Abstract
C32H48O5, orthorhombic, P212121 (no. 19), a = 7.1564(10) Å, b = 17.542(3) Å, c = 23.134(4) Å, V = 2904.2(8) Å3, Z = 4, Rgt(F) = 0.0555, wRref(F2) = 0.1343, T = 273(2) K.
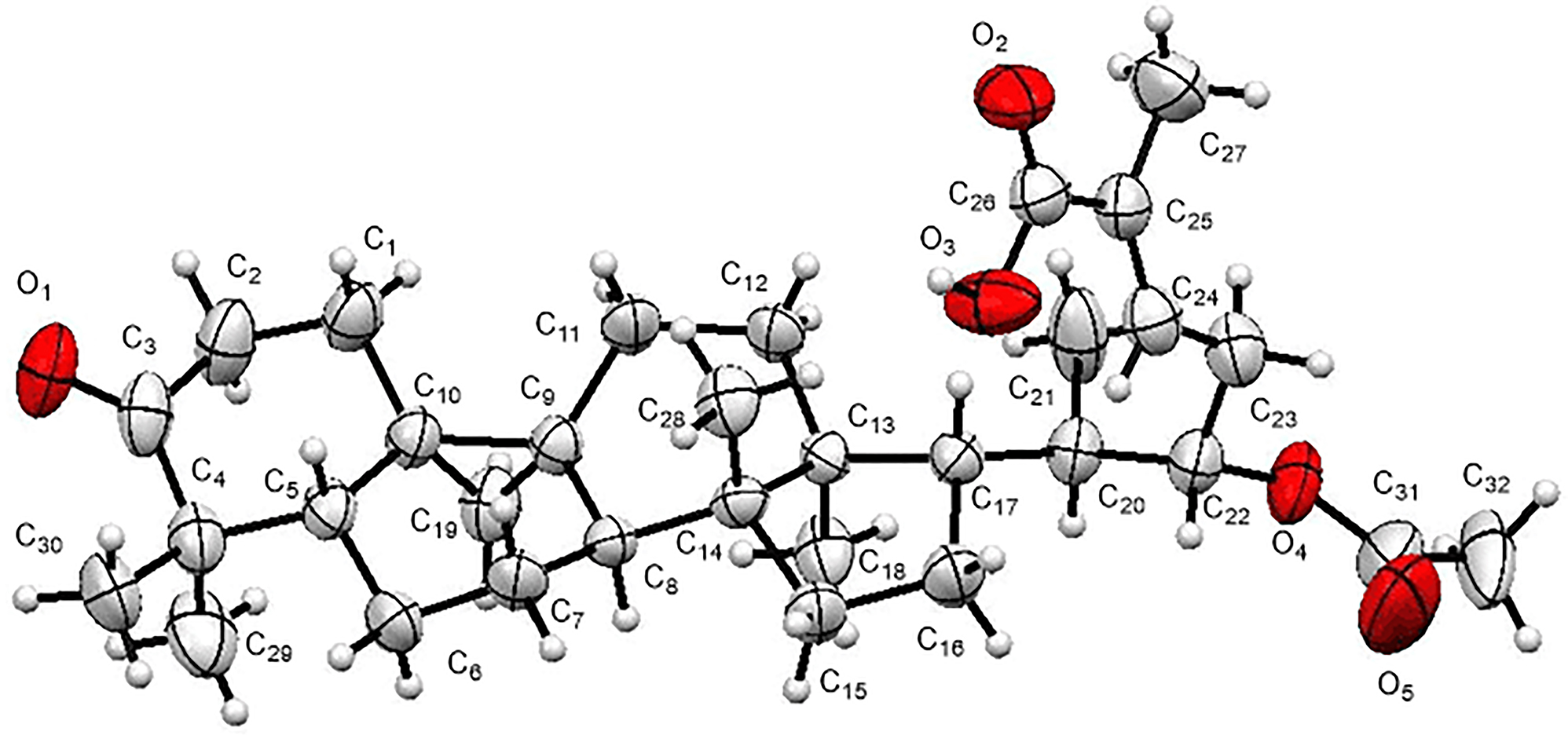
The molecular structure is shown in the figure. Table 1 contains crystallographic data and Table 2 contains the list of the atoms including atomic coordinates and displacement parameters.
Data collection and handling.
| Crystal: | Colourless needle |
| Size: | 0.25 × 0.23 × 0.21 mm |
| Wavelength: | Mo Kα radiation (0.71073 Å) |
| μ: | 0.08 mm−1 |
| Diffractometer, scan mode: | Bruker APEX-II, φ and ω |
| θmax, completeness: | 27.8°, 98 % |
| N(hkl)measured, N(hkl)unique, Rint: | 30,980, 6722, 0.076 |
| Criterion for Iobs, N(hkl)gt: | Iobs > 2 σ (Iobs), 3814 |
| N(param)refined: | 345 |
| Programs: | Olex2, 1 Bruker, 2 SHELX, 3 Diamond 4 |
Fractional atomic coordinates and isotropic or equivalent isotropic displacement parameters (Å2).
