Ginkgetin inhibits proliferation of human leukemia cells via the TNF-α signaling pathway
Abstract
Ginkgetin is known to be an anticancer agent in many studies. However, its effectiveness in treating chronic lymphoblastic leukemia (CLL) remains unknown. The present study aimed to evaluate the effects of ginkgetin on the growth of the K562 cell line. The MTT assay was employed to examine the proliferation of K562, and a terminal deoxynucleotidyl transferase-mediated deoxyuridine triphosphate nick end labeling (TUNEL) staining was conducted to detect the apoptotic rates. Furthermore, changes of tumor necrosis factor-α (TNF-α) were detected by Western blot analysis. Ginkgetin inhibited the proliferation of K562 cells in a dose- and time-dependent manner. Concentrations of ginkgetin required to induce 50% death of K562 at 24, 48 and 72 h were 38.9, 31.3 and 19.2 μM, respectively. Moreover, treatment of ginkgetin increased K562 apoptosis in vitro along with increased levels of TNF-α. Interestingly, anti-TNF-α antibody prevented ginkgetin-induced K562 cell apoptosis and growth inhibition via deactivation of caspase-8, caspase-9 and caspase-3. Concomitantly, downregulation of TNF-α by etanercept in vivo attenuated ginkgetin-induced inhibitory effects on the tumor growth in an xenograft mouse model. Our results indicate that ginkgetin effectively inhibits K562 cell proliferation, and TNF-α plays a key role in ginkgetin-induced cell apoptosis.
1 Introduction
Chronic lymphoblastic leukemia (CLL) is one kind of leukemia, accounting for 25% of adult leukemia and 25% of non-Hodgkin’s lymphoma [1]. The incidence of CLL in the United States is 4.5 cases per 100,000 people, and the overall 5-year survival rate is 81.7% [1], [2]. Currently, the first-line therapy of CLL is induction of chemotherapy, such as continuous infusion of chlorambucil and fludarabine. For patients with high risk of relapse, hematopoietic stem cell transplantation is usually applied. However, chemotherapeutic therapy kills the tumor cells with an impairment of the immune system, and not all patients are able to tolerate aggressive therapies [3], [4]. Thus, it is critical to develop a novel therapeutic agent that is less toxic and more specific to CLL.
Tumor necrosis factor-α (TNF-α), produced by several types of cells, is a pleiotropic cytokine with important functions in the progression of inflammatory diseases [5]. Currently, several biologic agents targeting TNF-α have been approved to treat rheumatoid arthritis, inflammatory bowel disease, psoriasis, psoriatic arthritis and juvenile idiopathic arthritis [6]. The TNF-α/TNF-α receptor (TNF-R) signaling pathway affects the growth of tumor cells. Apoptosis, referred to as programmed cell death, is a basic manner to control the number of cells. It has been previously demonstrated that the interaction of TNF and TNF-R induces apoptosis through two distinct caspase-8 activation pathways [7], [8]. TNF-α stimulation causes apoptosis in tumor cells along with decreased levels of Bcl-2 and increased levels of Bax, cleaved caspase-3 and cleaved PARP [9]. Therefore, TNF-α increases apoptosis and causes growth inhibition of tumor cells.
A number of studies have suggested important roles of plant-derived active ingredients in the prevention of tumorigenesis. Ginkgo biloba has been used in medicine and as a source of food for a long time in Asia [10]. Ginkgetin is the active flavonoid isolated from the leaves and bark of G. biloba. In previous pharmacological reports [11], [12], [13], [14], ginkgetin was demonstrated to exert anti-inflammatory, antifungal, anti-influenza and neuroprotective activities. Interestingly, some reports showed ginkgetin effectively inhibits tumor growth both in vivo and in vitro, such as prostate cancer, osteosarcoma, medulloblastoma and ovarian adenocarcinoma [15], [16], [17], [18]. However, whether the antitumor effect of ginkgetin in solid tumor can be replicated in hematological malignancies has never been reported. Therefore, the present study was mainly conducted to investigate the effects of ginkgetin on the growth of human leukemia cells both in vivo and in vitro.
2 Materials and methods
2.1 Cell culture
The human CLL cell line K562 was obtained from the American Type Culture Collection (Manassas, VA, USA). The normal human peripheral blood mononuclear cells (PBMCs) were isolated from venous blood by the density gradient centrifugation method using Ficoll-Histopaque (Sigma-Aldrich, St. Louis, MO, USA). Informed consents were obtained from the donors of PBMCs, and ethical approval was obtained from the Ethics Committee of The First Affiliated Hospital of Zhejiang University in accordance with the Declaration of Helsinki. All cells were cultured in RPMI-1640 medium supplied with 10% fetal bovine serum, 100 U/mL penicillin, and 100 μg/mL streptomycin at 37°C in a humidified incubator containing 5% CO2.
2.2 MTT assay
To determine the cytotoxicity of ginkgetin (purity, >95%; Nanjing PuYi Biological Technology Co. Ltd., Nanjing, China), the cells (5×103 cells/well) were seeded in a 96-well plate. Drugs were added to the medium at various concentrations. Following 24, 48 and 72 h of incubation, 10 μL of MTT solution (Sigma-Aldrich, St. Louis, MO, USA) was added to the cell culture supernatant and incubated at 37°C for an additional 4 h. Finally, dimethyl sulfoxide (DMSO) (150 μL) was added to each well before the absorbance was measured at 570 nm. The cells’ viability was calculated as follows: [OD (drug)/OD (control)]×100%. The 50% inhibitory concentration (IC50) was calculated using SPSS17.0 (SPSS Inc., Chicago, IL, USA). Three replications of each experiment were performed.
