Abstract
C18H24F12N4P2, monoclinic, P21/n (no. 14), a = 8.3185(9) Å, b = 21.883(2) Å, c = 13.4345(15) Å, β = 103.4730(10)°, V = 2378.3(5) Å3, Z = 4, Rgt(F) = 0.0531, wRref(F2) = 0.1424, T = 296(2) K.
Table 1 contains crystallographic data and Table 2 contains the list of the atoms including atomic coordinates and displacement parameters.
Data collection and handling.
| Crystal: | Colorless block |
| Size | 0.20 × 0.14 × 0.13 mm |
| Wavelength: | Mo Kα radiation (0.71073 Å) |
| μ: | 0.28 mm−1 |
| Diffractometer, scan mode: | Bruker APEX-II, φ and ω |
| θmax, completeness: | 25.5°, 99 % |
| N(hkl)measured, N(hkl)unique, Rint: | 12957, 4654, 0.044 |
| Criterion for Iobs, N(hkl)gt: | Iobs > 2 σ(Iobs), 2189 |
| N(param)refined: | 454 |
| Programs: | Bruker [1], SHELX [2], [, 3], Diamond [4] |
Fractional atomic coordinates and isotropic or equivalent isotropic displacement parameters (Å2).
| Atom | x | y | z | Uiso*/Ueq |
|---|---|---|---|---|
| C1 | −0.2587 (11) | 0.0640 (5) | 0.0084 (2) | 0.191 (3) |
| H1A | −0.1472 | 0.0790 | −0.0081 | 0.287* |
| H1B | −0.3653 | 0.1015 | −0.0063 | 0.287* |
| H1C | −0.2841 | −0.0099 | 0.0071 | 0.287* |
| C2 | −0.2277 (8) | 0.0965 (4) | 0.05750 (19) | 0.1330 (19) |
| H2A | −0.1208 | 0.0578 | 0.0723 | 0.160* |
| H2B | −0.3392 | 0.0790 | 0.0742 | 0.160* |
| C3 | −0.0359 (6) | 0.2543 (4) | 0.08282 (13) | 0.0770 (11) |
| H3 | 0.0665 | 0.2176 | 0.0973 | 0.092* |
| C4 | −0.3035 (6) | 0.2891 (4) | 0.04575 (15) | 0.0909 (13) |
| H4 | −0.4223 | 0.2812 | 0.0296 | 0.109* |
| C5 | −0.2171 (6) | 0.3798 (4) | 0.05631 (14) | 0.0831 (12) |
| H5 | −0.2637 | 0.4465 | 0.0490 | 0.100* |
| C6 | 0.0991 (5) | 0.4301 (3) | 0.09713 (13) | 0.0759 (11) |
| H6A | 0.1112 | 0.4841 | 0.0733 | 0.091* |
| H6B | 0.2199 | 0.3937 | 0.1013 | 0.091* |
| C7 | 0.0581 (5) | 0.4814 (3) | 0.14343 (12) | 0.0589 (9) |
| C8 | −0.0909 (5) | 0.4497 (3) | 0.16896 (13) | 0.0669 (10) |
| H8 | −0.1683 | 0.3947 | 0.1578 | 0.080* |
| C9 | −0.1266 (5) | 0.4992 (3) | 0.21120 (14) | 0.0748 (11) |
| H9 | −0.2287 | 0.4776 | 0.2280 | 0.090* |
| C10 | −0.0134 (6) | 0.5793 (3) | 0.22832 (13) | 0.0749 (11) |
| H10 | −0.0376 | 0.6121 | 0.2567 | 0.090* |
| C11 | 0.1370 (5) | 0.6113 (3) | 0.20330 (13) | 0.0712 (10) |
| H11 | 0.2148 | 0.6656 | 0.2150 | 0.085* |
| C12 | 0.1736 (4) | 0.5630 (3) | 0.16064 (12) | 0.0619 (9) |
| C13 | 0.3395 (5) | 0.5978 (3) | 0.13295 (14) | 0.0861 (12) |
| H13A | 0.4430 | 0.5481 | 0.1385 | 0.103* |
| H13B | 0.3007 | 0.5965 | 0.0993 | 0.103* |
| C14 | 0.5630 (5) | 0.7254(3) | 0.17234 (14) | 0.0741 (11) |
| H14 | 0.6494 | 0.6771 | 0.1860 | 0.089* |
| C15 | 0.3200 (6) | 0.7932 (4) | 0.13238 (16) | 0.0922 (13) |
| H15 | 0.2075 | 0.8000 | 0.1134 | 0.111* |
| C16 | 0.4249 (7) | 0.8706 (4) | 0.15166 (17) | 0.0943 (13) |
| H16 | 0.3991 | 0.9419 | 0.1486 | 0.113* |
| C17 | 0.7331 (7) | 0.8837 (4) | 0.20233 (16) | 0.1075 (15) |
| H17A | 0.6850 | 0.9495 | 0.2140 | 0.129* |
| H17B | 0.7782 | 0.8424 | 0.2297 | 0.129* |
| C18 | 0.8873 (7) | 0.9050 (5) | 0.1748 (2) | 0.161 (3) |
| H18A | 0.8412 | 0.9359 | 0.1451 | 0.241* |
| H18B | 0.9529 | 0.8409 | 0.1688 | 0.241* |
| H18C | 0.9729 | 0.9528 | 0.1916 | 0.241* |
| N1 | −0.1893 (5) | 0.2108 (3) | 0.06249 (11) | 0.0823 (10) |
| N2 | −0.0468 (4) | 0.3561 (3) | 0.07993 (10) | 0.0682 (8) |
| N3 | 0.4082 (4) | 0.7021 (3) | 0.14572 (11) | 0.0729 (9) |
| N4 | 0.5761 (5) | 0.8274 (3) | 0.17660 (11) | 0.0771 (9) |
| P1a | 0.4597 (7) | 0.2120 (4) | 0.1793 (2) | 0.0593 (15) |
| P2a | 0.7963 (12) | 0.7189 (8) | 0.0430 (3) | 0.108 (3) |
| F1a | 0.5375 (13) | 0.2689 (8) | 0.2241 (3) | 0.121 (4) |
| F2a | 0.3941 (19) | 0.1501 (12) | 0.1348 (4) | 0.145 (5) |
