Abstract
Objective
Industrial effluents and agriculture biomass are main environmental hazards which are facing by developing country like Pakistan. Along with various other industrial applications, laccases are also involved in the oxidation of various industrial hazardous compounds to detoxify them. This study was designed to produce and purify laccase from ascomyceteous fungi, i.e. Alternaria alternata through solid stat fermentation.
Materials and methods
Abundantly available Sarkanda grass “Saccharum spontaneum” was used as agro-waste substrate for laccase production from fungus A. alternata. Previously only white rot fungi are familiar for laccase production and almost no work has been done on laccase production by A. alternata. In this research work, different physical and chemical parameters were optimized for maximum laccase production through solid state fermentation (SSF).
Results
Enzyme was purified and its molecular weight was determined by sodium dodecyl sulfate polyacrylamide gel electrophoresis (SDS-PAGE). Maximum laccase activity (21.87±0.0115 unit/mL) was detected on 7th day of incubation having pH 5 of the medium at 35°C. None of the added metal ions increased laccase production. Galactose and “yeast extract” used as optimum carbon and nitrogen source for highest laccase production.
Conclusion
A monomeric protein (laccase) having approximately 51 kDa molecular weight obtained after SDS-PAGE.
Özet
Amaç
Endüstriyel atıklar ve tarımsal biyokütle Pakistan gibi gelişmekte olan ülkelerde çevresel tehlikelerin başında gelmektedir. Lakkazlar, çeşitli endüstriyel uygulamalarla birlikte, bu tür tehlikeli bileşiklerin oksitlenmesinde rol almakta ve detoksifikasyonda kullanılmaktadır.
Materyal ve metod
Bu çalışmada bol miktarda bulunan sarkanda çimi “Saccharum spontaneum”, Alternaria alternata’dan lakkaz üretimi sırasında agro-atık madde olarak kullanıldı. Günümüzde sadece beyaz çürükçül mantarları lakkaz üretimi için kullanılmaktadır, Alternaria alternata tarafından lakkaz üretimi üzerine neredeyse hiç çalışma yapılmamıştır. Bu araştırma çalışmasında, katı hal fermantasyonu (SSF) ile maksimum lakkaz üretimi için farklı fiziksel ve kimyasal parametreler optimize edilmiştir.
Bulgular
Enzim saflaştırıldı ve molekül ağırlığı SDS-PAGE ile belirlendi. Maksimum lakkaz aktivitesi (21.87±0.0115 birim/mL), pH 5 ve 35°C’de inkübasyonun 7. gününde tespit edildi. Eklenen metal iyonlarından hiçbiri lakkaz üretimini arttırmadı. Galaktoz ve “maya özütü” en yüksek lakkaz üretimi için optimum karbon ve azot kaynağı olarak kullanılır.
Sonuç
SDS-PAGE sonrasında yaklaşık 51 kDa molekül ağırlığına sahip monomerik bir protein (Lakkaz) elde edildi.
Introduction
Now a day, fungal enzymes are gaining more attention due to their prospective use in the pollutants detoxification [1], [2]. The biological treatments of environmental wastes like dye through laccase enzyme replacing conventional physical and chemical methods which pose severe limitations [1], [3], [4], [5], [6], [7].
Laccases (benzenediol: oxygen oxidoreductases, EC 1.10.3.2) are tetrameric or dimeric glycoproteins having four copper atoms dispersed in redox sites [8]. They can oxidize extensive range of inorganic and organic materials, including phenols, methoxyphenols, aminophenols, organic pollutants and aromatic amines ascorbate with simultaneous four-electron reduction of oxygen to water [9], [10], [11].
The laccase have recently used in the fast progression of textile manufacturing, along with decolorization of textile effluents, it is also used to bleach fabrics and even to synthesize colorants [12]. Laccases have also vast applications in production of anti cancer drugs, cleaning agents, water purification system and in cosmetics [13], [14], [15], [16].
Previously, the production of most enzymes was mostly done with submerged fermentation (SmF) techniques. However, in the last decades the enzyme production trend is shifted towards the solid state fermentation (SSF) due to its improve digestibility of agricultural wastes [17]. The SSF have many advantages over SmF such as less waste production, required simpler bio processing apparatus and the direct availability of the fermented product for feeding [18].
Saccharum spontaneum, locally named as “Kana” is abundant non woody, wasteland wild plant dominating the rural areas of Pakistan with sharp, hairy and green leaves. Abundantly available agro waste, S. spontaneum can be used as substrate for laccase manufacturing in SSF [17]. The laccase production is mostly reported with Ascomyceteous, Deuteromyceteous and Basidiomyceteous fungi [19], [20]. Production of extracellular laccase is a key quality of various higher Basidiomyceteous fungi, principally which are involved in wood decay and breakdown of leaf litter. White rot fungi have been commonly used for the production of lingo cellulolytic enzymes by SSF [21], [22]. Monocillium indicum was the first Ascomycetes fungi reported for laccase production [23], [24], [25].
Developing countries like Pakistan have major problems of proper management of industrial effluents. Abundant volume of industrial effluents like toxic heavy metals and metalloids are disposed into rivers and canals without any prior treatment which poses great threats to biological systems [26]. Cheap laccases produced by local fungal strain can be very good option for reducing the pollution in Pakistan.
So far researchers have mainly focused on Basidiomycete, predominantly white rot fungi for laccase production. Very little work has been done on Ascomycetes and particularly no work has been reported on Alternaria alternata for laccase production through SSF. Keeping in mind these facts, and importance of laccase for world and Pakistan, the present research work was conducted to optimize the culture condition for maximum laccase production by A. alternata through SSF.
Materials and methods
Substrate
Sarkanda grass (S. spontaneum) was collected from a village (Chhimma) of Gujrat district, and used as an agricultural waste substrate for laccase Production through “SSF”. The substrate was washed; sun dried and then crushed to mesh size of 40 mm then kept in moisture free plastic bottle for further used.
Fermentative organism
Pure culture of A. alternata was procured from Department of Biochemistry and molecular biology, University of Gujrat. The potato dextrose agar (PDA) (70139, Sigma-Aldrich, USA) slants were used for fungal culture growth.
Maintenance of medium
The sterilized PDA media of pH 4.8 was prepared and transferred into autoclaved “petri plates” and “test tubes”. The spore of A. Alternata was transferred into slants in sterilized condition to avoid any unwanted contamination in medium. These slants were incubated aerobically for sporulation at 30°C for 3 days (72 h) then stored at 4°C for further use. Fungus was sub cultured periodically twice in the month to maintain the required spore suspension for fermentation process [27].
Inoculum development
The aseptic transfer of A. alternata from PDA slant to 500 mL capacity flasks were performed in 100 mL autoclaved inoculum medium which served as growth medium for fungi. These flasks were then incubated at 30°C for 3 days in “orbital shaker” at 120 rpm [28].