2.3 TUNEL assay
K562 cell apoptosis was detected by the terminal deoxynucleotidyl transferase-mediated deoxyuridine triphosphate nick end labeling (TUNEL) assay with a TUNEL Bright Green Apoptosis Detection Kit (Vazyme Biotech Co. Ltd., Nanjing, China). The cells stained with 4′,6-diamidino-2-phenylindole (DAPI) (Beyotime, Shanghai, China) and fluorescein-12-dUTP-labeled DNA were visualized directly by fluorescence microscopy (TCS SP8, Leica Microsystems, Wetzlar, Germany). Average ratios of total TUNEL-positive cells were calculated from 10 random microscopic fields for each in all groups (magnification, ×400).
2.4 Caspase activity analysis
Caspases-3, caspase-8 and caspase-9 activities were measured using caspase colorimetric assay kits (KeyGen BioTECH, Nanjing, China) according to a previously published paper [19]. Briefly, the cells were treated with ginkgetin for 48 h, and then the cultured solution was centrifuged to remove the supernatant. Finally, the cells were washed by ice-cold phosphate-buffered saline (PBS). K562 cells were lysed by the lysis buffer, and the extracted protein was quantified using a BCA kit. The samples were mixed with reaction buffer, and caspase-3, caspase-8 and caspase-9 substrates were added, respectively. Optical density (OD) values were measured at 400 nm by a spectrophotometer. Caspase-3, caspase-8 and caspase-9 activities were evaluated as follows: activity=[OD (drug)/OD (control)].
2.5 Western blot analysis
K562 cells were treated with ginkgetin for 48 h, and cellular protein was extracted. Approximately 50 μg of protein was separated by electrophoresis on a 12% sodium dodecul sulfate (SDS)-polyacrylamide gel, transferred to a polyvinylidene difluoride (PVDF) membrane (Millipore, Bedford, MD, USA) and blocked with 3% bovine serum albumin (BSA). The membranes were further incubated with primary antibody of TNF-α (Santa Cruz Biotechnology, Santa Cruz, CA, USA) overnight at 4°C. After washing, the bands were visualized using enhanced chemiluminescence (ECL) detection reagents (Pierce Biotechnology, Rockford, IL, USA) according to the manufacturer’s instructions.
The quantification of proteins was analyzed by IPP software (Media Cybernetics, Rockville, MD, USA).
2.6 Neutralization assay in vitro
To determine the effects of TNF-α on growth inhibitory effects by ginkgetin, K562 cells were treated with different concentration of TNF-α polyclonal antibody (PeproTech, Rocky Hill, NJ, USA) and 40 μM ginkgetin in vitro for 48 h. Afterward, the growth rates, apoptosis rates and caspases activity were evaluated.
2.7 Antitumor activity assessment in vivo
Five-week-old male BALB/c nude mice were obtained from the animal department of Zhejiang University (Hangzhou, China). The mice were maintained under a 12-h light/dark cycle and housed under controlled temperature (25±1°C) conditions. The animals were allowed access to laboratory food and water ad libitum. Institutional and national guidelines for the care and use of laboratory animals were followed. Moreover, the experiments in vivo were approved by the Ethics Committee of The First Affiliated Hospital of Zhejiang University. To induce tumors, K562 cells were suspended in serum-free RPMI-1640 and implanted into the axillary skin of the mice. Tumor volume was measured by an electronic caliper and calculated with the following formula: volume=1/2×length×width2. Animals with tumors greater than 6–9 mm in diameter were selected and randomly grouped as following: vehicle control, ginkgetin (20 mg/kg), ginkgetin (20 mg/kg)+etanercept (6.25 mg/kg; Pfizer, Thousand Oaks, CA, USA), and ginkgetin (20 mg/kg)+etanercept (12.5 mg/kg). Ginkgetin was orally administrated to the mice daily for 2 consecutive weeks, while etanercept was given to the mice by intraperitoneal injection (once in 3 days). At the end of study, tumor-bearing mice were sacrificed, and the tumor masses were imaged and weighted.
2.8 Statistical analysis
All experiments in vitro were performed in triplicate and repeated three times. Data were expressed as the mean±standard deviation (SD). Student’s t-test and one-way analysis of variance (ANOVA) (17.0 SPSS software; SPSS Inc., Chicago, IL, USA) were used for the assessment of the difference in different groups. A P-value <0.05 was considered as statistically significant.
3 Results
3.1 Ginkgetin inhibits K562 cell growth in vitro
As seen in Figure 1A, ginkgetin significantly reduced the proliferation of K562 cells in a dose- and time-dependent manner (P<0.01). IC50 values of ginkgetin at 24, 48 and 72 h were 38.9, 31.3 and 19.2 μM, respectively. Moreover, we found ginkgetin had no toxic effects on PBMCs even with a high concentration (80.0 μM). These results demonstrate that ginkgetin with antileukemia concentration does not harm healthy cells.
