Abstract
C21H22O7, orthorhombic, P212121 (no. 19), a = 8.3764(2) Å, b = 11.4813(2) Å, c = 19.2394(4) Å, V = 1850.29(7) Å3, Z = 4, R gt (F) = 0.0303, wRref(F2) = 0.0767, T = 100(2) K.
Table 1 contains crystallographic data and Table 2 contains the list of the atoms including atomic coordinates and displacement parameters.
Data collection and handling.
| Crystal: | Colourless prism |
| Size: | 0.90 × 0.46 × 0.25 mm |
| Wavelength: | Cu Kα radiation (1.54178 Å) |
| μ: | 0.87 mm−1 |
| Diffractometer, scan mode: | Bruker APEX-II, φ and ω |
| θmax, completeness: | 70.2°, 98 % |
| N(hkl)measured, N(hkl)unique, Rint: | 11,045, 3,330, 0.026 |
| Criterion for Iobs, N(hkl)gt: | Iobs > 2σ(Iobs), 3,329 |
| N(param)refined: | 261 |
| Programs: | SHELX, 1 , 2 Bruker 3 |
Fractional atomic coordinates and isotropic or equivalent isotropic displacement parameters (Å2).
| Atom | x | y | z | Uiso*/Ueq |
|---|---|---|---|---|
| O1 | 0.90516 (18) | 0.24091 (15) | 0.10268 (8) | 0.0359 (4) |
| O2 | 0.71203 (18) | 0.38378 (13) | 0.29307 (7) | 0.0281 (3) |
| H2 | 0.765 (4) | 0.428 (3) | 0.3191 (16) | 0.042* |
| O3 | 0.86513 (16) | 0.36087 (13) | 0.19294 (7) | 0.0281 (3) |
| O4 | 0.02314 (16) | 0.71704 (12) | 0.14335 (7) | 0.0235 (3) |
| O5 | −0.09294 (16) | 0.66746 (12) | 0.24419 (7) | 0.0268 (3) |
| O6 | 0.1437 (2) | 0.60737 (14) | −0.07805 (8) | 0.0344 (4) |
| O7 | 0.15015 (19) | 0.04105 (14) | 0.12228 (8) | 0.0313 (3) |
| H19 | 0.108 (4) | 0.094 (3) | 0.1432 (16) | 0.047* |
| C1 | 0.8147 (2) | 0.29838 (18) | 0.13758 (11) | 0.0259 (4) |
| C2 | 0.6408 (2) | 0.31547 (16) | 0.12981 (10) | 0.0205 (4) |
| C3 | 0.5908 (2) | 0.38679 (15) | 0.17990 (9) | 0.0178 (4) |
| C4 | 0.4208 (2) | 0.43003 (15) | 0.18376 (9) | 0.0165 (4) |
| C5 | 0.4040 (2) | 0.54104 (16) | 0.13672 (9) | 0.0177 (4) |
| H10 | 0.4008 | 0.5146 | 0.0872 | 0.021* |
| C6 | 0.2478 (2) | 0.59826 (15) | 0.15261 (9) | 0.0178 (4) |
| C7 | 0.1691 (2) | 0.68745 (16) | 0.10660 (10) | 0.0198 (4) |
| H9 | 0.2388 | 0.7579 | 0.1027 | 0.024* |
| C8 | 0.1301 (2) | 0.64241 (16) | 0.03568 (10) | 0.0217 (4) |
| C9 | 0.0129 (3) | 0.55650 (19) | 0.01791 (12) | 0.0327 (5) |
| H7 | −0.0591 | 0.5188 | 0.0487 | 0.039* |
| C10 | 0.0253 (3) | 0.53985 (19) | −0.05071 (12) | 0.0357 (5) |
| H1 | −0.0396 | 0.4880 | −0.0769 | 0.043* |
| C11 | 0.7290 (2) | 0.42068 (17) | 0.22551 (10) | 0.0217 (4) |
| H18 | 0.7453 | 0.5069 | 0.2241 | 0.026* |
| C12 | 0.0176 (2) | 0.65637 (16) | 0.20429 (10) | 0.0216 (4) |
| C13 | 0.1590 (2) | 0.58121 (16) | 0.20898 (9) | 0.0198 (4) |
| C14 | 0.1972 (2) | 0.49762 (18) | 0.26598 (9) | 0.0224 (4) |
| H5 | 0.1260 | 0.4290 | 0.2630 | 0.027* |
| H6 | 0.1802 | 0.5354 | 0.3116 | 0.027* |
| C15 | 0.3720 (2) | 0.45901 (17) | 0.25911 (9) | 0.0203 (4) |
| H4 | 0.3894 | 0.3894 | 0.2885 | 0.024* |
| H3 | 0.4419 | 0.5219 | 0.2768 | 0.024* |