After 72 h (3 days) incubation, the medium was subjected for spores count. For spores counting purpose, hemocytometer was used adopting the method of Kolmer (1959). The concentration of spores was maintained at 1.52× 108 spores per mL in the homogeneous spore suspension. This inoculum (5 mL) was transferred to the fermentation flask (500 mL) having wet substrate (S. spontaneum) for optimization of different factors for laccase production. For each parameter/factor, newly prepared inoculum was used.
Solid state fermentation
All experiments were executed in triplicate flasks having seventy percent moisturized substrate. The nourishing medium was autoclaved and after cooling the flasks, 5 mL inoculum was poured to each flask aseptically (in laminar air flow hood) with the help of autoclaved syringe. For fermentation process, the flasks were then incubated at 37°C under still culture condition.
Laccase production and extraction
Laccase was produced in 500 mL Erlenmeyer flasks using S. spontaneum as a lignocellulosic agricultural waste substrate under optimized growth condition. The inoculated flasks were placed in a temperature controlled still culture incubator at 30°C for 7 days.
At 7th day of incubation, 50 mM sodium acetate buffer of pH 4.5 (100 mL) was added into fermented biomass, and placed in shaker at 150 rpm for half an hour then harvested. The Whatman No. 1 paper was used to filter the contents and filtrates were centrifuged (Corning® LSE™ compact centrifuges, Thermofisher Scientific) at 3000×g for 15 min at 25°C. Resulting “clear supernatant” was used as a crude enzyme extract for laccase activity assessment and refining/purification [29].
Laccase assay
The crude enzyme extract assay was done by observing the oxidation of ABTS at 35°C on pH 4.5 [30]. Total reaction mixture of 2.1 mL, containing 1 mL of 50 mM acetate buffer of pH 4.5, 1 mL of 1 mM 2,2 azinobis (3-ethylebenz-thiazoline)-6 sulfonate (ABTS) and 0.1 mL of enzyme solution was used for laccase activity. Oxidation of ABTS was measured by taking absorbance at 420 nm (ε420 36,000 M cm−1) after 10 min using 1.00707 EMD Millipore UV-VIS spectrophotometer Pharo 300 Spectroquant® Sigma-Aldrich, USA. The enzyme activity (IU) was measured by converting absorbance into μM of ABTS oxidized per min.
Optimization of conditions
Conditions like fermentation period, temperature, pH, nitrogen source; additional carbon source and different metal ions and their concentrations were optimized for laccase production through SSF.
Fermentation period
Triplicate flasks sets, containing Sarkanda grass (0.5 g) as a substrate was moistened with 10 mL of distilled water. The pH was maintained to 4.5 and the flasks were autoclaved and inoculated with 3 mL inoculum of A. alternata and incubated at 35°C for 2–11 days under still culture conditions. Flasks were harvested after every day for laccase production.
Medium’s initial pH
The Sarkanda grass substrate was moistened with Krick basal medium of different pH and incubated at 35°C for 7 days for maximum laccase production. The pH which showed best laccase productivity used for next experiments.
Incubation temperature
Meshed Sarkanda grass (0.5 g) was moistened with 10 mL distilled water and pH set to 5. Autoclaved flasks were inoculated with fungus spores and triplicate sets of flasks were incubated at different temperatures. Incubation was done for 7 days (optimized incubation period) in the still culture conditions. The temperature at which fungus grows intensively and produced maximum quantity of laccase was selected as an optimum temperature.
Additional supplementary carbon source
Fungi and many other microbes utilize energy from carbon source for their growth and other biochemical events. Six carbon sources were supplemented to the previously optimized medium of Sarkanda grass. The source which showed the enhanced production of laccase by A. alternata was chosen for further research work.
Supplementary nitrogen source
The nitrogen source and its concentration is crucial for microorganism’s growth and metabolism. So, various nitrogen supplements provided to examine their enhancing/inhibiting impact on laccase production by A. alternata under optimum conditions. The additive showing maximum enhancement in laccase production was chosen as a best nitrogen source for laccase production.
Different metal ions with varying concentrations
Metal ions can act as inhibitor or stimulator for fungi for laccase production. Different metal ions with varying concentration were used to check maximum laccase activity by A. alternata.
Purification of laccase
Crude laccase extract was centrifuged at (3000×g) for 15 min at 40°C followed by the ammonium sulfate precipitation. Laccase was precipitated from aqueous solution by dissolving ammonium sulfate into the protein solutions. Ammonium sulfates with different concentrations (35, 40, 45, 50, 55, 60, 65, 70, 75 and 80%) were used. Ammonium sulfate was used for salting out process as it can be fully dissolved into water resulting in high ionic power. At high ionic concentration, added salt can remove hydration water from proteins resulting in protein coagulation. The concentration at which laccase assay revealed minimum absorbance (75%) was selected as an optimum concentration because of maximum protein coagulation at that concentration [31].
Dialysis
Dialysis was performed for the separation of required enzyme protein from ammonium sulfate and other impurities. Acetate buffer (0.016 M) was used for dialysis of sample which was achieved by 75% ammonium sulfate precipitation. The dialysis membrane was filled with enzyme sample and tied from both sides. Membrane was then placed in the beaker having 400 mL acetate buffer and stirred for 6 h. During stirring buffer was changed continuously after every 2 h. Then this dialyzed sample was passed through column and multiple vials were pooled for protein and laccase activity determination. Finally the high laccase activity sample was subjected for sodium dodecyl sulfate polyacrylamide gel electrophoresis (SDS-PAGE) [29].
SDS-PAGE
For estimation of molecular weight, the filtered laccase was run on SDS-PAGE following the method of Laemmli (1970). The approximate molecular weight of purified laccase was checked by comparing it against broad molecular weight range (29–98 kDa) markers (“Phosphorylase b”, 98 kDa; “Bovine Serum Albumin”, 68 kDa; “Ovalbumin”, 44 kDa. and Glutathione S-Transferase, 29 kDa).
Results and discussion
Fungus has unique features of different enzyme production on cheap and local agro-waste substrate [22], [32]. The enzyme manufacturing is a growing era of biotechnology. Yearly world sales figures of enzymes are near to billion dollars [33]. Patents and research articles associated with this field are increasing terrifically. Huge amounts of low cost enzymes are required for missive biotechnological applications. So a cheap and efficient method for enzyme production is to use lignocellulosic wastes which have substantial concentrations of soluble carbohydrates and can generate ligninolytic enzymes using suitable microorganism [34], [35], [36].
The vital elements which may affect the enzyme production through SSF includes: selection of suitable microorganism and substrate; size of the meshed substrate particles; optimization of the incubation period, temperature, pH of the medium and optimization of the nutrients maintenance in the SSF environment [37].
