | 0.0002 | 0.4351 | 0.095* |
| C64 | 0.8340 (3) | 0.0312 (3) | 0.5037 (2) | 0.0934 (16) |
| H64 | 0.8495 | −0.0197 | 0.5143 | 0.112* |
| C65 | 0.8413 (3) | 0.0930 (3) | 0.53541 (18) | 0.0909 (16) |
| H65 | 0.8609 | 0.0841 | 0.5678 | 0.109* |
| C66 | 0.8198 (2) | 0.1692 (3) | 0.52009 (13) | 0.0666 (11) |
| H66 | 0.8244 | 0.2112 | 0.5422 | 0.080* |
| C67 | 0.79125 (18) | 0.1828 (2) | 0.47167 (11) | 0.0436 (7) |
| C68 | 0.77291 (18) | 0.26548 (19) | 0.45476 (10) | 0.0407 (7) |
| C69 | 0.77531 (17) | 0.28291 (17) | 0.40020 (10) | 0.0341 (6) |
| C70 | 0.64904 (17) | 0.30409 (17) | 0.29423 (9) | 0.0330 (6) |
| C71 | 0.65591 (18) | 0.32185 (17) | 0.24138 (9) | 0.0344 (6) |
| C72 | 0.73149 (18) | 0.34339 (17) | 0.21996 (10) | 0.0370 (6) |
| C73 | 0.7230 (2) | 0.3544 (2) | 0.16815 (11) | 0.0469 (7) |
| H73 | 0.7710 | 0.3669 | 0.1525 | 0.056* |
| C74 | 0.6486 (2) | 0.3475 (2) | 0.14038 (11) | 0.0544 (9) |
| H74 | 0.6462 | 0.3559 | 0.1068 | 0.065* |
| C75 | 0.5769 (2) | 0.3279 (2) | 0.16269 (11) | 0.0553 (9) |
| C76 | 0.5805 (2) | 0.3146 (2) | 0.21218 (10) | 0.0455 (7) |
| H76 | 0.5317 | 0.3006 | 0.2265 | 0.055* |
Source of material
The ligand was synthesized from the reaction of benzoyl hydrazine (100 mg, 0.60 mmol) and benzoate (50 mg, 0.60 mmol) according to the procedure reported earlier [4]. Yield (0.020 g, 60%).
Experimental details
The C-bound H atoms were geometrically placed (C—H = 0.95–0.98 Å) and refined as riding with U iso(H) = 1.2–1.5 U eq(C). The N-bound H atoms were located in a difference Fourier map but were refined with a distance restraint of N—H = 0.88 ± 0.01 Å, and with U iso(H) set to 1.2 U equiv(N) [1], [2], [3].
Discussion
Aldooxime and acyl compounds both containing multiple O and N atoms can participate in coordination, so the coordination ability of them are very strong, then they can show diversity in their coordination forms. A part of the multinuclear complexes have been one of the research hotspots in recent years [4], [5], [6], [7]. A lot of multinuclear metal complexes with aldooxime and acyl compounds have been reported. In this paper, a three nuclear Ni complex is reported.
The title complex is composed of three Ni atoms, two acylhydrazine ligands, nine pyridines and one methanol molecule. It is the first trinuclear Ni complex with this specific salycilate ligand. The coordination geometry of three Ni atoms is octahedral. The Ni1 atoms is bound to two carbonyl oxygens, two hydrazine nitrogens of two ligands and two nitrogens of pyridines. The bond lengths of Ni1–O are 2.020 and 2.029 Å, which are similar to the corresponding values of the reference [7–10]. For the Ni2 and Ni3 atoms, the octahedral geometry is realized with one carbonyl oxygen atom, one phenolate oxygen atom, one hydrazine nitrogen of ligand and three pyridine nitrogen atoms. The average bond distances of Ni–N and Ni–O for Ni2 and Ni3 are 2.123 and 2.039 Å, which is shorter than the distances of Ni1 atoms. This difference may be attributed to the difference in stereochemistry between the central and terminal Ni atoms. The neighboring Ni–Ni interatomic distances are 4.766 and 4.780 Å, respectively [10].
