Abstract
During 2012, about 782,500 new liver cancer cases were diagnosed and 745,500 deaths occurred all around the world. Liver cancer is the 2nd major cause of cancer death in men around the world and in underdeveloped countries. Dysregulation of the balance between proliferation and cell death, hepatitis B virus infection and hepatitis C virus chronic infection, and carcinogenic micro RNAs mainly contribute to the development and progression of liver cancer. Under physiological status, Src maintained the foundation of cells. While in liver cancer, it is known that the basic activities of cells are apparently disturbed possibly by Src. The mechanisms underlying the development and progression of liver cancer is needed to elucidate. In this study, we summarized newly found regulation pathway of SRC signaling, and clinical experience with inhibitors of Src signaling, such as, novel molecules that directly or indirectly targeted Src signaling which can be utilized in the treatment of liver cancer.
1 Introduction
In men, liver cancer is much more common compared to women. Liver cancer is the 2nd major cause of cancer death in men around the world and in underdeveloped countries; it is mainly caused by infection, accounting for about 77 percent of liver cancer cases all around the world. While in developed countries, liver cancer is the 6th major causes of cancer mortality among men. During 2012, about 782,500 liver cancer cases were newly diagnosed and 745,500 deaths recorded all around the world, with China alone accounting for about fifty percent of the total cases and deaths. About seventy percent to ninety percent primary liver cancers occurring worldwide are hepatocellular carcinoma (HCC) [1].
HCC is one of the most fatal forms of cancer around the world, for its high metastatic potential, recurrence and resistance to drugs. Abnormal regulation of the equilibrium between cell death and proliferation is a predominately marker in human hepatic carcinogenesis. One of the most famous changes in HCC is that the Spred family of Ras/ERK inhibitors is abnormal regulated and the expression level of PTEN is also decayed or deleted in nearly half of the HCCs cases, which consequently result in the over-activation of associated survival kinases: ERKs and Akt. Changes in the expression level and protein activity of functional molecules involved in neutralizing apoptosis, such as NF-κB, Mcl-1, c-IAP1, and Bcl-X(L), have also been identified in HCC [2]. Hepatitis B virus (HBV) infects the liver and could cause acute or chronic liver diseases which might give rise to liver cirrhosis and hepatocellular carcinoma [3]. Hepatitis C virus (HCV) Chronic infection is one another virus related risk factor for hepatic carcinogenesis. HCV core, NS5A, NS4B and NS3, are four major HCV gene products which have demonstrated potential transformation activity in tissue culture and a few potential oncogenic pathways. It has been shown that the above pathways are changed by HCV protein products. HCV core and NS5A can accumulate in wild type β-catenin and target Wnt pathway signaling in HCCs [4]. Recent studies have indicated that miRNAs contribute to HCCs. MiR-21, miR-26, miR-29, miR-122, miR-151, miR-155, and miR-221/222 are crucial for the progression of HCCs [5].
v-SRC, the transforming protein of Rous Sarcoma Virus, and its anthropic homolog c-Src possesses prominent historical importance for it was the first protumor gene to be discovered and the protein was the first confirmed tyrosine kinase [6]. It is the prototypic member of a family of non-receptor membrane-associated tyrosine kinases including Src, Yes, Fyn, Lyn, Blk, Fgr, Hck, Yrk, and Lck. Among them, Src, Yes and Fyn are pervasively expressed in multiple tissues, with relative higher intensities in osteoclasts, platelets, and neurons [7] Src participates in the stabilizing of normal cell homeostasis and in extensiveness physiological functions involving cell survival and proliferation, managing of the cytoskeleton and cell shape, keeping of intercellular contacts, cell–matrix adhesion, and motility [8]. Under physiological conditions Src regulates basic activities of the cell, while in liver cancer, a process in which basic activities are profoundly perturbed, Src is definitely involved. The regulation of the Src family by Csk had been reviewed by Okada [9].
Recently, the Src signaling pathways in prostate cancer had been reviewed by Varkaris et al. [10] Illuminating the underlying mechanisms of SRC signaling in HCCs is required for improving diagnosis, therapy and prognosis. In this study, we reviewed newly found regulation pathway of SRC signaling, and novel molecules that directly or indirectly targeted Src signaling which can be utilized in the treatment of liver cancer.
2 The molecular basis of Src signaling regulation
The Src family kinases (SFKs) are comprised of nine structurally similar non-receptor protein tyrosine kinases (Src, Fyn, Yes, Lyn, Blk, Fgr, Hck, Yrk, and Lck) [8]. SFKs’ amino acid composition consists of a conserved arrangement of four different function domains, named Src homology 2/3/4 (SH2/3/4) domains, catalytic domain, and a C-terminal regulatory domain [11]. The N-terminal unique SH4 domain facilitates myristoylation and palmitoylation. The myristoylation of glycine residue of each of the SFKs is required for membrane attachment, regulating kinase activity and intracellular stability. The palmitoylation of cysteine residues of all the SFKs except Src and Blk is crucial for the membrane attachment and SFKs’ trafficking [12]. The SH4 domain is followed by a region that is unique to each family member. SH3 and SH2 domains allow the association and interaction with adaptor and signaling proteins involved in the formation of complexes. Specifically, the SH3 domain leads to Src complexes with proteins having peptide sequences rich in proline and other hydrophobic amino acids [13,14]. These protein-protein interactions form a polyproline type II helix that associates with the hydrophobic sequence of SH3 domain. The SH2 domain has high affinity for phosphotyrosine containing sequences (pTyr-Glu-Glu-Ile) [15]. Association of SH2 and peptides resembles a two-pronged plug (peptide) engaging a two-“holed” socket (SH2). Phosphotyrosine occupies the first “hole”, whereas isoleucine occupies the second SH2 “hole”. SH2 and SH3 domains further regulate the kinase activity through intramolecular interactions, and provide some specificity for protein-protein interactions of individual SFKs. The catalytic domain possesses the intrinsic tyrosine kinase activity of the molecule and is followed by a C-terminal regulatory domain [16]. The intricacy regulations of the activity of the SFKs have been summarized before [9,10,17].
