Abstract
In order to improve the alkali metals discharge capacity of slag, the gas-slag balance method was used to carry out the slag alkali metals discharge experiments, the effect of slag composition on alkali metals discharge performance of slag was studied, some suggestions were put forward to optimize the alkali metals discharge performance of slag and the extreme value model was established. The results show that the alkali metals discharge ratio of slag decreased with the increase in the binary basicity and mass fraction of TiO2, and increased with the increase in the mass fraction of MgO, Al2O3 and MnO. The change in slag composition led to the change in the solubility of alkali metal oxides in liquid slag, decomposition of alkali metal silicates, structure of the slag in liquid state and viscosity of the slag, and then affected the alkali metals discharge performance of slag. The ability of slag to absorb alkali metals was certain under the condition of fixed composition. With the help of slag alkali metals discharge extreme value model, whether the current slag meets the needs of blast furnace alkali metals discharge could be evaluated. The alkali metals discharge capacity of slag could be improved by optimizing the alkali metals discharge performance of slag combined with experiments and actual production.
1 Introduction
Greening and intelligent are the inevitable requirements for the transformation and upgrading of iron and steel industry, which will promote the development of blast furnace (BF) in the direction of large scale and refinement [1,2,3]. The large scale and refinement BF puts forward higher standards for the quality of raw materials and fuels, especially for the control of harmful elements (alkali metals, lead, zinc, sulfur and arsenic) in raw materials and fuels, there is an upper limit of furnace load [4,5,6,7,8].
Alkali metals in raw materials and fuels not only worsens the low temperature reduction degradation property of sinter and pellet, but also affects the coke melting loss reaction and reduces the post reaction strength of coke, so the amount of powder in BF increases, the permeability of BF decreases and the differential pressure of BF increases [9,10,11,12]. There is a phenomenon of “cyclic enrichment” after alkali metals enter BF, it will form “nodules” on the hot surface of the furnace lining after enrichment to a certain extent, and change the operation furnace profile of BF [13]. Alkali metals steam can even enter the refractory of BF [14,15,16,17,18,19], making the refractory brittle and spalling, which seriously affects the service life of BF [20,21,22,23,24,25].
In this study, the optimization of slag alkali metals discharge based on experiments was carried out to improve the alkali metals discharge capacity of slag, and the extreme value model was established to evaluate the alkali metals discharge capacity of slag. The slag alkali metals discharge experiments were designed by using the gas-slag balance method to explore the influence law of basicity and composition content on alkali metals discharge ratio of slag, and clarify the effect of basicity and composition on alkali metals discharge performance of slag. The optimization scheme of alkali metals discharge performance of slag was proposed based on the analysis of the experimental results. The extreme value model of slag alkali metals discharge was established to calculate the current extreme value of BF slag alkali metals discharge.
2 Experimental
2.1 Samples preparation and experimental setup
The mass fraction of actual slag composition in an ironmaking plant was taken as the benchmark for slag alkali metals discharge experiments. It was prepared with CaO, MgO, Al2O3, SiO2, MnO and TiO2 pure chemical reagent powder. Among them, alkali metals were mixed into slag in the form of K2CO3 and Na2CO3. Q6 The slag sample was 50 g, 3.10 g for K2CO3 and 5.40 g for Na2CO3. The weighed sample powder was poured into a mortar for mixing, and the mixed powder was packed into a high-purity graphite crucible (Ф64 mm × 90 mm). The mass fraction of the experimental slag is shown in Table 1. The vertical tubular furnace was used for slag alkali metals discharge experiments, the inside of the tubular furnace was a corundum tube, U-shaped silicon molybdenum rod was employed as the heating unit and the insulation material is alumina refractory fiber, and the maximum temperature can reach 1,600°C.
The mass fraction of slag sample
| Number | CaO | SiO2 | MgO | Al2O3 | MnO | TiO2 | Basicity |
|---|---|---|---|---|---|---|---|
| A-1 | 36.62 | 39.38 | 8.50 | 15.50 | 0.00 | 0.00 | 0.93 |
| A-2 | 37.81 | 38.19 | 8.50 | 15.50 | 0.00 | 0.00 | 0.99 |
| A-3 | 38.93 | 37.07 | 8.50 | 15.50 | 0.00 | 0.00 | 1.05 |
| A-4 | 39.98 | 36.02 | 8.50 | 15.50 | 0.00 | 0.00 | 1.11 |
| A-5 | 40.98 | 35.02 | 8.50 | 15.50 | 0.00 | 0.00 | 1.17 |
| A-6 | 41.92 | 34.08 | 8.50 | 15.50 | 0.00 | 0.00 | 1.23 |
| B-1 | 40.51 | 36.49 | 7.50 | 15.50 | 0.00 | 0.00 | 1.11 |
| B-3 | 39.45 | 35.55 | 9.50 | 15.50 | 0.00 | 0.00 | 1.11 |
| B-4 | 38.93 | 35.07 | 10.50 | 15.50 | 0.00 | 0.00 | 1.11 |
| B-5 | 38.40 | 34.60 | 11.50 | 15.50 | 0.00 | 0.00 | 1.11 |
| C-1 | 40.51 | 36.49 | 8.50 | 14.50 | 0.00 | 0.00 | 1.11 |
| C-2 | 40.24 | 36.26 | 8.50 | 15.00 | 0.00 | 0.00 | 1.11 |
| C-4 | 39.72 | 35.78 | 8.50 | 16.00 | 0.00 | 0.00 | 1.11 |
| C-5 | 39.45 | 35.55 | 8.50 | 16.50 | 0.00 | 0.00 | 1.11 |
| D-1 | 39.88 | 35.92 | 8.50 | 15.50 | 0.20 | 0.00 | 1.11 |
| D-2 | 39.77 | 35.83 | 8.50 | 15.50 | 0.40 | 0.00 | 1.11 |
| D-3 | 39.67 | 35.73 | 8.50 | 15.50 | 0.60 | 0.00 | 1.11 |
| D-4 | 39.56 | 35.64 | 8.50 | 15.50 | 0.80 | 0.00 | 1.11 |
| D-5 | 39.45 | 35.55 | 8.50 | 15.50 | 1.00 | 0.00 | 1.11 |
| E-1 | 39.45 | 35.55 | 8.50 | 15.50 | 0.00 | 1.00 | 1.11 |
| E-2 | 38.93 | 35.07 | 8.50 | 15.50 | 0.00 | 2.00 | 1.11 |
| E-3 | 38.40 | 34.60 | 8.50 | 15.50 | 0.00 | 3.00 | 1.11 |
| E-4 | 37.88 | 34.12 | 8.50 | 15.50 | 0.00 | 4.00 | 1.11 |
| E-5 | 37.35 | 33.65 | 8.50 | 15.50 | 0.00 | 5.00 | 1.11 |
Note: Bold values mainly represent the parameters changed in the experiment.
