Abstract
Introduction
Neural tube defects (NTDs) are severe congenital anomalies arising from incomplete closure of the neural tube during early embryogenesis. Among cranial NTDs, exencephaly, acrania, and anencephaly represent a progressive developmental continuum, leading to major cerebral destruction and absence of cranial structures.
Content
This narrative integrative review synthesizes embryological mechanisms, historical documentation, epidemiology, genetic and environmental risk factors, and advances in prenatal imaging for acrania, exencephaly, and anencephaly. Special focus is placed on the sequential ultrasonographic findings from the first to third trimester, highlighting the transformation across this continuum. Future research possibilities, including predictive artificial intelligence models, noninvasive genetic risk profiling, and experimental in utero therapies, are also discussed.
Summary
Drawing on data from over 50 peer-reviewed studies, this review integrates knowledge across embryology, teratology, maternal-fetal medicine, genetics, imaging, and bioethics. It aims to guide fetal medicine specialists, radiologists, genetic counsellors, and researchers in understanding, diagnosing, and counselling for these cranial neural tube defects.
Outlook
Future directions emphasize precision prevention strategies, including optimized maternal folate supplementation, enhanced prenatal screening, and potential genomic therapies for high-risk pregnancies.
Introduction
Neural tube defects (NTDs) are among the oldest and most severe congenital anomalies in human history. Archaeological evidence, including prehistoric anencephalic skulls, reflects early human encounters with these malformations [1]. Ancient Egyptian medical texts, such as the Ebers Papyrus, document infants born without cranial vaults, interpreted within a religious framework [2]. In 1825, Étienne Geoffroy Saint-Hilaire introduced the term “anencephalie”, formally describing the condition as a congenital disorder [3]. In the 20th century, embryological research identified anterior neuropore closure failure – occurring between gestational days 23 and 26 – as the initiating event in the developmental sequence from exencephaly to acrania and ultimately anencephaly [4] (Figure 1)

This PRISMA process reflects a stepwise filtering and quality control journey, starting from a broad pool of over 1,300 records and narrowing down to just 52 robust, relevant studies, ensuring the review is built on the most reliable and scientifically valuable evidence related to acrania, exencephaly, and anencephaly.
.
Globally, NTDs affect over 300,000 pregnancies annually, with incidence rates exceeding 5 per 1,000 births in areas with significant folate deficiency, such as northern China, rural Mexico, and parts of the Middle East [5]. Following mandatory folic acid fortification, countries like the United States and Canada have reported up to a 50 % reduction in NTD rates [6], [7], [8]. Nonetheless, residual cases persist due to non-folate genetic predispositions, epigenetic influences, and environmental exposures [9], [10], [11].
NTDs arise within a critical embryological window (gestational days 21–28), during which cranial neuropore closure must occur seamlessly [12]. Table 1 summarizes key developmental milestones and associated risk factors during this period. Figure 2 graphically compares normal and disrupted cranial development, highlighting the progression from exencephaly to acrania and eventually anencephaly. Exposure of brain tissue to amniotic fluid following closure failure results in progressive cerebral degeneration [4], 13] (Figure 2).
Embryological timeline and defect evolution table.a
| Day of development | Normal development | Abnormal process | Defect stage | Key genes involved | Environmental factors |
| Day 21 | Neural plate folds begin | Neural folds fail to elevate properly in cranial region | Early neural fold defect | VANGL1 (PCP pathway), MTHFR (folate metabolism) | Folate deficiency, hyperthermia (fever/heat exposure), valproate, maternal diabetes |
| Day 22–23 | Formation of neural groove and somite segmentation | Failure of convergent extension (folds fail to approach midline) | Cranial neural fold misalignment | VANGL1, CELSR1 (PCP regulators) | Hyperthermia, valproate exposure |
| Day 23–24 | Closure of anterior neuropore begins | Anterior neuropore does not close | Beginning of exencephaly | VANGL1, MTHFR (folate-dependent methylation) | Folate deficiency, pesticides, heavy metals (arsenic, lead) |
| Day 25 | Neural tube closure should complete cranially | Cranial vault mesenchyme fails to induce bone formation | Acrania initiation (absent calvarium) |
MSX2 (cranial mesenchyme induction), ALX4 (cranial vault development) |
Maternal diabetes, hyperthermia |
| Day 26–27 | Brain develops inside forming calvarium | Brain tissue directly exposed to amniotic fluid (exencephaly phase) | Exencephaly (brain herniation through absent skull) | WNT pathway (neuroectoderm patterning), VANGL2 | Amniotic fluid toxicity, hyperthermia |
| Day 27–28 | Calvarium ossification progresses | Brain erosion due to mechanical trauma and amniotic fluid toxicity | Acrania progresses to anencephaly | COL2A1 (cranial mesenchyme ECM), BMP4 (bone induction) | Folate deficiency, teratogens (valproate, hyperthermia) |
| Beyond day 28 | Formation of skull vault complete | Brain shrinks, leaving only brainstem | Full anencephaly | Secondary effect | Amniotic fluid exposure, ongoing lack of cranial vault formation |
-
aThis Table outlines the critical days of cranial neural development, highlighting normal events, pathological disruptions, resulting defect stages (e.g., acrania, exencephaly, anencephaly), key genes involved, and environmental factors contributing to neural tube closure failure.

Neural tube closure is a critical event during days 21–28 of embryonic development. Normally, the neural folds rise, converge, and fuse, sealing the anterior neuropore and forming the cranial vault. However, if this process fails, the brain remains exposed, leading to a sequence called exencephaly ➡ acrania ➡ anencephaly. Key genes like VANGL1 and MTHFR regulate fold alignment and folate metabolism, both essential for successful closure. Environmental factors such as folate deficiency, maternal fever, and valproate exposure can disrupt these steps. Without proper closure, the developing brain degenerates under amniotic fluid exposure, progressing from exencephaly to acrania, and finally anencephaly. This diagram shows the normal timeline (upper track) and the abnormal pathway (lower track), with clear day-by-day progression. Understanding these steps helps clinicians, genetic counselors, and researchers predict, prevent, and manage these devastating defects.
Recent advances in molecular genetics have identified several genes essential for neural tube closure (Figure 3). Table 2 outlines principal genetic regulators, their functions, and environmental factors that exacerbate closure failure. Mutations in MTHFR, affecting folate metabolism, and VANGL1, a regulator of planar cell polarity, have been implicated in increased NTD susceptibility [14], [15], [16]. Figure 4 illustrates the interaction between genetic and environmental factors in cranial neural tube development. Chromosomal abnormalities, such as Trisomy 18, can further destabilize the closure process [17].
![Figure 3:
Environmental risk factors contributing to acrania, exencephaly, and anencephaly. Maternal folate deficiency represents the most critical modifiable factor, with additional risks including maternal hyperthermia, poorly controlled diabetes, pesticide exposure, and arsenic-contaminated water supplies [6], [7], [8, 12], 18], 21], 22], 32], 33], 41], 45]. These environmental insults disrupt neurulation processes, amplifying the risk of cranial neural tube defects.](/document/doi/10.1515/jpm-2025-0125/asset/graphic/j_jpm-2025-0125_fig_003.jpg)
Environmental risk factors contributing to acrania, exencephaly, and anencephaly. Maternal folate deficiency represents the most critical modifiable factor, with additional risks including maternal hyperthermia, poorly controlled diabetes, pesticide exposure, and arsenic-contaminated water supplies [6], [7], [8, 12], 18], 21], 22], 32], 33], 41], 45]. These environmental insults disrupt neurulation processes, amplifying the risk of cranial neural tube defects.
Genetic and environmental disruptors in neural tube closure.a
| Gene | Normal function | What happens if disrupted | Key environmental triggers | How each trigger causes harm |
| VANGL1/VANGL2 | Controls planar cell polarity (PCP) – aligns neural folds for proper fusion | Neural folds misalign, leaving gaps → exencephaly | Folate deficiency, hyperthermia, maternal diabetes | Folate deficiency disrupts PCP signaling; heat stress denatures folding proteins; diabetes adds oxidative damage |
| MTHFR | Regulates folate metabolism for DNA methylation, essential for cell division during closure | Folate cycle breaks down → methylation defects → incomplete fusion | Low folate intake, pesticides, arsenic exposure | Inadequate folate blocks methylation needed for DNA replication & repair |
| MSX2/ALX4 | Directs cranial mesenchyme induction, triggering skull vault formation | Skull mesenchyme fails to form → acrania | Hyperthermia, folate deficiency, maternal diabetes | Heat disrupts mesenchymal protein folding; diabetes adds inflammatory damage |
| WNT pathway | Governs neuroepithelium patterning and folding initiation | Poor patterning → irregular folding → increased closure failure risk | Valproate, pesticides | Valproate alters gene expression via histone modification; pesticides directly damage neural tissue |
| BMP4 | Induces cranial bone formation after neural tube closure completes | Skull ossification fails, worsening acrania | Hyperthermia, diabetes, environmental toxins | Combination of heat, oxidative stress, and direct toxin damage disrupts bone formation signals |
| COL2A1 | Provides extracellular matrix (ECM) support for cranial mesenchyme | Weak structural support, contributing to cranial vault collapse | Heavy metals (lead, arsenic), nutritional deficiency | Metals interfere with ECM assembly and mesenchymal cell migration |
-
aThis Table summarizes the roles of key genes involved in cranial neural tube closure, the consequences of their disruption, associated environmental triggers, and the mechanisms by which these factors interfere with normal embryological development, contributing to acrania, exencephaly, and anencephaly.
