Abstract
C50H72Al2B2O4, triclinic,
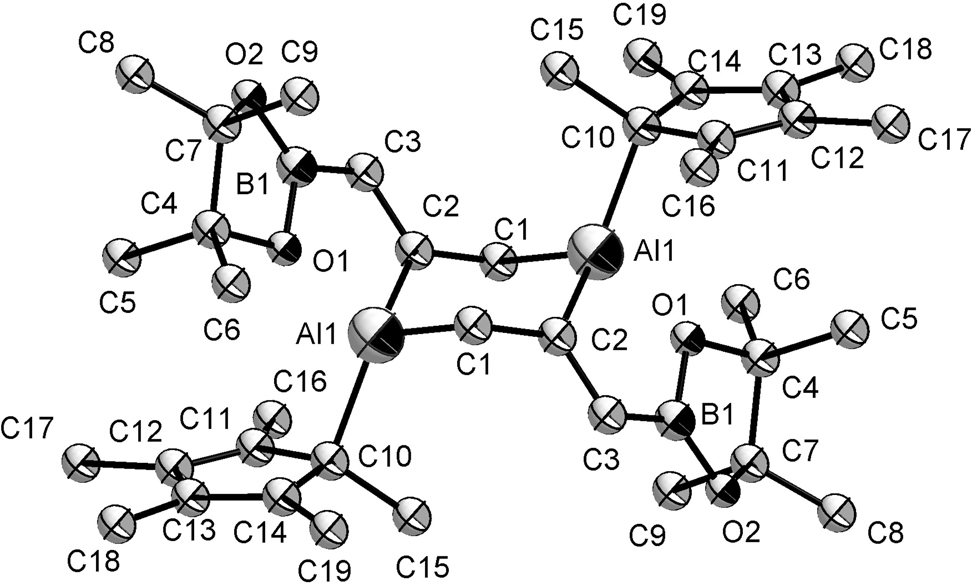
The molecular structure is shown in the figure. Table 1 contains crystallographic data and Table 2 contains the list of the atoms including atomic coordinates and displacement parameters.
Data collection and handling.
| Crystal: | Colourless needle |
| Size: | 0.42 × 0.12 × 0.10 mm |
| Wavelength: | Mo Kα radiation (0.71073 Å) |
| μ: | 0.10 mm−1 |
| Diffractometer, scan mode: | Bruker D8 Venture, φ and ω |
| θ max, completeness: | 27.2°, >99% |
| N(hkl)measured, N(hkl)unique, R int: | 42,277, 5315, 0.033 |
| Criterion for I obs, N(hkl)gt: | I obs > 2σ(I obs), 4777 |
| N(param)refined: | 271 |
| Programs: | Bruker [1], Olex2 [2], SHELX [3, 4] |
Fractional atomic coordinates and isotropic or equivalent isotropic displacement parameters (Å2).
| Atom | x | y | z | U iso*/U eq |
|---|---|---|---|---|
| Al1 | 0.39619 (4) | 0.42606 (3) | 0.56499 (3) | 0.02037 (10) |
| O1 | 0.40641 (9) | 0.52565 (8) | 0.71435 (6) | 0.02280 (17) |
| O2 | 0.55381 (10) | 0.70400 (9) | 0.86461 (7) | 0.0305 (2) |
| C1 | 0.71920 (13) | 0.47889 (12) | 0.51828 (9) | 0.0248 (2) |
| H1A | 0.713042 | 0.384922 | 0.471040 | 0.030* |
| H1B | 0.832323 | 0.520320 | 0.552821 | 0.030* |
| C2 | 0.62896 (13) | 0.49504 (11) | 0.60013 (9) | 0.0224 (2) |
| C3 | 0.68472 (13) | 0.58211 (12) | 0.70641 (10) | 0.0260 (2) |
| H3 | 0.793170 | 0.622626 | 0.738510 | 0.031* |
| C4 | 0.30514 (14) | 0.56349 (12) | 0.79205 (9) | 0.0250 (2) |
| C5 | 0.32607 (17) | 0.47445 (13) | 0.85167 (10) | 0.0344 (3) |
| H5A | 0.294593 | 0.382359 | 0.799196 | 0.052* |
| H5B | 0.259197 | 0.493902 | 0.902471 | 0.052* |
| H5C | 0.437939 | 0.489603 | 0.891843 | 0.052* |
| C6 | 0.13392 (15) | 0.54442 (14) | 0.72877 (11) | 0.0344 (3) |
| H6A | 0.127912 | 0.591054 | 0.681365 | 0.052* |
| H6B | 0.068583 | 0.579464 | 0.779332 | 0.052* |
| H6C | 0.094389 | 0.450404 | 0.684642 | 0.052* |
| C7 | 0.38627 (15) | 0.70880 (12) | 0.86420 (10) | 0.0288 (3) |
| C8 | 0.37385 (18) | 0.75889 (15) | 0.98023 (11) | 0.0412 (3) |
| H8A | 0.421626 | 0.703222 | 1.014249 | 0.062* |
| H8B | 0.261522 | 0.756177 | 0.980647 | 0.062* |
| H8C | 0.430377 | 0.849663 | 1.020791 | 0.062* |
| C9 | 0.33304 (19) | 0.80330 (14) | 0.81371 (13) | 0.0421 (3) |
| H9A | 0.403378 | 0.888684 | 0.854125 | 0.063* |
| H9B | 0.223431 | 0.814497 | 0.816517 | 0.063* |
