Abstract
Objectives
Preeclampsia (PE) is more common in multiple pregnancies, especially dichorionic twins, which often show birthweight discordance. The threshold of birthweight discordance linked to increased risk remains unclear, based largely on the Delphi consensus method. This study examines whether discordance independently predicts PE and identifies the most predictive threshold to guide prenatal care.
Methods
We conducted a retrospective population-based cohort study of 2,546 women with dichorionic twins who delivered in Slovenia between 2013 and 2022, using data from the National Perinatal Information System. The association between twin birthweight discordance, the main independent variable, and PE, the main outcome variable, was determined using multivariate logistic regression. We calculated the crude odds ratio (OR) and adjusted odds ratio (aOR) with a 95 % confidence interval. ROC analysis determined the area under the curve (AUC), the sensitivity, and the false positive rate for predicting PE. Two-sided p-values <0.05 were considered statistically significant.
Results
In our cohort PE occurred in 5.38 %. Compared with a birthweight discordance of <10 %, a discordance >25 % was identified as a statistically significant independent risk factor for PE development (aOR=1.66; 95 % CI 1.10–2.52; p=0.016). Birthweight discordance between 10 and 25 % was not significantly associated with PE. The AUC for predicting PE with birthweight discordance was significant at 55.2 % (51.6–58.8 %). However, at a birthweight discordance threshold of >25 %, only 10.2 % of pregnant women with PE could be identified with 5.8 % false-positive results.
Conclusions
This study revealed that a birthweight discordance >25 % in dichorionic twins significantly increases PE risk.
Introduction
Hypertensive disorders in pregnancy, including preeclampsia (PE), are among the most prevalent pregnancy complications and represent a leading contributor to maternal and perinatal morbidity and mortality globally [1], 2]. PE is estimated to affect approximately 2–8 % of pregnancies worldwide. In developing countries, it accounts for up to 26 % of maternal deaths, while in developed countries, PE is responsible for up to 16 % of maternal deaths [3]. Identifying risk factors for PE is therefore crucial for implementing effective preventive measures and clinical management strategies [4].
The incidence of twin gestations has been increasing in recent years, primarily due to rising maternal age at conception and the increased use of assisted reproductive technologies. This trend contributes to a higher prevalence of PE, as twin pregnancies are associated with a two- to threefold elevated risk of PE development [5], [6], [7]. The risk is particularly pronounced in dichorionic twin pregnancies, where each fetus has its own placenta. The augmented total placental mass is likely associated with an increased risk of suboptimal placentation, a key factor in the pathophysiology of PE [5], [8], [9], [10], [11]. Moreover, dichorionic twin pregnancies exhibit a higher incidence of placental abnormalities, which are often associated with increased intertwin birthweight discordance. Birthweight discordance in dichorionic twins may indicate impaired placental function in the smaller twin, which shares the same pathophysiological basis as PE [11], 12]. This poses a distinct clinical challenge for antenatal monitoring, early diagnosis, and timely intervention in twin pregnancies.
Few international studies have explored the impact of twin pregnancy and intertwin birthweight discordance on PE, with some also considering chorionicity [5], [6], [7], [8], [9], [10], [11], [12], [13], [14], [15], [16], [17], [18], [19], however their findings remain inconsistent. Additionally, there is no uniform definition across studies regarding the degree of discordance that significantly increases the risk of PE, current thresholds are based primarily on the Delphi consensus method. Due to these conflicting findings, further research is needed.
In this study, we aimed to determine whether birthweight discordance in dichorionic twin pregnancies is independently associated with the development of PE in the mother, regardless of other risk factors. Our main research hypothesis was that there would be a significant proportional association between birthweight discordance in dichorionic twins and the development of maternal PE.
Subjects and methods
We conducted a retrospective population-based cohort study of 2,546 women with dichorionic twins who gave birth in Slovenia from January 1st 2013 to December 31st 2022, and who met all the inclusion criteria outlined below. Anonymized data were obtained from the National Perinatal Information System of Slovenia (NPIS).
The data are collected at the time of delivery by midwives and attending doctors and are entered into the system according to strict methodological guidelines in all 14 maternity hospitals in Slovenia [20]. Data registration is legally required, as the NPIS also functions as the official birth registry of Slovenia.
Between 2013 and 2022, 2,671 dichorionic twin deliveries were recorded in Slovenia. For the purposes of this analysis, 61 cases were excluded due to perinatal mortality, 46 due to major structural anomalies, and 14 due to the coexistence of both conditions. An additional four pregnancies were excluded due to missing data on maternal height and pre-pregnancy BMI. The final analytical cohort comprised 2,546 dichorionic twin pregnancies.
The inclusion criteria comprised the presence of data for the selected variables in each pregnancy. We investigated birth weight discordance between twins as the main independent variable (%) and the occurrence of PE as the outcome variable (yes/no), as well as the following potential confounding variables: maternal age (years), parity (nullipara/multipara), mode of conception (spontaneous/IVF), pre-pregnancy body mass index (BMI) (<18.5, 18.5–25, 25–30, ≥30 kg/m2), presence of pre-pregnancy arterial hypertension (AH) (yes/no), presence of pre-pregnancy diabetes mellitus (DM) (yes/no), gestational age at delivery (completed week), birth weight below the 10th percentile for one or both fetuses (SGA) (yes/no) [21], presence of gestational DM (yes/no), gestational weight gain (kg), smoking during pregnancy (yes/no), preventive treatment with low-dose aspirin (yes/no), insulin therapy during pregnancy (yes/no), and sex of the twins (male-male/female-female/male-female). We defined nullipara as a woman with no history of viable pregnancies and multipara as a woman who had previously delivered at least once at ≥22 weeks of gestation or later.
PE was defined as new-onset hypertension after the 20th week of pregnancy and proteinuria or involvement of any other organ [3]. Hypertension was defined as a systolic blood pressure of 140 mmHg or higher and/or a diastolic blood pressure of 90 mmHg or higher on two separate measurements taken at least 4 h apart after the 20th week of pregnancy in a previously normotensive woman. Proteinuria was defined as the presence of 300 mg or more of protein in a 24-h urine sample, a protein/creatinine ratio of 0.3 mg/dL or more, or a protein level of 2+ or higher on a urine dipstick test. In the absence of proteinuria, PE was characterized as new-onset hypertension accompanied by at least one of the signs of organ failure (liver, kidney or lung) or neurologic symptoms.
