Abstract
C35H33ClN4O6, monoclinic,
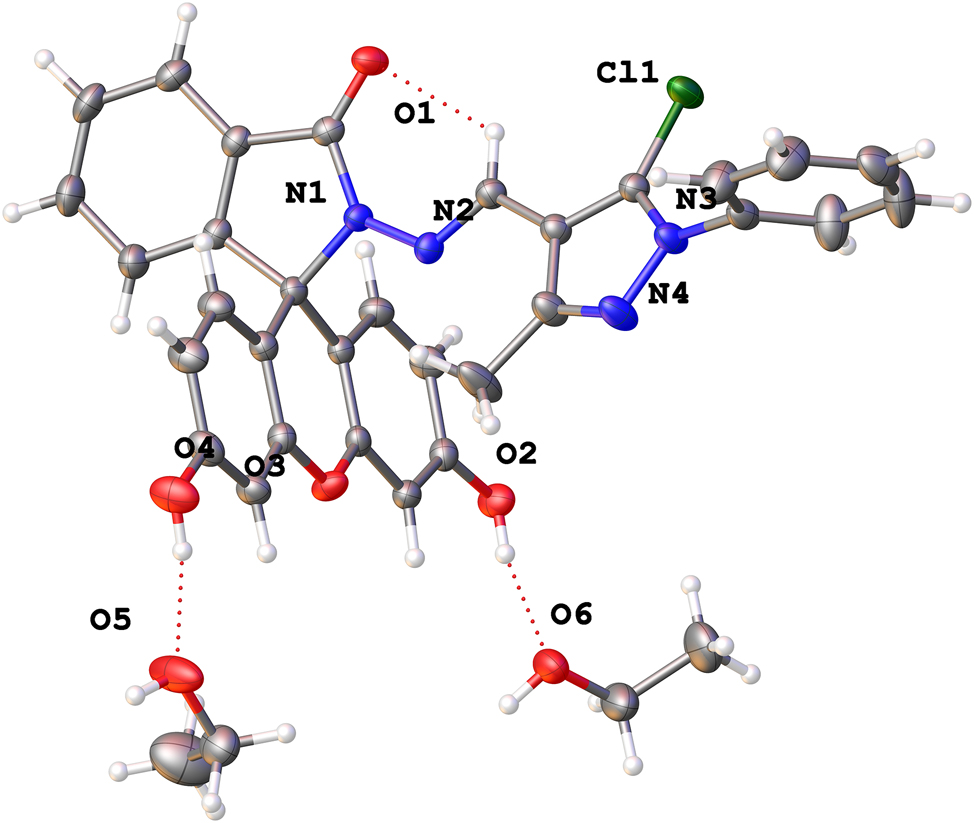
The molecular structure is shown in the figure. Table 1 contains the crystallographic data and the list of the atoms including atomic coordinates and displacement parameters can be found in the cif-file attached to this article.
Data collection and handling.
| Crystal: | Light colourless block |
| Size: | 0.28 × 0.15 × 0.10 mm |
| Wavelength: μ: |
Mo Kα radiation (0.71073 Å) 0.17 mm−1 |
| Diffractometer, scan mode: θmax, completeness: |
Bruker APEX-II, φ and ω scans 27.5°, 99 % |
| N(hkl)measured, N(hkl)unique, Rint: | 27117, 7343, 0.030 |
| Criterion for Iobs, N(hkl)gt: | Iobs > 2 σ(Iobs), 6,152 |
| N(param)refined: | 422 |
| Programs: | Bruker, 1 Olex2, 2 , 3 SHELX 4 , 5 |
1 Source of material
The title compound is a natural product, which has been obtained by extraction and isolation from fungal fermentation products. 6 All chemicals were of analytical grade and used as received without further purification. The preparation of the title compound is similar to the previously reported literature procedure. 6 Fluorescein hydrazide (200 mg, 577.46 μmol) and 5-chloro-3-methyl-1-phenyl-1H-pyrazole-4-carbaldehyde (127.42 mg, 577.46 μmol) were mixed in ethanol (15 mL) with addition of a catalytic amount of acetic acid. The reaction mixture was refluxed at 78 °C for 2.5 h and the reaction was shown to be completed by thin layer chromatography (TLC). After cooling to room temperature, the solution was filtered, and the colorless crystals were obtained by slow evaporating in ethanol at room temperature.