| Atom | x | y | z | Uiso*/Ueq |
|---|---|---|---|---|
| C1 | −0.0502 (5) | 0.7529 (2) | 0.26246 (16) | 0.0563 (10) |
| C2 | −0.1096 (5) | 0.8286 (3) | 0.23513 (18) | 0.0657 (12) |
| C3 | 0.0599 (5) | 0.8743 (2) | 0.22024 (17) | 0.0563 (10) |
| C4 | 0.1942 (5) | 0.8927 (2) | 0.26912 (16) | 0.0529 (10) |
| C5 | 0.2450 (4) | 0.8144 (2) | 0.29767 (14) | 0.0416 (8) |
| C6 | 0.3884 (5) | 0.8196 (2) | 0.34592 (16) | 0.0507 (9) |
| C7 | 0.4346 (4) | 0.7402 (2) | 0.36772 (15) | 0.0468 (9) |
| C8 | 0.2680 (4) | 0.70463 (19) | 0.39853 (14) | 0.0361 (8) |
| C9 | 0.0869 (4) | 0.70729 (18) | 0.36224 (14) | 0.0377 (8) |
| C10 | 0.0739 (5) | 0.76757 (19) | 0.31431 (14) | 0.0420 (8) |
| C11 | −0.0149 (5) | 0.6319 (2) | 0.35245 (15) | 0.0492 (9) |
| C12 | −0.0113 (5) | 0.5729 (2) | 0.40214 (14) | 0.0448 (8) |
| C13 | 0.1240 (4) | 0.59194 (17) | 0.45083 (13) | 0.0341 (7) |
| C14 | 0.3066 (4) | 0.62489 (19) | 0.42353 (13) | 0.0363 (7) |
| C15 | 0.4403 (5) | 0.6259 (2) | 0.47544 (15) | 0.0508 (9) |
| C16 | 0.3853 (5) | 0.5566 (2) | 0.51185 (16) | 0.0528 (10) |
| C17 | 0.2032 (4) | 0.52399 (19) | 0.48706 (14) | 0.0379 (8) |
| C18 | 0.0292 (5) | 0.64987 (19) | 0.49128 (15) | 0.0502 (9) |
| C19 | −0.0251 (5) | 0.7796 (2) | 0.37094 (15) | 0.0510 (9) |
| C20 | 0.0776 (5) | 0.4872 (2) | 0.53366 (14) | 0.0447 (8) |
| C21 | −0.1044 (5) | 0.4560 (3) | 0.5092 (2) | 0.0762 (14) |
| C22 | 0.1816 (5) | 0.4261 (2) | 0.56779 (14) | 0.0482 (9) |
| C23 | 0.2186 (6) | 0.3506 (2) | 0.53863 (17) | 0.0570 (10) |
| C24 | 0.3572 (5) | 0.3534 (2) | 0.48977 (16) | 0.0504 (9) |
| C25 | 0.3521 (5) | 0.3134 (2) | 0.44116 (16) | 0.0499 (9) |
| C26 | 0.5011 (6) | 0.3231 (2) | 0.39763 (16) | 0.0526 (10) |
| C27 | 0.2024 (7) | 0.2570 (3) | 0.42635 (19) | 0.0850 (15) |
| C28 | 0.3900 (5) | 0.5710 (2) | 0.37773 (15) | 0.0519 (9) |
| C29 | 0.0981 (7) | 0.9484 (2) | 0.3117 (2) | 0.0799 (14) |
| C30 | 0.3688 (6) | 0.9312 (2) | 0.24515 (19) | 0.0680 (12) |
| C31 | 0.1375 (10) | 0.3918 (3) | 0.6672 (2) | 0.0894 (17) |
| C32 | −0.0049 (11) | 0.3718 (3) | 0.71167 (18) | 0.117 (2) |
| H2 | 0.500854 | 0.844089 | 0.331679 | 0.061* |
| H3 | 0.539642 | 0.743012 | 0.394144 | 0.056* |
| H4 | 0.470682 | 0.708262 | 0.335372 | 0.056* |
| H5 | −0.160267 | 0.775300 | 0.371188 | 0.061* |
| H6 | 0.025064 | 0.817696 | 0.397044 | 0.061* |
| H7 | 0.022020 | 0.523525 | 0.386271 | 0.054* |
| H8 | −0.136115 | 0.568620 | 0.418157 | 0.054* |
| H9 | 0.483265 | 0.518399 | 0.510390 | 0.063* |
| H10 | 0.367231 | 0.571520 | 0.551826 | 0.063* |
| H11 | 0.569064 | 0.621968 | 0.462731 | 0.061* |
| H12 | 0.307216 | 0.568178 | 0.345139 | 0.078* |
| H13 | 0.509162 | 0.590163 | 0.365425 | 0.078* |
| H14 | 0.405446 | 0.521111 | 0.394077 | 0.078* |
| H15 | 0.425615 | 0.672596 | 0.497476 | 0.061* |
| H16 | 0.237735 | 0.483707 | 0.459724 | 0.045* |
| H17 | 0.263750 | 0.314896 | 0.567439 | 0.068* |
| H18A | −0.073784 | 0.625898 | 0.510676 | 0.075* |
| H18 | 0.457192 | 0.386770 | 0.494129 | 0.060* |
| H19 | 0.093027 | 0.283681 | 0.413378 | 0.127* |
| H20 | 0.246124 | 0.223904 | 0.396219 | 0.127* |
| H21A | −0.164980 | 0.424996 | 0.537775 | 0.114* |
| H21 | 0.172262 | 0.227400 | 0.459959 | 0.127* |
| H22 | 0.101241 | 0.330939 | 0.523856 | 0.068* |
| H23 | 0.300103 | 0.447191 | 0.581742 | 0.058* |
| H24 | −0.118004 | 0.399571 | 0.704209 | 0.175* |
| H25 | −0.029972 | 0.318115 | 0.710145 | 0.175* |
| H26 | 0.041531 | 0.384959 | 0.749306 | 0.175* |
| H27 | 0.044128 | 0.527500 | 0.561079 | 0.054* |
| H28 | −0.078223 | 0.425739 | 0.475554 | 0.114* |
| H29A | −0.184767 | 0.497590 | 0.498733 | 0.114* |
| H29 | −0.013742 | 0.925396 | 0.326541 | 0.120* |
| H32 | −0.015600 | 0.692294 | 0.469045 | 0.075* |
| H33 | 0.117985 | 0.667545 | 0.519365 | 0.075* |
| H34 | 0.038207 | 0.607965 | 0.318396 | 0.059* |
| H35A | −0.144521 | 0.643282 | 0.343778 | 0.059* |
| H35 | 0.697 (7) | 0.380 (3) | 0.379 (2) | 0.090 (17)* |