Funding source: Henan Province Science and Technology Research Project 501100017700
Award Identifier / Grant number: 212102210118
-
Author contributions: All the authors have accepted responsibility for the entire content of this submitted manuscript and approved submission.
-
Research funding: This work was funded by Henan Province Science and Technology Research Project (501100017700) and (212102210118).
-
Conflict of interest statement: The authors declare no conflicts of interest regarding this article.
References
1. Sheldrick, G. M. SMART and SAINT for Windows NT Software Reference Manuals, Version 5.0; Bruker Analytical X–Ray Systems: Madison, WI, 1997.Search in Google Scholar
2. Sheldrick, G. M. SADABS, a Software for Empirical Absorption Correction; University of Göttingen: Göttingen, Germany, 1997.Search in Google Scholar
3. Sheldrick, G. M. SHELXL Reference Manual, Version 5.1; Bruker Analytical X–Ray Systems: Madison, WI, 1997.Search in Google Scholar
4. Lin, S., Yang, M. X., Liu, S. X. Three novel trinuclear zinc(II)/nickel(II) complexes with pentadentate ligands N-nitrogenzoylsalicylhydrazidate. Polyhedron 2007, 26, 4793–4798; https://doi.org/10.1016/j.poly.2007.01.028.Search in Google Scholar
5. Yang, M. X., Lin, S., Chen, L. J., Liu, S. X. Synthesis and crystal structure of the trinuclear nickel(II) complex with Schiff base ligand N-butylsalicylhydrazide. Chin. J. Inorg. Chem. 2003, 19, 433–436.Search in Google Scholar
6. Wu, Q. J., Chen, X. H., Cai, B. Q., Wu, D. Synthesis and crystal structure of trinuclear nickel(II) complex with N-acetylsalicyhydrazide and imidazole ligands. Chin. J. Inorg. Chem. 2012, 28, 2650–2654.Search in Google Scholar
7. Yang, L. G., Wang, X., Luo, D., Liu, N. N., Tian, D. Y. The crystal structure of bis(5-chloro-2-oxido-N-(1-oxidoethylidene)benzohydrazonato)) hexkis(pyridine)trinickel(II)-pyridine, C63H57Cl2N13Ni3O6. Z. Kristallogr. – New Cryst. Struct. 2021, 236, 487–490; https://doi.org/10.1515/ncrs-2020-0600.Search in Google Scholar
8. Chen, X. H., Liu, S. X. Syntheses and crystal structures of two nickel complexes with N–substituted-salicyl ligand. Chin. J. Inorg. Chem. 2005, 21, 15–260.Search in Google Scholar
9. Yang, L. G., Wang, X., Zou, K., Hu, S. G., Liu, X. Z. The crystal structure bis(dimethylsulfoxide-κ1N-bis(μ2-(Z)-3-methyl-2-oxide-N-((Z)-oxide(phenyl) methylene)benzohydrazonato-κ5) trinickel(II)-dimethylsulfoxide(1/2), C48H56N6Ni3O10S4. Z. Kristallogr. N. Cryst. Struct. 2022, 237, 365–367.https://doi.org/10.1515/ncrs-2022-0014.Search in Google Scholar
10. Wang, X., Yang, L. G., Dong, Y. Z., Ye, Y. L., Wang, Y. F. The crystal structure of bis(5-chloro-2-oxide-N-(1-oxidopropylidene)benzohydrazonato-κ5N,O,O′:N′,O″)-octakis(pyridine-κ1N)trinickel(II). C60H56Cl2N12Ni3O6. Z. Kristallogr. N. Cryst. Struct. 2022, 237, 501–504.10.1515/ncrs-2022-0023Search in Google Scholar
© 2022 the author(s), published by De Gruyter, Berlin/Boston
This work is licensed under the Creative Commons Attribution 4.0 International License.