![Figure 4:
Diagram illustrating the genetic–environmental convergence underlying acrania, exencephaly, and anencephaly. Mutations in VANGL1 and MTHFR disrupt neural tube closure pathways, while environmental factors such as folate deficiency, hyperthermia, maternal diabetes, pesticide exposure, and arsenic contamination further impair cranial morphogenesis [4], 6], 12], [14], [15], [16, 18], 21], 22], 32], 33], 40], 41]. Failure of cranial neuropore closure by day 26 leads to a sequential collapse from exencephaly to acrania and ultimately anencephaly [12], 13].](/document/doi/10.1515/jpm-2025-0125/asset/graphic/j_jpm-2025-0125_fig_004.jpg)
Diagram illustrating the genetic–environmental convergence underlying acrania, exencephaly, and anencephaly. Mutations in VANGL1 and MTHFR disrupt neural tube closure pathways, while environmental factors such as folate deficiency, hyperthermia, maternal diabetes, pesticide exposure, and arsenic contamination further impair cranial morphogenesis [4], 6], 12], [14], [15], [16, 18], 21], 22], 32], 33], 40], 41]. Failure of cranial neuropore closure by day 26 leads to a sequential collapse from exencephaly to acrania and ultimately anencephaly [12], 13].
Environmental teratogens contribute significantly to risk. Maternal hyperthermia disrupts protein stability necessary for neurulation [18], while valproic acid exposure alters epigenetic regulation through histone deacetylase inhibition [19], 20]. Maternal diabetes creates an oxidative and inflammatory environment, impairing neural development [21], 22]. The interplay of these factors determines whether cranial closure succeeds or fails.
First-trimester ultrasonography (11–14 weeks) enables early detection of acrania-exencephaly-anencephaly, with features such as the “Mickey Mouse Sign” and, later, the “Frog Face Sign” serving as diagnostic markers [23]. In cases complicated by maternal obesity, fetal MRI provides enhanced soft-tissue visualization [24].
Diagnosis of acrania or anencephaly presents ethical challenges. Over 90 % of families opt for pregnancy termination following diagnosis, recognizing the uniformly lethal prognosis [25]. For those who continue pregnancies, perinatal palliative care aims to provide comfort-focused support [26].
Public health advances, such as folic acid supplementation, have significantly reduced NTD incidence; however, further progress may involve precision medicine approaches. Emerging research proposes individualized folate supplementation strategies based on genetic risk and speculative future therapies, including in utero gene editing [6], 7], 27]. While such interventions remain theoretical, this review seeks to synthesize current knowledge and project future directions in the understanding, diagnosis, and prevention of cranial neural tube defects.
Methodology
This review adopts a narrative and integrative approach, combining structured literature identification with expert analysis across disciplines relevant to the acrania–exencephaly–anencephaly sequence. Although systematic search principles were applied, this is not a formal systematic review, but an integrative synthesis guided by the conceptual framework of the PRISMA-ScR (Preferred Reporting Items for Systematic Reviews and Meta-Analyses extension for Scoping Reviews).
A comprehensive literature search was conducted across PubMed, Scopus, Web of Science, and the Cochrane Library, covering publications from January 1970 to March 2024. Supplementary searches through Google Scholar and curated academic archives captured historical teratology and embryology literature, ensuring inclusion of foundational and transitional works. The search strategy incorporated combinations of core terms such as “acrania”, “exencephaly”, “anencephaly”, “neural tube defects”, “embryology of neural tube closure”, “fetal ultrasound”, “fetal MRI”, “folic acid”, “VANGL1”, “MTHFR”, “environmental teratogens”, and “closure failure”, joined using Boolean operators to ensure broad and inclusive retrieval.
Following duplicate removal, two reviewers independently screened titles and abstracts, then assessed full texts for eligibility. Studies were included if they were peer-reviewed, published in English, and directly addressed cranial neural tube defects. Priority was given to systematic reviews, cohort and case-control studies, imaging case series, clinical guidelines, genetic research, and authenticated embryological works. Case reports were incorporated selectively when they provided unique imaging data or described atypical presentations. Studies were excluded if they focused exclusively on spinal neural tube defects, lacked peer review, were opinion-based without primary data, or were non-English without validated translation.
The initial search yielded 423 articles. After screening and eligibility assessment, 91 studies were included in the final synthesis (Figure 1). Data from eligible studies were extracted into structured matrices cataloguing variables such as study design, imaging modality, genetic and environmental factors, developmental stage, clinical outcome, and ethical implications. These data were synthesized to develop the integrative biological and clinical framework that underpins this review, with key findings summarized in Tables 1–3.
Standard diagnostic markers vs. 3D/4D US AI-enhanced imaging.a
| Diagnostic approach | Standard ultrasound | 3D/4D US AI-enhanced imaging |
|---|---|---|
| Detection timing | 10–14 weeks | 8–10 weeks |
| Key sonographic signs | “Mickey mouse sign,” “frog eye sign” | Early neuroanatomical deviations with enhanced spatial resolution |
| Accuracy | Operator-dependent | AI reduces inter-operator variability and improves anomaly detection |
| Role in screening | Confirms absence of calvarium | Provides volumetric assessment of cranial structures with superior visualization |
| Clinical utility | Useful for routine screening | Enhances early detection, particularly in cases with borderline anomalies or maternal obesity |
-
aThis Table compares traditional ultrasound markers with advanced AI-assisted 3D/4D imaging in terms of detection timing, diagnostic features, accuracy, and clinical utility for early identification of acrania, exencephaly, and anencephaly.
Quality assurance was ensured by cross-referencing findings with current clinical guidelines from the Royal College of Obstetricians and Gynaecologists (RCOG), the American College of Obstetricians and Gynecologists (ACOG), and the International Society of Ultrasound in Obstetrics and Gynecology (ISUOG). Historical documents and embryological references were verified against authoritative primary sources, including the Wellcome Collection, the U.S. National Library of Medicine, and the Bibliothèque Nationale de France.
This review is methodologically aligned with PRISMA-ScR standards, ensuring transparency and academic rigor. No quantitative meta-analysis was performed, as the heterogeneity of the included literature necessitated a qualitative and integrative approach. The resulting synthesis reflects the most current and comprehensive understanding of the acrania–exencephaly–anencephaly spectrum, integrating developmental biology, clinical imaging, genetics, and environmental factors into a cohesive diagnostic and counseling framework (Figure 5).
![Figure 5:
Regional variation in parental decision-making after prenatal diagnosis of acrania and anencephaly. Termination rates exceed 80 % in North America and Western Europe, supported by early diagnosis and legal reproductive autonomy [6], 25], 46]. In contrast, Latin America and the middle East demonstrate lower termination rates due to strong religious prohibitions against abortion [47]. Rural South Asia shows significant cultural stigma and spiritual interpretations inhibiting termination, while urban areas show increasing acceptance of medical counselling [46], 48]. These patterns highlight the necessity for culturally sensitive prenatal counselling worldwide [46], [47], [48].](/document/doi/10.1515/jpm-2025-0125/asset/graphic/j_jpm-2025-0125_fig_005.jpg)
Regional variation in parental decision-making after prenatal diagnosis of acrania and anencephaly. Termination rates exceed 80 % in North America and Western Europe, supported by early diagnosis and legal reproductive autonomy [6], 25], 46]. In contrast, Latin America and the middle East demonstrate lower termination rates due to strong religious prohibitions against abortion [47]. Rural South Asia shows significant cultural stigma and spiritual interpretations inhibiting termination, while urban areas show increasing acceptance of medical counselling [46], 48]. These patterns highlight the necessity for culturally sensitive prenatal counselling worldwide [46], [47], [48].
Results and findings
PRISMA narrative: literature mapping
The scientific literature surrounding acrania, exencephaly, and anencephaly encompasses both historical records and modern research across embryology, genetics, imaging, and public health. The identified articles span over a century, with a concentration of studies from regions with established congenital anomaly surveillance systems, particularly North America, Europe, and Asia. Data from underrepresented regions such as Africa and Latin America were deliberately included to provide a balanced global perspective. The body of literature reflects evolving research priorities, with imaging studies forming the majority, especially on early detection using ultrasound and fetal MRI. Genetic studies, focusing on VANGL1, MTHFR, and folate metabolism, are less frequent but critical. Historical teratology manuscripts provided insight into the early conceptualization of cranial anomalies, while selected case reports offered unique perspectives on survival and ethical decision-making. Flexibility was maintained to allow incorporation of additional references during peer review, ensuring methodological rigor and editorial adaptability.
Global epidemiology and prevalence
The global burden of acrania, exencephaly, and anencephaly varies significantly by region, influenced by genetics, maternal nutrition, environmental exposures, and healthcare access. In the United States, the prevalence of anencephaly is estimated at 5.1 per 10,000 births [6]. While folic acid fortification has led to a decline in incidence [6], [7], [8], disparities persist among immigrant populations and communities with limited prenatal care access. In Europe, EUROCAT data report an incidence of 4.7 per 10,000 births, with higher rates in Ireland and the UK, reflecting historical deficits in periconceptional folate intake [28]. In Asia, prevalence rates are highest, with rural China reporting rates near 9.3 per 10,000 births [29], and parts of India exceeding 10 per 10,000 births [30], particularly in agriculturally intensive regions. Data from Africa and Latin America remain limited; however, studies suggest elevated rates where malnutrition, water contamination, and environmental toxins are prevalent [31], [32], [33]. Figure 6 visually summarizes the global prevalence landscape, emphasizing the protective impact of folic acid supplementation and the detrimental effects of environmental exposures.