3 Expression of SFKs in liver cancer
As far as it’s known that, there is no somatic mutation in the SFK genes that have been detected [7,18]. Thus, dysregulation of SFKs must have played a crucial role in the etiology of various human diseases such as liver cancers. After analyzing 52 northern Chinese patients with HCC using immunohistochemistry, Zhao et al. [19] found that the expressions of t-Src and p-Src (phospho-Y416) in HCC tissue were significantly higher than those in adjacent nontumor tissue. In addition, the expression level of total Src was found to positively correlated with tumor stage, cellular differentiation, metastasis and the expression level of CA19-9 significantly, which is a tumor marker, and abnormal expressed in many types of cancer including hepatocellular carcinoma. While, the expression level of p-Y416 Src was only significantly correlated with tumor stage.
3.1 Roles of SFKs in liver cancer tumor cell growth
ASPP2 containing ankyrin repeat, SH3 domain, and proline-rich region protein (ASPP), was found to significantly inhibit FAK/Src/ERK signaling. It can directly interact with the CSK protein and activating the kinase activity of CSK, which consequently result in the downregulation of Snail expression mediated by activator protein 1 (AP1). Inhibition of ASPP2 induced stem-like properties of HCC cells and resistance to drugs, including the formation of hepatospheroids, expansion of side population fractions, expression of stem cell-associated genes, loss of chemosensitivity, and promoted carcinogenesis in mice model [20].
Hepatitis B virus (HBV)-X protein (HBx), a hepatitis B viral protein product, is a transactivator of several genes of the host, including alpha-fetoprotein (AFP) and alpha-fetoprotein receptor (AFPR) which accounts for HBV-induced carcinogenesis. The expressions of AFP and AFPR are associated with hepatocellular carcinoma (HCC) stem-like cells. HBx prefers to increase the expression levels of AFP and AFPR, which consequently activate Src expression. Ly294002 and GDC0941, which are Phosphatidylinositol 3-kinase(PI3K) inhibitors, can functionally suppress AFPR-induced increase of c-Src in AFPR positive HCC cell lines. Above all, it can confirm a pathway of AFPR/PI3K/Src [21].
It has been reported that transforming growth factor beta-interacting factor (TGIF) can inhibit arsenic trioxide-induced apoptosis of HepG2 cells. During this process, c-Src/AKT pathway is the key regulator that regulates TGIF-induced Nox4 activation and subsequent superoxide production [22].
Hepatic transmembrane 4 L six family member 5 (TM4SF5) enhance EMT process for the malignancy of cancer cells. TM4SF5/CD44 interaction increased the phosphorylation of residue 416 of c-Src/STAT3/Twist1/ Bmi1 signaling for spheroid formation. On the contrary, perturbation of the interaction, expression or activity of any component in the axis blocked the process of spheroid formation. TM4SF5 can bind directly to CD44 by their extracellular N-glycosylation modified domains. Besides, TM4SF5 can enhance self-renewal and circulating tumor cells (CTC) properties through depending on TM4SF5(+)/ CD44(+(TM4SF5-bound))/ALDH(+)/CD24(-) biomarkers during metastatic process of HCC [23].
3.2 Roles of SFKs in liver cancer development and metastasis
Tumor metastasis is a complicated process, involving elevated circulating and tumor-initiating activities of metastatic cancer cells, and considered as a dynamic procedure in the development of tumor which is associated with off normal migration and invasion activities of the cancer cells. Self-renewal and circulating tumor cell (CTC) characteristics for tumor metastasis are highly affected by epithelial-mesenchymal transition (EMT). Although tumor metastasis is a lethal procedure for tumor-bearing patients, which is initiated by blood circulation of tumor cells, the mechanism of self-renewal and circulating activities is still to be elucidated. Amoeboid and mesenchymal-type are two interconvertible modes of migration of tumor cells when they move as individual cells. The phosphorylation level of Src (tyrosine 416 phosphorylated) can be increased by CD147, which is also known as basigin (an extracellular matrix metalloproteinase inducer and determinant for the Ok blood group system). It has been found that this affection of CD147 is mainly modulated by focal adhesion kinase (FAK). Dedicator of cytokinesis protein 8 (DOCK8) is a GEF for Rac1, and functions as a major protein which drives mesenchymal-type migration. DOCK8 expression and Rac1 activation can be promoted by STAT3, whose phosphorylation is regulated by the activation of c-Src [24].
Liver fatty acid-binding protein (L-FABP) is rich in hepatocytes and is known to be involved in lipid metabolism. It was found that L-FABP is associated with VEGFR2 on lipid rafts activating the Akt/mTOR/ P70S6K/4EBP1 axis and Src/FAK/cdc42 axis, which resulted in the accumulation of VEGF-A and an increase in both angiogenic and migration activity [25].
It is known that cell surface GRP78 is a multiple functional receptor, which has been found to contribute to proliferation and anti-apoptotic signaling pathways as supported by emerging data. The association of cell surface GRP78 with alpha2M*, of which the activated form is a natural circulating ligand for cell surface GRP78, is found to contribute to the modulation of proliferation and survival of cells in cancers. GRP78 could interact with c-Src directly and consequently increased the phosphorylation level of c-Src at 416 tyrosine residue. The stimulation effect induced by the association of GRP78 with alpha2M* could be eliminated by PP2, which is the inhibitor of the tyrosine kinase activity of c-Src. In addition, the association of GRP78 with alpha2M* can enhance the interaction between c-Src and EGFR, and consequently increased the phosphorylation levels of EGFR at 1101 and 845 tyrosine residues, elevating the migration and invasion activity of HCCs. However, the binding of EGFR and Src was not affected by PP2 [26]. The development of acquired resistance to 5-FU, which is one of the standard first-line chemotherapeutic drugs for the therapy of hepatocellular carcinoma, restricts its clinical applications. Although several intense investigations have been conducted focusing on this phenomenon, acquired resistances to 5-FU are still remains to be resolved. Gu et al. [27] had reported that over-expression of cell surface GRP78 accounted for the acquired resistance to 5-FU in HCC through modulating the c-Src/LSF/TS pathway. Cell surface GRP78 binds directly to the ATPase domain of c-Src promoting the phosphorylation level of 416 tyrosine residue in c-Src, which results in the increase of LSF expression.
