Abstract
Activated carbons are widely used adsorbents for hydrogen sulfide (H2S) removal in the field of gas stream purification in industry. Five commercially available wood-, coal-, and coconut shell-based activated carbons, prepared by phosphoric acid activation and steam activation, were chosen as adsorbents in an effort to elucidate the effect of the pore structure on H2S adsorption of activated carbons in an oxygen-free gas stream. The adsorption experiments were conducted in an adsorption column, and the breakthrough curves of the activated carbons were examined. The results showed that adsorption of H2S by activated carbon under oxygen-free conditions was dependent on the microporous structure, particularly micropores with sizes of <1 nm. The surface area of CS-3 is 1,792 m2·g−1. The H2S breakthrough capacities of WS-1, BS-1, CS-1, CS-2, and CS-3 were 0.006, 0.01, 0.018, 0.021, and 0.026 g·cm−3 under oxygen-free conditions. The adsorption experiments of the KOH- and CuO-loaded activated carbons further confirmed the significance of micropores with sizes of <1 nm for H2S removal. Also, it was found that pre-adsorption or co-adsorption of carbon tetrachloride led to poisoning of the activated carbon for H2S adsorption due to a remarkable reduction in the micropore volume. Therefore, steam-activated materials with highly developed micropore structures were favorable adsorbents for H2S removal because of their high adsorption capacities and reduced fire risk in an oxygen-free gas stream.
1 Introduction
Hydrogen sulfide (H2S) is an acidic and poisonous gas that is frequently found at various concentrations in industrial gas streams such as natural gas, fermentation gas, syngas, and coal-derived gas [1,2,3,4,5]. It is problematic in industrial processes because it poisons catalysts and readily corrodes metallic equipment and pipelines. Accordingly, removal of H2S from industrial gas streams is an indispensable step in areas such as semiconductor manufacture, high-quality oil and gas, and green catalysis [6]. Adsorption is the most commonly used method to remove H2S from gas streams containing concentrations typically below 100 ppm.
Activated carbon is the most widely used adsorbent for H2S in the purification of gas streams [7,8,9]. Generally, removal of H2S requires that the activated carbon be loaded with a catalyst, which is commonly an alkaline substance or metal oxide, because the pristine-activated carbon has poor adsorption capacity for H2S [10,11,12,13,14]. Moreover, extensive investigations have demonstrated that H2S removal by activated carbon involves complex physical and chemical processes. Therefore, the adsorption capacity of activated carbon for H2S depends not only on its physicochemical properties but also on the adsorption parameters [15,16,17]. In particular, in the presence of oxygen, H2S can be oxidized to S, SO3 2−, or SO4 2− species during adsorption by activated carbon impregnated with the catalyst [18,19,20], resulting in remarkably enhanced removal of H2S. Accordingly, air or oxygen is a key factor in the removal of H2S by activated carbon and, thus usually pumped into the gas stream for industrial desulfurization.
However, oxygen and air are not permitted even at quite low concentrations in some gas streams, including natural gas, gaseous carbon dioxide in the beverage industry, and supercritical gas. Under such oxygen-free conditions, the adsorption capacity of activated carbon for H2S is dramatically decreased even if catalyst-loaded activated carbon is used. Unfavorably, the H2S-saturated activated carbon readily ignites during replacement of used adsorbent due to rapid oxidation in air [21,22]. Consequently, the development of efficient H2S removal technology using activated carbon in oxygen-free conditions is desirable for industrial desulfurization. Based on the adsorption mechanism of activated carbon, it can be inferred that surface chemistry and pore structure determine the adsorption capacity for H2S under oxygen-free conditions. It is generally accepted that a basic surface greatly benefits the adsorption of acidic H2S [23,24,25]. However, the effect of pore structure has yet to be addressed in detail. This investigation focused on the effect of the micropore structure of activated carbon on the ability to remove H2S based on detailed structural analysis. The results demonstrated the significance of micropores of different sizes in the adsorption of gaseous H2S by activated carbon, providing new insight into the adsorption process.
2 Experimental
2.1 Activated carbon adsorbents
Five commercially available activated carbons were selected as adsorbents. These included three coconut shell-based materials with different pore structures (three coconut carbon samples, named CS-1, CS-2, and CS-3, respectively), one bituminous-based activated carbon (bituminous carbon sample, named BS-1), and one wood-based activated carbon (wood carbon sample, named WS-1), which were all purchased from Purestar, China. CS-1, CS-2, CS-3, and BS-1 are manufactured by steam activation, and WS-1 is produced by phosphoric acid activation. All of the materials were crushed and sieved to obtain particles of 1.7–3.35 mm.
The activated carbons were modified by impregnating with a solution of Cu(NO3)2 or a KOH solution. The impregnated Cu2+ activated carbons were heat-treated in a N2 atmosphere at 450°C for 1 h to give 8% CuO-loaded activated carbons. The KOH-impregnated activated carbons were dried in an oven at 85°C. The resultant impregnated activated carbons were denoted as C-CuO and C-KOH.
2.2 Pore size analysis
Nitrogen adsorption–desorption tests were performed at 77 K using an adsorption analyzer (Autosorb IQ10, Quantachrome, USA). The surface area was calculated using the Brunauer–Emmett–Teller (BET) equation, and the total pore volume was obtained at a relative pressure of 0.99. The mesopore volume was obtained using the Barrett, Joyner, and Halenda method. The pore size distribution curves were analyzed using the quenched solid density functional theory (QSDFT) method in terms of the adsorption branch. The ratios of pore volumes of different sizes to the total pore volume were also evaluated based on the pore size distribution of the activated carbons.
