Abstract
Objectives
Post-mortem magnetic resonance imaging (pmMRI) has emerged as a promising, non-invasive alternative to conventional autopsy for detecting cardiac anomalies in fetal and perinatal deaths and we aimed to systematically evaluate the diagnostic accuracy of pmMRI in detecting cardiac anomalies in fetal and perinatal deaths.
Methods
A systematic search of electronic databases and grey literature was conducted following PRISMA 2020 guidelines to analyse studies comparing pmMRI findings to conventional autopsy. Data extraction and quality assessment were independently performed by two reviewers using the QUADAS-2 tool. Meta-analysis was conducted using a bivariate random-effects model to calculate pooled sensitivity, specificity, and diagnostic odds ratios (DOR).
Results
Sixteen studies were included in the systematic review, and 12 studies (n=1810 fetuses) were meta-analyzed. The pooled sensitivity and specificity of pmMRI for detecting cardiac anomalies were 76 % (95 % CI: 71–80 %) and 96 % (95 % CI: 95–97 %), respectively. The diagnostic odds ratio was 55.35 (95 % CI: 22.73–134.79), with an area under the SROC curve of 0.89, indicating excellent diagnostic performance. Subgroup analyses showed comparable sensitivity between 1.5 and 3 T scanners, though specificity was slightly higher for 1.5 T. Diagnostic accuracy was generally better in larger fetuses and when higher field strength MRI (9.4 T) was used.
Conclusions
pmMRI demonstrates high specificity and moderate sensitivity for detecting cardiac anomalies in fetal and perinatal deaths and may serve as a valuable non-invasive adjunct to conventional autopsy. Standardization of imaging protocols and further research into high-field pmMRI integration are recommended to optimize diagnostic reliability.
Introduction
Congenital heart disease (CHD) is the most frequently diagnosed birth defect, occurring in about 8–9.5 per 1,000 live births worldwide, although regional estimates range from 1.2 to 17 per 1,000 [1]. As one of the leading categories of congenital anomalies, CHD contributes substantially to fetal and perinatal mortality across the globe [2], 3]. Although conventional autopsy has been considered the gold standard for diagnosing structural cardiac anomalies after fetal/perinatal demise, post-mortem imaging has emerged as a promising, non-invasive alternative that may provide valuable diagnostic information while mitigating cultural, emotional, and religious concerns associated with conventional autopsy. A Norwegian study of 67 fetuses with isolated CHDs demonstrated high accuracy of prenatal ultrasound, but emphasized that post-mortem verification remains essential for quality assurance and parental counselling [4]. The study also highlighted the increasing difficulty of conducting conventional autopsies due to a trend toward earlier pregnancy terminations, particularly in the first trimester, creating a need for reliable, non-invasive post-mortem techniques for smaller fetal specimens.
Modalities such as post-mortem magnetic resonance imaging (pmMRI), computed tomography, and ultrasound are increasingly being integrated into fetal/perinatal death investigations [5]. Despite the growing use of post-mortem imaging, the diagnostic yield and accuracy of these techniques, particularly for detecting cardiac anomalies, remain incompletely defined. The heterogeneity in study populations, imaging protocols, reference standards, and outcome measures has made it challenging to draw definitive conclusions about the overall diagnostic yield of post-mortem imaging in this context. Given the critical role of accurate post-mortem diagnosis in guiding genetic counselling, understanding recurrence risk and informing future pregnancy management, it is essential to systematically evaluate the diagnostic performance of post-mortem imaging for cardiac anomalies [6]. This systematic review and meta-analysis aims to comprehensively assess the diagnostic accuracy of pmMRI in identifying cardiac anomalies in fetal and perinatal deaths and explore factors influencing diagnostic performance.
Methods
This study was conducted following the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) 2020 guidelines and aimed to assess the diagnostic accuracy of pmMRI in detecting cardiac anomalies in fetal/perinatal deaths, using conventional autopsy as the reference standard [7]. The review protocol was prospectively registered on the Open Science Framework registry (accessible at https://osf.io/dafcj/). A comprehensive literature search was carried out across major electronic databases using controlled vocabulary terms and free-text keywords related to “post-mortem imaging”, “autopsy”, “perinatal autopsy”, “fetal autopsy”, “magnetic resonance imaging”, “cardiac anomalies” and “diagnostic yield”. The detailed search string is presented in Supplementary Table 1. Additionally, grey literature sources such as Google Scholar, conference proceedings and reference lists of relevant articles were screened to ensure the inclusion of all pertinent studies. Diagnostic accuracy studies, cohort studies, and cross-sectional studies that directly compared pmMRI findings with autopsy results were included. The target population included fetal deaths (≥12 weeks of gestation) and perinatal deaths, defined as deaths occurring up to seven days postnatal. Studies were excluded if they focused exclusively on adult populations, did not report cardiac anomaly outcomes, utilized imaging modalities other than pmMRI, or provided insufficient data for calculating diagnostic outcomes.
The study screening process was performed using the open-access Rayyan software (https://www.rayyan.ai). After the removal of duplicates, two independent reviewers (SP, MM) screened the titles and abstracts for potential eligibility. Full-text articles from selected studies were then retrieved and evaluated against the inclusion criteria by two reviewers (SP, MM). Disagreements were resolved through discussion, and when consensus could not be reached, a third reviewer (AS) was consulted. Data from the included studies was independently extracted by two reviewers (MoM, PKK) using a pre-designed data extraction form, and cross-verified by the other two reviewers (SP, MM). The information extracted from each included study comprised of study characteristics (authors, publication year, country of origin, study design, sample size, etc.), population characteristics (gestational age range, birth weight, cause of death (when reported), demographic characteristics), imaging parameters (type of MRI equipment used, technical parameters, timing of imaging relative to death, imaging protocol, and interpreter experience level), outcome measures and any other relevant parameters.
The methodological quality and risk of bias of the included studies were assessed using the Quality Assessment of Diagnostic Accuracy Studies (QUADAS-2) tool [8]. ROBVIS (https://mcguinlu.shinyapps.io/robvis/) was utilized to generate the traffic light and the summary plots. Meta-analysis using a bivariate random-effects model was performed using Meta-Disc® (version 1.4, XI Cochrane Colloquium, Spain) [9] to calculate pooled estimates of sensitivity, specificity, and diagnostic odds ratios along with plotting of summary receiver operating characteristic (SROC) curves. Statistical heterogeneity was assessed using the I2 statistic and Cochran’s Q test, and potential sources of heterogeneity were explored through subgroup analyses. Values of positive predictive value (PPV), negative predictive value (NPV), and diagnostic odds ratio (DOR), if not reported, were derived using MedCalc Statistical Software version 19.2.6 [10]. Potential publication bias was assessed using funnel plot asymmetry.
Results
Details of the articles’ selection process are presented in the PRISMA flowchart (Figure 1). A total of 551 studies were retrieved from databases (including grey literature) and after manual removal of 301 duplicates, 240 studies were screened with abstracts and titles, and 49 full texts were retrieved. After the full-text screening, 16 studies were finally included in the systematic review, and results from 12 studies were pooled for the meta-analysis. The population characteristics, diagnostic metrics and pmMRI parameters of the included studies are summarized in Table 1 [11], [12], [13], [14], [15], [16], [17], [18], [19], [20], [21], [22], [23], [24], [25], [26], Supplementary Tables 2 and 3 respectively.

PRISMA flow-chart.
