Abstract
This study was conducted to fabricate scaffold from polylactic acid (PLA) and hydroxyapatite (HA) extracted from waste bovine bone for enhancing both mechanical and biocompatible properties. After pretreatment in dilute NaOH solution, the bone was calcined at 900°C for 6 h, ball milled and converted to HA. Factors that affect the formation of HA were investigated. Experimental results showed that HA particles with crystal size < 100 nm and 99% crystallinity could be obtained at 90°C, pH 11 and 35 mM H3PO4 solution followed by 4 h calcination at 900°C. By using non-solvent induced phase separation method, PLA scaffolds with pore size and surface area of 22.6 μm and 25.7 m2/g, respectively, containing different hydroxyapatite were successfully prepared. Tensile strength of scaffolds increased due to effective support by HA grafted collagen. PLA scaffolds containing HA were more degradable than PLA scaffolds and PLA scaffolds containing HA grafted collagen. Cell culture results showed that cell density increased significantly on porous scaffolds than that on the dense scaffolds. Moreover, cell density also increased significantly on the scaffold containing HA grafted collagen than that on the scaffold with pure HA.
1 Introduction
Tissue engineering method consists of harvesting cells from a patient, expanding them in culture, and seeding them into scaffolds which can later be implanted into the patient. The scaffolds known as artificial architecture for biosynthetic extracellular matrix (ECM) are used to create a 3D environment in order to promote cellular attachment, migration, proliferation and differentiation. Therefore, finding materials to produce scaffolds that possess not only properties suitable for cell penetration, capillary ingrowth, and adequate solute transport but also good mechanical and biological properties is a very important work. Some synthetic polymers have been applied to create scaffolds such as poly(glycolic acid) (PGA), poly(lactic acid) (PLA) [1], poly(lactic-co-glycolic acid) (PLGA) [2], poly(caprolactone) (PCL) [3], poly(propylene fumarate) (PPF), and poly(ethylene glycol) (PEG) [4]. However, mechanical properties of these synthetic polymers are very weak. Some fillers and reinforced materials such as calcium phosphate, ceramic and glass were mixed with polymers to enhance their mechanical properties [5, 6, 7, 8]. However, fillers and reinforced materials usually have low biocompatibility. Therefore, it is necessary to find potential materials that can be applied in scaffold. Recently hydroxyapatite is considered as a good material to replace ceramic [9].
Hydroxyapatite [Ca10(PO4)6(OH)2] with molar ratio of Ca/P from 1.58 to 1.67 is well known as the mineral component of animal bones [10]. Hydroxyapatite (HA) was found to have good biocompatibility with cells [11]. HA can be synthesized by numerous techniques such as sol–gel, chemical precipitation, hydrothermal, hydrolysis, and microemulsion [12]. HA synthesized from chemical such as CaO, Ca(NO3)2, (NH4)2HPO4 did not possess desired biological characteristics while these produced from natural materials such as egg shells or animal bones are very interesting since it is believed to be more biocompatible and biological safe [2, 3, 4, 5]. However, high temperature treatment during extraction process may cause structure breaking or loss of certain components which will partly affect the biological activity of HA. In Vietnam, the yearly amount of bovine bones produced from food processing industry is huge. These bones are often used to produce low economic value animal feed. In this study, waste bovine bone was converted into HA and then applied to produce biocompatible scaffold by using phase separation method.
A simple nonsolvent induced phase separation method was widely used to prepare polymeric membrane [13,14]. This method is also considered as a potential method to create polymeric porous structure, especially scaffolds used in tissue engineering. In the nonsolvent method, polymer was dissolved in organic solvents such as tetrehydrofuran (THF), chloroform to form homogeneous polymer solution. The solution was then separated by adding nonsolvent such as water, acetone and ethanol to form porous structure. In this work, the nonsolvent induced phase separation method was applied to create porous scaffold from polylactic acid (PLA) and hydroxyapatite for tissue engineering.
2 Materials and methods
2.1 Materials
Bone of adult bovine (2-3 years old) was collected from a local slaughterhouse. The bone was first cooked at 100°C for 24 h to remove tissue, then dried at 80-100°C for 24 h and cut into small sizes of about 10 mm × 5 mm × 5 mm. Chemicals such as NaOH, H3PO4 and NH3, 3-amino propyl triethoxysilane (APTES), glutaraldehyde (GA), and phosphate buffered saline (PBS) were supplied by Merck Vietnam for use in pretreatment and extraction of HA from bovine bone. Poly lactic acid (PLA) (CAS. 9051-89-2) was purchased from Nature Works. The solvents used in the preparation of scaffold such as chloroform (CHCl3) and ethanol were supplied by Merck, Vietnam. Standard hydroxyappatite (≥ 90%) was supplied by Sigma- Aldrich, Vietnam.
2.2 Extraction of hydroxyapatite from bovine bone
After pretreatment with 0.1 M NaOH for 24 h, the pretreated bone was washed with water and acetone to remove fats and other impurities. The bovine bone was then calcined at 900°C for 6 h in a furnace (Nabertherm, LHT 1750°C) to remove all organic components in the bone. The calcined bovine bone was then grinded into powder with particle size of 5 to 10 μm by using high speed mill ball. The bovine powder (10 g) was dispersed in 100 mL distilled water. The mixture was then reacted with aqueous H3PO4 solution. The reacted mixture was cooled to room temperature and neutralized by NH3 in 2 h. After reaction, the mixture was aged overnight before separating by filter. The sample was dried at 100°C before continuously calcining at high temperature to stabilize the structure of HA. Factors that affected HA formation such as reaction temperature, reaction time, pH and concentration of H3PO4 were investigated. X-ray diffraction (XRD), scanning electron microscopy (SEM), and Fourier transform infrared spectroscopy (FTIR) were applied for material characterization.
2.3 Preparation of hydroxyapatite grafted collagen
HA extracted from bovine bone was dispersed (1 wt%) in a co-solvent of ethanol/water (1:2 v/v). The suspension was added sequentially APTES 2 wt% and NH3 6 wt% while stirred at 100 rpm in an environment of N2 for 24 h. HA-APTES was collected and washed with distilled water 3 times. The HA-APTES was dispersed (1 wt%) into aqueous solution of 1% glutaraldehyde at room temperature and stirred at 100 rpm for 24 h. The mixture was filtered and washed by PBS solution to obtain the HA-APTES-GA particles. The particles were dispered 1% in PBS solution containing 100 mg/mL collagen extracted from catfish skin. The mixture was then stirred at 100 rpm for 24 h at 4°C. HA grafted collagen obtained was washed three times with PBS solution.
