Abstract
[SbCl3(C9H11N3S2)2], monoclinic, P21/n (no. 14), a = 13.8069(4) Å, b = 13.5739(6) Å, c = 14.0182(5) Å, β = 94.231(3)°, V = 2,620.04(17) Å3, Z = 4, R gt (F) = 0.0329, wRref(F2) = 0.0726, T = 153 K.
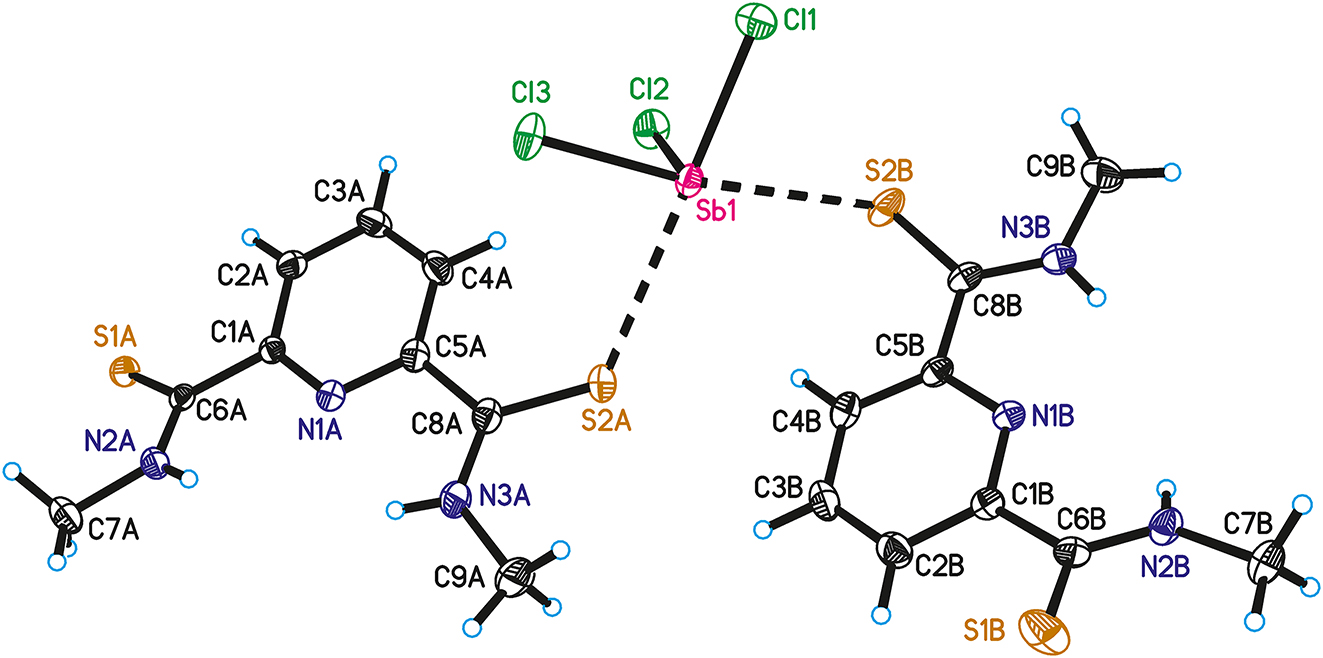
The molecular structure is shown in the figure. Table 1 contains crystallographic data and Table 2 contains the list of the atoms including atomic coordinates and displacement parameters.
Data collection and handling.
| Crystal: | Yellow prism |
| Size: | 0.32 × 0.28 × 0.20 mm |
| Wavelength: | Mo Kα radiation (0.71073 Å) |
| μ: | 1.70 mm−1 |
| Diffractometer, scan mode: | STOE IPDS 2, rotation method |
| θmax, completeness: | 27.5°, >99 % |
| N(hkl)measured, N(hkl)unique, Rint: | 34,269, 5,965, 0.039 |
| Criterion for Iobs, N(hkl)gt: | Iobs > 2 σ(Iobs), 5,231 |
| N(param)refined: | 309 |
| Programs: | X-RED, 1 X-AREA, 1 SHELX, 2 , 3 ORTEP-3 4 |
Fractional atomic coordinates and isotropic or equivalent isotropic displacement parameters (Å2).
