Abstract
Introduction
The tropical plant amylases involved in the fruit ripening stage is outstanding for their high activities in converting starch to sugars within a short period at high temperatures over 40°C.
Methods
The α amylase iso-enzymes from Ok-Rong mango (Mangifera indica Linn. cv. Ok-Rong) were purified in 2 steps, using 70% ammonium sulfate precipitation and affinity chromatography on a β-cyclodextrin sepharose 6B column, and characterized for biochemical properties.
Results
The enzyme was purified 105-fold with a final specific activity of 59.27 U mg−1. SDS-PAGE revealed two bands of 60 and 64 kDa. pI were supposed to be 4.6 and 5.0. Those were resolved into isoforms I and II by a zymographic method. They were matched with α amylase Amy1 from Vigna mungo and α amylase-like isoform I from Theobroma cacao after LC-MS/MS analysis. Isoforms I and II exhibited maximum activity at pH 4, retained more than 50% of their activity after 1 h of incubation at pH 5–9. Two isoforms showed high activity over a wide range of temperatures at 30°–90°C, with the highest activity at 70°C. They retained more than 50% of their activity at 30°C–40°C after 1 h of incubation. The enzymes were confirmed to be metalloenzymes by the effect of EDTA. In addition, limit-dextrin, amylopectin and soluble starch were suggested to be good substrates.
Conclusion
Two α amylase iso-enzymes were classified as members of the low-pI group of amylases with identical structure, properties and functions. They are mesophilic with high possibilities for application for many purposes.
Özet
Giriş
Meyve olgunlaştırma aşamasında yer alan tropikal bitki amilazları, 40OC’nin üzerindeki yüksek sıcaklıklarda, nişastayı şekerlere kısa sürede dönüştürme yönündeki yüksek aktiviteleri açısından göze çarpmaktadır.
Yöntemler
Ok-Rong mangosundan (Mangifera indica Linn. Cv. Ok-Rong) elde edilen alfa amilaz izo-enzimleri,% 70 amonyum sülfat çökeltme ve bir β-siklodekstrin sefaroz 6B kolonu üzerinde afinite kromatografisi kullanılarak 2 adımda saflaştırıldı ve biyokimyasal özellikler için karakterize edildi.
Bulgular
En son 59.27 U mg−1’lik bir nihai spesifik aktivite ile enzim 105 kat saflaştırıldı. SDS-PAGE, 60 ve 64 kDa’lık iki bant ortaya koymuştur. PI’nin 4.6 ve 5.0 olması gerekiyordu. Bunlar, zomografik bir yöntemle izoformlar I ve II’ye dönüştürüldü. LC-MS/MS analizinden sonra Theobroma kakao’sından Vigna mungo’dan α amilaz Amy1 ve α amilaz benzeri izoform I ile eşleştirildi. Isoformlar I ve II, pH 5’de enkübasyon 1 saat sonra aktivitelerinin% 50’sinden fazlasını koruyarak pH 4’te maksimum aktivite sergiledi. İki izoform, 30–90°C’de geniş bir sıcaklık aralığında yüksek etkinlik gösterdi ve en yüksek aktivite 70°C’de gerçekleşti. 1 saat inkübasyondan sonra aktivitelerinin% 50’sinden fazlasını 30–40°C’de muhafaza ettiler. Enzimlerin, EDTA etkisi ile metaloenzimler olduğu doğrulandı. Buna ek olarak, limit-dekstrin, amilopektin ve çözünür nişastanın iyi substrat olduğu öne sürülmüştür.
Sonuç
İki alfa amilaz izo-enzimi, düşük plaseboylu amilaz grubuna benzer yapı, özellikler ve fonksiyonlarla sınıflandırıldı. Birçok amaçla uygulama imkânı yüksek mezofiliklerdir.
Introduction
α Amylase (α-1,4-gluco-oligosaccharide, EC3.2.1.1) is a hydrolyzing enzyme that randomly cleaves an internal α-1,4-glycosidic linkage in starch and glycogen to produce malto-oligosaccharide and glucose [1]. This enzyme family is widely applied in many purposes including health promotions and industries, such as paper, sugar, textile, ethanol, etc., and it currently accounts for approximately 30% of the world’s industrial enzyme production [2]. Commonly available ones on the market are those from microorganisms, but there are limitations due to their specificities, activities and stabilities. Thus, novel amylases, especially from nature, is important. Those with high activity and stability have been sought out for many years. Recently, the enzymes involved in the fruit ripening stage is outstanding. The predominant carbohydrate in the raw stage of fruit is starch, which is replaced to a large extent by sugars, and α amylases are responsible for this process. Plant and fruit amylases are attractive because of their high activities in converting starch to sugars within a short period [3]. This always occurs at high temperatures over 40°C.
Mango (Mangifera indica Linn.) is a famous tropical fruit, commonly consumed as a dessert. Mango fruit mainly contains water and carbohydrates, with little protein and fat content. In the unripe stage, when carbohydrates is abundant, the carbohydrates are then replaced with sugars such as sucrose, glucose and fructose [4]. For starch content, amylose and amylopectin are the major ones in the fruit. Amylose is a glucose polymer linked by α (1–4) glycosidic bonds in linear structure. Amylopectin is a glucose polymer, as well, but linked by 94%–96% α (1–4) bonds in the linear portion and 4%6% α (1–6) bonds in the branch structure [5]. Ok-Rong mango (M. indica Linn. cv. Ok-Rong) is a native Thai mango commonly grown in all areas of Thailand (local name; Mamuang Ok-Rong). It is a large green tree that grows up to 20 m tall. This mango is edible, both in the green and ripe stage. The ripening one is defined by a soft texture, a yellow peel and unique sweet taste. For Ok-Rong mango, the plastid α amylase gene from Ok-Rong mango has been clarified to be a 594-amino acid residue sequence [6]. Three dimensional structure and catalytic site were computationally predicted. In this study, we intended to further study the α amylase iso-enzymes. They were purified by affinity chromatography using β-cyclodextrin, and characterized for its biochemical properties.
Materials and methods
Plant materials
Ok-Rong mangos (Mangifera indica Linn. cv. Ok-Rong) in the ripening stage were collected from Sri Sa Ket province (North eastern part of Thailand). The pulps were stored at −80°C before use.