2 Experimental details
The structure was treated with the Olex2 crystallographic software package, 2 , 3 solved with the SHELXT structure solution program and refined with the SHELXL refinement package. 4 , 5 Carbon-bound hydrogen atoms were placed in calculated positions and refined with riding coordinates, with Uiso(H) fixed at 1.2 times of Ueq(C) (Tables 1 and 2).
3 Comment
Fluorescein serves as an ideal fluorescent reporter for chemosensors due to its superior photophysical properties. 7 Moreover, certain fluorescein hydrazones derived from substituted benzaldehydes have demonstrated significant biological activity, by functioning as non-intercalative topoisomerase catalytic inhibitors. 8 Notably, fluorescein hydrazone derivatives incorporating aldehyde moieties have been developed as effective fluorescent probes for the detection of Hg2+, Zn2+ or Cu2+ ions. 9 , 10 , 11 Herein, we report the crystal structure of the title compound derived from fluorescein hydrazide and a substituted pyrazole-4-aldehyde, namely 5-chloro-3-methyl-1-phenyl-1H-pyrazole-4-carbaldehyde.
The title compound C35H33ClN4O6 was crystallized in P21/c with one C31H21 ClN4O4 and two solvent molecules C2H6O in its asymmetric unit. The molecule structure comprises one xanthene ring and one 1-phenyl-1H-pyrazole moieties. The geometry of the title structure was characterized with the bond angles and lengths. In particular, the bond angles for C23⋯N4⋯N3, O1⋯C20⋯N1, and C22⋯C24⋯Cl1 are 106.14 (13)°, 125.74(14)° and 129.00(12)°, respectively. For another, the bond lengths for N2⋯C30, N2⋯N1, O1⋯C20, CI1⋯C24, N4⋯C23 and C22⋯C24, are 1.283(2) Å, 1.3661(17) Å, 1.2306(19) Å, 1.6973(16) Å, 1.324(2) Å and 1.377(2) Å. All bond angles and lengths of the title structure are within the expected range and comparable with those of reported fluorescein hydrazones 12 , 13 , 14 and a directly related spiro compound. 15 In the crystal, hydrazine molecules are arranged in layers by ethanol molecules via hydrogen bonds O–H⋯O and O–H⋯N, with max D–A distance 2.9 Å and minimum angle 120°.
Acknowledgments
This work was supported by Jiangsu Provincial Drug Administration’s Scientific Research Program for Drugs (No. 202367), the Scientific Research Personnel Training Program of Kangda College of Nanjing Medical University (No. KD2022KYRC007), Science & Technology Funds of Kangda College of Nanjing Medical University (No. KD2023KYJJ017, KD2023KYJJ018 & KD2023KYJJ019), and the Science & Technology Funds of Lianyungang (No. JCYJ2320), Jiangsu Student’s foundation for innovation and entrepreneurship training program (No. 202413980013Y & 202413980053Y).
-
Author contributions: All the authors have accepted responsibility for the entire content of this submitted manuscript and approved submission.
-
Conflict of interest: The authors declare no conflicts of interest regarding this article.
