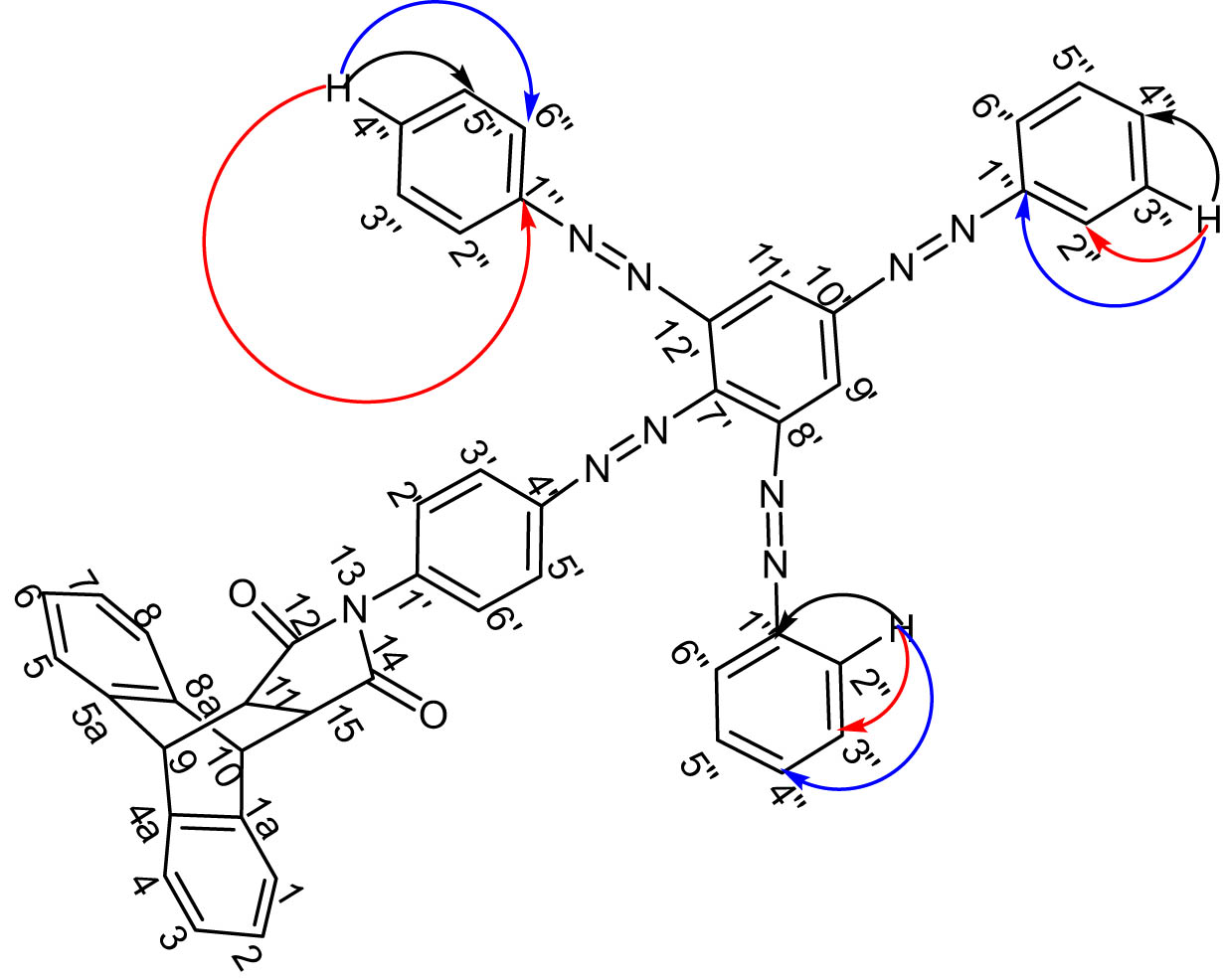
The present work describes the synthesis, characterization, and evaluation of the antibacterial activity of a new poly azo compound resulting from the coupling of a previously reported N -arylsuccinimid precursor 5 with the diazonium ion of aniline. This azo compound was characterized using its physical, elemental, and 1D and 2D spectroscopic data. The novel azo compound 7 (minimum inhibitory concentration [MIC] = 16–32 μg/mL) showed higher antibacterial activity than its precursor 5 (MIC = 32–64 μg/mL), although it was low compared to the reference drug ciprofloxacin (MIC = 0.5–4 μg/mL). Graphical abstract
Contents
- Research Articles