The expression deficiency of Caspase-8 is occurred in a few of tumors, which suggest that the perturbation of its function may contribute to the progression of cancer. Accordingly, it has been identified that Caspase-8 possessed several non-apoptotic functions and has the ability to regulate cell adhesion, migration and to facilitate the progression of tumor. Fianco et al. [28] found that Caspase-8 expression in hepatocellular carcinoma cell lines may promote cell transformations. In their studies, Caspase-8 is phosphorylated on amino acid Tyr380 residue in a c-Src dependent manner whose phosphorylation is very important for cell transformations and enhanced by hypoxic conditions. Interestingly, the function of Caspase-8 described above is distinct from its catalytic activity. Their study demonstrates another role for Caspase-8 in hepatic cancer, which suggests that the phosphorylation of Tyr380 residue of Caspase-8 may present a molecule switch to turn its role from tumor suppressor to tumor activator.
The newly found regulation network was summarized in Figure 1.
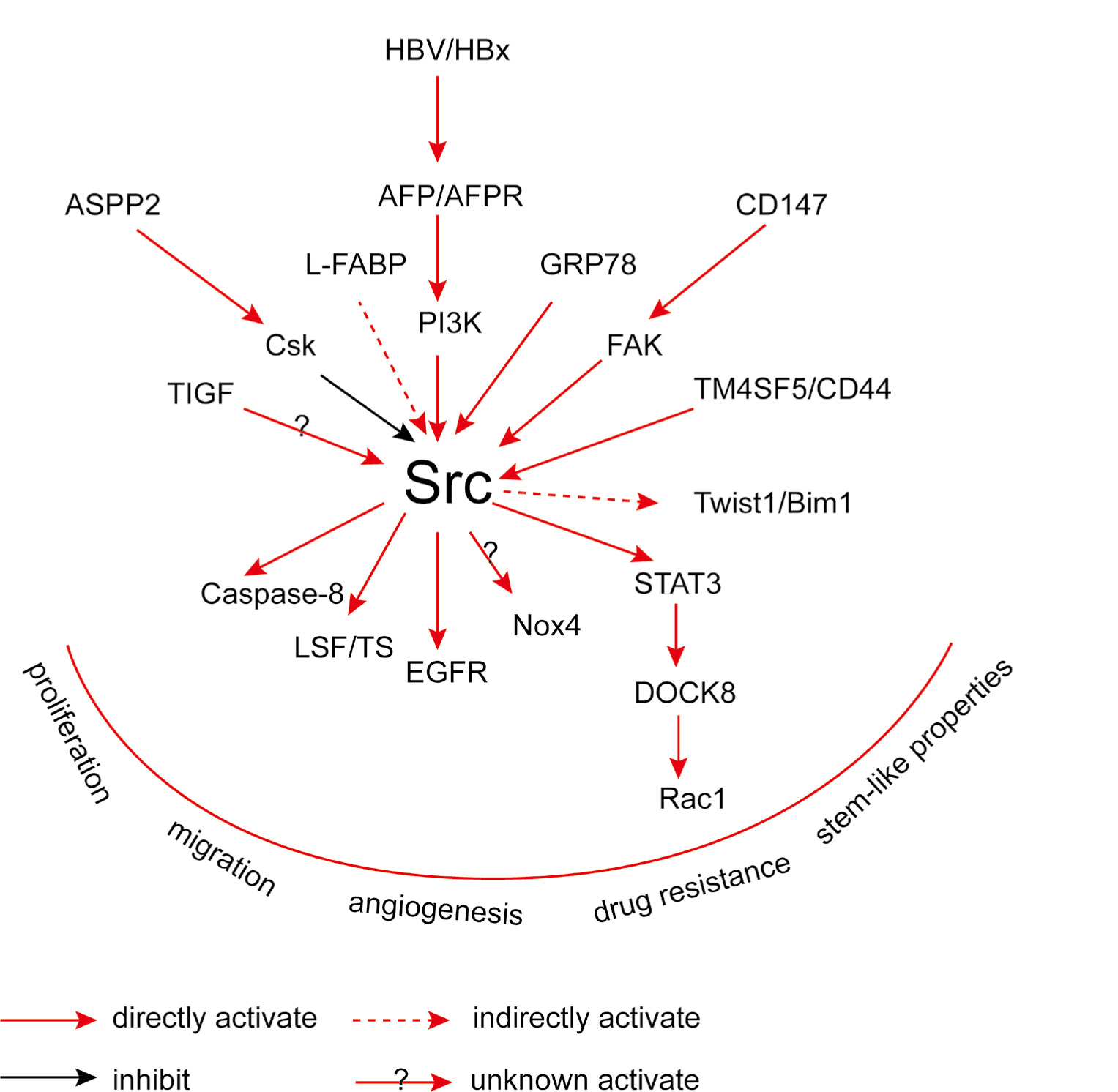
The plain scheme of the regulation network of Src signaling.
4 Clinical experience with inhibitors of Src signaling
Saracatinib, is the first Src inhibitor to exhibit inhibitory activity to Src signaling in human tumor tissues. Xiong et al. [29] had reported that the administration of saracatinib can reduce the metastasic potential in an orthotopic xenograft HCC model, while the proliferation of tumor cells was not affected. Saracatinib treatment can cause reduced phosphorylation of Src pathway signaling molecules, such as c-Src, STAT3 and FAK, in vitro and in vivo. Hepatitis B virus (HBV)-related hepatocellular carcinoma (HCC) often occurs in male subjects, which is mainly modulated by the enhancement of androgen receptor (AR) activity mediated by hepatitis B X (HBx). Using a HBx transgenic mice model, Wang et al. [30] showed that Sorafenib markedly blocked the HBx- mediated AR activity via stimulating phosphatase activity of SHP-1, which suppress the overactivation of Akt/GSK 3β and c-Src by HBx.
Cell cycle, regulation of gene expression, responses to cellular stress and many other cellular processes are dependent on proteasomes. Agents that inhibit these proteases are a promising anti-cancer agents. Bortezomib, a proteasome inhibitor, can effectively suppress protease activity in several cancers, including myeloma and lymphoma. While, solid tumors, including hepatocellular carcinoma are poorly inhibited by bortezomib [31]. Hou et al. [32] studied the mechanism behind this and concluded that reactive oxygen species (ROS) triggered the activation of the Src/EGFR/Akt signaling pathway by bortezomib. Based on these findings, the combination of bortezomib and saracatinib can synergistically induce hepatoma cell apoptosis.
SKLB-329, a multikinase inhibitor, could significantly suppress the growth of HCC cells via potently inhibiting angiogenesis-related kinases like VEGFR1, VEGFR2, VEGFR3, FGFR2, and c-Src. The inhibition effect of SKLB-329 is dependent on the depression of c-Src-modulated activities of FAK and Stat3. With a higher potency compared with sorafenib, SKLB-329 could perturb growth, migration, and invasion of endothelial cell, and inhibit tube formation significantly [33].