References
1. BRUKER. Saint, Apex2 and Sadabs; Bruker AXS Inc.: Madison, Wisconsin, USA, 2009.Search in Google Scholar
2. Dolomanov, O. V.; Bourhis, L. J.; Gildea, R. J.; Howard, J. A. K.; Puschmann, H. OLEX2: a Complete Structure Solution, Refinement and Analysis Program. J. Appl. Crystallogr. 2009, 42, 339–341; https://doi.org/10.1107/s0021889808042726.Search in Google Scholar
3. Bourhis, L. J.; Dolomanov, O. V.; Gildea, R. J.; Howard, J. A. K.; Puschmann, H. The Anatomy of a Comprehensive Constrained, Restrained Refinement Program for the Modern Computing Environment-Olex2 Dissected. Acta Crystallogr. 2015, 71, 59–75; https://doi.org/10.1107/s2053273314022207.Search in Google Scholar PubMed PubMed Central
4. Sheldrick, G. M. A Short History of SHELX. Acta Crystallogr. 2008, 64, 112–122; https://doi.org/10.1107/s0108767307043930.Search in Google Scholar PubMed
5. Sheldrick, G. M. Crystal Structure Refinement with SHELXL. Acta Crystallogr. 2015, 71, 3–8; https://doi.org/10.1107/s2053229614024218.Search in Google Scholar
6. Sandhu, N.; Singh, A. P.; Saxena, A.; Pandey, S. K.; Yadav, R. K. X-ray Crystallographic, Electrochemical, Quantum Chemical and Anti-microbial Analysis of Fluorescein Based Schiff Base. J. Mol. Struct. 2020, 1221, 128762; https://doi.org/10.1016/j.molstruc.2020.128762.Search in Google Scholar
7. Chen, X. Q.; Pradhan, T.; Wang, F.; Kim, J. S.; Yoon, J. Fluorescent Chemosensors Based on spiroring-opening of Xanthenes and Related Derivatives. Chem. Rev. 2012, 112, 1910–1956; https://doi.org/10.1021/cr200201z.Search in Google Scholar PubMed
8. Rahman, A. F. M. M.; Park, S.-E.; Kadi, A. A.; Kwon, Y. Fluorescein Hydrazones as Novel Nonintercalative Topoisomerase Catalytic Inhibitors with Low DNA Toxicity. J. Med. Chem. 2014, 57, 9139–9151; https://doi.org/10.1021/jm501263m.Search in Google Scholar PubMed
9. Roy, S.; Mondal, T.; Dey, D.; Mane, M. V.; Panja, S. S. A New thiophene- Appended fluorescein-hydrazone-based chromo-fluorogenic Sensor for the Screening of Hg2+ Ions in Real Water Samples. Chemistryselect 2021, 6, 10464–10479; https://doi.org/10.1002/slct.202102692.Search in Google Scholar
10. An, J.; Yan, M.; Yang, Z.; Li, T.; Zhou, Q.; An, J.; Yan, M.; Yang, Z.; Li, T.; Zhou, Q. A Turn-on Fluorescent Sensor for Zn(II) Based on fluorescein-coumarin Conjugate. Dyes Pigments 2013, 99, 1–5; https://doi.org/10.1016/j.dyepig.2013.04.018.Search in Google Scholar
11. Chen, X.; Li, Z.; Xiang, Y.; Tong, A. Salicylaldehyde Fluorescein Hydrazone: a Colorimetric Logic Chemosensor for pH and Cu(II). Tetrahedron Lett. 2008, 49, 4697–4700; https://doi.org/10.1016/j.tetlet.2008.05.137.Search in Google Scholar
12. Liu, B.; Xu, X.; Zhu, Z. Crystal Structure of (Z)-2-hydroxy-N′-(1-(o-tolyl)ethylidene)benzohydrazide, C16H16N2O2. Z. Kristallogr. – N. Cryst. Struct. 2025, 240, 423–424; https://doi.org/10.1515/ncrs-2025-0036.Search in Google Scholar
13. Zhang, L.; Wu, W.-N.; Wang, Y. Crystal Structure of (E)-2-(4-ethoxycarbonyl-3,5-dimethyl-2-(pyrrole-2-ylmethyleneamino)-3′,6′-dihydroxylspiro[isoindoline-1,9′-xanthen]-3-one-methanol (1/1), C31H29N3O7. Z. Kristallogr. – N. Cryst. Struct. 2021, 236, 239–241; https://doi.org/10.1515/ncrs-2020-0479.Search in Google Scholar
14. Polyakova, I. N.; Starikova, Z. A.; Parusnikov, B. V.; Krasavin, I. A.; Zhadanov, B. V. The Lactone Form of Fluorescein: Crystal Structure of the 1:1 Molecular Complex of Fluorescein with Methanol. J. Struct. Chem. 1984, 25, 752–757; https://doi.org/10.1007/bf00747920.Search in Google Scholar
15. Song, Y.-F.; Wu, W.-N.; Wang, Y. Crystal Structure of (E)-2-(((1H-pyrrol-2-yl)methylene)amino)-3′,6′-dihydroxyspiro[isoindoline-1,9′-xanthen]-3-one-methanol (1/2), C27H25N3O6. Z. Kristallogr. – N. Cryst. Struct. 2021, 236, 257–258; https://doi.org/10.1515/ncrs-2020-0480.Search in Google Scholar
© 2025 the author(s), published by De Gruyter, Berlin/Boston
This work is licensed under the Creative Commons Attribution 4.0 International License.
