Abstract
Introduction
Pregnancy poses an immunological paradox: the maternal immune system must tolerate a semi-allogeneic fetus while maintaining defense against infections. Rather than being an immunosuppressed state, gestation is now recognized as a dynamic, highly regulated immune condition.
Content
This review applies the cancer-derived immunoediting framework – elimination, equilibrium, and escape – to maternal–fetal immune tolerance. We examine how immune checkpoints, regulatory T cells, non-classical MHC molecules, and placental exosomes coordinate to create a localized tolerant environment. Integrating knowledge from oncology and reproductive immunology, this perspective provides a unifying concept for pregnancy immune regulation.
Summary
The immunoediting framework reinterprets obstetric disorders such as preeclampsia, recurrent pregnancy loss, and preterm birth as failures of distinct immune phases rather than isolated pathologies. This conceptual shift allows for a broader understanding of how immune balance influences implantation, placental development, and fetal growth.
Outlook
Adopting an immunoediting perspective highlights potential clinical advances, including immune checkpoint modulation, regulatory T-cell therapies, and exosome-based biomarkers, paving the way for innovative diagnostic and therapeutic strategies in pregnancy care.
Introduction
Pregnancy presents one of the most intriguing immunological paradoxes: the maternal immune system must tolerate a genetically distinct fetus while maintaining effective defense against infections and preserving systemic balance [1], [2], [3]. While previously viewed as a state of immunosuppression, it is now recognized as a dynamic and tightly regulated process of immune adaptation and surveillance [2], 4], 5].
This immune balance is maintained through several specialized mechanisms. Extravillous trophoblasts express non-classical major histocompatibility complex (MHC) class I molecules such as human leukocyte antigen G (HLA-G), which bind to inhibitory receptors on uterine natural killer (uNK) cells and antigen-presenting cells, promoting localized immune tolerance [4], [6], [7], [8]. Regulatory T cells (Tregs), which expand during early gestation, suppress inflammatory responses through cytokines like interleukin 10 (IL-10) and transforming growth factor beta (TGF-β), and through inhibitory molecules such as cytotoxic T lymphocyte-associated protein 4 (CTLA-4) [5], [9], [10], [11], [12]. Immune checkpoints – especially the programmed cell death protein 1 (PD-1)/programmed cell death ligand 1 (PD-L1) axis – play a key role by downregulating maternal cytotoxic responses. These checkpoints are expressed by trophoblasts and conveyed through placental exosomes [13], [14], [15], [16], [17], [18] (Table 1; Figure 1).
Summary of key literature on maternal-fetal immune tolerance.a
| Author/year | Focus area | Model/system | Key findings/insight | Strength | Limitation | Immunoediting phase |
| Dunn et al. 2004 [1] | Immunoediting concept | Cancer model (conceptual) | Introduced the elimination phase in immunoediting | Foundational concept | Not pregnancy-specific | Elimination |
| Moffett & Colucci, 2014 [12] | uNK cells | Human | Described the role of uNK cells in implantation and placental development | Human-focused and mechanistic | Focuses only on NK cells | Elimination |
| Zhou et al. 2016 [5] | Immune cell dynamics | Human | Linked dysregulated immune cells to recurrent miscarriage | Clinical relevance | Correlative, not mechanistic | Elimination |
| Vacca et al. 2011 [22] | NK precursors | Human decidua | Identified hematopoietic precursors differentiating into NK cells | Novel cellular source | Cell-specific focus | Elimination |
| Tilburgs et al. 2015 [3] | HLA-G | Human | Explains dual role of HLA-G in tolerance and immunity | Mechanistic depth | Focused on one molecule | Elimination |
| Aluvihare et al. 2004 [6] | Tregs | Murine | Tregs prevent fetal rejection | Experimental causality | Animal model | Equilibrium |
| Zenclussen et al. 2006 [9] | Tregs | Murine | Tregs promote fetal-maternal immune privilege | Model-based validation | Translational gap | Equilibrium |
| Samstein et al. 2012 [20] | Treg generation | Placental mammals | Describes extrathymic Treg development | Evolutionary and mechanistic insight | Requires further clinical correlation | Equilibrium |
| Mincheva-Nilsson & Baranov, 2014 [10] | Exosomes | Human | Exosomes carry immunosuppressive signals | Novel tolerance vector | In vitro bias | Equilibrium |
| Robertson et al. 2018 [56] | Tregs in implantation | Human/murine | Links Tregs with successful implantation | Broad relevance | Complex interactions | Equilibrium |
| Zhang et al. 2023 [13] | PD-L1 in RM | Human | Reduced PD-L1 in recurrent miscarriage | Clinical tissue evidence | No functional tests | Escape |
| Rong et al. 2021 [15] | PD-L1 and PE | Human | Low PD-L1 suppresses GM-CSF and promotes preeclampsia | Mechanistic and clinical | Pathway-specific | Escape |
| Zhang et al. 2019 [18] | PD-1/PD-L1 and macrophages | Human | PD-L1 shapes macrophage polarization | Links checkpoints to immune environment | Model needs expansion | Escape |
| Qian et al. 2024 [57] | PD-1/OX40 in RPL | Human | Differential checkpoint expression in RSA tissue | Checkpoint profiling | Correlative | Escape |
| Tian et al. 2025 [58] | PD-L1 and JAK/STAT5 | Human | Downregulated PD-L1 activates pro-inflammatory signaling | Pathway-level detail | Focused scope | Escape |
| Schreiber et al. 2011 [7] | Immunoediting phases | Cancer (conceptual) | Described elimination, equilibrium, escape | Cross-disciplinary insight | Not empirical in pregnancy | All |
| Mittal et al. 2014 [17] | Cancer immunoediting | Cancer | Refined immunoediting model | Established theoretical foundation | Requires obstetric translation | All |
| Costanzo et al. 2018 [11] | Onco-fetal immunology | Human (review) | Linked placental and tumor immune strategies | Conceptual synthesis | Theoretical | All |
| Zhu et al. 2022 [27] | TIM-3 checkpoint | Human | TIM-3 expression associated with maternal-fetal tolerance and RSA | Checkpoint relevance in human pathology | Primarily observational | Equilibrium |
| Zhang et al., 2021 [21] | Checkpoint therapy | Murine | Blocking CD28 prevents fetal rejection | Therapeutic intervention validated in vivo | Preclinical data only | Escape |
| Zhang et al. 2023 [59] | Memory Tregs | Human | Characterized FOXP3+ memory Tregs during pregnancy | Defines new regulatory subset | Lacks functional assays | Equilibrium |
| Luo et al. 2025 [53] | CD8+FoxP3+ Tregs | Human/Animal | Describes cytotoxic regulatory T cells with tolerogenic function | Expands Treg paradigm | Emerging topic | Equilibrium |
| Yin et al. 2025 [60] | Exosomal biomarkers | Human | Exosome content may serve as predictive biomarker in PE | Clinical utility | Validation needed | Escape |
| Qian et al. 2024 [62] | Checkpoint profiles | Human | Differential PD-1/OX40 in RSA | Checkpoint mapping | Correlational | Escape |
| Li et al. 2024 [61] | Exosome therapy | Review/Preclinical | Exosomes explored for immunotherapy delivery | Novel translational approach | Preclinical only | Escape |
| Green et al. 2021 [62] | Tregs in adverse outcomes | Systematic review | Lower Treg levels linked with miscarriage and PE | Meta-analysis support | No mechanistic data | Escape |
| Lu et al. 2025 [63] | Treg immunometabolism | Review | Metabolism affects Treg stability in inflammation | Molecular mechanism insight | Requires in vivo proof | Equilibrium |
| Zhao et al. 2025 [64] | Mitochondrial regulation | Review | Mitochondria control Treg suppressive capacity | Deep cellular mechanism | Conceptual only | Equilibrium |
| Zhao et al. 2022 [65] | Exosomes in cancer vs. Pregnancy | Review | Parallels in exosome-mediated immune editing | Cross-field insight | Theoretical | All |
-
aThis table summarizes key studies illustrating mechanisms of maternal-fetal immune tolerance, categorized by immunoediting phase and focus area, across both experimental and clinical models.

PRISMA-guided literature selection and screening for conceptual synthesis in fetal immune tolerance. This PRISMA-guided flowchart outlines our literature selection process for a conceptual review on fetal immune tolerance. From 3,212 identified records, 1,636 full-text articles were assessed, and 110 were included based on relevance to immune mechanisms in pregnancy. These studies were categorized into three immunoediting phases: elimination, equilibrium, and escape, adapting an oncology framework to the maternal-fetal context.
Interestingly, these immune adaptations mirror those used by tumors to escape immune surveillance [19], [20], [21], [22], [23], [24]. Both cancer and pregnancy create immune-privileged environments through tightly regulated suppression mechanisms [25], [26], [27], [28]. This similarity forms the basis of applying the immunoediting model to gestation – comprising three phases: elimination, equilibrium, and escape [19], 20], 22], 24] (Figure 2; Table 2).

Maternal-fetal immune regulation at the decidual interface. This illustration shows the complex immune interactions at the maternal-fetal interface. Extravillous trophoblasts invade the maternal decidua and interact with immune cells such as decidual NK (dNK) cells, macrophages, and regulatory T cells (Tregs). Key mechanisms include: HLA-G/KIR signaling promoting immune tolerance by modulating dNK cells. Ido production by macrophages suppressing T effector cells. FasL expression inducing apoptosis in activated maternal T cells. Cytokines like IL-15, IFN-γ, TNF-α, and VEGF coordinating vascular remodeling and immune modulation.
Immunoediting phases in pregnancy.a
| Immunoediting phase | Key immune players | Mechanisms | Outcome in normal pregnancy | Pathological shift | References |
| Elimination | uNK cells, macrophages, dendritic cells, T cells | Initial detection of fetal antigens; inflammatory cytokine production; immune cell recruitment | Promotes implantation and vascular remodeling | Implantation failure, recurrent miscarriage due to overactivation or insufficient tolerance | [1], 5], 12], 22], 30], 92] |
| Equilibrium | Regulatory T cells (CD4+FoxP3+, CD8+ Tregs), tolerogenic DCs, exosomes, PD-1/PD-L1 | Immune suppression via IL-10, TGF-β, PD-L1; exosomal delivery of tolerogenic molecules | Fetal tolerance, sustained placental growth | Loss of tolerance, immune imbalance, preeclampsia risk | [6], 9], 10], 20], 54], 96], 97] |
| Escape | Activated Th1/Th17 cells, dysfunctional Tregs, cytotoxic T cells, inflammatory macrophages | Checkpoint failure, reduced Treg function, pro-inflammatory cytokines (IFN-γ, TNF-α) | None – this phase reflects pathology | Preeclampsia, preterm labor, fetal growth restriction | [13], 15], 18], 30], 35], 58], 72], 98] |
-
aThis table summarizes the three immunoediting phases – Elimination, Equilibrium, and Escape – as applied to pregnancy. Each phase is characterized by distinct immune players, mechanisms, and outcomes in normal gestation or pathological conditions. Reference numbers correspond to the sources cited in the manuscript.
In pregnancy, elimination refers to early recognition of fetal antigens; equilibrium represents sustained tolerance through immune regulation; and escape involves breakdowns in tolerance, manifesting as complications like miscarriage or preeclampsia [12], [29], [30], [31], [32], [33] (Table 3). These phases map onto shifting immune landscapes across trimesters.
