Abstract
C16H25F3O5STe, monoclinic, P21/c (no. 14), a = 5.8214(2) Å, b = 16.0059(5) Å, c = 21.9311(7) Å, β = 94.760(3)°, V = 2036.42(12) Å3, Z = 4, R gt(F) = 0.0282, wR ref(F 2) = 0.0598, T = 293 K.
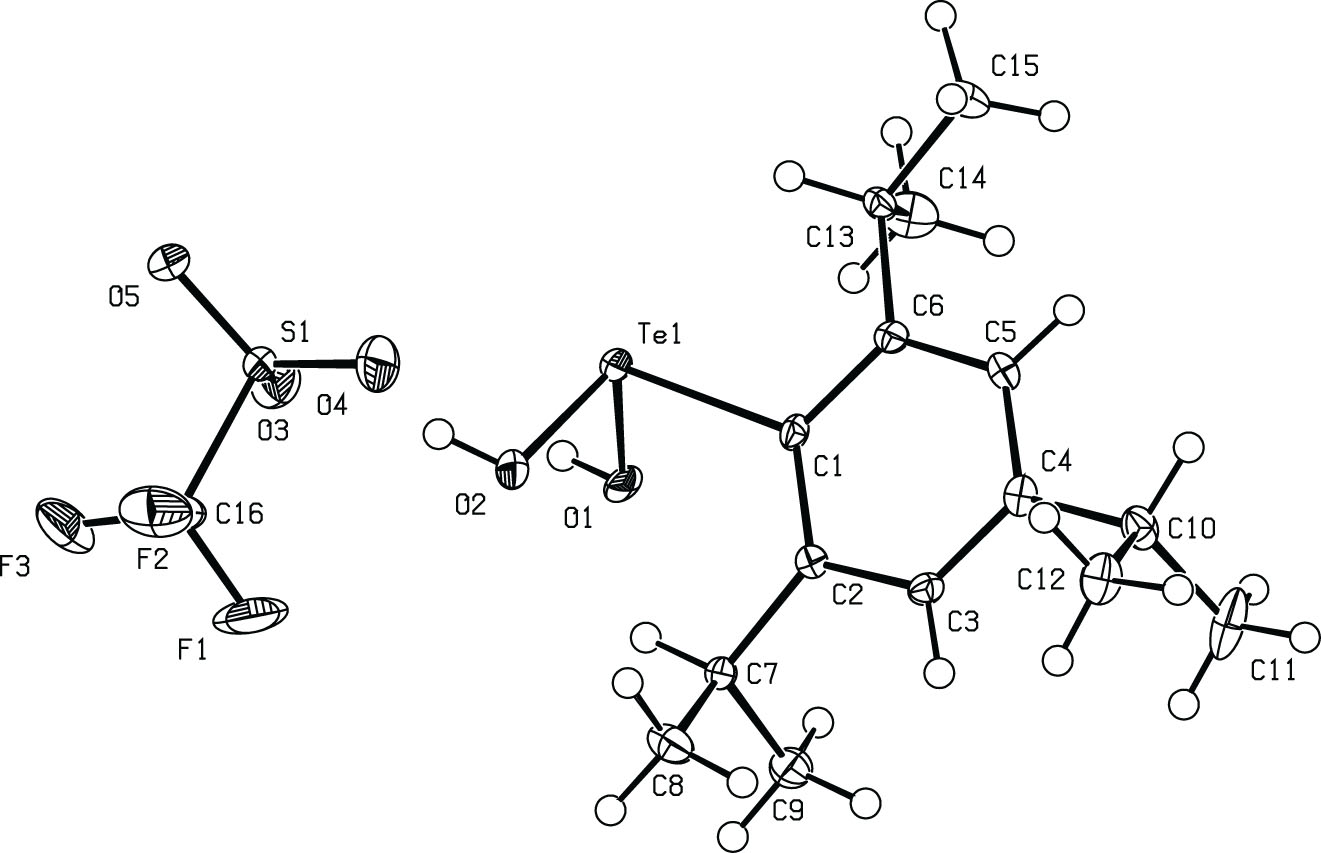
The molecular structure is shown in the figure. Table 1 contains crystallographic data and Table 2 contains the list of the atoms including atomic coordinates and displacement parameters.