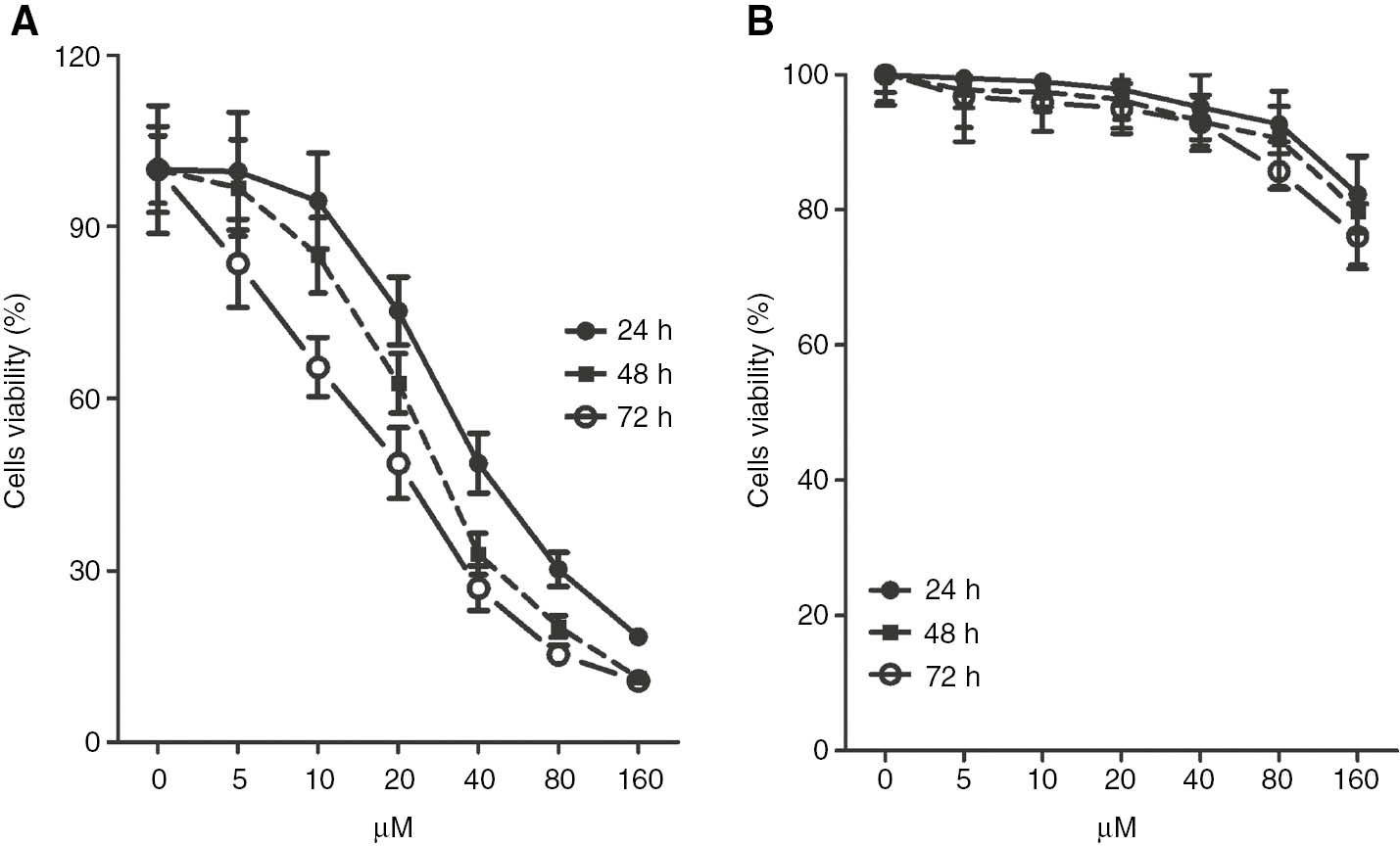
Effects of ginkgetin on the proliferation of K562 cells and PBMCs.
(A) K562 cells. (B) PBMCs. K562 cells and PBMCs were treated with increased concentrations of ginkgetin. Cell viability is expressed as percentages of survival to vehicle controls. The data are shown as mean±SD, n=3. Drug concentration resulting in 50% inhibition of growth (IC50) was determined on the basis of the nonlinear regression analysis from survival curves.
3.2 Ginkgetin increases K562 cell apoptosis in vitro
To study the effects of ginkgetin on apoptosis of K562 cells, the cells were treated with ginkgetin (10, 20 and 40 μM) for 48 h. DAPI/TUNEL staining was utilized to assess the apoptotic rate of K562 cells treated with ginkgetin. The results revealed that K562 cells treated with ginkgetin showed a higher rate of TUNEL-positive staining as compared with untreated cells (P<0.01; Figure 2). These data indicate ginkgetin obviously induces apoptosis in K562 cells.
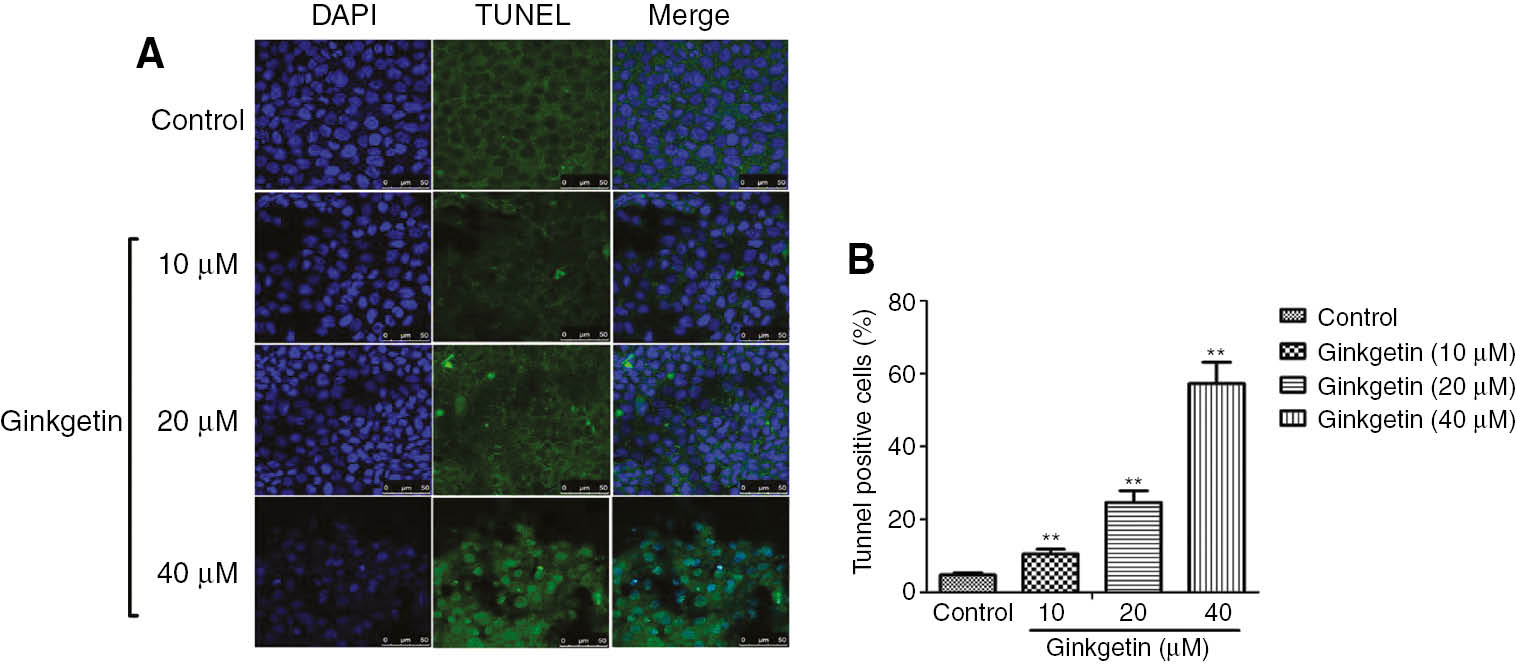
Effects of ginkgetin on the apoptosis of K562 cells.