Corosolic acid, extracted from the root of Actinidia chinensis, has a significant antagonism effect on HCC cells. Corosolic acid can directly interact with the ATP binding pocket of VEGFR2, and inhibit the kinase activity of VEGFR2. Depression of the VEGFR2/Src/FAK/CDC42 axis caused by Corosolic acid, subsequently decreased F-actin formation and migratory activity of the cells in vitro. An in vivo mouse model showed that Corosolic acid exhibited a significant effect on tumor growth with a relative low dose. Ku et al. [34] further demonstrated that the combination of Corosolic acid and sorafenib can synergistically affect tumor growth with a wide range of concentrations.
Antcin K, which is extracted from the fruiting body of basswood-cultivated A. cinnamomea, could inhibit the adhesion, migration and invasion abilities of Hep3B cells. The expression levels of integrin beta1, beta3, alpha5 and alpha V, and the phosphorylation of FAK, Src, PI3K, AKT, MEK, ERK, and JNK could be decreased by Antcin K. Coupled with the above findings; antcin K is a promising agent to lower the risk of liver cancer metastasis [35].
5 Conclusion
Src signaling plays a crucial role in maintaining cellular proliferation, invasion, migration, angiogenesis, and drug resistance of liver cancer. ASPP2, TIGF, L-FABP, GRP78, CD147, and TM4SF5/CD44 are newly found molecules that contribute to HCCs through Src signaling. Saracatinib is the first Src inhibitor to exhibit inhibitory activity of the Src signaling in human tumor tissues, and the first targeted drug approved by the FDA (food and drug administration) for the treatment of HCC. Drug resistance, side effects, toxicities, and many other problems need to be resolved to improve the outcome of treatment. Recently, many novel small molecules targeting Src signaling, or synergistic to TKIs were developed to overcome some of the aforementioned problems. ASPP2, TIGF, L-FABP, GRP78, CD147, and TM4SF5/CD44 are promising targets for the treatment of liver cancer.
Acknowledgements
This study was partly supported by the grants from the Medical Science and Technology Project of Zhejiang Province, (No.2015KYB210), Scientific Research Project of Zhejiang Provincial Department of Education, (Y201635902), Zhejiang Province Natural Fund (No.LY16H060002).
Conflict of interest: Authors declare that there are no conflicts of interest.
References
[1] Torre L.A., Bray F., Siegel R.L., Ferlay J., Lortet-Tieulent J., Jemal A., Global cancer statistics, 2012, Cancer J. Clin., 2015, 65, 87-108.10.3322/caac.21262Search in Google Scholar PubMed
[2] Fabregat I., Roncero C., Fernandez M., Survival and apoptosis: a dysregulated balance in liver cancer, Liver Int., 2007, 27, 155-162.10.1111/j.1478-3231.2006.01409.xSearch in Google Scholar PubMed
[3] Sarkar N., Chakravarty R., Hepatitis B Virus Infection, MicroRNAs and Liver Disease, Int. J. Mol. Sci., 2015, 16, 17746-17762.10.3390/ijms160817746Search in Google Scholar PubMed PubMed Central
[4] Levrero M., Viral hepatitis and liver cancer: the case of hepatitis C, Oncogene, 2006, 25, 3834-3847.10.1038/sj.onc.1209562Search in Google Scholar PubMed
[5] Braconi C., Henry J.C., Kogure T., Schmittgen T., Patel T., The role of microRNAs in human liver cancers, Seminars Oncol., 2011, 38, 752-763.10.1053/j.seminoncol.2011.08.001Search in Google Scholar PubMed PubMed Central
[6] Courtneidge S.A., Role of Src in signal transduction pathways. The Jubilee Lecture, Biochem. Soc. Trans., 2002, 30, 11-17.10.1042/bst0300011Search in Google Scholar
[7] Yeatman T.J., A renaissance for SRC, Nature Rev. Cancer, 2004, 4, 470-480.10.1038/nrc1366Search in Google Scholar PubMed
[8] Thomas S.M., Brugge J.S., Cellular functions regulated by Src family kinases, Ann. Rev. Cell Dev. Biol., 1997, 13, 513-609.10.1146/annurev.cellbio.13.1.513Search in Google Scholar PubMed
[9] Okada M., Regulation of the SRC family kinases by Csk, Int. J. Biol. Sci., 2012, 8, 1385-1397.10.7150/ijbs.5141Search in Google Scholar PubMed PubMed Central
[10] Varkaris A., Katsiampoura A.D., Araujo J.C., Gallick G.E., Corn P.G., Src signaling pathways in prostate cancer, Cancer Metastasis Rev., 2014, 33, 595-606.10.1007/s10555-013-9481-1Search in Google Scholar PubMed PubMed Central
[11] Sicheri F., Kuriyan J., Structures of Src-family tyrosine kinases, Curr. Opinion Structural Biol., 1997, 7, 777-785.10.1016/S0959-440X(97)80146-7Search in Google Scholar PubMed
[12] Patwardhan P., Resh M.D., Myristoylation and membrane binding regulate c-Src stability and kinase activity, Mol. Cell. Biol., 2010, 30, 4094-4107.10.1128/MCB.00246-10Search in Google Scholar PubMed PubMed Central
[13] Cohen G.B., Ren R., Baltimore D., Modular binding domains in signal transduction proteins, Cell, 1995, 80, 237-248.10.1016/0092-8674(95)90406-9Search in Google Scholar PubMed
[14] Moarefi I., LaFevre-Bernt M., Sicheri F., Huse M., Lee C.H., Kuriyan J., et al., Activation of the Src-family tyrosine kinase Hck by SH3 domain displacement, Nature, 1997, 385, 650-653.10.1038/385650a0Search in Google Scholar PubMed
[15] Moran M.F., Koch C.A., Anderson D., Ellis C., England L., Martin G.S., et al., Src homology region 2 domains direct proteinprotein interactions in signal transduction, Proc. Natl. Acad. Sci. USA, 1990, 87, 8622-8626.10.1073/pnas.87.21.8622Search in Google Scholar PubMed PubMed Central
