Abstract
Objectives
The purpose of the study is to estimate factors affecting survival in prenatally diagnosed hypoplastic left heart syndrome (HLHS) and echocardiographic features predicting poor prognosis and early neonatal death.
Methods
This study was designed as a retrospective cohort study. Cases of hypoplastic left heart syndrome diagnosed in the prenatal period between 2014 and 2023 were extracted from electronic medical records. Demographic data, echocardiographic features, results of genetic testing, pregnancy outcomes, and postnatal outcomes were analyzed.
Results
Eighty-three prenatally diagnosed fetal HLHS cases were analyzed. Overall, survival during the study period was 26.5 %, and survival among live births was 35.4 %. Survival analysis has shown that the majority of deaths occurred during the neonatal period. Out of 62 live births, 47 had Norwood procedures, six had balloon procedures and three had hybrid procedures. Eleven out of 47 who had the Norwood procedures went on to have a Glenn operation, and only three had full Fontan palliation. The presence of additional extra-cardiac anomaly, need for extracorporeal membrane oxygenation (ECMO), bidirectional flow at pulmonary veins on color Doppler, and low birth weight are associated with survival and early neonatal death. Tricuspid regurgitation, restrictive foramen ovale, and fetal growth restriction (FGR) are not associated with survival. HLHS evolved from critical aortic stenosis has better survival rates.
Conclusions
Extra-cardiac anomaly, need for ECMO, bidirectional flow at pulmonary veins, and low birth weight were negatively associated with survival and early neonatal death. The survival rate was higher among HLHS cases that had evolved from critical aortic stenosis.
Introduction
Hypoplastic left heart syndrome (HLHS) is a rare congenital heart malformation. The syndrome includes a hypoplastic left ventricle and its components, a hypoplastic left outflow tract, and a dominant single right ventricle [1]. Mitral valve atresia/hypoplasia/stenosis, and aortic atresia/hypoplasia/stenosis are possible pathologies associated with the hypoplastic left ventricle [2]. Congenital aortic stenosis (AS) can develop during mid-gestation leading to severe cardiac dysfunction, and a substantial proportion of affected fetuses will develop hypoplastic left heart syndrome at birth [3]. Because of the abnormal four-chamber view prenatal diagnosis rates for HLHS are high with fetal echocardiogram [4]. Newborns with HLHS are symptomatic within the first few days of life and the disease is fatal without intervention. There are two surgical procedures for treatment; univentricular palliation and primary cardiac transplantation. Univentricular palliation surgery has three stages: in neonatal period stage I (Norwood operation); at 6–8 months of age stage II (Glenn); and between 18 months and 5 years of age (most commonly around 4 years) stage III (Fontan) [1]. The hybrid procedure is also an option for stage I palliation. Survival is currently around 65 % at 5 years of age and 55 % at 10 years of age [5].
Prenatal counseling is important to give information to parents about mortality and long-term morbidity. The counseling should cover prenatal factors that increase poor prognosis. Additionally, the detection of prenatal factors that are associated with poor prognosis is important for the families that choose termination of pregnancy. Restrictive/intact atrial septum (RAS) and tricuspid regurgitation are considered prenatal factors for poor prognosis [6].
Most cases of hypoplastic left heart syndrome are diagnosed prenatally (53–88 %) [6]. Also, HLHS is responsible for 23 % of all cardiac deaths in the first week of life [7]. Our aim in this study is to estimate factors affecting survival, and echocardiographic features predicting poor prognosis and early neonatal death.
Subjects and methods
This study was designed as a retrospective cohort study. Cases of hypoplastic left heart syndrome (HLHS) diagnosed in the prenatal period between January 2014 and March 2023 were extracted from electronic medical records.
Our fetal medicine unit is a referral center for complex cardiac abnormalities. All cases referred undergo fetal echocardiography and detailed anatomy scan by fetal medicine experts. We collaborate with pediatric cardiology and every case is evaluated both by fetal medicine and pediatric cardiology. Women are followed up monthly with fetal echocardiography and routine antenatal care. By the time of delivery, a multidisciplinary team of fetal medicine, neonatology, and pediatric cardiology manages the peripartum and postnatal periods. All newborns are admitted to the neonatal intensive care unit (NICU) and then transferred to the pediatric cardiology intensive care unit if needed.