Immune checkpoints in pregnancy.a
| Checkpoint molecule | Source | Function at maternal-fetal interface | Role in tolerance | Dysregulation in disease | References |
| PD-1/PD-L1 | Trophoblasts, exosomes, decidual stromal cells | Suppresses maternal T cell activity; promotes M2 macrophage polarization | Maintains immune equilibrium and fetal protection | Reduced in preeclampsia and miscarriage | [13], 15], 18], 57], 72] |
| TIM-3 | T cells, NK cells, trophoblasts | Promotes immune exhaustion and tolerance via interaction with galectin-9 | Suppresses Th1/Th17 responses | Associated with recurrent miscarriage and inflammation | [27], 31], 98] |
| CTLA-4 | Regulatory T cells | Inhibits APC co-stimulation via CD80/CD86 | Promotes Treg-mediated suppression of effector responses | Reduced expression linked to pregnancy loss | [6], 9], 98] |
| OX40/OX40L | Activated T cells, decidual tissue | Modulates Treg stability and effector T cell survival | Balances immune activation and regulation | Altered expression in recurrent pregnancy loss | [58], 98] |
| FasL | Trophoblasts, exosomes | Induces apoptosis in activated maternal T cells | Immune silencing and protection of fetal cells | Not fully defined; under investigation | [10], 59], 79] |
| LILRB4 | Myeloid cells, decidual macrophages | Inhibits dendritic cell maturation and T cell activation | Promotes immune suppression and anti-inflammatory macrophage phenotype | Emerging evidence in preeclampsia and immune dysregulation | [43] |
| B7-H4 | Trophoblasts, endometrial epithelium | Suppresses T cell proliferation; promotes immune quiescence | Contributes to fetal immune privilege, similar to tumor escape | Proposed role in immune escape during inflammation | [23] |
| Galectin-9 | Trophoblasts, immune cells | Ligand for TIM-3; promotes T cell exhaustion and tolerance | Enhances TIM-3 mediated immune suppression | Reduced expression may impair tolerance signaling | [27], 31] |
-
aThis table outlines major immune checkpoints involved in pregnancy tolerance, including their sources, roles, and associations with pregnancy complications.
This review is the first to comprehensively apply the immunoediting framework to pregnancy by synthesizing insights from reproductive immunology, oncology, and exosome biology. We explore how immune checkpoints [13], 15], 17], [34], [35], [36], Tregs [5], [10], [11], [12], [13], [14], [15], [16], [17], [18], [19], [20], [21], [22], [23], [24], [25], [26], [27], [28], [29], [30], [31], [32], [33], [34], [35], [36], [37, 37], 38] (Table 4), HLA-G [4], 39], 40], and placenta-derived exosomes [41], [42], [43], [44], [45] (Figure 3) collectively shape maternal-fetal tolerance. Ultimately, we propose reclassifying common obstetric disorders – not as distinct pathologies – but as failures of immune editing. This shift opens new possibilities for diagnostics and therapies inspired by oncology, such as checkpoint modulators, Treg-based strategies, and exosome-targeted interventions [14], [46], [47], [48], [49] (Table 5).
Regulatory T cells in pregnancy.a
| Treg subtype | Source/Recruitment signals | Mechanism of action | Impact on pregnancy | Evidence from Models/Studies |
| CD4+FoxP3+ Tregs | Thymus, peripheral expansion via TGF-β, IL-2 | Suppress effector T cells; secrete IL-10, TGF-β; express CTLA-4 and PD-1 | Essential for implantation, tolerance, and fetal survival | [6], 9], 20], 96], 198] |
| Memory Tregs | Expansion from previous pregnancies or antigen exposure | Rapid recall and enhanced suppressive response at the maternal-fetal interface | Promotes improved tolerance in subsequent pregnancies | [20], 97] |
| CD8+FoxP3+ Tregs | Peripheral induction by fetal antigens and TGF-β | Suppress dendritic cell and T cell activation; cytotoxic regulation | Contributes to early maternal tolerance | [54], 97] |
| Induced (iTregs) | Peripheral naive CD4+ T cells under TGF-β/IL-2 influence | Promotes tolerance through suppressive cytokines and checkpoint expression | Maintains peripheral immune balance at the fetal interface | [20], 78], 98] |
| Tissue-resident Tregs (trTregs) | Localized expansion in decidua via local cytokines and antigens | Provide site-specific immune suppression; adapt to local signals | Ensure site-specific tolerance and placental development | [54], 97], 98] |
| Helios+/Helios− Tregs | Helios+: thymic (nTregs); helios−: Peripherally induced (iTregs) | Both suppress effector T cells, but differ in origin and cytokine profiles | Helios− iTregs are particularly important for fetal-specific tolerance | [56], 66], 98] |
| T-bet+ Tregs | Differentiation under IFN-γ and IL-12 signals in Th1 environment | Suppress Th1-type immune responses; maintain Th1/Treg balance | Prevent pro-inflammatory responses at maternal-fetal interface | [66], 97] |
| RORγt+ Tregs | Peripheral polarization influenced by IL-6 and microbiota | Control Th17 responses and mucosal tolerance | Contribute to immune balance and protection from inflammation | [66], 67], 97] |
-
aThis table summarizes the major subtypes and functional variants of regulatory T cells (Tregs) involved in pregnancy, detailing their origins, mechanisms, and roles in establishing and maintaining maternal-fetal tolerance.
![Figure 3:
Immunomodulatory roles of placental exosomes at the maternal-fetal interface. This illustration highlights how placental exosomes, released from syncytiotrophoblasts, shape maternal immune tolerance during pregnancy. These exosomes carry key immunoregulatory molecules: TRAIL and FasL induce apoptosis in activated lymphocytes. NKG2D-L downregulates the NKG2D receptor on NK cells, impairing cytotoxicity. TGF-β and PD-L1 promote the conversion of naive T cells into regulatory T cells (Tregs). Together, these exosomal signals suppress maternal immune activation, ensuring fetal survival in a semi-allogeneic environment. Figure adapted from Mincheva-Nilsson L and Baranov V [10].](/document/doi/10.1515/jpm-2025-0246/asset/graphic/j_jpm-2025-0246_fig_003.jpg)
Immunomodulatory roles of placental exosomes at the maternal-fetal interface. This illustration highlights how placental exosomes, released from syncytiotrophoblasts, shape maternal immune tolerance during pregnancy. These exosomes carry key immunoregulatory molecules: TRAIL and FasL induce apoptosis in activated lymphocytes. NKG2D-L downregulates the NKG2D receptor on NK cells, impairing cytotoxicity. TGF-β and PD-L1 promote the conversion of naive T cells into regulatory T cells (Tregs). Together, these exosomal signals suppress maternal immune activation, ensuring fetal survival in a semi-allogeneic environment. Figure adapted from Mincheva-Nilsson L and Baranov V [10].
Placenta-derived exosomes – immune cargo and Function.a
| Cargo component | Immune target | Function | Clinical correlation | References |
| PD-L1 | T cells, NK cells | Suppresses T cell activation and cytotoxicity; promotes immune tolerance | Reduced in preeclampsia and miscarriage | [10], 13], 72], 85] |
| HLA-G | NK cells, T cells | Induces immune tolerance by interacting with inhibitory receptors | Low levels linked with RSA and immune activation | [3], 10], 59] |
| miRNAs (e.g., miR-146a, miR-210) | Monocytes, T cells, DCs | Modulate cytokine release, inflammation, and T cell responses | Dysregulated miRNAs in preeclampsia and fetal growth restriction | [10], 78], 85] |
| FasL | Activated T cells | Induces apoptosis in effector T cells | Implicated in immune privilege; variable expression in disorders | [10], 59], 79] |
| Galectin-9 | TIM-3+ T cells, NK cells | Promotes T cell exhaustion and Th1 suppression | Impaired signaling linked with pregnancy loss | [27], 31], 78] |
| TGF-β | T cells, DCs, NK cells | Promotes Treg differentiation and suppresses effector responses | Key factor in establishing immune tolerance; reduced in preeclampsia | [10], 78], 98] |
| IL-10 | T cells, macrophages | Suppresses pro-inflammatory cytokine production and antigen presentation | Reduced IL-10 signaling implicated in miscarriage and PE | [10], 98] |
| Other miRNAs (e.g., miR-155, miR-223) | T cells, APCs | Regulate immune cell differentiation, activation, and cytokine profiles | Altered miRNA profiles linked to inflammation and fetal growth restriction | [78], 85] |
-
aThis table outlines key immunoregulatory components of placenta-derived exosomes, detailing their immune targets, functional roles in maternal-fetal tolerance, and clinical relevance to pregnancy disorders.
Methods
This review employed a multi-phase, integrative approach aimed at developing a comprehensive and theory-driven framework for understanding maternal-fetal immune tolerance through the lens of immunoediting. Rather than following a rigid systematic review or meta-analysis structure, this methodology draws upon elements of concept synthesis, scoping review, and translational modeling to capture the complexity and interdisciplinary nature of the topic [50]–55].
The development of the conceptual framework was guided by the hypothesis that the immunoediting model – originally proposed to describe tumor evolution under immune pressure [19], 20] – can be applied to pregnancy to reinterpret the immunological events governing fetal tolerance [51]. To construct this framework, the review integrated mechanistic, clinical, and theoretical findings from reproductive immunology [2], 3], placental exosome biology [41], immune checkpoint regulation [13], 15], 16], and oncology. This triangulated synthesis aimed to identify common immune mechanisms that contribute to either immune equilibrium or immune escape, depending on physiological or pathological context.
A comprehensive literature search was conducted using PubMed, Scopus, and Web of Science databases for peer-reviewed publications from January 2000 to March 2025 [52]. Search terms included combinations of “maternal-fetal immune tolerance”, “programmed death-ligand 1 (PD-L1) and placenta”, “regulatory T cells (Tregs) in pregnancy”, “immune checkpoint expression in trophoblasts”, “human leukocyte antigen-G (HLA-G)”, “tumor immune escape”, and “immunoediting”. Boolean logic was used to refine the search scope, and results were limited to full-text articles published in English. Both human and murine model studies were included, provided they offered mechanistic insight relevant to pregnancy or tumor immune regulation.
The inclusion criteria consisted of articles that demonstrated immunological relevance to maternal-fetal interaction, immune modulation, checkpoint biology, or clinical outcomes such as preeclampsia, recurrent pregnancy loss, or preterm labor [29], [30], [31], [32], [53]. Articles focusing solely on infectious disease, unrelated autoimmunity, or with inadequate experimental rigor were excluded. Additionally, non-English articles, abstracts without full data, and conference proceedings were omitted from final consideration.
The study selection process followed a three-tiered screening method. Titles and abstracts of all search results were first reviewed for relevance to the review’s objectives. Studies passing this initial screen were read in full and appraised for scientific quality, experimental depth, and alignment with the proposed immunoediting framework. Discrepancies in selection were resolved through discussion among the reviewing authors. A total of 110 articles meeting the criteria were retained for detailed thematic analysis (Figure 4 outlines this PRISMA-guided selection process).
![Figure 4:
Aire-driven T cell fate and immune regulation in pregnancy outcomes. This diagram shows how eTACs (extrathymic aire-expressing cells) guide naive T cells toward either tolerance or effector pathways, influencing pregnancy success. Treg and exhausted T cells promote fetal tolerance and support healthy pregnancy. Th1/Th17/Tfh2 cells drive inflammation, contributing to IUGR or fetal resorption. Figure adapted from Gillis-Buck E et al. [111].](/document/doi/10.1515/jpm-2025-0246/asset/graphic/j_jpm-2025-0246_fig_004.jpg)
Aire-driven T cell fate and immune regulation in pregnancy outcomes. This diagram shows how eTACs (extrathymic aire-expressing cells) guide naive T cells toward either tolerance or effector pathways, influencing pregnancy success. Treg and exhausted T cells promote fetal tolerance and support healthy pregnancy. Th1/Th17/Tfh2 cells drive inflammation, contributing to IUGR or fetal resorption. Figure adapted from Gillis-Buck E et al. [111].
Each study was thematically categorized according to one of three immunoediting phases – elimination, equilibrium, or escape – based on its findings and relevance to immune checkpoints, regulatory cell function, trophoblast signaling, or clinical outcomes (Table 6 summarizes these phases as applied to pregnancy). These themes were further refined into four major mechanistic domains: immune checkpoint regulation, regulatory T cell (Treg) dynamics, HLA-G and major histocompatibility complex (MHC) modulation, and placenta-derived exosomal signaling [57], 58], 63], [66], [67], [68], [69], [70], [71], [72], [73].