2.4 Preparation of scaffold
Nonsolvent induced phase separation method was applied to prepared scaffold. Poly lactic acid (PLA) was dissolved in chloroform at room temperature for 24 h. After that the polymer solution was casted onto a teflon mold (5 mm × 60 mm × 100 mm). The teflon mold was then immersed in a coagulation bath containing nonsolvent (ethanol/water) and PLA porous scaffold was obtained after 24 h.
2.5 Cell culture
2.5.1 Cell seeding
Fibrobast L929 standard cell suspension containing 20,000 cells/mL was prepared by diluting the cell suspension with serum medium. One milliliter of the cell solution was distributed in 24-well plate containing samples and incubated at 37°C in 5% CO2 atmosphere for 24 h. After a certain incubation time, the culture medium was removed and the unattached cells was washed by PBS solution. Cell density was characterized by both lactate dehydrogenase (LDH) assay and confocal laser scanning microscope.
2.5.2 Cell density by lactate dehydrogenase (LDH) assay
Cell density was determined by lactate dehydrogenase assay [15, 16, 17, 18]. The attached cells were lysed with 100 μL of 1% Triton X-100 in PBS. The reaction solution contained 0.3% NAD+, 0.27% diaphorase, 0.03% bovine serum albumin, 1.2% sucrose, 0.02% iodonitrotetrazolium, and 3.6% sodium lactate (all chemicals were purchased from Sigma). Optical density values were read at 490 nm by using enzyme-linked immunosorbent assay (ELISA, iMarkTM Absorbance Reader, BioRad). A standard curve was obtained by plotting the measured optical density of a series of cell solutions verses known cell densities.
2.5.3 Immunofluorescence staining for microscopy observation
The L-929 fibroblasts were fixed on the scaffold surfaces by immersion in a solution of 4% paraformaldehyde in PBS, pH 7.4, followed by permeabilization with 0.1% Triton X-100 at room temperature. Non-specific binding of proteins and antibodies was prevented by incubation with 0.5% BSA solution in PBS overnight at 4°C. The cells were stained for actin filaments with 165 nM of Rhodamine-FITC (Fluka, 77418) for 20 min at room temperature. The nuclei were stained with DAPI (Sigma, CAS. 28718-90-3) at 1:1000 dilutions for 20 min at room temperature.
2.5.4 Confocal laser scanning microscope
Confocal laser scanning microscope (LSCM, Leica-SP2, Germany, 40× 0.75 dry, 60× 0.75 oil or 100× 1.4 oil objective) was used to image the surface morphology of stained cells. Fluorescence with excitation wavelength (Ex) = 364 nm and emission wavelength (Em) = 454 nm were used to image cells stained by DAPI; and fluorescence with Ex = 495 nm and Em = 520 nm were used to capture images of cells stained by Phalloidin-FITC.
2.5.5 Statistical analyses
Variations of the amount of adherent and proliferate cells were statistically analyzed by performing one-way analysis of variance (ANOVA), executed by Minitab statistical software®. Fisher’s pairwise comparison test was applied to compare cell density on different samples for p < 0.05.
3 Results and discussion
3.1 Hydroxyapatite extraction from waste bovine bone
Bovine bone was pretreated to remove tissue, fats and other impurities. As shown in Figure 1, the color of bone was dark brown after cooking for 24 h at 100°C and it became brighter after treatment with NaOH solution. The bone was bright white and lost 45 wt% after calcined at 900°C.

Pictures of bovine bone treated by: (a) hot water at 100°C, 24 h; (b) 0.1 M NaOH solution, 48 h; (c) calcination at 900°C, 6 h; (d) ball milling for 30 min.
Chemical composition of the powder of bovine bone was analyzed by X-ray fluorescence (XRF). The results show that calcium and phosphorus are the two main components in bovine bone powder. CaO and P2O5 accounts for 44 wt% and 36 wt% of bovine bone powder, respectively (Table 1).
Chemical composition of calcined bovine bone powder.
| Composition | SiO2 | Al2O3 | Fe2O3 | CaO | MgO | SO3 | K2O | Na2O | Cl | P2O5 |
| Weight % | - | 0.49 | 0.01 | 44.10 | 1.41 | 1.05 | 0.02 | 3.69 | 0.04 | 36.2 |
3.1.1 Effect of concentration of H3PO4 on HA product
Results of XRD diffraction analysis of the calcined bovine bone show peaks of HA at 2θ (degree) at 10.8, 25.9, 31.7, 32.1, 34.1, 39.8, 46.7, 49.5, 50.5, 53.2; together with peaks of β-TCP and CaO at 2θ = 31.3, 33.67, 34.6, 35.85 and 23.4, respectively (Figure 2). It means that after calcining, crystal structure of HA, β-TCP and CaO still exist. Thus, in order to fully convert β-TCP and CaO that was created during calcination, H3PO4 with different concentrations was used to react with bovine bone powder. Results show that as concentration of H3PO4 increased, the peaks of β-TCP and CaO gradually disappeared and the intensity of peaks for HA increased. The peaks of β-TCP and CaO almost disappeared when using 35 mM H3PO4 and its characteristic peaks is not very different with the peaks of standard HA (Figures 2d and 2e). Calculated base on the Scherrer equation, the size of HA crystal was 83, 85, 86 and 88 nm and degree of crystallinity was 88%, 93%, 94% and 96% corresponding to concentration of H3PO4 0 mM, 25 mM, 30 mM and 35 mM, respectively (Table 2). Therefore, H3PO4 35 mM is considered as a good concentration to convert bovine bone to HA.

XRD diffraction of HA that was prepared in different concentrations of H3PO4: (a) 0 M, (b) 25 mM, (c) 30 mM, (d) 35 mM, and (e) Standard HA. Fixed condition: bovine powder/H3PO4 = 1 g/10 mL, reaction temperature 80°C, pH 11 and calcine temperature 900°C.