Articles in the same Issue
- Frontmatter
- New Crystal Structures
- Crystal structure of (S)-N-(10-((2,2-dimethoxyethyl)amino)-1,2,3-trimethoxy-9-oxo-5,6,7,9-tetrahydrobenzo[a]heptalen-7-yl)acetamide, C25H32N2O7
- The crystal structure of 6,6′-difluoro-3,3′-dimethyl-5,5′-di(10H-phenoxazin-10-yl)- [1,1′-biphenyl]-2,2′-dicarbonitrile, C40H24F2N4O2
- Crystal structure of poly[(di-ethylenediamine-κ2N,N′)cadmium(II) tetradedocyloxidohexavanadate] (V4+/V5+ = 2/1), C4H16CdN4O14V6
- The crystal structure of poly[bis(dimethylformamide-κ1N)-(μ4-2′,3,3″,5′-tetrakis(trifluoromethyl)-[1,1′:4′,1″-terphenyl]-4,4″-dicarboxylato-κ4 O,O′: O″,O‴)dicadmium(II)], C27H15CdF12NO5
- Crystal structure of bis(μ2-ferrocenylcarboxylato-O,O′)-(μ3-oxido-κ3O:O:O)-bis(μ2-salicyladoximato-κ2N,O,O′)-(μ2-isopropoxo)-tris(isopropoxy-κ1O trititanium(IV)), C48H55N2O13Fe2Ti3
- Crystal structure of 3-(diethylamino)-7,9,11-trimethyl-8-phenyl-6H,13H-12λ4,13λ4-chromeno[3′,4′:4,5]pyrrolo[1,2-c]pyrrolo[2,1-f][1,3,2]diazaborinin-6-one, C28H26BF2N3O2
- The crystal structure of catena-poly[aqua-μ2-2-nitro-benzene-1,3-dicarboxylato-κ2O,O′)-(1,10-phenanthroline-κ2N,N′)-zinc(II)], C20H13N3O7Zn
- Crystal structure of poly[diaqua-{μ3-1-(3-carboxylatophenyl)-4-oxo-1,4-dihydropyridazine-3-carboxylato-κ4O,O′:O′′:O′′′′}manganese(II)] hydrate
- Crystal structure of N′-((1-hydroxycyclohexyl)(phenyl)methyl)-2-methoxybenzohydrazide methanol solvate, C22H28N2O4
- The cocrystal of caffeic acid — progesterone — water (1/2/1), C51H70O9
- Crystal structure of (((oxido(quinolin-6-yl)methoxy)triphenyl-λ5-stibanyl)oxy)(quinolin-7-yl)methanolate
- Crystal structure of [(E)-6′-(diethylamino)-2-(2-(((E)-pyren-1-ylmethylene)amino)ethyl)-4′-(2-((E)-1,3,3-trimethylindolin-2-ylidene)ethylidene)-1′,2′,3′,4′-tetrahydrospiro[isoindoline-1,9′-xanthen]-3-one]-methanol, solvate C57H56N4O3
- The crystal structure of 1-(acridin-9-yl)pyrrolidine-2,5-dione, C17H22N2O2
- Crystal structure of N-(4-acetylphenyl)-2-(6-methoxynaphthalen-2-yl)propanamide, C22H21NO3
- The crystal structure of 5,10,15,20-tetrakis(4-(1H-1,2,4-triazol-1-yl)phenyl)porphyrin, C52H34N16
- Crystal structure of hexacarbonyl-μ2-[phenylmethanedithiolato-κ4S:S,S′:S′]diiron (Fe–Fe) C13H6Fe2O6S2
- Crystal structure of diiodo-bis(1-((2-propyl-1H-benzo[d]imidazol-1-yl)methyl)-1H-benzo[d][1,2,3]triazole-κ1N)cadmium(II), C34H34CdI2N10
- Crystal structure of (E)-(3-(3-bromophenyl)acryloyl)ferrocene, C19H15BrFeO
- Crystal structure of catena-poly(μ2-6-chloropyridine-2-carboxylato-κ3N,O:O′)(6-chloropyridine-2-carboxylato-κ2O,N)copper(II), C12H6Cl2N2O4Cu