| F3a | 0.6654 (14) | 0.1857 (15) | 0.1722 (6) | 0.120 (4) |
| F4a | 0.266 (2) | 0.2451 (13) | 0.1886 (6) | 0.178 (6) |
| F5a | 0.488 (2) | 0.3069 (10) | 0.1497 (5) | 0.126 (4) |
| F6a | 0.4387 (19) | 0.1105 (9) | 0.2069 (6) | 0.163 (6) |
| F7a | 0.9713 (13) | 0.7899 (10) | 0.0442 (4) | 0.148 (4) |
| F8a | 0.6270 (11) | 0.6403 (8) | 0.0423 (4) | 0.127 (4) |
| F9a | 0.9218 (18) | 0.6240 (9) | 0.0272 (5) | 0.146 (5) |
| F10a | 0.6697 (18) | 0.8043 (8) | 0.0610 (4) | 0.136 (4) |
| F11a | 0.7517 (14) | 0.7496 (11) | −0.0088 (3) | 0.129 (6) |
| F12a | 0.8503 (19) | 0.6903 (10) | 0.0958 (4) | 0.129 (4) |
| P3b | 0.7935 (7) | 0.7147 (6) | 0.04360 (18) | 0.051 (2) |
| F13b | 0.786 (2) | 0.7824 (12) | −0.0036 (5) | 0.138 (7) |
| F14b | 0.722 (2) | 0.6223 (6) | 0.0103 (4) | 0.137 (5) |
| F15b | 0.786 (2) | 0.6421 (13) | 0.0889 (5) | 0.144 (7) |
| F16b | 0.866 (3) | 0.8113 (9) | 0.0731 (5) | 0.175 (6) |
| F17b | 1.0006 (16) | 0.6802 (16) | 0.0354 (5) | 0.150 (7) |
| F18b | 0.5795 (13) | 0.7496 (16) | 0.0481 (4) | 0.147 (7) |
| P4b | 0.4495 (15) | 0.2036 (10) | 0.1771 (4) | 0.124 (4) |
| F19b | 0.2389 (17) | 0.2502 (9) | 0.1871 (4) | 0.084 (3) |
| F1Ab | 0.459 (3) | 0.2895 (17) | 0.1347 (7) | 0.172 (10) |
| F1Bb | 0.649 (2) | 0.1511 (18) | 0.1656 (8) | 0.156 (9) |
| F1Cb | 0.4338 (16) | 0.1244 (13) | 0.2212 (5) | 0.119 (5) |
| F1Db | 0.548 (2) | 0.2884 (13) | 0.2137 (7) | 0.190 (9) |
| F1Eb | 0.335 (2) | 0.1257 (13) | 0.1395 (6) | 0.163 (8) |
aOccupancy: 0.558(12).
bOccupancy: 0.442(12).
Source of material
To a stirred solution of 1-ethylimidazole (1.92 g, 0.02 mol) in acetonitrile (25 mL) at 75 °C was added dropwise α,α′-dibromo-o-xylene (2.64 g, 0.01 mol). The mixture was vigorously stirred at 75 °C for about 24 h. After the reaction has been completed (monitored by TLC), the acetonitrile top phase was decanted and the product was washed with ethyl acetate and diethyl ether 3 times respectively. Then residual solvent was removed, and the product was dried in vacuo at 60 °C for 1 h to give a white powder solid in 98.68% yield. The intermediate compound (1.14 g, 2.5 mmol) and KPF6 (0.92 g, 5 mmol) were dissolved in distilled water (25 mL). The mixture stirred for 6–8 h at 95 °C and then cooled slowly. The crystals of the target compound were obtained in 94.36% yield.
Experimental details
All H atoms were included in calculated positions and refined as riding atoms, with C–H = 0.90–0.97 Å with Uiso(H) = 1.5 Ueq(C) for methyl H atoms and 1.2 Ueq(C) for all other H atoms.
Comment
Ionic liquids (ILs) generally refer to a class of low-melting compounds composed of adjustable organic cations and inorganic or organic anions, and have different functions [5], [, 6]. ILs as a new type of environmentally friendly solvent, owing high catalytic efficiency at mild conditions, and can be recycled, etc., has been widely used in catalytic science, extraction and separation, biomass energy, resource conversion and other fields [7], [8], [9]. It is worth mentioning that various functional ionic liquids have been used to produce biodiesel highly efficiently and environmental friendly [10], [11], [12]. Our group is still focusing on the preparation of ionic liquid that are able to catalyze the reaction to biodiesel [13], [, 14]. We have demonstrated that diimidazole-based hexafluorophosphate exhibits the temperature-controlled characteristics in methanol and water [15]. In order to find an ionic liquid to catalyze the reaction with better catalytic and recycled utilization efficiency, we were engaged in synthesizing the novel ionic liquid catalyst with imidazole.