2.3 Adsorption test
The breakthrough curves were tested in a vertical adsorption tube (24 mm × 230 mm). Nitrogen containing 1% v/v H2S was used as the gas stream for testing breakthrough curves, which was obtained by diluting raw 5% v/v H2S with nitrogen or carbon tetrachloride (CTC)-loaded nitrogen. The gas stream was calibrated and monitored to maintain a total flow rate of 1,450 ± 20 mL·min−1. All tests used the same carbon volume to keep the same contact time of 5 s. The stream flow was maintained until a breakthrough of 50 ppmv was indicated. The time elapsed from the start to the breakthrough was recorded. The H2S detector was provided by Handa Technology (HD-P900-H2S). The H2S breakthrough capacities (BTC) of the adsorbents were calculated by integrating the areas of the breakthrough curves, expressed as the amount (g) of H2S removed from the vapor stream per volume (cm3) of carbon. The saturated adsorption capacity (SAC) was calculated by increased weight, expressed as the amount (g) of H2S removed from the vapor stream per weight (g) of carbon.
where BTC is the breakthrough capacity (g·cm−3), v is the flow rate (cm3·min−1), C 0 is the initial H2S concentration (ppmv), C t is the breakthrough concentration at time t (ppmv), and V is the volume of activated carbon (cm3).
2.4 Ignition temperature testing
The ignition temperatures of the activated carbons were determined according to the method described in the national testing standard of activated carbons (GB/T 7702.9-2008). The activated carbon was first placed in a vertical tube in an ignition point analyzer (FMX-K8, ZhongHuiTian Cheng, China) in direct contact with an air stream. The air stream was slowly heated until the activated carbon began to ignite. The temperatures of the carbon bed and the air entering the bed were recorded. The ignition temperature is defined as the point at which the carbon temperature suddenly rises above the temperature of the air entering the bed.
3 Results and discussion
3.1 Pore structures of activated carbons
Figure 1(a) shows the N2 adsorption/desorption isotherms of the activated carbons. It can be seen that CS-1, CS-2, and CS-3 all exhibited type I adsorption isotherms with no apparent hysteresis loop, indicative of predominantly micropore structures. The WS-1 and BS-1-activated carbons exhibited type IV adsorption isotherms, which had high adsorption capacity before the knee point in the curves and exhibited an obvious hysteresis loop. The results demonstrated that WS-1 and BS-1 had microporous/mesoporous structures. WS-1 was prepared by phosphoric acid activation, a chemical activation method, and therefore contained a high proportion of mesopores. The pore size distributions of these activated carbons are presented in Figure 1(b). A comparison of the pore size distribution curves indicated that the pores in CS-1, CS-2, and CS-3 were predominantly less than 3 nm, while WS-1 and BS-1 had a wide size distribution in the range of <15 nm, with the predominant pores of <2 nm. A further observation (the inset in Figure 1 (b)) demonstrated that the size of the predominant pores was <2 nm·min−1 CS-1, CS-2, and CS-3. Table 1 lists the pore parameters of the five activated carbons obtained in terms of their adsorption/desorption isotherms. As shown in Table 1, CS-2 and CS-3 had microporous structures but almost negligible mesopore volumes, and CS-1 had predominantly micropore structures with a minority of mesopores. BS-1 had developed micropore and mesopore structures. WS-1 is a type of porous carbon with a predominantly mesopore structure. Therefore, the five activated carbons possess different pore structures, depending on the raw materials and activation methods.

N2 adsorption isotherms (a) and pore size distribution (b) of activated carbons.
Porous parameters of the activated carbons
| BET surface area (m2·g−1) | Total pore volume (cm3·g−1) | Micropore volume (cm3·g−1) | Mesopore volume (cm3·g−1) | Average pore size (nm) | |
|---|---|---|---|---|---|
| WS-1 | 1,315 | 1.149 | 0.359 | 0.665 | 3.49 |
| BS-1 | 1,100 | 0.818 | 0.369 | 0.381 | 2.97 |
| CS-1 | 1,156 | 0.626 | 0.404 | 0.152 | 2.09 |
| CS-2 | 1,371 | 0.645 | 0.590 | 0.032 | 1.87 |
| CS-3 | 1,792 | 0.835 | 0.728 | 0.078 | 1.86 |
3.2 Effect of pore size on H2S adsorption
Figures 2 and 3 show the H2S breakthrough curves and SACs of the activated carbons. Figure 2 shows that the breakthrough times of WS-1, BS-1, CS-1, CS-2, and CS-3 are about 10, 27, 60, 72, and 95 min, respectively. Correspondingly, the H2S BTC values of WS-1, BS-1, CS-1, CS-2, and CS-3 were 0.006, 0.01, 0.018, 0.021, and 0.026 g·cm−3, respectively (Figure 2). The CS-3-activated carbon column exhibited the highest uptake ability toward H2S in an adsorption column, which was about 10 and 4.4 times that of WS-1 in terms of the breakthrough time and adsorption capacity. Based on the results shown in Table 1 and Figures 1 and 2, it is evident that the higher the micropore volume of activated carbons, the larger the adsorption capacity of H2S. This indicates that the micropores of activated carbons play a key role in removing H2S in the activated carbon adsorption column. Figure 4 shows that the adsorption capacity of activated carbons in a fixed bed increased almost linearly as the micropore volume increased, further confirming the significance of the micropores in the adsorption of H2S. Obviously, mesopores have a negligible contribution to the H2S uptake. Accordingly, although WS-1 and BS-1 had a much higher total pore volume than CS-1 and CS-2, they exhibited a much lower H2S adsorption capacity. Similarly, Table 1 and Figures 1 and 2 indicate that the surface area is not an exact parameter to evaluate the H2S adsorption capacity of activated carbons. Clearly, the breakthrough of H2S gas stream through an activated carbon column varied greatly dependent on the activated carbon species. WS-1 has the shortest breakthrough time of about 27 min and the least BTC value. In particular, the adsorption capacity of CS-3 toward H2S was about 4.4 times that of WS-1 in the fixed-bed adsorption process. This indicated that it had about 4.4 times the adsorption capacity. Moreover, we found that micropores played a key role in the adsorption of H2S. CS-3 had a much higher micropore volume (0.728 cm3·g−1) than that of WS-1 (0.359 cm3·g−1). Comparing CS-1, CS-2, and CS-3-activated carbons, which were all prepared by steam activation of coconut shell-based chars, it was found that the activated carbon with a higher micropore volume exhibited a longer breakthrough time.