| C16 | 0.2050 (3) | 0.66979 (18) | −0.02388 (10) | 0.0281 (4) |
| H8 | 0.2891 | 0.7249 | −0.0278 | 0.034* |
| C17 | 0.5390 (2) | 0.63057 (17) | 0.14357 (12) | 0.0286 (4) |
| H11 | 0.5131 | 0.6998 | 0.1160 | 0.043* |
| H12 | 0.6389 | 0.5964 | 0.1267 | 0.043* |
| H13 | 0.5512 | 0.6527 | 0.1925 | 0.043* |
| C18 | 0.3136 (2) | 0.33257 (16) | 0.15385 (10) | 0.0206 (4) |
| H16 | 0.2994 | 0.2717 | 0.1898 | 0.025* |
| H17 | 0.2070 | 0.3656 | 0.1435 | 0.025* |
| C19 | 0.3784 (2) | 0.27617 (17) | 0.08857 (10) | 0.0245 (4) |
| H14 | 0.3058 | 0.2472 | 0.0548 | 0.029* |
| C20 | 0.5347 (2) | 0.26624 (17) | 0.07738 (10) | 0.0240 (4) |
| H15 | 0.5752 | 0.2288 | 0.0371 | 0.029* |
| C21 | 0.0658 (3) | 0.0035 (2) | 0.06241 (13) | 0.0382 (5) |
| H22 | −0.0312 | −0.0379 | 0.0767 | 0.057* |
| H21 | 0.1336 | −0.0488 | 0.0351 | 0.057* |
| H20 | 0.0366 | 0.0713 | 0.0342 | 0.057* |
1 Source of materials
Aerial parts of Salvia hispanica L. (Lamiaceae family) were collected and identified by Min Fan, in July 2016. The air-dried powder (15 kg) was extracted with acetone to yield 0.6 kg of a crude extract, which was subjected to silica gel CC and eluted with PE-acetone (100:0 to 0:100, v/v) to give five fractions (A–E). Subsequently, fraction C (105 g) was separated by MCI CC, silica gel column and semipreparative HPLC (Agilent 1260) to afford the title compound (10 mg), which crystallized from methanol as colorless crystals.
2 Experimental details
Hydrogen atoms were placed in their geometrically idealized positions and constrained to ride on their parent atoms. Their Uiso values were set to 1.2Ueq of the parent atoms.
3 Comment
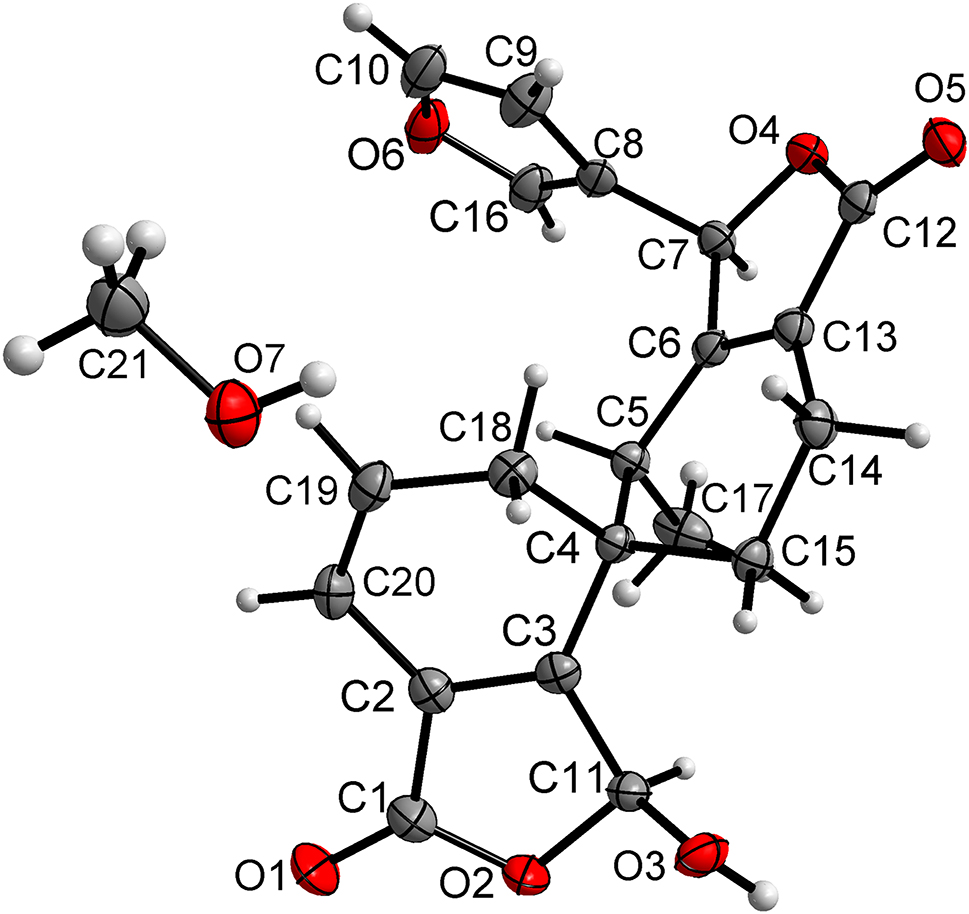
S. hispanica L. is an annual herbaceous plant belonging to the family Lamiaceae. 4 Its seed has been identified as a significant source of oil, protein, dietary fiber, minerals, and polyphenolic compounds. 5 This plant is known for its beneficial effects on health issues such as dyslipidemia, inflammation, cardiovascular diseases, and insulin resistance, all without inducing adverse effects. 6 While previous phytochemical research on S. hispanica L. has predominantly focused on its seeds, 7 , 8 there has been limited exploration of the aerial parts of this species. Our research team has undertaken a phytochemical analysis of S. hispanica L. grown in Yunnan, resulting in the isolation of several new compounds with unique structures and intriguing bioactivities. 9 , 10 , 11 This discovery has motivated us to delve deeper into studying this species further. Subsequently, we successfully isolated a new diterpenoid from this plant. The compound, illustrated in the figure, comprises two six-membered rings, A (atoms C2, C3, C4, C18, C19, C20) and B (atoms C4, C5, C6, C13, C14, C15), arranged in chair conformations.