Hypoplastic left heart syndrome was diagnosed as a significant underdevelopment of the left ventricle and the left ventricular outflow tract. The cardiac abnormalities that cause hypoplastic left heart such as double outlet right ventricle were excluded. Critical aortic stenosis evolving hypoplastic left heart syndrome was included, we defined it as hypoplastic left ventricle with endocardial fibroelastosis, in which the right ventricle forms the apex of the heart and has a stenotic aortic valve. Maternal (age, gravidity, parity, comorbidity, mode of conception, consanguinity, mode of delivery), fetal (time of birth, time of diagnosis, termination, results of genetic analysis, fetal loss), postnatal (birthweight, sex, APGAR scores, operation procedures, time of operation, need for extracorporeal membrane oxygenation (ECMO), length of NICU, survive after operation) and ultrasound findings (fetal growth, extra-cardiac abnormalities) were collected from medical records, also fetal echocardiography parameters were extracted from electronic fetal ecocardiography reports. Restricted foramen ovale (RAS) was defined as a narrow or sealed shape foramen ovale and the diagnosis was reviewed and confirmed by O.D. and I.K.Y. Gestational age was determined based on the last menstrual period (LMP) and dating by first-trimester crown-lump length (CRL) if there was a discrepancy or the date of LMP was unknown. Follow-up ultrasounds were reviewed for any change in diagnoses. All ultrasonographic assessments were done using the General Electric Voluson E6 ultrasound system. Surgical outcomes (if applicable), mortality, and survival were analyzed. Neonatal death was defined as death in the first 28 days of life, early neonatal death as death in the 7 days of life, and infant death as death in the first year.
Our primary outcome was estimating predictors of survival and determining which echocardiographic features and prenatal risk factors are associated with poor prognosis and neonatal death.
Measures of association for categorical variables were analyzed with Chi-square and Fisher Exact test. The distributional normality of the variables was assessed by using the Shapiro-Wilk test. Skewed distributions of continuous variables in groups were compared by the Wilcoxon-Rank Sum test. Logistic regression analysis was performed to calculate odds ratios (ORs) and 95 % confidence intervals (CIs) of binary outcomes. Linear regression analysis was performed to calculate the effect sizes of continuous variables. Cumulative survival was estimated using Kaplan-Meier analysis. All analyses were performed using STATA software, version 17.0 Basic Edition (Copyright 1985–2021 StataCorp LLC). A p-value of <0.05 was considered statistically significant.
Results
A total of 97 patients were diagnosed with HLHS in the prenatal period, during the study period. Nine patients were excluded because of insufficient data and five were excluded from the double outlet right ventricle with hypoplastic left ventricle. Eighty-three prenatally diagnosed fetal HLHS cases were analyzed. Case frequency based on the year of diagnosis is shown in Figure 1.

Distribution of prenatal diagnosis of HLHS based on year of diagnosis.
Overall, survival during the study period was 26.5 %, and survival among live births was 35.4 %. All patients who had aneuploidy as the result of genetic testing opted for termination of pregnancy, except one case with monosomy X. Out of 62 live births, 47 (75,8 %) had Norwood procedures, 6 (0.9 %) had balloon procedures and 3 (0.04 %) had hybrid procedures. Six neonates had died before any cardiac intervention because of hemodynamic collapse. Eleven out of 47 who had the Norwood procedures went on to have a Glenn operation, and only three had full Fontan palliation. Survival analysis has shown that the majority of deaths occurred during the neonatal period. Kaplan-Meier survival plot is shown in Figure 2.

Survival analysis of the study cohort.
Forty seven cases had Norwood operation, and 31 (65.9 %) died. Among 33 cases who could not survive until Glenn operation, therefore only had Norwood surgery, 30 (90.9 %) died, and three were still surviving and waiting for Glenn operation during the study period (Figure 3).