| H9C | 0.338048 | 0.767517 | 0.737991 | 0.063* |
| C10 | 0.32306 (14) | 0.23031 (11) | 0.50959 (9) | 0.0252 (2) |
| C11 | 0.42000 (14) | 0.18778 (11) | 0.58920 (10) | 0.0270 (2) |
| C12 | 0.32493 (16) | 0.15647 (12) | 0.64474 (10) | 0.0299 (3) |
| C13 | 0.16306 (15) | 0.17256 (12) | 0.60355 (11) | 0.0311 (3) |
| C14 | 0.15912 (14) | 0.21131 (11) | 0.52162 (10) | 0.0286 (3) |
| C15 | 0.3424 (2) | 0.16660 (13) | 0.39369 (11) | 0.0389 (3) |
| H15A | 0.275711 | 0.201585 | 0.347716 | 0.058* |
| H15B | 0.309708 | 0.071074 | 0.364405 | 0.058* |
| H15C | 0.454167 | 0.186522 | 0.395082 | 0.058* |
| C16 | 0.59319 (16) | 0.17856 (13) | 0.59948 (13) | 0.0386 (3) |
| H16A | 0.604346 | 0.103379 | 0.536336 | 0.058* |
| H16B | 0.638630 | 0.166699 | 0.665697 | 0.058* |
| H16C | 0.649648 | 0.259587 | 0.603117 | 0.058* |
| C17 | 0.3722 (2) | 0.11036 (16) | 0.73379 (13) | 0.0485 (4) |
| H17A | 0.335218 | 0.164231 | 0.796264 | 0.073* |
| H17B | 0.488414 | 0.118726 | 0.755526 | 0.073* |
| H17C | 0.323853 | 0.018062 | 0.707637 | 0.073* |
| C18 | 0.0254 (2) | 0.14867 (16) | 0.64662 (14) | 0.0492 (4) |
| H18A | 0.013440 | 0.058537 | 0.641626 | 0.074* |
| H18B | −0.072458 | 0.160984 | 0.603684 | 0.074* |
| H18C | 0.045442 | 0.210932 | 0.722610 | 0.074* |
| C19 | 0.01347 (17) | 0.22867 (15) | 0.45052 (13) | 0.0438 (4) |
| H19A | −0.000399 | 0.321058 | 0.480016 | 0.066* |
| H19B | −0.080318 | 0.173244 | 0.448215 | 0.066* |
| H19C | 0.026205 | 0.203002 | 0.376983 | 0.066* |
| B1 | 0.55911 (16) | 0.60839 (13) | 0.76847 (11) | 0.0252 (3) |
| C20 | 0.0721 (2) | 0.41111 (17) | 1.03287 (13) | 0.0473 (4) |
| H20 | 0.121595 | 0.349718 | 1.055378 | 0.057* |
| C21 | −0.0423 (2) | 0.36710 (17) | 0.93515 (13) | 0.0459 (4) |
| H21 | −0.071433 | 0.275517 | 0.890525 | 0.055* |
| C22 | −0.1146 (2) | 0.45620 (18) | 0.90213 (13) | 0.0474 (4) |
| H22 | −0.193308 | 0.425999 | 0.834842 | 0.057* |
| C23 | 0.9828 (2) | 1.00279 (16) | 0.89980 (14) | 0.0519 (4) |
| H23 | 0.971179 | 1.005006 | 0.830782 | 0.062* |
| C24 | 0.8590 (2) | 0.93987 (15) | 0.91984 (14) | 0.0498 (4) |
| H24 | 0.762560 | 0.898574 | 0.864584 | 0.060* |
| C25 | 0.8763 (2) | 0.93746 (15) | 1.01951 (15) | 0.0510 (4) |
| H25 | 0.791365 | 0.894679 | 1.033523 | 0.061* |
Source of material