2.2 Experimental procedure
The gas-slag (alkali metals steam and slag) balance method was employed to carry out the slag alkali metals discharge experiments. The high-purity graphite crucible containing the mixed powder sample was put into the tubular furnace, the upper part of the graphite crucible was covered with a graphite cover, which could inhibit the discharge of K and Na steam at high temperature, so that the slag could fully contact and react with alkali metals. Then, the tubular furnace was covered, and high-purity argon gas was introduced into the furnace with a flow rate of 3 L·min−1. The FP93 temperature control meter started to heat up after the temperature rise program was set. When the temperature rose to 1,500°C (the actual slag iron temperature in hearth, the highest liquidus temperature in the samples is 1,442°C), the temperature was kept for 2 h to make the slag composition uniform and fully react with alkali metals. After the reaction, the graphite crucible was taken out with crucible pliers and put into water for quenching, to keep the alkali metals content of slag in equilibrium. Then, the cooled slag sample was ground into powder, the mass fractions of K2O and Na2O in each slag sample were detected through chemical analysis under different experimental conditions, the alkali metals discharge ratio of slag was calculated to determine the alkali metals discharge capacity of slag under different conditions.
3 Results
3.1 Micro morphology of slag after reaction
After the reaction, the cooled slag sample was cut (as presented in Figure 1), the cross section of the sample was inlaid with resin into electron microscope samples and the micro morphology of the slag was analyzed by scanning electron microscope-energy dispersive spectrometer (SEM-EDS). As shown in Figure 2(a), holes of different sizes were found in the slag near the surface layer. According to the EDS results (Figure 2(b)), the mass fractions of K and Na were 3.66 and 5.34% in the hole and its surrounding area, respectively, which proved that the alkali metals once stayed in the hole. The hole presented a horn shape with large outer side and small inner side (Figure 2(c) and (d)), it was speculated that it might be caused by the separation of alkali metals steam from slag during high temperature experiment. Figure 3 shows the EDS results of the cross section of slag sample after reaction, it was clear from the figure that during the high temperature experiment, K and Na had been distributed throughout the slag after the experiment. The alkali metals steam that could not be absorbed would float up on the surface of the liquid slag and separated from the slag. This proved that the alkali metals discharge experiment of gas-slag balance method can evaluate the alkali metals discharge capacity of slag.

Macro morphology of slag after reaction.

Micro morphology of slag after reaction. (a) Holes of different sizes; (b) EDS results of point A; (c) horn shaped hole; (d) internal morphology of the hole.

EDS results of the cross section of slag sample after reaction. (a) Morphology of slag cross section; (b) EDS result of Ca; (c) EDS result of K; (d) EDS result of Na.
3.2 Influence of various factors on alkali metals discharge ratio of slag
The morphology of the slag with different binary basicity after reaction is shown in Figure 4. It is clear from the figure that when binary basicity was 0.93, the color of the slag was yellow and the surface was uneven after reaction, which proved that the slag had absorbed a large amount of the alkali metals. The sample with higher binary basicity had darker color and less alkali metals absorption. In addition, when binary basicity was 1.23, the slag had insufficient strength after cooling and was broken into small pieces. It shows that the slag might not be able to absorb so much of alkali steam at this time, and the alkali metals steam overflowed from the slag during the experiment, resulting in a large number of holes left in the slag after cooling, enabling the slag sample to break easily.

Morphology of slag with different binary basicity after reaction. (a) R = 0.93; (b) R = 0.99; (c) R = 1.05; (d) R = 1.11; (e) R = 1.17; (f) R = 1.23.
The alkali metals discharge ratio of slag can be calculated as follows:
where r is the alkali metals discharge ratio of slag, %;
The influence of binary basicity on slag alkali metals discharge is shown in Figure 5. When the binary basicity of slag increased from 0.93 to 1.23, the alkali metals discharge ratio of slag decreased from 85.03 to 68.16%. The alkali metals discharge ratio of slag generally showed a downward trend. The mass fraction of alkali metals in the slag decreased with the increase in the binary basicity, that is, the alkali metals discharge capacity of the slag decreased with the increase in the binary basicity. When the binary basicity increased from 0.99 to 1.11, the alkali metals discharge ratio of the slag decreased from 85.03 to 73.04%, the degree of decline was obvious. However, when the binary basicity was 1.17, the decrease in the slag alkali metals discharge ratio was small.

Effect of binary basicity on slag alkali metals discharge.
As shown in Figure 6, when the mass fraction of MgO increased from 7.5 to 11.5%, the alkali metals discharge ratio of the slag increased from 65.05 to 76.59%. It showed an upward trend, the alkali metals discharge capacity of the slag increased with the increase in the MgO mass fraction in the slag.
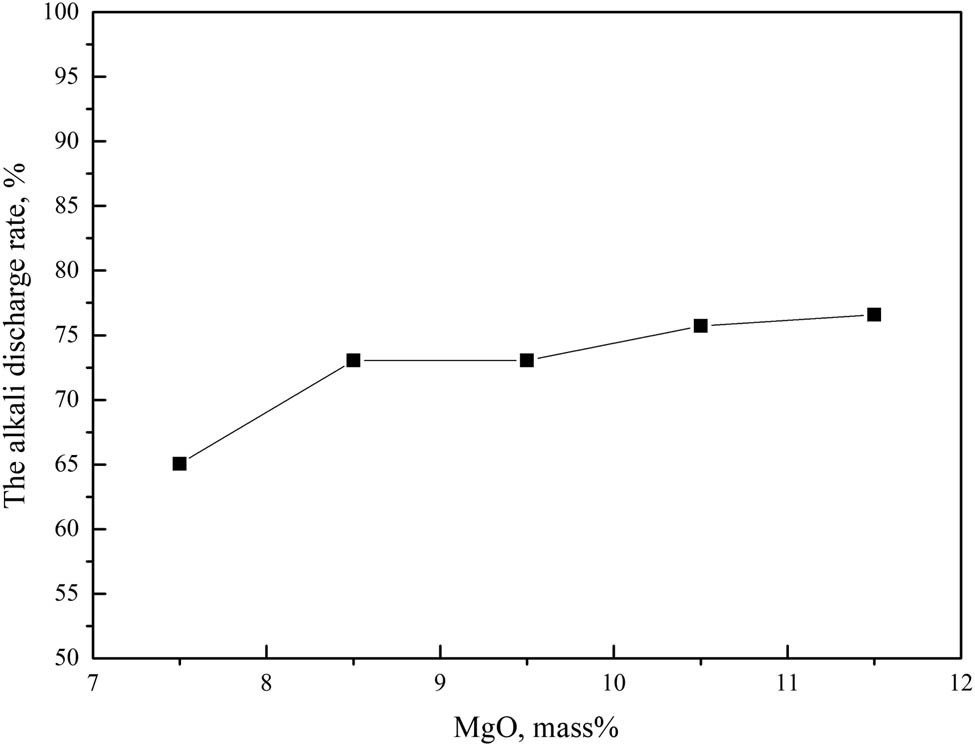
Effect of MgO on slag alkali metals discharge.