Data collection and handling.
| Crystal: | Colourless prism |
| Size: | 0.40 × 0.20 × 0.10 mm |
| Wavelength: | MoKα radiation (0.71073 Å) |
| μ: | 1.61 mm−1 |
| Diffractometer, scan mode: | XtaLAB Pro, ω |
| θ max, completeness: | 29.4°, >99 % |
| N(hkl)measured, N(hkl)unique, R int: | 29,146, 5013, 0.045 |
| Criterion for I obs, N(hkl)gt: | I obs > 2 σ(I obs), 4203 |
| N(param)refined: | 247 |
| Programs: | CrysAlis Pro [1], Shelx [2, 3], Olex2 [4] |
Fractional atomic coordinates and isotropic or equivalent isotropic displacement parameters (Å2).
| Atom | x | y | z | U iso*/U eq |
|---|---|---|---|---|
| Te1 | −0.04480 (3) | 0.63825 (2) | 0.44080 (2) | 0.01553 (5) |
| O1 | −0.3278 (3) | 0.64277 (11) | 0.38472 (8) | 0.0195 (4) |
| H1 | −0.409407 | 0.603844 | 0.397991 | 0.029* |
| O2 | 0.1371 (4) | 0.57150 (11) | 0.39152 (8) | 0.0244 (4) |
| H2 | 0.163239 | 0.524126 | 0.408242 | 0.037* |
| C1 | 0.0752 (4) | 0.75297 (14) | 0.40908 (10) | 0.0123 (4) |
| C2 | 0.1286 (4) | 0.76408 (14) | 0.34813 (10) | 0.0126 (4) |
| C3 | 0.2454 (4) | 0.83757 (15) | 0.33513 (11) | 0.0136 (5) |
| H3 | 0.286249 | 0.846110 | 0.295496 | 0.016* |
| C4 | 0.3029 (4) | 0.89836 (15) | 0.37915 (11) | 0.0136 (5) |
| C5 | 0.2299 (4) | 0.88745 (14) | 0.43773 (11) | 0.0126 (4) |
| H5 | 0.258289 | 0.929503 | 0.466638 | 0.015* |
| C6 | 0.1161 (4) | 0.81545 (14) | 0.45402 (10) | 0.0116 (4) |
| C7 | 0.0545 (4) | 0.70568 (15) | 0.29544 (10) | 0.0144 (5) |
| H7 | −0.022336 | 0.657393 | 0.312235 | 0.017* |
| C8 | 0.2586 (5) | 0.67339 (17) | 0.26213 (12) | 0.0220 (5) |
| H8A | 0.333167 | 0.719520 | 0.243930 | 0.033* |
| H8B | 0.203768 | 0.634701 | 0.230780 | 0.033* |
| H8C | 0.366564 | 0.645748 | 0.290848 | 0.033* |
| C9 | −0.1215 (4) | 0.75073 (17) | 0.25080 (12) | 0.0206 (5) |
| H9A | −0.250596 | 0.768218 | 0.272168 | 0.031* |
| H9B | −0.173365 | 0.713394 | 0.218270 | 0.031* |
| H9C | −0.050342 | 0.798736 | 0.234064 | 0.031* |
| C10 | 0.4404 (4) | 0.97502 (15) | 0.36397 (12) | 0.0181 (5) |
| H10 | 0.474577 | 1.006491 | 0.401968 | 0.022* |
| C11 | 0.3059 (5) | 1.03217 (17) | 0.31881 (16) | 0.0305 (7) |
| H11A | 0.273310 | 1.003370 | 0.280672 | 0.046* |
| H11B | 0.395538 | 1.081226 | 0.312270 | 0.046* |
| H11C | 0.163889 | 1.048065 | 0.334899 | 0.046* |
| C12 | 0.6711 (4) | 0.95028 (16) | 0.33997 (13) | 0.0203 (5) |
| H12A | 0.753061 | 0.913587 | 0.368777 | 0.030* |
| H12B | 0.761572 | 0.999520 | 0.334741 | 0.030* |
| H12C | 0.643070 | 0.922245 | 0.301368 | 0.030* |
| C13 | 0.0226 (4) | 0.80798 (15) | 0.51678 (10) | 0.0135 (5) |
| H13 | 0.041550 | 0.749671 | 0.529886 | 0.016* |
| C14 | −0.2363 (4) | 0.82733 (19) | 0.51168 (13) | 0.0246 (6) |
| H14A | −0.260547 | 0.884408 | 0.499334 | 0.037* |
| H14B | −0.296236 | 0.818629 | 0.550662 | 0.037* |
| H14C | −0.314191 | 0.791052 | 0.481841 | 0.037* |
| C15 | 0.1467 (4) | 0.86231 (18) | 0.56626 (11) | 0.0212 (5) |
| H15A | 0.310037 | 0.854169 | 0.566275 | 0.032* |
| H15B | 0.097601 | 0.847001 | 0.605464 | 0.032* |
| H15C | 0.110385 | 0.919979 | 0.558191 | 0.032* |
| S1 | 0.54239 (11) | 0.40927 (4) | 0.44132 (3) | 0.01662 (12) |
| O3 | 0.2948 (3) | 0.41948 (13) | 0.43240 (9) | 0.0274 (4) |
| O4 | 0.6718 (4) | 0.48501 (12) | 0.44131 (10) | 0.0328 (5) |
| O5 | 0.6168 (3) | 0.35064 (11) | 0.48920 (8) | 0.0206 (4) |
| C16 | 0.6107 (5) | 0.35701 (19) | 0.37101 (12) | 0.0258 (6) |