H2S breakthrough curves of WS-1, BS-1, CS-1, CS-2 and CS-3.

SACs of WS-1, BS-1, CS-1, CS-2 and CS-3.

Relationship between carbon porous properties and H2S adsorption capacity.
Furthermore, it is noteworthy that although there was only a small difference in the micropore volumes of BS-1 and CS-1 (0.369 and 0.404 cm3·g−1, respectively), CS-1 exhibited a much longer breakthrough time of 60 min compared to the 30 min of BS-1. The calculated adsorption capacity of CS-1 was also much higher than that of BS-1. This comparison indicated that the micropore size exerted a significant effect on the adsorption of H2S by activated carbon. In order to elucidate the effect of micropore size on H2S adsorption, a regression analysis was conducted. Based on the pore size distribution determined using the QSDFT model, the total pore volumes of the five activated carbons were calculated, together with the volumes of pores with diameters below 1, 2, and 5 nm. All of the results are listed in Table 2.
Analysis of different micropores
| Pore range (nm) | WS-1 | BS-1 | CS-1 | CS-2 | CS-3 | |||||
|---|---|---|---|---|---|---|---|---|---|---|
| Volume (cm3·g−1) | Ratio (%) | Volume (cm3·g−1) | Ratio (%) | Volume (cm3·g−1) | Ratio (%) | Volume (cm3·g−1) | Ratio (%) | Volume (cm3·g−1) | Ratio (%) | |
| <1 | 0.207 | 16.8 | 0.241 | 34.1 | 0.274 | 47.9 | 0.310 | 52.9 | 0.332 | 43.7 |
| <2 | 0.382 | 31 | 0.258 | 36.5 | 0.378 | 66.1 | 0.382 | 65.2 | 0.495 | 65.1 |
| <5 | 0.596 | 48.3 | 0.416 | 57.8 | 0.424 | 74.1 | 0.449 | 76.6 | 0.722 | 95.0 |
| Total | 1.234 | 0.707 | 0.572 | 0.586 | 0.76 | |||||
To analyze the relationship between the pore volume and H2S adsorption capacity, the data were plotted, as shown in Figure 5. Figure 5 shows that the SAC was significantly increased as the volume of pores with a diameter less than 1 nm increased, with the best regression coefficient of 0.9527. The R 2 values were much lower for volumes of pores of sizes less than 2 and 5 nm, indicating that the volume of pores with a size of <1 nm was most strongly correlated with the H2S adsorption capacity. In addition, the slope of the curve for the volume of pores with a size of <1 nm versus SAC was greater than that of the other two, further confirming that the micropores with a size of <1 nm greatly contribute to H2S adsorption. The adsorption theory predicted that the favorable pores in a porous adsorbent for adsorption are those with sizes of approximately three times the molecular size of the adsorbate [26,27,28]. The molecular diameter of H2S is about 0.36 nm [29], so the most effective pore size for adsorbing H2S is approximately 1 nm, which is consistent with the above-mentioned results. Accordingly, it is certain that the volume of pores with sizes of <1 nm had the most important effect on H2S adsorption.