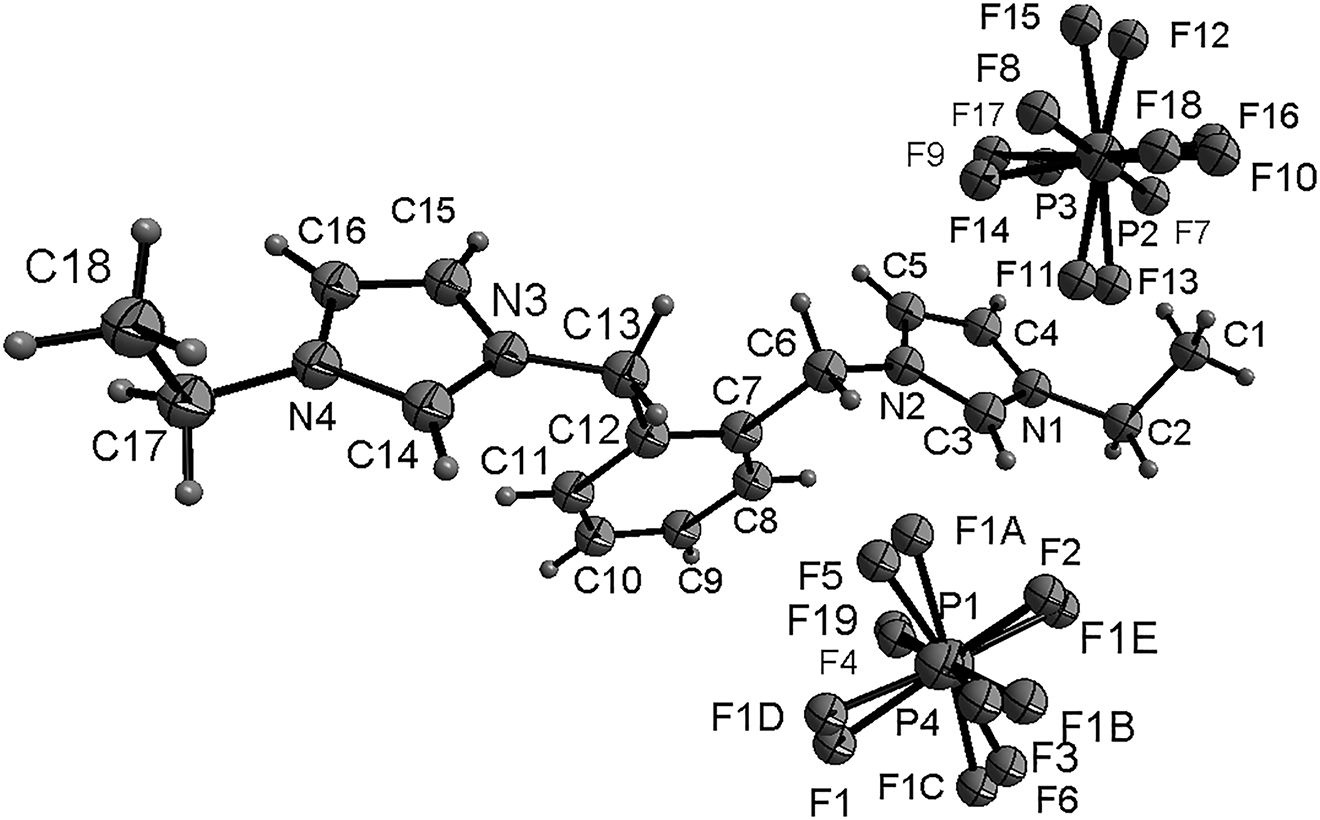
In the cation of the title compound bond lengths and angles are very similar to those given in the literature [16]. The atoms of imidazole ring are coplanar, and the dihedral angle of two imidazole rings and the phenyl group are 6.9(1)°, 82.0(1)° and 84.0(1)°, respectively. The torsion angles of C1–C2–N1–C3, C7–C6–N2–C3, C12–C13–N3–C14 and C18–C17–N4–C14 are −120.4(4)°, −102.4(4)°, 103.7(4)° and 86.8(6)°, respectively.
Funding source: National Natural Science Foundation of China
Award Identifier / Grant number: 31760193
Funding source: Project of Science and Technology Department of Jiangxi Province
Award Identifier / Grant number: 20192ACB60011
Award Identifier / Grant number: 20202BABL205003
Funding source: Key Research Foundation of Educational Department of Jiangxi Province of China
Award Identifier / Grant number: GJJ200404
Award Identifier / Grant number: GJJ190181
Funding source: Natural Science Foundation of Jiangxi Agriculture University
Award Identifier / Grant number: 202010410027
Acknowledgements
X-ray data were collected at Instrumental Analysis Center Nanchang Hangkong University, Nanchang, 330063, People’s Republic of China
Author contribution: All the authors have accepted responsibility for the entire content of this submitted manuscript and approved submission.
Research funding: National Natural Science Foundation of China (No. 31760193), Project of Science and Technology Department of Jiangxi Province (20192ACB60011 and 20202BABL205003), the Key Research Foundation of Educational Department of Jiangxi Province of China (GJJ200404, GJJ190181) and Natural Science Foundation of Jiangxi Agriculture University (No. 202010410027).
Conflict of interest statement: The authors declare no conflicts of interest regarding this article.