| F1 | 0.5475 (4) | 0.40322 (14) | 0.32249 (8) | 0.0473 (5) |
| F2 | 0.8352 (3) | 0.34227 (13) | 0.37165 (8) | 0.0397 (5) |
| F3 | 0.5018 (3) | 0.28402 (12) | 0.36496 (8) | 0.0414 (5) |
1 Source of material
A mixture of 2,4,6-tripropylbenzenetellurinic anhydride (0.7097 g, 0.9997 mmol) and trifluoromethanesulfonic anhydride (0.3103 g, 1.100 mmol) in dichloromethane (10 ml) was stirred at room temperature for 18 h under an argon atmosphere. After removing the solvent under reduced pressure, the resulting highly viscous residue was left at room temperature in the presence of air for three days. The precipitated crystals were then washed with hexane to obtain the final compound (0.4504 g, 0.8762 mmol, 44 %).
2 Experimental details
Hydrogen atoms were placed at predetermined positions and refined using a riding model. In this model, the C–H bond distances were set to 0.96 Å for methyl groups, 0.98 Å for methane groups, and 0.93 Å for aromatic groups. For methyl H atoms, the uncertainty displacement parameter (U iso(H)) was constrained to 1.5 times the equivalent isotropic displacement parameter (U eq(C)), and for all other H atoms, it was constrained to 1.2 times U eq(C).
3 Comment
Tellurinic acid is a fundamental organotellurium oxoacid that usually exists as complex aggregates because of highly polarized Te–O bonds that form intermolecular interactions. In 2008, the first well-defined tellurinic acid was isolated as a kinetically stabilized dimer by introducing bulky substituents onto the tellurium atom [5]. Recently, the isolation of a monomeric 2-(2-pyridyl)phenyltellurinic acid [ppyTe(O)OH], stabilized via intramolecular chalcogen bonding, has been reported [6]. Conversely, the sodium salt of tellurinic acid exists as a monomer without bulky substituents or stabilizing intramolecular bonds [7]. In this study, we report the crystal structure of a structurally rare dihydroxytelluronium compound, which can be considered to be a protonated form of tellurinic acid.
The asymmetric unit of the compound contains one ion pair associated with a medium-strength hydrogen bond (O2⋯O3 2.726 (3) Å) [8]. The donor–acceptor (O⋯O) distance is slightly shorter than that observed in the structurally related hydroxydiaryltelluronium trifluoromethanesulfonate [(8-Me2NC10H6)2TeOH]OTf (O⋯O 2.814 (6) Å) [9]. For the molecular structure, the geometry around the Te center in the dihydroxy(2,4,6-triisopropylphenyl)telluronium moiety is best described as a distorted tetrahedral with a CO2 donor set, considering the lone pair. The angles C1–Te1–O1, C1–Te1–O2, and O11–Te1–O2 are 92.57 (8)°, 94.97 (9)° and 98.15 (8)°, respectively. The two Te–OH bond lengths are not the same. The bond distance of Te1–O1 (1.974 (2) Å) is comparable to those observed in ppyTe(O)OH (Te–OH 1.966 (2) Å) and [(8-Me2NC10H6)2TeOH]OTf (Te–OH 1.957 (4) Å), whereas the Te1–O2 distance (1.903 (2) Å) is intermediate between those and Te=O (1.834 (2) Å in ppyTe(O)OH), indicating that the title compound was formed by the protonation of tellurinic acid. In the crystal lattice, O1 is hydrogen bonded to another TfO− (O1⋯O4 i 2.814 (3) Å, i = −1 + x, y, z), although it is slightly weaker than the O2⋯O3 hydrogen bond described above. As a result, hydrogen bonds alternately bind telluronium cations and trifluoromethanesulfonate anions, forming a column structure along the a-axis.