Therapeutic and diagnostic implications based on immunoediting.a
| Immune target | Proposed intervention | Status | Potential obstetric application | References |
| PD-1/PD-L1 pathway | Checkpoint agonists or exosomal PD-L1 supplementation | Preclinical | Prevention or treatment of miscarriage, immune-based diagnostics | [13], 15], 72], 78] |
| Regulatory T cells (Tregs) | Treg adoptive transfer or Treg-boosting agents (e.g., low-dose IL-2) | Preclinical/Experimental | Immunotherapy for recurrent pregnancy loss or preeclampsia | [6], 20], 78], 98] |
| TIM-3/Galectin-9 axis | Galectin-9 supplementation or TIM-3 agonism | Preclinical | Restoring tolerance in immune-mediated miscarriage | [27], 31], 98] |
| Exosomal miRNAs | Therapeutic delivery of regulatory miRNAs (e.g., miR-146a) | Exploratory | Diagnostic and therapeutic monitoring for preeclampsia, FGR | [10], 78], 85] |
| TGF-β/IL-10 signaling | Cytokine therapy or engineered exosomes | Experimental | Rebalancing inflammatory responses in complicated pregnancies | [10], 98] |
| CTLA-4 pathway | CTLA-4 agonists or enhancement of Treg-mediated suppression | Preclinical | Boosting tolerance in pregnancy loss or inflammatory complications | [6], 9], 96], 98] |
| OX40/OX40L signaling | OX40 modulation to enhance Treg stability or suppress effector T cells | Experimental | Immune rebalance in recurrent miscarriage | [58], 98] |
| Checkpoint combination therapy (PD-1 + TIM-3) | Dual checkpoint agonists or engineered exosomes | Exploratory | Restoring complex tolerance in severe immune-mediated pregnancy disorders | [31], 58], 98] |
| CD8+FoxP3+ Tregs | Expansion or adoptive transfer of cytotoxic Tregs | Preclinical | Enhancing early maternal-fetal tolerance | [9], 54] |
-
aThis table highlights key immune targets and emerging therapeutic or diagnostic strategies in pregnancy, based on the immunoediting framework. Each entry includes intervention type, development status, and potential clinical applications.
To support interpretation and communication of complex immunological concepts, the review also included several visual models and data representations. These included:
A conceptual diagram illustrating immunological crosstalk at the maternal-fetal interface (Figure 5).
A schematic of placental exosome-mediated immune suppression (Figure 6).

Immune cell crosstalk and checkpoint regulation at the maternal–fetal interface. This illustration compares the immune microenvironment in normal pregnancy across two anatomical contexts: (A) At the maternal–fetal interface, decidual immune cells such as Tregs, dendritic cells (DCs), group 3 ILCs (ILC3s), γδ T cells, and decidual NK cells (dNKs) interact closely with trophoblasts (cytotrophoblast and syncytiotrophoblast) of the chorionic villi. Immune tolerance is promoted by immune checkpoint molecules, including CTLA–CD80/86 signaling, which limits T cell activation. (B) Within the decidual stroma and uterine epithelium, immune populations such as MAIT cells, decidual macrophages (dM1, dM2), and dNK cells are modulated by stromal interactions and checkpoint regulators like Siglec-7, GdA, and CD200R, which help suppress inflammatory responses and support maternal tolerance.
![Figure 6:
PLZF and Eomes: Orchestrators of early-life T cell programming. PLZF and Eomes are key transcription factors that shape early T cell development. PLZF drives the formation of innate-like T cells such as iNKT cells, promoting quick, regulatory responses important in fetal life. Eomes supports the development of cytotoxic and memory-like T cells, priming the immune system for future challenges. Together, they help balance immune tolerance and defense in early life. Figure adapted from Rackaityte E and Halkias J [112].](/document/doi/10.1515/jpm-2025-0246/asset/graphic/j_jpm-2025-0246_fig_006.jpg)
PLZF and Eomes: Orchestrators of early-life T cell programming. PLZF and Eomes are key transcription factors that shape early T cell development. PLZF drives the formation of innate-like T cells such as iNKT cells, promoting quick, regulatory responses important in fetal life. Eomes supports the development of cytotoxic and memory-like T cells, priming the immune system for future challenges. Together, they help balance immune tolerance and defense in early life. Figure adapted from Rackaityte E and Halkias J [112].
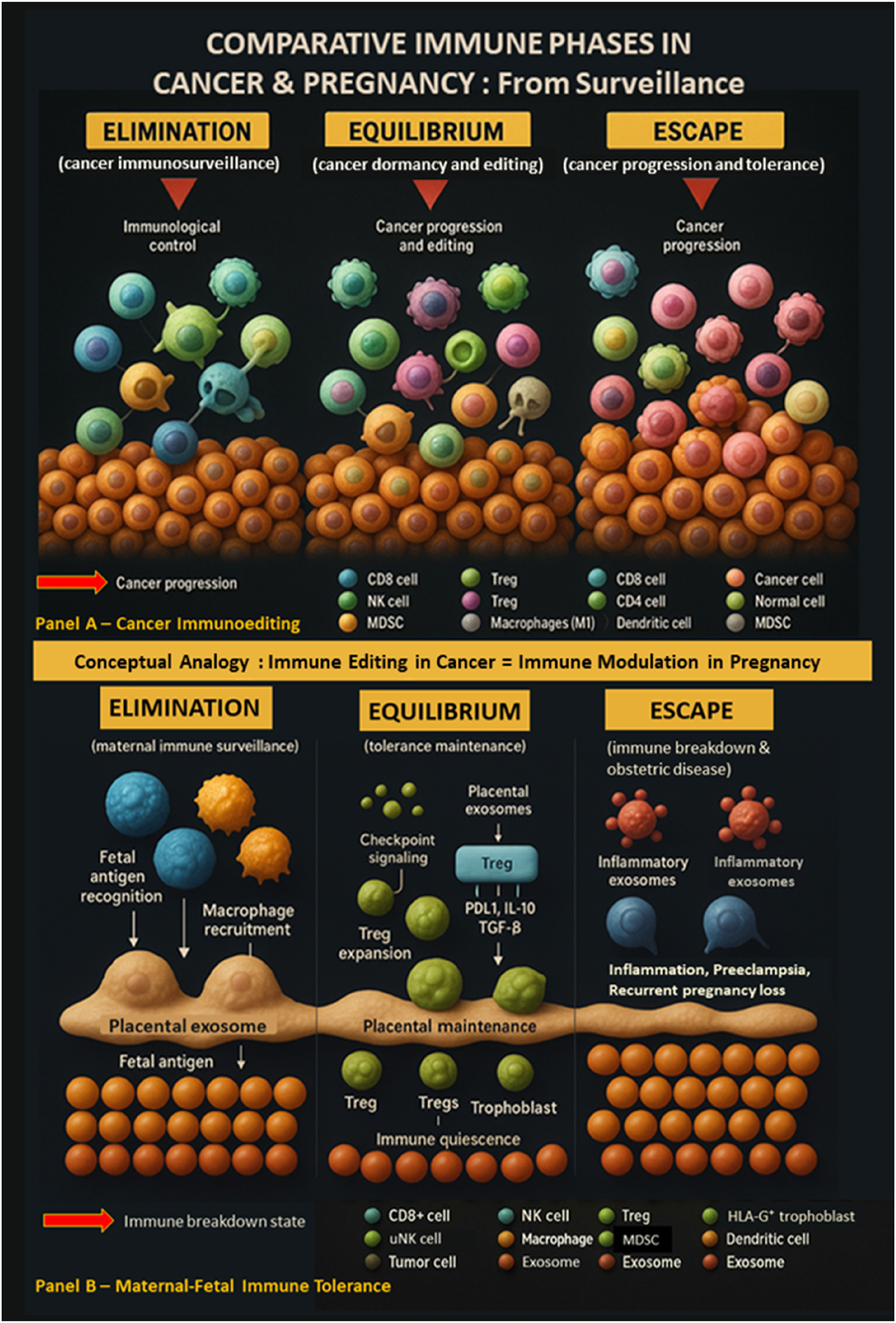
Comparative immune phases in cancer and pregnancy: A conceptual analogy of immunoediting and immune tolerance. This figure illustrates how cancer immunoediting and maternal-fetal immune tolerance follow similar immune phases: Elimination, equilibrium, and escape. In cancer (Panel A), immune cells first eliminate tumor cells. Surviving cells may persist in equilibrium under immune pressure, supported by regulatory elements like Tregs and MDSCs. In escape, tumors evade immune control and progress. In pregnancy (Panel B), maternal immune cells initially recognize fetal antigens. Tolerance is then maintained by Tregs, HLA-G+ trophoblasts, and immunosuppressive signals. When tolerance fails, immune activation leads to complications such as preeclampsia or pregnancy loss. This analogy highlights shared immune dynamics in cancer progression and pregnancy maintenance.

Temporal dynamics of immune regulation across pregnancy. This timeline illustrates the evolving landscape of immune regulation from implantation to delivery. Early pregnancy is marked by the rise of regulatory T cells (Tregs) and immunosuppressive exosomes. During the first and second trimesters, checkpoint molecules such as PD-1/PD-L1 and immunoregulatory cytokines (IL-10, TGF-β) maintain maternal-fetal tolerance. Toward term, uterine NK cells (uNK) and HLA-G interactions support placental stability and fetal protection, completing the immunological orchestration of gestation.

Pathway from immune dysregulation to pregnancy complications. This flowchart depicts how disruptions in immune regulation – such as checkpoint molecule loss (e.g., PD-1/PD-L1, Tim-3), regulatory T cell (Treg) dysfunction, and defective exosomal signaling – lead to immune activation. This dysregulated immune state contributes to key obstetric complications including recurrent pregnancy loss, preeclampsia, and preterm birth. The diagram highlights critical mechanistic links with potential for diagnostic and therapeutic intervention.

Placental exosome cargo in normal pregnancy vs. Preeclampsia. This comparative illustration highlights the molecular contents of placental exosomes under physiological and pathological conditions. In normal pregnancy, exosomes released by syncytiotrophoblasts carry immunoregulatory factors such as PD-L1, FasL, and TGF-β, which support maternal immune tolerance. In contrast, exosomes from preeclamptic placentas show altered cargo, including elevated HLA-G and pro-inflammatory microRNAs (e.g., miR-210, miR-155), contributing to immune dysregulation and endothelial dysfunction. These molecular shifts underscore the diagnostic and therapeutic potential of exosomal profiling in obstetric care.
Comparative immunoediting – pregnancy vs. cancer.a
| Category | Pregnancy | Cancer | References |
| Immunoediting trigger | Fetal alloantigens from the semi-allogeneic fetus | Tumor-specific neoantigens and altered self-proteins | [1], 7], 11], 17], 89], 98] |
| Tolerance mechanisms | Regulatory T cells, exosomes, immune checkpoints (PD-1, CTLA-4), HLA-G | Tregs, MDSCs, checkpoints (PD-1, CTLA-4), IDO, Galectin-9 | [11], 17], 89], 98] |
| Checkpoint roles | Facilitates maternal-fetal tolerance via immune silencing | Enables immune escape and tumor persistence | [13], 15], 17], 27], 98] |
| Treg function | Suppress maternal immune response to fetal antigens | Suppress anti-tumor immunity and promote tumor survival | [6], 9], 20], 96], 98] |
| Outcome if dysregulated | Recurrent miscarriage, preeclampsia, FGR | Tumor progression, metastasis | [5], 13], 15], 72], 98] |
-
aThis table compares the immunoediting processes in pregnancy and cancer, highlighting shared immune mechanisms such as tolerance induction, checkpoint roles, and regulatory T cell function, while emphasizing their distinct biological outcomes.
Immune cell profiles across pregnancy trimesters.a
| Trimester | Dominant immune cells | Functional role | Cytokine environment | References |
| 1st trimester | uNK cells, macrophages, iTregs, Th1 cells | Facilitate implantation, spiral artery remodeling, and immune tolerance initiation | Pro-inflammatory (TNF-α, IFN-γ, IL-1β) | [3], 5], 9], 12], 22], 30], 92] |
| 2nd trimester | Tregs (CD4+, memory), tolerogenic DCs, M2 macrophages | Sustain immune tolerance and promote placental development | Anti-inflammatory (IL-10, TGF-β) | [6], 20], 54], 78], 96], 98] |
| 3rd trimester | Th1/Th17 cells (mild rise), CD8+ T cells, inflammatory macrophages | Prepare for labor; reactivation of immune surveillance | Mildly pro-inflammatory (IL-6, IL-8, TNF-α) | [5], 13], 15], 35], 72], 92] |
-
aThis table summarizes immune cell dynamics across pregnancy trimesters, highlighting dominant cell types, their functions, and cytokine profiles relevant to each phase.