Purification of Ok-Rong mango α amylase
Sample preparation and all purification steps were carried out at 4°C. Mangos were peeled, sliced into small piece. Mangos pulps was homogenized using homogenizer. Large particles were removed by many layers of gauze. After being filtered, samples were centrifuged at 10,000 g for 15 min. The supernatant was ‘crude extract’ for next step purification and kept at −70°C until used. Protein contents were estimated by the Bradford method [7] using bovine serum albumin as the standard.
Ammonium sulfate fractionation
Protein was precipitated at 70% saturation of ammonium sulfate, centrifugated, dissolved and dialyzed against 50 mM sodium acetate buffer, pH 6.0, containing 5 mM CaCl2 to remove the ammonium sulfate salt.
Epoxy-activated Sepharose 6B affinity chromatography
The samples were applied to an affinity column containing epoxy-activated Sepharose 6B beads conjugated with β-cyclodextrin according to the method of Vretblad [8]. The sample (20 mL) was loaded onto the column (1.6 cm×20 cm) pre-equilibrated with 50 mM sodium acetate buffer, pH 6.0, containing 5 mM CaCl2. The column was washed with 20 mM sodium acetate buffer, pH 6.0, containing 25 mM CaCl2 and 200 mM NaCl. The proteins were eluted with 10 mg/mL β-cyclodextrin in washing buffer. The active fractions were pooled, dialyzed against 50 mM sodium acetate buffer, pH 6.0, containing 5 mM CaCl2 and concentrated by SpeedVac concentrator.
Electrophoresis and pI determination
SDS-PAGE
The proteins samples were separated on 13% acrylamide gel in a discontinuous buffer system as described by Laemmli [9] and visualized with Coomassie blue and silver staining.
2D-PAGE
Samples were separated on a non-linear pH 3–10 strip for isoelectric focusing (IEF), and then subjected to a second dimension (SDS-PAGE). The protein spots were analyzed using ImageMaster 2D Platinum Software (GE Healthcare, Germany) for their MW and pI.
Mass spectrometry
Protein bands were identified using LC-MS/MS technique. The single bands were individually cut and treated by in-gel digestion technique [10] before being subjected to a nano-liquid chromatography system (EASY-nLC II, Bruker), coupled to a QTOF mass spectrometer (MicrOTOFQ-II, Bruker) and equipped with an ESI nano-sprayer under service from the Khon Kaen University Research Instrument Center, Thailand. The peptide mass fingerprint was analyzed using information from the NCBI database and the MASCOT MS/MS Ion Search Program (www.matrixscience.com) with these searching parameters: (1) enzyme: trypsin; (2) carbamidomethyl (C) as the fixed modification and oxidation (M) as the variable modification; (3) a peptide charge state of +1, +2, and +3; and (4) instrument type: ESI-QUAD-TOF.
α Amylase activity assay
For assay in solution, method was modified from Bernfeld [11]. One hundred microliters sample was added to 250 μL of 2% soluble starch in 0.1 M sodium acetate buffer, pH 6, containing 50 mM CaCl2, incubated at 37°C for 10 min. The reaction was terminated with 250 μL of 3,5-dinitrosalicylic acid reagent in boiling water for 5 min. After cooling, the mixture was diluted with DDW, and determined for its absorbance at 540 nm. One unit of enzyme activity was defined as the amount of enzyme that produced 1 μ mol of reducing sugar in 1 min under the defined conditions.
For the zymographic method, the enzyme activity was detected on 13% SDS-PAGE containing soluble starch as substrate. Samples were loaded on the gel, run at 150 V for 1 h, refolded in 1% Triton-X 100 for 1 h. Gel was immersed in 1% starch for 1 h and washed with DDW for 2 min. The gel was stained with iodine solution for 5 min. The excess iodine was washed off with cold DDW. Gel was soaked in 1% acetic acid and observed for activity bands. Intensity of the clear zone was analyzed using ImageJ software (http://imagej.nih.gov/ij/links.html).
Characterization
Effect of pH and temperature on α amylase activity
pH effects were probed over the range of 2–11 using 0.1 M of the appropriate buffers containing 50 mM CaCl2. The buffers were glycine-hydrochloric buffer (for pH 2.0), citrate-phosphate buffer (pH 3.0–5.0), phosphate buffer (pH 6.0–8.0) and carbonate-bicarbonate buffer (pH 9.0–11.0). pH stability was assessed by pre-incubating the enzyme at a different pH for 1 h at 25°C and subjected to a zymographic method. The effects of temperature were examined in the range from 30°C to 90°C. The thermal stability was investigated by pre-incubating enzyme solution at a range from 30°C to 90°C, for 1 h. Experiments were performed in triplicate.
Effect of metal ions and chemical reagents on α amylase activity
The effects of various metal ions, inhibitors, and detergents were examined by pre-incubating the enzyme with 10 mM of each of the following metals: NaCl, CaCl2, KCl, ZnSO4, and HgCl2. Other reagent tests included 10 mM EDTA, 10 mM β-mercaptoethanol, and 1% Triton X-100. Samples were incubated at 25°C for 1 h. The remaining activity was determined by a zymographic method. Experiments were performed in triplicate.
Substrate specificity
The substrate specificity was investigated by incubating the enzyme solution with 1% (w/v) soluble starch, amylopectin, limit-dextrin, β-cyclodextrin and glucose using the zymographic method. Experiments were performed in triplicate.
Results
Purification of α amylases
From the β-cyclodextrin affinity chromatography profile, a single peak of protein with α amylase activity was obtained (Figure 1). The peak was in fractions 21–24. The α amylase from Ok-Rong mango was purified 105-fold with a 4.67% recovery yield and 59.27 U mg−1 of specific activity (Table 1). After being checked by SDS-PAGE and the zymographic method, the α amylase solution exhibited two protein bands with amylase activity toward starch (Figure 2).

Affinity chromatography elution profile of Ok-Rong mango α amylases from the β-cyclodextrin Sepharose 6B column. The elution of β-cyclodextrin is indicated by an arrow labeled β-CD. The α amylase activity fractions are indicated in a circle.