- Crystal structure of poly[diaqua-μ 3-(5-(3,5-dicarboxy-2,4,6-trimethylbenzyl)-2,4,6-trimethylisophthalato)-κ 6O,O′:O″,O‴:O‴′,O‴″) terbium(III)-monohydrate], C23H28TbO12
- Crystal structure of (E)-2-(((5-chloro-3-methyl-1-phenyl-1H-pyrazol-4-yl)methylene)amino)-3′,6′-dihydroxyspiro[isoindoline-1,9′-xanthen]-3-one – ethanol (1/2), C35H33ClN4O6
- The crystal structure of 3-(5-amino-3-phenylisoxazol-4-yl)-4-chloro-3-hydroxyindolin-2-one, C17H12ClN3O3
- The crystal structure of dimethylammonium 4-[2-(4-fluorophenyl)-4, 5-diphenyl-1H-imidazol-1-yl]benzenesulfonate, C29H26FN3O3S
- Crystal structure of (R)-2-ammonio-3-((5-carboxypentyl)thio)propanoate
- Crystal structure of 4-cyclohexyl-5-(thiophen-2-yl)-2,4-dihydro-3H-1,2,4-triazole-3-thione, C12H15N3S2
- The crystal structure of 4,6-bis(dimethylamino)-2-fluoroisophthalonitrile, C12H13FN4
- Hydrogen bonding in the crystal structure of nicotin-1,1′-dium tetrabromidomanganate(II)
- The crystal structure of bis(2-bromobenzyl)(2-((2-oxybenzylidene)amino)-4-methylpentanoato-κ3N, O,O′)tin(IV), C27H27Br2NO3Sn
- Crystal structure of (E)-(3-(p-tolyl)acryloyl)ferrocene, C20H18FeO
- Crystal structure of (E)-7-fluoro-2-((5-(4-methylpiperazin-1-yl)pyridin-2-yl)methylene)-3,4-dihydronaphthalen-1(2H)-one, C21H22FN3O
- Crystal structure of (E)-7-methoxy-2-((5-(4-methylpiperazin-1-yl)pyridin-2-yl)methylene)-3,4-dihydronaphthalen-1(2H)-one, C22H25N3O2
- The crystal structure of poly(bis(μ2-1,3,5-tri(1H-imidazol-1-yl)benzene-κ2N:N′)-(μ2-2,3,5,6-tetrafluoroterephthalato-κ2O:O′)-manganese(II), C38H24F4N12O4Mn
- Crystal structure of (3,4-dimethoxybenzyl)triphenylphosphonium bromide ethanol solvate, C29H32BrO3P
- Crystal structure of tetraethylammonium hydrogencarbonate – (diaminomethylene)thiourea – water (2/1/3)
- Crystal structure of N, N-Dimethyl-N′-tosylformimidamide, C10H14N2O2S
- The crystal structure of ethyl 2-methyl-5-oxo-4-(2-methoxyphenyl)-1,4,5,6,7,8-hexahydroquinoline-3-carboxylate, C20H23N2O4
- Crystal structure of bis(μ2-1,5-bis[(E)-1-(2-hydroxyphenyl)ethylidene] thiocarbonohydrazide)-bis(dimethylformamide)-dizinc(II) dimethylformamide solvate, C40H46N10O6S2Zn2⋅C3H7NO
- Crystal structure of azido-κ1N{hydridotris(3-tert-butyl-5-methylpyrazol-1-yl)borato-κ3N,N′,N″}copper(II), C24H40BCuN9
- The crystal structure of fac-tricarbonyl(1,10-phenanthroline-κ2N,N′)-(azido-κ1N)rhenium(I), C15H8N5O3Re
- Crystal structure of 4-((triphenylphosphonio)methyl)pyridin-1-ium tetrachloridozincate(II), C24H22Cl4NPZn