Additionally, Table 1 provides a curated summary of high-impact studies across the immunoediting spectrum, while Table 5 outlines the immune-relevant cargo of placenta-derived exosomes. Tables were also constructed to present key findings on immune checkpoints (Table 3), Treg subtypes (Table 4), and emerging therapeutic strategies (Table 9) [61], [74], [75], [76], [77], [78], [79], [80].
Immunological pathways in obstetric complications.a
| Disorder | Dysregulated pathways | Immune players involved | Biomarker potential | References |
| Preeclampsia (PE) | Checkpoint failure (PD-L1), reduced Tregs, pro-inflammatory cytokines | Tregs, NK cells, macrophages, exosomes | PD-L1, miR-210, IL-10, TGF-β | [13], 15], 72], 78], 85], 98] |
| Recurrent spontaneous abortion (RSA) | Loss of Tregs, reduced checkpoint signaling (TIM-3, PD-1), increased Th1/Th17 | Tregs, Th1 cells, Th17 cells, DCs | Galectin-9, PD-1, IL-17, IFN-γ | [5], 27], 31], 58], 97], 98] |
| Fetal growth restriction (FGR) | Exosomal miRNA imbalance, vascular dysfunction, inflammation | Trophoblasts, monocytes, macrophages | miR-155, miR-210, VEGF, IL-6 | [10], 35], 78], 85], 92] |
-
aThis table summarizes key immune dysregulations and associated biomarkers in major obstetric complications, linking them to specific immune pathways and cell types.
The synthesis of data across molecular, cellular, and systemic levels was designed to generate a coherent and translationally relevant model of pregnancy immunology. Emphasis was placed on identifying mechanisms that may serve as future biomarkers or therapeutic targets in obstetrics, drawing direct analogies to cancer immunotherapy where appropriate [81].
No experimental procedures involving human or animal subjects were performed as part of this review. Therefore, institutional ethics approval was not required. All analyzed studies were published in peer-reviewed journals and are assumed to have adhered to appropriate ethical standards at the time of their original publication. This comprehensive methodology ensured a rigorous, inclusive, and interdisciplinary synthesis of available evidence. By consolidating data from 110 high-quality sources, the review provides a robust immune-centric reinterpretation of pregnancy and its associated disorders through the immunoediting lens.
Results and findings
Literature selection and screening
A PRISMA-guided approach was applied to ensure methodological transparency and reproducibility. Database searches (PubMed, Web of Science, and Scopus) covering January 2000 to March 2025 yielded 3,212 records. After duplicate removal, 1,636 full-text articles were screened based on inclusion criteria focused on maternal-fetal immune tolerance, checkpoint biology, exosomal signaling, and complications such as preeclampsia, recurrent pregnancy loss (RPL), and preterm labor [19], 20], 27], 60], 77], 82], 83] (Figure 4).
Immune elimination: early immune engagement
The elimination phase represents early recognition of fetal antigens by the maternal immune system. This response involves uterine natural killer (uNK) cells, macrophages, and dendritic cells, which generate pro-inflammatory signals facilitating implantation [2], [3], [4], [5, 12], 30], 32] (Figure 5). Extravillous trophoblasts (EVTs) expressing HLA-C and HLA-G modulate these immune cells, promoting immune tolerance [3], 41], 42]. Insufficient HLA-G expression or excessive cytotoxic activity can disrupt implantation, causing early pregnancy loss [5], 34], 41], 42], 71] (Table 1). Similar to failed tumor immune elimination, unchecked immune activation at this stage can impair fetal survival [6], 29], 56], 59], 62], 65], 84]–108]. A key transition point toward equilibrium is expansion of regulatory T cells (Tregs), as demonstrated by Aluvihare et al. and Zenclussen et al. [6], 9] (Table 2, Figure 6).
Immune equilibrium: sustained tolerance
During equilibrium, fetal antigens persist, yet immune responses are actively regulated. Central mediators include CD4+FoxP3+ Tregs, recruited by trophoblast-secreted factors such as transforming growth factor beta (TGF-β) and interleukin 10 (IL-10), as well as placenta-derived exosomes [6], 8], 9], 20], 25], 26], 59], 60], 76], [83], [84], [85], [86], [87], [88], [89], [90], [91], [92], [93], [94], [95], [96], [97], [98], [99] (Table 4). Exosomes enriched in PD-L1, Fas ligand (FasL), and HLA-G suppress maternal cytotoxic T cell responses [10], 59], 65], 79], 86], 87] (Figure 7). Similar to tumor-derived exosomes, placental exosomes act as immune-modulatory packages that maintain tolerance [65], 78], 82], 88], [108], [109], [110] (Table 5). Breakdown of this phase – due to Treg insufficiency [97], 98], PD-L1 downregulation [13], 15], 18], 57], 72], [103], [104], [105], [106], or altered exosome composition [65], 78], 85], 88], 89] – is associated with RPL and preeclampsia [14], 35], 41], 56], [91], [92], [93] (Table 8, Figure 8).
Immune escape: failure of tolerance and disease onset
The escape phase parallels immune evasion in malignancies [17], 56], 88], [89], [90], [91], [92], [93], [94, 109], 110]. In pregnancy, failure of tolerance results in obstetric disorders such as preeclampsia, preterm birth, or fetal growth restriction (FGR). This phase is characterized by decreased PD-L1 expression, reduced Treg activity, and heightened Th1/Th17 cell responses [15], 30], 62], 72], [95], [96], [97], [98], [99], [100], [101], [102], [103]. Activated macrophages and dendritic cells regain antigen-presenting capacity, driving maternal cytotoxic T lymphocyte infiltration and systemic inflammation [18], 35], 63], 93], [102], [103], [104], [105], [106]. Exosomes lose tolerogenic signals and may carry damage-associated molecular patterns (DAMPs) [85], 89]. These processes closely resemble malignant immune escape pathways [88], 108], 110] (Figure 9, Table 9). Preclinical interventions – including PD-L1 or TIM-3 restoration and Treg cell transfer – have shown efficacy in rescuing pregnancies in animal models [21], 27], 31], 71], 111], 112] (Table 6).
PD-1/PD-L1 axis in gestation
The PD-1/PD-L1 pathway is a central regulator of maternal-fetal tolerance. PD-L1 expression on trophoblasts, exosomes, and decidual stromal cells suppresses maternal CD8+ T cell cytotoxicity [13], 15], 18], 57], 70], 74], 81], [103], [104], [105], [106]. PD-L1 blockade in mice induces fetal resorption, highlighting its physiological importance [21], 44]. In humans, PD-L1 downregulation is observed in preeclampsia and miscarriage [15], 58], 72], [104], [105], [106]. Beyond T cell suppression, PD-L1 drives M2 macrophage polarization, supporting placental vascular remodeling and nutrient exchange [18], 29], 33], 107] (Table 3). PD-L1 expression is modulated by hypoxia-inducible factors (HIFs) and nuclear receptor signaling [75], 107], mechanisms also exploited by tumors for immune evasion [19], 62], [106], [107], [108].
Regulatory T cells: immune calibration mechanism
Tregs are indispensable for maintaining maternal-fetal tolerance. Their early gestational expansion is triggered by fetal alloantigens and supported by cytokines such as IL-10, TGF-β, and inhibitory receptors like cytotoxic T lymphocyte-associated antigen 4 (CTLA-4) [6], 20], . Treg dysfunction is linked to preeclampsia, RPL, and intrauterine growth restriction (IUGR) [14], 35], 98]–102]. Their activity is sensitive to inflammatory signals and metabolic stress pathways [64], [66], [67], [68, 102], 103]. Notably, CD8+FoxP3+ Tregs have emerged as an additional regulatory subset, though understudied [54], 89], 110]. Animal studies demonstrate that Treg-based therapy can restore tolerance and prevent fetal loss [21], 96], 98], 111], 112] (Table 6, Figure 10).
Discussion
Rethinking pregnancy as a dynamic immunological process
For much of modern medical history, pregnancy was regarded as a passive immunological state characterized by maternal immune suppression to prevent fetal rejection. However, a growing body of human and animal studies now challenges this static model. Contemporary evidence reveals that pregnancy is, in fact, immunologically dynamic and governed by tightly regulated mechanisms of immune surveillance and tolerance. These insights are supported by a spectrum of studies across immunology, reproductive biology, and oncology, illustrating that gestation is an actively modulated state of immune equilibrium rather than immune dormancy [2], 3], 6], 12], 24], 56], 90] (Figure 4).
This review introduces the cancer-derived immunoediting model – comprising the sequential phases of elimination, equilibrium, and escape – as a unifying theoretical framework to interpret maternal-fetal immune interactions [1], 7], 17], 109] (Figure 5, Table 7). Applying this triphasic paradigm to gestation clarifies the immunological transitions that underpin implantation, tolerance, and obstetric pathology. The elimination phase maps onto early pro-inflammatory immune responses at implantation; the equilibrium phase reflects sustained immune regulation via Tregs, PD-L1 expression, and exosomal signaling; and the escape phase corresponds to breakdowns in tolerance that lead to clinical syndromes such as preeclampsia, recurrent pregnancy loss, and fetal growth restriction [5], 14], 30], 41], 92], 93], 98] (Tables 2 and 9).
Importantly, this model recasts immune dysfunction not as a secondary phenomenon but as a possible initiating factor in obstetric complications [5], 15], 30], 35], 71], 98] (Figure 9). It elevates the immune system from a background player to a primary determinant of gestational outcome, a role it also holds in cancer biology through tumor surveillance and immune escape [1], 88], 89].
Pregnancy vs. malignancy: limits of the analogy
While the immunoediting framework derived from oncology provides a valuable heuristic for understanding maternal-fetal immune tolerance, pregnancy and malignancy are fundamentally distinct biological processes [11], 88], 110]. A fetus is a semi-allogeneic but physiologically intended entity, the product of evolutionary pressure to support species survival [2], 3], 24], whereas a tumor is an abnormal, pathological proliferation of cells designed to escape immune surveillance [88], 89], 62].
The immune suppression observed in pregnancy is highly localized and temporally regulated, aimed at protecting both maternal and fetal well-being without compromising systemic host defense [90], 92], 101]. In contrast, tumors exploit immune escape pathways – such as PD-1/PD-L1 upregulation and regulatory T cell recruitment – for unchecked growth and dissemination, often at the expense of host survival [17], 88], 108], 109].
Moreover, the maternal-fetal interface involves a dual-patient dynamic, where therapeutic decisions must consider both maternal and fetal outcomes – a complexity not present in cancer immunotherapy [20], 96], 108]. Pregnancy also displays unique evolutionary adaptations, such as non-classical HLA-G expression and placental exosome-mediated immune regulation [10], 25], 59], 76], 79], that have no direct equivalent in cancer biology [73], 78], 88].
Thus, the analogy should be viewed as conceptual and hypothesis-generating, not as an assertion of equivalence. It serves to highlight overlapping mechanisms (e.g., PD-1/PD-L1, Treg induction, exosome-mediated signaling) while recognizing that pregnancy represents a symbiotic rather than a pathogenic state [11], 84], 87].
Translational opportunities from oncology to obstetrics
The convergence of immune mechanisms in pregnancy and cancer suggests novel opportunities for therapeutic crossover. Immune checkpoint inhibitors, Treg-based interventions, and exosome-targeted therapies – currently revolutionizing oncology – may hold translational promise for obstetrics [19], 44], 46], 78], 108], 111], 112] (Table 6). In particular, the PD-1/PD-L1 axis has emerged as a cornerstone of maternal-fetal tolerance. Its expression on trophoblasts and exosomes helps suppress cytotoxic T cell activity and maintain fetal viability [13], 15], 18], 57], 59], 72] (Table 3, Figure 6). Reduced PD-L1 levels in the placenta and maternal circulation are consistently associated with preeclampsia and pregnancy loss, supporting its role as both a mechanistic factor and a potential biomarker [15], 58], 72], 78], 85] (Figure 10).