Figure 7 depicts the effect of Al2O3 on the slag alkali metals discharge. When Al2O3 in the slag increased from 14.5 to 16.5%, the alkali metals discharge ratio of the slag increased from 62.72 to 73.15%. The mass fraction of alkali metals in the slag increased with the increase in Al2O3 mass fraction in the slag, the increase in Al2O3 could improve the alkali metals discharge capacity of the slag.

Effect of Al2O3 on slag alkali metals discharge.
It can be seen from Figure 8 that with the increase in MnO mass fraction in the slag, the alkali metals discharge ratio of the slag showed a slow upward trend. When the mass fraction of MnO increased from 0.2 to 0.6%, the alkali metals discharge ratio of slag increased from 73.26 to 73.60%, the alkali metals discharge capacity of the slag changed a little. When the mass fraction of MnO increased from 0.6 to 1.0%, the alkali metals discharge ratio of the slag increased from 73.60 to 77.48%, the alkali metals discharge capacity of the slag increased slowly.
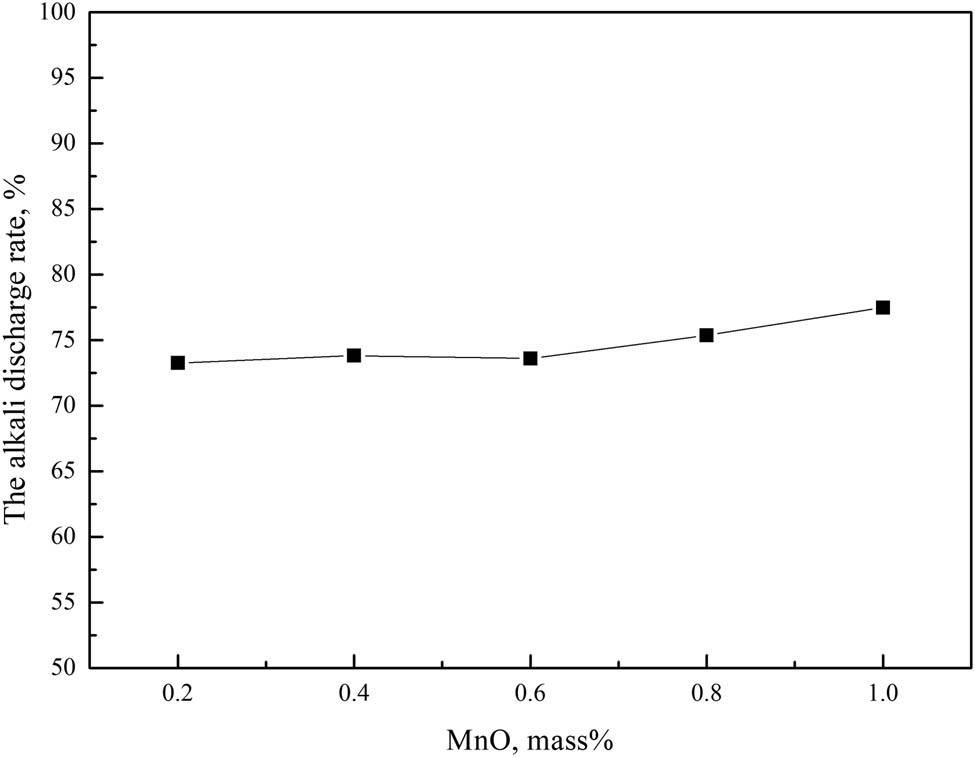
Effect of MnO on slag alkali metals discharge.
As shown in Figure 9, the alkali metals discharge ratio of the slag showed a downward trend with the increase in TiO2 mass fraction in the slag, the alkali metals discharge capacity of the BF slag decreased with the increase in TiO2 mass fraction in the slag. However, the degree of decline was different in different TiO2 mass fraction ranges. When the mass fraction of TiO2 was less than 3.0%, the alkali metals discharge ratio decreased from 85.14 to 83.37%, the decline was small. While, when the mass fraction of TiO2 was greater than 3.0%, the alkali metals discharge ratio decreased from 83.37 to 71.04%, the decline was large.
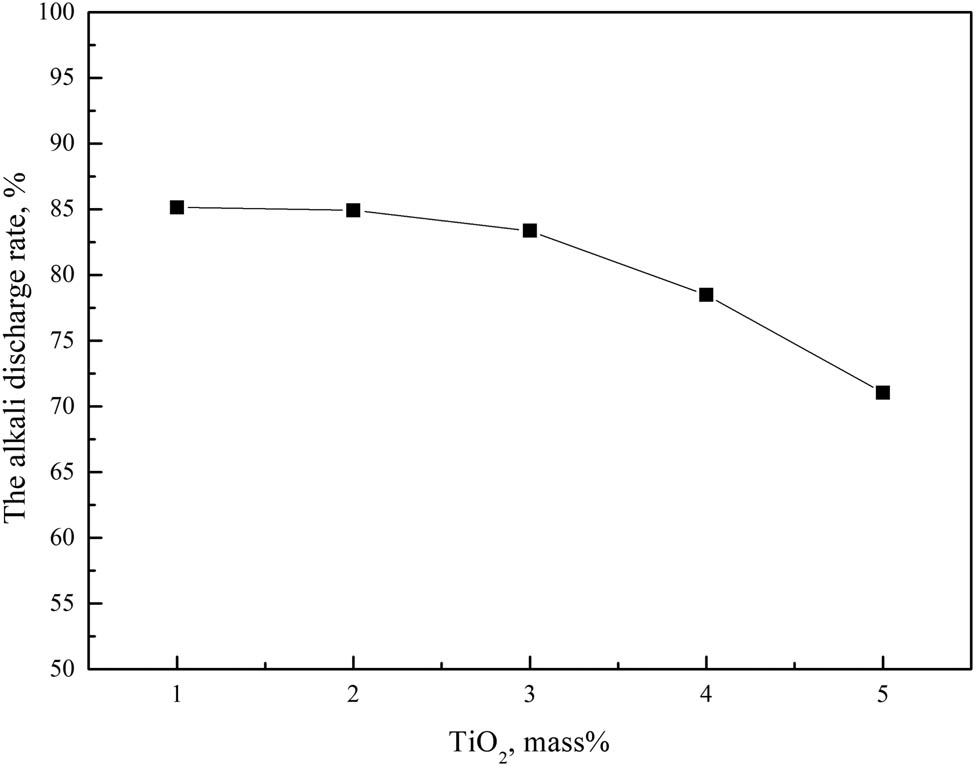
Effect of TiO2 on slag alkali metals discharge.