-
Author contributions: All the authors have accepted responsibility for the entire content of this submitted manuscript and approved submission.
-
Research funding: ‘Three Districts’ Science and Technology Talent Support Plan of Yunnan Province (KY2313135640, KY2313135540).
-
Conflict of interest statement: The authors declare no conflicts of interest regarding this article.
References
1. Sheldrick, G. M. Crystal Structure Refinement with SHELXL. Acta Crystallogr. 2015, C71, 3–8. https://doi.org/10.1107/s2053229614024218.Search in Google Scholar
2. Sheldrick, G. M. SHELXT – Integrated Space-Group and Crystal-Structure Determination. Acta Crystallogr. 2015, A71, 3–8; https://doi.org/10.1107/s2053273314026370.Search in Google Scholar
3. BRUKER. APEX2, SAINT and SADABS; Bruker AXS Inc.: Madison, Wisconsin, USA, 2012.Search in Google Scholar
4. Capitani, M. I.; Ixtaina, V. Y.; Nolasco, S. M.; Tomas, M. C. Microstructure, Chemical Composition and Mucilage Exudation of Chia (Salvia hispanica L.) Nutlets from Argentina. J. Sci. Food Agr. 2013, 93, 3856–3862; https://doi.org/10.1002/jsfa.6327.Search in Google Scholar PubMed
5. Reyes-Caudillo, E.; Tecante, A.; Valdivia-Lopez, M. A. Dietary Fibre Content and Antioxidant Activity of Phenolic Compounds Present in Mexican Chia (Salvia hispanica L.) Seeds. Food Chem. 2008, 107, 656–663; https://doi.org/10.1016/j.foodchem.2007.08.062.Search in Google Scholar
6. Vuksan, V.; Whitham, D.; Sievenpiper, J. L.; Jenkins, A. L.; Rogovik, A. L.; Bazinet, R. P.; Vidgen, E.; Hanna, A. Supplementation of Conventional Therapy with the Novel Grain Salba (Salvia hispanica L.) Improves Major and Emerging Cardiovascular Risk Factors in Type 2 Diabetes. Diabetes Care 2007, 30, 2804; https://doi.org/10.2337/dc07-1144.Search in Google Scholar PubMed
7. Zettel, V.; Kraemer, A.; Hecker, F.; Hitzmann, B. Influence of Gel from Ground Chia (Salvia hispanica L.) for Wheat Bread Production. Eur. Food Res. Technol. 2015, 240, 655–662; https://doi.org/10.1007/s00217-014-2368-8.Search in Google Scholar
8. Marineli, R. d. S.; Moura, C. S.; Moraes, E. A.; Lenquiste, S. A.; Lollo, P. C. B.; Morato, P. N.; Amaya-Farfan, J.; Marostica, M. R.Jr. Chia (Salvia hispanica L.) Enhances HSP, PGC-1α Expressions and Improves Glucose Tolerance in Diet-Induced Obese Rats. Nutrition 2015, 31, 740–748; https://doi.org/10.1016/j.nut.2014.11.009.Search in Google Scholar PubMed
9. Fan, M.; Luo, D.; Peng, L. Y.; Wu, X. D.; Ji, X.; Zhao, Q. S. Rearranged Neoclerodane Diterpenoids from the Aerial Parts of Salvia hispanica L. Fitoterapia 2020, 146, 104672; https://doi.org/10.1016/j.fitote.2020.104672.Search in Google Scholar PubMed