Production of laccase
A significant magnitude of an indigenous laccase was obtained from A. alternata in SSF of sarkanda grass (S. spontaneum) under optimized fermentation growth conditions. Maximum laccase activity (21.87±0.0115 units/mL) was attained when 5 g of sarkanda grass (40 meshed) was supplemented with 0.25 mg galactose, 0.25 mg yeast extract, inoculated with 3 mL of freshly prepared spore suspension of A. alternata and incubated at 35°C in SSF environment. Balaraju [38] reported laccase production through SSF using Oudemansiella radicata and activity was observed as 25.784 units/mL in rice bran. The next highest activity was observed at 11.473 units/mL in wheat bran. The nature and amount of the nutrients in the fermentation growth substrate have a strong effect on the extracellular laccase enzyme production. Therefore, a wide spectrum of fungi showed different growth and enzyme activity on different substrates in addition with some nutrient additions [29]. Different low cost agro-industrial substrates like wheat bran, rice straws, wheat straw, molasses and grasses are particularly appropriate for fungal growth and enzyme production under suitable fermentation environments [10].
Influence of incubation period on enzyme production
The incubation period for laccase production was observed with A. alternata at pH 5. The maximum laccase activity was obtained on 7th day and the production was decreased due to feed-back inhibition. The 6.04 units/mL laccase produced with 50% moisture content of grass substrate, depletion of nutrient in the medium was main cause of decreased laccase activity as shown in Figure 1. Even, ascomyceteous fungi are not well known for bulk laccase production as compare to Basidiomyceteous fungi but the present results indicated that A. alternata (Ascomyceteous fungi) produced significant amount of laccase. Similar results was obtained by Mabrouk [11] while using A. alternate as a fermentative organism he described the Guaiacol oxidation is one of the easiest qualitative assay for lignin degrading enzymes (LMEs) fungi. They described that Alternaria solani and A. alternata showed same oxidation and growth; they have optimum 19, 20 mm in “color zone diameter” and 20, 17 mm in “colony growth diameter”, respectively, at 7th day of incubation. Shyzophyllum commune IBL-06 manufactured highest laccase at 3rd day in solid SSF of banana stalk under optimum pH of 4.5 at temperature 35°C [10], [29]. Present results deviated from Gaitan [39] who reported the optimum laccase production of Pleurotus ostreatus at 21st day in SmF.

Influence of incubation time on laccase production by Alternaria alternata.
Influence of pH on laccase activity
The effect of different levels of pH, from 2 to 8 was examined on laccase production by A. alternata after 7th day of incubation at 35°C. The highest laccase activity was obtained at pH 5 on sarkanda grass which was found to be 6.9 units/mL and further rise in pH gradually decreased the laccase activity as shown in Figure 2. While, Johnsy and Kaviyarasan [40] described laccase production in submerged culture conditions by Lentinus kauffmanii and found maximum activity on pH 5.5 at 30°C. Balaraju [38] reported that laccase production through SSF/SmF using O. radicata, the highest activity was observed at 1.476 units/mL on 14th day at pH 7 under SmF. In SSF the highest activity (25.784 units/mL) was observed in rice bran while it was observed 11.473 units/mL in wheat bran.
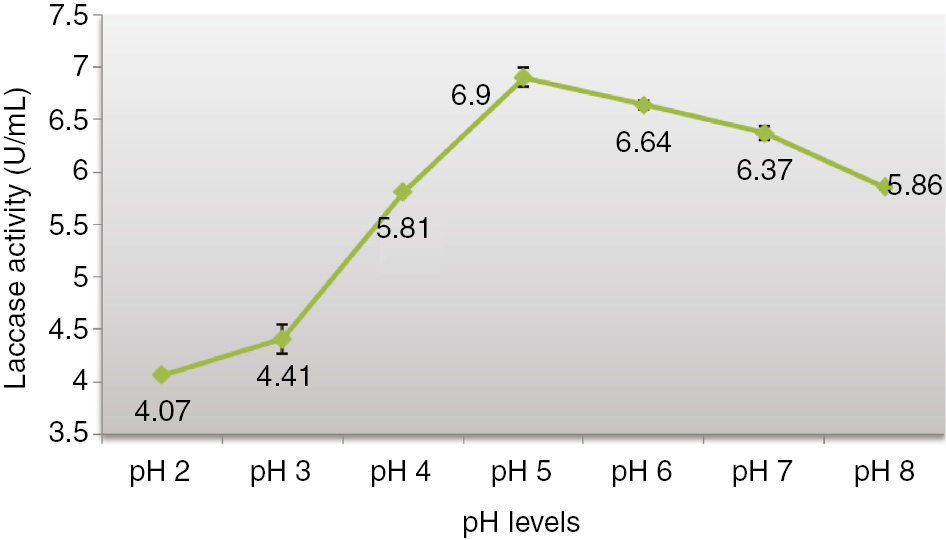
Influence of pH on production of laccase by Alternaria alternata in SSF.
Influence of temperature on enzyme production
Temperature influence on laccase production was assessed from 25 to 40°C as shown in Figure 3 and optimum temperature for maximum laccase activity was observed at 35°C, on pH 5 and 7th day of incubation by A. alternata. It can be seen that the further rise in temperature decreases the activity of laccase in harvested cultured filtrate. Similar results were observed by Elshafei [41] in which optimum temperature for laccase production by Penicillium martensii NRC 345 was observed as 30°C with specific activity of 7.21 U/mg proteins.
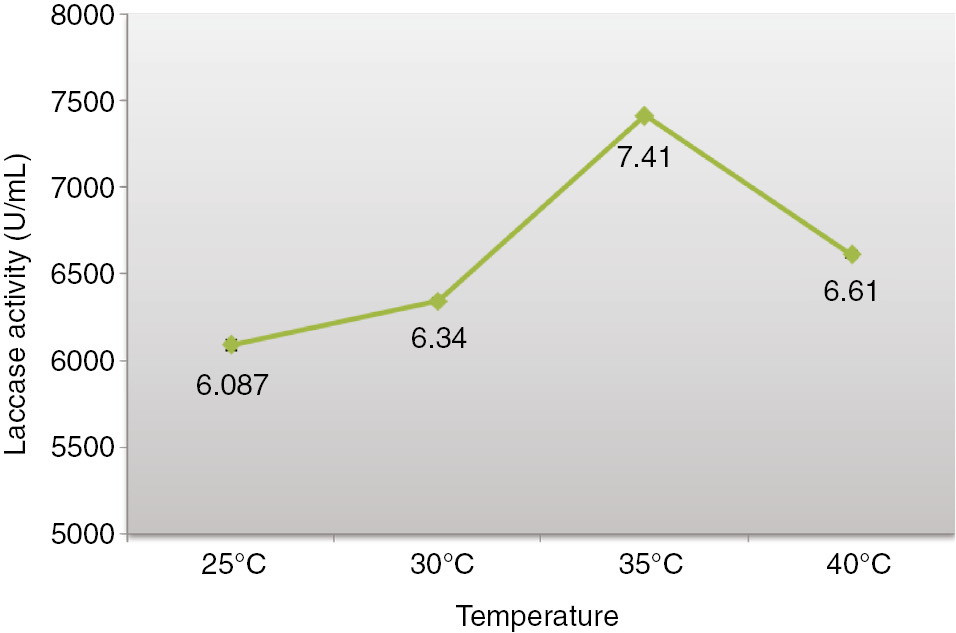
Impact of temperature on laccase formation Alternaria alternata in solid state bio-processing.