The synthesis of [Cp*Al]4(Cp* = 1,2,3,4,5-pentamethylcyclopenta-2,4-dien-1-yl) was performed according to the previously reported procedure [5]. A mixture of [Cp*Al]4 and Bpin–CH=C=CH2 (Bpin = 4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl) in C6D6 in a dried J–Young Tube was heated at 80 °C under argon for 2 h. The compound was crystallized from a 1/10 mixture of hexane/C6D6 to afford colorless crystals.
Experimental details
A suitable crystal was selected and mounted on a Bruker APEX–II CCD diffractometer. The crystal was kept at 170.0 K during data collection. Using OLEX2, the structure was solved with the ShelXT structure solution program using Intrinsic Phasing and refined with the ShelXT refinement package using Least Squares minimisation.
Comment
Due to transition metal-like reactivities, the development of low-valent and low-coordinated Al(I) compounds with high reactivity has made remarkable progress in recent years. In 1991, Schnöckel et al. reported that the first monovalent aluminum compound [Cp*Al]4 has successfully been synthesized, which opened a new chapter of aluminum chemistry [6], [7], [8]. Subsequent studies revealed that [Cp*Al]4 not only performs as a neutral ligand to stabilize transition metals [9], [10], [11], [12], but also exhibits high reactivity to activate small molecules, such as O2, S8, Se, Te, N2O, P4, and CO2 [13], [14], [15], [16]. Since diene is isoelectronic with CO2, we demonstrated that the C=C bond activation of Bpin–CH=C=CH2 by [Cp*Al]4 can be achieved to obtain the title compound. The title compound is a product of the activation of a C=C double bond in BPin–CH=C=CH2 by [Cp*Al]4. The structure of the product contains an Al–C–C–Al–C–C six-membered ring, in which the C–C bond length is 1.4792(15) Å and the Al–C bond length is 2.0160(12) Å. The C–C bond outside the ring has a bond length of 1.3672(17) Å. These bond lengths are all within the expected range.
Funding source: Zhejiang Provincial Natural Science Foundation
Award Identifier / Grant number: LQ20B010007
-
Author contributions: All the authors have accepted responsibility for the entire content of this submitted manuscript and approved submission.
-
Research funding: This work is granted by Zhejiang Provincial Natural Science Foundation (LQ20B010007).
-
Conflict of interest statement: The authors declare no conflicts of interest regarding this article.