Effect of reaction temperature on size and crystal degree of HA.
| H2SO4 concentration, mM | 0 | 25 | 30 | 35 |
|---|---|---|---|---|
| Size of crystal, nm | 84.8 | 83.6 | 86.4 | 88.3 |
| Degree of crystallinity, % | 88.7 | 94.4 | 93.1 | 95.9 |
3.1.2 Effects of reaction temperature on formation of HA
Reaction temperature was varied from 60°C to 90°C to investigate its effect on HA formation. From the XRD diffraction (Figure 3), HA samples synthesized at 60°C, 70°C and 80°C showed that besides the characteristic peaks of HA, there still existed a few characteristic peaks of β-TCP. At 90°C, only the characteristic peaks of HA appeared. This result shows that reaction rate increased with increasing reaction temperature and at 90°C high impurity HA was created. This agrees with the results of Luo [19]. Moreover, besides increasing intensity, the HA peaks also become sharper which means that crystallinity increased with increasing reaction temperature. As temperature was increased from 60°C to 90°C, the degree of crystallization of HA increased from 93.5% to 97.7%, accompanying with the increase of crystal size from 80.3 to 95.7 nm (Table 3). This result is consistent with the study of Wijesinghe et al. [20]. Therefore, 90°C is the suitable temperature to produce HA from bovine bone.
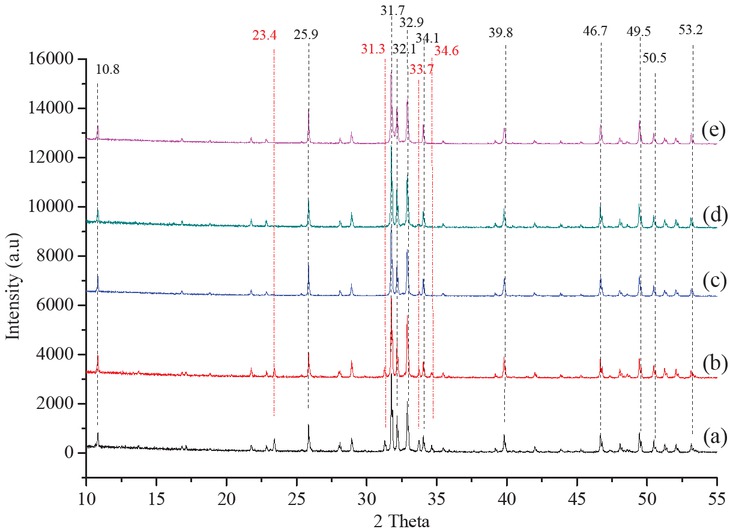
XRD diffraction of HA sample synthesized at (a) 60°C, (b) 70°C, (c) 80°C, (d) 90°C and (e) Standard HA. Fixed conditions: bovine powder/H3PO4 = 1:10 g/mL, H3PO4 (35 mM), pH 11 and calcine temperature 900°C.
Effect of reaction temperature on size and crystal degree of HA.
| Reaction temperature, °C | 60 | 70 | 80 | 90 |
|---|---|---|---|---|
| Size of crystal, nm | 80.3 | 86.5 | 88.3 | 95.7 |
| Degree of crystallinity, % | 93.5 | 94.8 | 95.9 | 97.7 |
3.1.3 Effect of pH on formation of HA
HA was synthesized at different pH values to investigate pH effect on the morphology and structure of HA. The XRD diffraction in all samples showed the characteristic peaks of HA appear (Figure 4). However, at pH 9, pH 10 and pH 12, there were few characteristic peaks of β-TCP. This result is consistent with the previous study of Peipei Wang et al. [21]. HA synthesized at pH 11 showed the characteristic peaks of HA and reach the highest crystal level of 97.7% (Table 4). The size of the crystal tends to increase with increasing pH and this is consistent with the study of Liu et al. [22].

XRD diffraction of HA sample synthesized at: (a) pH 9 (b) pH 10, (c) pH 11, (d) pH 12 and (e) Standard HA. Fixed conditions: bovine powder/H3PO4 = 1:10 g/mL, H3PO4 (35 mM), reaction temperature 90°C, calcine temperature 900°C.
Effect of reaction temperature on size and crystal degree of HA.
| pH value | 9 | 10 | 11 | 12 |
|---|---|---|---|---|
| Size of crystal, nm | 89.3 | 91.9 | 95.7 | 104.5 |
| Degree of crystallinity, % | 94.3 | 94.4 | 97.7 | 94.9 |
3.1.4 Effect of calcination temperature on HA formation
Temperature was varied from 700°C to 1000°C to investigate its effect on the formation of HA. Results of XRD diffraction analysis showed that the characteristic peaks of HA existed in all samples (Figure 5). The samples synthesized at 700°C and 800°C still showed small characteristic peaks of β-TCP phase and these peaks disappeared as temperature was increased to 900°C. At 900°C, only the characteristic peaks of HA appear and these peaks become narrower and sharper at 1000°C. Moreover, as temperature was increased from 700°C to 1000°C, crystallization degree increased from 83.7% to 99.0%, accompanying by the increase of crystal size from 90.6 nm to 101.8 nm (Table 5).

XRD diffraction of HA sample at: (a) 700°C, (b) 800°C, (c) 900°C, (d) 1000°C and (e) Standard HA. Fixed conditions: bovine powder/H3PO4 = 1:10 g/mL, H3PO4 (35 mM), reaction temperature 90°C and pH 11.
Effect of calcine temperature on crystal size and crystal degree of HA.
| Calcination temp., °C | 700 | 800 | 900 | 1000 |
|---|---|---|---|---|
| Size of crystal, nm | 90.6 | 92.5 | 95.7 | 101.8 |
| Degree of crystallinity, % | 83.7 | 95.9 | 97.7 | 99.0 |
3.2 Preparation of hydroxyapatite grafted collagen
HA prepared under optimal conditions of calcine temperature 1000°C, bovine powder/H3PO4 = 1:10 g/mL, H3PO4 (35 mM), reaction temperature 90°C and pH 11 was used for grafting collagen. SEM results (Figure 6) show that HA particles have asymmetric spherical form with size of about 1000 nm. The FTIR spectra (Figure 7) of the collagen grafted HA particles show the characteristic peak of HA–OH (3437 cm-1), PO43- (1042; 568 cm-1) and characteristic peak of –CO– (1997 cm-1) and –NH2 (3572 cm-1).

HA applied for grafting collagen was prepared under condition of calcine temperature 1000°C, bovine powder/H3PO4 = 1:10 g/mL, H3PO4, 0.35 M, reaction temperature 90°C and pH 11.

