Abstract
Objectives
The original French version of the “Douleur Neuropathique en 4 Questions (DN4) questionnaire” is a valid screening tool for the identification of neuropathic pain (NeP). The DN4 has been translated into English, but the reliability of the English version has not yet been investigated. The aim of this study was to investigate the 7-item DN4 questionnaire in regards to short-term reliability before (T0) and immediately after (T1) the clinical examination and long-term reliability one week later (T2).
Methods
A total of 222 participants (age 56.33 ± 16 years, 56 % female) were recruited from a Pain Management Department and Neurosurgery Spinal Clinic. For T2 measurements, the 7-item DN4 was sent by post with the “Patient Global Impression of Change Scale”. The scale detects possible changes of symptoms, scoring from “very much improved” (1) to “very much worse” (7). Only participants whose symptoms had not changed much (scores 3–5) were included in the T0-T2 analysis. Weighted Kappa was used to analyse the reliability of the DN4 total scores and unweighted Kappa for the DN4 classifications.
Results
Considering missing data and exclusions, data of 215 participants could be used for the T0-T1 and data of 103 participants for T0-T2 analysis. There was almost perfect agreement for the 7-item DN4 total score between T0-T1 (weighted k: 0.891, CI: 0.758–1.024) and T0-T2 (weighted k: 0.850, CI: 0.657–1.043). Classifications between neuropathic pain and no neuropathic pain showed almost perfect agreement (k: 0.835, CI: 0.755–0.915) for T0-T1 and substantial agreement (k: 0.733, CI: 0.598–0.868) for T0-T2.
Conclusions
The English 7-item DN4 is a reliable screening tool for neuropathic pain.
Ethical committee number
#RGS0000001759.
Introduction
The substantial burden of neuropathic pain (NeP), defined as “pain caused by a lesion or disease of the somatosensory system” [1], on patients’ health related quality of life, function and psychological wellness as well as on health care costs has been well documented [2, 3]. Early identification of NeP is crucial for timely and targeted evidence-based treatment. Numerous screening tools have been developed to assist clinicians in the identification of NeP, including the “Douleur Neuropathique en 4 Questions (DN4) questionnaire” [4].
The DN4 was originally developed in a French patient cohort with nociceptive and neuropathic pain. The questionnaire exists in two versions: The 10-item DN4 contains both sensory descriptors and signs related to a bedside sensory examination, the 7-item DN4 contains just sensory descriptors [4] and has been used as a self-reported screening tool [5]. The 7-item DN4 demonstrated a sensitivity of 78 % and specificity of 81.2 % in the identification of NeP [4] which was lower compared to the sensitivity (82.9 %) and specificity (89.9 %) of the 10-item DN4 (4). The original study did not assess the 7-item DN4’s test–retest reliability.
The DN4 questionnaire has been translated into numerous languages and validated: Thai [6], Spanish [7], Dutch [8], [9], [10], Portuguese [11], Turkish [12], Arabic [13, 14], Swedish [15], Italian [16], Persian [17], Greek [18], Korean [19], Hindi [20], Japanese [21] and Taiwanese [22]. For the 7-item DN4, sensitivity ranged from 70 to 97 % and specificity from 67 to 84 % [9, 10, 13, 14, 16, 20, 23], the reliability ranged from 0.79 to 0.97 (ICC) [10, 13, 20]. An English version of the 7-item DN4 exists, however its reliability and validity have never been explored.
The aim of this study was to investigate the reliability of the English version of the 7-item DN4 pre- and post-clinical consultation and at one week follow up. The reliability of the total DN4 score and the DN4 classifications (NeP, no NeP) as well as the reliability of each single item were examined, consistent with previous methodologies [4, 24].
Materials and methods
The study involved repeated measures to assess the short- and long-term reliability of the 7-item DN4 questionnaire at three time points. For the short-term reliability the questionnaire was filled out before (T0) and immediately after the clinical consultation (T1) and one week later (T2) for the long-term reliability.
Study population
Data collection took place from March to May 2019 at Sir Charles Gairdner Hospital in Perth, Australia. Patients presenting to the Pain Management Department and Neurosurgery Spinal Clinic were recruited. The inclusion criteria were the presence of pain and age ≥18 years. Patients were excluded if they were unable to read, write or speak English, had impaired vision, mental health illness like dementia and if they received pain interventional procedures such as injections on the day of recruitment. All participants were informed about the study protocol prior to giving written consent. The study was approved by the institutional ethics committee (#RGS0000001759) and adhered to the Declaration of Helsinki.
Sample size
A minimum of n=101 subjects has 80 % power to detect kappa=0.9 from a null value of 0.7, assuming p=0.5 positive ratings [25]. We anticipated to recruit around 200 patients considering a dropout rate of at least 30 % [24].
Measurement tools
Douleur Neuropatique en 4 Questions
The 7-item DN4 questionnaire consists of two questions. The first question asks if the pain has one or more of the following characteristics: the quality of burning, painful cold and electric shocks. The second question asks if the pain is associated with one or more of the following symptoms in the same area: the sensations of tingling, pins and needles, numbness and itching. Patients are asked to tick one answer, “yes” or “no” for each item in the two questions.
The final score of the DN4 is calculated with scoring 1 point for each positive (“yes”) answer. Results range between a minimum of 0 and a maximum of 7 points. As the English version of the 7-item DN4 had not yet been validated, we used the cut-off score of 3 from the original French study [4]. A total score of 3 or above is classified as having NeP and scores <3 are classified as having no NeP.