Elshafei [41] studied laccase production by P. ostreatus ARC280 on PDA medium and found the maximum fungal laccase production at 28°C.
Influence of supplementary carbon sources on production of laccase
Figure 4 illustrates the additional carbon source effect on laccase production. 0.25 g galactose was selected as a best carbon source in pre optimized medium such as 35°C, 7th day of incubation and pH 5 by A. alternata for laccase production. Additional galactose showed maximum activity of 11.93 U/mL in SSF of Sarkanda grass. Other sugars such as glucose, lactose, maltose, fructose and sucrose considerably increased the laccase activity as shown in Figure 4.
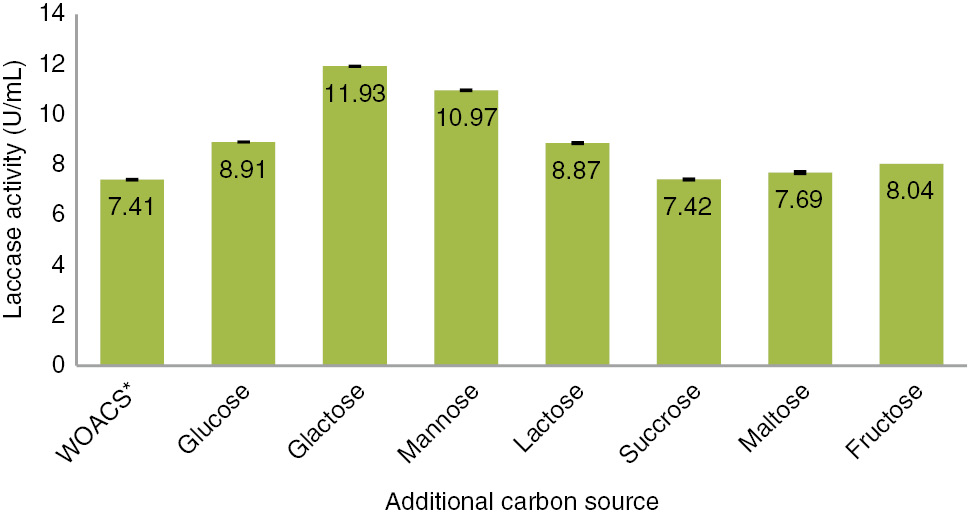
Influence of carbon source on laccase formation by Alternaria alternata.
*Without additional carbon source.
Elshafei [41] studied the production of laccase by P. martensii NRC 345 and effect of different carbon sources on laccase production. He used the concentration of 0.5% as a trial for every supplied carbon source. Each carbon source was supplied to the medium as the prime only carbon source. Like present study, they also described that galactose is an ideal carbon source for laccase production.
Effect of carbon sources on adaptation of the fungus for the formation of laccase is of vital importance. Elsayed [42], [43] studied laccase production by P. ostreatus ARC280 on PDA medium. For checking the impact of various carbon sources on the laccase production, he used galactose, fructose (monosaccharides), maltose, sucrose (disaccharides), and polysaccharides [carboxy methyl cellulose (CMC) and soluble starch]. They found soluble starch as best carbon source for maximum laccase production.
Effect of nitrogen sources on laccase production
The outcome of added nitrogen on laccase production by A. alternata during the 168 h (7 days) fermentation period at 35°C and constant moisture and inoculums size, is showing in Figure 5. Alternaria alternata was investigated for laccase production with urea, ammonium hepta molybdate, ammonium sulfate, yeast extract, ammonium chloride and ammonium per sulfate. Yeast extract (0.25 g) was found to support maximal production of laccase activity 21.44 mg/mL. On the other hand, ammonium per sulfate and ammonium chloride inhibited laccase formation and showed very low activity (Figure 5). D’Agostini et al. [44] evaluated the effect of concentrations of non-protein nitrogen sources, soybean hulls, urea and ammonium sulfate in solid substrate cultivation on laccase production. Elshafei [41] studied optimized laccase production by P. martensii NRC 345 using different nitrogen sources. Sodium nitrate showed maximum laccase activity while on the other hand, like present research, no enzyme production could be noticed with ammonium sulfate or ammonium chloride.
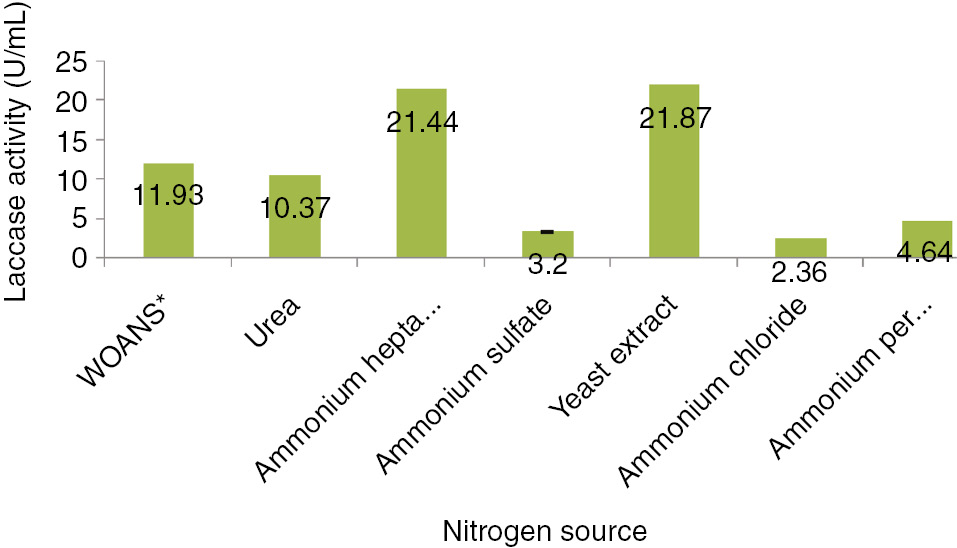
Influence of additional nitrogen source on laccase production by Alternaria alternata.
*Without additional nitrogen source.
Influence of metal ions on laccase production
Impact of different metals ions laccase production
All above mentioned Pre optimized conditions were subjected with different metal ions (0.3%) for laccase production by A. alternata as shown in Figure 6. It was noticed that laccase production decreased significantly as SSF medium was supplemented with various metals ions. Study showed that metals selections were not good for laccase production by this fungus. Figure 6 shows that CaCl2 was the least inhibitor for laccase production and KH2PO4 showed maximum inhibition. Used metal showed decreased production of laccase but it was even higher than many other reported studies regarding laccase production from ascomyceteous fungi [45], [46]. Its applicability will be definitely higher on effluents without these metal ions.