References
1. Bruker . SAINT, APEX2 and SADABS; Bruker AXS Inc.: Madison, Wisconsin, USA, 2009.Search in Google Scholar
2. Dolomanov, O. V., Bourhis, L. J., Gildea, R. J., Howard, J. A. K., Puschmann, H. OLEX2: a complete structure solution, refinement and analysis program. J. Appl. Crystallogr. 2009, 42, 339–341; https://doi.org/10.1107/s0021889808042726.Search in Google Scholar
3. Sheldrick, G. M. SHELXTL – Integrated space-group and crystal-structure determination. Acta Crystallogr. 2015, A71, 3–8; https://doi.org/10.1107/s2053273314026370.Search in Google Scholar PubMed PubMed Central
4. Sheldrick, G. M. Crystal structure refinement with SHELXL. Acta Crystallogr. 2015, C71, 3–8; https://doi.org/10.1107/s2053229614024218.Search in Google Scholar PubMed PubMed Central
5. Ganesamoorthy, C., Loerke, S., Gemel, C., Jerabek, P., Winter, M., Frenking, G., Fischer, R. A. Reductive elimination: a pathway to low-valent aluminium species. Chem. Commun. 2013, 49, 2858–2860; https://doi.org/10.1039/c3cc38584a.Search in Google Scholar PubMed
6. Dohmeier, C., Robl, C., Tacke, M., Schnöckel, H. The tetrameric aluminum(I) compound [{Al(η5-C5Me5)}4]. Angew. Chem. Int. Ed. Engl. 1991, 30, 564–565; https://doi.org/10.1002/anie.199105641.Search in Google Scholar
7. Zhong, M., Sinhababu, S., Roesky, H. W. The unique beta-diketiminate ligand in aluminum(I) and gallium(I) chemistry. Dalton Trans. 2020, 49, 1351–1364; https://doi.org/10.1039/c9dt04763h.Search in Google Scholar PubMed
8. Hicks, J., Vasko, P., Goicoechea, J. M., Aldridge, S. The aluminyl anion: a new generation of aluminium nucleophile. Angew. Chem. Int. Ed. 2021, 60, 1702–1713; https://doi.org/10.1002/ange.202007530.Search in Google Scholar
9. Steinke, T., Gemel, C., Cokoja, M., Winter, M., Fischer, R. A. AlCp* as a directing ligand: C–H and Si–H bond activation at the reactive intermediate [Ni(AlCp*)3]. Angew. Chem. Int. Ed. 2004, 43, 2299–2302; https://doi.org/10.1002/anie.200353114.Search in Google Scholar PubMed
10. Steinke, T., Gemel, C., Winter, M., Fischer, R. A. The clusters [Ma(ECp*)b] (M=Pd, Pt; E=Al, Ga, In): structures, fluxionality, and ligand exchange reactions. Chem. Eur J. 2005, 11, 1636–1646; https://doi.org/10.1002/chem.200400959.Search in Google Scholar PubMed
11. Steinke, T., Cokoja, M., Gemel, C., Kempter, A., Krapp, A., Frenking, G., Zenneck, U., Fischer, R. A. C–H activated isomers of [M(AlCp*)5] (M=Fe, Ru). Angew. Chem. Int. Ed. 2005, 44, 2943–2946; https://doi.org/10.1002/anie.200462834.Search in Google Scholar PubMed
12. Minasian, S. G., Krinsky, J. L., Williams, V. A., Arnold, J. A heterobimetallic complex with an unsupported uranium(III)-aluminum(I) bond: (CpSiMe3)3U–AlCp* (Cp* = C5Me5). J. Am. Chem. Soc. 2008, 130, 10086–10087; https://doi.org/10.1021/ja8042382.Search in Google Scholar PubMed
13. Schulz, S., Roesky, H. W., Koch, H. J., Sheldrick, G. M., Stalke, D., Kuhn, A. A simple synthesis of [(Cp*Al)4] and its conversion to the heterocubanes [(Cp*AlSe)4] and [(Cp*AlTe)4] (Cp* = η5–C5(CH3)5). Angew. Chem. Int. Ed. Engl. 1993, 32, 1729–1731; https://doi.org/10.1002/anie.199317291.Search in Google Scholar
14. Stelzer, A. C., Hrobárik, P., Braun, T., Kaupp, M., Braun-Cula, B. Completing the heterocubane family [Cp*AlE]4 (E = O, S, Se, and Te) by selective oxygenation and sulfuration of [Cp*Al]4: density functional theory calculations of [Cp*AlE]4 and reactivity of [Cp*AlO]4 toward hydrolysis. Inorg. Chem. 2016, 55, 4915–4923; https://doi.org/10.1021/acs.inorgchem.6b00462.Search in Google Scholar PubMed
15. Dohmeier, C., Schnöckel, H., Robl, C., Schneider, U., Ahlrichs, R. [P4(Cp*Al)6]: a compound with an unusual P4Al6 cage structure. Angew. Chem. Int. Ed. Engl. 1994, 33, 199–200; https://doi.org/10.1002/anie.199401991.Search in Google Scholar
16. Wang, B., Zhuang, Y., Tu, D., Shen, L., Zhang, Z. F., Su, M. D., Li, Y., Wu, D., Roesky, H. W. Deoxygenating reduction of CO2 by [Cp*Al]4 to form a (Al3O2C)2 cluster featuring two ketene moieties. Inorg. Chem. 2022, 61, 14500–14505; https://doi.org/10.1021/acs.inorgchem.2c01587.Search in Google Scholar PubMed
© 2022 the author(s), published by De Gruyter, Berlin/Boston
This work is licensed under the Creative Commons Attribution 4.0 International License.