[16] Hanks S.K., Quinn A.M., Hunter T., The protein kinase family: conserved features and deduced phylogeny of the catalytic domains, Science, 1988, 241, 42-52.10.1126/science.3291115Search in Google Scholar PubMed
[17] Brown M.T., Cooper J.A., Regulation, substrates and functions of src, Biochim. Biophys. Acta, 1996, 1287, 121-149.10.1016/0304-419X(96)00003-0Search in Google Scholar PubMed
[18] Frame M.C., Newest findings on the oldest oncogene; how activated src does it, J. Cell Sci., 2004, 117, 989-998.10.1242/jcs.01111Search in Google Scholar PubMed
[19] Zhao R., Wu Y., Wang T., Zhang Y., Kong D., Zhang L., et al., Elevated Src expression associated with hepatocellular carcinoma metastasis in northern Chinese patients, Oncol. Lett., 2015, 10, 3026-3034.10.3892/ol.2015.3706Search in Google Scholar PubMed PubMed Central
[20] Xu L., Tong X., Zhang S., Yin F., Li X., Wei H., et al., ASPP2 suppresses stem cell-like characteristics and chemoresistance by inhibiting the Src/FAK/Snail axis in hepatocellular carcinoma, Tumour Biol., 2016, 10, 3669-13677.10.1007/s13277-016-5246-0Search in Google Scholar PubMed
[21] Zhu M., Guo J., Li W., Xia H., Lu Y., Dong X., et al., HBx induced AFP receptor expressed to activate PI3K/AKT signal to promote expression of Src in liver cells and hepatoma cells, BMC Cancer, 2015, 15, 362.10.1186/s12885-015-1384-9Search in Google Scholar PubMed PubMed Central
[22] Liu Z.M., Tseng H.Y., Tsai H.W., Su F.C., Huang H.S., Transforming growth factor beta-interacting factor-induced malignant progression of hepatocellular carcinoma cells depends on superoxide production from Nox4, Free Radical Biol. & Med., 2015, 84, 54-64.10.1016/j.freeradbiomed.2015.03.028Search in Google Scholar PubMed
[23] Lee D., Na J., Ryu J., Kim H.J., Nam S.H., Kang M., et al., Interaction of tetraspan(in) TM4SF5 with CD44 promotes self-renewal and circulating capacities of hepatocarcinoma cells, Hepatology, 2015, 61, 1978-1997.10.1002/hep.27721Search in Google Scholar PubMed
[24] Wang S.J., Cui H.Y., Liu Y.M., Zhao P., Zhang Y., Fu Z.G., et al., CD147 promotes Src-dependent activation of Rac1 signaling through STAT3/DOCK8 during the motility of hepatocellular carcinoma cells, Oncotarget, 2015, 6, 243-257.10.18632/oncotarget.2801Search in Google Scholar PubMed PubMed Central
[25] Ku C.Y., Liu Y.H., Lin H.Y., Lu S.C., Lin J.Y., Liver fatty acid-binding protein (L-FABP) promotes cellular angiogenesis and migration in hepatocellular carcinoma, Oncotarget, 2016, 7, 18229-18246.10.18632/oncotarget.7571Search in Google Scholar PubMed PubMed Central
[26] Zhao S., Li H., Wang Q., Su C., Wang G., Song H., et al., The role of c-Src in the invasion and metastasis of hepatocellular carcinoma cells induced by association of cell surface GRP78 with activated alpha2M, BMC Cancer, 2015, 15, 389.10.1186/s12885-015-1401-zSearch in Google Scholar PubMed PubMed Central
[27] Gu Y.J., Li H.D., Zhao L., Zhao S., He W.B., Rui L., et al., GRP78 confers the resistance to 5-FU by activating the c-Src/LSF/ TS axis in hepatocellular carcinoma, Oncotarget, 2015, 6, 33658-33674.10.18632/oncotarget.5603Search in Google Scholar PubMed PubMed Central
[28] Fianco G., Cenci C., Barila D., Caspase-8 expression and its Src-dependent phosphorylation on Tyr380 promote cancer cell neoplastic transformation and resistance to anoikis, Exp. Cell Res., 2016, 347 (1), 114-122.10.1016/j.yexcr.2016.07.013Search in Google Scholar PubMed
[29] Xiong J., Wu J.S., Mao S.S., Yu X.N., Huang X.X., Effect of saracatinib on pulmonary metastases from hepatocellular carcinoma, Oncol. Rep., 2016, 36 (3), 1483-1490.10.3892/or.2016.4968Search in Google Scholar PubMed
[30] Wang S.H., Yeh S.H., Shiau C.W., Chen K.F., Lin W.H., Tsai T.F., et al., Sorafenib Action in Hepatitis B Virus X-Activated Oncogenic Androgen Pathway in Liver through SHP-1, J. Natl. Cancer Instit., 2015, 107.10.1093/jnci/djv190Search in Google Scholar PubMed
[31] Lara P.N., Jr., Davies A.M., Mack P.C., Mortenson M.M., Bold R.J., Gumerlock P.H., et al., Proteasome inhibition with PS-341 (bortezomib) in lung cancer therapy, Seminars Oncol., 2004, 31, 40-46.10.1053/j.seminoncol.2003.12.013Search in Google Scholar PubMed
[32] Hou J., Cui A., Song P., Hua H., Luo T., Jiang Y., Reactive oxygen species-mediated activation of the Src-epidermal growth factor receptor-Akt signaling cascade prevents bortezomib-induced apoptosis in hepatocellular carcinoma cells, Mol. Med. Rep., 2015, 11, 712-718.10.3892/mmr.2014.2736Search in Google Scholar PubMed
[33] Zhong L., Fu X.Y., Zou C., Yang L.L., Zhou S., Yang J., et al., A preclinical evaluation of a novel multikinase inhibitor, SKLB-329, as a therapeutic agent against hepatocellular carcinoma, Int. J. Cancer, 2014, 135, 2972-2983.10.1002/ijc.28944Search in Google Scholar PubMed
[34] Ku C.Y., Wang Y.R., Lin H.Y., Lu S.C., Lin J.Y., Corosolic Acid Inhibits Hepatocellular Carcinoma Cell Migration by Targeting the VEGFR2/Src/FAK Pathway, PloS One, 2015, 10, e0126725.10.1371/journal.pone.0126725Search in Google Scholar PubMed PubMed Central
[35] Huang Y.L., Chu Y.L., Ho C.T., Chung J.G., Lai C.I., Su Y.C., et al., Antcin K, an Active Triterpenoid from the Fruiting Bodies of Basswood-Cultivated Antrodia cinnamomea, Inhibits Metastasis via Suppression of Integrin-Mediated Adhesion, Migration, and Invasion in Human Hepatoma Cells, J. Agri. Food Chem., 2015, 63, 4561-4569.10.1021/jf5059304Search in Google Scholar PubMed
© 2016 Hong Ren et al.