| H36 | 0.244166 | 0.737361 | 0.432058 | 0.043* |
| H37 | 0.338930 | 0.850184 | 0.377339 | 0.061* |
| H39 | 0.435111 | 0.896070 | 0.220833 | 0.102* |
| H40 | 0.447899 | 0.946680 | 0.276543 | 0.102* |
| H41 | 0.333286 | 0.975143 | 0.222978 | 0.102* |
| H42 | 0.181568 | 0.959516 | 0.343044 | 0.120* |
| H43 | 0.067019 | 0.994702 | 0.291814 | 0.120* |
| H44 | −0.160122 | 0.724603 | 0.274435 | 0.068* |
| H45 | 0.016765 | 0.722460 | 0.234239 | 0.068* |
| H46 | −0.187126 | 0.856950 | 0.262019 | 0.079* |
| H47 | −0.182247 | 0.818998 | 0.200495 | 0.079* |
| H58 | 0.306453 | 0.785016 | 0.267052 | 0.050* |
| O1 | 0.0904 (4) | 0.89191 (18) | 0.17016 (12) | 0.0732 (9) |
| O2 | 0.0611 (4) | 0.40969 (15) | 0.61723 (10) | 0.0650 (8) |
| O3 | 0.3059 (8) | 0.3876 (3) | 0.67335 (19) | 0.156 (2) |
| O4 | 0.6241 (4) | 0.3774 (2) | 0.40968 (14) | 0.0803 (10) |
| O5 | 0.5121 (5) | 0.28645 (17) | 0.35414 (12) | 0.0786 (9) |
1 Source of material
As a medicinal herb, Pteris wallichiana C. Agardh has been used for the treatment of dysentery and traumatic injury for a long time. 5 Thus, we conducted a phytochemical investigation on this plant for a better development. The whole plant of Pteris wallichiana C. Agardh (47.5 kg) was collected from Changning County, Yunnan Province. The air-dried plant material was respectively extracted by 95 % methanol for twice, 85 % methanol for twice and 70 % methanol for once (seven days each time) at room temperature. The total extract (about 10.3 kg) was obtained by concentration under vacuum. Then the obtained extract was dissolved in warm water and extracted with isovolumic petroleum ether, ethyl acetate and n-butanol, respectively, to provide three fractions. The ethyl acetate fraction (304 g) was subjected to MCI column chromatography, eluting with methanol-water system (volume radio 50–100 %) to obtain four fractions (A–D). After repeated silica gel column chromatography, the title compound (24 mg) was purified and isolated from fraction B. The compound was further recrystallized with methanol to obtain a white needle-like crystal suitable for X-ray diffraction.
2 Experimental details
The carbon-bound hydrogen atoms were placed in their geometrically idealized positions and constrained to ride on their parent atoms with d(C–H) = 0.93–0.98 Å, Uiso(H) = 1.5 times Ueq(C) and 1.2 times Ueq(O).
3 Comment
The Pteris Wallichiana C. Agardh is a member of Pteris genus in the family Pteridaceae. It is a ethnomedicine that has the functions of clearing away heat and relieving dysentery, stabilizing shock and stopping bleeding. Thus, it is used for the treatment of dysentery, wind shock in children and traumatic bleeding. The previous studies have shown that this genus has good biological activities. 6 , 7 , 8 , 9 , 10 In order to better develop and utilize the Pteris wallichiana, its chemical constituents were preliminarily studied and the title compound was obtained. The compound has three ketone groups, one hydroxyl group and one double bond. The bond lengths and bond angles derived from the title structure are within the normal ranges. The ketone bonds were determined by the distance of 1.219(4) Å(C3–O3), 1.327(4) Å(C26–O2), 1.216(7) Å(C31–O5). The hydroxyl group was identified by the distance of 1.196(4) Å(C26′–O3′). And the double bond was identified by the distance of 1.326(5) Å (C24′–C25′). In general all bond lengths and angles are in the expected ranges. 11 , 12
Funding source: National Natural Science Foundation of China
Award Identifier / Grant number: 81660723
Funding source: National Natural Science Foundation of China
Award Identifier / Grant number: 82360689
Funding source: Natural-Scientific Research Program of Department of Education of Guizhou Province, Qianjiaoji
Award Identifier / Grant number: (2023)070
-
Conflict of interest statement: The authors declare no conflicts of interest regarding this article.