Articles in the same Issue
- Frontmatter
- New Crystal Structures
- Crystal structure of undecacalcium decaarsenide, Ca11As10
- Crystal structure of catena-poly[diiodido-(μ2-1,5-dimethyl-2-phenyl-4-((pyridin-4- ylmethylene)amino)-1,2-dihydro-3H -pyrazol-3-one-κ2 N: O)zinc(II)], C17H16I2N4OZn
- The crystal structure of 5,10,15,20-tetrakis(4-(tert-butyl)phenyl)porphyrin-21,23-diido-κ4 N 4-naphthalocyanido-κ4 N 4-neodymium(IV) - chloroform (1/6) C114H90N12Cl18Nd
- The crystal structure of 1-(4-bromophenyl)-3-(2-chlorobenzyl)urea, C14H12BrClN2O
- Crystal structure of bis[benzyl(methyl)carbamodithioato-κ 2 S,S′]-di-n-butyltin(IV), C26H38N2S4Sn
- Crystal structure of (E)-3-(2-(4-(diethylamino)-2-hydroxystyryl)-3,3-dimethyl-3H-indol-1-ium-1-yl)propane-1-sulfonate – methanol (1/2), C25H32N2O4S⋅2CH3OH
- Synthesis and crystal structure of {(N′,N″-(((ethane-1,2-diylbis(oxy))bis(2,1-phenylene))-bis(methaneylylidene))bis(2-hydroxybenzohydrazonato)-κ6 N 2 O 4}copper(II), C30H24CuN4O6
- The crystal structure of ((E)-2,4-dibromo-6-(((5-(nitro)-2-oxidophenyl)imino)methyl)phenolato-κ 3 N,O,O′)tris(pyridine-κN)manganese(II), C28H21Br2MnN5O4
- Synthesis and crystal structure of 1-((3R,10S,13R,17S)-10,13-dimethyl-3-(p-tolylamino)hexadecahydro-1H-cyclopenta-[a]phenanthren-17-yl)ethan-1-one, C28H41NO
- The crystal structure of 3-nitrobenzene-1,2-dicarboxylic acid—pyrazine-2-carboxamide(1/1), C13H10N4O7
- Crystal structure of poly[tetrakis(μ3-2-aminonicotinato-κ3N,O,O′)-(μ2-oxalato-κ4 O,O′:O″,O′″)-(μ4-oxalato-κ6 N:N′:O,O′:O″,O′″)dicopper(I)-disamarium(III)], [SmCu(C6N2H5O2)2(C2O4)] n
- The crystal structure of 2,3,4-trihydroxybenzoic- acid—pyrazine-2-carboxamide—water (1/1/1), C12H13N3O7
- Crystal structure of N-ethyl-4-[3-(trifluoromethyl)-phenyl]piperazine-1-carbothioamide, C14H18F3N3S
- The crystal structure of 3-anilino-1,4-diphenyl-4H-1,2,4-triazol-1-ium iodide, C20H17N4I
- The crystal structure of (tris(2-benzimidazolylmethyl)amine)-benzoato-copper(II) perchlorate monohydrate, CuC31H28N7O7Cl
- Crystal structure of [2-hydroxy-3-methyl-benzoato-k1 O-triphenyltin(IV)], C26H22O3Sn
- Crystal structure of diaqua-bis(4-(hydroxymethyl)-benzoato-k1 O)zinc(II), C16H18O8Zn
- The crystal structure of dicarbonyl-(N-nitroso-N-oxido-phenylamine-κ 2 O,O)-rhodium(I), C8H5N2O4Rh
- The crystal structure of oxalic acid – 2-ethoxybenzamide (2/1), C20H24N2O8
- The crystal structure of ethyl 7-ethyl-5-methyl-4,7-dihydrotetrazolo[1,5-a]pyrimidine-6-carboxylate, C10H15N5O2
- Crystal structure of poly[(N,N-dimethylacetamide-κO) (μ4-2-nitroisophthalato-κ 4 O:O′:O″:O′″)manganese(II)], C11H10N2O7Mn
- Crystal structure of 14-O-acetyldelcosine, C26H41NO8