References
1. Bruker. APEX2, SAINT and SADABS; Bruker AXS Inc.: Madison, Wisconsin, USA, 2009.Suche in Google Scholar
2. Sheldrick, G. M. A short history of SHELX. Acta Crystallogr. 2008, A64, 112–122; https://doi.org/10.1107/s0108767307043930.Suche in Google Scholar
3. Sheldrick, G. M. Crystal structure refinement with SHELXL. Acta Crystallogr. 2015, C71, 3–8; https://doi.org/10.1107/s2053229614024218.Suche in Google Scholar
4. Brandenburg, K. DIAMOND. Visual Crystal Structure Information System. Ver. 4.0; Crystal Impact: Bonn, Germany, 2015.Suche in Google Scholar
5. Dong, K., Liu, X. M., Dong, H. F., Zhang, X. P., Zhang, S. J. Multiscale studies on ionic liquids. Chem. Rev. 2017, 117, 6636–6695; https://doi.org/10.1021/acs.chemrev.6b00776.Suche in Google Scholar
6. Berthod, A., Ruiz-ángel, M. J., Carda-Broch, S. Recent advances on ionic liquid uses in separation techniques. J. Chromatogr. A 2014, 1559, 2–16.10.1016/j.chroma.2017.09.044Suche in Google Scholar
7. Jin, M. M., Niu, Q. T., Si, C. D., Lv, Z. G., Guo, H. Y., Guo, Z. M. Peroxotungstate-based ionichybrid as a triphase heterogeneous catalyst for efficient benzyl alcohol oxidation under mild conditions. Catal. Lett. 2020, 150, 1692–1706; https://doi.org/10.1007/s10562-019-03071-4.Suche in Google Scholar
8. Kohantorabi, M., Giannakis, S., Gholami, M. R. Supported PtxPd1−x bimetallic nanoparticles on ionic liquid-functionalized SiO2@graphene oxide nanocomposite and its application as an effective multiphasic catalyst. Appl. Catal. 2019, 579, 30–43; https://doi.org/10.1016/j.apcata.2019.04.015.Suche in Google Scholar
9. Wang, J. H., Cheng, D. H., Chen, X. W., Du, Z., Fang, Z. L. Direct extraction of doublestranded DNA into ionic liquid 1-butyl-3- methylimidazolium hexafluorophosphate and its quantification. Anal. Chem. 2007, 79, 620–625; https://doi.org/10.1021/ac061145c.Suche in Google Scholar
10. Li, Y., Hu, S. G., Cheng, J. H., Lou, W. Y. Acidic ionic liquid catalyzed esterification of oleic acid for biodiesel synthesis. Chin. J. Catal. 2014, 35, 396–406; https://doi.org/10.1016/s1872-2067(14)60005-x.Suche in Google Scholar
11. Rafiee, E., Eavani, S. A new organic-inorganic hybrid ionic liquid polyoxometalate for biodiesel production. J. Mol. Liq. 2014, 199, 96–101; https://doi.org/10.1016/j.molliq.2014.08.034.Suche in Google Scholar
12. Kong, J. H., Lan, Y. D., Chen, J., Huang, C. G., Xiong, W. M. Preparation and component analysis of biodiesel catalyzed by functionalized dication ionic liquid. Acta Agric. Univ. Jiangxiensis 2016, 38, 386–390.Suche in Google Scholar
13. Ting, H., Nie, X. L., Chen, J., Zhao, W., Xiong, W. M. Crystal structure of 3-(2-ethoxy-2-oxoethyl)-1-methyl-1H-imidazol-3-ium hexafluoridophosphate(V), C8H13F6N2O2P. Z. Kristallogr. NCS 2019, 234, 1077–1079.10.1515/ncrs-2019-0273Suche in Google Scholar
14. Zhao, W., Chen, J., Xiong, W. M., Lan, Y. D., Nie, X. L. Crystal structure of 1,1′-(hexane-1,6-diyl)bis(3-ethyl-1H-imidazol-3-ium) bis(hexafluorido phosphate), C16H28F12N4P2. Z. Kristallogr. NCS 2019, 234, 609–611.10.1515/ncrs-2018-0527Suche in Google Scholar
15. Huang, T., Zhao, W., Zhang, X. H., Nie, X. L., Chen, J., Xiong, W. M. Synthesis and characterization of diimidazole-based hexafluorophosphate ionic liquids. J. Mol. Liq. 2020, 320, 114465; https://doi.org/10.1016/j.molliq.2020.114465.Suche in Google Scholar
16. Zhou, Y. H., Huang, T., Nie, X. L., Chen, J., Xiong, W. M. Crystal structure of 3,3′-(1,2-phenylenebis(methylene))bis(1-methyl-1H-imidazol-3-ium) bis(hexafluoridophosphate), C16H20F12N4P2. Z. Kristallogr. NCS 2020, 235, 1217–1219.10.1515/ncrs-2020-0268Suche in Google Scholar
© 2021 Wen Zhao et al., published by De Gruyter, Berlin/Boston
This work is licensed under the Creative Commons Attribution 4.0 International License.