4 Discussion
4.1 Influence mechanism of slag composition on alkali metals discharge performance
Binary basicity. The alkali metals discharge capacity of the slag decreased with the increase in binary basicity. The mechanism: (1) The solubility of alkali metal oxides in the liquid slag decreased with the increase in binary basicity, so the alkali metals absorption capacity of the slag decreased after the increase in binary basicity. (2) Alkali metals were dissolved in the liquid slag and existed in the form of silicate, the following reactions occurred [26]: K2SiO3 + CaO + C = CaO·SiO2 + CO(g) + 2 K(g). The mass fraction of CaO in the slag also increased with the increase in binary basicity, which would promote the reaction to the right, accelerate the decomposition of alkali metals silicate and reduce the alkali metals absorption capacity of the slag. When the binary basicity was 1.17, the decrease in the slag alkali metals discharge ratio was small, which might be related to the change in slag viscosity. In a certain range, the complex anion groups in the slag were disintegrated into simple ion units to reduce the viscosity after the binary basicity increased. The kinetics of alkali metals discharge chemical reaction were improved, this was conducive to the alkali metals discharge reaction, and some adverse effects on alkali metals discharge were offset. However, when the binary basicity was too high (1.23), heterogeneous high melting point compounds such as 2CaO·SiO2 were formed in the slag, which increased the viscosity and further worsened the alkali metals discharge capacity of the slag. Therefore, excessive slag viscosity was unfavorable to alkali metals discharge.
MgO. The alkali metals discharge capacity of the slag increased with the increase in MgO mass fraction in the slag. The mechanism: (1) MgO could improve the solubility of alkali metals oxides in the liquid slag [27]. (2) The existence of MgO increased the free oxygen ion O2− in the liquid slag, the complex network ion structure and heterogeneous high melting point compounds in the slag were disintegrated. The structure of slag in the liquid state was simplified, so the viscosity of the slag was reduced. The components had greater diffusion kinetic energy in the liquid slag after the slag viscosity reduced, the kinetics of alkali metals discharge chemical reaction was improved, so the alkali metals discharge capacity of the slag was increased.
Al 2 O 3 . The increase in Al2O3 mass fraction could improve the alkali metals discharge capacity of the slag. The mechanism: (1) Al2O3 could form nepheline with high melting point (such as KAlSiO4) with SiO2 and alkali metals in the slag, so that part of the alkali metals could be solidified in the slag [28,29]. (2) Al2O3 could form CaO·Al2O3 with CaO, reducing the mass fraction of free CaO and inhibiting the decomposition of alkali metal silicates, which made more alkali metals stay in the slag, so as to increase the alkali metals discharge capacity of the slag.
MnO. The alkali metals discharge ratio of the slag increased slowly with the increase in MnO mass fraction in the slag. The mechanism was mainly because MnO reduced the viscosity of the slag: (1) MnO could separate the free oxygen ion O2− from the slag, the free oxygen ion dissociated the complex network structure of the slag into a simple structure and reduced the viscosity of the slag. (2) MnO and SiO2 formed manganese olivine (2MnO·SiO2) with low melting point in the slag, the superheat degree of the slag was increased, the force between the components in the liquid slag was reduced, so the viscosity of the slag was reduced. (3) The reduction of some oxides in the slag would release some CO gas, and the alkali metals steam would overflow from the slag with CO. When the mass fraction of MnO was high (higher than 0.06%), it could inhibit the reduction of some oxides: 2MnO + Ti = 2Mn + TiO2, the foaming probability in the liquid slag was reduced [30,31]. The overflow of some alkali metals steam was restrained, so the alkali metals discharge capacity of the slag was improved.
TiO 2 . The alkali metals discharge ratio of the slag showed a downward trend with the increase in TiO2 content in the slag. The mechanism: (1) The acidity of TiO2 in the slag was weaker than that of SiO2, which was only about three fifths of that of SiO2. When the mass fraction of TiO2 increased, the acidity of the slag was reduced, so the alkali metals discharge capacity was relatively reduced. (2) Some alkali metals could be dissolved by high melting point compound (perovskite) and low melting point compound (diopside) formed by TiO2 in the slag. However, TiO2 could precipitate solid particles such as TiC in the slag under high temperature and carbon containing conditions [32]. Once solid particles appeared in the liquid slag, the properties of Newtonian fluid would be changed and the viscosity of the slag would increase rapidly, so the kinetic conditions of alkali metals discharge chemical reaction were deteriorated, this effect completely masked the solid solution. In particular, when the mass fraction of TiO2 was high (higher than 3%), the viscosity increased greatly, so the alkali metals discharge capacity of the slag decreased significantly.
4.2 Optimization of slag alkali metals discharge performance
The alkali metals discharge performance of the slag was optimized based on the experimental results and theoretical analysis.
The alkali metals discharge capacity of the slag decreased with the increase in binary basicity under the condition of experimental slag system, the alkali metals discharge ratio of slag decreased little when the binary basicity was 1.17. Therefore, in the actual production process, on the premise of ensuring the production of qualified pig iron, it is suggested that the binary basicity should be controlled at about 1.17, considering desulfurization, alkali metals discharge and viscosity.
The alkali metals discharge capacity of the slag increased with the increase in MgO mass fraction in the slag. The mass fraction of MgO in the slag should not exceed 12%, in order to ensure the smooth operation of the BF, prevent the formation of high melting point periclase and spinel in the slag, deteriorate the fluidity of the slag and worsen the dynamic conditions of the reaction in the furnace. The mass fraction of MgO in the BF slag is recommended to be controlled at 7.5–10.5% according to the actual slag system.
