Abstract
Matrix metalloproteinases (MMPs), a class of zinc-contained endopeptidases, are closely involved in tumor growth, infiltration, metastasis, and angiogenesis. By virtue of the specifically enzymatic hydrolysis, MMPs have been widely used to turn on imaging and/or therapy function of elaborately designed enzyme-responsive nanoprobes, which is expected to realize precise diagnosis and treatment of cancer. This review systematically summarizes the classifications of MMPs, their substrates and recognized sequences, and overexpressed tumor types. The advances of MMPs-responsive nanoprobes for cancer diagnosis and therapy are focused, including trigger mechanism, design principle, and various imaging or therapy modes. Finally, this review analyzes the challenges of MMPs-responsive nanoprobes in clinical application, and provides constructive opinions for future study.
1 Introduction
According to the report from International Agency for Research on Cancer under World Health Organization, there were 9.96 million cancer deaths worldwide in 2020 [1]. Cancer has become one of the major threats to human life and health. To study precise diagnostic methods and effective treatments are of great significance for saving cancer patients or improving their quality of life. Compared with normal tissue, the tumor tissue has a complex microenvironment including weak acidic pH, hypoxia, high glutathione content, increased oxidative stress, and overexpression of enzymes, such as matrix metalloproteinase (MMP), cathepsin B, phospholipase, hyaluronidase [2–7]. Among these enzymes, MMPs are a class of zinc-dependent endopeptidases that can degrade the extracellular matrix (ECM) and basement membrane, and hence induce tumor spread [8,9]. As a representative endogenous stimulant, the abnormally expressed MMPs are closely related to tumor growth, infiltration, metastasis, as well as angiogenesis, and therefore are extensively attracted as anti-cancer targets in enzyme-responsive nanoprobe-based imaging and therapeutic strategies [10].
Compared with traditional small-molecule drugs, MMPs-responsive nanoprobes have the following advantages: (1) highly specific binding affinity with target molecules to improve the imaging or therapeutic effect; (2) less influence of the peptide substrate on physicochemical properties of the nanoprobes; (3) being relatively stable in body and keeping appropriate clearance period in blood circulation without causing significant immune response; and (4) feasible coupling imaging molecules or therapeutic drugs to achieve visualization of treatment [11]. The design principle of MMPs-responsive nanoprobes mainly depends on the high selectivity and catalytic activity of the enzymes on their substrates [12]. The enzyme-specific substrate is designed as nanoprobes with quenched imaging or therapeutic function. When the nanoprobes arrive in tumor site, the substrate is hydrolyzed by high concentration of MMPs, which results in a changed structure, and therefore activates diagnosis and therapy [13–15]. Specifically, in the presence of MMPs, hydrolysis of the enzyme-specific substrate causes changes in structure, conformation, hydrophobicity, hydrophilicity, and charge of the nanoprobes, which gives rise to effect on size, morphology, stability, and other properties of the nanoprobes. Finally, the nanoprobes are decomposed, gathered, rearranged, or self-assembled exposing the previously masked functional groups [16].
In this review, we systematically summarize the classifications of MMPs, types of substrates, recognized sequences, and overexpressed tumor types. Then, the advances of MMPs-based cancer imaging and treatment are reviewed, including mechanism of turning off/on function, design principle, as well as single mode/multimode imaging and therapy. Finally, we discuss the challenges in clinical application of MMPs-responsive nanoprobes, and provide suggestions for future study.
2 MMPs
MMPs, a class of zinc and calcium ions-dependent endogenous proteolytic enzymes, are synthesized and secreted by fibroblasts, macrophages, neutrophils, and tumor cells [17–19]. Studies have found that there are at least 23 MMPs in human body, which can be divided into six categories according to their structures and substrate sensitivity: (1) collagenases, including MMP-1, MMP-8, MMP-13, and MMP-18; (2) gelatinases, including MMP-2 and MMP-9; (3) stromelysins, including MMP-3, MMP-10, and MMP-11; (4) matrilysins, including MMP-7 and MMP-26; (5) membrane-type metalloproteinases, including MMP-14, MMP-15, MMP-16, MMP-17, MMP-24, and MMP-25; (6) other MMPs, including MMP-12, MMP-19, MMP-20, MMP-23, and MMP-28 [20–22].
MMPs play a critical role in cancer progression. On the one hand, ECM is the natural barrier for cancer cells invasion and metastasis, while MMPs participate in degradation of ECM, which promotes tumor to break the tissue barrier [10]. On the other hand, MMPs are also considered as vascular modulators, which can control the neovascularization required in the growth, progression, and spread of cancer [23]. As shown in Table 1, we summarize several MMPs that have been investigated extensively at present, and enumerate their overexpressed cancer types, relative biological functions, and degraded substrates.
Contribution of MMPs to cancer progression and their substrate
| Enzymes | Overexpressed cancer types | Function | Substrates | Ref. |
|---|---|---|---|---|
| MMP-1 | Oral cancer, colorectal cancer, breast cancer, prostate cancer, and bladder cancer | ECM degradation | Type I, II, III, VII, and X collagens, gelatin | [24–30] |
| MMP-2 | Breast cancer, gastric cancer osteosarcoma, prostate cancer, pancreatic cancer, liver cancer, and colon cancer | Promotes tumor growth, invasion, and metastasis and angiogenesis | Type IV, V collagens, gelatin, and elastin | [31–36] |
| MMP-7 | Gastric cancer, pancreatic cancer, colorectal cancer, esophageal cancer, gallbladder cancer, bladder cancer, ovarian cancer, breast cancer, lung cancer, and melanoma | Inhibits cancer cells apoptosis, reduces cell adhesion, and angiogenesis | Type IV collagen, casein, laminin, fibronectin, gelatins, elastin, and proteoglycans | [36–38] |
| MMP-9 | Cervical cancer, ovarian cancer, breast cancer, pancreatic cancer, prostate cancer, lung cancer, melanoma, osteosarcoma, and neuroblastoma | Promotes tumor growth, invasion, and metastasis and angiogenesis | Type IV and V collagen, gelatins, elastin, fibrillin, and osteonectin | [31–36,39] |
| MMP-14 | Melanoma, neuroblastoma, small cell lung cancer, squamous cell carcinoma of the tongue, head, and neck cancer, pancreatic cancer, bladder cancer, breast cancer, colorectal cancer, and ovarian cancer | Proliferation, invasion, metastasis, and angiogenesis. | Type Ⅰ and Ⅱ collagen, fibronectin, laminin, hyaluronan, fibrin, and proteoglycan | [40–47] |
2.1 MMP-1
MMP-1 belongs to the interstitial collagenase, and its substrates mainly include collagen types I, II, III, and V [24]. As a collagenase related with cancer, MMP-1 can promote tumor progress by degrading ECM [25]. Currently, upregulated MMP-1 expression has been found in a variety of cancer tissue specimens, and its high expression has been associated with poor prognosis of oral cancer, colorectal cancer, breast cancer, prostate cancer, and bladder cancer [26–30].
2.2 MMP-2/9
MMP-2 and MMP-9, both of which belong to gelatinases, play key roles in the physiological and pathological process of tumors [31]. On the one hand, they can specifically degrade the main structural components of ECM and basement membrane, such as gelatin and type IV collagen, which help tumor cells to infiltrate from the missing basement membrane into the surrounding tissues, thus promoting the invasion and metastasis of tumor cells [32,33]. On the other hand, studies have shown that MMP-2 and MMP-9 can promote the expression of vascular endothelial factors, which in turn reduce the formation of pannus, increase the permeability of the vascular wall, promote angiogenesis, and hence improve tumor growth [23,34,39]. MMP-2 and MMP-9, the most important MMPs, are abnormally expressed in various tumors, such as pancreatic cancer, breast cancer, bladder cancer, and cervix cancer [35,36].
2.3 MMP-7
MMP-7, also known as matrilysins, is the member of the matrix metalloprotease family with the smallest molecular weight [37]. MMP-7 is specifically expressed in various human tumors, including gastric cancer, pancreatic cancer, colorectal cancer, esophageal cancer, gallbladder cancer, bladder cancer, ovarian cancer, breast cancer, lung cancer, and other malignant tumors [36,38]. MMP-7 promotes tumor invasion and metastasis by inhibiting apoptosis, decreasing cell adhesion, and inducing angiogenesis [38].
2.4 MMP-14
MMP-14, also known as MT1-MMP, is a membrane-type MMP [40]. It has the widest substrate hydrolysis range, and the degradable zymogram involves type I collagen, type II collagen, fibronectin, laminin, hyaluronan, fibrin, proteoglycan, etc., [41]. Clinical studies have found that over-expressed MMP-14 is associated with poor prognosis in many kinds of cancer patients, such as melanoma, neuroblastoma, small cell lung cancer, pancreatic cancer, bladder cancer, breast cancer, and so on [42,43]. MMP14 is involved in the degradation and remodeling of ECM, promotion of tumor angiogenesis and regulation of cell adhesion, and affects the cellular microenvironment [42]. It is a key enzyme for tumor cell infiltration and migration. Studies have demonstrated that MMP-14 plays a role in the activation of pro-MMP2 and pro-MMP13, the upregulation of VEGF gene expression, and the mediation of CD44 shedding [44–47].
3 Applications in cancer imaging
MMPs-responsive nanoprobes have been widely used in cancer diagnosis. Through ingenious design, researchers have developed various nanoprobes that can realize non-invasive detection, real-time imaging, and long-term tracking in the living body. Table 2 illustrates the construction of MMPs-responsive nanoprobes and their application in cancer diagnosis.
Illustration of MMPs-responsive nanoprobes and their cancer diagnostic applications
| MMP types | Nanoprobes | Substrate | Diagnosis | Cancer | Ref. |
|---|---|---|---|---|---|
| MMP-2 | Dab-PLGVRGY-FITC | PLGVRGY | FI | Gastric cancer | [48] |
| MMP-2/9 | I780BP-PEG12 | GPLGVRGKGG | FI | Pancreatic cancer | [49] |
| MMP-2 | SPN-MMP-RGD | GGPLGVRGK | FI | Gastric cancer | [50] |
| MMP-2/7/9 | Au-Se | DGPLGVRG, VPLSLTMG, GPLGLRGG | FI | Hepatoma | [51] |
| MMP-14 | QD-FRET | AHLR | FI | Breast cancer | [52] |
| MMP-2 | Au-QD NCs | VLGPG | FI | Lung cancer | [53] |
| MMP-2 | PEG-PepMMP2-MNP-Gd | KPLGLAGC | PA/MRI | Breast cancer | [54] |
| MMP-2 | FMNS@Au | PLGVR | FI/SERS | Breast cancer, osteosarcoma | [55] |
| MMP-14 | LI-COR | RSCitG-HPhe-YLY | PET/NIRF | Glioma | [56] |
3.1 Fluorescence imaging (FI)
FI has shown considerable promise in applications of medical diagnosis, drug delivery, and image-guided surgery, which attributed to its benefits of non-invasive detection, high sensitivity, in situ operability, and high temporal resolution [57]. Owing to the high resolution and good biocompatibility of fluorescent probes, FI has become an attractive method for study of biomolecules, signal pathways, and biological reactions [58]. In recent years, a variety of fluorescent dyes have been developed, including organic dyes such as small molecule dyes, aggregation induced emission (AIE) nanoparticles, semiconductor polymer nanoparticles, and inorganic materials such as semiconductor quantum dots (QDs), metal nanoclusters, rare earth doped nanoparticles, and nanodiamonds [59,60]. At present, MMP-responsive fluorescent probes are mostly constructed based on fluorescence resonance energy transfer (FRET) principle to form an energy acceptor-polypeptide-energy donor system [61]. As shown in Figure 1, when the distance between energy donor and receptor is less than 10 nm, energy transfer occurs between the two, resulting in fluorescence quenching. When enzyme hydrolyzes the substrate polypeptide, the FRET system is damaged, thereby turning on the fluorescence and realizing detection of tissues [12,62].
