Abstract
Textural properties of the solid sorbents are critical to tuning their CO2 capture performance. In this work, we studied the effect of fiber density (in turn, pore size, distribution, and accessibility) on CO2 capture capacity and kinetics. CO2 solid sorbents were prepared by physisorption of tetraethylenepentamine (TEPA) molecules on dendritic fibrous nanosilica (DFNS) with varying fiber density. Among the various DFNS, the DFNS with moderate fiber density [DFNS-3] showed the best CO2 capture capacity under the flue gas condition. The maximum CO2 capture capacity achieved was 24.3 wt % (5.53 mmol/g) at 75 °C for DFNS-3 under humid gas conditions. Fiber density also played a role in the kinetics of CO2 capture. DFNS-1 with dense fiber density needed ∼10.4 min to reach 90 % capture capacity, while DFNS-3 (moderate fiber density) needed only 6.4 min, which further decreased to 5.9 min for DFNS-5 with lightly dense fibers. The DFNS-impregnated TEPA also showed good recyclability during 21 adsorption and desorption cycles under humid and dry conditions. The total CO2 capture capacity of DFNS-3 (14.7) in 21 cycles was 108.9 and 105.0 mmol/g under humid and dry conditions, respectively. Adsorption lifetime calculation and recyclability confirmed the fiber density-dependent CO2 capture performance.
Introduction
The CO2 content in the earth’s atmosphere is continuously increasing due to the burning of fossil fuels. CO2 accumulation into the environment is causing global warming and climate change [1, 2]. To mitigate the impact of global warming, CO2 concentrations in the environment must be reduced [3, 4]. CO2 capture, storage, and utilization are considered effective strategies to reduce CO2 emissions into the environment [5], [6], [7], [8], [9], [10], [11]. In the field of CO2 capture and storage, many technologies have been developed and amine-based technology is used by industries for the last two decades to capture CO2. Aqueous amines such as monoethanolamine (MEA), diethanolamine (DEA), and other polyamines are generally used [12], [13], [14]. But this liquid amine technology is associated with many disadvantages and limitations, such as liquid amines are not thermally stable and degrade into small toxic chemicals which are environmentally hazardous, high-energy requirements to regenerate sorbents, and corrosive nature. The best alternative to aqueous amine sorbents is the use of porous solid sorbents [12, 15], [16], [17], [18]. Several porous materials such as zeolites [19, 20], MOF [21], porous carbon [22, 23], metal oxide [18, 24], and silica were used to develop solid sorbents [25]. Solid sorbent has many advantages over liquid amine sorbent such as solid sorbent requires less energy to regenerate, is easy to handle, has good capture capacity at high temperatures, has good thermal stability even in the presence of moisture, and has good selectivity for CO2 gas over the other gas [26].
Several reports used silica as a support for the synthesis of amine-based solid sorbent for CO2 capture [27]. Silica supports have many advantages over other porous support because of their easy synthesis, chemically inert nature, high surface area, and biocompatibility. Extensive work has been reported on the silica-amine solid sorbent for CO2 capture and varieties of silica such as MCM-41, SBA-15, and DFNS are being used [28], [29], [30]. Silica-based solid amine sorbent can effectively capture CO2 gas with high capacity and fast kinetics and the presence of moisture cannot affect the working capacity [31], [32], [33]. Apart from the high surface area, accessibility of the inner surface area is more important, and pore size and its distribution have a huge impact on the diffusion of gases molecules or reactants [34], [35], [36]. Hence optimization of pore size is another important parameter of CO2 capture, separation, and catalysis [37]. Zeleňák et al. reported the effect of pore size of amine-modified ordered mesoporous silica on CO2 capture [38]. SBA-12 showed fast responses to CO2 capture in comparison to MCM-41 and SBA-15; this might be due to the three-dimensionally connected pores in SBA-12 [38]. SIFSIX-3-Zn with a pore size of 0.384 nm showed outstanding CO2 uptake and selectivity compared to other MOFs [39]. Although MOFs are highly porous and have good CO2 capture capacity; however, MOFs can only capture at high pressure and low temperature, which limits their applications for practical use.
Dendritic fibrous nanosilica (DFNS) has many advantages over MOFs, and other silica-based materials because of its fibrous morphology, three-dimensional connected channels, and open structure [30, 40], [41], [42]. A unique feature of DFNS is that its high surface area originates from its fibrous morphology and not from mesoporous tubular pores (like in SBA-15 or MCM-41) [30]. We have demonstrated the successful utilization of fibrous morphology of DFNS compared with conventional ordered mesoporous silica i.e. MCM-41 or SBA-15 for various applications [23, 29, 30, 42], [43], [44], [45], [46], [47], [48]. In our previous report, we showed DFNS-based sorbents possess several advantages over conventional silica-based sorbents, such as i) high amine loading, ii) minimum reduction in surface area after functionalization, and iii) more accessibility of amine sites resultant into enhancing CO2 capture efficiency (capture capacity, kinetics, and recyclability) [49]. In this work, we studied the role of the fiber density of DFNS in its CO2 capture performance. The DFNS-amine sorbent was synthesized according to our previous study, where we found that physisorption is the best method for the synthesis of DFNS-based sorbents, using TEPA as the amine molecules, and toluene as solvent [49, 50].
Results and discussion
Synthesis of DFNS-amine with varying fiber densities
Five types of DFNS with varying fiber density from dense to moderate to light were prepared and then evaluated the effect of fiber density on the CO2 capture capacity. These five DFNS with different surface areas, fiber density, and pore volume (Fig. 1, Table 1) were synthesized by our previously reported procedure [40]. Fig. 1 shows SEM images, particle size distribution, and corresponding TEM images of five different DFNS. From DFNS-1 to DFNS-5, the fiber density decreased, and qualitative analyses were made only based on TEM and SEM images. As the fiber density decreases, the surface area and pore volume increase until DFNS-3; however, DFNS-4 has a lower surface area compared to DFNS-3 & 5, but with almost equal pore volume (Table 1). This was attributed to the increase in the pores size distribution in the range of 5–20 nm.