Discordance was defined as the difference in birth weight between the twins. It was calculated using the following formula:
Birth weight discordance was analyzed both as a continuous variable (%) and as a categorical variable (<10 %, 10–25 %, >25 %).
Exclusion criteria included the presence of congenital anomalies in one or both fetuses, perinatal death of one twin, and missing or implausible data for the included variables (values more than 3 SD from the mean).
Statistical analyses
We described the demographic and clinical characteristics of women with and without PE using descriptive statistics: numbers and frequencies for categorical variables and means with standard deviation (SD) for continuous (linear) variables. The association between birth weight discordance and PE was analyzed using logistic regression. Confounding variables that showed a significance level (p-value) of <0.1 in univariate logistic analysis were included. We calculated adjusted odds ratios (aOR) with 95 % confidence intervals (CI). Receiver operating characteristic (ROC) analysis was used to calculate the area under the curve (AUC), as well as sensitivity and false positive predictive value for the prediction of PE based on birth weight discordance. Linear regression was used to assess the correlation between gestational age at delivery and birth weight discordance. Two-tailed p-values <0.05 were considered statistically significant.
No patients were involved in the design or implementation of the study. There are no plans to involve patients in dissemination.
For statistical calculations, we used IBM SPSS Statistics for Windows, Version 29.0. (IBM Corporation, Armonk, New York, USA).
Ethical approval
The research was approved by the National Medical Ethics Committee of the Republic of Slovenia on January, 16th, 2024 (No. 0120–559/2023/3).
Results
The clinical and demographic characteristics of women with and without PE from our cohort are presented in Table 1. The incidence of PE in the cohort was 5.38 %. Compared to those without PE, women with PE were more often nulliparous, had higher mean pre-pregnancy BMI, had a greater prevalence of pre-gestational AH and pre-gestational DM, and were more likely to be treated with insulin. The mean gestational age at delivery was lower in the PE group. Additionally, in pregnancies affected by PE, birthweight discordance increased by 0.17 % for each week of shorter gestation, whereas in those without PE, the increase was only 0.05 % per week. Overall, the mean birthweight discordance was greater in the PE group.
Demographic and clinical characteristics of pregnant women and dichorionic twin pregnancies (n=2,546).
| Variables | Women without PE | Women with PE | OR | 95 % CI | p-Value |
|---|---|---|---|---|---|
| (n=2,409) (94.62) | (n=137) (5.38) | ||||
| Maternal age, years | 32.08 (4.89) | 33.16 (5.37) | 0.090 | ||
| Nulliparity | 1,263 (52.43) | 100 (72.99) | 2.45 | 1.67–3.60 | <0.001 |
| Conception via IVF | 673 (27.94) | 46 (33.58) | 1.30 | 0.91–1.88 | 0.154 |
| Pre-pregnancy BMI, kg/m2a | 24.24 (4.67) | 26.02 (5.82) | 1.07 | 1.04–1.11 | <0.001 |
| Pre-pregnancy hypertension | 15 (0.62) | 12 (8.76) | 15.32 | 7.02–33.43 | <0.001 |
| Pre-pregnancy diabetes | 7 (0.29) | 2 (1.46) | 5.08 | 1.05–24.71 | 0.025 |
| Gestational age at delivery, weeks | 35.49 (2.60) | 34.80 (2.02) | 0.91 | 0.87–0.97 | <0.001 |
| SGA (in one or both twins) | 201 (8.34) | 14 (10.22) | 1.25 | 0.71–2.21 | 0.443 |
| Gestational diabetes | 351 (14.57) | 24 (17.52) | 0.91 | 0.79–1.96 | 0.344 |
| GWG, kgb | 15.50 (6.45) | 16.53 (7.42) | 1.02 | 0.99–1.05 | 0.051 |
| Smoking during pregnancy | 197 (8.18) | 9 (6.57) | 0.79 | 0.40–1.58 | 0.502 |
| Preventive treatment with low-dose aspirin | 121 (5.02) | 10 (7.30) | 1.49 | 0.76–2.91 | 0.241 |
| Insulin treatment during pregnancy | 61 (5.02) | 8 (5.84) | 2.39 | 1.12–5.09 | 0.020 |
| Sex of twins | |||||
| Male-male | 735 (30.51) | 38 (27.74) | Ref | ||
| Female-male | 1,042 (43.25) | 55 (40.15) | 1.02 | 0.67–1.56 | 0.924 |
| Female-female | 632 (26.23) | 44 (32.12) | 1.35 | 0.86–2.10 | 0.192 |
| Birth weight discordance, % | 10.87 (8.34) | 12.44 (8.82) | 1.02 | 1.00–1.04 | 0.041 |
-
Results are presented according to the type of variable as mean (SD) or number (%). Odds ratios for BMI (kg/m2), gestational age at delivery (weeks), GWG (kg), and birth weight discordance (%) are presented per unit increase. BMI, body mass index; CI, confidence interval; SGA, small for gestational age; GWG, gestational weight gain; IVF, in vitro fertilization; OR, odds ratio; PE, preeclampsia; SD, standard deviation.
Moreover, BMI and gestational weight gain (GWG) were categorized as shown in Table 2. Compared to the group without PE, the group with PE included a higher proportion of women with a BMI over 30 kg/m2 (21.90 vs. 11.50 %) and more women with excessive GWG (70.80 vs. 58.16 %).
