Abstract
This study presents an eco-friendly approach for synthesizing chitosan-coated superparamagnetic iron oxide nanoparticles (Ch-SPIONs) using Carica papaya bark extract as a green reducing and stabilizing agent. The phytochemicals present in the bark extract facilitate the formation of SPIONs with well-defined magnetic characteristics. Comprehensive characterization through UV-visible spectroscopy, X-ray diffraction, scanning and transmission electron microscopy, energy-dispersive X-ray spectroscopy, and Fourier-transform infrared spectroscopy confirmed the successful synthesis of Ch-SPIONs. The nanoparticles exhibited an average size range of 50–80 nm with a rectangular morphology and a UV-vis absorption peak at 300 nm. The antioxidant potential of Ch-SPIONs was evaluated, revealing concentration-dependent scavenging activity that surpassed standard antioxidants at higher concentrations. Antibacterial assays demonstrated significant efficacy against Klebsiella pneumoniae (15 mm inhibition zone) and Staphylococcus aureus (20 mm inhibition zone). Furthermore, in vitro cytotoxicity studies on HeLa cervical cancer cells showed dose-dependent cell death, with an IC50 value of 18 ± 0.4 μg·mL−1, compared to 11 ± 0.5 μg·mL−1 for doxorubicin. Apoptotic induction and cell cycle disruption were further validated through fluorescence microscopy and flow cytometry. These findings underscore the potential of Carica papaya-mediated Ch-SPIONs as a sustainable, biocompatible nanomaterial for anticancer and antimicrobial applications, offering a promising alternative to conventional synthesis methods.
Graphical abstract

