Abstract
A polypeptide band with an apparent molecular weight of 55,000 was phosphorylated in vitro in whole-cell lysates of Trypanosoma equiperdum. This band corresponds to tubulin as demonstrated by immunoprecipitation of the phosphorylated polypeptide from T. equiperdum extracts when anti-α and anti-β tubulin monoclonal antibodies were employed. A parasite protein kinase CK2 was in charge of modifying tubulin given that common mammalian CK2 inhibitors such as emodin and GTP, hindered the phosphorylation of tubulin and exogenously added casein. Interestingly, a divalent cation-dependent translocation of the T. equiperdum tubulin and the CK2 responsible for its phosphorylation was noticed, suggesting a direct interaction between these two proteins. Additionally, this fraction of tubulin and its kinase coeluted using separations based on parameters as different as charge (DEAE-Sepharose anion-exchange chromatography) and size (Sephacryl S-300 gel filtration chromatography). Analyses by non-denaturing polyacrylamide gel electrophoresis and immunoblot of the purified and radioactively labeled fraction containing both tubulin and the CK2 enzyme, established the phosphorylation of a single band that was recognized by anti-CK2 α-subunit and anti-tubulin antibodies. All these findings revealed a physical association between a pool of tubulin and a CK2 in T. equiperdum.
1 Introduction
Trypanosoma equiperdum is the etiological vehicle of dourine, a venereal disease of horses and other equids that is characterized by inflammation of the genitalia, cutaneous plaques and neurological signs [1]. Trypanosoma equiperdum belongs to the subgenus Trypanozoon and is very closely related to Trypanosoma brucei and Trypanosoma evansi [2].
Microtubules are structures composed of α- and β-tubulin dimers. Besides being the key constituents of the flagellum, the mitotic spindle and the microtubule-organizing centers in trypanosomes, these polymers are implicated in the assembly of the protozoan cytoskeleton [3]. CK2 (previously known as casein kinase II) is a serine/threonine protein kinase that belongs to the CMGC group [4]. Mammalian CK2s are heterotetramers built of two α and/or α’ catalytic subunits that are bound to two β regulatory subunits. We have formerly reported that the major phosphorylated polypeptide in whole-cell homogenates of the TeAp-N/D1 strain of T. equiperdum (aka TEVA1) corresponded to a 55-kDa band [5]. This band was recognized by specific anti-α and anti-β tubulin monoclonal antibodies; and inhibition experiments using emodin, heparin and 2,3-bisphosphoglycerate, three well-known mammalian CK2 inhibitors, indicated the involvement of a CK2 enzyme in the phosphorylation of the 55-kDa polypeptide band [5]. Here, we established a physical association between a pool of tubulin and a CK2 enzyme in T. equiperdum.
2 Experimental
2.1 Materials
Reagents were purchased from the following sources: [γ-32P] adenosine-5′-triphosphate (ATP) (3000 Ci/mmol), New England Nuclear (Wellesley, MA, USA) or Amersham (Pittsburgh, PA, USA); rabbit polyclonal anti-human CK2 α-subunit antibodies directed against the conserved sequence delimited by amino acids 74–89, Calbiochem (San Diego, CA, USA); dephosphorylated casein, emodin, guanosine-5′-triphosphate (GTP), benzamidine, leupeptin, L-trans-epoxysuccinyl-leucylamido(4-guanidino)butane (E-64), phenylmethylsulfonylfluoride (PMSF), fibrous diethylaminoethyl (DEAE)-cellulose, mouse monoclonal anti-chicken embryo brain α-tubulin antibodies (Clone DM1A), mouse monoclonal anti-rat brain β-tubulin antibodies (Clone TUB 2.1), anti-rabbit immunoglobulin G (IgG) (whole molecule) alkaline phosphatase conjugate, anti-mouse IgG (whole molecule) alkaline phosphatase conjugate, Sigma (Saint Louis, MO, USA); immunoglobulin-binding protein G-coupled agarose, Gibco BRL (Grand Island, NY, USA); P81 phosphocellulose chromatography paper, Whatman (Pittsburgh, PA, USA); polyvinylidene difluoride (PVDF) membranes, Millipore Corporation (Billerica, MA, USA); DEAE-Sepharose, Sephacryl S-300, Pharmacia (Pittsburgh, PA, USA). All other chemicals were of the highest quality grade available.
2.2 Parasites
Sprague-Dawley rats were infected with cryopreserved T. equiperdum parasites from the Venezuelan TeAp-N/D1 strain (TEVA1) [6]. When the number of parasites reached ~107 trypanosomes/mL of blood, the rats were bled by cardiac puncture using 2% EDTA as an anticoagulant. Trypanosomes were purified by anion-exchange chromatography using a fibrous DEAE-cellulose column [7].
2.3 Homogenization of T. equiperdum parasites and preparation of parasite soluble and particulate fractions
Trypanosoma equiperdum parasites (~107) were extracted in the presence of either divalent cations or chelating agents [8] and the corresponding soluble and particulate fractions were separated by centrifugation at 100,000×g, for 1 h, at 4°C.