This work is licensed under the Creative Commons Attribution-NonCommercial-NoDerivatives 3.0 License.
Articles in the same Issue
- Regular article
- Purification of polyclonal IgG specific for Camelid’s antibodies and their recombinant nanobodies
- Regular article
- Antioxidative defense mechanism of the ruderal Verbascum olympicum Boiss. against copper (Cu)-induced stress
- Regular article
- Polyherbal EMSA ERITIN Promotes Erythroid Lineages and Lymphocyte Migration in Irradiated Mice
- Regular article
- Expression and characterization of a cutinase (AnCUT2) from Aspergillus niger
- Regular article
- The Lytic SA Phage Demonstrate Bactericidal Activity against Mastitis Causing Staphylococcus aureus
- Regular article
- MafB, a target of microRNA-155, regulates dendritic cell maturation
- Regular article
- Plant regeneration from protoplasts of Gentiana straminea Maxim
- Regular article
- The effect of radiation of LED modules on the growth of dill (Anethum graveolens L.)
- Regular article
- ELF-EMF exposure decreases the peroxidase catalytic efficiency in vitro
- Regular article
- Cold hardening protects cereals from oxidative stress and necrotrophic fungal pathogenesis
- Regular article
- MC1R gene variants involvement in human OCA phenotype
- Regular article
- Chondrogenic potential of canine articular cartilage derived cells (cACCs)
- Regular article
- Cloning, expression, purification and characterization of Leishmania tropica PDI-2 protein
- Regular article
- High potential of sub-Mediterranean dry grasslands for sheep epizoochory
- Regular article
- Identification of drought, cadmium and root-lesion nematode infection stress-responsive transcription factors in ramie
- Regular article
- Herbal supplement formula of Elephantopus scaber and Sauropus androgynus promotes IL-2 cytokine production of CD4+T cells in pregnant mice with typhoid fever
- Regular article
- Caffeine effects on AdoR mRNA expression in Drosophila melanogaster
- Regular article
- Effects of MgCl2 supplementation on blood parameters and kidney injury of rats exposed to CCl4
- Regular article
- Effective onion leaf fleck management and variability of storage pathogens
- Regular article
- Improving aeration for efficient oxygenation in sea bass sea cages. Blood, brain and gill histology
- Regular article
- Biogenic amines and hygienic quality of lucerne silage
- Regular article
- Isolation and characterization of lytic phages TSE1-3 against Enterobacter cloacae
- Regular article
- Effects of pH on antioxidant and prooxidant properties of common medicinal herbs
- Regular article
- Relationship between cytokines and running economy in marathon runners
- Regular article
- Anti-leukemic activity of DNA methyltransferase inhibitor procaine targeted on human leukaemia cells
- Regular article
- Research Progress in Oncology. Highlighting and Exploiting the Roles of Several Strategic Proteins in Understanding Cancer Biology
- Regular article
- Ectomycorrhizal communities in a Tuber aestivum Vittad. orchard in Poland
- Regular article
- Impact of HLA-G 14 bp polymorphism and soluble HLA-G level on kidney graft outcome
- Regular article
- In-silico analysis of non-synonymous-SNPs of STEAP2: To provoke the progression of prostate cancer
- Regular article
- Presence of sequence and SNP variation in the IRF6 gene in healthy residents of Guangdong Province
- Regular article
- Environmental and economic aspects of Triticum aestivum L. and Avena sativa growing
- Regular article
- A molecular survey of Echinococcus granulosus sensu lato in central-eastern Europe
- Topical Issue on Cancer Signaling, Metastasis and Target Therapy
- Molecular genetics related to non-Hodgkin lymphoma
- Topical Issue on Cancer Signaling, Metastasis and Target Therapy
- Roles of long noncoding RNAs in Hepatocellular Carcinoma
- Topical Issue on Cancer Signaling, Metastasis and Target Therapy
- Advancement of Wnt signal pathway and the target of breast cancer
- Topical Issue on Cancer Signaling, Metastasis and Target Therapy
- A tumor suppressive role of lncRNA GAS5 in human colorectal cancer
- Topical Issue on Cancer Signaling, Metastasis and Target Therapy
- The role of E-cadherin - 160C/A polymorphism in breast cancer
- Topical Issue on Cancer Signaling, Metastasis and Target Therapy
- The proceedings of brain metastases from lung cancer
- Topical Issue on Cancer Signaling, Metastasis and Target Therapy
- Newly-presented potential targeted drugs in the treatment of renal cell cancer
- Topical Issue on Cancer Signaling, Metastasis and Target Therapy
- Decreased expression of miR-132 in CRC tissues and its inhibitory function on tumor progression
- Topical Issue on Cancer Signaling, Metastasis and Target Therapy
- The unusual yin-yang fashion of RIZ1/RIZ2 contributes to the progression of esophageal squamous cell carcinoma
- Topical Issue on Cancer Signaling, Metastasis and Target Therapy
- Human papillomavirus infection mechanism and vaccine of vulva carcinoma
- Topical Issue on Cancer Signaling, Metastasis and Target Therapy
- Abnormal expressed long non-coding RNA IRAIN inhibits tumor progression in human renal cell carcinoma cells
- Topical Issue on Cancer Signaling, Metastasis and Target Therapy
- UCA1, a long noncoding RNA, promotes the proliferation of CRC cells via p53/p21 signaling
- Topical Issue on Cancer Signaling, Metastasis and Target Therapy
- Forkhead box 1 expression is upregulatedin non-small cell lung cancer and correlateswith pathological parameters
- Topical Issue on Cancer Signaling, Metastasis and Target Therapy
- The development of potential targets in the treatment of non-small cell lung cancer
- Topical Issue on Cancer Signaling, Metastasis and Target Therapy
- Low expression of miR-192 in NSCLC and its tumor suppressor functions in metastasis via targeting ZEB2
- Topical Issue on Cancer Signaling, Metastasis and Target Therapy
- Downregulation of long non-coding RNA MALAT1 induces tumor progression of human breast cancer through regulating CCND1 expression
- Topical Issue on Cancer Signaling, Metastasis and Target Therapy
- Post-translational modifications of EMT transcriptional factors in cancer metastasis
- Topical Issue on Cancer Signaling, Metastasis and Target Therapy