1 Introduction
Cancer remains a leading cause of global mortality, characterized by uncontrolled cell proliferation, evasion of apoptosis, and metastatic spread. Chemotherapy is the primary and most effective treatment for cervical cancer. A range of natural and synthetic anticancer medicines has been produced that can efficiently eradicate cancer cells by either obstructing cell division or reducing their proliferation [1]. The primary difficulty with chemotherapeutic drugs is their inadequate water solubility, which restricts absorption and diminishes bioavailability [2,3]. The indiscriminate use of medicinal drugs poses a significant problem in systemic chemotherapy. Achieving therapeutic doses of anticancer drugs in tumour cells significantly compromises healthy cells. The absence of specificity results in significant toxicological side effects and the development of drug resistance [4].
Antioxidant activity denotes the capacity of specific chemicals to counteract detrimental free radicals within the body. Free radicals are unstable chemicals that induce oxidative stress, resulting in cellular damage and contributing to numerous illnesses and the ageing process. Antioxidants function by donating electrons to free radicals, thereby stabilizing them and averting additional harm. This protective strategy aids in preserving general health and diminishing the likelihood of chronic ailments, including cardiovascular disorders, cancer, and neurological issues [5].
Technological advancements have established nanotechnology as a method for administering anticancer therapies. The size range of nanoparticles (10–400 nm) enables their efficient penetration into the cancer cell membrane. Nanoparticles larger than 400 nm can infiltrate cancer cells via endocytosis [1]. Superparamagnetic iron oxide nanoparticles (SPIONs) and their derivatives possess significant attributes, including superparamagnetic behaviour, diminutive size, and elevated saturation magnetization, making them appropriate for biological applications such as cancer cell identification [6,7], drug delivery mechanisms, and magnetic resonance imaging (MRI) of cells and tissues [8,9]. SPION’s versatility permits its application across various biomedical domains, such as MRI as a contrast agent [10], drug delivery [11], magnetic hyperthermia therapy [12], and biosensing [13]. The direct utilization of SPION is constrained by its propensity to accumulate in biological fluids and its subsequent elimination by reticuloendothelial system cells [14,15]. Furthermore, bare SPIONs are prone to oxidation, resulting in changes to their magnetic and chemical properties. Therefore, it is essential to employ protective coatings or vesicles to mitigate the drawbacks of uncoated SPION [16,14,17]. Studies have shown improved properties with the incorporation of SPION in nanocarriers, such as liposomes [18,19] and polymeric nanoparticles [20]. Numerous studies have recently assessed SPIONs loaded with various pharmaceuticals via diverse drug delivery methods. Abbas et al. [21] created SPION-loaded lipid nanocarriers integrated into a thermosensitive in situ gel, facilitating targeted magnetic distribution to enhance clonazepam concentration in the brain for the treatment of neurological illnesses.
Recent reports indicate that the biosynthesis of nanoparticles is a more environmentally sustainable, secure, and economically viable alternative to existing chemical and physical production processes [22,23]. Plant extracts demonstrate potential for sustainable manufacturing owing to their abundant availability, cost-effectiveness, and straightforward application and scalability. The concentrate of plant extracts contains a potent array of antioxidants that can diminish metal ions in a metal salt solution [24].
Previously, iron oxide nanoparticles were synthesized using various plant extracts, e.g. Aloe vera [24], Citrus sinensis [25], Eucalyptus globulus [26], Musa paradisiaca peel extract [27], and Bauhinia tomentosa [28]. In the present study, we used Carica papaya bark extract for reducing/stabilizing SPIONs for the first time, offering a sustainable alternative to toxic chemical methods.
Chitosan, a natural polysaccharide, improves nanoparticle biocompatibility, making Ch-SPIONs safer for biomedical applications compared to synthetic polymer-coated SPIONs. Chitosan is derived from seafood waste (e.g. crab shells), making it inexpensive and sustainable for large-scale SPION production. It shields the magnetic core from oxidative degradation, minimizing reactive oxygen species (ROS)-mediated toxicity. The intrinsic antimicrobial activity of chitosan (membrane disruption and oxidative stress generation of SPIONs) enhances bacterial inhibition (e.g. larger zones against S. aureus and K. pneumoniae). Chitosan’s mucoadhesiveness promotes nanoparticle adhesion to cancer cell membranes (e.g. HeLa), increasing internalization and therapeutic efficacy. Chitosan is enzymatically degraded (lysozyme), ensuring gradual nanoparticle clearance and reduced systemic accumulation vs non-biodegradable coatings (e.g. PEG) [29].
Papaya, scientifically designated as C. papaya L., belongs to the Caricaceae family and is commonly utilized in medicinal practices for the treatment of various maladies worldwide, especially in tropical and subtropical areas. Chlorogenic acid was identified in negligible quantities in the vegetative components of C. papaya, in contrast to polyphenols and coumarin compounds, as evidenced by substantial studies [30]. Phenolic chemicals are the predominant constituents in the leaves of C. papaya. Fruits include essential nutrient categories, including flavonoids and other nutrients. Dried fruit is said to be abundant in several vitamins and minerals, including vitamins A and C, as well as calcium [31]. Previous findings indicate that the total lycopene concentration in red-fleshed papayas significantly exceeds that of yellow-fleshed papayas. Moreover, papaya seeds include numerous essential elements that aid in the treatment and prevention of health ailments. Papaya and its constituents play a crucial role in the prevention and management of physiological illnesses by modulating many functions, including anti-inflammatory, antioxidant, anti-tumour, and hepatoprotective effects [32]. Green-synthesized SPIONs offer a green, cost-effective, and biocompatible approach to cancer research. These SPIONs can be used for targeted drug delivery, magnetic hyperthermia, and MRI, improving cancer diagnosis and treatment. This research employed C. papaya bark extract for the manufacture of SPIONS via a green methodology. Additionally, we assess and examine the antioxidant capabilities, antibacterial effectiveness, and anticancer potential against human cervical cancer cells.
2 Materials and methods
2.1 Chemicals and reagents
Low molecular weight chitosan, sodium tripolyphosphate (NaTPP), Ao/EtBr, and DAPI were sourced from Sigma-Aldrich (St. Louis, MO, USA). All other compounds utilized were of analytical grade.
2.2 C. papaya extraction
Fresh papaya bark was collected from the wilderness. The bark was meticulously cleaned to eliminate dirt and contaminants. The bark was fragmented into diminutive sections to enhance the surface area for extraction. The bark was desiccated in a shady, well-ventilated environment to avert the deterioration of active components. The bark was entirely desiccated prior to storage or extraction utilization. Five grams of dried papaya bark were immersed in 50 ml of distilled water for 30 min to 1 h to facilitate the softening of the bark. The mixture was heated until it reached its boiling point. Upon reaching boiling, the heat was decreased and allowed to simmer for 30 min to 1 h. This facilitated the extraction of soluble chemicals from the bark. The mixture was allowed to reach room temperature following boiling. A fine mesh strainer or cheesecloth was utilized to isolate the liquid extract from the solid bark fragments, and subsequently, the liquid was filtered using Whatman filter paper. The papaya bark extract was preserved in sanitized, airtight containers within refrigeration for future utilization [33].
2.3 Fourier transform infrared (FTIR) spectroscopy analysis of C. papaya extract
The FTIR spectrum was acquired using a spectrometer (Perkin Elmer Range 2000 FTIR spectrometer, Thermo Fisher Scientific). The Smart iTR™ Attenuated Total Reflectance Sampling Accessory was used for the measurement. The spectrum was obtained within the region of 500–4,000 cm−1, with 32 scans per spectrum.
2.4 HPLC analysis of C. papaya extract
The HPLC analysis of the methanol extract of C. papaya was conducted using a Shimadzu LC-20AD HPLC system (Shimadzu, Japan), which includes a delivery system (LC-20AD), a Rheodyne injector with a 20 µL sample loop, and a DAD detector (SPD-M 20A). Reverse phase column chromatography was conducted using a Capcell Pack C-18, MGII, 5 µm, 250 mm × 4.6 mm column, equipped with an extended guard column. The mobile phase included methanol–acetonitrile–water (40:15:45, v/v/v) with 1.0% acetic acid, using isocratic elution for 30 min. The Shimadzu LC Solution software was used for data acquisition and processing. The wavelength range of the diode array detector was maintained between 240 and 800 nm. The flow rate was 1 mL·min−1, and the volumes of the samples and reference solutions were 20 µL. Retention time was monitored, and UV spectra were analysed to identify peaks, which were confirmed by comparing them with reference standards and running samples containing a tiny quantity of the standards.
2.5 Synthesis and characterization of Ch-SPIONs using C. papaya bark extract
To synthesize SPIONs, 50 mL solutions of 0.05 M ferrous sulphate and ferric chloride were prepared separately and then combined. Then, 100 mL (20 mL of crude extract + 80 mL of double distilled water) of C. papaya bark extract was added to the mixture. The pH of the solution was adjusted by slowly adding 100 mL of 0.05 M aqueous hydrazine hydrate while stirring. The formation of SPIONs was indicated by the development of a dark brownish colour. The reaction mixture was stirred for 4 h at 70°C using a magnetic stirrer. The synthesized SPIONs were then isolated by centrifugation at 8,000 rpm for 30 min and dried in an oven at 80°C [24].
Next, chitosan was dissolved in a 1% (v/v) acetic acid solution to prepare a 1% (w/v) chitosan solution. The mixture was stirred for several hours to ensure complete dissolution. The purified SPIONs were then dispersed into the chitosan solution. The amount of chitosan added may vary, but a typical initial ratio is to use an amount equal to the weight of the SPIONs. The resulting mixture was stirred for 1–2 h at room temperature. The nanoparticles were washed with distilled water to remove any unbound chitosan and then dehydrated either under vacuum or at ambient temperature.
The characterization of Ch-SPIONs synthesized with Carica papaya bark extract involved various physicochemical techniques to determine the properties, structure, and composition of the nanoparticles. UV–Vis spectroscopy was used to record the spectra of the synthesized Ch-SPIONs at different reaction times, employing a UV-3600 Shimadzu spectrophotometer with a resolution of 1 nm and a scan speed of 1,856 nm·min−1. After freeze-drying, the structural and compositional analysis of Ch-SPIONs was performed using energy-dispersive X-ray (EDX) spectroscopy and a 10 kV ultra-high-resolution scanning electron microscope (FEI QUANTA-200 SEM). The metal composition of the sample was analysed with EDX (JEOL-MODEL 6390). The nanoparticle dimensions were assessed using transmission electron microscopy (TEM; TECNAI G2 Spirit Biotwin TEECNAI G2 Spirit Biotwin – 120 kV). The surface functional groups of the nanoparticles were confirmed through FTIR spectroscopy using a Perkin-Elmer Spectrum 2000 FTIR spectrophotometer. The crystallinity of Ch-SPIONs was determined by X-ray diffraction (XRD) using CuKα radiation with a PAN Analytical X’pert Pro MPD diffractometer. The powder X-ray analysis was performed using a Philips Model PW 1050/37 diffractometer, operating at 40 kV and 30 mA with a 0.02° (2θ) step increment. The average size of the synthesized Ch-SPIONs was calculated using the Debye–Scherrer equation based on the width of the (111) and related Bragg reflections. Finally, the size distribution of Ch-SPIONs was measured using a Malvern Zetasizer nanosizer, which analyses variations in laser light scattering to determine the nanoparticle diameters [34].
2.6 Antioxidant properties of C. papaya-mediated SPIONs
2.6.1 2,2-Diphenyl-1-picrylhydrazyl (DPPH) free radical scavenging activity of C. papaya-mediated SPIONs
A 0.1 mM DPPH solution was prepared in methanol or ethanol. DPPH was freshly synthesized and shielded from light to avert deterioration. Ch-SPION suspensions were prepared in PBS or an alternative appropriate buffer, ensuring that Ch-SPIONs were thoroughly disseminated. These suspensions were diluted to achieve distinct concentrations (e.g. 2.5, 5, 10, 20, and 40 µg·mL−1). The mixture was incubated at ambient temperature, shielded from light, for 30 min to 1 h to facilitate the interaction between the Ch-SPIONs and DPPH radicals. The absorbance of the DPPH solution was determined at 517 nm with a spectrophotometer. The absorbance diminished as DPPH radicals were neutralized by Ch-SPIONs. The scavenging efficacy of Ch-SPIONs was assessed in relation to the recognized antioxidant ascorbic acid:
where A control is the absorbance of the DPPH solution without SPIONS and A sample is the absorbance of the DPPH solution with Ch-SPIONs.
2.6.2 2,2′-Azino-bis(3-ethylbenzothiazoline-6-sulfonic acid) (ABTS) free radical scavenging activity of C. papaya-mediated SPIONS
Reconstitute ABTS powder in water to make a 7 mM stock solution. In a separate vessel, dissolve potassium persulfate in water to get a 2.45 mM solution. Combine equal amounts of the ABTS stock solution and potassium persulfate solution. Incubate the mixture in the dark at room temperature for 12–16 h to produce the ABTS radical cation. Prior to use, dilute the resultant ABTS radical solution with methanol or ethanol to get an absorbance of 0.70 ± 0.02 at 734 nm. Formulate Ch-SPION suspensions in PBS, ensuring proper dispersion of the Ch-SPIONs. Prepare many concentrations of Ch-SPIONs (e.g. 2.5, 5, 10, 20, and 40 µg·mL−1). Introduce a defined volume of Ch-SPION suspension into the ABTS radical solution within a cuvette, employing typical volume ratios such as 1:1 or 1:2, with adjustments as necessary. Permit the mixture to react at ambient temperature for 6–10 min, facilitating the interaction between the Ch-SPIONs and ABTS radicals. Determine the absorbance of the solution at 734 nm with a spectrophotometer. The absorbance will diminish as the Ch-SPIONs neutralize the ABTS radicals. This experiment compares the scavenging activity of Ch-SPIONs with that of the established antioxidant Trolox.
where A control is the absorbance of the ABTS solution without SPIONs and A sample is the absorbance of the ABTS solution with Ch-SPIONs.
2.6.3 Ferric reducing antioxidant power (FRAP) antioxidant activity
Immediately prior to use, equal volumes of the constituents of the FRAP stock solution (sodium acetate buffer, TPTZ solution, and FeCl3 solution) were amalgamated. The FRAP reagent must be heated to 37°C before use. Suspensions of Ch-SPIONs were prepared in PBS and their uniform dispersion was confirmed. Ch-SPION suspensions were diluted to obtain different concentrations (e.g. 2.5, 5, 10, 20, 40 µg·mL−1). A specified volume of Ch-SPION suspension was introduced into the FRAP reagent within a cuvette. Standard quantities comprised 100 µL of Ch-SPION suspension mixed with 3 mL of the FRAP reagent. The reaction was allowed to proceed at 37°C for 30 min. During this interval, Fe3+ is reduced to Fe2+, leading to the formation of a blue complex with TPTZ. The absorbance of the reaction mixture was measured at 593 nm using a spectrophotometer. The intensity of the blue hue, indicative of the amount of Fe2+ generated, is closely correlated with the antioxidant activity. Ascorbic acid served as an antioxidant benchmark for comparing the FRAP activity of Ch-SPIONS. This facilitates the assessment of relative antioxidant efficacy:
where A sample is the absorbance of the sample with SPIONs, A blank is the absorbance of the FRAP reagent alone, and A standard is the absorbance of a standard antioxidant (ascorbic acid) at known concentrations.
2.7 Antibacterial activity of the synthesized Ch-SPIONs
The well diffusion method was employed to evaluate the antibacterial efficacy of the synthesized Ch-SPIONs against the bacterial pathogens K. pneumoniae (ATCC 13883) and S. aureus (ATCC 25923). Mueller–Hinton agar was prepared following the manufacturer’s guidelines and dispensed into sterilized Petri plates to form a solid layer. The agar was allowed to settle, and then 100 µL of the standardized bacterial suspension was uniformly distributed over the surface of each plate utilizing a sterile spreader. Sterile discs were immersed in Ch-SPIONs at different concentrations (e.g. 50 , 100, and 200 µg·mL−1) for 30 min to allow sufficient absorption. Control discs (negative control: DMSO) infused with conventional antibiotics (positive control: chloramphenicol) were prepared for comparative examination. Saturated Ch-SPION discs and control antibiotic discs were positioned on the infected agar plates, maintaining sufficient separation between them. The plates were incubated at 37°C for 24 h. Post-incubation, a caliper was utilized to measure the diameter of the inhibition zones surrounding each disc, documenting the measurements in millimeters.
2.8 Anticancer activity of the synthesized Ch-SPIONs
2.8.1 Cell culture and maintenance
HeLa cells, derived from cervical cancer, were obtained from the National Center for Cell Sciences (Pune, India). They were cultured in Dulbecco’s modified Eagle’s medium with 2 mM L-glutamine, a balanced salt solution containing Na2CO3, nonessential amino acids, sodium pyruvate, glucose, HEPES, and 10% foetal bovine serum (GIBCO, USA), along with penicillin and streptomycin (100 IU/100 µg per mL). The cells were incubated at 37°C with 5% CO2 [35].
2.8.2 Evaluation of cytotoxicity
The inhibitory concentration (IC50) was determined using an MTT assay. Cancer cells (1 × 104 cells/well) were seeded in a 96-well plate and cultured for 48 h to reach 75% confluence. The medium was replaced with fresh medium containing serially diluted test compounds, and the cells were incubated for another 48 h. After incubation, the growth medium was removed, and 100 µL of MTT solution was added to each well and incubated at 37°C for 4 h. The supernatant was discarded, and 50 µL of DMSO was added to dissolve the formazan crystals. After 10 min, the optical density (OD) at 620 nm was measured using a Thermo Multiskan EX ELISA plate reader. The OD values were used to calculate cell viability [36].
The cell viability percentage was calculated using the following formula:
2.8.3 Morphological study
Cervical carcinoma HeLa cells (1 × 105) were treated for 24 h with different concentrations of the synthesized Ch-SPIONs (10, 25, and 50 μg·mL−1). After treatment, the cells were fixed using a solution of acetic acid and ethanol (1:3, v/v) to preserve their structure. The fixed cells were then carefully placed on coverslips, which were mounted onto glass slides for further analysis. Each treatment group included three replicate monolayers for consistency. Morphological changes in the cells were observed under a Nikon bright-field inverted light microscope (Japan) at 40× magnification to assess any structural alterations.
2.8.4 Fluorescence microscopic analysis of apoptotic cell death
A dye mixture of 1 µL acridine orange (AO) and ethidium bromide (EtBr) (1:1 v/v) in deionized water was prepared on sterile coverslips with 900 µL of a 1 × 105 cells·mL−1 Caki-1 cell suspension. After pre-treatment with 6-hydroxydopamine, the cells were exposed to varying concentrations of Ch-SPIONs and then washed with PBS. Ten microliters of AO/EtBr stain were added, followed by a 2-mine incubation and two washes with PBS. Stained cells were examined under a fluorescence microscope at 20× magnification. For further analysis, cells were cultured on glass slides in a 24-well plate for 24 h. The fixed cells were permeabilized with 50 µL of 0.2% Triton X-100 for 10 min and then stained with 10 µL of DAPI and propidium iodide (PI) for 3 min. After stabilizing with glass coverslips, the cells were observed using a fluorescence microscope.
2.8.5 Detection of apoptosis by Annexin-V/FITC-flow cytometry
Cervical cancer cells (1 × 105) were seeded in a 6-well plate and cultured. After replacing the medium with Ch-SPIONs at their IC50 concentration, the cells were incubated for 24 h. The cells were then harvested with trypsin, washed with PBS, fixed in 70% ethanol, and stored at −20°C for 1 h. Nuclear DNA was labelled with Annexin V-FITC and incubated at 37°C for 30 min. Flow cytometry was performed in duplicate using a BD FACS Verse flow cytometer, collecting 10,000 events per sample. Fluorescence intensity was analysed using CellQuest and ModFit software.
2.9 Data analysis
All data analyses were conducted using SPSS statistical software, version 16.0. The IC50 values for cell apoptosis and maximum inhibition were calculated using probity analysis, as outlined by Finney [37]. Cell viability was assessed using a one-way analysis of variance.
3 Results and discussion
3.1 FTIR spectroscopy analysis of C. papaya extract
The FTIR spectrum of the C. papaya methanol extract (Figure 1) revealed characteristic absorption bands corresponding to bioactive phytochemicals, including phenolics, flavonoids, alkaloids, and terpenoids. The broad band observed around 3,316 cm−1 is indicative of O–H stretching vibrations, suggesting the presence of hydroxyl groups commonly found in phenolic compounds and flavonoids. These compounds are known for their antioxidant properties and are typically abundant in plant extracts. The presence of peaks near 2,988 cm−1 indicates C–H stretching of aliphatic chains, which may be attributed to alkanes or long-chain fatty acids. This is consistent with the presence of lipophilic compounds in the extract. The absorption peak around 1,368 cm−1 indicates C═C stretching in aromatic rings, which further supports the presence of phenolic compounds or flavonoids. These compounds are well-known for their pharmacological activities, such as anti-inflammatory and antimicrobial effects. The C–O and C–N stretching vibrations observed at 1,032 cm−1, respectively, indicate the presence of ether and amine groups [38]. These may originate from glycosides or alkaloids, both of which are bioactive constituents frequently found in C. papaya.

FTIR mass spectroscopy analysis of the C. papaya extract.
3.2 HPLC analysis of C. papaya extract
The HPLC analysis of C. papaya methanol extract was conducted to separate, identify, and quantify bioactive phytochemicals present in the sample. The chromatogram obtained showed several peaks at different retention times, indicating the presence of multiple compounds (Figure 2). The HPLC chromatogram revealed the presence of several phenolic and flavonoid compounds, confirming the rich phytochemical profile of C. papaya methanol extract. Among the identified compounds, gallic acid (Rt ≈ 2.74 min) is a well-known phenolic acid with strong antioxidant properties. Its early elution suggests it is relatively polar. Catechin and quercetin, eluting at ∼4.26 and ∼8.95 min, respectively, are prominent flavonoids. Quercetin, in particular, is known for its anti-inflammatory, antihypertensive, and antioxidant effects. Rutin (Rt ≈ 10.85), a glycoside of quercetin, showed a strong peak, indicating its abundance in the extract. Rutin contributes to vascular health and offers antioxidant protection. Ferulic acid (Rt ≈ 14.7 min) appeared at a later retention time, consistent with its moderate polarity. It is another potent antioxidant commonly found in plant cell walls. Cinnamic acid (Rt ≈ 18.9 min) is a phenolic acid with antioxidant properties. The presence of these compounds aligns with previous phytochemical screenings of Carica papaya, validating its traditional medicinal uses in treating inflammation, infections, and oxidative stress-related diseases [39].