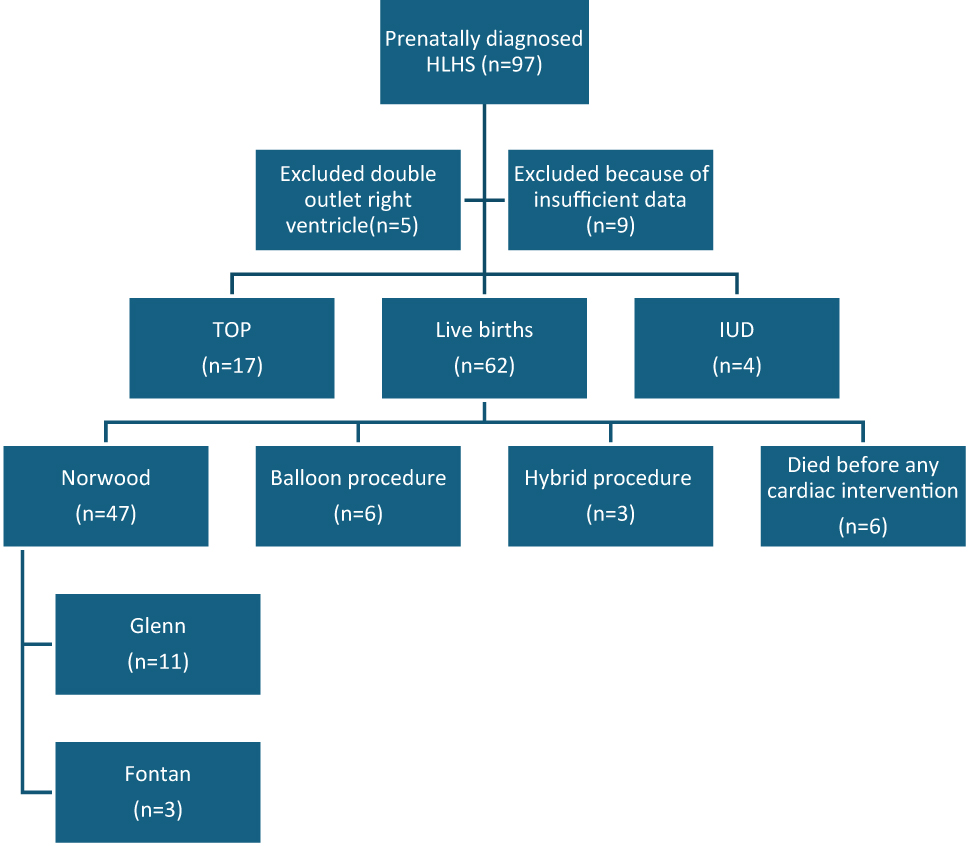
Distribution of cases.
Clinical characteristics and pregnancy outcomes of the study cohort are shown in Table 1. Twenty two patients had genetic sampling, and 15 of 22 had normal karyotype and microarray analysis. There were Turner syndrome in two patients, Trisomy 13 in one patient, and Trisomy 18 in two patients. Two patients had copy number variants: 11p.11.12 1.7 Mb duplication heterozygot variant, 15q11.1q11.2 2 Mb deletion heterozygot variant. In the study cohort, there were 62 live births, 17 terminancy of pregnancies, and four intra-uterine fetal demises. Thirty one of 41 post-natal deaths were neonatal death.
Clinical characteristics and pregnancy outcomes of the study cohort.
| Age, years | 28 ± 6 |
| Gravidity | 2 (1–8) |
| Parity | 1 (0–6) |
| Mode of conception | |
| Spontaneous | 81 (97.5) |
| IVF | 2 (2.4) |
| Consangineous marriage | 13 (16) |
| GA at diagnosis/referral, weeks | 26 (12–39) |
| Genetic testing | 22 (26) |
| Genetic abnormalitya | |
| Absent | 15 (68.1) |
| Trisomy 13 | 1 (4.5) |
| Trisomy 18 | 2 (9.1) |
| Monosomy X | 2 (9.1) |
| CNVb | 2 (20) |
| Fetal sex | |
| Female | 21 (27.3) |
| Male | 56 (72.7) |
| Outcome of pregnancy | |
| TOP | 17 (20.5) |
| Livebirth | 62 (74.7) |
| IUFD | 4 (4.8) |
| GA at delivery, weeks | 38 ± 2 |
| Birthweight, grams | 3,062 ± 561 |
| Preterm birth | 6 (9.1) |
| Before 34th week | 1 (1.5) |
| Between 34 and 37th week | 5 (7.6) |
| Mode of delivery | |
| Vaginal birth | 29 (43.9) |
| Cesarean section | 37 (56.1) |
| Postnatal death | 40 (64.5) |
| Neonatal death | 31 (50) |
| Infant death | 9 (14.5) |
| APGAR | |
| 1st minute APGAR | 8 (6–8) |
| 5th minute APGAR | 9 (8–9) |
-
IVF, in vitro fertilization; GA, gestational age; CNV, copy number variant; TOP, termination of pregnancy; IUFD, intrauterine fetal demise. Data presented as mean ± SD, median (range), or n (percentage). aOut of 22 cases who had pre- or postnatal genetic testing. bOut of 10 cases who had chromosomal microarray analysis.