Purification of the α amylases from Ok-Rong mango.
| Procedure | Protein (mg) | Activity (U) | Specific activity (U mg protein−1) | Yield (%) | Purification (fold) |
|---|---|---|---|---|---|
| Crude extract | 1020.75 | 570.00 | 0.56 | 100 | 1 |
| 70%Amomonium sulfate precipitation | 76.35 | 123.33 | 1.62 | 21.63 | 2.89 |
| β-cyclodextrin Sepharose 6B | 0.45 | 26.67 | 59.27 | 4.67 | 105.83 |
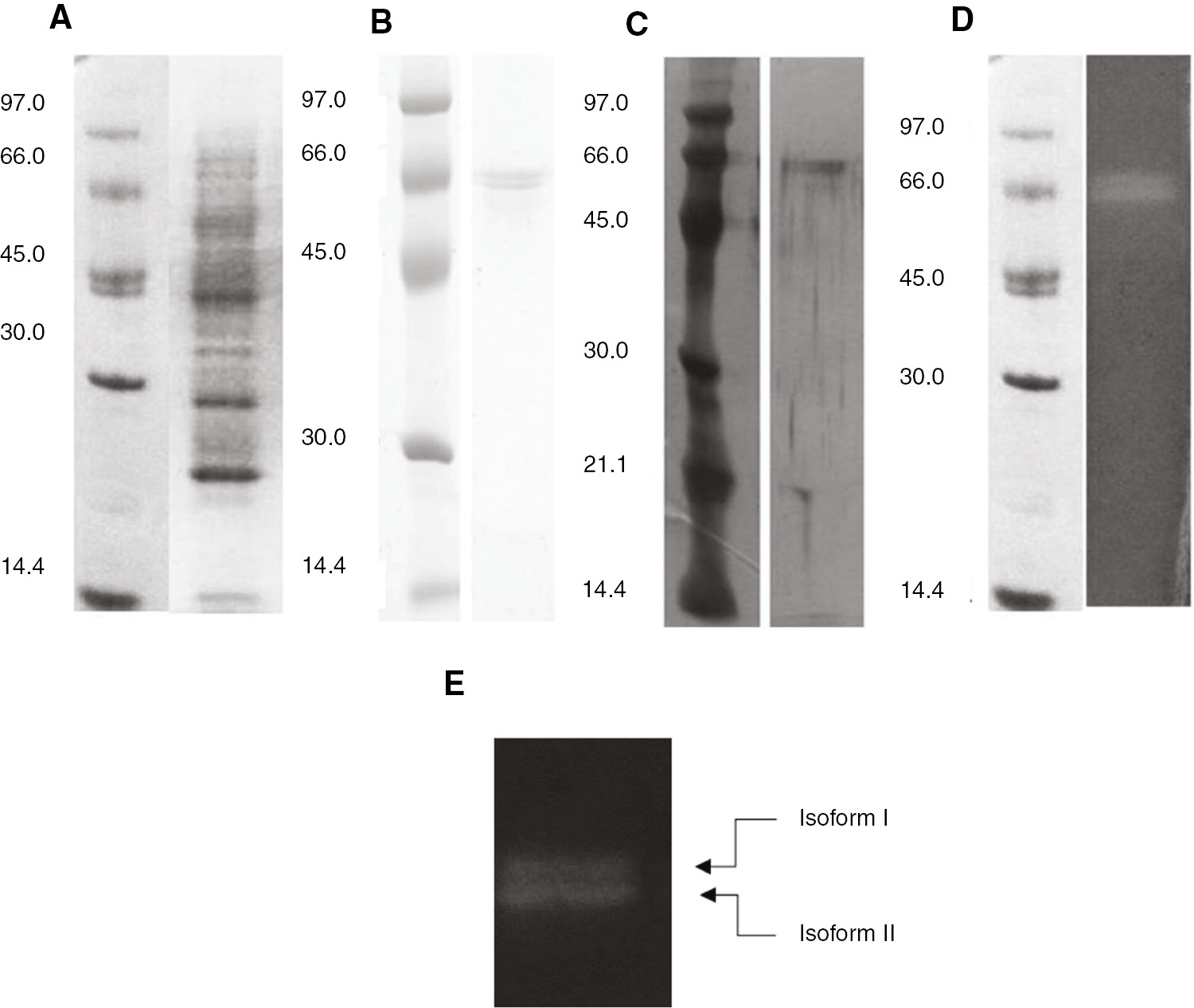
SDS PAGE and zymogram analysis of purified amylases from Ok-Rong mango. (A) Crude protein stained with Coomassie blue; (B) purified amylase proteins stained with Coomassie blue; (C) purified amylase protein stained with silver stain; (D) amylase activity detection by zymogram staining. These results represent the two isoforms of α amylase, (E).
Molecular weight and pI determination
Two isoforms of α amylase were successfully separated on a 2D-PAGE profile. After being analyzed with ImageMaster 2D Platinum Software, the upper band weighed 64 kDa and had a pI of 5.0. The lower band was found to be a 60 kDa protein with a pI of 4.6 (Figure 3).

Isoelectric point analysis of the two α amylase isoforms.
Protein identification
The two protein bands were separately cut for mass spectrometry analysis by LC-MS/MS. Peptide mass fingerprint analyzes of these isoforms were matched with Amy1, the α amylase from Vigna mungo (for the upper band), and the α amylase-like isoform I from Theobroma cacao (for the lower band). These are secreted amylases classified as members of Family 1 of α amylases (Table 2).
LC-MS/MS analysis of the two isoforms of α amylase from Ok-Rong mango.
| Sample | Protein annotation (organism) | Accession number | Mr theor./exp. | Peptide sequence | Mascot score |
|---|---|---|---|---|---|
| Isoform I (upper) | Alpha amylase, Amy1 (Vigna mungo) | P17859 | 1079.5/1079.5 | K.TEIGFDGWR.F | 53 |
| Isoform II (lower) | Alpha amylase-like isoform I (Theobroma cacao) | gi|1009134141 | 994.4/995.4 1079.5/1079.5 | K.AGGWYNSLK.N K.TEIGFDGWR.F | 40 53 |
Enzyme characterization
Effect of pH and temperature on α amylase activity
The effect of pH was examined at 37°C using pH values ranging from 2 to 11. The maximum activity of the two isoforms was observed at pH 4 (Figure 4A). To probe the pH stability, the enzyme solution was pre-incubated with different buffers for 1 h. The residual relative activity was higher than 50% at pH 5–9 after pre-incubation and had a decrease of 80% by pH 11 (Figure 4B). To study the effect of temperature, the α amylase activities of two isoforms were determined at different temperatures ranging from 30°C to 90°C. The α amylase activity remained higher than 80% at 40°C–70°C. The optimal temperature was 70°C (Figure 5A). For temperature stability, α amylases activities were studied by pre-incubating the enzyme at different temperatures for 1 h. The α amylase activity remained higher than 80% over the range of 30°C–40°C (Figure 5B).