2.4 In vitro protein kinase assay
Endogenous kinase and casein kinase activities were assayed according to De Lima et al. [8] by using liquid-scintillation counting and autoradiography following sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE). The effect of two CK2 inhibitors, emodin (100 μM) and GTP (150 μM), was evaluated in the absence or presence of casein.
2.5 Immunoprecipitation
The soluble fraction obtained after parasite homogenization in the presence of chelating agents (500 μg of total protein) was phosphorylated with [γ-32P] ATP. The sample was incubated with a mixture of anti-α and anti-β tubulin monoclonal antibodies (1/100 dilution each) for 1 h, at 4°C and the immune complex was collected by centrifugation using protein G-agarose beads. Controls were carried out using pre-immune mouse serum or a polyclonal antibody directed against the soluble form of a variant surface glycoprotein (VSG) purified from the TeAp-N/D1 strain of T. equiperdum [9].
2.6 Purification of tubulin and the kinase responsible for its phosphorylation from T. equiperdum
Trypanosoma equiperdum parasites (~1010) were lysed on ice by sonication with 50 mM Tris-HCl (pH 8.0), 10 mM EGTA, 10 mM EDTA, 50 μM PMSF, 10 μM leupeptin, 10 μM E-64 and 1 mM benzamidine. After centrifugation at 100,000×g, for 1 h, at 4°C, the soluble fraction was loaded onto a DEAE-Sepharose anion-exchange column (30 mL) that had been equilibrated with 50 mM Tris-HCl (pH 8.0), 0.1 mM EDTA, 2 mM EGTA, 0.2 M NaCl, 50 μM PMSF, 10 μM leupeptin, 10 μM E-64 and 1 mM benzamidine. Proteins were eluted with a linear gradient from 0 to 2 M NaCl in the same buffer.
The tubulin- and tubulin kinase-enriched fractions eluting from the DEAE-Sepharose were pooled, concentrated and applied to a Sephacryl S-300 size-exclusion column (36 mL) previously equilibrated with 50 mM Tris-HCl (pH 8.0), 150 mM NaCl, 5 mM β-mercaptoethanol and protease inhibitors (50 μM PMSF, 10 μM leupeptin, 10 μM E-64 and 1 mM benzamidine). The column was run at a flow rate of 150 μL/min.
2.7 Other procedures
Protein concentration was determined using bovine serum albumin as protein standard [10]. SDS-PAGE was carried out on 1.5 mm thick slab gels containing 12% polyacrylamide [11]. Non-denaturing gel electrophoresis was performed on 0.75 mm thick, linear 2%–15% polyacrylamide gradient slab gels [12]. Coomassie Blue R-250 or silver staining was used for protein visualization on gels. For western blot analyses, proteins separated by SDS-PAGE or native electrophoresis were electrotransferred from the gels to PVDF sheets [13].
3 Results
The major phosphorylated polypeptide in whole-cell lysates of T. equiperdum corresponded to a 55-kDa polypeptide band (Figure 1). Exogenously added casein was clearly phosphorylated, demonstrating the presence of casein kinases in T. equiperdum (Figure 1). Inhibition experiments in the presence of two well-known mammalian protein kinase CK2 inhibitors, emodin and GTP, indicated the participation of a CK2 enzyme in the phosphorylation of both the 55-kDa polypeptide band and casein (Figure 1).
![Figure 1: SDS-PAGE separation of whole-cell lysates from T. equiperdum. CBs, polypeptide profile revealed by Coomassie Blue staining. Shown are parasite homogenates in the absence (1) or presence of dephosphorylated casein (2). In vitro kinase activity was assayed in the presence of [γ-32P] ATP and the labeled gel was dried and analyzed by autoradiography (Autorad.). Shown are the endogenously phosphorylated bands in the absence (1), or in the presence of either 100 μM emodin (7) or 150 μM GTP (8). Control experiments containing just the vehicles used to dissolve emodin (0.01 M NaOH and 0.1% dimethyl sulfoxide) and GTP [50 mM Tris-HCl (pH 8.0)] were included in lanes 2 and 3, respectively. Casein kinase activity was determined by including 1 mg/mL of dephosphorylated casein to the reaction mixtures, in the absence (4) or presence of either 100 μM emodin (5) or 150 μM GTP (6).](/document/doi/10.1515/znc-2017-0019/asset/graphic/j_znc-2017-0019_fig_001.jpg)
SDS-PAGE separation of whole-cell lysates from T. equiperdum. CBs, polypeptide profile revealed by Coomassie Blue staining. Shown are parasite homogenates in the absence (1) or presence of dephosphorylated casein (2). In vitro kinase activity was assayed in the presence of [γ-32P] ATP and the labeled gel was dried and analyzed by autoradiography (Autorad.). Shown are the endogenously phosphorylated bands in the absence (1), or in the presence of either 100 μM emodin (7) or 150 μM GTP (8). Control experiments containing just the vehicles used to dissolve emodin (0.01 M NaOH and 0.1% dimethyl sulfoxide) and GTP [50 mM Tris-HCl (pH 8.0)] were included in lanes 2 and 3, respectively. Casein kinase activity was determined by including 1 mg/mL of dephosphorylated casein to the reaction mixtures, in the absence (4) or presence of either 100 μM emodin (5) or 150 μM GTP (6).