A comparison of prenatal ultrasonographic and echocardiographic features based on survival is shown in Table 2. Fetal growth restriction (p: 0.311), tricuspid valve regurgitation (p: 0.596), restrictive interatrial septum (RAS) (p: 0.234) are not associated with survival, and extracardiac anomaly (p: 0.05), bidirectional flow across pulmonary veins (p: 0.037) are negatively associated with survival. HLHS secondary to critical aortic stenosis has a higher survival rate (p: 0.023).
Comparison of prenatal ultrasonographic and echocardiographic features based on survival.
| Postnatal death (n=40) | Survival (n=22) | p-Value | |
|---|---|---|---|
| FGR | 5 (12.5) | 1 (4.5) | 0.311 |
| Extracardiac anomaly | 12 (30) | 2 (9.1) | 0.05 |
| Tricuspid valve regurgitation | 21 (52.5) | 10 (45.4) | 0.596 |
| Restrictive foramen ovale | 15 (37.5) | 5 (22.7) | 0.234 |
| HLHS secondary to criticial aortic stenosis | 6 (15) | 9 (40.9) | 0.023 |
| Bidirectional flow across pulmonary veins | 7 (17.5) | 0 | 0.037 |
-
FGR, fetal growth restriction; HLHS, hypoplastic left heart syndrome. Data is presented as n (percentage).
A comparison of maternal and neonatal characteristics based on survival is shown in Table 3. Birthweight under 3,000 g (p: 0.002), length of stay at neonatal intensive care unit (NICU) (p: 0.0004), and need for extracorporeal membrane oxygenation (ECMO) (p: 0.037) are negatively associated with survival. Also, logistic and linear regression analysis to estimate predictors of postnatal death is detailed in Table 4.
Comparison of maternal and neonatal characteristics based on survival.
| Postnatal death (n=40) | Survival (n=22) | p-Value | |
|---|---|---|---|
| Birthweight, g | 2,935 ± 484 | 3,353 ± 451 | 0.002 |
| Maternal age, years | 27 ± 6 | 29 ± 5 | 0.157 |
| GA at diagnosis/referral, weeks | 29 (14–39) | 30 (16–39) | 0.288 |
| GA at delivery, weeks | 38 (32–40) | 38 (37–40) | 0.802 |
| Age at first intervention, daysa | 4 ± 2 | 5 ± 3 | 0.031 |
| LOS at NICU, daysb | 10 (1–38) | 18 (8–55) | 0.0004 |
| Need for ECMO | 7 (17.5) | 0 | 0.037 |
-
GA, gestational age; LOS, length of stay; NICU, neonatal intensive care unit; ECMO, extracorporeal membrane oxygenation. Data presented as mean ± SD, median (range), or n (percentage). aEither Norwood surgery or angiographic balloon procedure as a part of a hybrid surgical approach. bTotal duration pre- and post-surgery (if applicable).
Logistic and linear regression analysis to estimate predictors of postnatal death.
| OR (95 % CI) | |
|---|---|
| Extracardiac anomaly | 4.3 (0.9–22.6) |
| HLHS secondary to criticial aortic stenosis | 0.3 (0.1–0.8) |
| Age at first intervention, daysa | 0.05 (0.005–0.1) |
| LOS at NICUb | 0.02 (0.007–0.02) |
| Birthweight, gc | 0.03 (0.01–0.06) |
-
OR, odds ratio; CI, confidence interval; HLHS, hypoplastic left heart syndrome; LOS, length of stay; NICU, neonatal intensive care unit. The predictors ‘bidirectional flow across pulmonary veins’ and ‘Need for ECMO’ were not added to the analysis since they predict postnatal death perfectly from a statistical point of view. aEither Norwood surgery or angiographic balloon procedure as a part of a hybrid surgical approach. bTotal duration pre- and post-surgery (if applicable). cResults of linear regression for birthweight were reported as per 100 g difference.
In our study, four patients had gastrointestinal, eight patients had central nervous system, seven patients had genitourinary, eight patients had musculoskeletal, seven patients had placenta-umbilical cord, three patients had facial anomalies, and 10 patients had multisystem anomalies. The presence of an additional extra-cardiac anomaly is associated with survival and neonatal death (p: 0.05). Extra-cardiac malformations are detailed in Table 5.