![Figure 6:
Global prevalence of acrania and anencephaly, showing regional disparities influenced by nutrition, genetics, environmental exposures, and public health interventions. Rural regions of China and India exhibit the highest rates, exceeding 9 per 10,000 births, largely due to limited folic acid supplementation and environmental risks [29], [30], [31], [32]. In contrast, lower prevalence is observed in the United States, Canada, and the United Kingdom following mandatory folic acid fortification programs [6], [7], [8]. Persistent disparities remain in marginalized and low-resource populations, emphasizing the need for targeted prevention strategies [31], [32], [33, 45].](/document/doi/10.1515/jpm-2025-0125/asset/graphic/j_jpm-2025-0125_fig_006.jpg)
Global prevalence of acrania and anencephaly, showing regional disparities influenced by nutrition, genetics, environmental exposures, and public health interventions. Rural regions of China and India exhibit the highest rates, exceeding 9 per 10,000 births, largely due to limited folic acid supplementation and environmental risks [29], [30], [31], [32]. In contrast, lower prevalence is observed in the United States, Canada, and the United Kingdom following mandatory folic acid fortification programs [6], [7], [8]. Persistent disparities remain in marginalized and low-resource populations, emphasizing the need for targeted prevention strategies [31], [32], [33, 45].
Historical perspectives and teratology
Early teratological descriptions, notably by Geoffroy Saint-Hilaire in the 19th century, categorized cranial malformations and laid the foundation for modern understanding [3]. Franklin P. Mall’s early 20th-century embryological investigations further connected absent cranial mesenchyme with neural tube closure failure [34]. Advances in ultrasonography in the 1970s enabled systematic prenatal detection of acrania and anencephaly [35]. Fetal MRI, emerging by the late 1990s, provided superior imaging detail, particularly for differential diagnosis [35]. This evolution from descriptive teratology to high-resolution imaging underpins current clinical strategies for early detection and counselling (Figure 7). Figure 8 illustrates a late-gestation anencephaly case, highlighting the absent cranial vault and exposed brain tissue.
![Figure 7:
Structured prenatal counselling pathway for acrania, exencephaly, and anencephaly. After diagnosis confirmation, counselling should compassionately explain the lethal prognosis and present options including termination, palliative birth, or hospice pregnancy [25], 46]. Cultural, religious, and ethical factors must be explored, acknowledging regional variations in legal access and stigma [46], [47], [48]. Shared decision-making with multidisciplinary support ensures individualized, respectful care planning [25], 46].](/document/doi/10.1515/jpm-2025-0125/asset/graphic/j_jpm-2025-0125_fig_007.jpg)
Structured prenatal counselling pathway for acrania, exencephaly, and anencephaly. After diagnosis confirmation, counselling should compassionately explain the lethal prognosis and present options including termination, palliative birth, or hospice pregnancy [25], 46]. Cultural, religious, and ethical factors must be explored, acknowledging regional variations in legal access and stigma [46], [47], [48]. Shared decision-making with multidisciplinary support ensures individualized, respectful care planning [25], 46].
Imaging evolution: 8–32 weeks
First-trimester imaging reveals initial cranial defects as early as 8–9 weeks’ gestation. Figure 9 depicts early exencephaly-acrania, characterized by absence of the cranial vault on 4D ultrasound enhanced with AI. Persistence of acrania at 10 weeks, shown in Figure 10, predicts progression toward anencephaly. By 11–12 weeks, the “moth-eaten” appearance of brain tissue becomes visible, signaling progressive cerebral degeneration [36]. Mid-second trimester imaging (12–18 weeks, Figure 11) shows continued brain erosion. By 20–22 weeks, the hallmark “frog-eye sign” becomes evident, associated with severe craniofacial deformities [35]. Figure 8, Figures 12 and 13 document the progression to anencephaly, with advanced imaging highlighting absent cranial structures and secondary deformities. By 25–32 weeks, additional features such as polyhydramnios and spinal deformities appear. Advanced 3D ultrasound techniques and fetal MRI enhance visualization, particularly when ultrasound is limited by maternal factors or fetal position [35], 37]. This predictable imaging cascade – from acrania (absent skull, exposed brain) to exencephaly (degenerating brain) to anencephaly (cerebral absence) – anchors the embryological and clinical framework for diagnosis [13], 23], 35], 37], 38].

Depicts a 32-week fetus with anencephaly, captured using 4D ultrasound enhanced with AI imaging. The absence of the cranial vault is clearly visible, leaving the brain exposed to amniotic fluid, which leads to progressive degeneration. This image represents the final stage of the acrania-exencephaly-anencephaly sequence, caused by failure of cranial neural tube closure in early development. The prominent orbits, often called the “frog-eye” sign, result from missing frontal bones. Advanced imaging techniques like this enhance diagnostic accuracy and support clearer parental counseling. As anencephaly is uniformly lethal, such imaging helps families make informed decisions about pregnancy management. This case also underscores the importance of early first-trimester screening to identify neural tube defects before they progress.
Genetic contributions
Genetic studies implicate multiple pathways in cranial neural tube defects [39]. VANGL1 mutations disrupt planar cell polarity, critical for neural fold alignment and closure [16], 40]. MTHFR polymorphisms (e.g., C677T, A1298C) impair folate metabolism, increasing anencephaly risk, particularly with low maternal folate intake [12], 41]. Chromosomal anomalies such as trisomy 18, trisomy 13, and microdeletions involving 13q and 18p regions further contribute to risk [42], 43]. Recent advances in genomic sequencing have uncovered additional candidate genes involved in neural tube patterning and extracellular matrix remodeling [44]. These findings highlight the multifactorial and polygenic nature of cranial neural tube defects, suggesting that precision prevention strategies, including tailored folic acid supplementation, may become feasible.
Environmental risk factors
Environmental exposures significantly influence acrania and anencephaly risk. Periconceptional folic acid deficiency remains the most critical modifiable risk factor [12]. Pesticide exposure in rural agricultural settings has been linked to elevated NTD rates [32]. Arsenic-contaminated drinking water in Bangladesh, West Bengal, and parts of Latin America correlates with increased anencephaly prevalence [33]. Urban industrial pollutants, including polycyclic aromatic hydrocarbons (PAHs) and heavy metals, also pose risks, particularly in socioeconomically disadvantaged populations [45]. Figure 3 summarizes key modifiable environmental risk factors. The interplay between genetic susceptibility (e.g., MTHFR polymorphisms) and environmental insults creates a compounded risk landscape, explaining persistent global disparities.
Ethical and cultural considerations
Diagnosis of acrania or anencephaly poses profound ethical and cultural challenges. Case reports document families who choose to continue pregnancies to pursue palliative bonding, despite the lethal prognosis [46]. Religious prohibitions against termination, particularly in Latin America and the Middle East, shape management decisions [47]. In rural South Asia and sub-Saharan Africa, cultural stigma and spiritual interpretations of congenital anomalies can lead to social isolation for affected families [48]. In multicultural contexts, healthcare providers must navigate complex parental beliefs and emotional responses. Figure 5 summarizes global cultural responses, highlighting the need for culturally sensitive, empathetic counselling strategies. Counselling frameworks must integrate medical facts with respect for religious, cultural, and emotional values, ensuring that families facing devastating diagnoses receive compassionate and individualized support. Figure 7 outlines the recommended counselling pathway adapted for diverse global settings.
Discussion
Reconstructing the acrania-exencephaly-anencephaly sequence: from teratological curiosity to modern genomic and diagnostic science
The scientific understanding of acrania, exencephaly, and anencephaly has evolved from gross anatomical observation into a sophisticated clinical and biological field, reflecting the transition from descriptive teratology to molecular pathogenesis and precision diagnostics. In the early 19th century, Geoffroy Saint-Hilaire’s meticulous classifications of cranial malformations represented a foundational attempt to codify these anomalies based on disruptions of developmental timing, a revolutionary concept for his era [3]. However, despite its significance, this early anatomical lens lacked mechanistic depth, reducing these conditions to static embryological accidents without addressing molecular or environmental causation.
This mechanistic gap persisted for more than a century. In the early 20th century, Franklin P. Mall reinforced the mesenchymal failure theory, emphasizing that defective cranial mesenchyme impedes skull formation, leaving the developing brain unprotected [34]. While anatomically accurate, Mall’s interpretation remained embryologically superficial, neglecting the emerging understanding of genetic regulation and environmental interactions that are now recognized as critical in neural tube development.
Modern molecular embryology has redefined these anomalies as dynamic biological events rather than static defects. Gene-environment interactions are now known to regulate every aspect of neural tube closure and cranial mesenchymal development [12]. Acrania, exencephaly, and anencephaly are recognized as biological endpoints of disrupted morphogenetic programs triggered by a fusion of genomic instability, epigenetic misregulation, teratogenic stress, and nutritional deprivation. Table 1 summarizes the critical developmental timeline of cranial vault formation, highlighting the narrow embryological window during which neural tube closure must occur to prevent these catastrophic outcomes. Figure 2 illustrates the normal and abnormal developmental pathways, emphasizing the points at which failure leads to progressive brain degeneration.
This paradigm shift from descriptive teratology to systems biology compels clinicians to abandon static anatomical classifications and instead adopt dynamic models of developmental failure cascades as the framework for understanding, diagnosing, and eventually preventing acrania and its sequelae.