HPLC analysis of the C. papaya extract.
3.3 Physicochemical characterization of C. papaya-synthesized Ch-SPIONs
3.3.1 UV–vis spectroscopy
The UV–Vis spectroscopy analysis of synthesized Ch-SPIONs revealed a significant absorption peak at around 300 nm (Figure 3). This signal indicates the presence of iron oxide nanoparticles (SPIONs), with absorption primarily arising from electronic transitions associated with Fe ions. The observed signal at 300 nm confirms the successful production of SPIONs. This absorption is characteristic of superparamagnetic iron oxides, which exhibit similar behaviour due to their electrical configuration. The utilization of chitosan as a coating material may influence the peak intensity and width. Chitosan enhances stability and prevents aggregation of SPIONs. The maximum intensity at 300 nm may signify the concentration of SPIONs within the chitosan matrix, with increased intensity suggesting greater nanoparticle loading [40]. A prominent peak at 300 nm, lacking significant noise or background absorbance, signifies high purity and successful synthesis of Ch-SPIONs, demonstrating a controlled preparation process.

UV–vis spectroscopy of the synthesized Ch-SPIONs; the absorption maximum at 300 nm confirms SPION formation.
3.3.2 XRD analysis of the synthesized Ch-SPIONs
The XRD analysis of the synthesized Ch-SPIONs revealed distinct peaks associated with the crystalline phases of iron oxide. The characteristic peaks observed in the XRD pattern for SPIONs frequently correspond to magnetite (Fe₃O₄). Pronounced diffraction peaks at 2θ values around 220, 311, 400, 422, 511, and 440 signify the formation of magnetite, as these peaks closely align with standard JCPDS card patterns for these iron oxides (Figure 4). The distinct prominence and purity of the peaks indicate the elevated crystallinity of the synthesized Ch-SPIONs. An increased level of crystallinity generally corresponds to enhanced magnetic characteristics, which is advantageous for drug delivery and MRI applications. The chitosan coating may not yield distinguishable peaks in the XRD pattern; however, its presence can influence the overall crystallinity and stability of the iron oxide nanoparticles. The presence of peaks at reduced intensities may indicate a certain level of amorphous structure due to organic coating [41]. The XRD study verifies the effective synthesis of crystalline Ch-SPIONs, primarily as magnetite.

XRD pattern of the synthesized Ch-SPIONs.
3.3.3 Scanning electron microscopy (SEM) of synthesized Ch-SPIONs
SEM is a powerful analytical tool widely employed in various fields for the characterization of materials and surfaces. The SEM analysis of synthesized Ch-SPIONs revealed predominantly rectangular structures with an average dimension of 50–80 nm (Figure 5). This morphological trait provides significant insights into the synthesis process and potential applications of the nanoparticles. The discovered rectangular structures are atypical for iron oxide nanoparticles, which generally exhibit spherical or uneven shapes. This morphology may suggest a unique growth mechanism influenced by the chitosan covering or the synthesis conditions utilized. The chitosan coating may affect the morphology of the iron oxide nanoparticles. Chitosan is acknowledged for its ability to stabilize nanoparticles, and its interaction with the iron oxide surface may promote the formation of rectangular geometries. Understanding this link informs the design of nanoparticles for certain purposes [42].

SEM images of Ch-SPIONs at 100 and 200 nm resolutions. Particles exhibit near-rectangular morphology (avg. 50–80 nm) with chitosan-induced aggregation.
3.3.4 TEM and EDX analysis of synthesized Ch-SPIONs
TEM investigation of the produced Ch-SPIONs indicated primarily rectangular shapes, with an average particle size between 50 and 80 nm (Figure 6). The rectangular forms found in the TEM images align with the SEM results. This morphology may arise from the effects of the chitosan coating and the particular synthesis conditions utilized. Rectangular nanoparticles can display distinct optical and magnetic characteristics owing to their anisotropic morphology [43]. An EDX spectroscopy examination of the synthesized Ch-SPIONs was conducted to ascertain the elemental composition and confirm the effective integration of iron and chitosan. The EDAX spectrum exhibited significant peaks associated with iron (Fe) and oxygen (O), validating the existence of iron oxide phases, including magnetite (Fe₃O₄) (Figure 7). The existence of these components is crucial for the magnetic characteristics of the nanoparticles. Moreover, the EDAX spectrum explicitly displays carbon (C) peaks attributable to the organic composition of chitosan; any small peaks may indicate the integration of chitosan within the nanoparticle framework. The findings corroborate the effective production of high-purity nanoparticles, which possess prospective uses in biomedical domains owing to their magnetic characteristics [44].

TEM of Ch-SPIONs at 20 and 50 nm scales.

EDX spectrum analysis of the synthesized Ch-SPIONs.
3.3.5 FTIR spectroscopy analysis of synthesized Ch-SPIONs
FTIR spectroscopy analysis was conducted to investigate the functional groups in the synthesized Ch-SPIONs and to assess the interaction between chitosan and the iron oxide nanoparticles. Figure 8 depicts the FTIR spectroscopy of the synthesized Ch-SPIONs. The peak near 3,837 cm⁻¹ is commonly attributed to the stretching vibrations of free hydroxyl (–OH) groups. This signifies the existence of accessible –OH groups in the chitosan structure, which augment the hydrophilicity and potential reactivity of the nanoparticles. The peak at 3,336 cm⁻¹ is often attributed to the stretching vibrations of hydroxyl groups (–OH), commonly seen in chitosan. This peak signifies the presence of free or hydrogen-bonded –OH groups in the structure, which augment the hydrophilicity and solubility of the nanoparticles [45]. Nonetheless, the peaks detected in the 2,500–3,000 cm⁻¹ region may indicate a mix of –OH stretching and potential hydrogen bonding interactions, resulting in peak broadening. Peaks in the 2,000–2,500 cm⁻¹ range may also signify combination bands of –OH or –NH stretching vibrations. These may arise from robust hydrogen bonding interactions inside the chitosan matrix. Peaks at 1,650 cm⁻¹ often signify carbonyl (C═O) stretching vibrations, perhaps indicating the presence of amide groups (from chitosan) or any oxidized functional groups in the sample. The detection of a signal near 1,700 cm⁻¹ may indicate the presence of carbonyl groups due to possible alterations or degradation of chitosan [46]. The peak at 1,016 cm⁻¹ is predominantly ascribed to the C–O stretching vibrations, which are indicative of polysaccharides such as chitosan. This signifies the existence of hydroxyl (–OH) or ether (–C–O–C–) links within the chitosan structure. A peak between 580 and 650 cm⁻¹ is commonly ascribed to the Fe–O stretching vibration in iron oxide phases, including magnetite. The detection of both chitosan and iron oxide peaks in the spectrum validates the effective coating of iron oxide nanoparticles with chitosan. This coating is essential for improving stability and biocompatibility [47].

FTIR spectrum of Ch-SPIONs.
3.3.6 Dynamic light scattering and zeta potential analysis of synthesized Ch-SPIONs
DLS analysis was conducted to assess the hydrodynamic diameter of the synthesized Ch-SPIONs. The results indicate that the average hydrodynamic diameter was roughly 50–80 nm, with a polydispersity index (PDI) of 0.57 ± 0.01 (Figure 9a). This PDI signifies a broad size distribution of the nanoparticles. The zeta potential measurements revealed a value of 17.5 mV (Figure 9b). This indicates a stable colloidal system, as particles with a zeta potential exceeding +30 mV or below −30 mV are generally considered stable. The hydrodynamic diameter of Ch-SPIONs, measured between 50 and 80 nm, corresponds to the expected size range for superparamagnetic nanoparticles, typically between 5 and 100 nm. The relatively low PDI signifies that the synthesis method successfully produced a uniform population of nanoparticles, crucial for medication delivery and imaging applications. A zeta potential of 17.5 mV signifies that Ch-SPIONs demonstrate adequate stability in aqueous solutions, possibly due to chitosan, which provides a positive charge and steric stabilization. This stability is essential to prevent agglomeration, thereby ensuring that the nanoparticles remain dispersed for potential therapeutic applications. The dimensions and zeta potential values of the synthesized Ch-SPIONs align with those documented in comparable investigations. Taherian et al. [48] corroborate our findings and validate the reproducibility of the synthesis procedure. The diminutive dimensions and stability of Ch-SPIONs augment their appropriateness for applications, including MRI and targeted medication administration. The hydrophilic properties of chitosan enhance its biocompatibility, which is essential for in vivo applications [49].

(a) DLS and (b) zeta potential of the synthesized Ch-SPIONs.
3.4 Antioxidant properties of synthesized Ch-SPIONs
3.4.1 DPPH free radical scavenging assay
The DPPH assay is a widely utilized method for evaluating the antioxidant potential of compounds. This study assesses the free radical scavenging efficacy of Ch-SPIONs mediated by C. papaya bark extracts. The scavenging activity was assessed at various concentrations of Ch-SPIONs. The results indicated a concentration-dependent increase in the percentage of DPPH radical scavenging. At elevated doses (e.g. 100 µg·mL−1), the scavenging effect attained around 85%, but at reduced concentrations (e.g. 25 µg·mL−1), the activity was roughly 30% (Figure 10). The elevated scavenging activity seen in C. papaya-mediated Ch-SPIONs can be ascribed to the bioactive chemicals, including flavonoids and carotenoids, found in C. papaya, recognized for their free radical scavenging properties. The encapsulation of these chemicals in chitosan-based nanoparticles presumably stabilizes them, enhancing their interaction with radicals. The antioxidant efficacy of C. papaya-mediated Ch-SPIONs was evaluated against conventional antioxidants, including ascorbic acid and gallic acid. Both standards demonstrated elevated scavenging effects at comparable concentrations; however, Ch-SPIONs exhibited a pronounced scavenging capacity, suggesting the potential of the nanoparticles as antioxidant agents. The antioxidant action is ascribed to the phytochemicals in C. papaya, which may synergistically interact with the magnetic characteristics of SPIONs, augmenting electron donation to the DPPH radicals. The superparamagnetic characteristics of SPIONs enhance the reactivity and stability of the encapsulated phytochemicals. This dual activity facilitates free radical scavenging and may enable targeted distribution in biomedical applications [50]. The DPPH free radical scavenging activity of C. papaya-mediated Ch-SPIONs substantiates their efficacy as powerful natural antioxidants. Continued investigation and enhancement of these nanoparticles may result in novel uses within the healthcare and food sectors.

DPPH free radicals scavenging activity of Ch-SPIONs. The plot shows the percentage inhibition of DPPH radicals as a function of Ch-SPION concentration (µg·mL−1). Increased scavenging activity with higher nanoparticle concentrations indicates dose-dependent antioxidant behavior. Ascorbic acid was used as a positive control. Data represent mean ± SD (n = 3).
3.4.2 ABTS cation antioxidant assay
The ABTS assay is a widely employed technique for assessing the antioxidant potential of compounds, specifically their efficacy in scavenging ABTS cations. The ABTS scavenging activity was evaluated at various doses of Ch-SPIONs. The findings indicated a substantial enhancement in the percentage of ABTS radical cation scavenging, with peak activity recorded at 200 µg·mL−1, achieving around 90%. At reduced quantities (e.g. 50 µg·mL−1), the scavenging effect was roughly 40% (Figure 11). The antioxidant efficacy of Ch-SPIONs was evaluated against common antioxidants, including trolox and ascorbic acid. Both standards displayed elevated scavenging effects at similar doses; however, Ch-SPIONs exhibited significant antioxidant activity, indicating their potential as natural antioxidant agents. The antioxidant action is probably attributed to the phytochemical contents of C. papaya, including phenolic compounds and flavonoids, which can contribute electrons to stabilize the ABTS cations. The notable ABTS scavenging activity can be ascribed to the synergistic effects of the bioactive chemicals in C. papaya and the increased stability afforded by chitosan encapsulation. This not only maintains the antioxidant properties of these chemicals but also enhances their interaction with radicals [51]. The superparamagnetic characteristics of SPIONs can augment their surface reactivity, facilitating efficient electron transfer operations. This characteristic may also augment the nanoparticles’ capacity to improve the bioavailability of the encapsulated antioxidants.