Galán-Caridad et al. [5] have shown that both the phosphorylated 55-kDa band and the kinase responsible for its phosphorylation, predominantly remained in the parasite particulate fraction when 1 mM CaCl2 and 1mM MgCl2 were included in the homogenization buffer. As the presence or absence of divalent cations affected the solubilization of a pool of tubulin and the CK2 responsible for its phosphorylation in Trypanosoma cruzi epimastigotes [8], an analogous experiment was performed here with T. equiperdum. When T. equiperdum parasites were homogenized in the presence of both 5 mM CaCl2 and 5 mM MgCl2, the kinase apparently responsible for phosphorylating the 55-kDa band, as well as the pool of phosphorylated 55-kDa polypeptide, remained associated with the parasite particulate fraction (Figure 2, buffer A). However, the kinase activity and its phosphorylated substrate were released into the cytosolic fraction when T. equiperdum parasites were extracted in the presence of 10 mM EDTA and 10 mM EGTA (Figure 2, buffer B). Similar results were obtained on the phosphorylation of casein when this artificial substrate was included in the reaction mixtures (data not shown). The use of a mixture of anti-α and anti-β tubulin monoclonal antibodies demonstrated the same cation-dependent solubilization of tubulin (Figure 2C). These results revealed a differential cellular compartmentalization of the pools of both proteins depending on the presence or absence of divalent cations and suggested that the 55-kDa band and the enzymatic activity in charge of its phosphorylation might correspond to tubulin and protein kinase CK2, respectively, as previously reported for T. cruzi epimastigotes [14, 15].
![Figure 2: Effect of divalent cations and chelating agents on the solubility of tubulin and tubulin kinase from T. equiperdum. Whole-cell homogenates (H), soluble (S) and particulate (P) fractions were obtained from T. equiperdum parasites extracted with either 50 mM Tris-HCl (pH 8.0), 5 mM MgCl2 and 5 mM CaCl2 (buffer A) or 50 mM Tris-HCl (pH 8.0), 10 mM EGTA and 10 mM EDTA (buffer B) and separated by SDS-PAGE. Panel A shows the resulting polypeptide profile. Phosphorylation assays were performed with [γ-32P] ATP and the [32P]-labeled phosphopolypeptides were separated by SDS-PAGE on a 12% gel and electrotransferred to a PVDF membrane. The membrane was exposed to X-ray film at −80°C, using an intensifying screen and visualized by autoradiography (panel B). Western blot employing a mixture of anti-α and β tubulin monoclonal antibodies (panel C).](/document/doi/10.1515/znc-2017-0019/asset/graphic/j_znc-2017-0019_fig_002.jpg)
Effect of divalent cations and chelating agents on the solubility of tubulin and tubulin kinase from T. equiperdum. Whole-cell homogenates (H), soluble (S) and particulate (P) fractions were obtained from T. equiperdum parasites extracted with either 50 mM Tris-HCl (pH 8.0), 5 mM MgCl2 and 5 mM CaCl2 (buffer A) or 50 mM Tris-HCl (pH 8.0), 10 mM EGTA and 10 mM EDTA (buffer B) and separated by SDS-PAGE. Panel A shows the resulting polypeptide profile. Phosphorylation assays were performed with [γ-32P] ATP and the [32P]-labeled phosphopolypeptides were separated by SDS-PAGE on a 12% gel and electrotransferred to a PVDF membrane. The membrane was exposed to X-ray film at −80°C, using an intensifying screen and visualized by autoradiography (panel B). Western blot employing a mixture of anti-α and β tubulin monoclonal antibodies (panel C).
To confirm that the 55-kDa band corresponded to phosphorylated tubulin, T. equiperdum homogenates were phosphorylated in vitro and analyzed after immunoprecipitation with specific anti-tubulin antibodies. The in vitro phosphorylation assay of the parasite extract was either analyzed directly by SDS-PAGE and autoradiography, or immunoprecipitated with the mixture of anti-α and anti-β tubulin monoclonal antibodies prior to separation by SDS-PAGE and autoradiography (Figure 3). As shown in Figure 3 (lane 2), a prominent 55-kDa polypeptide was phosphorylated in whole-cell lysates of T. equiperdum. The mixture of both antibodies precipitated the phosphorylated 55-kDa polypeptide, indicating that it contained phosphorylated forms of the parasite α- and β-tubulin (Figure 3, lane 3). No signal was detected in control assays using either pre-immune mouse serum (data not shown), or an unrelated mouse polyclonal antibody directed against a VSG from the TeAp-N/D1 strain of T. equiperdum (Figure 3, lane 4).