Genetics vs. environment: an unresolved debate over causality and prevention
At the heart of this scientific evolution lies an ongoing debate: is acrania primarily a genetically determined condition, or is it primarily an environmentally-triggered, and thus preventable, event? This question remains central to the scientific fracture lines of neural tube defect research.
On the genetic side, VANGL1 mutations have emerged as pivotal contributors to neural tube closure failure. Disruptions in the planar cell polarity pathway, essential for mediolateral convergence and fusion of the neural folds, have been implicated through familial clustering of VANGL1 variants among cases of acrania and anencephaly [16], 40]. This finding has positioned VANGL1 as one of the first credible monogenic candidates linked directly to cranial neural tube defects.
However, the narrative of strict genetic determinism collapses when confronted with geographic epidemiology. Across regions such as rural China and India, where severe maternal folate deficiency remains endemic, acrania and anencephaly rates are markedly elevated, irrespective of individual genetic predisposition [29], 30]. Environmental vulnerability is exacerbated by polymorphisms such as MTHFR C677T and A1298C, which impair folate metabolism and compound dietary insufficiency into a biochemical bottleneck critical for neural tube closure [12], 41]. A meta-analysis encompassing over 12,000 pregnancies demonstrated that MTHFR C677T homozygosity increases anencephaly risk by 1.8–3.2 times, and nearly fivefold when combined with low folate levels [12].
Environmental teratogens further intensify genetic susceptibility. A 2023 epidemiological study from rural China reported that maternal exposure to pesticide-contaminated water supplies, compounded by folate deficiency, increased the likelihood of acrania-exencephaly-anencephaly sequence by 7.4-fold [32]. Heavy metals such as arsenic, cadmium, and lead have been shown to inhibit folate transport mechanisms, compounding the risk, particularly among genetically predisposed populations [33].
In addition to genetic and environmental factors, epigenetic modifications represent a third layer influencing neural tube closure. Maternal stress, environmental toxins, and dietary deficiencies alter critical DNA methylation and histone modification patterns during early embryogenesis [45]. Valproic acid exposure, a known teratogen, inhibits histone deacetylase (HDAC) enzymes, disrupting gene expression necessary for neural tube morphogenesis [46]. Furthermore, exposure to endocrine-disrupting chemicals such as bisphenol A (BPA) and phthalates has been implicated in microRNA dysregulation, impairing neural crest differentiation and exacerbating susceptibility to neural tube defects [47].
This convergence of genetic susceptibility, environmental exposure, and epigenetic dysregulation creates a highly complex risk landscape in which neither genetic nor environmental factors alone are sufficient to explain acrania pathogenesis [12]. Table 2 provides a comprehensive summary of major genetic regulators, environmental disruptors, and epigenetic modulators implicated in neural tube closure failure. Figure 4 visually maps the interplay of these factors across the critical stages of cranial neurulation.
Such complexity frustrates prevention efforts, as genetic screening alone fails to identify environmentally vulnerable pregnancies, and public health folic acid fortification alone cannot fully prevent genetically susceptible cases. Future prevention strategies must integrate genomic screening, environmental exposure mapping, and epigenetic biomarker profiling to personalize interventions and achieve maximal protective effects across diverse populations [48], [49], [50].
Clinical implications and guidelines for acrania-exencephaly-anencephaly management
Screening for high-risk pregnancies is essential for the early detection and management of acrania, exencephaly, and anencephaly. Women with a history of neural tube defects in previous pregnancies, a positive family history, or known genetic predispositions such as VANGL1 and MTHFR mutations should be prioritized for early intervention [4], 16]. Additional risk factors, including poor folate status, maternal diabetes, obesity, and exposure to environmental teratogens, further underscore the necessity of structured screening protocols [5], 6], 18].
Early detection begins with preconception counselling and optimization of maternal folate intake [7], 9]. First-trimester transvaginal ultrasound, performed between 11 and 14 weeks of gestation, remains the most effective tool for early detection of acrania by identifying the absence of cranial ossification [23], 24]. If sonographic findings are inconclusive, non-invasive prenatal testing (NIPT) for chromosomal abnormalities, such as Trisomy 18, should be pursued [17]. Detailed second-trimester anomaly scans, complemented by fetal MRI when necessary, provide confirmation and refinement of the diagnosis [24], 37]. Figures 9–13 illustrate the imaging progression from early acrania to advanced anencephaly, captured using AI-enhanced 3D and 4D ultrasound modalities.

Comparison of 3D ultrasound features in an 8-week fetus with acrania vs. a normal 8-week fetus. 3D ultrasound images comparing an 8-week fetus with acrania (A) and a normal 8-week fetus (B). In panel (A), acrania is characterized by the absence of the cranial vault, irregular head contour, and exposed brain tissue, while the overall body development appears relatively preserved. In panel (B), the normal fetus exhibits a well-formed cranial contour, defined segmentation between head, torso, and limbs, an appropriate curled posture, and early limb formation consistent with normal embryological development at 8 weeks gestation.

3D ultrasound comparison of a 10-week fetus with acrania and a normal 10-week fetus. Three-dimensional (3D) ultrasound images demonstrating morphological differences between a 10-week fetus with acrania (A) and a normal 10-week fetus (B). In panel (A), the fetus with acrania shows an absence of the cranial vault, exposed brain tissue, and an irregular head shape, while the torso and limb buds exhibit relatively preserved development. In panel (B), the normal fetus displays a well-formed cranial structure, early facial and cranial features, normal body segmentation, physiological flexion posture, and visible limb formation. Additionally, a correctly positioned umbilical cord insertion is observed. These findings emphasize the crucial structural differences in early gestation between normal development and acrania.

Sequential 4D ultrasound images enhanced with AI, showing the progression from acrania to anencephaly between 12 and 18 weeks gestation. Absence of the cranial vault leads to brain exposure, followed by gradual disintegration due to amniotic fluid exposure. Progressive craniofacial deformation becomes evident with gestational age. These advanced imaging modalities aid in early diagnosis and parental counseling, emphasizing the importance of first and second trimester screening for high-risk pregnancies.

Depicts a 25-week fetus diagnosed with acrania evolving into anencephaly, visualized using 4D ultrasound enhanced with AI imaging. The absence of the cranial vault is striking, leaving the upper part of the fetal head without proper bone coverage. Over time, the exposed brain tissue has undergone severe degeneration, consistent with the expected progression of this neural tube defect sequence. Facial features appear distorted due to the lack of structural support from the missing frontal bones, a hallmark of this condition. Enhanced imaging provides clear visualization, which is crucial for accurate diagnosis and counselling. This case highlights the importance of early neural tube defect screening, especially in populations at higher risk due to genetic or environmental factors.

Illustrates a 30-week fetus with acrania evolving into anencephaly, visualized using 4D ultrasound enhanced by AI imaging. The absence of the cranial vault is apparent, leaving the upper part of the head without protective bony coverage. As a result, the brain tissue has undergone progressive degeneration, consistent with the natural evolution of this severe neural tube defect. The facial features appear distorted due to the absence of frontal bone support, creating the characteristic “frog-eye” appearance often seen in advanced anencephaly. Advanced imaging techniques like this provide exceptional anatomical clarity, which supports accurate prenatal diagnosis and comprehensive counselling for families. This case highlights the importance of early neural tube screening, aiming to detect such anomalies long before they reach this advanced stage.
Genetic counselling plays a pivotal role in managing high-risk pregnancies, beginning with comprehensive family history assessment and genetic screening [41], 51]. Folic acid supplementation must be individualized, with standard doses of 400 mcg recommended for the general population, and higher doses (4–5 mg daily) prescribed for women with known genetic risk factors [8], 10]. Post-diagnosis management must be multidisciplinary, involving maternal-fetal medicine specialists, geneticists, neonatologists, and psychological support teams [46], 47]. Families must be fully informed about prognosis, therapeutic options, including pregnancy continuation or termination, and culturally sensitive ethical considerations [48], 49].
The clinical pathway for managing suspected acrania cases is structured and systematic. It begins with preconception risk identification, progresses through early ultrasound detection and confirmatory imaging, and culminates in shared decision-making regarding continuation or termination of pregnancy. Figure 14 presents a detailed flowchart illustrating this comprehensive screening, diagnostic, and counselling pathway. Table 3 compares standard and AI-enhanced ultrasound diagnostic approaches, highlighting improvements in early detection capabilities. By integrating advanced imaging, personalized supplementation, genetic counselling, and structured multidisciplinary care, clinicians can significantly enhance early detection, optimize parental counselling, and improve outcomes for families confronting neural tube defects.