10. Fan, M.; Zhu, Y.; Zhang, Z. J.; Du, R. N.; Zhu, Q. F.; Wu, X. D.; Zhao, Q. S. Salvihispin A and Its Glycoside, Two Neo-Clerodane Diterpenoids with Neurotrophic Activities from Salvia hispanica L. Tetrahedron Lett. 2018, 59, 143–146; https://doi.org/10.1016/j.tetlet.2017.12.010.Search in Google Scholar
11. Fan, M.; Luo, D.; Peng, L. Y.; Li, X. N.; Wu, X. D.; Ji, X.; Zhao, Q. S. Neo-clerodane Diterpenoids from Aerial Parts of Salvia hispanica L. and Their Cardioprotective Effects. Phytochemistry 2019, 166, 112065; https://doi.org/10.1016/j.phytochem.2019.112065.Search in Google Scholar PubMed
© 2024 the author(s), published by De Gruyter, Berlin/Boston
This work is licensed under the Creative Commons Attribution 4.0 International License.
Articles in the same Issue
- Frontmatter
- Editorial
- Editorial 2024 – New developments and changes of Zeitschrift für Kristallographie – New Crystal Structures
- New Crystal Structures
- Hydrogen bonding and π⋅⋅⋅halogen interactions in the crystal structure of bis(theophyllinium) hexachloridoplatinate(IV) monohydrate
- The crystal structure of 6-amino-2-carboxypyridin-1-ium perchlorate, C6H7ClN2O6
- Crystal structure of poly[(μ4-(3-amino-1H-1,2,4-triazol-1-yl)benzene-1,3-dicarboxylato-κ 4 N:O:O':O')(1-methylpyrroldin-2-one-κ1O)dicopper(II)] – 1-methylpyrroldin-2-one (1/3), C40H48Cu2N12O12
- The crystal structure of 18-crown-6-k6O6(2,4,5-trinitroimidazol-1-ido-k1O)potassium(I)
- Crystal structure of poly[tetraaqua-bis(μ2-5-bromoisophthalato-κ3O,O′:O″)-(μ2-1,5-bis(imidazol-2-methyl)pentane-κ2N:N′)dicadmium(II)] dihydrate
- Crystal structure of (5R,6S,E)-5-acetoxy-2-methyl-6-((2aR,3R,5aS,5bS,11aR,12aS)-2a,5a,8,8-tetramethyl-9-oxotetradecahydro-1H,12H-cyclopenta[a]cyclopropa[e]phenanthren-3-yl)hept-2-enoic acid, C32H48O5
- The crystal structure of poly[diaqua-bis(μ2 -thiocyanato-κ2N:O)cobalt(II) monohydrate
- The crystal structure of 1,3,5-tri(1H-imidazol-1-yl)benzene–2,3,5,6-tetrachlorobenzene-1,4-dicarboxylic acid (1/1)
- Crystal structure of dichlorido-bis(1-[(2-ethyl-benzimidazole-1-yl)methyl]-1H–benzotriazole) cadmium(II), C32H32CdN10OCl2
- The crystal structure of N′-(tert-butyl)-N′-(3,5-dimethylbenzoyl)-3-methoxy-N,2-dimethylbenzohydrazide, C23H30N2O3
- Crystal stucture of 3-benzamido-N-(2-bromo-4-(perfluoropropan-2-yl)-6-(trifluoromethyl)phenyl)-2-fluorobenzamide
- Crystal structure of bis(μ-benzeneselenolato)-(tetracarbonyl)-{μ-[N-(diphenylphosphanyl)-N-(3-ethynylphenyl)-P,P-diphenylphosphinous amide]} diiron, C48H35Fe2NO4P2Se2
- The crystal structure of 2′-(p-tolyl)-4′H-spiro[isochromane-1,1′-naphthalene]-3,4′-dione, C25H18O3
- The crystal structure of poly[hexaqua-tetrakis(μ4-pyridine-2,4-dicarboxylate-κ5N: O: O′: O″: O‴)-bi(μ2-pyridine-2,4-dicarboxylate-κ3N: O: O′)-digadolinium(III)tricopper (II)], [Gd2Cu3(C7H3NO4)6(H2O)6] n
- Crystal structure of poly[bis(4-(4-(pyridin-4-yl)phenyl)pyridin-1-ium-κ1N)-(μ4-benzene-1,2,4,5-tetracarboxylato-κ5O:O′: O″:O‴:O⁗)-(μ2-2,5-dicarboxyterephthalato-κ2O:O′)dizinc(II)], C52H32N4O16Zn2