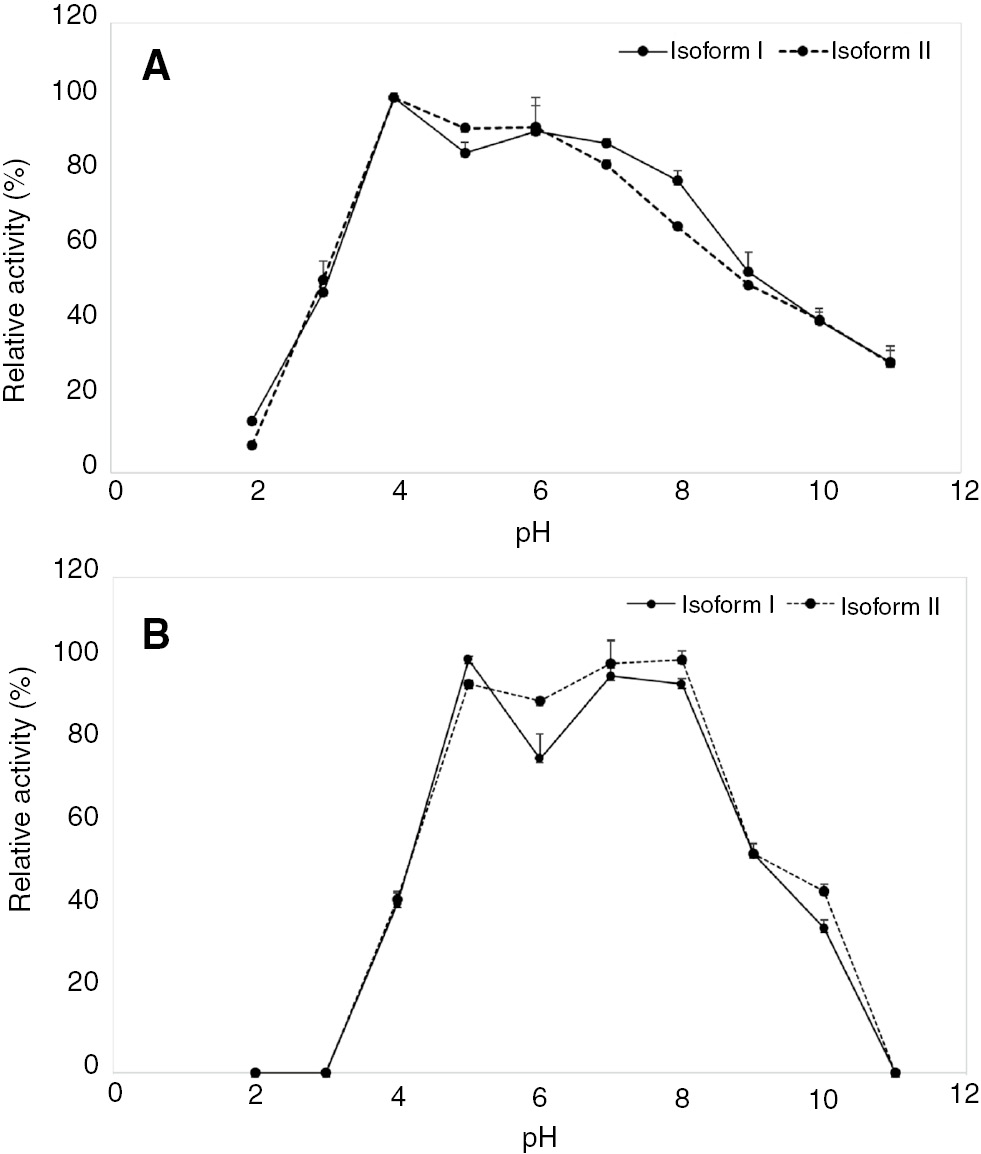
Optimal pH (A) and pH stability (B) of the two isoforms of Ok-Rong mango α amylase (solid line, isoform I and dotted line, isoform II). The optimal pH was detected using various buffers. The relative activity was observed as the percentage of the optimum activity. To probe stability, the enzyme was incubated at 25°C for 1 h, and its remaining activity was assayed. Each point represents the mean±SD of three independent experiments.