Characteristics of included studies on accuracy of pmMRI in detecting cardiac anomalies in fetal/perinatal loss [11], [12], [13], [14], [15], [16], [17], [18], [19], [20], [21], [22], [23], [24], [25], [26].
| Author, year | Country | Study design | Fetuses evaluated by both pmMRI and autopsy | Mode of fetal loss | Mean fetal weight at evaluation | Gestational age range (mean or median) | Delivery to pmMRI interval | Delivery to autopsy interval | MRI field strength & equipment | Expertise and experience of the radiologist | Organ system(s) evaluated | Key points/remarks | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | Gupta et al. [11] | India | Prospective | 162 | TOP, stillbirths | NM | 15–40.5 weeks (mean: 24 weeks) | Median: 24.6 h (0.5–119 h) | Median: 27.3 h (2–145 h) | 1.5 T (Achieva 1.5 T, Philips) with MIA | Two radiologists (8 years) | Brain, cardiovascular, pulmonary, abdominal, genitourinary, musculoskeletal | 6 excluded due to technical issues |
| 2 | Ruegger et al. [12] | Switzerland | Prospective | 84 | TOP, stillbirths | Median: 520 g | Range not specified (median: 23.4 weeks) | Mean 49 h (±29) | Mean 78 h (±32) | 3 T (Achieva, Philips), with robotic CT-guided biopsy | One forensic radiologist (10 years) | Brain, cardiovascular, pulmonary, gastrointestinal, genitourinary, musculoskeletal | Overcalls upon cardiac pmMRI included, VSD [3], ASD [2] and one case each of AVSD, coarctation, complex CHD, hypoplastic left heart syndrome, partial anomalous venous drainage, biventricular hypertrophy |
| 3 | Tang et al. a[13] | United Kingdom | Prospective | 8 | TOP | NM | 12+6–18+0 weeks (median: 13+4 weeks) | >2 weeks (performed on formalin fixed ex vivo organs) | NM | 9.4 T (Bruker Biospec system, Bruker Biospin) | Radiologist + cardiothoracic surgeon (15 years) | Cardiovascular system |
|
| 4 | Staicu et al. [14] | Romania | Prospective | 32 (8 with cardiac evaluation) | TOP | Mean: 433.77 g (range: 180–981 g) | 18–23 weeks (mean: 20.5 weeks) | Same/next day (exact time range NM) | Same/next day (exact time range NM) | 3 T (Discovery MR750w GEM, GE Healthcare) | One pediatric radiologist | Nervous, cardiac, vascular, respiratory, gastrointestinal, renal, skeletal, facial abnormalities | In the study cohort, out of eight cardiac abnormalities identified by prenatal ultrasound, 5 were confirmed by pmMRI |
| 5 | Shelmerdine et al. [15] | United Kingdom | Prospective | 88 | TOP, IUFD, miscarriage, neonatal deaths | Range: 56–4060 g | 26–29 weeks | 10 (0–35) | 12 (4–47) | 1.5 T (Avanto, Siemens) and US machine (UGEO HM70A, Samsung) | MRI: pediatric radiologist; USG: pediatric radiology research fellow |
Brain, cardiovascular, thoracic, abdominal | For cardiac anomalies, pmUS missed 4 diagnoses (one case each of dilated cardiomyopathy, DORV, cardiac hypertrophy, dysplastic pulmonary valve, cardiomegaly) and overcalled one case of VSD, while pmMRI missed two cardiac anomalies (one case each of cardiomegaly and VSD) and overcalled one case of DORV |
| 6 | Ulm et al. [16] | Austria | Retrospective | 219 | TOP, IUFD, neonatal death | Mean: 444 g (range: 10–3800 g) | 12–41 weeks (mean: 22 weeks) | 0–24 h | 24–48 h | 3 T (Magnetom Trio and Magnetom Vida, Siemens) | 2 radiologists experienced in cardiac imaging (2 years) | Cardiovascular system | Concluded that pmMRI can detect CHD at any gestational age, with diagnostic rates high in fetuses with a birthweight of >100 g and gestational age of >20 weeks |
| 7 | Shelmerdine et al. [17] | United Kingdom | Retrospective | 81 | TOP, miscarriages, stillbirths | Median: 346 g (range: 16–3480 g) | 14–41 weeks (median: 21 weeks) | Median: 6 days (range 0–36 days) | Median: 9 days (range 1–42 days) | 1.5 scanner (Avanto, Siemens) | MRI – pediatric radiologist (>6 years), USG- fetal medicine & genetic specialist | Brain, cardiovascular, thoracic, abdominal, musculoskeletal | pmMR had a significantly higher concordance Rate with autopsy than ultrasound (89.0 vs. 93.8 %; p<0.001). |
| 8 | Kang et al. [18] | Belgium | Prospective | 118 (80 with cardiac evaluation) | TOP, miscarriages, stillbirths | NM | 13–41 weeks | Median 1 day (range 0–7) | Median 4 days (range 0–7) | 3 T (Philips Achieva, Philips), and US machine (Voluson E8, GE Healthcare) | MRI – radiologists, USG – fetal medicine experts | Brain, cardiovascular, thoracic, abdominal, spinal |
|
| 9 | Shruthi et al. [19] | India | Prospective | 516 | TOP, stillbirths | Range: 210–2,960 g | ≥20 weeks (mean: 25 weeks) | 67.4 % within 5 days; all within 22 days | 48.8 % within 30 days; all within 49 days | 1.5 T (Achieva, Philips) | 3 radiologists experienced in fetal medicine (18 years/12 years/7 years) | Nervous, cardiovascular, gastrointestinal, renal, musculoskeletal | With regard to malformations of the cardiovascular system, pmMRI provided partial diagnosis of complex CHD in six cases |
| 10 | Ashwin et al. [20] | United Kingdom | Retrospective | 201 (180 with cardiac evaluation) | TOP, stillbirths, neonatal deaths | NM | Range not specified (Mean: 25.1±7.7 weeks) | Mean: ∼6.6 days | NM | 1.5 T scanner (Avanto, Siemens) | One pediatric radiologist | Brain and spine, cardiovascular, thoracic, abdominal, musculoskeletal | Upon pmMRI, there were 5 overcalls (3 RVOT anomalies, 1 Large pericardial effusion, 1 dilated heart) and 8 misses (including 3 small VSD, 1 RVOT anomalies/Fallot, 1 Dilated heart, 1 PAPVD) |
| 11 | Kang et al. [21] | Belgium | Prospective | 92 | TOP, miscarriage, stillbirths | NM | 12.0–41.1 weeks (mean: 25.0 weeks) | 0–5 days | 0–7 days | 1.5 T (Avanto, Siemens); 3 T (Achieva, Philips) | 2 radiologists (10 years) | Brain, spine, cardiovascular, thoracic, abdominal | Diagnostic error was lower for 3 T than for 1.5 T pmMRI for all anatomic structures and organ groups, mainly for fetuses <20 weeks, particularly for thoracic and abdominal organs |
| 12 | Taylor et al. a[22] | United Kingdom | Prospective | 277 | TOP, miscarriage, stillbirths | NM | 22–30 weeks | NM | NM | 1.5 T (Avanto, Siemens) | Pediatric radiologist (12 years) | Cardiovascular system |
|
| 13 | Sandaite et al. a[23] | Belgium | Retrospective | 24 | TOP | NM | 12+5–34+6 (median: 22 weeks) | Median 6 h and 30 min (range 1 h and 30 min–19 h) | NM | 3 T (Magnetom Trio, Siemens) | Cardiac radiologist (10 years) | Cardiovascular system |
|
| 14 | Votino et al. [24] | Belgium | Prospective | 24 | TOP | NM | ≤20 weeks | NM | NM | 9.4 T (Bruker Biospec system, Bruker Biospin) | Single radiologist with fetal MRI expertise (10 years) | Cardiovascular system | High-field MRI at 9.4 T seems to be an acceptable alternative approach to invasive stereomicroscopic autopsy for fetuses with CHD at or below 20 weeks’ gestation |
| 15 | Cannie et al. a[25] | Belgium | Prospective | 33 | TOP, IUFD | NM | ≥20 weeks | NM | NM | 1.5 T (Avanto, Siemens); CT scanner (GE 64-detector, GE Healthcare) | Single radiologist | Whole body and multiple organ systems (head, thorax, abdomen, etc.) | Confidence score of pmMRI ranged from 50 to 100 for detecting cardiovascular anomalies |
| 16 | Breeze et al. a[26] | United Kingdom | Prospective | 44 | TOP, IUFD, miscarriages | Range: 61–3270 g | 16–40 weeks (median: 21.5 weeks) | NM | NM | 1.5 T (GE signa, GE Healthcare) with percutaneous organ biopsies, using 16-18 G biopsy needle | Radiologist with pediatric neuroradiology expertise | Liver, lung, heart, spleen, kidney adrenal, and thymus | Out of the 10 cases, where conventional autopsy provided superior diagnostic information to that of MIA, there were 4 cases of cardiac anomalies |
-
aStudies included in systematic review alone. ASD, atrial septal defect; AVSD, atrioventricular septal defect; CHD, congenital heart disease; CT, computer tomography; DORV, double-outlet right ventricle; MRI, magnetic resonance imaging; MIA, minimally invasive autopsy; NM, not mentioned; pmMRI, post-mortem MRI; PAPVD, partial anomalous pulmonary venous drainage; RVOT, right ventricular outflow tract; T, tesla; TGA, transposition of the great arteries; TOP, termination of pregnancy; US, ultrasonography; VSD, ventricular septal defect.