Funding source: Tokai University
Acknowledgments
The authors wish to thank Enago (www.enago.jp) for the English language review.
-
Author contributions: All authors have accepted responsibility for the entire content of this submitted manuscript and approved submission.
-
Research funding: This research was supported in part by a doctoral dissertation grant from Tokai University.
-
Conflict of interest statement: The authors declare no conflicts of interest regarding this article.
References
1. Rigaku Oxford Diffraction. CrysAlisPro; Rigaku Corporation: Tokyo, Japan, 2015.Search in Google Scholar
2. Sheldrick, G. M. SHELXTL – integrated space-group and crystal-structure determination. Acta Crystallogr. 2015, A71, 3–8. https://doi.org/10.1107/S2053273314026370.Search in Google Scholar PubMed PubMed Central
3. Sheldrick, G. M. Crystal structure refinement with SHELXL. Acta Crystallogr. 2015, C71, 3–8. https://doi.org/10.1107/S2053229614024218.Search in Google Scholar PubMed PubMed Central
4. Dolomanov, O. V., Bourhis, L. J., Gildea, R. J., Howard, J. A. K., Puschmann, H. OLEX2: a complete structure solution, refinement and analysis program. J. Appl. Crystallogr. 2009, 42, 339–341. https://doi.org/10.1107/S0021889808042726.Search in Google Scholar
5. Beckmann, J., Finke, P., Hesse, M., Wettig, B. Well-defined stibonic and tellurinic acids. Angew. Chem. Int. Ed. 2008, 47, 9982–9984. https://doi.org/10.1002/anie.200803997.Search in Google Scholar PubMed
6. Deka, R., Sarkar, A., Butcher, R. J., Junk, P. C., Turner, D. R., Deacon, G. B., Singh, H. B. Isolation of the novel example of a monomeric organotellurinic acid. Dalton Trans. 2020, 49, 1173–1180. https://doi.org/10.1039/C9DT04013G.Search in Google Scholar PubMed
7. Beckmann, J., Duthie, A., Gesing, T. M., Koehne, T., Lork, E. Depolymerization of aryltellurinic anhydrides with sodium hydroxide. Synthesis and structure of the hydrated sodium aryltellurinates [Na(H2O)4](RTeO2) (R = 4-MeOC6H4, 8-Me2NC10H6). Organometallics 2012, 31, 3451–3454. https://doi.org/10.1021/om300130a.Search in Google Scholar
8. Steiner, T. The hydrogen bond in the solid state. Angew. Chem. Int. Ed. 2002, 41, 49–76. https://doi.org/10.1002/1521–3773(20020104)41:1%3C48::AID–ANIE48%3E3.0.CO,2–U.10.1002/1521-3773(20020104)41:1<48::AID-ANIE48>3.0.CO;2-USearch in Google Scholar
9. Beckmann, J., Bolsinger, J., Duthie, A., Finke, P. New series of intramolecularly coordinated diaryltellurium compounds. Rational synthesis of the diarylhydroxytelluronium triflate [(8-Me2NC10H6)2Te(OH)](O3SCF3). Organometallics 2012, 31, 238–245. https://doi.org/10.1021/om2008259.Search in Google Scholar
© 2023 the author(s), published by De Gruyter, Berlin/Boston
This work is licensed under the Creative Commons Attribution 4.0 International License.