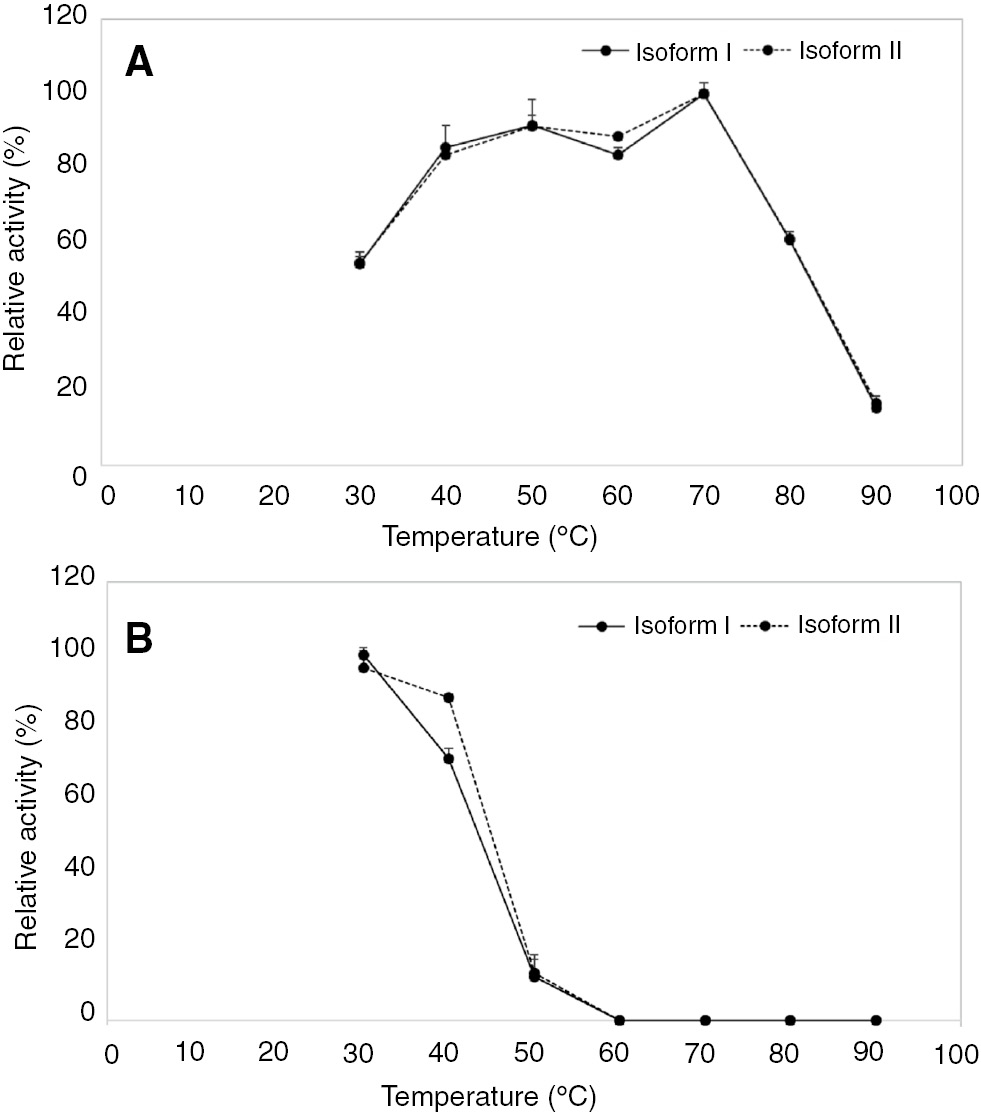
Optimal temperature (A) and temperature stability (B) of two isoforms of Ok-Rong mango α amylase (solid line, isoform I and dotted line, isoform II). The relative activity was observed as the percentage of optimum activity. To probe stability, the enzyme was incubated at different temperatures for 1 h, and the remaining activity was assayed. Each point represents the mean±SD of three independent experiments.
Effect of metal ions and reagents on α amylase activity
Zinc ion did not affect α amylase activity, whereas Hg2+ completely inhibited the enzyme activity (Table 3). β-Mercaptoethanol had some effect on the activity. The enzyme solution retained more than 80% of its activity. Both isoforms of α amylase were completely inhibited by 10 mM EDTA.
Effect of various metal ions on the purified amylases from Ok-Rong mango.
| Treatment | Relative activity (%) | |
|---|---|---|
| Isoform I | Isoform II | |
| EDTA | 0± 0.0 | 0± 0.0 |
| Triton-X 100 | 103± 2.5 | 109± 0.6 |
| β-mercaptoethanol | 84± 3.9 | 88± 0.9 |
| Hg2+ | 0± 0.0 | 0± 0.0 |
| Na+ | 94± 1.3 | 102± 6.0 |
| Co2+ | 84± 4.2 | 94± 0.5 |
| Zn2+ | 99± 2.6 | 97± 2.5 |
| Ca2+ | 106± 6.5 | 107± 2.4 |
| Non | 100 | 100 |
Values are expressed as the mean±standard error of three replicates.
Substrate specificity
Limit-dextrin was relatively found to be the best substrate (Table 4). Amylopectin and soluble starch were good substrates for α amylase from Ok-Rong mango.
Substrate specificity of the amylases from Ok-Rong mango.
| Substrate | Relative activity (%) |
|---|---|
| Limit- dextrin | 100 |
| Amylopectin | 84±1.1 |
| Starch | 82±2.0 |
| Glycogen | 20±1.7 |
| Maltose | 21±2.8 |
| β-cyclodextrin | 0±0.0 |
Values are expressed as the mean±standard error of three replicates.
Discussion
The finding for novel enzyme still be necessary in the field of enzyme technology for the best one. Plant is one of the most common sources for amylase enzyme studies due to its important role for basic metabolism including rapid degradation of starch during germination and fruit ripening. Mangos, M. indica, normally accumulated starch in pulp as the main storage. Recently, there are one report described that the saliva amylase was able to degraded mango starch. It showed obvious pits and cracks due to the action of amylase in the stomach under the observation by optical microscopy [12]. For amylases in mango fruits, not only pulp, mango peel also exhibited α amylase activity during ripening stage [13]. However, strains and varieties vary their basic biochemical properties. For Alphonso mango, the hydrolase including α amylase showed relatively high activity and gradually increased with maximum levels of activity around climacteric stage [14]. It was observed to be increased in the middle stage of ripening process in Ashwina hybrid mango, as well [15]. This related with an increase in amylase activity only on the 6th day after harvesting in Haden Mango [16]. The α-type amylase has been detected to bind and degrade starch granules in the early stage of ripening, but it degrade soluble starch for all the period of ripening stage in Keitt mangos [17]. Whereas the β-type amylase exhibited significantly higher activity than the α-type for the early stage just only for starch granule degradation. For application in post-harvesting technology, mango have to be carefully harvested and kept to prevent from any texture damages. Those including spongy tissues in Tommy Atkins mango resulted in three times amylase activity lower than that of healthy pulp [18] and the remaining of unhydrolyzed starch. Our previous study revealed that amylase in Ok-Rong mango pulp showed significantly high activity among Thai fruits during ripening stage (data not shown). Therefore, this study started from the purification of amylase using β-cyclodextrin affinity chromatography. This column was successful for purifying many plant α amylases in just one step [19, 20]. Bands from SDS-PAGE and zymographic analysis were 60 and 64 kDa with apparent α amylase activity. For plant amylases, masses in range 45–70 kDa with pI 4.5–5.5 are quite common (Table 5). They were classified into the low-pI group α amylases [26].