Artikel in diesem Heft
- Frontmatter
- New Crystal Structures
- The crystal structure of bis(μ2-5-chloro-2-oxido-N-(1-oxidoethylidene)benzohydrazonato-κ5N,O,O′:N′,O′′)hexkis(pyridine-κ1N)trinickel(II) - pyridine (1/1), C63H57Cl2N13Ni3O6
- Crystal structure of [(μ2-succinato κ3O,O′:O′′)-bis-(5,5,7,12,12,14-hexamethyl-1,4,8,11-tetraazacyclotetradecane)]dinickel(II)] diperchlorate, dihydrate C36H82Cl2N8Ni2O15
- Crystal structure of catena-poly[aquabis(3-nitrobenzoato-κ2O:O′)-(μ2-pyrazine-N: N′)cadmium(II)], C18H14N4O9Cd
- Crystal structure of 4-(2,2-difluoroethyl)-2,4,6-trimethylisoquinoline-1,3(2H,4H)-dione, C14H15F2NO2
- The crystal structure of thioxanthen-9-one-10,10-dioxide, C13H8O3S – a second polymorph
- Crystal structure of (E)-2-((2-methoxy-3-pyridyl)methylene)-7-fluoro-3,4-dihydronaphthalen-1(2H)-one, C17H14FNO2
- The crystal structure of diaquahydrogen 2,5-dimethylbenzenesulphonate, C8H14O5S
- The crystal structure of N-(4-(cyclohexylimino)pent-2-en-2-yl)cyclohexanamine, C17H30N2
- The twinned crystal structure of 1,3-phenylenedimethanaminium dibromide, C8H14Br2N2
- Crystal structure of 2,4,7,9-tetranitro-10H-benzofuro[3,2-b]indole – dimethyl sulfoxide (1/1), C16H11N5O10S
- Crystal structure of 2,6-bis(2-(pyridin-3-yl)ethyl)pyrrolo[3,4-f]isoindole-1,3,5,7(2H,6H)-tetraone, C24H18N4O4
- The crystal structure of 3,4-dichlorobenzoic acid chloride, C7H3Cl3O
- Crystal structure of 1,1′-(1,4-phenylenebis(methylene))bis(pyridin-1-ium) bis(1,2-dicyanoethene-1,2-dithiolato-k2S:S)zinc(II), C26H18N6ZnS4
- Crystal structure of tetrakis(μ-naphthalene-1-carboxylato-κ2O,O′)bis(methanol)copper(II), C46H36Cu2O10
- Crystal structure of 9-methyl-3-methylene-1,2,3,9-tetrahydro-4H-carbazol-4-one, C14H13NO
- Crystal structure of bis(amino(carbamothioylamino)methaniminium) 3-nitrophthalate monohydrate, C12H19N9O7S2
- Crystal structure of 3,3′-(1,2-phenylene-bis(methylene))bis(1-ethyl-1H-imidazol-3-ium) bis(hexafluorophosphate), C18H24F12N4P2
- The crystal structure of 5-hydroxy-6,8-dimethoxy-2-methyl-4H-benzo[g]chromen-4-one– rubrofusarin B, C16H14O5
- The crystal structure of bis(ethanol-kO)- bis(6-aminopicolinato-k2N,O)manganese(II), C16H22O6N4Mn
- The crystal structure of 3,3′-((carbonylbis(azanediyl))bis(ethane-2,1-diyl)) bis(1-methyl-1H-benzo[d]imidazol-3-ium) tetrafluoroborate monohydrate, C21H28N6O3B2F8
- Crystal structure of dimethanol-dichlorido-bis( μ2-2-(((1,5-dimethyl-3-oxo-2- phenyl-2,3-dihydro-1H-pyrazol-4-yl)imino)methyl)phenolato- κ4O:O,O′,N)dinickel (II), C20H24ClNiN3O4
- The crystal structure of methyl 5-(trifluoromethyl)-1H-pyrrole-2-carboxylate, C7H6F3NO2
- Crystal structure of (OC‐6‐13)‐aqua‐tris (3‐bromopyridine‐κ1N)‐bis(trifluoroacetato‐κ1O)cadmium(II) C19H14Br3CdF6N3O5
- Crystal structure of methyl (E)-3-(4-(2-ethoxy-2-oxoethoxy)phenyl) acrylate, C14H16O5
- Crystal structure of methyl 4-acetoxy-3,5-dimethoxybenzoate, C12H14O6
- The crystal structure of 2-(1H-benzimidazol-2-yl)-3-bromo-5-chlorophenol, C13H8BrClN2O
- The crystal structure of bis(μ2-5-chloro-N-(2-methyl-1-oxidopropylidene)-2-oxidobenzohydrazonate-κ5N,O,O′:N′,O′′)pentakis(pyridine-κ1N)tricopper(II), C47H45Cl2N9Cu3O6
- Synthesis and crystal structure of catena-poly[aqua-bis(nitrato-κ2O:O′)- (μ2-((1 H-imidazol-1-yl)methyl)benzene-κ2 N,N′)-H2O-κ2O]cadmium(II), C14H16N6O7Cd
- The crystal structure of pentakis(carbonyl)-{μ-[2,3-bis(sulfanyl)propan-1-olato]}-(triphenylphosphane)diiron (Fe–Fe)C26H21Fe2O6PS2
- Crystal structure of ethyl-2-(3-benzoylthioureido)propanoate, C13H16N2O3S
- Crystal structure of 2-methoxy-4b,5,14,15-tetrahydro-6H-isoquinolino[2′,1′:1,6] pyrazino[2,3-b]quinoxaline, C19H18N4O
- Crystal structure of 2,2′-[ethane-1,2-diylbis(azanylylidenemethylylidene)]bis(6-chlorophenol), C16H14Cl2N2O2