(A) Apoptosis of K562 cells was detected by DAPI/TUNEL staining. (B) Apoptosis rates are quantified and expressed as TUNEL-positive rates in K562 cells. The data are shown as mean±SD, n=3. **P<0.01 as compared to the control (0 μM).
3.3 Ginkgetin increases TNF-α expression in K562 cells
TNF-α-mediated signaling pathway plays an important role in the proliferation of tumor cells. Here, we investigated whether ginkgetin affects the TNF-α level in K562 cells. The results showed K562 cells treated with ginkgetin for 24–72 h exhibited high levels of TNF-α, as compared with control cells (P<0.01; Figure 3).
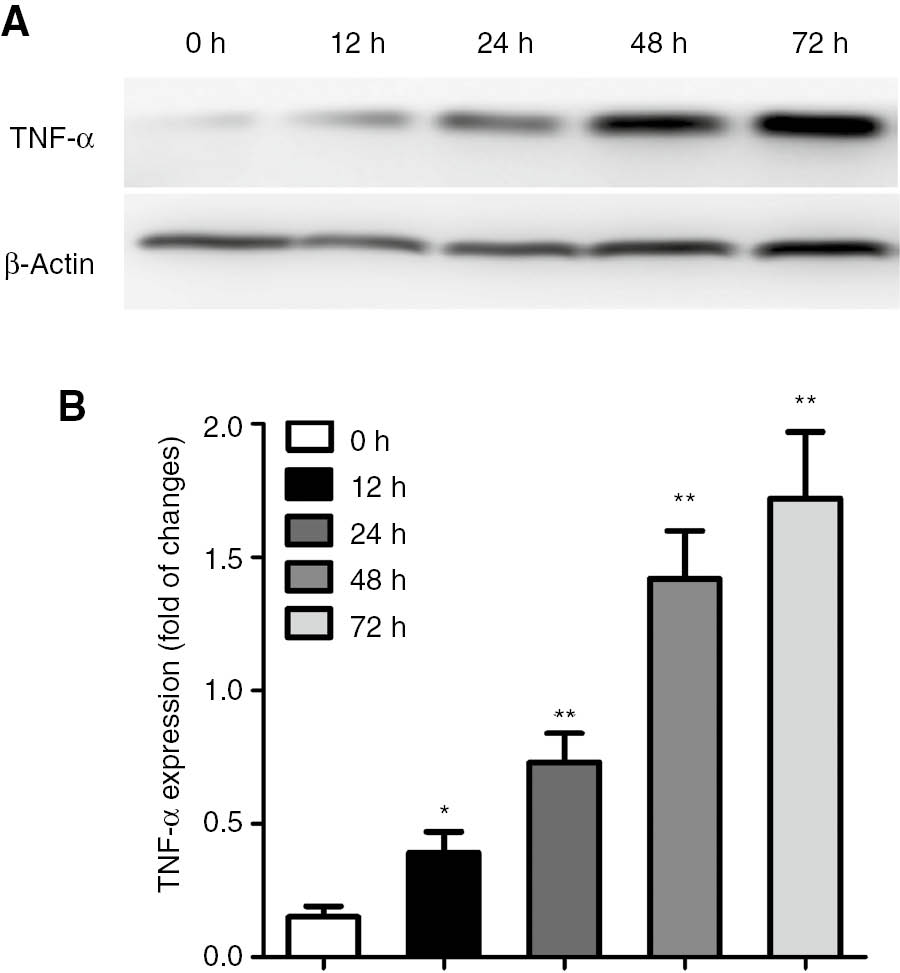
Effects of ginkgetin on the expression of TNF-α in K562 cells.
(A) The levels of TNF-α in K562 cells were measured by Western blotting. (B) Treatment with ginkgetin obviously increased TNF-α levels in K562 cells. The data are shown as mean±SD, n=3. *P<0.05, **P<0.01 as compared to the 0 h.
3.4 Neutralization of TNF-α attenuates inhibitory effects of ginkgetin on the growth of K562 cells
To determine the effects of TNF-α on ginkgetin-induced growth inhibition, we blocked the TNF-α signaling pathway using anti-TNF-α antibody. Our data showed that ginkgetin-induced growth inhibition and apoptosis could be significantly inhibited by increased levels of anti-TNF-α antibody (P<0.01; Figure 4A and B). Moreover, we observed ginkgetin treatment resulted in the activation of caspase-3, caspase-8 and caspase-9 in K562 cells (P<0.01; Figure 4C–E), which could be inhibited by TNF-α antibody (P<0.01; Figure 4C–E). Our results suggest ginkgetin activates both extrinsic and intrinsic apoptosis pathways in K562 cells.