Extra-cardiac malformations.
| Gastrointestinal | 4 (0.04 %) |
| Intestinal volvulus | 1 |
| Echogenic bowel | 1 |
| Hepatic calcifications | 1 |
| Anal atresia | 1 |
| Central nervous system | 8 (0.09 %) |
| Ventriculomegaly | 1 |
| Choroid plexus cysts | 3 |
| Arachnoid cyst | 1 |
| Alobar holoprosencephaly | 1 |
| Agenesis of the corpus callosum | 2 |
| Genitourinary | 7 (0.08 %) |
| Renal agenesis | 1 |
| Pelviectasis | 1 |
| Horse-shoe kidney | 1 |
| Duplicated collecting system | 1 |
| Megacystis | 1 |
| Multicystic dysplastic kidney | 1 |
| Hypospadias | 1 |
| Musculoskeletal | 8 (0.09 %) |
| Brachycephaly | 1 |
| Polydactyly | 1 |
| Clinodactyly | 1 |
| Hemivertebrae | 1 |
| Clubfoot | 1 |
| Diaphragmatic hernia | 1 |
| Diaphragmatic eventration | 1 |
| Omphalocele | 1 |
| Placenta-umbilical cord anomalies | 7 (0.08 %) |
| Single umbilical artery | 5 |
| Placenta previa | 2 |
| Facial | 3 (0.03 %) |
| Micrognathia | 1 |
| Cleft lip and palate | 1 |
| Hypoplasia of nasal bone | 1 |
-
Data is presented as n (percentage).
Discussion
Prenatal counseling is important and termination of pregnancy is one of the options. So it is important to understand the poor prognostic factors in utero for counseling with parents. In our study, we found low birth weight, bidirectional flow at pulmonary veins on color Doppler, and extra-cardiac abnormality as significant poor prognostic factors. Critical aortic stenosis evolving HLHS group has better prognosis and survival rates.
In literature; tricuspid regurgitation (TR) is one of the prognostic factors. Tricuspid regurgitation has been considered a significant mortality risk factor, particularly for HLHS patients who develop TR at an earlier age [8]. Tricuspid regurgitation requiring surgical treatment occurs in up to 25 % of patients with hypoplastic left heart syndrome within 10 years and has been identified as a risk factor for death [9]. In our study, there are 21 newborns with TR in the postnatal death group and 10 newborns in the survival group. Nine patients have TR in the early neonatal death group. However, TR is not associated with survival statistically.
Restrictive/intact atrial septum is seen in about 6 % of all HLHS patients and continues to increase mortality rates in the single-ventricle palliation group for hypoplastic left heart syndrome [10]. Also, a recent meta-analysis shows that RAS does not differ in survival rates and neonatal death [11]. In our study, RAS was found in 15 patients and 7 of 15 died in the neonatal period, but RAS is not associated with survival, statistically. The pulmonary vascular changes secondary to RAS, resulting in pulmonary arterial hypertension, complicate the early perioperative period and may be important for survival [12]. Bidirectional flow across pulmonary veins on color Doppler may be a good emergent intervention predictor [13]. In our study in the RAS group, there is bidirectional flow across pulmonary veins in seven patients. None of the cases with bidirectional flow across pulmonary veins had survived, therefore the predictor ‘bidirectional flow across pulmonary veins’ was not added to the analysis since it predicts postnatal death perfectly from a statistical point of view.
Total anomalous pulmonary venous connection (TAPVC) was found in five patients in postnatal echocardiography. One of five patients was diagnosed as TAPVC prenatally. Two of five died postnatally before surgery, and two of five died in 7 days of life after surgery. One of five died 38 days after surgery. TAPVC seems to be one of the poor prognostic factors. In a recent study; thirty-day mortality after stage 1 pallation surgery was significantly increased in patients with TAPVC vs. without TAPVC [14].
Levoatrial cardinal vein (LACV) is an anomalous connection between the left atrium or pulmonary veins and any systemic vein that can be seen in HLHS cases. LACV in the presence of HLHS has an unfavorable prognosis [15]. In our study, one fetus that was diagnosed HLHS with mitral atresia and aortic atresia had LACV, mild tricuspid regurgitation, restrictive atrial septum, bidirectional flow across pulmonary veins, and pericardial effusion as additional echocardiographic findings. Postnatal echocardiography confirmed the diagnosis and obstructive TAPVC was detected. Unfortunately, the neonate died after 3 h because of cardiovascular collapse.