- EZH2 Expression and its Correlation with Clinicopathological Features in Patients with Colorectal Carcinoma
- Topical Issue on Cancer Signaling, Metastasis and Target Therapy
- The association between EGFR expression and clinical pathology characteristics in gastric cancer
- Topical Issue on Cancer Signaling, Metastasis and Target Therapy
- The peiminine stimulating autophagy in human colorectal carcinoma cells via AMPK pathway by SQSTM1
- Topical Issue on Cancer Signaling, Metastasis and Target Therapy
- Activating transcription factor 3 is downregulated in hepatocellular carcinoma and functions as a tumor suppressor by regulating cyclin D1
- Topical Issue on Cancer Signaling, Metastasis and Target Therapy
- Progress toward resistance mechanism to epidermal growth factor receptor tyrosine kinase inhibitor
- Topical Issue on Cancer Signaling, Metastasis and Target Therapy
- Effect of miRNAs in lung cancer suppression and oncogenesis
- Topical Issue on Cancer Signaling, Metastasis and Target Therapy
- Role and inhibition of Src signaling in the progression of liver cancer
- Topical Issue on Cancer Signaling, Metastasis and Target Therapy
- The antitumor effects of mitochondria-targeted 6-(nicotinamide) methyl coumarin
- Special Issue on CleanWAS 2015
- Characterization of particle shape, zeta potential, loading efficiency and outdoor stability for chitosan-ricinoleic acid loaded with rotenone
- Special Issue on CleanWAS 2015
- Genetic diversity and population structure of ginseng in China based on RAPD analysis
- Special Issue on CleanWAS 2015
- Optimizing the extraction of antibacterial compounds from pineapple leaf fiber
- Special Issue on CleanWAS 2015
- Identification of residual non-biodegradable organic compounds in wastewater effluent after two-stage biochemical treatment
- Special Issue on CleanWAS 2015
- Remediation of deltamethrin contaminated cotton fields: residual and adsorption assessment
- Special Issue on CleanWAS 2015
- A best-fit probability distribution for the estimation of rainfall in northern regions of Pakistan
- Special Issue on CleanWAS 2015
- Artificial Plant Root System Growth for Distributed Optimization: Models and Emergent Behaviors
- Special Issue on CleanWAS 2015
- The complete mitochondrial genomes of two weevils, Eucryptorrhynchus chinensis and E. brandti: conserved genome arrangement in Curculionidae and deficiency of tRNA-Ile gene
- Special Issue on CleanWAS 2015
- Characteristics and coordination of source-sink relationships in super hybrid rice
- Special Issue on CleanWAS 2015
- Construction of a Genetic Linkage Map and QTL Analysis of Fruit-related Traits in an F1 Red Fuji x Hongrou Apple Hybrid
- Special Issue on CleanWAS 2015
- Effects of the Traditional Chinese Medicine Dilong on Airway Remodeling in Rats with OVA-induced-Asthma
- Special Issue on CleanWAS 2015
- The effect of sewage sludge application on the growth and absorption rates of Pb and As in water spinach
- Special Issue on CleanWAS 2015
- Effectiveness of mesenchymal stems cells cultured by hanging drop vs. conventional culturing on the repair of hypoxic-ischemic-damaged mouse brains, measured by stemness gene expression
Articles in the same Issue
- Regular article
- Purification of polyclonal IgG specific for Camelid’s antibodies and their recombinant nanobodies
- Regular article
- Antioxidative defense mechanism of the ruderal Verbascum olympicum Boiss. against copper (Cu)-induced stress
- Regular article
- Polyherbal EMSA ERITIN Promotes Erythroid Lineages and Lymphocyte Migration in Irradiated Mice
- Regular article
- Expression and characterization of a cutinase (AnCUT2) from Aspergillus niger
- Regular article
- The Lytic SA Phage Demonstrate Bactericidal Activity against Mastitis Causing Staphylococcus aureus
- Regular article
- MafB, a target of microRNA-155, regulates dendritic cell maturation
- Regular article
- Plant regeneration from protoplasts of Gentiana straminea Maxim
- Regular article
- The effect of radiation of LED modules on the growth of dill (Anethum graveolens L.)
- Regular article
- ELF-EMF exposure decreases the peroxidase catalytic efficiency in vitro
- Regular article
- Cold hardening protects cereals from oxidative stress and necrotrophic fungal pathogenesis
- Regular article
- MC1R gene variants involvement in human OCA phenotype
- Regular article
- Chondrogenic potential of canine articular cartilage derived cells (cACCs)
- Regular article
- Cloning, expression, purification and characterization of Leishmania tropica PDI-2 protein
- Regular article
- High potential of sub-Mediterranean dry grasslands for sheep epizoochory
- Regular article
- Identification of drought, cadmium and root-lesion nematode infection stress-responsive transcription factors in ramie
- Regular article
- Herbal supplement formula of Elephantopus scaber and Sauropus androgynus promotes IL-2 cytokine production of CD4+T cells in pregnant mice with typhoid fever
- Regular article
- Caffeine effects on AdoR mRNA expression in Drosophila melanogaster
- Regular article
- Effects of MgCl2 supplementation on blood parameters and kidney injury of rats exposed to CCl4
- Regular article
- Effective onion leaf fleck management and variability of storage pathogens
- Regular article
- Improving aeration for efficient oxygenation in sea bass sea cages. Blood, brain and gill histology
- Regular article
- Biogenic amines and hygienic quality of lucerne silage
- Regular article
- Isolation and characterization of lytic phages TSE1-3 against Enterobacter cloacae
- Regular article
- Effects of pH on antioxidant and prooxidant properties of common medicinal herbs
- Regular article
- Relationship between cytokines and running economy in marathon runners
- Regular article
- Anti-leukemic activity of DNA methyltransferase inhibitor procaine targeted on human leukaemia cells
- Regular article
- Research Progress in Oncology. Highlighting and Exploiting the Roles of Several Strategic Proteins in Understanding Cancer Biology
- Regular article
- Ectomycorrhizal communities in a Tuber aestivum Vittad. orchard in Poland
- Regular article
- Impact of HLA-G 14 bp polymorphism and soluble HLA-G level on kidney graft outcome
- Regular article