| Atom | x | y | z | Uiso*/Ueq |
|---|---|---|---|---|
| Sb1 | 0.64366 (2) | 0.24601 (2) | 0.53951 (2) | 0.01768 (6) |
| Cl1 | 0.78386 (6) | 0.14400 (6) | 0.57836 (6) | 0.02799 (17) |
| Cl2 | 0.67120 (6) | 0.33885 (6) | 0.68384 (5) | 0.02716 (16) |
| Cl3 | 0.53975 (6) | 0.13044 (6) | 0.61169 (6) | 0.03006 (18) |
| S1A | 0.08071 (5) | 0.39812 (6) | 0.86473 (5) | 0.02098 (15) |
| S2A | 0.45442 (6) | 0.36408 (9) | 0.47917 (7) | 0.0397 (2) |
| N1A | 0.23472 (17) | 0.34382 (17) | 0.64476 (17) | 0.0166 (5) |
| N2A | 0.05054 (18) | 0.3262 (2) | 0.69011 (19) | 0.0208 (5) |
| H1N | 0.069 (3) | 0.310 (3) | 0.638 (3) | 0.024 (10)* |
| N3A | 0.2649 (2) | 0.3541 (2) | 0.46135 (19) | 0.0243 (6) |
| H2N | 0.211 (3) | 0.348 (3) | 0.488 (3) | 0.051 (13)* |
| C1A | 0.2180 (2) | 0.34679 (19) | 0.7378 (2) | 0.0148 (5) |
| C2A | 0.2914 (2) | 0.3423 (2) | 0.8109 (2) | 0.0197 (6) |
| H2A | 0.276750 | 0.345868 | 0.875933 | 0.024* |
| C3A | 0.3865 (2) | 0.3326 (2) | 0.7867 (2) | 0.0240 (6) |
| H3A | 0.438097 | 0.326279 | 0.834938 | 0.029* |
| C4A | 0.4047 (2) | 0.3324 (2) | 0.6912 (2) | 0.0235 (6) |
| H4A | 0.469456 | 0.327467 | 0.672841 | 0.028* |
| C5A | 0.3277 (2) | 0.3395 (2) | 0.6223 (2) | 0.0186 (6) |
| C6A | 0.1134 (2) | 0.3545 (2) | 0.7597 (2) | 0.0158 (5) |
| C7A | −0.0541 (2) | 0.3225 (3) | 0.6984 (3) | 0.0315 (8) |
| H7A | −0.068097 | 0.279183 | 0.751592 | 0.047* |
| H7B | −0.086215 | 0.296775 | 0.638804 | 0.047* |
| H7C | −0.078327 | 0.388874 | 0.710227 | 0.047* |
| C8A | 0.3439 (2) | 0.3505 (2) | 0.5183 (2) | 0.0204 (6) |
| C9A | 0.2612 (3) | 0.3709 (3) | 0.3583 (2) | 0.0327 (8) |
| H9A | 0.273164 | 0.440766 | 0.345879 | 0.049* |
| H9B | 0.197014 | 0.352417 | 0.329416 | 0.049* |
| H9C | 0.311049 | 0.330898 | 0.330463 | 0.049* |
| S1B | 0.59722 (6) | 0.32279 (8) | −0.03756 (7) | 0.0350 (2) |
| S2B | 0.80156 (6) | 0.40692 (6) | 0.48082 (5) | 0.02731 (17) |
| N1B | 0.73429 (17) | 0.40148 (17) | 0.20157 (17) | 0.0166 (5) |
| N2B | 0.76630 (19) | 0.4087 (2) | 0.01454 (18) | 0.0218 (5) |
| H3N | 0.800 (3) | 0.440 (3) | 0.059 (3) | 0.023 (9)* |
| N3B | 0.88898 (19) | 0.4177 (2) | 0.31998 (19) | 0.0219 (5) |
| H4N | 0.888 (3) | 0.421 (3) | 0.262 (3) | 0.035 (11)* |
| C1B | 0.6576 (2) | 0.4000 (2) | 0.1375 (2) | 0.0180 (6) |
| C2B | 0.5622 (2) | 0.4135 (2) | 0.1621 (2) | 0.0245 (6) |
| H2B | 0.509376 | 0.413291 | 0.114652 | 0.029* |
| C3B | 0.5472 (2) | 0.4273 (3) | 0.2575 (2) | 0.0272 (7) |
| H3B | 0.483334 | 0.437372 | 0.276534 | 0.033* |
| C4B | 0.6256 (2) | 0.4264 (2) | 0.3255 (2) | 0.0222 (6) |
| H4B | 0.616374 | 0.434493 | 0.391498 | 0.027* |
| C5B | 0.7176 (2) | 0.4134 (2) | 0.2941 (2) | 0.0175 (5) |
| C6B | 0.6791 (2) | 0.3809 (2) | 0.0363 (2) | 0.0196 (6) |
| C7B | 0.8055 (3) | 0.3971 (3) | −0.0785 (2) | 0.0284 (7) |
| H7D | 0.856477 | 0.346790 | −0.074292 | 0.043* |
| H7E | 0.832859 | 0.459950 | −0.098104 | 0.043* |
| H7F | 0.753427 | 0.376911 | −0.125787 | 0.043* |
| C8B | 0.8070 (2) | 0.4124 (2) | 0.3621 (2) | 0.0184 (6) |
| C9B | 0.9852 (2) | 0.4178 (3) | 0.3698 (3) | 0.0340 (8) |
| H9D | 0.994569 | 0.478947 | 0.406517 | 0.051* |
| H9E | 1.034461 | 0.413155 | 0.323167 | 0.051* |
| H9F | 0.991378 | 0.361325 | 0.413434 | 0.051* |
1 Source of materials
The synthesis of the title compound was performed using standard Schlenk technique 5 with Argon (5.9 Nippon gases) as inert gas. Pyridine-2,6-dithiocarbomethylamide was prepared according to a literature procedure. 6
In a representative experiment 495 mg (2.2 mmol) of pyridine-2,6-dithiocarbomethylamide was dissolved in 10 mL THF (purified in Mbraun SPS 800, VWR AnalaR). The solution was cooled in an ice-ethanol bath to −10 °C. 2 mL of n-butyllithium (2.5 M in hexanes, Merck) were added slowly. The solution turns red and was allowed to reach room temperature over 20 min. Afterward the solution was cooled down to −10 °C again and 0.5 g of SbCl3 (Merck, distilled before use, 2.2 mmol) dissolved in THF (40 mass%) and 10 mL of pure THF were added. The solution turned yellowish-green immediately. The solution was stirred at room temperature overnight. The solvent was removed under reduced pressure and was replaced with 10 mL of chloroform (Fisher scientific, stabilized with amylene, dried over molecular sieves 3 Å and activated alumina). The suspension was filtrated, the residue was washed with additional 5 mL of CHCl3. The solvent was removed under reduced pressure from the filtrate. 1 mL of CDCl3 was added for NMR. NMR analysis of the bulk product showed the formation of a product mixture. From the residue suspension orange crystals of the title compound suitable for X-ray diffraction were obtained after 6 weeks standing at room temperature. Large yellow prisms, decomposition point: 157 °C.