-
Open AccessDesign, synthesis, and antiproliferative activity of novel 1,2,4-triazole-chalcone compoundsOctober 27, 2023
-
November 15, 2023
-
December 12, 2023
- Erratum
- SI: Undergraduate Research in the Synthesis of Biologically Active Small Molecules and Their Applications
-
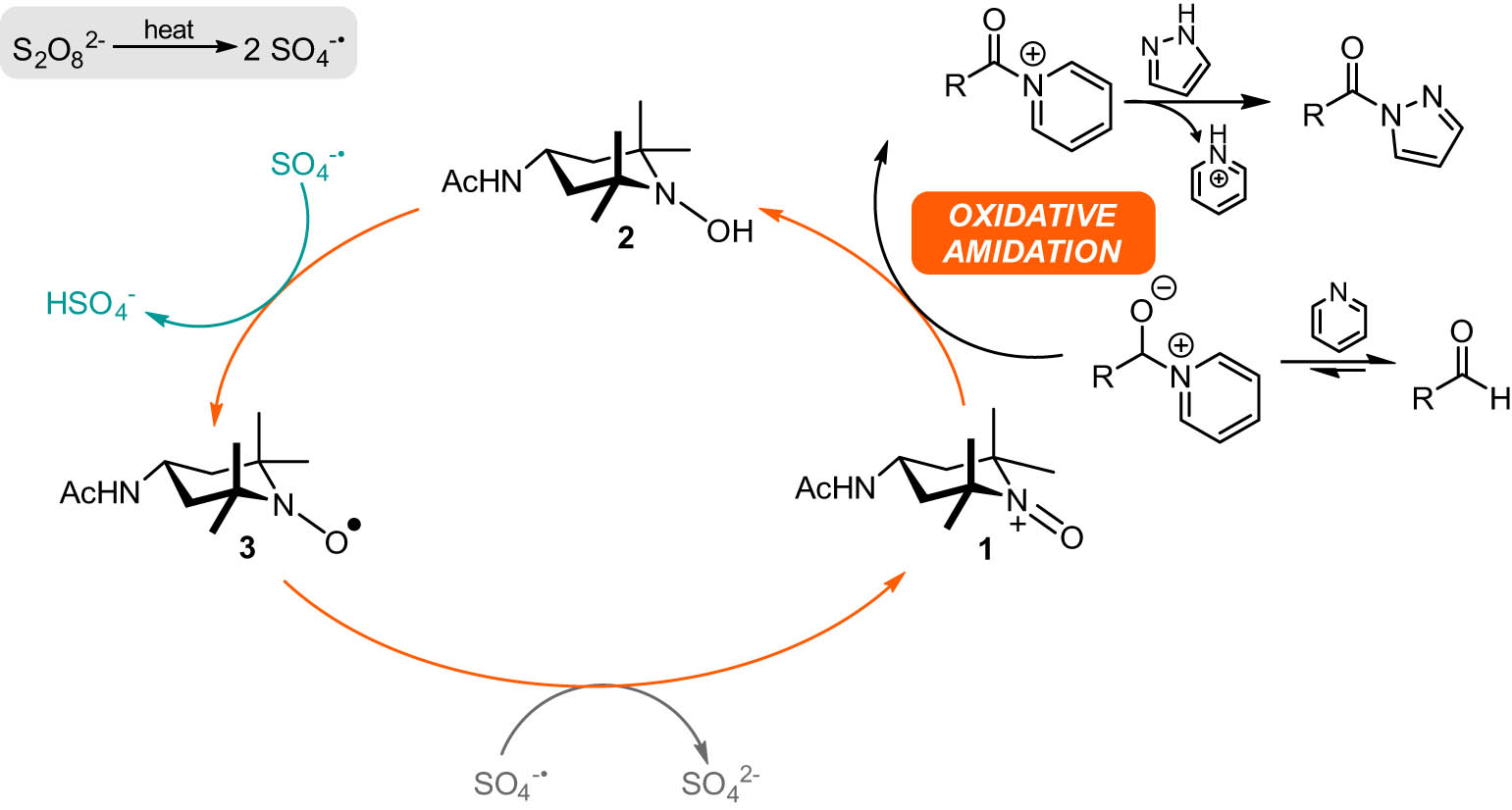
Open AccessPreparation of novel acyl pyrazoles and triazoles by means of oxidative functionalization reactionsFebruary 7, 2023
- SI: Development of Heterocycles for Biomedical and Bioanalytical Applications
-
Open AccessInfluence of octreotide on apoptosis and metabolome expression in lipopolysaccharide-induced A549 cellsFebruary 28, 2023
-
April 20, 2023