The alkali metals discharge ratio of the slag increased with the increase in Al2O3 mass fraction in the slag; however, the slag viscosity increased with the increase in Al2O3 mass fraction. Therefore, it is suggested that in the actual production process of BF, slag desulfurization, slag alkali metals discharge and slag viscosity should be comprehensively considered, and the mass fraction of Al2O3 should be kept below 16%.
The alkali metals discharge ratio of the slag could be improved by increasing the mass fraction of MnO in the slag. However, the fluidity of the slag would increase with the high MnO content in it, which was unfavorable to the long service life of the hearth. Therefore, it is suggested that the mass fraction of MnO in slag should be controlled below 0.6%.
The alkali metals discharge capacity of the slag decreased with the increase in TiO2 content in the slag. When the mass fraction of TiO2 in the slag was high, solid particles such as TiC were precipitated in the slag, which made the viscosity of the slag increase rapidly. Therefore, it is recommended to reduce the mass fraction of TiO2 in the slag as much as possible, during normal production of BF. Subsequently, the mass fraction of TiO2 in the slag shall be appropriately increased according to the need of corrosion resistance and hearth protection.
4.3 Extreme value model of slag alkali metals discharge
BF slag itself has the ability to discharge alkali metals, but under the condition of fixed composition, the ability of the slag system to absorb alkali metals is limited. If the alkali metals load in the furnace exceeds the alkali metals discharge capacity of the slag system, the alkali metals will be “recycled and enriched” in the BF, which will have a series of adverse effects on the BF. Therefore, it is necessary to study the extreme value model of slag alkali metals discharge. The model is used to calculate the current extreme value of BF slag alkali metals discharge, evaluate the current situation of BF alkali metals discharge and judge whether the current slag can meet the needs of BF alkali metals discharge.
First, the mass fractions of K2O and Na2O in the slag are detected under the condition of existing slag composition. Take the highest value of the sum of different alkali metals’ mass fractions:
where w max is the highest value of total mass fraction of the slag alkali metals, %; and w(j) is the mass fraction of components in the slag, %.
The extreme value of alkali metals discharged with the slag is calculated under the condition of existing slag composition:
where m max is the extreme value of alkali metals discharged with the slag, kg·t−1; and R is the slag ratio of the BF, kg·t−1.
The alkali metals load of the BF is calculated according to the existing raw material conditions:
where L is the alkali metals load of the BF, kg·tHM−1; m i is the mass of different kinds of raw materials and fuels in the BF, kg·tHM−1; and w i (j) is the alkali metals content in different kinds of raw materials and fuels, %.
The alkali metals load of the BF is compared with the extreme value of the slag alkali metals discharge, if L < m max, the BF slag can meet the needs of the alkali metals discharge; if L > m max, the BF slag under the existing conditions cannot complete the alkali metals discharge task, and it will produce “cyclic enrichment” in the BF. The existing slag composition needs to be adjusted to meet the needs of alkali metals discharge. The slag composition can be adjusted in the laboratory according to the slag alkali metals discharge performance optimization scheme. The mass fraction of alkali metals in the slag is determined by the gas-slag balance experimental method in this article, and the extreme value of alkali metals discharge is calculated. If the experimental slag can meet the needs of alkali metals discharge, it can be put into practice in BF production. If the experimental slag still does not meet the alkali metals discharge needs, continue to adjust until it can meet the alkali metals discharge needs of the BF. The adjusted technical route is presented in Figure 10.

Technical route for adjusting slag alkali metals discharge performance.
5 Conclusion
The influence direction and range of various factors on the alkali metals discharge ratio of the slag were different. The alkali metals discharge ratio of the slag decreased with the increase in binary basicity and mass fraction of TiO2. The alkali metals discharge ratio of the slag showed an upward trend with the increase in mass fraction of MgO, Al2O3 and MnO.
The influence mechanism of slag composition on alkali metals discharge ratio: the alkali metals discharge performance of the slag was mainly affected by the solubility of alkali metal oxides in the liquid slag, the decomposition of alkali metal silicates, structure of slag in the liquid state and the viscosity of the slag.
In actual production, the ability of the slag system to absorb alkali metals was limited when the composition was fixed. With the help of the extreme value model of the slag alkali metals discharge, whether the current slag can meet the needs of the BF alkali metals discharge could be evaluated. The alkali metals discharge capacity of the slag could be improved by optimizing the alkali metals discharge performance of the slag combined with experiments and actual production.
-
Funding information: The project was supported by Open Fund of The State Key Laboratory of Refractories and Metallurgy (G202005), Natural Science Foundation–Steel and Iron Foundation of Hebei Province (E2020209069, E2020209208).
-
Author contributions: Yong Deng contributed to the conception of the study; Yan-bo Chen, Li-da Chen performed the experiment; Ran Liu, Xing-min Guo helped perform the analysis with constructive discussions.
-
Conflict of interest: Authors state no conflict of interest.