-
Author contribution: All the authors have accepted responsibility for the entire content of this submitted manuscript and approved submission.
-
Research funding: This work was funded by National Natural Science Foundation of China [No. 81660723], the National Natural Science Foundation of China [No. 82360689], the Natural-Scientific Research Program of Department of Education of Guizhou Province, Qianjiaoji [No. (2023)070].
References
1. Dolomanov, O. V.; Bourhis, L. J.; Gildea, R. J.; Howard, J. A. K.; Puschmann, H. OLEX2: A Complete Structure Solution, Refinement and Analysis Program. J. Appl. Crystallogr. 2009, 42, 339–341.10.1107/S0021889808042726Search in Google Scholar
2. Bruker. APEX2, SAINT and SADABS; Bruker AXS Inc.: Madison, Wisconsin, USA, 2012.Search in Google Scholar
3. Sheldrick, G. M. Crystal Structure Refinement with SHELXL. Acta Crystallogr. 2015, C71, 3–8.10.1107/S2053229614024218Search in Google Scholar PubMed PubMed Central
4. Brandenburg, K. DIAMOND. Visual Crystal Structure Information System. Ver. 4.0; Crystal Impact: Bonn, Germany, 2015.Search in Google Scholar
5. Pan, L. T. Medicinal Fern Plants in Guizhou; Guizhou Science and Technology Publishing House: Guiyang, China, 2012.Search in Google Scholar
6. Luo, X. K.; Li, C. J.; Luo, P.; Lin, X.; Ma, H.; Seeram, N. P.; Song, C.; Xu, J.; Gu, Q. Pterosin Sesquiterpenoids from Pteris cretica as Hypolipidemic Agents via Activating Liver X Receptors. J. Nat. Prod. 2016, 79, 3014–3021.10.1021/acs.jnatprod.6b00558Search in Google Scholar PubMed
7. Ni, G.; Fu, N. J.; Zhang, D.; Yang, H. Z.; Chen, X. G.; Yu, D. Q. An Unusual Dihydrobenzofuroisocoumarin and Ent–Kaurane Diterpenoids from Pteris multifida. J. Asian Nat. Prod. Res. 2015, 17, 423–429.10.1080/10286020.2015.1040777Search in Google Scholar PubMed
8. Wang, Y. S.; Li, F. Y.; Huang, R.; Li, Y.; Feng, X. F.; Yang, J. H. Chemical Constituents of Pteris multifida. Chem. Nat. Compd. 2013, 49, 629–631.10.1007/s10600-013-0695-9Search in Google Scholar
9. Li, W. F.; Wang, J.; Zhang, J. J.; Song, X.; Ku, C. F.; Zou, J.; Li, J. X.; Rong, L. J.; Pan, L. T.; Zhang, H. J.; Henrin, A. A New Anti–HIV Ent-Kaurane Diterpene from Pteris henryi. Int. J. Mol. Sci. 2015, 16, 27978–27987.10.3390/ijms161126071Search in Google Scholar PubMed PubMed Central
10. Shi, Y. S.; Zhang, Y.; Hu, W. Z.; Zhang, L. H.; Chen, X.; Zhang, N.; Li, G.; Tan, L. Y. Cytotoxic Diterpenoids from Pteris ensiformis. J. Asian Nat. Prod. Res. 2017, 19, 188–193.10.1080/10286020.2016.1274307Search in Google Scholar PubMed
11. Xiong, H.-P.; Wu, Z.-J.; Chen, F.-T.; Chen, W.-S. 24-Methylenecycloartanone. Acta Crystallogr. 2010, E66, o260–o261.10.1107/S1600536809055123Search in Google Scholar PubMed PubMed Central
12. Zhao, X.-T.; Yu, M.-H.; Su, S.-Y.; Shi, X.-L.; Lei, C.; Hou, A.-J. Cycloartane Triterpenoids from Pseudolarix Amabilis and Their Antiviral Activity. Phytochemistry 2020, 171, 112229.10.1016/j.phytochem.2019.112229Search in Google Scholar PubMed
© 2024 the author(s), published by De Gruyter, Berlin/Boston
This work is licensed under the Creative Commons Attribution 4.0 International License.