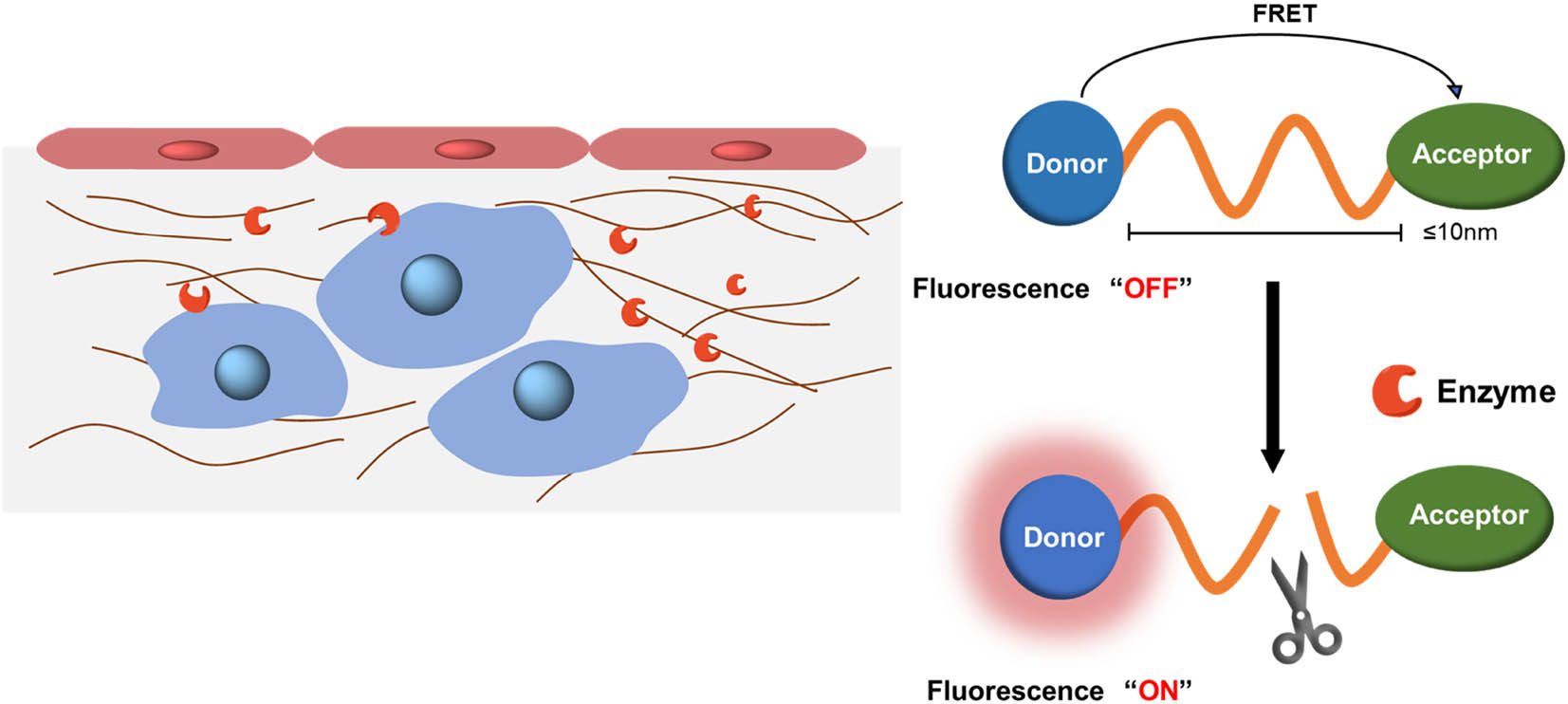
FRET system based on enzyme response.
3.1.1 Organic fluorescent probes
Organic small molecule fluorescent reagents are a class of biological probes with excellent performance, featuring high sensitivity, good biocompatibility, and fast reaction time. Luan et al. developed Dab-GPLGVRGY-FITC fluorescent probes for rapid and accurate imaging of MMP-2 overexpressed gastric cancers [48]. Upon cleavage of GPLGVRGY by MMP-2, separation of Dab/FITC pairs decomposed the FRET system and activated fluorescence emission of FITC. Their results demonstrated that the nanoprobes possessed excellent sensitivity to MMP-2, which measured approximately 40-fold fluorescence enhancement at 520 nm with a detection limit of 42 ng·mL−1. Xu et al. synthesized MMP-activatable I780BP-PEG12 probes by labeling the MMP substrate peptide GPLGVRGKGG with near-infrared dye IR780 and quencher BHQ-3. Benefiting from the targetability of substrate peptides and long wavelengths of near-infrared fluorophores, the I780BP-PEG12 probes exhibited highly sensitive detection of MMPs in tumor areas as low as nanomolar concentrations, which allowed deep tissue imaging and dynamic non-invasive evaluation. Their results showed that the I780BP-PEG12 probes provided significant fluorescence enhancement in early stage of pancreatic ductal adenocarcinoma model with only 3 mm of tumor diameter [49].
In addition to fluorescence enhancement, the ratiometric fluorescent probes have unique advantages in detection of biological systems with high accuracy. The ratio type fluorescent probes show a change in enzyme activity by a proportional change in fluorescence intensity at two wavelengths [63]. Zeng et al. Designed ratiometric near-infrared fluorescent nanoprobes (SPN-MMP-RGD) for sensitive detection of MMP-2 activity in vitro [50]. As shown in Figure 2a, after self-assembly of semiconductor nanoparticles (SPNs) based on near-infrared absorption organic polymer (PCPDTBT), the nanoprobes were further modified with MMP-2 cleavable peptide GGPLGVRGK linked fluorophore Cy5.5 and its quencher QSY21, as well as labeled with cRGD ligand that could bind to tumor αvβ3 integrin. The results showed that upon interaction with MMP-2, fluorescence of SPN-MMP-RGD nanoprobes at 690 nm could activate fluorescence due to broken FRET structure between Cy5.5 and QSY21, while the fluorescence of PCPDTBT at 830 nm remained unchanged (Figure 2b), resulting in a significant enhancement of fluorescence ratio between 690 and 830 nm (I 690/I 830) by about 176-fold (Figure 2c).
![Figure 2
(a) Scheme of preparation and activation of ratiometric NIR fluorescence of SPN-MMP-RGD toward MMP-2 under irradiation by 660 nm. (b) Fluorescence spectra and (c) fluorescence intensity ratio (I
690/I
830) of SPN-MMP-RGD (28 µg·mL−1 PCPDTBT) before and after incubation with MMP-2 (8 nM). Reprinted with permission from Zeng et al. [50]. Copyright 2021, Wiley-VCH.](/document/doi/10.1515/revac-2022-0044/asset/graphic/j_revac-2022-0044_fig_002.jpg)
(a) Scheme of preparation and activation of ratiometric NIR fluorescence of SPN-MMP-RGD toward MMP-2 under irradiation by 660 nm. (b) Fluorescence spectra and (c) fluorescence intensity ratio (I 690/I 830) of SPN-MMP-RGD (28 µg·mL−1 PCPDTBT) before and after incubation with MMP-2 (8 nM). Reprinted with permission from Zeng et al. [50]. Copyright 2021, Wiley-VCH.
3.1.2 Inorganic fluorescent probes
Inorganic fluorescent nanomaterials have become a hotspot in the field of bioimaging due to their high fluorescence intensity, good photostability, and large Stokes shift, which include three major categories: QDs, rare earth fluorescent materials, and noble metal nanoclusters [64–66]. Gold nanoparticles (AuNPs) are widely used for FI owing to their good biocompatibility and high quenching efficiency. Guo et al. constructed a novel tricolor fluorescence nanoplatforms based on a high-fidelity gold-selenium (Au-Se) bond for simultaneously imaging and in situ monitoring the expression level of MMP-2/7/9 protein [51]. The nanoprobes were conjugated with three peptide substrates labeled with fluorescein isothiocyanate (FITC), 5-carboxytetramethylrhodamine (5-TAMRA), and cyanine 5 (Cy5), respectively. When hydrolyzed by MMP-2/7/9 in tumor, the previously quenched fluorescence by FRET was recovered, so that the biomarker MMP-2/7/9 could be visualized to elucidate the invasion and migration behaviors of tumor cells in an inflammatory environment. QDs are fluorescent nanocrystals with excellent characteristics for biological imaging, including adjustable spectral range, strong anti-photobleaching ability, and easy surface modification [67]. For instance, Chung et al. developed quantum dot-based fluorescence resonance energy transfer (QD-FRET) nanosensors to visualize the activity of MT1-MMP on cell membranes. Via FRET and penetrated QD signals, the nanosensors could profile cancer cells [52]. Pham-Nguyen et al. reported a tumor sensing system based on AuNPs and QDs via an MMP-cleavable linker. The fluorescence of the QDs was quenched by AuNPs based on FRET, allowing fluorescence monitoring of tumor-specific enzymes in vivo levels (Figure 3a) [53]. When AuNPs and QDs reached tumor with overexpression of MMP-2, the methoxy PEG as a protective layer was removed and the azide fraction was exposed, which clicked with QDs to form Au-QD nanoclusters. The fluorescence of QDs was quenched by AuNPs due to FRET effect. As shown in Figure 3b, fluorescence quenching of QDs occurred within 1 h after exposure to 0.25–1.0 nM of MMP-2. Furthermore, it was demonstrated that the FRET efficiency was proportional to the MMP-2 level (Figure 3c).
![Figure 3
(a) Scheme of preparation and enzyme-responsive cleavage of nanoclusters for real-time imaging. (b) IVIS images and (c) FRET efficiency of Au-QD nanoclusters at various incubation times and MMP-2 concentrations. Reprinted with permission from Pham-Nguyen et al. [53]. Copyright 2022, American Chemical Society.](/document/doi/10.1515/revac-2022-0044/asset/graphic/j_revac-2022-0044_fig_003.jpg)
(a) Scheme of preparation and enzyme-responsive cleavage of nanoclusters for real-time imaging. (b) IVIS images and (c) FRET efficiency of Au-QD nanoclusters at various incubation times and MMP-2 concentrations. Reprinted with permission from Pham-Nguyen et al. [53]. Copyright 2022, American Chemical Society.
3.2 Multimodal imaging
FI has high sensitivity but is limited by tissue penetration depth, while magnetic resonance imaging (MRI) has high soft tissue resolution, unlimited tissue penetration, but low sensitivity. In addition, computed tomography has high spatial resolution but carries radiation risks [68]. Therefore, compared with single imaging, integrating the complementary advantages of multiple imaging modalities can provide more comprehensive and detailed information for precise diagnosis of tumors.
MRI contrast agents combined with photoacoustic imaging agents are expected to produce complementary imaging effects. Melanin has become a potential photoacoustic contrast agent due to its excellent photoacoustic properties and near-infrared absorption capability. Meng et al. chelated Gd3+ on melanin nanoparticles, making them possess dual-mode MRI/PA imaging ability [54]. In addition, due to modification of MMP-2 peptide substrate, the nanoparticles were hydrolyzed under MMP-2 enzyme to trigger exposure of the hydrophobic end, leading to nanoparticles accumulated in tumor with longer time of retention, finally showing an excellent imaging effect.