Comparative information on the properties of plant α amylases.
| Source | pH | Temp (°C) | Mw (kDa) | pI | Ref. |
|---|---|---|---|---|---|
| Malted barley | |||||
| α 1 | 5.5 | 55 | 52 | 5.1 | [21, 22] |
| α 2 | – | – | – | 6.2–6.4 | |
| Pearl millet | |||||
| α 1 | 4.4–4.8 | 55 | 31 | 4.8 | [21, 22] |
| α 2 | – | – | 46 | 5.2 | |
| α 3 | – | – | 53 | 6.2 | |
| Malted | |||||
| α 1 | 4.6 | – | 47–51 | – | [21, 22] |
| Rice | |||||
| α 1 | – | – | 48 | 4.6 | [21, 22] |
| Immature wheat | |||||
| α 1 | – | – | – | 4.65 | [21, 22] |
| α 2 | 3.6–5.75 | – | 52–54 | 4.84 | |
| α 3 | – | – | – | 5.11 | |
| Malted wheat | |||||
| α 1 | 5.5 | – | 41.5–42.5 | 6.16 | [21, 22] |
| α 2 | 5.7 | – | – | 6.2 | |
| α 3 | 5.5 | – | – | 6.05 | |
| Mung bean | |||||
| α 1 | 5.6 | 65 | 46 | – | [23] |
| Malted finger millet | |||||
| α 1 | 5.0 | 45 | 47 | – | [21, 24] |
| α 2 | 5.5 | 50 | 47 | – | |
| α 3 | 5.0 | 40 | 47 | – | |
| Azuki bean | |||||
| α 1 | 5.3 | 70 | 47 | 5.2 | [25] |
| α 2 | 5.2 | 70 | 44 | 5.2 | |
| Ok–Rong mango | |||||
| α 1 | 4.0 | 70 | 64 | 5.0 | This study |
| α 2 | 4.0 | 70 | 60 | 4.6 | |
These two isozymes in this study were identified as members of Family 1 of α amylases. Those are found in all plants especially cereal grain endosperm [27] with varieties structure and properties (isozymes) even in one individual organisms. There are five α amylase isozymes in apple [28], two isozymes in root beet [29], and two isozymes in azuki bean [25]. Pea (Pisum sativum) seedlings contained two to seven isozymes [30, 31]. For Keitt mango, at least three bands of amylolytic activity were observed. They became gradually higher in fruit extract at 3 and 5 days after harvesting [17].
Many α amylases in plants showed activity toward starch over a broad range of temperatures, 40°C–70°C (Table 5). The medium optimal temperature, at 70°C, also found for two α amylase isozymes from azuki bean including two isozymes of this report. These enzymes can be classified as mesophilic α amylases, active at 50°C–70°C. Optimal pH of the two Ok-Rong amylase isozymes were four. These corresponded with the increasing of hydrolases activity for carbohydrate degradation as ripening progresses where pH was increasing from 3.4 to 6.0 [32].
α Amylase is a metalloenzyme that contains at least one activating and stabilizing Ca2+ ion [33]. Calcium plays an important role in maintaining and stabilizing the structure of α amylase by interacting with negatively charge amino acid residues, such as aspartic and glutamic acid, especially those in their catalytic triad [34]. In previous reports, α amylases from Haden mango, P. sativum, Vigna mungo, V. radiata and V. angularis were shown to be inhibited by 5–10 mM EDTA [16, 19, 20, 23, 25, 35]. Related to this study, both isoforms of α amylase were completely inhibited by 10 mM EDTA. The effects of EDTA indicate that the metal ion is required for this enzyme. The addition of 10 mM Zn2+, a known inhibitor of α amylase from many sources [36], showed no effects on the enzyme. α Amylase from Chokanan mango exhibit 61%remaining activity in 5 mM Zn2+ solution [13]. α Amylase from Pachyrhizus erosus L. tuber has 42.5% activity after the addition of 10 mM Zn2+, whereas 100 mM Zn2+ loss 90% activity [37]. Hg2+ completely inhibited the activity. Ranwala and Miller reported the complete inhibition in tulip by 2 mM Hg2+ and Ag+, as well [38]. The addition of β-mercaptoethanol, in this study, slightly affected the activity of the α amylases. The study of the disulfide bridges in cold-active α amylase demonstrated that none of disulfide bonds are important in stabilizing the native structure of the enzyme [39]. However, charged amino acids play an important role in the conformation of amylases. Salt-bridges are often abundant in thermostable proteins predicting of their role in enzyme stability [40].
Limit-dextrin, amylopectin and starch were suggested to be good substrates for Ok-Rong mango α amylases. The enzymes showed low activity toward glycogen, a high branch-point polysaccharide. These results related to the endo-acting property of α amylase.
Conclusions
α Amylases from Ok-Rong mango (M. indica) were purified by β-cyclodextrin affinity chromatography for the first time with a final specific activity of 59.27 U mg−1. The two isozymes of α amylase from Ok-Rong mango were characterized. Both were major enzymes and classified as members of the low-pI group of amylases with identical structure, properties and functions. They are mesophilic. It is highly possible that these enzymes could be applied for many purposes, including industries where a variety of enzymatic reactions is needed.
Acknowledgement
This work was supported by the Higher Education Research Promotion and National Research University Project of Thailand, Office of the Higher Education Commission, through the Biofuel Cluster of Khon Kaen University.