FTIR spectra of collagen grafted HA sample.
3.3 Preparation of biocompatible composite scaffold
Scaffolds in different concentrations of HA and HA grafted collagen were prepared by using nonsolvent induced phase separation method. PLA solution (125 g/L) was prepared in chloroform at room temperature. The polymer solution was mixed with HA and casted onto a teflon mold (5 mm × 60 mm × 100 mm). The teflon mold was then immersed into a coagulation bath (25°C, volume ratio ethanol to water 96/4; volume ratio solvent to nonsolvent 1/20). PLA porous scaffolds with different HA contents were obtained after for 24 h. SEM images show porous structure and its average pore size is 22.6 ± 4 μm (Figure 8). BET measurement resulted in a surface area of 25.7 m2/g.

SEM images of scaffolds prepared in different HA contents: (a) 0%, (b) 5%, (c) 10%, (d) 15%, (e) 20%.
Mechanical property of scaffolds was evaluated by tensile strength measured on a Zwick/Roell BDO equipment – FB050TN. Tensile strengths were 88.5 MPa, 76.5 MPa, 62.3 MPa, 40.1 MPa, and 10.6 MPa for scaffold samples containing 0%, 5%, 10%, 15%, and 20% HA, respectively (Figure 9). The results mean that the interface of HA particles and PLA was not compatible leading to decrease in tensile strength when HA content was increased. Compatibility was significantly improved for collagen grafted HA. Tensile strength of scaffold increased with increasing HA grafted collagen from 5% to 10% and after that decreased from 15% to 20%.

Tensile strength of scaffold containing HA and HA grafted collagen.
3.4 Degradation of scaffold in PBS solution
Scaffold samples containing 10% particles were prepared with size of 1 × 1 cm2 and immersed in 10 mL PBS solution at 37°C. After 7 days, degradation of scaffolds was 0.45%, 0.5% and 0.75% for PLA, PLA-HA-Col and PLA-HA, respectively. The degradation of scaffold increased with time. After 28 days, degradation was 1.75%, 3.4% and 4.3% for PLA, PLA-HA-Col, and PLA-HA, respectively. The results show that all scaffolds could be degraded in PBS solution (Figure 10). The degradation is highest for the PLA scaffolds containing HA. However, after grafting with collagen the degradation is lower than PLA scaffold, meaning that the interaction between PLA and HA-Coll is better than that of PLA and HA.

Degradation of scaffold in PBS solution.
3.5 Cell responses on scaffolds
Biocompatibility of scaffold was studied based on cell responses. PLA scaffolds containing different concentrations of HA particle and collagen grafted HA particle were evaluated by directly culturing L929 cell line on the samples and characterized by lactate dehydrogenase assay and confocal microscopy. Cell density is shown in Figure 11. Cell density increased significantly on porous scaffolds compared to that on dense scaffold. Cell density also increased sightly as HA content was increased from 0% to 10% and increased significantly as HA content reached 15%. Moreover, cell density also increased significantly on scaffold containing HA grafted collagen compared to that on scaffold with pure HA.
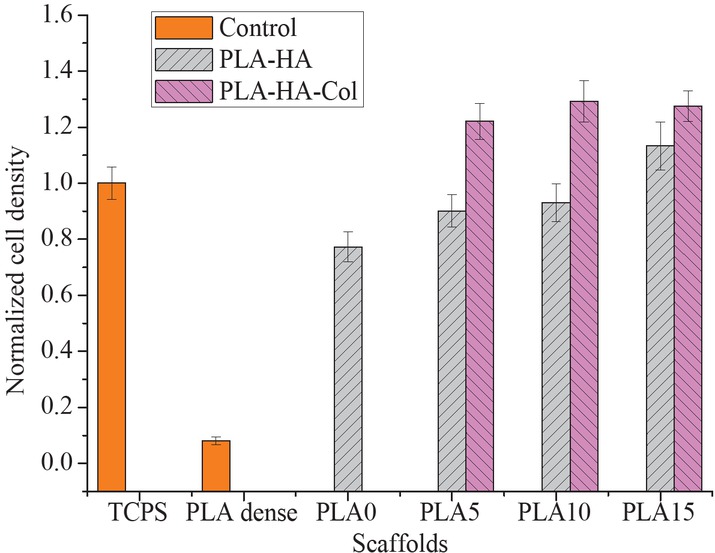
LDH results for cell density on pristine (dense PLA scaffold), PLA-HA scaffold and PLA-HA-Col scaffold at different concentrations of HA particles.
Confocal microscopy images show that fibroblasts attached on PLA dense and PLA scaffold without HA with limited number after 24 h cultivation (Figures 12a and 12b). It is similar to LDH results, confocal microscopy images also show that cell number increased approximately with increasing HA concentration in porous scaffold (Figures 12d-f), suggesting that cell proliferation was promoted by HA. Moreover, it was observed that higher cell density was found on surfaces containing HA grafted with collagen (Figures 12g-k).