![Fig. 1:
SEM images, pore size distribution, and TEM images of DFNS-1 (a, b, c, d), DFNS-2 (e, f, g, h), DFNS-3 (I, j, k, l), DFNS-4 (m, n, o, p), DFNS-5 (q, r, s, t). Some images are reproduced from our own Ref. [23].](/document/doi/10.1515/pac-2023-0103/asset/graphic/j_pac-2023-0103_fig_001.jpg)
SEM images, pore size distribution, and TEM images of DFNS-1 (a, b, c, d), DFNS-2 (e, f, g, h), DFNS-3 (I, j, k, l), DFNS-4 (m, n, o, p), DFNS-5 (q, r, s, t). Some images are reproduced from our own Ref. [23].
Nitrogen contents, and textural properties of DFNS and DFNS-TEPAads.
| Sample name | Nitrogen contents (mmol/g) | BET surface area (m2/g) | Pore volume (cm3/g) | Reduction in BET surface area (%) | Reduction in pore volume (%) | ||
|---|---|---|---|---|---|---|---|
| Before amine loading | After amine loading | Before amine loading | After amine loading | ||||
| DFNS-1 | 16.5 | 610 | 74 | 1.06 | 0.26 | 88.0 | 75.4 |
| DFNS-2 | 17.1 | 710 | 108 | 1.27 | 0.31 | 84.8 | 75.5 |
| DFNS-3 | 17.1 | 1380 | 158 | 2.22 | 0.45 | 88.6 | 79.7 |
| DFNS-4 | 17.4 | 980 | 247 | 2.07 | 0.71 | 74.8 | 65.7 |
| DFNS-5 | 17.1 | 1070 | 162 | 1.81 | 0.40 | 85.0 | 78.0 |
These DFNS were then loaded with tetraethylenepentamine (TEPA) using the physisorption method [49]. The textural properties of five DFNS and their corresponding amine sorbents are summarized in Table 1. There was around 84 % decreased in the surface area and 75 % decrease in the pore volume of all DFNS after amine loading. The textural properties, such as N2 sorption isotherm and BJH pore sizes distribution before and after amine loading, are shown in Fig. S1. The DFNS before and after amine loading showed a type 4 isotherm (Fig. S1). The BJH desorption pore size distribution of DFNS (Fig. S1) showed two wide pore size distributions, narrow pore size distribution around 2.5–5 nm, and another wide pore distribution around 5–25 nm. As the fiber density decreased, 0.5–5 nm pores reduced and pores around 5–25 increased (Fig. S1). After amine loading, pores were still visible; however, with reduced numbers (Fig. S1), all five DFNS showed around 17 mmol/g N contents (Table 1). The stability and morphology after amine loading were studied using SEM (Fig. S2) and found that the morphology of DFNS remains unchanged after amine loading.
Role of DFNS fiber density on CO2 adsorption and desorption
DFNS-Amine sorbents were evaluated based on the effect of amine loading on textural properties, CO2 capture capacity, kinetics, the effect of temperature on CO2 capture capacity, and recyclability under dry and humid conditions.
All DFNS-TEPAads sorbents were analyzed for CO2 capture capacity at 75 °C according to the program given in Fig. 2 using TGA. The sorbents were first activated by removing the adsorbed gases and moisture at 100 °C under the flow of N2 gas (150 mL/min). After 60 min at 100 °C, the sample temperature was stabilized at 75 °C for 30 min in N2. After that, the gas flow was changed to 15 % CO2 in N2 (150 mL/min) at 75 °C, and flow was maintained for 60 min. CO2 capture capacity with time was determined by weight gain in TGA. The desorption of gas was carried out at 110 °C under the flow of 150 mL N2 for 15 min. Three subsequent adsorption cycles for each sorbent were carried out and Table 2 depicts the average values of CO2 capture from three adsorption-desorption cycles. Change in CO2 capture capacities with change in fiber density of DFNS clearly indicated the key role of fiber density on CO2 capture performance. As the fiber density decreased from DFNS-1 to DFNS-5, CO2 capture capacity increased. The surface area and pore volume of DFNS-5 lie in between the DFNS-3 and DFNS-4 the CO2 capture capacity of DFNS-4 was found to be in good agreement with fiber density. This was due to more space between the fibers (pore volume) available to accommodate more CO2 molecules which increased CO2 capture capacity (Table 2, Figs. S3 and S4). Also, more amines were available for CO2 capture due to an increase in their accessibility due to the separation between fiber at less dense DFNS. CO2 capture is diffusion limited, and as the diffusion of CO2 molecules increases in less dense DFNS, it directly results in more CO2 capture capacity.

The schematic of the CO2 adsorption-desorption using TGA.
CO2 capture performance of DFNS-TEPAads sorbents at 75 °C using 15 % CO2.
| Sample name | Nitrogen contents (mmol/g) | CO2 capture capacity (wt %) | CO2 capture capacity (mmol/g) | Amine efficiency (CO2 mmol per mmol of N) |
|---|---|---|---|---|
| DFNS-1 | 16.5 | 14.2 | 3.22 | 0.19 |
| DFNS-2 | 17.1 | 18.9 | 4.29 | 0.25 |
| DFNS-3 | 17.1 | 19.5 | 4.43 | 0.26 |
| DFNS-4 | 17.4 | 20.4 | 4.63 | 0.27 |
| DFNS-5 | 17.1 | 18.1 | 4.11 | 0.24 |
Role DFNS fiber density vs. surface area and pore volume in CO2 capture performance
The difference in CO2 capture capacity could be due to the difference in surface area or pore volume and not fiber density (Fig. 3). To study this aspect, DFNS-3, and DFNS-4 were further studied to understand the precise effect of fiber density on CO2 capture capacity. Both DFNS-3 and DFNS-4 have similar N contents i.e., 17.1 mmol/g and there was around similar reduction in surface area and pore volume, i.e., both having similar surface area and pore volume. Hence these two sorbents are ideal for the understanding role of fiber density. To optimize the TEPA loading on the DFNS-3 and DFNS-4, a series of amine-loaded sorbents were prepared and evaluated for CO2 capture performance (Table 3). The CO2 capture capacity of the DFNS-3 and DFNS-4 series first increased with an increase in TEPA loading and then decreased after the optimal concentration of amine loading. In the case of the DFNS-3 series, DFNS-3 (14.7 mmol/g) was found to be better than the 17.1 and 12.4 mmol/g N contents, respectively (Table 3 and Fig. S5). Whereas, in the DFNS-4 series, the DFNS-4 (17.4 mmol/g) was found best sorbent. DFNS-3 (14.7) even with lower N content was found better in terms of CO2 capture capacity than DFNS-4 (17.4) with higher N content. These results clearly indicated that a combination of amine loading with optimized fiber density is key to maximizing the adsorption capacity of DFNS-based solid amine sorbents.

