Abstract
C26H20.50N1.50O3PRh, monoclinic, C2/c (no. 15), a = 30.5215(9) Å, b = 8.7529(2) Å, c = 22.4219(11) Å, β = 132.137(1)∘, V = 4441.9(3) Å3, Z = 8, Rgt(F) = 0.0305, wRref(F2) = 0.0615, T = 100 K.
Table 1 contains crystallographic data and Table 2 contains the list of the atoms including atomic coordinates and displacement parameters.
Data collection and handling.
| Crystal: | Yellow prism |
| Size: | 0.27 × 0.16 × 0.07 mm |
| Wavelength: μ: |
Mo Kα radiation (0.71073 Å) 0.87 mm−1 |
| Diffractometer, scan mode: θmax, completeness: |
Bruker D8 Venture 4K Kappa Photon III, 28.6°, >99 % |
| N(hkl)measured, N(hkl)unique, Rint: | 26405, 5483, 0.038 |
| Criterion for Iobs, N(hkl)gt: | Iobs > 2 σ(Iobs), 4951 |
| N(param)refined: | 296 |
| Programs: | SHELX [1], Bruker [2], Olex2 [3], Diamond [4] |
Fractional atomic coordinates and isotropic or equivalent isotropic displacement parameters (Å2).
| Atom | x | y | z | Uiso*/Ueq |
|---|---|---|---|---|
| C1 | 0.81872 (10) | 0.4047 (3) | 0.62648 (13) | 0.0153 (4) |
| H1 | 0.837763 | 0.347957 | 0.674450 | 0.018* |
| C2 | 0.85249 (10) | 0.5006 (3) | 0.62218 (14) | 0.0174 (5) |
| H2 | 0.893922 | 0.508779 | 0.666021 | 0.021* |
| C3 | 0.82481 (11) | 0.5841 (3) | 0.55304 (14) | 0.0184 (5) |
| H3 | 0.846476 | 0.654318 | 0.549031 | 0.022* |
| C4 | 0.76381 (10) | 0.5635 (3) | 0.48841 (14) | 0.0169 (5) |
| H4 | 0.744073 | 0.617541 | 0.439508 | 0.020* |
| C5 | 0.73279 (10) | 0.4648 (2) | 0.49623 (13) | 0.0126 (4) |
| C6 | 0.66681 (10) | 0.4328 (3) | 0.42786 (13) | 0.0151 (4) |
| C7 | 0.75118 (10) | 0.2124 (3) | 0.66684 (13) | 0.0150 (4) |
| C8 | 0.55065 (9) | 0.2210 (2) | 0.45739 (12) | 0.0112 (4) |
| C9 | 0.53199 (10) | 0.2017 (2) | 0.38178 (13) | 0.0130 (4) |
| H9 | 0.556804 | 0.151090 | 0.376540 | 0.016* |
| C10 | 0.47660 (10) | 0.2567 (3) | 0.31319 (13) | 0.0162 (4) |
| H10 | 0.464233 | 0.242286 | 0.261801 | 0.019* |
| C11 | 0.43953 (10) | 0.3318 (3) | 0.31884 (14) | 0.0172 (5) |
| H11 | 0.402264 | 0.369502 | 0.272007 | 0.021* |
| C12 | 0.45759 (10) | 0.3508 (3) | 0.39321 (14) | 0.0183 (5) |
| H12 | 0.432560 | 0.401487 | 0.398004 | 0.022* |
| C13 | 0.51286 (10) | 0.2958 (2) | 0.46237 (13) | 0.0151 (4) |
| H13 | 0.524720 | 0.309757 | 0.513470 | 0.018* |
| C14 | 0.62103 (10) | −0.0532 (2) | 0.53614 (13) | 0.0115 (4) |
| C15 | 0.67291 (11) | −0.1380 (3) | 0.59380 (14) | 0.0165 (5) |
| H15 | 0.708259 | −0.087089 | 0.637389 | 0.020* |
| C16 | 0.67252 (12) | −0.2956 (3) | 0.58706 (15) | 0.0210 (5) |
| H16 | 0.707592 | −0.352592 | 0.625892 | 0.025* |
| C17 | 0.62051 (12) | −0.3703 (3) | 0.52314 (15) | 0.0219 (5) |
| H17 | 0.620605 | −0.477991 | 0.518071 | 0.026* |
| C18 | 0.56823 (11) | −0.2880 (3) | 0.46644 (15) | 0.0203 (5) |
| H18 | 0.532773 | −0.339758 | 0.423804 | 0.024* |
| C19 | 0.56853 (10) | −0.1297 (3) | 0.47295 (14) | 0.0172 (5) |
| H19 | 0.533186 | −0.073306 | 0.434598 | 0.021* |