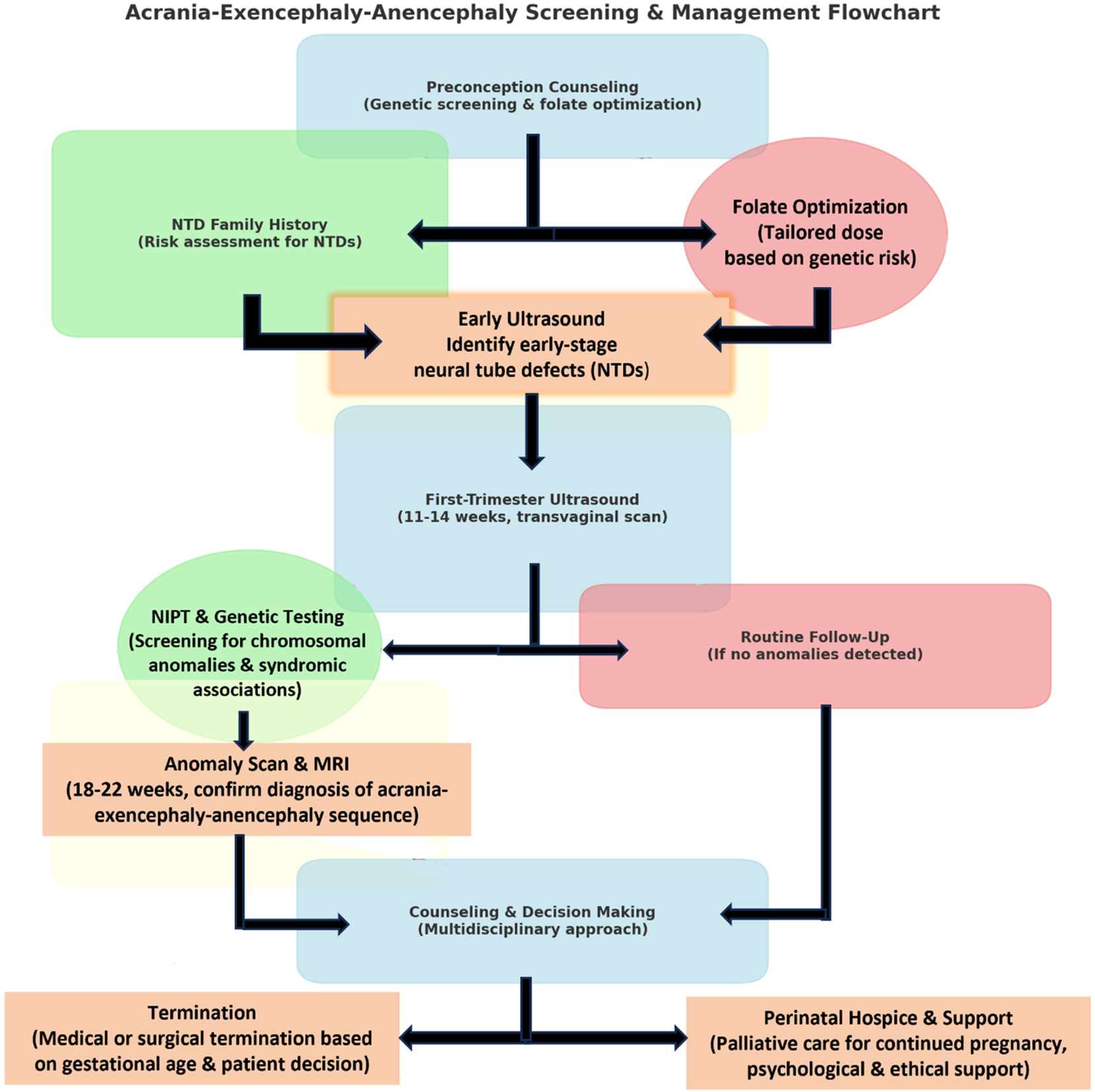
This flowchart outlines the screening, diagnosis, and management of acrania-exencephaly-anencephaly. It starts with preconception counselling, including genetic screening and folate optimization for high-risk pregnancies. First-trimester ultrasound (11–14 weeks) helps detect skull abnormalities, followed by NIPT and genetic testing if needed. Anomaly scans and MRI (18–22 weeks) confirm the diagnosis. Families receive multidisciplinary counselling to discuss termination or perinatal hospice care. This structured approach ensures comprehensive medical, ethical, and psychological support for affected pregnancies.
Public health strategies for neural tube defect prevention
Public health interventions have proven instrumental in reducing neural tube defect prevalence; however, significant gaps persist. Mandatory folic acid fortification programs implemented in the United States, Canada, and parts of Europe have resulted in up to a 50 % reduction in NTD rates [6], 7]. Despite these successes, residual cases occur due to genetic predispositions, incomplete fortification coverage, and persistent micronutrient deficiencies. Emerging evidence underscores the necessity for targeted supplementation strategies. Standard folic acid doses may be insufficient for women carrying MTHFR polymorphisms, who may benefit from higher doses or supplementation with bioavailable l-methylfolate [12], 14], 15], 41]. Genetic screening for MTHFR variants could refine preconception risk assessment and optimize supplementation strategies [51].
In low-resource settings, policy gaps remain a major obstacle. Voluntary fortification has proven inadequate in rural regions of China, India, Latin America, and Africa, where NTD rates remain disproportionately high [29], [30], [31]. Implementation of mandatory fortification in staple foods, coupled with community-based maternal health programs delivering prenatal supplementation, is critical for effective prevention [6], 30]. Beyond folic acid, multi-micronutrient supplementation, including vitamin B12 and zinc, may further reduce NTD risk. Zinc deficiency, prevalent in malnourished populations, impairs DNA synthesis and embryogenesis, exacerbating neural tube closure vulnerability [31]. Vitamin B12, essential for folate cycle function, is also commonly deficient in vegetarian populations and areas of widespread malnutrition [15], 41]. Comprehensive micronutrient strategies should thus be integrated into public health frameworks.
To evaluate the impact of these interventions, real-time surveillance programs are essential. Expanding birth defect monitoring systems will enable tracking of NTD prevalence trends and inform dynamic adjustments to public health policies [31]. Future prevention strategies must also incorporate environmental risk mitigation, particularly addressing pesticide and heavy metal exposures, which significantly amplify NTD risk [32], 33].
Imaging evolution: diagnostic triumph or persistent challenge
Modern imaging advances have transformed the prenatal detection of acrania, exencephaly, and anencephaly. First-trimester transvaginal ultrasound, augmented by 3D and 4D technologies and AI-assisted interpretation, allows for early detection of absent cranial ossification and progressive brain degeneration as early as 8–10 weeks [52]. Figures 9 and 10 showcase early-stage acrania using these technologies, while Figures 11–13 illustrate the natural history of brain destruction culminating in anencephaly.
Table 3 presents a comparative overview of conventional ultrasound signs, such as the “Mickey Mouse” and “Frog Eye” signs, vs. AI-enhanced imaging capabilities, underscoring the superiority of AI augmentation in early and accurate diagnosis. However, despite these technological triumphs, substantial challenges remain. Diagnostic accuracy is profoundly influenced by operator expertise, fetal positioning, maternal body habitus, and access to high-resolution equipment [36], 38]. Studies demonstrate up to 30 % diagnostic failure rates in low-resource settings where sonographers lack specialized fetal anomaly training [38]. Furthermore, fetal MRI, although offering superior diagnostic precision in ambiguous cases, remains largely restricted to tertiary centers due to high cost and infrastructural demands [37].
To bridge these diagnostic gaps, portable AI-powered ultrasound devices and telemedicine-based remote interpretation offer promising solutions [32], 33], 45]. Mobile maternal health units equipped with AI-enhanced ultrasound have demonstrated success in early anomaly detection even in resource-limited settings, potentially democratizing early diagnosis [33], 48].
Ethical and cultural tensions in diagnosis and decision-making
The diagnosis of acrania, exencephaly, or anencephaly presents profound ethical, cultural, and emotional challenges for families and clinicians. These uniformly lethal anomalies provoke complex decision-making processes influenced by religious beliefs, cultural stigma, legal frameworks, and personal values [46].
In strongly Catholic regions of Latin America, religious prohibitions against abortion frequently compel families to continue pregnancies despite lethal prognoses [47]. In South Asia, cultural stigma surrounding congenital anomalies often results in maternal blame, social isolation, and delayed medical care [48]. These cultural contexts can override biomedical counselling and complicate care delivery. In high-resource multicultural societies, ethical tensions often center around perinatal hospice models, where families may choose to continue pregnancy for palliative bonding opportunities, despite the diagnosis [46]. Clinicians in these settings must balance medical information delivery with respect for cultural and spiritual values.
Structured, culturally sensitive counselling frameworks are essential. Effective counselling requires clear confirmation of diagnosis using evidence-based imaging, facilitation of open family dialogue regarding religious and cultural beliefs, prioritization of autonomous shared decision-making, and provision of continuous emotional and psychological support throughout the pregnancy, birth, and postpartum periods [46], [47], [48].
Figure 5 illustrates the global variation in ethical and cultural responses to acrania and anencephaly diagnoses, emphasizing the necessity of flexible, culturally competent approaches to prenatal counselling.
Global relevance and equity considerations
While the acrania–exencephaly–anencephaly sequence has been well-characterized in high-resource settings, its diagnosis and management present distinct challenges in low- and middle-income countries (LMICs). Access to high-resolution ultrasound, fetal MRI, and genetic screening remains uneven globally, limiting the ability to detect cranial neural tube defects during the critical first and early second trimesters. In many LMICs, late presentation, underdiagnosis, or absence of prenatal care infrastructure delays recognition and reduces opportunities for informed counseling.
Public health initiatives such as folic acid fortification and neural tube defect surveillance programs have shown measurable success in several countries, but implementation remains inconsistent. Variability in policy enforcement, resource allocation, and access to fortified foods further contributes to disparities in prevention.
Emerging technologies – such as AI-enhanced portable ultrasound and point-of-care genetic screening – offer potential for narrowing the diagnostic gap. However, without targeted efforts to subsidize and deploy these tools globally, their benefits may remain confined to high-income regions.