Schematic diagram of DFNS with different fiber densities and effect on CO2 capture capacity.
Amine loading and CO2 capture of DFNS-3 and DFNS-4 at 75 °C using 15 % CO2.
| Sample name | Nitrogen contents (mmol/g) | CO2 capture capacity (wt %) | CO2 capture capacity (mmol/g) | Amine efficiency (mmol of CO2/mmol of N) | BET surface area (m2/g) | Pore volume (cm3/g) |
|---|---|---|---|---|---|---|
| DFNS-3 | 17.1 | 19.5 | 4.43 | 0.26 | 158 | 0.45 |
| 14.7 | 22.1 | 5.0 | 0.34 | 119 | 0.36 | |
| 14.1 | 22.1 | 5.0 | 0.35 | 107 | 0.37 | |
| 12.4 | 9.0 | 2.0 | 0.16 | 232 | 0.65 | |
| DFNS-4 | 18.9 | 19.2 | 4.36 | 0.23 | 143 | 0.45 |
| 18.2 | 19.4 | 4.40 | 0.24 | 186 | 0.58 | |
| 17.4 | 20.4 | 4.63 | 0.27 | 247 | 0.71 | |
| 14.9 | 14.9 | 3.38 | 0.22 | 292 | 0.88 | |
| 13.2 | 14.8 | 3.36 | 0.22 | 315 | 0.84 |
Why DFNS-3 (14.7) showed better CO 2 capture capacity than DFNS-4 (17.4) even after low N contents? The reason is the better pore volume of DFNS-3; DFNS-3 with higher pore volume can accommodate more CO2 molecules. Also, fiber density decides the diffusion time of CO2 molecules within pores. To study this aspect, we measured the capture kinetics of all DFNS-TEPA sorbents (Fig. 4a). The difference in time required to achieve 90 % capture capacity of all five samples suggests the clear effect of fiber density on CO2 capture capacity. From DFNS-1 to DFNS-5, the time to achieve 90 % CO2 capture capacity decreased from DFNS-1 i.e., 10.38 min to DFNS-2 i.e., 6.57 min. In the case of DFNS-3 to DFNS-4, capture kinetics was found to be similar ∼6.4 min, while it decreased for DFNS-5 to 5.49 min. When we compared the two best sorbents from both series, DFNS-3 (14.7) and DFNS-4 (17.4) (Table 3, Figs. 4b and S6), even after low N contents and more CO2 capture capacity in the case of DFNS-3 (14.7), it required nearly same time to achieve 90 % of the maximum CO2 capture capacity, that of DFNS-4 (17.4). These results further confirm the key role of fiber density in controlling the diffusion of CO2 molecules, as well as the accessibility of amines and hence in turn, CO2 capture kinetics (Fig. 4b).
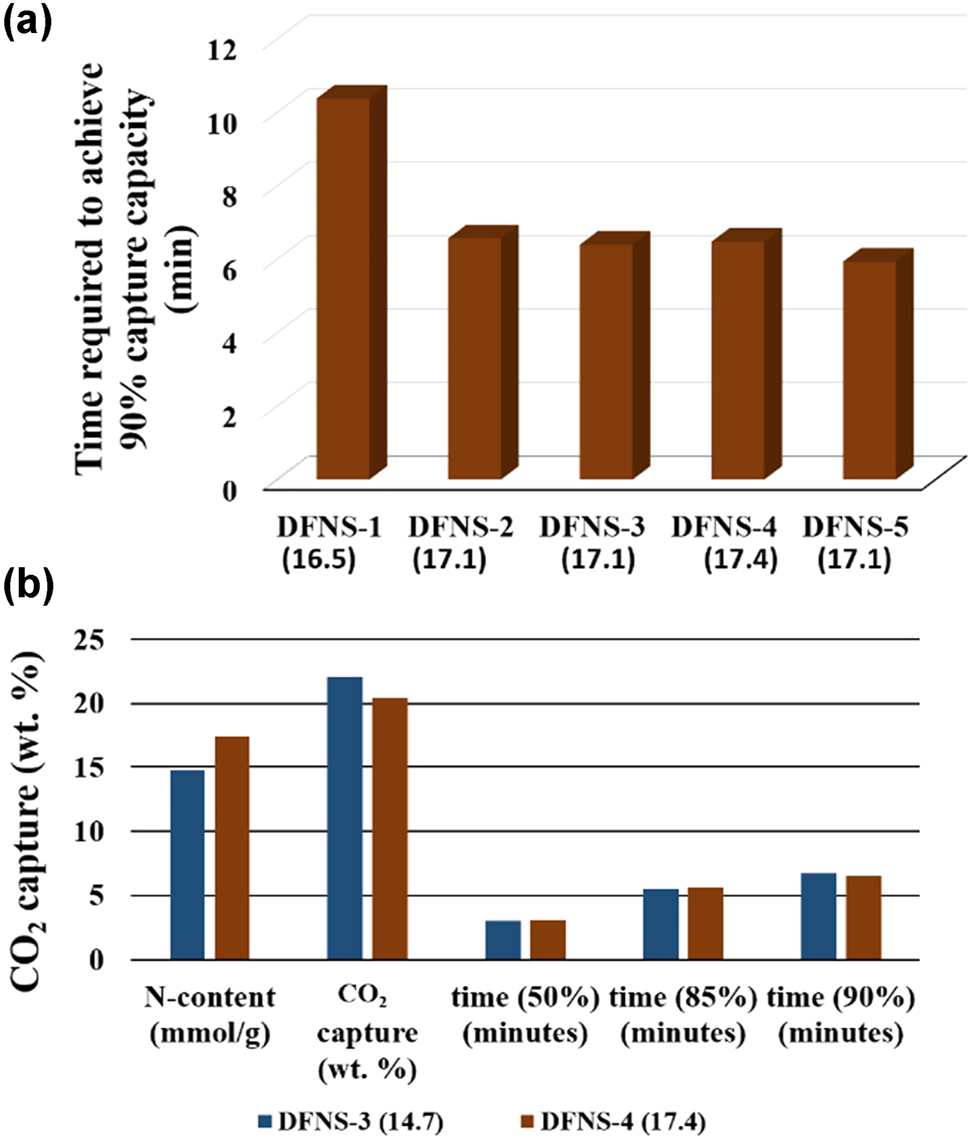
CO2 capture performance. (a) Time required to achieve 90% of maximum capture capacity, (b) comparison between the two best sorbents DFNS-3 (14.7) and DFNS-4 (17.4).