Articles in the same Issue
- Frontmatter
- New Crystal Structures
- Crystal structure of poly[diaqua-(μ4-3,3′-di(1H-1,2,4-triazol-1-yl)-[1,1′-biphenyl]-4,4′-dicarboxylate-N:N′:O:O′)cadmium(II)], C18H14N6O6Cd
- Crystal structure of (8R,8′S,13S,13′R)-8,8′-bis(hydroxymethyl)-9,9′,10,10′-tetramethoxy-5,5′,6,6′,8,8′,13,13′-octahydro-[13,13′-bi[1,3]dioxolo[4,5-g]isoquinolino[3,2-a]isoquinoline]-7,7′-diium chloride-methanol (1/2), C46H58N2O14Cl2
- The crystal structure of 8-methoxy-2,2-diphenyl-tosyl-1,2-dihydro-2λ4,3λ4-[1,3,2]diazaborolo[4,5,1-ig]quinoline, C29H25BN2O3S
- Crystal structure of aqua-(5,5,7,12,12,14-hexamethyl-1,4,8,11-tetraazacyclotetradecane-κ4N,N′,N″,N‴)copper(II) 5-carboxyisophthalate tetrahydrate, C25H50N4CuO11
- The crystal structure of 1-(naphthalen-2-ylsulfonyl)-2,2-diphenyl-1,2-dihydro-2λ4,3λ4-[1,3,2]diazaborolo[4,5,1-ij]quinoline, C31H23BN2O2S
- Crystal structure of iodido-(η6-benzene) (1-(pyridin-2-yl)-N-(p-fluoro-methanamine)-κ2N,Nʹ)ruthenium(II) hexaflourophosphate, (C18H15F7IN2RuP)
- The crystal structure of 1-(3-oxo-1-phenyl-3-(p-tolyl) propylidene)-1,3-dihydro-2H-inden-2-one, C25H20O2
- Crystal structure of tricyclohexyl[4-(4H-1,2,4-triazol-4-yl)-benzoato-κO]tin(IV), C27H39N3O2Sn
- Crystal structure of [triaqua-(8-carboxymethoxy-quinoline-2-carboxylate-κ4N,O,O,O)cadmium(II)]monohydrate, C12H15NO9Cd
- Crystal structure of ethyl 2-((4-(3,5-dimethylisoxazol-4-yl)-2,6-difluorophenyl)amino)benzoate, C20H18F2N2O3
- The crystal structure of 2-(hydroxymethyl)-2-(4H-1,2,4-triazol-4-yl)propane-1,3-diol, C6H11N3O3
- The crystal structure of 1,2-bis(2,4-dinitrophenyl) hydrazine, C12H8N6O8
- Crystal structure of 1-(2,6-dichloro-4-(3,5-dimethylisoxazol-4-yl)phenyl)-1,2-dihydro-4H-benzo[d][1,3]oxazin-4-one, C19H14Cl2N2O3
- The crystal structure of 5-amino-5-oxo-4-(1-oxo-4-(2-oxopyrrolidin-1-yl)isoindolin-2-yl)pentanoic acid, C17H19N3O5
- Crystal structure of N2,N6-bis(2-(((Z)-5-bromo-2-hydroxybenzylidene)amino) phenyl)pyridine-2,6-dicarboxamide, C33H23Br2N5O4
- The crystal structure of (E)-2-methoxy-6-(((5-methyl-1,3,4-thiadiazol-2-yl)imino)methyl)phenol, C11H11N3O2S
- The crystal structure of 3-((tert-butyldiphenylsilyl)methyl)-5,5-diphenyl-6-(p-tolyl) tetrahydro-2H-pyran-2-one, C41H42O2Si
- Crystal structure of 9-fluoro-4-(6-methoxypyridin-3-yl)-5,6-dihydrobenzo[h]quinazolin-2-amine, C18H15FN4O
- The crystal structure of 2-bromo-5-(4-cyanophenoxy)benzyl 1-methyl-1,2,5,6-tetrahydropyridine-3-carboxylate, C21H19BrN2O3
- Crystal structure of 3,3′-(1,4-phenylenebis(methylene))bis(1-isopropyl-1H-imidazol-3-ium) bis(hexafluorophosphate(V)), C10H14F6N2P
- The crystal structure of 2,2-di(thiophen-3-yl)-1-tosyl-1,2-dihydro-2λ4,3λ4-[1,3,2]diazaborolo[4,5,1-ig]quinoline, C24H19BN2O2S3
- Crystal structure of 5-bromo-1-(2-iodobenzoyl)-1H-indole-3-carbaldehyde, C16H9BrINO2