Articles in the same Issue
- Frontmatter
- New Crystal Structures
- Crystal structure of undecacalcium decaarsenide, Ca11As10
- Crystal structure of catena-poly[diiodido-(μ2-1,5-dimethyl-2-phenyl-4-((pyridin-4- ylmethylene)amino)-1,2-dihydro-3H -pyrazol-3-one-κ2 N: O)zinc(II)], C17H16I2N4OZn
- The crystal structure of 5,10,15,20-tetrakis(4-(tert-butyl)phenyl)porphyrin-21,23-diido-κ4 N 4-naphthalocyanido-κ4 N 4-neodymium(IV) - chloroform (1/6) C114H90N12Cl18Nd
- The crystal structure of 1-(4-bromophenyl)-3-(2-chlorobenzyl)urea, C14H12BrClN2O
- Crystal structure of bis[benzyl(methyl)carbamodithioato-κ 2 S,S′]-di-n-butyltin(IV), C26H38N2S4Sn
- Crystal structure of (E)-3-(2-(4-(diethylamino)-2-hydroxystyryl)-3,3-dimethyl-3H-indol-1-ium-1-yl)propane-1-sulfonate – methanol (1/2), C25H32N2O4S⋅2CH3OH
- Synthesis and crystal structure of {(N′,N″-(((ethane-1,2-diylbis(oxy))bis(2,1-phenylene))-bis(methaneylylidene))bis(2-hydroxybenzohydrazonato)-κ6 N 2 O 4}copper(II), C30H24CuN4O6
- The crystal structure of ((E)-2,4-dibromo-6-(((5-(nitro)-2-oxidophenyl)imino)methyl)phenolato-κ 3 N,O,O′)tris(pyridine-κN)manganese(II), C28H21Br2MnN5O4
- Synthesis and crystal structure of 1-((3R,10S,13R,17S)-10,13-dimethyl-3-(p-tolylamino)hexadecahydro-1H-cyclopenta-[a]phenanthren-17-yl)ethan-1-one, C28H41NO
- The crystal structure of 3-nitrobenzene-1,2-dicarboxylic acid—pyrazine-2-carboxamide(1/1), C13H10N4O7
- Crystal structure of poly[tetrakis(μ3-2-aminonicotinato-κ3N,O,O′)-(μ2-oxalato-κ4 O,O′:O″,O′″)-(μ4-oxalato-κ6 N:N′:O,O′:O″,O′″)dicopper(I)-disamarium(III)], [SmCu(C6N2H5O2)2(C2O4)] n
- The crystal structure of 2,3,4-trihydroxybenzoic- acid—pyrazine-2-carboxamide—water (1/1/1), C12H13N3O7
- Crystal structure of N-ethyl-4-[3-(trifluoromethyl)-phenyl]piperazine-1-carbothioamide, C14H18F3N3S
- The crystal structure of 3-anilino-1,4-diphenyl-4H-1,2,4-triazol-1-ium iodide, C20H17N4I
- The crystal structure of (tris(2-benzimidazolylmethyl)amine)-benzoato-copper(II) perchlorate monohydrate, CuC31H28N7O7Cl
- Crystal structure of [2-hydroxy-3-methyl-benzoato-k1 O-triphenyltin(IV)], C26H22O3Sn
- Crystal structure of diaqua-bis(4-(hydroxymethyl)-benzoato-k1 O)zinc(II), C16H18O8Zn
- The crystal structure of dicarbonyl-(N-nitroso-N-oxido-phenylamine-κ 2 O,O)-rhodium(I), C8H5N2O4Rh
- The crystal structure of oxalic acid – 2-ethoxybenzamide (2/1), C20H24N2O8
- The crystal structure of ethyl 7-ethyl-5-methyl-4,7-dihydrotetrazolo[1,5-a]pyrimidine-6-carboxylate, C10H15N5O2
- Crystal structure of poly[(N,N-dimethylacetamide-κO) (μ4-2-nitroisophthalato-κ 4 O:O′:O″:O′″)manganese(II)], C11H10N2O7Mn
- Crystal structure of 14-O-acetyldelcosine, C26H41NO8