Articles in the same Issue
- Frontmatter
- Editorial
- Editorial 2024 – New developments and changes of Zeitschrift für Kristallographie – New Crystal Structures
- New Crystal Structures
- Hydrogen bonding and π⋅⋅⋅halogen interactions in the crystal structure of bis(theophyllinium) hexachloridoplatinate(IV) monohydrate
- The crystal structure of 6-amino-2-carboxypyridin-1-ium perchlorate, C6H7ClN2O6
- Crystal structure of poly[(μ4-(3-amino-1H-1,2,4-triazol-1-yl)benzene-1,3-dicarboxylato-κ 4 N:O:O':O')(1-methylpyrroldin-2-one-κ1O)dicopper(II)] – 1-methylpyrroldin-2-one (1/3), C40H48Cu2N12O12
- The crystal structure of 18-crown-6-k6O6(2,4,5-trinitroimidazol-1-ido-k1O)potassium(I)
- Crystal structure of poly[tetraaqua-bis(μ2-5-bromoisophthalato-κ3O,O′:O″)-(μ2-1,5-bis(imidazol-2-methyl)pentane-κ2N:N′)dicadmium(II)] dihydrate
- Crystal structure of (5R,6S,E)-5-acetoxy-2-methyl-6-((2aR,3R,5aS,5bS,11aR,12aS)-2a,5a,8,8-tetramethyl-9-oxotetradecahydro-1H,12H-cyclopenta[a]cyclopropa[e]phenanthren-3-yl)hept-2-enoic acid, C32H48O5
- The crystal structure of poly[diaqua-bis(μ2 -thiocyanato-κ2N:O)cobalt(II) monohydrate
- The crystal structure of 1,3,5-tri(1H-imidazol-1-yl)benzene–2,3,5,6-tetrachlorobenzene-1,4-dicarboxylic acid (1/1)
- Crystal structure of dichlorido-bis(1-[(2-ethyl-benzimidazole-1-yl)methyl]-1H–benzotriazole) cadmium(II), C32H32CdN10OCl2
- The crystal structure of N′-(tert-butyl)-N′-(3,5-dimethylbenzoyl)-3-methoxy-N,2-dimethylbenzohydrazide, C23H30N2O3
- Crystal stucture of 3-benzamido-N-(2-bromo-4-(perfluoropropan-2-yl)-6-(trifluoromethyl)phenyl)-2-fluorobenzamide
- Crystal structure of bis(μ-benzeneselenolato)-(tetracarbonyl)-{μ-[N-(diphenylphosphanyl)-N-(3-ethynylphenyl)-P,P-diphenylphosphinous amide]} diiron, C48H35Fe2NO4P2Se2
- The crystal structure of 2′-(p-tolyl)-4′H-spiro[isochromane-1,1′-naphthalene]-3,4′-dione, C25H18O3
- The crystal structure of poly[hexaqua-tetrakis(μ4-pyridine-2,4-dicarboxylate-κ5N: O: O′: O″: O‴)-bi(μ2-pyridine-2,4-dicarboxylate-κ3N: O: O′)-digadolinium(III)tricopper (II)], [Gd2Cu3(C7H3NO4)6(H2O)6] n
- Crystal structure of poly[bis(4-(4-(pyridin-4-yl)phenyl)pyridin-1-ium-κ1N)-(μ4-benzene-1,2,4,5-tetracarboxylato-κ5O:O′: O″:O‴:O⁗)-(μ2-2,5-dicarboxyterephthalato-κ2O:O′)dizinc(II)], C52H32N4O16Zn2
- The crystal structure of 4-(3-carboxy-1-ethyl-6-fluoro-4-oxo-1,4-dihydroquinolin-7-yl)piperazin-1-ium 2-carboxy-6-nitrobenzoate monohydrate, C24H25FN4O10
- Crystal structure of dichlorido-(1-((3,5-dimethyl-2,3-dihydro-1H-1,2,3-triazol-1-yl)methyl)-1H-benzo[d][1,2,3]triazole-k1N)zinc(II), C22H24ZnN12Cl2
- The crystal structure of (3-chlorothiophene-2-carboxylato-κ2O, O′)-(2,2′-dipyridyl-κ2N,N′)lead(II), C20H12Cl2N2O4S2Pb
- Synthesis and crystal structure of (Z)-4-((1-(3-fluorophenyl)-1H-1,2,3-triazol-4-yl)methylene)-5-methyl-2-phenyl-2,4-dihydro-3H-pyrazol-3-one, C19H14FN5O
- The crystal structure of the coordination compound catena-poly[(18-crown-6-ether-κ6O6)(4,5-dinitroimidazolato-κ1O)potassium(I)]
- Crystal structure of 7-(diethylamino)-3-(trifluoroacetyl)-2H-chromen-2-one, C15H14F3NO3
- Crystal structure of dichlorido-1-[(2-ethylimidazole-1-yl)methyl]-1H–benzotriazole κ1N zinc(II), C24H26ZnN10Cl2
- Crystal and molecular structure of 5-bromopyridine-2,3-diamine
- Crystal structure of catena-poly[bis(μ2-1-(3-carboxyphenyl)-5-methyl-4-oxo-1,4-dihydropyridazine-3-carboxylato-k3-O,O′:O″)hexaqua-dicobalt tetrahydrate], C26H36N4O20Co2