Effect of adsorption temperature on CO2 capture capacity
The adsorption temperature can affect both chemical adsorption and diffusion of CO2 molecules. Hence, capture capacity and rate of adsorption can be affected by adsorption temperature. We evaluated the two best sorbents DFNS-3 (14.7), and DFNS-4 (17.4) for their CO2 capture performance at four different temperatures. Both the sorbents showed an increase in CO2 capture capacity with an increase in adsorption temperature and reached maximum CO2 capture capacity at 75 °C (Figs. 5 and S7). The increase in CO2 capture capacity can be attributed to increased temperature as it facilitated the chemical reaction between the amine and CO2 molecules due to the exothermic nature of the reaction and as well as help in the diffusion of CO2 molecule [13, 51]. With A further increase in adsorption temperature to 100 °C, the CO2 capture capacity was decreased. The CO2 adsorption and desorption processes are reversible at all temperatures. At temperatures up to 75 °C, the forward process is more favorable while at high temperatures (100 °C) the reverse reaction is more favorable, resulting into increase desorption of the adsorbed CO2 [52].

CO2 capture capacity at four different temperatures (a) DFNS-3 (14.7); (b) DFNS-4 (17.4). Effect of temperature on CO2 capture capacity and adsorption lifetime (c) adsorption time of DFNS-3 (14.7), (d) adsorption time of DFNS-4 (17.4).
Kinetics of CO2 adsorptions were still fast at all four temperatures and sorbents saturated within 5 min (Figs. 5, S8 and S9). However, as the adsorption temperature increased, the curve became more rounded around 6 min (Fig. 5) and the saturation time increased with an increase in the temperature. The increase of diffusion with an increase in the adsorption temperature can be seen by the adsorption kinetics for different adsorption capacities. For particular sorbents to calculate the time required to reach a particular adsorption capacity, we fixed two values 10.0 and 8.0 wt % to get an idea about how much time a sorbent took to reach at a fixed value at a controlled temperature. From these data, we observed that total time increased slightly as temperature increased from 30 to 50 °C but was constant for 75 °C and increased again at 100 °C (Fig. 5c and d). This is because at 100 °C, desorption of CO2 molecules also started, and sorbents took more time to reach the maximum CO2 capture capacity at 100 °C temperature.
DFNS-TEPA sorbents stability and recyclability
We also studied how long a sorbent DFNS-3 (14.7) can hold captured CO2 at different temperatures; these experiments were carried out to find out the best storage/desorption temperature. We studied CO2-saturated sample stability at three temperatures 30, 50, and 75 °C for 15 h without a flow of any gas. For these experiments, first, we captured the CO2 at 75 °C and after 1 h exposure to CO2, we hold samples at different temperatures (30, 50, and 75 °C) to check the thermal stability of amine-CO2 adduct for 24 h. We maintained the temperature for 24 h without a flow of any gas. As the holding temperature increased, desorption kinetics increased (Fig. 6). At 30 °C, the loss of CO2 was around 3.8 % in 23.4 h; however, as the temperature increased from 30 to 100 °C the rate of CO2 desorption increased. It was 13.1 % in 22.3 h at 50 °C, 100 % loss in 4.8 h at 75 °C, and 100 % in 3.3 h at 100 °C.

Desorption study of sorbent DFNS-3 (14.7) at different temperatures (a) 30 °C, (b) 50 °C, (c) 75 °C, and (d) 100 °C.
Good sorbents must show stability under moist conditions. For this study, 21 CO2 adsorption-desorption cycles were conducted on DFNS-3 (14.7) and DFNS-4 (17.4) under dry and humid conditions (Fig. 7). For each step, the adsorption of CO2 gas at 75 °C followed by the desorption of CO2 gas at 110 °C under N2 flow. During the recyclability study, both sorbents showed an increase in CO2 capture capacity after 1st cycle. DFNS-3 (14.7) showed a CO2 capture capacity increase from 18.8 to 23.4 wt % from the first to the second cycle, which was maintained up to 10th cycle under dry conditions. After 10th cycle of CO2 capture, there was a slow reduction in CO2 capture capacity, and in the 21st cycle, the DFNS-3 (14.7) still showed better CO2 capture capacity than its first cycle. DFNS-4 (17.4) (Fig. 7a) also showed similar behavior in the recyclability study.

Sorbent recyclability under (a) dry, (b) humid conditions. (c) Total CO2 capture capacity in 21 cycles under dry and humid conditions at 75 °C using 15 % CO2 in N2.