2 Experimental details
The carbon-bound H atoms were geometrically placed (C–H = 0.95–0.98 Å) and refined as riding atoms with Uiso(H) = 1.2–1.5 Ueq(C). The N-bound H-atoms were located in a difference Fourier map and their positions and isotropic displacement parameters were refined using isotropic approximation.
3 Comment
Pyridine-2,6-dithiocarbomethylamide has been used as chelate ligand to prepare several nickel, iron, and copper complexes. 6 , 7 , 8 , 9 Herein we describe the crystal structure of an adduct formed out of pyridine-2,6-dithiocarbomethylamide and antimony trichloride. The crystal structure was obtained as a side product during our work about chelate complexes of antimony. 10 , 11
The asymmetric unit of the crystal structure contains one molecule antimony trichloride, SbCl3, and two molecules of pyridine-2,6-dithiocarbomethylamide. The formation of a chelate complex, as initially intended, was not observed. Instead an adduct or simple coordination compound was formed, wherein three pyridine-2,6-dithiocarbomethylamide molecules coordinate via one sulfur atom each at the antimony atom. The bond lengths Sb1–Cl1 [2.4095(8) Å], Sb1–Cl2 [2.3901(8) Å], and Sb1–Cl3 [2.3995(8) Å] correspond well with the sum of covalent radii of 2.40 Å [r(Sb) = 1.41 Å, r(Cl) = 0.99 Å]. 12 The atomic distances Sb1⋯S2A [3.128(1) Å], Sb1⋯S2B [3.2349(8) Å] and Sb1⋯S1A [3.2050(8) Å] are substantial longer than the sum of covalent radii of 2.45 Å [r(S) = 1.04 Å], but shorter than the sum of van der Waals radii of 3.86 Å [r(Sb) = 2.06 Å, r(S) = 1.80 Å]. 13 The last interaction between Sb1 and S1A is not shown in the figure, since the sulfur atom belongs to a symmetry equivalent molecule of pyridine-2,6-dithiocarbomethylamide on the position x + 1/2, −y + 1/2, z − 1/2. The interactions with three chlorine and three sulfur atoms making the antimony atom hexacoordinated. The coordination geometry at the antimony atom can be classified as distorted octahedral. The trans bond angles vary between 169.68(3)° for Cl3–Sb1–S2B and 175.32(3)° for Cl1–Sb1–S2A. The cis bond angles in the distorted octahedron vary from 77.67(2)° for Cl3–Sb1–S1A to 111.57(2)° for S2B–Sb1–S1A.
The thiocarbomethylamide units of the ligand molecules have conformational flexibility to rotate around the bonds C1–C6 and C5–C8 (in both molecules A and B). Both groups adopt a conformation in the solid state where the sulfur atoms are oriented away from the pyridine nitrogen atom. The same conformation of thiocarbomethylamide units was observed in the crystal structure of pyridine-2,6-dithiocarbomethylamide. 14 This conformation allows the formation of hydrogen bonds between the N–H groups at N2 and N3 with the pyridine nitrogen atom N1 (in molecules A and B). On the other side, the sulfur atoms are oriented in lateral positions at the pyridine-2,6-dithiocarbomethylamide and allow coordination with the antimony atom in the present structure. Further hydrogen bonds are formed between N–H respectively C–H groups and sulfur atoms, forming a complicated threedimensional network in the solid state.
There are a few related crystal structures containing pyridine-2,6-dithiocarbomethylamide as ligand in nickel and copper complexes. 7 , 8 , 9 However, a comparison of these structures with the title compound is not fruitful, since the ligand molecule acts in these complexes as chelate ligand.
Acknowledgments
The authors thank TU Bergakademie Freiberg (Freiberg, Germany) for financial support. Open Access Funding by the Publication Fund of the TU Bergakademie Freiberg.