Systematic review/qualitative synthesis
The 16 included studies collectively provide a comprehensive overview of the diagnostic performance of pmMRI in detecting cardiac anomalies in fetal and perinatal loss. The included studies demonstrate significant geographic clustering, with the majority originating from European centres (Belgium: 4 studies, United Kingdom: 4 studies) and two studies from India. This distribution reflects the established expertise in postmortem imaging within European tertiary centres, though the growing contribution from Asian centres suggests expanding global adoption of pmMRI techniques. Sample sizes varied considerably, from as few as eight fetuses to as many as 516, introducing some heterogeneity in the precision of diagnostic accuracy estimates across studies. The gestational age of fetuses included in the studies ranged from 12 to 41 weeks, with a majority of cases falling within the second trimester. Where reported, fetal weights demonstrate considerable variation (16–4,060 g), with several studies noting improved diagnostic performance in larger fetuses.
The interval from delivery (TOP/miscarriages/stillbirths) to imaging varied widely, with some studies performing pmMRI within a few hours and others extending beyond two weeks, particularly for formalin-fixed ex vivo imaging. The time to autopsy also ranged from same-day examinations to several days post-delivery. While most studies attempted to minimize this interval, the heterogeneity in timing protocols may influence diagnostic accuracy through post-mortem tissue changes and decomposition artifacts. The imaging modalities utilized predominantly involved 1.5 Tesla (T) and 3 T MRI scanners, with select studies [13], 24] employing ultra-high field 9.4 T MRI, particularly for early gestational age fetuses and ex vivo imaging. The integration of complementary modalities, including CT-guided biopsy, demonstrates an evolving multimodal approach to minimally invasive autopsy, suggesting that pmMRI may be most effective when employed as part of a comprehensive imaging strategy rather than as a standalone diagnostic tool [12]. Radiologists involved in these studies typically had substantial experience in fetal or cardiac imaging, ranging from 2 to 15 years.
The sensitivity of pmMRI in detecting cardiac anomalies, ranged widely from 50 % (Shelmerdine et al.) to 93.2 % (Gupta et al.) [11], 17]. In contrast, specificity was consistently high across studies, typically ranging from 75 to 100 %. Several studies, including those by Kang et al. and Shruthi et al., reported perfect specificity, implying that pmMRI is highly reliable in correctly excluding cardiac anomalies in non-affected cases [18], 19], 21]. This variability suggests that factors such as imaging protocols, operator expertise, and lesion characteristics/case complexity may significantly influence diagnostic performance. The DOR, a combined measure of sensitivity and specificity, varied across studies, with the highest observed by Shelmerdine et al. and Gupta et al. [11], 15]. It is to be noted that, recent studies generally reported higher diagnostic accuracy, likely reflecting advances in imaging technology and increased radiological expertise over time. The PPV was notably high across most studies, often exceeding 70 %, and in some cases reaching 100 %, suggesting that cardiac anomalies identified on pmMRI are highly likely to be true findings. The NPV was similarly strong, typically above 85 %. However, studies like Ruegger et al. and Shruthi et al. reported significant false negatives, showing that pmMRI may still miss certain cardiac anomalies, particularly small or complex lesions [12], 19].
Meta-analysis for pooled diagnostic metrics
A total of 12 studies, comprising of data of 1810 fetuses in total, were included in the diagnostic accuracy meta-analysis wherein the pooled sensitivity of pmMRI for detecting fetal cardiac anomalies was 0.76 (95 % CI: 0.71–0.80) (Figure 2A). The pooled specificity was high at 0.96 (95 % CI: 0.95–0.97) (Figure 2B). However, significant heterogeneity was observed for both pooled sensitivity (Chi-square=37.83, p=0.0001; I2=70.9 %) and specificity (Chi-square=120.44, p<0.0001; I2=90.9 %). The SROC demonstrated a high area under the curve (AUC) of 0.89 (SE=0.045) and a Q* index of 0.82 (SE=0.047), indicating excellent overall diagnostic performance of pmMRI in detecting fetal cardiac anomalies (Figure 3). Analysis of the diagnostic threshold using the Spearman correlation coefficient revealed no significant threshold effect (r=0.063, p=0.845). Moses’ regression model yielded an intercept of 3.03 (p=0.0008) and a slope of −0.48 (p=0.062), suggesting some variation in diagnostic accuracy across studies but no clear threshold bias. The pooled DOR was 55.35 (95 % CI: 22.73–134.79) (Supplementary Figure 1). Notably, some studies such as Shruthi M et al. and Ulm et al. demonstrated extremely high DORs (>300), while others such as Staicu et al. had much lower DORs (∼5.0) [14], 16], 19]. The DOR results also showed considerable heterogeneity (Chi-square=37.25, p=0.0001; I2=70.5 %). The pooled positive likelihood ratio (PLR) was 14.06 (95 % CI: 6.61–29.89), indicating that fetuses with cardiac anomalies were approximately 14 times more likely to test positive on pmMRI than those without anomalies, while, the pooled negative likelihood ratio (NLR) was 0.32 (95 % CI: 0.24–0.43), suggesting a moderate ability to rule out cardiac anomalies when pmMRI is negative (Supplementary Figure 2). Individual NLRs ranged from 0.08 to 0.59 [11], 17]. Heterogeneity for NLR (Chi-square=28.55, p=0.0027; I2=61.5 %) was lower than that for PLR (Chi-square=66.93, p<0.0001; I2=83.6 %).

Pooled sensitivity (A) and specificity (B) of pmMRI in detecting fetal cardiac anomalies as compared to conventional autopsy.