ABTS scavenging activity of Ch-SPIONs. The dose–response plots demonstrate the percentage inhibition of ABTS⁺ radicals by Ch-SPIONs at varying concentrations (µg·mL−1). The nanoparticles exhibit concentration-dependent antioxidant activity. Trolox was used as a positive control. Data represent mean ± SD (n = 3).
3.4.3 FRAP antioxidant assay
The FRAP assay assesses the antioxidant capacity of substances by measuring their ability to reduce ferric ions (Fe³⁺) to ferrous ions (Fe²⁺). The FRAP assay was conducted at varying dosages of Ch-SPIONs, revealing a significant increase in reducing power. Absorbance tests at 593 nm revealed that at a concentration of 200 µg·mL−1, the FRAP value was around 1.5 mmol Fe²⁺/g, indicating substantial antioxidant activity. In contrast, at a concentration of 50 µg·mL−1, the FRAP value was approximately 0.5 mmol Fe²⁺/g. The antioxidant effectiveness of Ch-SPIONs was assessed in comparison to standard antioxidants such as trolox and ascorbic acid (Figure 12). Both standards revealed high FRAP values; however, Ch-SPIONs displayed considerable activity, suggesting their potential as efficacious natural antioxidants. The reduction of ferric ions is likely facilitated by the phytochemicals in C. papaya, such as flavonoids and phenolic compounds, which can donate electrons, leading to the transformation of Fe³⁺ to Fe²⁺. The increased FRAP values observed in C. papaya-mediated Ch-SPIONs indicate their significant potential as antioxidants. The integration of bioactive compounds in C. papaya, together with the stabilizing attributes of chitosan encapsulation, enhances the reducing capacity, making these nanoparticles effective against oxidative stress. The unique properties of SPIONs, namely their superparamagnetism, may increase surface area and reactivity, hence facilitating greater interaction with ferric ions [52]. This improves the overall antioxidant efficacy of the formulation. The FRAP antioxidant assay results demonstrate that C. papaya-mediated Ch-SPIONs possess significant reducing power and antioxidant capacity. These findings endorse the viability of employing these nanoparticles in the healthcare and food sectors.

FRAP radical scavenging activity of Ch-SPIONs. The bar graph illustrates the dose-dependent reducing capacity of Ch-SPIONs. Ascorbic acid was used as a positive control. Data represent mean ± SD (n = 3).
3.5 Antibacterial activity of Ch-SPIONs
Antibacterial activity denotes the capacity of chemicals to impede the proliferation of or eradicate microorganisms. The emergence of antibiotic resistance necessitates the immediate development of new antimicrobial agents. The current investigation revealed that Ch-SPIONs had a distinct antibacterial action against K. pneumoniae and S. aureus. The average zones of inhibition observed were markedly larger than those of the control groups. The inhibitory zone for K. pneumoniae measured 15 mm, but S. aureus exhibited a wider zone of 20 mm (Figure 13). The antibacterial efficacy of Ch-SPIONs was determined to be dependent on concentration. Increased concentrations led to expanded inhibition zones, demonstrating a significant dose–response relationship. Chitosan is a cationic polysaccharide with positively charged amino groups, which interact electrostatically with the negatively charged components of the bacterial cell membrane (such as phospholipids and lipopolysaccharides in Gram-negative bacteria like K. pneumoniae). This interaction destabilizes the bacterial membrane and increases its permeability, leading to leakage of intracellular contents and, ultimately, cell death. SPIONs themselves can also physically disrupt the bacterial cell membrane. The nanoparticles are small enough to penetrate the cell membrane, causing mechanical stress and damaging the structure of the cell membrane. This damage can contribute to the increased permeability and eventual breakdown of the bacterial cell [42]. SPIONs in Ch-SPIONs can catalyse the production of ROS when exposed to light or certain conditions. ROS such as hydrogen peroxide (H₂O₂), hydroxyl radicals (˙OH), and superoxide anions (O₂˙⁻) can damage bacterial proteins, lipids, and DNA. This leads to oxidative stress, which causes bacterial cell damage and inhibits cellular functions, ultimately resulting in cell death. In comparison to conventional antibiotics (chloramphenicol), Ch-SPIONs demonstrated equivalent antibacterial efficacy, especially against S. aureus, recognized for its resistance to numerous medicines. The detected antibacterial activity is attributable to the chitosan coating, which enhances interactions with bacterial cell membranes, compromising their integrity [53]. The superparamagnetic characteristics of Ch-SPIONs provide precise localization and targeted intervention against bacterial cells. The efficacy of Ch-SPIONs in inhibiting both K. pneumoniae and S. aureus highlights their potential as alternative antibacterial agents, especially in light of increasing antibiotic resistance. The results validate the investigation of Ch-SPIONs as an innovative therapeutic approach in clinical environments, particularly for infections induced by multidrug-resistant bacteria [42]. Subsequent investigations should concentrate on in vivo studies and the refinement of formulations to improve efficacy and safety.

Antibacterial activity of the synthesized Ch-SPIONs: (a) K. pneumoniae and (b). S. aureus.
3.6 Anticancer potential of the synthesized Ch-SPIONs
3.6.1 Cytotoxicity by the MTT assay
Cervical cancer persists as a major worldwide health issue, and the pursuit of effective therapeutic agents is ongoing. This study assesses the anticancer efficacy of chitosan-encapsulated Ch-SPIONs synthesized from C. papaya extracts, concentrating on their cytotoxic effects against cervical cancer cell lines, evaluated via the MTT assay (Figure 14), with IC50 values provided in Table 1. The MTT experiment demonstrated a pronounced dose-dependent cytotoxicity of Ch-SPIONs on cervical cancer cells. Figure 14 illustrates that Ch-SPIONs effectively inhibited the proliferation of HeLa cells at particular concentrations (10, 25, 50 µg·mL−1), with an IC50 value of 18 µg·mL−1, in contrast to 11 µg·mL−1 for doxorubicin (the reference standard). Ch-SPIONs showed superior inhibitory efficacy on cell proliferation compared to doxorubicin.

Effect of the synthesized Ch-SPIONs on HeLa cells by using the MTT assay.
Cytotoxic activity of complexes (µg·mL−1) by the MTT assay
| Sample | Cells | (Inhibitory concentration/IC50) | |
|---|---|---|---|
| Sample | Dox | ||
| Ch-SPIONs | HeLa | 18 ± 0.4 | 11 ± 0.5 |
IC50 – values of the respective samples (at 24 h).
3.6.2 Morphological changes in HeLa cells treated with Ch-SPIONs
Figure 15 illustrates the morphological changes in HeLa cells following 24 h of treatment with the IC50 concentration of Ch-SPIONs. Microscopic examination of the treated HeLa cells revealed distinct morphological alterations, including cell shrinkage, rounding, and detachment from the substrate, all of which are indicative of apoptosis induction. These cytotoxic effects of Ch-SPIONs on HeLa cells can be attributed to multiple factors. The observed changes suggest that Ch-SPIONs effectively triggered apoptosis in HeLa cells (Figure 15b–d), whereas the untreated control cells showed no significant changes (Figure 15a). The bioactive compounds from C. papaya may exert direct cytotoxic effects through the induction of oxidative stress, leading to apoptosis. The superparamagnetic nature of SPIONs facilitates enhanced cellular uptake, potentially leading to increased accumulation within cancer cells and subsequent cytotoxic effects [54]. Chitosan not only enhances the stability and biocompatibility of the nanoparticles but may also promote cellular internalization, thereby increasing the local concentration of the active compounds at the target site. The significant reduction in cell viability suggests that Ch-SPIONs could be a promising adjunct to existing cervical cancer therapies, potentially enhancing treatment efficacy while minimizing side effects associated with conventional chemotherapeutics [55]. The synthesized Ch-SPIONs exhibit significant anticancer potential against cervical cancer cells, as demonstrated by the MTT assay. Their dose-dependent cytotoxicity, combined with the induction of morphological changes characteristic of apoptosis, underscores their promise as a therapeutic option.