Treg-based therapies, such as adoptive transfer or pharmacological expansion of regulatory T cells, have been shown in preclinical models to restore immune tolerance and prevent fetal resorption [20], 21], 96], 98] (Table 4). The emerging recognition of CD8+FoxP3+ Tregs and memory Treg subsets further broadens the therapeutic landscape [54], 97], 99]. Additionally, placenta-derived exosomes, enriched with tolerogenic proteins such as HLA-G, FasL, and PD-L1, offer a promising delivery system for localized immunomodulation [10], 25], 59], 76]–97] (Table 5). This exosome-based approach mirrors current efforts in oncology to use nanotherapeutics for immune reprogramming [83], 89], 108], 111], 112].
However, the objective in pregnancy is not immune activation but precisely timed immune modulation. Interventions must preserve systemic maternal immunity while selectively enhancing tolerance at the maternal-fetal interface [2], 24], 90]. Such nuanced interventions require new models of immune timing and immune profiling, tailored specifically to the pregnant state.
Clinical implications for obstetric practice
Framing pregnancy through the lens of immunoediting also opens pathways for clinical application. Immune-based diagnostics, widely used in oncology, could be adapted for obstetrics to improve early detection and risk stratification. PD-L1 levels in maternal blood and placental tissue have already demonstrated predictive value for preeclampsia and recurrent pregnancy loss [13], 15], 57], 58], 72]. Likewise, shifts in the Th17/Treg balance mirror immune dysregulation patterns observed in autoimmunity and graft rejection, and may be leveraged to identify pregnancies at risk for immune-mediated complications [30], 98], 100].
Exosomes in maternal circulation – detectable in the first trimester – carry immunological cargo such as PD-L1, HLA-G, cytokines, and microRNAs that reflect the status of immune regulation at the maternal-fetal interface [10], 25], 76], 85], 59] (Figure 10). Profiling this exosomal content could provide a minimally invasive method for longitudinal immune surveillance during pregnancy [78], [79], [80] (Table 5). Integration of such immune diagnostics into prenatal care would allow clinicians to identify immune deviations before they translate into clinical disease.
Ethical and policy considerations
The adaptation of immunotherapeutic approaches to pregnancy raises complex ethical and regulatory questions. Unlike cancer treatment, which targets pathology within a single host, obstetric immunomodulation must account for the well-being of both mother and fetus. Immune interventions must be precisely calibrated to avoid unintended consequences. While enhancing tolerance may prevent fetal loss, overmodulation could impair maternal defense mechanisms or reduce vaccine efficacy [8], 14], 64], 66]. Conversely, insufficient control may trigger fetal rejection or contribute to placental insufficiency [35], 72], 92] (Table 9).
Regulatory frameworks and clinical trial designs must therefore incorporate dual-host considerations, with long-term maternal and fetal safety as a central concern [20], 98], 110]. Furthermore, equitable access to emerging diagnostic technologies, such as immune checkpoint assays and exosome profiling platforms, must be ensured. Many of the populations most affected by immune-mediated pregnancy complications – such as those experiencing eclampsia in low-resource settings – face systemic barriers to care [14], 35], 92]. Expanding access will require international policy support, investment in affordable technologies, and inclusive research practices [79], 85].
Call to action – rethinking obstetric immunology
Pregnancy must be recognized as a programmable immune state rather than an immune-suppressed condition. Clinicians and researchers should adopt immune profiling, develop immune-based diagnostic tools, and explore targeted immunotherapies to prevent and manage pregnancy complications.
Key takeaways
Novel Perspective: Pregnancy framed as an immune-edited process rather than passive immune suppression.
Interdisciplinary Integration: Uses oncology principles (PD-1/PD-L1, Tregs, exosomes) to interpret obstetric immunology.
Translational Potential: Diagnostics (immune profiling), therapies (Treg-based, checkpoint-targeted), and biomarkers (exosomes).
Limitations: Conceptual nature, limited direct validation, experimental nature of proposed interventions.
Ethics & Policy: Dual-host considerations and equitable access are essential for clinical translation.
Implementation checklist for future research
Perform longitudinal immune profiling (checkpoint expression, Treg subsets) across pregnancy.
Expand in vivo studies on placental exosome signaling and immune regulation.
Design clinical trials for immune-targeted therapies with strict ethical oversight.
Develop non-invasive biomarker panels (PD-L1, HLA-G, exosomal miRNAs).
Build ethical and policy frameworks ensuring equitable access and patient-centered consent.
Strengths, limitations, and future directions
This review presents a novel and integrative perspective by systematically applying the cancer-derived immunoediting model to pregnancy. By framing maternal-fetal immune interactions through the phases of elimination, equilibrium, and escape, the model organizes a wide range of molecular and clinical observations into a coherent immunological structure. This includes key processes such as immune checkpoint signaling, regulatory T cell (Treg) dynamics, and placental exosome activity, all of which are linked to clinical outcomes like preeclampsia, recurrent pregnancy loss (RPL), and preterm labor. Rather than portraying pregnancy as a static state of tolerance, this model recasts it as a dynamic, time-sensitive immune process shaped by surveillance and regulation.
A major strength of this framework lies in its interdisciplinary reach. Drawing from 110 studies across both human and animal research, it integrates mechanistic findings on PD-1/PD-L1 pathways, Treg biology, non-classical MHC molecules (HLA-G), and exosomal signaling at the maternal-fetal interface. These components are synthesized into a temporally structured immune model that parallels immune escape in oncology, offering both theoretical clarity and potential clinical applications.
From a translational standpoint, the model highlights new opportunities for diagnostics and intervention. Immune profiling using markers like PD-L1 or Treg signatures, already in use in cancer medicine, could be adapted for early detection and risk stratification in pregnancy. Preclinical studies suggest that Treg-based therapies – such as adoptive cell transfer or pharmacologic expansion – may help restore tolerance and prevent fetal rejection in high-risk pregnancies. Similarly, the immunoregulatory capacity of placental exosomes could be harnessed through engineering approaches that deliver tolerogenic agents. These translational extensions demonstrate the broader utility of viewing pregnancy as an immune-edited process rather than an immunological anomaly.
However, this model also has important limitations. Its application to pregnancy is primarily conceptual and interpretive. While analogies with tumor immune escape provide a useful framework, direct empirical validation in human gestation remains limited. Fundamental biological differences between fetal symbiosis and tumorigenesis – including their evolutionary goals and host contexts – warrant careful distinction. Furthermore, many of the proposed interventions, such as checkpoint-targeted therapies and exosome engineering, are still experimental and untested in human pregnancy. Their safety, optimal timing, and long-term effects on both mother and fetus must be thoroughly evaluated in large, well-characterized cohorts.
To move from theory to practice, future research should focus on longitudinal immune profiling across all trimesters of pregnancy. This includes mapping changes in checkpoint expression, cytokine networks, and Treg subsets to distinguish healthy immune trajectories from those associated with pathology. More in vivo studies are needed to elucidate the role of exosomes under both normal and inflammatory conditions. Immunomodulatory therapies should be tested in rigorously designed clinical trials with ethical oversight that addresses the dual-patient nature of pregnancy and considers both immediate and long-term fetal outcomes.
The development of non-invasive immune biomarkers, such as circulating PD-L1, HLA-G, or exosomal microRNAs, could allow for early detection of immune imbalance and enable more personalized management strategies. Equally important are policy and ethical frameworks that can support these innovations. Informed consent processes must reflect the complexity of immune interventions in pregnancy, while regulatory guidance should prioritize maternal-fetal safety. Global access must also be addressed, particularly in settings where immune-mediated complications are most prevalent and resources are scarce.
Conclusions
This review redefines pregnancy not as a passive state of immune suppression but as a dynamic, immune-edited process governed by phases of elimination, equilibrium, and escape – concepts originally derived from oncology. By synthesizing evidence from 110 studies, we demonstrate that immune checkpoints (PD-1/PD-L1), regulatory T cells (Tregs), HLA-G/MHC signaling, and placenta-derived exosomes orchestrate maternal-fetal immune tolerance. When these mechanisms fail, immune escape pathways emerge, manifesting as obstetric complications including preeclampsia, recurrent pregnancy loss, and preterm birth.
The proposed immunoediting framework provides both conceptual clarity and translational opportunity. It suggests that pregnancy complications may often stem from primary immune dysregulation, rather than secondary consequences of placental dysfunction alone. This opens new frontiers for immune-based diagnostics, non-invasive biomarkers, and targeted immunomodulation informed by advances in oncology.
However, moving from theory to clinical application will require longitudinal immune profiling, in vivo validation, and carefully regulated clinical trials designed for the unique dual-patient context of pregnancy. Ethical considerations – including patient consent, fetal safety, and equitable access – must be central to these efforts. Ultimately, adopting an immune-centric perspective has the potential to transform obstetric care from reactive disease management to precision-based early intervention, improving outcomes for both mother and child.
Acknowledgments
The authors acknowledge the invaluable support of the Indonesian Society of Obstetrics & Gynecology (ISOG/POGI) and Indonesian Association of Maternal Fetal Medicine (IAMFM/HKFM) in facilitating this review article.
-
Research ethics: Not applicable.
-
Informed consent: Not applicable.
-
Author contributions: The authors have accepted responsibility for the entire content of this manuscript and approved its submission.
-
Use of Large Language Models, AI and Machine Learning Tools: None declared.
-
Conflict of interest: The authors state no conflict of interest.
-
Research funding: None declared.
-
Data availability: Not applicable.