- The crystal structure of (Z)-3-((2-(2-(2-aminophenoxy)ethoxy)phenyl)amino)-1-phenylbut-2-en-1-one, C24H24N2O3
- The crystal structure of 10-(3,5-di(pyridin-4-yl)phenyl)-10H-phenoxazine dihydrate, C28H23N3O3
- Crystal structure of poly[dipoly[aqua-di(µ2-pyrazin-2-olato-κ2N:N′) zinc(II)], C8H8N4O3Zn
- Crystal structure of poly[tetra(μ2-cyanido-κ2N:O)-bis(N,N-dimethylformamide-κO)-manganese(II)-platinum(II)], C10H14MnN6O2Pt
- The crystal structure of aqua-chlorido-6,6′-((ethane-1,2-diylbis(azaneylylidene))bis(methaneylylidene))bis(2,4-dichlorophenolato-κ4N,N′,O,O′)manganese(III), C16H12Cl5MnN2O3
- Crystal structure of [di(µ2-cyanido)-dicyanido-bis(dimethyl sulfoxide-κO)- bis(2,2′-(ethane-1,2-diylbis(azanylylidenemethanylylidene))diphenolato-κ4,N,N′,O,O′)- dimanganese(III)-platinum(II)], C40H40Mn2N8O6PtS2
- The crystal structure of (azido)-κ1N-6,6′-((cyclohexane-1,2-diylbis(azanylylidene)) bis(methanylylidene))bis(3-bromophenolato-κ4N,N,O,O)-(methanol)-manganese(III)–methanol(1/1), C22H26Br2MnN5O4
- Crystal structure of 7-chloro-N-(4-iodobenzyl)-1,2,3,4-tetrahydroacridin-9-amine, C20H18ClIN2
- Crystal structure of catena-poly[(1,4,8,11-tetraazacyclotetradecane-κ4N,N′,N″,N′′′)-bis(μ2-thiocyanato-κ2N:S)-bis(thiocyanato-κS)-nickel(II)palladium(II)], C14H24N8NiPdS4
- Crystal structure of 3-chloro-4-(4-ethylpiperazin-1-yl)aniline monohydrate, C12H20ClN3O
- Crystal structure of the 2D coordination polymer poly[diaqua-bis(μ2-3- methoxyisonicotinato-κ2N:O)cobalt(II)] — dimethylformamide (1/1), C20H30CoN4O10
- Crystal structure of 4-[(5-chloro-2-hydroxybenzylidene)amino]-3-propyl-1H-1,2,4-triazole-5(4H)-thione, C12H13ClN4OS
- Crystal structure of N-(5-(2-(benzyl(1-(4-methoxyphenyl)propan-2-yl)amino)-1-hydroxyethyl)-2-(benzyloxy)phenyl)formamide, C33H36N2O4
- Crystal structure of 3-(methoxycarbonyl)-7-oxabicyclo[2.2.1]heptane-2-carboxylic acid, C9H12O5
- The crystal structure of 1-((dimethylamino)(3-nitrophenyl)methyl)naphthalen-2-ol, C19H18N2O3
- Crystal structure of catena-poly[di(μ2-cyanido-κ2C:N)-dicyanido-tetrakis(dimethyl sulfoxide-κO)-manganese(II)-platinum(II)], C12H24MnN4O4PtS4
- Crystal structure of 4-amino-N-(2-pyrimidinyl)benzenesulfonamide–1,4-dioxane (1/1), C14H18N4O4S
- Crystal structure of bis{1-[(benzotriazol-1-yl)methyl]-1H-1,3-(2-methyl-imidazol)}di-chloridomercury(II), [Hg(C11H11N5)2Cl2], C22H22N10Cl2Hg
- Crystal structure of 2, 3-bis((4-methylbenzoyl)oxy) succinic acid–N, N-dimethylformamide (1/1), C23H25NO9
- Crystal structure of catena-poly[bis(4-(4-carboxyphenoxy)benzoato-κ1O)-μ2-(1,4-bis(1-imidazolyl)benzene-κ2N:N′)cobalt(II)], C40H28N4O10Co
- Crystal structure of 1H-imidazol-3-ium poly[aqua-(μ4-glutarato-κ6O,O′:O′:O′′,O′′′:O′′′)-(nitrato-κ2O,O′)strontium(II)], C8H13N3O8Sr
- Crystal structure of (R)-6-(benzo[b]thiophen-5-yl)-2-methyl-2,6-dihydrobenzo [5,6] silino[4,3,2-cd]indole, C23H17NSSi
- Crystal structure of catena-poly[bis(μ2-thiocyanato-κ2N:S)-(2-(5-methyl-1H-pyrazol-3-yl)pyridine-κ2N,N′)cadmium(II)]–dioxane (1/1), C15H17CdN5O2S2
- Crystal structure of poly[aqua-(μ2-1,4-bis(2′-carboxylatophenoxy)benzene-κ2O:O′)-(μ2-4,4′-bipyridione-κ2N:N′)cadmium(II)] monhydrate, C30H22CdN2O7⋅H2O
- Crystal structure of catena-poly[tetraaqua-(μ2-4,4′-bipyridine-k2N:N′)-bis(μ2-4′-methyl-[1,1′-biphenyl]-3,5-dicarboxylato-k4O,O′:O″,O′″)dicadmium(II)] dihydrate, C20H20NO7Cd
- Crystal structure of 1‐tert‐butyl‐3‐(2,6‐diisopropyl‐4‐phenoxyphenyl)‐2-methylisothiourea, C24H34N2OS
- Crystal structure of catena-poly[triaqua-(μ2-1,3-di(1H-imidazol-1-yl)propane-κ2N:N′)-(4,4′-(1H-1,2,4-triazole-3,5-diyl)dibenzoato-κ1O)cobalt(II)] — N,N′-dimethylformamide (1/1), C28H34N8O8Co
- Crystal structure of tetraaqua-bis(1,4-di(1H-imidazol-1-yl)benzene-κ1N)manganese(II) 2,3-dihydroxyterephthalate, C32H32MnN8O10