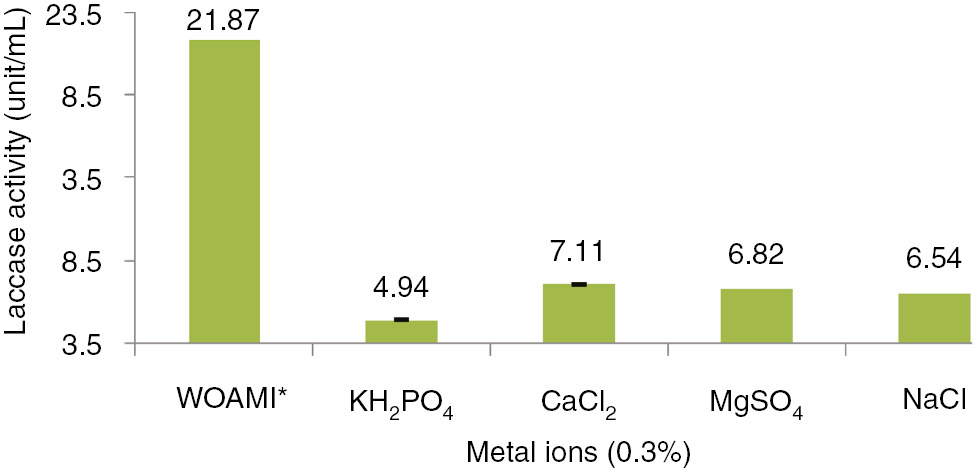
Influence of different metals on laccase production by Alternaria alternata.
*Without additional metal ions.
Mahmoud et al. [45] studied laccase production by Streptomyces lydicus in SmF and showed that CuSO4 acted as laccase production inducer and Fe2+, Zn2+, Mn2+, EDTA and arbutin diminished the enzyme production, while L-cystein, sodium azide and Ag+ totally blocked the enzyme production. As copper ions are the part of laccase enzyme, so increase of laccase production with additional Cu2+ can be well interpreted in this regard.
Impact of different metals with different concentrations on laccase production
Effect of laccase activity was determined by selecting different concentrations of metals (0.1, 0.3, 0.5, 0.7 and 0.9%). Results revealed that none of the used concentration improved laccase activity (Table 1). ANOVA was used to analyze and determine the variability of data (Table 2). Calculated F-value and p-value (0.004) showed that the data was significant.
Laccase activities with metals having different concentrations.
| Metals | Metal conc. (%) | Enzyme activity (unit/mL) | SD |
|---|---|---|---|
| MgSO4 | 0.1 | 6.39 | ±0.008 |
| 0.3 | 6.82 | ±0.017 | |
| 0.5 | 6.05 | ±0.035 | |
| 0.7 | 5.21 | ±0.035 | |
| 0.9 | 4.23 | ±0.000 | |
| NaCl | 0.1 | 5.85 | ±0.008 |
| 0.3 | 6.54 | ±0.035 | |
| 0.5 | 6.65 | ±0.005 | |
| 0.7 | 6.99 | ±0.017 | |
| 0.9 | 3.85 | ±0.008 | |
| KH2PO4 | 0.1 | 4.53 | ±0.116 |
| 0.3 | 4.94 | ±0.000 | |
| 0.5 | 6.82 | ±0.030 | |
| 0.7 | 6.05 | ±0.005 | |
| 0.9 | 5.03 | ±0.026 | |
| CaCl2 | 0.1 | 6.88 | ±0.020 |
| 0.3 | 7.11 | ±0.035 | |
| 0.5 | 6.89 | ±0.008 | |
| 0.7 | 5.98 | ±0.017 | |
| 0.9 | 3.22 | ±0.030 |
Bold value shows that 0.3% CaCl2 has shown maximum laccase production as compared to all other used metals.
Results of ANOVA for supplementary metal ions (with different concentrations) on laccase production.
| Source | DF | SS | MS | F | p |
|---|---|---|---|---|---|
| C1 | 4 | 15.912 | 3.978 | 6.18 | 0.004 |
| Error | 15 | 9.651 | 0.643 | ||
| Total | 19 | 25.563 |
DF, degree of freedom; SS, sum of squares within groups; MS, mean square; p-value, (calculated probability); F Value, F-Statistics.
SDS-PAGE
A novel monomeric protein having approximately 51 kDa molecular weight obtained when purified laccase was resolved on SDS-PAGE (Figure 7). Enzyme was single polypeptide protein as SDS-PAGE depicted. This characterized enzyme was little bit similar to Koli et al. [36], who investigated laccase production from purpureus spp. They isolated and purified single polypeptide chain enzyme of 40 kDa molecular weight on SDS-PAGE. Kunamneni et al. [45] reviewed that the molecular mass of the single polypeptide chain laccase ranges from about 50 kDa to 100 kDa. From different white rot fungi, molecular weight of laccase in the range of 55–90 kDa have earlier been reported [13], [47], [48], [49], [50].