At present, dual-mode imaging strategies are commonly used including FI/MRI, PA/MRI, CT/MRI, and FI/SERS, etc. Among these strategies, the FI/SERS detection platform has proved to be a very reliable analytical tool in the field of biosensing. Taking advantage of the high sensitivity and fluorescence visualization of SERS, Liu and colleagues designed a novel hybrid nanosensor to accurately monitor MMP-2 activity in cell secretions and human serum samples [55]. Specifically, the prepared nanosensor (FMNS@Au) were based on biological self-assembly, in which MMP-2 peptide substrate (PLGVR) acts as a bridge between AuNP and fluorescent magnetic nanospheres (FMNS), forming a FRET system. Under the action of MMP-2, the FRET system was damaged, meanwhile the “hot spot” effect of SERS was weakened, which resulted in recovery of fluorescence signal and reduction in SERS signal, and thereby improving detection sensitivity of MMP-2.
Surgery is the main treatment of cancer, and the extent of surgical resection is closely related to the prognosis of patients. Accurate tumor resection, especially for the tumors located in complex position in body, such as glioma, is an urgent problem to be solved. Currently, multimodal imaging-guided surgery has become a hot research field. MMP-14 is overexpressed in glioma, which can be used as a biomarker for molecular imaging of glioma. Kasten et al. designed dual-mode imaging probes for MMP-14 targeting, and applied them in near-infrared fluorescence (NIRF)/PET-guided resection of glioma (Figure 4a) [56]. The probes consist of two peptide sequences, one is MMP14 substrate peptide with near-infrared fluorophore IRDye800 and the quencher QC-1-NHS attached to each end, and thus can be used for MMP-14 activatable NIRF imaging, and the other is peptide labeled with radioactive elements 64Cu(ii) or 68Ga(iii) for PET imaging (Figure 4b). As shown in Figure 4c, U87 and U251 cells showed high levels of MMP-14 expression by immunofluorescence, demonstrating the ability of these cell lines to detect enzyme substrates. This was also confirmed by the results of Figure 4d, where the NIRF signals of U87 and U251 cells were higher under the NIRF microscope when the substrate peptide and the substrate-binding peptide were incubated with the glioma cell line in vitro. In addition, Figure 4e–g illustrates that the MMP-14 probe, after intravenous injection in mice bearing in situ PDX JX12 glioma, shows superior NIRF/PET imaging capability with significant contrast at the tumor site relative to normal brain tissue. The results indicated that PET and NIRF signals were linearly related and co-localized with MMP-14 expression in resected tumors in orthotopic patient-derived xenograft glioma tumors.
![Figure 4
(a) Scheme for dual-modality PET/NIRF imaging of glioma (GBM) with an MMP-14 activatable peptide. (b) Structure of MMP-14 substrate-binding peptide probe. (c) Immunofluorescence signal quantification of MMP-14 in GBM adherent cells cultured in vitro. (d) Cell-associated NIRF signals (red) of glioma cells (D54, U87, U251) after incubation with substrate binding peptide (top), substrate peptide (middle), or buffer control without peptide (bottom) for 1 h. (e) NIRF imaging of mouse tissue sections harboring in situ PDX JX12 glioma tumor 1 h after intravenous injection of substrate-binding peptide. (f) PET images in mice bearing in situ PDX JX12 glioma tumors 4 h after intravenous injection of 64Cu substrate binding peptide. (g) In vitro biodistribution showing whole brain activity 5.5 h after intravenous injection of 64Cu substrate binding peptide or 64Cu substrate binding peptide + blockade (non-labeled binding peptide) in mice bearing in situ PDX JX12 glioma tumors. Reprinted with the permission from Kasten et al. [56]. Copyright 2019, Springer Nature.](/document/doi/10.1515/revac-2022-0044/asset/graphic/j_revac-2022-0044_fig_004.jpg)
(a) Scheme for dual-modality PET/NIRF imaging of glioma (GBM) with an MMP-14 activatable peptide. (b) Structure of MMP-14 substrate-binding peptide probe. (c) Immunofluorescence signal quantification of MMP-14 in GBM adherent cells cultured in vitro. (d) Cell-associated NIRF signals (red) of glioma cells (D54, U87, U251) after incubation with substrate binding peptide (top), substrate peptide (middle), or buffer control without peptide (bottom) for 1 h. (e) NIRF imaging of mouse tissue sections harboring in situ PDX JX12 glioma tumor 1 h after intravenous injection of substrate-binding peptide. (f) PET images in mice bearing in situ PDX JX12 glioma tumors 4 h after intravenous injection of 64Cu substrate binding peptide. (g) In vitro biodistribution showing whole brain activity 5.5 h after intravenous injection of 64Cu substrate binding peptide or 64Cu substrate binding peptide + blockade (non-labeled binding peptide) in mice bearing in situ PDX JX12 glioma tumors. Reprinted with the permission from Kasten et al. [56]. Copyright 2019, Springer Nature.
4 Applications in cancer therapy
Up to now, various methods including surgery, chemotherapy, radiotherapy, immunotherapy, phototherapy, and thermal therapy have been widely applied in cancer treatment, but therapeutic effect of these methods is still limited due to metastasis of cancer cells and poor prognosis [69]. MMPs response-based intelligent nanoplatform has become the focus in cancer treatment [70]. Enzyme-specific substrate peptides are used as carriers to load chemotherapeutic drugs, radiotherapy sensitizers, photosensitizers, or photothermal agents through physical adsorption or chemical bonding, and to develop multi-functional nanoplatforms to address the difficult problems in tumor treatment [71]. The latest advances in MMPs-responsive cancer treatments are systematically summarized in Table 3.
Representative development of nanoparticles responsive to MMP and their application in cancer therapy
| Enzyme | Nanocarrier | Substrate | Drug | Therapy | Tumor | Ref. |
|---|---|---|---|---|---|---|
| MMP-14 | MC-T-DOX | KRRQLGLPALSβAla | DOX | Chemotherapy | Pancreatic cancer | [72] |
| MMP-2 | ELP-CPP-Do | PLGALG | DOX | Chemotherapy | Breast cancer | [73] |
| MMP-2/9 | RPM@NLQ | CYGGRGNG | PTX | Chemotherapy | Breast cancer | [74] |
| MMP-2 | T-PFRT | GPLGVRGK | / | PDT | Breast cancer | [75] |
| MMP-2 | CeOx-EGPLGVRGK-PPa | EGPLGVRGK | / | PDT | Liver cancer | [76] |
| MMP-9 | Au@Res | GPLG | / | PTT | Liver cancer | [77] |
| MMP-2 | P/ML-NNG | GPLGIAGQG | / | Immunotherapy | Melanoma | [78] |
| MMP-2 | PEG-MP9-aPDL1 | PLGLAG | / | Immunotherapy | Colon cancer | [79] |
| MMP-2 | AFT/2-BP@PLGA@MD | PLGA | / | Immunotherapy | Breast cancer | [80] |
| MMP-2 | PMRA (DPPA-1-PLGLAG-RRRRRRRR-PyTPA), | PLGLAG | / | Immunotherapy | Colon cancer | [81] |
| MMP-2 | MA-pepA-Ce6 | GPLGVRGDK | / | PDT, immunotherapy | Breast cancer | [82] |
| MMP-2 | NIA-D1@R848 | PLGLAG | / | Radiotherapy, immunotherapy | Colon cancer | [83] |
| MMP-2 | A/Au@MSMs-P | PVGLIG | abemaciclib | PTT, immunotherapy, chemotherapy | Breast cancer | [84] |
4.1 Chemotherapy
Traditional chemotherapeutic drugs lack targeting, which will cause toxicity to normal tissues [16]. Taking advantage of specially overexpressed MMPs in tumor, targeted drug delivery system can accurately deliver and control release drugs in tumor site, thereby reducing side effects and improving the treatment effect [85]. Doxorubicin (DOX) is an anthracycline that has been used as first-line drug in clinical treatment of malignant tumors such as breast cancer, malignant lymphoma, and lung cancer [86]. Due to the concomitant toxic and side effects, such as cardiac toxicity and liver injury, DOX is seriously restricted in clinical treatment of cancer [87]. For improving pancreatic cancer treatment and reducing side effects of DOX, Wei et al. developed an intelligent response-type nanovesicle MC-T-DOX loaded with DOX (Figure 5) [72]. After intravenously injecting MC-T-DOX in pancreatic cancer model, MC-T-DOX was activated by MT1-MMP from tumor endothelial cells (ECs) to release selegiline, thereby promoting ECs migration and angiogenesis, and improving the accumulation and distribution of MC-T-DOX at tumor site. The DOX was then released under thermal triggered by MC-T-DOX, which increased bioavailability. Ryu et al. further improved DOX delivery system by carrying an MMP-2 responsive polypeptide linker, which showed better cell penetration and could induce more cancer cell death [73]. Paclitaxel (PTX) is another first-line chemotherapeutic drug. Duan et al. designed MMP-triggered liposomes that sequentially loaded quercetin and PTX for fibrotic tumor microenvironment (TME) remodeling and chemotherapy boosting [74]. After administration, the liposomes specifically accumulated in stroma-rich tumor sites, and the two drugs were released when undergoing MMP digestion.
![Figure 5
Scheme of MC-T-DOX enhances tumor blood perfusion and drug delivery in pancreatic cancer. MT1-MMP, membrane type 1-matrix metalloproteinase; MC, MT1-MMP-activated cilengitide. Reprinted by permission from Wei et al. [72]. Copyright 2020, Wiley-VCH.](/document/doi/10.1515/revac-2022-0044/asset/graphic/j_revac-2022-0044_fig_005.jpg)
Scheme of MC-T-DOX enhances tumor blood perfusion and drug delivery in pancreatic cancer. MT1-MMP, membrane type 1-matrix metalloproteinase; MC, MT1-MMP-activated cilengitide. Reprinted by permission from Wei et al. [72]. Copyright 2020, Wiley-VCH.
4.2 Phototherapy
Phototherapy, including photodynamic therapy (PDT) and photothermal therapy, is a non-invasive treatment with features of high safety and high selectivity [88]. PDT utilizes photosensitizers to produce cytotoxic reactive oxygen species under irradiation of near-infrared light to induce apoptosis [89]. However, most of the photosensitizers lack cancer targeting capability and tissue penetration. It has been clarified that MMPs-responsive probes can improve the penetration of photosensitizers and achieve PDT in deep tumor [90]. Based on this strategy, Liang et al. overcame physiological barriers caused by TME and delivered zinc phthalocyanine photosensitizers (ZnPc) into deep tumor, which enhanced therapeutic efficiency [75]. As shown in Figure 6a, the nanoprobes consisted of two parts: (1) ferritin nanocage (PFRT) encapsulated ZnPc and (2) dendritic mesoporous silicon nanoparticles (DMSN) loaded oxygen-supplied hemoglobin (Hb)/matrix remodeling reagent (iTGFb). The two parts were linked by MMP-2 substrate peptide to form core-satellite nanoframeworks (T-PFRT) with convertible dimensions. After the nanoframeworks were cleavaged into two parts by MMP-2, DMSN released Hb and iTGFb led to normalization of TEM, which further promoted deep tissue penetration of PFRT, and finally improved PDT effect of ZnPc. As shown in Figure 6b, compared with addition of enzyme inhibitor SB-3CT, DMSN- PFRT showed more deep penetration of released PFRT in tumor spheroids. The tumor size curve in Figure 6c indicates that laser irradiated T-PFRT showed superior tumor suppression over the other groups. Similarly, Fan et al. combined catalase mimetics (Cerium oxide, CeOx) and photosensitizers with MMP-2 peptide substrate to overcome tumor hypoxia in PDT [76]. When the peptide (EGPLGVRGK) was cut by MMP-2 at cleavage site between V and G, the smart nanoprobes changed from “silent state” before reaching cancer cell to “activated state” inside cell, thus turning on fluorescence and generating 1O2.