- The crystal structure of monocarbonyl-2-carboxypyridinato-κ2N,O-triphenylphosphine-rhodium(I) acetonitrile solvate, C26H20.50N1.50O3PRh
- Crystal structure of dichlorido-tetrakis(1-(2,4-dichlorophenyl)-4,4-dimethyl-2-(1,2,4-triazol-1-yl)pent-1-en-3-ol-κ1N)manganese(II), C60H68O4N12Cl10Mn
- Crystal structure of 3-(tert-butyldiphenylsilyl)-1-(2,6-dichlorophenyl)-2,2-diphenylpropan-1-ol, C37H36Cl2OSi
- Crystal structure of langite from Mine du Pradet (France)
- The crystal structure of 5′-(furan-2-yl)-3′-((4-methylphenyl)sulfonamido)-3′,4′,5′,6′-tetrahydro-[1,1′:3′,1″-terphenyl]-4′-carboxylic acid, C30H27NO5S
- Synthesis and crystal structure of bis{2-(((4-acetophenone)imino)methyl)-4-fluorophenolato-κ2N,O}zinc(II), C30H22F2N2O4Zn
- The crystal structure of poly[(tripyridine-κ3N,N′,N″) μ3-(pyridine-3,4-dicarboxylate-κ3N:O:O′) manganese(II)], C22H22N4O8Mn
- The crystal structure of (E)-4-chloro-N′-(1-(4-hydroxyphenyl)propylidene)benzohydrazide, C16H15ClN2O2
- Synthesis and crystal structure of bis{2-(tert-butyl)-6-((E)-((4-((E)-1-(methoxyimino) ethyl)phenyl)imino)methyl)phenolato-κ2N,O}cobalt(II), C40H46CoN4O4
- Crystal structure of tetraaqua-[(1-(carboxymethyl)-1H-pyrazole-3-carboxylato-κ2N,O)cobalt(II)], C6H12CoN2O8
- (6R,7S)-2,3,13-trimethoxy-6,7-dimethyl-5,6,7,8-tetrahydrobenzo[3′,4′]cycloocta [1′,2′:4,5]benzo[1,2-d][1,3]dioxol-1-ol, C22H26O6
- Crystal structure of 2-((2,6-dichloro-4-(3,5-dimethylisoxazol-4-yl)phenyl)amino)benzoic acid, C18H14Cl2N2O3
- Crystal structure of (5aS,6aS,8aR,9R,11aS, 11bS,13R,13aS)-1,1,8a,11a-tetramethyl-9-((S)-1-((S)-5-methyl-6-oxo-3,6-dihydro-2H-pyran-2-yl)ethyl)-3-oxo-1,7,8,8a,9,10,11,11a,11b,12,13,13a-dodecahydro-3H,6H-cyclopenta[5,6]cyclopropa[1,8a]naphtho[2,1-c]oxepin-13-yl acetate, C32H44O6
- Crystal structure of catena-poly[triaqua-(μ2-1-(4-carboxylatophenyl)-4-oxo-1,4-dihydropyridazine-3-carboxylato-O,O′:O″)cobalt(II)], C12H12N2O8Co
- Crystal structure of 3-[(furan-2-ylmethyl)-amino]-2-(2,3,4,5-tetrafluoro-benzoyl)-acrylic acid ethyl ester, C17H13F4NO4
- Crystal structure of methyl 4-(2-ethoxy-2-oxoethoxy)-3-methoxybenzoate, C13H16O6
- Crystal structure of 4-bromo-2-(4-chlorophenyl)-1-methyl-5-(trifluoromethyl)-1H-pyrrole-3-carbonitrile, C13H7BrClF3N2
- The crystal structure of triaqua-(8-carboxymethoxy-quinoline-2-carboxylate-κ3N,O,O)nickel(II) monohydrate, C12H15NO9Ni
- Crystal structure of dihydroxy(2,4,6-triisopro-pylphenyl)telluronium trifluoromethanesulfonate, C16H25F3O5STe
- The crystal structure of 1-(carboxymethyl)-1H-imidazole 3-oxide
- The crystal structure of 1,3,5-tris(dibromomethyl)benzene, C9H6Br6
- Crystal structure of (Z)-3-(4-methoxyphenyl)-4-(5-methyl-1-phenyl-1H-1,2,3-triazol-4-yl)-N-phenylthiazol-2(3H)-imine, C25H21N5OS
- Crystal structure of (Z)-3-(3-(4-hydroxyphenyl)-2-(phenylimino)-2,3-dihydrothiazol-4-yl)-2H-chromen-2-one, C24H16N2O3S