References
[1] Li, X. C. Road map to high-quality development of iron and steel industry in new age. Iron and Steel, Vol. 54, No. 1, 2019, pp. 1–7.Search in Google Scholar
[2] Xiao, P. Innovation practice and future prospects of blast furnace ironmaking technology. Iron and Steel, Vol. 56, No. 6, 2021, pp. 10–14, 34.Search in Google Scholar
[3] Yang, T. J., J. L. Zhang, Z. J. Liu, and K. J. Li. Development of ironmaking industry at the new situation. Ironmaking, Vol. 39, No. 5, 2010, pp. 1–9.Search in Google Scholar
[4] Zhang, S. Z., D. H. Gao, P. F. Wang, and G. P. Luo. Balance and control of potassium and sodium in No. 6 blast furnace of Baotou steel. China Metallurgy, Vol. 29, No. 3, 2019, pp. 28–31.Search in Google Scholar
[5] Liu, W. W., Y. H. Qi, K. X. Jiao, and H. K. Li. Balance and control of alkali metal in No.6 blast furnace of Tisco. China Metallurgy, Vol. 30, No. 12, 2020, pp. 72–76.Search in Google Scholar
[6] Zheng, P. C., J. L. Zhang, Z. J. Liu, and Y. B. Chen. Effect of alkali metals on thermal properties of coke. China Metallurgy, Vol. 27, No. 5, 2017, pp. 19–28.Search in Google Scholar
[7] Gan, M. Y. and Z. H. Tang. Control practice of harmful elements in blast furnace of Liugang. Ironmaking, Vol. 39, No. 4, 2020, pp. 35–38.Search in Google Scholar
[8] Si, J. C. and H. Y. Wei. Control practice of harmful elements in blast furnace of Liugang. Ironmaking, Vol. 35, No. 1, 2016, pp. 54–57.Search in Google Scholar
[9] Jiao, K. X., J. L. Zhang, Z. J. Liu, C. L. Chen, and Y. X. Liu. Analysis of blast furnace hearth sidewall erosion and protective layer formation. ISIJ International, Vol. 56, No. 11, 2016, pp. 1956–1963.10.2355/isijinternational.ISIJINT-2016-168Search in Google Scholar
[10] Dastidar, M. G., A. Bhattacharyya, B. K. Sarkar, D. Rajib, M. K. Mitra, and J. Schenk. The effect of alkali on the reaction kinetics and strength of blast furnace coke. Fuel, Vol. 268, 2020, id. 117388.10.1016/j.fuel.2020.117388Search in Google Scholar
[11] Wang, G. Influence of alkali metals on metallurgical properties of raw materials in blast furnace. Shanxi Metallurgy, Vol. 41, No. 5, 2018, pp. 17–19.Search in Google Scholar
[12] Xu, L. Effect of alkali metals on metallurgical properties of blast furnace raw materials. China Steel Focus, Vol. No. 15, 2019, pp. 15–16.Search in Google Scholar
[13] Xia, Z. H., Y. Sun, L. Chen, and J. W. Ge. Lump detection of No. 2 blast furnace in Shagang and recovery of furnace condition. Ironmaking, Vol. 36, No. 3, 2017, pp. 47–48.Search in Google Scholar
[14] Wang, T. S., Y. W. Li, S. B. Sang, Y. B. Xu, and H. Wang. Effect of pitch powder addition on the microstructure and properties of carbon blocks for blast furnace. Ceramics International, Vol. 45, No. 1, 2019, pp. 634–643.10.1016/j.ceramint.2018.09.221Search in Google Scholar
[15] Dai, Y. J., Y. C. Yin, X. F. Xu, S. L. Jin, Y. W. Li, and H. Harmuth. Effect of the phase transformation on fracture behaviour of fused silica refractories. Journal of the European Ceramic Society, Vol. 38, No. 16, 2018, pp. 5601–5609.10.1016/j.jeurceramsoc.2018.08.040Search in Google Scholar
[16] Dai, Y. J., Y. W. Li, X. F. Xu, Q. Y. Zhu, W. Yan, S. L. Jin, et al. Fracture behaviour of magnesia refractory materials in tension with the Brazilian test. Journal of the European Ceramic Society, Vol. 39, No. 16, 2019, pp. 5433–5441.10.1016/j.jeurceramsoc.2019.07.026Search in Google Scholar
[17] Dai, Y. J., Y. W. Li, S. L. Jin, H. Harmuth, Y. Wen, and X. F. Xu. Mechanical and fracture investigation of magnesia refractories with acoustic emission-based method. Journal of the European Ceramic Society, Vol. 40, No. 1, 2020, pp. 181–191.10.1016/j.jeurceramsoc.2019.09.010Search in Google Scholar
[18] Cheng, Y., T. B. Zhu, Y. W. Li, S. B. Sang, N. Liao, Z. P. Xie, et al. Microstructure and mechanical properties of oscillatory pressure sintered WC ceramics with different carbon sources. Ceramics International, Vol. 47, No. 8, 2021, pp. 11793–11798.10.1016/j.ceramint.2021.01.020Search in Google Scholar
[19] Chen, Q. L., T. B. Zhu, Y. W. Li, Y. Chen, N. Liao, L. P. Pan, et al. Enhanced performance of low-carbon MgO–C refractories with nano-sized ZrO2–Al2O3 composite powder. Ceramics International, Vol. 47, No. 14, 2021, pp. 20178–20186.10.1016/j.ceramint.2021.04.024Search in Google Scholar
[20] Peng, W. D., Z. Chen, W. Yan, S. Schafföner, G. Q. Li, Y. W. Li, et al. Advanced lightweight periclase-magnesium aluminate spinel refractories with high mechanical properties and high corrosion resistance. Construction and Building Materials, Vol. 291, No. 11, 2021, id. 123388.10.1016/j.conbuildmat.2021.123388Search in Google Scholar
[21] Xu, X. F., Y. W. Li, Y. J. Dai, T. B. Zhu, L. P. Pan, and J. Szczerba. Influence of graphite content on fracture behavior of MgO–C refractories based on wedge splitting test with digital image correlation method and acoustic emission. Ceramics International, Vol. 47, No. 9, 2021, pp. 12742–12752.10.1016/j.ceramint.2021.01.134Search in Google Scholar
[22] Liu, G. F., N. Liao, M. Nath, Y. W. Li, and S. B. Sang. Optimized mechanical properties and oxidation resistance of low carbon Al2O3-C refractories through Ti3AlC2 addition. Journal of the European Ceramic Society, Vol. 41, No. 4, 2021, pp. 2948–2957.10.1016/j.jeurceramsoc.2020.11.038Search in Google Scholar
[23] Chang, Q. M., X. W. Li, H. W. Ni, W. Y. Zhu, C. G. Pan, and S. D. Hu. Modeling on dry centrifugal granulation process of molten blast furnace slag. ISIJ International, Vol. 55, No. 7, 2015, pp. 1361–1366.10.2355/isijinternational.55.1361Search in Google Scholar
[24] Wang, Q. H., G. He, S. X. Deng, J. Liu, X. Y. Li, J. Q. Li, et al. Wetting behavior and reaction mechanism of molten Si in contact with silica substrate. Ceramics International, Vol. 45, No. 17, 2019, pp. 21365–21372.10.1016/j.ceramint.2019.07.123Search in Google Scholar
[25] Wang, T. S., S. B. Sang, and Y. W. Li. Effect of artificial graphite and nickel nitrate on the microstructure and properties of carbon blocks for blast furnace. Key Engineering Materials, Vol. 768, No. 4, 2018, pp. 267–273.10.4028/www.scientific.net/KEM.768.267Search in Google Scholar
[26] Lyu, Q., F. M. Li, L. N. Gu, and D. H. Hou. Experimental Investigation on dealkalization and desulphurization of Alkaliferous BF slag. Journal of Northeastern University, Vol. 28, No. 11, 2007, pp. 1590–1593.Search in Google Scholar
[27] Wang, Y. C., S. R. Na, C. Y. Chen, and R. J. Wang. Experimental Investigation on improving the capacity of removing Alkali metals of baogang blast furnace slag. Journal of Inner Mongolia University of Science and Technology, Vol. 20, No. 2, 2001, pp. 104–106, 117.Search in Google Scholar
[28] Peng, Q. C., B. Q. Chen, J. Peng, and L. M. Zhao. Discussion on alkali discharge of high Al2O3 BF slag in Xiangtan iron & steel corp. Research on Iron & Steel, Vol. 34, No. 3, 2006, pp. 10–14.Search in Google Scholar
[29] Zhu, G. Y., J. L. Zhang, R. Mao, and X. Yuan. Orthogonal experimental study on removal of Alkalis through blast furnace slag. Research on Iron & Steel, Vol. 41, No. 6, 2013, pp. 19–23.Search in Google Scholar
[30] Qv, Y. P. and H. G. Du. Effect of MnO on foaming behavior of high titanium blast furnace slag. Journal of Northeastern University, Vol. 23, No. 8, 2002, pp. 769–772.Search in Google Scholar
[31] Li, Y. H., T. P. Lou, and Z. T. Sui. Effects of CaO and MnO on the crystallization of the Perovskite phase in the Ti-bearing blast furnace slag. Journal of Iron and Steel Research, Vol. 12, No. 3, 2000, pp. 1–4.Search in Google Scholar
[32] Chen, G. Y., J. L. Kang, S. J. Wu, L. X. Liu, F. Zhang, J. Peng, et al. Effect of TiO2 on slag viscosity. Journal of Inner Mongolia University of Science and Technology, Vol. 37, No. 4, 2018, pp. 338–342, 372.Search in Google Scholar
© 2022 Yan-bo Chen et al., published by De Gruyter
This work is licensed under the Creative Commons Attribution 4.0 International License.