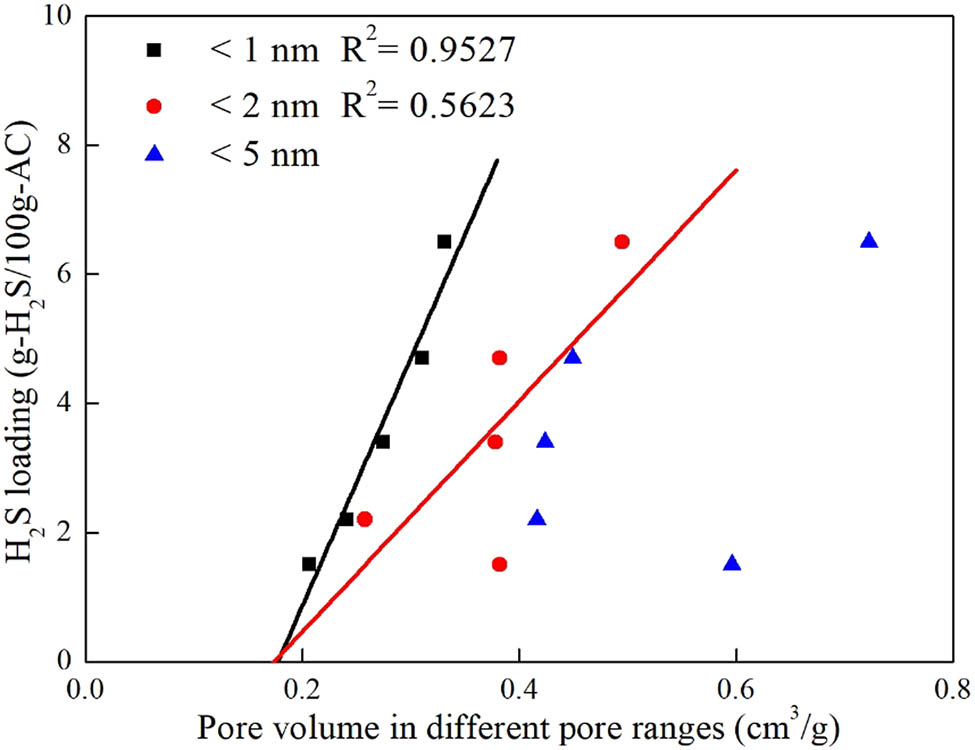
Relationship between the pore volume and H2S adsorption capacity.
Additionally, it is noted that the micropore volume of BS-1 and WS-1 is approximate, and WS-1 has a higher surface area than BS-1, but BS-1 has a much longer breakthrough time and higher adsorption capacity toward H2S in a gas flow. This discrepancy is caused by their surface chemistry. WS-1 was prepared by phosphoric acid activation of wood sawdust, and thus, is characteristic of an acidic surface. BS-1 was fabricated by steam activation of coal, and thus, is of a basic surface. For the acidic H2S, the basic surface favors the uptake of H2S on the surface of activated carbons.
3.3 Effect of loaded activated carbon and pre-adsorption on H2S adsorption
Generally, H2S is not a single gas in the adsorption process, and many other substances are accompanied by the H2S adsorption process, like the biogas, natural gas, and NH3 [30,31,32]. Organic substances are important factors in adsorption. Previously, KOH and CuO were commonly used to improve the uptake of H2S by activated carbon because H2S adsorption could be remarkably promoted by providing basic surfaces for KOH-loaded activated carbons and active sites of chemically adsorbing H2S for the loaded CuO-activated carbons. Therefore, we further investigated the H2S adsorption ability of KOH or CuO-loaded CS-3. The N2 adsorption isotherms of CS-3, C-KOH, and C-CuO are shown in Figure 6, and their pore parameters are listed in Table 3. It is evident that the micropore volume of the activated carbon was remarkably reduced by almost 50% after loading KOH or CuO. Particularly, the pores with sizes of <1 nm were sharply reduced to a nearly negligible value. As a result, the H2S adsorption capacity was decreased from 6.5 g/100 g for CS-3 to 4.4 g/100 g for the KOH-loaded CS-3, and to 3.8 g/100 g for the CuO-loaded CS-3, as shown in Figure 7. Nevertheless, it should be noted that the H2S adsorption experiments in this study were conducted in an oxygen-free stream. In an oxygen-free atmosphere, the catalytic ability of KOH and CuO to convert H2S into S, SO3 2− or SO4 2− was inhibited in the process of removing H2S, which commonly works in an oxygen or air-containing stream [33,34,35].

N2 adsorption isotherms for CS-3, C-KOH, and C-CuO.
Porous properties of CS-3, C-KOH, and C-CuO
| BET surface area (m2·g−1) | Total pore volume (cm3·g−1) | Micropore volume (cm3·g−1) | Pore volume below 1 nm (cm3·g−1) | |
|---|---|---|---|---|
| CS-3 | 1,792 | 0.835 | 0.728 | 0.332 |
| C-KOH | 1,216 | 0.809 | 0.389 | 0.0748 |
| C-CuO | 972 | 0.607 | 0.361 | 0.0591 |

Adsorption capacities of CS-3, C-KOH, and C-CuO.
Considering that the gas stream usually contains organic substances other than H2S, we tested the effect of CTC on the adsorption of H2S by activated carbon. Surprisingly, the breakthrough time was sharply decreased to almost zero when CS-3 pre-adsorbed gaseous CTC or the adsorption was conducted with a gaseous mixture of CTC and H2S. We described this phenomenon as the poisoning of activated carbon for H2S adsorption. This was due to the adsorbed CTC that can occupy to some degree the micropores with sizes of less than 1 nm because the molecular size of CTC was 0.59 nm [36]. This poisoning phenomenon further illustrates the importance of micropores of smaller sizes in the H2S adsorption process. Therefore, it can be predicted that the organic substances of the smaller molecular dimensions contained in the H2S stream produce an obvious detriment on the H2S adsorption (Figure 8).

The effect of CCl4 on H2S adsorption.
3.4 Risk of spent carbon fire
In the industrial process, fire risk is another important issue to be considered in the process of activated carbon for H2S removal. We therefore tested the ignition temperatures of WS-1, BS-1, and CS-3 microporous activated carbons and the KOH- and CuO-loaded activated carbons, C-KOH and C-CuO, as shown in Table 4. It was found that loading KOH or CuO led to an obvious decrease in the ignition temperature. This may be due to catalytic oxidation of carbon by air or oxygen in the presence of KOH or CuO. Clearly, the BS-1 and CS-3 microporous activated carbon materials were safer and exhibited an outstanding advantage in the large-scale industrial process.