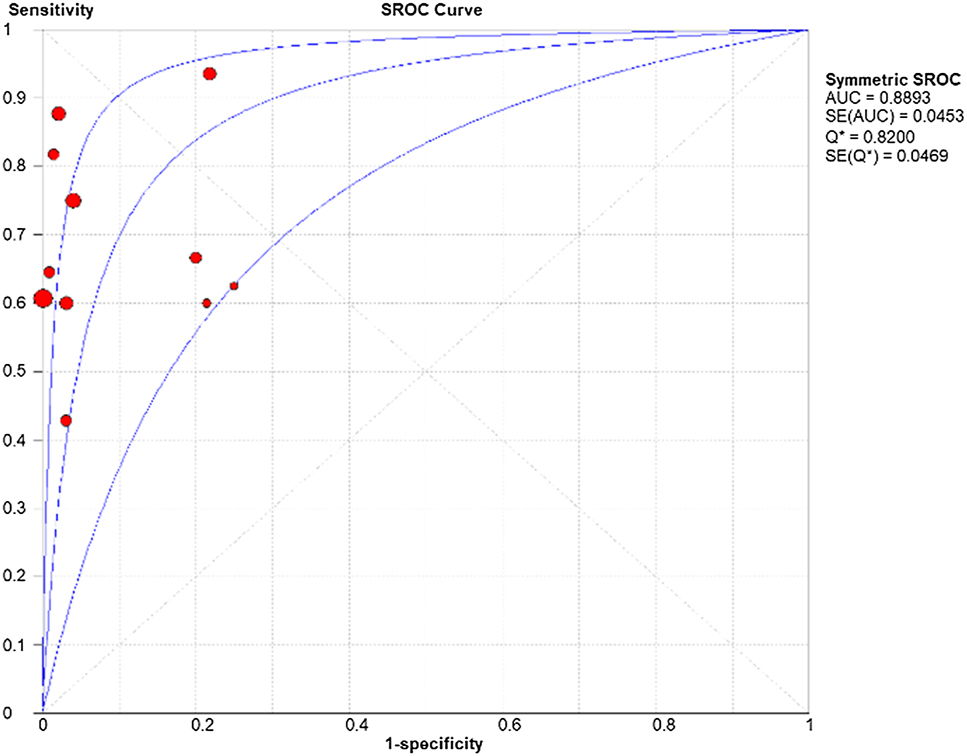
SROC of the diagnostic accuracy of pmMRI in detecting fetal cardiac anomalies.
Subgroup analysis
In the subgroup analysis of studies using 1.5 T pmMRI, seven studies were included [11], 15], 17], [19], [20], [21], [22]. The pooled sensitivity for detecting fetal cardiac anomalies was 0.76 (95 % CI: 0.70–0.82), while the pooled specificity was notably high at 0.97 (95 % CI: 0.96–0.98). The pooled PLR, was 22.50 (95 % CI: 8.23–61.54), indicating a strong ability to confirm cardiac anomalies when the test is positive, while the pooled NLR was 0.31 (95 % CI: 0.21–0.46), suggesting a moderate-to-high capacity to rule out disease in negative cases. Furthermore, the pooled DOR was 76.47 (95 % CI: 37.03–157.88), reflecting high overall diagnostic accuracy. Threshold effect analysis revealed no significant evidence of a threshold phenomenon (Spearman correlation coefficient=0.357, p=0.432). The SROC (from Moses’ model) had a non-significant slope (b=−0.158, p=0.29), supporting the absence of substantial threshold variation. Overall, the findings indicate that 1.5 T MRI offers high specificity and good diagnostic odds, though some variability in sensitivity across studies suggests the potential influence of case mix, gestational age, or imaging protocols (Supplementary Figures 3–6).
Four studies [12], 14], 16], 18] utilized 3 T pmMRI, and the pooled sensitivity of this subgroup was 0.76 (95 % CI: 0.67–0.84), and the pooled specificity was 0.93 (95 % CI: 0.89–0.96). While the sensitivity was similar to the 1.5 T subgroup, specificity was slightly lower, albeit still high. The PLR was 11.25 (95 % CI: 1.50–84.39), and the NLR was 0.31 (95 % CI: 0.17–0.57), both indicating good diagnostic performance, though with wider confidence intervals reflecting variability and smaller sample size. The pooled DOR was 40.41 (95 % CI: 3.87–421.53), suggesting a strong overall diagnostic capability, albeit with a wider range compared to the 1.5 T group. The SROC yielded an AUC of 0.78 and a Q index of 0.72*, reflecting fair to good discriminative ability. No significant threshold effect was found (Spearman correlation coefficient=−0.400, p=0.600), and the Moses model slope was non-significant (p=0.259), suggesting no major threshold bias. Overall, 3 T pmMRI showed comparable sensitivity but slightly lower specificity than 1.5 T, with wider confidence intervals and more heterogeneity – likely due to fewer studies and differing imaging protocols or case mix. Nonetheless, the diagnostic odds suggest good diagnostic utility of 3 T scanners in detecting fetal cardiac anomalies (Supplementary Figures 7–10).
Additionally, one study by Votino et al., evaluated pmMRI at an ultra-high field strength of 9.4 T, which demonstrated superior visualization of fetal cardiac structures even in early gestation (≤20 weeks) [24]. The 9.4 T MRI was able to delineate the four-chamber view, outflow tracts, aortic arch, and systemic veins in nearly all cases, with a reported sensitivity of 75 % and specificity of 100 % for outflow tract anomalies. This underscores the potential for ultra-high-field MRI to improve diagnostic accuracy, particularly in early gestation, though limited availability currently restricts its routine use.
Publication bias and risk of bias assessment
The inverse funnel plot (Figure 4) demonstrated noticeable asymmetry, indicating a potential risk of publication bias within the included studies. Visual inspection reveals notable asymmetry, suggesting the possibility of publication bias or small-study effects, where smaller studies with non-significant or favourable outcomes may be underrepresented. The pseudo 95 % confidence limits, shown as an inverted funnel, indicate where studies would typically fall in the absence of bias and the distribution of points outside this funnel further supports the presence of potential bias. The quality assessment using the QUADAS-2 tool is presented in Figure 5. Although, few of the included studies were limited by relatively small sample sizes, non-consecutive sampling strategy, and retrospective study design, the included studies generally ranged from low to moderate risk of bias.

Inverse funnel plot for assessment of publication bias.

Risk of bias assessment of included studies using QUADAS-2 tool.
Discussion
Cardiac abnormalities are identified in as many as 35 % of fetal autopsies, yet only approximately half of these defects are detected before birth, thereby signifying the role of postmortem cardiac assessment [27], 28]. Postmortem imaging of congenital heart defects in fetal and perinatal cases utilizes several complementary modalities, each offering unique advantages for detailed cardiac assessment [29]. Postmortem ultrasound (pmUS) is accessible and portable, often used for initial screening, but its diagnostic utility is limited postmortem due to low resolution and tissue decomposition and gas formation that can obscure cardiac structures [30], 31]. Postmortem computed tomography (pmCT) provides rapid imaging but it lacks the soft tissue resolution required for detailed cardiac assessment. Postmortem CT angiography (pmCTA) improves vascular visualization through contrast injection and specialized postmortem perfusion techniques, offering a better evaluation of great vessels and outflow tract anomalies, however, it is technically demanding and not widely available [32]. Postmortem micro-CT offers extremely high-resolution imaging ideal for very small fetuses, yet it is resource-intensive and less widely accessible [33]. Considering the clinical applicability, non-invasiveness, and diagnostic detail, pmMRI currently represents the most practical imaging modality for routine postmortem cardiac assessment in fetal and perinatal deaths. It offers superior soft tissue contrast with multi-planar, three-dimensional visualization, thereby enabling detailed visualization of complex cardiac anatomy, including septal defects, valvular abnormalities, great vessel anomalies, and syndromic malformations, without the technical barriers of contrast administration [4], 5].