| C20 | 0.62651 (9) | 0.1837 (2) | 0.62738 (12) | 0.0115 (4) |
| C21 | 0.65422 (10) | 0.3133 (2) | 0.67615 (13) | 0.0131 (4) |
| H21 | 0.674813 | 0.381447 | 0.669352 | 0.016* |
| C22 | 0.65155 (10) | 0.3418 (3) | 0.73390 (13) | 0.0145 (4) |
| H22 | 0.670851 | 0.428687 | 0.767809 | 0.017* |
| C23 | 0.62063 (10) | 0.2433 (3) | 0.74239 (13) | 0.0164 (4) |
| H23 | 0.618001 | 0.264431 | 0.781466 | 0.020* |
| C24 | 0.59342 (10) | 0.1141 (3) | 0.69464 (13) | 0.0173 (5) |
| H24 | 0.572650 | 0.046900 | 0.701491 | 0.021* |
| C25 | 0.59645 (10) | 0.0833 (3) | 0.63745 (13) | 0.0143 (4) |
| H25 | 0.578217 | −0.005711 | 0.605016 | 0.017* |
| C26 | 0.500000 | 0.5957 (4) | 0.250000 | 0.0335 (9) |
| H26Aa | 0.524930 | 0.558337 | 0.305399 | 0.050* |
| H26Ba | 0.515531 | 0.558337 | 0.226075 | 0.050* |
| H26Ca | 0.459539 | 0.558337 | 0.218526 | 0.050* |
| C27 | 0.500000 | 0.7608 (4) | 0.250000 | 0.0202 (7) |
| N1 | 0.75958 (8) | 0.3880 (2) | 0.56519 (11) | 0.0121 (4) |
| N2 | 0.500000 | 0.8911 (4) | 0.250000 | 0.0268 (7) |
| O1 | 0.64029 (7) | 0.490 (2) | 0.36194 (10) | 0.0231 (4) |
| O2 | 0.64361 (7) | 0.34217 (18) | 0.44512 (9) | 0.0151 (3) |
| O3 | 0.78578 (8) | 0.1820 (2) | 0.73428 (10) | 0.0239 (4) |
| P1 | 0.62483 (2) | 0.15424 (6) | 0.54650 (3) | 0.00971 (11) |
| Rh1 | 0.69905 (2) | 0.26665 (2) | 0.56228 (2) | 0.01013 (5) |
-
aOccupancy: 0.5
1 Source of materials
The complex was synthesized starting with the reduction of hydrated RhCl3 in DMF which was refluxed for approximately 20 min to give a yellow solution of di-μ-chloro-tetracarbonyldirhodium(I), [RhCl(CO)2]2 [5], [6], [7].
The addition of an equivalent amount of pyridine-2-carboxylic acid (picH) to the aforementioned yellow solution followed by addition of ice water to precipitate the dicarbonylrhodium(I) complex, [Rh I (pic)(CO)2].
[Rh(pic)(CO)(PPh3)] was synthesized by dissolving [Rh(pic)(CO)2] (0.023 g, 0.078 mmol) in 5 cm3 of acetone. Triphenylphosphine (0.0239 g, 0.078 mmol) was added to the aforementioned solution with stirring. Some ice water was added dropwise to precipitate the product. Yellow prism crystals were obtained from recrystallization in acetonitrile.
2 Experimental details
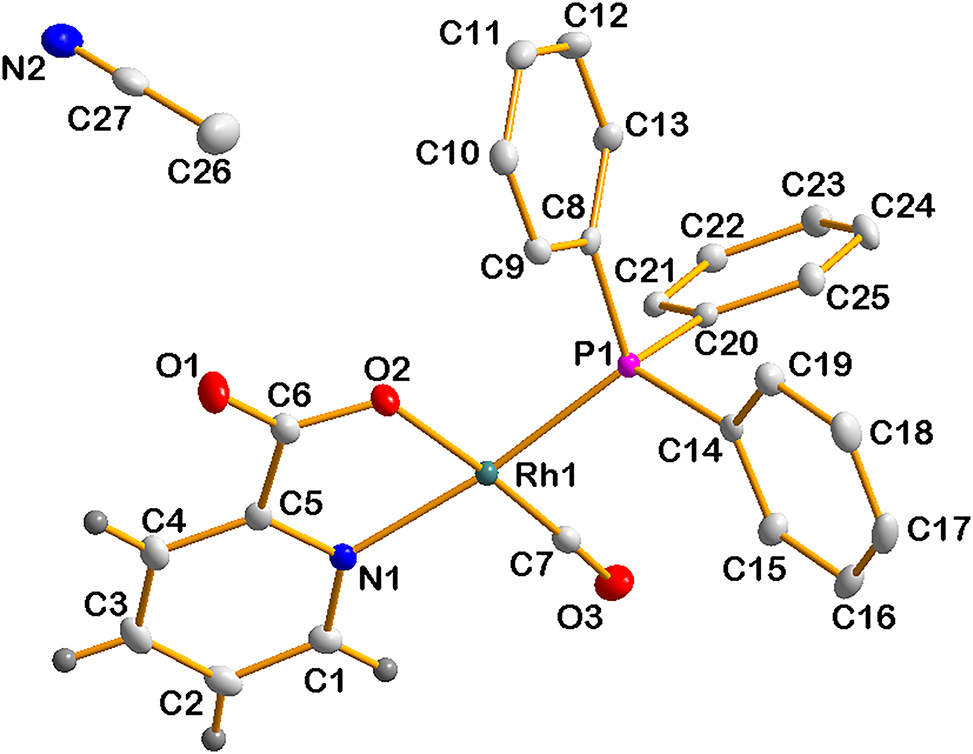
All H-atoms were positioned on geometrically idealized positions and refined using the riding model with fixed C–H distances for aromatic C–H of 0.93 Å(C–H) [Uiso (H) = 1.2 Ueq], for methyl C–H of 0.96 Å (C–H) [Uiso(H) = 1.5 Ueq]. The graphics were obtained using the DIAMOND [4] program with 50 % probability ellipsoids for all non-hydrogen atoms. The highest peak is located 0.89 Å from P1 and the deepest hole is situated 0.71 Å from C 23. Some hydrogen atoms in the figure are omitted for clarity.
3 Comment