References
1. Cantarel BL, Coutinho PM, Rancurel C, Bernard T, Lombard V, Henrissat B. The carbohydrate-Active Enzymes data base (CAZy): An export resource for Glycogenomics. Nucl Acids Res 2009; 37:233–8.10.1093/nar/gkn663Suche in Google Scholar
2. van der Maarel MJ, van der Veen B, Uitdehaag JC, Leemhuis H, Dijkhuizen L. Properties and applications of starch converting enzymes of the α-amylase family. J Biotechnol 2002;94:137–55.10.1016/S0168-1656(01)00407-2Suche in Google Scholar
3. Posoongnoen S, Ubonbal R, Thammasirirak S, Daduang J, Minami H, Yamamoto K, et al. α-Amylase from Mon Thong durian (Durio zibethinus Murr. cv. Mon Thong)- nucleotide sequence analysis, cloning and expression. Plant Biotechnol 2015;32:1–10.10.5511/plantbiotechnology.14.1122aSuche in Google Scholar
4. Ollé D, Lozano FY, Brillouet JM. Isolation and characterization of soluble polysaccharides and insoluble cell wall material of the pulp from four mango (Mangifera indica L.) cultivars. J Agric Food Chem 1996;44:2658–62.10.1021/jf9507506Suche in Google Scholar
5. Biliaderis CG. The Structure and interactions of starch with food constituents. Can J Physiol Pharmacol 1991;69:60–78.10.1139/y91-011Suche in Google Scholar
6. Ubonbal R, Posoongnoen S, Daduang J, Klaynongsruang, Daduang S. Amino acid sequence of amylase type alpha, MiAmy, from Ok-Rong mango (Mangifera indica Linn. cv. Ok-Rong). Am J Biochem Biotechnol 2015;11:119–26.10.3844/ajbbsp.2015.119.126Suche in Google Scholar
7. Kruger NJ. The Bradford method for protein quantitation. Methods Mol Biol 1994;32:9–15.10.1385/0-89603-268-X:9Suche in Google Scholar
8. Vretblad P. Immobilization of ligands for biospecific affinity chromatographic via their hydroxyl group, the cyclohexaamylose-β-amylase system. FEBS Lett 1974;46:86–9.10.1016/0014-5793(74)80431-XSuche in Google Scholar
9. Laemmli UK. Cleavage of structural proteins during the assembly of bend of bacteriophage T4. Nature 1970;227:680–5.10.1038/227680a0Suche in Google Scholar PubMed
10. Shevchenko A, Tomas H, Havlis J, Olsen JV, Mann M. In-gel digestion for mass spectrometric characterization of proteins and proteomes. Nat Protoc 2007;1:2856–60.10.1038/nprot.2006.468Suche in Google Scholar PubMed
11. Bernfeld P. Amylases α and β. Methods Enzymol 1955;1:149–58.10.1016/0076-6879(55)01021-5Suche in Google Scholar
12. Wu P, Dhital S, Williams BA, Chen XD, Gidley MJ. Rheological and microstructural properties of porcine gastric digesta and diets containing pectin or mango powder. Carbohydr Polym 2016;148:216–26.10.1016/j.carbpol.2016.04.037Suche in Google Scholar
13. Mehrnoush A, Yazid AM. Characterization of novel amylase enzyme from mango (Mangifera indica cv. Chokanan) peel. J Food Agric Environ 2013;11:47–50.Suche in Google Scholar
14. Yashoda HM, Prabha TN, Tharanathan RN. Mango ripening – role of carbohydrases in tissue softening. Food Chem 2007;102:691–8.10.1016/j.foodchem.2006.06.001Suche in Google Scholar
15. Hossain MA, Rana MM, Kimura Y, Roslan HA. Changes in biochemical characteristics and activities of ripening associated enzymes in mango fruit during the storage at different temperatures. BioMed Res Int 2014;2014:232969.10.1155/2014/232969Suche in Google Scholar
16. Fuchs Y, Pesis E, Zauberman G. Changes in amylase activity, starch and sugar contents in mango fruit pulp. J Hortic Sci 1980;13:155–60.10.1016/0304-4238(80)90080-1Suche in Google Scholar
17. Peroni FH, Koike C, Louro RP, Purgatto E, do Nascimento JR, Lajolo FM, et al. Mango starch degradation. II. The binding of alpha-amylase and beta-amylase to the starch granule. J Agric Food Chem 2008;56:7416–21.10.1021/jf800469wSuche in Google Scholar
18. Lima LD, Chitarra AB, Chitarra MIF. Changes in amylase activity, starch and sugar contents in mango fruit pulp of cv. Tommy Atkins with spongy tissue. Braz Arch Biol Technol 2001;44:59–62.10.1590/S1516-89132001000100008Suche in Google Scholar
19. Koshiba T, Minamikawa T. Purification by affinity chromatography of α-amylase a main amylase in cotyledons of germinating Vigna mungo seeds. Plant Cell Physiol 1981;22:979–87.Suche in Google Scholar
20. Beers EP, Duke SH. Characterization of α-amylase from shoots and cotyledons of pea (Pisum sativum L.) seedlings. Plant Physiol 1990;92:1154–63.10.1104/pp.92.4.1154Suche in Google Scholar
21. Muralickrishna G, Nirmalab M. Cereal amylases – an overview. Carbohydr Polym 2005;60:165–73.10.1016/j.carbpol.2004.12.002Suche in Google Scholar
22. Kruger JE, Lineback DR. Carbohydrate degrading enzymes in cereals. In: Kruger JE, Lineback DR, Stautter CE, editors. Enzymes and their role in the cereal technology. AACC International, Minnesota, 1987:117–39.Suche in Google Scholar
23. Tripathi P, Leggio LL, Mansfeld J, Hofmann RU, Kayastha AM. α-Amylase from mung beans (Vigna radiata)–correlation of biochemical properties and tertiary structure by homology modeling. Phytochemistry 2007;68:1623–31.10.1016/j.phytochem.2007.04.006Suche in Google Scholar
24. Nirmala M, Muralikrishna G. Three α -amylases from malted finger millet (Ragi, Eleusine coracana, Indaf-15–purification and partial characterization. Phytochemistry 2003;62:21–30.10.1016/S0031-9422(02)00443-0Suche in Google Scholar
25. Mar SS, Mori H, Lee J, Fukuda K, Saburi W, Fukuhara A, et al. Purification, characterization and sequence analysis of two α-amylase isoforms from azuki bean (Vigna angularis) showing different affinity β-cyclodextrin sepharose. Biosci Biotechnol Biochem 2003;67:1080–93.10.1271/bbb.67.1080Suche in Google Scholar
26. Jacobsen JV, Higgins TJ. Characterization of the α-amylases synthesized by aleurone layers of Himalaya barley in response to gibberellic acid. Plant Physiol 1982;70:1647–53.10.1104/pp.70.6.1647Suche in Google Scholar