Key findings in the context of existing literature
Although earlier studies consistently showed poor diagnostic yield of pmMRI for detecting cardiovascular anomalies, this was largely attributable to the use of conventional 2-D MRI sequences, which had a very low sensitivity of approximately 12 % for identifying structural cardiac defects [29]. For instance, Alderliesten et al., utilizing 1 T MRI scanner and 2D sequences, found no cardiac defects on postmortem MRI in 26 fetuses, despite five anomalies being confirmed at autopsy, resulting in 0 % sensitivity [34]. It is now recognized that the use of high-resolution, 3-D postmortem MRI can significantly improve diagnostic accuracy [35]. Three-dimensional imaging allows detailed visualization of complex cardiac structures in multiple planes, while longer scan times and higher resolution in the postmortem setting reduce partial volume effects, enabling better detection of small cardiac anomalies. This systematic review and meta-analysis represent the first comprehensive evaluation of the diagnostic accuracy of pmMRI for detecting cardiac anomalies in fetal and perinatal deaths. Our findings demonstrate that pmMRI exhibits good overall diagnostic performance, with a pooled sensitivity of 76 % and excellent specificity of 96 % when compared to conventional autopsy as the reference standard. The high specificity reported across the included studies, with some demonstrating perfect specificity highlights its excellent capacity to correctly rule out cardiac anomalies when absent, reducing unnecessary anxiety for families already coping with perinatal loss. Furthermore, the high area under the SROC curve (0.89) and substantial diagnostic odds ratio (55.35) support pmMRI as a reliable, non-invasive adjunct to conventional autopsy, especially in settings where consent for autopsy is not granted. Conversely, the moderate sensitivity suggests that pmMRI should be interpreted cautiously when negative, particularly in cases where clinical suspicion for cardiac anomalies remains high based on other factors such as family history, associated anomalies, or antenatal imaging findings.
Technical limitations and challenges of cardiac pmMRI
While cardiac pmMRI shows good overall diagnostic metrics for CHD, several technical limitations persist. One key challenge is fetal size. In the MARIAS study by Taylor et al. 10 % of cardiac pmMRI scans were non-diagnostic, all involving smaller fetuses under 24 weeks of gestation [22]. This issue might be addressed by developing pmCTA or using high-field MRI, which holds promise for improving image clarity, particularly in small fetuses and complex cardiac cases. In this context, Jawad et al. concluded that the diagnostic yield of pmMRI is significantly influenced by fetal body weight, with weight emerging as the strongest predictor of diagnostic success, surpassing the impact of gestational age [36]. They established that pmMRI is highly likely to provide diagnostic-quality images across all major organ systems, including the cardiovascular system, when fetal body weight is at least 535 g, with a 95 % confidence that 90 % of cases would yield diagnostic images. The diagnostic adequacy of pmMRI varied across organ systems, with the musculoskeletal system requiring the lowest body weight for acceptable imaging, while the cardiovascular and thoracic systems needed significantly higher weights for reliable assessment [36].
Another limitation is the occurrence of false positives. In the study by Taylor et al. apart from fetuses ≤24 weeks, only one significant misdiagnosis (cor triatriatum) upon pmMRI was reported, with most false positives involving minor septal defects, coarctation, aortic stenosis, or partial anomalous pulmonary venous return [22]. These misinterpretations may result from suboptimal image quality, over-interpretation of findings, or potentially from cardiac anomalies being overlooked at conventional autopsy. Furthermore, the location and complexity of cardiac lesions may significantly influence diagnostic performance. Votino et al. [24] reported that pmMRI achieved 100 % sensitivity in detecting situs and aortic arch abnormalities, while sensitivity decreased to 75 % for outflow tract anomalies and further to 66.7 % for four-chamber defects, while Shelmerdine et al. [17] and Rüegger et al. [12] found outflow tract anomalies, valve abnormalities, and coronary artery anomalies challenging to assess due to the absence of perfusion and dynamic motion in the post-mortem heart. Small ventricular and atrial septal defects are frequently missed due to limited resolution and tissue collapse post-mortem (Table 2) [15], 17], 35], 36]. Subtle conditions such as coarctation of the aorta, cor triatriatum and mild forms of hypoplastic left heart syndrome may go undetected [29], 36] while conduction system abnormalities remain beyond the detection capabilities of MRI and require histological evaluation [37]. These limitations underscore the importance of combining pmMRI with histopathology or high-resolution imaging when evaluating suspected CHD.
| Cardiac anomaly | Reason for missed diagnosis on pmMRI | |
|---|---|---|
| 1 | Small ventricular septal defects | Limited spatial resolution; collapsed ventricles postmortem obscure defect margins |
| 2 | Atrial septal defects | Difficult to visualize due to thin septal tissue and absence of contrast |
| 3 | Outflow tract anomalies (e.g., DORV, TGA) | Orientation and collapsed vessels make accurate interpretation difficult |
| 4 | Valvular abnormalities (e.g., Ebstein anomaly) | Valve leaflets collapse and lack motion; morphology not reliably evaluated |
| 5 | Coarctation of the aorta | Mild narrowing may be missed in absence of functional assessment |
| 6 | Coronary artery anomalies | Small size and no vascular contrast limit visualization |
| 7 | Total anomalous pulmonary venous return | Pulmonary veins hard to trace without blood flow; may appear normal |
| 8 | Hypoplastic left heart syndrome (mild forms) | Collapsed chambers postmortem may mimic or obscure hypoplasia |
| 9 | Conduction system abnormalities | Not visualized on MRI; requires histological or molecular methods |
-
DORV, double-outlet right ventricle; pmMRI, post-mortem magnetic resonance imaging; TGA, transposition of the great arteries.
In one of the included studies (Tang et al.), the investigators performed pmMRI of ex vivo organs fixed in 10 % formalin [13]. Tissue fixation effects can alter tissue contrast and signal characteristics compared to living tissue. Formalin fixation, commonly used for specimen preservation, changes T1 and T2 relaxation times, potentially affecting diagnostic accuracy and requiring adjusted imaging protocols [38].
Sources of heterogeneity
In the meta-analysis, significant heterogeneity was observed across studies for most diagnostic metrics, with I2 values exceeding 70 % for sensitivity, specificity, and diagnostic odds ratio and subgroup analyses provided meaningful insights into potential sources of this variability. Notably, studies utilizing 1.5 T pmMRI demonstrated consistently high specificity and a higher DOR, with relatively lower heterogeneity compared to the overall analysis. This suggests that 1.5 T scanners may offer more reliable diagnostic performance, potentially due to standardized imaging protocols and broader clinical familiarity. Conversely, studies employing 3 T pmMRI displayed comparable sensitivity but slightly reduced specificity and greater heterogeneity, likely reflecting differences in image acquisition protocols, interpreter expertise, and patient populations. Although 3 T pmMRI remains a valuable diagnostic option, these findings suggest a need for protocol standardization and operator training to improve diagnostic consistency. The single study using ultra-high-field 9.4 T MRI included in meta-analysis, demonstrated superior anatomical visualization of cardiac structures, even in early gestation. While this highlights the potential for future improvements in post-mortem imaging, the limited availability and high cost of ultra-high-field MRI currently restrict its widespread clinical use.
Study’s strengths and limitations
This systematic review followed rigorous methodology following PRISMA guidelines with a pre-registered review protocol. Subgroup analyses exploring the impact of MRI field strength and imaging parameters were performed. However, several limitations warrant consideration. The potential publication bias, with possible underrepresentation of smaller studies with negative or non-significant results and significant heterogeneity, limits the generalizability of pooled estimates. This heterogeneity likely reflects several factors including variations in study populations, gestational age ranges, types of cardiac anomalies studied, imaging protocols, and interpreter experience levels. Gestational age at death and fetal weight likely affect imaging quality and anatomical visualization, with earlier gestations and low fetal weight potentially more challenging to interpret [22], [23], [24, 36]. The spectrum of cardiac anomalies studied also varies considerably between studies, with some focusing on major structural defects while others include minor anomalies that may be more difficult to detect on imaging.
The relatively small number of studies in subgroup analyses reduces the precision of field strength comparisons. Also, only two studies employed ultra-high-field MRI (9.4 T), thereby restricting robust conclusions about its diagnostic superiority, over 1.5 and 3 T equipment. Furthermore, there was a predominance of studies from high-resource settings, for example, we encountered 8 studies from Europe, which may limit applicability in low- and middle-income countries where pmMRI access is limited. Additionally, interpreter experience and availability of specialized pediatric or fetal cardiac imaging expertise likely influence diagnostic accuracy but were inconsistently reported across studies.