The synthesis of Rh(I) and Rh(III) complexes have been discussed intensively, and a massive number of studies involve the synthesis of the precursor complex [Rh(L,L′–BID)(CO)2], of which one of the carbonyl ligands may be substituted by tertiary phosphine ligands to form [Rh(L,L′–BID)(CO) (PX3)], L,LN′–BID denotes monocharged bidentate ligands with different donor atoms such as O,O′, O,N, O,S, and N,S, with PX3 demonstrating different neutral monodentate phosphine ligands. In this study the complex [Rh(pic)(CO) (PPh3)] (pic- = picolinic acid) has been synthesized as part of a study to gain a better understanding of the electronic and steric influence of the phosphine ligands on the oxidative addition of iodomethane [8], [9], [10], [11], [12], [13]. The coordination mode in the title complex is similar to the complexes reported previously [14]. Leipoldt et al. [15] had determined a structure similar to the title complex.
A distortion from the ideal square planar geometry in the title structure, as well as in the complex published by Leipoldt et al. is evidenced by the angles around the coordination polyhedron, which differ from 90°. The P1–Rh1–O2, P1–Rh1–C7, and O2–Rh1–N1 bond angles of 93.4(1)∘, 90.4(2)∘ and 80.2(2)∘ for the title structure and the P1–Rh1–O1, P1–Rh1–C2 and O1–Rh1–N1 bond angles of 90.9(1)∘, 93.5(3)∘ and 78.9(2)∘ for the complex by Leipoldt et al. The Rh1–P1, Rh1–O2 and Rh1–N1 bond distances of 2.269(1) Å, 2.028(1) Å and 2.093(0) Å respectively for the title structure, Rh1–P1, Rh1–O1 and Rh1–N1 of 2.262(2) Å, 2.066(7) Å and 2.088(6) Å respectively for the complex by Leipoldt et al. The structure crystallizes with an acetonitrile solvate molecule in the crystal lattice while the structure by Leipoldt et al. crystallizes with methanol solvate. The title structure has been collected at 100 K while the structure by Leipoldt et al. was collected at 298 K. Leipoldt showed that the complex crystallized in the triclinic system with Z = 2, while for the title structure the space group has been determined as the monoclinic C2/c with Z = 8.
The lengthening of Rh1–N1 compared to Rh1–O2 in the title structure and Rh1–N1 and Rh1–O1 for the complex by Leipoldt is due to the strong trans influence of the P atom.
-
Author contributions: All the authors have accepted responsibility for the entire content of this submitted manuscript and approved submission.
-
Competing interests: The authors declare no conflicts of interest regarding this article.
-
Research funding: The authors acknowledge funding under the Swiss–South Africa joint research program (SSAJRP) from the SANRF (A. Roodt: UID: 107802) as well as from the Competitive Program for Rated Researchers of the SANRF (A. Roodt: UID: 111698), from the South African Department of Science Innovation (DSI) and the Department of Science and Technology (DST) respectively, “Department of Science and Innovation, Republic of South Africa” and “Department of Science and Technology, Republic of South Africa”.