- The crystal structure of 4-(3-carboxy-1-ethyl-6-fluoro-4-oxo-1,4-dihydroquinolin-7-yl)piperazin-1-ium 2-carboxy-6-nitrobenzoate monohydrate, C24H25FN4O10
- Crystal structure of dichlorido-(1-((3,5-dimethyl-2,3-dihydro-1H-1,2,3-triazol-1-yl)methyl)-1H-benzo[d][1,2,3]triazole-k1N)zinc(II), C22H24ZnN12Cl2
- The crystal structure of (3-chlorothiophene-2-carboxylato-κ2O, O′)-(2,2′-dipyridyl-κ2N,N′)lead(II), C20H12Cl2N2O4S2Pb
- Synthesis and crystal structure of (Z)-4-((1-(3-fluorophenyl)-1H-1,2,3-triazol-4-yl)methylene)-5-methyl-2-phenyl-2,4-dihydro-3H-pyrazol-3-one, C19H14FN5O
- The crystal structure of the coordination compound catena-poly[(18-crown-6-ether-κ6O6)(4,5-dinitroimidazolato-κ1O)potassium(I)]
- Crystal structure of 7-(diethylamino)-3-(trifluoroacetyl)-2H-chromen-2-one, C15H14F3NO3
- Crystal structure of dichlorido-1-[(2-ethylimidazole-1-yl)methyl]-1H–benzotriazole κ1N zinc(II), C24H26ZnN10Cl2
- Crystal and molecular structure of 5-bromopyridine-2,3-diamine
- Crystal structure of catena-poly[bis(μ2-1-(3-carboxyphenyl)-5-methyl-4-oxo-1,4-dihydropyridazine-3-carboxylato-k3-O,O′:O″)hexaqua-dicobalt tetrahydrate], C26H36N4O20Co2
- Crystal structure of thiocyanate-κ1N-bis(μ1-2,6-diformyl-4-methylphenol oxime-κ2N,O)-manganese(III) acetonitrile solvate, C21H21MnN6O6S
- The crystal structure of pyrrolidin-1-yl pivalate, C9H13NO4
- The crystal structure of 2,2′-(2,2-diphenylethene-1,1-diyl)bis(1,4-dimethoxybenzene), C30H28O4
- Crystal structure of bis(benzyltrimethylammonium) tetrathiotungstate(VI), {(C6H5CH2)(CH3)3N}2[WS4]
- The crystal structure of ethyl (Z)-2-(ethoxymethylene)-3-oxobutanoate, C9H14O4
- The crystal structure of (E)-6-bromo-3,5-dimethyl-2-(1-phenylprop-1-en-2-yl)-3Himidazo[4,5b]pyridine, C17H16BrN3
- Crystal structure of (3S,3′S,4R,4′S)-3′-(furan-3-yl)-3-hydroxy-4′-methyl-3,5,6′,7′-tetrahydro-1H,3′H-4,5′-spirobi[isobenzofuran]-1,1′(4′H)-dione-methanol (1/1), C21H22O7
- Cocrystal structure of progesterone-isophthalic acid, C25H33O4
- The crystal structure of 3-(6-fluoro-1H-indol-3-yl)-1-methylquinoxalin-2(1H)-one, C17H12FN3O
- Crystal structure of S-(4-carboxybutyl)- l -cysteine
- The cocrystal of 2,2′-(hydrazine-1,1-diyl)bis(1H-imidazole-4,5-dicarbonitrile)– methanol (2/3)
- Crystal structure of (1′R,2′S,4′R,6′S)-4,6-dihydroxy-1′,8′,8′-trimethyl-3-(3-methylbutanoyl)-4′,8′,6′,1′,7,2′-hexahydro-1H-4′,6′-methanoxanthene-8-carbaldehyde, C23H30O5
- Crystal structure of (3,6-di(2-pyridyl)-4-methylphenyl pyridazine-k 2 N,N′)-bis(1-phenyl-pyrazole-κ 2 C,N) iridium(III) hexafluorophosphate, C39H29F6IrN8P
- Crystal structure of 1,5-bis[(E)-1-(2-hydroxyphenyl)ethylidene]thiocarbonohydrazide dimethyl sulfoxide monosolvate, C17H18N4O2S·C2H6OS