Patient Global Impression of Change Scale
The Patient Global Impression of Change (PGIC) Scale was used for the detection of changes in the participants’ pain condition between T0 and T2 [24, 26]. The PGIC is commonly used in clinical research for measuring a person’s own impression of change occurring over time [24, 26]. Patients rated any change on the numeric 7-point scale between “very much improved” (1) and “very much worse” (7). No change is stated with “4”. Participants scoring <3 or >5 were excluded from our T0-T2 statistical analysis as this indicated a change in symptoms.
Study protocol
Participants completed the DN4 before seeing the clinician (T0) and immediately after their consultation (T1) as well as one week afterwards (T2). Based on our observation that patients reported multiple pain areas, we asked them to answer the questionnaire in regards to their main pain area. Participants were reminded that a copy of the DN4 as well as the PGIC, together with a pre-paid postage envelope would be sent via registered post. Participants were instructed to complete the DN4 and PGIC seven days after their consultation and to return them via post. Documents were dispatched and at time point T2 participants were contacted by phone to remind them to complete the questionnaires. A maximum of two phone calls was conducted.
Missing data
Participants who failed to complete the DN4 at T0 were excluded from the study. If they failed only at time point T1 they were still included for the statistical analysis of long-term reliability (T0-T2), as long as they were not excluded due to changes indicated in the PGIC (<3 or >5). If they failed to complete the DN4 only at time point T2, they were still included for statistical analysis of the short-term reliability (T0-T1). Failures at filling out the questionnaire could have been not ticking a box or ticking two boxes (yes and no).
Statistical analysis
Data analysis was performed using SPSS vs. 26. Means with standard deviations were calculated for the DN4 total scores. Paired t-tests were performed to detect statistically significant differences between the means of total scores between the different time points.
Weighted kappa was used to analyze agreement of the total scores between T0-T1 and T0-T2. Unweighted kappa was calculated for the agreement in classifying patients into having NeP or no NeP and for the agreement of the seven single items which were binary variables (yes or no). Kappa values between 0.41 and 0.60 indicate moderate agreement, values between 0.61 and 0.80 indicate a substantial agreement and values between 0.81 and 1.00 indicate an almost perfect agreement [27]. For all kappa values, 95 % confidence intervals (CIs) were calculated. Bland-Altman plots were used to graphically investigate the agreement of measures taken on the same individual at two different time points and to assess the presence of possible systematic bias.
Results
Characteristics of study population
Two hundred and twenty-three participants were enrolled in the study. One participant could not be included because he failed to fill out the questionnaire correctly at time point T0. Accordingly, information on 222 participants (age range 18–90 years; 56 % female) was available, i.e. 222 participants had questionnaires included in at least one reliability analysis. The demographic and clinical characteristics for the cohort are shown in Table 1.
Clinical and demographic characteristics of the study population.
| Age in years, mean, SD | 56.3 (15.8) |
| Gender, n female/n male, % | 124 (55.9 %)/98 (44.1 %) |
| Pain duration in years (n=210), mean (SD) | 10.48 (10.8) |
| Main pain area, n, % | |
| Head | 6 (2.7) |
| Neck | 27 (12.2) |
| Back | 104 (46.8) |
| Upper limb | 29 (13.1) |
| Lower limb | 41 (18.5) |
| Other | 15 (6.8) |
| Clinical diagnosis, n, %a | |
| Stenosis (spinal canal, foraminal) | 64 (28.8) |
| Other changes of the spine | 24 (10.8) |
| Disc prolapse/protrusion | 24 (10.8) |
| Chronic back pain | 28 (12.6) |
| Inflammatory joint diseases | 33 (14.9) |
| Fibromyalgia | 7 (3.2) |
| Fractures | 7 (3.2) |
| Complex regional pain syndrome | 4 (1.8) |
| Post op | 6 (2.7) |
| Trauma | 7 (3.2) |
| Oncological diseases | 5 (2.3) |
| Otherb | 34 (15.3) |
| Unknown | 5 (2.3) |
| Pain medication, n, % | 181 (81.5) |
| Daily | 147 (66.2) |
| As required | 34 (15.3) |
-
SD, standard deviation; op, operation. aTotal numbers are greater than the total number of participants as some participants have more than one diagnosis. bOther includes: Crohn’s disease, diabetes mellitus, rare syndromes, neuropathic pain, central sensitization.
The mean symptom duration was 10.5 (±10.8) years, ranging from six weeks to 61 years. The participants’ main pain areas and clinical diagnoses are documented in Table 1. One hundred and eighty-one (82 %) participants reported the presence of multiple pain areas. The intake of pain medication was recorded, however for the purpose of the study, the specific name and dosage of medication were not documented.
T0-T1 reliability
Two hundred and twenty-two participants completed the DN4 at T0, of whom 215 (94.8 %) also completed the questionnaire correctly at T1. Hence, 215 were included in the T0-T1 analysis. There was a statistically significant difference between the mean total DN4 score at T0 (3.7 ± 1.9) and T1 (3.4 ± 2.0) (p<0.001) (Table 2). The weighted kappa of the DN4 total scores between T0 and T1 was 0.891 (95 % CI: 0.758–1.024). Figure 1 displays a Bland-Altman plot for the total DN4 score at T0 and T1. As the limits of agreement (−1.540 and 1.987) contain zero, there is no obvious discrepancy in agreement of the two measures and no evidence of systematic bias. Three outliers were apparent, showing a reduction in the DN4 score from T0-T1. One patient demonstrated a reduction from 6 to 0 points, and the two other outliers a reduction of 4 points (4–0 and 5–1).