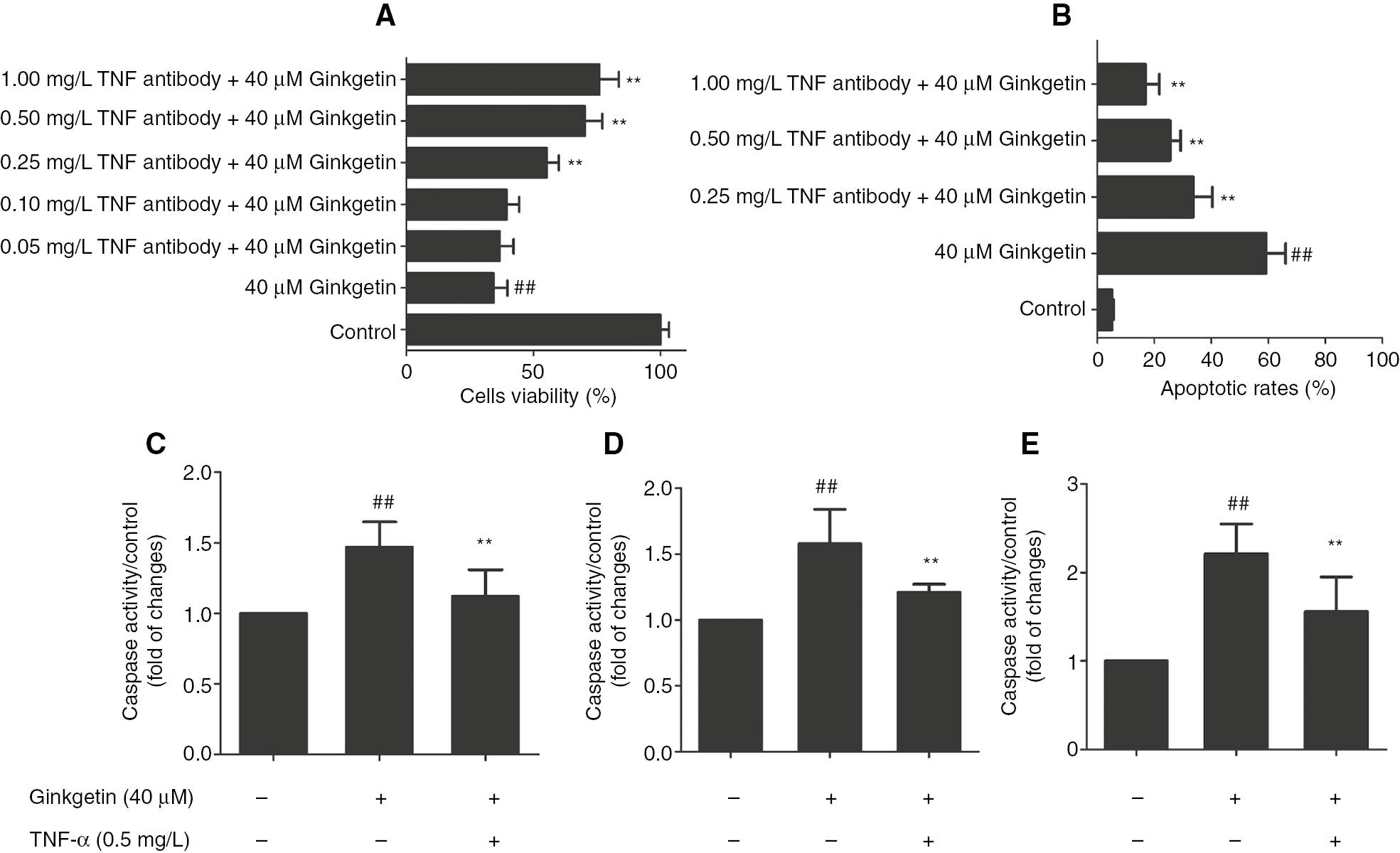
Effects of TNF-α-neutralizing antibody on the growth of ginkgetin-treated K562 cells.
(A) Viability of K562 cells was measured by MTT assay. (B) Apoptosis rates of K562 cells were measured by DAPI/TUNEL staining. (C) Caspase-3, (D) caspase-8 and (E) caspase-9 activities of K562 cells. The data are shown as mean±SD, n=3. ##P<0.01 as compared to control; **P<0.01 as compared to ginkgetin.
3.5 Etanercept attenuates anti-CLL activity of ginkgetin in a xenograft mouse model
To investigate anti-CLL effects of ginkgetin in vivo, K562 cells were subcutaneously transplanted into nude mice. As shown in Figure 5, ginkgetin treatment significantly suppressed tumor growth in the mice transplanted with K562 cells (P<0.01; Figure 5). Moreover, we found that the mice that received etanercept and ginkgetin showed a greater size of tumor tissue as compared to the mice treated with ginkgetin alone (P<0.01; Figure 5). Undoubtedly, reduction in the TNF-α expression attenuates inhibitory effects of ginkgetin on K562 growth in vivo.