Morphological analysis of synthesized Ch-SPIONS -treated HeLa cells for 24 h: (a) Control, (b) 10 µg·mL−1, (c) 25 µg·mL−1, and (d) 50 µg·mL−1. The scale bar represents 50 µm.
3.6.3 AO/EtBr staining for fluorescence microscopy investigation
The apoptogenic effect of synthesized Ch-SPIONs on HeLa cancer cells was evaluated using fluorescence microscopy. No agglomeration of Ch-SPIONs was detected during the experiment, as the majority of the nanoparticles were adequately distributed at all evaluated low concentrations. To assess apoptosis induction in HeLa cells, the cells were exposed to the IC50 concentration of synthesized Ch-SPIONs and subsequently stained with AO/EtBr. The findings indicated that viable cells emitted green fluorescence, whereas non-viable cells displayed red/orange fluorescence. Untreated control cells exhibited a substantial percentage of viable cells (Figure 16a). Conversely, HeLa cells subjected to Ch-SPIONs demonstrated heightened cellular damage, evidenced by nuclear shrinkage, membrane blebbing, and nuclear degradation, manifesting as orangish bodies (Figure 16b–d). Cells undergoing apoptosis showed a combination of green (live cells) and orange/red (apoptotic cells) fluorescence, with a higher proportion of red fluorescence correlating with increased concentrations of Ch-SPIONs. The presence of orange/red fluorescence signified late apoptotic or necrotic cells, especially at higher concentrations. The superparamagnetic properties of SPIONs, in combination with bioactive compounds from C. papaya, likely contribute to the generation of ROS, which induces oxidative stress and triggers apoptotic pathways [56]. Overall, the AO/EtBr staining results demonstrate that synthesized Ch-SPIONs effectively induce apoptosis in cervical cancer cells, highlighting their potential as an effective anticancer agent.
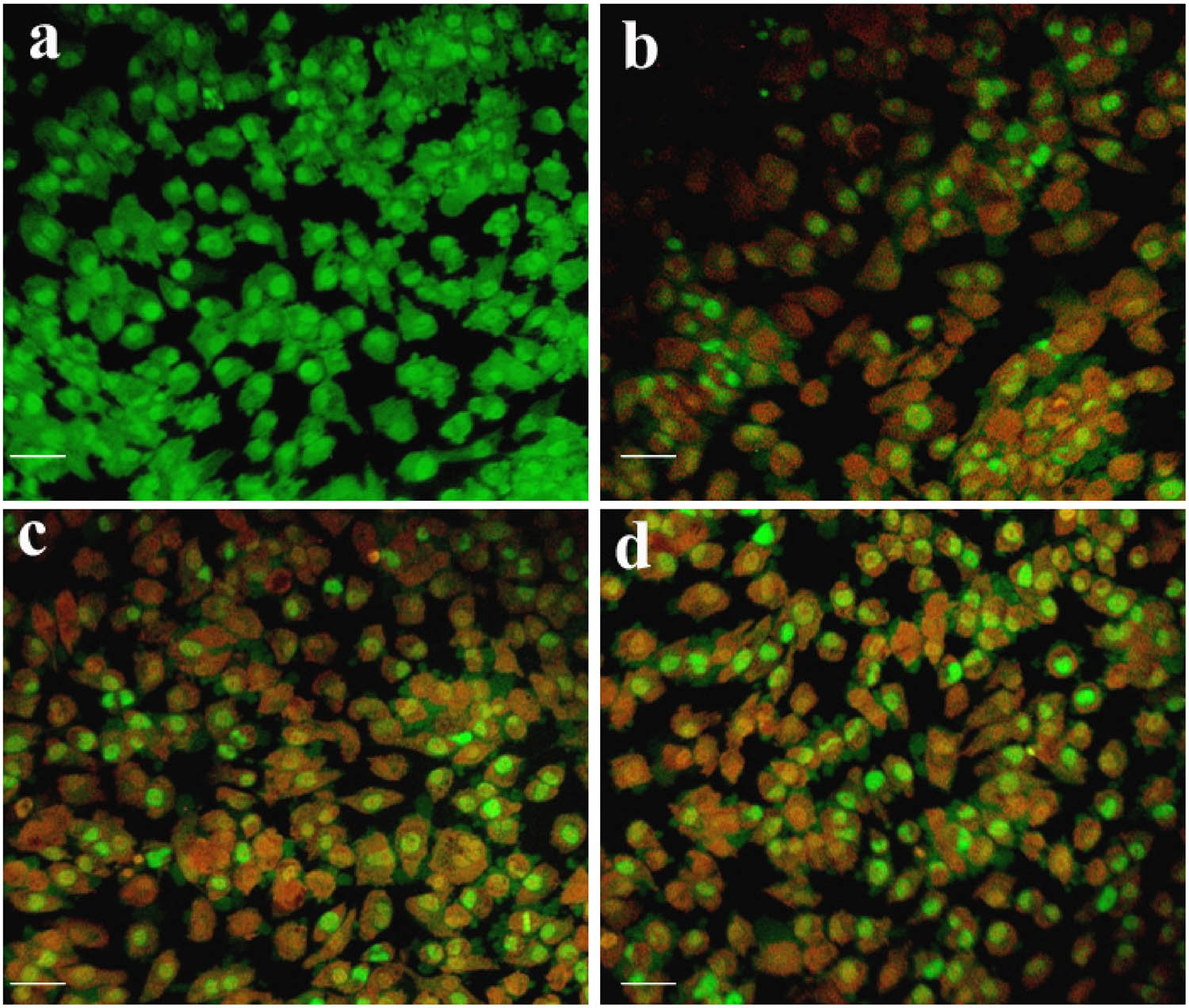
AO/EtBr staining assay of thesynthesized Ch-SPION-treated HeLa cells: (a) Control, (b) 10 µg·mL−1, (c) 25 µg·mL−1, and (d) 50 µg·mL−1. The scale bar represents 50 µm.
3.6.4 Fluorescence microscopy analysis of nuclear fragmentation: DAPI staining
We also assessed Ch-SPIONs using the DAPI staining method. Figure 4 presents fluorescence microscopy images of cells labelled with DAPI following a 24-h period, comparing conditions with and without Ch-SPION treatment. Figure 17a illustrates that untreated cells exhibited no significant changes, with intact nuclei demonstrating uniform blue fluorescence. In contrast, cells treated with Ch-SPIONs exhibited brighter fluorescence signals (Figure 17b–d), with notable nuclear condensation and fragmentation, indicative of late-stage apoptosis. The number of cells with altered nuclear morphology increased markedly, correlating with treatment duration. Normal nuclei appeared bright blue and intact. Cells exhibiting signs of apoptosis showed fragmented nuclei, with a higher proportion of cells displaying irregular shapes and condensed chromatin. Some cells showed diffuse staining and irregular nuclear structures, indicative of necrosis, particularly at higher concentrations [57]. The significant induction of nuclear fragmentation and condensation, as observed by DAPI staining, suggests that Ch-SPIONs effectively trigger apoptotic pathways in cervical cancer cells. The findings align with previous studies, indicating that nanoparticles can induce oxidative stress and apoptosis through various mechanisms. The DAPI staining results indicate that the synthesized Ch-SPIONs effectively induce apoptosis in cervical cancer cells, evidenced by significant nuclear changes and increased percentages of apoptotic nuclei.

DAPI staining assay of the synthesized Ch-SPION-treated HeLa cells: (a) Control, (b) 10 µg·mL−1, (c) 25 µg·mL−1, and (d) 50 µg·mL−1. The scale bar represents 50 µm.
3.6.5 Fluorescence microscopy analysis of PI staining to assess cell membrane integrity and viability
This research examines the anticancer properties of synthesized Ch-SPIONs on cervical cancer cells, utilizing PI staining to evaluate cell membrane integrity and viability. Cervical cancer cells were treated with varying concentrations of Ch-SPIONs (10, 25, and 50 µg·mL−1) for 24 h. Following treatment, cells were stained with PI, which binds to DNA in cells with compromised membranes. Untreated HeLa cells displayed minimal red fluorescence, indicating intact cell membranes and high viability (Figure 18a). After treatment with 10 µg·mL−1 Ch-SPIONs, around 25% of cells were PI-positive, indicating early membrane damage (Figure 18b). After treatment with 25 µg·mL−1 Ch-SPIONs, approximately 60% of the cells exhibited red fluorescence (Figure 18c). When treated with 50 µg·mL−1 Ch-SPIONs, more than 90% of the cells showed significant PI uptake, suggesting extensive necrosis and loss of viability (Figure 18d). Microscopic examination revealed that treated cells exhibited swelling, irregular shapes, and disrupted membrane structures, indicating cell death. In contrast, control cells appeared healthy with intact morphology. The significant increase in PI-positive cells indicates that Ch-SPIONs induce cell death primarily through necrosis, as evidenced by compromised membrane integrity. This may result from oxidative stress generated by the nanoparticles, leading to cellular damage. PI staining serves as a reliable method for assessing cell viability, effectively distinguishing between viable cells and those with compromised membranes [58]. This technique complements other assays and provides a visual confirmation of the cytotoxic effects. The findings indicate that synthesized Ch-SPIONs effectively induce necrosis in cervical cancer cells, as demonstrated by increased PI staining. This underscores their potential as an effective anticancer agent.

PI staining assay of the synthesized Ch-SPION-treated HeLa cells: (a) Control, (b) 10 µg·mL−1, (c) 25 µg·mL−1, and (d) 50 µg·mL−1. The scale bar represents 50 µm.
3.6.6 Apoptotic analysis of cervical cancer cells by flow cytometry
Dead cell proportions were evaluated through flow cytometry employing Annexin V/FITC staining. The proportion of surviving cells in the control group remained constant, whereas the percentage of dead cells markedly increased at elevated concentrations of Ch-SPIONs, especially during the late apoptotic phase. Figure 19 illustrates a significant decrease in viable cell counts at concentrations of 10, 25, and 50 µg·mL−1. Subsequent analysis indicated an accumulation of cells in the sub-G1 phase, a characteristic feature of apoptosis. The proportion of cells in this phase was markedly elevated in the Ch-SPION-treated group relative to the control group. The increase in early and late apoptotic cells indicates that Ch-SPIONs may induce apoptosis through intrinsic pathways, likely involving mitochondrial dysfunction. The accumulation of cells in the sub-G1 phase substantiates this hypothesis, suggesting that Ch-SPIONs induce programmed cell death. The distinctive characteristics of Ch-SPIONs, including biocompatibility and magnetic targeting abilities, could improve their effectiveness in the direct delivery of therapeutic agents to tumour cells, thus elevating local drug concentration and reducing systemic side effects [59]. The findings indicate that Ch-SPIONs may serve as an effective approach for cervical cancer therapy, potentially enhancing treatment outcomes via targeted apoptosis induction.