Artikel in diesem Heft
- Frontmatter
- New Crystal Structures
- The crystal structure of bis(μ2-5-chloro-2-oxido-N-(1-oxidoethylidene)benzohydrazonato-κ5N,O,O′:N′,O′′)hexkis(pyridine-κ1N)trinickel(II) - pyridine (1/1), C63H57Cl2N13Ni3O6
- Crystal structure of [(μ2-succinato κ3O,O′:O′′)-bis-(5,5,7,12,12,14-hexamethyl-1,4,8,11-tetraazacyclotetradecane)]dinickel(II)] diperchlorate, dihydrate C36H82Cl2N8Ni2O15
- Crystal structure of catena-poly[aquabis(3-nitrobenzoato-κ2O:O′)-(μ2-pyrazine-N: N′)cadmium(II)], C18H14N4O9Cd
- Crystal structure of 4-(2,2-difluoroethyl)-2,4,6-trimethylisoquinoline-1,3(2H,4H)-dione, C14H15F2NO2
- The crystal structure of thioxanthen-9-one-10,10-dioxide, C13H8O3S – a second polymorph
- Crystal structure of (E)-2-((2-methoxy-3-pyridyl)methylene)-7-fluoro-3,4-dihydronaphthalen-1(2H)-one, C17H14FNO2
- The crystal structure of diaquahydrogen 2,5-dimethylbenzenesulphonate, C8H14O5S
- The crystal structure of N-(4-(cyclohexylimino)pent-2-en-2-yl)cyclohexanamine, C17H30N2
- The twinned crystal structure of 1,3-phenylenedimethanaminium dibromide, C8H14Br2N2
- Crystal structure of 2,4,7,9-tetranitro-10H-benzofuro[3,2-b]indole – dimethyl sulfoxide (1/1), C16H11N5O10S
- Crystal structure of 2,6-bis(2-(pyridin-3-yl)ethyl)pyrrolo[3,4-f]isoindole-1,3,5,7(2H,6H)-tetraone, C24H18N4O4
- The crystal structure of 3,4-dichlorobenzoic acid chloride, C7H3Cl3O
- Crystal structure of 1,1′-(1,4-phenylenebis(methylene))bis(pyridin-1-ium) bis(1,2-dicyanoethene-1,2-dithiolato-k2S:S)zinc(II), C26H18N6ZnS4
- Crystal structure of tetrakis(μ-naphthalene-1-carboxylato-κ2O,O′)bis(methanol)copper(II), C46H36Cu2O10
- Crystal structure of 9-methyl-3-methylene-1,2,3,9-tetrahydro-4H-carbazol-4-one, C14H13NO
- Crystal structure of bis(amino(carbamothioylamino)methaniminium) 3-nitrophthalate monohydrate, C12H19N9O7S2
- Crystal structure of 3,3′-(1,2-phenylene-bis(methylene))bis(1-ethyl-1H-imidazol-3-ium) bis(hexafluorophosphate), C18H24F12N4P2
- The crystal structure of 5-hydroxy-6,8-dimethoxy-2-methyl-4H-benzo[g]chromen-4-one– rubrofusarin B, C16H14O5
- The crystal structure of bis(ethanol-kO)- bis(6-aminopicolinato-k2N,O)manganese(II), C16H22O6N4Mn
- The crystal structure of 3,3′-((carbonylbis(azanediyl))bis(ethane-2,1-diyl)) bis(1-methyl-1H-benzo[d]imidazol-3-ium) tetrafluoroborate monohydrate, C21H28N6O3B2F8
- Crystal structure of dimethanol-dichlorido-bis( μ2-2-(((1,5-dimethyl-3-oxo-2- phenyl-2,3-dihydro-1H-pyrazol-4-yl)imino)methyl)phenolato- κ4O:O,O′,N)dinickel (II), C20H24ClNiN3O4
- The crystal structure of methyl 5-(trifluoromethyl)-1H-pyrrole-2-carboxylate, C7H6F3NO2
- Crystal structure of (OC‐6‐13)‐aqua‐tris (3‐bromopyridine‐κ1N)‐bis(trifluoroacetato‐κ1O)cadmium(II) C19H14Br3CdF6N3O5
- Crystal structure of methyl (E)-3-(4-(2-ethoxy-2-oxoethoxy)phenyl) acrylate, C14H16O5
- Crystal structure of methyl 4-acetoxy-3,5-dimethoxybenzoate, C12H14O6
- The crystal structure of 2-(1H-benzimidazol-2-yl)-3-bromo-5-chlorophenol, C13H8BrClN2O
- The crystal structure of bis(μ2-5-chloro-N-(2-methyl-1-oxidopropylidene)-2-oxidobenzohydrazonate-κ5N,O,O′:N′,O′′)pentakis(pyridine-κ1N)tricopper(II), C47H45Cl2N9Cu3O6
- Synthesis and crystal structure of catena-poly[aqua-bis(nitrato-κ2O:O′)- (μ2-((1 H-imidazol-1-yl)methyl)benzene-κ2 N,N′)-H2O-κ2O]cadmium(II), C14H16N6O7Cd
- The crystal structure of pentakis(carbonyl)-{μ-[2,3-bis(sulfanyl)propan-1-olato]}-(triphenylphosphane)diiron (Fe–Fe)C26H21Fe2O6PS2
- Crystal structure of ethyl-2-(3-benzoylthioureido)propanoate, C13H16N2O3S
- Crystal structure of 2-methoxy-4b,5,14,15-tetrahydro-6H-isoquinolino[2′,1′:1,6] pyrazino[2,3-b]quinoxaline, C19H18N4O
- Crystal structure of 2,2′-[ethane-1,2-diylbis(azanylylidenemethylylidene)]bis(6-chlorophenol), C16H14Cl2N2O2