Risk of developing preeclampsia in dichorionic twin pregnancies with birth weight discordance.
| Variables | Women without PE (n=2,409) (94.62) | Women with PE (n=137) (5.38) | aORc | 95 % CI | p-Value |
|---|---|---|---|---|---|
| Birth weight discordance | |||||
| <10 % | 1,311 (54.42) | 69 (50.36) | Ref | ||
| 10–25 % | 942 (39.10) | 51 (37.23) | 0.91 | 0.69–1.20 | 0.498 |
| >25 % | 156 (6.48) | 17 (12.41) | 1.66 | 1.10–2.52 | 0.016 |
| Pre-pregnancy hypertension | 15 (0.62) | 12 (8.76) | 10.82 | 5.96–19.66 | <0.001 |
| Nulliparity | 1,263 (52.43) | 100 (72.99) | 2.52 | 1.89–3.35 | <0.001 |
| Insulin treatment during pregnancy | 61 (5.02) | 8 (5.84) | 2.10 | 1.14–3.84 | 0.017 |
| GWGa | |||||
| Insufficient | 267 (11.08) | 14 (10.22) | 1.35 | 0.83–2.20 | 0.230 |
| Adequate | 740 (30.72) | 26 (18.98) | Ref | ||
| Excessive | 1,401 (58.16) | 97 (70.80) | 2.11 | 1.52–2.93 | <0.001 |
| Pre-pregnancy BMIb | |||||
| Underweight | 80 (3.32) | 1 (0.73) | 0.31 | 0.08–1.28 | 0.106 |
| Normal weight | 1,529 (63.47) | 78 (56.93) | Ref | ||
| Overweight | 523 (21.71) | 28 (20.44) | 0.97 | 0.70–1.35 | 0.862 |
| Obese | 277 (11.50) | 30 (21.90) | 1.86 | 1.33–2.60 | <0.001 |
| Maternal age, years | 32.08 (4.89) | 33.16 (5.37) | 1.05 | 1.02–1.07 | <0.001 |
| Gestational age at delivery, weeks | 35.49 (2.60) | 34.80 (2.02) | 0.94 | 0.90–0.99 | <0.001 |
| Pre-pregnancy diabetes | 7 (0.29) | 2 (1.46) | 1.60 | 0.46–5.51 | 0.458 |
-
Results are presented according to the type of variable as mean (SD) or number (%). Odds ratios for BMI (kg/m2), gestational age at delivery (weeks), GWG (kg), and birth weight discordance (%) are presented per unit increase. aOR, adjusted odds ratio; BMI, body mass index; CI, confidence interval; GWG, gestational weight gain; IVF, in vitro fertilization; PE, preeclampsia; SD, standard deviation. aGWG, groups: insufficient, adequate (reference category), excessive [22]. bPre-pregnancy BMI, groups: underweight (<18.5), normal weight (18.5–24.9) (reference category), overweight (25–29.9), obesity (>30). caOR, calculated with adjustment for parity, maternal age, pre-pregnancy BMI, pre-pregnancy hypertension, pre-pregnancy diabetes, insulin treatment during pregnancy; GWG, and gestational age at delivery.
Multivariable analysis
For the purposes of multivariate logistic regression, birthweight discordance (%) was categorized as shown in Table 2, with discordance less than 10 % used as the reference group. Models were adjusted for parity, maternal age, pre-pregnancy BMI, pre-gestational AH, pre-gestational DM, insulin treatment during pregnancy, GWG, and gestational age at delivery.
A birthweight discordance greater than 25 % was identified as a statistically significant independent risk factor for the development of PE (p=0.016). Pregnant women with discordance >25 % had a 1.7-fold higher risk of developing PE (95 % CI 1.10–2.52; p=0.016) compared to those with discordance <10 %, regardless of other risk factors. Discordance between 10 and 25 % was not found to be statistically significant. Other variables independently associated with increased PE risk included chronic hypertension, nulliparity, excessive GWG, obesity, and advanced maternal age. Conversely, the likelihood of PE decreased with increasing gestational age (aOR=0.94; 95 % CI 0.90–0.99; p<0.001).
In a subanalysis, we examined the correlation between gestational age and birth weight discordance. Among women with PE, twin birthweight discordance decreased linearly with advancing gestational age at delivery as shown in Figure 1, indicating that higher discordance was associated with earlier onset of PE and earlier delivery. However, gestational age at birth explained only 8.6 % of the variance in birthweight discordance. In pregnancies without PE, discordance remained constant around 10 %, with a negligible association to gestational age.

Linear regression curves between gestational age at delivery and birth weight discordance in twin pregnancies with (red) and without (blue) preeclampsia.
Prediction of preeclampsia using twin birthweight discordance
We assessed the predictive value of birth weight discordance for identifying PE with the ROC curve shown in Figure 2. Although statistically significant, the AUC was low at 55.2 % (95 % CI 51.6%–58.8 %; p=0.004), indicating limited discriminatory power. At a discordance threshold greater than 25 %, we could identify 10.2 % of PE cases, with a false positive rate of 5.8 %.
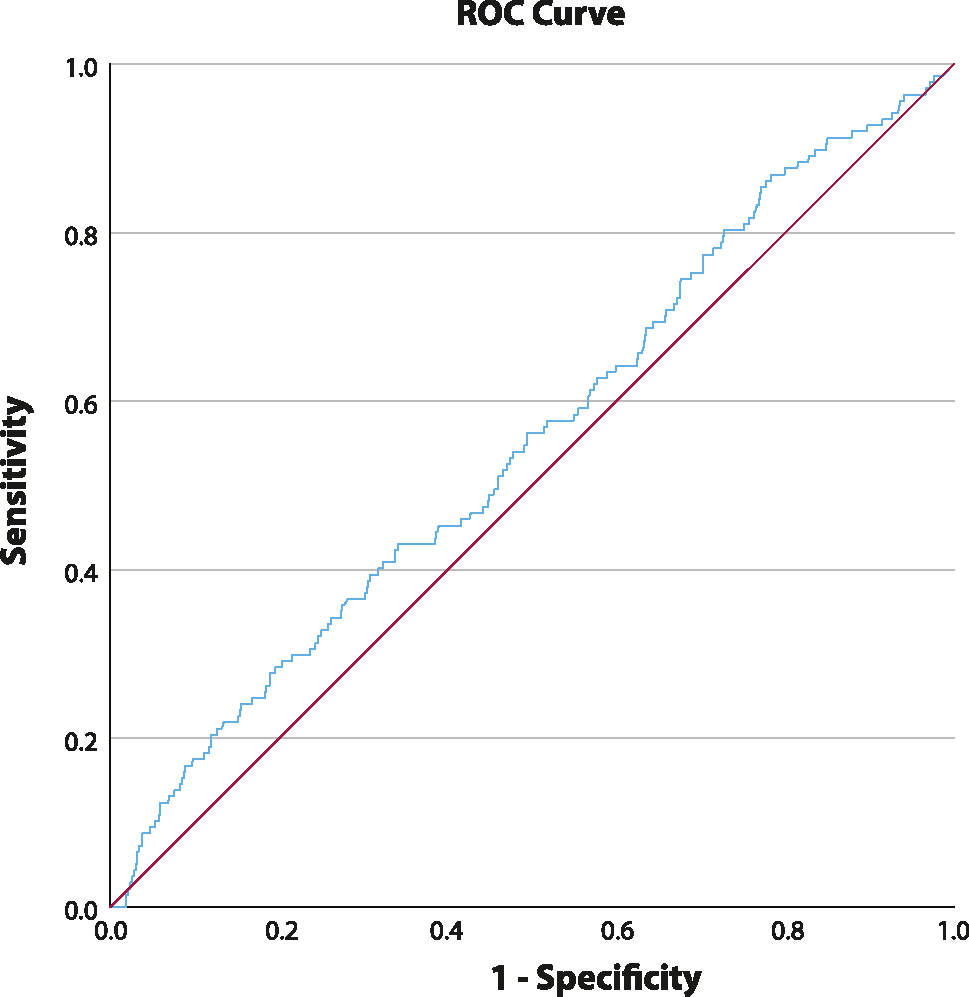
ROC curve for preeclampsia prediction in birth weight discordant dichorionic twins. Area under the curve (AUC)=55.2 % (51.6–58.8 %).