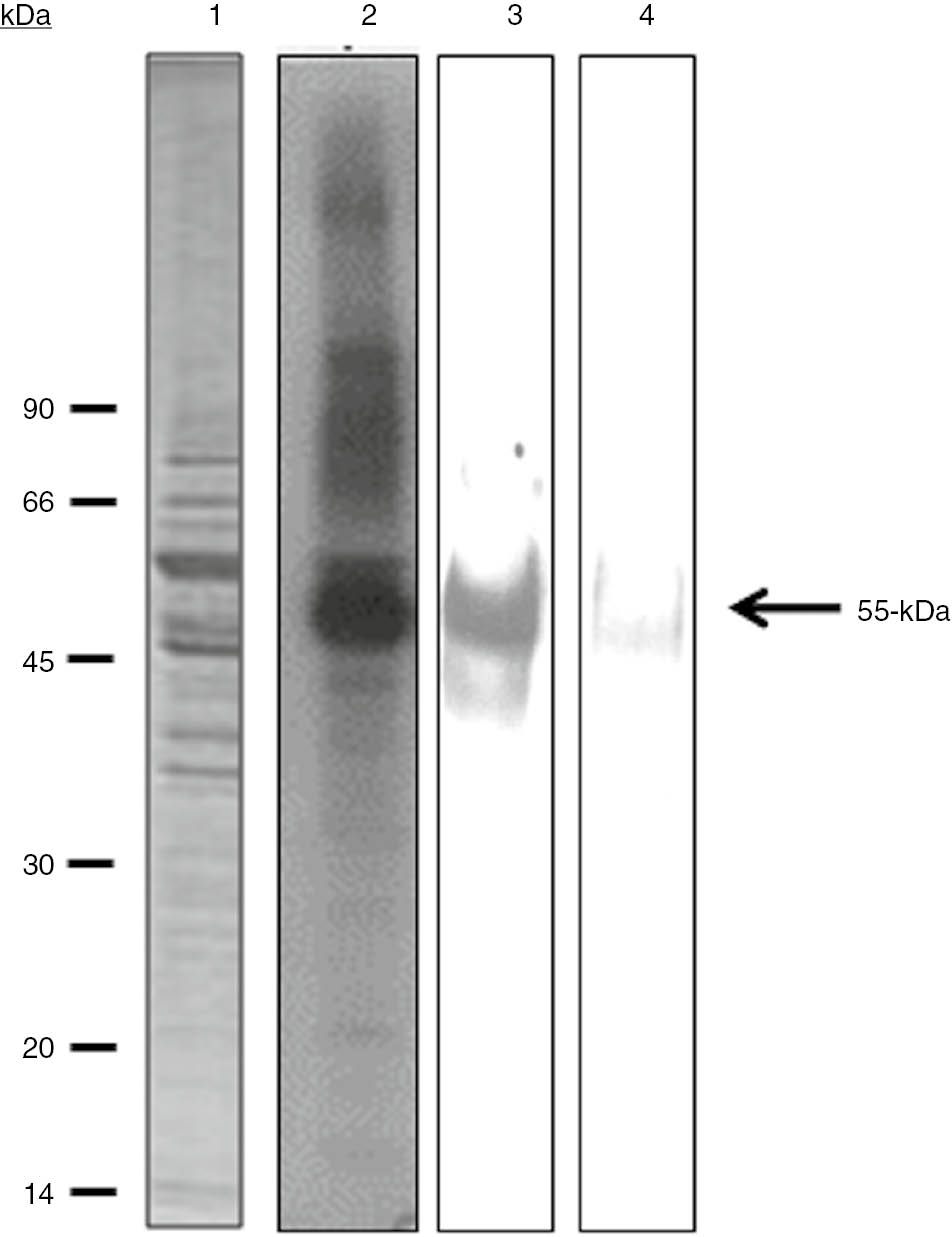
Immunoprecipitation with anti-tubulin monoclonal antibodies. The clarified soluble fraction was obtained from T. equiperdum parasites extracted in the presence of 10 mM EDTA and 10 mM EGTA. A sample of the parasite soluble fraction was phosphorylated in vitro and directly analyzed by SDS-PAGE followed by autoradiography (2) or immunoprecipitated by either a mixture of anti-α and β tubulin monoclonal antibodies (3) or a polyclonal antibody directed against the soluble form of a VSG from the same strain of T. equiperdum (4). The polypeptide profile revealed by Coomassie blue staining is shown in lane 1.