-
Author contribution: All the authors have accepted responsibility for the entire content of this submitted manuscript and approved submission.
-
Conflict of interest statement: The authors declare no conflicts of interest regarding this article.
-
Research funding: TU Bergakademie Freiberg (Freiberg, Germany).
References
1. Stoe & Cie. X-RED (Version 1.53) and X-AREA (Version 1.55): Darmstadt, Germany, 2009.Search in Google Scholar
2. Sheldrick, G. M. SHELXT – Integrated Space-Group and Crystal-Structure Determination. Acta Crystallogr. 2015, A71, 3–8; https://doi.org/10.1107/s2053273314026370.Search in Google Scholar
3. Sheldrick, G. M. Crystal Structure Refinement with SHELXL. Acta Crystallogr. 2015, C71, 3–8; https://doi.org/10.1107/s2053229614024218.Search in Google Scholar
4. Farrugia, L. J. ORTEP-3 for Windows – A Version of ORTEPIII with a Graphical User Interface (GUI). J. Appl. Crystallogr. 1997, 30, 565; https://doi.org/10.1107/S0021889897003117.Search in Google Scholar
5. Böhme, U. Inertgastechnik – Arbeiten unter Schutzgas in der Chemie, 1. Auflage; Walter de Gruyter GmbH: Berlin/Boston, 2020. ISBN: 978-3-11-062703-9.10.1515/9783110627046Search in Google Scholar
6. Gagliardi, E.; Popitsch, A. Über das koordinationschemische Verhalten von Pyridin-2,6-di-thiocarbomethylamid. Monatsh. Chem. 1972, 103, 1337–1348; https://doi.org/10.1007/bf00904517.Search in Google Scholar
7. Kratky, C.; Schurz, J.; Gagliardi, E.; Popitsch, A. Crystal Structure and Spectra of the Ni(II) Complex of Pyridine-2,6-Dithio-Carbomethylamide. Monatsh. Chem. 1981, 112, 721–730; https://doi.org/10.1007/bf00899775.Search in Google Scholar
8. Kratky, C.; Jorde, C.; Popitsch, A. Structure of Cu(II) Complexes of Pyridine-2,6-Dithiocarbomethylamide. Monatsh. Chem. 1982, 113, 933–940; https://doi.org/10.1007/bf00799234.Search in Google Scholar
9. Kratky, C.; Jorde, C.; Popitsch, A. Crystal Structures of Cu(II) Complexes of Pyridine-2,6-Dithiocarbomethylamide. Monatsh. Chem. 1983, 114, 829–838; https://doi.org/10.1007/bf00799945.Search in Google Scholar
10. Böhme, U.; Herbig, M. New Complexes of Antimony(III) with Tridentate O,E,O–Ligands (E = O, S, Se, Te, NH, NMe) Derived from N–Methyldiethanolamine. Molecules 2023, 28, 4959. https://doi.org/10.3390/molecules28134959.Search in Google Scholar PubMed PubMed Central
11. Knerr, S.; Böhme, U.; Herbig, M. Coordinative Unsaturation in an Antimony(III)–Complex with the 2-Salicylidenaminophenolato Ligand: Synthesis, Crystal Structure, Spectroscopic Analysis, and DFT Studies. Crystals 2023, 13, 1300. https://doi.org/10.3390/cryst13091300.Search in Google Scholar
12. Pauling, L. Die Natur der Chemischen Bindung; Verl. Chemie: Weinheim: Germany, 1962.Search in Google Scholar
13. Mantina, M.; Chamberlin, A. C.; Valero, R.; Cramer, C. J.; Truhlar, D. G. Consistent van der Waals Radii for the Whole Main Group. J. Phys. Chem. A 2009, 113, 5806–5812; https://doi.org/10.1021/jp8111556.Search in Google Scholar PubMed PubMed Central
14. Popitsch, A.; Gagliardi, E.; Schurz, J.; Kratky, C. Crystal Structure and Vibrational Spectra of Pyridine-2,6-Dithio-Carbomethylamide. Monatsh. Chem. 1981, 112, 537–545; https://doi.org/10.1007/bf00899668.Search in Google Scholar
© 2024 the author(s), published by De Gruyter, Berlin/Boston
This work is licensed under the Creative Commons Attribution 4.0 International License.