Discussion
In this large population-based cohort study of women with dichorionic twin pregnancies, birthweight discordance greater than 25 % was significantly associated with the development of PE, irrespective of other risk factors. Additionally, the gestational age at delivery increases as birthweight discordance decreases. Using ROC curve analysis to assess the predictive value of various levels of birthweight discordance for PE, we confirmed that discordance can distinguish between pregnancies with and without PE, however, the AUC, although statistically significant, was modest (55.2 %). A discordance greater than 25 % identified only 10.2 % of PE cases with a false-positive rate of 5.8 %. Nevertheless, we confirmed that the >25 % threshold for clinically relevant discordance, previously established through the Delphi consensus method, was appropriate, as this level proved to offer the best balance between sensitivity and specificity.
The incidence of PE in our cohort was 5.38 %, which is lower than the incidence of up to 15 % reported in other studies on dichorionic diamniotic twin pregnancies [23]. Three factors may account for this difference. First, in Slovenia the standardized definition of PE, as specified in this article, is applied rigorously, which contributes to a lower incidence even in singleton pregnancies compared with rates typically reported in international literature. Second, our data represent the entire Slovenian population rather than a selected cohort from one or more tertiary centers that predominantly manage high-risk pregnancies. Consistently, other nationwide Slovenian studies have also reported a PE incidence of approximately 5 % or lower [24], 25]. Third, demographic differences between populations may contribute to variation in PE incidence [26].
The impact of hypertensive disorders during pregnancy, including PE, on the development of discordant twins has been known and confirmed in several studies [27], 28]. However, opinions are divided regarding the influence of birth weight discordance on the development of PE, partly due to varying definitions of clinically significant discordance [5], [6], [7], [8], [9], [10], [11], [12], [13], [14], [15], [16], [17], [18], [19]. In our cohort, all discordances (>10 %) accounted for 45.80 %, while larger discordances (>25 %) represented 6.79 %. The lower incidence of significant discordance compared to some other studies may be explained by the fact that we studied only dichorionic twins. Qiao and Lin et al. reported higher rates (17.6 and 12.1 %, respectively), but their studies also included monochorionic twins, which carry a higher risk of discordance due to a shared placenta [8], 16].
In our study, birth weight discordance among dichorionic twins was significantly associated with PE in univariate analysis. For the multivariate analysis, we categorized birth weight discordance based on NICE guidelines, which recommend more frequent and detailed monitoring of pregnancies with fetal growth discordance over 20 %, and referral to a tertiary obstetric center for discordance over 25 % [29]. We also referenced the study by Khalil et al., which categorizes discordance into <25 % and ≥25 % based on Delphi consensus [30]. After adjusting for other PE risk factors, birth weight discordance >25 % remained statistically significant in our analysis, confirming our hypothesis that discordance in dichorionic twin pregnancies contributes to PE development. Moreover, greater discordance was associated with a higher risk of PE. These findings align with some comparable international studies [7], 8], 13], 31], although others found no significant association [6], 16], 19]. Qiao et al. found an association between discordance and PE only in dichorionic twins, defining discordance as a weight difference >20 % according to ACOG guidelines [8]. Kim and colleagues also observed this association using ultrasound measurements of discordance between the 20th–24th and 28th–32nd weeks of pregnancy, while Maric et al. found no such association in the 11th–14th weeks [7], 19]. It is likely that PE begins to develop early in pregnancy but may not yet affect fetal growth markedly enough to be detected by ultrasound in the first trimester. Even a placenta affected by early PE could still meet the relatively low demands of the fetus during this stage. These findings open opportunities for further research on discordance throughout pregnancy in larger cohorts.
Dai et al. also found a statistically significant association between birth weight discordance >20 % in dichorionic twins and early-onset PE [11]. Unlike our study, they identified IVF as a risk factor for PE, whereas in our univariate logistic regression, this association was not statistically significant (OR=1.3; 95 % CI 0.91–1.88; p=0.154).
Previous studies often link discordance with growth restriction (SGA) in one or both fetuses. In our dichorionic cohort, SGA of one or both fetuses were not statistically significant in univariate analysis (OR=1.25; 95 % CI 0.71–2.21; p=0.443). This contrasts with the findings of Giorgione et al., who identified a significant association between discordance and PE only when growth restriction (SGA or IUGR) was also present [6]. Lin et al. also confirmed the link between SGA and PE, but excluded women with chronic pre-pregnancy hypertension from their study, which may explain the different outcomes [16]. Pre-pregnancy hypertension is a known risk factor for PE, which we also confirmed in our multivariate logistic regression analysis (aOR=10.82; 95 % CI 5.96–19.66; p<0.001) [3].
Our findings are consistent with the study by Zhu et al., which used a restricted cubic spline (RCS) model to demonstrate a nonlinear relationship between birth weight discordance and PE. They categorized discordance into four groups: <15 %, 15–20 %, 20–25 %, and >25 %. After adjusting for other risk factors, only discordance >25 % remained statistically significant and was suggested by the authors as a threshold for intervention [31]. In conclusion, greater discordance in dichorionic twins is an important predictive factor for the development of PE, supporting the need for close monitoring of such pregnancies.