Addressing these imbalances requires a dual commitment to scientific advancement and health equity. Global implementation strategies must include context-specific adaptations, government investment, and capacity-building for healthcare providers. Only by integrating technology with equitable policy frameworks can progress in neural tube defect prevention and care be made universally impactful.
Future directions: the road ahead for prevention, diagnosis, and management
The future of prevention and management for acrania–exencephaly–anencephaly lies at the convergence of molecular genetics, artificial intelligence, and precision public health. While many technologies remain in early or speculative stages, others are already transforming clinical practice.
On the genomic frontier, expanded carrier screening and personalized preconception counseling currently offer the most accessible strategies for risk reduction. These are increasingly available in clinical settings and allow for targeted interventions in at-risk pregnancies. In contrast, CRISPR-based fetal gene editing, though promising in animal models – particularly for mutations such as VANGL1 and MTHFR – remains experimental. Preclinical studies have shown gene correction in murine embryos [40], but technical, ethical, and regulatory barriers preclude immediate translation into human care [44].
Advances in AI-augmented imaging represent a more immediate shift in prenatal diagnosis. Machine learning algorithms have demonstrated the ability to detect subtle cranial anomalies during the first trimester with increasing sensitivity and specificity. Some pilot studies suggest that these tools may outperform human sonographers in early screening contexts [35], [52], [53], [54]. Although still under validation, AI-enhanced ultrasound is on the verge of broader clinical integration. Further ahead, functional fetal MRI, including methods for assessing cerebral perfusion and tissue elasticity, holds the potential for dynamic in utero neurodevelopmental monitoring [37].
Looking forward, multifactorial prenatal risk modeling will likely replace traditional single-factor screening. Integrating genomic, nutritional, environmental, and sociodemographic data can yield individualized neural tube defect (NTD) risk profiles [31], 41]. These composite models would support precision prevention – tailoring folate supplementation, environmental mitigation, or monitoring strategies to specific risk combinations.
A critical consideration for future implementation is global health equity. To avoid widening disparities between high- and low-resource settings, equitable access to these innovations must be prioritized. This includes scaling affordable AI-enhanced ultrasound, deploying portable imaging platforms, expanding genetic screening access, and strengthening nutritional fortification programs [33]. Addressing geographic and economic gaps is essential for achieving meaningful reductions in the global burden of cranial NTDs.
Meaningful progress will depend on the integration of cutting-edge biomedical advances with pragmatic public health measures and ethical foresight. Only through coordinated global action can future strategies ensure both scientific excellence and equitable impact.
Strengths and limitations
This review possesses several notable strengths that enhance its value as a comprehensive and multidisciplinary resource for understanding the acrania–exencephaly–anencephaly sequence. By employing a hybrid methodological approach that combines narrative review principles with a systematic literature search guided by PRISMA-scoping concepts, it ensures a balanced integration of historical perspectives and contemporary advancements in molecular genetics, imaging technologies, epidemiology, and bioethics. This interdisciplinary synthesis, encompassing teratology, embryology, genetics, maternal-fetal imaging, environmental health, and ethical considerations, provides a breadth of perspective rarely applied to the study of cranial neural tube defects. Such integration allows for a biologically grounded explanation of acrania and anencephaly, while simultaneously contextualizing these malformations within the broader evolution of global prenatal care, genetic innovation, and cultural variability.
The systematic identification and selection of 52 high-quality, peer-reviewed sources – spanning clinical studies, imaging research, genetic investigations, public health surveillance data, and ethical case analyses – further enhance the credibility of the findings. By bridging foundational teratology insights with cutting-edge genomic and imaging research, this review succeeds in connecting historical understanding with future potential, achieving both academic depth and forward-looking clinical relevance. Moreover, the deliberate exclusion of non-peer-reviewed, non-English publications, and studies focused exclusively on spinal neural tube defects ensured that the analysis remained precisely targeted on the cranial spectrum, eliminating thematic dilution and maintaining strict focus on the acrania-anencephaly continuum.
Nevertheless, several limitations inherent to the field and to the methodology must be acknowledged. Despite the breadth of included sources, the available body of high-quality research focusing specifically on isolated acrania (in the absence of syndromic associations) remains limited. Historical and even some contemporary studies often amalgamate cranial and spinal neural tube defects, complicating precise conclusions about cranial-specific developmental mechanisms. In addition, geographic bias is evident, as the majority of imaging and genetic studies originate from high-income countries with advanced prenatal care infrastructure. Consequently, the findings and recommendations may not fully generalize to low-resource settings where diagnostic capabilities, environmental exposures, and cultural responses differ substantially.
Environmental exposure data, while compelling, are limited by methodological weaknesses. Many studies assessing maternal folate deficiency, pesticide exposure, or heavy metal contamination rely on retrospective self-reporting or ecological estimations, introducing recall bias and potential exposure misclassification. Furthermore, comprehensive studies that simultaneously assess genetic polymorphisms (such as MTHFR or VANGL1 variants) alongside quantified environmental exposures within the same cohorts are scarce. As a result, the full nature of gene–environment interactions in acrania and anencephaly remains incompletely characterized.
The reliance on first-trimester ultrasound as the primary modality for early detection introduces another limitation. Although expert-performed scans in tertiary referral centers report near-perfect sensitivity for acrania, substantial operator variability persists in community and rural settings, leading to diagnostic inconsistencies. Consequently, the real-world effectiveness of early detection strategies likely falls below the idealized rates reported in controlled research environments. This review’s discussion of imaging capabilities therefore reflects best-practice standards rather than global averages, and diagnostic access disparities remain a major challenge.
Finally, the ethical and cultural analyses presented, while informed by case reports and regional studies, inherently reflect specific socio-cultural contexts. They may not capture the full global diversity of ethical frameworks, particularly among indigenous populations, undocumented immigrant communities, and conflict-affected regions, where access to consistent prenatal care is irregular and biomedical norms may differ markedly.
Taken together, these strengths and limitations highlight that although this review represents the most comprehensive, multidisciplinary, and historically anchored analysis currently available, significant research gaps remain. Addressing these gaps – particularly through prospective, genetically and environmentally integrated cohort studies, the development of cross-cultural ethical counselling frameworks, and the expansion of affordable access to advanced imaging and genomic screening technologies in low-resource environments – will define the next frontier in the prevention and management of cranial neural tube defects.
Conclusions
The acrania–exencephaly–anencephaly sequence, once considered a static embryological anomaly, has become a model for understanding developmental failure at the intersection of genetics, environment, diagnostics, and ethics. From Saint-Hilaire’s early classifications to modern genomic discoveries involving VANGL1 and MTHFR, these anomalies trace the evolution of fetal medicine. Acrania’s biological origins are multifactorial, shaped by complex gene–environment interactions that demand a systems biology approach. No single intervention – whether folic acid fortification, genetic screening, or imaging – can fully prevent these conditions, requiring personalized risk prediction models instead. Despite technological advances in ultrasound and MRI, diagnostic disparities persist, particularly in low-resource settings, necessitating AI-driven standardization and broader access to imaging. Ethically, acrania and anencephaly force families into culturally and religiously influenced decisions, highlighting the need for counselling models that balance medical facts with ethical pluralism. Future progress will depend on building equitable, precision public health systems that integrate genomics, environmental monitoring, and proactive prenatal surveillance. Reducing the burden of cranial neural tube defects requires fusing molecular medicine with pragmatic global health strategies. This review urges interdisciplinary collaboration across fetal medicine, genetics, public health, and bioethics. The future of acrania prevention will be shaped by how boldly we embrace innovation, cultural sensitivity, and shared global responsibility.
Acknowledgments
The authors acknowledge the invaluable support of the Indonesian Society of Obstetrics & Gynecology (POGI) and Indonesian Society of Maternal-Fetal Medicine (HKFM) in facilitating this original article.
-
Research ethics: Not applicable.
-
Informed consent: Not applicable.
-
Author contributions: The authors have accepted responsibility for the entire content of this manuscript and approved its submission.
-
Use of Large Language Models, AI and Machine Learning Tools: None declared.
-
Conflict of interest: The authors state no conflict of interest.
-
Research funding: None declared.
-
Data availability: Not applicable.