Articles in the same Issue
- Frontmatter
- Editorial
- Editorial 2024 – New developments and changes of Zeitschrift für Kristallographie – New Crystal Structures
- New Crystal Structures
- Hydrogen bonding and π⋅⋅⋅halogen interactions in the crystal structure of bis(theophyllinium) hexachloridoplatinate(IV) monohydrate
- The crystal structure of 6-amino-2-carboxypyridin-1-ium perchlorate, C6H7ClN2O6
- Crystal structure of poly[(μ4-(3-amino-1H-1,2,4-triazol-1-yl)benzene-1,3-dicarboxylato-κ 4 N:O:O':O')(1-methylpyrroldin-2-one-κ1O)dicopper(II)] – 1-methylpyrroldin-2-one (1/3), C40H48Cu2N12O12
- The crystal structure of 18-crown-6-k6O6(2,4,5-trinitroimidazol-1-ido-k1O)potassium(I)
- Crystal structure of poly[tetraaqua-bis(μ2-5-bromoisophthalato-κ3O,O′:O″)-(μ2-1,5-bis(imidazol-2-methyl)pentane-κ2N:N′)dicadmium(II)] dihydrate
- Crystal structure of (5R,6S,E)-5-acetoxy-2-methyl-6-((2aR,3R,5aS,5bS,11aR,12aS)-2a,5a,8,8-tetramethyl-9-oxotetradecahydro-1H,12H-cyclopenta[a]cyclopropa[e]phenanthren-3-yl)hept-2-enoic acid, C32H48O5
- The crystal structure of poly[diaqua-bis(μ2 -thiocyanato-κ2N:O)cobalt(II) monohydrate
- The crystal structure of 1,3,5-tri(1H-imidazol-1-yl)benzene–2,3,5,6-tetrachlorobenzene-1,4-dicarboxylic acid (1/1)
- Crystal structure of dichlorido-bis(1-[(2-ethyl-benzimidazole-1-yl)methyl]-1H–benzotriazole) cadmium(II), C32H32CdN10OCl2
- The crystal structure of N′-(tert-butyl)-N′-(3,5-dimethylbenzoyl)-3-methoxy-N,2-dimethylbenzohydrazide, C23H30N2O3
- Crystal stucture of 3-benzamido-N-(2-bromo-4-(perfluoropropan-2-yl)-6-(trifluoromethyl)phenyl)-2-fluorobenzamide
- Crystal structure of bis(μ-benzeneselenolato)-(tetracarbonyl)-{μ-[N-(diphenylphosphanyl)-N-(3-ethynylphenyl)-P,P-diphenylphosphinous amide]} diiron, C48H35Fe2NO4P2Se2
- The crystal structure of 2′-(p-tolyl)-4′H-spiro[isochromane-1,1′-naphthalene]-3,4′-dione, C25H18O3
- The crystal structure of poly[hexaqua-tetrakis(μ4-pyridine-2,4-dicarboxylate-κ5N: O: O′: O″: O‴)-bi(μ2-pyridine-2,4-dicarboxylate-κ3N: O: O′)-digadolinium(III)tricopper (II)], [Gd2Cu3(C7H3NO4)6(H2O)6] n
- Crystal structure of poly[bis(4-(4-(pyridin-4-yl)phenyl)pyridin-1-ium-κ1N)-(μ4-benzene-1,2,4,5-tetracarboxylato-κ5O:O′: O″:O‴:O⁗)-(μ2-2,5-dicarboxyterephthalato-κ2O:O′)dizinc(II)], C52H32N4O16Zn2
- The crystal structure of 4-(3-carboxy-1-ethyl-6-fluoro-4-oxo-1,4-dihydroquinolin-7-yl)piperazin-1-ium 2-carboxy-6-nitrobenzoate monohydrate, C24H25FN4O10
- Crystal structure of dichlorido-(1-((3,5-dimethyl-2,3-dihydro-1H-1,2,3-triazol-1-yl)methyl)-1H-benzo[d][1,2,3]triazole-k1N)zinc(II), C22H24ZnN12Cl2
- The crystal structure of (3-chlorothiophene-2-carboxylato-κ2O, O′)-(2,2′-dipyridyl-κ2N,N′)lead(II), C20H12Cl2N2O4S2Pb
- Synthesis and crystal structure of (Z)-4-((1-(3-fluorophenyl)-1H-1,2,3-triazol-4-yl)methylene)-5-methyl-2-phenyl-2,4-dihydro-3H-pyrazol-3-one, C19H14FN5O
- The crystal structure of the coordination compound catena-poly[(18-crown-6-ether-κ6O6)(4,5-dinitroimidazolato-κ1O)potassium(I)]
- Crystal structure of 7-(diethylamino)-3-(trifluoroacetyl)-2H-chromen-2-one, C15H14F3NO3
- Crystal structure of dichlorido-1-[(2-ethylimidazole-1-yl)methyl]-1H–benzotriazole κ1N zinc(II), C24H26ZnN10Cl2
- Crystal and molecular structure of 5-bromopyridine-2,3-diamine
- Crystal structure of catena-poly[bis(μ2-1-(3-carboxyphenyl)-5-methyl-4-oxo-1,4-dihydropyridazine-3-carboxylato-k3-O,O′:O″)hexaqua-dicobalt tetrahydrate], C26H36N4O20Co2