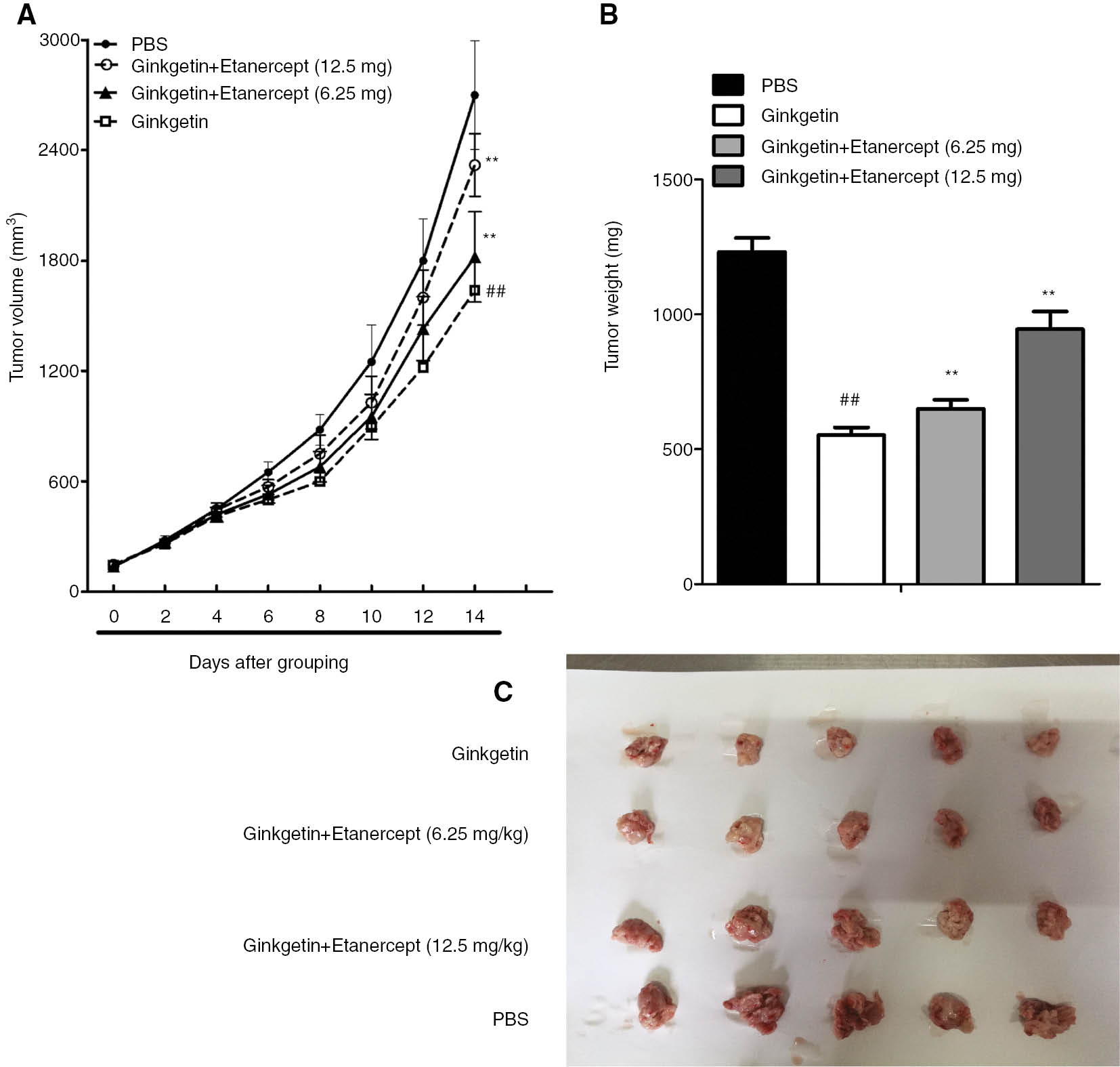
Effects of etanercept and ginkgetin on K562 growth in a xenograft mouse model.
(A) Tumor volume. (B) Tumor weight. The mice implanted with K562 cells were treated with ginkgetin and etanercept. Tumors were removed at 2 weeks after drug administration. The images of tumor tissues are showed in (C). The data are shown as mean±SD, n=5. ##P<0.01 as compared to control (PBS); **P<0.01 as compared to ginkgetin (20 mg/kg).
4 Discussion
Ginkgetin, as the major active constituents in G. biloba leaves, possesses several antitumor properties, such as in prostate cancer, osteosarcoma, medulloblastoma and ovarian adenocarcinoma [15], [16], [17], [18]. However, whether ginkgetin could effectively inhibit CLL growth has not been studied. In the present study, we found ginkgetin suppressed the growth of K562 cells in a dose- and time-dependent manner and induced apoptosis in K562 cells. In previous reports, ginkgetin was reported to inhibit human ovarian adenocarcinoma cells with 50% inhibition occurring at 1.8 mg/mL [20]. Moreover, the concentrations of ginkgetin required to induce 50% death in HeLa and FS-5 were 5.2 and 8.3 mg/mL, respectively [18]. For inhibiting medulloblastoma cells, the IC50 value was 14.65 μM [21]. In our research, we found the concentration of ginkgetin required to induce 50% death in K562 cells varied from 19.2 μM (10.86 mg/L) to 38.9 μM (22.0 mg/L). These data suggest K562 cells are more sensitive than HeLa and FS-5 in response to ginkgetin treatment. Moreover, the toxic effects of ginkgetin on human PBMCs are very low, even with a high concentration (80.0 μM). In in vivo research, ginkgetin was proved to reduce tumor size in a K562 transplanted mouse model. Ginkgetin treatment exhibited high selectivity to K562 cells rather than PBMCs, but we did not focus on the side effects of ginkgetin based on animal welfare in the present study. It is well known that side effects of the drug in tumor-bearing animals are affected by several factors, such as susceptibility differences in tumor and normal cells, activity, pharmacokinetic differences, and distribution of drug in the tumor tissue. Therefore, there is a continual need to identify side effects of ginkgetin in vivo, compared with that of first-line therapy of CLL, such as chlorambucil or fludarabine.