References
1. Salari, N, Fatahi, B, Fatahian, R, Mohammadi, P, Rahmani, A, Darvishi, N, et al.. Global prevalence of congenital anencephaly: a comprehensive systematic review and meta-analysis. Reprod Health 2022;19:201. https://doi.org/10.1186/s12978-022-01509-4.Search in Google Scholar PubMed PubMed Central
2. Smith, GE. The papyrus ebers: the greatest Egyptian medical document. London: University College London Press; 1912. https://archive.org/details/ancientegyptianm0000unse_w1u0.Search in Google Scholar
3. Saint-Hilaire, E. Histoire Générale et Particulière des Anomalies de l’Organisation chez l’Homme et les Animaux. Paris: JB Baillière; 1825. https://books.google.com/books?id=GCKLtAEACAAJ.Search in Google Scholar
4. Copp, AJ, Stanier, P, Greene, ND. Neural tube defects: recent advances, unsolved questions, and controversies. Lancet Neurol 2013;12:799–810. https://doi.org/10.1016/S1474-4422(13)70110-8.Search in Google Scholar PubMed PubMed Central
5. Kancherla, V, Black, RE, Cherian, T, Barkley, A, Berry, RJ, Blencowe, H, et al.. A global update on the prevalence of neural tube defects for 2020: a systematic review and meta-analysis. Ann N Y Acad Sci 2023;1510:12–31. https://doi.org/10.1111/nyas.14847.Search in Google Scholar PubMed
6. Williams, J, Mai, CT, Mulinare, J, Isenburg, J, Flood, TJ, Ethen, M, et al.. Updated estimates of neural tube defects prevented by mandatory folic acid fortification–United States, 1995–2011. MMWR Morb Mortal Wkly Rep 2015;64:1–5. https://www.cdc.gov/mmwr/preview/mmwrhtml/mm6401a2.html.Search in Google Scholar
7. Crider, KS, Bailey, LB, Berry, RJ. Folic acid food fortification–Its history, effect, concerns, and future directions. Nutrients 2011;3:370–84. https://doi.org/10.3390/nu3030370.Search in Google Scholar PubMed PubMed Central
8. Botto, LD, Moore, CA, Khoury, MJ, Erickson, JD. Neural-tube defects. N Engl J Med 1999;341:1509–19. https://doi.org/10.1056/NEJM199911113412006.Search in Google Scholar PubMed
9. MRC Vitamin Study Research Group. Prevention of neural tube defects: results of the medical research council vitamin study. Lancet 1991;338:131–7. https://doi.org/10.1016/0140-6736(91)90133-A.Search in Google Scholar
10. Czeizel, AE, Dudas, I. Prevention of the first occurrence of neural-tube defects by periconceptional vitamin supplementation. N Engl J Med 1992;327:1832–5. https://doi.org/10.1056/NEJM199212243272602.Search in Google Scholar PubMed
11. Salih, MA, Murshid, WR, Seidahmed, MZ. Al-Ani ZR. Epidemiology, prenatal management, and prevention of neural tube defects. Saudi Med J 2014;35(Suppl 1):S15–28. https://smj.org.sa/content/smj/35/Supplement_1/S15.full.pdf.Search in Google Scholar
12. Greene, ND, Copp, AJ. Neural tube defects. Annu Rev Neurosci 2014;37:221–42. https://doi.org/10.1146/annurev-neuro-062012-170354.Search in Google Scholar PubMed PubMed Central
13. Blaas, HG, Eik-Nes, SH, Vainio, T, Isaksen, CV, Ulriksen, M, Rønnaug, S, et al.. Three-dimensional ultrasound for detailed examination of identified fetal anomalies. Ultrasound Obstet Gynecol 2000;15:177–85. https://doi.org/10.1046/j.1469-0705.2000.00073.x.Search in Google Scholar
14. Blom, HJ, Shaw, GM, den Heijer, M, Finnell, RH. Neural tube defects and folate: case far from closed. Nat Rev Neurosci 2006;7:724–31. https://doi.org/10.1038/nrn1986.Search in Google Scholar PubMed PubMed Central
15. Molloy, AM, Kirke, PN, Troendle, JF, Burke, H, Sutton, M, Brody, LC, et al.. Maternal vitamin B12 status and risk of neural tube defects in a population with high neural tube defect prevalence and no folic acid fortification. Pediatrics 2009;123:917–23. https://doi.org/10.1542/peds.2008-1173.Search in Google Scholar PubMed PubMed Central
16. Kibar, Z, Torban, E, McDearmid, JR, Reynolds, A, Berghout, J, Mathieu, M, et al.. Mutations in VANGL1 associated with neural-tube defects. N Engl J Med 2007;356:1432–7. https://doi.org/10.1056/NEJMoa060651.Search in Google Scholar PubMed
17. Viora, E, Sciarrone, A, Bastonero, S, Errante, G, Bonavita, G, Campogrande, M. Correlation between first trimester fetal malformations and chromosomal abnormalities by ultrasound examination and chorionic villus sampling. Prenat Diagn 2007;27:557–65. https://doi.org/10.1002/pd.1744.Search in Google Scholar PubMed
18. Moretti, ME, Bar-Oz, B, Chu, MW, Fried, S, Koren, G. Maternal hyperthermia and the risk for neural tube defects in offspring: systematic review and meta-analysis. Epidemiology 2005;16:216–19. https://doi.org/10.1097/01.ede.0000152903.55579.15.Search in Google Scholar PubMed
19. Lammer, EJ, Sever, LE, Oakley, GPJr. Teratogen update: valproic acid. Teratology 1987;35:465–73. https://doi.org/10.1002/tera.1420350409.Search in Google Scholar
20. Wyszynski, DF, Nambisan, M, Surve, T, Alsdorf, RM, Smith, CR, Holmes, LB. Increased rate of major malformations in offspring exposed to valproate during pregnancy. Neurology 2005;64:961–5. https://doi.org/10.1212/01.WNL.0000154516.43630.C5.Search in Google Scholar PubMed
21. Correa, A, Gilboa, SM, Besser, LM, Botto, LD, Moore, CA, Hobbs, CA, et al.. Diabetes mellitus and birth defects. Am J Obstet Gynecol 2008;199:237.e1–237.e9. https://doi.org/10.1016/j.ajog.2008.06.028.Search in Google Scholar PubMed PubMed Central
22. Padmanabhan, R. Etiology, pathogenesis and prevention of neural tube defects. Congenital Anom 2006;46:55–67. https://doi.org/10.1111/j.1741-4520.2006.00104.x.Search in Google Scholar PubMed
23. Bronshtein, M, Gover, A, Zimmer, EZ. First trimester diagnosis of fetal acrania using transvaginal sonography. J Clin Ultrasound 1991;19:396–9. https://doi.org/10.1002/jcu.1870190513.Search in Google Scholar PubMed
24. Glenn, OA. MR imaging of the fetal brain. Clin Perinatol 2009;36:869–88. https://doi.org/10.1016/j.clp.2009.08.011.Search in Google Scholar
25. Boyd, PA, Tondi, F, Hicks, NR, Chamberlain, PF. Autopsy after termination of pregnancy for fetal anomaly: retrospective study of 1000 cases. BMJ 2004;328:137. https://doi.org/10.1136/bmj.37952.582614.EE.Search in Google Scholar
26. Sumner, S, Kavanaugh, K, Moro, T. Extending palliative care into pregnancy and the immediate newborn period. J Perinat Neonatal Nurs 2006;20:113–16. https://doi.org/10.1097/00005237-200601000-00018.Search in Google Scholar PubMed
27. Cohen, AR, Couto, J, Cummings, JJ, Johnson, MP, Liechty, KW, Flake, AW, et al.. Fetal surgery for myelomeningocele: trial design and initial clinical experience at the university of Pittsburgh. J Neurosurg Pediatr 2015;16:404–10. https://doi.org/10.3171/2015.2.PEDS14517.Search in Google Scholar
28. Loane, M, Dolk, H, Kelly, A, Teljeur, C, Greenlees, R, Densem, J, et al.. Trends over time in the prevalence of major congenital anomalies in Europe: analysis of a large database of population-based registries, 1980 to 2012. Eur J Epidemiol 2019;34:521–31. https://doi.org/10.1007/s10654-018-0475-5.Search in Google Scholar
29. Zhang, T, Lou, J, Zhong, Y, Jin, L, Zheng, X. Prevalence and risk factors of neural tube defects in five counties in Shanxi province, China. Birth Defects Res 2021;113:207–16. https://doi.org/10.1002/bdr2.1821.Search in Google Scholar PubMed PubMed Central
30. Bhide, P, Sagoo, GS, Moorthie, S, Burton, H, Kar, A. Systematic review of birth prevalence of neural tube defects in India. Birth Defects Res A Clin Mol Teratol 2013;97:437–43. https://doi.org/10.1002/bdra.23145.Search in Google Scholar PubMed
31. Cavalli, L, Copp, AJ, Greene, ND. Environmental factors influencing prenatal development: neural tube defects and folate metabolism. Environ Res 2022;204:111948. https://doi.org/10.1016/j.envres.2021.111948.Search in Google Scholar PubMed PubMed Central
32. Ren, A, Qiu, X, Jin, L, Ma, J, Li, Z, Zhang, L, et al.. Association of selected persistent organic pollutants in the placenta with neural tube defects. Proc Natl Acad Sci U S A 2011;108:12770–5. https://doi.org/10.1073/pnas.1105209108.Search in Google Scholar PubMed PubMed Central
33. Tsuji, JS, Croft, J, Silva, M, Ray, M, Plunkett, LM, Prophete, C, et al.. Anencephaly: an overview of environmental risk factors and public health implications. Environ Health Perspect 2017;125:106–13. https://doi.org/10.1289/EHP158.Search in Google Scholar PubMed PubMed Central
34. Mall, FP. Developmental anomalies observed in human embryos. J Morphol 1908;19:1–28. https://doi.org/10.1002/jmor.1050190102.Search in Google Scholar
35. Levine, D, Barnes, PD, Madsen, JR, Abbott, J, Edelman, RR. Fetal central nervous system anomalies: MR imaging augments sonographic diagnosis. Radiology 1999;213:635–42. https://doi.org/10.1148/radiology.213.2.r99nv08635.Search in Google Scholar
36. Bronshtein, M, Zimmer, EZ. Exencephaly–anencephaly sequence: early diagnosis by transvaginal sonography and in utero evolution. Obstet Gynecol 1991;77:453–5. https://doi.org/10.1097/00006250-199103000-00020.Search in Google Scholar