However, DFNS-4 (17.4) showed more reduction in CO2 capture capacity compared to DFNS-3 (14.7) in 21 cycles. Nevertheless, after 21 cycles under dry conditions, DFNS-4 (17.4) still showed CO2 capture capacity similar to DFNS-3 (14.7). There is always a loss of amines during every adsorption-desorption cycle, but even with a decrease in N contents, the sorbents still showed similar CO2 capture capacity to its first cycle. This indicates excellent amine accessibility and diffusion of CO2 molecules in the DFNS-TEPA sorbents. With the number of adsorption-desorption cycles, amine accessibility increase, and diffusion of CO2 molecules also increase, which lead to maintaining capture capacity for a long time. These results were also indicating about the role of fiber density in the accessibility of amines and efficient diffusion of CO2 molecules.
The recyclability of sorbents under humid conditions is the real parameter to mimic the flue gas condition. The effect of humidity on CO2 capture was studied using the pre-humidified CO2 gas. The humid CO2 was generated at room temperature by passing CO2 gas through the two water bubblers. There was approximately 1 wt % increase in CO2 capture capacity. Notably, under humid conditions, DFNS-3 (14.7) showed much better recyclability than DFNS-4 (17.4) (Fig. 7b). For a comparison under humid conditions, DFNS-3 (14.7) has 18.6 wt % in the 1st cycle and 20.8 in 21st cycle, indicating no decrease in CO2 capture capacity in 21 cycles. DFNS-4 (17.4) showed a decrease in CO2 capture capacity, which was 17.4 in 1st cycle and 15.9 wt % in the 21st cycle, a 8.6 % decrease. We observed DFNS-3 (14.7) was more stable and recyclable as compared to DFNS-4 (17.4). From recyclability data, it was revealed that fiber density not only affects the CO2 capture capacity but also affects the stability of sorbents during the adsorption-desorption steps.
We also calculated the total CO2 capture capacity in 21 cycles under dry and humid conditions. We found that DFNS-3 (14.7) captured 108.5 and 105 mmol/g (Fig. 7c, Tables S1 and S2) total CO2 in 21 cycles under humid and dry conditions, respectively. DFNS-4 (17.4) captured 90.6 and 88.2 mmol/g (Fig. 7c, Tables S1 and S2) of total CO2 under dry and humid conditions, respectively. Their result further confirmed the fiber density effect of DFNS on CO2 capture capacity.
After the recyclability study, both sorbents were then re-characterized to estimate amine loss after 21 cycles during humid and dry conditions (Table 4 and S10). DFNS-3 (14.7) showed 4 mmol/g nitrogen loss in 21 cycles, whereas DFNS-4 (17.4) showed 3 mmol/g N loss. This further indicates the role of fiber density in stabilizing the amine molecules. The selectivity of the sorbents toward the different gases is also a very important parameter in deciding the industrial application of sorbents for CO2 capture. The selectivity study was carried out under similar conditions of CO2 capture in TGA. Both sorbents showed 100 % selectivity for CO2 over N2 and O2 (Fig. S11). Overall comparisons are summarized in Table 5.
Characterization of DFNS-3 (14.7) and DFNS-4 (17.4) after 21 cycles of adsorption-desorption.
| Sorbents | N-contents (mmol/g) | BET surface area (m2/g) | Pore volume (cm3/g) | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Before 21 cycles | After 21 cycles | Before 21 cycles | After 21 cycles | Before 21 cycles | After 21 cycles | ||||
| Under dry condition | Under humid condition | Under dry condition | Under humid condition | Under dry condition | Under humid condition | ||||
| DFNS-3 (14.7) | 14.7 | 9 | 9.7 | 119 | 295 | 161 | 0.36 | 0.79 | 0.52 |
| DFNS-4 (17.4) | 17.4 | 14.4 | 12.7 | 247 | 257 | 208 | 0.71 | 0.77 | 0.66 |
Comparison between DFNS-3 (14.7) and DFNS-4 (17.4) sorbents (CO2 capture at 75 °C using 15 % CO2 in N2).
| S. N. | Properties | DFNS-3 (14.7) | DFNS-4 (17.4) |
|---|---|---|---|
| 1 | Nitrogen contents (mmol/g) | 14.7 | 17.4 |
| 2 | Reduction in BET surface area after amine loading | 1386–119 m2/g Absolute loss −1267 m2/g (91.4 % loss) |
982–247 m2/g Absolute loss −735 m2/g (74.8 % loss) |
| 3 | Reduction in volume after amine loading | 2.22–0.36 cm3 g−1 (83.7 loss) | 2.07–0.716 cm3 g−1 (65.7 loss) |
| 4 | CO2 capture capacity under dry conditions (best value during recyclability) | 5.31 mmol/g | 4.75 mmol/g |
| 5 | CO2 capture capacity under humid conditions (best value during recyclability value) | 5.52 mmol/g | 4.75 mmol/g |
| 6 | Reduction in CO2 capture capacity in 21 cycles under dry condition | 1st cycle-18.8 wt % 21st cycle-20.9 wt % Ideally no loss |
1st cycle-18 wt % 21st cycle-17.2 wt % 4.4 % loss |
| 7 | Reduction in CO2 capture capacity in 21 cycles under humid conditions | 1st cycle-18.6 wt % 21st cycle-20.8 wt % Ideally no loss |