Apoptosis is the main form of cell death and served as a potential target for preventing the proliferation of malignant tumor cells [22]. Apoptosis can be triggered through either the extrinsic or intrinsic pathway. Briefly, the extrinsic pathway begins with the stimulation of death receptors on the cell membrane, and the intrinsic pathway is initiated by the dysfunction of mitochondria. Death receptors are members of the TNF-R superfamily. The receptors activate extrinsic apoptotic pathways in response to extracellular death signals, such as TNF-α and Fas ligand stimulation. The extrinsic pathway is activated along with caspase-8, which directly cleaves and activates the effector caspase-3. In the intrinsic pathway, a variety of extra- and intracellular stresses, such as oxidative stress and cytotoxic drug treatment, promote activation of caspase-9, followed by cleaving and activation of caspase-3 [23]. The strategy to specifically activate the extrinsic pathway in malignant cells is attractive for cancer therapy. Unfortunately, the clinical application of TNF-α and Fas is hampered by severe side effects [24], [25]. In previous investigations, it was shown that apoptosis induced by ginkgetin is mediated through activation of caspases [18]. Moreover, several studies found ginkgetin inhibited signal transducer and activator of transcription 3 (STAT3) signaling pathway by induction of SHP-1 and phosphatase and tensin homolog (PTEN) proteins, thus attenuating STAT3 phosphorylation and tumorigenesis [15], [16], [26]. However, to the best of our knowledge, the underlying mechanisms of ginkgetin-induced apoptosis in CLL cells remain as yet unidentified. In the present study, we observed neutralization of TNF-α by antibody, and blocking the TNF-α signaling pathway by etanercept significantly attenuates ginkgetin-induced inhibitory effects on K562 cell growth both in vitro and in vivo. Therefore, our results indicate ginkgetin inhibits K562 cell proliferation via modulating of the TNF-α signaling pathway.
In conclusion, our results provide novel insights into the anti-CLL activity of ginkgetin, and supply a theoretical basis for its clinical application. Moreover, ginkgetin-induced growth inhibition of K562 cells may be the result of activating the TNF-α pathway. Our conclusion further enriches the antitumor mechanism of ginkgetin.
Acknowledgments
This work was supported by the Natural Science Foundation of China (91429302).
Disclosure of interest: The authors report no conflicts of interest.
References
1. Rai KR, Jain P. Chronic lymphocytic leukemia (CLL) – then and now. Am J Hematol 2016;91:330–40.10.1002/ajh.24282Suche in Google Scholar PubMed
2. Hallek M, Cheson BD, Catovsky D, Caligaris-Cappio F, Dighiero G, Döhner H, et al. International Workshop on chronic lymphocytic leukemia. Guidelines for the diagnosis and treatment of chronic lymphocytic leukemia: a report from the International Workshop on chronic lymphocytic leukemia updating the National Cancer Institute-Working Group 1996 guidelines. Blood 2008;111:5446–56.10.1182/blood-2007-06-093906Suche in Google Scholar PubMed PubMed Central
3. Hallek M. Chronic lymphocytic leukemia: 2015 update on diagnosis, risk stratification, and treatment. Am J Hematol 2015;90:446–60.10.1002/ajh.23979Suche in Google Scholar PubMed
4. Smolewski P, Witkowska M, Korycka-Wołowiec A. New insights into biology, prognostic factors, and current therapeutic strategies in chronic lymphocytic leukemia. ISRN Oncol 2013;2013:740615.10.1155/2013/740615Suche in Google Scholar PubMed PubMed Central
5. Olmos G, Lladó J. Tumor necrosis factor alpha: a link between neuroinflammation and excitotoxicity. Mediators Inflamm 2014;2014:861231.10.1155/2014/861231Suche in Google Scholar PubMed PubMed Central
6. Kalliolias GD, Ivashkiv LB. TNF biology, pathogenic mechanisms and emerging therapeutic strategies. Nat Rev Rheumatol 2016;12:49–62.10.1038/nrrheum.2015.169Suche in Google Scholar PubMed PubMed Central
7. Wang CY, Mayo MW, Korneluk RG, Goeddel DV, Baldwin AS Jr. NF-kappaB antiapoptosis: induction of TRAF1 and TRAF2 and c-IAP1 and c-IAP2 to suppress caspase-8 activation. Science 1998;281:1680–3.10.1126/science.281.5383.1680Suche in Google Scholar PubMed
8. Wang L, Du F, Wang X. TNF-alpha induces two distinct caspase-8 activation pathways. Cell 2008;133:693–703.10.1016/j.cell.2008.03.036Suche in Google Scholar PubMed
9. Chen D, Liu J, Lu L, Huang Y, Wang Y, Wang M, et al. Emodin attenuates TNF-α-induced apoptosis and autophagy in mouse C2C12 myoblasts though the phosphorylation of Akt. Int Immunopharmacol 2016;34:107–13.10.1016/j.intimp.2016.02.023Suche in Google Scholar PubMed
10. Isah T. Rethinking Ginkgo biloba L.: medicinal uses and conservation. Pharmacogn Rev 2015;9:140–8.10.4103/0973-7847.162137Suche in Google Scholar PubMed PubMed Central
11. Wang YQ, Wang MY, Fu XR, Peng-Yu, Gao GF, Fan YM, et al. Neuroprotective effects of ginkgetin against neuroinjury in Parkinson’s disease model induced by MPTP via chelating iron. Free Radic Res 2015;49:1069–80.10.3109/10715762.2015.1032958Suche in Google Scholar PubMed
12. Zhou HF, Xie C, Jian R, Kang J, Li Y, Zhuang CL, et al. Biflavonoids from caper (Capparis spinosa L.) fruits and their effects in inhibiting NF-kappa B activation. J Agric Food Chem 2011;59:3060–5.10.1021/jf105017jSuche in Google Scholar PubMed