Clinical recommendations and future directions
The evidence generated from this meta-analysis supports integrating pmMRI into routine perinatal death investigation protocols, particularly when conventional autopsy is declined or delayed, a standard protocol, as deduced from included studies and other existing literature is presented in Table 3 [11], [12], [13], [14], [15], [16], [17], [18], [19], [20], [21], [22], [23], [24], [25], [26, 37], 39]. pmMRI can be considered a highly specific tool for confirming cardiac anomalies in fetal and perinatal deaths, thereby playing a crucial role in supporting genetic counselling, and assessing the likelihood of recurrence. However, pmMRI should be viewed as complementary to, rather than a replacement for, conventional autopsy when the latter is acceptable to families. The moderate sensitivity suggests that negative pmMRI results should not preclude conventional autopsy, along with genetic testing, when detailed cardiac evaluation is clinically indicated. Standardization of imaging protocols and interpreter training is essential to reduce variability and improve diagnostic reliability across centres. The use of either 1.5 T and 3 T pmMRI scanners remains acceptable with protocol optimization and further research is needed to explore the incremental value of higher field strengths, that is 9.4 T, advanced imaging techniques, and artificial intelligence-assisted interpretation. Studies investigating the optimal timing of imaging relative to death, the impact of body preparation techniques, and the development of specialized fetal cardiac MRI sequences may further enhance diagnostic performance. Additionally, cost-effectiveness analyses comparing pmMRI to the conventional autopsy, considering both direct costs and indirect benefits such as improved family acceptance and genetic counselling outcomes, would provide valuable information for healthcare policy decisions.
Standard protocol for pmMRI in fetal cardiac evaluation [11], [12], [13], [14], [15], [16], [17], [18], [19], [20], [21], [22], [23], [24], [25], [26, 37], 39].
| Component | Recommendations |
|---|---|
| Magnetic field strength | 3 T preferred (1.5 T acceptable) for higher spatial resolution |
| MRI coil | Pediatric body or surface coil to optimize signal-to-noise ratio |
| Timing of scan | Within 24–72 h post-delivery; body stored at 4 °C to minimize decomposition |
| Patient preparation | Fetus wrapped and positioned in supine/prone; no embalming prior to imaging |
| Key sequences |
|
| Cardiac imaging planes |
|
| Interpretation | By pediatric radiologist or experienced fetal imaging specialist Correlation with prenatal imaging and clinical history recommended |
| Reporting format | Structured report including cardiac anatomy, chamber size, valve morphology, septa, and great vessel orientation |
| Adjunct modalities |
|
Conclusions
To conclude, pmMRI emerged as a valuable diagnostic modality for detecting cardiac anomalies in fetal and perinatal deaths, offering high specificity and moderate sensitivity. While pmMRI cannot completely replace conventional autopsy for comprehensive cardiac evaluation, it offers a less invasive alternative that may be more acceptable to families while providing clinically meaningful diagnostic information. Both 1.5 T and 3 T MRI systems demonstrate acceptable diagnostic performance, making pmMRI accessible across different healthcare settings. However, significant heterogeneity between studies highlights the need for standardized imaging protocols and quality assurance measures to optimize diagnostic consistency. Effective clinical integration of post-mortem MRI depends on a multidisciplinary approach involving fetal medicine specialists, pediatric cardiologists, radiologists, and genetic counsellors, supported by a quality assurance system to integrate prenatal diagnoses and improve future detection rates. Standardized protocols, dedicated training, cost analyses, and regulatory support will be essential for successful clinical adoption.
-
Research ethics: The local Institutional Review Board deemed the study exempt from review.
-
Informed consent: Not applicable.
-
Author contributions: SP: Concept, Design, definition of intellectual content, literature search and review, data extraction, data analysis, manuscript preparation, approval of final draft; MM: Concept, Design, definition of intellectual content, literature search and review, data extraction, data analysis, manuscript review and approval of final draft, correspondence, MoM and PKK: Literature search and review, data extraction, manuscript review and approval of final draft; AS: Data extraction, manuscript review and approval of final draft. All the authors have contributed substantially to the drafting the work or revising it critically for important intellectual content; AND Final approval of the version to be published; AND Agreement to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
-
Use of Large Language Models, AI and Machine Learning Tools: None declared.
-
Conflict of interest: The authors state no conflict of interest.
-
Research funding: None declared.
-
Data availability: Not applicable.
References
1. Hasan, AA, Abu Lehyah, NAA, Al Tarawneh, MK, Abbad, MY, Fraijat, AG, Al-Jammal, RA, et al.. Incidence and types of congenital heart disease at a referral hospital in Jordan: retrospective study from a tertiary center. Front Pediatr 2023;11:1261130. https://doi.org/10.3389/fped.2023.1261130.Search in Google Scholar PubMed PubMed Central
2. Nijkamp, JW, Sebire, NJ, Bouman, K, Korteweg, FJ, Erwich, JJHM, Gordijn, SJ. Perinatal death investigations: what is current practice? Semin Fetal Neonatal Med 2017;22:167–75. https://doi.org/10.1016/j.siny.2017.02.005.Search in Google Scholar PubMed PubMed Central
3. Jepson, BM, Metz, TD, Miller, TA, Son, SL, Ou, Z, Presson, AP, et al.. Pregnancy loss in major fetal congenital heart disease: incidence, risk factors and timing. Ultrasound Obstet Gynecol 2023;62:75–87. https://doi.org/10.1002/uog.26231.Search in Google Scholar PubMed
4. Struksnæs, C, Blaas, HK, Eik-Nes, SH, Tegnander, E, Vogt, C. Postmortem assessment of isolated congenital heart defects remains essential following termination of pregnancy. Pediatr Dev Pathol 2021;24:422–9. https://doi.org/10.1177/10935266211016184.Search in Google Scholar PubMed
5. Ashby, C, Razzak, AN, Kogler, A, Amireh, A, Dempsey, J, Lin, KK, et al.. The practicality of post-mortem imaging in prenatal, perinatal, and pediatric cases. Cureus 2022;14:e28859. https://doi.org/10.7759/cureus.28859.Search in Google Scholar PubMed PubMed Central
6. Mangla, M, Kumar, N. Recurrence risks in congenital anomalies: a comprehensive guide for parental counselling. NeoReviews 2024;25:e793–803. https://doi.org/10.1542/neo.25-12-e793.Search in Google Scholar PubMed
7. Page, MJ, McKenzie, JE, Bossuyt, PM, Boutron, I, Hoffmann, TC, Mulrow, CD, et al.. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ 2021;372:n71. https://doi.org/10.1136/bmj.n71.Search in Google Scholar PubMed PubMed Central
8. Whiting, P, Rutjes, AW, Reitsma, JB, Bossuyt, PMM, Kleijnen, J. The development of QUADAS: a tool for the quality assessment of studies of diagnostic accuracy included in systematic reviews. BMC Med Res Methodol 2003;3:25. https://doi.org/10.1186/1471-2288-3-25.Search in Google Scholar PubMed PubMed Central