- Crystal structure of (S)-4-(2-(4-(2-acetyl-5-chlorophenyl)-3-methoxy-6-oxopyridazin-1(6H)-yl)-3-phenylpropanamido)benzoic acid monohydrate, C29H26ClN3O7
- The crystal structure of 1,3-bis(2,4-dinitro-1H-imidazol-1-yl)propane
- Crystal structure of 4-chlorobenzyl (S)-2-(6-methoxynaphthalen-2-yl)propanoate, C21H19ClO3
- Crystal structure of 1-(5-(benzo[d][1,3]dioxol-5-yl)-4-benzyl-1-(4-bromophenyl)-4,5-dihydro-1H-1,2,4-triazol-3-yl)ethan-1-one, C24H20BrN3O3
- The crystal structure of (Z)-3′-(2-(1-(3,4-dimethyl-phenyl)-3-methyl-5-oxo-1,5-dihydro-4H-pyrazol-4-ylidene)hydrazinyl)-2′-hydroxy-[1,1′-biphenyl]-3-carboxylicacid ─ methanol (1/1), C26H26N4O5
- Crystal structure of (S)-1-phenylpropan-1-aminium (S)-(1-phenylpropyl)carbamate C19H26N2O2
- Synthesis and crystal structure of methyl 2-((5-bromo-4-(4-cyclopropylnaphthalen-1-yl)-4H-1,2,4-triazol-3-yl)thio)acetate, C18H16BrN3O2S
- The crystal structure of trichlorobis(pyridine-2,6-dithio-κS-carbomethylamido)antimony(III), [SbCl3(C9H11N3S2)2]
- Crystal structure of 1,8-dihydroxy-3-{[(triphenylstannyl)oxy]carbonyl} anthracene-9,10-dione, C33H22O6Sn
- The crystal structure of (E)-4-(2-(pyridin-4-ylmethylene)hydrazine-1-carbonyl)pyridin-1-ium-2-olate dihydrate, C12H14N4O4
- The crystal structure of 6-amino-pyridinium-2-carboxylate, C6H6N2O2
- The crystal structure of catena-poly[aqua-nitrato-κ3O,O:O′′-(1,10-phenanthroline-κ2N,N′)sodium(I)], C24H18N6O7Na2
- Retractions
- Retraction of: Crystal structure of bis[diaquaisonicotinatosamarium(III)]-µ-isonicotinato-[diisonicotinatocopper(II)], CuSm2(C6H4NO2)8(H2O)4
- Retraction of: Crystal structure of aqua(2,2′-bipyridine-k 2 N:N′)(nitrato)-(4-aminobenzoato)cadmium(II) nitrate, [Cd(H2O)(NO3)(C10H8N2)(C7H7NO2)][NO3]
Articles in the same Issue
- Frontmatter
- Editorial
- Editorial 2024 – New developments and changes of Zeitschrift für Kristallographie – New Crystal Structures
- New Crystal Structures
- Hydrogen bonding and π⋅⋅⋅halogen interactions in the crystal structure of bis(theophyllinium) hexachloridoplatinate(IV) monohydrate
- The crystal structure of 6-amino-2-carboxypyridin-1-ium perchlorate, C6H7ClN2O6
- Crystal structure of poly[(μ4-(3-amino-1H-1,2,4-triazol-1-yl)benzene-1,3-dicarboxylato-κ 4 N:O:O':O')(1-methylpyrroldin-2-one-κ1O)dicopper(II)] – 1-methylpyrroldin-2-one (1/3), C40H48Cu2N12O12
- The crystal structure of 18-crown-6-k6O6(2,4,5-trinitroimidazol-1-ido-k1O)potassium(I)
- Crystal structure of poly[tetraaqua-bis(μ2-5-bromoisophthalato-κ3O,O′:O″)-(μ2-1,5-bis(imidazol-2-methyl)pentane-κ2N:N′)dicadmium(II)] dihydrate
- Crystal structure of (5R,6S,E)-5-acetoxy-2-methyl-6-((2aR,3R,5aS,5bS,11aR,12aS)-2a,5a,8,8-tetramethyl-9-oxotetradecahydro-1H,12H-cyclopenta[a]cyclopropa[e]phenanthren-3-yl)hept-2-enoic acid, C32H48O5
- The crystal structure of poly[diaqua-bis(μ2 -thiocyanato-κ2N:O)cobalt(II) monohydrate