27. Stanley D, Kevin JF, Elspeth AM. Plant α-amylases: functions and roles in carbohydrate metabolism. Biologia (Bratisl) 2005;60:65–71.Suche in Google Scholar
28. Stanley D, Fitzgerald AM, Farnden KJF, MacRae EA. Characterisation of putative α-amylases from apple (Malus domestica) and Arabidopsis thaliana. Biologia (Bratisl) 2002;57:137–48.Suche in Google Scholar
29. Masuda H, Takahashi T, Sugawara S. Purification and properties of starch hydrolyzing enzymes in mature roots of sugar beets. Plant Physiol 1987;84:361–5.10.1104/pp.84.2.361Suche in Google Scholar
30. Yomo H, Varner JE. Control of the formation of amylases and proteases in the cotyledons of germinating peas. Plant Physiol 1973;51:708–13.10.1104/pp.51.4.708Suche in Google Scholar
31. Monerri C, Guardiola JL. Estudio electrofordtico de las amilasas del guisante. Revista Agroquímica y de Tecnologia de Alimentos 1986;26:424–34.Suche in Google Scholar
32. Yoshoda HM, Prabha TN, Tharanathan RN. Mango ripening: chemical and structural characterization of pectic and hemicellulosic polysaccharides. Carbohydr Res 2005;340:1335–42.10.1016/j.carres.2005.03.004Suche in Google Scholar
33. Vallee Bl, Stein EA, Summeerwell EA, Fischer EH. Metal content of α amylase of various organisms. J Biol Chem 1959;234: 2901–5.10.1016/S0021-9258(18)69691-7Suche in Google Scholar
34. Rogers JC. Conserved amino acid sequence domain in alpha amylase from plant, mammal and bacteria. Biochem Biophys Res Commun 1985;128:470–6.10.1016/0006-291X(85)91702-4Suche in Google Scholar
35. Lily MK, Bahuguna A, Bhatt KK, Dangwall K. Production, partial purification and characterization of α-Amylase from high molecular weight polycyclic aromatic hydrocarbons (HMW-PAHs) degrading Bacillus subtilis BMT4i (MTCC 9447). Turk J Biochem 2012;37:463–70.10.5505/tjb.2012.07279Suche in Google Scholar
36. Savchenko A, Vielle C, Kang S, Zeikus G. Pyrococcus furiosus α-amylase is stabilized by calcium and zinc. Biochemistry 2002;41:6193–201.10.1021/bi012106sSuche in Google Scholar PubMed
37. Noman AS, Hoque MA, Sen PK, Karim MR. Purification and some properties of α-amylase from post-harvest Pachyrhizus erosus L. tuber. Food Chem 2006;99:444–9.10.1016/j.foodchem.2005.07.056Suche in Google Scholar
38. Rawanla AP, Miller WB. Purification and characterization of an endoamylase from tulip (Tulipa gesneriana) bulbs. Physiol Plant 2000;109:388–95.10.1034/j.1399-3054.2000.100404.xSuche in Google Scholar
39. Siddiqui KS, Poljak A, Guilhaus M, Feller G, D’Amico S, Gerday C, et al. Role of disulfide bridges in the activity and stability of a cold-active α –amylase. J Bacteriol 2005;187:6206–12.10.1128/JB.187.17.6206-6212.2005Suche in Google Scholar
40. Jensen MT, Gottschalk TE, Svensson B. Differences in conformational stability of barley alpha-amylase isozymes 1 and 2. Role of charged groups and isozyme 2 specific salt-bridges. J Cereal Sci 2003;38:289–300.10.1016/S0733-5210(03)00032-8Suche in Google Scholar
©2017 Walter de Gruyter GmbH, Berlin/Boston
Artikel in diesem Heft
- Frontmatter
- Opinion Paper
- How can I protect my telomeres and slow aging?
- Research Articles
- Biochemical changes in the leaves of mungbean (Vigna radiata) plants infected by phytoplasma
- Optimization of process parameters and kinetic modeling for the enantioselective kinetic resolution of (R,S)-2-pentanol
- Determination of petroleum biodegradation by bacteria isolated from drilling fluid, waste mud pit and crude oil
- Variations in the fatty acid compositions of the liver and gonad tissue of spiny eel (Mastacembelus mastacembelus) from Atatürk Dam Lake
- Purification and characterization of two isoforms of native α amylase from Ok-Rong mango (Mangifera indica Linn. cv. Ok-Rong)
- Purification, immobilization and characterization of thermostable α-amylase from a thermophilic bacterium Geobacillus sp. TF14
- Development and characterization of a formulation based covalent conjugation with polyacrylic acid and recombinant major surface antigen (SAG1) of Toxoplasma gondii
- Reasearch Article
- Effect of elevation and phenological stages on essential oil composition of Stachys
- Letter to the Editor
- Some errors in the measurement of neutrophil-to-lymphocyte ratio
- Indices
- Reviewers 2017
- Yazar Dizini/Author Index
Artikel in diesem Heft
- Frontmatter
- Opinion Paper
- How can I protect my telomeres and slow aging?
- Research Articles
- Biochemical changes in the leaves of mungbean (Vigna radiata) plants infected by phytoplasma
- Optimization of process parameters and kinetic modeling for the enantioselective kinetic resolution of (R,S)-2-pentanol
- Determination of petroleum biodegradation by bacteria isolated from drilling fluid, waste mud pit and crude oil
- Variations in the fatty acid compositions of the liver and gonad tissue of spiny eel (Mastacembelus mastacembelus) from Atatürk Dam Lake
- Purification and characterization of two isoforms of native α amylase from Ok-Rong mango (Mangifera indica Linn. cv. Ok-Rong)
- Purification, immobilization and characterization of thermostable α-amylase from a thermophilic bacterium Geobacillus sp. TF14
- Development and characterization of a formulation based covalent conjugation with polyacrylic acid and recombinant major surface antigen (SAG1) of Toxoplasma gondii
- Reasearch Article
- Effect of elevation and phenological stages on essential oil composition of Stachys
- Letter to the Editor
- Some errors in the measurement of neutrophil-to-lymphocyte ratio
- Indices
- Reviewers 2017
- Yazar Dizini/Author Index