![Figure 6
(a) The structure and composition of nanoframeworks T-PFRT. (b) Penetration depth of different formulations in 3D tumor spheroid. (c) Tumor growth curve in different groups. G1, PBS; G2, T-PFRT without laser irradiation; G3, DMSN-PFRT (w/o pep) with laser irradiation; G4, DMSN-PFRT with laser irradiation; G5, iTGFbDMSN-PFRT with laser irradiation; G6, OxyHbDMSN-PFRT with laser irradiation; G7, T-PFRT with laser irradiation. Reprinted with permission from Liang et al. [75]. Copyright 2021, Wiley-VCH.](/document/doi/10.1515/revac-2022-0044/asset/graphic/j_revac-2022-0044_fig_006.jpg)
(a) The structure and composition of nanoframeworks T-PFRT. (b) Penetration depth of different formulations in 3D tumor spheroid. (c) Tumor growth curve in different groups. G1, PBS; G2, T-PFRT without laser irradiation; G3, DMSN-PFRT (w/o pep) with laser irradiation; G4, DMSN-PFRT with laser irradiation; G5, iTGFbDMSN-PFRT with laser irradiation; G6, OxyHbDMSN-PFRT with laser irradiation; G7, T-PFRT with laser irradiation. Reprinted with permission from Liang et al. [75]. Copyright 2021, Wiley-VCH.
Different from PDT, photothermal therapy (PTT) is a method that converts the absorbed near-infrared light into thermal energy by photothermal agent to ablate cancer cells [91].The principle of PTT is that cancer cells have lower heat tolerance than normal cells, and heat can cause irreversible damage to cancer cell membranes and trigger protein denaturation [92]. Although PTT has made great progress in cancer treatment, non-specific PTT reagents have certain limitations, such as inefficiency accumulation in tumor, low photothermal conversion, and inevitable damage to normal tissue [93,94]. In order to minimize the damage to non-pathological tissues, Wu et al. modified gold nanorods with MMP-9 sensitive peptide sequence (GPLG) [77]. Compared with nude gold nanorods, peptide modified gold nanorods showed enhanced accumulation in tumor, and therefore improved treatment efficiency.
4.3 Immunotherapy
Immunotherapy is a method of killing cancer cells by activating human autoimmune system [95]. Utilizing enzyme-specific hydrolysis to construct nanocarriers for immunotherapeutic drug delivery is a common strategy for MMPs-based immunotherapy. Liu et al. designed NF-κB pathway inhibitor IMD-0354 and programmed cell death protein 1 (PD-1) antibody co-loaded enzyme-responsive nanoparticles. Under decomposition by MMP-2, the nanoparticles released drugs could promote immune checkpoint-blocking immunotherapy by inducing polarization of tumor-associated macrophages and inhibiting the programmed cell death protein-1 (PD-1)/Programmed cell death 1-ligand 1 (PD-L1) pathway [78]. Similarly, Lu et al. exploited the abundant MMP-2 in TME to initiate tumor lysis and release tumor-associated antigen, addressing the problem of colorectal cancer insensitivity to anti-PD-L1 immunotherapy due to the lack of neoantigen [79]. In addition, D-peptide antagonist (DPPA-1), an immune checkpoint inhibitor, is often applied in immunotherapy as a polypeptide sequence with PD-1/PD-L1 blocking function due to its convenience in modification and strong penetration [80]. For example, Dai et al. used MMP-2 substrate peptide to immobilize immune checkpoint inhibitor DPPA-1 on outer membrane (Figure 7a). High expression of MMP-2 in tumor could reactively release DPPA-1 to block PD-1/PD-L1 pathway and activate anti-tumor immune response, thereby greatly inhibiting tumor growth and metastasis (Figure 7b). Compared with control group, tumor fluorescence intensity of the DIR@PLGA@MD group was significantly increased, which indicates excellent tumor targeting ability of the nanoprobes (Figure 7c). Figure 7d shows that AFT/2BP@PLGA@MD exhibited significant tumor inhibition (71.9%) and improved survival. In addition, AFT/2-BP@PLGA@MD significantly inhibited metastatic nodules in lung after 34 days (Figure 7e). Currently, coupling MMP-substrate peptides with DPPA-1 peptides to achieve responsive release in TME is a common strategy [81]. In conclusion, enzyme-mediated delivery system can improve drug efficacy, reduce immunotoxicity, and provide a platform for synergistic cancer immunotherapy.
![Figure 7
(a) Scheme of preparation of AFT/2-BP@PLGA@MD nanoparticles. (b) The antitumor mechanism of AFT/2-BP@PLGA@MD. (c) FI of mice administration by various nanoparticles labeled with DIR. Fluorescence images of tumors and main organs of mice intravenously injected at 48 h and quantitative analysis of fluorescence intensity of ex vivo images. (d) Mice survival curves. (e) Images of metastatic nodules in lungs. Reprinted with permission from Wang et al. [80]. Copyright 2022, American Chemical Society.](/document/doi/10.1515/revac-2022-0044/asset/graphic/j_revac-2022-0044_fig_007.jpg)
(a) Scheme of preparation of AFT/2-BP@PLGA@MD nanoparticles. (b) The antitumor mechanism of AFT/2-BP@PLGA@MD. (c) FI of mice administration by various nanoparticles labeled with DIR. Fluorescence images of tumors and main organs of mice intravenously injected at 48 h and quantitative analysis of fluorescence intensity of ex vivo images. (d) Mice survival curves. (e) Images of metastatic nodules in lungs. Reprinted with permission from Wang et al. [80]. Copyright 2022, American Chemical Society.
4.4 Combination therapy
Despite the fact that individual medicines have anti-tumor effects, it is still necessary to construct multifunctional therapeutic platform to deal with the complex TME, and enhance therapeutic efficiency [96]. It has been demonstrated that combination of two or more therapeutic techniques can achieve more effective treatment and minimize incidence of tumor recurrence. Because of convenient modification, the MMPs substrate-based responsive strategy can integrate multiple drugs or treatment modalities to achieve multifunctional combination therapy.
As a representative, Hu et al. prepared enzyme-responsive MA-PEPA-Ce6 nanoparticles, on which PD-L1 inhibitor (metformin, MET) and photosensitizer (chlorin, CE6) were loaded via substrate peptide GPLGVRGDK [82]. After degraded by MMP-2 in tumor, the exposed VRGDK-Ce6 could specially bind with integrin αvβ3 receptor, and induce a strong anti-tumor immune effect under laser irradiation. Meanwhile, the released MET in acidic TME could further amplify the anti-tumor immune response, therefore combinedly inhibiting tumor growth. Self-assemble of amphiphilic peptides containing both hydrophilic and hydrophobic groups is another effective strategy for drug carriers. As shown in Figure 8a, Zhu et al. synthesized amphiphilic polypeptide by using MMP-2-sensitive peptide PLGLAG to graft hydrophobic radiotherapy sensitizers 2-(2-nitroimidazol1-yl) acetic acid (NIA) and hydrophilic PD-L1 antagonist DPPA-1 [83]. Further, as-synthesized amphiphilic peptides were co-assembled with hydrophobic immune adjuvant R848 to form nanoparticles. As shown in Figure 8b–d, MMP-2 responsive release of NIA, R848, and D1 peptide dramatically enhanced radiation sensitivity of tumor cells, promoted maturation of dendritic cells, and blocked PD-1/PD-L1 pathway, respectively. Therefore, both primary tumor and distal tumor were inhibited, and long-term immune protection was generated to prevent tumor recurrence.
![Figure 8
(a) Antitumor mechanism of NIA-D1@R848. (b) Pictures of primary tumors and abscopal tumors after different treatments at day 23; (A–F) represent mice treated with saline, RT, R848, NIA-D1@R848, NIA-D1@R848 + RT, and C16-D1@R848 + RT at an equivalent R848 dose of 0.5 mg·kg−1, respectively; scale bar = 1 cm. (c) Growth curve of primary and abscopal tumors. (d) Tumor images and growth curve of tumors from control naive mice and NIA-D1@R848 + RT cured mice. Scale bar = 1 cm. Reprinted with the permission from Zhu et al. [83]. Copyright 2022, Wiley-VCH.](/document/doi/10.1515/revac-2022-0044/asset/graphic/j_revac-2022-0044_fig_008.jpg)
(a) Antitumor mechanism of NIA-D1@R848. (b) Pictures of primary tumors and abscopal tumors after different treatments at day 23; (A–F) represent mice treated with saline, RT, R848, NIA-D1@R848, NIA-D1@R848 + RT, and C16-D1@R848 + RT at an equivalent R848 dose of 0.5 mg·kg−1, respectively; scale bar = 1 cm. (c) Growth curve of primary and abscopal tumors. (d) Tumor images and growth curve of tumors from control naive mice and NIA-D1@R848 + RT cured mice. Scale bar = 1 cm. Reprinted with the permission from Zhu et al. [83]. Copyright 2022, Wiley-VCH.
For considerable enhancing treatment efficiency, three or more therapeutic medications have been successfully mounted on the same nanoplatform. Gao et al. designed novel nanoplatforms consisting of abemaciclib-loaded photothermal nanoparticles (A/Au@MSMs) and MMP-2 substrate peptide-conjugated anti-PD-1 antibody [84]. The nanoplatforms carried chemical-photothermal-immune synergistic therapy in one treatment, which completely eliminated tumor and showed great potential for cancer treatment in future.
5 Integration of diagnosis and treatment
Integration of diagnosis and treatment will visually evaluate and real-time monitor therapeutic effect, and is of great significance for precision medicine. MMPs-responsive nanoprobes have been continuously optimized to construct high performance of imaging and therapy functions in one platform, which aims to provide patients with more personalized treatment plans and better prognosis means (Table 4).
Examples of MMPs-responsive nanoprobes for integration of diagnosis and treatment
| Enzyme | Nanocarrier | Substrate | Diagnosis | Therapy | Tumor | Ref. |
|---|---|---|---|---|---|---|
| MMP-9 | PMP@USPIO/DOX | GPLGL | MRI | PTT, chemotherapy | Breast cancer | [97] |
| MMPs | TM@P/DOX | KCQGWIGQPGCK | MRI | PTT, chemotherapy | Breast cancer | [98] |
| MMP-2 | PEG2000-R8-PLGLAG-EK6 | PLGLAG | FI | Chemotherapy | Pancreatic cancer | [99] |
| MMP-2 | T-MAN | GGPLGVRGK | FI, MRI | PTT | Gastric cancer | [100] |
| MMP-2 | DSP@Ce6@Gd | PLGVRGRGDC | FI, MRI | PTT | Lung cancer | [101] |
| MMP-2/9 | TRA-GNRs-DM1 | CRDPLGLAGDRC | CT, PT, PA | PTT | Breast cancer | [102] |
5.1 Single mode image guided tumor therapy
MRI has been widely used in cancer diagnosis due to its advantages of non-invasive detection, high spatial resolution, and infinite tissue penetration depth [103]. Commonly used MRI contrast agents in clinic include T1 contrast agents such as gadolinium-based (Gd3+) or manganese-based (Mn2+) small molecular, and T2 contrast agents such as superparamagnetic iron oxide nanoparticles (SPIONs) [104,105]. The synergy of MRI contrast agents with cancer therapy show wide application prospect in tumor detection and drug release monitoring. Chen et al. designed novel MMP-9 responsive nanoplatform (PMP@USPIO/DOX, PMPSD) with T2–T1 conversion characteristics for tumor imaging and synergistic chemo-photothermal therapy [97]. Under physiological condition, the ultrasmall superparamagnetic iron oxide (USPIO) was in aggregated state and acted as T2 contrast agent. However, when polypeptide chains were cleaved by MMP-9 in TME, the released USPIO transformed into T1 contrast agent, which could obtain non-invasive imaging and quantitative analysis of MMP-9. In addition, the nanoplatform also shows excellent photothermal performance. Wang et al. synthesized multifunctional metal–organic frameworks modified with MMPs substrate peptide-conjugated bone targeting peptide and cell penetrating peptide for MRI guided chemo-photothermal therapy of bone tumor. The multifunctional metal-organic frameworks showed increased uptake in bone tumor cells, therefore enhancing photothermal-chemotherapeutic efficacy [98].