References
1. Dunn, GP, Old, LJ, Schreiber, RD. The immunobiology of cancer immunoediting and immunoediting. Immunity 2004;21:137–48. https://doi.org/10.1016/j.immuni.2004.07.017.Search in Google Scholar PubMed
2. Mor, G, Aldo, P, Alvero, AB. The unique immunological and microbial aspects of pregnancy. Nat Rev Immunol 2017;17:469–82. https://doi.org/10.1038/nri.2017.64.Search in Google Scholar PubMed
3. Tilburgs, T, Evans, JH, Crespo, ÂC, Strominger, JL. The HLA-G cycle provides for both NK tolerance and immunity at the maternal-fetal interface. Proc Natl Acad Sci USA 2015;112:13312–7. https://doi.org/10.1073/pnas.1517724112.Search in Google Scholar PubMed PubMed Central
4. Lanier, LL. NK cell recognition. Annu Rev Immunol 2005;23:225–74. https://doi.org/10.1146/annurev.immunol.23.021704.115526.Search in Google Scholar PubMed
5. Zhou, Y, Fu, B, Xu, X, Zhang, J. The role of immune cells in recurrent spontaneous abortion. Reprod Sci 2016;23:1215–25. https://doi.org/10.1177/1933719116630410.Search in Google Scholar PubMed PubMed Central
6. Aluvihare, VR, Kallikourdis, M, Betz, AG. Regulatory T cells mediate maternal tolerance to the fetus. Nat Immunol 2004;5:266–71. https://doi.org/10.1038/ni1037.Search in Google Scholar PubMed
7. Schreiber, RD, Old, LJ, Smyth, MJ. Cancer immunoediting: integrating immunity’s roles in cancer suppression and promotion. Science 2011;331:1565–70. https://doi.org/10.1126/science.1203486.Search in Google Scholar PubMed
8. La Rocca, C, Carbone, F, Longobardi, S, Matarese, G. The immunology of pregnancy: regulatory T cells control maternal immune tolerance toward the fetus. Immunol Lett 2014;162:41–8. https://doi.org/10.1016/j.imlet.2014.06.006.Search in Google Scholar PubMed
9. Zenclussen, AC, Gerlof, K, Zenclussen, ML, Ritschel, S, Zambon Bertoja, A, Fest, S, et al.. Regulatory T cells induce a privileged tolerant microenvironment at the fetal-maternal interface. Eur J Immunol 2006;36:82–94. https://doi.org/10.1002/eji.200535428.Search in Google Scholar PubMed
10. Mincheva-Nilsson, L, Baranov, V. Placenta-derived exosomes and syncytiotrophoblast microparticles and their role in human reproduction: immune modulation for pregnancy success. Am J Reprod Immunol 2014;72:440–57. https://doi.org/10.1111/aji.12230.Search in Google Scholar PubMed PubMed Central
11. Costanzo, V, Bardelli, A, Siena, S, Abrignani, S. Exploring the links between cancer and placenta development. Open Biol. 2018;8:180081. https://doi.org/10.1098/rsob.180081.Search in Google Scholar PubMed PubMed Central
12. Moffett, A, Colucci, F. Uterine NK cells: active regulators at the maternal-fetal interface. J Clin Investig 2014;124:1872–9. https://doi.org/10.1172/JCI68107.Search in Google Scholar PubMed PubMed Central
13. Zhang, Y, Wang, H, Qiu, P, Jiang, J, Wu, X, Mei, J, et al.. Decidual macrophages derived NO downregulates PD-L1 in trophoblasts leading to decreased Treg cells in recurrent miscarriage. Front Immunol 2023;14:1180154. https://doi.org/10.3389/fimmu.2023.1180154.Search in Google Scholar PubMed PubMed Central
14. Saito, S, Nakashima, A, Shima, T, Ito, M. Clinical implication of recent advances in our understanding of immune cell behavior in preeclampsia. J Reprod Immunol 2007;76:1–8. https://doi.org/10.1016/j.jri.2007.05.002.Search in Google Scholar PubMed
15. Rong, QX, Wang, F, Guo, ZX, Hu, Y, An, SN, Luo, M, et al.. GM-CSF mediates immune evasion via upregulation of PD-L1 expression in extranodal natural killer/T cell lymphoma. Mol Cancer 2021;20:80. https://doi.org/10.1186/s12943-021-01374-y.Search in Google Scholar PubMed PubMed Central
16. Guleria, I, Sayegh, MH. Maternal acceptance of the fetus: true human tolerance. J Immunol 2005;174:3345–51. https://doi.org/10.4049/jimmunol.174.6.3345.Search in Google Scholar
17. Mittal, D, Gubin, MM, Schreiber, RD, Smyth, MJ. New insights into cancer immunoediting and its three component phases – elimination, equilibrium and escape. Curr Opin Immunol 2014;27:16–25. https://doi.org/10.1016/j.coi.2014.01.004.Search in Google Scholar PubMed PubMed Central
18. Zhang, Y, Ma, L, Hu, X, Ji, J, Mor, G, Liao, A. The role of the PD-1/PD-L1 axis in macrophage differentiation and function during pregnancy. Hum Reprod 2019;34:25–36. https://doi.org/10.1093/humrep/dey330.Search in Google Scholar PubMed PubMed Central
19. Dong, H, Strome, SE, Salomao, DR, Tamura, H, Hirano, F, Flies, DB, et al.. Tumor-associated B7-H1 promotes T-cell apoptosis: a potential mechanism of immune evasion. Nat Med 2002;8:793–800. https://doi.org/10.1038/nm730.Search in Google Scholar PubMed
20. Samstein, RM, Josefowicz, SZ, Arvey, A, Treuting, PM, Rudensky, AY. Extrathymic generation of regulatory T cells in placental mammals mitigates maternal-fetal conflict. Cell. 2012;150:29–38. https://doi.org/10.1016/j.cell.2012.05.031.Search in Google Scholar PubMed PubMed Central
21. Zhang, T, Fresnay, S, Welty, E, Sangrampurkar, N, Rybak, E, Zhou, H, et al.. Selective CD28 blockade attenuates acute and chronic rejection of murine cardiac allografts in a CTLA-4-dependent manner. Am J Transplant 2011;11:1599–609. https://doi.org/10.1111/j.1600-6143.2011.03624.x.Search in Google Scholar PubMed PubMed Central
22. Vacca, P, Vitale, C, Montaldo, E, Conte, R, Cantoni, C, Fulcheri, E, et al.. CD34+ hematopoietic precursors are present in human decidua and differentiate into natural killer cells upon interaction with stromal cells. Proc Natl Acad Sci USA 2011;108:2402–7. https://doi.org/10.1073/pnas.1016257108.Search in Google Scholar PubMed PubMed Central
23. Yu, J, Yan, Y, Li, S, Xu, Y, Parolia, A, Rizvi, S, et al.. Progestogen-driven B7-H4 contributes to onco-fetal immune tolerance. Cell 2024;187:4713–32.e19. https://doi.org/10.1016/j.cell.2024.06.012.Search in Google Scholar PubMed PubMed Central
24. Wang, J, Han, T, Zhu, X. Role of maternal-fetal immune tolerance in the establishment and maintenance of pregnancy. Chin Med J (Engl). 2024;137:1399–406. https://doi.org/10.1097/CM9.0000000000003114.Search in Google Scholar PubMed PubMed Central
25. Burlingham, WJ. Extracellular vesicles in fetal-maternal immune tolerance. Biomed J 2024;47:100785. https://doi.org/10.1016/j.bj.2024.100785.Search in Google Scholar PubMed PubMed Central
26. Joo, JS, Lee, D, Hong, JY. Multi-layered mechanisms of immunological tolerance at the maternal-fetal interface. Immune Netw 2024;24:e30. https://doi.org/10.4110/in.2024.24.e30.Search in Google Scholar PubMed PubMed Central
27. Zhu, W, Tan, YQ, Wang, FY. Tim-3: an inhibitory immune checkpoint is associated with maternal-fetal tolerance and recurrent spontaneous abortion. Clin Immunol 2022;245:109185. https://doi.org/10.1016/j.clim.2022.109185.Search in Google Scholar PubMed
28. Blazheva, S, Pachkova, S, Bodurska, T, Ivanov, P, Blazhev, A, Lukanov, T, et al.. Unlocking the uterine code: microbiota, immune cells, and therapy for recurrent reproductive failure. Microorganisms 2024;12:547. https://doi.org/10.3390/microorganisms12030547.Search in Google Scholar PubMed PubMed Central
29. Guo, XL, Wang, YD, Liu, YJ, Chu, L, Zhu, H, Hu, Y, et al.. Fetal hepatocytes protect the HSPC genome via fetuin-A. Nature. 2025;637:402–11. https://doi.org/10.1038/s41586-024-08307-x. Erratum in: Nature. 2025;639:E3. https://doi.org/10.1038/s41586-025-08714-8Search in Google Scholar PubMed PubMed Central
30. Wang, W, Sung, N, Gilman-Sachs, A, Kwak-Kim, J. T helper (Th) cell profiles in pregnancy and recurrent pregnancy losses: Th1/Th2/Th9/Th17/Th22/Tfh cells. Front Immunol 2020;11:2025. https://doi.org/10.3389/fimmu.2020.02025.Search in Google Scholar PubMed PubMed Central
31. Meng, X, Luo, Y, Cui, L, Wang, S. Involvement of Tim-3 in maternal-fetal tolerance: a review of current understanding. Int J Biol Sci 2025;21:789–801. https://doi.org/10.7150/ijbs.106115.Search in Google Scholar PubMed PubMed Central
32. Negishi, Y, Takahashi, H, Kuwabara, Y, Takeshita, T. Innate immune cells in reproduction. J Obstet Gynaecol Res 2018;44:2025–36. https://doi.org/10.1111/jog.13759.Search in Google Scholar PubMed
33. Cai, S, Xue, B, Li, S, Wang, X, Zeng, X, Zhu, Z, et al.. Methionine regulates maternal-fetal immune tolerance and endometrial receptivity by enhancing embryonic IL-5 secretion. Cell Rep 2025;44:115291. https://doi.org/10.1016/j.celrep.2025.115291.Search in Google Scholar PubMed
34. Rizzuto, G. B cell responses to the placenta and fetus. Annu Rev Pathol 2025;20:33–58. https://doi.org/10.1146/annurev-pathmechdis-111523-023459.Search in Google Scholar PubMed PubMed Central
35. Jacobsen, DP, Fjeldstad, HE, Olsen, MB, Sugulle, M, Staff, AC. Microchimerism and pregnancy complications with placental dysfunction. Semin Immunopathol 2025;47:21. https://doi.org/10.1007/s00281-025-01045-w.Search in Google Scholar PubMed PubMed Central
36. Prescott, S, Billeci, N, Gotcher, M, Patel, S, Almon, A, Morgan, H, et al.. Tryptophan as a biomarker of pregnancy-related immune expression and modulation: an integrative review. Front Reprod Health 2025;6:1453714. https://doi.org/10.3389/frph.2024.1453714.Search in Google Scholar PubMed PubMed Central
37. Gougis, P, Hamy, AS, Jochum, F, Bihan, K, Carbonnel, M, Salem, JE, et al.. Immune checkpoint inhibitor use during pregnancy and outcomes in pregnant individuals and newborns. JAMA Netw Open 2024;7:e245625. https://doi.org/10.1001/jamanetworkopen.2024.5625.Search in Google Scholar PubMed PubMed Central
38. Zych, M, Kniotek, M, Roszczyk, A, Dąbrowski, F, Jędra, R, Zagożdżon, R. Surface immune checkpoints as potential biomarkers in physiological pregnancy and recurrent pregnancy loss. Int J Mol Sci 2024;25:9378. https://doi.org/10.3390/ijms25179378.Search in Google Scholar PubMed PubMed Central
39. Huang, J, Feng, L, Huang, J, Zhang, G, Liao, S. Unveiling sialoglycans’ immune mastery in pregnancy and their intersection with tumor biology. Front Immunol 2024;15:1479181. https://doi.org/10.3389/fimmu.2024.1479181.Search in Google Scholar PubMed PubMed Central
40. Vanders, RL, Gomez, HM, Daly, K, Wark, PA, Horvat, JC, Hansbro, PM. Immune checkpoints are suppressed during pregnancy following influenza A virus infection. Am J Physiol Lung Cell Mol Physiol 2024;327:L890–904. https://doi.org/10.1152/ajplung.00391.2023.Search in Google Scholar PubMed
41. Meggyes, M, Feik, T, Nagy, DU, Polgar, B, Szereday, L. CD8 and CD4 positive NKT subpopulations and immune-checkpoint pathways in early-onset preeclampsia and healthy pregnancy. Int J Mol Sci 2023;24:1390. https://doi.org/10.3390/ijms24021390.Search in Google Scholar PubMed PubMed Central
42. Wang, X, Li, L, Liu, D, Jin, Y, Zhao, X, Li, S, et al.. LILRB4 as a novel immunotherapeutic target for multiple diseases. Biochem Pharmacol 2025;233:116762. https://doi.org/10.1016/j.bcp.2025.116762.Search in Google Scholar PubMed
43. Hu, X, Lai, S, Liao, A. Immune checkpoint for pregnancy. Semin Immunopathol 2025;47:26. https://doi.org/10.1007/s00281-025-01051-y.Search in Google Scholar PubMed
44. Li, P, Zeng, L, Yan, X, Zhu, Z, Gu, Q, He, X, et al.. Molecular and cellular morphology of placenta unveils new mechanisms of reproductive immunology. J Adv Res 2025;77:605–21.https://doi.org/10.1016/j.jare.2025.01.025.Search in Google Scholar PubMed PubMed Central