In the multiple logistic regression analysis, we also accounted for the effect of gestational age at delivery, despite its seemingly counterintuitive role, since severe PE often necessitates early iatrogenic delivery. As predicted by the multiple logistic regression, gestational age appears to play a protective role in the development of PE (pOR=0.94; 95 % CI 0.90–0.99; p<0.001). Using linear regression, we demonstrated an inverse correlation between gestational age at delivery and birth weight discordance, indicating that the smaller the discordance, the more advanced the gestational age at delivery. Specifically, each week less of gestation is associated with a 0.17 % increase in birth weight discordance. Therefore, higher discordance at lower gestational ages requires careful monitoring of both the pregnant woman and the fetuses. This association was also reported by Ferrazzani et al. In a single-center retrospective study of 476 twin pregnancies, they found that birth weight discordance greater than 25 % was associated with earlier gestational age at delivery, suggesting that more discordant twins were delivered earlier or required earlier induction of labor. One of the limitations of their study was that PE was only considered when hypertension was accompanied by proteinuria [15].
To assess the predictive value of intertwin birth weight discordance for PE, we constructed a ROC curve representing the trade-off between the sensitivity and specificity of the diagnostic test. Although birth weight discordance alone demonstrated limited discriminatory power between pregnancies complicated by PE and those without, it nonetheless appears to play a significant role in PE development, particularly in the presence of additional maternal risk variables such as nulliparity, advanced maternal age, obesity, pre-existing hypertension, and chronic comorbidities, highlighting the cumulative nature of PE pathogenesis. ROC analysis further validated the 25 % discordance threshold proposed by the Delphi consensus method, at which the majority of surveyed experts would initiate intensified maternal-fetal monitoring. With birth weight discordance above 25 %, we identified 10.2 % of women with PE at a false positive rate of 5.8 %, supporting the Delphi-selected cutoff even in dichorionic twins. While lowering the discordance threshold increases sensitivity, it simultaneously elevates the false-positive rate, potentially leading to unnecessary clinical interventions that may adversely affect fetal outcomes. Therefore, we consider the Delphi method and the 25 % discordance threshold to be appropriate and clinically significant.
A key strength of our study, in comparison to prior research, is the large cohort of pregnant women with dichorionic diamniotic twin pregnancies, covering a longer time span of 10 years. Data were obtained from the NPIS, which is completed by trained medical personnel following standardized protocols for variable entry. Given that NPIS collects information from all 14 Slovenian maternity hospitals, our findings are generalizable to the entire Slovenian population, rather than being limited to a specific region or center. An additional advantage is that our study focused exclusively on dichorionic diamniotic twin pregnancies, thereby minimizing confounding effects related to chorionicity on the pathogenesis of PE. Prior studies examining the impact of chorionicity on PE have yielded inconsistent and conflicting results [5], 8], 32]. The study employed a categorical approach to birth weight discordance, enabling us to examine statistically significant associations with PE incidence within groups defined by different levels of discordance. By refraining from predefining a single cutoff for discordance, we were able to identify the precise threshold that was most strongly associated with PE development. This approach was useful for assessing the clinical relevance of a discordance threshold using the Delphi consensus method.
One of the main limitations of our study is the absence of data on PE in previous pregnancies, including the gestational age at which it occurred, as this information is not available in the NPIS. However, PE in a previous pregnancy has long been recognized as a risk factor for developing PE in a subsequent pregnancy [33], 34]. We cannot exclude the possibility of human and technical error related to the computerized database, which may have unknowingly affected the study results. Despite the large sample size, certain conditions that could influence the occurrence of PE (for example antiphospholipid syndrome, pre-pregnancy kidney disease) were so rare that they could not be included in the statistical analysis [35], 36]. Another limitation is the retrospective observational design of our study, which does not allow for establishing causality. Additionally, NPIS data do not allow differentiation between FGR and SGA fetuses. Therefore, we could not include FGR data in the statistical analysis, which may have influenced the association between birthweight discordance in twins and the incidence of PE, as suggested by the studies of Giorgione et al. and Lin et al. [6], 16]. As mentioned, in our study SGA did not reach the threshold for inclusion in the multivariate analysis in the univariate analysis. Moreover, differences arising from the study cohorts must be considered, as our cohort was based on the entire population. A further limitation is that our study was focused on discordance in birth weights of twins rather than ultrasound-estimated fetal weight at the time of PE onset, which would be more clinically relevant. This highlights the need for further research, particularly prospective studies assessing the value of routine fetal growth monitoring via ultrasound as a screening tool for identifying high-risk pregnancies.
Conclusions
Our study revealed that a birthweight discordance between dichorionic twins >25 % significantly adds to an increased risk of PE, independently of other confounding factors. Birth weight discordance with hypertension, nulliparity, excessive gestational weight gain, obesity, and maternal age as significant independent risk factors may improve the predictive value for the development of PE in dichorionic twins. Conversely, the likelihood of developing PE decreased with increasing gestational age. As the growth of dichorionic twins is routinely monitored by ultrasound, an intertwin growth discordance exceeding 25 % may serve as an early indicator of the potential development of PE. Therefore, understanding the risk of developing PE in discordant dichorionic twin pregnancy can help in prenatal monitoring, diagnostics and intervention. Combined with other risk factors, this can significantly impact the incidence of PE-related complications and lead to a better perinatal outcome. Given the current paucity and inconsistency of research in this area, our results contribute meaningfully to clinical knowledge. Understanding the role of birthweight discordance provides a specific and actionable target for preventive strategies aimed at mitigating the risk of PE in twin pregnancies.
-
Research ethics: The research was approved by the National Medical Ethics Committee of the Republic of Slovenia on January, 16th, 2024 (No. 0120–559/2023/3).
-
Informed consent: Not applicable.
-
Author contributions: TPS conceived the study. IV contributed to data collection. SČ, AR TPS and IV contributed to data analysis. SČ and AR wrote the first version of the manuscript. TPS and IV contributed to manuscript critical revision. All the authors have accepted responsibility for the entire content of this submitted manuscript and approved submission.
-
Use of Large Language Models, AI and Machine Learning Tools: None declared.
-
Conflict of interest: The authors state no conflict of interest.
-
Research funding: None declared.
-
Data availability: The research data are stored at the Research Department of Division of Obstetrics and Gynecology, UMC Ljubljana and available upon reasonable request.