In addition to MRI, near-infrared FI-guided cancer therapy can supply high-sensitive optical signal of focus, thereby expecting to achieve precise treatment and real time prognosis evaluation. Chen et al. designed an image-guided drug delivery strategy using supramolecular AIE nanodots to achieve targeted drug release within tumor cells [99]. As shown in Figure 9a, the nanodots are composed of three parts, including two groups of functional alpha-cyclodextrin (alpha-CD) modified with anticancer drug gemcitabine (GEM) and AIE luminescent agent TPR, and a group of cell-penetrating peptide RRRRRRRRRR (R8) and zwitterionic stealth sequence EKEKEKEKEKEKEKEK (EK6) connected by MMP-2 peptide substrate PLGLAG. After MMP-2 degradation in tumor, the shielding sequence EK6 was removed to expose R8 to enhance intracellular internalization, and then release of GEM was triggered by intracellular reduction microenvironment. Compared to the control group, the nanodots showed enhanced fluorescence signaling and antitumor activity, demonstrating the targeting of MMP-2 in tumors (Figure 9b–e).
![Figure 9
(a) Illustration of preparation of α-CD-TPR-GEM-mmp (+) NP. (b) Fluorescence images of main organs and tumors of mice after administration of nanoparticles for 24 h. (c) Quantitative fluorescent signals of main organs and tumors in (b). (d) In vivo bioluminescence images of orthotopic pancreatic tumor-bearing mice in different treatment groups. (e) Tumor growth curves through quantitatively analyzing the in vivo bioluminescence signals from pancreatic tumors in different treatment groups. Reprinted with permission from Chen et al. [99]. Copyright 2020, American Chemical Society.](/document/doi/10.1515/revac-2022-0044/asset/graphic/j_revac-2022-0044_fig_009.jpg)
(a) Illustration of preparation of α-CD-TPR-GEM-mmp (+) NP. (b) Fluorescence images of main organs and tumors of mice after administration of nanoparticles for 24 h. (c) Quantitative fluorescent signals of main organs and tumors in (b). (d) In vivo bioluminescence images of orthotopic pancreatic tumor-bearing mice in different treatment groups. (e) Tumor growth curves through quantitatively analyzing the in vivo bioluminescence signals from pancreatic tumors in different treatment groups. Reprinted with permission from Chen et al. [99]. Copyright 2020, American Chemical Society.
5.2 Multimodal image-guided tumor therapy
Multimode imaging-guided therapy can overcome limitations of single-mode imaging and show higher application prospects. Shi et al. constructed a novel Gd3+ doped CuS magnetic SPNs (T-MAN), and then covalently modified with cRGD-targeted peptide, NIR fluorophore (Cy5.5), and quencher QSY21-labeled MMP-2 cleavable peptide substrate (Figure 10a) [100]. Following intravenous administration, T-MAN was actively delivered to gastric cancer tissues mediated by αvβ3 integrin and specifically cleaved by MMP-2 in ECM, which produced significantly enhanced NIR fluorescence and T1-weighted MR contrast signals to accurately depict gastric tumors (Figure 10b). Under the guidance of NIR/MR bimodal imaging, in situ gastric tumors were ablated by 808 nm NIR laser. Figure 10c–e indicated that when interacting with MMP-2, T-MAN exhibited a large fluorescence turn-on ratio at 690 nm (∼185-fold), a high r1 relaxation value (∼60.0 mM−1·s−1), and preferential tumor accumulation (∼23.4% ID%/g at 12 h).
![Figure 10
(a) Scheme of preparation of T-MAN nanodisks. (b) Fluorescence/MR imaging and PTT of in situ orthotopic gastric tumors. (c) Fluorescence spectra of T-MAN (0.5 μM MMP-2 substrate) incubated with MMP-2 (10 nM) for different times at 37°C. (d) r1 value of T-MAN and Dotarem at 1 T. (e) Distribution of T-MAN in MKN45 tumors and main organs of mice after administration. Reprinted with permission from Shi et al. [100]. Copyright 2019, American Chemical Society.](/document/doi/10.1515/revac-2022-0044/asset/graphic/j_revac-2022-0044_fig_010.jpg)
(a) Scheme of preparation of T-MAN nanodisks. (b) Fluorescence/MR imaging and PTT of in situ orthotopic gastric tumors. (c) Fluorescence spectra of T-MAN (0.5 μM MMP-2 substrate) incubated with MMP-2 (10 nM) for different times at 37°C. (d) r1 value of T-MAN and Dotarem at 1 T. (e) Distribution of T-MAN in MKN45 tumors and main organs of mice after administration. Reprinted with permission from Shi et al. [100]. Copyright 2019, American Chemical Society.
In addition to PTT, PDT-based diagnosis and treatment is also a research focus. For instance, Yang et al. modified amphiphilic DSPE-PEG2000 with MMP-2 substrate peptide PLGVRGRGDC and gadolinium chelating agent DTPA (diethylenetriamine pentaacetic acid), respectively, to form functionalized nanoparticles by self-assembly. Then, photosensitizers Ce6 and gadolinium ions were loaded on the nanoparticles. Their results showed MMP-triggered targeting peptides could specifically deliver Ce6 photosensitizers into A549 tumor cells and effectively ablate cancer cells under laser irradiation with guidance of NIRF/MR dual-mode imaging [101]. Furthermore, by using MMP-2/9 sensitive peptide as linker, Liu et al. prepared ultra-small gold nanoparticles with functions of three-mode imaging including CT, PA, and photothermal imaging, as well as high performance of photothermal therapeutic effect [102].
6 Conclusion and perspective
As an important cancer marker, MMPs participate in the whole process of cancer occurrence and development, including tumor growth, proliferation, invasion, metastasis, and angiogenesis. The combination of emerged nanotechnology with enzyme-responsive strategies shows great potential for targeted cancer diagnosis and treatment. In this work, we first introduced the types of cancer in which MMPs are overexpressed and their biological functions in cancer. Then, we focused on the advances of MMP-responsive nanoprobes in tumor imaging and treatment, and gave representative examples of integrated diagnosis and treatment probes.
Although most MMPs responsive strategies show promising results, they still face many challenges in clinical application. First, the MMPs have many uncertainties. On the one hand, the enzymes have slow recognition response to the substrate peptide and long degradation process; on the other hand, type and content of the proteases in body fluctuate greatly and have complex functions, as a result, other proteases degrade nanoprobes in advance to interfere the targeting effect. Second, the current design of multifunctional response systems mostly relies on building multiple components with different “diagnosis” or “treatment” functions together, and the obtained final system structures are complex, with poor circulation stability and easy to collapse during blood transportation. In addition, there are unpredictable interference between different components and even potential toxicity. Therefore, how to improve the sensitivity of nanoprobes to MMPs, enhance the specificity, improve the stability of transport in the body, optimize the synergy between functional groups, ensure the biocompatibility of nanoprobes to reduce toxic and side effects, and other issues still need efforts from various aspects. In addition, the excessive production cost has become one of the obstacles to the clinical application of enzyme-responsive nanoprobes. As a potential direction, MMP-based responsive nanoprobes are expected to open up new ideas for the diagnosis and treatment of cancer.
-
Funding information: This work was supported by the National Natural Science Foundation of China (31971292, 32025021, 51873225, 32171359), the Zhejiang Province Financial Supporting (2020C03110), the Key Scientific and Technological Special Project of Ningbo City (2020Z094), and the Science & Technology Bureau of Ningbo City (202003N4001), and the Users with Excellence Program of Hefei Science Center, CAS (2020HSC-UE006).
-
Author contributions: Qiuye Wang: writing – original draft and writing – review and editing; Haijing Cui: literature survey and writing – review and editing; Ning Gan: review; Xuehua Ma: writing – review and editing; Wenzhi Ren: supervision, conceptualization, and writing – review and editing; Aiguo Wu: resources, funding acquisition, and supervision.
-
Conflict of interest: Authors state no conflict of interest.