- Crystal structure of thiocyanate-κ1N-bis(μ1-2,6-diformyl-4-methylphenol oxime-κ2N,O)-manganese(III) acetonitrile solvate, C21H21MnN6O6S
- The crystal structure of pyrrolidin-1-yl pivalate, C9H13NO4
- The crystal structure of 2,2′-(2,2-diphenylethene-1,1-diyl)bis(1,4-dimethoxybenzene), C30H28O4
- Crystal structure of bis(benzyltrimethylammonium) tetrathiotungstate(VI), {(C6H5CH2)(CH3)3N}2[WS4]
- The crystal structure of ethyl (Z)-2-(ethoxymethylene)-3-oxobutanoate, C9H14O4
- The crystal structure of (E)-6-bromo-3,5-dimethyl-2-(1-phenylprop-1-en-2-yl)-3Himidazo[4,5b]pyridine, C17H16BrN3
- Crystal structure of (3S,3′S,4R,4′S)-3′-(furan-3-yl)-3-hydroxy-4′-methyl-3,5,6′,7′-tetrahydro-1H,3′H-4,5′-spirobi[isobenzofuran]-1,1′(4′H)-dione-methanol (1/1), C21H22O7
- Cocrystal structure of progesterone-isophthalic acid, C25H33O4
- The crystal structure of 3-(6-fluoro-1H-indol-3-yl)-1-methylquinoxalin-2(1H)-one, C17H12FN3O
- Crystal structure of S-(4-carboxybutyl)- l -cysteine
- The cocrystal of 2,2′-(hydrazine-1,1-diyl)bis(1H-imidazole-4,5-dicarbonitrile)– methanol (2/3)
- Crystal structure of (1′R,2′S,4′R,6′S)-4,6-dihydroxy-1′,8′,8′-trimethyl-3-(3-methylbutanoyl)-4′,8′,6′,1′,7,2′-hexahydro-1H-4′,6′-methanoxanthene-8-carbaldehyde, C23H30O5
- Crystal structure of (3,6-di(2-pyridyl)-4-methylphenyl pyridazine-k 2 N,N′)-bis(1-phenyl-pyrazole-κ 2 C,N) iridium(III) hexafluorophosphate, C39H29F6IrN8P
- Crystal structure of 1,5-bis[(E)-1-(2-hydroxyphenyl)ethylidene]thiocarbonohydrazide dimethyl sulfoxide monosolvate, C17H18N4O2S·C2H6OS
- Crystal structure of (S)-4-(2-(4-(2-acetyl-5-chlorophenyl)-3-methoxy-6-oxopyridazin-1(6H)-yl)-3-phenylpropanamido)benzoic acid monohydrate, C29H26ClN3O7
- The crystal structure of 1,3-bis(2,4-dinitro-1H-imidazol-1-yl)propane
- Crystal structure of 4-chlorobenzyl (S)-2-(6-methoxynaphthalen-2-yl)propanoate, C21H19ClO3
- Crystal structure of 1-(5-(benzo[d][1,3]dioxol-5-yl)-4-benzyl-1-(4-bromophenyl)-4,5-dihydro-1H-1,2,4-triazol-3-yl)ethan-1-one, C24H20BrN3O3
- The crystal structure of (Z)-3′-(2-(1-(3,4-dimethyl-phenyl)-3-methyl-5-oxo-1,5-dihydro-4H-pyrazol-4-ylidene)hydrazinyl)-2′-hydroxy-[1,1′-biphenyl]-3-carboxylicacid ─ methanol (1/1), C26H26N4O5
- Crystal structure of (S)-1-phenylpropan-1-aminium (S)-(1-phenylpropyl)carbamate C19H26N2O2
- Synthesis and crystal structure of methyl 2-((5-bromo-4-(4-cyclopropylnaphthalen-1-yl)-4H-1,2,4-triazol-3-yl)thio)acetate, C18H16BrN3O2S
- The crystal structure of trichlorobis(pyridine-2,6-dithio-κS-carbomethylamido)antimony(III), [SbCl3(C9H11N3S2)2]
- Crystal structure of 1,8-dihydroxy-3-{[(triphenylstannyl)oxy]carbonyl} anthracene-9,10-dione, C33H22O6Sn
- The crystal structure of (E)-4-(2-(pyridin-4-ylmethylene)hydrazine-1-carbonyl)pyridin-1-ium-2-olate dihydrate, C12H14N4O4
- The crystal structure of 6-amino-pyridinium-2-carboxylate, C6H6N2O2
- The crystal structure of catena-poly[aqua-nitrato-κ3O,O:O′′-(1,10-phenanthroline-κ2N,N′)sodium(I)], C24H18N6O7Na2
- Retractions
- Retraction of: Crystal structure of bis[diaquaisonicotinatosamarium(III)]-µ-isonicotinato-[diisonicotinatocopper(II)], CuSm2(C6H4NO2)8(H2O)4
- Retraction of: Crystal structure of aqua(2,2′-bipyridine-k 2 N:N′)(nitrato)-(4-aminobenzoato)cadmium(II) nitrate, [Cd(H2O)(NO3)(C10H8N2)(C7H7NO2)][NO3]