- The crystal structure of poly[(1,10-phenanthroline-κ2 N,N′)-(μ 4-2-chlorobenzene-1,3-dicarboxylato-κ5 O:O′:O″:O‴) cadmium(II)] monohydrate, C20H13CdClN2O5
- Crystal structure of propane-1,3-diylbis(diphenylphosphine sulfide) ethanol solvate, C27H26P2S2
- Crystal structure of bis{[(4-diethylamino-2-hydroxy-benzylidene)-hydrazinocarbonylmethyl]-trimethylammonium} tetrabromozincate, C32H54N8O4ZnBr4
- Synthesis and crystal structure of dimethyl 2,2′-(2,5-bis(4-hydroxyphenyl)-2,5-dihydrofuran-3,4-diyl)dibenzoate, C34H30O7
- Synthesis and crystal structure of 2-(2-oxo-2-phenylethyl)-4H-chromen-4-one, C17H12O3
- The crystal structure of tetra(imidazole-κ1 N)zinc(II) μ2-oxido-hexaoxido-divanadium(VI) C12H16N8O6V2Zn
- Crystal structure of S-2-(1-(5-methylpyridin-2-ylamino)octyl)-3-hydroxynaphthalene-1,4-dione, C24H28N2O3
- Crystal structure of bis(μ2-5-chloro-2-oxido-N-(1-oxido-2-oxo-2-phenylethylidene)-benzohydrazonato-κ5 N,O,O′:N′,O′′)-oktakis(pyridine-κ1 N)trinickel(II) – methanol – pyridine (1/1/1) C76H65N13Cl2Ni3O9
- The crystal structure of methyl 3,5-diaminobenzoate, C8H10N2O2
- Crystal structure of 10-(9H-carbazol-9-yl)-5H-dibenzo[a,d][7]annelen-5-one, C27H17NO
- Crystal structure of ethyl 1-(2-hydroxyethyl)-4-((4-methoxyphenyl)amino)-5-oxo-2,5-dihydro-1H-pyrrole-3-carboxylate, C16H20N2O5
- The crystal structure of 1-(4-bromophenyl)-3-cycloheptylurea, C14H19BrN2O
- The crystal structure of 1,4-bis(1,2,3,4,5-pentamethylcyclopenta-2,4-dien-1-yl)-3,6-bis ((4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)methylene)-1,4-dialuminacyclohexane – benzene (1/2), C50H72Al2B2O4
- Crystal structure of bis(μ 3-diphenylphosphinato)-tetrakis(μ 2-diphenylphosphinato)-bis(diphenylphosphinato)-bis(μ 2-hydroxo)dicopper(II)-ditin(IV), C104H100O18P8Cu2Sn2
- Crystal structure of 3-((3,4-dichloroisothiazol-5-yl)methoxy)benzo[d] isothiazole 1,1-dioxide, C11H6Cl2N2O3S2
- Synthesis and crystal structure of 2-(2-(2-fluorophenyl)-2-oxoethyl)-4H-chromen-4-one, C17H11FO3
- The crystal structure of tris(carbonyl)-bis(carbonyl)-[μ-propane-1,2- dithiolato]-(benzyldiphenylphosphine)diiron (Fe—Fe), C27H23Fe2O5PS2
- Crystal structure of 1-(2-(4-chlorophenethyl)-2-hydroxy-3,3-dimethylbutyl)-1H-1,2,4-triazol-4-ium nitrate, C16H23N4O4Cl
- The crystal structure of 3,3′-disulfanediyldi(1H-1,2,4-triazol-5-amine) monohydrate, C4H8N8OS2
- The crystal structure of trans-[bis(4-methylpyridine-κN)bis(quinoline-2-carboxylato- κ 2 N,O)cadmium(II)], C32H26CdN4O4
- The crystal structure of ethyl 2′-hydroxy-4′,6′-dimethoxy-3-(4-methoxynaphthalen-1-yl)-5-oxo-2,3,4,5-tetrahydro-[1,1′-biphenyl]-4-carboxylate, C28H28O7