7-Item DN4 scores (mean/SD) and classifications at each time-point for patients included in the baseline (T0) to immediately post-consultation (T1) comparison, and baseline (T0) to one-week follow up (T2) comparison.
| T0-T1 (n=215) | p-Value | T0-T2 (n=103) | p-Value | |||
|---|---|---|---|---|---|---|
| T0 | T1 | T0 | T2 | |||
| DN4 total score | 3.7 (1.9) | 3.4 (2.0) | <0.001 | 3.4 (2.1) | 3.3 (2.2) | 0.350 |
| DN4 classification, n, % | ||||||
| Neuropathic pain | 152 (71) | 147 (68) | 0.600 | 65 (63) | 62 (60) | 0.185 |
| No neuropathic pain | 63 (29) | 68 (32) | 38 (37) | 41 (40) | ||
-
DN4, Douleur Neuropathique en 4 Questions; SD, standard deviation.
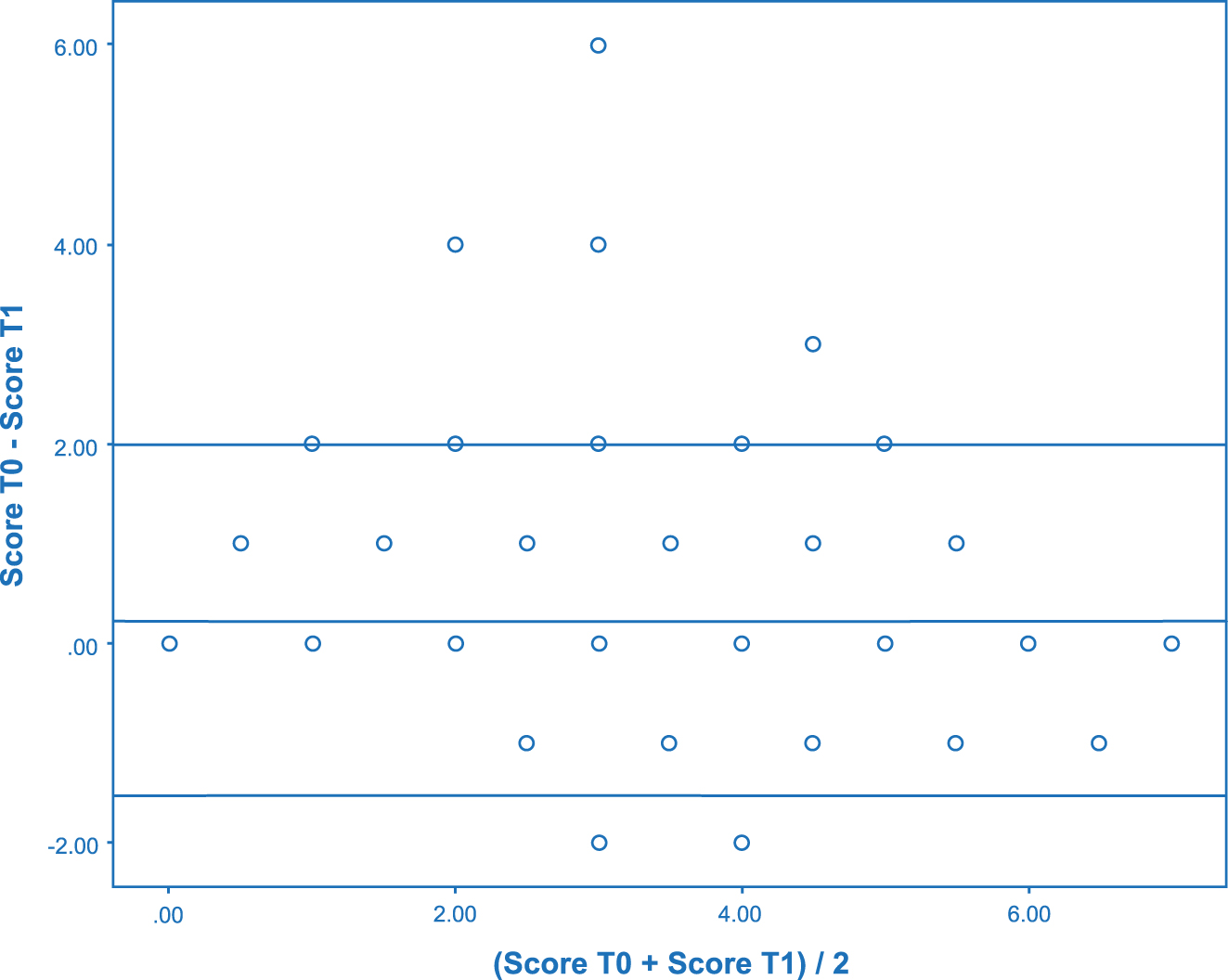
Bland-Altmann plot for total DN4 scores at T0-T1.