References
1. Sheldrick, G. M. A short history of SHELX. Acta Crystallogr. 2008, A64, 112–122; https://doi.org/10.1107/s0108767307043930.Suche in Google Scholar PubMed
2. Bruker. SAINT-plus (Version 7.12) and SADABS (Version 2004/1); Bruker AXS Inc.: Madison, WI, USA, 2004.Suche in Google Scholar
3. Dolomanov, O. V., Bourhis, L. J., Gildea, R. J., Howard, J. A. K., Puschmann, H. OLEX2: a complete structure solution, refinement and analysis program. J. Appl. Cryst. 2009, 42, 339–341; https://doi.org/10.1107/s0021889808042726.Suche in Google Scholar
4. Brandenburg, K. DIAMOND. Visual crystal structure information system. 3.0c; Crystal Impact: Bonn, Germany, 2005.Suche in Google Scholar
5. Singh, M. M., Szafran, Z., Pike, R. M. Microscale synthesis of trans-chlorocarbonylbis(triphenylphosphine)rhodium(l) and its reactions-a versatile metal carbonyl complex. J. Chem. Educ. 1990, 67, A180–A182; https://doi.org/10.1021/ed067pa180.Suche in Google Scholar
6. Goswami, K., Singh, M. M. Few carbonyl oximato compounds of rhodium. J. Inorg. Nucl. Chem. 1977, 39, 1718–1719; https://doi.org/10.1016/0022-1902(77)80141-3.Suche in Google Scholar
7. Goswami, K., Singh, M. M. Di and monocarbonyl complexes of rhodium(I) containing singly charged bidentate ligands. Transition Met. Chem. 1981, 5, 83–85; https://doi.org/10.1007/bf01396876.Suche in Google Scholar
8. Elmakki, M. A. E., Alexander, O. T., Venter, G. J. S., Venter, J. A., Roodt, A. Structural study and iodomethane oxidative addition mechanistic investigation on model rhodium(I) carbonylation catalysts activated by indole-2-/indoline-2-carboxylate bidentate ligands. Inorganics 2022, 10, 1–24.10.3390/inorganics10120251Suche in Google Scholar
9. Warsink, S., Kotze, P. D. R., van Rensburg, J. M. J., Venter, J. A., Otto, S., Botha, E., Roodt, A. Kinetic-mechanistic and solid-state study of the oxidative addition and migratory insertion of iodomethane to [rhodium(S,O–BdiPT or N,O-ox)(CO) (PR1R2R3)] complexes. Eur. J. Inorg. Chem. 2018, 2018, 3615–3625; https://doi.org/10.1002/ejic.201800293.Suche in Google Scholar
10. Elmakki, M. A., Koen, R., Venter, J. A., Drost, R. Crystal structure of dicarbonyl(pyridine-2-olate-1-oxido-κ2O,O’)rhodium(I), C7H4NO4Rh. Z. Kristallogr. N. Cryst. Struct. 2016, 231, 703–705; https://doi.org/10.1515/ncrs-2015-0240.Suche in Google Scholar
11. Brink, A., Roodt, A., Steyl, G., Visser, H. G. Steric vs. electronic anomaly bserved from iodomethane oxidative addition to tertiary phosphine modified rhodium(I) acetylacetonato complexes following progressive phenyl replacement by cyclohexyl [PR3 = PPh3, PPh2Cy, PPhCy2, PCy3]. Dalton Trans. 2010, 39, 5572–5578; https://doi.org/10.1039/b922083f.Suche in Google Scholar PubMed
12. Elmakki, M. A. E., Alexander, O. T., Venter, G. J. S., Venter, J. A., Roodt, A. Synthesis and structural determination of [Rh(opo)(CO)(PR3)] complexes (opo- = 2-oxopyridin-1-olate) and in situ isomeric behavior from preliminary kinetic study of iodomethane oxidative addition. J. Coord. Chem. 2021, 74, 444–466; https://doi.org/10.1080/00958972.2021.1879385.Suche in Google Scholar
13. Venter, J. A., Leipoldt, J. G., van Eldik, R. Solvent, temperature, and pressure dependence of the oxidative addition of iodomethane to complexes of the type RhI(β-diketonate)(CO)(PPh3). Inorg. Chem. 1991, 30, 2207–2209; https://doi.org/10.1021/ic00009a046.Suche in Google Scholar
14. Elmakki, M. A., Koen, R., Drost, R. M., Alexander, O. T., Venter, G. J. S., Venter, J. A. Crystal structure of carbonyl 2-oxopyridin-1(2H-olato-κ2O,O′)(triphenylphosphine-κP)rhodium(I), C24H19NO3PRh. Z. Kristallogr. N. Cryst. Struct. 2016, 231, 781–783; https://doi.org/10.1515/ncrs-2015-0266.Suche in Google Scholar
15. Leipoldt, J. G., Lamprecht, G. J., Graham, D. E. The crystal structure of the methanol solvate of 2-carboxypyridinatocarbonyltriphenylphosphinerhodium(I). Inorg. Chim. Acta 1985, 101, 123–126; https://doi.org/10.1016/s0020-1693(00)85997-6.Suche in Google Scholar
© 2023 the author(s), published by De Gruyter, Berlin/Boston
This work is licensed under the Creative Commons Attribution 4.0 International License.