- The crystal structure of poly[(1,10-phenanthroline-κ2 N,N′)-(μ 4-2-chlorobenzene-1,3-dicarboxylato-κ5 O:O′:O″:O‴) cadmium(II)] monohydrate, C20H13CdClN2O5
- Crystal structure of propane-1,3-diylbis(diphenylphosphine sulfide) ethanol solvate, C27H26P2S2
- Crystal structure of bis{[(4-diethylamino-2-hydroxy-benzylidene)-hydrazinocarbonylmethyl]-trimethylammonium} tetrabromozincate, C32H54N8O4ZnBr4
- Synthesis and crystal structure of dimethyl 2,2′-(2,5-bis(4-hydroxyphenyl)-2,5-dihydrofuran-3,4-diyl)dibenzoate, C34H30O7
- Synthesis and crystal structure of 2-(2-oxo-2-phenylethyl)-4H-chromen-4-one, C17H12O3
- The crystal structure of tetra(imidazole-κ1 N)zinc(II) μ2-oxido-hexaoxido-divanadium(VI) C12H16N8O6V2Zn
- Crystal structure of S-2-(1-(5-methylpyridin-2-ylamino)octyl)-3-hydroxynaphthalene-1,4-dione, C24H28N2O3
- Crystal structure of bis(μ2-5-chloro-2-oxido-N-(1-oxido-2-oxo-2-phenylethylidene)-benzohydrazonato-κ5 N,O,O′:N′,O′′)-oktakis(pyridine-κ1 N)trinickel(II) – methanol – pyridine (1/1/1) C76H65N13Cl2Ni3O9
- The crystal structure of methyl 3,5-diaminobenzoate, C8H10N2O2
- Crystal structure of 10-(9H-carbazol-9-yl)-5H-dibenzo[a,d][7]annelen-5-one, C27H17NO
- Crystal structure of ethyl 1-(2-hydroxyethyl)-4-((4-methoxyphenyl)amino)-5-oxo-2,5-dihydro-1H-pyrrole-3-carboxylate, C16H20N2O5
- The crystal structure of 1-(4-bromophenyl)-3-cycloheptylurea, C14H19BrN2O
- The crystal structure of 1,4-bis(1,2,3,4,5-pentamethylcyclopenta-2,4-dien-1-yl)-3,6-bis ((4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)methylene)-1,4-dialuminacyclohexane – benzene (1/2), C50H72Al2B2O4
- Crystal structure of bis(μ 3-diphenylphosphinato)-tetrakis(μ 2-diphenylphosphinato)-bis(diphenylphosphinato)-bis(μ 2-hydroxo)dicopper(II)-ditin(IV), C104H100O18P8Cu2Sn2
- Crystal structure of 3-((3,4-dichloroisothiazol-5-yl)methoxy)benzo[d] isothiazole 1,1-dioxide, C11H6Cl2N2O3S2
- Synthesis and crystal structure of 2-(2-(2-fluorophenyl)-2-oxoethyl)-4H-chromen-4-one, C17H11FO3
- The crystal structure of tris(carbonyl)-bis(carbonyl)-[μ-propane-1,2- dithiolato]-(benzyldiphenylphosphine)diiron (Fe—Fe), C27H23Fe2O5PS2
- Crystal structure of 1-(2-(4-chlorophenethyl)-2-hydroxy-3,3-dimethylbutyl)-1H-1,2,4-triazol-4-ium nitrate, C16H23N4O4Cl
- The crystal structure of 3,3′-disulfanediyldi(1H-1,2,4-triazol-5-amine) monohydrate, C4H8N8OS2
- The crystal structure of trans-[bis(4-methylpyridine-κN)bis(quinoline-2-carboxylato- κ 2 N,O)cadmium(II)], C32H26CdN4O4
- The crystal structure of ethyl 2′-hydroxy-4′,6′-dimethoxy-3-(4-methoxynaphthalen-1-yl)-5-oxo-2,3,4,5-tetrahydro-[1,1′-biphenyl]-4-carboxylate, C28H28O7