References
1. Wu, P, Green, M, Myers, JE. Hypertensive disorders of pregnancy. BMJ 2023;381:e071653. https://doi.org/10.1136/bmj-2022-071653.Search in Google Scholar PubMed
2. Say, L, Chou, D, Gemmill, A, Tunçalp, Ö, Moller, AB, Daniels, J, et al.. Global causes of maternal death: a WHO systematic analysis. Lancet Glob Health 2014;2:e323–333. https://doi.org/10.1016/s2214-109x(14)70227-x.Search in Google Scholar PubMed
3. Gestational Hypertension and Preeclampsia: ACOG Practice Bulletin. Number 222. Obstet Gynecol 2020;135:e237–60.10.1097/AOG.0000000000003891Search in Google Scholar PubMed
4. Premru-Srsen, T, Kocic, Z, Fabjan Vodusek, V, Geršak, K, Verdenik, I. Total gestational weight gain and the risk of preeclampsia by pre-pregnancy body mass index categories: a population-based cohort study from 2013 to 2017. J Perinat Med 2019;47:585–91. https://doi.org/10.1515/jpm-2019-0008.Search in Google Scholar PubMed
5. Chantanahom, N, Phupong, V. Clinical risk factors for preeclampsia in twin pregnancies. PLoS ONE 2021;16:e0249555. https://doi.org/10.1371/journal.pone.0249555.Search in Google Scholar PubMed PubMed Central
6. Giorgione, V, Bhide, A, Bhate, R, Reed, K, Khalil, A. Are twin pregnancies complicated by weight discordance or fetal growth restriction at higher risk of preeclampsia? J Clin Med 2020;9:3276. https://doi.org/10.3390/jcm9103276.Search in Google Scholar PubMed PubMed Central
7. Kim, HM, Cha, HH, Seong, WJ, Lee, HJ, Kim, MJ. Prediction of maternal complications and neonatal outcome in dichorionic diamniotic twins with fetal weight discordancy measured by ultrasonography. Sci Rep 2022;12:14889. https://doi.org/10.1038/s41598-022-18864-8.Search in Google Scholar PubMed PubMed Central
8. Qiao, P, Zhao, Y, Jiang, X, Xu, C, Yang, Y, Bao, Y, et al.. Impact of growth discordance in twins on preeclampsia based on chorionicity. Am J Obstet Gynecol 2020;223:572.e1–8. https://doi.org/10.1016/j.ajog.2020.03.024.Search in Google Scholar PubMed
9. Sparks, TN, Cheng, YW, Phan, N, Caughey, AB. Does risk of preeclampsia differ by twin chorionicity? J Matern Fetal Neonatal Med 2013;26:1273–7. https://doi.org/10.3109/14767058.2013.777701.Search in Google Scholar PubMed
10. Sarno, L, Maruotti, GM, Donadono, V, Saccone, G, Martinelli, P. Risk of preeclampsia: comparison between dichorionic and monochorionic twin pregnancies. J Matern Fetal Neonatal Med 2014;27:1080–1. https://doi.org/10.3109/14767058.2013.847414.Search in Google Scholar PubMed
11. Dai, F, Pan, S, Lan, Y, Tan, H, Li, J, Hua, Y. Pregnancy outcomes and risk factors for preeclampsia in dichorionic twin pregnancies after in vitro fertilization: a five-year retrospective study. BMC Pregnancy Childbirth 2022;22:830. https://doi.org/10.1186/s12884-022-05184-y.Search in Google Scholar PubMed PubMed Central
12. Kent, EM, Breathnach, FM, Gillan, JE, McAuliffe, FM, Geary, MP, Daly, S, et al.. Placental pathology, birthweight discordance, and growth restriction in twin pregnancy: results of the ESPRiT Study. Am J Obstet Gynecol 2012;207:220.e1–5. https://doi.org/10.1016/j.ajog.2012.06.022.Search in Google Scholar PubMed
13. Chen, W, Li, Z, Wang, D, Wang, Z. The association between neonatal birthweight discordance and preeclampsia in twin pregnancy. J Obstet Gynaecol Res 2024;50:961–9. https://doi.org/10.1111/jog.15937.Search in Google Scholar PubMed
14. Jahanfar, S, Lim, K. Adverse maternal outcomes and birth weight discordance in twin gestation: British Columbia, Canadian data. Int J Womens Health 2017;9:871–8. https://doi.org/10.2147/ijwh.s115515.Search in Google Scholar PubMed PubMed Central
15. Ferrazzani, S, Merola, A, De Carolis, S, Carducci, B, Paradisi, G, Caruso, A. Birth weight in pre-eclamptic and normotensive twin pregnancies: an analysis of discordance and growth restriction. Hum Reprod Oxf Engl 2000;15:210–7. https://doi.org/10.1093/humrep/15.1.210.Search in Google Scholar PubMed
16. Lin, D, Luo, BC, Chen, G, Fan, D, Huang, Z, Li, P, et al.. The association of hypertensive disorders of pregnancy with small for gestational age and intertwin birthweight discordance. J Clin Hypertens Greenwich Conn 2021;23:1354–62.10.1111/jch.14257Search in Google Scholar PubMed PubMed Central
17. Fox, NS, Saltzman, DH, Oppal, S, Klauser, CK, Gupta, S, Rebarber, A. The relationship between preeclampsia and intrauterine growth restriction in twin pregnancies. Am J Obstet Gynecol 2014;211:422.e1–5. https://doi.org/10.1016/j.ajog.2014.05.035.Search in Google Scholar PubMed
18. Hollier, LM, McIntire, DD, Leveno, KJ. Outcome of twin pregnancies according to intrapair birth weight differences. Obstet Gynecol 1999;94:1006–10. https://doi.org/10.1097/00006250-199912000-00019.Search in Google Scholar
19. Maric, T, Singh, N, Duncan, K, Thorpe-Beeston, GJ, Savvidou, MD. First-trimester fetal growth discordance and development of preeclampsia in dichorionic twin pregnancies. J Perinat Med 2015;43:755–9. https://doi.org/10.1515/jpm-2013-0260.Search in Google Scholar PubMed
20. Babnik, J, Blejec, T, Cetin-Lovšin, I, Lovšin, B, Lučovnik, M, Kavšek, G, et al.. Perinatal information system (PIS): definitions and methodology guidelines for receiving data via e-transfer [Online]. NIJZ; 2019. https://nijz.si/wp-content/uploads/2021/12/peris-metodoloska-navodila-2020-v2-2.pdf [Accessed 28 Jul 2024].Search in Google Scholar