9. Zamora, J, Abraira, V, Muriel, A, Khan, K, Coomarasamt, A. Meta-DiSc: a software for meta-analysis of test accuracy data. BMC Med Res Methodol 2006;6:31. https://doi.org/10.1186/1471-2288-6-31.Search in Google Scholar PubMed PubMed Central
10. MedCalc Statistical Software version 19.2.6 (MedCalc Software bv, Ostend, Belgium; https://www.medcalc.org; 2020).Search in Google Scholar
11. Gupta, N, Langeh, N, Sharma, A, Mridha, A, Jana, M, Barwad, A, et al.. Minimally invasive autopsy in the evaluation of fetal malformations and stillbirths: a feasibility study. Birth Defects Res 2024;116:e2280. https://doi.org/10.1002/bdr2.2280.Search in Google Scholar PubMed
12. Rüegger, CM, Gascho, D, Bode, PK, Bruder, E, Haslinger, C, Ross, S, Virtopsy Study Group, et al.. Post-mortem magnetic resonance imaging with computed tomography-guided biopsy for foetuses and infants: a prospective, multicentre, cross-sectional study. BMC Pediatr 2022;22:464. https://doi.org/10.1186/s12887-022-03519-4.Search in Google Scholar PubMed PubMed Central
13. Tang, H, Zhang, Y, Dai, C, Ru, T, Li, J, Chen, J, et al.. Postmortem 9.4-T MRI for fetuses with congenital heart defects diagnosed in the first trimester. Front Cardiovasc Med 2022;8:764587. https://doi.org/10.3389/fcvm.2021.764587.Search in Google Scholar PubMed PubMed Central
14. Staicu, A, Albu, C, Popa-Stanila, R, Bondor, CI, Rotar, IC, Stamatian, F, et al.. Diagnostic value of virtual autopsy using pm-MRI at 3T on malformed second trimester fetuses vs classic autopsy. PLoS One 2021;16:e0260357. https://doi.org/10.1371/journal.pone.0260357.Search in Google Scholar PubMed PubMed Central
15. Shelmerdine, SC, Sebire, NJ, Arthurs, OJ. Diagnostic accuracy of postmortem ultrasound vs postmortem 1.5-T MRI for non-invasive perinatal autopsy. Ultrasound Obstet Gynecol 2021;57:449–58. https://doi.org/10.1002/uog.22012.Search in Google Scholar PubMed PubMed Central
16. Ulm, B, Dovjak, GO, Scharrer, A, Muin, DA, Zimpfer, D, Prayer, D, et al.. Diagnostic quality of 3 Tesla postmortem magnetic resonance imaging in fetuses with and without congenital heart disease. Am J Obstet Gynecol 2021;225:189. https://doi.org/10.1016/j.ajog.2021.02.030.Search in Google Scholar PubMed
17. Shelmerdine, SC, Arthurs, OJ, Gilpin, I, Norman, W, Jones, R, Taylor, AM, et al.. Is traditional perinatal autopsy needed after detailed fetal ultrasound and post-mortem MRI? Prenat Diagn 2019;39:818–29. https://doi.org/10.1002/pd.5448.Search in Google Scholar PubMed
18. Kang, X, Sanchez, TC, Arthurs, OJ, Bevilacqua, E, Cannie, MM, Segers, V, et al.. Postmortem fetal imaging: prospective blinded comparison of two-dimensional ultrasound with magnetic resonance imaging. Ultrasound Obstet Gynecol 2019;54:791–9. https://doi.org/10.1002/uog.20217.Search in Google Scholar PubMed
19. Shruthi, M, Gupta, N, Jana, M, Mridha, AR, Kumar, A, Agarwal, R, et al.. Conventional vs virtual autopsy with postmortem MRI in phenotypic characterization of stillbirths and fetal malformations. Ultrasound Obstet Gynecol 2018;51:236–45. https://doi.org/10.1002/uog.17468.Search in Google Scholar PubMed
20. Ashwin, C, Hutchinson, JC, Kang, X, Langan, D, Jones, R, Norman, W, et al.. Learning effect on perinatal post-mortem magnetic resonance imaging reporting: single reporter diagnostic accuracy of 200 cases. Prenat Diagn 2017;37:566–74. https://doi.org/10.1002/pd.5043.Search in Google Scholar PubMed
21. Kang, X, Cannie, MM, Arthurs, OJ, Segers, V, Fourneau, C, Bevilacqua, E, et al.. Post-mortem whole-body magnetic resonance imaging of human fetuses: a comparison of 3-T vs. 1.5-T MR imaging with classical autopsy. Eur Radiol 2017;27:354253. https://doi.org/10.1007/s00330-016-4725-4.Search in Google Scholar PubMed
22. Taylor, AM, Sebire, NJ, Ashworth, MT, Schievano, S, Scott, RJ, Wade, A, et al.. Magnetic resonance imaging autopsy study collaborative group. Postmortem cardiovascular magnetic resonance imaging in fetuses and children: a masked comparison study with conventional autopsy. Circulation 2014;129:1937–44. https://doi.org/10.1161/circulationaha.113.005641.Search in Google Scholar
23. Sandaite, I, Dymarkowski, S, De Catte, L, Moerman, P, Gewillig, M, Fedele, L, et al.. Fetal heart pathology on postmortem 3-T magnetic resonance imaging. Prenat Diagn 2014;34:223–9. https://doi.org/10.1002/pd.4283.Search in Google Scholar PubMed
24. Votino, C, Jani, J, Verhoye, M, Bessieres, B, Fierens, Y, Segers, V, et al.. Postmortem examination of human fetal hearts at or below 20 weeks’ gestation: a comparison of high-field MRI at 9.4 T with lower-field MRI magnets and stereomicroscopic autopsy. Ultrasound Obstet Gynecol 2012;40:437–44. https://doi.org/10.1002/uog.11191.Search in Google Scholar PubMed
25. Cannie, M, Votino, C, Moerman, P, Vanheste, R, Segers, V, Van Berkel, K, et al.. Acceptance, reliability and confidence of diagnosis of fetal and neonatal virtuopsy compared with conventional autopsy: a prospective study. Ultrasound Obstet Gynecol 2012;39:659–65. https://doi.org/10.1002/uog.10079.Search in Google Scholar PubMed
26. Breeze, AC, Jessop, FA, Set, PA, Whitehead, AL, Cross, JJ, Lomas, DJ, et al.. Minimally-invasive fetal autopsy using magnetic resonance imaging and percutaneous organ biopsies: clinical value and comparison to conventional autopsy. Ultrasound Obstet Gynecol 2011;37:317–23. https://doi.org/10.1002/uog.8844.Search in Google Scholar PubMed
27. Grant, EK, Evans, MJ. Cardiac findings in fetal and pediatric autopsies: a five-year retrospective review. Pediatr Dev Pathol 2009;12:103–10. https://doi.org/10.2350/08-03-0440.1.Search in Google Scholar PubMed
28. Tegnander, E, Williams, W, Johansen, OJ, Blaas, HG, Eik-Nes, SH. Prenatal detection of heart defects in a non-selected population of 30,149 fetuses--detection rates and outcome. Ultrasound Obstet Gynecol 2006;27:252–65. https://doi.org/10.1002/uog.2710.Search in Google Scholar PubMed
29. Taylor, AM, Arthurs, OJ, Sebire, NJ. Postmortem cardiac imaging in fetuses and children. Pediatr Radiol 2015;45:549–55. https://doi.org/10.1007/s00247-014-3164-0.Search in Google Scholar PubMed PubMed Central
30. Isaksen, CV, Eik-Nes, SH, Blaas, HG, Tegnander, E, Torp, SH. Comparison of prenatal ultrasound and postmortem findings in fetuses and infants with congenital heart defects. Ultrasound Obstet Gynecol 1999;13:117–26. https://doi.org/10.1046/j.1469-0705.1999.13020117.x.Search in Google Scholar PubMed
31. Shelmerdine, SC, Sebire, NJ, Arthurs, OJ. Perinatal post mortem ultrasound (PMUS): a practical approach. Insights Imaging 2019;10:35. https://doi.org/10.1186/s13244-019-0723-9.Search in Google Scholar PubMed PubMed Central
32. Votino, C, Cannie, M, Segers, V, Dobrescu, O, Dessy, H, Gallo, V, et al.. Virtual autopsy by computed tomographic angiography of the fetal heart: a feasibility study. Ultrasound Obstet Gynecol 2012;39:679–84. https://doi.org/10.1002/uog.11150.Search in Google Scholar PubMed
33. Hutchinson, JC, Arthurs, OJ, Ashworth, MT, Ramsey, AT, Mifsud, W, Lombardi, CM, et al.. Clinical utility of postmortem microcomputed tomography of the fetal heart: diagnostic imaging vs macroscopic dissection. Ultrasound Obstet Gynecol 2016;47:58–64. https://doi.org/10.1002/uog.15764.Search in Google Scholar PubMed
34. Alderliesten, ME, Peringa, J, van der Hulst, VP, Blaauwgeers, HL, van Lith, JM. Perinatal mortality: clinical value of postmortem magnetic resonance imaging compared with autopsy in routine obstetric practice. BJOG 2003;110:378–82. https://doi.org/10.1046/j.1471-0528.2003.02076.x.Search in Google Scholar