Apoptotic analysis by flow cytometry of the synthesized Ch-SPION-treated HeLa cells: (a) Control, (b) 10 µg·mL−1, (c) 25 µg·mL−1, and (d) 50 µg·mL−1.
4 Conclusion
Ch-SPIONs exhibit strong antioxidant properties, capable of neutralizing ROS and mitigating oxidative stress, which is implicated in various diseases, including cancer and neurodegenerative disorders. This positions Ch-SPIONs as a potential strategy for preventing oxidative damage and supporting overall health. The synthesized nanoparticles show significant antibacterial effects against a broad spectrum of bacteria. Their ability to disrupt bacterial membranes and inhibit biofilm formation makes them effective against bacterial infections, including antibiotic-resistant strains. This presents Ch-SPIONs as a potential alternative to traditional antibiotics in the fight against antimicrobial resistance (AMR). Ch-SPIONs demonstrate promising anticancer effects in cervical cancer cells, inducing cell death and reducing cell viability. The combination of chitosan’s bioactivity, the magnetic properties of SPIONs, and the bioactive compounds from C. papaya bark extract enhances the anticancer efficacy. These nanoparticles also open possibilities for magnetic targeting and controlled drug delivery, offering a more precise, localized treatment with reduced side effects compared to conventional chemotherapy. The use of C. papaya bark extract for synthesizing the nanoparticles provides a sustainable, eco-friendly approach to nanoparticle production. This method avoids toxic chemicals and offers a biocompatible material that can be safely used in medical applications. Overall, this research highlights chitosan-coated SPIONs as multifunctional nanoparticles with promising applications in antioxidant therapy, antimicrobial treatments, and cancer therapy. Their biocompatibility, sustainability, and synergistic effects make them a novel and versatile tool for addressing complex health issues. This study lays the foundation for further exploration and development of Ch-SPIONs as effective and targeted therapeutic agents for treating bacterial infections and cancer, as well as for providing preventative care against oxidative stress-related diseases.
Acknowledgment
We acknowledge the work was supported by Princess Nourah bint Abdulrahman University Researchers Supporting Project (PNURSP2025R105) Princess Nourah bint Abdulrahman University, Riyadh, Saudi Arabia.
-
Funding information: This work was supported by Princess Nourah bint Abdulrahman University Researchers Supporting Project (PNURSP2025R105) Princess Nourah bint Abdulrahman University, Riyadh, Saudi Arabia.
-
Author contributions: Conceptualization: Hanan Nasser Altamimi and Rasha Assad Assiri. Methods and data interpretation of results: Hanan Nasser Altamimi, Asmatanzeem Bepari, Shaik Kalimulla Niazi, Mohammed Al-Zharani, Ghadah Asaad Assiri, Mujeeb Ahmed Shaikh, ND Manickam Paulpandi. Original draft and final manuscript: Hanan Nasser Altamimi and Rasha Assad Assiri.
-
Conflict of interest: Authors state no conflict of interest.
-
Data availability statement: The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
References
[1] Khan FA, Albalawi R, Pottoo FH. Trends in targeted delivery of nanomaterials in colon cancer diagnosis and treatment. Med Res Rev. 2022;42(1):227–58.10.1002/med.21809Suche in Google Scholar PubMed
[2] Jadid MFS, Jafari-Gharabaghlou D, Bahrami MK, Bonabi E, Zarghami N. Enhanced anti-cancer effect of curcumin loaded-niosomal nanoparticles in combination with heat-killed Saccharomyces cerevisiae against human colon cancer cells. J Drug Delivery Sci Technol. 2023;80:104167.10.1016/j.jddst.2023.104167Suche in Google Scholar
[3] Soto KM, Mendoza S, López-Romero JM, Gasca-Tirado JR, Manzano-Ramírez A. Gold nanoparticles: Synthesis, application in colon cancer therapy and new approaches-review. Green Chem Lett Rev. 2021;14(4):665–78.10.1080/17518253.2021.1998648Suche in Google Scholar
[4] You X, Kang Y, Hollett G, Chen X, Zhao W, Gu Z, et al. Polymeric nanoparticles for colon cancer therapy: overview and perspectives. J Mater Chem B. 2016;4(48):7779–92.10.1039/C6TB01925KSuche in Google Scholar
[5] Siddeeg A, AlKehayez NM, Abu-Hiamed HA, Al-Sanea EA, Al-Farga AM. Mode of action and determination of antioxidant activity in the dietary sources: An overview. Saudi J Biol Sci. 2021;28(3):1633–44.10.1016/j.sjbs.2020.11.064Suche in Google Scholar PubMed PubMed Central
[6] Kuznetsov AA, Filippov VI, Alyautdin RN, Torshina NL, Kuznetsov OA. Application of magnetic liposomes for magnetically guided transport of muscle relaxants and anti-cancer photodynamic drugs. J Magn Magn Mater. 2001;225(1–2):95–100.10.1016/S0304-8853(00)01235-XSuche in Google Scholar
[7] Tuli HS, Kashyap D, Bedi SK, Kumar P, Kumar G, Sandhu SS. Molecular aspects of metal oxide nanoparticle (MO-NPs) mediated pharmacological effects. Life Sci. 2015;143:71–9.10.1016/j.lfs.2015.10.021Suche in Google Scholar PubMed
[8] Mornet S, Vasseur S, Grasset F, Veverka P, Goglio G, Demourgues A, et al. Magnetic nanoparticle design for medical applications. Prog Solid State Chem. 2006;34(2–4):237–47.10.1016/j.progsolidstchem.2005.11.010Suche in Google Scholar
[9] Hu F, Wei L, Zhou Z, Ran Y, Li Z, Gao M. Preparation of biocompatible magnetite nanocrystals for in vivo magnetic resonance detection of cancer. Adv Mater. 2006;18(19):2553–6.10.1002/adma.200600385Suche in Google Scholar
[10] Lu H, Chen A, Zhang X, Wei Z, Cao R, Zhu Y, et al. A pH-responsive T1-T2 dual-modal MRI contrast agent for cancer imaging. Nat Commun. 2022;13(1):7948.10.1038/s41467-022-35655-xSuche in Google Scholar PubMed PubMed Central
[11] Azarmi M, Maleki H, Nikkam N, Malekinejad H. Novel neurolisteriosis therapy using SPION as a drivable nanocarrier in gallic acid delivery to CNS. J Controlled Release. 2023;353:507–17.10.1016/j.jconrel.2022.12.006Suche in Google Scholar PubMed
[12] Liu X, Zhang Y, Wang Y, Zhu W, Li G, Ma X, et al. Comprehensive understanding of magnetic hyperthermia for improving antitumor therapeutic efficacy. Theranostics. 2020;10(8):3793.10.7150/thno.40805Suche in Google Scholar PubMed PubMed Central
[13] Kim S, Kim J, Im J, Kim M, Kim T, Wang SX, et al. Magnetic supercluster particles for highly sensitive magnetic biosensing of proteins. Microchim Acta. 2022;189(7):256.10.1007/s00604-022-05354-xSuche in Google Scholar PubMed PubMed Central
[14] Senapati S, Mahanta AK, Kumar S, Maiti P. Controlled drug delivery vehicles for cancer treatment and their performance. Signal Transduct Target Ther. 2018;3(1):7.10.1038/s41392-017-0004-3Suche in Google Scholar PubMed PubMed Central
[15] Zhi D, Yang T, Yang J, Fu S, Zhang S. Targeting strategies for superparamagnetic iron oxide nanoparticles in cancer therapy. Acta Biomater. 2020;102:13–34.10.1016/j.actbio.2019.11.027Suche in Google Scholar PubMed
[16] Pucci C, Degl’Innocenti A, Gümüş MB, Ciofani G. Superparamagnetic iron oxide nanoparticles for magnetic hyperthermia: Recent advancements, molecular effects, and future directions in the omics era. Biomater Sci. 2022;10(9):2103–21.10.1039/D1BM01963ESuche in Google Scholar
[17] Reddy LH, Arias JL, Nicolas J, Couvreur P. Magnetic nanoparticles: design and characterization, toxicity and biocompatibility, pharmaceutical and biomedical applications. Chem Rev. 2012;112(11):5818–78.10.1021/cr300068pSuche in Google Scholar PubMed
[18] Amstad E, Kohlbrecher J, Müller E, Schweizer T, Textor M, Reimhult E. Triggered release from liposomes through magnetic actuation of iron oxide nanoparticle containing membranes. Nano Lett. 2011;11(4):1664–70.10.1021/nl2001499Suche in Google Scholar PubMed
[19] Salvatore A, Montis C, Berti D, Baglioni P. Multifunctional magnetoliposomes for sequential controlled release. ACS nano. 2016;10(8):7749–60.10.1021/acsnano.6b03194Suche in Google Scholar PubMed
[20] Ye F, Barrefelt Å, Asem H, Abedi-Valugerdi M, El-Serafi I, Saghafian M, et al. Biodegradable polymeric vesicles containing magnetic nanoparticles, quantum dots and anticancer drugs for drug delivery and imaging. Biomaterials. 2014;35(12):3885–94.10.1016/j.biomaterials.2014.01.041Suche in Google Scholar PubMed
[21] Abbas H, Refai H, El Sayed N. Superparamagnetic iron oxide–loaded lipid nanocarriers incorporated in thermosensitive in situ gel for magnetic brain targeting of clonazepam. J Pharm Sci. 2018;107(8):2119–27.10.1016/j.xphs.2018.04.007Suche in Google Scholar PubMed
[22] Ahmad W, Ahmed S, Kumar S. Facile one step microwave assisted biofabrication of Fe2O3 NPs: potential application as solar light-driven photocatalyst in the photodegradation of acridine orange. Int J Environ Anal Chem. 2024;(104):1–16.10.1080/03067319.2024.2344707Suche in Google Scholar
[23] Ahmad W, Joshi H, Ahmed S, Kumar S, Wilson I. Parmelia perlata mediated microwave-assisted one-pot green synthesis of NiO nanoparticles a noble approach: Antibacterial and photocatalytic activity evaluation. Chem Phys Lett. 2024;853:141524.10.1016/j.cplett.2024.141524Suche in Google Scholar
[24] Rahmani R, Gharanfoli M, Gholamin M, Darroudi M, Chamani J, Sadri K, et al. Plant-mediated synthesis of superparamagnetic iron oxide nanoparticles (SPIONs) using aloe vera and flaxseed extracts and evaluation of their cellular toxicities. Ceram Int. 2020;46(3):3051–8.10.1016/j.ceramint.2019.10.005Suche in Google Scholar
[25] Eldeeb BA, El-Raheem WMA, Elbeltagi S. Green synthesis of biocompatible Fe3O4 magnetic nanoparticles using Citrus Sinensis peels extract for their biological activities and magnetic-hyperthermia applications. Sci Rep. 2023;13(1):19000.10.1038/s41598-023-46287-6Suche in Google Scholar PubMed PubMed Central
[26] Andrade-Zavaleta K, Chacon-Laiza Y, Asmat-Campos D, Raquel-Checca N. Green synthesis of superparamagnetic iron oxide nanoparticles with Eucalyptus globulus extract and their application in the removal of heavy metals from agricultural soil. Molecules. 2022;27(4):1367.10.3390/molecules27041367Suche in Google Scholar PubMed PubMed Central
[27] Peréz DL, Puentes I, Romero GM, Gaona IS, Vargas CP, Rincón RJ. Synthesis of superparamagnetic iron oxide nanoparticles coated with polyethylene glycol as potential drug carriers for cancer treatment. J Nanopart Res. 2024;26(1):2.10.1007/s11051-023-05900-5Suche in Google Scholar
[28] Lakshminarayanan S, Shereen MF, Niraimathi KL, Brindha P, Arumugam A. One-pot green synthesis of iron oxide nanoparticles from Bauhinia tomentosa: Characterization and application towards synthesis of 1, 3 diolein. Sci Rep. 2021;11(1):8643.10.1038/s41598-021-87960-ySuche in Google Scholar PubMed PubMed Central
[29] Kafali M, Şahinoğlu OB, Tufan Y, Orsel ZC, Aygun E, Alyuz B, et al. Antibacterial properties and osteoblast interactions of microfluidically synthesized chitosan–SPION composite nanoparticles. J Biomed Mater Res Part A. 2023;111(11):1662–77.Suche in Google Scholar
[30] Ghosh S, Ahire M, Patil S, Jabgunde A, Bhat Dusane M, Joshi BN, et al. Antidiabetic activity of Gnidia glauca and Dioscorea bulbifera: potent amylase and glucosidase inhibitors. Evidence‐Based Complementary Altern Med. 2012;2012(1):929051.10.1155/2012/929051Suche in Google Scholar PubMed PubMed Central
[31] Ajitha B, Reddy YAK, Reddy S. Green synthesis and characterization of silver nanoparticles using Lantana camara leaf extract. Mater Sci Eng: C. 2015;49:373–81.10.1016/j.msec.2015.01.035Suche in Google Scholar PubMed
[32] Sathya TA, Viswanathan S, Kolar AB, Jahirhussain G, Alagumanian S, Sobana S, et al. Environmental profiling of gold nanoparticles by flavonoids fractionalization from Carrica papaya leaf extract for photocatalytic debasement of organic contaminants and it’s cyto-toxic analysis. Environ Res. 2024;259:119445.10.1016/j.envres.2024.119445Suche in Google Scholar PubMed
[33] Rajaganesh R, Murugan K, Panneerselvam C, Jayashanthini S, Roni M, Suresh U, et al. Fern-synthesized silver nanocrystals: towards a new class of mosquito oviposition deterrents? Res Vet Sci. 2016;109:40–51.10.1016/j.rvsc.2016.09.012Suche in Google Scholar PubMed
[34] Rajaganesh R, Murugan K. Anti-dengue potential and mosquitocidal effect of marine green algae–stabilized Mn-doped superparamagnetic iron oxide nanoparticles (Mn-SPIONs): An eco-friendly approach. Environ Sci Pollut Res. 2024;31(13):19575–94.10.1007/s11356-024-32413-ySuche in Google Scholar PubMed
[35] Paulpandi M, Rajaganesh R, Kavithaa K, Kalaiarasi G, Arul N. Curcumin-loaded a-lactalbumin nanoparticles neuroprotective effects against permethrine-induced neurotoxicity. Inorg Chem Commun. 2023;158:111586.10.1016/j.inoche.2023.111586Suche in Google Scholar