Artikel in diesem Heft
- Frontmatter
- New Crystal Structures
- Crystal structure of poly[diaqua-(μ4-3,3′-di(1H-1,2,4-triazol-1-yl)-[1,1′-biphenyl]-4,4′-dicarboxylate-N:N′:O:O′)cadmium(II)], C18H14N6O6Cd
- Crystal structure of (8R,8′S,13S,13′R)-8,8′-bis(hydroxymethyl)-9,9′,10,10′-tetramethoxy-5,5′,6,6′,8,8′,13,13′-octahydro-[13,13′-bi[1,3]dioxolo[4,5-g]isoquinolino[3,2-a]isoquinoline]-7,7′-diium chloride-methanol (1/2), C46H58N2O14Cl2
- The crystal structure of 8-methoxy-2,2-diphenyl-tosyl-1,2-dihydro-2λ4,3λ4-[1,3,2]diazaborolo[4,5,1-ig]quinoline, C29H25BN2O3S
- Crystal structure of aqua-(5,5,7,12,12,14-hexamethyl-1,4,8,11-tetraazacyclotetradecane-κ4N,N′,N″,N‴)copper(II) 5-carboxyisophthalate tetrahydrate, C25H50N4CuO11
- The crystal structure of 1-(naphthalen-2-ylsulfonyl)-2,2-diphenyl-1,2-dihydro-2λ4,3λ4-[1,3,2]diazaborolo[4,5,1-ij]quinoline, C31H23BN2O2S
- Crystal structure of iodido-(η6-benzene) (1-(pyridin-2-yl)-N-(p-fluoro-methanamine)-κ2N,Nʹ)ruthenium(II) hexaflourophosphate, (C18H15F7IN2RuP)
- The crystal structure of 1-(3-oxo-1-phenyl-3-(p-tolyl) propylidene)-1,3-dihydro-2H-inden-2-one, C25H20O2
- Crystal structure of tricyclohexyl[4-(4H-1,2,4-triazol-4-yl)-benzoato-κO]tin(IV), C27H39N3O2Sn
- Crystal structure of [triaqua-(8-carboxymethoxy-quinoline-2-carboxylate-κ4N,O,O,O)cadmium(II)]monohydrate, C12H15NO9Cd
- Crystal structure of ethyl 2-((4-(3,5-dimethylisoxazol-4-yl)-2,6-difluorophenyl)amino)benzoate, C20H18F2N2O3
- The crystal structure of 2-(hydroxymethyl)-2-(4H-1,2,4-triazol-4-yl)propane-1,3-diol, C6H11N3O3
- The crystal structure of 1,2-bis(2,4-dinitrophenyl) hydrazine, C12H8N6O8
- Crystal structure of 1-(2,6-dichloro-4-(3,5-dimethylisoxazol-4-yl)phenyl)-1,2-dihydro-4H-benzo[d][1,3]oxazin-4-one, C19H14Cl2N2O3
- The crystal structure of 5-amino-5-oxo-4-(1-oxo-4-(2-oxopyrrolidin-1-yl)isoindolin-2-yl)pentanoic acid, C17H19N3O5
- Crystal structure of N2,N6-bis(2-(((Z)-5-bromo-2-hydroxybenzylidene)amino) phenyl)pyridine-2,6-dicarboxamide, C33H23Br2N5O4
- The crystal structure of (E)-2-methoxy-6-(((5-methyl-1,3,4-thiadiazol-2-yl)imino)methyl)phenol, C11H11N3O2S
- The crystal structure of 3-((tert-butyldiphenylsilyl)methyl)-5,5-diphenyl-6-(p-tolyl) tetrahydro-2H-pyran-2-one, C41H42O2Si
- Crystal structure of 9-fluoro-4-(6-methoxypyridin-3-yl)-5,6-dihydrobenzo[h]quinazolin-2-amine, C18H15FN4O
- The crystal structure of 2-bromo-5-(4-cyanophenoxy)benzyl 1-methyl-1,2,5,6-tetrahydropyridine-3-carboxylate, C21H19BrN2O3
- Crystal structure of 3,3′-(1,4-phenylenebis(methylene))bis(1-isopropyl-1H-imidazol-3-ium) bis(hexafluorophosphate(V)), C10H14F6N2P
- The crystal structure of 2,2-di(thiophen-3-yl)-1-tosyl-1,2-dihydro-2λ4,3λ4-[1,3,2]diazaborolo[4,5,1-ig]quinoline, C24H19BN2O2S3
- Crystal structure of 5-bromo-1-(2-iodobenzoyl)-1H-indole-3-carbaldehyde, C16H9BrINO2