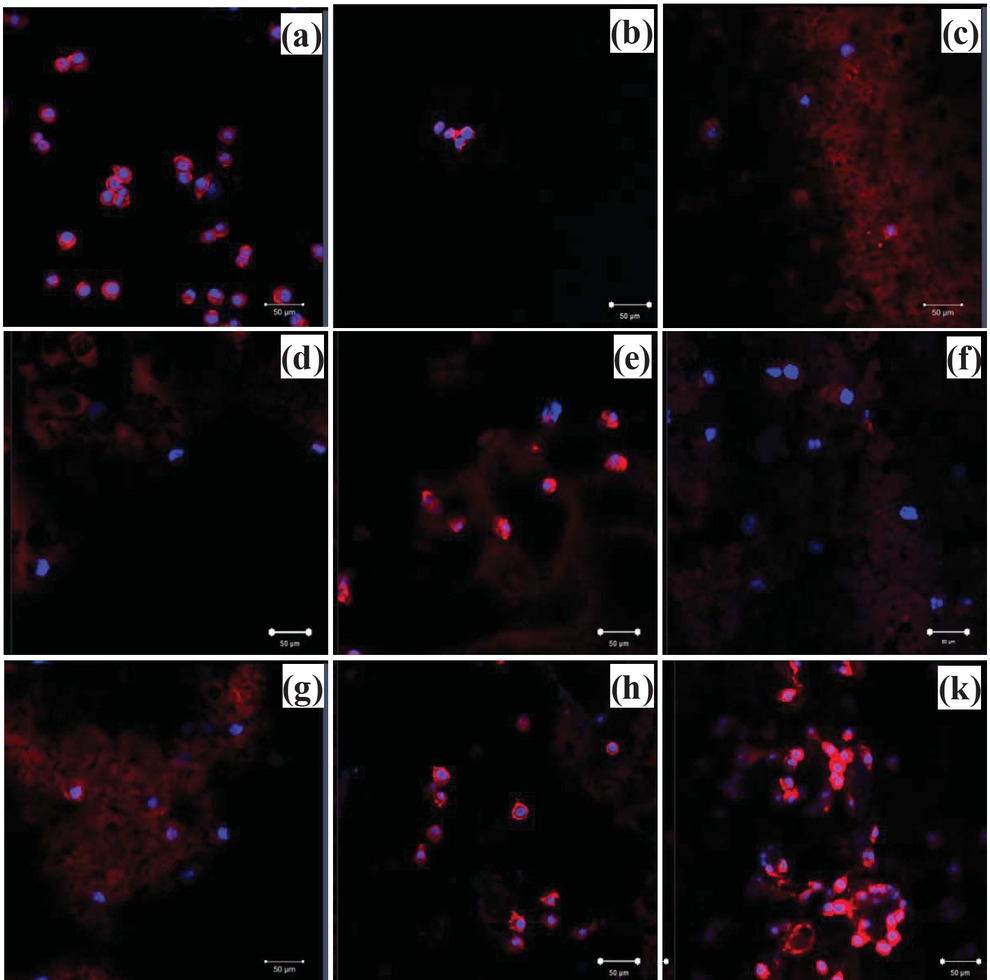
Confocal laser scanning microscopy images of L-929 fibroblasts on scaffolds, cultured for 24 h: (a) TCPS, (b) PLA dense scaffold without pore and (c) porous scaffold without HA. (d), (e) and (f) are the images from PLA scaffol ds that contained HA ranging from 5%, 10% to 15% respectively and (g), (h) and (k) are the images from PLA scaffolds that contained HA grafted collagen ranging from 5%, 10% to 15%, respectively (scale bar = 50 μm).
4 Conclusion
An effective method was sucessfully applied to produce hydroxyapatite from waste bovine bone. It was possible to create HA particles with a crystal size < 100 nm with a crystallinity of 99% under conditions of H3PO4 35 mM, reaction temperature 90°C, pH 11, calcination at 900°C for 4 h. Bioccompatible PLA scaffolds prepared by combining HA promoted cell growth effectively depending on HA content. Collagen extracted from catfish was successfully grafted on HA surface and it enhanced cell proliferation compared to pure HA.
Acknowledgments
The authors would like to express great appreciation to Vietnam Ministry of Education and Training for financial support (Grant No.: B2017-TCT-20ĐT).
References
[1] Huang R., Zhu X., Tu H., Wan A., The crystallization behavior of porous poly(lactic acid) prepared by modified solvent casting/particulate leaching technique for potential use of tissue engineering scaffold. Mater. Lett., 2014, 136, 126-129.10.1016/j.matlet.2014.08.044Search in Google Scholar
[2] Patrício T., Domingos M., Gloria A., Bártolo P., Characterisation of PCL and PCL/PLA scaffolds for tissue engineering. Procedia CIRP, 2013, 5, 110-114.10.1016/j.procir.2013.01.022Search in Google Scholar
[3] Johari N., Fathi M.H., Golozar M.A., Fabrication, characterization and evaluation of the mechanical properties of poly (ε-caprolactone)/nano-fluoridated hydroxyapatite scaffold for bone tissue engineering. Compos. Part B-Eng., 2012, 3, 1671-1675.10.1016/j.compositesb.2012.01.013Search in Google Scholar
[4] Huang R., Zhu X., Zhao T., Wan A., Preparation of tissue engineering porous scaffold with poly(lactic acid) and polyethylene glycol solution blend by solvent-casting/particulate-leaching. Mater. Res. Express, 2014, 1, 045403.10.1088/2053-1591/1/4/045403Search in Google Scholar
[5] López-Álvarez M., Rodríguez-Valencia C., Serra J., González P., Bio-inspired ceramics: promising scaffolds for bone tissue engineering. Procedia Engineer., 2013, 59, 51-58.10.1016/j.proeng.2013.05.093Search in Google Scholar
[6] Roohani-Esfahani S.I., Chen Y., Shi J., Zreiqat H., Fabrication and characterization of a new, strong and bioactive ceramic scaffold for bone regeneration. Mater. Lett., 2013, 107, 378-381.10.1016/j.matlet.2013.06.046Search in Google Scholar
[7] Chen Q., Baino F., Spriano S., Pugno N.M., Vitale-Brovarone C., Modelling of the strength–porosity relationship in glass-ceramic foam scaffolds for bone repair. J. Eur. Ceram. Soc., 2014, 34, 2663-2673.10.1016/j.jeurceramsoc.2013.11.041Search in Google Scholar
[8] Sabree I., Gough J.E., Derby B., Mechanical properties of porous ceramic scaffolds: Influence of internal dimensions. Ceram. Int., 2015, 41, 8425-8432.10.1016/j.ceramint.2015.03.044Search in Google Scholar
[9] Sadat-Shojai M., Khorasani M.T., Dinpanah-Khoshdargi E., Jamshidi A., Synthesis methods for nanosized hydroxyapatite with diverse structures. Acta Biomater., 2013, 9, 7591-7621.10.1016/j.actbio.2013.04.012Search in Google Scholar PubMed
[10] Mostafa N.Y., Characterization, thermal stability and sintering of hydroxyapatite powders prepared by different routes. Mater. Chem. Phys., 2005, 94, 333-341.10.1016/j.matchemphys.2005.05.011Search in Google Scholar
[11] Sobczak A., Kowalski Z., Wzorek Z., Preparation of hydroxyapatite from animal bones. Acta Bioeng. Biomech., 2009, 11, 23-28.10.2478/v10026-009-0010-5Search in Google Scholar
[12] Barakat N.A.M., Khalil K.A., Sheikh F.A., Omran A.M., Gaihre B., Khil S.M., et al., Physiochemical characterizations of hydroxyapatite extracted from bovine bones by three different methods: Extraction of biologically desirable HAp. Mater. Sci. Eng. C, 2008, 28, 1381-1387.10.1016/j.msec.2008.03.003Search in Google Scholar
[13] Reuvers A.J., van den Berg J.W.A., Smolders CA., Formation of membranes by means of immersion precipitation: Part I. A model to describe mass transfer during immersion precipitation. J. Membrane Sci., 1987, 34, 45-65.10.1016/S0376-7388(00)80020-4Search in Google Scholar
[14] Wang Y., Liu Z., Huang Y., Han B., Yang G., Micropatterned polymer surfaces induced by nonsolvent. Langmuir, 2006, 22, 1928-1931.10.1021/la051646dSearch in Google Scholar
[15] Tsai W.B., Grunkemeier J.M., Horbett T.A., Human plasma fibrinogen adsorption and platelet adhesion to polystyrene. J. Biomed. Mater. Res., 1999, 44, 130-139.10.1002/(SICI)1097-4636(199902)44:2<130::AID-JBM2>3.0.CO;2-9Search in Google Scholar
[16] Tsai W.B., Lin J.H., Modulation of morphology and functions of human hepatoblastoma cells by nano-grooved substrata. Acta Biomater., 2009, 5, 1442-1454.10.1016/j.actbio.2009.01.002Search in Google Scholar
[17] Tsai W.-B., Chen R.P.Y., Wei K.L., Chen Y.R., Liao T.Y., Liu H.L., et al., Polyelectrolyte multilayer films functionalized with peptides for promoting osteoblast functions. Acta Biomater., 2009, 5, 3467-3477.10.1016/j.actbio.2009.05.034Search in Google Scholar
[18] Tamada Y., Kulik E.A., Ikada Y., Simple method for platelet counting. Biomaterials, 1995, 16, 259-261.10.1016/0142-9612(95)92126-QSearch in Google Scholar
[19] Luo J., Chen J., Li W., Huang Z., Chen C., Temperature effect on hydroxyapatite preparation by co-precipitation method under carbamide influence. Matec Web of Conf., 2015, 26, 1-5.10.1051/matecconf/20152601007Search in Google Scholar