1st cycle-17.4 wt % 21st cycle-15.9 wt % 8.6 % loss |
| 8 | Total CO2 capture in 21 cycles under dry condition | 105 mmol/g | 90.6 mmol/g |
| 9 | Total CO2 capture in 21 cycles under humid condition | 108 mmol/g | 88.2 mmol/g |
| 10 | BET surface area after 21 cycles under dry condition | 295 m2/g | 257 m2/g |
| 11 | Pore volume after 21 cycles under humid condition | 161 m2/g | 208 m2/g |
| 12 | Total loss of N-contents in 21 cycles under dry condition | 9 mmol/g | 14.4 mmol/g |
| 13 | Total loss of N-contents in 21 cycles under humid condition | 9.7 mmol/g | 12.7 mmol/g |
| 14 | Amine efficiency (mmol of CO2/mmol of N) in dry condition | 0.36 | 0.27 |
| 15 | Amine efficiency (mmol of CO2/mmol of N) in humid condition | 0.37 | 0.27 |
Conclusions
Fiber density-controlled solid amine sorbents were prepared via the physisorption of TEPA on dendritic fibrous nanosilica with varying fiber density. We observed the unique role of fiber density on CO2 capture performance of these DFNS-based sorbents. All three aspects, capture capacity, kinetics, and stability, were tuned by tuning the fiber density of the sorbent. The CO2 capture capacity of 24.3 wt % (5.53 mmol/g) was achieved under humid gas conditions by simply tuning fiber density. Fiber density also impacted the kinetics of CO2 capture, with dense fiber density needing ∼10.4 min to reach 90 % capture capacity, while moderate fiber density needed only 6.4 min and only 5.9 min for lightly dense fibers. We found that DFNS fiber density dictated the time required to achieve 90 % of the total CO2 capture capacity. DFNS-1 took 10.38 min to reach 90 %, while DFNS-3 reached the same value in 6.4 min, it is further decreased to 5.94 min for DFNS-5. Varying distances between the fibers allow CO2 molecules to diffuse faster at low fiber density conditions. Fiber density also played a role in the stability of sorbents, and they showed good recyclability during 21 adsorption and desorption cycles under humid and dry conditions. The total capture capacity in 21 cycles was found to be 105 and 108 mmol/g for DFNS-3 (14.7) under dry and humid conditions, respectively. The better performance of DFNS-3 was due to the better diffusion of CO2 molecules into the fibers of DFNS and the accessibility of amines. The better performance of DFNS is invariably due to the unique fibrous morphology, highly accessible surface area, and pore volume.
Article note:
A collection of invited papers by the winners of the 2020 and 2022 IUPAC ChemRAWN VII Prize for Green Chemistry.
Acknowledgments
We acknowledge the SEM facility of TIFR, Mumbai. We sincerely thank Mr. L Borde and Ms. B Chalke’s assisting with EM measurements.
References
[1] S. Solomon, G.-K. Plattner, R. Knutti, P. Friedlingstein. Proc. Natl. Acad. Sci. U. S. A. 106, 1704 (2009), https://doi.org/10.1073/pnas.0812721106.Suche in Google Scholar PubMed PubMed Central
[2] D. A. Lashof, D. R. Ahuja. Nature 344, 529 (1990), https://doi.org/10.1038/344529a0.Suche in Google Scholar
[3] G. Iyer, Y. Ou, J. Edmonds, A. A. Fawcett, N. Hultman, J. McFarland, J. Fuhrman, S. Waldhoff, H. McJeon. Nat. Clim. Change 12, 1092 (2022), https://doi.org/10.1038/s41558-022-01517-z.Suche in Google Scholar
[4] P. R. Liu, A. E. Raftery. Commun. Earth Environ. 2, 29 (2021), https://doi.org/10.1038/s43247-021-00097-8.Suche in Google Scholar PubMed PubMed Central
[5] N. Mac Dowell, P. S. Fennell, N. Shah, G. C. Maitland. Nat. Clim. Change 7, 243 (2017), https://doi.org/10.1038/nclimate3231.Suche in Google Scholar
[6] H. Chalmers. Nat. Clim. Change 9, 348 (2019), https://doi.org/10.1038/s41558-019-0462-4.Suche in Google Scholar
[7] L. Whitmarsh, D. Xenias, C. R. Jones. Palgrave Commun. 5, 17 (2019), https://doi.org/10.1057/s41599-019-0217-x.Suche in Google Scholar
[8] H. Chand, P. Choudhary, A. Kumar, A. Kumar, V. Krishnan. J. CO2 Util. 51, 101646 (2021), https://doi.org/10.1016/j.jcou.2021.101646.Suche in Google Scholar
[9] A. Kumar, A. Kumar, V. Krishnan. ACS Catal. 10, 10253 (2020), https://doi.org/10.1021/acscatal.0c02947.Suche in Google Scholar
[10] P. Gabrielli, M. Gazzani, M. Mazzotti. Ind. Eng. Chem. Res. 59, 7033 (2020), https://doi.org/10.1021/acs.iecr.9b06579.Suche in Google Scholar
[11] C. Hepburn, E. Adlen, J. Beddington, E. A. Carter, S. Fuss, N. Mac Dowell, J. C. Minx, P. Smith, C. K. Williams. Nature 575, 87 (2019), https://doi.org/10.1038/s41586-019-1681-6.Suche in Google Scholar PubMed
[12] A. Samanta, A. Zhao, G. K. H. Shimizu, P. Sarkar, R. Gupta. Ind. Eng. Chem. Res. 51, 1438 (2012), https://doi.org/10.1021/ie200686q.Suche in Google Scholar
[13] M. T. Dunstan, F. Donat, A. H. Bork, C. P. Grey, C. R. Müller. Chem. Rev. 121, 12681 (2021), https://doi.org/10.1021/acs.chemrev.1c00100.Suche in Google Scholar PubMed