45. Sharon, E. Immune checkpoint inhibitors and pregnancy: considerations for treating medically complex patients with the best available therapy. Semin Perinatol 2025;49:152045. https://doi.org/10.1016/j.semperi.2025.152045.Search in Google Scholar PubMed
46. Kitazawa, R, Kitazawa, S. On the similarity between postpartum autoimmune abnormalities and immune-related adverse events (irAE) by immune checkpoint inhibitors (ICI): the placenta as a PD-L1-rich immune-tolerant organ. Acta Histochem Cytoc 2025;58:1–8. https://doi.org/10.1267/ahc.24-00056EP.Search in Google Scholar PubMed PubMed Central
47. Shabir, I, Foster, C. Immune checkpoint inhibitors impact fertility: a review. Rev Recent Clin Trials 2025;20:185–90. https://doi.org/10.2174/0115748871340496250130054721.Search in Google Scholar PubMed
48. Wiley, KS, Martínez, LE, Kwon, D, Knorr, DA, Epeldegui, M, Fox, MM. Regulatory B-cells are associated negatively with regulatory T-cells and positively with cytokines in peripheral blood of pregnant women. Am J Reprod Immunol 2025;93:e70027. https://doi.org/10.1111/aji.70027.Search in Google Scholar PubMed PubMed Central
49. Vilbois, S, Xu, Y, Ho, PC. Metabolic interplay: tumor macrophages and regulatory T cells. Trends Cancer 2024;10:242–55. https://doi.org/10.1016/j.trecan.2023.11.007.Search in Google Scholar PubMed
50. Zong, Y, Deng, K, Chong, WP. Regulation of Treg cells by cytokine signaling and co-stimulatory molecules. Front Immunol 2024;15:1387975. https://doi.org/10.3389/fimmu.2024.1387975.Search in Google Scholar PubMed PubMed Central
51. Yue, Y, Ren, Y, Lu, C, Li, P, Zhang, G. Epigenetic regulation of human FOXP3+ Tregs: from homeostasis maintenance to pathogen defense. Front Immunol 2024;15:1444533. https://doi.org/10.3389/fimmu.2024.1444533.Search in Google Scholar PubMed PubMed Central
52. Morgun, EI, Govorova, IA, Chernysheva, MB, Machinskaya, MA, Vorotelyak, EA. Mini-review: Tregs as a tool for therapy – obvious and non-obvious challenges and solutions. Cells 2024;13:1680. https://doi.org/10.3390/cells13201680.Search in Google Scholar PubMed PubMed Central
53. Luo, S, Larson, JH, Blazar, BR, Abdi, R, Bromberg, JS. Foxp3+CD8+ regulatory T cells: bona fide Tregs with cytotoxic function. Trends Immunol 2025;46:324–37. https://doi.org/10.1016/j.it.2025.02.010.Search in Google Scholar PubMed
54. Indrio, F, Salatto, A. Gut microbiota–bone axis. Ann Nutr Metab 2025;81:47–56. https://doi.org/10.1159/000541999.Search in Google Scholar PubMed
55. Fisher, MS, Sennikov, SV. T-regulatory cells for the treatment of autoimmune diseases. Front Immunol 2025;16:1511671. https://doi.org/10.3389/fimmu.2025.1511671.Search in Google Scholar PubMed PubMed Central
56. Robertson, SA, Care, AS, Moldenhauer, LM. Regulatory T cells in embryo implantation and the immune response to pregnancy. J Clin Investig 2018;128:4224–35. https://doi.org/10.1172/JCI122182.Search in Google Scholar PubMed PubMed Central
57. Qian, C, Pan, C, Liu, J, Wu, L, Pan, J, Liu, C, et al.. Differential expression of immune checkpoints (OX40/OX40L and PD-1/PD-L1) in decidua of unexplained recurrent spontaneous abortion women. Hum Immunol 2024;85:110745. https://doi.org/10.1016/j.humimm.2023.110745.Search in Google Scholar PubMed
58. Tian, Y, Peng, X, Yang, X. Decreased PD-L1 contributes to preeclampsia by suppressing GM-CSF via the JAK2/STAT5 signal pathway. Sci Rep. 2025;15:3124. https://doi.org/10.1038/s41598-025-87349-1. Erratum in: Sci Rep. 2025;15:8851. https://doi.org/10.1038/s41598-025-93453-zSearch in Google Scholar PubMed PubMed Central
59. Zhang, YH, Sun, HX. Immune checkpoint molecules in pregnancy: focus on regulatory T cells. Eur J Immunol 2020;50:160–9. https://doi.org/10.1002/eji.201948382.Search in Google Scholar PubMed
60. Yin, L, Zhang, Y, Fu, G, Huang, H, Su, H, Zhang, Y, et al.. Knowledge mapping of exosomes in preeclampsia: a bibliometric analysis (2008–2023). Front Endocrinol 2025;16:1546554. https://doi.org/10.3389/fendo.2025.1546554.Search in Google Scholar PubMed PubMed Central
61. Li, G, Zhang, S, Zou, Y, Ai, H, Zheng, X, Qian, K, et al.. The therapeutic potential of exosomes in immunotherapy. Front Immunol 2024;15:1424081. https://doi.org/10.3389/fimmu.2024.1424081.Search in Google Scholar PubMed PubMed Central
62. Green, S, Politis, M, Rallis, KS, Saenz de Villaverde Cortabarria, A, Efthymiou, A, Mureanu, N, et al.. Regulatory T cells in pregnancy adverse outcomes: a systematic review and meta-analysis. Front Immunol 2021;12:737862. https://doi.org/10.3389/fimmu.2021.737862.Search in Google Scholar PubMed PubMed Central
63. Lu, Y, Wang, Y, Ruan, T, Wang, Y, Ju, L, Zhou, M, et al.. Immunometabolism of Tregs: mechanisms, adaptability, and therapeutic implications in diseases. Front Immunol 2025;16:1536020. https://doi.org/10.3389/fimmu.2025.1536020.Search in Google Scholar PubMed PubMed Central
64. Zhao, X, Zhang, J, Li, C, Kuang, W, Deng, J, Tan, X, et al.. Mitochondrial mechanisms in Treg cell regulation: implications for immunotherapy and disease treatment. Mitochondrion 2025;80:101975. https://doi.org/10.1016/j.mito.2024.101975.Search in Google Scholar PubMed
65. Zhao, Y, Liu, L, Sun, R, Cui, G, Guo, S, Han, S, et al.. Exosomes in cancer immunoediting and immunotherapy. Asian J Pharm Sci 2022;17:193–205. https://doi.org/10.1016/j.ajps.2021.12.001.Search in Google Scholar PubMed PubMed Central
66. Hassan, M, Elzallat, M, Mohammed, DM, Balata, M, El-Maadawy, WH. Exploiting regulatory T cells (Tregs): cutting-edge therapy for autoimmune diseases. Int Immunopharmacol 2025;155:114624. https://doi.org/10.1016/j.intimp.2025.114624.Search in Google Scholar PubMed
67. Zhang, M, Ma, J, Li, M. Original antigenic sin in CD4+ T cells. Immunology 2025;175:165–79. https://doi.org/10.1111/imm.13916.Search in Google Scholar PubMed
68. Arneth, B. Molecular mechanisms of immune regulation: a review. Cells 2025;14:283. https://doi.org/10.3390/cells14040283.Search in Google Scholar PubMed PubMed Central
69. Meggyes, M, Miko, E, Szigeti, B, Farkas, N, Szereday, L. The importance of the PD-1/PD-L1 pathway at the maternal-fetal interface. BMC Pregnancy Childbirth 2019;19:74. https://doi.org/10.1186/s12884-019-2218-6.Search in Google Scholar PubMed PubMed Central
70. Duan, B, Feng, Q, Li, L, Huang, J. CircDDX21 alleviates trophoblast dysfunction and Treg differentiation in recurrent spontaneous abortion via miR-520a-5p/FOXP3/PD-L1 axis. J Assist Reprod Genet 2024;41:3539–57. https://doi.org/10.1007/s10815-024-03281-9.Search in Google Scholar PubMed PubMed Central
71. Elahi, S, Elahi, Z, Bozorgmehr, N, Rosero, EP, Rahmati, A, Abouda, A. Galectin-3 regulates erythropoiesis and enhances the immunoregulatory properties of CD71+ erythroid cells across developmental stages. J Immunol 2025:vkaf020. https://doi.org/10.1093/jimmun/vkaf020.Search in Google Scholar PubMed PubMed Central
72. Gao, Y, Chi, Y, Chen, Y, Wang, W, Li, H, Zheng, W, et al.. Multi-omics analysis of human mesenchymal stem cells shows cell aging that alters immunomodulatory activity through the downregulation of PD-L1. Nat Commun 2023;14:4373. https://doi.org/10.1038/s41467-023-39958-5.Search in Google Scholar PubMed PubMed Central
73. Kiriyama, Y, Nochi, H. Regulation of PD-L1 expression by nuclear receptors. Int J Mol Sci 2023;24:9891. https://doi.org/10.3390/ijms24129891.Search in Google Scholar PubMed PubMed Central
74. Lee, KWA, Chan, LKW, Hung, LC, Phoebe, LKW, Park, Y, Yi, KH. Clinical applications of exosomes: a critical review. Int J Mol Sci 2024;25:7794. https://doi.org/10.3390/ijms25147794.Search in Google Scholar PubMed PubMed Central
75. Arya, SB, Collie, SP, Parent, CA. The ins-and-outs of exosome biogenesis, secretion, and internalization. Trends Cell Biol 2024;34:90–108. https://doi.org/10.1016/j.tcb.2023.06.006.Search in Google Scholar PubMed PubMed Central
76. Chen, YF, Luh, F, Ho, YS, Yen, Y. Exosomes: a review of biologic function, diagnostic and targeted therapy applications, and clinical trials. J Biomed Sci 2024;31:67. https://doi.org/10.1186/s12929-024-01055-0.Search in Google Scholar PubMed PubMed Central
77. Al-Madhagi, H. The landscape of exosomes biogenesis to clinical applications. Int J Nanomed 2024;19:3657–75. https://doi.org/10.2147/IJN.S463296.Search in Google Scholar PubMed PubMed Central
78. Ranjan, P, Verma, SK. Exosomes isolation, purification, and characterization. Methods Mol Biol 2024;2835:173–80. https://doi.org/10.1007/978-1-0716-3995-5_16.Search in Google Scholar PubMed
79. Singh, S, Paul, D, Nath, V, A, R. Exosomes: current knowledge and future perspectives. Tissue Barriers 2024;12:2232248. https://doi.org/10.1080/21688370.2023.2232248.Search in Google Scholar PubMed PubMed Central
80. Wang, JS, Schellenberg, SJ, Demeros, A, Lin, AY. Exosomes in review: a new Frontier in CAR-T cell therapies. Neoplasia 2025;62:101147. https://doi.org/10.1016/j.neo.2025.101147.Search in Google Scholar PubMed PubMed Central
81. Caramelo, A, Polónia, A, Vale, J, Curado, M, Campelos, S, Nascimento, V, et al.. Demonstrating the interference of tissue processing in the evaluation of tissue biomarkers: the case of PD-L1. Pathol Res Pract 2023;248:154605. https://doi.org/10.1016/j.prp.2023.154605.Search in Google Scholar PubMed
82. Duan, L, Lin, W, Zhang, Y, Jin, L, Xiao, J, Wang, H, et al.. Exosomes in autoimmune diseases: a review of mechanisms and diagnostic applications. Clin Rev Allergy Immunol 2025;68:5. https://doi.org/10.1007/s12016-024-09013-2.Search in Google Scholar PubMed
83. Krylova, SV, Feng, D. The machinery of exosomes: biogenesis, release, and uptake. Int J Mol Sci 2023;24:1337. https://doi.org/10.3390/ijms24021337.Search in Google Scholar PubMed PubMed Central
84. Tsai, CH, Chuang, YM, Li, X, Yu, YR, Tzeng, SF, Teoh, ST, et al.. Immunoediting instructs tumor metabolic reprogramming to support immune evasion. Cell Metab 2023;35:118–33.e7. https://doi.org/10.1016/j.cmet.2022.12.003.Search in Google Scholar PubMed PubMed Central
85. Liu, S, Sun, Q, Ren, X. Novel strategies for cancer immunotherapy: counter-immunoediting therapy. J Hematol Oncol 2023;16:38. https://doi.org/10.1186/s13045-023-01430-8.Search in Google Scholar PubMed PubMed Central
86. Roerden, M, Spranger, S. Cancer immune evasion, immunoediting and intratumour heterogeneity. Nat Rev Immunol 2025;25:353–69. https://doi.org/10.1038/s41577-024-01111-8.Search in Google Scholar PubMed
87. Lan, J, Cai, D, Gou, S, Bai, Y, Lei, H, Li, Y, et al.. The dynamic role of ferroptosis in cancer immunoediting: implications for immunotherapy. Pharmacol Res 2025;214:107674. https://doi.org/10.1016/j.phrs.2025.107674.Search in Google Scholar PubMed
88. Zingoni, A, Antonangeli, F, Sozzani, S, Santoni, A, Cippitelli, M, Soriani, A. The senescence journey in cancer immunoediting. Mol Cancer 2024;23:68. https://doi.org/10.1186/s12943-024-01973-5.Search in Google Scholar PubMed PubMed Central
89. Li, X, Zhou, J, Fang, M, Yu, B. Pregnancy immune tolerance at the maternal-fetal interface. Int Rev Immunol 2020;39:247–63. https://doi.org/10.1080/08830185.2020.1777292.Search in Google Scholar PubMed
90. Riley, JK. Trophoblast immune receptors in maternal-fetal tolerance. Immunol Investig 2008;37:395–426. https://doi.org/10.1080/08820130802206066.Search in Google Scholar PubMed