[20] Wijesinghe W., Mantilaka M., Premalal E., Herath H., Mahalingam S., Edirisinghe M., et al., Facile synthesis of both needle-like and spherical hydroxyapatite nanoparticles: Effect of synthetic temperature and calcination on morphology, crystallite size and crystallinity. Mater. Sci. Eng. C., 2014, 42, 83-90.10.1016/j.msec.2014.05.032Search in Google Scholar
[21] Wang P., Li C., Gong H., Jiang X., Wang H., Li K., Effects of synthesis conditions on the morphology of hydroxyapatite nanoparticles produced by wet chemical process. Powder Technol., 2010, 203, 315-321.10.1016/j.powtec.2010.05.023Search in Google Scholar
[22] Liu J., Ye X., Wang H., Zhu M., Wang B., Yan H., The influence of pH and temperature on the morphology of hydroxyapatite synthesized by hydrothermal method. Ceram. Int., 2003, 29, 629-633.10.1016/S0272-8842(02)00210-9Search in Google Scholar
© 2020 Ho et al., published by De Gruyter
This work is licensed under the Creative Commons Attribution 4.0 International License.
Articles in the same Issue
- Obituary for Prof. Dr. Jun-ichi Yoshida
- Regular Articles
- Optimization of microwave-assisted manganese leaching from electrolyte manganese residue
- Crustacean shell bio-refining to chitin by natural deep eutectic solvents
- The kinetics of the extraction of caffeine from guarana seed under the action of ultrasonic field with simultaneous cooling
- Biocomposite scaffold preparation from hydroxyapatite extracted from waste bovine bone
- A simple room temperature-static bioreactor for effective synthesis of hexyl acetate
- Biofabrication of zinc oxide nanoparticles, characterization and cytotoxicity against pediatric leukemia cell lines
- Efficient synthesis of palladium nanoparticles using guar gum as stabilizer and their applications as catalyst in reduction reactions and degradation of azo dyes
- Isolation of biosurfactant producing bacteria from Potwar oil fields: Effect of non-fossil fuel based carbon sources
- Green synthesis, characterization and photocatalytic applications of silver nanoparticles using Diospyros lotus
- Dielectric properties and microwave heating behavior of neutral leaching residues from zinc metallurgy in the microwave field
- Green synthesis and stabilization of silver nanoparticles using Lysimachia foenum-graecum Hance extract and their antibacterial activity
- Microwave-induced heating behavior of Y-TZP ceramics under multiphysics system
- Synthesis and catalytic properties of nickel salts of Keggin-type heteropolyacids embedded metal-organic framework hybrid nanocatalyst
- Preparation and properties of hydrogel based on sawdust cellulose for environmentally friendly slow release fertilizers
- Structural characterization, antioxidant and cytotoxic effects of iron nanoparticles synthesized using Asphodelus aestivus Brot. aqueous extract
- Phase transformation involved in the reduction process of magnesium oxide in calcined dolomite by ferrosilicon with additive of aluminum
- Green synthesis of TiO2 nanoparticles from Syzygium cumini extract for photo-catalytic removal of lead (Pb) in explosive industrial wastewater
- The study on the influence of oxidation degree and temperature on the viscosity of biodiesel
- Prepare a catalyst consist of rare earth minerals to denitrate via NH3-SCR
- Bacterial nanobiotic potential
- Green synthesis and characterization of carboxymethyl guar gum: Application in textile printing technology
- Potential of adsorbents from agricultural wastes as alternative fillers in mixed matrix membrane for gas separation: A review
- Bactericidal and cytotoxic properties of green synthesized nanosilver using Rosmarinus officinalis leaves
- Synthesis of biomass-supported CuNi zero-valent nanoparticles through wetness co-impregnation method for the removal of carcinogenic dyes and nitroarene
- Synthesis of 2,2′-dibenzoylaminodiphenyl disulfide based on Aspen Plus simulation and the development of green synthesis processes
- Catalytic performance of the biosynthesized AgNps from Bistorta amplexicaule: antifungal, bactericidal, and reduction of carcinogenic 4-nitrophenol
- Optical and antimicrobial properties of silver nanoparticles synthesized via green route using honey
- Adsorption of l-α-glycerophosphocholine on ion-exchange resin: Equilibrium, kinetic, and thermodynamic studies
- Microwave-assisted green synthesis of silver nanoparticles using dried extracts of Chlorella vulgaris and antibacterial activity studies
- Preparation of graphene oxide/chitosan complex and its adsorption properties for heavy metal ions
- Green synthesis of metal and metal oxide nanoparticles from plant leaf extracts and their applications: A review
- Synthesis, characterization, and electrochemical properties of carbon nanotubes used as cathode materials for Al–air batteries from a renewable source of water hyacinth
- Optimization of medium–low-grade phosphorus rock carbothermal reduction process by response surface methodology
- The study of rod-shaped TiO2 composite material in the protection of stone cultural relics
- Eco-friendly synthesis of AuNPs for cutaneous wound-healing applications in nursing care after surgery
- Green approach in fabrication of photocatalytic, antimicrobial, and antioxidant zinc oxide nanoparticles – hydrothermal synthesis using clove hydroalcoholic extract and optimization of the process
- Green synthesis: Proposed mechanism and factors influencing the synthesis of platinum nanoparticles
- Green synthesis of 3-(1-naphthyl), 4-methyl-3-(1-naphthyl) coumarins and 3-phenylcoumarins using dual-frequency ultrasonication
- Optimization for removal efficiency of fluoride using La(iii)–Al(iii)-activated carbon modified by chemical route
- In vitro biological activity of Hydroclathrus clathratus and its use as an extracellular bioreductant for silver nanoparticle formation
- Evaluation of saponin-rich/poor leaf extract-mediated silver nanoparticles and their antifungal capacity
- Propylene carbonate synthesis from propylene oxide and CO2 over Ga-Silicate-1 catalyst