37. Whitby, EH, Paley, MN, Davies, NP, Wilkinson, ID, Sprigg, A, Griffiths, PD. Ultrafast magnetic resonance imaging of the fetus: an increasing role in prenatal diagnosis. Lancet 2001;358:1131–4. https://doi.org/10.1016/S0140-6736(01)06145-3.Search in Google Scholar
38. Jurkovic, D, Gruboeck, K, Campbell, S. Ultrasound imaging of early pregnancy failure. Br Med Bull 1997;53:322–43. https://doi.org/10.1093/oxfordjournals.bmb.a011611.Search in Google Scholar PubMed
39. Greene, ND, Stanier, P, Copp, AJ. Genetics of human neural tube defects. Hum Mol Genet 2009;18:R113–29. https://doi.org/10.1093/hmg/ddp347.Search in Google Scholar PubMed PubMed Central
40. Lei, YP, Zhang, T, Li, H, Wu, BL, Jin, L, Wang, HY, et al.. VANGL2 mutations in human cranial neural-tube defects. N Engl J Med 2010;362:2232–5. https://doi.org/10.1056/NEJMoa0906940.Search in Google Scholar
41. Boyles, AL, Billups, AV, Deak, KL, Siegel, DG, Mehltretter, L, Slifer, SH, et al.. Neural tube defects and folate pathway genes: family-Based association tests of gene-gene and gene-environment interactions. Environ Health Perspect 2006;114:1547–52. https://doi.org/10.1289/ehp.9015.Search in Google Scholar
42. Stoll, C, Dott, B, Alembik, Y, Roth, MP. Associated malformations in cases with neural tube defects. Genet Couns 2018;29:235–51.Search in Google Scholar
43. Kennedy, D, Chitayat, D, Winsor, E, Silver, M, Toi, A. Prenatal detection and natural history of trisomy 13, trisomy 18, and trisomy 21: the 17-year experience of a single prenatal diagnosis center. Prenat Diagn 2015;35:821–9. https://doi.org/10.1002/pd.4603.Search in Google Scholar PubMed PubMed Central
44. Lemay, P, De Sandre-Giovannoli, A, Toutain, A, Gubler, MC, Benoit, G, Bieth, E, et al.. Whole-exome sequencing identifies novel candidate genes for congenital anomalies of the kidney and urinary tract. J Med Genet 2021;58:456–67. https://doi.org/10.1136/jmedgenet-2020-107267.Search in Google Scholar
45. Zhang, X, Li, S, Wu, S, Hao, Y, Xu, Y, Wang, H, et al.. Environmental exposure to heavy metals and risk for neural tube defects: a systematic review and meta-analysis. Ecotoxicol Environ Saf 2019;173:237–45. https://doi.org/10.1016/j.ecoenv.2019.02.063.Search in Google Scholar PubMed
46. Wool, C, Northam, S, Pinto, C, Kautz, D. Hospice and palliative care for pregnant women with a lethal fetal diagnosis: a systematic review. J Obstet Gynecol Neonatal Nurs 2018;47:613–23. https://doi.org/10.1016/j.jogn.2018.04.134.Search in Google Scholar PubMed PubMed Central
47. Brown, SD, Lyerly, AD, Little, MO. Prenatal decision making in religious contexts: a theoretical framework for understanding religious influences on maternal health care decisions. Perspect Biol Med 2012;55:396–407. https://doi.org/10.1353/pbm.2012.0037.Search in Google Scholar PubMed
48. Acharya, K, Richards, N, De Souza, R, Sah, N. Exploring social stigma faced by mothers of children with birth defects in rural Nepal. BMC Public Health 2017;17:762. https://doi.org/10.1186/s12889-017-4795-7.Search in Google Scholar
49. Detrait, ER, George, TM, Etchevers, HC, Gilbert, JR, Vekemans, M, Speer, MC. Human neural tube defects: developmental biology, epidemiology, and genetics. Neurotoxicol Teratol 2005;27:515–24. https://doi.org/10.1016/j.ntt.2004.12.007.Search in Google Scholar PubMed PubMed Central
50. World Health Organization (WHO). Birth defects surveillance: atlas of selected congenital anomalies. Geneva: WHO Press; 2014. https://apps.who.int/iris/handle/10665/110223.Search in Google Scholar
51. Kibar, Z, Capra, V, Gros, P. Toward understanding the genetic basis of neural tube defects. Clin Genet 2007;71:295–310. https://doi.org/10.1111/j.1399-0004.2007.00799.x.Search in Google Scholar PubMed
52. Blaas, HG, Eriksson, AG, Salvesen, KA, Isaksen, CV, Eik-Nes, SH. Visualization of the brain in first-trimester fetuses using transvaginal ultrasound. Ultrasound Obstet Gynecol 2009;33:620–7. https://doi.org/10.1002/uog.6369.Search in Google Scholar PubMed
53. Bachnas, MA, Andonotopo, W, Dewantiningrum, J, Adi Pramono, MB, Stanojevic, M, Kurjak, A. The utilization of artificial intelligence in enhancing 3D/4D ultrasound analysis of fetal facial profiles. J Perinat Med 2024;52:899–913. https://doi.org/10.1515/jpm-2024-0347.Search in Google Scholar PubMed
54. Andonotopo, W, Bachnas, MA, Dewantiningrum, J, Adi Pramono, MB, Stanojevic, M, Kurjak, A. AI and early diagnostics: mapping fetal facial expressions through development, evolution, and 4D ultrasound. J Perinat Med 2025;53:263–85. https://doi.org/10.1515/jpm-2024-0602.Search in Google Scholar PubMed
© 2025 the author(s), published by De Gruyter, Berlin/Boston
This work is licensed under the Creative Commons Attribution 4.0 International License.
Articles in the same Issue
- Frontmatter
- Reviews
- Fetal neurobehavior and consciousness: a systematic review of 4D ultrasound evidence and ethical challenges
- Prenatal maternal stress and long-term neurodevelopmental outcomes: a narrative review
- From exencephaly to anencephaly: a catastrophic continuum of neural tube defects from embryogenesis to ultrasonographic diagnosis
- Opinion Papers
- Fetoception: a window into maternal interoception?
- Rationale for the use of fetal ventriculosubgaleal shunts for the treatment of aqueduct stenosis
- Original Articles – Obstetrics
- The fetal occiput-spine angle measurement during first stage of labor as a predictor for vaginal delivery, a systematic review and meta-analysis
- Hemorrhage-related maternal morbidity of secondary compared to primary postpartum hemorrhage
- First-trimester maternal serum PAPP-A levels and hyperemesis gravidarum: unraveling the link – a meta-analysis
- Antepartum cerebroplacental ratio in low risk pregnancy and its relationship with adverse perinatal outcome: a prospective cohort study
- Liver fibrosis markers as predictors of adverse outcomes in pregnancy-related hypertensive disorders
- Endocrine disrupting chemicals: translating mechanisms into perinatal risk assessment
- Low uterine segment thickness in the prediction of cesarean delivery after induction of labor
- Original Articles – Fetus
- Fetal adrenal gland size in preeclamptic pregnancies with and without fetal growth restriction: an ultrasonographic assessment
- Evaluation of safety and performance of CentaFlow™ in the assessment of fetal growth restriction – a randomized trial and prospective cohort study
- Advantages of fully automated AI-enhanced algorithm (5D CNS+™) for generating a fetal neurosonogram in clinical routine
- Reference ranges for fetal tricuspid and mitral annular plane systolic excursions
- FetalDenseNet: multi-scale deep learning for enhanced early detection of fetal anatomical planes in prenatal ultrasound
- Original Articles – Neonates
- Incidence and mortality trends in congenital diaphragmatic hernia in the United States
Articles in the same Issue
- Frontmatter
- Reviews
- Fetal neurobehavior and consciousness: a systematic review of 4D ultrasound evidence and ethical challenges
- Prenatal maternal stress and long-term neurodevelopmental outcomes: a narrative review
- From exencephaly to anencephaly: a catastrophic continuum of neural tube defects from embryogenesis to ultrasonographic diagnosis
- Opinion Papers
- Fetoception: a window into maternal interoception?
- Rationale for the use of fetal ventriculosubgaleal shunts for the treatment of aqueduct stenosis
- Original Articles – Obstetrics
- The fetal occiput-spine angle measurement during first stage of labor as a predictor for vaginal delivery, a systematic review and meta-analysis
- Hemorrhage-related maternal morbidity of secondary compared to primary postpartum hemorrhage
- First-trimester maternal serum PAPP-A levels and hyperemesis gravidarum: unraveling the link – a meta-analysis
- Antepartum cerebroplacental ratio in low risk pregnancy and its relationship with adverse perinatal outcome: a prospective cohort study
- Liver fibrosis markers as predictors of adverse outcomes in pregnancy-related hypertensive disorders
- Endocrine disrupting chemicals: translating mechanisms into perinatal risk assessment
- Low uterine segment thickness in the prediction of cesarean delivery after induction of labor
- Original Articles – Fetus
- Fetal adrenal gland size in preeclamptic pregnancies with and without fetal growth restriction: an ultrasonographic assessment
- Evaluation of safety and performance of CentaFlow™ in the assessment of fetal growth restriction – a randomized trial and prospective cohort study
- Advantages of fully automated AI-enhanced algorithm (5D CNS+™) for generating a fetal neurosonogram in clinical routine
- Reference ranges for fetal tricuspid and mitral annular plane systolic excursions
- FetalDenseNet: multi-scale deep learning for enhanced early detection of fetal anatomical planes in prenatal ultrasound
- Original Articles – Neonates
- Incidence and mortality trends in congenital diaphragmatic hernia in the United States