Safety parameters of CS-3, BS-1, C-KOH, and C-CuO
| Samples | Ignition temperature (°C) | Spent carbon fire |
|---|---|---|
| CS-3 | 407 | No |
| BS-1 | 459 | No |
| WS-1 | 394 | No |
| C-KOH | 232 | Burn directly |
| C-CuO | 360 | Burn directly |
4 Conclusions
The five activated carbon materials were prepared by phosphoric acid activation and steam activation from coal, coconut shell, and wood. Their breakthrough curves and H2S adsorption capacities were examined using an adsorption column in an oxygen-free stream. The micropore volume of CS-3 is 0.728 cm3·g−1 and the average pore size is 1.86 nm. The H2S breakthrough capacity of coconut shell-based steam-activated carbon was 0.026 g·cm−3 under oxygen-free conditions. The results showed that the micropores with sizes of <1 nm were the main contributors to the uptake of H2S, with mesopores having nearly negligible contribution. The adsorption experiments of the KOH- and CuO-loaded activated carbons and pre-adsorption experiments of tetrachloride further confirmed the significance of the micropores with sizes of <1 nm. In addition, the steam-activated carbons exhibited higher safety due to a higher ignition temperature. The loading of KOH and CuO apparently reduced the ignition temperature of the activated carbons.
Acknowledgments
This study was performed in the framework of the research work in the State Key Laboratory of Chemistry and Utilization of Agriculture and Forestry Biomass in China.
-
Funding information: This study was funded by the National Key Research and Development Program of China (No. 2019YFB1503804).
-
Author contributions: Bin Liu, conceptualization, methodology, formal analysis, software, data curation, resources, writing – original draft, writing – review and editing, and visualization. Songlin Zuo, writing – original draft, writing – review and editing, methodology, supervision, and funding acquisition. The manuscript was written through the equal contributions of all authors. All authors read and approved the final manuscript.
-
Conflict of interest: The authors state no conflict of interest.
-
Compliance with ethical standards: We declare that accepted principles of ethical and professional conduct have been followed.
-
Consent to participate: Not applicable.
-
Consent to publish: Not applicable.
-
Data availability statement: The datasets used in the current available from the corresponding author on reasonable request.
References
[1] Tian, X. F., L. M. Wang, P. Zhang, D. Fu, and Z. Y. Wang. A high efficient absorbent for the separation of H2S from low partial pressure coke oven gas. Environmental Science and Pollution Research, Vol. 28, 2021, pp. 5822–5832.10.1007/s11356-020-10968-wSearch in Google Scholar PubMed
[2] Lin, Y., Y. Li, Z. Xu, J. Guo, and T. Zhu. Carbon consumption and adsorption-regeneration of H2S on activated carbon for coke oven flue gas purification. Environmental Science and Pollution Research, Vol. 28, 2021, pp. 60557–60568.10.1007/s11356-021-14914-2Search in Google Scholar PubMed
[3] Andrade, I. M., G. Moreno, and G. Quijano. Theoretical framework for the estimation of H2S concentration in biogas produced from complex sulfur-rich substrates. Environmental Science and Pollution Research, Vol. 27, 2020, pp. 15959–15966.10.1007/s11356-019-04846-3Search in Google Scholar PubMed
[4] Kandola, I., A. Pogrebnoi, and T. Pogrebnaya. Oldoinyo Lengai Volcanic ash for removal of hydrogen sulfide and ammonia from biogas. Journal of Materials Science & Chemical Engineering, Vol. 6, 2018, pp. 78–93.10.4236/msce.2018.64010Search in Google Scholar
[5] Castellini, E., F. Bernini, L. Sebastianelli, C. I. Sainz-Diaz, and M. Borsari. Interlayer-confined Cu(II) complex as an efficient and long-lasting catalyst for oxidation of H2S on Montmorillonite, minerals. Minerals, Vol. 10, 2020, pp. 1–13.10.3390/min10060510Search in Google Scholar
[6] Zagoruiko, A. and P. Mikenin. Decomposition of hydrogen sulfide into elements in the cyclic chemisorption-catalytic regime. Catalysis Today, Vol. 378, 2021, pp. 176–188.10.1016/j.cattod.2020.12.004Search in Google Scholar
[7] Li, X., X. Q. Wang, L. L. Wang, P. Ning, Y. X. Ma, L. Zhong, et al. Efficient removal of carbonyl sulfur and hydrogen sulfide from blast furnace gas by one-step catalytic process with modified activated carbon. Applied Surface Science, Vol. 579, 2022, id. 152189.10.1016/j.apsusc.2021.152189Search in Google Scholar
[8] Li, K., C. Wang, P. Ning, K. Li, X. Sun, X. Song, and et al. Surface characterization of metal oxides-supported activated carbon fiber catalysts for simultaneous catalytic hydrolysis of carbonyl sulfide and carbon disulfide. Journal of Environmental Sciences, Vol. 11, 2020, pp. 44–54.10.1016/j.jes.2020.03.019Search in Google Scholar PubMed
[9] Yang, C., H. Ye, J. Byun, Y. Hou, and X. Wang. N-Rich carbon catalysts with economic feasibility for the selective oxidation of hydrogen sulfide to sulfur. Environmental Science and Technology, Vol. 54, 2020, pp. 12621–12630.10.1021/acs.est.0c02967Search in Google Scholar PubMed
[10] Habeeb, O. A., K. Ramesh, and G. A. M. Ali. Modeling and optimization for H2S adsorption from wastewater using coconut shell based activated carbon. Australian Journal of Basic & Applied Sciences, Vol. 10, 2016, pp. 136–147.Search in Google Scholar