References
[1] Sung H, Ferlay J, Siegel RL, Laversanne M, Soerjomataram I, Jemal A, et al. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA-A Cancer J Clin. 2021;71(3):209–49. 10.3322/caac.21660.Search in Google Scholar PubMed
[2] Cathcart J, Pulkoski-Gross A, Cao J. Targeting matrix metalloproteinases in cancer: Bringing new life to old ideas. Genes Dis. 2015;2(1):26–34. 10.1016/j.gendis.2014.12.002.Search in Google Scholar PubMed PubMed Central
[3] Gondi CS, Rao JS. Cathepsin B as a cancer target. Expert Opin Ther Targets. 2013;17(3):281–91. 10.1517/14728222.2013.740461.Search in Google Scholar PubMed PubMed Central
[4] Lokeshwar VB, Mirza S, Jordan A. Targeting hyaluronic acid family for cancer chemoprevention and therapy. Adv Cancer Res. 2014;123:35–65. 10.1016/b978-0-12-800092-2.00002-2.Search in Google Scholar PubMed PubMed Central
[5] Muntimadugu E, Kommineni N, Khan W. Exploring the potential of nanotherapeutics in targeting tumor microenvironment for cancer therapy. Pharmacol Res. 2017;126:109–22. 10.1016/j.phrs.2017.05.010.Search in Google Scholar PubMed
[6] Quach ND, Arnold RD, Cummings BS. Secretory phospholipase A(2) enzymes as pharmacological targets for treatment of disease. Biochem Pharmacol. 2014;90(4):338–48. 10.1016/j.bcp.2014.05.022.Search in Google Scholar PubMed PubMed Central
[7] Shahriari M, Zahiri M, Abnous K, Taghdisi SM, Ramezani M, Alibolandi M. Enzyme responsive drug delivery systems in cancer treatment. J Controlled Rel. 2019;308:172–89. 10.1016/j.jconrel.2019.07.004.Search in Google Scholar PubMed
[8] Raeeszadeh-Sarmazdeh M, Do LD, Hritz BG. Metalloproteinases and their inhibitors: Potential for the development of new therapeutics. Cells. 2020;9(5):1313. 10.3390/cells9051313.Search in Google Scholar PubMed PubMed Central
[9] Kessenbrock K, Plaks V, Werb Z. Matrix metalloproteinases: Regulators of the tumor microenvironment. Cell. 2010;141(1):52–67. 10.1016/j.cell.2010.03.015.Search in Google Scholar PubMed PubMed Central
[10] Niland S, Riscanevo AX, Eble JA. Matrix metalloproteinases shape the tumor microenvironment in cancer progression. Int J Mol Sci. 2022;23(1):146. 10.3390/ijms23010146.Search in Google Scholar PubMed PubMed Central
[11] Ge Z, Liu S. Functional block copolymer assemblies responsive to tumor and intracellular microenvironments for site-specific drug delivery and enhanced imaging performance. Chem Soc Rev. 2013;42(17):7289–325. 10.1039/c3cs60048c.Search in Google Scholar PubMed
[12] Liu HW, Chen LL, Xu CY, Li Z, Zhang HY, Zhang XB, et al. Recent progresses in small-molecule enzymatic fluorescent probes for cancer imaging. Chem Soc Rev. 2018;47(18):7140–80. 10.1039/c7cs00862g.Search in Google Scholar PubMed
[13] Xiong J, Gao H. Matrix metalloproteases-responsive nanomaterials for tumor targeting diagnosis and treatment. J Microencapsul. 2017;34(5):440–53. 10.1080/02652048.2017.1343873.Search in Google Scholar PubMed
[14] Wang M, Gao B, Wang X, Li W, Feng Y. Enzyme-responsive strategy as a prospective cue to construct intelligent biomaterials for disease diagnosis and therapy. Biomater Sci. 2022;10(8):1883–903. 10.1039/d2bm00067a.Search in Google Scholar PubMed
[15] Yao QX, Huang ZT, Liu DD, Chen JL, Gao Y. Enzyme-instructed supramolecular self-assembly with anticancer activity. Adv Mater. 2019;31:1804814. 10.1002/adma.201804814.Search in Google Scholar PubMed
[16] Yao Q, Kou LF, Tu Y, Zhu L. MMP-responsive “Smart” drug delivery and tumor targeting. Trends Pharmacol Sci. 2018;39(8):766–81. 10.1016/j.tips.2018.06.003.Search in Google Scholar PubMed
[17] Agren MS, auf dem Keller U. Matrix metalloproteinases: How much can they do? Int J Mol Sci. 2020;21(8):2678. 10.3390/ijms21082678.Search in Google Scholar PubMed PubMed Central
[18] Shohrati M, Hosseini RH, Esfandiari MA, Najafian N, Najafian B, Golbedagh A. Serum matrix metalloproteinase levels in patients exposed to sulfur mustard. Iran Red Crescent Med J. 2014;16(3):e15129. 10.5812/ircmj.15129.Search in Google Scholar PubMed PubMed Central
[19] Tampa M, Georgescu SR, Mitran MI, Mitran CI, Matei C, Caruntu A, et al. Current perspectives on the role of matrix metalloproteinases in the pathogenesis of basal cell carcinoma. Biomolecules. 2021;11(6):903. 10.3390/biom11060903.Search in Google Scholar PubMed PubMed Central
[20] Garcia-Fernandez N, Jacobs-Cacha C, Mora-Gutierrez JM, Vergara A, Orbe J, Soler MJ. Matrix metalloproteinases in diabetic kidney disease. J Clin Med. 2020;9(2):472. 10.3390/jcm9020472.Search in Google Scholar PubMed PubMed Central
[21] Kapoor C, Vaidya S, Wadhwan V, Hitesh, Kaur G, Pathak A. Seesaw of matrix metalloproteinases (MMPs). J Cancer Res Ther. 2016;12(1):28–35. 10.4103/0973-1482.157337.Search in Google Scholar PubMed
[22] Pittayapruek P, Meephansan J, Prapapan O, Komine M, Ohtsuki M. Role of matrix metalloproteinases in photoaging and photocarcinogenesis. Int J Mol Sci. 2016;17:868. 10.3390/ijms17060868.Search in Google Scholar PubMed PubMed Central
[23] Quintero-Fabian S, Arreola R, Becerril-Villanueva E, Torres-Romero JC, Arana-Argaez V, Lara-Riegos J, et al. Role of matrix metalloproteinases in angiogenesis and cancer. Front Oncol. 2019;9:1370. 10.3389/fonc.2019.01370.Search in Google Scholar PubMed PubMed Central
[24] Arakaki PA, Marques MR, Santos M. MMP-1 polymorphism and its relationship to pathological processes. J Biosci. 2009;34(2):313–20. 10.1007/s12038-009-0035-1.Search in Google Scholar PubMed
[25] Nikolov A, Popovski N, Hristova I. Collagenases MMP-1, MMP-13, and tissue inhibitors TIMP-1, TIMP-2: Their role in healthy and complicated pregnancy and potential as preeclampsia biomarkers-a brief review. Appl Sci-Basel. 2020;10(21):7713. 10.3390/app10217731.Search in Google Scholar
[26] Chang YT, Chu LJ, Liu YC, Chen CJ, Wu SF, Chen CH, et al. Verification of saliva matrix metalloproteinase-1 as a strong diagnostic marker of oral cavity cancer. Cancers. 2020;12(8):2273. 10.3390/cancers12082273.Search in Google Scholar PubMed PubMed Central
[27] Dey S, Ghosh N, Saha D, Kesh K, Gupta A, Swarnakar S. Matrix metalloproteinase-1 (MMP-1) promoter polymorphisms are well linked with lower stomach tumor formation in eastern Indian population. PLoS One. 2014;9(2):e88040. 10.1371/journal.pone.0088040.Search in Google Scholar PubMed PubMed Central
[28] Liu XK, Lin LY, Tseng FY, Tan YC, Li J, Feng L, et al. Label-free electrochemical immunosensor based on gold nanoparticle/polyethyleneimine/reduced graphene oxide nanocomposites for the ultrasensitive detection of cancer biomarker matrix metalloproteinase-1. Analyst. 2021;146(12):4066–79. 10.1039/d1an00537e.Search in Google Scholar PubMed
[29] Pulukuri SMK, Rao JS. Matrix metalloproteinase-1 promotes prostate tumor growth and metastasis. Int J Oncol. 2008;32(4):757–65. 10.3892/ijo.32.4.757.Search in Google Scholar
[30] Wang J, Ye CY, Lu DM, Chen YX, Jia YL, Ying XG, et al. Matrix metalloproteinase-1 expression in breast carcinoma: a marker for unfavorable prognosis. Oncotarget. 2017;8(53):91379–90. 10.18632/oncotarget.20557.Search in Google Scholar PubMed PubMed Central
[31] Pezeshkian Z, Nobili S, Peyravian N, Shojaee B, Nazari H, Soleimani H, et al. Insights into the Role of Matrix Metalloproteinases in Precancerous Conditions and in Colorectal Cancer. Cancers. 2021;13(24):6226. 10.3390/cancers13246226.Search in Google Scholar PubMed PubMed Central
[32] Baidya SK, Amin SA, Jha T. Outline of gelatinase inhibitors as anti-cancer agents: A patent mini-review for 2010-present. Eur J Medicinal Chem. 2021;213:113044. 10.1016/j.ejmech.2020.113044.Search in Google Scholar PubMed
[33] Cabral-Pacheco GA, Garza-Veloz I, Castruita-De la Rosa C, Ramirez-Acuna JM, Perez-Romero BA, Guerrero-Rodriguez JF, et al. The roles of matrix metalloproteinases and their inhibitors in human diseases. Int J Mol Sci. 2020;21(24):9739. 10.3390/ijms21249739.Search in Google Scholar PubMed PubMed Central
[34] Mook ORF, Frederiks WM, Van Noorden CJF. The role of gelatinases in colorectal cancer progression and metastasis. Biochimica Et Biophysica Acta-Reviews Cancer. 2004;1705(2):69–89. 10.1016/j.bbcan.2004.09.006.Search in Google Scholar PubMed
[35] Das S, Amin SA, Jha T. Inhibitors of gelatinases (MMP-2 and MMP-9) for the management of hematological malignancies. Eur J Med Chem. 2021;223:113623. 10.1016/j.ejmech.2021.113623.Search in Google Scholar PubMed
[36] Wieczorek E, Jablonska E, Wasowicz W, Reszka E. Matrix metalloproteinases and genetic mouse models in cancer research: A mini-review. Tumor Biol. 2015;36(1):163–75. 10.1007/s13277-014-2747-6.Search in Google Scholar PubMed PubMed Central
[37] Liu Z, Tan RJ, Liu YH. The many faces of matrix metalloproteinase-7 in kidney diseases. Biomolecules. 2020;10(6):960. 10.3390/biom10060960.Search in Google Scholar PubMed PubMed Central
[38] Liao HY, Da CM, Liao B, Zhang HH. Roles of matrix metalloproteinase-7 (MMP-7) in cancer. Clin Biochem. 2021;92:9–18. 10.1016/j.clinbiochem.2021.03.003.Search in Google Scholar PubMed
[39] Augoff K, Hryniewicz-Jankowska A, Tabola R, Stach K. MMP9: A tough target for targeted therapy for cancer. Cancers. 2022;14(7):1847. 10.3390/cancers14071847.Search in Google Scholar PubMed PubMed Central
[40] Vos MC, van der Wurff AAM, van Kuppevelt TH, Massuger L. The role of MMP-14 in ovarian cancer: A systematic review. J Ovarian Res. 2021;14(1):101. 10.1186/s13048-021-00852-7.Search in Google Scholar PubMed PubMed Central
[41] Gong YX, Chippada-Venkata UD, Oh WK. Roles of matrix metalloproteinases and their natural inhibitors in prostate cancer progression. Cancers. 2014;6(3):1298–327. 10.3390/cancers6031298.Search in Google Scholar PubMed PubMed Central
[42] Knapinska AM, Fields GB. The expanding role of MT1-MMP in cancer progression. Pharm (Basel). 2019;12(2):77. 10.3390/ph12020077.Search in Google Scholar PubMed PubMed Central
[43] Pahwa S, Stawikowski M, Fields G. Monitoring and inhibiting MT1-MMP during cancer initiation and progression. Cancers. 2014;6(1):416–35. 10.3390/cancers6010416.Search in Google Scholar PubMed PubMed Central
[44] Gifford V, Itoh Y. MT1-MMP-dependent cell migration: Proteolytic and non-proteolytic mechanisms. Biochem Soc Trans. 2019;47:811–26. 10.1042/bst20180363.Search in Google Scholar PubMed PubMed Central
[45] Itoh Y, Seiki M. MT1-MMP: A potent modifier of pericellular microenvironment. J Cell Physiol. 2006;206(1):1–8. 10.1002/jcp.20431.Search in Google Scholar PubMed