- Environmentally benevolent synthesis and characterization of silver nanoparticles using Olea ferruginea Royle for antibacterial and antioxidant activities
- Eco-synthesis and characterization of titanium nanoparticles: Testing its cytotoxicity and antibacterial effects
- A novel biofabrication of gold nanoparticles using Erythrina senegalensis leaf extract and their ameliorative effect on mycoplasmal pneumonia for treating lung infection in nursing care
- Phytosynthesis of selenium nanoparticles using the costus extract for bactericidal application against foodborne pathogens
- Temperature effects on electrospun chitosan nanofibers
- An electrochemical method to investigate the effects of compound composition on gold dissolution in thiosulfate solution
- Trillium govanianum Wall. Ex. Royle rhizomes extract-medicated silver nanoparticles and their antimicrobial activity
- In vitro bactericidal, antidiabetic, cytotoxic, anticoagulant, and hemolytic effect of green-synthesized silver nanoparticles using Allium sativum clove extract incubated at various temperatures
- The green synthesis of N-hydroxyethyl-substituted 1,2,3,4-tetrahydroquinolines with acidic ionic liquid as catalyst
- Effect of KMnO4 on catalytic combustion performance of semi-coke
- Removal of Congo red and malachite green from aqueous solution using heterogeneous Ag/ZnCo-ZIF catalyst in the presence of hydrogen peroxide
- Nucleotide-based green synthesis of lanthanide coordination polymers for tunable white-light emission
- Determination of life cycle GHG emission factor for paper products of Vietnam
- Parabolic trough solar collectors: A general overview of technology, industrial applications, energy market, modeling, and standards
- Structural characteristics of plant cell wall elucidated by solution-state 2D NMR spectroscopy with an optimized procedure
- Sustainable utilization of a converter slagging agent prepared by converter precipitator dust and oxide scale
- Efficacy of chitosan silver nanoparticles from shrimp-shell wastes against major mosquito vectors of public health importance
- Effectiveness of six different methods in green synthesis of selenium nanoparticles using propolis extract: Screening and characterization
- Characterizations and analysis of the antioxidant, antimicrobial, and dye reduction ability of green synthesized silver nanoparticles
- Foliar applications of bio-fabricated selenium nanoparticles to improve the growth of wheat plants under drought stress
- Green synthesis of silver nanoparticles from Valeriana jatamansi shoots extract and its antimicrobial activity
- Characterization and biological activities of synthesized zinc oxide nanoparticles using the extract of Acantholimon serotinum
- Effect of calcination temperature on rare earth tailing catalysts for catalytic methane combustion
- Enhanced diuretic action of furosemide by complexation with β-cyclodextrin in the presence of sodium lauryl sulfate
- Development of chitosan/agar-silver nanoparticles-coated paper for antibacterial application
- Preparation, characterization, and catalytic performance of Pd–Ni/AC bimetallic nano-catalysts
- Acid red G dye removal from aqueous solutions by porous ceramsite produced from solid wastes: Batch and fixed-bed studies
- Review Articles
- Recent advances in the catalytic applications of GO/rGO for green organic synthesis
Articles in the same Issue
- Obituary for Prof. Dr. Jun-ichi Yoshida
- Regular Articles
- Optimization of microwave-assisted manganese leaching from electrolyte manganese residue
- Crustacean shell bio-refining to chitin by natural deep eutectic solvents
- The kinetics of the extraction of caffeine from guarana seed under the action of ultrasonic field with simultaneous cooling
- Biocomposite scaffold preparation from hydroxyapatite extracted from waste bovine bone
- A simple room temperature-static bioreactor for effective synthesis of hexyl acetate
- Biofabrication of zinc oxide nanoparticles, characterization and cytotoxicity against pediatric leukemia cell lines
- Efficient synthesis of palladium nanoparticles using guar gum as stabilizer and their applications as catalyst in reduction reactions and degradation of azo dyes
- Isolation of biosurfactant producing bacteria from Potwar oil fields: Effect of non-fossil fuel based carbon sources
- Green synthesis, characterization and photocatalytic applications of silver nanoparticles using Diospyros lotus
- Dielectric properties and microwave heating behavior of neutral leaching residues from zinc metallurgy in the microwave field
- Green synthesis and stabilization of silver nanoparticles using Lysimachia foenum-graecum Hance extract and their antibacterial activity
- Microwave-induced heating behavior of Y-TZP ceramics under multiphysics system
- Synthesis and catalytic properties of nickel salts of Keggin-type heteropolyacids embedded metal-organic framework hybrid nanocatalyst
- Preparation and properties of hydrogel based on sawdust cellulose for environmentally friendly slow release fertilizers
- Structural characterization, antioxidant and cytotoxic effects of iron nanoparticles synthesized using Asphodelus aestivus Brot. aqueous extract
- Phase transformation involved in the reduction process of magnesium oxide in calcined dolomite by ferrosilicon with additive of aluminum
- Green synthesis of TiO2 nanoparticles from Syzygium cumini extract for photo-catalytic removal of lead (Pb) in explosive industrial wastewater
- The study on the influence of oxidation degree and temperature on the viscosity of biodiesel
- Prepare a catalyst consist of rare earth minerals to denitrate via NH3-SCR
- Bacterial nanobiotic potential
- Green synthesis and characterization of carboxymethyl guar gum: Application in textile printing technology
- Potential of adsorbents from agricultural wastes as alternative fillers in mixed matrix membrane for gas separation: A review