[14] C. Halliday, T. A. Hatton. Ind. Eng. Chem. Res. 60, 9313 (2021), https://doi.org/10.1021/acs.iecr.1c00597.Suche in Google Scholar
[15] M. Khraisheh, F. Almomani, G. Walker. Sci. Rep. 10, 269 (2020), https://doi.org/10.1038/s41598-019-57151-x.Suche in Google Scholar PubMed PubMed Central
[16] J. J. Vericella, S. E. Baker, J. K. Stolaroff, E. B. Duoss, J. O. Hardin, J. Lewicki, E. Glogowski, W. C. Floyd, C. A. Valdez, W. L. Smith, J. H. Satcher, W. L. Bourcier, C. M. Spadaccini, J. A. Lewis, R. D. Aines. Nat. Commun. 6, 6124 (2015), https://doi.org/10.1038/ncomms7124.Suche in Google Scholar PubMed
[17] S. Lee, T. P. Filburn, M. Gray, J.-W. Park, H.-J. Song. Ind. Eng. Chem. Res. 47, 7419 (2008), https://doi.org/10.1021/ie8006984.Suche in Google Scholar
[18] R. Chang, X. Wu, O. Cheung, W. Liu. J. Mater. Chem. A 10, 1682 (2022), https://doi.org/10.1039/d1ta07697c.Suche in Google Scholar
[19] P. Murge, S. Dinda, S. Roy. Langmuir 35, 14751 (2019), https://doi.org/10.1021/acs.langmuir.9b02259.Suche in Google Scholar PubMed
[20] N. Czuma, I. Casanova, P. Baran, J. Szczurowski, K. Zarębska. Sci. Rep. 10, 1825 (2020), https://doi.org/10.1038/s41598-020-58591-6.Suche in Google Scholar PubMed PubMed Central
[21] Z. Hu, Y. Wang, B. B. Shah, D. Zhao. Adv. Sustain. Syst. 3, 1800080 (2019), https://doi.org/10.1002/adsu.201800080.Suche in Google Scholar
[22] B. Zhao, M. Borghei, T. Zou, L. Wang, L.-S. Johansson, J. Majoinen, M. H. Sipponen, M. Österberg, B. D. Mattos, O. J. Rojas. ACS Nano 15, 6774 (2021), https://doi.org/10.1021/acsnano.0c10307.Suche in Google Scholar PubMed PubMed Central
[23] B. Singh, A. Maity, V. Polshettiwar. ChemistrySelect 3, 10684 (2018), https://doi.org/10.1002/slct.201802341.Suche in Google Scholar
[24] G. A. Mutch, S. Shulda, A. J. McCue, M. J. Menart, C. V. Ciobanu, C. Ngo, J. A. Anderson, R. M. Richards, D. Vega-Maza. J. Am. Chem. Soc. 140, 4736 (2018), https://doi.org/10.1021/jacs.8b01845.Suche in Google Scholar PubMed
[25] E. E. Ünveren, B. Ö. Monkul, Ş. Sarıoğlan, N. Karademir, E. Alper. Petroleum 3, 37 (2017), https://doi.org/10.1016/j.petlm.2016.11.001.Suche in Google Scholar
[26] J. Wang, L. Huang, R. Yang, Z. Zhang, J. Wu, Y. Gao, Q. Wang, D. O’Hare, Z. Zhong. Energy Environ. Sci. 7, 3478 (2014), https://doi.org/10.1039/c4ee01647e.Suche in Google Scholar
[27] Y. Fan, X. Jia. Energy Fuels 36, 1252 (2022), https://doi.org/10.1021/acs.energyfuels.1c03788.Suche in Google Scholar
[28] B. Singh, J. Na, M. Konarova, T. Wakihara, Y. Yamauchi, C. Salomon, M. B. Gawande. Bull. Chem. Soc. Jpn. 93, 1459 (2020), https://doi.org/10.1246/bcsj.20200136.Suche in Google Scholar
[29] A. Maity, V. Polshettiwar. ChemSusChem 10, 3866 (2017), https://doi.org/10.1002/cssc.201701076.Suche in Google Scholar PubMed PubMed Central
[30] V. Polshettiwar. Acc. Chem. Res. 55, 1395 (2022), https://doi.org/10.1021/acs.accounts.2c00031.Suche in Google Scholar PubMed
[31] J. Wu, X. Zhu, F. Yang, T. Ge, R. Wang. J. Chem. Eng. 425, 131409 (2021), https://doi.org/10.1016/j.cej.2021.131409.Suche in Google Scholar
[32] A. Cherevotan, J. Raj, S. C. Peter. J. Mater. Chem. A 9, 27271 (2021), https://doi.org/10.1039/d1ta05961k.Suche in Google Scholar
[33] L. Lin, Y. Meng, T. Ju, S. Han, F. Meng, J. Li, Y. Du, M. Song, T. Lan, J. Jiang. J. Environ. Manag. 325, 116438 (2023), https://doi.org/10.1016/j.jenvman.2022.116438.Suche in Google Scholar PubMed
[34] A. C. Forse, K. A. Colwell, M. I. Gonzalez, S. Benders, R. M. Torres-Gavosto, B. Blümich, J. A. Reimer, J. R. Long. Chem. Mater. 32, 3570 (2020), https://doi.org/10.1021/acs.chemmater.0c00745.Suche in Google Scholar
[35] T. Shi, Y. Zheng, T. Wang, P. Li, Y. Wang, D. Yao. ChemPhysChem 19, 130 (2018), https://doi.org/10.1002/cphc.201700842.Suche in Google Scholar PubMed
[36] L. Estevez, D. Barpaga, J. Zheng, S. Sabale, R. L. Patel, J.-G. Zhang, B. P. McGrail, R. K. Motkuri. Ind. Eng. Chem. Res. 57, 1262 (2018), https://doi.org/10.1021/acs.iecr.7b03879.Suche in Google Scholar
[37] M. Sevilla, A. S. M. Al-Jumialy, A. B. Fuertes, R. Mokaya. ACS Appl. Mater. Interfaces 10, 1623 (2018), https://doi.org/10.1021/acsami.7b10433.Suche in Google Scholar PubMed
[38] V. Zeleňák, M. Badaničová, D. Halamová, J. Čejka, A. Zukal, N. Murafa, G. Goerigk. J. Chem. Eng. 144, 336 (2008), https://doi.org/10.1016/j.cej.2008.07.025.Suche in Google Scholar
[39] O. Shekhah, Y. Belmabkhout, Z. Chen, V. Guillerm, A. Cairns, K. Adil, M. Eddaoudi. Nat. Commun. 5, 4228 (2014), https://doi.org/10.1038/ncomms5228.Suche in Google Scholar PubMed PubMed Central
[40] N. Bayal, B. Singh, R. Singh, V. Polshettiwar. Sci. Rep. 6, 24888 (2016), https://doi.org/10.1038/srep24888.Suche in Google Scholar PubMed PubMed Central