[46] Suarez H, Lopez-Martin S, Toribio V, Zamai M, Hernandez-Riquer MV, Genis L, et al. Regulation of MT1-MMP Activity through its association with ERMs. Cells. 2020;9(2):348. 10.3390/cells9020348.Search in Google Scholar PubMed PubMed Central
[47] Thakur V, Bedogni B. The membrane tethered matrix metalloproteinase MT1-MMP at the forefront of melanoma cell invasion and metastasis. Pharmacol Res. 2016;111:17–22. 10.1016/j.phrs.2016.05.019.Search in Google Scholar PubMed
[48] Luan F, Yu Z, Yin L, Leng X, Shi Y, Wang J, et al. Accurate detection of matrix metalloproteinase-2 activity in clinical gastric cancer tissues using a fluorescent probe. Anal Methods. 2019;11(11):1516–21. 10.1039/c8ay02789g.Search in Google Scholar
[49] Xu T, Chang D, Cai Y, Min S, Ma Y, Mao H, et al. Targeting of an antecedent proteinase by an activatable probe with deep tissue penetration facilitates early visualization and dynamic malignancy evaluation of orthotopic pancreatic ductal adenocarcinoma (PDAC). Biomater Sci. 2019;7(8):3320–33. 10.1039/c9bm00441f.Search in Google Scholar PubMed
[50] Zeng W, Wu L, Sun Y, Wang Y, Wang J, Ye D. Ratiometric imaging of MMP-2 activity facilitates tumor detection using activatable near-infrared fluorescent semiconducting polymer nanoparticles. Small. 2021;17(36):e2101924. 10.1002/smll.202101924.Search in Google Scholar PubMed
[51] Guo W, Gao X, Zhan R, Zhao Z, Xu K, Tang B. Tricolor imaging of MMPs to investigate the promoting roles of inflammation on invasion and migration of tumor cells. Talanta. 2021;222:121525. 10.1016/j.talanta.2020.121525.Search in Google Scholar PubMed
[52] Chung EY, Ochs CJ, Wang Y, Lei L, Qin Q, Smith AM, et al. Activatable and cell-penetrable multiplex FRET nanosensor for profiling MT1-MMP activity in single cancer cells. Nano Lett. 2015;15(8):5025–32. 10.1021/acs.nanolett.5b01047.Search in Google Scholar PubMed PubMed Central
[53] Pham-Nguyen OV, Shin J, Park Y, Jin S, Kim SR, Jung YM, et al. Fluorescence-shadowing nanoparticle clusters for real-time monitoring of tumor progression. Biomacromolecules. 2022;23(8):3130–41. 10.1021/acs.biomac.2c00169.Search in Google Scholar PubMed PubMed Central
[54] Meng T, Fan B, Li Q, Peng X, Xu J, Zhang R. Matrix metalloproteinase-initiated aggregation of melanin nanoparticles as highly efficient contrast agent for enhanced tumor accumulation and dual-modal imaging. J Mater Chem B. 2020;8(43):9888–98. 10.1039/d0tb01651a.Search in Google Scholar PubMed
[55] Liu L, Chu H, Yang J, Sun Y, Ma P, Song D. Construction of a magnetic-fluorescent-plasmonic nanosensor for the determination of MMP-2 activity based on SERS-fluorescence dual-mode signals. Biosens Bioelectron. 2022;212:114389. 10.1016/j.bios.2022.114389.Search in Google Scholar PubMed
[56] Kasten BB, Jiang K, Cole D, Jani A, Udayakumar N, Gillespie GY, et al. Targeting MMP-14 for dual PET and fluorescence imaging of glioma in preclinical models. Eur J Nucl Med Mol Imaging. 2020;47(6):1412–26. 10.1007/s00259-019-04607-x.Search in Google Scholar PubMed PubMed Central
[57] Imamura T, Saitou T, Kawakami R. In vivo optical imaging of cancer cell function and tumor microenvironment. Cancer Sci. 2018;109(4):912–8. 10.1111/cas.13544.Search in Google Scholar PubMed PubMed Central
[58] Hou SG, Liang L, Deng SH, Chen JF, Huang Q, Cheng Y, et al. Nanoprobes for super-resolution fluorescence imaging at the nanoscale. Sci China-Chemistry. 2014;57(1):100–6. 10.1007/s11426-013-5014-6.Search in Google Scholar
[59] Yang RQ, Lou KL, Wang PY, Gao YY, Zhang YQ, Chen M, et al. Surgical navigation for malignancies guided by near-infrared-II fluorescence imaging. Small Methods. 2021;5:2001066. 10.1002/smtd.202001066.Search in Google Scholar PubMed
[60] Bouccara S, Sitbon G, Fragola A, Loriette V, Lequeux N, Pons T. Enhancing fluorescence in vivo imaging using inorganic nanoprobes. Curr OpBiotechnol. 2015;34:65–72. 10.1016/j.copbio.2014.11.018.Search in Google Scholar PubMed
[61] Wang ZS, Li CH, Wei YN. Application of fluorescence in studying therapeutic enzymes. Therapeutic Enzymes Funct Clin Implic. 2019;1148:105–14. 10.1007/978-981-13-7709-9_5.Search in Google Scholar PubMed
[62] Wu LL, Huang CS, Emery B, Sedgwick AC, Bull SD, He XP, et al. Forster resonance energy transfer (FRET)-based small-molecule sensors and imaging agents. Chem Soc Rev. 2020;49(15):5110–39. 10.1039/c9cs00318e.Search in Google Scholar PubMed PubMed Central
[63] Gui RJ, Jin H, Bu XN, Fu YX, Wang ZH, Liu QY. Recent advances in dual-emission ratiometric fluorescence probes for chemo/biosensing and bioimaging of biomarkers. Coord Chem Rev. 2019;383:82–103. 10.1016/j.ccr.2019.01.004.Search in Google Scholar
[64] Zheng YK, Lai LM, Liu WW, Jiang H, Wang XM. Recent advances in biomedical applications of fluorescent gold nanoclusters. Adv Colloid Interface Sci. 2017;242:1–16. 10.1016/j.cis.2017.02.005.Search in Google Scholar PubMed
[65] Yang YM. Upconversion nanophosphors for use in bioimaging, therapy, drug delivery and bioassays. Microchimica Acta. 2014;181(3-4):263–94. 10.1007/s00604-013-1139-8.Search in Google Scholar
[66] Ma F, Li CC, Zhang CY. Development of quantum dot-based biosensors: Principles and applications. J Mater Chem B. 2018;6(39):6173–90. 10.1039/c8tb01869c.Search in Google Scholar PubMed
[67] Park Y, Jeong S, Kim S. Medically translatable quantum dots for biosensing and imaging. J Photochem Photobiol C-Photochem Rev. 2017;30:51–70. 10.1016/j.jphotochemrev.2017.01.002.Search in Google Scholar
[68] Yuan H, Liang HY, Hou PD, Li J. Advanced nanomaterials for multimodal molecular imaging. Chem Res ChUniversities. 2021;37(4):840–5. 10.1007/s40242-021-1196-1.Search in Google Scholar
[69] Gong F, Yang NL, Wang XW, Zhao Q, Chen Q, Liu Z, et al. Tumor microenvironment-responsive intelligent nanoplatforms for cancer theranostics. Nano Today. 2020;32. 10.1016/j.nantod.2020.100851.Search in Google Scholar
[70] Zhang ZT, Huang-Fu MY, Xu WH, Han M. Stimulus-responsive nanoscale delivery systems triggered by the enzymes in the tumor microenvironment. Eur J Pharm Biopharm. 2019;137:122–30. 10.1016/j.ejpb.2019.02.009.Search in Google Scholar PubMed
[71] Zhang C, Wu W, Li RQ, Qiu WX, Zhuang ZN, Cheng SX, et al. Peptide-based multifunctional nanomaterials for tumor imaging and therapy. Adv Funct Mater. 2018;28(50):1804492. 10.1002/adfm.201804492.Search in Google Scholar
[72] Wei Y, Song S, Duan N, Wang F, Wang Y, Yang Y, et al. MT1-MMP-activated liposomes to improve tumor blood perfusion and drug delivery for enhanced pancreatic cancer therapy. Adv Sci. 2020;7(17):1902746. 10.1002/advs.201902746.Search in Google Scholar PubMed PubMed Central
[73] Ryu JS, Kratz F, Raucher D. Cell-penetrating doxorubicin released from elastin-like polypeptide kills doxorubicin-resistant cancer cells in in vitro study. Int J Mol Sci. 2021;22(3):1126. 10.3390/ijms22031126.Search in Google Scholar PubMed PubMed Central
[74] Duan H, Liu C, Hou Y, Liu Y, Zhang Z, Zhao H, et al. Sequential delivery of quercetin and paclitaxel for the fibrotic tumor microenvironment remodeling and chemotherapy potentiation via a dual-targeting hybrid micelle-in-liposome system. ACS Appl Mater Interfaces. 2022;14(8):10102–16. 10.1021/acsami.1c23166.Search in Google Scholar PubMed
[75] Liang T, Zhang B, Xing Z, Dong Y, Xu H, Chen X, et al. Adapting and remolding: Orchestrating tumor microenvironment normalization with photodynamic therapy by size transformable nanoframeworks. Angew Chem Int Ed. 2021;60(20):11464–73. 10.1002/anie.202102180.Search in Google Scholar PubMed
[76] Fan Y, Li P, Hu B, Liu T, Huang Z, Shan C, et al. A smart photosensitizer-cerium oxide nanoprobe for highly selective and efficient photodynamic therapy. Inorg Chem. 2019;58(11):7295–302. 10.1021/acs.inorgchem.9b00363.Search in Google Scholar PubMed
[77] Wu L, Lin B, Yang H, Chen J, Mao Z, Wang W, et al. Enzyme-responsive multifunctional peptide coating of gold nanorods improves tumor targeting and photothermal therapy efficacy. Acta Biomaterialia. 2019;86:363–72. 10.1016/j.actbio.2019.01.026.Search in Google Scholar PubMed
[78] Liu Y, Liang S, Jiang D, Gao T, Fang Y, Fu S, et al. Manipulation of TAMs functions to facilitate the immune therapy effects of immune checkpoint antibodies. J Controlled Rel. 2021;336:621–34. 10.1016/j.jconrel.2021.07.009.Search in Google Scholar PubMed
[79] Lu L, Zhang H, Zhou Y, Lin J, Gao W, Yang T, et al. Polymer chimera of stapled oncolytic peptide coupled with anti-PD-L1 peptide boosts immunotherapy of colorectal cancer. Theranostics. 2022;12(7):3456–73. 10.7150/thno.71129.Search in Google Scholar PubMed PubMed Central
[80] Wang X, Zhu X, Li B, Wei X, Chen Y, Zhang Y, et al. Intelligent biomimetic nanoplatform for systemic treatment of metastatic triple-negative breast cancer via enhanced EGFR-targeted therapy and immunotherapy. ACS Appl Mater Interfaces. 2022;14:23152–63. 10.1021/acsami.2c02925.Search in Google Scholar PubMed
[81] Dai J, Dong X, Liu R, Chen B, Dong X, Wang Q, et al. A peptide-AIEgen nanocomposite mediated whole cancer immunity cycle-cascade amplification for improved immunotherapy of tumor. Biomaterials. 2022;285:121528. 10.1016/j.biomaterials.2022.121528.Search in Google Scholar PubMed
[82] Hu C, He X, Chen Y, Yang X, Qin L, Lei T, et al. Metformin mediated PD‐L1 downregulation in combination with photodynamic‐immunotherapy for treatment of breast cancer. Adv Funct Mater. 2021;31(11). 10.1002/adfm.202007149.Search in Google Scholar
[83] Zhu X, Wang X, Li B, Zhang Y, Chen Y, Zhang W, et al. A three-in-one assembled nanoparticle containing peptide-radio-sensitizer conjugate and TLR7/8 agonist can initiate the cancer-immunity cycle to trigger antitumor immune response. Small. 2022;18(20):e2107001. 10.1002/smll.202107001.Search in Google Scholar PubMed
[84] Gao T, Zhang Z, Liang S, Fu S, Mu W, Guan L, et al. Reshaping antitumor immunity with chemo‐photothermal integrated nanoplatform to augment checkpoint blockade‐based cancer therapy. Adv Funct Mater. 2021;31(28). 10.1002/adfm.202100437.Search in Google Scholar
[85] Li Y, Zhang C, Li G, Deng G, Zhang H, Sun Y, et al. Protease-triggered bioresponsive drug delivery for the targeted theranostics of malignancy. Acta Pharm Sin B. 2021;11(8):2220–42. 10.1016/j.apsb.2021.01.017.Search in Google Scholar PubMed PubMed Central
[86] Matyszewska D. Drug delivery systems in the transport of doxorubicin. Surf Innov. 2014;2(4):201–10. 10.1680/si.13.00040.Search in Google Scholar
[87] Yu J, Qi H-L, Zhang H, Zhao Z-Y, Jing Z, Nie Z-Y. Morin inhibits dox-induced vascular inflammation by regulating PTEN/AKT/NF-kappaB pathway. Inflammation. 2022;15:1–3. 10.1007/s10753-022-01701-5.Search in Google Scholar PubMed