[36] Kavithaa K, Paulpandi M, Ponraj T, Murugan K, Sumathi S. Induction of intrinsic apoptotic pathway in human breast cancer (MCF-7) cells through facile biosynthesized zinc oxide nanorods. Karbala Int J Mod Sci. 2016;2(1):46–55.10.1016/j.kijoms.2016.01.002Suche in Google Scholar
[37] Finney DJ. Probit analysis. London: Cambridge University; 1971. p. 68–78.Suche in Google Scholar
[38] Adil M, Filimban FZ, Ambrin, Quddoos A, Sher AA, Naseer M. Phytochemical screening, HPLC analysis, antimicrobial and antioxidant effect of Euphorbia parviflora L.(Euphorbiaceae Juss.). Sci Rep. 2024;14(1):5627.10.1038/s41598-024-55905-wSuche in Google Scholar PubMed PubMed Central
[39] Shah M, Linyan Z, Qasim M, Zewdie Y, Qiu L, Ayaz M. HPLC/MALDI-TOF-MS characterization and nutraceutical potential of Seriphedium quettense extracts: A polarity directed solvent extraction and antioxidant kinetics. J Mol Liq. 2025;426:127349.10.1016/j.molliq.2025.127349Suche in Google Scholar
[40] Tsave O, Psarrou M, Kastrinaki G, Papachristou E, Papi R, Zaspalis V, et al. Functional polysaccharide-coated SPIONs for in vitro mRNA delivery in breast cancer cells. Mater Adv. 2024;5:5410–32.10.1039/D4MA00010BSuche in Google Scholar
[41] Hoffmann N, Tortella G, Hermosilla E, Fincheira P, Diez MC, Lourenço IM, et al. Comparative toxicity assessment of eco-friendly synthesized superparamagnetic iron oxide nanoparticles (SPIONs) in plants and aquatic model organisms. Minerals. 2022;12(4):451.10.3390/min12040451Suche in Google Scholar
[42] Kafali M, Şahinoğlu OB, Tufan Y, Orsel ZC, Aygun E, Alyuz B, et al. Antibacterial properties and osteoblast interactions of microfluidically synthesized chitosan–SPION composite nanoparticles. J Biomed Mater Res Part A. 2023;111(11):1662–77.10.1002/jbm.a.37575Suche in Google Scholar PubMed
[43] Justin C, Samrot AV, Sruthi PD, Sahithya CS, Bhavya KS, Saipriya C. Preparation, characterization and utilization of coreshell super paramagnetic iron oxide nanoparticles for curcumin delivery. PLoS One. 2018;13(7):e0200440.10.1371/journal.pone.0200440Suche in Google Scholar PubMed PubMed Central
[44] Sayahi MH, Sepahdar A, Bazrafkan F, Dehghani F, Mahdavi M, Bahadorikhalili S. Ionic liquid modified SPION@ chitosan as a novel and reusable superparamagnetic catalyst for green one-pot synthesis of pyrido [2, 3-d] pyrimidine-dione derivatives in water. Catalysts. 2023;13(2):290.10.3390/catal13020290Suche in Google Scholar
[45] Farmanbar N, Mohseni S, Darroudi M. Green synthesis of chitosan-coated magnetic nanoparticles for drug delivery of oxaliplatin and irinotecan against colorectal cancer cells. Polym Bull. 2022;79(12):10595–613.10.1007/s00289-021-04066-1Suche in Google Scholar
[46] Jabali MK, Allafchian AR, Jalali SAH, Shakeripour H, Mohammadinezhad R, Rahmani F. Design of a pDNA nanocarrier with ascorbic acid modified chitosan coated on superparamagnetic iron oxide nanoparticles for gene delivery. Colloids Surf, A. 2022;632:127743.10.1016/j.colsurfa.2021.127743Suche in Google Scholar
[47] Lang X, Wang T, Sun M, Chen X, Liu Y. Advances and applications of chitosan-based nanomaterials as oral delivery carriers: A review. Int J Biol Macromol. 2020;154:433–45.10.1016/j.ijbiomac.2020.03.148Suche in Google Scholar PubMed
[48] Taherian A, Esfandiari N, Rouhani S. Breast cancer drug delivery by novel drug-loaded chitosan-coated magnetic nanoparticles. Cancer Nanotechnol. 2021;12:1–20.10.1186/s12645-021-00086-8Suche in Google Scholar
[49] Tyagi N, Gupta, Khan Z, Neupane YR, Mangla B, Mehra N, et al. Superparamagnetic iron-oxide nanoparticles synthesized via green chemistry for the potential treatment of breast cancer. Molecules. 2023;28(5):2343.10.3390/molecules28052343Suche in Google Scholar PubMed PubMed Central
[50] Vinothini P, Malaikozhundan B, Krishnamoorthi R, Senthamarai MD, Shanthi D. Potential inhibition of biofilm forming bacteria and fungi and DPPH free radicals using Tamarindus indica fruit extract assisted iron oxide nanoparticle. Inorg Chem Commun. 2023;156:111206.10.1016/j.inoche.2023.111206Suche in Google Scholar
[51] Turin-Moleavin IA, Fifere A, Lungoci AL, Rosca I, Coroaba A, Peptanariu D, et al. In vitro and in vivo antioxidant activity of the new magnetic-cerium oxide nanoconjugates. Nanomaterials. 2019;9(11):1565.10.3390/nano9111565Suche in Google Scholar PubMed PubMed Central
[52] Ilie CI, Spoiala A, Chircov C, Dolete G, Oprea OC, Vasile BS, et al. Antioxidant, antitumoral, antimicrobial, and prebiotic activity of magnetite nanoparticles loaded with bee pollen/bee bread extracts and 5-fluorouracil. Antioxidants. 2024;13(8):895.10.3390/antiox13080895Suche in Google Scholar PubMed PubMed Central
[53] Seabra AB, Pelegrino MT, Haddad S. Antimicrobial applications of superparamagnetic iron oxide nanoparticles: Perspectives and challenges. Nanostructures for Antimicrobial Therapy. Elsevier; 2017. p. 531–50.10.1016/B978-0-323-46152-8.00024-XSuche in Google Scholar
[54] Yusefi M, Shameli K, Ali RR, Pang SW, Teow SY. Evaluating anticancer activity of plant-mediated synthesized iron oxide nanoparticles using Punica granatum fruit peel extract. J Mol Struct. 2020;1204:127539.10.1016/j.molstruc.2019.127539Suche in Google Scholar
[55] Alkahtane AA, Alghamdi HA, Aljasham AT, Alkahtani S. A possible theranostic approach of chitosan-coated iron oxide nanoparticles against human colorectal carcinoma (HCT-116) cell line. Saudi J Biol Sci. 2022;29(1):154–60.10.1016/j.sjbs.2021.08.078Suche in Google Scholar PubMed PubMed Central
[56] Bharathi VU, Thambidurai S. Green synthesized chitosan-coated iron oxide nanocomposite using Cissus quadrangularis plant extract for antibacterial, antioxidant and anticancer applications. Inorg Chim Acta. 2024;572:122293.10.1016/j.ica.2024.122293Suche in Google Scholar
[57] Mickymaray S, Al Aboody MS, Eraqi MM, Alhoqail WA, Alothaim AS, Suresh K, et al. Chitosan-encapsulated nickel oxide, tin dioxide, and farnesol nanoparticles: antimicrobial and anticancer properties in breast cancer cells. Int J Biol Macromol. 2023;248:125799.10.1016/j.ijbiomac.2023.125799Suche in Google Scholar PubMed
[58] Farghadani R, Lim HY, Abdulla MA, Rajarajeswaran J. Novel indole Schiff base β-diiminato compound as an anti-cancer agent against triple-negative breast cancer: In vitro anticancer activity evaluation and in vivo acute toxicity study. Bioorganic Chem. 2024;152:107730.10.1016/j.bioorg.2024.107730Suche in Google Scholar PubMed
[59] Girija NS, Neethi B, Sakthimanipriya L, Sinekha MA, Shanmugapriya P, Meenakumari R. In-vitro cytotoxic, antiproliferative and apoptotic activity of siddha formulation Nandhi Mezhugu (NM) against Hela cell line. Appl Biochem Biotechnol. 2024;196(3):1612–22.10.1007/s12010-023-04657-wSuche in Google Scholar PubMed
© 2025 the author(s), published by De Gruyter
This work is licensed under the Creative Commons Attribution 4.0 International License.
Artikel in diesem Heft
- Synthesis of N,S co-doped carbon quantum dots – metal complex for the detection of fluoride (F−) ion in adults and Children’s toothpastes
- Research Articles
- Optimized green synthesis of silver nanoparticles from guarana seed skin extract with antibacterial potential
- Green adsorbents for water remediation: Removal of Cr(vi) and Ni(ii) using Prosopis glandulosa sawdust and biochar
- Green approach for the synthesis of zinc oxide nanoparticles from methanolic stem extract of Andrographis paniculata and evaluation of antidiabetic activity: In silico GSK-3β analysis
- Development of a green and rapid ethanol-based HPLC assay for aspirin tablets and feasibility evaluation of domestically produced bioethanol in Thailand as a sustainable mobile phase
- A facile biodegradation of polystyrene microplastic by Bacillus subtilis
- Enhanced synthesis of fly ash-derived hydrated sodium silicate adsorbents via low-temperature alkaline hydrothermal treatment for advanced environmental applications
- Impact of metal nanoparticles biosynthesized using camel milk on bacterial growth and copper removal from wastewater
- Preparation of Co/Cr-MOFs for efficient removal of fleroxacin and Rhodamine B
- Applying nanocarbon prepared from coal as an anode in lithium-ion batteries
- Improved electrochemical synthesis of Cu–Fe/brass foil alloy followed by combustion for high-efficiency photoelectrodes and hydrogen production in alkaline solutions
- Precipitation of terephthalic acid from post-consumer polyethylene terephthalate waste fractions
- Biosynthesized zinc oxide nanoparticles: Multifunctional potential applications in anticancer, antibacterial, and B. subtilis DNA gyrase docking
- Anticancer and antimicrobial effects of green-synthesized silver nanoparticles using Teucrium polium leaves extract
- Green synthesis of eco-friendly bioplastics from Chlorella and Lithothamnion algae for safe and sustainable solutions for food packaging
- Optimizing coal water slurry concentration via synergistic coal blending and particle size distribution
- Green synthesis of Ag@Cu and silver nanowire using Pterospermum heterophyllum extracts for surface-enhanced Raman scattering
- Green synthesis of copper oxide nanoparticles from Algerian propolis: Exploring biochemical, structural, antimicrobial, and anti-diabetic properties
- Simultaneous quantification of mefenamic acid and paracetamol in fixed-dose combination tablet dosage forms using the green HPTLC method
- Green synthesis of titanium dioxide nanoparticles using green tea (Camellia sinensis) extract: Characteristics and applications
- Pharmaceutical properties for green fabricated ZnO and Ag nanoparticle-mediated Borago officinalis: In silico predications study
- Synthesis and optimization of gemcitabine-loaded nanoparticles by using Box–Behnken design for treating prostate cancer: In vitro characterization and in vivo pharmacokinetic study
- A comparative analysis of single-step and multi-step methods for producing magnetic activated carbon from palm kernel shells: Adsorption of methyl orange dye
- Sustainable green synthesis of silver nanoparticles using walnut septum waste: Characterization and antibacterial properties
- Efficient electrocatalytic reduction of CO2 to CO over Ni/Y diatomic catalysts
- Greener and magnetic Fe3O4 nanoparticles as a recyclable catalyst for Knoevenagel condensation and degradation of industrial Congo red dye
- Recycling of HDPE-giant reed composites: Processability and performance
- Fabrication of antibacterial chitosan/PVA nanofibers co-loaded with curcumin and cefadroxil for wound healing
- Cost-effective one-pot fabrication of iron(iii) oxychloride–iron(iii) oxide nanomaterials for supercapacitor charge storage
- Novel trimetallic (TiO2–MgO–Au) nanoparticles: Biosynthesis, characterization, antimicrobial, and anticancer activities
- Green-synthesized chromium oxide nanoparticles using pomegranate husk extract: Multifunctional bioactivity in antioxidant potential, lipase and amylase inhibition, and cytotoxicity
- Therapeutic potential of sustainable zinc oxide nanoparticles biosynthesized using Tradescantia spathacea aqueous leaf extract
- Chitosan-coated superparamagnetic iron oxide nanoparticles synthesized using Carica papaya bark extract: Evaluation of antioxidant, antibacterial, and anticancer activity of HeLa cervical cancer cells
- Antioxidant potential of peptide fractions from tuna dark muscle protein isolate: A green enzymatic approach
- Clerodendron phlomoides leaf extract-mediated synthesis of selenium nanoparticles for multi-applications
- Optimization of cellulose yield from oil palm trunks with deep eutectic solvents using response surface methodology
- Nitrogen-doped carbon dots from Brahmi (Bacopa monnieri): Metal-free probe for efficient detection of metal pollutants and methylene blue dye degradation
- High energy density pseudocapacitor based on a nanoporous tungsten(VI) oxide iodide/poly(2-amino-1-mercaptobenzene) composite
- Green synthesized Ag–Cu nanocomposites as an improved strategy to fight multidrug-resistant bacteria by inhibition of biofilm formation: In vitro and in silico assessment study
- In vitro evaluation of antibacterial activity and associated cytotoxicity of biogenic silver nanoparticles using various extracts of Tabernaemontana ventricosa
- Fabrication of novel composite materials by impregnating ZnO particles into bacterial cellulose nanofibers for antimicrobial applications
- Solidification floating organic drop for dispersive liquid–liquid microextraction estimation of copper in different water samples
- Kinetics and synthesis of formation of phosphate composites from low-grade phosphorites in the presence of phosphate–siliceous shales and oil sludge
- Removal of minocycline and terramycin by graphene oxide and Cr/Mn base metal–organic framework composites