[41] V. Polshettiwar, D. Cha, X. Zhang, J. M. Basset. Angew. Chem. Int. Ed. 49, 9652 (2010), https://doi.org/10.1002/anie.201003451.Suche in Google Scholar PubMed
[42] R. Verma, R. Belgamwar, P. Chatterjee, R. Bericat-Vadell, J. Sa, V. Polshettiwar. ACS Nano 17, 4526 (2023), https://doi.org/10.1021/acsnano.2c10470.Suche in Google Scholar PubMed
[43] A. S. Lilly Thankamony, C. Lion, F. Pourpoint, B. Singh, A. J. Perez Linde, D. Carnevale, G. Bodenhausen, H. Vezin, O. Lafon, V. Polshettiwar. Angew. Chem. Int. Ed. 54, 2190 (2015), https://doi.org/10.1002/anie.201406463.Suche in Google Scholar PubMed
[44]. U. Patil, A. Fihri, A.-H. Emwas, V. Polshettiwar. Chem. Sci. 3, 2224 (2012), https://doi.org/10.1039/c2sc20356a.Suche in Google Scholar
[45] M. Bouhrara, C. Ranga, A. Fihri, R. R. Shaikh, P. Sarawade, A.-H. Emwas, M. N. Hedhili, V. Polshettiwar. ACS Sustain. Chem. Eng. 1, 1192 (2013), https://doi.org/10.1021/sc400126h.Suche in Google Scholar
[46] A. Maity, R. Belgamwar, V. Polshettiwar. Nat. Protoc. 14, 2177 (2019), https://doi.org/10.1038/s41596-019-0177-z.Suche in Google Scholar PubMed
[47] K. Mishra Amit, R. Belgamwar, R. Jana, A. Datta, V. Polshettiwar. Proc. Natl. Acad. Sci. U. S. A. 117, 6383 (2020), https://doi.org/10.1073/pnas.1917237117.Suche in Google Scholar PubMed PubMed Central
[48] A. Maity, S. Chaudhari, J. J. Titman, V. Polshettiwar. Nat. Commun. 11, 3828 (2020), https://doi.org/10.1038/s41467-020-17711-6.Suche in Google Scholar PubMed PubMed Central
[49] B. Singh, V. Polshettiwar. J. Mater. Chem. A 4, 7005 (2016), https://doi.org/10.1039/c6ta01348a.Suche in Google Scholar
[50] B. Singh, V. Polshettiwar. Nanoscale 11, 5365 (2019), https://doi.org/10.1039/c8nr10119a.Suche in Google Scholar PubMed
[51] M. S. Khosrowshahi, M. A. Abdol, H. Mashhadimoslem, E. Khakpour, H. B. M. Emrooz, S. Sadeghzadeh, A. Ghaemi. Sci. Rep. 12, 8917 (2022), https://doi.org/10.1038/s41598-022-12596-5.Suche in Google Scholar PubMed PubMed Central
[52] E. R. Monazam, L. J. Shadle, R. Siriwardane. Ind. Eng. Chem. Res. 50, 10989 (2011), https://doi.org/10.1021/ie201214q.Suche in Google Scholar
Supplementary Material
This article contains supplementary material (https://doi.org/10.1515/pac-2023-0103).
© 2023 IUPAC & De Gruyter. This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License. For more information, please visit: http://creativecommons.org/licenses/by-nc-nd/4.0/
Artikel in diesem Heft
- Frontmatter
- In this issue
- Editorial
- Preface for special issue of ICPOC-25 in Hiroshima
- Conference papers
- Control of chirality inversion kinetics of triple-helical metallocryptands
- Cooperativity in molecular recognition of feet-to-feet-connected biscavitands
- Bis-periazulene: remaining non-alternant isomer of pyrene
- Closed-shell and open-shell dual nature of singlet diradical compounds
- Anticancer activity and DNA interaction of bis(pyridyl)allene-derived metal complexes
- Reactivity of electrophilic cyclopropanes
- m-Quinodimethane-based fused-ring triplet hydrocarbons
- White light emission from an upconverted emission based on triplet-triplet annihilation with rose bengal as the sensitizer
- Fluorosumanenes as building blocks for organic crystalline dielectrics
- Recent advances in developing tetrathiafulvalene analogs of electrode materials: discovery of an in-cell polymerization technique
- The current landscape of author guidelines in chemistry through the lens of research data sharing
- Role of fiber density of amine functionalized dendritic fibrous nanosilica on CO2 capture capacity and kinetics
Artikel in diesem Heft
- Frontmatter
- In this issue
- Editorial
- Preface for special issue of ICPOC-25 in Hiroshima
- Conference papers
- Control of chirality inversion kinetics of triple-helical metallocryptands
- Cooperativity in molecular recognition of feet-to-feet-connected biscavitands
- Bis-periazulene: remaining non-alternant isomer of pyrene
- Closed-shell and open-shell dual nature of singlet diradical compounds
- Anticancer activity and DNA interaction of bis(pyridyl)allene-derived metal complexes
- Reactivity of electrophilic cyclopropanes
- m-Quinodimethane-based fused-ring triplet hydrocarbons
- White light emission from an upconverted emission based on triplet-triplet annihilation with rose bengal as the sensitizer
- Fluorosumanenes as building blocks for organic crystalline dielectrics
- Recent advances in developing tetrathiafulvalene analogs of electrode materials: discovery of an in-cell polymerization technique
- The current landscape of author guidelines in chemistry through the lens of research data sharing
- Role of fiber density of amine functionalized dendritic fibrous nanosilica on CO2 capture capacity and kinetics