91. Mellor, AL, Munn, DH. Immunology at the maternal-fetal interface: lessons for T cell tolerance and suppression. Annu Rev Immunol 2000;18:367–91. https://doi.org/10.1146/annurev.immunol.18.1.367.Search in Google Scholar PubMed
92. Sun, Y, Wu, S, Zhou, Q, Li, X. Trophoblast-derived interleukin 9 mediates immune cell conversion and contributes to maternal-fetal tolerance. J Reprod Immunol 2021;148:103379. https://doi.org/10.1016/j.jri.2021.103379.Search in Google Scholar PubMed
93. Bai, K, Lee, CL, Liu, X, Li, J, Cao, D, Zhang, L, et al.. Human placental exosomes induce maternal systemic immune tolerance by reprogramming circulating monocytes. J Nanobiotechnol 2022;20:86. https://doi.org/10.1186/s12951-022-01283-2.Search in Google Scholar PubMed PubMed Central
94. Chen, Z, Zhang, Y, Kwak-Kim, J, Wang, W. Memory regulatory T cells in pregnancy. Front Immunol 2023;14:1209706. https://doi.org/10.3389/fimmu.2023.1209706.Search in Google Scholar PubMed PubMed Central
95. Zhang, YJ, Shen, L, Zhang, T, Muyayalo, KP, Luo, J, Mor, G, et al.. Immunologic memory in pregnancy: focusing on memory regulatory T cells. Int J Biol Sci 2022;18:2406–18. https://doi.org/10.7150/ijbs.70629.Search in Google Scholar PubMed PubMed Central
96. Barbaro, G, Inversetti, A, Cristodoro, M, Ticconi, C, Scambia, G, Di Simone, N. HLA-G and recurrent pregnancy loss. Int J Mol Sci 2023;24:2557. https://doi.org/10.3390/ijms24032557.Search in Google Scholar PubMed PubMed Central
97. Arnaiz-Villena, A, Juarez, I, Suarez-Trujillo, F, López-Nares, A, Vaquero, C, Palacio-Gruber, J, et al.. HLA-G: function, polymorphisms and pathology. Int J Immunogenet 2021;48:172–92. https://doi.org/10.1111/iji.12513.Search in Google Scholar PubMed
98. Amodio, G, Gregori, S. HLA-G genotype/expression/disease association studies: success, hurdles, and perspectives. Front Immunol 2020;11:1178. https://doi.org/10.3389/fimmu.2020.01178.Search in Google Scholar PubMed PubMed Central
99. Contini, P, Murdaca, G, Puppo, F, Negrini, S. HLA-G expressing immune cells in immune mediated diseases. Front Immunol 2020;11:1613. https://doi.org/10.3389/fimmu.2020.01613.Search in Google Scholar PubMed PubMed Central
100. Ferreira, LMR, Meissner, TB, Tilburgs, T, Strominger, JL. HLA-G: at the interface of maternal-fetal tolerance. Trends Immunol 2017;38:272–86. https://doi.org/10.1016/j.it.2017.01.009.Search in Google Scholar PubMed
101. Rouas-Freiss, N, Moreau, P, LeMaoult, J, Papp, B, Tronik-Le Roux, D, Carosella, ED. Role of the HLA-G immune checkpoint molecule in pregnancy. Hum Immunol 2021;82:353–61. https://doi.org/10.1016/j.humimm.2021.01.003.Search in Google Scholar PubMed
102. Wu, Q, You, L, Nepovimova, E, Heger, Z, Wu, W, Kuca, K, et al.. Hypoxia-inducible factors: master regulators of hypoxic tumor immune escape. J Hematol Oncol 2022;15:77. https://doi.org/10.1186/s13045-022-01292-6.Search in Google Scholar PubMed PubMed Central
103. Jiang, X, Wang, J, Deng, X, Xiong, F, Ge, J, Xiang, B, et al.. Role of the tumor microenvironment in PD-L1/PD-1-mediated tumor immune escape. Mol Cancer 2019;18:10. https://doi.org/10.1186/s12943-018-0928-4.Search in Google Scholar PubMed PubMed Central
104. Wang, H, Sun, P, Yuan, X, Xu, Z, Jiang, X, Xiao, M, et al.. Autophagy in tumor immune escape and immunotherapy. Mol Cancer 2025;24:85. https://doi.org/10.1186/s12943-025-02277-y.Search in Google Scholar PubMed PubMed Central
105. Pathania, AS, Prathipati, P, Challagundla, KB. New insights into exosome mediated tumor-immune escape: clinical perspectives and therapeutic strategies. Biochim Biophys Acta Rev Cancer 2021;1876:188624. https://doi.org/10.1016/j.bbcan.2021.188624.Search in Google Scholar PubMed PubMed Central
106. Liu, L, Wang, Q, Qiu, Z, Kang, Y, Liu, J, Ning, S, et al.. Noncoding RNAs: the shot callers in tumor immune escape. Signal Transduct Targeted Ther 2020;5:102. https://doi.org/10.1038/s41392-020-0194-y.Search in Google Scholar PubMed PubMed Central
107. Poggi, A, Musso, A, Dapino, I, Zocchi, MR. Mechanisms of tumor escape from immune system: role of mesenchymal stromal cells. Immunol Lett 2014;159:55–72. https://doi.org/10.1016/j.imlet.2014.03.001.Search in Google Scholar PubMed
108. Marin-Acevedo, JA, Kimbrough, EO, Lou, Y. Next generation of immune checkpoint inhibitors and beyond. J Hematol Oncol 2021;14:45. https://doi.org/10.1186/s13045-021-01056-8.Search in Google Scholar PubMed PubMed Central
109. Zhuang, B, Shang, J, Yao, Y. HLA-G: an important mediator of maternal-fetal immune-tolerance. Front Immunol 2021;12:744324. https://doi.org/10.3389/fimmu.2021.744324.Search in Google Scholar PubMed PubMed Central
110. Zhang, L, Long, X, Yin, Y, Wang, J, Zhu, H, Chen, J, et al.. Histone methyltransferase Nsd2 ensures maternal-fetal immune tolerance by promoting regulatory T-cell recruitment. Cell Mol Immunol 2022;19:634–43. https://doi.org/10.1038/s41423-022-00849-2.Search in Google Scholar PubMed PubMed Central
111. Gillis-Buck, E, Miller, H, Sirota, M, Sanders, SJ, Ntranos, V, Anderson, MS . Extrathymic Aire-expressing cells support maternal-fetal tolerance. Sci Immunol. 2021;6:eabf1968.10.1126/sciimmunol.abf1968Search in Google Scholar PubMed PubMed Central
112. Rackaityte, E, Halkias, J. Mechanisms of fetal T cell tolerance and immune regulation. Front Immunol. 2020;11:588.10.3389/fimmu.2020.00588Search in Google Scholar PubMed PubMed Central
© 2025 the author(s), published by De Gruyter, Berlin/Boston
This work is licensed under the Creative Commons Attribution 4.0 International License.
Articles in the same Issue
- Frontmatter
- Reviews
- Immunoediting in pregnancy: a new paradigm for understanding fetal tolerance and obstetric disease
- Virtual fetal holography for parental counseling and education: applications, limitations, and future directions
- Original Articles – Obstetrics
- RNA biomarkers in hypertensive disorders of pregnancy: systematic review
- Second pregnancy vaginal birth after cesarean- impact of maternal age on outcomes from a retrospective cohort study
- Analysis of changes in serum VEGF, β-hCG, and sFlt-1 levels in women with placenta accreta spectrum and the impact on prognosis
- The adjunctive role of the neutrophil-to-lymphocyte ratio in risk stratification for clinical chorioamnionitis in term pregnancies with meconium-stained amniotic fluid
- Clinical utility of chromosomal microarray and whole exome sequencing in evaluating genetic causes for pregnancy loss using products of conception specimens
- The association of antiphospholipid syndrome under medical treatment with adverse pregnancy outcomes
- Effects of virtual reality on fear of birth, length of labor, and fetal well-being: a randomized controlled trial
- Antenatal corticosteroid prophylaxis in women with increased sFlt-1/PlGF ratio in the clinical routine – A retrospective analysis
- Early unfavorable outcomes of second-trimester selective feticide for complicated monochorionic twins: single-operator experiences
- Original Articles – Fetus
- Maternal circulating sFlt-1/placental growth factor is a biomarker of fetal death associated with placental lesions of maternal vascular malperfusion
- Intra-partum and perinatal outcomes in fetuses exhibiting ZigZag pattern on cardiotocography trace: a systematic review and meta-analysis
- Assessment of fetal adrenal gland and thymothoracic ratio in preterm premature membrane rupture: a prospective case-control study
- Fetal brain in fetal growth restriction: alterations in cortical morphometry and volume
- Diagnostic yield of post-mortem magnetic resonance imaging for cardiac anomalies in fetal and perinatal deaths: a systematic review and meta-analysis
- Original Articles – Neonates
- Risk factors and analysis of retinopathy of prematurity in monochorionic diamniotic twins
- Association between FAR, PAR, APRI and adverse neonatal outcomes in pregnancies complicated by intrahepatic cholestasis
- Incidence and predictors of high vitamin D in premature infants with very low birth weight
- Urinary immune biomarkers for late-onset sepsis in preterm very low birth weight neonates – a diagnostic accuracy study
- Letters to the Editor
- Histological chorioamnionitis and maternal inflammatory biomarkers: implications beyond clinical diagnosis
- Response to Letter to the Editor
- Erratum
- Erratum to: Gestational diabetes mellitus: the role of IGF-1 and leptin in cord blood
Articles in the same Issue
- Frontmatter
- Reviews
- Immunoediting in pregnancy: a new paradigm for understanding fetal tolerance and obstetric disease
- Virtual fetal holography for parental counseling and education: applications, limitations, and future directions
- Original Articles – Obstetrics
- RNA biomarkers in hypertensive disorders of pregnancy: systematic review
- Second pregnancy vaginal birth after cesarean- impact of maternal age on outcomes from a retrospective cohort study
- Analysis of changes in serum VEGF, β-hCG, and sFlt-1 levels in women with placenta accreta spectrum and the impact on prognosis
- The adjunctive role of the neutrophil-to-lymphocyte ratio in risk stratification for clinical chorioamnionitis in term pregnancies with meconium-stained amniotic fluid
- Clinical utility of chromosomal microarray and whole exome sequencing in evaluating genetic causes for pregnancy loss using products of conception specimens
- The association of antiphospholipid syndrome under medical treatment with adverse pregnancy outcomes
- Effects of virtual reality on fear of birth, length of labor, and fetal well-being: a randomized controlled trial
- Antenatal corticosteroid prophylaxis in women with increased sFlt-1/PlGF ratio in the clinical routine – A retrospective analysis
- Early unfavorable outcomes of second-trimester selective feticide for complicated monochorionic twins: single-operator experiences
- Original Articles – Fetus
- Maternal circulating sFlt-1/placental growth factor is a biomarker of fetal death associated with placental lesions of maternal vascular malperfusion
- Intra-partum and perinatal outcomes in fetuses exhibiting ZigZag pattern on cardiotocography trace: a systematic review and meta-analysis
- Assessment of fetal adrenal gland and thymothoracic ratio in preterm premature membrane rupture: a prospective case-control study
- Fetal brain in fetal growth restriction: alterations in cortical morphometry and volume
- Diagnostic yield of post-mortem magnetic resonance imaging for cardiac anomalies in fetal and perinatal deaths: a systematic review and meta-analysis
- Original Articles – Neonates
- Risk factors and analysis of retinopathy of prematurity in monochorionic diamniotic twins
- Association between FAR, PAR, APRI and adverse neonatal outcomes in pregnancies complicated by intrahepatic cholestasis
- Incidence and predictors of high vitamin D in premature infants with very low birth weight
- Urinary immune biomarkers for late-onset sepsis in preterm very low birth weight neonates – a diagnostic accuracy study
- Letters to the Editor
- Histological chorioamnionitis and maternal inflammatory biomarkers: implications beyond clinical diagnosis
- Response to Letter to the Editor
- Erratum
- Erratum to: Gestational diabetes mellitus: the role of IGF-1 and leptin in cord blood