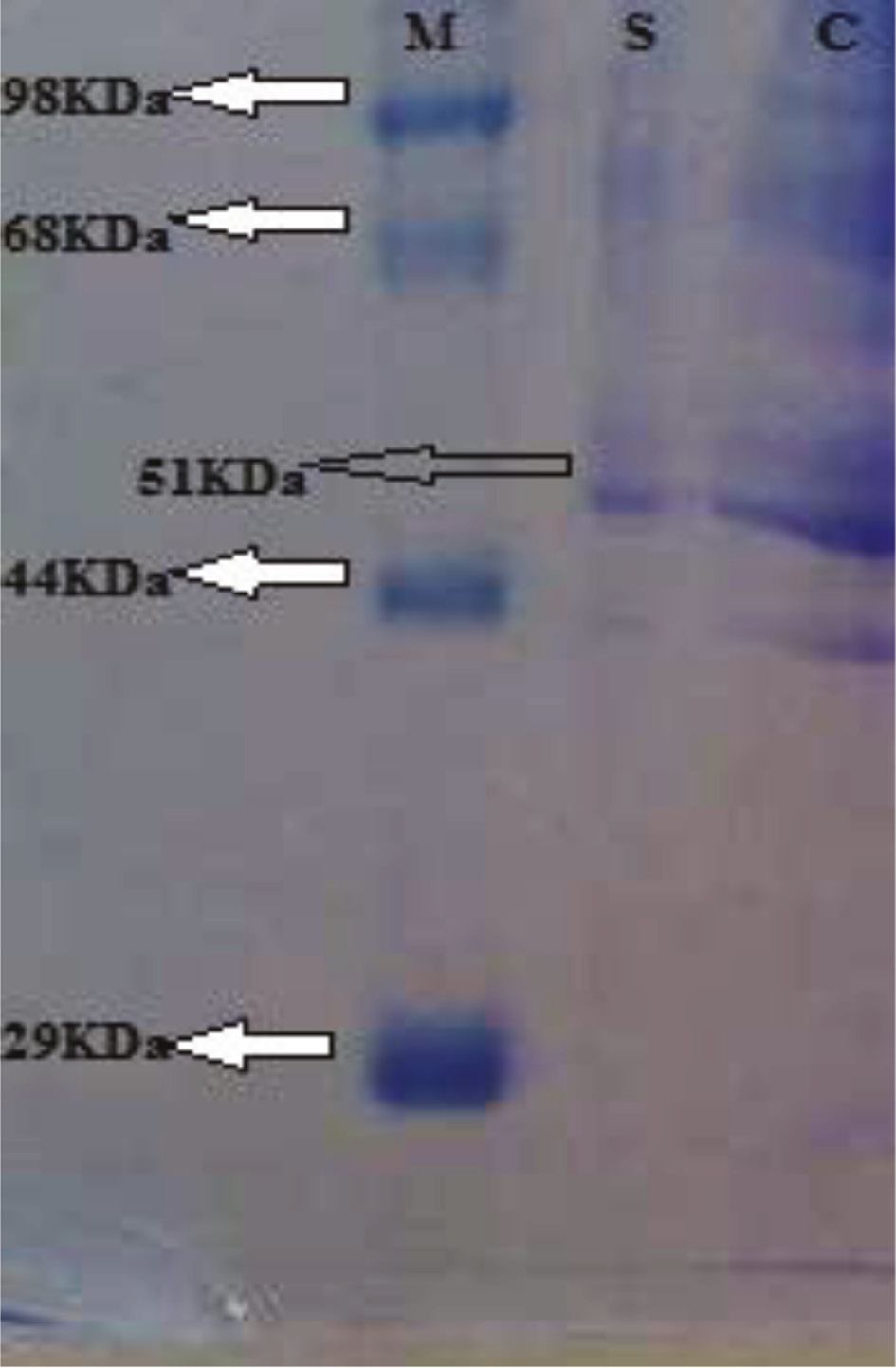
Molecular weight assessment of partially purified laccase by SDS-PAGE.
Lane M, molecular weights of standard marker; Lane S, purified laccase (51 kDa); Lane C, crude enzyme.
Conclusion
Present research work illustrated that not only basidiomycete fungi produce laccase, rather ascomyceteous fungus also produce significant amount of laccase if these microorganism are provided with suitable conditions. Alternaria alternata and Sarkanda grass (agro-waste) is easily available in Pakistan. So instead of investing huge amount of money on import of laccase, this enzyme can be produced by cheap methods as described in this study. One of the main problems of our country is inappropriate management of industrial wastes. In these circumstances laccase can be a better option for this problem. As laccase also utilized in cancer drug development, present research will also helpful in the form of futuristic cancer drugs production. This enzyme can be produced at pilot scale level by further in-depth analysis of physical and chemical parameters. Fungus can be genetically engineered for boosting the enzyme production and environmental/nutritional parameters can be optimized for industrial scale production of enzyme.
Conflict of interest: The authors have no conflict of interest.
References
1. Mani P, Keshavarz T, Chandra TS, Kyazze G. Decolourisation of acid orange 7 in a microbial fuel cell with a laccase-based biocathode: Influence of mitigating pH changes in the cathode chamber. Enzyme Microb Technol 2017;96:170–6.10.1016/j.enzmictec.2016.10.012Suche in Google Scholar
2. Shraddha R, Shekher R, Sehgal S, Kamthania M, Kumar A. Laccase: microbial sources, production, purification, and potential biotechnological applications. Enzyme Res 2011;2011:217861.10.4061/2011/217861Suche in Google Scholar
3. Schroyen M, Van Hulle SW, Holemans S, Vervaeren H, Raes K. Laccase enzyme detoxifies hydrolysates and improves biogas production from hemp straw and miscanthus. Bioresour Technol 2017;244:597–604.10.1016/j.biortech.2017.07.137Suche in Google Scholar
4. Cristovao RO, Tavares AP, Ribeiro A, Loureiro JM, Boaventura RA, Macedo EA. Kinetic modelling and simulation of laccase catalysed degradation of reactive textile dyes. Bioresour Technol 2008;99:4768–74.10.1016/j.biortech.2007.09.059Suche in Google Scholar
5. Tavares AP, Cristovao RO, Gamelas JA, Loureiro JM, Boaventuraa RA, Macedoa EA. Sequential decolourization of reactive textile dyes by laccase mediator system. J Chem Technol Biotechnol 2009;84:442–6.10.1002/jctb.2060Suche in Google Scholar
6. Shi Y, Chai L, Tang C, Yang Z, Zhang H, Chen R, et al. Characterization and genomic analysis of kraft lignin biodegradation by the beta-proteobacterium Cupriavidus basilensis B-8. Biotechnol Biofuels 2013;6:1.10.1186/1754-6834-6-1Suche in Google Scholar
7. Le T, Murugesan K, Lee C, Vu C, Chang Y, Jeon J. Degradation of synthetic pollutants in real wastewater using laccase encapsulated in core–shell magnetic copper alginate beads. Bioresour Technol 2016;216:203–10.10.1016/j.biortech.2016.05.077Suche in Google Scholar
8. Sitarz AK, Mikkelsen JD, Meyer AS. Structure, functionality and tuning up of laccases for lignocellulose and other industrial applications. Crit Rev Biotechnol 2016;36:70–86.10.3109/07388551.2014.949617Suche in Google Scholar
9. D’Souza TM, Boominathan K, Reddy CA. Isolation of laccase gene-specific sequences from white rot and brown rot fungi by PCR. Appl Environ Microbiol 1996;62:3739–44.10.1128/aem.62.10.3739-3744.1996Suche in Google Scholar
10. Asgher M, Iqbal HM, Asad MJ. Kinetic characterization of purified laccase produced from Trametes versicolor IBL-04 in solid state bio-processing of corncobs. BioResources 2012;7:1171–88.10.15376/biores.7.1.1171-1188Suche in Google Scholar
11. Atalla MM, Zeinab HK, Eman RH, Amani AY, Abeer AA. Screening of some marine-derived fungal isolates for lignin degrading enzymes (LDEs) production. Agric Biol J N Am 2010;1:591–9.Suche in Google Scholar
12. Setti L, Giuliani S, Spinozzi G, Pifferi PG. Laccase catalyzedoxidative coupling of 3-methyl 2-benzothiazolinone hydrazine and methoxyphenols. Enzyme Microb Technol 1999;25:285–9.10.1016/S0141-0229(99)00059-9Suche in Google Scholar
13. Yaropolov AI, Skorobogatko OV, Vartanov SS, Varfolomeyev SD. Laccase: properties, catalytic mechanism and applicability. Appl Biochem Biotechnol 1994;49:257–80.10.1007/BF02783061Suche in Google Scholar
14. Burton S. Laccases and phenol oxidases in organic synthesis. Curr Org Chem 2002;7:1317–31.10.2174/1385272033486477Suche in Google Scholar
15. Arora DS, Sharma RK. Ligninolytic fungal laccases and their biotechnological applications. Appl Biochem Biotechnol 2010;160:1760–88.10.1007/s12010-009-8676-ySuche in Google Scholar PubMed
16. Yang LR, Wang F, Guo C, Liu CZ. Magnetic mesoporous silica nanoparticles: fabrication and their laccase immobilization performance. Bioresour Technol 2010;101:8931–5.10.1016/j.biortech.2010.06.115Suche in Google Scholar PubMed
17. Shieh CH, Barnett SM, Hira AU. Production of enzymes and single cell protein from rice hulls. In: Linko P, Larinkari J, editors. Food Processing Engineering Vol 2. Enzyme Engineering in Food Processing. London: Applied Science Publishers, 1980:289–94.10.1007/978-94-011-7795-5_32Suche in Google Scholar
18. Walker JR, Ferrar PH. Inhibition of diphenol oxidases: a comparative study. J Food Biochem 1996;20:15–30.10.1111/j.1745-4514.1996.tb00582.xSuche in Google Scholar