- The crystal structure of 1,3,5-tri(1H-imidazol-1-yl)benzene–2,3,5,6-tetrachlorobenzene-1,4-dicarboxylic acid (1/1)
- Crystal structure of dichlorido-bis(1-[(2-ethyl-benzimidazole-1-yl)methyl]-1H–benzotriazole) cadmium(II), C32H32CdN10OCl2
- The crystal structure of N′-(tert-butyl)-N′-(3,5-dimethylbenzoyl)-3-methoxy-N,2-dimethylbenzohydrazide, C23H30N2O3
- Crystal stucture of 3-benzamido-N-(2-bromo-4-(perfluoropropan-2-yl)-6-(trifluoromethyl)phenyl)-2-fluorobenzamide
- Crystal structure of bis(μ-benzeneselenolato)-(tetracarbonyl)-{μ-[N-(diphenylphosphanyl)-N-(3-ethynylphenyl)-P,P-diphenylphosphinous amide]} diiron, C48H35Fe2NO4P2Se2
- The crystal structure of 2′-(p-tolyl)-4′H-spiro[isochromane-1,1′-naphthalene]-3,4′-dione, C25H18O3
- The crystal structure of poly[hexaqua-tetrakis(μ4-pyridine-2,4-dicarboxylate-κ5N: O: O′: O″: O‴)-bi(μ2-pyridine-2,4-dicarboxylate-κ3N: O: O′)-digadolinium(III)tricopper (II)], [Gd2Cu3(C7H3NO4)6(H2O)6] n
- Crystal structure of poly[bis(4-(4-(pyridin-4-yl)phenyl)pyridin-1-ium-κ1N)-(μ4-benzene-1,2,4,5-tetracarboxylato-κ5O:O′: O″:O‴:O⁗)-(μ2-2,5-dicarboxyterephthalato-κ2O:O′)dizinc(II)], C52H32N4O16Zn2
- The crystal structure of 4-(3-carboxy-1-ethyl-6-fluoro-4-oxo-1,4-dihydroquinolin-7-yl)piperazin-1-ium 2-carboxy-6-nitrobenzoate monohydrate, C24H25FN4O10
- Crystal structure of dichlorido-(1-((3,5-dimethyl-2,3-dihydro-1H-1,2,3-triazol-1-yl)methyl)-1H-benzo[d][1,2,3]triazole-k1N)zinc(II), C22H24ZnN12Cl2
- The crystal structure of (3-chlorothiophene-2-carboxylato-κ2O, O′)-(2,2′-dipyridyl-κ2N,N′)lead(II), C20H12Cl2N2O4S2Pb
- Synthesis and crystal structure of (Z)-4-((1-(3-fluorophenyl)-1H-1,2,3-triazol-4-yl)methylene)-5-methyl-2-phenyl-2,4-dihydro-3H-pyrazol-3-one, C19H14FN5O
- The crystal structure of the coordination compound catena-poly[(18-crown-6-ether-κ6O6)(4,5-dinitroimidazolato-κ1O)potassium(I)]
- Crystal structure of 7-(diethylamino)-3-(trifluoroacetyl)-2H-chromen-2-one, C15H14F3NO3
- Crystal structure of dichlorido-1-[(2-ethylimidazole-1-yl)methyl]-1H–benzotriazole κ1N zinc(II), C24H26ZnN10Cl2
- Crystal and molecular structure of 5-bromopyridine-2,3-diamine
- Crystal structure of catena-poly[bis(μ2-1-(3-carboxyphenyl)-5-methyl-4-oxo-1,4-dihydropyridazine-3-carboxylato-k3-O,O′:O″)hexaqua-dicobalt tetrahydrate], C26H36N4O20Co2
- Crystal structure of thiocyanate-κ1N-bis(μ1-2,6-diformyl-4-methylphenol oxime-κ2N,O)-manganese(III) acetonitrile solvate, C21H21MnN6O6S
- The crystal structure of pyrrolidin-1-yl pivalate, C9H13NO4
- The crystal structure of 2,2′-(2,2-diphenylethene-1,1-diyl)bis(1,4-dimethoxybenzene), C30H28O4
- Crystal structure of bis(benzyltrimethylammonium) tetrathiotungstate(VI), {(C6H5CH2)(CH3)3N}2[WS4]
- The crystal structure of ethyl (Z)-2-(ethoxymethylene)-3-oxobutanoate, C9H14O4