Column chromatography was employed in an effort to separate the parasite tubulin from its kinase. Trypanosoma equiperdum parasites were homogenized in the presence of 10 mM EDTA and 10 mM EGTA, in order to extract both proteins into the cytosolic fraction. Following ultracentrifugation, the clarified fraction was chromatographed on a DEAE-Sepharose anion-exchange column. As seen in Figure 4A, the absorbance profile showed a protein peak eluting when the ionic strength was increased to 0.5–0.7 M NaCl, which overlapped with the peak of endogenous phosphorylation. Coomassie blue staining following SDS-PAGE confirmed that the peak eluting from the DEAE-Sepharose column contained a polypeptide band of 55 kDa (Figure 4B), which corresponded to tubulin, as was shown by immunoblots employing anti-α and anti-β tubulin monoclonal antibodies (Figure 4D). Aliquots of the resulting fractions were subjected to phosphorylation assays and analysis by autoradiography showed that the activity responsible for phosphorylating tubulin (Figure 4C) and exogenously added casein (Figure 4E) coincided with this protein peak. Moreover, emodin inhibited the phosphorylation of both tubulin and casein, indicating again that tubulin kinase corresponded to a CK2 enzyme. All these results indicated that tubulin and its kinase were eluting together.
![Figure 4: DEAE-Sepharose chromatography. The soluble fraction from T. equiperdum parasites extracted in the presence of 10 mM EDTA and 10 mM EGTA was applied to a DEAE-Sepharose column and after extensive washing the proteins were eluted with a linear gradient from 0 to 2 M NaCl. (A) Fractions were collected and spectrophotometrically assayed for protein (■). Endogenous protein kinase (♦) activity was determined by liquid-scintillation counting after spotting aliquots of the reaction mixtures on P81 phosphocellulose filters. Also shown is the eluting salt concentration of the resulting fractions. (B) Aliquots of the column starting material (S) and eluted fractions 11–18 were analyzed by SDS-PAGE followed by Coomassie Blue staining. Identical aliquots were analyzed using either an in vitro phosphorylation assay in the presence of [γ-32P] ATP and the radioactively labeled gel was dried and subjected to autoradiography (C), or by immunoblot using a mixture of anti-α and anti-β tubulin antibodies (D). (E) Fractions containing the co-eluting tubulin and protein kinase responsible for its phosphorylation were combined and kinase-assayed without (–) or with (+) emodin, in the presence of casein as an exogenous substrate. Shown is an autoradiograph.](/document/doi/10.1515/znc-2017-0019/asset/graphic/j_znc-2017-0019_fig_004.jpg)
DEAE-Sepharose chromatography. The soluble fraction from T. equiperdum parasites extracted in the presence of 10 mM EDTA and 10 mM EGTA was applied to a DEAE-Sepharose column and after extensive washing the proteins were eluted with a linear gradient from 0 to 2 M NaCl. (A) Fractions were collected and spectrophotometrically assayed for protein (■). Endogenous protein kinase (♦) activity was determined by liquid-scintillation counting after spotting aliquots of the reaction mixtures on P81 phosphocellulose filters. Also shown is the eluting salt concentration of the resulting fractions. (B) Aliquots of the column starting material (S) and eluted fractions 11–18 were analyzed by SDS-PAGE followed by Coomassie Blue staining. Identical aliquots were analyzed using either an in vitro phosphorylation assay in the presence of [γ-32P] ATP and the radioactively labeled gel was dried and subjected to autoradiography (C), or by immunoblot using a mixture of anti-α and anti-β tubulin antibodies (D). (E) Fractions containing the co-eluting tubulin and protein kinase responsible for its phosphorylation were combined and kinase-assayed without (–) or with (+) emodin, in the presence of casein as an exogenous substrate. Shown is an autoradiograph.
An endogenously phosphorylated 60-kDa polypeptide band was also obtained in the fractions containing tubulin and CK2 (Figure 4C). However, the phosphorylation of the 60-kDa substrate was not affected by emodin (Figure 4E) or GTP (data not shown), indicating that a protein kinase different than CK2 was responsible for its phosphorylation. Accordingly, the 60-kDa polypeptide and its corresponding protein kinase co-purified with tubulin and CK2 following anion-exchange chromatography.
The DEAE-Sepharose fractions containing the co-eluting tubulin and the CK2 responsible for its phosphorylation were combined, concentrated and chromatographed using a Sephacryl S-300 size-exclusion column. As illustrated in Figure 5, the absorbance profile showed a protein peak that overlapped with the peak of endogenous phosphorylation. SDS-PAGE showed that this peak contained a 55-kDa band (Figure 5 inset, top), which was identified as tubulin, by immunoblotting using specific anti-tubulin monoclonal antibodies (Figure 6A). Analysis by autoradiography confirmed that this peak also contained the protein kinase responsible for phosphorylating both tubulin (Figure 5 inset, middle) and exogenous casein (Figure 5 inset, bottom). Moreover, emodin was capable of inhibiting the phosphorylation of both tubulin and casein (data not shown), indicating again that the protein kinase corresponded to a CK2. Thus, tubulin and its kinase also co-purified in the same protein peak after gel filtration. A major protein band was clearly observed when the Sephacryl S-300 fractions containing the co-eluting tubulin and CK2, were electrophoresed in a non-denaturing polyacrylamide gel (Figure 6B, lane 1). When the purified fractions were immunoblotted with anti-human CK2 α-subunit antibodies, the same protein band was recognized (Figure 6B, lane 2). Incubation of the PVDF membrane with a mixture of anti-α and anti-β tubulin monoclonal antibodies also identified the same slow-migrating protein band (Figure 6B, lane 3). This band migrated with an identical relative mobility as the phosphorylated band that appeared following phosphorylation of the same sample (Figure 6B, lane 4). All these results clearly established that this band contained a complex of the parasite tubulin and CK2. When the purified T. equiperdum tubulin-CK2 complex was electrophoresed in a denaturing polyacrylamide gel and then western blotted with anti-human CK2 α-subunit polyclonal antibodies, no recognition of polypeptide bands was observed (data not shown), suggesting that the protein three-dimensional native conformation is important for its recognition by these antibodies. Similarly, no recognition of CK2 was obtained when immunoblots using anti-CK2 α-subunit antibodies were carried out on the T. cruzi tubulin-CK2 complex under denaturing conditions [8].