- Crystal structure of thiocyanate-κ1N-bis(μ1-2,6-diformyl-4-methylphenol oxime-κ2N,O)-manganese(III) acetonitrile solvate, C21H21MnN6O6S
- The crystal structure of pyrrolidin-1-yl pivalate, C9H13NO4
- The crystal structure of 2,2′-(2,2-diphenylethene-1,1-diyl)bis(1,4-dimethoxybenzene), C30H28O4
- Crystal structure of bis(benzyltrimethylammonium) tetrathiotungstate(VI), {(C6H5CH2)(CH3)3N}2[WS4]
- The crystal structure of ethyl (Z)-2-(ethoxymethylene)-3-oxobutanoate, C9H14O4
- The crystal structure of (E)-6-bromo-3,5-dimethyl-2-(1-phenylprop-1-en-2-yl)-3Himidazo[4,5b]pyridine, C17H16BrN3
- Crystal structure of (3S,3′S,4R,4′S)-3′-(furan-3-yl)-3-hydroxy-4′-methyl-3,5,6′,7′-tetrahydro-1H,3′H-4,5′-spirobi[isobenzofuran]-1,1′(4′H)-dione-methanol (1/1), C21H22O7
- Cocrystal structure of progesterone-isophthalic acid, C25H33O4
- The crystal structure of 3-(6-fluoro-1H-indol-3-yl)-1-methylquinoxalin-2(1H)-one, C17H12FN3O
- Crystal structure of S-(4-carboxybutyl)- l -cysteine
- The cocrystal of 2,2′-(hydrazine-1,1-diyl)bis(1H-imidazole-4,5-dicarbonitrile)– methanol (2/3)
- Crystal structure of (1′R,2′S,4′R,6′S)-4,6-dihydroxy-1′,8′,8′-trimethyl-3-(3-methylbutanoyl)-4′,8′,6′,1′,7,2′-hexahydro-1H-4′,6′-methanoxanthene-8-carbaldehyde, C23H30O5
- Crystal structure of (3,6-di(2-pyridyl)-4-methylphenyl pyridazine-k 2 N,N′)-bis(1-phenyl-pyrazole-κ 2 C,N) iridium(III) hexafluorophosphate, C39H29F6IrN8P
- Crystal structure of 1,5-bis[(E)-1-(2-hydroxyphenyl)ethylidene]thiocarbonohydrazide dimethyl sulfoxide monosolvate, C17H18N4O2S·C2H6OS
- Crystal structure of (S)-4-(2-(4-(2-acetyl-5-chlorophenyl)-3-methoxy-6-oxopyridazin-1(6H)-yl)-3-phenylpropanamido)benzoic acid monohydrate, C29H26ClN3O7
- The crystal structure of 1,3-bis(2,4-dinitro-1H-imidazol-1-yl)propane
- Crystal structure of 4-chlorobenzyl (S)-2-(6-methoxynaphthalen-2-yl)propanoate, C21H19ClO3
- Crystal structure of 1-(5-(benzo[d][1,3]dioxol-5-yl)-4-benzyl-1-(4-bromophenyl)-4,5-dihydro-1H-1,2,4-triazol-3-yl)ethan-1-one, C24H20BrN3O3
- The crystal structure of (Z)-3′-(2-(1-(3,4-dimethyl-phenyl)-3-methyl-5-oxo-1,5-dihydro-4H-pyrazol-4-ylidene)hydrazinyl)-2′-hydroxy-[1,1′-biphenyl]-3-carboxylicacid ─ methanol (1/1), C26H26N4O5
- Crystal structure of (S)-1-phenylpropan-1-aminium (S)-(1-phenylpropyl)carbamate C19H26N2O2
- Synthesis and crystal structure of methyl 2-((5-bromo-4-(4-cyclopropylnaphthalen-1-yl)-4H-1,2,4-triazol-3-yl)thio)acetate, C18H16BrN3O2S
- The crystal structure of trichlorobis(pyridine-2,6-dithio-κS-carbomethylamido)antimony(III), [SbCl3(C9H11N3S2)2]
- Crystal structure of 1,8-dihydroxy-3-{[(triphenylstannyl)oxy]carbonyl} anthracene-9,10-dione, C33H22O6Sn
- The crystal structure of (E)-4-(2-(pyridin-4-ylmethylene)hydrazine-1-carbonyl)pyridin-1-ium-2-olate dihydrate, C12H14N4O4
- The crystal structure of 6-amino-pyridinium-2-carboxylate, C6H6N2O2
- The crystal structure of catena-poly[aqua-nitrato-κ3O,O:O′′-(1,10-phenanthroline-κ2N,N′)sodium(I)], C24H18N6O7Na2
- Retractions
- Retraction of: Crystal structure of bis[diaquaisonicotinatosamarium(III)]-µ-isonicotinato-[diisonicotinatocopper(II)], CuSm2(C6H4NO2)8(H2O)4
- Retraction of: Crystal structure of aqua(2,2′-bipyridine-k 2 N:N′)(nitrato)-(4-aminobenzoato)cadmium(II) nitrate, [Cd(H2O)(NO3)(C10H8N2)(C7H7NO2)][NO3]