Articles in the same Issue
- Frontmatter
- New Crystal Structures
- Crystal structure of undecacalcium decaarsenide, Ca11As10
- Crystal structure of catena-poly[diiodido-(μ2-1,5-dimethyl-2-phenyl-4-((pyridin-4- ylmethylene)amino)-1,2-dihydro-3H -pyrazol-3-one-κ2 N: O)zinc(II)], C17H16I2N4OZn
- The crystal structure of 5,10,15,20-tetrakis(4-(tert-butyl)phenyl)porphyrin-21,23-diido-κ4 N 4-naphthalocyanido-κ4 N 4-neodymium(IV) - chloroform (1/6) C114H90N12Cl18Nd
- The crystal structure of 1-(4-bromophenyl)-3-(2-chlorobenzyl)urea, C14H12BrClN2O
- Crystal structure of bis[benzyl(methyl)carbamodithioato-κ 2 S,S′]-di-n-butyltin(IV), C26H38N2S4Sn
- Crystal structure of (E)-3-(2-(4-(diethylamino)-2-hydroxystyryl)-3,3-dimethyl-3H-indol-1-ium-1-yl)propane-1-sulfonate – methanol (1/2), C25H32N2O4S⋅2CH3OH
- Synthesis and crystal structure of {(N′,N″-(((ethane-1,2-diylbis(oxy))bis(2,1-phenylene))-bis(methaneylylidene))bis(2-hydroxybenzohydrazonato)-κ6 N 2 O 4}copper(II), C30H24CuN4O6
- The crystal structure of ((E)-2,4-dibromo-6-(((5-(nitro)-2-oxidophenyl)imino)methyl)phenolato-κ 3 N,O,O′)tris(pyridine-κN)manganese(II), C28H21Br2MnN5O4
- Synthesis and crystal structure of 1-((3R,10S,13R,17S)-10,13-dimethyl-3-(p-tolylamino)hexadecahydro-1H-cyclopenta-[a]phenanthren-17-yl)ethan-1-one, C28H41NO
- The crystal structure of 3-nitrobenzene-1,2-dicarboxylic acid—pyrazine-2-carboxamide(1/1), C13H10N4O7
- Crystal structure of poly[tetrakis(μ3-2-aminonicotinato-κ3N,O,O′)-(μ2-oxalato-κ4 O,O′:O″,O′″)-(μ4-oxalato-κ6 N:N′:O,O′:O″,O′″)dicopper(I)-disamarium(III)], [SmCu(C6N2H5O2)2(C2O4)] n
- The crystal structure of 2,3,4-trihydroxybenzoic- acid—pyrazine-2-carboxamide—water (1/1/1), C12H13N3O7
- Crystal structure of N-ethyl-4-[3-(trifluoromethyl)-phenyl]piperazine-1-carbothioamide, C14H18F3N3S
- The crystal structure of 3-anilino-1,4-diphenyl-4H-1,2,4-triazol-1-ium iodide, C20H17N4I
- The crystal structure of (tris(2-benzimidazolylmethyl)amine)-benzoato-copper(II) perchlorate monohydrate, CuC31H28N7O7Cl
- Crystal structure of [2-hydroxy-3-methyl-benzoato-k1 O-triphenyltin(IV)], C26H22O3Sn
- Crystal structure of diaqua-bis(4-(hydroxymethyl)-benzoato-k1 O)zinc(II), C16H18O8Zn
- The crystal structure of dicarbonyl-(N-nitroso-N-oxido-phenylamine-κ 2 O,O)-rhodium(I), C8H5N2O4Rh
- The crystal structure of oxalic acid – 2-ethoxybenzamide (2/1), C20H24N2O8
- The crystal structure of ethyl 7-ethyl-5-methyl-4,7-dihydrotetrazolo[1,5-a]pyrimidine-6-carboxylate, C10H15N5O2
- Crystal structure of poly[(N,N-dimethylacetamide-κO) (μ4-2-nitroisophthalato-κ 4 O:O′:O″:O′″)manganese(II)], C11H10N2O7Mn
- Crystal structure of 14-O-acetyldelcosine, C26H41NO8