- Bactericidal and cytotoxic properties of green synthesized nanosilver using Rosmarinus officinalis leaves
- Synthesis of biomass-supported CuNi zero-valent nanoparticles through wetness co-impregnation method for the removal of carcinogenic dyes and nitroarene
- Synthesis of 2,2′-dibenzoylaminodiphenyl disulfide based on Aspen Plus simulation and the development of green synthesis processes
- Catalytic performance of the biosynthesized AgNps from Bistorta amplexicaule: antifungal, bactericidal, and reduction of carcinogenic 4-nitrophenol
- Optical and antimicrobial properties of silver nanoparticles synthesized via green route using honey
- Adsorption of l-α-glycerophosphocholine on ion-exchange resin: Equilibrium, kinetic, and thermodynamic studies
- Microwave-assisted green synthesis of silver nanoparticles using dried extracts of Chlorella vulgaris and antibacterial activity studies
- Preparation of graphene oxide/chitosan complex and its adsorption properties for heavy metal ions
- Green synthesis of metal and metal oxide nanoparticles from plant leaf extracts and their applications: A review
- Synthesis, characterization, and electrochemical properties of carbon nanotubes used as cathode materials for Al–air batteries from a renewable source of water hyacinth
- Optimization of medium–low-grade phosphorus rock carbothermal reduction process by response surface methodology
- The study of rod-shaped TiO2 composite material in the protection of stone cultural relics
- Eco-friendly synthesis of AuNPs for cutaneous wound-healing applications in nursing care after surgery
- Green approach in fabrication of photocatalytic, antimicrobial, and antioxidant zinc oxide nanoparticles – hydrothermal synthesis using clove hydroalcoholic extract and optimization of the process
- Green synthesis: Proposed mechanism and factors influencing the synthesis of platinum nanoparticles
- Green synthesis of 3-(1-naphthyl), 4-methyl-3-(1-naphthyl) coumarins and 3-phenylcoumarins using dual-frequency ultrasonication
- Optimization for removal efficiency of fluoride using La(iii)–Al(iii)-activated carbon modified by chemical route
- In vitro biological activity of Hydroclathrus clathratus and its use as an extracellular bioreductant for silver nanoparticle formation
- Evaluation of saponin-rich/poor leaf extract-mediated silver nanoparticles and their antifungal capacity
- Propylene carbonate synthesis from propylene oxide and CO2 over Ga-Silicate-1 catalyst
- Environmentally benevolent synthesis and characterization of silver nanoparticles using Olea ferruginea Royle for antibacterial and antioxidant activities
- Eco-synthesis and characterization of titanium nanoparticles: Testing its cytotoxicity and antibacterial effects
- A novel biofabrication of gold nanoparticles using Erythrina senegalensis leaf extract and their ameliorative effect on mycoplasmal pneumonia for treating lung infection in nursing care
- Phytosynthesis of selenium nanoparticles using the costus extract for bactericidal application against foodborne pathogens
- Temperature effects on electrospun chitosan nanofibers
- An electrochemical method to investigate the effects of compound composition on gold dissolution in thiosulfate solution
- Trillium govanianum Wall. Ex. Royle rhizomes extract-medicated silver nanoparticles and their antimicrobial activity
- In vitro bactericidal, antidiabetic, cytotoxic, anticoagulant, and hemolytic effect of green-synthesized silver nanoparticles using Allium sativum clove extract incubated at various temperatures
- The green synthesis of N-hydroxyethyl-substituted 1,2,3,4-tetrahydroquinolines with acidic ionic liquid as catalyst
- Effect of KMnO4 on catalytic combustion performance of semi-coke
- Removal of Congo red and malachite green from aqueous solution using heterogeneous Ag/ZnCo-ZIF catalyst in the presence of hydrogen peroxide
- Nucleotide-based green synthesis of lanthanide coordination polymers for tunable white-light emission
- Determination of life cycle GHG emission factor for paper products of Vietnam
- Parabolic trough solar collectors: A general overview of technology, industrial applications, energy market, modeling, and standards
- Structural characteristics of plant cell wall elucidated by solution-state 2D NMR spectroscopy with an optimized procedure
- Sustainable utilization of a converter slagging agent prepared by converter precipitator dust and oxide scale
- Efficacy of chitosan silver nanoparticles from shrimp-shell wastes against major mosquito vectors of public health importance
- Effectiveness of six different methods in green synthesis of selenium nanoparticles using propolis extract: Screening and characterization
- Characterizations and analysis of the antioxidant, antimicrobial, and dye reduction ability of green synthesized silver nanoparticles
- Foliar applications of bio-fabricated selenium nanoparticles to improve the growth of wheat plants under drought stress
- Green synthesis of silver nanoparticles from Valeriana jatamansi shoots extract and its antimicrobial activity
- Characterization and biological activities of synthesized zinc oxide nanoparticles using the extract of Acantholimon serotinum
- Effect of calcination temperature on rare earth tailing catalysts for catalytic methane combustion
- Enhanced diuretic action of furosemide by complexation with β-cyclodextrin in the presence of sodium lauryl sulfate
- Development of chitosan/agar-silver nanoparticles-coated paper for antibacterial application
- Preparation, characterization, and catalytic performance of Pd–Ni/AC bimetallic nano-catalysts
- Acid red G dye removal from aqueous solutions by porous ceramsite produced from solid wastes: Batch and fixed-bed studies
- Review Articles
- Recent advances in the catalytic applications of GO/rGO for green organic synthesis