35. Thayyil, S, Chandrasekaran, M, Chitty, LS, Wade, A, Skordis-Worrall, J, Bennett-Britton, I, et al.. Diagnostic accuracy of post-mortem magnetic resonance imaging in fetuses, children and adults: a systematic review. Eur J Radiol 2010;75:e142–8. https://doi.org/10.1016/j.ejrad.2009.10.007.Search in Google Scholar PubMed
36. Jawad, N, Sebire, NJ, Wade, A, Taylor, AM, Chitty, LS, Arthurs, OJ. Body weight lower limits of fetal postmortem MRI at 1.5 T. Ultrasound Obstet Gynecol 2016;48:92–7. https://doi.org/10.1002/uog.14948.Search in Google Scholar PubMed
37. Thayyil, S, Sebire, NJ, Chitty, LS, Wade, A, Chong, W, Olsen, O, et al.. MARIAS collaborative group. Post-mortem MRI versus conventional autopsy in fetuses and children: a prospective validation study. Lancet 2013;382:223–33. https://doi.org/10.1016/s0140-6736-13-60134-8.Search in Google Scholar
38. Birkl, C, Langkammer, C, Golob-Schwarzl, N, Leoni, M, Haybaeck, J, Goessler, W, et al.. Effects of formalin fixation and temperature on MR relaxation times in the human brain. NMR Biomed 2016;29:458–65. https://doi.org/10.1002/nbm.3477.Search in Google Scholar PubMed
39. Royal College of Radiologists, British Society of Paediatric Radiology. Standards for radiological autopsy practice in perinatal and paediatric cases. London: The Royal College of Radiologists; 2017. Available from: https://www.rcr.ac.uk/publication/standards-radiological-autopsy-practice-perinatal-and-paediatric-cases.Search in Google Scholar
Supplementary Material
This article contains supplementary material (https://doi.org/10.1515/jpm-2025-0528).
© 2026 the author(s), published by De Gruyter, Berlin/Boston
This work is licensed under the Creative Commons Attribution 4.0 International License.
Articles in the same Issue
- Frontmatter
- Reviews
- Immunoediting in pregnancy: a new paradigm for understanding fetal tolerance and obstetric disease
- Virtual fetal holography for parental counseling and education: applications, limitations, and future directions
- Original Articles – Obstetrics
- RNA biomarkers in hypertensive disorders of pregnancy: systematic review
- Second pregnancy vaginal birth after cesarean- impact of maternal age on outcomes from a retrospective cohort study
- Analysis of changes in serum VEGF, β-hCG, and sFlt-1 levels in women with placenta accreta spectrum and the impact on prognosis
- The adjunctive role of the neutrophil-to-lymphocyte ratio in risk stratification for clinical chorioamnionitis in term pregnancies with meconium-stained amniotic fluid
- Clinical utility of chromosomal microarray and whole exome sequencing in evaluating genetic causes for pregnancy loss using products of conception specimens
- The association of antiphospholipid syndrome under medical treatment with adverse pregnancy outcomes
- Effects of virtual reality on fear of birth, length of labor, and fetal well-being: a randomized controlled trial
- Antenatal corticosteroid prophylaxis in women with increased sFlt-1/PlGF ratio in the clinical routine – A retrospective analysis
- Early unfavorable outcomes of second-trimester selective feticide for complicated monochorionic twins: single-operator experiences
- Original Articles – Fetus
- Maternal circulating sFlt-1/placental growth factor is a biomarker of fetal death associated with placental lesions of maternal vascular malperfusion
- Intra-partum and perinatal outcomes in fetuses exhibiting ZigZag pattern on cardiotocography trace: a systematic review and meta-analysis
- Assessment of fetal adrenal gland and thymothoracic ratio in preterm premature membrane rupture: a prospective case-control study
- Fetal brain in fetal growth restriction: alterations in cortical morphometry and volume
- Diagnostic yield of post-mortem magnetic resonance imaging for cardiac anomalies in fetal and perinatal deaths: a systematic review and meta-analysis
- Original Articles – Neonates
- Risk factors and analysis of retinopathy of prematurity in monochorionic diamniotic twins
- Association between FAR, PAR, APRI and adverse neonatal outcomes in pregnancies complicated by intrahepatic cholestasis
- Incidence and predictors of high vitamin D in premature infants with very low birth weight
- Urinary immune biomarkers for late-onset sepsis in preterm very low birth weight neonates – a diagnostic accuracy study
- Letters to the Editor
- Histological chorioamnionitis and maternal inflammatory biomarkers: implications beyond clinical diagnosis
- Response to Letter to the Editor
- Erratum
- Erratum to: Gestational diabetes mellitus: the role of IGF-1 and leptin in cord blood
Articles in the same Issue
- Frontmatter
- Reviews
- Immunoediting in pregnancy: a new paradigm for understanding fetal tolerance and obstetric disease
- Virtual fetal holography for parental counseling and education: applications, limitations, and future directions
- Original Articles – Obstetrics
- RNA biomarkers in hypertensive disorders of pregnancy: systematic review
- Second pregnancy vaginal birth after cesarean- impact of maternal age on outcomes from a retrospective cohort study
- Analysis of changes in serum VEGF, β-hCG, and sFlt-1 levels in women with placenta accreta spectrum and the impact on prognosis
- The adjunctive role of the neutrophil-to-lymphocyte ratio in risk stratification for clinical chorioamnionitis in term pregnancies with meconium-stained amniotic fluid
- Clinical utility of chromosomal microarray and whole exome sequencing in evaluating genetic causes for pregnancy loss using products of conception specimens
- The association of antiphospholipid syndrome under medical treatment with adverse pregnancy outcomes
- Effects of virtual reality on fear of birth, length of labor, and fetal well-being: a randomized controlled trial
- Antenatal corticosteroid prophylaxis in women with increased sFlt-1/PlGF ratio in the clinical routine – A retrospective analysis
- Early unfavorable outcomes of second-trimester selective feticide for complicated monochorionic twins: single-operator experiences
- Original Articles – Fetus
- Maternal circulating sFlt-1/placental growth factor is a biomarker of fetal death associated with placental lesions of maternal vascular malperfusion
- Intra-partum and perinatal outcomes in fetuses exhibiting ZigZag pattern on cardiotocography trace: a systematic review and meta-analysis
- Assessment of fetal adrenal gland and thymothoracic ratio in preterm premature membrane rupture: a prospective case-control study
- Fetal brain in fetal growth restriction: alterations in cortical morphometry and volume
- Diagnostic yield of post-mortem magnetic resonance imaging for cardiac anomalies in fetal and perinatal deaths: a systematic review and meta-analysis
- Original Articles – Neonates
- Risk factors and analysis of retinopathy of prematurity in monochorionic diamniotic twins
- Association between FAR, PAR, APRI and adverse neonatal outcomes in pregnancies complicated by intrahepatic cholestasis
- Incidence and predictors of high vitamin D in premature infants with very low birth weight
- Urinary immune biomarkers for late-onset sepsis in preterm very low birth weight neonates – a diagnostic accuracy study
- Letters to the Editor
- Histological chorioamnionitis and maternal inflammatory biomarkers: implications beyond clinical diagnosis
- Response to Letter to the Editor
- Erratum
- Erratum to: Gestational diabetes mellitus: the role of IGF-1 and leptin in cord blood