Based on the cut-off score of 3, the DN4 classified 152 patients as having NeP and 63 patients as not having NeP at T0 (Table 2). At T1, 147 patients were identified as having NeP and 68 as not having NeP. The DN4 classifications from T0 and T1 demonstrated a weighted kappa score of 0.835 (95 % CI: 0.755–0.915) (Table 3). For individual DN4 items, weighted kappa values ranged between 0.767 (95 % CI: 0.667–0.867) and 0.835 (95 % CI: 0.759–0.911) (Table 3).
Agreement estimates (kappa, weighted kappa) and 95 % confidence intervals (CIs) for DN4 total scores, classifications and individual items at time-point comparisons of T0-T1 and T0-T2.
| T0-T1 (n=215) | T0-T2 (n=103) | |
|---|---|---|
| Kappa (95 % CI) | Kappa (95 % CI) | |
| DN4 total scorea | 0.891 (0.758–1.024) | 0.850 (0.657–1.043) |
| DN4 classificationsb | 0.835 (0.755–0.915) | 0.733 (0.598–0.868) |
| Burningb | 0.835 (0.759–0.911) | 0.714 (0.575–0.853) |
| Painful coldb | 0.767 (0.677–0.867) | 0.651 (0.490–0.812) |
| Electric shocksb | 0.795 (0.715–0.876) | 0.784 (0.664–0.904) |
| Tinglingb | 0.798 (0.714–0.882) | 0.657 (0.508–0.806) |
| Pins and needlesb | 0.809 (0.731–0.887) | 0.582 (0.423–0.741) |
| Numbnessb | 0.811 (0.731–0.891) | 0.722 (0.587–0.857) |
| Itchingb | 0.807 (0.717–0.897) | 0.706 (0.543–0.869) |
-
aWeighted kappa. bUnweighted kappa.
T0-T2 reliability
One hundred and thirty-two participants completed the DN4 at T2. After excluding 29 participants due to missing data or a PGIC score of <3 or >5, a total of 103 participants were included in the T0-T2 analysis. There was no statistically significant difference between the mean total DN4 score at T0 (3.4 ± 2.1) and T2 (3.3 ± 2.2) (p=0.350) (Table 2). The weighted kappa of the DN4 total scores between T0 and T2 was 0.850 (95 % CI: 0.657–1.043) (Table 3). Figure 2 displays a Bland-Altman plot for the total DN4 score at T0 and T2. As the limits of agreement (−2.155 and 2.369) contain zero, there is no obvious discrepancy in agreement of the two measures and no evidence of systematic bias. There were four outliers evident. Two patients showed a reduction in the DN4 score (4–0 and 7–4) between T0 and T2 and two showed a score increase (2–5 and 4–7).
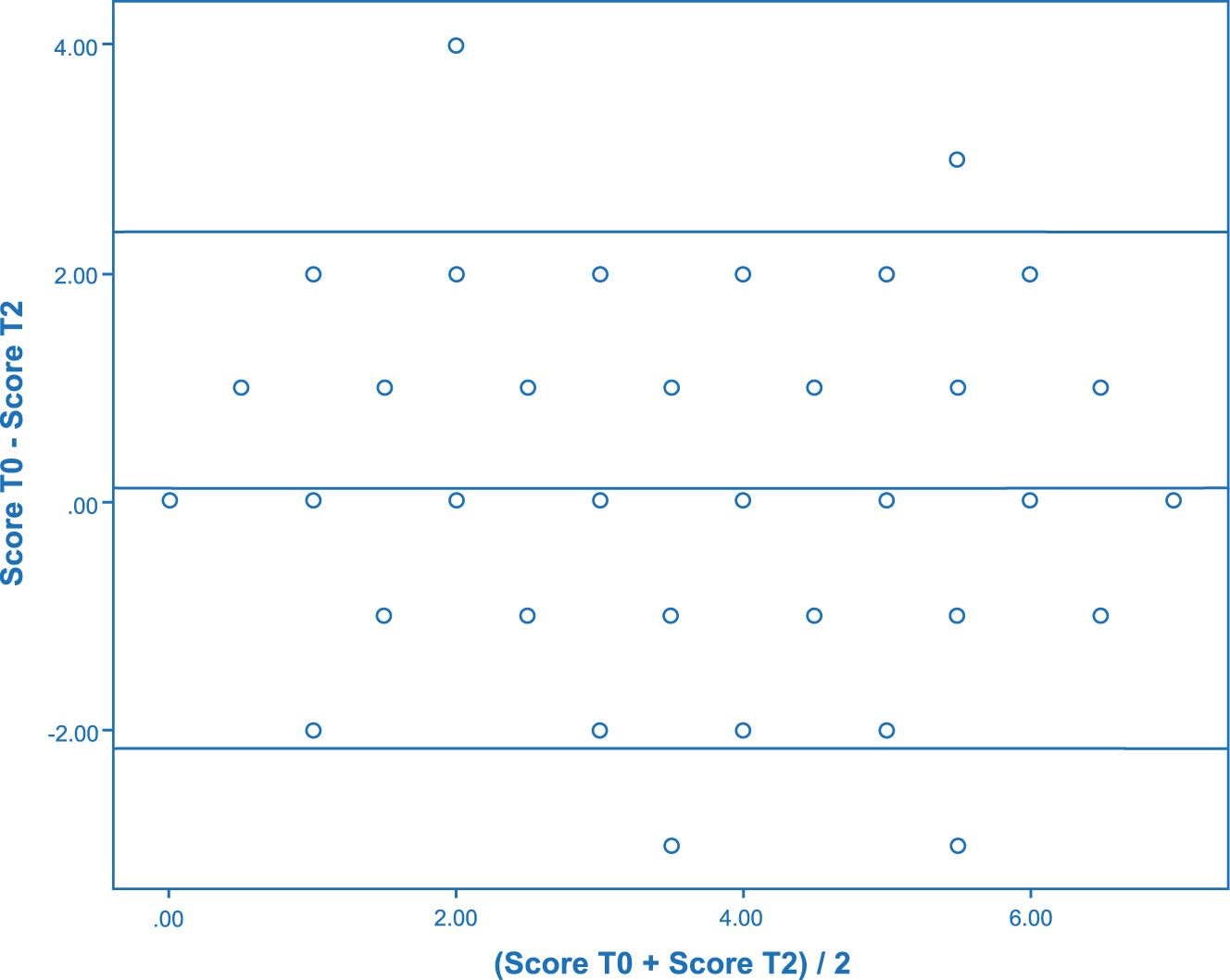
Bland-Altmann plot for total DN4 scores at T0-T2.