21. Melamed, N, Baschat, A, Yinon, Y, Athanasiadis, A, Mecacci, F, Figueras, F, idr FIGO, (international Federation of Gynecology and obstetrics) initiative on fetal growth: best practice advice for screening, diagnosis, and management of fetal growth restriction. Int J Gynaecol Obstet Off Organ Int Fed Gynaecol Obstet 2021;152:3–57. https://doi.org/10.1002/ijgo.13522.Search in Google Scholar PubMed PubMed Central
22. American College of Obstetricians and Gynecologists. ACOG Committee opinion no. 548: weight gain during pregnancy. Obstet Gynecol 2013;121:210–2. https://doi.org/10.1097/01.aog.0000425668.87506.4c.Search in Google Scholar PubMed
23. Lyu, S, Gao, L, Sun, J, Zhao, X, Wu, Y, Hua, R, idr. The association between maternal complications and small for gestational age in twin pregnancies using singleton and twin birth weight references. J Matern-Fetal Neonatal Med Off J Eur Assoc Perinat Med Fed Asia Ocean Perinat Soc Int Soc Perinat Obstet 2022;35:7459–65. https://doi.org/10.1080/14767058.2021.1949703.Search in Google Scholar PubMed
24. Lučovnik, M, Tul, N, Verdenik, I, Novak, Z, Blickstein, I. Risk factors for preeclampsia in twin pregnancies: a population-based matched case-control study. J Perinat Med 2012;40:379–82. https://doi.org/10.1515/jpm-2011-0252.Search in Google Scholar PubMed
25. Lučovnik, M, Blickstein, I, Verdenik, I, Steblovnik, L, Trojner Bregar, A, Tul, N. Impact of pre-gravid body mass index and body mass index change on preeclampsia and gestational diabetes in singleton and twin pregnancies. J Matern-Fetal Neonatal Med Off J Eur Assoc Perinat Med Fed Asia Ocean Perinat Soc Int Soc Perinat Obstet 2014;27:1901–4. https://doi.org/10.3109/14767058.2014.892069.Search in Google Scholar PubMed
26. Fasanya, HO, Hsiao, CJ, Armstrong-Sylvester, KR, Beal, SG. A critical review on the use of race in understanding racial disparities in preeclampsia. J Appl Lab Med 2021;6:247–56. https://doi.org/10.1093/jalm/jfaa149.Search in Google Scholar PubMed PubMed Central
27. Sparks, TN, Nakagawa, S, Gonzalez, JM. Hypertension in dichorionic twin gestations: how is birthweight affected? J Matern-Fetal Neonatal Med Off J Eur Assoc Perinat Med Fed Asia Ocean Perinat Soc Int Soc Perinat Obstet. 2017;30:380–5. https://doi.org/10.3109/14767058.2016.1174209.Search in Google Scholar PubMed
28. Wang, Y, Zeng, H, Liu, J, Zhang, F. Gestational hypertensive disease and birthweight discordance in twin pregnancies: a systematic review and meta-analysis. J Matern-Fetal Neonatal Med Off J Eur Assoc Perinat Med Fed Asia Ocean Perinat Soc Int Soc Perinat Obstet. 2022;35:8869–77. https://doi.org/10.1080/14767058.2021.2005572.Search in Google Scholar PubMed
29. Twin and triplet pregnancy [Online]. London: National Institute for Health and Care Excellence (NICE); 2024. (National Institute for Health and Care Excellence: Guidelines). http://www.ncbi.nlm.nih.gov/books/NBK546070/ [Accessed 29. Jan 2025].Search in Google Scholar
30. Khalil, A, Beune, I, Hecher, K, Wynia, K, Ganzevoort, W, Reed, K, et al.. Consensus definition and essential reporting parameters of selective fetal growth restriction in twin pregnancy: a Delphi procedure. Ultrasound Obstet Gynecol Off J Int Soc Ultrasound Obstet Gynecol 2019;53:47–54. https://doi.org/10.1002/uog.19013.Search in Google Scholar PubMed
31. Zhu, J, An, P, Zhao, H, Zhao, Y, Zhou, J, Zhou, Q, et al.. Optimal cutoffs of growth discordance for the risk of preeclampsia in twin pregnancies: a single-center retrospective cohort study. Front Cardiovasc Med [Online] 2023;9. https://www.frontiersin.org/journals/cardiovascular-medicine/articles/10.3389/fcvm.2022.1073729/full [Accessed 14 Aug 2024].10.3389/fcvm.2022.1073729Search in Google Scholar PubMed PubMed Central
32. Chen, W, Li, Z, Wang, D, Wang, Z. The association between neonatal birthweight discordance and preeclampsia in twin pregnancy. J Obstet Gynaecol Res 2024;50:961–9. https://doi.org/10.1111/jog.15937.Search in Google Scholar PubMed
33. Dildy, GA, Belfort, MA, Smulian, JC. Preeclampsia recurrence and prevention. Semin Perinatol 2007;31:135–41. https://doi.org/10.1053/j.semperi.2007.03.005.Search in Google Scholar PubMed
34. Bernardes, TP, Mol, BW, Ravelli, ACJ, van den Berg, PP, Boezen, HM, Groen, H. Recurrence risk of preeclampsia in a linked population-based cohort: effects of first pregnancy maximum diastolic blood pressure and gestational age. Pregnancy Hypertens 2019;15:32–6. https://doi.org/10.1016/j.preghy.2018.10.010.Search in Google Scholar PubMed
35. Al Khalaf, S, Bodunde, E, Maher, GM, O’Reilly, ÉJ, McCarthy, FP, O’Shaughnessy, MM, et al.. Chronic kidney disease and adverse pregnancy outcomes: a systematic review and meta-analysis. Am J Obstet Gynecol 2022;226:656–70.e32. https://doi.org/10.1016/j.ajog.2021.10.037.Search in Google Scholar PubMed
36. D’Ippolito, S, Barbaro, G, Paciullo, C, Tersigni, C, Scambia, G, Di Simone, N. Antiphospholipid syndrome in pregnancy: new and old pathogenetic mechanisms. Int J Mol Sci 2023;24:3195. https://doi.org/10.3390/ijms24043195.Search in Google Scholar PubMed PubMed Central
© 2025 the author(s), published by De Gruyter, Berlin/Boston
This work is licensed under the Creative Commons Attribution 4.0 International License.