13. Park H, Kim YH, Chang HW, Kim HP. Anti-inflammatory activity of the synthetic C-C biflavonoids. J Pharm Pharmacol 2006;58:1661–7.10.1211/jpp.58.12.0014Suche in Google Scholar PubMed
14. Miki K, Nagai T, Suzuki K, Tsujimura R, Koyama K, Kinoshita K, et al. Anti-influenza virus activity of biflavonoids. Bioorg Med Chem Lett 2007;17:772–5.10.1016/j.bmcl.2006.10.075Suche in Google Scholar PubMed
15. Xiong M, Wang L, Yu HL, Han H, Mao D, Chen J, et al. Ginkgetin exerts growth inhibitory and apoptotic effects on osteosarcoma cells through inhibition of STAT3 and activation of caspase-3/9. Oncol Rep 2016;35:1034–40.10.3892/or.2015.4427Suche in Google Scholar PubMed
16. Jeon YJ, Jung SN, Yun J, Lee CW, Choi J, Lee YJ, et al. Ginkgetin inhibits the growth of DU-145 prostate cancer cells through inhibition of signal transducer and activator of transcription 3 activity. Cancer Sci 2015;106:413–20.10.1111/cas.12608Suche in Google Scholar PubMed PubMed Central
17. You OH, Kim SH, Kim B, Sohn EJ, Lee HJ, Shim BS, et al. Ginkgetin induces apoptosis via activation of caspase and inhibition of survival genes in PC-3 prostate cancer cells. Bioorg Med Chem Lett 2013;23:2692–5.10.1016/j.bmcl.2013.02.080Suche in Google Scholar PubMed
18. Su Y, Sun CM, Chuang HH, Chang PT. Studies on the cytotoxic mechanisms of ginkgetin in a human ovarian adenocarcinoma cell line. Naunyn Schmiedebergs Arch Pharmacol 2000;362: 82–90.10.1007/s002100000240Suche in Google Scholar PubMed
19. Huang J, Peng K, Wang L, Wen B, Zhou L, LuoT, et al. Ginsenoside Rh2 inhibits proliferation and induces apoptosis in human leukemia cells via TNF-α signaling pathway. Acta Biochim Biophys Sin (Shanghai) 2016;48:750–5.10.1093/abbs/gmw049Suche in Google Scholar PubMed
20. Sun CM, Syu WJ, Huang YT, Chen CC, Ou JC. Selective cytotoxicity of ginkgetin from Selaginella moellendorffii. J Nat Prod 1997;60:382–4.10.1021/np960608eSuche in Google Scholar PubMed
21. Ye ZN, Yu MY, Kong LM, Wang WH, Yang YF, Liu JQ, et al. Biflavone ginkgetin, a novel Wnt inhibitor, suppresses the growth of medulloblastoma. Nat Prod Bioprospect 2015;5:91–7.10.1007/s13659-015-0056-4Suche in Google Scholar PubMed PubMed Central
22. Guicciardi ME, Gores GJ. Life and death by death receptors. FASEB J 2009;23:1625–37.10.1096/fj.08-111005Suche in Google Scholar PubMed PubMed Central
23. Ren X, Zhang Y, Li C, Wang H, Jiang Z, Zhang Z, et al. Enhancement of baicalin by hexamethylene bisacetamide on the induction of apoptosis contributes to simultaneous activation of the intrinsic and extrinsic apoptotic pathways in human leukemia cells. Oncol Rep 2013;30:2071–80.10.3892/or.2013.2684Suche in Google Scholar PubMed
24. Walczak H, Krammer PH. The CD95 (APO-1/Fas) and the TRAIL (APO-2L) apoptosis systems. Exp Cell Res 2000;256:58–66.10.1006/excr.2000.4840Suche in Google Scholar PubMed
25. Ashkenazi A, Pai RC, Fong S, Leung S, Lawrence DA, Marsters SA, et al. Safety and antitumor activity of recombinant soluble Apo2 ligand. J Clin Invest 1999;104:155–62.10.1172/JCI6926Suche in Google Scholar PubMed PubMed Central
26. Baek SH, Lee JH, Ko JH, Lee H, Nam D, Lee SG, et al. Ginkgetin blocks constitutive STAT3 activation and induces apoptosis through induction of SHP-1 and PTEN tyrosine phosphatases. Phytother Res 2016;30:567–76.10.1002/ptr.5557Suche in Google Scholar PubMed
©2017 Walter de Gruyter GmbH, Berlin/Boston
Artikel in diesem Heft
- Frontmatter
- Ginkgetin inhibits proliferation of human leukemia cells via the TNF-α signaling pathway
- Effects of a water extract of Lepidium meyenii root in different models of persistent pain in rats
- Interaction of tubulin and protein kinase CK2 in Trypanosoma equiperdum
- Synthesis, biological activity and molecular modeling study of new Schiff bases incorporated with indole moiety
- A dihydrochalcone derivative and further steroidal saponins from Sansevieria trifasciata Prain
- Chemical composition and biological activities of leaf and fruit essential oils from Eucalyptus camaldulensis
- Fungal production of the polysaccharide pullulan from a plant hydrolysate
- Acetogenins and alkaloids during the initial development of Annona muricata L. (Annonaceae)
- Corrigendum
- Corrigendum to: Ginkgetin inhibits proliferation of human leukemia cells via the TNF-α signaling pathway
Artikel in diesem Heft
- Frontmatter
- Ginkgetin inhibits proliferation of human leukemia cells via the TNF-α signaling pathway
- Effects of a water extract of Lepidium meyenii root in different models of persistent pain in rats
- Interaction of tubulin and protein kinase CK2 in Trypanosoma equiperdum
- Synthesis, biological activity and molecular modeling study of new Schiff bases incorporated with indole moiety
- A dihydrochalcone derivative and further steroidal saponins from Sansevieria trifasciata Prain
- Chemical composition and biological activities of leaf and fruit essential oils from Eucalyptus camaldulensis
- Fungal production of the polysaccharide pullulan from a plant hydrolysate
- Acetogenins and alkaloids during the initial development of Annona muricata L. (Annonaceae)
- Corrigendum
- Corrigendum to: Ginkgetin inhibits proliferation of human leukemia cells via the TNF-α signaling pathway