- The crystal structure of monocarbonyl-2-carboxypyridinato-κ2N,O-triphenylphosphine-rhodium(I) acetonitrile solvate, C26H20.50N1.50O3PRh
- Crystal structure of dichlorido-tetrakis(1-(2,4-dichlorophenyl)-4,4-dimethyl-2-(1,2,4-triazol-1-yl)pent-1-en-3-ol-κ1N)manganese(II), C60H68O4N12Cl10Mn
- Crystal structure of 3-(tert-butyldiphenylsilyl)-1-(2,6-dichlorophenyl)-2,2-diphenylpropan-1-ol, C37H36Cl2OSi
- Crystal structure of langite from Mine du Pradet (France)
- The crystal structure of 5′-(furan-2-yl)-3′-((4-methylphenyl)sulfonamido)-3′,4′,5′,6′-tetrahydro-[1,1′:3′,1″-terphenyl]-4′-carboxylic acid, C30H27NO5S
- Synthesis and crystal structure of bis{2-(((4-acetophenone)imino)methyl)-4-fluorophenolato-κ2N,O}zinc(II), C30H22F2N2O4Zn
- The crystal structure of poly[(tripyridine-κ3N,N′,N″) μ3-(pyridine-3,4-dicarboxylate-κ3N:O:O′) manganese(II)], C22H22N4O8Mn
- The crystal structure of (E)-4-chloro-N′-(1-(4-hydroxyphenyl)propylidene)benzohydrazide, C16H15ClN2O2
- Synthesis and crystal structure of bis{2-(tert-butyl)-6-((E)-((4-((E)-1-(methoxyimino) ethyl)phenyl)imino)methyl)phenolato-κ2N,O}cobalt(II), C40H46CoN4O4
- Crystal structure of tetraaqua-[(1-(carboxymethyl)-1H-pyrazole-3-carboxylato-κ2N,O)cobalt(II)], C6H12CoN2O8
- (6R,7S)-2,3,13-trimethoxy-6,7-dimethyl-5,6,7,8-tetrahydrobenzo[3′,4′]cycloocta [1′,2′:4,5]benzo[1,2-d][1,3]dioxol-1-ol, C22H26O6
- Crystal structure of 2-((2,6-dichloro-4-(3,5-dimethylisoxazol-4-yl)phenyl)amino)benzoic acid, C18H14Cl2N2O3
- Crystal structure of (5aS,6aS,8aR,9R,11aS, 11bS,13R,13aS)-1,1,8a,11a-tetramethyl-9-((S)-1-((S)-5-methyl-6-oxo-3,6-dihydro-2H-pyran-2-yl)ethyl)-3-oxo-1,7,8,8a,9,10,11,11a,11b,12,13,13a-dodecahydro-3H,6H-cyclopenta[5,6]cyclopropa[1,8a]naphtho[2,1-c]oxepin-13-yl acetate, C32H44O6
- Crystal structure of catena-poly[triaqua-(μ2-1-(4-carboxylatophenyl)-4-oxo-1,4-dihydropyridazine-3-carboxylato-O,O′:O″)cobalt(II)], C12H12N2O8Co
- Crystal structure of 3-[(furan-2-ylmethyl)-amino]-2-(2,3,4,5-tetrafluoro-benzoyl)-acrylic acid ethyl ester, C17H13F4NO4
- Crystal structure of methyl 4-(2-ethoxy-2-oxoethoxy)-3-methoxybenzoate, C13H16O6
- Crystal structure of 4-bromo-2-(4-chlorophenyl)-1-methyl-5-(trifluoromethyl)-1H-pyrrole-3-carbonitrile, C13H7BrClF3N2
- The crystal structure of triaqua-(8-carboxymethoxy-quinoline-2-carboxylate-κ3N,O,O)nickel(II) monohydrate, C12H15NO9Ni
- Crystal structure of dihydroxy(2,4,6-triisopro-pylphenyl)telluronium trifluoromethanesulfonate, C16H25F3O5STe
- The crystal structure of 1-(carboxymethyl)-1H-imidazole 3-oxide
- The crystal structure of 1,3,5-tris(dibromomethyl)benzene, C9H6Br6
- Crystal structure of (Z)-3-(4-methoxyphenyl)-4-(5-methyl-1-phenyl-1H-1,2,3-triazol-4-yl)-N-phenylthiazol-2(3H)-imine, C25H21N5OS
- Crystal structure of (Z)-3-(3-(4-hydroxyphenyl)-2-(phenylimino)-2,3-dihydrothiazol-4-yl)-2H-chromen-2-one, C24H16N2O3S