Patients in our study population prefer to continue pregnancy because of their cultural, socioeconomic, and religious beliefs. Hence, the number of genetic samples and termination of pregnancy are low. In literature Turner syndrome (45, X; monosomy X) and deletions of 11q are mostly associated with left-sided lesions and HLHS [16]. We found Turner syndrome in two patients, Trisomy 13 in one patient, and Trisomy 18 in two patients. Fifteen of the patients had normal karyotype. Two patients had copy number variants; 11p.11.12 1.7 Mb duplication heterozygous variant, 15q11.1q11.2 2 Mb deletion heterozygous variant. HLHS was detected more frequently in male fetuses, consistent with the literature.
Although postnatal survival rates have increased compared to the past with developing surgical techniques, HLHS remains a major disease-causing neonatal death. There has been a significant increase in overall survival to 61 % [6]. Some centers are reporting a Stage 1 Norwood palliation survival rate of 90 % [17]. Some centers report following a Norwood, an interstage mortality rate of 5–15 % [18]. Despite this in our study survival among live births was 35.4 %. Eleven out of 47 who had the Norwood procedures went on to have a Glenn operation, and only three had full Fontan palliation.
The low number of the study cohort, and excluding fetal interventions such as atrial septostomy or aortic valvuloplasty are the limitations of the study. Also, we did not analyze long-term morbidity for survivors.
In conclusion, extra-cardiac anomaly, need for ECMO, bidirectional flow at pulmonary veins, and low birth weight are negatively associated with survival and early neonatal death in our study. Critical aortic stenosis evolving HLHS has a better prognosis. Tricuspid regurgitation and restrictive interatrial septum are not associated with survival and early neonatal death but should be evaluated in HLHS patients. A careful fetal echocardiogram may be performed to identify the total anomalous pulmonary venous connection and levoatrial cardinal vein.
Survival rates are reported to be increasing day by day, our rates were found to be low in our study, incompatible with the literature. Factors affecting postnatal prognosis should be evaluated with careful fetal echocardiographic examination. Parents should be informed about risk factors.
-
Research ethics: Research involving human subjects complied with all relevant national regulations, and institutional policies and is in accordance with the tenets of the Helsinki Declaration (as revised in 2013), and has been approved by the authors’ Institutional Review Board (Zeynep Kamil Women and Children Diseases Training and Research Hospital) (Decision-Nr.: 137/2022).
-
Informed consent: Informed consent was obtained from all individuals included in this study.
-
Author contributions: All authors have accepted responsibility for the entire content of this manuscript and approved its submission.
-
Use of Large Language Models, AI and Machine Learning Tools: None declared.
-
Conflict of interest: The authors state no conflict of interest.
-
Research funding: None declared.
-
Data availability: Not applicable.
References
1. Ma, J, Yuan, Y, Zhang, L, Chen, S, Cao, H, Hong, L, et al.. Evaluation of right ventricular function and myocardial microstructure in fetal hypoplastic left heart syndrome. J Clin Med 2022;11:4456. https://doi.org/10.3390/jcm11154456.Search in Google Scholar PubMed PubMed Central
2. Wald, RM, Mertens, LL. Hypoplastic left heart syndrome across the lifespan: clinical considerations for care of the fetus, child, and adult. Can J Cardiol 2022;38:930–45. https://doi.org/10.1016/j.cjca.2022.04.028.Search in Google Scholar PubMed
3. Ong, CW, Ren, M, Wiputra, H, Mojumder, J, Chan, WX, Tulzer, A, et al.. Biomechanics of human fetal hearts with critical aortic stenosis. Ann Biomed Eng 2021;49:1364–79. https://doi.org/10.1007/s10439-020-02683-x.Search in Google Scholar PubMed PubMed Central