[11] Yu, Q. F., M. Li, P. Ning, H. Yi, and X. Tang. Characterization of metal oxide-modified walnut-shell activated carbon and its application for phosphine adsorption: Equilibrium, regeneration, and mechanism studies. Journal of Wuhan University of Technology Materials Science Edition, Vol. 34, 2019, pp. 487–495.10.1007/s11595-019-2078-ySearch in Google Scholar
[12] Anisuzzama, S. M., D. Krishnaiah, C. G. Joseph, and S. Abang. Dynamic simulation of hydrogen sulfide adsorption in a packed bed column of activated carbon. Journal of Applied Sciences, Vol. 14, 2014, pp. 3294–3299.10.3923/jas.2014.3294.3299Search in Google Scholar
[13] Rattanaphan, S., T. Rungrotmongkol, and P. Kongsune. Biogas improving by adsorption of CO2 on modified waste tea activated carbon. Renewable Energy, Vol. 145, 2020, pp. 622–631.10.1016/j.renene.2019.05.104Search in Google Scholar
[14] Georgiadis, A. G., N. D. Charisiou, and M. A. Goula. Removal of hydrogen sulfide from various industrial gases: A review of the most promising adsorbing materials. Catalysts, Vol. 10, 2020, pp. 2–36.10.3390/catal10050521Search in Google Scholar
[15] Choi, D. Y., J. W. Lee, and S. C. Jang. Adsorption dynamics of hydrogen sulfide in impregnated activated carbon bed. Adsorption, Vol. 14, 2008, pp. 533–538.10.1007/s10450-008-9118-9Search in Google Scholar
[16] Zeng, F., X. Liao, J. Lu, D. Pan, and W. Zhang. Effect of iron salt modification on the adsorption of hydrogen sulfide by sludge-based activated carbon. Desalination and Water Treatment, Vol. 202, 2020, pp. 61–70.10.5004/dwt.2020.26150Search in Google Scholar
[17] Zhang, Y., Z. Zhang, X. Liu, L. Liu, and J. Peng. Desulfurization process and kinetics of Fe/activated carbon removal of H2S at low temperature. ChemistrySelect, Vol. 5, 2020, pp. 8386–8393.10.1002/slct.202001834Search in Google Scholar
[18] Zagoruiko, A. N., S. Vasiliy, and K. Gennady. Kinetics of H2S selective oxidation by oxygen at the carbon nanofibrous catalyst. Reaction Kinetics Mechanisms & Catalysis, Vol. 123, 2018, pp. 1–15.10.1007/s11144-017-1339-zSearch in Google Scholar
[19] Qin, H., D. X. Wu, and Q. Wang. Research on adsorption of H2S by modified activated carbon. Advanced Materials Research, Vol. 463–464, 2012, pp. 128–132.10.4028/www.scientific.net/AMR.463-464.128Search in Google Scholar
[20] Ciahotny, K. and V. Kyselova. Hydrogen sulfide removal from biogas using carbon impregnated with oxidants. Energy & Fuels, Vol. 33, 2019, pp. 5316–5321.10.1021/acs.energyfuels.9b00624Search in Google Scholar
[21] Xiao, Y., S. Wang, D. Wu, and Y. Quan. Experimental and simulation study of hydrogen sulfide adsorption on impregnated activated carbon under anaerobic conditions. Journal of Hazardous Materials, Vol. 153, 2008, pp. 1193–1200.10.1016/j.jhazmat.2007.09.081Search in Google Scholar PubMed
[22] Karthikeyan, S., C. J. Magthalin, M. Mahesh, C. Anandan, and G. Sekaran. Synthesis of reactive iron impregnated nanoporous activated carbon and its application in anaerobic biological treatment to enhance biodegradability of ortho-phenylenediamine. Journal of Chemical Technology & Biotechnology, Vol. 90, 2015, pp. 1013–1026.10.1002/jctb.4403Search in Google Scholar
[23] Shen, F., J. Liu, C. Gu, and D. Wu. Roles of oxygen functional groups in hydrogen sulfide adsorption on activated carbon surface: A density functional study. Industrial & Engineering Chemistry Research, Vol. 58, 2019, pp. 5526–5532.10.1021/acs.iecr.9b00507Search in Google Scholar
[24] Borguet, E., S. Kwon, W. Feng, and R. Vidic. Adsorption of hydrogen sulfide onto activated carbon fibers: Effect of pore structure and surface chemistry. Environmental Science & Technology, Vol. 39, 2005, pp. 9744–9749.10.1021/es0507158Search in Google Scholar PubMed
[25] Bagreev, A., J. A. Menendez, I. Dukhno, Y. Tarasenko, and T. J. Bandosz. Bituminous coal-based activated carbons modified with nitrogen as adsorbents of hydrogen sulfide. Carbon, Vol. 42, 2004, pp. 469–476.10.1016/j.carbon.2003.10.042Search in Google Scholar
[26] Zhang, X., A. Li, Z. Jiang, and Q. Zhang. Adsorption of dyes and phenol from water on resin adsorbents: Effect of adsorbate size and pore size distribution. Journal of Hazardous Materials, Vol. 137, 2006, pp. 1115–1122.10.1016/j.jhazmat.2006.03.061Search in Google Scholar PubMed
[27] Llewellyn, P. L., Y. Grillet, F. Schuth, H. Reichert, and K. K. Unger. Effect of pore size on adsorbate condensation and hysteresis within a potential model adsorbent: M41S. Microporous Materials, Vol. 3, 1994, pp. 345–349.10.1016/0927-6513(94)00042-5Search in Google Scholar
[28] Papadopoulos, G. K. and J. H. Petropoulos. Model study of the effect of pore structure and condensation on multilayer adsorbate transport in porous media. Langmuir the Acs Journal of Surfaces & Colloids, Vol. 23, 2007, pp. 12932–12936.10.1021/la702237vSearch in Google Scholar PubMed
[29] Shah, M. S., M. Tsapatsis, and J. I. Siepmann. Hydrogen sulfide capture: From absorption in polar liquids to oxide, zeolite and metal-organic framework adsorbents and membranes. Chemical Reviews, Vol. 117, 2017, pp. 9755–9803.10.1021/acs.chemrev.7b00095Search in Google Scholar PubMed