Articles in the same Issue
- Frontmatter
- New Crystal Structures
- Crystal structure of undecacalcium decaarsenide, Ca11As10
- Crystal structure of catena-poly[diiodido-(μ2-1,5-dimethyl-2-phenyl-4-((pyridin-4- ylmethylene)amino)-1,2-dihydro-3H -pyrazol-3-one-κ2 N: O)zinc(II)], C17H16I2N4OZn
- The crystal structure of 5,10,15,20-tetrakis(4-(tert-butyl)phenyl)porphyrin-21,23-diido-κ4 N 4-naphthalocyanido-κ4 N 4-neodymium(IV) - chloroform (1/6) C114H90N12Cl18Nd
- The crystal structure of 1-(4-bromophenyl)-3-(2-chlorobenzyl)urea, C14H12BrClN2O
- Crystal structure of bis[benzyl(methyl)carbamodithioato-κ 2 S,S′]-di-n-butyltin(IV), C26H38N2S4Sn
- Crystal structure of (E)-3-(2-(4-(diethylamino)-2-hydroxystyryl)-3,3-dimethyl-3H-indol-1-ium-1-yl)propane-1-sulfonate – methanol (1/2), C25H32N2O4S⋅2CH3OH
- Synthesis and crystal structure of {(N′,N″-(((ethane-1,2-diylbis(oxy))bis(2,1-phenylene))-bis(methaneylylidene))bis(2-hydroxybenzohydrazonato)-κ6 N 2 O 4}copper(II), C30H24CuN4O6
- The crystal structure of ((E)-2,4-dibromo-6-(((5-(nitro)-2-oxidophenyl)imino)methyl)phenolato-κ 3 N,O,O′)tris(pyridine-κN)manganese(II), C28H21Br2MnN5O4
- Synthesis and crystal structure of 1-((3R,10S,13R,17S)-10,13-dimethyl-3-(p-tolylamino)hexadecahydro-1H-cyclopenta-[a]phenanthren-17-yl)ethan-1-one, C28H41NO
- The crystal structure of 3-nitrobenzene-1,2-dicarboxylic acid—pyrazine-2-carboxamide(1/1), C13H10N4O7
- Crystal structure of poly[tetrakis(μ3-2-aminonicotinato-κ3N,O,O′)-(μ2-oxalato-κ4 O,O′:O″,O′″)-(μ4-oxalato-κ6 N:N′:O,O′:O″,O′″)dicopper(I)-disamarium(III)], [SmCu(C6N2H5O2)2(C2O4)] n
- The crystal structure of 2,3,4-trihydroxybenzoic- acid—pyrazine-2-carboxamide—water (1/1/1), C12H13N3O7
- Crystal structure of N-ethyl-4-[3-(trifluoromethyl)-phenyl]piperazine-1-carbothioamide, C14H18F3N3S
- The crystal structure of 3-anilino-1,4-diphenyl-4H-1,2,4-triazol-1-ium iodide, C20H17N4I
- The crystal structure of (tris(2-benzimidazolylmethyl)amine)-benzoato-copper(II) perchlorate monohydrate, CuC31H28N7O7Cl
- Crystal structure of [2-hydroxy-3-methyl-benzoato-k1 O-triphenyltin(IV)], C26H22O3Sn
- Crystal structure of diaqua-bis(4-(hydroxymethyl)-benzoato-k1 O)zinc(II), C16H18O8Zn
- The crystal structure of dicarbonyl-(N-nitroso-N-oxido-phenylamine-κ 2 O,O)-rhodium(I), C8H5N2O4Rh
- The crystal structure of oxalic acid – 2-ethoxybenzamide (2/1), C20H24N2O8
- The crystal structure of ethyl 7-ethyl-5-methyl-4,7-dihydrotetrazolo[1,5-a]pyrimidine-6-carboxylate, C10H15N5O2
- Crystal structure of poly[(N,N-dimethylacetamide-κO) (μ4-2-nitroisophthalato-κ 4 O:O′:O″:O′″)manganese(II)], C11H10N2O7Mn
- Crystal structure of 14-O-acetyldelcosine, C26H41NO8