Articles in the same Issue
- Frontmatter
- New Crystal Structures
- Crystal structure of (S)-N-(10-((2,2-dimethoxyethyl)amino)-1,2,3-trimethoxy-9-oxo-5,6,7,9-tetrahydrobenzo[a]heptalen-7-yl)acetamide, C25H32N2O7
- The crystal structure of 6,6′-difluoro-3,3′-dimethyl-5,5′-di(10H-phenoxazin-10-yl)- [1,1′-biphenyl]-2,2′-dicarbonitrile, C40H24F2N4O2
- Crystal structure of poly[(di-ethylenediamine-κ2N,N′)cadmium(II) tetradedocyloxidohexavanadate] (V4+/V5+ = 2/1), C4H16CdN4O14V6
- The crystal structure of poly[bis(dimethylformamide-κ1N)-(μ4-2′,3,3″,5′-tetrakis(trifluoromethyl)-[1,1′:4′,1″-terphenyl]-4,4″-dicarboxylato-κ4 O,O′: O″,O‴)dicadmium(II)], C27H15CdF12NO5
- Crystal structure of bis(μ2-ferrocenylcarboxylato-O,O′)-(μ3-oxido-κ3O:O:O)-bis(μ2-salicyladoximato-κ2N,O,O′)-(μ2-isopropoxo)-tris(isopropoxy-κ1O trititanium(IV)), C48H55N2O13Fe2Ti3
- Crystal structure of 3-(diethylamino)-7,9,11-trimethyl-8-phenyl-6H,13H-12λ4,13λ4-chromeno[3′,4′:4,5]pyrrolo[1,2-c]pyrrolo[2,1-f][1,3,2]diazaborinin-6-one, C28H26BF2N3O2
- The crystal structure of catena-poly[aqua-μ2-2-nitro-benzene-1,3-dicarboxylato-κ2O,O′)-(1,10-phenanthroline-κ2N,N′)-zinc(II)], C20H13N3O7Zn
- Crystal structure of poly[diaqua-{μ3-1-(3-carboxylatophenyl)-4-oxo-1,4-dihydropyridazine-3-carboxylato-κ4O,O′:O′′:O′′′′}manganese(II)] hydrate
- Crystal structure of N′-((1-hydroxycyclohexyl)(phenyl)methyl)-2-methoxybenzohydrazide methanol solvate, C22H28N2O4
- The cocrystal of caffeic acid — progesterone — water (1/2/1), C51H70O9
- Crystal structure of (((oxido(quinolin-6-yl)methoxy)triphenyl-λ5-stibanyl)oxy)(quinolin-7-yl)methanolate
- Crystal structure of [(E)-6′-(diethylamino)-2-(2-(((E)-pyren-1-ylmethylene)amino)ethyl)-4′-(2-((E)-1,3,3-trimethylindolin-2-ylidene)ethylidene)-1′,2′,3′,4′-tetrahydrospiro[isoindoline-1,9′-xanthen]-3-one]-methanol, solvate C57H56N4O3
- The crystal structure of 1-(acridin-9-yl)pyrrolidine-2,5-dione, C17H22N2O2
- Crystal structure of N-(4-acetylphenyl)-2-(6-methoxynaphthalen-2-yl)propanamide, C22H21NO3
- The crystal structure of 5,10,15,20-tetrakis(4-(1H-1,2,4-triazol-1-yl)phenyl)porphyrin, C52H34N16
- Crystal structure of hexacarbonyl-μ2-[phenylmethanedithiolato-κ4S:S,S′:S′]diiron (Fe–Fe) C13H6Fe2O6S2
- Crystal structure of diiodo-bis(1-((2-propyl-1H-benzo[d]imidazol-1-yl)methyl)-1H-benzo[d][1,2,3]triazole-κ1N)cadmium(II), C34H34CdI2N10
- Crystal structure of (E)-(3-(3-bromophenyl)acryloyl)ferrocene, C19H15BrFeO
- Crystal structure of catena-poly(μ2-6-chloropyridine-2-carboxylato-κ3N,O:O′)(6-chloropyridine-2-carboxylato-κ2O,N)copper(II), C12H6Cl2N2O4Cu