19. Assavanig A, Amornkitticharoen B, Ekpaisal N, Meevootisom V, Flegel TW. Isolation, characterization and function of laccase from Trichoderma. Appl Microbiol Biotech 1992;38:198–202.10.1007/BF00174468Suche in Google Scholar
20. Hildén K, Hakala TK, Lundell T. Thermotolerant and thermostable laccases. Biotechnol Lett 2009;31:1117–28.10.1007/s10529-009-9998-0Suche in Google Scholar PubMed
21. Zeng XK, Cai YJ, Liao XR, Zeng XL, Li WX, Zhang DB. Decolorization of synthetic dyes by crude laccase from a newly isolated trametes trogii strain cultivated on solid agro-industrial residue. J Hazard Mater 2011;187:517–25.10.1016/j.jhazmat.2011.01.068Suche in Google Scholar PubMed
22. Kent KT. Effects of microorganisms on lignin. Annu Rev Phytopathol 1971;9:185–210.10.1146/annurev.py.09.090171.001153Suche in Google Scholar
23. Froehner SC, Eriksson KE. Purification and properties of Neurospora crassa laccase. J Bacteriol 1974;120:458–65.10.1128/jb.120.1.458-465.1974Suche in Google Scholar PubMed PubMed Central
24. Thakker GD, Evans CS, Koteswara KR. Purification and characterization of laccase from Monocillium indicum Saxena. Appl Microbiol Biotechnol 1992;37:321–3.10.1007/BF00210986Suche in Google Scholar
25. Sun J, Guo N, Niu LL, Wang QF, Zang YP, Zu YG, et al. Production of laccase by a new myrothecium verrucaria MD-R-16 isolated from Pigeon Pea [Cajanus cajan (L.) Millsp.] and its application on dye decolorization. Molecules 2017;22:673.10.3390/molecules22040673Suche in Google Scholar
26. Das A, Singh J, Yogalakshmi KN. Laccase immobilized magnetic iron nanoparticles: Fabrication and its performance evaluation in chlorpyrifos degradation. Int Biodeterior Biodegradation 2017;117:183–9.10.1016/j.ibiod.2017.01.007Suche in Google Scholar
27. Oguntinein G, Vlach D, Moo-Young M. Production of celluloytic enzymes by Neurospora sitophila grown on cellulosic materials. Bioresour Technol 1992;39:277–83.10.1016/0960-8524(92)90217-LSuche in Google Scholar
28. Pointing SB, Pelling AL, Smith GJ, Hyde KD, Reddy CA. Screening of basidiomycetes and xylariaceous fungi for lignin peroxidase and laccase gene-specific sequences. Mycol Res 2005;109: 115–24.10.1017/S0953756204001376Suche in Google Scholar
29. Irshad M, Asgher M, Sheikh MA, Nawaz H. Purification and characterization of laccase produced by Schyzophylum commune IBL-06 in solid state culture of banana stalks. BioResources 2011;6:2861–73.10.15376/biores.6.3.2861-2873Suche in Google Scholar
30. Wilson DK, Rudolph FB, Quiocho FA. Atomic structure of adenosine deaminase complexed with a transition-state analog: understanding catalysis and immunodeficiency mutations. Science 1991;252:1278–85.10.1126/science.1925539Suche in Google Scholar
31. Green AA, Hughes W. Protein fractionation on the basis of solubility in aqueous solutions of salts and organic solvents. Methods Enzymol 1955;1:67–90.10.1016/0076-6879(55)01014-8Suche in Google Scholar
32. Layman P. Promising new markets emerging for commercial enzymes. Chem Eng News 1990;68:17–8.10.1021/cen-v068n039.p017Suche in Google Scholar
33. Elisashvili V, Penninckx M, Kachlishvili E, Asatiani M, Kvestiadze G. Use of Pleurotus dryinus for lignocellulolytic enzymes production in submerged fermentation of mandarin peels and tree leaves. Enzyme Microb Technol 2006;38:998–1004.10.1016/j.enzmictec.2005.08.033Suche in Google Scholar
34. Moldes D, Lorenzo M, Sanroman MA. Di Verent proportions of laccase isoenzymes produced by submerged cultures of Trametes versicolor grown on lignocellulosic wastes. Biotechnol Lett 2004;26:327–30.10.1023/B:BILE.0000015452.40213.bfSuche in Google Scholar
35. Nandal P, Ravella SR, Kuhad RC. Laccase production by Coriolopsis caperata RCK2011: optimization under solid state fermentation by Taguchi DOE methodology. Sci Rep 2013;3:1386–93.10.1038/srep01386Suche in Google Scholar PubMed PubMed Central
36. Koli SH, Suryawanshi RK, Patil CD, Patil SV. Fluconazole treatment enhances extracellular release of red pigments in the fungus Monascus purpureus. FEMS Microbiol Lett 2017;364:FNX058.10.1093/femsle/fnx058Suche in Google Scholar PubMed
37. Balaraju K, Park K, Jahagirdar S, Kaviyarasan V. Production of cellulase and laccase enzymes by Oudemansiella radicata using agro wastes under solid-state and submerged conditions. Res Biotechnol 2010;1:21–8.Suche in Google Scholar
38. Gaitan IJ, Medina SC, González JC, Rodríguez A, Espejo ÁJ, Osma JF, et al. Evaluation of toxicity and degradation of a chlorophenol mixture by the laccase produced by Trametes pubescens. Bioresour Technol 2011;102:3632–5.10.1016/j.biortech.2010.11.040Suche in Google Scholar PubMed
39. Johnsy G, Kaviyarasan V. Effect of nutritional and environmental conditions on production of extracellular laccase under submerged culture conditions in Lentinus kauffmanii. J Curr Pharm Res 2011;3:105–9.Suche in Google Scholar
40. Elshafei AM, Hassan MM, Haroun BM, Elsayed MA, Othman AM. Optimization of Laccase Production from Penicillium martensii NRC 345. Adv Life Sci 2012;2:31–7.10.5923/j.als.20120201.05Suche in Google Scholar
41. Elsayed MA, Hassan MM, Elshafei AM, Haroun BM, Othman AM. Optimization of cultural and nutritional parameters for the production of laccase by Pleurotus ostreatus ARC280. Br Biotechnol J 2012;2;115.10.9734/BBJ/2012/1305Suche in Google Scholar
42. Revankar MS, Lele SS. Enhanced production of laccase using a new isolate of white rot fungus WR-1. Process Biochem 2006;41:581–8.10.1016/j.procbio.2005.07.019Suche in Google Scholar