Articles in the same Issue
- Frontmatter
- New Crystal Structures
- Crystal structure of poly[diaqua-(μ4-3,3′-di(1H-1,2,4-triazol-1-yl)-[1,1′-biphenyl]-4,4′-dicarboxylate-N:N′:O:O′)cadmium(II)], C18H14N6O6Cd
- Crystal structure of (8R,8′S,13S,13′R)-8,8′-bis(hydroxymethyl)-9,9′,10,10′-tetramethoxy-5,5′,6,6′,8,8′,13,13′-octahydro-[13,13′-bi[1,3]dioxolo[4,5-g]isoquinolino[3,2-a]isoquinoline]-7,7′-diium chloride-methanol (1/2), C46H58N2O14Cl2
- The crystal structure of 8-methoxy-2,2-diphenyl-tosyl-1,2-dihydro-2λ4,3λ4-[1,3,2]diazaborolo[4,5,1-ig]quinoline, C29H25BN2O3S
- Crystal structure of aqua-(5,5,7,12,12,14-hexamethyl-1,4,8,11-tetraazacyclotetradecane-κ4N,N′,N″,N‴)copper(II) 5-carboxyisophthalate tetrahydrate, C25H50N4CuO11
- The crystal structure of 1-(naphthalen-2-ylsulfonyl)-2,2-diphenyl-1,2-dihydro-2λ4,3λ4-[1,3,2]diazaborolo[4,5,1-ij]quinoline, C31H23BN2O2S
- Crystal structure of iodido-(η6-benzene) (1-(pyridin-2-yl)-N-(p-fluoro-methanamine)-κ2N,Nʹ)ruthenium(II) hexaflourophosphate, (C18H15F7IN2RuP)
- The crystal structure of 1-(3-oxo-1-phenyl-3-(p-tolyl) propylidene)-1,3-dihydro-2H-inden-2-one, C25H20O2
- Crystal structure of tricyclohexyl[4-(4H-1,2,4-triazol-4-yl)-benzoato-κO]tin(IV), C27H39N3O2Sn
- Crystal structure of [triaqua-(8-carboxymethoxy-quinoline-2-carboxylate-κ4N,O,O,O)cadmium(II)]monohydrate, C12H15NO9Cd
- Crystal structure of ethyl 2-((4-(3,5-dimethylisoxazol-4-yl)-2,6-difluorophenyl)amino)benzoate, C20H18F2N2O3
- The crystal structure of 2-(hydroxymethyl)-2-(4H-1,2,4-triazol-4-yl)propane-1,3-diol, C6H11N3O3
- The crystal structure of 1,2-bis(2,4-dinitrophenyl) hydrazine, C12H8N6O8
- Crystal structure of 1-(2,6-dichloro-4-(3,5-dimethylisoxazol-4-yl)phenyl)-1,2-dihydro-4H-benzo[d][1,3]oxazin-4-one, C19H14Cl2N2O3
- The crystal structure of 5-amino-5-oxo-4-(1-oxo-4-(2-oxopyrrolidin-1-yl)isoindolin-2-yl)pentanoic acid, C17H19N3O5
- Crystal structure of N2,N6-bis(2-(((Z)-5-bromo-2-hydroxybenzylidene)amino) phenyl)pyridine-2,6-dicarboxamide, C33H23Br2N5O4
- The crystal structure of (E)-2-methoxy-6-(((5-methyl-1,3,4-thiadiazol-2-yl)imino)methyl)phenol, C11H11N3O2S
- The crystal structure of 3-((tert-butyldiphenylsilyl)methyl)-5,5-diphenyl-6-(p-tolyl) tetrahydro-2H-pyran-2-one, C41H42O2Si
- Crystal structure of 9-fluoro-4-(6-methoxypyridin-3-yl)-5,6-dihydrobenzo[h]quinazolin-2-amine, C18H15FN4O
- The crystal structure of 2-bromo-5-(4-cyanophenoxy)benzyl 1-methyl-1,2,5,6-tetrahydropyridine-3-carboxylate, C21H19BrN2O3
- Crystal structure of 3,3′-(1,4-phenylenebis(methylene))bis(1-isopropyl-1H-imidazol-3-ium) bis(hexafluorophosphate(V)), C10H14F6N2P
- The crystal structure of 2,2-di(thiophen-3-yl)-1-tosyl-1,2-dihydro-2λ4,3λ4-[1,3,2]diazaborolo[4,5,1-ig]quinoline, C24H19BN2O2S3
- Crystal structure of 5-bromo-1-(2-iodobenzoyl)-1H-indole-3-carbaldehyde, C16H9BrINO2