Articles in the same Issue
- Frontmatter
- Editorial
- Editorial 2024 – New developments and changes of Zeitschrift für Kristallographie – New Crystal Structures
- New Crystal Structures
- Hydrogen bonding and π⋅⋅⋅halogen interactions in the crystal structure of bis(theophyllinium) hexachloridoplatinate(IV) monohydrate
- The crystal structure of 6-amino-2-carboxypyridin-1-ium perchlorate, C6H7ClN2O6
- Crystal structure of poly[(μ4-(3-amino-1H-1,2,4-triazol-1-yl)benzene-1,3-dicarboxylato-κ 4 N:O:O':O')(1-methylpyrroldin-2-one-κ1O)dicopper(II)] – 1-methylpyrroldin-2-one (1/3), C40H48Cu2N12O12
- The crystal structure of 18-crown-6-k6O6(2,4,5-trinitroimidazol-1-ido-k1O)potassium(I)
- Crystal structure of poly[tetraaqua-bis(μ2-5-bromoisophthalato-κ3O,O′:O″)-(μ2-1,5-bis(imidazol-2-methyl)pentane-κ2N:N′)dicadmium(II)] dihydrate
- Crystal structure of (5R,6S,E)-5-acetoxy-2-methyl-6-((2aR,3R,5aS,5bS,11aR,12aS)-2a,5a,8,8-tetramethyl-9-oxotetradecahydro-1H,12H-cyclopenta[a]cyclopropa[e]phenanthren-3-yl)hept-2-enoic acid, C32H48O5
- The crystal structure of poly[diaqua-bis(μ2 -thiocyanato-κ2N:O)cobalt(II) monohydrate
- The crystal structure of 1,3,5-tri(1H-imidazol-1-yl)benzene–2,3,5,6-tetrachlorobenzene-1,4-dicarboxylic acid (1/1)
- Crystal structure of dichlorido-bis(1-[(2-ethyl-benzimidazole-1-yl)methyl]-1H–benzotriazole) cadmium(II), C32H32CdN10OCl2
- The crystal structure of N′-(tert-butyl)-N′-(3,5-dimethylbenzoyl)-3-methoxy-N,2-dimethylbenzohydrazide, C23H30N2O3
- Crystal stucture of 3-benzamido-N-(2-bromo-4-(perfluoropropan-2-yl)-6-(trifluoromethyl)phenyl)-2-fluorobenzamide
- Crystal structure of bis(μ-benzeneselenolato)-(tetracarbonyl)-{μ-[N-(diphenylphosphanyl)-N-(3-ethynylphenyl)-P,P-diphenylphosphinous amide]} diiron, C48H35Fe2NO4P2Se2
- The crystal structure of 2′-(p-tolyl)-4′H-spiro[isochromane-1,1′-naphthalene]-3,4′-dione, C25H18O3
- The crystal structure of poly[hexaqua-tetrakis(μ4-pyridine-2,4-dicarboxylate-κ5N: O: O′: O″: O‴)-bi(μ2-pyridine-2,4-dicarboxylate-κ3N: O: O′)-digadolinium(III)tricopper (II)], [Gd2Cu3(C7H3NO4)6(H2O)6] n
- Crystal structure of poly[bis(4-(4-(pyridin-4-yl)phenyl)pyridin-1-ium-κ1N)-(μ4-benzene-1,2,4,5-tetracarboxylato-κ5O:O′: O″:O‴:O⁗)-(μ2-2,5-dicarboxyterephthalato-κ2O:O′)dizinc(II)], C52H32N4O16Zn2
- The crystal structure of 4-(3-carboxy-1-ethyl-6-fluoro-4-oxo-1,4-dihydroquinolin-7-yl)piperazin-1-ium 2-carboxy-6-nitrobenzoate monohydrate, C24H25FN4O10
- Crystal structure of dichlorido-(1-((3,5-dimethyl-2,3-dihydro-1H-1,2,3-triazol-1-yl)methyl)-1H-benzo[d][1,2,3]triazole-k1N)zinc(II), C22H24ZnN12Cl2
- The crystal structure of (3-chlorothiophene-2-carboxylato-κ2O, O′)-(2,2′-dipyridyl-κ2N,N′)lead(II), C20H12Cl2N2O4S2Pb
- Synthesis and crystal structure of (Z)-4-((1-(3-fluorophenyl)-1H-1,2,3-triazol-4-yl)methylene)-5-methyl-2-phenyl-2,4-dihydro-3H-pyrazol-3-one, C19H14FN5O
- The crystal structure of the coordination compound catena-poly[(18-crown-6-ether-κ6O6)(4,5-dinitroimidazolato-κ1O)potassium(I)]
- Crystal structure of 7-(diethylamino)-3-(trifluoroacetyl)-2H-chromen-2-one, C15H14F3NO3
- Crystal structure of dichlorido-1-[(2-ethylimidazole-1-yl)methyl]-1H–benzotriazole κ1N zinc(II), C24H26ZnN10Cl2
- Crystal and molecular structure of 5-bromopyridine-2,3-diamine
- Crystal structure of catena-poly[bis(μ2-1-(3-carboxyphenyl)-5-methyl-4-oxo-1,4-dihydropyridazine-3-carboxylato-k3-O,O′:O″)hexaqua-dicobalt tetrahydrate], C26H36N4O20Co2