43. D’Agostini ÉC, Mantovani TR, Valle JS, Paccola-Meirelles LD, Colauto NB, Linde GA. Low carbon/nitrogen ratio increases laccase production from basidiomycetes in solid substrate cultivation. Scientia Agricola 2011;68:295–300.10.1590/S0103-90162011000300004Suche in Google Scholar
44. Mahmoud MG, Rifat HM, Sayed EI, Beih FM, Selim MS. Effect of inducers and process parameters on laccase production by locally isolated marine Streptomyces lydicus from Red Sea, Egypt. Int J Chem Tech Res 2013;5:15–23.Suche in Google Scholar
45. Kunamneni A, Ballesteros A, Plou FJ, Alcalde M. Fungal laccase – a versatile enzyme for biotechnological applications. Commun Curr Res Educ Top Trends Appl Microbiol 2007;1:233–45.Suche in Google Scholar
46. Vetchinkina EP, Loshchinina EA, Vodolazov IR, Kursky VF, Dykman LA, Nikitina VE. Biosynthesis of nanoparticles of metals and metalloids by basidiomycetes. Preparation of gold nanoparticles by using purified fungal phenol oxidases. Appl Microbiol Biotechnol 2017;101;1047–62.10.1007/s00253-016-7893-xSuche in Google Scholar PubMed
47. Murugesan K, Nam IH, Kim YM, Chang YS. Decolorization of reactive dyes by a thermostable laccase produced by Ganoderma lucidum in solid state culture. Enzyme Microb Technol 2007;40:1662–72.10.1016/j.enzmictec.2006.08.028Suche in Google Scholar
48. Salony SM, Bisaria VS. Decolorization and detoxification of textile dyes and black liquor by laccase of Cyathus bulleri. J Sci Ind Res 2007;66:684–9.Suche in Google Scholar
49. Quaratino D, Federici F, Petruccioli M, Fenice M, D’Annibale A. Production, purification and partial characterisation of a novel laccase from the white-rot fungus Panus tigrinus CBS 577.79. Antonie van Leeuwenhoek 2007;91:57–69.10.1007/s10482-006-9096-4Suche in Google Scholar PubMed
50. Couto SR, Herrera JL. Industrial and biotechnological applications of laccases: a review. Biotechnol Adv 2006;24:500–13.10.1016/j.biotechadv.2006.04.003Suche in Google Scholar PubMed
©2018 Walter de Gruyter GmbH, Berlin/Boston
Artikel in diesem Heft
- Frontmatter
- Research Articles
- Effects of calcium hydroxide and N-acetylcysteine on MMP-2, MMP-9, TIMP-1 and TIMP-2 in LPS-stimulated macrophage cell lines
- Synthesis of fused 1,4-dihydropyridines as potential calcium channel blockers
- Optimization of fermentation conditions for efficient ethanol production by Mucor hiemalis
- Covalent immobilization of an alkaline protease from Bacillus licheniformis
- Major biological activities and protein profiles of skin secretions of Lissotriton vulgaris and Triturus ivanbureschi
- Optimized production, purification and molecular characterization of fungal laccase through Alternaria alternata
- Adsorption of methyl violet from aqueous solution using brown algae Padina sanctae-crucis
- Protective effect of dexpanthenol (vitamin B5) in a rat model of LPS-induced endotoxic shock
- Purification and biochemical characterization of a β-cyanoalanine synthase expressed in germinating seeds of Sorghum bicolor (L.) moench
- Molecular cloning and in silico characterization of two alpha-like neurotoxins and one metalloproteinase from the maxilllipeds of the centipede Scolopendra subspinipes mutilans
- Improvement of delta-endotoxin production from local Bacillus thuringiensis Se13 using Taguchi’s orthogonal array methodology
- Enhancing vitamin B12 content in co-fermented soy-milk via a Lotka Volterra model
- Species and number of bacterium may alternate IL-1β levels in the odontogenic cyst fluid
- Rheo-chemical characterization of exopolysaccharides produced by plant growth promoting rhizobacteria
- Benzo(a)pyrene degradation pathway in Bacillus subtilis BMT4i (MTCC 9447)
- Indices
- Reviewers 2018
- Yazar Dizini/Author Index
Artikel in diesem Heft
- Frontmatter
- Research Articles
- Effects of calcium hydroxide and N-acetylcysteine on MMP-2, MMP-9, TIMP-1 and TIMP-2 in LPS-stimulated macrophage cell lines
- Synthesis of fused 1,4-dihydropyridines as potential calcium channel blockers
- Optimization of fermentation conditions for efficient ethanol production by Mucor hiemalis
- Covalent immobilization of an alkaline protease from Bacillus licheniformis
- Major biological activities and protein profiles of skin secretions of Lissotriton vulgaris and Triturus ivanbureschi
- Optimized production, purification and molecular characterization of fungal laccase through Alternaria alternata
- Adsorption of methyl violet from aqueous solution using brown algae Padina sanctae-crucis
- Protective effect of dexpanthenol (vitamin B5) in a rat model of LPS-induced endotoxic shock
- Purification and biochemical characterization of a β-cyanoalanine synthase expressed in germinating seeds of Sorghum bicolor (L.) moench
- Molecular cloning and in silico characterization of two alpha-like neurotoxins and one metalloproteinase from the maxilllipeds of the centipede Scolopendra subspinipes mutilans
- Improvement of delta-endotoxin production from local Bacillus thuringiensis Se13 using Taguchi’s orthogonal array methodology
- Enhancing vitamin B12 content in co-fermented soy-milk via a Lotka Volterra model
- Species and number of bacterium may alternate IL-1β levels in the odontogenic cyst fluid
- Rheo-chemical characterization of exopolysaccharides produced by plant growth promoting rhizobacteria
- Benzo(a)pyrene degradation pathway in Bacillus subtilis BMT4i (MTCC 9447)
- Indices
- Reviewers 2018
- Yazar Dizini/Author Index