- Crystal structure of poly[diaqua-μ 3-(5-(3,5-dicarboxy-2,4,6-trimethylbenzyl)-2,4,6-trimethylisophthalato)-κ 6O,O′:O″,O‴:O‴′,O‴″) terbium(III)-monohydrate], C23H28TbO12
- Crystal structure of (E)-2-(((5-chloro-3-methyl-1-phenyl-1H-pyrazol-4-yl)methylene)amino)-3′,6′-dihydroxyspiro[isoindoline-1,9′-xanthen]-3-one – ethanol (1/2), C35H33ClN4O6
- The crystal structure of 3-(5-amino-3-phenylisoxazol-4-yl)-4-chloro-3-hydroxyindolin-2-one, C17H12ClN3O3
- The crystal structure of dimethylammonium 4-[2-(4-fluorophenyl)-4, 5-diphenyl-1H-imidazol-1-yl]benzenesulfonate, C29H26FN3O3S
- Crystal structure of (R)-2-ammonio-3-((5-carboxypentyl)thio)propanoate
- Crystal structure of 4-cyclohexyl-5-(thiophen-2-yl)-2,4-dihydro-3H-1,2,4-triazole-3-thione, C12H15N3S2
- The crystal structure of 4,6-bis(dimethylamino)-2-fluoroisophthalonitrile, C12H13FN4
- Hydrogen bonding in the crystal structure of nicotin-1,1′-dium tetrabromidomanganate(II)
- The crystal structure of bis(2-bromobenzyl)(2-((2-oxybenzylidene)amino)-4-methylpentanoato-κ3N, O,O′)tin(IV), C27H27Br2NO3Sn
- Crystal structure of (E)-(3-(p-tolyl)acryloyl)ferrocene, C20H18FeO
- Crystal structure of (E)-7-fluoro-2-((5-(4-methylpiperazin-1-yl)pyridin-2-yl)methylene)-3,4-dihydronaphthalen-1(2H)-one, C21H22FN3O
- Crystal structure of (E)-7-methoxy-2-((5-(4-methylpiperazin-1-yl)pyridin-2-yl)methylene)-3,4-dihydronaphthalen-1(2H)-one, C22H25N3O2
- The crystal structure of poly(bis(μ2-1,3,5-tri(1H-imidazol-1-yl)benzene-κ2N:N′)-(μ2-2,3,5,6-tetrafluoroterephthalato-κ2O:O′)-manganese(II), C38H24F4N12O4Mn
- Crystal structure of (3,4-dimethoxybenzyl)triphenylphosphonium bromide ethanol solvate, C29H32BrO3P
- Crystal structure of tetraethylammonium hydrogencarbonate – (diaminomethylene)thiourea – water (2/1/3)
- Crystal structure of N, N-Dimethyl-N′-tosylformimidamide, C10H14N2O2S
- The crystal structure of ethyl 2-methyl-5-oxo-4-(2-methoxyphenyl)-1,4,5,6,7,8-hexahydroquinoline-3-carboxylate, C20H23N2O4
- Crystal structure of bis(μ2-1,5-bis[(E)-1-(2-hydroxyphenyl)ethylidene] thiocarbonohydrazide)-bis(dimethylformamide)-dizinc(II) dimethylformamide solvate, C40H46N10O6S2Zn2⋅C3H7NO
- Crystal structure of azido-κ1N{hydridotris(3-tert-butyl-5-methylpyrazol-1-yl)borato-κ3N,N′,N″}copper(II), C24H40BCuN9
- The crystal structure of fac-tricarbonyl(1,10-phenanthroline-κ2N,N′)-(azido-κ1N)rhenium(I), C15H8N5O3Re
- Crystal structure of 4-((triphenylphosphonio)methyl)pyridin-1-ium tetrachloridozincate(II), C24H22Cl4NPZn