- In-silico analysis of non-synonymous-SNPs of STEAP2: To provoke the progression of prostate cancer
- Regular article
- Presence of sequence and SNP variation in the IRF6 gene in healthy residents of Guangdong Province
- Regular article
- Environmental and economic aspects of Triticum aestivum L. and Avena sativa growing
- Regular article
- A molecular survey of Echinococcus granulosus sensu lato in central-eastern Europe
- Topical Issue on Cancer Signaling, Metastasis and Target Therapy
- Molecular genetics related to non-Hodgkin lymphoma
- Topical Issue on Cancer Signaling, Metastasis and Target Therapy
- Roles of long noncoding RNAs in Hepatocellular Carcinoma
- Topical Issue on Cancer Signaling, Metastasis and Target Therapy
- Advancement of Wnt signal pathway and the target of breast cancer
- Topical Issue on Cancer Signaling, Metastasis and Target Therapy
- A tumor suppressive role of lncRNA GAS5 in human colorectal cancer
- Topical Issue on Cancer Signaling, Metastasis and Target Therapy
- The role of E-cadherin - 160C/A polymorphism in breast cancer
- Topical Issue on Cancer Signaling, Metastasis and Target Therapy
- The proceedings of brain metastases from lung cancer
- Topical Issue on Cancer Signaling, Metastasis and Target Therapy
- Newly-presented potential targeted drugs in the treatment of renal cell cancer
- Topical Issue on Cancer Signaling, Metastasis and Target Therapy
- Decreased expression of miR-132 in CRC tissues and its inhibitory function on tumor progression
- Topical Issue on Cancer Signaling, Metastasis and Target Therapy
- The unusual yin-yang fashion of RIZ1/RIZ2 contributes to the progression of esophageal squamous cell carcinoma
- Topical Issue on Cancer Signaling, Metastasis and Target Therapy
- Human papillomavirus infection mechanism and vaccine of vulva carcinoma
- Topical Issue on Cancer Signaling, Metastasis and Target Therapy
- Abnormal expressed long non-coding RNA IRAIN inhibits tumor progression in human renal cell carcinoma cells
- Topical Issue on Cancer Signaling, Metastasis and Target Therapy
- UCA1, a long noncoding RNA, promotes the proliferation of CRC cells via p53/p21 signaling
- Topical Issue on Cancer Signaling, Metastasis and Target Therapy
- Forkhead box 1 expression is upregulatedin non-small cell lung cancer and correlateswith pathological parameters
- Topical Issue on Cancer Signaling, Metastasis and Target Therapy
- The development of potential targets in the treatment of non-small cell lung cancer
- Topical Issue on Cancer Signaling, Metastasis and Target Therapy
- Low expression of miR-192 in NSCLC and its tumor suppressor functions in metastasis via targeting ZEB2
- Topical Issue on Cancer Signaling, Metastasis and Target Therapy
- Downregulation of long non-coding RNA MALAT1 induces tumor progression of human breast cancer through regulating CCND1 expression
- Topical Issue on Cancer Signaling, Metastasis and Target Therapy
- Post-translational modifications of EMT transcriptional factors in cancer metastasis
- Topical Issue on Cancer Signaling, Metastasis and Target Therapy
- EZH2 Expression and its Correlation with Clinicopathological Features in Patients with Colorectal Carcinoma
- Topical Issue on Cancer Signaling, Metastasis and Target Therapy
- The association between EGFR expression and clinical pathology characteristics in gastric cancer
- Topical Issue on Cancer Signaling, Metastasis and Target Therapy
- The peiminine stimulating autophagy in human colorectal carcinoma cells via AMPK pathway by SQSTM1
- Topical Issue on Cancer Signaling, Metastasis and Target Therapy
- Activating transcription factor 3 is downregulated in hepatocellular carcinoma and functions as a tumor suppressor by regulating cyclin D1
- Topical Issue on Cancer Signaling, Metastasis and Target Therapy
- Progress toward resistance mechanism to epidermal growth factor receptor tyrosine kinase inhibitor
- Topical Issue on Cancer Signaling, Metastasis and Target Therapy
- Effect of miRNAs in lung cancer suppression and oncogenesis
- Topical Issue on Cancer Signaling, Metastasis and Target Therapy
- Role and inhibition of Src signaling in the progression of liver cancer
- Topical Issue on Cancer Signaling, Metastasis and Target Therapy
- The antitumor effects of mitochondria-targeted 6-(nicotinamide) methyl coumarin
- Special Issue on CleanWAS 2015
- Characterization of particle shape, zeta potential, loading efficiency and outdoor stability for chitosan-ricinoleic acid loaded with rotenone
- Special Issue on CleanWAS 2015
- Genetic diversity and population structure of ginseng in China based on RAPD analysis
- Special Issue on CleanWAS 2015
- Optimizing the extraction of antibacterial compounds from pineapple leaf fiber
- Special Issue on CleanWAS 2015
- Identification of residual non-biodegradable organic compounds in wastewater effluent after two-stage biochemical treatment
- Special Issue on CleanWAS 2015
- Remediation of deltamethrin contaminated cotton fields: residual and adsorption assessment
- Special Issue on CleanWAS 2015
- A best-fit probability distribution for the estimation of rainfall in northern regions of Pakistan
- Special Issue on CleanWAS 2015
- Artificial Plant Root System Growth for Distributed Optimization: Models and Emergent Behaviors
- Special Issue on CleanWAS 2015
- The complete mitochondrial genomes of two weevils, Eucryptorrhynchus chinensis and E. brandti: conserved genome arrangement in Curculionidae and deficiency of tRNA-Ile gene
- Special Issue on CleanWAS 2015
- Characteristics and coordination of source-sink relationships in super hybrid rice
- Special Issue on CleanWAS 2015
- Construction of a Genetic Linkage Map and QTL Analysis of Fruit-related Traits in an F1 Red Fuji x Hongrou Apple Hybrid
- Special Issue on CleanWAS 2015
- Effects of the Traditional Chinese Medicine Dilong on Airway Remodeling in Rats with OVA-induced-Asthma
- Special Issue on CleanWAS 2015
- The effect of sewage sludge application on the growth and absorption rates of Pb and As in water spinach
- Special Issue on CleanWAS 2015
- Effectiveness of mesenchymal stems cells cultured by hanging drop vs. conventional culturing on the repair of hypoxic-ischemic-damaged mouse brains, measured by stemness gene expression