[88] Zou JH, Li L, Yang Z, Chen XY. Phototherapy meets immunotherapy: A win-win strategy to fight against cancer. Nanophotonics. 2021;10(12):3229–45. 10.1515/nanoph-2021-0209.Search in Google Scholar
[89] Kubrak T, Karakula M, Czop M, Kawczyk-Krupka A, Aebisher D. Advances in management of bladder cancer-the role of photodynamic therapy. Molecules. 2022;27(3):731. 10.3390/molecules27030731.Search in Google Scholar PubMed PubMed Central
[90] Nasseri B, Alizadeh E, Bani F, Davaran S, Akbarzadeh A, Rabiee N, et al. Nanomaterials for photothermal and photodynamic cancer therapy. Appl Phys Rev. 2022;9(1):011317. 10.1063/5.0047672.Search in Google Scholar
[91] Zou LL, Wang H, He B, Zeng LJ, Tan T, Cao HQ, et al. Current approaches of photothermal therapy in treating cancer metastasis with nanotherapeutics. Theranostics. 2016;6(6):762–72. 10.7150/thno.14988.Search in Google Scholar PubMed PubMed Central
[92] Chen JQ, Ning CY, Zhou ZN, Yu P, Zhu Y, Tan GX, et al. Nanomaterials as photothermal therapeutic agents. Prog Mater Sci. 2019;99:1–26. 10.1016/j.pmatsci.2018.07.005.Search in Google Scholar PubMed PubMed Central
[93] Han HS, Choi KY. Advances in nanomaterial-mediated photothermal cancer therapies: Toward clinical applications. Biomedicines. 2021;9(3):305. 10.3390/biomedicines9030305.Search in Google Scholar PubMed PubMed Central
[94] Liu YJ, Bhattarai P, Dai ZF, Chen XY. Photothermal therapy and photoacoustic imaging via nanotheranostics in fighting cancer. Chem Soc Rev. 2019;48(7):2053–108. 10.1039/c8cs00618k.Search in Google Scholar PubMed PubMed Central
[95] Li M, Zhao X, Dai J, Yu Z. Peptide therapeutics and assemblies for cancer immunotherapy. Sci China Mater. 2019;62(11):1759–81. 10.1007/s40843-019-9451-7.Search in Google Scholar
[96] Shrestha B, Wang LJ, Brey EM, Uribe GR, Tang L. Smart nanoparticles for chemo-based combinational therapy. Pharmaceutics. 2021;13(6):853. 10.3390/pharmaceutics13060853.Search in Google Scholar PubMed PubMed Central
[97] Chen A, Lu H, Cao R, Zhu Y, Li Y, Ge R, et al. A novel MMP-responsive nanoplatform with transformable magnetic resonance property for quantitative tumor bioimaging and synergetic chemo-photothermal therapy. Nano Today. 2022;45:101524. 10.1016/j.nantod.2022.101524.Search in Google Scholar
[98] Wang Y, Cui J, Chen J, Wan J, Liang Y, Qi M, et al. Novel bone tumor cell targeting nanosystem for chemo-photothermal therapy of malignant bone tumors. Chem Eng J. 2022;446:136905. 10.1016/j.cej.2022.136905.Search in Google Scholar
[99] Chen X, Gao H, Deng Y, Jin Q, Ji J, Ding D. Supramolecular aggregation-induced emission nanodots with programmed tumor microenvironment responsiveness for image-guided orthotopic pancreatic cancer therapy. ACS Nano. 2020;14(4):5121–34. 10.1021/acsnano.0c02197.Search in Google Scholar PubMed
[100] Shi H, Sun Y, Yan R, Liu S, Zhu L, Liu S, et al. Magnetic semiconductor Gd-doping CuS nanoparticles as activatable nanoprobes for bimodal imaging and targeted photothermal therapy of gastric tumors. Nano Lett. 2019;19(2):937–47. 10.1021/acs.nanolett.8b04179.Search in Google Scholar PubMed
[101] Yang L, Tang J, Yin H, Yang J, Xu B, Liu Y, et al. Self-assembled nanoparticles for tumor-triggered targeting dual-mode NIRF/MR imaging and photodynamic therapy applications. ACS Biomater Sci & Eng. 2022;8(2):880–92. 10.1021/acsbiomaterials.1c01418.Search in Google Scholar PubMed
[102] Liu Y, Ye Z, Yang W, Hu Y, Zhao Y, Li Z, et al. A triple enhanced permeable gold nanoraspberry designed for positive feedback interventional therapy. J Controlled Rel. 2022;345:120–37. 10.1016/j.jconrel.2022.03.010.Search in Google Scholar PubMed
[103] Sim AJ, Kaza E, Singer L, Rosenberg SA. A review of the role of MRI in diagnosis and treatment of early stage lung cancer. Clin Transl Radiat Oncol. 2020;24:16–22. 10.1016/j.ctro.2020.06.002.Search in Google Scholar PubMed PubMed Central
[104] Lu HW, Xu YJ, Qiao RR, Lu ZW, Wang P, Zhang XD, et al. A novel clustered SPIO nanoplatform with enhanced magnetic resonance T2 relaxation rate for micro-tumor detection and photothermal synergistic therapy. Nano Res. 2020;13(8):2216–25. 10.1007/s12274-020-2839-0.Search in Google Scholar
[105] Zhang WZ, Liu L, Chen HM, Hu K, Delahunty I, Gao S, et al. Surface impact on nanoparticle-based magnetic resonance imaging contrast agents. Theranostics. 2018;8(9):2521–48. 10.7150/thno.23789.Search in Google Scholar PubMed PubMed Central
© 2022 Qiuye Wang et al., published by De Gruyter
This work is licensed under the Creative Commons Attribution 4.0 International License.
Articles in the same Issue
- Research Articles
- Abnormal retention of s-triazine herbicides on porous graphitic carbon
- One-factor-at-a-time method combined with ICP-MS for determining 11 elements in soy sauce and their migration from the containing glass bottles
- Analysis of initiator content of prepreg by near-infrared spectroscopy
- Simultaneous MEKC-DAD and smart spectrophotometric assays of thiocolchicoside and etoricoxib in challenging concentration ratio mixtures
- Alternative analytical methods for ibrutinib quantification in pharmaceutical formulation: A statistical comparison
- Chemometric determination of common cold infection drugs in human urine
- An effective, novel, and cheap carbon paste electrode for naproxen estimation
- Fabrication of ultra-sensitive carbon paste electrode with nanocomposite CdS modification for electroanalysis of rafoxanide in dosage form and biological fluids
- Purification and characterisation of phytochemicals extracted from Rhizophora mucronata: Their efficacy against Pseudomonas aeruginosa infection in Catla catla
- Review Articles
- Recent applications of quantitative analytical FTIR spectroscopy in pharmaceutical, biomedical, and clinical fields: A brief review
- Review of characteristics and analytical methods for determination of indomethacin
- A review of the application of comprehensive two-dimensional gas chromatography MS-based techniques for the analysis of persistent organic pollutants and ultra-trace level of organic pollutants in environmental samples
- Enrichment and analysis of glycated proteins
- Round robin tests of secondary raw materials: A systematic review of performance parameters
- Paper-based microfluidic devices: Fabrication, detection, and significant applications in various fields
- Applications of headspace solid-phase microextraction in human biological matrix analysis
- Chemometrics and infrared spectroscopy – A winning team for the analysis of illicit drug products
- Canagliflozin: A review with specific focus on analytical methods in biological matrices and pharmaceuticals
- RNA-based isothermal amplification technology and its clinical application in pathogen infection
- Detection of diarrheal shellfish toxins
- Special Issue: Nanomaterials with mimetic enzymatic properties for label-free sensors and biosensors (Guest Editors: Gang Wei and Zhiqiang Su)
- Preparation of cuprous oxide-supported silver-modified reduced graphene oxide nanocomposites for non-enzymatic electrochemical sensor
- Recent advances in matrix metalloproteinases-responsive nanoprobes for cancer diagnosis and therapy
- A simple salicylaldehyde-bearing pyrazine as a turn-on fluorescent chemosensor for Al3+ and Zn2+ recognition and its applications
- Preparation of silver nanosheet-assembled film as a surface-enhanced Raman scattering substrate
- Synthesis of group I–III–VI semiconductor quantum dots and its application in food safety testing
Articles in the same Issue
- Research Articles
- Abnormal retention of s-triazine herbicides on porous graphitic carbon
- One-factor-at-a-time method combined with ICP-MS for determining 11 elements in soy sauce and their migration from the containing glass bottles
- Analysis of initiator content of prepreg by near-infrared spectroscopy
- Simultaneous MEKC-DAD and smart spectrophotometric assays of thiocolchicoside and etoricoxib in challenging concentration ratio mixtures
- Alternative analytical methods for ibrutinib quantification in pharmaceutical formulation: A statistical comparison
- Chemometric determination of common cold infection drugs in human urine
- An effective, novel, and cheap carbon paste electrode for naproxen estimation
- Fabrication of ultra-sensitive carbon paste electrode with nanocomposite CdS modification for electroanalysis of rafoxanide in dosage form and biological fluids
- Purification and characterisation of phytochemicals extracted from Rhizophora mucronata: Their efficacy against Pseudomonas aeruginosa infection in Catla catla
- Review Articles
- Recent applications of quantitative analytical FTIR spectroscopy in pharmaceutical, biomedical, and clinical fields: A brief review
- Review of characteristics and analytical methods for determination of indomethacin
- A review of the application of comprehensive two-dimensional gas chromatography MS-based techniques for the analysis of persistent organic pollutants and ultra-trace level of organic pollutants in environmental samples
- Enrichment and analysis of glycated proteins
- Round robin tests of secondary raw materials: A systematic review of performance parameters
- Paper-based microfluidic devices: Fabrication, detection, and significant applications in various fields
- Applications of headspace solid-phase microextraction in human biological matrix analysis
- Chemometrics and infrared spectroscopy – A winning team for the analysis of illicit drug products
- Canagliflozin: A review with specific focus on analytical methods in biological matrices and pharmaceuticals
- RNA-based isothermal amplification technology and its clinical application in pathogen infection
- Detection of diarrheal shellfish toxins
- Special Issue: Nanomaterials with mimetic enzymatic properties for label-free sensors and biosensors (Guest Editors: Gang Wei and Zhiqiang Su)
- Preparation of cuprous oxide-supported silver-modified reduced graphene oxide nanocomposites for non-enzymatic electrochemical sensor
- Recent advances in matrix metalloproteinases-responsive nanoprobes for cancer diagnosis and therapy
- A simple salicylaldehyde-bearing pyrazine as a turn-on fluorescent chemosensor for Al3+ and Zn2+ recognition and its applications
- Preparation of silver nanosheet-assembled film as a surface-enhanced Raman scattering substrate
- Synthesis of group I–III–VI semiconductor quantum dots and its application in food safety testing