- The crystal structure of poly[(1,10-phenanthroline-κ2 N,N′)-(μ 4-2-chlorobenzene-1,3-dicarboxylato-κ5 O:O′:O″:O‴) cadmium(II)] monohydrate, C20H13CdClN2O5
- Crystal structure of propane-1,3-diylbis(diphenylphosphine sulfide) ethanol solvate, C27H26P2S2
- Crystal structure of bis{[(4-diethylamino-2-hydroxy-benzylidene)-hydrazinocarbonylmethyl]-trimethylammonium} tetrabromozincate, C32H54N8O4ZnBr4
- Synthesis and crystal structure of dimethyl 2,2′-(2,5-bis(4-hydroxyphenyl)-2,5-dihydrofuran-3,4-diyl)dibenzoate, C34H30O7
- Synthesis and crystal structure of 2-(2-oxo-2-phenylethyl)-4H-chromen-4-one, C17H12O3
- The crystal structure of tetra(imidazole-κ1 N)zinc(II) μ2-oxido-hexaoxido-divanadium(VI) C12H16N8O6V2Zn
- Crystal structure of S-2-(1-(5-methylpyridin-2-ylamino)octyl)-3-hydroxynaphthalene-1,4-dione, C24H28N2O3
- Crystal structure of bis(μ2-5-chloro-2-oxido-N-(1-oxido-2-oxo-2-phenylethylidene)-benzohydrazonato-κ5 N,O,O′:N′,O′′)-oktakis(pyridine-κ1 N)trinickel(II) – methanol – pyridine (1/1/1) C76H65N13Cl2Ni3O9
- The crystal structure of methyl 3,5-diaminobenzoate, C8H10N2O2
- Crystal structure of 10-(9H-carbazol-9-yl)-5H-dibenzo[a,d][7]annelen-5-one, C27H17NO
- Crystal structure of ethyl 1-(2-hydroxyethyl)-4-((4-methoxyphenyl)amino)-5-oxo-2,5-dihydro-1H-pyrrole-3-carboxylate, C16H20N2O5
- The crystal structure of 1-(4-bromophenyl)-3-cycloheptylurea, C14H19BrN2O
- The crystal structure of 1,4-bis(1,2,3,4,5-pentamethylcyclopenta-2,4-dien-1-yl)-3,6-bis ((4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)methylene)-1,4-dialuminacyclohexane – benzene (1/2), C50H72Al2B2O4
- Crystal structure of bis(μ 3-diphenylphosphinato)-tetrakis(μ 2-diphenylphosphinato)-bis(diphenylphosphinato)-bis(μ 2-hydroxo)dicopper(II)-ditin(IV), C104H100O18P8Cu2Sn2
- Crystal structure of 3-((3,4-dichloroisothiazol-5-yl)methoxy)benzo[d] isothiazole 1,1-dioxide, C11H6Cl2N2O3S2
- Synthesis and crystal structure of 2-(2-(2-fluorophenyl)-2-oxoethyl)-4H-chromen-4-one, C17H11FO3
- The crystal structure of tris(carbonyl)-bis(carbonyl)-[μ-propane-1,2- dithiolato]-(benzyldiphenylphosphine)diiron (Fe—Fe), C27H23Fe2O5PS2
- Crystal structure of 1-(2-(4-chlorophenethyl)-2-hydroxy-3,3-dimethylbutyl)-1H-1,2,4-triazol-4-ium nitrate, C16H23N4O4Cl
- The crystal structure of 3,3′-disulfanediyldi(1H-1,2,4-triazol-5-amine) monohydrate, C4H8N8OS2
- The crystal structure of trans-[bis(4-methylpyridine-κN)bis(quinoline-2-carboxylato- κ 2 N,O)cadmium(II)], C32H26CdN4O4
- The crystal structure of ethyl 2′-hydroxy-4′,6′-dimethoxy-3-(4-methoxynaphthalen-1-yl)-5-oxo-2,3,4,5-tetrahydro-[1,1′-biphenyl]-4-carboxylate, C28H28O7