Articles in the same Issue
- Frontmatter
- Editorial
- Editorial 2024 – New developments and changes of Zeitschrift für Kristallographie – New Crystal Structures
- New Crystal Structures
- Hydrogen bonding and π⋅⋅⋅halogen interactions in the crystal structure of bis(theophyllinium) hexachloridoplatinate(IV) monohydrate
- The crystal structure of 6-amino-2-carboxypyridin-1-ium perchlorate, C6H7ClN2O6
- Crystal structure of poly[(μ4-(3-amino-1H-1,2,4-triazol-1-yl)benzene-1,3-dicarboxylato-κ 4 N:O:O':O')(1-methylpyrroldin-2-one-κ1O)dicopper(II)] – 1-methylpyrroldin-2-one (1/3), C40H48Cu2N12O12
- The crystal structure of 18-crown-6-k6O6(2,4,5-trinitroimidazol-1-ido-k1O)potassium(I)
- Crystal structure of poly[tetraaqua-bis(μ2-5-bromoisophthalato-κ3O,O′:O″)-(μ2-1,5-bis(imidazol-2-methyl)pentane-κ2N:N′)dicadmium(II)] dihydrate
- Crystal structure of (5R,6S,E)-5-acetoxy-2-methyl-6-((2aR,3R,5aS,5bS,11aR,12aS)-2a,5a,8,8-tetramethyl-9-oxotetradecahydro-1H,12H-cyclopenta[a]cyclopropa[e]phenanthren-3-yl)hept-2-enoic acid, C32H48O5
- The crystal structure of poly[diaqua-bis(μ2 -thiocyanato-κ2N:O)cobalt(II) monohydrate
- The crystal structure of 1,3,5-tri(1H-imidazol-1-yl)benzene–2,3,5,6-tetrachlorobenzene-1,4-dicarboxylic acid (1/1)
- Crystal structure of dichlorido-bis(1-[(2-ethyl-benzimidazole-1-yl)methyl]-1H–benzotriazole) cadmium(II), C32H32CdN10OCl2
- The crystal structure of N′-(tert-butyl)-N′-(3,5-dimethylbenzoyl)-3-methoxy-N,2-dimethylbenzohydrazide, C23H30N2O3
- Crystal stucture of 3-benzamido-N-(2-bromo-4-(perfluoropropan-2-yl)-6-(trifluoromethyl)phenyl)-2-fluorobenzamide
- Crystal structure of bis(μ-benzeneselenolato)-(tetracarbonyl)-{μ-[N-(diphenylphosphanyl)-N-(3-ethynylphenyl)-P,P-diphenylphosphinous amide]} diiron, C48H35Fe2NO4P2Se2
- The crystal structure of 2′-(p-tolyl)-4′H-spiro[isochromane-1,1′-naphthalene]-3,4′-dione, C25H18O3
- The crystal structure of poly[hexaqua-tetrakis(μ4-pyridine-2,4-dicarboxylate-κ5N: O: O′: O″: O‴)-bi(μ2-pyridine-2,4-dicarboxylate-κ3N: O: O′)-digadolinium(III)tricopper (II)], [Gd2Cu3(C7H3NO4)6(H2O)6] n
- Crystal structure of poly[bis(4-(4-(pyridin-4-yl)phenyl)pyridin-1-ium-κ1N)-(μ4-benzene-1,2,4,5-tetracarboxylato-κ5O:O′: O″:O‴:O⁗)-(μ2-2,5-dicarboxyterephthalato-κ2O:O′)dizinc(II)], C52H32N4O16Zn2
- The crystal structure of 4-(3-carboxy-1-ethyl-6-fluoro-4-oxo-1,4-dihydroquinolin-7-yl)piperazin-1-ium 2-carboxy-6-nitrobenzoate monohydrate, C24H25FN4O10
- Crystal structure of dichlorido-(1-((3,5-dimethyl-2,3-dihydro-1H-1,2,3-triazol-1-yl)methyl)-1H-benzo[d][1,2,3]triazole-k1N)zinc(II), C22H24ZnN12Cl2
- The crystal structure of (3-chlorothiophene-2-carboxylato-κ2O, O′)-(2,2′-dipyridyl-κ2N,N′)lead(II), C20H12Cl2N2O4S2Pb
- Synthesis and crystal structure of (Z)-4-((1-(3-fluorophenyl)-1H-1,2,3-triazol-4-yl)methylene)-5-methyl-2-phenyl-2,4-dihydro-3H-pyrazol-3-one, C19H14FN5O
- The crystal structure of the coordination compound catena-poly[(18-crown-6-ether-κ6O6)(4,5-dinitroimidazolato-κ1O)potassium(I)]
- Crystal structure of 7-(diethylamino)-3-(trifluoroacetyl)-2H-chromen-2-one, C15H14F3NO3
- Crystal structure of dichlorido-1-[(2-ethylimidazole-1-yl)methyl]-1H–benzotriazole κ1N zinc(II), C24H26ZnN10Cl2
- Crystal and molecular structure of 5-bromopyridine-2,3-diamine
- Crystal structure of catena-poly[bis(μ2-1-(3-carboxyphenyl)-5-methyl-4-oxo-1,4-dihydropyridazine-3-carboxylato-k3-O,O′:O″)hexaqua-dicobalt tetrahydrate], C26H36N4O20Co2