Artikel in diesem Heft
- Frontmatter
- New Crystal Structures
- Crystal structure of poly[diaqua-(μ4-3,3′-di(1H-1,2,4-triazol-1-yl)-[1,1′-biphenyl]-4,4′-dicarboxylate-N:N′:O:O′)cadmium(II)], C18H14N6O6Cd
- Crystal structure of (8R,8′S,13S,13′R)-8,8′-bis(hydroxymethyl)-9,9′,10,10′-tetramethoxy-5,5′,6,6′,8,8′,13,13′-octahydro-[13,13′-bi[1,3]dioxolo[4,5-g]isoquinolino[3,2-a]isoquinoline]-7,7′-diium chloride-methanol (1/2), C46H58N2O14Cl2
- The crystal structure of 8-methoxy-2,2-diphenyl-tosyl-1,2-dihydro-2λ4,3λ4-[1,3,2]diazaborolo[4,5,1-ig]quinoline, C29H25BN2O3S
- Crystal structure of aqua-(5,5,7,12,12,14-hexamethyl-1,4,8,11-tetraazacyclotetradecane-κ4N,N′,N″,N‴)copper(II) 5-carboxyisophthalate tetrahydrate, C25H50N4CuO11
- The crystal structure of 1-(naphthalen-2-ylsulfonyl)-2,2-diphenyl-1,2-dihydro-2λ4,3λ4-[1,3,2]diazaborolo[4,5,1-ij]quinoline, C31H23BN2O2S
- Crystal structure of iodido-(η6-benzene) (1-(pyridin-2-yl)-N-(p-fluoro-methanamine)-κ2N,Nʹ)ruthenium(II) hexaflourophosphate, (C18H15F7IN2RuP)
- The crystal structure of 1-(3-oxo-1-phenyl-3-(p-tolyl) propylidene)-1,3-dihydro-2H-inden-2-one, C25H20O2
- Crystal structure of tricyclohexyl[4-(4H-1,2,4-triazol-4-yl)-benzoato-κO]tin(IV), C27H39N3O2Sn
- Crystal structure of [triaqua-(8-carboxymethoxy-quinoline-2-carboxylate-κ4N,O,O,O)cadmium(II)]monohydrate, C12H15NO9Cd
- Crystal structure of ethyl 2-((4-(3,5-dimethylisoxazol-4-yl)-2,6-difluorophenyl)amino)benzoate, C20H18F2N2O3
- The crystal structure of 2-(hydroxymethyl)-2-(4H-1,2,4-triazol-4-yl)propane-1,3-diol, C6H11N3O3
- The crystal structure of 1,2-bis(2,4-dinitrophenyl) hydrazine, C12H8N6O8
- Crystal structure of 1-(2,6-dichloro-4-(3,5-dimethylisoxazol-4-yl)phenyl)-1,2-dihydro-4H-benzo[d][1,3]oxazin-4-one, C19H14Cl2N2O3
- The crystal structure of 5-amino-5-oxo-4-(1-oxo-4-(2-oxopyrrolidin-1-yl)isoindolin-2-yl)pentanoic acid, C17H19N3O5
- Crystal structure of N2,N6-bis(2-(((Z)-5-bromo-2-hydroxybenzylidene)amino) phenyl)pyridine-2,6-dicarboxamide, C33H23Br2N5O4
- The crystal structure of (E)-2-methoxy-6-(((5-methyl-1,3,4-thiadiazol-2-yl)imino)methyl)phenol, C11H11N3O2S
- The crystal structure of 3-((tert-butyldiphenylsilyl)methyl)-5,5-diphenyl-6-(p-tolyl) tetrahydro-2H-pyran-2-one, C41H42O2Si
- Crystal structure of 9-fluoro-4-(6-methoxypyridin-3-yl)-5,6-dihydrobenzo[h]quinazolin-2-amine, C18H15FN4O
- The crystal structure of 2-bromo-5-(4-cyanophenoxy)benzyl 1-methyl-1,2,5,6-tetrahydropyridine-3-carboxylate, C21H19BrN2O3
- Crystal structure of 3,3′-(1,4-phenylenebis(methylene))bis(1-isopropyl-1H-imidazol-3-ium) bis(hexafluorophosphate(V)), C10H14F6N2P
- The crystal structure of 2,2-di(thiophen-3-yl)-1-tosyl-1,2-dihydro-2λ4,3λ4-[1,3,2]diazaborolo[4,5,1-ig]quinoline, C24H19BN2O2S3
- Crystal structure of 5-bromo-1-(2-iodobenzoyl)-1H-indole-3-carbaldehyde, C16H9BrINO2