- The crystal structure of poly[(1,10-phenanthroline-κ2 N,N′)-(μ 4-2-chlorobenzene-1,3-dicarboxylato-κ5 O:O′:O″:O‴) cadmium(II)] monohydrate, C20H13CdClN2O5
- Crystal structure of propane-1,3-diylbis(diphenylphosphine sulfide) ethanol solvate, C27H26P2S2
- Crystal structure of bis{[(4-diethylamino-2-hydroxy-benzylidene)-hydrazinocarbonylmethyl]-trimethylammonium} tetrabromozincate, C32H54N8O4ZnBr4
- Synthesis and crystal structure of dimethyl 2,2′-(2,5-bis(4-hydroxyphenyl)-2,5-dihydrofuran-3,4-diyl)dibenzoate, C34H30O7
- Synthesis and crystal structure of 2-(2-oxo-2-phenylethyl)-4H-chromen-4-one, C17H12O3
- The crystal structure of tetra(imidazole-κ1 N)zinc(II) μ2-oxido-hexaoxido-divanadium(VI) C12H16N8O6V2Zn
- Crystal structure of S-2-(1-(5-methylpyridin-2-ylamino)octyl)-3-hydroxynaphthalene-1,4-dione, C24H28N2O3
- Crystal structure of bis(μ2-5-chloro-2-oxido-N-(1-oxido-2-oxo-2-phenylethylidene)-benzohydrazonato-κ5 N,O,O′:N′,O′′)-oktakis(pyridine-κ1 N)trinickel(II) – methanol – pyridine (1/1/1) C76H65N13Cl2Ni3O9
- The crystal structure of methyl 3,5-diaminobenzoate, C8H10N2O2
- Crystal structure of 10-(9H-carbazol-9-yl)-5H-dibenzo[a,d][7]annelen-5-one, C27H17NO
- Crystal structure of ethyl 1-(2-hydroxyethyl)-4-((4-methoxyphenyl)amino)-5-oxo-2,5-dihydro-1H-pyrrole-3-carboxylate, C16H20N2O5
- The crystal structure of 1-(4-bromophenyl)-3-cycloheptylurea, C14H19BrN2O
- The crystal structure of 1,4-bis(1,2,3,4,5-pentamethylcyclopenta-2,4-dien-1-yl)-3,6-bis ((4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)methylene)-1,4-dialuminacyclohexane – benzene (1/2), C50H72Al2B2O4
- Crystal structure of bis(μ 3-diphenylphosphinato)-tetrakis(μ 2-diphenylphosphinato)-bis(diphenylphosphinato)-bis(μ 2-hydroxo)dicopper(II)-ditin(IV), C104H100O18P8Cu2Sn2
- Crystal structure of 3-((3,4-dichloroisothiazol-5-yl)methoxy)benzo[d] isothiazole 1,1-dioxide, C11H6Cl2N2O3S2
- Synthesis and crystal structure of 2-(2-(2-fluorophenyl)-2-oxoethyl)-4H-chromen-4-one, C17H11FO3
- The crystal structure of tris(carbonyl)-bis(carbonyl)-[μ-propane-1,2- dithiolato]-(benzyldiphenylphosphine)diiron (Fe—Fe), C27H23Fe2O5PS2
- Crystal structure of 1-(2-(4-chlorophenethyl)-2-hydroxy-3,3-dimethylbutyl)-1H-1,2,4-triazol-4-ium nitrate, C16H23N4O4Cl
- The crystal structure of 3,3′-disulfanediyldi(1H-1,2,4-triazol-5-amine) monohydrate, C4H8N8OS2
- The crystal structure of trans-[bis(4-methylpyridine-κN)bis(quinoline-2-carboxylato- κ 2 N,O)cadmium(II)], C32H26CdN4O4
- The crystal structure of ethyl 2′-hydroxy-4′,6′-dimethoxy-3-(4-methoxynaphthalen-1-yl)-5-oxo-2,3,4,5-tetrahydro-[1,1′-biphenyl]-4-carboxylate, C28H28O7