4. Toma, D, Moldovan, E, Gozar, L. The impact of prenatal diagnosis in the evolution of newborns with congenital heart disease. J Crit Care Med 2023;9:6–11. https://doi.org/10.2478/jccm-2023-0007.Search in Google Scholar PubMed PubMed Central
5. Birla, AK, Brimmer, S, Short, WD, Olutoye, OO, Shar, JA, Lalwani, S, et al.. Current state of the art in hypoplastic left heart syndrome. Front Cardiovasc Med 2022;9:878266. https://doi.org/10.3389/fcvm.2022.878266.Search in Google Scholar PubMed PubMed Central
6. Ramcharan, T, Quintero, DB, Stickley, J, Poole, E, Miller, P, Desai, T, et al.. Medium-term outcome of prenatally diagnosed hypoplastic left-heart syndrome and impact of a restrictive atrial septum diagnosed in-utero. Pediatr Cardiol 2023;44:1217–25. https://doi.org/10.1007/s00246-023-03184-z.Search in Google Scholar PubMed
7. Yabrodi, M, Mastropietro, CW. Hypoplastic left heart syndrome: from comfort care to long-term survival. Pediatr Res 2017;81:142–9. https://doi.org/10.1038/pr.2016.194.Search in Google Scholar PubMed PubMed Central
8. Ross, CJ, Mir, A, Burkhart, HM, Holzapfel, GA, Lee, CH. Tricuspid valve regurgitation in hypoplastic left heart syndrome: current insights and future perspectives. J Cardiovasc Dev Dis 2023;10:111. https://doi.org/10.3390/jcdd10030111.Search in Google Scholar PubMed PubMed Central
9. Pigula, FA, Mettler, B. Management of tricuspid regurgitation in patients with hypoplastic left heart syndrome. Semin Thorac Cardiovasc Surg 2017;29:64–9. https://doi.org/10.1053/j.semtcvs.2017.02.004.Search in Google Scholar PubMed
10. Sood, V, Zampi, JD, Romano, JC. Hypoplastic left heart syndrome with an intact atrial septum. JTCVS Open 2020;1:51–6. https://doi.org/10.1016/j.xjon.2020.03.001.Search in Google Scholar PubMed PubMed Central
11. Mustafa, HJ, Aghajani, F, Jawwad, M, Shah, N, Abuhamad, A, Khalil, A. Fetal cardiac intervention in hypoplastic left heart syndrome with intact or restrictive atrial septum, systematic review, and meta-analysis. Prenat Diagn 2024;44:747–57. https://doi.org/10.1002/pd.6420.Search in Google Scholar PubMed
12. Manning, N, Archer, N. Fetal pulmonary venous Doppler flow patterns in hypoplastic left heart syndrome. Heart 2008;94:1374–5. https://doi.org/10.1136/hrt.2007.138412.Search in Google Scholar PubMed
13. Jadczak, A, Respondek-Liberska, M, Sokołowski, Ł, Chrzanowski, J, Rizzo, G, Araujo Júnior, E, et al.. Hypoplastic left heart syndrome with prenatally diagnosed foramen ovale restriction: diagnosis, management and outcome. J Matern Fetal Neonatal Med 2022;35:291–8. https://doi.org/10.1080/14767058.2020.1716717.Search in Google Scholar PubMed
14. Heinisch, PP, Kido, T, Burri, M, Kornyeva, A, Mertin, J, Vodiskar, J, et al.. Impact of total anomalous pulmonary venous connection in staged single ventricle palliation. Ann Thorac Surg 2023;115:1213–21. https://doi.org/10.1016/j.athoracsur.2022.07.021.Search in Google Scholar PubMed
15. Hellmund, A, Berg, C, Herberg, U, Geipel, A, Kempe, A, Gembruch, U. Levoatrial cardinal vein in a series of five prenatal cases with hypoplastic left heart syndrome and intact atrial septum. Ultraschall Med 2017;38:206–11. https://doi.org/10.1055/s-0034-1399695.Search in Google Scholar PubMed
16. Theis, JL, Olson, TM. Whole genome sequencing in hypoplastic left heart syndrome. J Cardiovasc Dev Dis 2022;9:117. https://doi.org/10.3390/jcdd9040117.Search in Google Scholar PubMed PubMed Central
17. Roeleveld, PP, Axelrod, DM, Klugman, D, Jones, MB, Chanani, NK, Rossano, JW, et al.. Hypoplastic left heart syndrome: from fetus to fontan. Cardiol Young 2018;28:1275–88. https://doi.org/10.1017/s104795111800135x.Search in Google Scholar
18. Rao, PS. Management of congenital heart disease: state of the art-part II-cyanotic heart defects. Children 2019;6:54. https://doi.org/10.3390/children6040054.Search in Google Scholar PubMed PubMed Central
© 2025 the author(s), published by De Gruyter, Berlin/Boston
This work is licensed under the Creative Commons Attribution 4.0 International License.