- The crystal structure of monocarbonyl-2-carboxypyridinato-κ2N,O-triphenylphosphine-rhodium(I) acetonitrile solvate, C26H20.50N1.50O3PRh
- Crystal structure of dichlorido-tetrakis(1-(2,4-dichlorophenyl)-4,4-dimethyl-2-(1,2,4-triazol-1-yl)pent-1-en-3-ol-κ1N)manganese(II), C60H68O4N12Cl10Mn
- Crystal structure of 3-(tert-butyldiphenylsilyl)-1-(2,6-dichlorophenyl)-2,2-diphenylpropan-1-ol, C37H36Cl2OSi
- Crystal structure of langite from Mine du Pradet (France)
- The crystal structure of 5′-(furan-2-yl)-3′-((4-methylphenyl)sulfonamido)-3′,4′,5′,6′-tetrahydro-[1,1′:3′,1″-terphenyl]-4′-carboxylic acid, C30H27NO5S
- Synthesis and crystal structure of bis{2-(((4-acetophenone)imino)methyl)-4-fluorophenolato-κ2N,O}zinc(II), C30H22F2N2O4Zn
- The crystal structure of poly[(tripyridine-κ3N,N′,N″) μ3-(pyridine-3,4-dicarboxylate-κ3N:O:O′) manganese(II)], C22H22N4O8Mn
- The crystal structure of (E)-4-chloro-N′-(1-(4-hydroxyphenyl)propylidene)benzohydrazide, C16H15ClN2O2
- Synthesis and crystal structure of bis{2-(tert-butyl)-6-((E)-((4-((E)-1-(methoxyimino) ethyl)phenyl)imino)methyl)phenolato-κ2N,O}cobalt(II), C40H46CoN4O4
- Crystal structure of tetraaqua-[(1-(carboxymethyl)-1H-pyrazole-3-carboxylato-κ2N,O)cobalt(II)], C6H12CoN2O8
- (6R,7S)-2,3,13-trimethoxy-6,7-dimethyl-5,6,7,8-tetrahydrobenzo[3′,4′]cycloocta [1′,2′:4,5]benzo[1,2-d][1,3]dioxol-1-ol, C22H26O6
- Crystal structure of 2-((2,6-dichloro-4-(3,5-dimethylisoxazol-4-yl)phenyl)amino)benzoic acid, C18H14Cl2N2O3
- Crystal structure of (5aS,6aS,8aR,9R,11aS, 11bS,13R,13aS)-1,1,8a,11a-tetramethyl-9-((S)-1-((S)-5-methyl-6-oxo-3,6-dihydro-2H-pyran-2-yl)ethyl)-3-oxo-1,7,8,8a,9,10,11,11a,11b,12,13,13a-dodecahydro-3H,6H-cyclopenta[5,6]cyclopropa[1,8a]naphtho[2,1-c]oxepin-13-yl acetate, C32H44O6
- Crystal structure of catena-poly[triaqua-(μ2-1-(4-carboxylatophenyl)-4-oxo-1,4-dihydropyridazine-3-carboxylato-O,O′:O″)cobalt(II)], C12H12N2O8Co
- Crystal structure of 3-[(furan-2-ylmethyl)-amino]-2-(2,3,4,5-tetrafluoro-benzoyl)-acrylic acid ethyl ester, C17H13F4NO4
- Crystal structure of methyl 4-(2-ethoxy-2-oxoethoxy)-3-methoxybenzoate, C13H16O6
- Crystal structure of 4-bromo-2-(4-chlorophenyl)-1-methyl-5-(trifluoromethyl)-1H-pyrrole-3-carbonitrile, C13H7BrClF3N2
- The crystal structure of triaqua-(8-carboxymethoxy-quinoline-2-carboxylate-κ3N,O,O)nickel(II) monohydrate, C12H15NO9Ni
- Crystal structure of dihydroxy(2,4,6-triisopro-pylphenyl)telluronium trifluoromethanesulfonate, C16H25F3O5STe
- The crystal structure of 1-(carboxymethyl)-1H-imidazole 3-oxide
- The crystal structure of 1,3,5-tris(dibromomethyl)benzene, C9H6Br6
- Crystal structure of (Z)-3-(4-methoxyphenyl)-4-(5-methyl-1-phenyl-1H-1,2,3-triazol-4-yl)-N-phenylthiazol-2(3H)-imine, C25H21N5OS
- Crystal structure of (Z)-3-(3-(4-hydroxyphenyl)-2-(phenylimino)-2,3-dihydrothiazol-4-yl)-2H-chromen-2-one, C24H16N2O3S