- Crystal structure of thiocyanate-κ1N-bis(μ1-2,6-diformyl-4-methylphenol oxime-κ2N,O)-manganese(III) acetonitrile solvate, C21H21MnN6O6S
- The crystal structure of pyrrolidin-1-yl pivalate, C9H13NO4
- The crystal structure of 2,2′-(2,2-diphenylethene-1,1-diyl)bis(1,4-dimethoxybenzene), C30H28O4
- Crystal structure of bis(benzyltrimethylammonium) tetrathiotungstate(VI), {(C6H5CH2)(CH3)3N}2[WS4]
- The crystal structure of ethyl (Z)-2-(ethoxymethylene)-3-oxobutanoate, C9H14O4
- The crystal structure of (E)-6-bromo-3,5-dimethyl-2-(1-phenylprop-1-en-2-yl)-3Himidazo[4,5b]pyridine, C17H16BrN3
- Crystal structure of (3S,3′S,4R,4′S)-3′-(furan-3-yl)-3-hydroxy-4′-methyl-3,5,6′,7′-tetrahydro-1H,3′H-4,5′-spirobi[isobenzofuran]-1,1′(4′H)-dione-methanol (1/1), C21H22O7
- Cocrystal structure of progesterone-isophthalic acid, C25H33O4
- The crystal structure of 3-(6-fluoro-1H-indol-3-yl)-1-methylquinoxalin-2(1H)-one, C17H12FN3O
- Crystal structure of S-(4-carboxybutyl)- l -cysteine
- The cocrystal of 2,2′-(hydrazine-1,1-diyl)bis(1H-imidazole-4,5-dicarbonitrile)– methanol (2/3)
- Crystal structure of (1′R,2′S,4′R,6′S)-4,6-dihydroxy-1′,8′,8′-trimethyl-3-(3-methylbutanoyl)-4′,8′,6′,1′,7,2′-hexahydro-1H-4′,6′-methanoxanthene-8-carbaldehyde, C23H30O5
- Crystal structure of (3,6-di(2-pyridyl)-4-methylphenyl pyridazine-k 2 N,N′)-bis(1-phenyl-pyrazole-κ 2 C,N) iridium(III) hexafluorophosphate, C39H29F6IrN8P
- Crystal structure of 1,5-bis[(E)-1-(2-hydroxyphenyl)ethylidene]thiocarbonohydrazide dimethyl sulfoxide monosolvate, C17H18N4O2S·C2H6OS
- Crystal structure of (S)-4-(2-(4-(2-acetyl-5-chlorophenyl)-3-methoxy-6-oxopyridazin-1(6H)-yl)-3-phenylpropanamido)benzoic acid monohydrate, C29H26ClN3O7
- The crystal structure of 1,3-bis(2,4-dinitro-1H-imidazol-1-yl)propane
- Crystal structure of 4-chlorobenzyl (S)-2-(6-methoxynaphthalen-2-yl)propanoate, C21H19ClO3
- Crystal structure of 1-(5-(benzo[d][1,3]dioxol-5-yl)-4-benzyl-1-(4-bromophenyl)-4,5-dihydro-1H-1,2,4-triazol-3-yl)ethan-1-one, C24H20BrN3O3
- The crystal structure of (Z)-3′-(2-(1-(3,4-dimethyl-phenyl)-3-methyl-5-oxo-1,5-dihydro-4H-pyrazol-4-ylidene)hydrazinyl)-2′-hydroxy-[1,1′-biphenyl]-3-carboxylicacid ─ methanol (1/1), C26H26N4O5
- Crystal structure of (S)-1-phenylpropan-1-aminium (S)-(1-phenylpropyl)carbamate C19H26N2O2
- Synthesis and crystal structure of methyl 2-((5-bromo-4-(4-cyclopropylnaphthalen-1-yl)-4H-1,2,4-triazol-3-yl)thio)acetate, C18H16BrN3O2S
- The crystal structure of trichlorobis(pyridine-2,6-dithio-κS-carbomethylamido)antimony(III), [SbCl3(C9H11N3S2)2]
- Crystal structure of 1,8-dihydroxy-3-{[(triphenylstannyl)oxy]carbonyl} anthracene-9,10-dione, C33H22O6Sn
- The crystal structure of (E)-4-(2-(pyridin-4-ylmethylene)hydrazine-1-carbonyl)pyridin-1-ium-2-olate dihydrate, C12H14N4O4
- The crystal structure of 6-amino-pyridinium-2-carboxylate, C6H6N2O2
- The crystal structure of catena-poly[aqua-nitrato-κ3O,O:O′′-(1,10-phenanthroline-κ2N,N′)sodium(I)], C24H18N6O7Na2
- Retractions
- Retraction of: Crystal structure of bis[diaquaisonicotinatosamarium(III)]-µ-isonicotinato-[diisonicotinatocopper(II)], CuSm2(C6H4NO2)8(H2O)4
- Retraction of: Crystal structure of aqua(2,2′-bipyridine-k 2 N:N′)(nitrato)-(4-aminobenzoato)cadmium(II) nitrate, [Cd(H2O)(NO3)(C10H8N2)(C7H7NO2)][NO3]