- The crystal structure of (E)-6-bromo-3,5-dimethyl-2-(1-phenylprop-1-en-2-yl)-3Himidazo[4,5b]pyridine, C17H16BrN3
- Crystal structure of (3S,3′S,4R,4′S)-3′-(furan-3-yl)-3-hydroxy-4′-methyl-3,5,6′,7′-tetrahydro-1H,3′H-4,5′-spirobi[isobenzofuran]-1,1′(4′H)-dione-methanol (1/1), C21H22O7
- Cocrystal structure of progesterone-isophthalic acid, C25H33O4
- The crystal structure of 3-(6-fluoro-1H-indol-3-yl)-1-methylquinoxalin-2(1H)-one, C17H12FN3O
- Crystal structure of S-(4-carboxybutyl)- l -cysteine
- The cocrystal of 2,2′-(hydrazine-1,1-diyl)bis(1H-imidazole-4,5-dicarbonitrile)– methanol (2/3)
- Crystal structure of (1′R,2′S,4′R,6′S)-4,6-dihydroxy-1′,8′,8′-trimethyl-3-(3-methylbutanoyl)-4′,8′,6′,1′,7,2′-hexahydro-1H-4′,6′-methanoxanthene-8-carbaldehyde, C23H30O5
- Crystal structure of (3,6-di(2-pyridyl)-4-methylphenyl pyridazine-k 2 N,N′)-bis(1-phenyl-pyrazole-κ 2 C,N) iridium(III) hexafluorophosphate, C39H29F6IrN8P
- Crystal structure of 1,5-bis[(E)-1-(2-hydroxyphenyl)ethylidene]thiocarbonohydrazide dimethyl sulfoxide monosolvate, C17H18N4O2S·C2H6OS
- Crystal structure of (S)-4-(2-(4-(2-acetyl-5-chlorophenyl)-3-methoxy-6-oxopyridazin-1(6H)-yl)-3-phenylpropanamido)benzoic acid monohydrate, C29H26ClN3O7
- The crystal structure of 1,3-bis(2,4-dinitro-1H-imidazol-1-yl)propane
- Crystal structure of 4-chlorobenzyl (S)-2-(6-methoxynaphthalen-2-yl)propanoate, C21H19ClO3
- Crystal structure of 1-(5-(benzo[d][1,3]dioxol-5-yl)-4-benzyl-1-(4-bromophenyl)-4,5-dihydro-1H-1,2,4-triazol-3-yl)ethan-1-one, C24H20BrN3O3
- The crystal structure of (Z)-3′-(2-(1-(3,4-dimethyl-phenyl)-3-methyl-5-oxo-1,5-dihydro-4H-pyrazol-4-ylidene)hydrazinyl)-2′-hydroxy-[1,1′-biphenyl]-3-carboxylicacid ─ methanol (1/1), C26H26N4O5
- Crystal structure of (S)-1-phenylpropan-1-aminium (S)-(1-phenylpropyl)carbamate C19H26N2O2
- Synthesis and crystal structure of methyl 2-((5-bromo-4-(4-cyclopropylnaphthalen-1-yl)-4H-1,2,4-triazol-3-yl)thio)acetate, C18H16BrN3O2S
- The crystal structure of trichlorobis(pyridine-2,6-dithio-κS-carbomethylamido)antimony(III), [SbCl3(C9H11N3S2)2]
- Crystal structure of 1,8-dihydroxy-3-{[(triphenylstannyl)oxy]carbonyl} anthracene-9,10-dione, C33H22O6Sn
- The crystal structure of (E)-4-(2-(pyridin-4-ylmethylene)hydrazine-1-carbonyl)pyridin-1-ium-2-olate dihydrate, C12H14N4O4
- The crystal structure of 6-amino-pyridinium-2-carboxylate, C6H6N2O2
- The crystal structure of catena-poly[aqua-nitrato-κ3O,O:O′′-(1,10-phenanthroline-κ2N,N′)sodium(I)], C24H18N6O7Na2
- Retractions
- Retraction of: Crystal structure of bis[diaquaisonicotinatosamarium(III)]-µ-isonicotinato-[diisonicotinatocopper(II)], CuSm2(C6H4NO2)8(H2O)4
- Retraction of: Crystal structure of aqua(2,2′-bipyridine-k 2 N:N′)(nitrato)-(4-aminobenzoato)cadmium(II) nitrate, [Cd(H2O)(NO3)(C10H8N2)(C7H7NO2)][NO3]