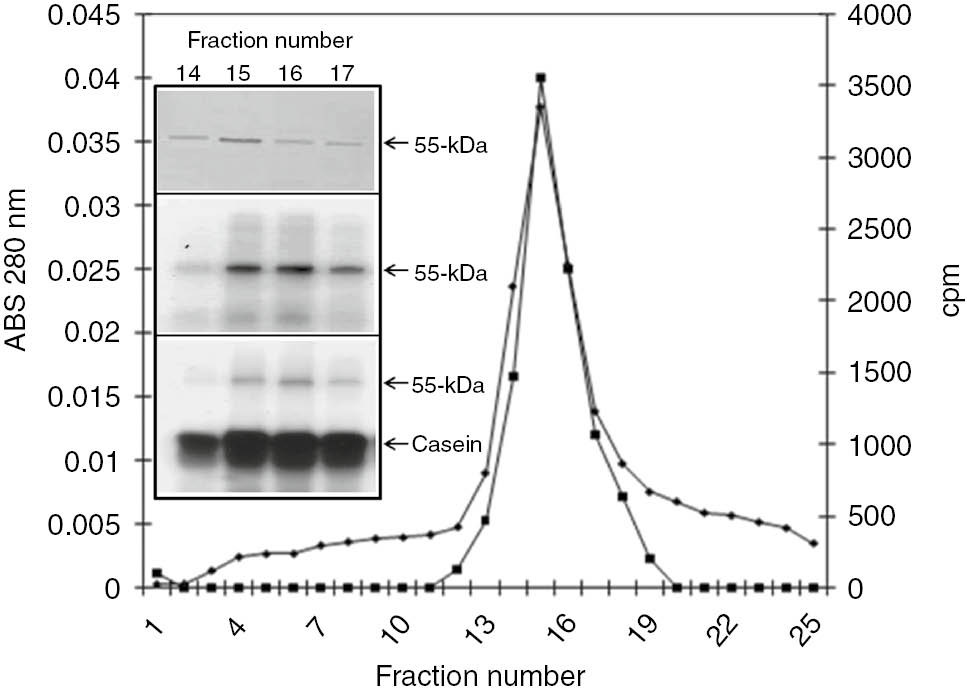
Sephacryl S-300 gel filtration chromatography. The tubulin and tubulin kinase-enriched fractions from the DEAE-Sepharose column were concentrated and subjected to size-exclusion chromatography on a Sephacryl S-300 column. Fractions were collected and assayed for protein (■) and endogenous protein kinase (♦). Inset: Protein staining using Coomassie Blue (top), autoradiography of the phosphorylated fractions (middle) and casein kinase activity visualized by autoradiography (bottom).
![Figure 6: Association of a pool of tubulin and the CK2 responsible for its phosphorylation. (A) Fractions containing the protein peak from the Sephacryl S-300 column were combined and analyzed by western blot using either anti-α tubulin or anti-β tubulin monoclonal antibodies. (B) Native electrophoresis of the purified CK2-tubulin complex previously phosphorylated with [γ-32P] ATP. Protein staining using silver (1), immunoblot using either an anti-CK2 α-subunit polyclonal antibody (2) or a mixture of anti-α and anti-β tubulin monoclonal antibodies (3), analysis of [32P] labeling by autoradiography (4).](/document/doi/10.1515/znc-2017-0019/asset/graphic/j_znc-2017-0019_fig_006.jpg)
Association of a pool of tubulin and the CK2 responsible for its phosphorylation. (A) Fractions containing the protein peak from the Sephacryl S-300 column were combined and analyzed by western blot using either anti-α tubulin or anti-β tubulin monoclonal antibodies. (B) Native electrophoresis of the purified CK2-tubulin complex previously phosphorylated with [γ-32P] ATP. Protein staining using silver (1), immunoblot using either an anti-CK2 α-subunit polyclonal antibody (2) or a mixture of anti-α and anti-β tubulin monoclonal antibodies (3), analysis of [32P] labeling by autoradiography (4).