- The crystal structure of (Z)-3-((2-(2-(2-aminophenoxy)ethoxy)phenyl)amino)-1-phenylbut-2-en-1-one, C24H24N2O3
- The crystal structure of 10-(3,5-di(pyridin-4-yl)phenyl)-10H-phenoxazine dihydrate, C28H23N3O3
- Crystal structure of poly[dipoly[aqua-di(µ2-pyrazin-2-olato-κ2N:N′) zinc(II)], C8H8N4O3Zn
- Crystal structure of poly[tetra(μ2-cyanido-κ2N:O)-bis(N,N-dimethylformamide-κO)-manganese(II)-platinum(II)], C10H14MnN6O2Pt
- The crystal structure of aqua-chlorido-6,6′-((ethane-1,2-diylbis(azaneylylidene))bis(methaneylylidene))bis(2,4-dichlorophenolato-κ4N,N′,O,O′)manganese(III), C16H12Cl5MnN2O3
- Crystal structure of [di(µ2-cyanido)-dicyanido-bis(dimethyl sulfoxide-κO)- bis(2,2′-(ethane-1,2-diylbis(azanylylidenemethanylylidene))diphenolato-κ4,N,N′,O,O′)- dimanganese(III)-platinum(II)], C40H40Mn2N8O6PtS2
- The crystal structure of (azido)-κ1N-6,6′-((cyclohexane-1,2-diylbis(azanylylidene)) bis(methanylylidene))bis(3-bromophenolato-κ4N,N,O,O)-(methanol)-manganese(III)–methanol(1/1), C22H26Br2MnN5O4
- Crystal structure of 7-chloro-N-(4-iodobenzyl)-1,2,3,4-tetrahydroacridin-9-amine, C20H18ClIN2
- Crystal structure of catena-poly[(1,4,8,11-tetraazacyclotetradecane-κ4N,N′,N″,N′′′)-bis(μ2-thiocyanato-κ2N:S)-bis(thiocyanato-κS)-nickel(II)palladium(II)], C14H24N8NiPdS4
- Crystal structure of 3-chloro-4-(4-ethylpiperazin-1-yl)aniline monohydrate, C12H20ClN3O
- Crystal structure of the 2D coordination polymer poly[diaqua-bis(μ2-3- methoxyisonicotinato-κ2N:O)cobalt(II)] — dimethylformamide (1/1), C20H30CoN4O10
- Crystal structure of 4-[(5-chloro-2-hydroxybenzylidene)amino]-3-propyl-1H-1,2,4-triazole-5(4H)-thione, C12H13ClN4OS
- Crystal structure of N-(5-(2-(benzyl(1-(4-methoxyphenyl)propan-2-yl)amino)-1-hydroxyethyl)-2-(benzyloxy)phenyl)formamide, C33H36N2O4
- Crystal structure of 3-(methoxycarbonyl)-7-oxabicyclo[2.2.1]heptane-2-carboxylic acid, C9H12O5
- The crystal structure of 1-((dimethylamino)(3-nitrophenyl)methyl)naphthalen-2-ol, C19H18N2O3
- Crystal structure of catena-poly[di(μ2-cyanido-κ2C:N)-dicyanido-tetrakis(dimethyl sulfoxide-κO)-manganese(II)-platinum(II)], C12H24MnN4O4PtS4
- Crystal structure of 4-amino-N-(2-pyrimidinyl)benzenesulfonamide–1,4-dioxane (1/1), C14H18N4O4S
- Crystal structure of bis{1-[(benzotriazol-1-yl)methyl]-1H-1,3-(2-methyl-imidazol)}di-chloridomercury(II), [Hg(C11H11N5)2Cl2], C22H22N10Cl2Hg
- Crystal structure of 2, 3-bis((4-methylbenzoyl)oxy) succinic acid–N, N-dimethylformamide (1/1), C23H25NO9
- Crystal structure of catena-poly[bis(4-(4-carboxyphenoxy)benzoato-κ1O)-μ2-(1,4-bis(1-imidazolyl)benzene-κ2N:N′)cobalt(II)], C40H28N4O10Co
- Crystal structure of 1H-imidazol-3-ium poly[aqua-(μ4-glutarato-κ6O,O′:O′:O′′,O′′′:O′′′)-(nitrato-κ2O,O′)strontium(II)], C8H13N3O8Sr
- Crystal structure of (R)-6-(benzo[b]thiophen-5-yl)-2-methyl-2,6-dihydrobenzo [5,6] silino[4,3,2-cd]indole, C23H17NSSi
- Crystal structure of catena-poly[bis(μ2-thiocyanato-κ2N:S)-(2-(5-methyl-1H-pyrazol-3-yl)pyridine-κ2N,N′)cadmium(II)]–dioxane (1/1), C15H17CdN5O2S2
- Crystal structure of poly[aqua-(μ2-1,4-bis(2′-carboxylatophenoxy)benzene-κ2O:O′)-(μ2-4,4′-bipyridione-κ2N:N′)cadmium(II)] monhydrate, C30H22CdN2O7⋅H2O
- Crystal structure of catena-poly[tetraaqua-(μ2-4,4′-bipyridine-k2N:N′)-bis(μ2-4′-methyl-[1,1′-biphenyl]-3,5-dicarboxylato-k4O,O′:O″,O′″)dicadmium(II)] dihydrate, C20H20NO7Cd
- Crystal structure of 1‐tert‐butyl‐3‐(2,6‐diisopropyl‐4‐phenoxyphenyl)‐2-methylisothiourea, C24H34N2OS
- Crystal structure of catena-poly[triaqua-(μ2-1,3-di(1H-imidazol-1-yl)propane-κ2N:N′)-(4,4′-(1H-1,2,4-triazole-3,5-diyl)dibenzoato-κ1O)cobalt(II)] — N,N′-dimethylformamide (1/1), C28H34N8O8Co
- Crystal structure of tetraaqua-bis(1,4-di(1H-imidazol-1-yl)benzene-κ1N)manganese(II) 2,3-dihydroxyterephthalate, C32H32MnN8O10