- Crystal structure of thiocyanate-κ1N-bis(μ1-2,6-diformyl-4-methylphenol oxime-κ2N,O)-manganese(III) acetonitrile solvate, C21H21MnN6O6S
- The crystal structure of pyrrolidin-1-yl pivalate, C9H13NO4
- The crystal structure of 2,2′-(2,2-diphenylethene-1,1-diyl)bis(1,4-dimethoxybenzene), C30H28O4
- Crystal structure of bis(benzyltrimethylammonium) tetrathiotungstate(VI), {(C6H5CH2)(CH3)3N}2[WS4]
- The crystal structure of ethyl (Z)-2-(ethoxymethylene)-3-oxobutanoate, C9H14O4
- The crystal structure of (E)-6-bromo-3,5-dimethyl-2-(1-phenylprop-1-en-2-yl)-3Himidazo[4,5b]pyridine, C17H16BrN3
- Crystal structure of (3S,3′S,4R,4′S)-3′-(furan-3-yl)-3-hydroxy-4′-methyl-3,5,6′,7′-tetrahydro-1H,3′H-4,5′-spirobi[isobenzofuran]-1,1′(4′H)-dione-methanol (1/1), C21H22O7
- Cocrystal structure of progesterone-isophthalic acid, C25H33O4
- The crystal structure of 3-(6-fluoro-1H-indol-3-yl)-1-methylquinoxalin-2(1H)-one, C17H12FN3O
- Crystal structure of S-(4-carboxybutyl)- l -cysteine
- The cocrystal of 2,2′-(hydrazine-1,1-diyl)bis(1H-imidazole-4,5-dicarbonitrile)– methanol (2/3)
- Crystal structure of (1′R,2′S,4′R,6′S)-4,6-dihydroxy-1′,8′,8′-trimethyl-3-(3-methylbutanoyl)-4′,8′,6′,1′,7,2′-hexahydro-1H-4′,6′-methanoxanthene-8-carbaldehyde, C23H30O5
- Crystal structure of (3,6-di(2-pyridyl)-4-methylphenyl pyridazine-k 2 N,N′)-bis(1-phenyl-pyrazole-κ 2 C,N) iridium(III) hexafluorophosphate, C39H29F6IrN8P
- Crystal structure of 1,5-bis[(E)-1-(2-hydroxyphenyl)ethylidene]thiocarbonohydrazide dimethyl sulfoxide monosolvate, C17H18N4O2S·C2H6OS
- Crystal structure of (S)-4-(2-(4-(2-acetyl-5-chlorophenyl)-3-methoxy-6-oxopyridazin-1(6H)-yl)-3-phenylpropanamido)benzoic acid monohydrate, C29H26ClN3O7
- The crystal structure of 1,3-bis(2,4-dinitro-1H-imidazol-1-yl)propane
- Crystal structure of 4-chlorobenzyl (S)-2-(6-methoxynaphthalen-2-yl)propanoate, C21H19ClO3
- Crystal structure of 1-(5-(benzo[d][1,3]dioxol-5-yl)-4-benzyl-1-(4-bromophenyl)-4,5-dihydro-1H-1,2,4-triazol-3-yl)ethan-1-one, C24H20BrN3O3
- The crystal structure of (Z)-3′-(2-(1-(3,4-dimethyl-phenyl)-3-methyl-5-oxo-1,5-dihydro-4H-pyrazol-4-ylidene)hydrazinyl)-2′-hydroxy-[1,1′-biphenyl]-3-carboxylicacid ─ methanol (1/1), C26H26N4O5
- Crystal structure of (S)-1-phenylpropan-1-aminium (S)-(1-phenylpropyl)carbamate C19H26N2O2
- Synthesis and crystal structure of methyl 2-((5-bromo-4-(4-cyclopropylnaphthalen-1-yl)-4H-1,2,4-triazol-3-yl)thio)acetate, C18H16BrN3O2S
- The crystal structure of trichlorobis(pyridine-2,6-dithio-κS-carbomethylamido)antimony(III), [SbCl3(C9H11N3S2)2]
- Crystal structure of 1,8-dihydroxy-3-{[(triphenylstannyl)oxy]carbonyl} anthracene-9,10-dione, C33H22O6Sn
- The crystal structure of (E)-4-(2-(pyridin-4-ylmethylene)hydrazine-1-carbonyl)pyridin-1-ium-2-olate dihydrate, C12H14N4O4
- The crystal structure of 6-amino-pyridinium-2-carboxylate, C6H6N2O2
- The crystal structure of catena-poly[aqua-nitrato-κ3O,O:O′′-(1,10-phenanthroline-κ2N,N′)sodium(I)], C24H18N6O7Na2
- Retractions
- Retraction of: Crystal structure of bis[diaquaisonicotinatosamarium(III)]-µ-isonicotinato-[diisonicotinatocopper(II)], CuSm2(C6H4NO2)8(H2O)4
- Retraction of: Crystal structure of aqua(2,2′-bipyridine-k 2 N:N′)(nitrato)-(4-aminobenzoato)cadmium(II) nitrate, [Cd(H2O)(NO3)(C10H8N2)(C7H7NO2)][NO3]