Articles in the same Issue
- Research Articles
- Numerical and experimental research on solidification of T2 copper alloy during the twin-roll casting
- Discrete probability model-based method for recognition of multicomponent combustible gas explosion hazard sources
- Dephosphorization kinetics of high-P-containing reduced iron produced from oolitic hematite ore
- In-phase thermomechanical fatigue studies on P92 steel with different hold time
- Effect of the weld parameter strategy on mechanical properties of double-sided laser-welded 2195 Al–Li alloy joints with filler wire
- The precipitation behavior of second phase in high titanium microalloyed steels and its effect on microstructure and properties of steel
- Development of a huge hybrid 3D-printer based on fused deposition modeling (FDM) incorporated with computer numerical control (CNC) machining for industrial applications
- Effect of different welding procedures on microstructure and mechanical property of TA15 titanium alloy joint
- Single-source-precursor synthesis and characterization of SiAlC(O) ceramics from a hyperbranched polyaluminocarbosilane
- Carbothermal reduction of red mud for iron extraction and sodium removal
- Reduction swelling mechanism of hematite fluxed briquettes
- Effect of in situ observation of cooling rates on acicular ferrite nucleation
- Corrosion behavior of WC–Co coating by plasma transferred arc on EH40 steel in low-temperature
- Study on the thermodynamic stability and evolution of inclusions in Al–Ti deoxidized steel
- Application on oxidation behavior of metallic copper in fire investigation
- Microstructural study of concrete performance after exposure to elevated temperatures via considering C–S–H nanostructure changes
- Prediction model of interfacial heat transfer coefficient changing with time and ingot diameter
- Design, fabrication, and testing of CVI-SiC/SiC turbine blisk under different load spectrums at elevated temperature
- Promoting of metallurgical bonding by ultrasonic insert process in steel–aluminum bimetallic castings
- Pre-reduction of carbon-containing pellets of high chromium vanadium–titanium magnetite at different temperatures
- Optimization of alkali metals discharge performance of blast furnace slag and its extreme value model
- Smelting high purity 55SiCr automobile suspension spring steel with different refractories
- Investigation into the thermal stability of a novel hot-work die steel 5CrNiMoVNb
- Residual stress relaxation considering microstructure evolution in heat treatment of metallic thin-walled part
- Experiments of Ti6Al4V manufactured by low-speed wire cut electrical discharge machining and electrical parameters optimization
- Effect of chloride ion concentration on stress corrosion cracking and electrochemical corrosion of high manganese steel
- Prediction of oxygen-blowing volume in BOF steelmaking process based on BP neural network and incremental learning
- Effect of annealing temperature on the structure and properties of FeCoCrNiMo high-entropy alloy
- Study on physical properties of Al2O3-based slags used for the self-propagating high-temperature synthesis (SHS) – metallurgy method
- Low-temperature corrosion behavior of laser cladding metal-based alloy coatings on EH40 high-strength steel for icebreaker
- Study on thermodynamics and dynamics of top slag modification in O5 automobile sheets
- Structure optimization of continuous casting tundish with channel-type induction heating using mathematical modeling
- Microstructure and mechanical properties of NbC–Ni cermets prepared by microwave sintering
- Spider-based FOPID controller design for temperature control in aluminium extrusion process
- Prediction model of BOF end-point P and O contents based on PCA–GA–BP neural network
- Study on hydrogen-induced stress corrosion of 7N01-T4 aluminum alloy for railway vehicles
- Study on the effect of micro-shrinkage porosity on the ultra-low temperature toughness of ferritic ductile iron
- Characterization of surface decarburization and oxidation behavior of Cr–Mo cold heading steel
- Effect of post-weld heat treatment on the microstructure and mechanical properties of laser-welded joints of SLM-316 L/rolled-316 L
- An investigation on as-cast microstructure and homogenization of nickel base superalloy René 65
- Effect of multiple laser re-melting on microstructure and properties of Fe-based coating
- Experimental study on the preparation of ferrophosphorus alloy using dephosphorization furnace slag by carbothermic reduction
- Research on aging behavior and safe storage life prediction of modified double base propellant
- Evaluation of the calorific value of exothermic sleeve material by the adiabatic calorimeter
- Thermodynamic calculation of phase equilibria in the Al–Fe–Zn–O system
- Effect of rare earth Y on microstructure and texture of oriented silicon steel during hot rolling and cold rolling processes
- Effect of ambient temperature on the jet characteristics of a swirl oxygen lance with mixed injection of CO2 + O2
- Research on the optimisation of the temperature field distribution of a multi microwave source agent system based on group consistency
- The dynamic softening identification and constitutive equation establishment of Ti–6.5Al–2Sn–4Zr–4Mo–1W–0.2Si alloy with initial lamellar microstructure
- Experimental investigation on microstructural characterization and mechanical properties of plasma arc welded Inconel 617 plates
- Numerical simulation and experimental research on cracking mechanism of twin-roll strip casting
- A novel method to control stress distribution and machining-induced deformation for thin-walled metallic parts
- Review Article
- A study on deep reinforcement learning-based crane scheduling model for uncertainty tasks
- Topical Issue on Science and Technology of Solar Energy
- Synthesis of alkaline-earth Zintl phosphides MZn2P2 (M = Ca, Sr, Ba) from Sn solutions
- Dynamics at crystal/melt interface during solidification of multicrystalline silicon
- Boron removal from silicon melt by gas blowing technique
- Removal of SiC and Si3N4 inclusions in solar cell Si scraps through slag refining
- Electrochemical production of silicon
- Electrical properties of zinc nitride and zinc tin nitride semiconductor thin films toward photovoltaic applications
- Special Issue on The 4th International Conference on Graphene and Novel Nanomaterials (GNN 2022)
- Effect of microstructure on tribocorrosion of FH36 low-temperature steels