[30] Alayande, A. B., H. Jee, and D. Kang. Membrane and adsorption technologies for efficient hydrogen sulfide removal from biogas:a review focused on the advancement of key components. Transactions of The Institution of Chemical Engineers, Process Safety and Environmental Protection, Part B, Vol. 186, 2024, pp. 448–473.10.1016/j.psep.2024.04.018Search in Google Scholar
[31] Turk, A., E. Sakalis, and J. Lessuck. Ammonia injection enhances capacity of activated carbon for hydrogen sulfide and methyl mercaptan. Environmental Science & Technology, Vol. 23, 1989, pp. 1242–1245.10.1021/es00068a008Search in Google Scholar
[32] Abdirakhimov, M., M. H. Al-Rashed, and W. Janusz. Hydrogen sulfide adsorption from natural gas using silver-modified 13X molecular sieve. Materials, Vol. 17, 2024, id. 165.10.3390/ma17010165Search in Google Scholar PubMed PubMed Central
[33] Bandosz, T. J. On the adsorption/oxidation of hydrogen sulfide on activated carbons at ambient temperatures. Journal of Colloid & Interface Science, Vol. 246, 2002, pp. 1–20.10.1006/jcis.2001.7952Search in Google Scholar PubMed
[34] Sitthikhankaew, R., D. Chadwick, S. Assabumrungrat, and N. Laosiripojana. Effects of humidity, O2, and CO2 on H2S adsorption onto upgraded and KOH impregnated activated carbons. Fuel Processing Technology, Vol. 124, 2014, pp. 249–257.10.1016/j.fuproc.2014.03.010Search in Google Scholar
[35] Xiao, Y., S. Wang, D. Wu, and Y. Quan. Catalytic oxidation of hydrogen sulfide over unmodified and impregnated activated carbon. Separation & Purification Technology, Vol. 59, 2008, pp. 326–332.10.1016/j.seppur.2007.07.042Search in Google Scholar
[36] Sing, K. W. and R. Williams. The use of molecular probes for the characterization of nanoporous adsorbents. Particle & Particle Systems Characterization, Vol. 21, 2004, pp. 71–79.10.1002/ppsc.200400923Search in Google Scholar
© 2025 the author(s), published by De Gruyter
This work is licensed under the Creative Commons Attribution 4.0 International License.
Articles in the same Issue
- Research Articles
- Endpoint carbon content and temperature prediction model in BOF steelmaking based on posterior probability and intra-cluster feature weight online dynamic feature selection
- Thermal conductivity of lunar regolith simulant using a thermal microscope
- Multiobjective optimization of EDM machining parameters of TIB2 ceramic materials using regression and gray relational analysis
- Research on the magnesium reduction process by integrated calcination in vacuum
- Microstructure stability and softening resistance of a novel Cr-Mo-V hot work die steel
- Effect of bonding temperature on tensile behaviors and toughening mechanism of W/(Ti/Ta/Ti) multilayer composites
- Exploring the selective enrichment of vanadium–titanium magnetite concentrate through metallization reduction roasting under the action of additives
- Effect of solid solution rare earth (La, Ce, Y) on the mechanical properties of α-Fe
- Impact of variable thermal conductivity on couple-stress Casson fluid flow through a microchannel with catalytic cubic reactions
- Effects of hydrothermal carbonization process parameters on phase composition and the microstructure of corn stalk hydrochars
- Wide temperature range protection performance of Zr–Ta–B–Si–C ceramic coating under cyclic oxidation and ablation environments
- Influence of laser power on mechanical and microstructural behavior of Nd: YAG laser welding of Incoloy alloy 800
- Aspects of thermal radiation for the second law analysis of magnetized Darcy–Forchheimer movement of Maxwell nanomaterials with Arrhenius energy effects
- Use of artificial neural network for optimization of irreversibility analysis in radiative Cross nanofluid flow past an inclined surface with convective boundary conditions
- The interface structure and mechanical properties of Ti/Al dissimilar metals friction stir lap welding
- Significance of micropores for the removal of hydrogen sulfide from oxygen-free gas streams by activated carbon
- Experimental and mechanistic studies of gradient pore polymer electrolyte fuel cells
- Microstructure and high-temperature oxidation behaviour of AISI 304L stainless steel welds produced by gas tungsten arc welding using the Ar–N2–H2 shielding gas
- Mathematical investigation of Fe3O4–Cu/blood hybrid nanofluid flow in stenotic arteries with magnetic and thermal interactions: Duality and stability analysis
- Influence on hexagonal closed structure and mechanical properties of outer heat treatment cycle and plasma arc transfer Ti54Al23Si8Ni5XNb+Ta coating for Mg alloy by selective laser melting process
- Effect of rare-earth yttrium doping on the microstructure and texture of hot-rolled non-oriented electrical steel
- Study on the rheological behavior and microstructure evolution of isothermal compression of high-chromium cast steel
- Analysis of CO2–O2 jet characteristics of post-combustion oxygen lance in converter under the influence of multiple parameters
- Source-modulated controllable growth and mechanism exploration of 2D MoS2 deposited by NaCl-assisted CVD
- Topical Issue on Conference on Materials, Manufacturing Processes and Devices - Part II