Based on the cut-off score of 3, the DN4 classified 65 patients as having NeP and 38 patients as not having NeP at T0 (Table 2). At T2, 62 patients were identified as having NeP and 41 patients as not having NeP. The classifications at these two time points demonstrated a weighted kappa score of 0.733 (95 % CI: 0.598–0.868) (Table 3). Kappa scores for the individual items varied between 0.582 (95 % CI: 0.423–0.741) and 0.850 (95 % CI: 0.657–1.043) (Table 3).
Discussion
This is the first study to investigate the reliability of the English language version of the 7-item DN4 in a large cohort of patients with chronic pain. The results showed almost perfect agreement for the 7-item DN4 total score and DN4 pain classification at pre- and post-clinical consultation (T0-T1) and almost perfect agreement for the 7-item DN4 total score and substantial agreement for DN4 pain classifications at one week follow-up (T0-T2).
A representative sample with a broad age range and balanced gender distribution was achieved. We did not exclude specific diseases or pain presentations compared to other studies which did not include diseases associated with diffuse pain, such as fibromyalgia [4, 9], [10], [11, 14, 17, 19, 21]. In addition, we did not define minimal pain intensity whereas many studies included only participants with moderate to severe pain intensity [4, 9, 11, 15, 17], [18], [19]. Our study population would reflect the type of pain presentations seen in everyday clinical practice and, in combination with a much larger sample size compared to others [4, 7, 10], [11], [12], [13], [14], [15], [16], [17], [18], [19], [20], [21], allows for greater generalizability to a wider range of people suffering from persistent pain. We also did not define a minimum pain duration. However, it is evident from our results that most patients were chronic pain patients comparable with cohorts in the original study [4] and other studies [7, 9], [10], [11, 13], [14], [15, 17], [18], [19], [20], [21].
Out of three studies investigating the reliability of the 7-item DN4 questionnaire [10, 13, 20], one study used the questionnaire in interview format [13] and only two studies are comparable with our methodology, applying the DN4 as a self-reported tool [10, 20]. The Hindi version was assessed using only one follow-up period of three days [20]. The results were comparable to ours with an almost perfect agreement for the overall score (ICC of 0.92) and substantial to almost perfect agreement for the individual items (ICC between 0.71 and 0.96). Their follow-up sample consisted of only 82 subjects. By contrast, the Dutch version also used two follow-up periods, longer than ours, lasting two weeks and three months [10]. T0 starting not before but directly after the clinical consultation. Their results were slightly worse than ours, demonstrating almost perfect (ICC 0.85) and substantial agreement (ICC 0.79) respectively which may relate to the longer time frame between measurements.
In our cohort, the difference between the DN4 total score between T0 and T1 was statistically significant, however this was not clinically significant, as the mean still fell into the same NeP classification and the agreement of DN4 classifications was almost perfect between the two time points. DN4 classifications between T0-T2 showed substantial agreement. Unfortunately our results cannot be compared to other studies using the 7-item DN4 questionnaire as they did not report on this comparison [10, 20]. With respect to the classifications of the English painDETECT [24], better to equal results were observed. While the measurement period T0-T2 showed equally substantial agreement (weighted Kappa of 0.691), the results for T0-T1 in our study were better in contrast to the substantial agreement of the painDETECT (weighted kappa of 0.771).
We observed outliers in the time interval T0-T1 as well as T0-T2. A 65-year-old woman with chronic low back pain had a total score of 6 at T0, whereas a score of 0 was obtained at T1. While she reported all symptoms except “burning” as present at T0, no symptoms seemed to be present at T1. Two other outliers, a 19-year-old woman and a 55-year-old woman with degenerative joint disease, also had reductions (both minus 4 points) in the total score. We assume that the visit to the health professional somehow had an effect on the symptoms of the subjects mentioned. For the interval T0-T2, two outliers related to a reduction in the DN4 score and two showed a score increase. Since all subjects included in the long-term analysis stated on the PGIC that their complaints had not changed or had changed only slightly, these results remain difficult to explain.
The individual DN4 symptom descriptors showed substantial to almost perfect agreement in the short-term interval and substantial agreement for the long-term interval except for the symptom “pins and needles” (moderate agreement). It remains unclear why this item was less stable as other studies did not report the same observation [13, 20].