- Microfluidic preparation of ceramide E liposomes and properties
- Therapeutic potential of Anamirta cocculus (L.) Wight & Arn. leaf aqueous extract-mediated biogenic gold nanoparticles
- Antioxidant-rich Micromeria imbricata leaf extract as a medium for the eco-friendly preparation of silver-doped zinc oxide nanoparticles with antibacterial properties
- Influence of different colors with light regime on Chlorella sp., biomass, pigments, and lipids quantity and quality
- Experimental vibrational analysis of natural fiber composite reinforced with waste materials for energy absorbing applications
- Green synthesis of sea buckthorn-mediated ZnO nanoparticles: Biological applications and acute nanotoxicity studies
- Production of liquid smoke by consecutive electroporation and microwave-assisted pyrolysis of empty fruit bunches
- Synthesis of MPAA based on polyacrylamide and gossypol resin and applications in the encapsulation of ammophos
- Application of iron-based catalysts in the microwave treatment of environmental pollutants
- Enhanced adsorption of Cu(ii) from wastewater using potassium humate-modified coconut husk biochar
- Adsorption of heavy metal ions from water by Fe3O4 nano-particles
- Green synthesis of parsley-derived silver nanoparticles and their enhanced antimicrobial and antioxidant effects against foodborne resistant bacteria
- Unwrapping the phytofabrication of bimetallic silver–selenium nanoparticles: Antibacterial, Anti-virulence (Targeting magA and toxA genes), anti-diabetic, antioxidant, anti-ovarian, and anti-prostate cancer activities
- Optimizing ultrasound-assisted extraction process of anti-inflammatory ingredients from Launaea sarmentosa: A novel approach
- Eggshell membranes as green carriers for Burkholderia cepacia lipase: A biocatalytic strategy for sustainable wastewater bioremediation
- Research progress of deep eutectic solvents in fuel desulfurization
- Enhanced electrochemical synthesis of Ni–Fe/brass foil alloy with subsequent combustion for high-performance photoelectrode and hydrogen production applications
- Valorization of baobab fruit shell as a filler fiber for enhanced polyethylene degradation and soil fertility
- Valorization of Agave durangensis bagasse for cardboard-type paper production circular economy approach
- Green priming strategies using seaweed extract and citric acid to improve early growth and antioxidant activity in lentil
- Gold extraction from oxide ore using iron ion-thiourea-additive
- Development of loess-derived P-type molecular sieve as a sustainable antibacterial agent
- Review Article
- Sustainable innovations in garlic extraction: A comprehensive review and bibliometric analysis of green extraction methods
- Natural sustainable coatings for marine applications: advances, challenges, and future perspectives
- Integration of traditional medicinal plants with polymeric nanofibers for wound healing
- Rapid Communication
- In situ supported rhodium catalyst on mesoporous silica for chemoselective hydrogenation of nitriles to primary amines
- Special Issue: Valorisation of Biowaste to Nanomaterials for Environmental Applications
- Valorization of coconut husk into biochar for lead (Pb2+) adsorption
- Corrigendum
- Corrigendum to “An updated review on carbon nanomaterials: Types, synthesis, functionalization and applications, degradation and toxicity”
Artikel in diesem Heft
- Synthesis of N,S co-doped carbon quantum dots – metal complex for the detection of fluoride (F−) ion in adults and Children’s toothpastes
- Research Articles
- Optimized green synthesis of silver nanoparticles from guarana seed skin extract with antibacterial potential
- Green adsorbents for water remediation: Removal of Cr(vi) and Ni(ii) using Prosopis glandulosa sawdust and biochar
- Green approach for the synthesis of zinc oxide nanoparticles from methanolic stem extract of Andrographis paniculata and evaluation of antidiabetic activity: In silico GSK-3β analysis
- Development of a green and rapid ethanol-based HPLC assay for aspirin tablets and feasibility evaluation of domestically produced bioethanol in Thailand as a sustainable mobile phase
- A facile biodegradation of polystyrene microplastic by Bacillus subtilis
- Enhanced synthesis of fly ash-derived hydrated sodium silicate adsorbents via low-temperature alkaline hydrothermal treatment for advanced environmental applications
- Impact of metal nanoparticles biosynthesized using camel milk on bacterial growth and copper removal from wastewater
- Preparation of Co/Cr-MOFs for efficient removal of fleroxacin and Rhodamine B
- Applying nanocarbon prepared from coal as an anode in lithium-ion batteries
- Improved electrochemical synthesis of Cu–Fe/brass foil alloy followed by combustion for high-efficiency photoelectrodes and hydrogen production in alkaline solutions
- Precipitation of terephthalic acid from post-consumer polyethylene terephthalate waste fractions
- Biosynthesized zinc oxide nanoparticles: Multifunctional potential applications in anticancer, antibacterial, and B. subtilis DNA gyrase docking
- Anticancer and antimicrobial effects of green-synthesized silver nanoparticles using Teucrium polium leaves extract
- Green synthesis of eco-friendly bioplastics from Chlorella and Lithothamnion algae for safe and sustainable solutions for food packaging
- Optimizing coal water slurry concentration via synergistic coal blending and particle size distribution
- Green synthesis of Ag@Cu and silver nanowire using Pterospermum heterophyllum extracts for surface-enhanced Raman scattering
- Green synthesis of copper oxide nanoparticles from Algerian propolis: Exploring biochemical, structural, antimicrobial, and anti-diabetic properties
- Simultaneous quantification of mefenamic acid and paracetamol in fixed-dose combination tablet dosage forms using the green HPTLC method
- Green synthesis of titanium dioxide nanoparticles using green tea (Camellia sinensis) extract: Characteristics and applications
- Pharmaceutical properties for green fabricated ZnO and Ag nanoparticle-mediated Borago officinalis: In silico predications study
- Synthesis and optimization of gemcitabine-loaded nanoparticles by using Box–Behnken design for treating prostate cancer: In vitro characterization and in vivo pharmacokinetic study
- A comparative analysis of single-step and multi-step methods for producing magnetic activated carbon from palm kernel shells: Adsorption of methyl orange dye
- Sustainable green synthesis of silver nanoparticles using walnut septum waste: Characterization and antibacterial properties
- Efficient electrocatalytic reduction of CO2 to CO over Ni/Y diatomic catalysts
- Greener and magnetic Fe3O4 nanoparticles as a recyclable catalyst for Knoevenagel condensation and degradation of industrial Congo red dye
- Recycling of HDPE-giant reed composites: Processability and performance
- Fabrication of antibacterial chitosan/PVA nanofibers co-loaded with curcumin and cefadroxil for wound healing
- Cost-effective one-pot fabrication of iron(iii) oxychloride–iron(iii) oxide nanomaterials for supercapacitor charge storage
- Novel trimetallic (TiO2–MgO–Au) nanoparticles: Biosynthesis, characterization, antimicrobial, and anticancer activities
- Green-synthesized chromium oxide nanoparticles using pomegranate husk extract: Multifunctional bioactivity in antioxidant potential, lipase and amylase inhibition, and cytotoxicity
- Therapeutic potential of sustainable zinc oxide nanoparticles biosynthesized using Tradescantia spathacea aqueous leaf extract
- Chitosan-coated superparamagnetic iron oxide nanoparticles synthesized using Carica papaya bark extract: Evaluation of antioxidant, antibacterial, and anticancer activity of HeLa cervical cancer cells
- Antioxidant potential of peptide fractions from tuna dark muscle protein isolate: A green enzymatic approach
- Clerodendron phlomoides leaf extract-mediated synthesis of selenium nanoparticles for multi-applications
- Optimization of cellulose yield from oil palm trunks with deep eutectic solvents using response surface methodology
- Nitrogen-doped carbon dots from Brahmi (Bacopa monnieri): Metal-free probe for efficient detection of metal pollutants and methylene blue dye degradation
- High energy density pseudocapacitor based on a nanoporous tungsten(VI) oxide iodide/poly(2-amino-1-mercaptobenzene) composite
- Green synthesized Ag–Cu nanocomposites as an improved strategy to fight multidrug-resistant bacteria by inhibition of biofilm formation: In vitro and in silico assessment study
- In vitro evaluation of antibacterial activity and associated cytotoxicity of biogenic silver nanoparticles using various extracts of Tabernaemontana ventricosa
- Fabrication of novel composite materials by impregnating ZnO particles into bacterial cellulose nanofibers for antimicrobial applications
- Solidification floating organic drop for dispersive liquid–liquid microextraction estimation of copper in different water samples
- Kinetics and synthesis of formation of phosphate composites from low-grade phosphorites in the presence of phosphate–siliceous shales and oil sludge
- Removal of minocycline and terramycin by graphene oxide and Cr/Mn base metal–organic framework composites
- Microfluidic preparation of ceramide E liposomes and properties
- Therapeutic potential of Anamirta cocculus (L.) Wight & Arn. leaf aqueous extract-mediated biogenic gold nanoparticles
- Antioxidant-rich Micromeria imbricata leaf extract as a medium for the eco-friendly preparation of silver-doped zinc oxide nanoparticles with antibacterial properties
- Influence of different colors with light regime on Chlorella sp., biomass, pigments, and lipids quantity and quality
- Experimental vibrational analysis of natural fiber composite reinforced with waste materials for energy absorbing applications
- Green synthesis of sea buckthorn-mediated ZnO nanoparticles: Biological applications and acute nanotoxicity studies
- Production of liquid smoke by consecutive electroporation and microwave-assisted pyrolysis of empty fruit bunches
- Synthesis of MPAA based on polyacrylamide and gossypol resin and applications in the encapsulation of ammophos
- Application of iron-based catalysts in the microwave treatment of environmental pollutants
- Enhanced adsorption of Cu(ii) from wastewater using potassium humate-modified coconut husk biochar
- Adsorption of heavy metal ions from water by Fe3O4 nano-particles
- Green synthesis of parsley-derived silver nanoparticles and their enhanced antimicrobial and antioxidant effects against foodborne resistant bacteria
- Unwrapping the phytofabrication of bimetallic silver–selenium nanoparticles: Antibacterial, Anti-virulence (Targeting magA and toxA genes), anti-diabetic, antioxidant, anti-ovarian, and anti-prostate cancer activities
- Optimizing ultrasound-assisted extraction process of anti-inflammatory ingredients from Launaea sarmentosa: A novel approach
- Eggshell membranes as green carriers for Burkholderia cepacia lipase: A biocatalytic strategy for sustainable wastewater bioremediation
- Research progress of deep eutectic solvents in fuel desulfurization
- Enhanced electrochemical synthesis of Ni–Fe/brass foil alloy with subsequent combustion for high-performance photoelectrode and hydrogen production applications
- Valorization of baobab fruit shell as a filler fiber for enhanced polyethylene degradation and soil fertility
- Valorization of Agave durangensis bagasse for cardboard-type paper production circular economy approach
- Green priming strategies using seaweed extract and citric acid to improve early growth and antioxidant activity in lentil
- Gold extraction from oxide ore using iron ion-thiourea-additive
- Development of loess-derived P-type molecular sieve as a sustainable antibacterial agent
- Review Article
- Sustainable innovations in garlic extraction: A comprehensive review and bibliometric analysis of green extraction methods
- Natural sustainable coatings for marine applications: advances, challenges, and future perspectives
- Integration of traditional medicinal plants with polymeric nanofibers for wound healing
- Rapid Communication
- In situ supported rhodium catalyst on mesoporous silica for chemoselective hydrogenation of nitriles to primary amines
- Special Issue: Valorisation of Biowaste to Nanomaterials for Environmental Applications
- Valorization of coconut husk into biochar for lead (Pb2+) adsorption
- Corrigendum
- Corrigendum to “An updated review on carbon nanomaterials: Types, synthesis, functionalization and applications, degradation and toxicity”