- Effects of heat treatment on microstructure and properties of CrVNiAlCu high-entropy alloy
- Enhanced bioactivity and degradation behavior of zinc via micro-arc anodization for biomedical applications
- Study on the parameters optimization and the microstructure of spot welding joints of 304 stainless steel
- Research on rotating magnetic field–assisted HRFSW 6061-T6 thin plate
- Efficient preparation and evaluation of dry gas sealed spiral grooves
- Special Issue on A Deep Dive into Machining and Welding Advancements - Part II
- Microwave hybrid process-based fabrication of super duplex stainless steel joints using nickel and stainless steel filler materials
- Special Issue on Polymer and Composite Materials and Graphene and Novel Nanomaterials - Part II
- Low-temperature corrosion performance of laser cladded WB-Co coatings in acidic environment
- Special Issue for the conference AMEM2025
- Effect of thermal effect on lattice transformation and physical properties of white marble
Articles in the same Issue
- Research Articles
- Endpoint carbon content and temperature prediction model in BOF steelmaking based on posterior probability and intra-cluster feature weight online dynamic feature selection
- Thermal conductivity of lunar regolith simulant using a thermal microscope
- Multiobjective optimization of EDM machining parameters of TIB2 ceramic materials using regression and gray relational analysis
- Research on the magnesium reduction process by integrated calcination in vacuum
- Microstructure stability and softening resistance of a novel Cr-Mo-V hot work die steel
- Effect of bonding temperature on tensile behaviors and toughening mechanism of W/(Ti/Ta/Ti) multilayer composites
- Exploring the selective enrichment of vanadium–titanium magnetite concentrate through metallization reduction roasting under the action of additives
- Effect of solid solution rare earth (La, Ce, Y) on the mechanical properties of α-Fe
- Impact of variable thermal conductivity on couple-stress Casson fluid flow through a microchannel with catalytic cubic reactions
- Effects of hydrothermal carbonization process parameters on phase composition and the microstructure of corn stalk hydrochars
- Wide temperature range protection performance of Zr–Ta–B–Si–C ceramic coating under cyclic oxidation and ablation environments
- Influence of laser power on mechanical and microstructural behavior of Nd: YAG laser welding of Incoloy alloy 800
- Aspects of thermal radiation for the second law analysis of magnetized Darcy–Forchheimer movement of Maxwell nanomaterials with Arrhenius energy effects
- Use of artificial neural network for optimization of irreversibility analysis in radiative Cross nanofluid flow past an inclined surface with convective boundary conditions
- The interface structure and mechanical properties of Ti/Al dissimilar metals friction stir lap welding
- Significance of micropores for the removal of hydrogen sulfide from oxygen-free gas streams by activated carbon
- Experimental and mechanistic studies of gradient pore polymer electrolyte fuel cells
- Microstructure and high-temperature oxidation behaviour of AISI 304L stainless steel welds produced by gas tungsten arc welding using the Ar–N2–H2 shielding gas
- Mathematical investigation of Fe3O4–Cu/blood hybrid nanofluid flow in stenotic arteries with magnetic and thermal interactions: Duality and stability analysis
- Influence on hexagonal closed structure and mechanical properties of outer heat treatment cycle and plasma arc transfer Ti54Al23Si8Ni5XNb+Ta coating for Mg alloy by selective laser melting process
- Effect of rare-earth yttrium doping on the microstructure and texture of hot-rolled non-oriented electrical steel
- Study on the rheological behavior and microstructure evolution of isothermal compression of high-chromium cast steel
- Analysis of CO2–O2 jet characteristics of post-combustion oxygen lance in converter under the influence of multiple parameters
- Source-modulated controllable growth and mechanism exploration of 2D MoS2 deposited by NaCl-assisted CVD
- Topical Issue on Conference on Materials, Manufacturing Processes and Devices - Part II
- Effects of heat treatment on microstructure and properties of CrVNiAlCu high-entropy alloy
- Enhanced bioactivity and degradation behavior of zinc via micro-arc anodization for biomedical applications
- Study on the parameters optimization and the microstructure of spot welding joints of 304 stainless steel
- Research on rotating magnetic field–assisted HRFSW 6061-T6 thin plate
- Efficient preparation and evaluation of dry gas sealed spiral grooves
- Special Issue on A Deep Dive into Machining and Welding Advancements - Part II
- Microwave hybrid process-based fabrication of super duplex stainless steel joints using nickel and stainless steel filler materials
- Special Issue on Polymer and Composite Materials and Graphene and Novel Nanomaterials - Part II
- Low-temperature corrosion performance of laser cladded WB-Co coatings in acidic environment
- Special Issue for the conference AMEM2025
- Effect of thermal effect on lattice transformation and physical properties of white marble