- The crystal structure of monocarbonyl-2-carboxypyridinato-κ2N,O-triphenylphosphine-rhodium(I) acetonitrile solvate, C26H20.50N1.50O3PRh
- Crystal structure of dichlorido-tetrakis(1-(2,4-dichlorophenyl)-4,4-dimethyl-2-(1,2,4-triazol-1-yl)pent-1-en-3-ol-κ1N)manganese(II), C60H68O4N12Cl10Mn
- Crystal structure of 3-(tert-butyldiphenylsilyl)-1-(2,6-dichlorophenyl)-2,2-diphenylpropan-1-ol, C37H36Cl2OSi
- Crystal structure of langite from Mine du Pradet (France)
- The crystal structure of 5′-(furan-2-yl)-3′-((4-methylphenyl)sulfonamido)-3′,4′,5′,6′-tetrahydro-[1,1′:3′,1″-terphenyl]-4′-carboxylic acid, C30H27NO5S
- Synthesis and crystal structure of bis{2-(((4-acetophenone)imino)methyl)-4-fluorophenolato-κ2N,O}zinc(II), C30H22F2N2O4Zn
- The crystal structure of poly[(tripyridine-κ3N,N′,N″) μ3-(pyridine-3,4-dicarboxylate-κ3N:O:O′) manganese(II)], C22H22N4O8Mn
- The crystal structure of (E)-4-chloro-N′-(1-(4-hydroxyphenyl)propylidene)benzohydrazide, C16H15ClN2O2
- Synthesis and crystal structure of bis{2-(tert-butyl)-6-((E)-((4-((E)-1-(methoxyimino) ethyl)phenyl)imino)methyl)phenolato-κ2N,O}cobalt(II), C40H46CoN4O4
- Crystal structure of tetraaqua-[(1-(carboxymethyl)-1H-pyrazole-3-carboxylato-κ2N,O)cobalt(II)], C6H12CoN2O8
- (6R,7S)-2,3,13-trimethoxy-6,7-dimethyl-5,6,7,8-tetrahydrobenzo[3′,4′]cycloocta [1′,2′:4,5]benzo[1,2-d][1,3]dioxol-1-ol, C22H26O6
- Crystal structure of 2-((2,6-dichloro-4-(3,5-dimethylisoxazol-4-yl)phenyl)amino)benzoic acid, C18H14Cl2N2O3
- Crystal structure of (5aS,6aS,8aR,9R,11aS, 11bS,13R,13aS)-1,1,8a,11a-tetramethyl-9-((S)-1-((S)-5-methyl-6-oxo-3,6-dihydro-2H-pyran-2-yl)ethyl)-3-oxo-1,7,8,8a,9,10,11,11a,11b,12,13,13a-dodecahydro-3H,6H-cyclopenta[5,6]cyclopropa[1,8a]naphtho[2,1-c]oxepin-13-yl acetate, C32H44O6
- Crystal structure of catena-poly[triaqua-(μ2-1-(4-carboxylatophenyl)-4-oxo-1,4-dihydropyridazine-3-carboxylato-O,O′:O″)cobalt(II)], C12H12N2O8Co
- Crystal structure of 3-[(furan-2-ylmethyl)-amino]-2-(2,3,4,5-tetrafluoro-benzoyl)-acrylic acid ethyl ester, C17H13F4NO4
- Crystal structure of methyl 4-(2-ethoxy-2-oxoethoxy)-3-methoxybenzoate, C13H16O6
- Crystal structure of 4-bromo-2-(4-chlorophenyl)-1-methyl-5-(trifluoromethyl)-1H-pyrrole-3-carbonitrile, C13H7BrClF3N2
- The crystal structure of triaqua-(8-carboxymethoxy-quinoline-2-carboxylate-κ3N,O,O)nickel(II) monohydrate, C12H15NO9Ni
- Crystal structure of dihydroxy(2,4,6-triisopro-pylphenyl)telluronium trifluoromethanesulfonate, C16H25F3O5STe
- The crystal structure of 1-(carboxymethyl)-1H-imidazole 3-oxide
- The crystal structure of 1,3,5-tris(dibromomethyl)benzene, C9H6Br6
- Crystal structure of (Z)-3-(4-methoxyphenyl)-4-(5-methyl-1-phenyl-1H-1,2,3-triazol-4-yl)-N-phenylthiazol-2(3H)-imine, C25H21N5OS
- Crystal structure of (Z)-3-(3-(4-hydroxyphenyl)-2-(phenylimino)-2,3-dihydrothiazol-4-yl)-2H-chromen-2-one, C24H16N2O3S