Articles in the same Issue
- Research Articles
- Numerical and experimental research on solidification of T2 copper alloy during the twin-roll casting
- Discrete probability model-based method for recognition of multicomponent combustible gas explosion hazard sources
- Dephosphorization kinetics of high-P-containing reduced iron produced from oolitic hematite ore
- In-phase thermomechanical fatigue studies on P92 steel with different hold time
- Effect of the weld parameter strategy on mechanical properties of double-sided laser-welded 2195 Al–Li alloy joints with filler wire
- The precipitation behavior of second phase in high titanium microalloyed steels and its effect on microstructure and properties of steel
- Development of a huge hybrid 3D-printer based on fused deposition modeling (FDM) incorporated with computer numerical control (CNC) machining for industrial applications
- Effect of different welding procedures on microstructure and mechanical property of TA15 titanium alloy joint
- Single-source-precursor synthesis and characterization of SiAlC(O) ceramics from a hyperbranched polyaluminocarbosilane
- Carbothermal reduction of red mud for iron extraction and sodium removal
- Reduction swelling mechanism of hematite fluxed briquettes
- Effect of in situ observation of cooling rates on acicular ferrite nucleation
- Corrosion behavior of WC–Co coating by plasma transferred arc on EH40 steel in low-temperature
- Study on the thermodynamic stability and evolution of inclusions in Al–Ti deoxidized steel
- Application on oxidation behavior of metallic copper in fire investigation
- Microstructural study of concrete performance after exposure to elevated temperatures via considering C–S–H nanostructure changes
- Prediction model of interfacial heat transfer coefficient changing with time and ingot diameter
- Design, fabrication, and testing of CVI-SiC/SiC turbine blisk under different load spectrums at elevated temperature
- Promoting of metallurgical bonding by ultrasonic insert process in steel–aluminum bimetallic castings
- Pre-reduction of carbon-containing pellets of high chromium vanadium–titanium magnetite at different temperatures
- Optimization of alkali metals discharge performance of blast furnace slag and its extreme value model
- Smelting high purity 55SiCr automobile suspension spring steel with different refractories
- Investigation into the thermal stability of a novel hot-work die steel 5CrNiMoVNb
- Residual stress relaxation considering microstructure evolution in heat treatment of metallic thin-walled part
- Experiments of Ti6Al4V manufactured by low-speed wire cut electrical discharge machining and electrical parameters optimization
- Effect of chloride ion concentration on stress corrosion cracking and electrochemical corrosion of high manganese steel
- Prediction of oxygen-blowing volume in BOF steelmaking process based on BP neural network and incremental learning
- Effect of annealing temperature on the structure and properties of FeCoCrNiMo high-entropy alloy
- Study on physical properties of Al2O3-based slags used for the self-propagating high-temperature synthesis (SHS) – metallurgy method
- Low-temperature corrosion behavior of laser cladding metal-based alloy coatings on EH40 high-strength steel for icebreaker
- Study on thermodynamics and dynamics of top slag modification in O5 automobile sheets
- Structure optimization of continuous casting tundish with channel-type induction heating using mathematical modeling
- Microstructure and mechanical properties of NbC–Ni cermets prepared by microwave sintering
- Spider-based FOPID controller design for temperature control in aluminium extrusion process
- Prediction model of BOF end-point P and O contents based on PCA–GA–BP neural network
- Study on hydrogen-induced stress corrosion of 7N01-T4 aluminum alloy for railway vehicles
- Study on the effect of micro-shrinkage porosity on the ultra-low temperature toughness of ferritic ductile iron
- Characterization of surface decarburization and oxidation behavior of Cr–Mo cold heading steel
- Effect of post-weld heat treatment on the microstructure and mechanical properties of laser-welded joints of SLM-316 L/rolled-316 L
- An investigation on as-cast microstructure and homogenization of nickel base superalloy René 65
- Effect of multiple laser re-melting on microstructure and properties of Fe-based coating
- Experimental study on the preparation of ferrophosphorus alloy using dephosphorization furnace slag by carbothermic reduction
- Research on aging behavior and safe storage life prediction of modified double base propellant
- Evaluation of the calorific value of exothermic sleeve material by the adiabatic calorimeter
- Thermodynamic calculation of phase equilibria in the Al–Fe–Zn–O system
- Effect of rare earth Y on microstructure and texture of oriented silicon steel during hot rolling and cold rolling processes
- Effect of ambient temperature on the jet characteristics of a swirl oxygen lance with mixed injection of CO2 + O2
- Research on the optimisation of the temperature field distribution of a multi microwave source agent system based on group consistency
- The dynamic softening identification and constitutive equation establishment of Ti–6.5Al–2Sn–4Zr–4Mo–1W–0.2Si alloy with initial lamellar microstructure
- Experimental investigation on microstructural characterization and mechanical properties of plasma arc welded Inconel 617 plates
- Numerical simulation and experimental research on cracking mechanism of twin-roll strip casting
- A novel method to control stress distribution and machining-induced deformation for thin-walled metallic parts
- Review Article
- A study on deep reinforcement learning-based crane scheduling model for uncertainty tasks
- Topical Issue on Science and Technology of Solar Energy
- Synthesis of alkaline-earth Zintl phosphides MZn2P2 (M = Ca, Sr, Ba) from Sn solutions
- Dynamics at crystal/melt interface during solidification of multicrystalline silicon
- Boron removal from silicon melt by gas blowing technique
- Removal of SiC and Si3N4 inclusions in solar cell Si scraps through slag refining
- Electrochemical production of silicon
- Electrical properties of zinc nitride and zinc tin nitride semiconductor thin films toward photovoltaic applications
- Special Issue on The 4th International Conference on Graphene and Novel Nanomaterials (GNN 2022)
- Effect of microstructure on tribocorrosion of FH36 low-temperature steels