Articles in the same Issue
- Frontmatter
- Review
- Sex differences in lung function of adolescents or young adults born prematurely or of very low birth weight: a systematic review
- Original Articles – Obstetrics
- Shifts in peak month of births and socio-economic factors: a study of divided and reunified Germany 1950–2022
- The predictive role of serial transperineal sonography during the first stage of labor for cesarean section
- Gestational weight gain and obstetric outcomes in women with obesity in an inner-city population
- Placental growth factor as a predictive marker of preeclampsia in twin pregnancy
- Learning curve for the perinatal outcomes of radiofrequency ablation for selective fetal reduction: a single-center, 10-year experience from 2013 to 2023
- External validation of a non-invasive vaginal tool to assess the risk of intra-amniotic inflammation in pregnant women with preterm labor and intact membranes
- Placental fetal vascular malperfusion in maternal diabetes mellitus
- The importance of the cerebro-placental ratio at term for predicting adverse perinatal outcomes in appropriate for gestational age fetuses
- Comparing achievability and reproducibility of pulsed wave Doppler and tissue Doppler myocardial performance index and spatiotemporal image correlation annular plane systolic excursion in the cardiac function assessment of normal pregnancies
- Characteristics of the pregnancy and labour course in women who underwent COVID-19 during pregnancy
- Original Articles – Fetus
- Sonographic visualization and measurement of the fetal optic chiasm and optic tract and association with the cavum septum pellucidum
- The association among fetal head position, fetal head rotation and descent during the progress of labor: a clinical study of an ultrasound-based longitudinal cohort study in nulliparous women
- Fetal hypoplastic left heart syndrome: key factors shaping prognosis
- The value of ultrasound spectra of middle cerebral artery and umbilical artery blood flow in adverse pregnancy outcomes
- Original Articles – Neonates
- A family-centric, comprehensive nurse-led home oxygen programme for neonatal chronic lung disease: home oxygen policy evaluation (HOPE) study
- Effects of a respiratory function indicator light on visual attention and ventilation quality during neonatal resuscitation: a randomised controlled crossover simulation trial
- Short Communication
- Incidence and awareness of dysphoric milk ejection reflex (DMER)
Articles in the same Issue
- Frontmatter
- Review
- Sex differences in lung function of adolescents or young adults born prematurely or of very low birth weight: a systematic review
- Original Articles – Obstetrics
- Shifts in peak month of births and socio-economic factors: a study of divided and reunified Germany 1950–2022
- The predictive role of serial transperineal sonography during the first stage of labor for cesarean section
- Gestational weight gain and obstetric outcomes in women with obesity in an inner-city population
- Placental growth factor as a predictive marker of preeclampsia in twin pregnancy
- Learning curve for the perinatal outcomes of radiofrequency ablation for selective fetal reduction: a single-center, 10-year experience from 2013 to 2023
- External validation of a non-invasive vaginal tool to assess the risk of intra-amniotic inflammation in pregnant women with preterm labor and intact membranes
- Placental fetal vascular malperfusion in maternal diabetes mellitus
- The importance of the cerebro-placental ratio at term for predicting adverse perinatal outcomes in appropriate for gestational age fetuses
- Comparing achievability and reproducibility of pulsed wave Doppler and tissue Doppler myocardial performance index and spatiotemporal image correlation annular plane systolic excursion in the cardiac function assessment of normal pregnancies
- Characteristics of the pregnancy and labour course in women who underwent COVID-19 during pregnancy
- Original Articles – Fetus
- Sonographic visualization and measurement of the fetal optic chiasm and optic tract and association with the cavum septum pellucidum
- The association among fetal head position, fetal head rotation and descent during the progress of labor: a clinical study of an ultrasound-based longitudinal cohort study in nulliparous women
- Fetal hypoplastic left heart syndrome: key factors shaping prognosis
- The value of ultrasound spectra of middle cerebral artery and umbilical artery blood flow in adverse pregnancy outcomes
- Original Articles – Neonates
- A family-centric, comprehensive nurse-led home oxygen programme for neonatal chronic lung disease: home oxygen policy evaluation (HOPE) study
- Effects of a respiratory function indicator light on visual attention and ventilation quality during neonatal resuscitation: a randomised controlled crossover simulation trial
- Short Communication
- Incidence and awareness of dysphoric milk ejection reflex (DMER)