4 Discussion
In this study we demonstrated a direct physical association between a pool of tubulin and a CK2 enzyme in T. equiperdum. This protein-protein interaction between a pool of tubulin and a CK2 enzyme is probably common among trypanosomatids as a similar finding was reported in T. cruzi [8, 14, 15].
In a variety of biological systems, CK2 has been shown to participate in the preservation of the morphology and polarity of the cell and in the regulation of the cytoskeleton [16], for example, CK2 was found to interact with microtubules from rat brain [17, 18]. Co-immunoprecipitation and far Western blot analyses revealed that CK2 α and α’ catalytic subunits are capable of binding to tubulin [19]; however, there was no ligation of the CK2 β regulatory subunit to tubulin [19]. By using RNA interference experiments, Lim et al. [20] have shown that CK2 is a microtubule-associated protein that mediates microtubule integrity. Carneiro et al. [21] revealed that incubation of rat retinas with a CK2 inhibitor led to disruption of their microtubules and to blockage of nuclear migration of retinal progenitor cells during development. In addition, Kramerov et al. [22] reported that incubation of cultured human astrocytes and vascular endothelial cells with CK2 inhibitors induced alterations in both cell shape and cytoskeleton organization. All these findings suggest that the phosphorylation by CK2 of microtubule elements seems to facilitate microtubule assembly and dynamics. By analogy, the formation of the tubulin-CK2 complex together with the concomitant CK2-dependent phosphorylation of tubulin might assist trypanosome microtubule stabilization and integrity and might participate in the modulation of microtubule cytoskeleton reorganization in trypanosomatids.
Acknowledgments
Supported by Fondo Nacional de Ciencia, Tecnología e Innovación (FONACIT), grants No. LAB-2000001639, S1-2001000807 and 2013001659, Caracas, Venezuela; and by Decanato de Investigación y Desarrollo (DID), Universidad Simón Bolívar, grant No. S1-IC-CB-007-14, Caracas, Venezuela.
References
1. Brun R, Hecker H, Lun ZR. Trypanosoma evansi and T. equiperdum: distribution, biology, treatment and phylogenetic relationship (a review). Vet Parasitol 1998;79:95–107.10.1016/S0304-4017(98)00146-0Suche in Google Scholar
2. Gibson W. Species concepts for trypanosomes: from morphological to molecular definitions? Kinetoplastid Biol Dis 2003;2:10.10.1186/1475-9292-2-10Suche in Google Scholar
3. Gull K. The cytoskeleton of trypanosomatid parasites. Annu Rev Microbiol 1999;53:629–55.10.1146/annurev.micro.53.1.629Suche in Google Scholar
4. Hanks SK, Hunter T. Protein kinases 6. The eukaryotic protein kinase superfamily: kinase (catalytic) domain structure and classification. FASEB J 1995;9:576–96.10.1096/fasebj.9.8.7768349Suche in Google Scholar
5. Galán-Caridad JM, Calabokis M, Uzcanga G, Aponte F, Bubis J. Identification of casein kinase 1, casein kinase 2, and cAMP-dependent protein kinase-like activities in Trypanosoma evansi. Mem Inst Oswaldo Cruz 2004;99:845–54.10.1590/S0074-02762004000800011Suche in Google Scholar
6. Sánchez E, Perrone TM, Recchimuzzi G, Cardozo I, Biteau N, Mijares A, et al. Molecular characterization and classification of Trypanosoma spp. Venezuelan isolates based on microsatellite markers and kinetoplast maxicircle genes. Parasit Vectors 2015;8:536.10.1186/s13071-015-1129-2Suche in Google Scholar
7. Lanham SM, Godfrey DG. Isolation of salivarian trypanosomes from man and other mammals using DEAE-cellulose. Exp Parasitol 1970;28:521–34.10.1016/0014-4894(70)90120-7Suche in Google Scholar
8. De Lima AR, Medina R, Uzcanga GL, Noris Suárez K, Contreras VT, Navarro MC, et al. Tight binding between a pool of the heterodimeric α/β tubulin and a protein kinase CK2 in Trypanosoma cruzi epimastigotes. Parasitology 2006;132:511–23.10.1017/S0031182005009352Suche in Google Scholar
9. Uzcanga GL, Pérez-Rojas Y, Camargo R, Izquier A, Noda JA, Chacín R, et al. Serodiagnosis of bovine trypanosomosis caused by non-tsetse transmitted Trypanosoma (Duttonella) vivax parasites using the soluble form of a Trypanozoon variant surface glycoprotein antigen. Vet Parasitol 2016;218:31–42.10.1016/j.vetpar.2016.01.007Suche in Google Scholar
10. Bradford MM. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 1976;72:248–54.10.1016/0003-2697(76)90527-3Suche in Google Scholar
11. Laemmli UK. Cleavage of structural proteins during assembly of the head of bacteriophage T4. Nature 1970;227:680–5.10.1038/227680a0Suche in Google Scholar