Articles in the same Issue
- Frontmatter
- Editorial
- The fetus as a patient in the 21st century: science, ethics, technology and global responsibility
- Vision, Education, and the Future of Perinatal Medicine
- Opening – field direction, education, and AI
- Quo vadis neonatologia? Where is neonatology heading in the 21st century?
- Shaping the future: advancing maternal-fetal medicine through educational standards and innovations
- Integrating generative AI in perinatology: applications for literature review
- Maternal Hemodynamics, Fetal Physiology, and Surveillance
- Core fetal physiology and maternal-fetal interaction
- Cardiac output-guided maternal positioning may protect the fetal oxygen supply and thereby reduce pregnancy complications
- Effect of antenatal betamethasone on fetal heart rate short-term variability in growth restricted fetuses
- Umbilical venous flow and maternal hemodynamics as predictors of impaired fetal growth in gestational diabetes: a prospective study
- The impact of maternal cardiovascular status prior to labor on birth outcomes: an observational study
- Complex Pregnancies, Placenta, and Fetal Therapy
- Twins, placental disease, fetal intervention, and periviability
- Complications in monochorionic twin pregnancies
- Association of discordance in birth weights of dichorionic twins with the incidence of preeclampsia in pregnant women
- Management and outcomes of periviable infants in Slovenia: a decade of experience
- Successful management of severe hemolytic disease of the fetus and newborn (HDFN) due to anti-Kell
- Systems of Care, Screening, and Population-Level Perinatal Medicine
- Public health, structured care, and national data
- Structured stillbirth management in Slovenia: outcomes and comparison with international guidelines
- Newborn screening for rare diseases: expanding the paradigm in the genomic era
- Ten years of experience with screening for diabetes in pregnancy according to IADPSG criteria in Slovenia
- Gestational diabetes and fetal macrosomia: a dissenting opinion
- Advanced Prenatal Diagnosis
- Imaging and fetal anomaly detectionata
- Detection of isolated fetal limb anomalies using 3D/4D ultrasound
- Ethics, Professional Responsibility, and Patient-Centered Counseling
- The moral and communicative core of “Fetus as a Patient”
- The fetus as a patient: professional responsibility in contemporary Perinatal Medicine
- Placenta-oriented counseling: challenges and opportunities in obstetric practice
- Patient education materials: improving readability to advance health equity
- Global Health, Pandemic, and Humanitarian Perinatal Medicine
- COVID-19, war, immunity, and respectful care
- Clinical factors in SARS-CoV-2 antibody response in unvaccinated mothers
- Serum vitamin D and inflammatory markers in SARS-CoV-2 positive pregnant women
- Perceptions of respectful maternity care in Ukraine during a time of war
- Role of prelabour midwifery consultation in enhancing maternal satisfaction and preparedness for birth
- Obstetric Decision-Making and Postpartum Outcomes
- Clinical controversies and maternal outcomes
- Should we conduct a trial of labor in women with a macrosomic fetus?
- Postpartum maternal complications: a retrospective single-center study
- Annual Reviewer Acknowledgment
- Reviewer Acknowledgment
Articles in the same Issue
- Frontmatter
- Editorial
- The fetus as a patient in the 21st century: science, ethics, technology and global responsibility
- Vision, Education, and the Future of Perinatal Medicine
- Opening – field direction, education, and AI
- Quo vadis neonatologia? Where is neonatology heading in the 21st century?
- Shaping the future: advancing maternal-fetal medicine through educational standards and innovations
- Integrating generative AI in perinatology: applications for literature review
- Maternal Hemodynamics, Fetal Physiology, and Surveillance
- Core fetal physiology and maternal-fetal interaction
- Cardiac output-guided maternal positioning may protect the fetal oxygen supply and thereby reduce pregnancy complications
- Effect of antenatal betamethasone on fetal heart rate short-term variability in growth restricted fetuses
- Umbilical venous flow and maternal hemodynamics as predictors of impaired fetal growth in gestational diabetes: a prospective study
- The impact of maternal cardiovascular status prior to labor on birth outcomes: an observational study
- Complex Pregnancies, Placenta, and Fetal Therapy
- Twins, placental disease, fetal intervention, and periviability
- Complications in monochorionic twin pregnancies
- Association of discordance in birth weights of dichorionic twins with the incidence of preeclampsia in pregnant women
- Management and outcomes of periviable infants in Slovenia: a decade of experience
- Successful management of severe hemolytic disease of the fetus and newborn (HDFN) due to anti-Kell
- Systems of Care, Screening, and Population-Level Perinatal Medicine
- Public health, structured care, and national data
- Structured stillbirth management in Slovenia: outcomes and comparison with international guidelines
- Newborn screening for rare diseases: expanding the paradigm in the genomic era
- Ten years of experience with screening for diabetes in pregnancy according to IADPSG criteria in Slovenia
- Gestational diabetes and fetal macrosomia: a dissenting opinion
- Advanced Prenatal Diagnosis
- Imaging and fetal anomaly detectionata
- Detection of isolated fetal limb anomalies using 3D/4D ultrasound
- Ethics, Professional Responsibility, and Patient-Centered Counseling
- The moral and communicative core of “Fetus as a Patient”
- The fetus as a patient: professional responsibility in contemporary Perinatal Medicine
- Placenta-oriented counseling: challenges and opportunities in obstetric practice
- Patient education materials: improving readability to advance health equity
- Global Health, Pandemic, and Humanitarian Perinatal Medicine
- COVID-19, war, immunity, and respectful care
- Clinical factors in SARS-CoV-2 antibody response in unvaccinated mothers
- Serum vitamin D and inflammatory markers in SARS-CoV-2 positive pregnant women
- Perceptions of respectful maternity care in Ukraine during a time of war
- Role of prelabour midwifery consultation in enhancing maternal satisfaction and preparedness for birth
- Obstetric Decision-Making and Postpartum Outcomes
- Clinical controversies and maternal outcomes
- Should we conduct a trial of labor in women with a macrosomic fetus?
- Postpartum maternal complications: a retrospective single-center study
- Annual Reviewer Acknowledgment
- Reviewer Acknowledgment