The majority of our patients had multiple pain areas. In order to guarantee the validity of the questionnaire, patients were asked to indicate their main pain area and to answer the DN4 questions in regards to their main pain area. They were also told that the following DN4 questionnaires should also be completed for the same pain area. Similar observations of the presence of multiple pain areas were made in the English painDETECT study [24], but not reported in any studies using the DN4. One DN4 study used a body chart to verify the location of pain [11]; another study defined that the questionnaire should be completed in regards to the pain which is the reason for current physician consultation [7]. However, subjects in our study had difficulty identifying a main area of pain due to multiple areas of pain with varying intensity. Hence, it cannot be conclusively ruled out that the repeated questionnaires were completed for a different pain area. While in our study the presence of multiple pain areas seemed not to affect the reliability, it has to be considered that the discriminative ability of NeP screening tools is reliable only when applied to one pain area [28].
We encountered some difficulties in our study. Some participants did not understand the meaning of used terminologies in the DN4, such as “painful cold”. The investigators were not supposed to assist the participants, as the 7-item DN4 was used as a self-reported tool. At most, they suggested that the character of pain might not have been experienced if it was unknown to the participant. Although there is an instruction on the DN4 that each question has to be answered with either “yes” or “no”, some participants had to be explicitly instructed to tick one of the options. In the Dutch study [10], help was provided to complete the survey, however, questions were rarely asked. In the Arabic version [14], subjects were asked at the end of the questionnaire whether they had understood the questions.
The time frame between T0 and T2 varied amongst individuals, as delays in mail delivery were not uncommon and participants could not always complete the questionnaire on time after one week. A solution could be to digitise the questionnaire and this would also minimise the risk of missing data.
Conclusions
The English version of the 7-item DN4 has been shown to be a reliable tool for measurements of short-term (same day) and long-term reliability (one week). The validity of the questionnaire has yet to be investigated.
Acknowledgements
We would like to thank the staff of the Sir Charles Gairdner Hospital Department of Pain Management and Neurosurgery Spinal Clinic for their support. We would also like to thank all participants.
-
Research funding: Authors state no funding was involved with this research.
-
Author contributions: All authors have accepted responsibility for the entire content of this manuscript and approved its submission.
-
Competing interests: Authors state no conflict of interest.
-
Informed consent: Informed consent has been obtained from all individuals included in this study.
-
Ethical approval: Research involving human subjects complied with all relevant national regulations, institutional policies and is in accordance with the tenets of the Helsinki Declaration (as amended in 2013), and has been approved by the authors‘ Institutional Review Board (IRB #RGS0000001759).
References
1. Jensen, TS, Baron, R, Haanpää, M, Kalso, E, Loeser, JD, Rice, ASC, et al.. A new definition of neuropathic pain. Pain 2011;152:2204–5. https://doi.org/10.1016/j.pain.2011.06.017.Search in Google Scholar PubMed
2. Attal, N, Lanteri-Minet, M, Laurent, B, Fermanian, J, Bouhassira, D. The specific disease burden of neuropathic pain: results of a French nationwide survey. Pain 2011;152:2836–43. https://doi.org/10.1016/j.pain.2011.09.014.Search in Google Scholar PubMed
3. Doth, AH, Hansson, PT, Jensen, MP, Taylor, RS. The burden of neuropathic pain: a systematic review and meta-analysis of health utilities. Pain 2010;149:338–44. https://doi.org/10.1016/j.pain.2010.02.034.Search in Google Scholar PubMed
4. Bouhassira, D, Attal, N, Alchaar, H, Boureau, F, Brochet, B, Bruxelle, J, et al.. Comparison of pain syndromes associated with nervous or somatic lesions and development of a new neuropathic pain diagnostic questionnaire (DN4). Pain 2005;114:29–36. https://doi.org/10.1016/j.pain.2004.12.010.Search in Google Scholar PubMed
5. Bouhassira, D, Lantéri-Minet, M, Attal, N, Laurent, B, Touboul, C. Prevalence of chronic pain with neuropathic characteristics in the general population. Pain 2008;136:380–7. https://doi.org/10.1016/j.pain.2007.08.013.Search in Google Scholar PubMed
6. Chaudakshetrin, P, Prateepayanich, P, Chira-Adisai, W, Tassanawipas, W, Leechavengvongs, S, Kitisomprayoonkul, W. Cross-cultural adaption to the Thai language of the neuropathic pain diagnostic questionnaire (DN4). J Med Assoc Thai 2007;90:1860.Search in Google Scholar
7. Perez, C, Galvez, R, Huelbes, S, Insausti, J, Bouhassira, D, Diaz, S, et al.. Validity and reliability of the Spanish version of the DN4 (Douleur Neuropatique 4 questions) questionnaire for differential diagnosis of pain syndromes associated to a neuropathic or somatic component. Health Qual Life Outcome 2007;5:1–10.10.1186/1477-7525-5-66Search in Google Scholar PubMed PubMed Central