12. Schagger H, Cramer WA, Von Jagow G. Analysis of molecular masses and oligomeric states of protein complexes by blue native electrophoresis and isolation of membrane protein complexes by two-dimensional native electrophoresis. Anal Biochem 1994;217:220–30.10.1006/abio.1994.1112Suche in Google Scholar
13. Towbin H, Staehelin T, Gordon J. Electrophoretic transfer of proteins from polyacrylamide gels to nitrocellulose sheets: procedure and some applications. Biotechnology 1979;24:145–9.10.1073/pnas.76.9.4350Suche in Google Scholar
14. Uzcanga G, Galán-Caridad JM, Noris Suarez K, Bubis J. Divalent cation hinder the solubilization of a tubulin kinase activity from Trypanosoma cruzi epimastigotes. Biol Res 2003;36:367–79.10.4067/S0716-97602003000300008Suche in Google Scholar
15. Casas B, Calabokis M, Kurz L, Galán-Caridad JM, Bubis J, Gonzatti MI. Trypanosoma cruzi: in vitro phosphorylation of tubulin by a protein kinase CK2-like enzyme. Exp Parasitol 2002;101:129–37.10.1016/S0014-4894(02)00110-8Suche in Google Scholar
16. Canton DA. Litchfield DW. The shape of things to come: an emerging role for protein kinase CK2 in the regulation of cell morphology and the cytoskeleton. Cell Signal 2006;18:267–75.10.1016/j.cellsig.2005.07.008Suche in Google Scholar
17. Serrano L, Hernández MA, Díaz-Nido J, Avila J. Association of casein kinase II with microtubules. Exp Cell Res 1989;181:263–72.10.1016/0014-4827(89)90200-0Suche in Google Scholar
18. Díaz-Nido J, Avila J. Protein kinases associated with isolated mitotic spindles from mammalian cells: identification of a casein kinase II-like enzyme. Second Messengers Phosphoproteins 1992;14:39–53.Suche in Google Scholar
19. Faust M, Schuster N, Montenarh M. Specific binding of protein kinase CK2 catalytic subunits to tubulin. FEBS Lett 1999;462: 51–6.10.1016/S0014-5793(99)01492-1Suche in Google Scholar
20. Lim AC, Tiu SY, Li Q, Qi RZ. Direct regulation of microtubule dynamics by protein kinase CK2. J Biol Chem 2004;279:4433–9.10.1074/jbc.M310563200Suche in Google Scholar PubMed
21. Carneiro AC, Fragel-Madeira L, Silva-Neto MA, Linden R. A role for CK2 upon interkinetic nuclear migration in the cell cycle of retinal progenitor cells. Dev Neurobiol 2008;68:620–31.10.1002/dneu.20613Suche in Google Scholar PubMed
22. Kramerov AA, Golub AG, Bdzhola VG, Yarmoluk SM, Ahmed K, Bretner M, et al. Treatment of cultured human astrocytes and vascular endothelial cells with protein kinase CK2 inhibitors induces early changes in cell shape and cytoskeleton. Mol Cell Biochem 2011;349:125–37.10.1007/s11010-010-0667-3Suche in Google Scholar PubMed PubMed Central
©2017 Walter de Gruyter GmbH, Berlin/Boston
Artikel in diesem Heft
- Frontmatter
- Ginkgetin inhibits proliferation of human leukemia cells via the TNF-α signaling pathway
- Effects of a water extract of Lepidium meyenii root in different models of persistent pain in rats
- Interaction of tubulin and protein kinase CK2 in Trypanosoma equiperdum
- Synthesis, biological activity and molecular modeling study of new Schiff bases incorporated with indole moiety
- A dihydrochalcone derivative and further steroidal saponins from Sansevieria trifasciata Prain
- Chemical composition and biological activities of leaf and fruit essential oils from Eucalyptus camaldulensis
- Fungal production of the polysaccharide pullulan from a plant hydrolysate
- Acetogenins and alkaloids during the initial development of Annona muricata L. (Annonaceae)
- Corrigendum
- Corrigendum to: Ginkgetin inhibits proliferation of human leukemia cells via the TNF-α signaling pathway
Artikel in diesem Heft
- Frontmatter
- Ginkgetin inhibits proliferation of human leukemia cells via the TNF-α signaling pathway
- Effects of a water extract of Lepidium meyenii root in different models of persistent pain in rats
- Interaction of tubulin and protein kinase CK2 in Trypanosoma equiperdum
- Synthesis, biological activity and molecular modeling study of new Schiff bases incorporated with indole moiety
- A dihydrochalcone derivative and further steroidal saponins from Sansevieria trifasciata Prain
- Chemical composition and biological activities of leaf and fruit essential oils from Eucalyptus camaldulensis
- Fungal production of the polysaccharide pullulan from a plant hydrolysate
- Acetogenins and alkaloids during the initial development of Annona muricata L. (Annonaceae)
- Corrigendum
- Corrigendum to: Ginkgetin inhibits proliferation of human leukemia cells via the TNF-α signaling pathway