8. Van Seventer, R, Vos, C, Meerding, W, Mear, I, Le Gal, M, Bouhassira, D, et al.. Linguistic validation of the DN4 for use in international studies. Eur J Pain 2010;14:58–63. https://doi.org/10.1016/j.ejpain.2009.01.005.Search in Google Scholar PubMed
9. van Seventer, R, Vos, C, Giezeman, M, Meerding, W-J, Arnould, B, Regnault, A, et al.. Validation of the Dutch version of the DN4 diagnostic questionnaire for neuropathic pain. Pain Pract 2013;13:390–8. https://doi.org/10.1111/papr.12006.Search in Google Scholar PubMed
10. Timmerman, H, Steegers, MAH, Huygen, F, Goeman, JJ, van Dasselaar, NT, Schenkels, MJ, et al.. Investigating the validity of the DN4 in a consecutive population of patients with chronic pain. PLoS One 2017;12:e0187961. https://doi.org/10.1371/journal.pone.0187961.Search in Google Scholar PubMed PubMed Central
11. Santos, JG, Brito, JO, de Andrade, DC, Kaziyama, VM, Ferreira, KA, Souza, I, et al.. Translation to Portuguese and validation of the Douleur Neuropatique 4 Questionnaire. J Pain 2010;11:484–90. https://doi.org/10.1016/j.jpain.2009.09.014.Search in Google Scholar PubMed
12. Unal-Cevik, I, Sarioglu-Ay, S, Evcik, D. A comparison of the DN4 and LANSS questionnaires in the assessment of neuropathic pain: validity and reliability of the Turkish version of DN4. J Pain 2010;11:1129–35. https://doi.org/10.1016/j.jpain.2010.02.003.Search in Google Scholar PubMed
13. Harifi, G, Ouilki, I, El Bouchti, I, Ait Ouazar, M, Belkhou, A, Younsi, R, et al.. Validity and reliability of the Arabic adapted version of the DN4 questionnaire (Douleur Neuropatique 4 Questions) for differential diagnosis of pain syndromes with a neuropathic or somatic component. Pain Pract 2011;11:139–47. https://doi.org/10.1111/j.1533-2500.2010.00399.x.Search in Google Scholar PubMed
14. Chatila, N, Pereira, B, Maarrawi, J, Dallel, R. Validation of a new Arabic version of the neuropathic pain diagnostic questionnaire (DN4). Pain Pract 2017;17:78–87. https://doi.org/10.1111/papr.12419.Search in Google Scholar PubMed
15. Hallström, H, Norrbrink, C. Screening tools for neuropathic pain: can they be of use in individuals with spinal cord injury? Pain 2011;152:772–9. https://doi.org/10.1016/j.pain.2010.11.019.Search in Google Scholar PubMed
16. Spallone, V, Morganti, R, D’Amato, C, Greco, C, Cacciotti, L, Marfia, GA. Validation of DN4 as a screening tool for neuropathic pain in painful diabetic polyneuropathy. Diabet Med 2012;29:578–85. https://doi.org/10.1111/j.1464-5491.2011.03500.x.Search in Google Scholar PubMed
17. Madani, SP, Fateh, HR, Forogh, B, Fereshtehnejad, SM, Ahadi, T, Ghaboussi, P, et al.. Validity and reliability of the Persian (Farsi) version of the DN4 (Douleur Neuropatique 4 Questions) questionnaire for differential diagnosis of neuropathic from non-neuropathic pains. Pain Pract 2014;14:427–36. https://doi.org/10.1111/papr.12088.Search in Google Scholar PubMed
18. Sykioti, P, Zis, P, Vadalouca, A, Siafaka, I, Argyra, E, Bouhassira, D, et al.. Validation of the Greek version of the DN4 diagnostic questionnaire for neuropathic pain. Pain Pract 2015;15:627–32. https://doi.org/10.1111/papr.12221.Search in Google Scholar PubMed
19. Kim, HJ, Park, JH, Bouhassira, D, Shin, JH, Chang, BS, Lee, CK, et al.. Validation of the Korean version of the DN4 diagnostic questionnaire for neuropathic pain in patients with lumbar or lumbar-radicular pain. Yonsei Med J 2016;57:449–54. https://doi.org/10.3349/ymj.2016.57.2.449.Search in Google Scholar PubMed PubMed Central
20. Gudala, K, Ghai, B, Bansal, D. Hindi version of short form of douleur ` 4 (S-DN4) questionnaire for assessment of neuropathic pain component: a cross-cultural validation study. Korean J Pain 2017;30:197–206. https://doi.org/10.3344/kjp.2017.30.3.197.Search in Google Scholar PubMed PubMed Central
21. Matsuki, Y, Sukenaga, N, Miyagi, K, Tsunetoh, T, Mizogami, M, Shigemi, K, et al.. Reliability and validity of the Japanese translation of the DN4 Diagnostic Questionnaire in patients with neuropathic pain. Anaesthesia 2018;32:403–8. https://doi.org/10.1007/s00540-018-2495-7.Search in Google Scholar PubMed
22. Wang, YF, Yang, CC, Ro, LS, Tsai, YC, Lin, KP, Sun, WZ, et al.. Development and validation of a Taiwan version of the DN4-T questionnaire. Chin Med J 2019;82:623–7. https://doi.org/10.1097/jcma.0000000000000129.Search in Google Scholar PubMed
23. Gudala, K, Ghai, B, Bansal, D. Usefulness of four commonly used neuropathic pain screening questionnaires in patients with chronic low back pain: a cross-sectional study. Korean J Pain 2017;30:51–8. https://doi.org/10.3344/kjp.2017.30.1.51.Search in Google Scholar PubMed PubMed Central
24. Tampin, B, Bohne, T, Callan, M, Kvia, M, Melsom Myhre, A, Neoh, E, et al.. Reliability of the English version of the painDETECT questionnaire. Curr Med Res Opin 2017;33:741–8. https://doi.org/10.1080/03007995.2017.1278682.Search in Google Scholar PubMed
25. Sim, J, Wright, CC. The kappa statistic in reliability studies: use, interpretation, and sample size requirements. Phys Ther 2005;85:257–68. https://doi.org/10.1093/ptj/85.3.257.Search in Google Scholar
26. Bouhassira, D, Attal, N, Fermanian, J, Alchaar, H, Gautron, M, Masquelier, E, et al.. Development and validation of the neuropathic pain symptom inventory. Pain 2004;108:248–57. https://doi.org/10.1016/j.pain.2003.12.024.Search in Google Scholar PubMed
27. Landis, JR, Koch, GG. The measurement of observer agreement for categorical data. Biometrics 1977;33:159–74. https://doi.org/10.2307/2529310.Search in Google Scholar
28. Bouhassira, D, Attal, N. Diagnosis and assessment of neuropathic pain: the saga of clinical tools. Pain 2011;152:74–83. https://doi.org/10.1016/j.pain.2010.11.027.Search in Google Scholar PubMed
© 2023 Walter de Gruyter GmbH, Berlin/Boston
Articles in the same Issue
- Frontmatter
- Systematic Review
- Comparison of the effectiveness of eHealth self-management interventions for pain between oncological and musculoskeletal populations: a systematic review with narrative synthesis
- Topical Review
- Shifting the perspective: how positive thinking can help diminish the negative effects of pain
- Clinical Pain Researches
- Pain acceptance and psychological inflexibility predict pain interference outcomes for persons with chronic pain receiving pain psychology
- A feasibility trial of online Acceptance and Commitment Therapy for women with provoked vestibulodynia
- Relations between PTSD symptom clusters and pain in three trauma-exposed samples with pain
- Short- and long-term test–retest reliability of the English version of the 7-item DN4 questionnaire – a screening tool for neuropathic pain
- Chronic post-thoracotomy pain after lung cancer surgery: a prospective study of preoperative risk factors
- Pain sensitivity after Roux-en-Y gastric bypass – associations with chronic abdominal pain and psychosocial aspects
- Barriers in chronic prostatitis/chronic pelvic pain syndrome (CP/CPPS) management: perspectives from health practitioners
- Observational studies
- Spontaneous self-affirmation: an adaptive coping strategy for people with chronic pain
- COVID-19 and processes of adjustment in people with persistent pain: the role of psychological flexibility
- Presence and grade of undertreatment of pain in children with cerebral palsy
- Sex-related differences in migraine clinical features by frequency of occurrence: a cross-sectional study
- Recurrent headache, stomachache, and backpain among adolescents: association with exposure to bullying and parents’ socioeconomic status
- Original Experimentals
- Temporal stability and responsiveness of a conditioned pain modulation test
- Anticipatory postural adjustments mediate the changes in fear-related behaviors in individuals with chronic low back pain
- The role of spontaneous vs. experimentally induced attentional strategies for the pain response to a single bout of exercise in healthy individuals
- Acute exercise of painful muscles does not reduce the hypoalgesic response in young healthy women – a randomized crossover study
- Short Communications
- Nation-wide decrease in the prevalence of pediatric chronic pain during the COVID-19 pandemic
- A multidisciplinary transitional pain service to improve pain outcomes following trauma surgery: a preliminary report
Articles in the same Issue
- Frontmatter
- Systematic Review
- Comparison of the effectiveness of eHealth self-management interventions for pain between oncological and musculoskeletal populations: a systematic review with narrative synthesis
- Topical Review
- Shifting the perspective: how positive thinking can help diminish the negative effects of pain
- Clinical Pain Researches
- Pain acceptance and psychological inflexibility predict pain interference outcomes for persons with chronic pain receiving pain psychology
- A feasibility trial of online Acceptance and Commitment Therapy for women with provoked vestibulodynia
- Relations between PTSD symptom clusters and pain in three trauma-exposed samples with pain
- Short- and long-term test–retest reliability of the English version of the 7-item DN4 questionnaire – a screening tool for neuropathic pain
- Chronic post-thoracotomy pain after lung cancer surgery: a prospective study of preoperative risk factors
- Pain sensitivity after Roux-en-Y gastric bypass – associations with chronic abdominal pain and psychosocial aspects
- Barriers in chronic prostatitis/chronic pelvic pain syndrome (CP/CPPS) management: perspectives from health practitioners
- Observational studies
- Spontaneous self-affirmation: an adaptive coping strategy for people with chronic pain
- COVID-19 and processes of adjustment in people with persistent pain: the role of psychological flexibility
- Presence and grade of undertreatment of pain in children with cerebral palsy
- Sex-related differences in migraine clinical features by frequency of occurrence: a cross-sectional study
- Recurrent headache, stomachache, and backpain among adolescents: association with exposure to bullying and parents’ socioeconomic status
- Original Experimentals
- Temporal stability and responsiveness of a conditioned pain modulation test
- Anticipatory postural adjustments mediate the changes in fear-related behaviors in individuals with chronic low back pain
- The role of spontaneous vs. experimentally induced attentional strategies for the pain response to a single bout of exercise in healthy individuals
- Acute exercise of painful muscles does not reduce the hypoalgesic response in young healthy women – a randomized crossover study
- Short Communications
- Nation-wide decrease in the prevalence of pediatric chronic pain during the COVID-19 pandemic
- A multidisciplinary transitional pain service to improve pain outcomes following trauma surgery: a preliminary report

